Abstract
Background:
Academic reports have confirmed Moringa oleifera leaves to possess significant antioxidant capacities; however, such studies are unavailable for its ripe seeds even though they are more desirous for consumption due to their sweet taste.
Objective:
In this study, we investigated antioxidant capacities of four polar extracts (crude water, ethanol, butanol, and aqueous residue) from the plant's ripe seeds.
Materials and Methods:
Phytochemicals were extracted from the ripe seeds of M. oleifera using ethanol and water solvents at initial stage. Butanol and aqueous residue were then subsequently fractioned out from the ethanol extract. Phenolic and flavonoid contents of the polar extracts were determined. Then, their antioxidant capacities were quantified by 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assays. Finally, gas chromatography-mass spectrometry (GC-MS) analyses of the extracts were performed.
Results:
DPPH and ABTS tests showed that the polar extracts possess significant antioxidant capacities that ranged from 29 to 35.408 μM Trolox equivalence antioxidant capacity (TEAC)/mg sample and 7 to 29 μM TEAC/mg sample, respectively. The antioxidant capacities of the extracts corresponded to their phenolic and flavonoid contents that varied from 13.61 to 20.42 mg gallic acid equivalence/g sample and 0.58 to 9.81 mg quercetin equivalence/g sample, respectively. Finally, GC-MS analyses revealed antimicrobial phenolic compounds, 4-hydroxybenzaldehyde in crude water extract and 4-hydroxybenzene acetonitrile in the ethanol and butanol extracts, and aqueous residue.
Conclusion:
Our results established that M. oleifera ripe seeds have significant antioxidant activity which may be due to its phenolic and nonphenolic compounds content.
SUMMARY
In this study, polar phytochemicals from ripe seeds of Moringa oleifera were extracted by water and ethanol solvents, and butanol extract and aqueous residue were subsequently fractioned out of the ethanol extract. The four polar extracts were shown to have significant antioxidant capacities which correspond to their phenolic contents. Further, antimicrobial compounds 4-hydroxybenzaldehyde and 4-hydroxybenzene acetonitrile were identified in the extracts by gas chromatography-mass spectrometry analyses.
Abbreviations used: ABTS: 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); DPPH: 2,2-diphenyl-1-picrylhydrazyl; TEAC: Trolox equivalence antioxidant capacity; QE: Quercetin equivalence; GAE: Gallic acid equivalence; GC-MS: Gas chromatography-mass spectrometry.
Keywords: 4-hydroxybenzaldehyde, 4-hydroxybenzene acetonitrile, antioxidant, gas chromatography-mass spectrometry, Moringa oleifera ripe seeds, phenolics
INTRODUCTION
Plants are natural reservoirs of numerous phytochemicals that are sources of nutrition and medicine for humans. An important group of phytochemicals of plants is antioxidants. Plant antioxidants are chemical compounds such as polyphenols, vitamins, and fatty acids that can react with unstable oxidants such as free radical species (reactive oxygen and nitrogen species) and stabilize them. High level of oxidants in human body system results to oxidative stress which is the cause of many degenerative diseases such as cancer, neurological disorder, and Alzheimer, cardiovascular, and pulmonary diseases among others.[1] Hence, there is need for internal and external sources of antioxidants to combat oxidative stress in the body system to prevent the development of the diseases and keep the body healthy.
Moringa oleifera is one of the plants that are commonly consumed by many Africans and Asians which have high content of antioxidants. Consumers of this plant usually refer to it as a “wonderful tree or miracle tree” because they believe it is highly nutritious and serves as cure to many diseases such as diabetes, cancer, insomnia to mention few. All of the plant parts (leaves, root, bark, and seeds) can be consumed by eaten raw, in curry, or decoctions.[2,3] There are many extensive studies that have been reported in various academic publications on the antioxidant capacities and phenolic contents of the leaves of the plant; however, such information and properties of the plant whitish ripe seed are unavailable even though it is widely consumed as well.[4,5,6]
Hence, phenolic contents and antioxidant capacities of different polar fractions of M. oleifera ripe seeds were determined in this study. In addition, gas chromatography-mass spectrometry (GC-MS) profiling of volatile compounds in all the fractions of the plant seeds was performed to identify phenolic compounds and other antioxidants in the samples.
MATERIALS AND METHODS
Extraction and fractionation of crude phenolic compounds from Moringa oleifera seeds
The extraction method employed has been previously described by Adebayo et al.[7] The whitish ripe seeds were purchased from HERBAGUS and de-coated. The naked seeds were mechanically macerated to powder. Ten grams of the powder was mixed with water, and the mixture was gently agitated at room temperature for 24 h. Another 100 g of the powder was mixed with 80% ethanol. The mixture was left at room temperature for 24 h with continuous mild agitation. Then, the crude water and ethanol extracts were filtered out from their mixtures using Whatman #1 filter paper. Ethanol was evaporated off the filtrate (extract) by a rotary evaporator. The extract was then sequentially fractionated using four solvents (hexane, dichloromethane, chloroform, and butanol) based on their degrees of polarity. The water extract, crude ethanolic extract, butanol fraction, and purified aqueous residue were then lyophilized and stored in 4°C until further use.
Total phenolic content
Total phenolic contents of the samples were determined by Folin–Ciocalteu colorimetric method. Stock solutions of 1 mg/ml concentration of samples and standard (gallic acid) dissolved in 50% (v/v) methanol were prepared. Twenty microliters of sample and standard (of eight different serial dilutions of stock solution) were separately mixed with 100 μl of 25% (v/v) Folin–Ciocalteu reagent and allowed to stand for 3–5 min. Eighty microliters of Na2 CO3 was added making a total of 200 μl solution. The solutions were incubated for 1 h in the dark and absorbance values were read at 765 nm. A calibration curve of absorbance values against varying concentrations of gallic acid standard was plotted. A regression equation of the curve, absorbance value = 0.0248 (gallic acid concentration) + 0.0003 with R2 value, 0.9865 was obtained. Total phenolic contents of the samples in mg gallic acid equivalence (GAE)/g of sample were calculated using the equation.
Total flavonoid content
A stock solution of 1 mg/ml of quercetin (standard) was prepared using 50% methanol solvent, and the previous samples' stock solutions were used. Procedure employed for the assay was as described by Lamaison and Carnat,[8] using AlCl3.6H2O with slight changes. Forty microliters of samples and quercetin (of six different dilutions) were separately mixed with 200 μl 2% (w/v) AlCl3.6H2O. The mixture was incubated for 10 min at ambient temperature, and absorbance values at 440 nm were obtained. Calibration curve for quercetin standard was plotted. Regression equation of the curve, absorbance value = 0.0401 (quercetin concentration) − 0.0017 with R2 value, 0.9992 was obtained. The equation was used to calculate the quercetin content in 1 g of sample (mg quercetin equivalence [QE]/g of sample).
2,2-diphenyl-1-picrylhydrazil free radical scavenging assay
Antioxidant activities of all the polar fractions of seeds of M. oleifera were determined by 2,2-diphenyl-1-picrylhydrazil (DPPH) free radical scavenging assay using the protocol of Brand-Williams et al.,[9] with slight changes. Briefly, 180 μl of 0.2 mM DPPH methanolic solution was mixed and reacted with 12 μl of the samples (4 mg/ml), Trolox (of six different serial dilutions, 0–125 μM), and 50% methanol (blank) separately in 96-microwell plate for 30 min at 37°C in the dark. Then, absorbance value for each mixture was read at 517 nm after incubation. Percentage of radical scavenging activity was calculated from absorbance values using the following equation.
Percentage of radical scavenging activity (%) = ([AB − AS/C] ÷ AB) ×100
Where AB = Absorbance of blank, AS = Absorbance of sample, and AC = Absorbance of control (Trolox).
Calibration curve of radical scavenging activity (%) against concentration of Trolox standard was plotted. Regression equation of the curve, radical scavenging activity = 0.7488 (Trolox concentration) + 0.076 with R2 value, 0.998 was obtained. Trolox equivalence antioxidant capacity (TEAC) of each sample was calculated using the equation and expressed as μM Trolox equivalent/mg of sample (μM TE/mg sample).
2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) antioxidant capacity
Equal volumes of 7 mM 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) solution and 0.2 mM potassium persulfate were reacted together for 16–18 h in the dark to form ABTS+ solution. Then, the working solution of ABTS+ was diluted with 50% methanol. A volume of 180 μl ABTS+ working solution was mixed with 20 μl of different concentrations Trolox standard and the polar samples separately in 96-well plate. The mixtures were incubated for 6 min in the dark after which their absorbance values were read at 754 nm. The TEAC of each sample was determined as previously described above in DPPH method.
Gas chromatography-mass spectrometry analysis
GC-MS analyses of the samples were performed using an Agilent GC system (7890A series) equipped with split less/split injector coupled with 5% phenyl methyl silox capillary column that has 30 m × 250 μm film dimension and 0.25 μm thickness. The column is attached to a mass detector. Helium (He) was the carrier gas with 10 ml/min flow rate. The injector and detector temperatures were fixed at 280°C. The column temperature was 70°C and held for 2 min followed by an increase to 200°C at a rate of 8°C/min for 2 min. Then, the temperature was further increased to 250°C at 10°C/min and held for 2 min. Total run time was approximately 27.2 min. Mass spectra were acquired by MS in scan mode at 70 eV, in a range of 30–800 m/z. The injection volume was 1 μl of 2.5 mg/ml of each of the samples with exception of aqueous residue (5 mg/ml). Each sample was run thrice and intermittently with methanol as blank.
Identification of phytochemical compounds
Volatile compounds of the polar samples were identified by matching their obtained mass spectra with mass spectra of compounds in the National Institute of Standards and Technology library. Matchings with >80% quality were taken as true identification.
RESULTS AND DISCUSSION
M. oleifera is among the most common plants usually consumed by Africans and Asians as herbal medicine for curing many diseases associated with oxidative stress such as cancer and neurodegenerative disorder among others.[2] In this study, antioxidant capacities of the plant ripe seed extracts by four polar solvents were quantified by ABTS and DPPH tests. The phenolic and flavonoid contents of the seed samples were also determined because they are major contributors to the overall antioxidant activity of plants.[10] Total phenolic and flavonoid contents of M. oleifera seeds of different polar fractions yields are displayed in Figure 1. Crude water extract (20.42 mg GAE/g sample) has the highest amount of phenolics, and aqueous residue (13.61 mg GAE/g sample) has the least even though all fractions have significant amount just like other parts of the plant.[6] The flavonoid contents of the fractions are in the following order: crude ethanolic extract 9.81 mg QE/g sample > crude water extract 1.28 mg QE/g sample > butanol fraction 0.83 mg QE/g sample > aqueous residue 0.58 mg QE/g sample. Flavonoid content amounts were lower than phenolic amounts for all fractions, which is expected and showed that the results are theoretically correct as flavonoids are subgroups of phenolics which contribute to the overall amount of the phenolics.[10]
Figure 1.
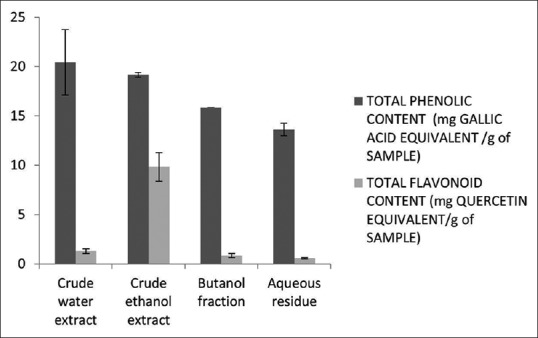
Total phenolic and flavonoid contents of Moringa oleifera ripe seeds. Total phenolic and flavonoid contents are expressed in mg gallic acid equivalence/g of sample (mg gallic acid equivalence/g sample) and mg quercetin equivalence/g of sample (quercetin equivalence/g of sample). Values were expressed as mean ± standard deviation
The antioxidant capacities of the polar fractions of M. oleifera seeds are shown in Figure 2. Based on DPPH tests, butanol fraction (35.408 μM TEAC/mg sample) has the highest scavenging effects while crude water extract and aqueous residue (29 μM TEAC/mg sample) have the lowest, while according to the ABTS antioxidant activity tests, crude water extract (29 μM TEAC/mg sample) has the highest antioxidant capacity while aqueous residue (7 μM TEAC/mg sample) has the least. ABTS and DPPH results of the polar fractions of M. oleifera seeds did not correlate and this may be due to different action mechanisms of the tests for determining antioxidant effects;[11] however, the two tests revealed that all the samples from M. oleifera seeds have high and significant antioxidant capacities. Thus, the plant seed is a good source of antioxidant.
Figure 2.
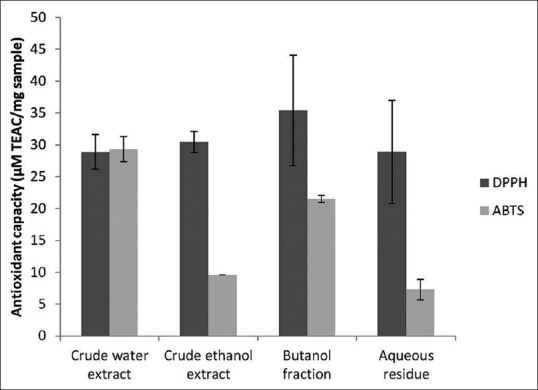
Antioxidant capacities of the polar fractions of Moringa oleifera ripe seeds. 2,2-diphenyl-1-picrylhydrazyl and 2,2'-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) radical scavenging activities of the polar extracts are expressed in Trolox equivalence antioxidant capacity. Values were expressed as mean ± standard deviation
GC-MS analyses were performed to profile volatile compounds present in all the polar fractions of M. oleifera ripe seeds. The chromatograms of the compounds identified in the samples are displayed in Figures 3–6. This helps to identify volatile phenolic compounds in the fractions and also determine the efficiency of purification of M. oleifera seed phenolic compounds in the crude ethanolic extract by the sequential fractionation employed from volatile nonphenolic compounds such as lipids and sugars. A total of 25 compounds were identified in all the fractions which include five phenols, ten esters, four alkenes, one alkane, three carbohydrates, and one pyran [Figures 3–6]. Two phenolic compounds are the most abundant among the identified volatile compounds in all the polar samples and these are 4-hydroxybenzaldehyde in crude water extract (42.10%) and 4-hydroxybenzeneacetonitrile in crude ethanolic extract (29.76%), butanol extract (16.29%), and aqueous residue (38.52%). The GC-MS results also revealed that the aqueous residue of M. oleifera seeds has the highest abundance of identified phenolic compound and the least abundance of nonphenolic interferences among the crude ethanolic extract and its fractions which suggest that the sequential fractionation employed in the experiment was effective in purifying volatile phenols of the seeds from other nonvolatile compounds. The esters such as hexadecanoic acid, methyl ester, and 9-octadecenoic acid (Z)-, methyl ester identified in the crude water and ethanolic extracts might have also contributed to their observed high antioxidant activities.[12]
Figure 3.
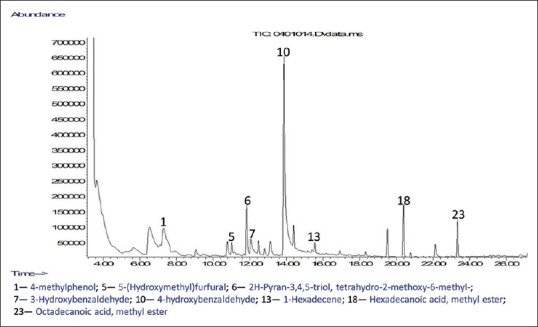
Chromatogram of compounds identified in crude water extract of Moringa oleifera ripe seeds
Figure 6.
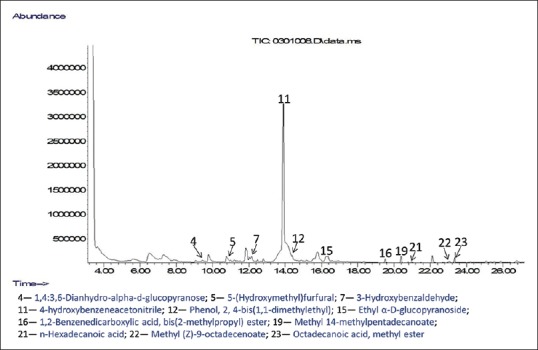
Chromatogram of compounds identified in aqueous residue of Moringa oleifera ripe seeds
Figure 4.
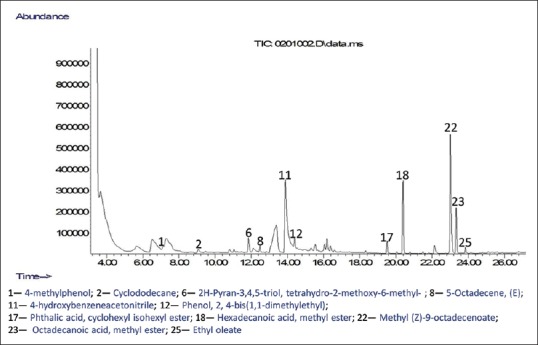
Chromatogram of compounds identified in crude ethanolic extract of Moringa oleifera ripe seeds
Figure 5.
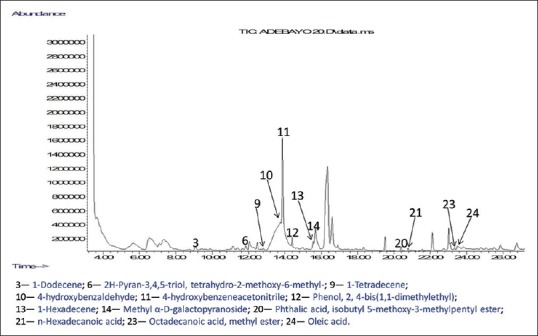
Chromatogram of compounds identified in butanol extract of Moringa oleifera ripe seeds
Moreover, phenolic compounds identified in the samples have certain biological functions and activities that are worth mentioning. For instance, 4-hydroxybenzaldehyde identified in the crude water extract has been certified to be safe for consumption as a flavoring agent by expert panel of the Flavor and Extract Manufacturers Association.[13] Isolated 4-hydroxybenzaldehyde from rhizome and pseudostem of Alpinia conchigera has a significant antibacterial effect on several strains of Staphylococcus aureus and antifungal activity on Microsporum canis, Candida albicans and Trichophyton rubrum.[14] Furthermore, 4-hydroxybenzeneacetonitrile identified in the crude ethanol extract and its fractions was found to be toxic to pathogenic ciliated protozoan, Tetrahymena pyriformis.[15]
CONCLUSION
Polar fractions of M. oleifera ripe seeds have significant antioxidant activities due to their phenolic and nonphenolic compounds content.
Financial support and sponsorship
The research was supported by Fundamental Research Grant Scheme (FRGS) (grant number: 203/CIPPT/6711340) and I.A.A. is grateful to Universiti Sains Malaysia for awarding him USM fellowship.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Pham-Huy LA, He H, Pham-Huy C. Free radicals, antioxidants in disease and health. Int J Biomed Sci. 2008;4:89–96. [PMC free article] [PubMed] [Google Scholar]
- 2.Fahey JW. Moringa oleifera: A review of the medical evidence for its nutritional, therapeutic, and prophylactic properties. Part 1. Trees Life J. 2005;1:1–15. [Google Scholar]
- 3.Fuglie LJ, Gomez Zambrano J, Aleman RJ, Hunter IS, Furst M, Garcia SA, et al. The Miracle Tree, Moringa oleifera: Republica Dominicana. Centro Para el Desarrollo Agropecuario y Forestal. 1999 [Google Scholar]
- 4.Vongsak B, Sithisarn P, Gritsanapan W. Bioactive contents and free radical scavenging activity of Moringa oleifera leaf extract under different storage conditions. Ind Crops Prod. 2013;49:419–21. [Google Scholar]
- 5.Verma AR, Vijayakumar M, Mathela CS, Rao CV. In vitro and In vitro antioxidant properties of different fractions of Moringa oleifera leaves. Food Chem Toxicol. 2009;47:2196–201. doi: 10.1016/j.fct.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 6.Sreelatha S, Padma PR. Antioxidant activity and total phenolic content of Moringa oleifera leaves in two stages of maturity. Plant Foods Hum Nutr. 2009;64:303–11. doi: 10.1007/s11130-009-0141-0. [DOI] [PubMed] [Google Scholar]
- 7.Adebayo IA, Arsad H, Samian MR. Antiproliferative effect on breast cancer (MCF7) of Moringa oleifera seed extracts. Afr J Tradit Complement Altern Med. 2017;14:282–7. doi: 10.21010/ajtcam.v14i2.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lamaison J, Carnat A. Levels of principal flavonoids in flowers and leaves of Crataegus-Monogyna Jacq and Crataegus-Laevigata (Poiret) Dc (Rosaceae) Pharm Acta Helv. 1990;65:315–20. [Google Scholar]
- 9.Brand-Williams W, Cuvelier ME, Berset C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol. 1995;28:25–30. [Google Scholar]
- 10.Dai J, Mumper RJ. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules. 2010;15:7313–52. doi: 10.3390/molecules15107313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Prior RL, Wu X, Schaich K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J Agric Food Chem. 2005;53:4290–302. doi: 10.1021/jf0502698. [DOI] [PubMed] [Google Scholar]
- 12.Bartosz G. Food Oxidants and Antioxidants: Chemical, Biological, and Functional Properties. Boca Raton: CRC Press; 2013. [Google Scholar]
- 13.Amendment FA. 20 Flavoring. Food Technol. 2001;55:34–55. [Google Scholar]
- 14.Aziz AN, Ibrahim H, Rosmy Syamsir D, Mohtar M, Vejayan J, Awang K, et al. Antimicrobial compounds from Alpinia conchigera. J Ethnopharmacol. 2013;145:798–802. doi: 10.1016/j.jep.2012.12.024. [DOI] [PubMed] [Google Scholar]
- 15.Schultz TW, Netzeva TI. In Predicting Chemical Toxicity and Fate. Boca Raton: CRC Press; 2004. Development and evaluation of QSARs for ecotoxic endpoints: The benzene response-surface model for Tetrahymena toxicity. Modeling Environmental Fate and Toxicity; pp. 265–84. [Google Scholar]


