Abstract
There remains a significant un-met need to reduce the extent of myocardial injury caused by ischaemia and reperfusion injury in patients experiencing an ST-elevation MI. Although nitric oxide is central to many cardioprotective strategies currently undergoing investigation, cardioprotection from the delivery of nitrates/nitrites has been inconsistently observed. The route of administration appears to be a critical variable. The glyceryl trinitrate (GTN) patch is commonly used as a simple and practical means of delivering nitric oxide to patients with ischaemic heart disease, but whether acute cardioprotection can be achieved by application of a GTN patch has not been investigated before. Here, we use a mouse model to demonstrate that a GTN patch is highly cardioprotective when applied immediately prior to 40 min occlusion of the left anterior coronary artery followed by 2 h reperfusion, reducing infarct size from 54 ± 4% in control mice, to 28 ± 4% (P < 0.001, N = 7). The degree of protection was similar to that achieved with a standard remote ischaemic preconditioning protocol. Furthermore, and of greater potential clinical relevance, a GTN patch was also protective when applied well after the initiation of ischaemia and 15 min prior to reperfusion (28 ± 4 vs 59 ± 4%; P < 0.01, N = 5). Confirmatory experiments verified the expected effect increase in plasma nitrite levels and decrease in blood pressure. The simplicity and rapidity of GTN patch application (easily applied in an ambulance or cardiac catheterization laboratory), and low cost (potentially relevant to low-income countries), make it attractive for further investigation.
Keywords: Nitrate, Nitric oxide, Cardioprotection, Mouse, Ischaemia, Reperfusion
Introduction
There remains a significant un-met need to reduce the extent of myocardial injury caused by ischaemia and reperfusion injury in patients experiencing an ST-elevation MI (STEMI) [11–13, 15]. The ideal cardioprotective modality would be simple, effective, and able to be easily and rapidly delivered by the first responder. One promising procedure that is being investigated at present is remote ischaemic preconditioning (RIPC), in which several brief episodes of ischaemia and reperfusion are applied to a limb, thereby signalling to the heart to stimulate cardioprotection. RIPC has shown promise in reducing infarct size in STEMI patients in numerous proof of concept studies and is now undergoing investigation in a large, multi-centre, randomized trial [3, 14, 28]. However, the application of RIPC takes at least 30 min. Although the precise mechanism of RIPC is still under investigation, circulating nitrite originating from RIPC limb is believed to contribute to its cardioprotection [29, 36].
Organic nitrates, such as glyceryl trinitrate (GTN), are a highly effective means of rapidly delivering nitrate and nitrite into the blood stream, and are potent vasodilators. The transdermal GTN patch is widely used to reduce angina in patients with acute and chronic ischaemic syndromes due to coronary artery disease. Application of a GTN patch has been shown to induce delayed cardioprotection when applied to rabbits 72 h prior to infarction [19]. However, whether a GTN patch is protective when applied at a more clinically relevant time-point, such as during ischaemia, has never been investigated. We have investigated this hypothesis using an in vivo mouse model of ischaemia and reperfusion.
Materials and methods
All animals received humane care in accordance with the United Kingdom Home Office Guide on the Operation of Animal (Scientific Procedures) Act of 1986. The investigation conforms to the guidelines from Directive 2010/63/EU of the European Parliament on the protection of animals used for scientific purposes or the NIH guidelines. All experiments were approved by the appropriate ethics committee and have, therefore, been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
10- to 12-week-old C57Bl/6 male mice were anaesthetized by i.p. injection of 80 mg/kg pentobarbitone at a concentration of 20 mg/ml in 0.9% (w/v) saline and maintained at 36.5 ± 0.5 °C on a heating mat. Surgery was started once pedal and tail reflexes were abolished and depth of anaesthesia was monitored throughout. Mice were intubated using a 19G cannula and ventilated using a MiniVent, type 845, Small Animal Ventilator (Harvard Apparatus, Kent, UK), supplemented with either room air or 100% oxygen, at a flow rate of 1.0 l/min with 2 cmH2O PEEP, stroke volume 200 µl at 130 strokes/min. After orotracheal intubation, the left common carotid artery running parallel to the trachea was carefully dissected and isolated, taking care not to damage the vagus nerve. The artery was cannulated using a thinned tip polyethylene tube (OD 0.96 mm) that was pre-filled with saline containing 10 units of heparin and was connected a calibrated pressure transducer. Mean arterial blood pressure was recorded using Labchart software.
A transdermal 5 mg GTN patch for human use was cut into eight equal pieces, and a single piece (containing 0.6 mg GTN) was applied to the depilated abdomen of an anaesthetized mouse. Inactive tape was applied to control mice. The change in blood pressure was measured after 10 min. The blood was then immediately removed by cardiac puncture, directly into a syringe containing sodium citrate. Plasma was obtained by centrifugation at 13,000×g for 15 min. Blood plasma was used for determination of total nitrate and nitrite (NOx) concentrations using ozone chemiluminescence. Plasma was filtered using Sartorius Vivaspin 500 3000 MWCO PES (Sartorius Stedim Biotech, Germany) at 4 °C, 14,000g for 60 min. Prior to use, filters were washed twice with low NOx containing 18 MΩ dH2O. An NO analyser (NOA 280A, Sievers, UK) was used to measure NO based on the gas-phase chemiluminescent reaction between NO and ozone. To determine total NOx concentration, samples were added to the purge vessel containing 0.1 M vanadium(III) chloride in 1 M hydrochloric acid refluxing at 95 °C under nitrogen. Nitrite concentration was determined by addition of samples to the purge vessel containing 0.09 M potassium iodide in glacial acetic acid under nitrogen at room temperature. Both of these conditions result in NO generation in the gas phase which is carried from the purge vessel to the NOA analyser where it reacts with ozone to emit a photon of light which is detected by the analyser. Nitrate concentration was calculated by subtraction of the nitrite concentration from the total NOx [4, 20].
In a second set of experiments, the left anterior descending (LAD) coronary artery of anaesthetized mice was occluded (verified by ST elevation, hypokinesia and pallor) for 40 min followed by 2 h reperfusion, after which infarct size was measured by tetrazolium staining and expressed as a percentage of area at risk, determined using Evan’s blue. A GTN patch (1/8) or inactive tape was applied 10 min prior to ischaemia and left throughout the experiment. Remote ischaemic preconditioning (RIPC) was induced using a 6-mm lumen custom vascular occluder (Kent Scientific, CT, USA) around the right hind limb inflated to 250 mmHg to induce three cycles of 5 min ischaemia followed by 5 min reperfusion.
In a third set of experiments, ischaemia and reperfusion was performed as described above, but the patch (1/8) was applied after 25 min ischaemia, 15 min prior to reperfusion. All mice (i.e.: both control and those receiving the GTN patch) were transfused via the jugular vein with 250 µl blood from a donor mouse during reperfusion, in order to ensure blood pressure was maintained.
The results are shown as mean ± standard deviation of the mean. Statistical comparison of the groups was made by two-way ANOVA, with Bonferroni correction for multiple comparisons. A significance value of P < 0.05 was considered significant.
Results
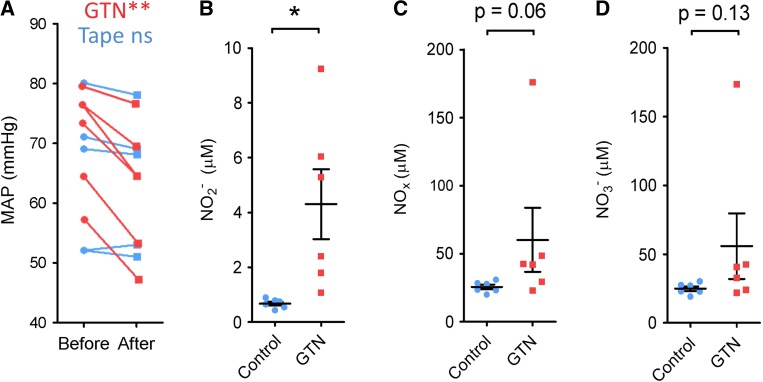
In anaesthetized mice, application of the GTN patch resulted in a 13 ± 2% decrease in mean arterial blood pressure (MAP) after 10 min (P < 0.01, N = 7) (Fig. 1a). Plasma nitrite levels were confirmed to be 6 ± 2-fold greater than in mice receiving the control tape (P < 0.05, N = 6-7) (Fig. 1b). Plasma nitrate and total nitrate + nitrite levels were similarly increased, although the difference was not significant (Fig. 1c, d).
Fig. 1.
a The mean arterial blood pressure of anaesthetized mice decreased significantly 10 min after adhesion of a GTN patch to the abdomen (red), but not after control adhesive tape (blue). **P < 0.01 by paired T test. b–d In mice treated with a GTN patch, plasma nitrite (NO2−) concentration was significantly elevated after 10 min (*P < 0.05 by unpaired T test). The difference in plasma nitrate (NO3−) and total nitrates and nitrites was not significant
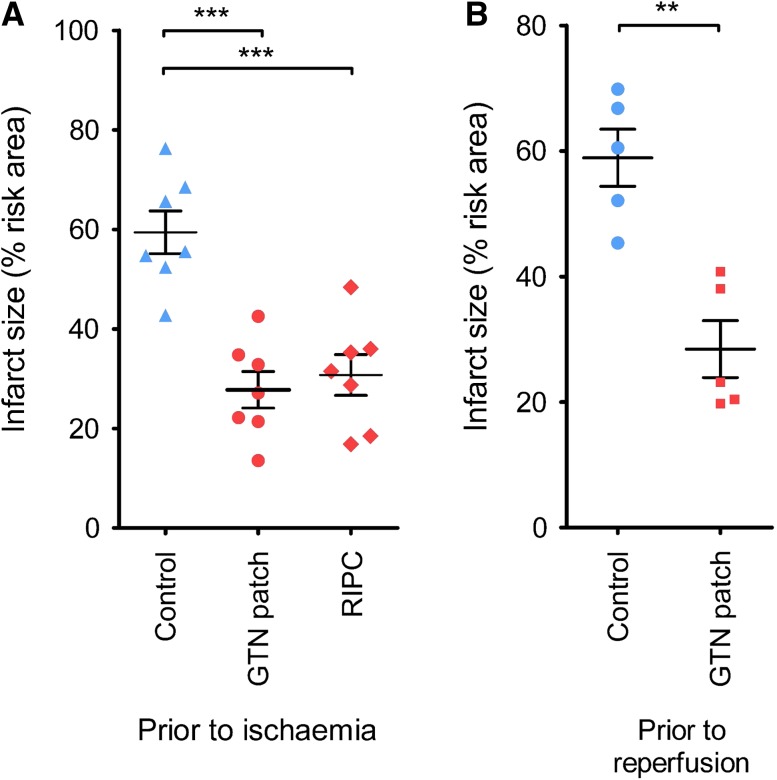
In a second set of experiments, application of a GTN patch 10 min prior to 40 min ischaemia followed by 2 h reperfusion significantly reduced myocardial infarct size from 54 ± 4 to 28 ± 4% (P < 0.001, N = 7) (Fig. 2a). The degree of protection was equivalent to a positive control consisting of a standard remote ischaemic preconditioning protocol of three cycles of 5 min hind-limb, blood-flow occlusion (Fig. 2a). It was important to verify that the GTN patch would reduce infarct size when applied prior to reperfusion, as this better reflects the potential scenario in which it would be applied to a patient in an ambulance. Application of the GTN patch 15 min prior to reperfusion was equally effective at reducing infarct size (28 ± 4 vs 59 ± 4%; P < 0.01, N = 5) (Fig. 2b).
Fig. 2.
a Infarct size as a percentage of the area at risk was significantly decreased by a GTN patch applied 10 min prior to ischaemia. A positive control of remote ischaemic preconditioning was equally protective (RIPC). b GTN patch applied 15 min prior to reperfusion was cardioprotective. ***P < 0.001 one-way ANOVA and Tukey post-test. **P < 0.01 by unpaired T test
Discussion
These results suggest that the simple application of a transdermal GTN patch may be an effective means of protecting the heart against ischaemia and reperfusion injury, whether it is applied prior to ischaemia, or prior to reperfusion. Organic nitrates such as GTN are rapidly metabolized into nitrite, which is then reduced to the active vasodilator molecule, nitric oxide. Although nitric oxide is central to many cardioprotective strategies, including remote preconditioning, cardioprotection from the delivery of nitrates/nitrites has been inconsistently observed. The route and timing of administration appears to be a critical variable [19, 31]. For example, i.v. infusion of sodium nitrite at the time of reperfusion did not reproducibly protect animals in the CAESAR trial, but intracoronary or oral nitrite has successfully reduced infarct size in other animal studies [7, 16, 32, 36]. A recent systematic review of nitric oxide therapies given at reperfusion, which included 21 animal studies, found significant protection in the majority of studies, with a mean overall reduction in infarct size of 17.93% (95% confidence interval: 22.05, 13.81) [2]. The exceptions in which no protection was observed included 5 studies: two in which 2 µg/kg/min GTN was administered to rabbits or pigs via continuous IV infusion starting 5 or 10 min prior to reperfusion [25, 30]; a study in rats in which 4 mg/kg NaNO2 was administered IV starting 10 s after the onset of reperfusion [1]; a study from 1997 in which acidified NaNO2 was infused IV in adult male mongrel dogs starting at the time of reperfusion [37]; and a study in which inhalation of 80 ppm NO was initiated 0.5 min prior to reperfusion in mice [27].
In humans, a 4-h intravenous infusion of GTN protects the heart against ischaemia during coronary angioplasty conducted 24 h later, as evidenced by reduced ST-segment elevation, ischaemic dysfunction, and pain [24]. However, how well these clinical endpoints relate to experimentally used endpoints of myocardial damage caused by ischaemia and reperfusion injury, and the precise relationship between GTN and preconditioning are not entirely clear [18]. Several studies of STEMI patients from the thrombolytic era, as well as a more recent study, have not found evidence for infarct size reduction after nitrate or nitrite infusion [2]. Interestingly, in a recent study, intracoronary but not i.v. infusion of nitrites reduced infarct size in STEMI patients with completely occluded arteries at admission [21, 33], in whom reperfusion injury might be expected to be a significant factor. GTN is sometimes administered to patients undergoing surgical coronary revascularization, at the discretion of the anaesthesiologist. However, when comparing patients who do not receive GTN to those who receive 0.042 ± 0.024 mg/kg GTN, no evidence for a reduction peri-procedural injury was found, as determined by cTnI release [23]. Surprisingly, the simple, practical, and cost-effective method of inducing acute cardioprotection by simply applying a GTN patch prior to reperfusion had not been previously investigated.
Nitrates and nitric oxide can protect the heart via multiple possible mechanisms, but this appears to be independent from their vasodilatory effects [5]. Nitric oxide can directly nitrosate proteins and also activate a pathway involving nitric oxide-sensitive guanylyl cyclase and cGMP-dependent protein kinase type I (PKGI) in cardiomyocytes [6, 9]. Recently, these have been shown to activate cardioprotection via large-conductance, Ca2+-dependent potassium (BK) channels [8, 17].
The GTN patch used here was designed to deliver 5 mg/day, so the 1/8 patch we used is expected to deliver 0.026 mg/h. However, we did not perform a dose–response experiment to establish the optimal dose of GTN. Basal levels of plasma nitrite vary considerably and decrease with increasing numbers of cardiovascular risk factors [22]. Plasma nitrates are also affected by dietary nitrates. Dietary inorganic nitrate ingestion or supplementation causes a dose-dependent elevation in plasma nitrite concentration with a consequent decrease in blood pressure in healthy volunteers. A meta-analysis found a significant association of the dose of inorganic nitrate supplementation with decline in systolic blood pressure, with a factor of − 0.12 mmHg/1.0 mmol nitrate (P < 0.05) [34]. Although it would be of interest to test for an association between infarct size and plasma nitrite, this was not possible in our study since the quantity of blood necessary for the nitrite assay made it impossible to perform infarction using the same mice.
When using this approach, it is important to be aware that excessive nitrate concentrations are potentially damaging, and in addition to nitrate tolerance, can cause nitro-oxidative stress. Endothelial dysfunction, formation of DNA adducts, and even apoptotic death of vascular cells has been seen when 10–50 mg/kg/day nitrate was administered to rats for 3 days [26]. Furthermore, prolonged exposure to GTN can induce tolerance. For example, 7 days of transdermal GTN exposure abolished pacing-induced preconditioning in conscious rabbits [35] and abrogated the effectiveness of remote ischaemic conditioning in rat myocardium and human volunteers [10]. Interestingly, however, direct myocardial protection with glyceryl trinitrate (GTN) may be preserved in the state of vascular nitrate tolerance [5].
Clinical studies are required to establish the potential efficacy of using the transdermal GTN patch in the setting of patients who are experiencing an STEMI. These will necessitate careful determination of the appropriate dose and timing of administration. However, its simplicity and rapidity (easily applied in an ambulance or cardiac catheterization laboratory), and low cost (potentially relevant to low-income countries), make it attractive for further investigation.
Funding
NIHR Biomedical Research Council (SD), British Heart Foundation PG/15/52/31598 (SD, DH) and the The Hatter Foundation.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no competing interests.
References
- 1.Baker JE, Su J, Fu X, Hsu A, Gross GJ, Tweddell JS, Hogg N. Nitrite confers protection against myocardial infarction: role of xanthine oxidoreductase, NADPH oxidase and K(ATP) channels. J Mol Cell Cardiol. 2007;43:437–444. doi: 10.1016/j.yjmcc.2007.07.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bice JS, Jones BR, Chamberlain GR, Baxter GF. Nitric oxide treatments as adjuncts to reperfusion in acute myocardial infarction: a systematic review of experimental and clinical studies. Basic Res Cardiol. 2016;111:23. doi: 10.1007/s00395-016-0540-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bromage DI, Pickard JM, Rossello X, Ziff OJ, Burke N, Yellon DM, Davidson SM. Remote ischaemic conditioning reduces infarct size in animal in vivo models of ischaemia-reperfusion injury: a systematic review and meta-analysis. Cardiovasc Res. 2017;113:288–297. doi: 10.1093/cvr/cvw219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bush PA, Gonzalez NE, Griscavage JM, Ignarro LJ. Nitric oxide synthase from cerebellum catalyzes the formation of equimolar quantities of nitric oxide and citrulline from l-arginine. Biochem Biophys Res Commun. 1992;185:960–966. doi: 10.1016/0006-291X(92)91720-B. [DOI] [PubMed] [Google Scholar]
- 5.Csont T, Ferdinandy P. Cardioprotective effects of glyceryl trinitrate: beyond vascular nitrate tolerance. Pharmacol Ther. 2005;105:57–68. doi: 10.1016/j.pharmthera.2004.10.001. [DOI] [PubMed] [Google Scholar]
- 6.Davidson SM, Yellon DM. Cardioprotection—is NO the answer? A renewed look at nitric oxide signalling in cardiomyocytes. Cardiovasc Res. 2018 doi: 10.1093/cvr/cvy073. [DOI] [PubMed] [Google Scholar]
- 7.Duranski MR, Greer JJ, Dejam A, Jaganmohan S, Hogg N, Langston W, Patel RP, Yet SF, Wang X, Kevil CG, Gladwin MT, Lefer DJ. Cytoprotective effects of nitrite during in vivo ischemia-reperfusion of the heart and liver. J Clin Investig. 2005;115:1232–1240. doi: 10.1172/JCI22493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Frankenreiter S, Bednarczyk P, Kniess A, Bork NI, Straubinger J, Koprowski P, Wrzosek A, Mohr E, Logan A, Murphy MP, Gawaz M, Krieg T, Szewczyk A, Nikolaev VO, Ruth P, Lukowski R. cGMP-elevating compounds and ischemic conditioning provide cardioprotection against ischemia and reperfusion injury via cardiomyocyte-specific BK channels. Circulation. 2017;136:2337–2355. doi: 10.1161/CIRCULATIONAHA.117.028723. [DOI] [PubMed] [Google Scholar]
- 9.Frankenreiter S, Groneberg D, Kuret A, Krieg T, Ruth P, Friebe A, Lukowski R. Cardioprotection by ischemic postconditioning and cGMP-elevating agents involves cardiomyocyte nitric oxide-sensitive guanylyl cyclase. Cardiovasc Res. 2018 doi: 10.1093/cvr/cvy039. [DOI] [PubMed] [Google Scholar]
- 10.Hauerslev M, Mork SR, Pryds K, Contractor H, Hansen J, Jespersen NR, Johnsen J, Heusch G, Kleinbongard P, Kharbanda R, Botker HE, Schmidt MR. Influence of long-term treatment with glyceryl trinitrate on remote ischemic conditioning. Am J Physiol Heart Circ Physiol. 2018 doi: 10.1152/ajpheart.00114.2018. [DOI] [PubMed] [Google Scholar]
- 11.Hausenloy DJ, Barrabes JA, Botker HE, Davidson SM, Di Lisa F, Downey J, Engstrom T, Ferdinandy P, Carbrera-Fuentes HA, Heusch G, Ibanez B, Iliodromitis EK, Inserte J, Jennings R, Kalia N, Kharbanda R, Lecour S, Marber M, Miura T, Ovize M, Perez-Pinzon MA, Piper HM, Przyklenk K, Schmidt MR, Redington A, Ruiz-Meana M, Vilahur G, Vinten-Johansen J, Yellon DM, Garcia-Dorado D. Ischaemic conditioning and targeting reperfusion injury: a 30 year voyage of discovery. Basic Res Cardiol. 2016;111:70. doi: 10.1007/s00395-016-0588-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hausenloy DJ, Baxter G, Bell R, Botker HE, Davidson SM, Downey J, Heusch G, Kitakaze M, Lecour S, Mentzer R, Mocanu MM, Ovize M, Schulz R, Shannon R, Walker M, Walkinshaw G, Yellon DM. Translating novel strategies for cardioprotection: the Hatter workshop recommendations. Basic Res Cardiol. 2010;105:677–686. doi: 10.1007/s00395-010-0121-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hausenloy DJ, Garcia-Dorado D, Botker HE, Davidson SM, Downey J, Engel FB, Jennings R, Lecour S, Leor J, Madonna R, Ovize M, Perrino C, Prunier F, Schulz R, Sluijter JPG, Van Laake LW, Vinten-Johansen J, Yellon DM, Ytrehus K, Heusch G, Ferdinandy P. Novel targets and future strategies for acute cardioprotection: position paper of the European Society of Cardiology Working Group on cellular biology of the heart. Cardiovasc Res. 2017;113:564–585. doi: 10.1093/cvr/cvx049. [DOI] [PubMed] [Google Scholar]
- 14.Hausenloy DJ, Kharbanda R, Rahbek Schmidt M, Moller UK, Ravkilde J, Okkels Jensen L, Engstrom T, Garcia Ruiz JM, Radovanovic N, Christensen EF, Sorensen HT, Ramlall M, Bulluck H, Evans R, Nicholas J, Knight R, Clayton T, Yellon DM, Botker HE. Effect of remote ischaemic conditioning on clinical outcomes in patients presenting with an ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Eur Heart J. 2015;36:1846–1848. [PubMed] [Google Scholar]
- 15.Hausenloy DJ, Yellon DM. Ischaemic conditioning and reperfusion injury. Nat Rev Cardiol. 2016;13:193–209. doi: 10.1038/nrcardio.2016.5. [DOI] [PubMed] [Google Scholar]
- 16.Hendgen-Cotta UB, Merx MW, Shiva S, Schmitz J, Becher S, Klare JP, Steinhoff HJ, Goedecke A, Schrader J, Gladwin MT, Kelm M, Rassaf T. Nitrite reductase activity of myoglobin regulates respiration and cellular viability in myocardial ischemia-reperfusion injury. Proc Natl Acad Sci USA. 2008;105:10256–10261. doi: 10.1073/pnas.0801336105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Heusch G. Cardioprotection is alive but remains enigmatic: the nitric oxide–protein kinases–mitochondria signaling axis. Circulation. 2017;136:2356–2358. doi: 10.1161/CIRCULATIONAHA.117.031978. [DOI] [PubMed] [Google Scholar]
- 18.Heusch G. Nitroglycerin and delayed preconditioning in humans: yet another new mechanism for an old drug? Circulation. 2001;103:2876–2878. doi: 10.1161/01.CIR.103.24.2876. [DOI] [PubMed] [Google Scholar]
- 19.Hill M, Takano H, Tang XL, Kodani E, Shirk G, Bolli R. Nitroglycerin induces late preconditioning against myocardial infarction in conscious rabbits despite development of nitrate tolerance. Circulation. 2001;104:694–699. doi: 10.1161/hc3201.092218. [DOI] [PubMed] [Google Scholar]
- 20.Ignarro LJ, Fukuto JM, Griscavage JM, Rogers NE, Byrns RE. Oxidation of nitric oxide in aqueous solution to nitrite but not nitrate: comparison with enzymatically formed nitric oxide from l-arginine. Proc Natl Acad Sci USA. 1993;90:8103–8107. doi: 10.1073/pnas.90.17.8103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jones DA, Pellaton C, Velmurugan S, Rathod KS, Andiapen M, Antoniou S, van Eijl S, Webb AJ, Westwood MA, Parmar MK, Mathur A, Ahluwalia A. Randomized phase 2 trial of intracoronary nitrite during acute myocardial infarction. Circ Res. 2015;116:437–447. doi: 10.1161/CIRCRESAHA.116.305082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kleinbongard P, Dejam A, Lauer T, Jax T, Kerber S, Gharini P, Balzer J, Zotz RB, Scharf RE, Willers R, Schechter AN, Feelisch M, Kelm M. Plasma nitrite concentrations reflect the degree of endothelial dysfunction in humans. Free Radic Biol Med. 2006;40:295–302. doi: 10.1016/j.freeradbiomed.2005.08.025. [DOI] [PubMed] [Google Scholar]
- 23.Kleinbongard P, Thielmann M, Jakob H, Peters J, Heusch G, Kottenberg E. Nitroglycerin does not interfere with protection by remote ischemic preconditioning in patients with surgical coronary revascularization under isoflurane anesthesia. Cardiovasc Drugs Ther. 2013;27:359–361. doi: 10.1007/s10557-013-6451-3. [DOI] [PubMed] [Google Scholar]
- 24.Leesar MA, Stoddard MF, Dawn B, Jasti VG, Masden R, Bolli R. Delayed preconditioning-mimetic action of nitroglycerin in patients undergoing coronary angioplasty. Circulation. 2001;103:2935–2941. doi: 10.1161/01.CIR.103.24.2935. [DOI] [PubMed] [Google Scholar]
- 25.Liu X, Huang Y, Pokreisz P, Vermeersch P, Marsboom G, Swinnen M, Verbeken E, Santos J, Pellens M, Gillijns H, Van de Werf F, Bloch KD, Janssens S. Nitric oxide inhalation improves microvascular flow and decreases infarction size after myocardial ischemia and reperfusion. J Am Coll Cardiol. 2007;50:808–817. doi: 10.1016/j.jacc.2007.04.069. [DOI] [PubMed] [Google Scholar]
- 26.Mikhed Y, Fahrer J, Oelze M, Kroller-Schon S, Steven S, Welschof P, Zinssius E, Stamm P, Kashani F, Roohani S, Kress JM, Ullmann E, Tran LP, Schulz E, Epe B, Kaina B, Munzel T, Daiber A. Nitroglycerin induces DNA damage and vascular cell death in the setting of nitrate tolerance. Basic Res Cardiol. 2016;111:52. doi: 10.1007/s00395-016-0571-4. [DOI] [PubMed] [Google Scholar]
- 27.Nagasaka Y, Fernandez BO, Garcia-Saura MF, Petersen B, Ichinose F, Bloch KD, Feelisch M, Zapol WM. Brief periods of nitric oxide inhalation protect against myocardial ischemia-reperfusion injury. Anesthesiology. 2008;109:675–682. doi: 10.1097/ALN.0b013e318186316e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pickard JM, Botker HE, Crimi G, Davidson B, Davidson SM, Dutka D, Ferdinandy P, Ganske R, Garcia-Dorado D, Giricz Z, Gourine AV, Heusch G, Kharbanda R, Kleinbongard P, MacAllister R, McIntyre C, Meybohm P, Prunier F, Redington A, Robertson NJ, Suleiman MS, Vanezis A, Walsh S, Yellon DM, Hausenloy DJ. Remote ischemic conditioning: from experimental observation to clinical application: report from the 8th Biennial Hatter Cardiovascular Institute Workshop. Basic Res Cardiol. 2015;110:453. doi: 10.1007/s00395-014-0453-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rassaf T, Totzeck M, Hendgen-Cotta UB, Shiva S, Heusch G, Kelm M. Circulating nitrite contributes to cardioprotection by remote ischemic preconditioning. Circ Res. 2014;114:1601–1610. doi: 10.1161/CIRCRESAHA.114.303822. [DOI] [PubMed] [Google Scholar]
- 30.Salloum FN, Takenoshita Y, Ockaili RA, Daoud VP, Chou E, Yoshida K, Kukreja RC. Sildenafil and vardenafil but not nitroglycerin limit myocardial infarction through opening of mitochondrial K(ATP) channels when administered at reperfusion following ischemia in rabbits. J Mol Cell Cardiol. 2007;42:453–458. doi: 10.1016/j.yjmcc.2006.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schulz R, Kelm M, Heusch G. Nitric oxide in myocardial ischemia/reperfusion injury. Cardiovasc Res. 2004;61:402–413. doi: 10.1016/j.cardiores.2003.09.019. [DOI] [PubMed] [Google Scholar]
- 32.Shiva S, Sack MN, Greer JJ, Duranski M, Ringwood LA, Burwell L, Wang X, MacArthur PH, Shoja A, Raghavachari N, Calvert JW, Brookes PS, Lefer DJ, Gladwin MT. Nitrite augments tolerance to ischemia/reperfusion injury via the modulation of mitochondrial electron transfer. J Exp Med. 2007;204:2089–2102. doi: 10.1084/jem.20070198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Siddiqi N, Neil C, Bruce M, MacLennan G, Cotton S, Papadopoulou S, Feelisch M, Bunce N, Lim PO, Hildick-Smith D, Horowitz J, Madhani M, Boon N, Dawson D, Kaski JC, Frenneaux M, Investigators N Intravenous sodium nitrite in acute ST-elevation myocardial infarction: a randomized controlled trial (NIAMI) Eur Heart J. 2014;35:1255–1262. doi: 10.1093/eurheartj/ehu096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Siervo M, Lara J, Ogbonmwan I, Mathers JC. Inorganic nitrate and beetroot juice supplementation reduces blood pressure in adults: a systematic review and meta-analysis. J Nutr. 2013;143:818–826. doi: 10.3945/jn.112.170233. [DOI] [PubMed] [Google Scholar]
- 35.Szilvassy Z, Ferdinandy P, Nagy I, Jakab I, Koltai M. The effect of continuous versus intermittent treatment with transdermal nitroglycerin on pacing-induced preconditioning in conscious rabbits. Br J Pharmacol. 1997;121:491–496. doi: 10.1038/sj.bjp.0701163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Totzeck M, Hendgen-Cotta UB, Rassaf T. Nitrite-nitric oxide signaling and cardioprotection. Adv Exp Med Biol. 2017;982:335–346. doi: 10.1007/978-3-319-55330-6_18. [DOI] [PubMed] [Google Scholar]
- 37.Tripathi Y, Hegde BM, Raghuveer CV. Effect of superoxide dismutase and acidified sodium nitrite on infarct size following ischemia and reperfusion in dogs. Indian J Physiol Pharmacol. 1997;41:248–256. [PubMed] [Google Scholar]