Abstract
Objectives
The aim of this study was to explore the prognostic value of ki67 as a marker in patients with non-muscle invasive bladder cancer (NMIBC) treated with BCG.
Methods
Studies were systematically retrieved from the relevant databases (Web of Science, PubMed, Cochrane Library and Embase), and the expiry date was May 2017. The research steps referred to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis statement.
Results
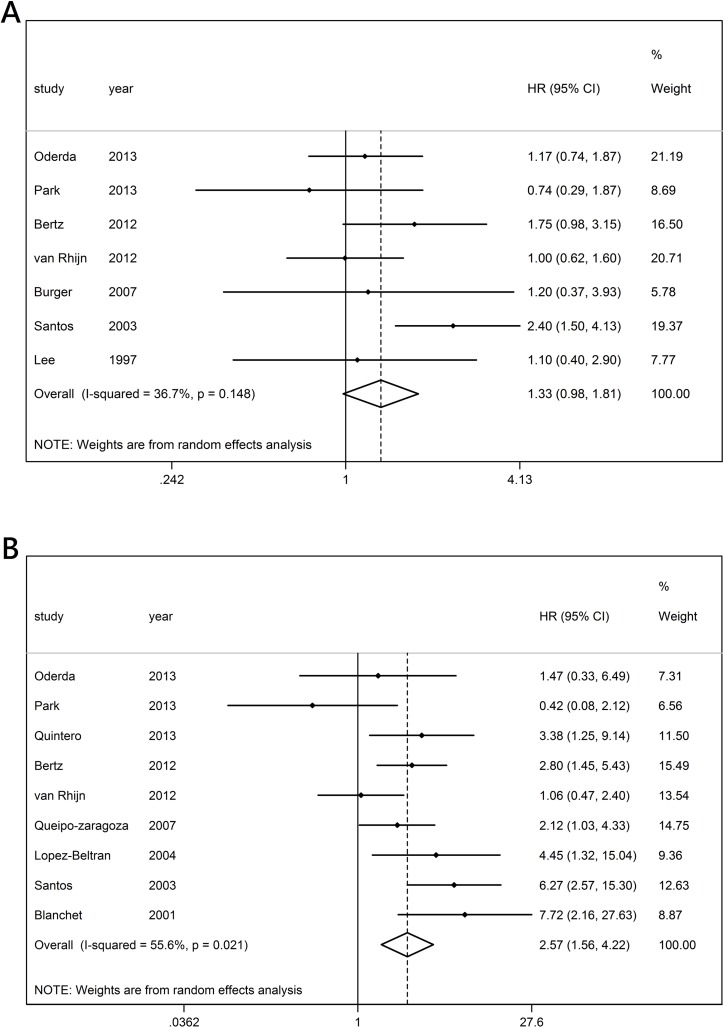
A total of 11 studies that complied with the inclusion criteria were included. The expression of ki67 was not statistically significantly associated with recurrence-free survival (RFS) (HR 1.331; 95% CI 0.980 to 1.809). No significant heterogeneity was found among all included studies (I2=36.7%, p=0.148). The expression of ki67 was statistically significantly associated with progression-free survival (PFS) (HR 2.567; 95% CI 1.562 to 4.219), and the overexpression of ki67 was the risk factor for PFS. Significant heterogeneity was noted among all the included studies (I2=55.6%, p=0.021). The studies that might cause heterogeneity were excluded using the Galbraith plot, and then the meta-analysis was performed again. The results showed that the expression of ki67 was still associated with PFS (HR 2.922; 95% CI 2.002 to 4.266).
Conclusions
The overexpression of ki67 was the risk factor for PFS, and the relationship between the expression of ki67 and RFS was not statistically significant in patients with NMIBC treated with BCG intravesical immunotherapy. Well-designed, prospective, with a large sample size are still needed to validate the findings.
Keywords: Ki67, meta-analysis, non-muscular-invasive bladder cancer, prognosis
Strengths and limitations of this study.
This meta-analysis and systematic review was performed via a strict literature search. It was the first meta-analysis to evaluate the prognostic value of ki67 in patients with NMIBC after transurethral resection and BCG intravesical immunotherapy.
The number of studies considered in the final meta-analysis was 11. This small sample size limited the potential analyses. The research did not consider the surgical skills mentioned in published studies.
Despite a systematic search strategy, the inclusion criteria excluded non-English documents and had language bias. The meta-regression analysis suggested no bias, but a selection bias was likely.
These limitations notwithstanding, the research can guide the follow-up research on immunohistochemical markers and clinical practice in NMIBC.
Introduction
Bladder cancer is one of the most common clinical urological tumours. It is a direct threat to the survival of patients with the disease. The incidence of bladder cancer varies across the world, with the highest rate in the developed communities.1 There was a total of 429 800 new cases of bladder cancer and 165 100 deaths worldwide in 2012. Bladder cancer occurs mostly in men, and about 10-fold variation in incidence rates has been reported internationally.2 About 70% of these patients have non-muscle invasive bladder cancer (NMIBC).3 Four major organisational guidelines on NMIBC, including the American Urological Association/Society of Urologic Oncology, the European Association of Urology, the National Comprehensive Cancer Network, and the National Institute for Health and Care Excellence guidelines, recommend that proper initial transurethral resection (TUR) of bladder tumour is a critical step in the initial management and staging of the disease.4 However, TUR surgery alone cannot solve the postoperative problems of NMIBC because of high recurrence rate and disease development.5 Postoperative TUR associated with BCG intravesical immunotherapy can prevent postoperative recurrence of NMIBC and significantly reduce the moderate and high risk of development of NMIBC.6 7 However, postoperative BCG intravesical immunotherapy still has some problems. The failure rate of BCG intravesical therapy in NMIBC is about 40%–50%.8 Furthermore, BCG has toxic side effects, such as hepatitis, pneumonitis, epididymitis/orchitis, abscess formation, bladder contracture, ureteral obstruction, BCG sepsis, leucopenia and haematuria.9 Therefore, BCG therapy should be individually performed, and the patients in whom BCG therapy is ineffective should be recognised in time. These patients or those with poor prognosis should receive radical cystectomy or any other therapy in time to avoid futile treatment and to alleviate pain. However, it is still difficult to recognise patients with no effect of TUR postoperative BCG intravesical immunotherapy due to the heterogeneity of bladder cancer and the individuality of patients.10 Therefore, it is necessary to find the prognostic factors for patients with NMIBC undergoing TUR and receiving BCG therapy.
The recurrence rate of bladder cancer treated with different therapies is between 50% and 80%, and about 15% of the low-grade tumour recurrence involves high-grade tumours.11 Cystoscopy should be performed periodically in patients so that the recurrent lesion can be detected in time. A reliable prognostic molecular marker can reduce the pain caused by cystoscopy. The absence of reliable prognostic markers for NMIBC, makes it hard to decide on the postoperative therapy in the clinic,12 which depends mainly on clinical guidelines and the physician’s experience. Currently, some of the published studies on immunohistochemical markers have evaluated the prognostic value of BCG intravesical immunotherapy on the patients receiving TUR first. The main immunohistochemical markers include ki67, p53, p27, pRb, CD9, CD20, E2F1 and so on.13 14 However, no immunohistochemical marker has been confirmed so far. The prognostic value of the ki67 antigen for the survival of patients with NMIBC receiving BCG intravesical immunotherapy has been controversial. For example, Kruger15 reported that the ki67 antigen was an independent predictive factor for the recurrence of pT1 stage tumour, but Oderde16 believed that ki67 was an independent predictive factor for the recurrence of all NMIBCs. Zlotta17 reported that ki67 antigen had no independent prognostic value in patients receiving BCG therapy. Saint18 retrospected on the recent 25-year published studies and believed that the independent prognostic factor for bladder cancer in patients receiving BCG therapy was not clear. An international consensus group listed various bladder cancer prognostic indexes by reviewing PubMed and considered that although some markers (such as ki67 and p53) could predict the recurrence and development of bladder cancer, the data still had heterogeneity. Thus, strict test criteria and clear statistical methods should be established for further evaluation.19
A meta-analysis can enlarge the sample size by integrating independent studies with small sample size, further increase the statistical efficacy and reduce the wrong conclusion caused by the small sample size.20 The aim of this study was to explore the prognostic value of ki67 as a marker in patients with NMIBC treated with BCG. Based on the literature search, this study was the first meta-analysis to evaluate the prognostic value of ki67 in patients with NMIBC treated with BCG.
Methods
This meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (online supplementary table S1).21 The present meta-analysis did not need approval because all the included published studies were approved by the ethics committees in the respective research institutes.
bmjopen-2017-019635supp003.pdf (213.3KB, pdf)
Literature retrieval strategy
The comprehensive literature search was performed on Web of Science, PubMed, Cochrane Library and Embase databases for relevant studies. The last quest was updated on 24 May 2017, with hand-searching to identify any potentially eligible studies that might have been missed. The following search strategy was adopted for each database: (“Urinary Bladder Neoplasms”(Mesh) OR “bladder cancer” OR “bladder carcinoma” OR “bladder tumour”) AND (“BCG Vaccine”(Mesh) OR “BCG” OR “Bacillus Calmette–Guérin”) AND (“ki67 antigen”(Mesh) OR “ki-67” OR “ki67” OR “MBI-1”). Filters were as follows: retrospective, array research, clinical trial, controlled clinical trial and randomised controlled trial. Free word retrieval strategy was used. The contents included the reference lists and relevant suggestive references while searching (online supplementary file S1).
bmjopen-2017-019635supp006.pdf (176.9KB, pdf)
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) Prospective or retrospective published studies evaluating the prognostic relationship between the expression of ki67 and NMIBC treated with BCG. (2) The expression of ki67 in tissues detected by immunohistochemistry analysis. (3) HRs and 95% CIs directly obtained from the published studies. (4) Published English studies. The exclusion criteria were as follows: (1) Review, systematic evaluation, case report, editorial and specialist experience. (2) Studies with no human subjects. (3) Published studies in which data could not be extracted or those having wrong data.
Data extraction and evaluation of literature quality
Based on the aforementioned criteria, two reviewers independently screened the published studies by reading titles and abstracts and got preliminary conclusions. If the conclusions were not consistent, the literature was discussed by all the authors to decide on its inclusion. Relevant information from the included published studies, such as first author, publication time, research country, sex, case number, age, follow-up date, disease stage, cut-off values, recurrence-free survival (RFS) and progression-free survival (PFS), was extracted. The Newcastle-Ottawa Scale (NOS) was used to evaluate the quality of all the published studies.22 Scores 0–3, 4–5 and 6–8 were accepted as low quality, medium quality and high quality, respectively.
Statistical methods
The measuring time and method of ki67 complied with the standard of clinical routine and pathological examination. Tumour tissue samples were taken in accordance with the standard surgical procedure and used for immunohistochemical analysis. RFS and PFS were the traditionally used statistical parameters. PFS was defined as the time from the beginning of treatment to the first progression. RFS was defined as the time from the removal of the lesion (or the randomisation of the clinical trial) until the recurrence or death of the tumour. The impact of the expression of ki67 on survival was quantified using the combined HRs and 95% CIs. The HR and 95% CI of each study were directly extracted from the original published study. Besides, Parmar and Tierney’s23 method was used to extract the data because some of the published studies did not directly provide HR and 95% CI. For example, some studies provided only the survival curve. In this meta-analysis, the DerSimonian-Laird random-effects model24 was used because only the random-effects model is suitable for large heterogeneity. Similar to traditional methods, HR>1 was considered as the prognostic risk factor for the overexpression of ki67, and HR<1 was a protective factor. A 95% CI<1 indicated a statistically significant difference in the relationship between the overexpression of ki67 and prognosis.
Heterogeneity was calculated according to χ2-based Q test and I 2 statistic,25 and was judged using the I 2 value (low heterogeneity: I2<25%; moderate heterogeneity: I2=25%–50%; large heterogeneity: I2>50%). Besides, a p value >0.05 was considered as low heterogeneity. Then, subgroup analysis based on regions, sample size, follow-up period, tumour grading, cut-off value, publication time and patient age was performed. A value of 1% was considered to be statistically significant in the subgroup analysis. A Galbraith plot was used to search published studies with heterogeneity,26 and the meta-analysis was again performed after excluding these published studies. Meanwhile, the factors causing heterogeneity were also explored using the residual maximum likelihood based random-effects meta-regression analysis.27 All the statistical analyses were performed using Stata V.12.0 software (StataCorp, College Station, Texas, USA), and two-sided test was used to evaluate the p value.
Estimation of publication bias
Begg’s plot and Egger’s test method were used to find possible publication bias. A p value <0.05 was considered to indicate publication bias.
Results
Literature screening
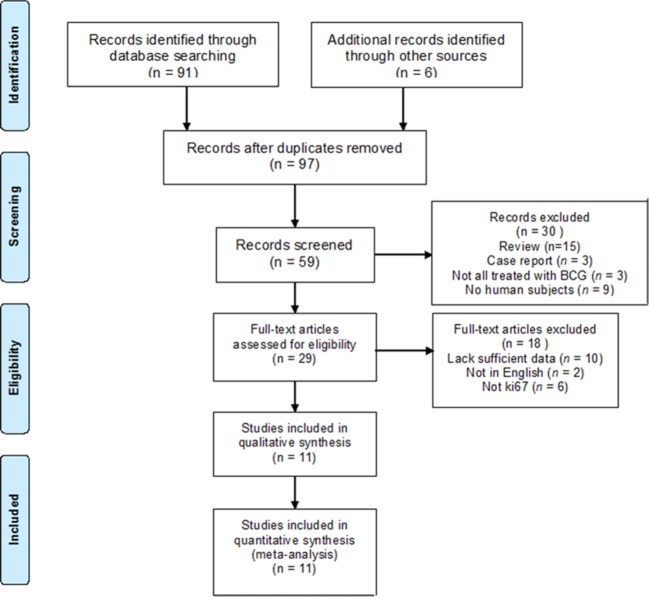
A total of 97 published studies were retrieved. Furthermore, 68 of them were excluded after duplicates were removed and records screened, and 18 were excluded after reading the full text (10 published studies from which HR and 95% CI could not be obtained, 2 non-English studies and 6 that did not use ki67 detection). Finally, 11 published studies were included in the meta-analysis (figure 1).
Figure 1.
Flow diagram of study selection.
Basic characteristics and quality evaluation of included published studies
The 11 included studies were published between 1997 and 2013, and the countries included Italy, South Korea, Spain, Germany, New Zealand, Canada, Portugal and France. The largest sample size was 309, and the smallest was 32. A total of 1321 patients were enrolled in this study. The follow-up period was more than 36 months, and the longest was 229 months. T1 was the main tumour grading, and the cut-off value ranged from 10.4% to 40%. Seven published studies reported patients' RFS, and nine reported PFS (table 1). One literature was scored as six stars by NOS, seven as seven stars and three as eight stars. The median of the NOS Score was 7 (table 2).
Table 1.
Main characteristics of all studies included in this meta-analysis
| Study | Year | Country | Male/ female |
No. of patients |
Age (years) |
Follow-up (month) | Stage | Cut-off | Survival analysis |
| Oderda16 | 2013 | Italy | 166/26 | 192 | 73.2 (SD 11.9) | 100 (2–229) | All NMIBC | 20% | RFS |
| Park14 | 2013 | Korea | 53/8 | 61 | 66 (31–85) | 60 (6–217) | T1G3 | 10.4% | RFS/PFS |
| Quintero45 | 2013 | Spain | 143/21 | 164 | 61 (29–93) | 75 (60–144) | Ta | 13% | PFS |
| Bertz12 | 2012 | Germany | 237/72 | 309 | 71.7 (38–87) | 49 (5–172) | pT1 | 15% | RFS/PFS |
| van Rhijn29 | 2012 | The Netherlands, Canada | 105/24 | 129 | 68.8 (SD 9.9) | 78 (39.6–110.4) | T1 | 25% | RFS/PFS |
| Burger46 | 2007 | Germany | 45/21 | 71 | 71 (52–94) | 39 (1–133) | T1/Ta | 15% | RFS |
| Queipo-Zaragoza47 | 2007 | Spain | 71/12 | 83 | 68.1 (SD 8.5) | All>36 | T1G3 | 40% | PFS |
| Lopez-Beltran48 | 2004 | Spain | 49/2 | 51 | 69.96 (49–89) | 63.82 (60–144) | T1G3 | 13% | PFS |
| Santos28 | 2003 | Portugal | 115/44 | 159 | 66 (21–88) | 46.5 (4–123) | pTa/pT1 | 18% | RFS/PFS |
| Blanchet49 | 2001 | France | - | 70 | 62.6 (21–84) | 64 (12–111) | pT1/pTa | 13% | PFS |
| Lee50 | 1997 | Korea | 28/4 | 32 | 57.1 (30–81) | All >24 | T1G2–3 | 20% | RFS |
NMIBC, non-muscle invasive bladder cancer; no., number; PFS, progression-free survival; RFS, recurrence-free survival.
Table 2.
Quality of the included studies assessed by the Newcastle-Ottawa Scale
| Study | Selection | Comparability | Exposure | Scores | |||||
| Adequate definition of cases |
Representativeness of cases |
Selection of controls |
Definition of controls |
Control for important factor |
Ascertainment of exposure |
Same method to ascertain as for cases and controls |
Non-response rate | ||
| Oderda16 | — | ☆ | ☆ | ☆ | ☆☆ | ☆ | ☆ | ☆ | 8 |
| Park14 | — | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | ☆ | 7 |
| Quintero45 | — | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | — | 6 |
| Bertz12 | — | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | ☆ | 7 |
| van Rhijn29 | — | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | ☆ | 7 |
| Burger46 | ☆ | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | — | 7 |
| Queipo-Zaragoza47 | — | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | ☆ | 7 |
| Lopez-Beltran48 | — | ☆ | ☆ | ☆ | ☆☆ | ☆ | ☆ | ☆ | 8 |
| Santos28 | — | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | ☆ | 7 |
| Blanchet49 | — | ☆ | ☆ | ☆ | ☆☆ | ☆ | ☆ | ☆ | 8 |
| Lee50 | — | ☆ | ☆ | — | ☆☆ | ☆ | ☆ | ☆ | 7 |
Influence of the expression of ki67 on RFS
Seven published studies reported the expression of ki67 and PFS results of patients with NMIBC treated with BCG. The meta-analysis indicated that ki67 had no statistically significant association with RFS (HR 1.331; 95% CI 0.980 to 1.809), and no heterogeneity among the included studies was reported (I 2=36.7%, p=0.148) (figure 2A). The subgroup analysis was performed based on the regions, sample size, follow-up period, stage, cut-off value, publication time and age. Meanwhile, all the original published analyses on the association between the expression of ki67 and RFS in patients with NMIBC treated with BCG were multivariate, and the HRs were adjusted. The stratification analysis by region indicated that ki67 was also significantly associated with RFS in Caucasians and a follow-up period shorter than 60 months (HR 1.441, 95% CI 1.014 to 2.047; HR 1.853, 95% CI 1.316 to 2.607) (table 3).
Figure 2.
Forest plots of HRs estimated for the relationship between the expression of ki67 and recurrence-free survival (A) or progression-free survival (B) among patients with non-muscle invasive bladder cancer treated with BCG.
Table 3.
Subgroup results of RFS and heterogeneity test
| Variables | Analysis number | HR (95% CI) | Heterogeneity test | ||
| Q | P | I2 (%) | |||
| Total RFS | 7 | 1.331 (0.980 to 1.809) | 9.48 | 0.148 | 36.7 |
| Region | |||||
| Asian | 2 | 0.892 (0.454 to 1.752) | 0.32 | 0.570 | 0.0 |
| Caucasian | 5 | 1.441 (1.014 to 2.047) | 7.52 | 0.111 | 46.8 |
| Sample size | |||||
| >100 | 4 | 1.466 (0.986 to 2.181) | 7.44 | 0.059 | 59.7 |
| ≤100 | 3 | 0.959 (0.534 to 1.725) | 0.51 | 0.777 | 0.0 |
| Follow-up (month) | |||||
| ≥60 | 3 | 1.036 (0.758 to 1.415) | 0.79 | 0.674 | 0.0 |
| <60 | 4 | 1.853 (1.316 to 2.607) | 2.62 | 0.453 | 0.0 |
| Stage | |||||
| All NMIBC | 3 | 1.575 (0.915 to 2.711) | 4.44 | 0.109 | 54.9 |
| Others | 4 | 1.153 (0.821 to 1.620) | 3.21 | 0.360 | 6.6 |
| Cut-off | |||||
| 15% | 2 | 1.625 (0.963 to 2.743) | 0.31 | 0.575 | 0.0 |
| Others | 5 | 1.252 (0.839 to 1.869) | 8.56 | 0.073 | 53.3 |
| Publication year | |||||
| ≥2012 | 4 | 1.164 (0.874 to 1.550) | 3.20 | 0.362 | 6.3 |
| <2012 | 3 | 1.774 (1.046 to 3.008) | 2.57 | 0.277 | 22.1 |
| Patient age (year) | |||||
| ≥70 | 3 | 1.352 (0.955 to 1.913) | 1.16 | 0.559 | 0.0 |
| <70 | 4 | 1.256 (0.717 to 2.198) | 8.32 | 0.040 | 63.9 |
NMIBC, non-muscle invasive bladder cancer; RFS, recurrence-free survival.
Influence of the expression of ki67 on PFS
A total of nine published studies reported the expression of ki67 and PFS results of patients with NMIBC treated with BCG. The meta-analysis indicated that ki67 had no statistically significant association with RFS (HR 2.567, 95% CI 1.562 to 4.219), and the overexpression of ki67 was the risk factor for PFS. Statistically significant heterogeneity was found among all the included studies (I2=55.6%, p=0.021) (figure 2B). The subgroup analysis was performed based on the regions, sample size, follow-up period, stage, cut-off value, publication time and age. However, the data extracted from six original published analyses on the association between the expression of ki67 and PFS in patients with NMIBC treated with BCG were multivariate with adjusted HRs, whereas the data from three original published analyses were univariate with unadjusted HRs. In the stratified analyses by region, sample size, follow-up time, stage, cut-off, publication year and patient age, significant associations were observed in the studies with the Caucasian subgroup, sample size >100, follow-up period <60 months, other cut-offs and two subgroups based on age (HR 1.97, 95% CI 1.04 to 3.74; HR 2.37, 95% CI 1.23 to 4.55; HR 2.49, 95% CI 1.19 to 5.21; HR 2.515, 95% CI 1.382 to 4.576; HR 2.800, 95% CI 1.447 to 5.418; and HR 2.654, 95% CI 1.381 to 5.100, respectively). However, significant associations were also observed in both multivariate and univariate analyses (HR 2.10, 95% CI 1.07 to 1.12; HR 2.80, 95% CI 1.65 to 7.85, respectively), and the effect size suggested the same outcomes (HR 2.567, 95% CI 1.562 to 4.219) (table 4).
Table 4.
Subgroup results of PFS and heterogeneity test
| Variables | Analysis number | HR (95% CI) | Heterogeneity test | ||
| Q | P value | I2 (%) | |||
| Total PFS | 9 | 2.567 (1.562 to 4.219) | 18.10 | 0.021 | 55.6 |
| Region | |||||
| Asian | 1 | 0.421 (0.084 to 2.114) | 0.00 | ||
| Caucasian | 8 | 2.883 (1.830 to 4.544) | 12.99 | 0.072 | 46.1 |
| Sample size | |||||
| >100 | 5 | 2.559 (1.372 to 4.774) | 9.26 | 0.055 | 56.8 |
| ≤100 | 4 | 2.536 (0.943 to 6.818) | 8.75 | 0.033 | 65.7 |
| Follow-up (month) | |||||
| ≥60 | 6 | 2.153 (0.984 to 4.710) | 13.08 | 0.023 | 61.8 |
| <60 | 3 | 3.158 (1.774 to 5.623) | 3.56 | 0.169 | 43.8 |
| Stage | |||||
| All NMIBC | 3 | 4.673 (1.938 to 11.264) | 3.29 | 0.193 | 39.2 |
| Others | 6 | 2.044 (1.213 to 15.040) | 9.58 | 0.088 | 47.8 |
| Cut-off | |||||
| 15% | 1 | 2.800 (1.447 to 5.418) | 0.00 | ||
| Others | 8 | 2.515 (1.382 to 4.576) | 17.92 | 0.012 | 60.9 |
| Publication year | |||||
| ≥2012 | 5 | 1.685 (0.883 to 3.215) | 8.04 | 0.090 | 50.2 |
| <2012 | 4 | 4.176 (2.209 to 7.884) | 5.00 | 0.172 | 40.0 |
| Patient age (year) | |||||
| ≥70 | 2 | 2.519 (1.377 to 4.606) | 0.60 | 0.438 | 0.0 |
| <70 | 7 | 2.654 (1.381 to 5.100) | 17.40 | 0.008 | 65.5 |
| Multivariate/univariate | |||||
| Multivariate | 6 | 2.101 (1.070 to 1.121) | 13.83 | 0.031 | 63.8 |
| Univariate | 3 | 2.803 (1.652 to 7.856) | 3.38 | 0.001 | 40.8 |
NMIBC, non-muscle invasive bladder cancer; PFS, progression-free survival.
Galbraith plot
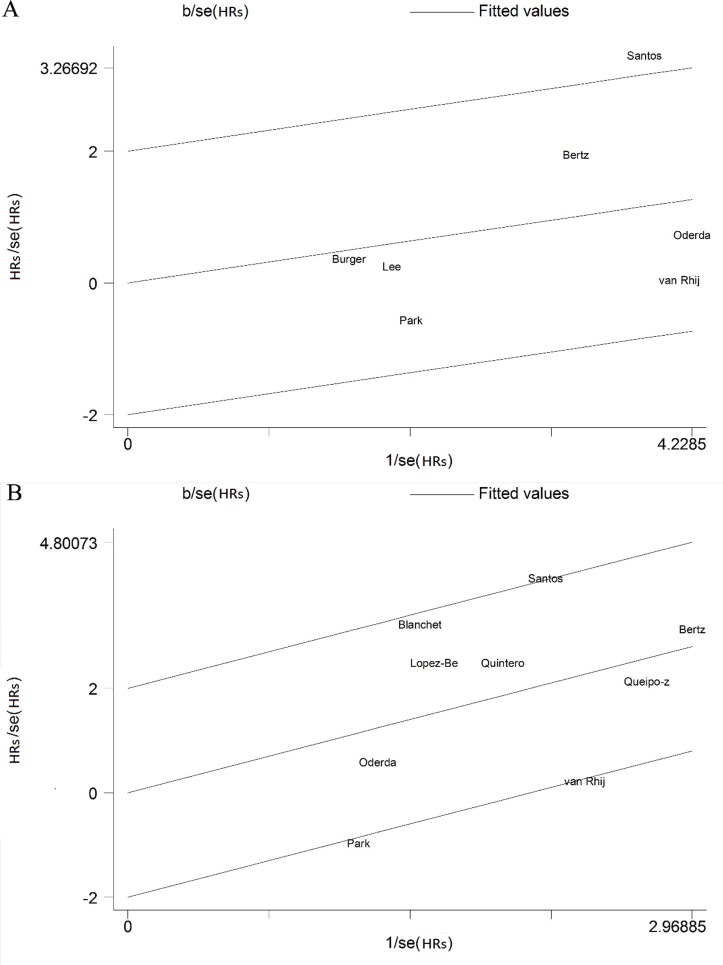
Using Galbraith plot (figure 3A), it was found that the study by Santos28 was the main reason for the heterogeneity of RFS. After the aforementioned study was excluded, the remaining RFS studies had no significant heterogeneity according to the new meta-analysis (I 2=0.0%, p=0.667). However, the expression of ki67 still had no statistically significant association with RFS (HR 1.161, 95% CI 0.896 to 1.504) (online supplementary figure S1). Using the Galbraith plot (figure 3B), it was found that the Santos,28 Park14 and van Rhijn29 studies were the main reason for the heterogeneity of PFS. After these studies were excluded, the remaining RFS studies had no significant heterogeneity according to the new meta-analysis (I 2=0.0%, p=0.497). The expression of ki67 still had a statistically significant association with PFS (HR 2.922, 95% CI 2.002 to 4.266) (online supplementary figure S2).
Figure 3.
Galbraith plot analysis was used to evaluate heterogeneity and recurrence-free survival (A) or progression-free survival (B).
bmjopen-2017-019635supp001.pdf (629.6KB, pdf)
bmjopen-2017-019635supp002.pdf (601.8KB, pdf)
Meta-regression analysis
The meta-regression analysis indicated that the factors influencing heterogeneity (publication time, research region, sample size, stage, cut-off value, age and follow-up period) might not be the reason for RFS heterogeneity. Publication time was the reason for heterogeneity of PFS (P=0.036), but other factors were not (online supplementary table S2).
bmjopen-2017-019635supp004.pdf (176.3KB, pdf)
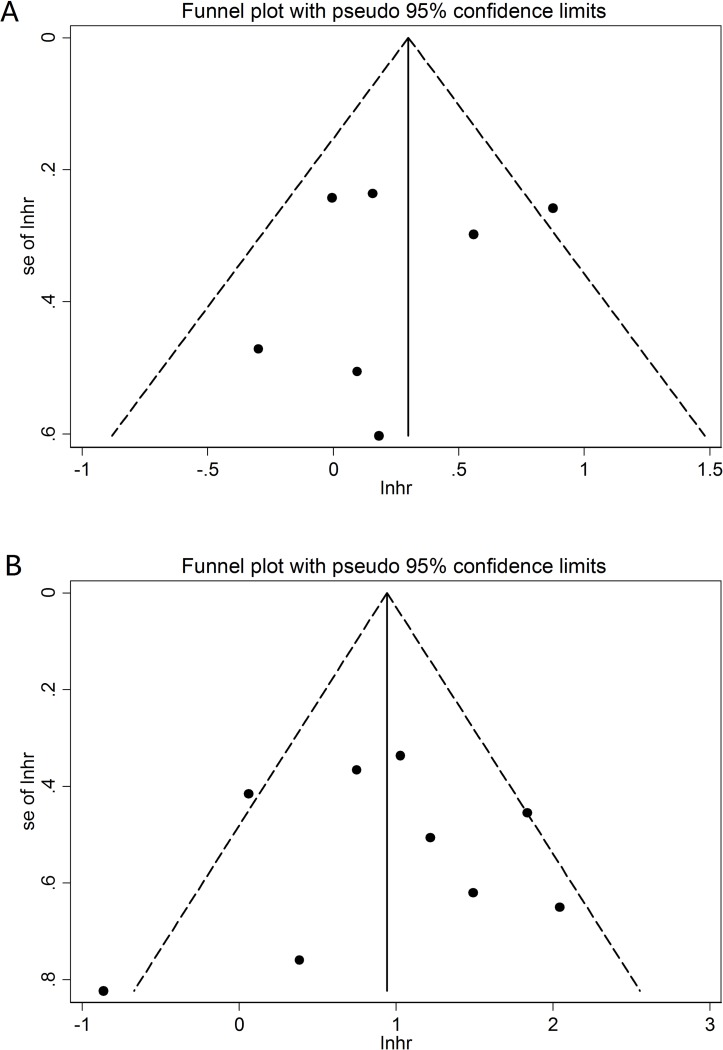
Publication bias
The funnel plot (figure 4A) was basically symmetrical for RFS. The results of Begg’s test and Egger’s test showed p=0.761 (Begg’s) and p=0.601 (Egger’s). The funnel plot (figure 4B) was also basically symmetrical for PFS. The results of Begg’s test and Egger’s test showed p=0.917 (Begg’s) and p=0.964 (Egger’s).
Figure 4.
Funnel plots of the expression of ki67 and recurrence-free survival (A) or progression-free survival (B).
Discussion
A total of 11 published studies with 1321 cases complying with the inclusion criteria were included in this meta-analysis. The results of the meta-analysis indicated that the expression of ki67 had no statistically significant association with RFS, but it was significantly associated with PFS. The overexpression of ki67 was the risk factor for PFS. It suggested that ki67 was the prognostic predictive marker in patients with NMIBC treated with BCG. Besides, the aforementioned conditions did not change after excluding the published studies, possibly leading to heterogeneity and reperforming of the meta-analysis. It further proved that the result of the aforementioned meta-analysis was stable, that is, the overexpression of ki67 was the risk factor for PFS. In the Caucasian subgroup for PFS, racial classification and regional factors might be crucial in the prognosis of patients with NMIBC after BCG therapy. This might be related to the existence of different drug gene susceptibilities in people belonging to different races and living areas. The two subgroups were based on age in PFS, suggesting that age might be the important factor influencing the prognosis of bladder cancer. This also complies with our clinical practice. The elder the patient, the worse the prognosis. Several factors led to heterogeneity in the aforementioned subgroup analysis: (1) Due to the influence of race and environment, the documents included in this study came from different regions and countries. A large number of studies confirmed the differences in disease susceptibility between ethnic groups and regions. (2) Differences existed in the operation of healthcare workers in TUR and BGC intravesical immunotherapy because of different regions and different clinicians, such as surgical clearance of the tumour. It is not often easy to completely remove a tumour with a broad-based surface and also depends on the surgeon’s experience and surgical skills. In addition, the quality of BCG manufacturers may vary from region to region. (3) Different literature might include the bias of research object, research design, measuring instrument and so on. However, in general, heterogeneity did not affect the conclusion. Besides, the meta-regression analysis indicated publication time as the reason for PFS heterogeneity. It might be related to the improvement in testing technology, research level, and the quality and number of published studies, facilitating follow-up studies. As all the original data extracted from published studies on the association between the expression of ki67 and RFS in patients with NMIBC treated with BCG were multivariate, the result was considered to be precise because the HRs were adjusted, excluding the confounding factors such as age and gender. However, the original data extracted from published analyses on the association between the expression of ki67 and PFS were both multivariate and univariate. It was believed that the aforementioned adjustments did not have a significant impact on meta-analyses. Besides, according to the funnel plot, Begg’s test and Egger’s test, the included studies had no statistically significant publication bias. Thus, the reliability of the present meta-analysis was high.
In 2016, the European Association of Urology30 recommended a scoring system for the prognostic evaluation of NMIBC based on six clinical and pathological factors proposed by the European Organisation for the Research and Treatment of Cancer (EORTC) Genito-Urinary Group, including number of tumours, tumour size, prior recurrence rate, T category, presence of concurrent carcinoma in situ and tumour grade (online supplementary table S3). The tumours were categorised into low-risk, intermediate-risk and high-risk using this assessment system to evaluate the prognosis. For the patients after BCG therapy, the European Association of Urology recommended another risk calculator developed by Club Urologico Espanol de Tratamiento Oncologico (CUETO) and EORTC. This calculator was based on gender, age, recurrent tumour, number of tumours, T category, associated Tis and grade. The CUETO risk calculator can be found at http://www.aeu.es/Cueto.html. The recommended level was B grade for the two scales for patients with NMIBC, whether used alone or combined. The two scales could be used together in the clinic. When using the CUETO Scale, the calculated recurrent risk was lower than that from the EORTC Scale,31 which might be related to the special design in the CUETO Scale for the patients receiving BCG intravesical immunotherapy. However, the scoring system that depended only on clinical and pathological factors could not accurately evaluate the prognosis of patients with bladder cancer in the T1 stage due to the independence of disease condition in each patient.32 The markers regulated at the genetic level may judge the prognosis of patients with bladder cancer with the development of precision medicine. A reliable marker helps recognise, in time, the high-risk patients who have failed BCG intravesical immunotherapy. Hence, these patients can undergo radical cystectomy or other treatments, in time. Unfortunately, no prognostic marker has currently been applied in the clinic. The results of this study potentially help to remind clinicians that patients with high expression of ki67 may need to develop more personalised follow-up plans, such as shorter follow-up and cystoscopy cycles. Patients with high risk of clinical evaluation of the guidelines and overexpression of ki67 may need to promptly change the treatment strategy.
bmjopen-2017-019635supp005.pdf (165KB, pdf)
Ki67 is a nucleoprotein that can be detected in the cell cycles except during the G0 phase.33 The expression of human ki67 protein is closely related to proliferation. Therefore, it is an ideal marker to confirm the growth fraction of specific cell colonies.34 Ki67 is a widely known amplified biomarker. The ki67 monoclonal antibody can be detected by the immunohistochemical method.35 Ki67 has been proved to be a good proliferation marker in different cancers, including bladder cancer.36
So far, some meta-analyses have studied the effect of ki67 on the prognostic quality of life of patients with oesophageal cancer, breast cancer, epithelial ovarian cancer and so on.37–39 Some studies have also focused on other aspects of bladder cancer. Using meta-analysis, Luo40 believed that a high reactivity of ki67 could predict the poor prognosis in patients with bladder cancer. The univariate analysis showed that cancer-specific survival, disease-free survival, overall survival, PFS and RFS had a significant association with poor prognosis in patients with a high reactivity of ki67. However, this study included all types of bladder tumours and all the therapies for NMIBC. Currently, the bladder cancer treated in the clinic is mainly NMIBC. Thus, most of the applied therapy is TUR combined with installations of chemotherapy or BCG intravesical immunotherapy based on the patients' conditions. Therefore, this analysis had a certain limitation in the prognosis of patients with NMIBC after BCG intravesical immunotherapy.
Currently, few evidence-based studies focused on the prognosis of patients with NMIBC after BCG intravesical immunotherapy. Using meta-analysis, Zhou41analysed the association between the expression of p53 and quality of life of patients with NMIBC after BCG intravesical immunotherapy. They believed that the overexpression of p53 in patients with NMIBC treated with BCG might be associated with RFS, especially in the Asian population. Similarly, Du42 also carried out a meta-analysis on the relationship between p53 status and NMIBC in the T1 stage and believed that the overexpression of p53 might be related to the development of NMIBC. The present study indicated that the overexpression of ki67 was the risk factor for PFS, but the expression of ki67 had no statistically significant association with RFS. P53 is the most common inactivated tumour suppressor gene in tumour cells.43 The inactivation of p53 may cause cell abnormal hyperplasia and cancerisation. The variation in p53 results in enhanced proliferation, invasion and metabolism.44 The increase in the expression of ki67, as a cell proliferation marker suggests enhanced proliferation.34 As a tumour suppressor gene with complicated function, p53 has a wider range of effects. The accuracy in the prediction of quality of life may not be more appropriate compared with ki67. The genetic difference between Asians and Caucasians suggests that different prediction systems should be built for different races. Besides, p27, E2F1, ezrin and CK20 were also studied in other investigations for predicting NMIBC prognosis, which could be explored further comparing the advantages of using them alone or combined.
However, this study still had some limitations. First, the published studies included involved different populations, used similar detection equipment and had different cut-off values. All these reasons might have led to heterogeneity. Further, the sample size of the meta-analysis also limited its significance. Second, the meta-analysis included published English studies. Although Begg’s test and Egger’s test did not suggest publication bias, this study was still influenced by some bias. Finally, the surgical skills were different in the different studies, affecting judgement regarding the effectiveness of BCG.
Conclusions
The overexpression of ki67 was the risk factor for PFS in patients with NMIBC after TUR and BCG intravesical immunotherapy, but the relationship between the expression of ki67 and RFS was not statistically significant. Due to the aforementioned limitations of the present study, randomised controlled trials with a large sample size are still required to validate the results.
Supplementary Material
Footnotes
Contributors: YH, NW and XZ conceived and designed the experiments. XC and YD extracted the data. YH, NW and XZ analysed the data. ZD, JW and XC contributed reagents/materials/analysis tools. YH and NW wrote the paper. XZ critically revised the report.
Funding: This study was supported by grants from the International S&T Cooperation Program of China (ISTCP) (Grant No.2014DFE30010).
Disclaimer: The contents of the present study are solely the responsibility of the author. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing interests: None declared.
Patient consent: Not required.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: Extra data can be accessed via the Dryad data repository at http://datadryad.org/ with https://doi.org/10.5061/dryad.hf06q72.
References
- 1. Ploeg M, Aben KK, Kiemeney LA. The present and future burden of urinary bladder cancer in the world. World J Urol 2009;27:289–93. 10.1007/s00345-009-0383-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87–108. 10.3322/caac.21262 [DOI] [PubMed] [Google Scholar]
- 3. Ro JY, Staerkel GA, Ayala AG. Cytologic and histologic features of superficial bladder cancer. Urol Clin North Am 1992;19:435–53. [PubMed] [Google Scholar]
- 4. Woldu SL, Bagrodia A, Lotan Y. Guideline of guidelines: non-muscle-invasive bladder cancer. BJU Int 2017;119:371–80. 10.1111/bju.13760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Brausi M, Collette L, Kurth K, et al. Variability in the recurrence rate at first follow-up cystoscopy after TUR in stage Ta T1 transitional cell carcinoma of the bladder: a combined analysis of seven EORTC studies. Eur Urol 2002;41:523–31. 10.1016/S0302-2838(02)00068-4 [DOI] [PubMed] [Google Scholar]
- 6. Järvinen R, Kaasinen E, Sankila A, et al. Long-term efficacy of maintenance bacillus Calmette-Guérin versus maintenance mitomycin C instillation therapy in frequently recurrent TaT1 tumours without carcinoma in situ: a subgroup analysis of the prospective, randomised FinnBladder I study with a 20-year follow-up. Eur Urol 2009;56:260–5. 10.1016/j.eururo.2009.04.009 [DOI] [PubMed] [Google Scholar]
- 7. Sylvester RJ, Brausi MA, Kirkels WJ, et al. Long-term efficacy results of EORTC genito-urinary group randomized phase 3 study 30911 comparing intravesical instillations of epirubicin, bacillus Calmette-Guérin, and bacillus Calmette-Guérin plus isoniazid in patients with intermediate- and high-risk stage Ta T1 urothelial carcinoma of the bladder. Eur Urol 2010;57:766–73. 10.1016/j.eururo.2009.12.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gual Frau J, Palou J, Rodríguez O, et al. Failure of Bacillus Calmette-Guérin therapy in non-muscle-invasive bladder cancer: definition and treatment options. Arch Esp Urol 2016;69:423–33. [PubMed] [Google Scholar]
- 9. Boyd LA. Intravesical Bacillus Calmette-Guerin for treating bladder cancer. Urol Nurs 2003;23:189–91. 99; quiz 92. [PubMed] [Google Scholar]
- 10. Alkhateeb SS, Neill M, Bar-Moshe S, et al. Long-term prognostic value of the combination of EORTC risk group calculator and molecular markers in non-muscle-invasive bladder cancer patients treated with intravesical Bacille Calmette-Guérin. Urol Ann 2011;3:119–26. 10.4103/0974-7796.84954 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Mariappan P, Smith G. A surveillance schedule for G1Ta bladder cancer allowing efficient use of check cystoscopy and safe discharge at 5 years based on a 25-year prospective database. J Urol 2005;173:1108–11. 10.1097/01.ju.0000149163.08521.69 [DOI] [PubMed] [Google Scholar]
- 12. Bertz S, Otto W, Denzinger S, et al. Combination of CK20 and Ki-67 immunostaining analysis predicts recurrence, progression, and cancer-specific survival in pT1 urothelial bladder cancer. Eur Urol 2014;65:218–26. 10.1016/j.eururo.2012.05.033 [DOI] [PubMed] [Google Scholar]
- 13. Kim K, Cho YM, Park BH, et al. Histological and immunohistochemical markers for progression prediction in transurethrally resected high-grade non-muscle invasive bladder cancer. Int J Clin Exp Pathol 2015;8:743–50. [PMC free article] [PubMed] [Google Scholar]
- 14. Park J, Song C, Shin E, et al. Do molecular biomarkers have prognostic value in primary T1G3 bladder cancer treated with bacillus Calmette-Guerin intravesical therapy? Urol Oncol 2013;31:849–56. 10.1016/j.urolonc.2011.06.004 [DOI] [PubMed] [Google Scholar]
- 15. Krüger S, Thorns C, Stöcker W, et al. Prognostic value of MCM2 immunoreactivity in stage T1 transitional cell carcinoma of the bladder. Eur Urol 2003;43:138–45. 10.1016/S0302-2838(02)00580-8 [DOI] [PubMed] [Google Scholar]
- 16. Oderda M, Ricceri F, Pisano F, et al. Prognostic factors including Ki-67 and p53 in Bacillus Calmette-Guérin-treated non-muscle-invasive bladder cancer: a prospective study. Urol Int 2013;90:184–90. 10.1159/000343431 [DOI] [PubMed] [Google Scholar]
- 17. Zlotta AR, Noel JC, Fayt I, et al. Correlation and prognostic significance of p53, p21WAF1/CIP1 and Ki-67 expression in patients with superficial bladder tumors treated with bacillus Calmette-Guerin intravesical therapy. J Urol 1999;161:792–8. 10.1016/S0022-5347(01)61770-1 [DOI] [PubMed] [Google Scholar]
- 18. Saint F, Salomon L, Quintela R, et al. Do prognostic parameters of remission versus relapse after Bacillus Calmette-Guérin (BCG) immunotherapy exist?. analysis of a quarter century of literature. Eur Urol 2003;43:351–61. discussion 60-1 10.1016/S0302-2838(03)00048-4 [DOI] [PubMed] [Google Scholar]
- 19. Habuchi T, Marberger M, Droller MJ, et al. Prognostic markers for bladder cancer: International Consensus Panel on bladder tumor markers. Urology 2005;66:64–74. 10.1016/j.urology.2005.08.065 [DOI] [PubMed] [Google Scholar]
- 20. Button KS, Ioannidis JP, Mokrysz C, et al. Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci 2013;14:365–76. 10.1038/nrn3475 [DOI] [PubMed] [Google Scholar]
- 21. Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 2009;6:e1000100 10.1371/journal.pmed.1000100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 2010;25:603–5. 10.1007/s10654-010-9491-z [DOI] [PubMed] [Google Scholar]
- 23. Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 1998;17:2815–34. 10.1002/(SICI)1097-0258(19981230)17:24<2815::AID-SIM110>3.0.CO;2-8 [DOI] [PubMed] [Google Scholar]
- 24. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177–88. 10.1016/0197-2456(86)90046-2 [DOI] [PubMed] [Google Scholar]
- 25. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med 2002;21:1539–58. 10.1002/sim.1186 [DOI] [PubMed] [Google Scholar]
- 26. Bax L, Ikeda N, Fukui N, et al. More than numbers: the power of graphs in meta-analysis. Am J Epidemiol 2009;169:249–55. 10.1093/aje/kwn340 [DOI] [PubMed] [Google Scholar]
- 27. Xu X, Cheng Y, Li S, et al. Dietary carrot consumption and the risk of prostate cancer. Eur J Nutr 2014;53:1615–23. 10.1007/s00394-014-0667-2 [DOI] [PubMed] [Google Scholar]
- 28. Santos L, Amaro T, Costa C, et al. Ki-67 index enhances the prognostic accuracy of the urothelial superficial bladder carcinoma risk group classification. Int J Cancer 2003;105:267–72. 10.1002/ijc.11049 [DOI] [PubMed] [Google Scholar]
- 29. van Rhijn BW, Liu L, Vis AN, et al. Prognostic value of molecular markers, sub-stage and european organisation for the research and treatment of cancer risk scores in primary T1 bladder cancer. BJU Int 2012;110:1169–76. 10.1111/j.1464-410X.2012.10996.x [DOI] [PubMed] [Google Scholar]
- 30. Babjuk M, Böhle A, Burger M, et al. EAU Guidelines on Non-Muscle-invasive Urothelial Carcinoma of the Bladder: Update 2016. Eur Urol 2017;71:447–61. 10.1016/j.eururo.2016.05.041 [DOI] [PubMed] [Google Scholar]
- 31. Fernandez-Gomez J, Madero R, Solsona E, et al. Predicting nonmuscle invasive bladder cancer recurrence and progression in patients treated with bacillus Calmette-Guerin: the CUETO scoring model. J Urol 2009;182:2195–203. 10.1016/j.juro.2009.07.016 [DOI] [PubMed] [Google Scholar]
- 32. Kiemeney LA, Witjes JA, Heijbroek RP, et al. Predictability of recurrent and progressive disease in individual patients with primary superficial bladder cancer. J Urol 1993;150:60–4. 10.1016/S0022-5347(17)35397-1 [DOI] [PubMed] [Google Scholar]
- 33. Gerdes J, Lemke H, Baisch H, et al. Cell cycle analysis of a cell proliferation-associated human nuclear antigen defined by the monoclonal antibody Ki-67. J Immunol 1984;133:1710–5. [PubMed] [Google Scholar]
- 34. Scholzen T, Gerdes J. The Ki-67 protein: from the known and the unknown. J Cell Physiol 2000;182:311–22. 10.1002/(SICI)1097-4652(200003)182:3<311::AID-JCP1>3.0.CO;2-9 [DOI] [PubMed] [Google Scholar]
- 35. Gerdes J, Li L, Schlueter C, et al. Immunobiochemical and molecular biologic characterization of the cell proliferation-associated nuclear antigen that is defined by monoclonal antibody Ki-67. Am J Pathol 1991;138:867–73. [PMC free article] [PubMed] [Google Scholar]
- 36. Rodríguez-Alonso A, Pita-Fernández S, González-Carreró J, et al. p53 and ki67 expression as prognostic factors for cancer-related survival in stage T1 transitional cell bladder carcinoma. Eur Urol 2002;41:182–9. discussion 88-9 10.1016/S0302-2838(01)00038-0 [DOI] [PubMed] [Google Scholar]
- 37. McCormick Matthews LH, Noble F, Tod J, et al. Systematic review and meta-analysis of immunohistochemical prognostic biomarkers in resected oesophageal adenocarcinoma. Br J Cancer 2015;113:107–18. 10.1038/bjc.2015.179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Gui Y, Xu S, Yang X, et al. A meta-analysis of biomarkers for the prognosis of triple-negative breast cancer patients. Biomark Med 2016;10:771–90. 10.2217/bmm-2015-0064 [DOI] [PubMed] [Google Scholar]
- 39. Qiu HL, Deng SZ, Li C, et al. High expression of KIF14 is associated with poor prognosis in patients with epithelial ovarian cancer. Eur Rev Med Pharmacol Sci 2017;21:239–45. [PubMed] [Google Scholar]
- 40. Luo Y, Zhang X, Mo M, et al. High Ki-67 immunohistochemical reactivity correlates with poor prognosis in bladder carcinoma: a comprehensive meta-analysis with 13,053 patients involved. Medicine 2016;95:e3337 10.1097/MD.0000000000003337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Zhou X, Zhang G, Tian Y. p53 Status correlates with the risk of recurrence in non-muscle invasive bladder cancers treated with Bacillus Calmette-Guérin: a meta-analysis. PLoS One 2015;10:e0119476 10.1371/journal.pone.0119476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Du J, Wang SH, Yang Q, et al. p53 status correlates with the risk of progression in stage T1 bladder cancer: a meta-analysis. World J Surg Oncol 2016;14:137 10.1186/s12957-016-0890-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Fridman JS, Lowe SW. Control of apoptosis by p53. Oncogene 2003;22:9030–40. 10.1038/sj.onc.1207116 [DOI] [PubMed] [Google Scholar]
- 44. Muller PA, Vousden KH, Norman JC. p53 and its mutants in tumor cell migration and invasion. J Cell Biol 2011;192:209–18. 10.1083/jcb.201009059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Quintero A, Alvarez-Kindelan J, Luque RJ, et al. Ki-67 MIB1 labelling index and the prognosis of primary TaT1 urothelial cell carcinoma of the bladder. J Clin Pathol 2006;59:83–8. 10.1136/jcp.2004.022939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Burger M, Denzinger S, Hartmann A, et al. Mcm2 predicts recurrence hazard in stage Ta/T1 bladder cancer more accurately than CK20, Ki67 and histological grade. Br J Cancer 2007;96:1711–5. 10.1038/sj.bjc.6603784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Queipo-Zaragoza JA, Ruiz-Cerda JL, Vera-Donoso CD, et al. Prognostic value of p53, Ki-67, microstaging and microvessel density in pT1G3 bladder tumors: creation of risk groups for progression. Scand J Urol Nephrol 2007;41:283–9. 10.1080/00365590601183055 [DOI] [PubMed] [Google Scholar]
- 48. Lopez-Beltran A, Luque RJ, Alvarez-Kindelan J, et al. Prognostic factors in stage T1 grade 3 bladder cancer survival: the role of G1-S modulators (p53, p21Waf1, p27kip1, Cyclin D1, and Cyclin D3) and proliferation index (ki67-MIB1). Eur Urol 2004;45:606–12. 10.1016/j.eururo.2003.11.011 [DOI] [PubMed] [Google Scholar]
- 49. Blanchet P, Droupy S, Eschwege P, et al. Prospective evaluation of Ki-67 labeling in predicting the recurrence and progression of superficial bladder transitional cell carcinoma. Eur Urol 2001;40:169–75. 10.1159/000049768 [DOI] [PubMed] [Google Scholar]
- 50. Lee E, Park I, Lee C. Prognostic markers of intravesical bacillus Calmette-Guérin therapy for multiple, high-grade, stage T1 bladder cancers. Int J Urol 1997;4:552–6. 10.1111/j.1442-2042.1997.tb00307.x [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2017-019635supp003.pdf (213.3KB, pdf)
bmjopen-2017-019635supp006.pdf (176.9KB, pdf)
bmjopen-2017-019635supp001.pdf (629.6KB, pdf)
bmjopen-2017-019635supp002.pdf (601.8KB, pdf)
bmjopen-2017-019635supp004.pdf (176.3KB, pdf)
bmjopen-2017-019635supp005.pdf (165KB, pdf)