Abstract
The efficient transduction of specific genes into natural killer (NK) cells has been a major challenge. Successful transductions are critical to defining the role of the gene of interest in the development, differentiation, and function of NK cells. Recent advances related to chimeric antigen receptors (CARs) in cancer immunotherapy accentuate the need for an efficient method to deliver exogenous genes to effector lymphocytes. The efficiencies of lentiviral-mediated gene transductions into primary human or mouse NK cells remain significantly low, which is a major limiting factor. Recent advances using cationic polymers, such as polybrene, show an improved gene transduction efficiency in T cells. However, these products failed to improve the transduction efficiencies of NK cells. This work shows that dextran, a branched glucan polysaccharide, significantly improves the transduction efficiency of human and mouse primary NK cells. This highly reproducible transduction methodology provides a competent tool for transducing human primary NK cells, which can vastly improve clinical gene delivery applications and thus NK cell-based cancer immunotherapy.
Keywords: Immunology, Issue 131, Transduction, primary NK cells, dextran, lentivirus, genetically modified, immunotherapy
Introduction
Natural killer (NK) cells are the major lymphocytic population of the innate immune system1. NK cells function as the first-line defenders of the host immune response against tumors and infections2,3,4. NK cells also play a central role in the development of tolerance through the secretion of potent cytokines and chemokines5. Due to their potent ability to target and eliminate tumor cells, multiple clinical trials are being conducted to evaluate donor-derived human NK cells as an adoptive immunotherapy for cancer6,7. In contrast to T cells, the developmental biology of NK cells has yet to be well-characterized8. This lack of knowledge is partially due to the absence of efficient techniques that deliver genes of interest to mouse or human primary NK cells. For these reasons, most NK-cell studies have been conducted in cell lines, rather than in primary cells. Therefore, the need for a reliable and efficient protocol to transduce primary NK cells with genes of interest is crucial.
The overall goal of this study was to formulate a consistent and reliable method by which primary human or murine NK cells could be transduced with lenti- or retroviruses.
Earlier studies that attempted to address this problem have been performed, largely using the transient transformation of primary NK cells. This includes plasmid transfection9,10, Epstein-Barr Virus (EBV)/retroviral hybrid vector11, vaccinia vectors12,13, and Ad5/F35 chimeric adenoviral vectors14. Despite the modest efficiency of these techniques, the transient nature of transduction makes them unsuitable for the long-term utilization of the genetically modified NK cells. A few recent studies have used retroviral vectors to transduce NK cells, requiring multiple cycles of infection to achieve an acceptable level of gene expression11,15. In contrast to retroviral vectors, lentiviral vectors can use host-cell nuclear import machinery to translocate the viral pre-integration complex into the nucleus. This is a major limiting factor in the replication of the virus in non-dividing cells, which include primary NK cells.
Interactions between different cell-surface receptors and viral particles permit viral uptake into the cell. The initial engagements between the viral envelope proteins and their cognate host receptors could be limited because of the potential negative charges existing between these two. The rationale behind many transduction techniques is that the addition of cationic polymers, such as polybrene (Pb), protamine sulfate (PS), or dextran, could give a positive charge to the cell-surface receptors and thereby augment the binding of viral envelope proteins. This will increase the fusion efficiency and the uptake of the viral particles by the cells16. Although it has been reported that Pb or PS can improve gene transfer in T cells17, their application did not have any effect in the transduction efficiency of primary NK cells. Moreover, a comparative analysis between these reagents using primary NK cells has not been performed. In this study, the transduction efficiencies of the three cationic polymers were compared. The results show that, among these three cationic polymers, only dextran significantly enhances efficient viral transduction into both mouse and human primary NK cells.
Protocol
All animal protocols followed the humane and ethical treatment of animals and were approved by the Institutional Animal Care and Use Committee (IACUC) within the Biomedical Research Center (BRC) of the Medical College of Wisconsin (MCW), Milwaukee, WI. The use of human peripheral blood mononuclear cells (PBMCs) was approved by the Institutional Review Board (IRB) of the Blood Research Institute of the Blood Center of Wisconsin, Milwaukee, WI.
1. Mice, cell lines, and vectors
Obtain C57BL/6 mice from commercial vendors. Maintain the mouse colonies in pathogen-free conditions and use female and male mice between the ages of 6 and 12 weeks. Obtain de-identified human PBMCs from IRB-approved sources.
Anesthetize the animals witha mixture of 20-30% v/v isoflurane in propylene glycol (1, 2-propanediol, USP grade) to anesthetize the mice. Apply vet ointment to the eyes to prevent dryness while the mice are under anesthesia.
- To euthanize the animals, perform cervical dislocation after the induction of anesthesia.
- Restrain the mice by firmly grasping the base of the tail with one hand. Place a sturdy stick-type pen or the thumb and first finger of the other hand against the back of the neck, at the base of the skull.
- To produce the dislocation, push the hand restraining the head of the animal forward and push down while pulling backward with the hand holding the tail base. Verify the effectiveness of dislocation by feeling for a separation of cervical tissue.
Keep the animals in the chamber/cage for at least 5 min and remove them only when respiratory activity is absent or when there is a lack of a detectable heartbeat.
Obtain K562 and YAC-1 cells from commercial vendors and maintain them in RPMI1640 medium containing 10% heat-inactivated FBS. Test these cell lines periodically to exclude the possibility of mycoplasma contamination.
2. Preparation and titration of lentiviral vectors
Culture 5 × 106 293T cells overnight in a T75 cm2 flask containing a solution of 20 mL of RPMI1640 medium with 10% FBS, 100 U/mL penicillin, 100 µg/mL streptomycin, 1 mM sodium pyruvate, 5% of 7.5% sodium bicarbonate solution, and 0.001% β-mercaptoethanol. Place the flask in a 37 °C incubator infused with 5.2% CO2.
- Harvest the 293T cells using trypsin (0.025%)/EDTA (1 mM) in phosphate-buffered saline (PBS). Add 5 mL of Trypsin/EDTA in PBS and incubate the flasks for 10 min in a 37 °C an incubator to allow the detachment of 293T cells from the T75 cm2 flasks.
- Collect the detached cells and wash them twice in PBS to remove any traces of trypsin and EDTA. Count the cells with a hemocytometer and adjust the cell number to one million cells per mL.
- Transfect the 293T cells18 with 3.95 µg of psPAX2, 1.32 µg of pMD2G, and 5.26 µg of pLEP-GFP-Puro (generated in-house at BRI by cloning EF1alpha promoter from pWPI in place of a CMV promoter in pLenti CMV-GFP-Puro)19.
- Prepare 1 mL of 150 mM NaCl plus 63 µL of 0.6 mg/mL polyethylenimine (PEI; 25,000 kD, linear). Mix well and then add the plasmids and mix. Incubate for 20 min at room temperature and then add it to cells.
- 16 h post-transfection, replace the transfection medium with fresh medium plus 4.5 mM sodium butyrate. Harvest the supernatant containing virus 48 h post-transfection and concentrate by overnight centrifugation at 5,000 × g. See the Materials Table.
Determine the viral titers using 293T cells by performing serial dilutions and assay by flow cytometry 72 h post-transduction20. Use the expression of green fluorescent protein (GFP) as a measure to quantify the viral titers.
3. Purification and expansion of murine primary NK cells
Purify murine primary NK cells21. Briefly, pass single-cell spleen suspensions through nylon wool columns to deplete the adherent populations consisting of B cells and macrophages.
Culture the non-adherent populations eluted from the nylon wool column that contains murine NK cells with 1,000 U/mL interleukin (IL)-2 in RPMI1640 medium with 10% FBS, 100 U/mL penicillin, 100 µg/mL streptomycin, 1 mM sodium pyruvate, 5% of 7.5% sodium bicarbonate solution, and 0.001% β-mercaptoethanol (RPMI1640 complete medium).
Change the medium from the flasks on day 4 of this culture to remove the non-adherent T and NKT cells; cells that remain adhered to the flasks are largely NK cells. Add 20 mL of fresh RPMI1640 complete medium and 1,000 U/mL IL-2 to replenish the flasks.
Check the purity of the murine NK cell cultures on day 7 by flow cytometry with NK cell-specific markers20 using preparations with more than 95% of CD3-NK1.1+ population.
4. Purification and expansion of human primary NK cells
Isolate PBMCs by density gradient from the buffy coats of healthy human volunteers. Briefly, carefully layer 35 mL of half-diluted (with HBSS) cells over 15 mL of density gradient in a 50-mL canonical tube. Centrifuge at 400 × g for 30 min at 20 °C in a swinging bucket rotor without brake.
Use the commercial antibody-based negative selection kits to purify human primary NK cells according to the manufacturer's protocol.
Following the isolation, determine the purity of the NK cell preparations by immunofluorescence analyses. Stain NK cell preparations with 2 µg/mL anti-CD3 and 2 µg/mL anti-CD56 antibodies at 4 °C for 20 min. Wash the cells twice with PBS NK cell populations determined by the absence of CD3 and the presence of CD56 on the cell surface.
Incubate the cell preparations with antibodies for 20 min at 4 °C, wash with PBS, and analyze in a flow cytometer. Use NK cell preparations with more than 85% purity for the gene transduction assays20.
5. Transduction of murine and human primary NK cells with lentivirus
Suspend mouse or human primary NK cells in a 24-well plate at 0.5 × 105/mL of medium in the presence of GFP lentivirus supernatant at 5, 10, and 20 multiplicity of infection (MOI) and in the presence of Pb (8 µg/mL), PS (8 µg/mL) or dextran (8 µg/mL).
Centrifuge the plates at 1,000 × g for 60 min.
Without decanting the supernatant, culture the cells overnight (16-18 h) in a 37 °C incubator infused with 5.2% CO2.
Wash with 10 mL of PBS and resuspend in 2 mL of complete RPMI1640 culture medium in the presence of IL-2 (300 U/mL).
- Test the human and mouse primary NK cell-mediated cytotoxicity against K562 and YAC-1, respectively by performing 51Chromium (Cr)-release assays at varied effector-to-target-cell ratios22.
- Briefly, give one million target cells 50 µCi of radiolabeled sodium chromate 51Cr. During the 4-h incubation time, they uptake the 51Cr into cellular proteins. At the end of the incubation, wash the cells to remove any unincorporated label.
- Calculate specific tumor cell lysis by the amount of absolute, spontaneous, and experimental release of 51Cr from the target cells. NOTE: The calculation of the percentage of specific lysis from pentaplicate experiments was done using the following equation: % specific lysis = ((51Cr mean experimental release - 51Cr mean spontaneous release) / (51Cr mean maximal release - 51Cr mean spontaneous release)) х 100, where "51Cr mean spontaneous release" is the 51Cr released from target cells in the absence of NK cells and "51Cr mean maximal release" is the 51Cr released from target cells upon lysis by 2 N hydrochloric acid (HCl).
- Harvest the transduced NK cells on day 7 by gently tapping the flasks.
- Activate the harvested cells with titrated concentrations of plate-bound anti-NKG2D (A10) mAb by coating 96-well, highly protein-absorbent polystyrene plates with 2.5 µg/mL concentrations of anti-NKG2D mAb overnight.
- Wash each well with 100 µL of PBS three times before the addition of primary NK cells.
Collect culture supernatants using a multi-channel pipette between 16 and 18 h post-activation to quantify cytokines such as IFN-γ. Generate standard curves using the recombinant cytokines provided with enzyme-linked immunosorbent assay (ELISA) kits.
- Test the NK cell viability using annexin-V/7-amino-actinomycin D (7-AAD) staining23 and determine the percent of necrotic cells among the transformed NK cells using a flow cytometer.
- To perform this assay, harvest the murine and human NK cells four days after transduction, wash two times with 10 mL of cold PBS at 500 × g for 5 min each, and incubate with an Annexin-V (PE)/7-AAD kit.
Use appropriate flow cytometry software to analyze the data. Select the live-cell population and analyze the expression of GFP (FITC channel) as a measure of viral transduction24.
Representative Results
Dextran induces the efficient gene transfer of lentiviral vector in primary human and murine NK cells
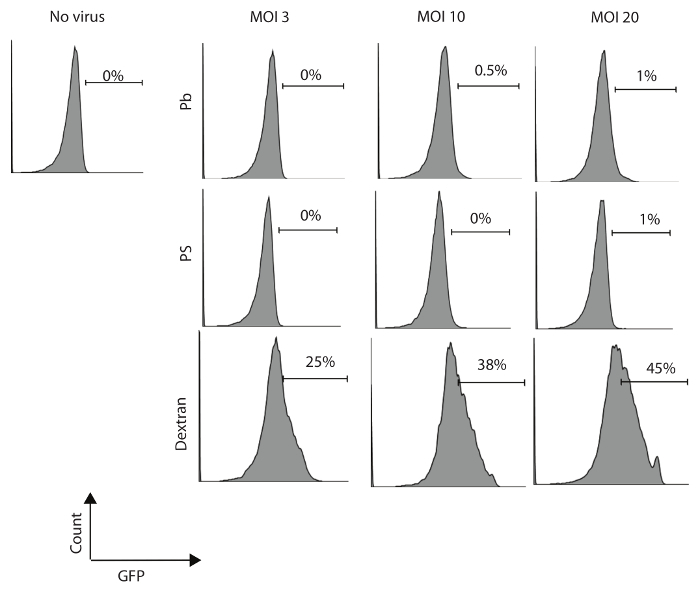
Human NK cells were isolated and purified from PBMC (with a purity of more than 85%) and incubated overnight with rIL-2 300 U/mL. These primary NK cells were then transduced with GFP lentivirus at varied multiplicities of infection (MOI; 3, 10, and 20 IU per cell) in 24-well plates in the presence of 8 µg/mL Pb, PS, or dextran. Cells were centrifuged at 1,000 × g for 60 min and cultured (in the presence of virus) overnight at 37 °C in a CO2 incubator. Cells were washed and resuspended in fresh RPMI1640 complete medium with rIL-2 300 U/mL for seven days. The transduction efficiency was evaluated by flow cytometry for GFP expression seven days after transduction. Results show that dextran enhances the efficiency of lentiviral transduction in human NK cells and increases the viral titer, which can also improve the percentage of transduced NK cells (Figure 1).
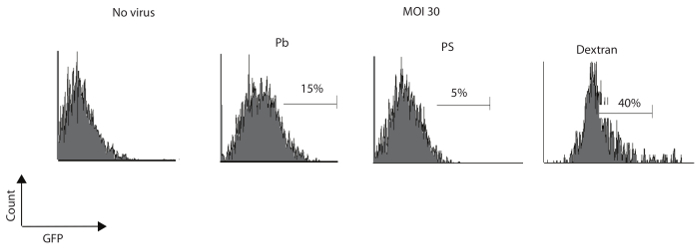
Murine NK cells were isolated and cultured as described in the earlier section. The above protocol was used to analyze and compare the transduction efficiency of Pb, Ps, and dextran. Results in Figure 2 show that dextran can augment the efficiency of lentiviral vectors compared to Pb or PS.
Transduction of NK cells with dextran does not affect their ability to mediate effector functions
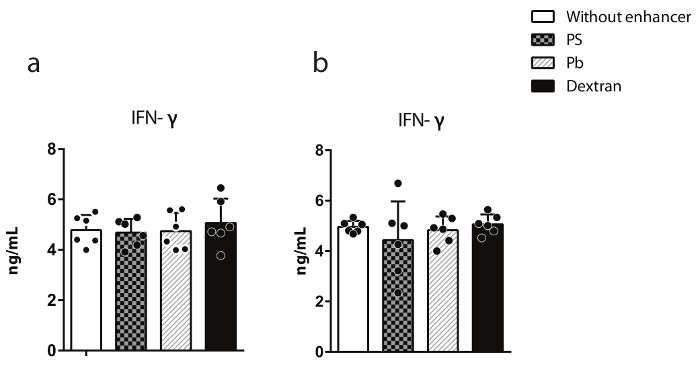
The cytotoxic capacity of transduced human and murine NK cells was examined by 51Cr-release assays against K562 and YAC-1 as the target cells. Results presented in Figure 3a and b reveal that the transduction of NK cells by dextran does not negatively alter the killing potential of transformed NK cells compared to non-transduced NK cells. Additionally, the cytokine production from transduced primary NK cells was analyzed. IFN-γ generation was measured using an ELISA. Transduced human primary NK cells were co-cultured with K562 for 24 h, and the supernatants were collected to measure IFN-γ. Results presented in Figure 4a reveal that dextran has no impact on the ability of transduced NK cells to produce cytokines. As an independent validation, transduced NK cells were activated with titrated concentrations of plate-bound anti-NKG2D (A10) mAb for 18 h. Culture supernatants were collected, and IFN-γ generation was measured by ELISA. Results shown in Figure 4b reveal that the transduction of murine NK cells with dextran has no effect on their ability to produce cytokines.

Transduction of NK cells with dextran does not affect their cell viability
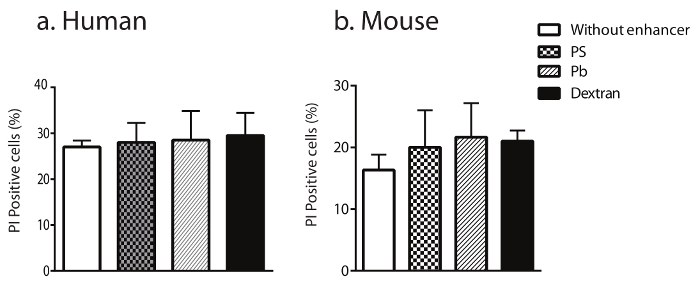
The influence of Pb, PS, or dextran on the viability of transduced NK cells was evaluated using a propidium iodide (PI) assay and subsequent flow analyses. Results in Figure 5a and b demonstrate that, although transducing primary human and mouse NK cells with viruses can induce necrosis in about 20% of NK cells, the addition of dextran did not augment this necrosis compared to Pb or PS.
Statistical analyses were performed with a two-tailed unpaired Student's t-test. P-values ≤ 0.05 were considered significant.
Figure 1: Dextran has a higher efficiency of transduction of human primary NK cells. Primary human NK cells were transformed with MOI of 3, 10, or 20 IU/cell and were cultured for seven days in the presence of Pb (8 µg/mL), PS (8 µg/mL), or dextran (8 µg/mL). Percentages of GFP-positive NK cells were determined by flow cytometry on day seven following the transduction. One of three independent experiments is shown. Please click here to view a larger version of this figure.
Figure 2: Dextran has a higher efficiency of transduction in murine NK cells. Primary murine NK cells were transformed with an MOI of 30 IU/cell and were cultured for seven days in the presence of Pb (8 µg/mL), PS (8 µg/mL), or dextran (8 µg/mL). Percentages of GFP-expressing cells were determined by flow cytometry at seven days following the transduction. One of three independent experiments is shown. Please click here to view a larger version of this figure.
Figure 3: Dextran does not modify the NK cell-mediated cytotoxic activity of transformed cells. Primary NK cells were transformed with an MOI of 10 IU/cell and were cultured for seven days. YAC-1 and K562 were used as target cells to determine the cytotoxic potentials of murine (a) and human (b) NK cells, respectively. The data shown are representatives of two independent experiments. The data shown are averages with SEM. Please click here to view a larger version of this figure.
Figure 4: Dextran does not alter the IFN-γ production of primary NK cells. a) Murine NK cells were transduced with an MOI of 30 IU/cell and cultured for seven days. NK cells were stimulated with 2.5 µg/mL of plate-bound anti-NKG2D antibody for 18 h, and IFN-γ was quantified in culture supernatants using an ELISA. b) Transduced human NK cells were cultured with K562 cells for 24 h, and the IFN-γ production was measured in culture supernatants. The data presented are representative of three independent experiments. The data shown are averages with SEM. Please click here to view a larger version of this figure.
Figure 5: Transduction of NK cells with dextran does not alter their cell viability. The viability of transformed human (a) and mouse (b) NK cells was quantified after staining for Annexin-V (PE)/7-AAD positive cells. The data shown are averages with SEM. Please click here to view a larger version of this figure.
Discussion
This study demonstrates that use of dextran as a cationic polymer agent enhances the lentiviral transduction efficiency of both murine and human primary NK cells. Additionally, other cationic agents, such as Pb or PS, have no discernible effect on the delivery of viral vectors into human primary NK cells. Previously, it has been demonstrated that Pb can augment gene transduction in human T cells17. These results, however, suggest that neither Pb nor PS have a similar efficiency on human primary NK cells. In this study, Pb improved the transduction efficacy only modestly in murine NK cells. It has been shown that the inhibition of intracellular antiviral defense mechanisms using BX795, an inhibitor of TBK1/IKKɛ, and PS as an enhancer can increase the lentiviral transduction efficiency25. Nevertheless, the results showed that this inhibitor has no effect on the transduction efficacy in murine or human primary NK cells (data not shown).
The results demonstrate that dextran can induce the efficient transduction of primary human and mouse NK cells, while PS and Pb have no effect. The results also show that dextran does not alter the effector functions of NK cells, such as anti-tumor cytotoxicity and the production of pro-inflammatory cytokines. Thus, these results prove that the viability of these primary NK cells was not altered by the use of dextran.
Multiple studies analyzed and compared the transduction efficiency of retroviral vectors using different cationic polymers, with varied outcomes26,27,28. One earlier study compared the transduction efficacy of these polymers using lentiviral vectors; however, this was tested in CD4+ T cells16. In one of these studies, it has been shown that Pb has a better capacity of transduction on transformed B cells and dendritic cells compared to dextran26. In another study, it has been shown that dextran facilitates a higher transduction efficiency than other enhancers in human B cells and T cells16. The current study is the first of its kind, to our knowledge, that analyzed and compared the transduction capacity of different polymers in both human and murine primary NK cells.
Dextran-based gene transductions may require a minimum of two rounds of transductions. These results show that one round of transduction is enough to reach up to 40% of positively transduced cells, which is increased up to 100% following two rounds of transduction (data not shown). One of the major challenges in transducing NK cells is the ability of these cells to preserve the expression of the transduced gene. The current results demonstrate that the transduction of primary NK cells with dextran is stable and that the NK cells that are cultured in the presence of IL-2 can retain the vector and maintain transgene expression for four weeks (data not shown). Additional experiments are required to analyze the transduction efficiency and to examine the concurrent expression of multiple transgenes.
The clinical efficacy of immune cell-based cancer therapies is being validated by multiple institutions. CAR-transduced T cells provide renewed promise for the disease- and relapse-free recovery of cancer patients compared to conventional therapies. As part of innate immune responses, NK cells do not require prior sensitization to mediate their effector functions, including anti-tumor cytotoxicity. Due to their rapid response, NK cells are an ideal effector lymphocyte subset to mediate an efficient cell-based cancer immunotherapy. Despite their positive attributes in tumor recognition and elimination, the technical hurdles regarding the genetic manipulation to deliver transgenes limit the fullest clinical utilization of NK cells.
Electroporation of mRNA for exogenous gene expression is highly efficient and has better outcomes. However, mRNA utility is limited, as they allow only the transient expression of genes of interest and thereby are not suitable for clinical applications. The retroviral transduction of NK cells is less efficient as it requires multiple rounds of transduction; moreover, retroviral vectors cannot transduce into non-dividing cells. The only promising approach for stable transgene delivery is the use of lentiviral vectors.
Altogether, this study provides an efficient method to deliver transgenes into primary NK cells, without impairing their effector functions. This protocol makes NK cell-based cancer immunotherapy highly efficient and applicable to emerging clinical trials. Future experiments are needed to validate the efficacy of this method in NK cell-based cancer immunotherapies.
Disclosures
The authors claim no financial conflict of interest.
Acknowledgments
We thank Lucia Sammarco and her Lulu's Lemonade Stand for inspiration, motivation, and support. This work was supported in part by NIH R01 AI102893 and NCI R01 CA179363 (S.M.); NHLBI-HL087951 (S.R.); NIH-CA151893-K08 (M.J.R.); NCI 1R01CA164225 (L.W); the Alex Lemonade Stand Foundation (S.M.); the HRHM Program of the MACC Fund (S.M.; S.R.; M.S.T); the Nicholas Family Foundation (S.M.); the Gardetto Family (S.M.); the Hyundai Scholars Program (M.S.T.); Hyundai Hope on Wheels (S.R.); the MACC Fund (M.S.T. and S.M.); the Children's Research Institute, MCW (S.R.); and the Kathy Duffey Fogerty Award (M.J.R.).
References
- Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat. Immunol. 2008;9:503–510. doi: 10.1038/ni1582. [DOI] [PubMed] [Google Scholar]
- Zitvogel L, Tesniere A, Kroemer G. Cancer despite immunosurveillance: immunoselection and immunosubversion. Nat. Rev. Immunol. 2006;6:715–727. doi: 10.1038/nri1936. [DOI] [PubMed] [Google Scholar]
- Arina A, et al. Cellular liaisons of natural killer lymphocytes in immunology and immunotherapy of cancer. Expert. Opin. Biol. Ther. 2007;7:599–615. doi: 10.1517/14712598.7.5.599. [DOI] [PubMed] [Google Scholar]
- Manilay JO, Sykes M. Natural killer cells and their role in graft rejection. Curr. Opin. Immunol. 1998;10:532–538. doi: 10.1016/s0952-7915(98)80219-7. [DOI] [PubMed] [Google Scholar]
- Raulet DH, Vance RE. Self-tolerance of natural killer cells. Nat. Rev. Immunol. 2006;6:520–531. doi: 10.1038/nri1863. [DOI] [PubMed] [Google Scholar]
- Chouaib S, et al. Improving the outcome of leukemia by natural killer cell-based immunotherapeutic strategies. Front Immunol. 2014;5:95. doi: 10.3389/fimmu.2014.00095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dulphy N, et al. Underground Adaptation to a Hostile Environment: Acute Myeloid Leukemia vs. Natural Killer Cells. Front Immunol. 2016;7:94. doi: 10.3389/fimmu.2016.00094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran J, Kung SK. Lentiviral vectors mediate stable and efficient gene delivery into primary murine natural killer cells. Mol. Ther. 2007;15:1331–1339. doi: 10.1038/sj.mt.6300184. [DOI] [PubMed] [Google Scholar]
- Maasho K, Marusina A, Reynolds NM, Coligan JE, Borrego F. Efficient gene transfer into the human natural killer cell line, NKL, using the Amaxa nucleofection system. J. Immunol. Methods. 2004;284:133–140. doi: 10.1016/j.jim.2003.10.010. [DOI] [PubMed] [Google Scholar]
- Trompeter HI, Weinhold S, Thiel C, Wernet P, Uhrberg M. Rapid and highly efficient gene transfer into natural killer cells by nucleofection. J. Immunol. Methods. 2003;274:245–256. doi: 10.1016/s0022-1759(02)00431-3. [DOI] [PubMed] [Google Scholar]
- Becknell B, et al. Efficient infection of human natural killer cells with an EBV/retroviral hybrid vector. J. Immunol. Methods. 2005;296:115–123. doi: 10.1016/j.jim.2004.11.012. [DOI] [PubMed] [Google Scholar]
- Jiang K, et al. Syk regulation of phosphoinositide 3-kinase-dependent NK cell function. J. Immunol. 2002;168:3155–3164. doi: 10.4049/jimmunol.168.7.3155. [DOI] [PubMed] [Google Scholar]
- Burshtyn DN, et al. Conserved residues amino-terminal of cytoplasmic tyrosines contribute to the SHP-1-mediated inhibitory function of killer cell Ig-like receptors. J. Immunol. 1999;162:897–902. [PubMed] [Google Scholar]
- Schroers R, et al. Gene transfer into human T lymphocytes and natural killer cells by Ad5/F35 chimeric adenoviral vectors. Exp. Hematol. 2004;32:536–546. doi: 10.1016/j.exphem.2004.03.010. [DOI] [PubMed] [Google Scholar]
- Imai C, Iwamoto S, Campana D. Genetic modification of primary natural killer cells overcomes inhibitory signals and induces specific killing of leukemic cells. Blood. 2005;106:376–383. doi: 10.1182/blood-2004-12-4797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denning W, et al. Optimization of the transductional efficiency of lentiviral vectors: effect of sera and polycations. Mol. Biotechnol. 2013;53:308–314. doi: 10.1007/s12033-012-9528-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamers CH, Willemsen RA, Luider BA, Debets R, Bolhuis RL. Protocol for gene transduction and expansion of human T lymphocytes for clinical immunogene therapy of cancer. Cancer Gene Ther. 2002;9:613–623. doi: 10.1038/sj.cgt.7700477. [DOI] [PubMed] [Google Scholar]
- Campeau E, et al. A versatile viral system for expression and depletion of proteins in mammalian cells. PLoS. ONE. 2009;4:e6529. doi: 10.1371/journal.pone.0006529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segura MM, Garnier A, Durocher Y, Ansorge S, Kamen A. New protocol for lentiviral vector mass production. Methods Mol. Biol. 2010;614:39–52. doi: 10.1007/978-1-60761-533-0_2. [DOI] [PubMed] [Google Scholar]
- Rajasekaran K, et al. Signaling by Fyn-ADAP via the Carma1-Bcl-10-MAP3K7 signalosome exclusively regulates inflammatory cytokine production in NK cells. Nat. Immunol. 2013;14:1127–1136. doi: 10.1038/ni.2708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Regunathan J, Chen Y, Wang D, Malarkannan S. NKG2D receptor-mediated NK cell function is regulated by inhibitory Ly49 receptors. Blood. 2005;105:233–240. doi: 10.1182/blood-2004-03-1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Awasthi A, et al. Rap1b facilitates NK cell functions via IQGAP1-mediated signalosomes. J. Exp. Med. 2010;207:1923–1938. doi: 10.1084/jem.20100040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajasekaran K, et al. Transforming Growth Factor-{beta}-activated Kinase 1 Regulates Natural Killer Cell-mediated Cytotoxicity and Cytokine Production. J Biol. Chem. 2011;286:31213–31224. doi: 10.1074/jbc.M111.261917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolkowicz R, Nolan GP, Curran MA. Lentiviral vectors for the delivery of DNA into mammalian cells. Methods Mol. Biol. 2004;246:391–411. doi: 10.1385/1-59259-650-9:391. [DOI] [PubMed] [Google Scholar]
- Sutlu T, et al. Inhibition of intracellular antiviral defense mechanisms augments lentiviral transduction of human natural killer cells: implications for gene therapy. Hum. Gene Ther. 2012;23:1090–1100. doi: 10.1089/hum.2012.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornetta K, Anderson WF. Protamine sulfate as an effective alternative to polybrene in retroviral-mediated gene-transfer: implications for human gene therapy. J. Virol. Methods. 1989;23:187–194. doi: 10.1016/0166-0934(89)90132-8. [DOI] [PubMed] [Google Scholar]
- Toyoshima K, Vogt PK. Enhancement and inhibition of avian sarcoma viruses by polycations and polyanions. Virology. 1969;38:414–426. doi: 10.1016/0042-6822(69)90154-8. [DOI] [PubMed] [Google Scholar]
- Jensen M, et al. The bi-specific CD3 x NCAM antibody: a model to preactivate T cells prior to tumour cell lysis. Clin. Exp. Immunol. 2003;134:253–263. doi: 10.1046/j.1365-2249.2003.02300.x. [DOI] [PMC free article] [PubMed] [Google Scholar]


