Abstract
Humic acid is natural biological organic, which has a high effect on plant growth and quality. However, the mechanisms of the promoting effect of humic acid on the volatile composition were rarely reported. In this study, the effects of soil application of humic acid on the chemical composition and nutrients uptake of Thymus vulgaris were investigated. Treatments comprised 0, 50, 75 and 100 g m−2. Essential oil was extracted by hydrodistillation and analyzed using GC–MS and GC–FID. Essential oil content was enhanced by increase of the humic acid level and its content ranged from 0.8% (control) to 2.0% (75 g m−2). Thirty-two volatile compounds were identified and these compounds were considerably affected by humic acid. The highest percentage of thymol (74.15%), carvacrol (6.20%), p-cymene (4.24%), borneol (3.42%), trans-caryophyllene (1.70%) and cis-sabinene hydrate (1.35%) as major compounds were observed in T. vulgaris under 100 g m−2 humic acid. There was a linear relationship (R2 = 97%) between humic acid levels and thymol as a major compound. The oils were dominated by oxygenated monoterpenes followed by monoterpene hydrocarbons and sesquiterpene hydrocarbons. Based on the path coefficient analysis, the highest direct effects on essential oil content were observed in monoterpene esters (3.465) and oxygenated sesquiterpenes (3.146). The humic acid application also enhanced the uptake of N, P, K, Mg and Fe in garden thyme. The highest N (2.42%), P (0.75%), K (2.63%), Mg (0.23%) and Fe (1436.58 ppm) were observed in medium supplemented with 100 g m−2 humic acid. In all, the utilization of humic acid could positively change nutrients uptake, essential oil content and its major constituents in T. vulgaris.
Keywords: Essential oil, Humic acid, Nutrients, Thymol
Introduction
The Thymus genus consisting of 215 species, is one of the most popular medicinal plants that belongs to the Lamiaceae family. Thyme is small shrub and herbaceous perennial in the Mediterranean region which has been recognized as the center of the thyme genus (Cronquist 1988). Thymus, with the popular Persian name of “Avishan or Azorbe,” consists of 14 native species which are found wild in many regions of Iran. Thymus vulgaris L. is a medicinal and aromatic plant which grows in the northern part of the Western Mediterranean region (Al-Ramamneh 2009), and it is commercially cultivated in many countries (Al-Ramamneh 2009; Badi et al. 2004; Letchamo et al. 1995). The inflorescence and volatile constituents of thyme are commonly used as the herbal tea, flavoring agents and for many medicinal purposes (Stahl-Biskup and Saez 2003). In Iran, decoction and infusion of the flowers, stem, and leaves of thyme are used as digestive, anti-inflammatory, carminative, expectorant and antispasmodic (Ghasemi Pirbalouti 2009; Nickavar et al. 2005). The essential oil from the shoots and leaves of T. vulgaris contains mainly monoterpenes, and sesquiterpenes (Ghasemi Pirbalouti et al. 2013, 2014). Previous studies have shown thymol, carvacrol, p-cymene and γ-terpinene as the major components of the essential oils from garden thyme (Ghasemi Pirbalouti et al. 2014; Nickavar et al. 2005).
Humic substances are the blackish- or brownish-colored organic compositions with large molecular weights and complex structures constructed by the decomposition of plant or animal remain (Lee et al. 2004). It has also been applied as soil amendments to ameliorate chemical and physical attributes (Suh et al. 2014). Humic substances include humic acid, humin, and fulvic acid depending on its solvability at various pH (Lee et al. 2004). It is demonstrated that humic substances, irrespective of their nature, origin, and characteristics, usually enhance plant yields, seed germination, physico-chemical characteristics and directly or indirectly stimulate absorption by roots (Nardi et al. 2002; Nikbakht et al. 2008).
Humic acid is known to contain carboxyl, alcoholic hydroxyl, ketone, quinoide and phenolic hydroxyl (Canellas et al. 2015). However, the chemical structures of humic substances have not clearly been clarified because of the complexity of the molecules (Lee et al. 2004). Consequently, the functional properties of molecules with various structures and reaction mechanisms are also not well known (Mora et al. 2010).
Nardi et al. (2002) proposed that humic acid could directly influence plant growth components such as cell permeability, respiration, photosynthesis, and cell elongation. Previous researches have shown other effects of humic substances on fruits (Arancon et al. 2006; Pilanal and Kaplan 2003), vegetables (Tuefenkci et al. 2006; Yildirim 2007), cereals (Delfine et al. 2005; Jones et al. 2007) and Lolium perenne (Verlinden et al. 2010). This was followed by a reduce in the incidence of plant disease (Naidu et al. 2013; Olivares et al. 2015; Singh et al. 2010). In addition to the notable changes on nutrient uptake and plant primary metabolism, secondary metabolism may also be strongly affected by humic substances (Canellas et al. 2015; Schiavon et al. 2010).
Varanini and Pinton (1995) reported that humic acid enhanced absorption of micro- and macro-elements in plants. Humic acid can enhance plant growth by promoting the bioavailability of nutrients via reform of the soil environment at the roots (Chen et al. 2004). Humic acid was the main source of nitrogen (N), phosphorus (P), and potassium (K) (Panuccio et al. 2001). Haghighi et al. (2014) showed that humic acid had advantageous effects on nutrient uptake, particularly in availability and transport of micronutrients.
In all, the effect of humic acid on the nutrients uptake and volatile composition of thyme has rarely been reported. Therefore, this study aimed to examine the effects of application to soil of humic acid on the nutrients uptake and chemical composition of garden thyme as new natural agricultural supplements.
Materials and methods
Plant material and cultivation
Garden thyme (T. vulgaris L. German cultivar ‘Deutscher Winter’) seedlings (F1) were purchased from a Pakan Seed Company, Isfahan, Iran. Seedlings were transferred on April 5 and harvested (about 25 cm heights) before flowering on October 6, 2016, at the greenhouse of medicinal plants garden, Hamadan (48°51′ N 34°80′ E), Iran. The unicolor and uniform plantlets (about 10 cm heights) were cultured in plots (1 × 1 m) filled with clay loam soil (36.25% clay, 30% silt, and 33.75% sand) and perlite (2:1). In addition, soil mixture depth was 30 cm. The chemical properties of the soil mixture are shown in Table 1. Each experimental plot accommodated nine plants. Three replications per treatment were involved in a randomized complete block design. Garden thyme was grown under controlled conditions in the greenhouse (day/night 16/8 h, light intensity 150 µmol m−2 s−1, 28 ± 2 °C/22 ± 2 °C day/night, and relative humidity 55–75%). Humic acid [containing nitrogen (N), 1.3%; carbon (C), 62.2%; oxygen (O), 36.8%; and hydrogen (H), 2.4%] was purchased from the Kimia Pars Shayankar Company, Tehran, Iran. The humic acid powder was spread and mixed well in plots. Treatments were as follows: (1) control, (2) 50 g m−2, (3) 75 g m−2and (4) 100 g m−2. In this study, no systemic pesticide and additional fertilizer were used during the entire experiment, and weed control was done manually. The plots were irrigated when 50% of soil available water was depleted (irrigation intervals varied from 7 to 9 days). Samples were aerial parts of nine plants in each plot and before the flowering stage were harvested.
Table 1.
Properties of the soil used in this study
| Characteristic | Unit | Content |
|---|---|---|
| Clay | % | 36.25 |
| Silt | % | 30 |
| Sand | % | 33.75 |
| Texture | – | Clay-loam |
| N (total) | mg kg−1 | 0.076 |
| P (available) | mg kg−1 | 5.33 |
| K (available) | mg kg−1 | 273 |
| EC | ds m−1 | 1.41 |
| pH | – | 7.32 |
| Organic matter | % | 1.19 |
EC electrical conductivity
Essential oil isolation
The essential oil of air-dried leaf samples (30 g) was isolated by hydrodistillation (250 ml of water in 500 ml round bottomed flask) for 3 h, using a Clevenger-type apparatus as recommended method in British pharmacopeia (British Pharmacopeia, 1998). The essential oil was dried over anhydrous sodium sulfate (Na2SO4) and kept at 4 °C in the dark vial until analyzed and tested. The essential oil content was calculated in % (v/w) of air dry leaf matter.
GC–FID analysis
GC analysis was performed using a Thermoquest gas chromatograph with a flame ionization detector (FID). The analysis was carried out on the fused silica capillary DB-5 column (30 m × 0.25 mm i.d.; film thickness 0.25 µm). The injector and detector temperatures were kept at 250 and 300 °C, respectively. N was used as the carrier gas at a flow rate of 1.1 ml min−1; oven temperature program was 60–250 °C at the rate of 4 °C min−1 and finally held isothermally for 10 min; split ratio was 1:50.
GC–MS analysis
The GC–MS analysis was carried out by use of Thermoquest-Finnigan gas chromatograph equipped with fused silica capillary DB-5 column (60 m × 0.25 mm i.d.; film thickness 0.25 µm) coupled with a TRACE mass (Manchester, UK). Helium was used as carrier gas with the ionization voltage of 70 eV. Ion source and interface temperatures were 200 and 250 °C, respectively. Mass range was from 35 to 456 amu. Oven temperature program was the same given above for the GC.
Identification and quantification of the oil components
The constituents of essential oils were identified by calculation of their retention indices under temperature-programmed conditions for n-alkanes (C6–C24) and the oil on a DB-5 column under the same chromatographic conditions. Identification of individual compounds was done by comparison of their mass spectra with those of the internal reference mass spectra library (Adams and Wiley 7.0) or with authentic compounds and confirmed by comparison of their retention indices with authentic compounds or with those reported in the literature (Adams 1997). For quantification purposes, relative area percentages obtained by FID were used without the use of correction factors.
Plant analysis
Fully expanded leaves were collected, washed thoroughly with tap water and then deionized water, dried at 65 °C to constant weight, and ground to determine their mineral composition. The determination of organically bound N in the leaf samples was based on the Kjeldahl method (Schuman et al. 1973). The extraction of K, P, magnesium (Mg), and iron (Fe) from the plant tissue material was performed using 1 N hydrochloric acid (HCl) after dry ashing at 550 °C for 5 h (Savvas and Gizas 2002). Total K was directly measured by flame spectrophotometry using an Evans Electro Selenium LTB (Plaza et al. 2016) flame photometer. The concentrations of Mg and Fe in the leaf extracts were determined by atomic absorption spectrophotometry (Perkin-Elmer, Waltham, Mass., USA), while P was determined by the vanadomolybdophosphoric colorimetric method (Hogue et al. 1970), using a Helios Gamma spectrophotometer.
Data analysis
The experiment was conducted in a one-way analysis of variance with three replications. Data were subjected to analysis of variance and means were separated by Duncan’s multiple range test (DMRT) at P < 0.05 significance level in SAS (Software Version 9.1 SAS). Correlation and path analysis were performed in SPSS (IBM SPSS Software Version 22) and Path2 software, respectively. Path analysis was carried out using the procedure suggested by Dewey and Lu (1959), which has been used to quantify a perceived biological relationship through partitioning of correlation coefficients into direct and indirect effects.
Results and discussion
This study examined the effects of various levels of humic acid (control, 50, 75 and 100 g m−2) on the chemical composition of T. vulgaris. Results showed that with the increase of humic acid levels, the essential oil content was enhanced compared to the control (Table 2). The essential oil content ranged from 0.8% (control) to 2.0% (75 g m−2). The chemical composition of the garden thyme was evaluated using both GC–FID and GC–MS techniques. Quantity and quality of chemical compositions were affected by humic acid treatments (Table 2). The GC–MS analysis resulted in the identification of thirty-two constituents of the oil composition. Their sums constitution of the oils were 98.64% (control), 99.55% (50 g m−2), 99.52% (75 g m−2) and 99.80% (100 g m−2).
Table 2.
Effect of soil application of humic acid on oil chemical compositions of Thymus vulgaris
| No. | Compound | RIa | %b | ANOVAd | |||
|---|---|---|---|---|---|---|---|
| 1c | 2 | 3 | 4 | ||||
| 1. | (E)-2-Hexenal | 867.44 | 0.14 | 0.16 | 0.14 | 0.15 | – |
| Aliphatic aldehydes | – | 0.14 a | 0.16 a | 0.14 a | 0.15 a | ns | |
| 2. | α-Thujene | 925 | 0.23 | 0.27 | 0.27 | 0.26 | – |
| 3. | α-Pinene | 931.97 | 0.25 | 0.25 | 0.23 | 0.27 | – |
| 4. | Camphene | 947.09 | 0.23 | 0.20 | 0.20 | 0.25 | – |
| 5. | Sabinene | 970.93 | 0.04 | 0.07 | 0.03 | 0.06 | – |
| 6. | β-Myrcene | 987.20 | 0.16 | 0.18 | 0.19 | 0.20 | – |
| 7. | α-Terpinene | 1014.66 | 0.35 | 0.50 | 0.55 | 0.48 | – |
| 8. | p-Cymene | 1022.22 | 3.46b | 4.26a | 3.71b | 4.24a | * |
| 9. | Limonene | 1025.77 | 0.10 | 0.12 | 0.18 | 0.21 | – |
| 10. | γ-Terpinene | 1054.66 | 2.24b | 2.35b | 2.79a | 2.40b | * |
| 11. | α-Terpinolene | 1084.88 | 0.06 | 0.07 | 0.10 | 0.05 | – |
| Monoterpene hydrocarbons | – | 7.12 a | 8.27 b | 8.25 b | 8.42 a | * | |
| 12. | 1-Octen-3-ol | 975 | 0.93 | 0.99 | 0.92 | 0.9 | – |
| Aliphatic alcohols | – | 0.93 a | 0.99 a | 0.92 a | 0.9 a | ns | |
| 13. | 1,8-Cineole | 1028.44 | 0.58 | 0.45 | 0.47 | 0.53 | – |
| 14. | cis-Sabinene hydrate | 1063.55 | 1.33a | 1.18a | 1.24a | 1.35a | ns |
| 15. | Linalool | 1096 | 1.73a | 1.65a | 1.64a | 1.58a | ns |
| 16. | Borneol | 1168.10 | 3.12b | 3.11b | 3.19b | 3.42a | * |
| 17. | 4-Terpineol | 1179.74 | 0.63 | 0.76 | 0.67 | 0.60 | – |
| 18. | α-Terpineol | 1199.56 | 0.17 | 0.30 | 0.20 | 0.12 | – |
| 19. | Thymol | 1297.87 | 73.08b | 73.28ab | 73.78ab | 74.15a | * |
| 20. | Carvacrol | 1303.13 | 6.18a | 5.99a | 6.03a | 6.20a | ns |
| Oxygenated monoterpenes | – | 86.82 b | 86.72 b | 87.22 ab | 87.95 a | * | |
| 21. | Bornyl formate | 1228.01 | 0.18 | 0.13 | 0.14 | 0.12 | – |
| 22. | Linalyl acetate | 1263.83 | 0.11 | 0.12 | 0.16 | 0.13 | – |
| 23. | Thymol acetate | – | – | – | – | 0.09 | – |
| 24. | Geranyl propanoate | 1464.22 | 0.25 | 0.21 | 0.19 | 0.22 | – |
| 25. | Isobornyl propionate | 1368.62 | 0.11 | 0.1 | 0.11 | 0.13 | – |
| 26. | Citronellyl propionate | 1432.92 | 0.06 | 0.06 | – | 0.08 | – |
| Monoterpenes esters | – | 0.71 ab | 0.62 ab | 0.06 b | 0.77 a | * | |
| 27. | trans-Caryophyllene | 1412.19 | 1.69a | 1.61a | 1.49a | 1.70a | ns |
| 28. | α-Humulene | 1446.34 | 0.09 | 0.08 | – | 0.06 | – |
| 29. | γ-Cadinene | 1468.30 | – | 0.05 | – | – | – |
| 30. | τ-Cadinene | 1505.57 | 0.12 | 0.11 | 0.1 | 0.1 | – |
| 31. | δ-Cadinene | 1515.02 | 0.22 | 0.25 | 0.23 | 0.22 | – |
| Sesquiterpene hydrocarbons | – | 2.12 a | 2.1 a | 1.82 a | 2.08 a | ns | |
| 32. | Ledene oxide-(II) | 1575.53 | 0.8 | 0.65 | 0.57 | 0.91 | – |
| Oxygenated sesquiterpenes | – | 0.8 c | 0.65 b | 0.57 b | 0.91 a | * | |
| Total | – | 98.64 | 99.55 | 99.52 | 99.80 | ||
| Essential oil content (v/w) | – | 0.8c | 1.5b | 2a | 1.8a | * | |
aRI: retention indices relative to C6–C24 n-alkanes on a DB-5 column
bRelative percentage obtained from peak area
cDifferent levels of humic acid: 1. 0 g m−2; 2. 50 g m−2; 3. 75 g m−2; 4. 100 g m−2
dAnalysis of variance; values of major compounds with same letters in a row are not significantly different at 5% level using Duncan’s multiple range test
Values of essential oil families are shown in bold
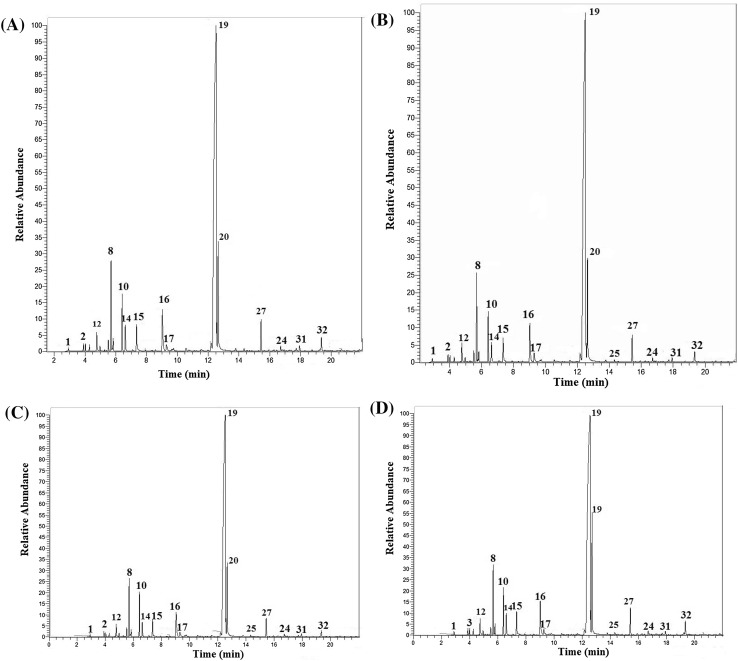
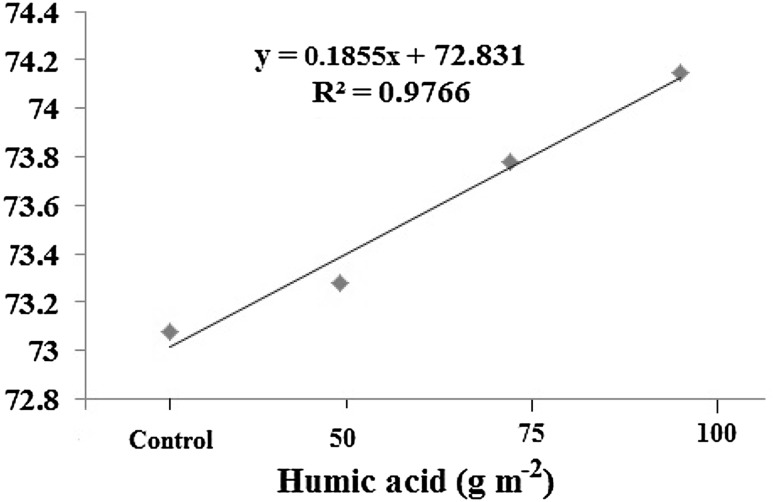
The major identified compounds in the oil from T. vulgaris under humic acid application were thymol, carvacrol, p-cymene, borneol, γ-terpinene, trans-caryophyllene and cis-sabinene hydrate, respectively. Above-mentioned major compounds in thyme species have previously been reported (Amiri 2011; Ghasemi Pirbalouti et al. 2013; Rahimmalek et al. 2009; Sajjadi and Khatamsaz 2003). Results of the statistical analysis showed that there were considerable differences between humic acid treatments in major volatile compounds (Table 2). The soil application of humic acid noticeably increased thymol, p-Cymene and borneol percentages, but there were no statistically significant correlation between humic acid treatments and carvacrol and linalool percentages. GC–FID chromatograms of the essential oils from different levels of humic acid with identified major compounds are presented in Fig. 1. The peaks of some compounds were overlapped and not separated. Therefore, these compounds are not shown in Fig. 1. The results showed that the most compounds amount affected by humic acid (Table 2). These increases in some main constituents were considerable. The highest percentage of thymol (74.15%), carvacrol (6.20%), p-cymene (4.24%), borneol (3.42%), trans-caryophyllene (1.70%) and cis-sabinene hydrate (1.35%) were observed in T. vulgaris under 100 g m−2 humic acid. The lowest percentage of thymol, carvacrol and p-cymene were observed in control treatments. However, there were no significant differences between treatment 2 and 3 and control. A linear relationship (R2 = 97%) was found between humic acid levels and thymol as a major compound (Fig. 2). Thymol, as a major constituent of the essential oil of the thyme, is an oxygenated monoterpene which configured in chloroplasts from freshly fixed carbon (Bohlmann et al. 1998) and its levels maybe related to CO2 formation and acquisition of photosynthesis intermediates (Loreto et al. 1996).
Fig. 1.
Gas chromatography-flame ionization detector (GC–FID) chromatograms of the essential oil from Thymus vulgaris affected by a 0 g m−2, b 50 g m−2, c 75 g m−2, d 100 g m−2. Compounds codes are shown in Table 1
Fig. 2.
Regression relationship between humic acid levels and thymol as a major compound in the oil of Thymus vulgaris
As shown in Table 2, identified compounds based on chemical families have been classified into seven groups: (1) aliphatic aldehydes, (2) monoterpene hydrocarbons, (3) aliphatic alcohols, (4) oxygenated monoterpenes, (5), monoterpenes esters (6) sesquiterpene hydrocarbons and (7) oxygenated sesquiterpenes. Similarly, previous researches indicated that major chemical groups acquired from the essential oils of T. vulgaris were oxygenated monoterpenes and monoterpene hydrocarbons, and the amounts of other groups were low (Alavi-Samani et al. 2015; Amiri 2011; Ghasemi Pirbalouti et al. 2014). The results showed that humic acid treatments had significant effects on monoterpene hydrocarbons, oxygenated monoterpenes, monoterpenes esters and Oxygenated sesquiterpenes. The highest monoterpene hydrocarbons (8.42%), oxygenated monoterpenes (87.95%), monoterpenes esters (0.77%) and Oxygenated sesquiterpenes (0.91%) were noticeably affected by medium supplemented with 100 g m−2 humic acid. While humic acid application had no significant effects on aliphatic aldehydes, aliphatic alcohols, and sesquiterpene hydrocarbons. Previously, the effect of humic acid application on other secondary metabolites has been reported. Schiavon et al. (2010) showed that humic acid increased the expression of the phenylalanine ammonialyase that catalyzes the first phase in the biosynthesis of phenylpropanoid, by transforming tyrosine to p-coumaric acid and phenylalanine to trans-cinnamic acid. The expression of these enzymes was accompanied by phenol repletion in leaves. It has been reported that the compounds related to the shikimic pathway (flavonoids, some alkaloids such as isoquinoline alkaloids, tocopherols, and phenols) are agitated by humic acid (Schiavon et al. 2010). In addition, an enzymatic system such as peroxidase and non-enzymatic antioxidant systems such as glutathione, ascorbate, phenols, alkaloids, carotenoids and tocopherols have been affected by humic acid treatments (Canellas et al. 2015; Pizzeghello et al. 2001).
The correlations coefficients among chemical essential oil families and essential oil content in T. vulgaris are presented in Table 3. Oxygenated sesquiterpenes were positively correlated with monoterpenes esters, sesquiterpene hydrocarbons, and essential oil content. Correlation between monoterpene hydrocarbons and oxygenated monoterpenes were significantly positive. Also, there were significantly positive correlations between aliphatic aldehydes with monoterpene hydrocarbons and aliphatic alcohols. In addition, monoterpenes esters were significantly correlated to sesquiterpene hydrocarbons, oxygenated sesquiterpenes, and essential oil content. But no positive correlation was observed between other indices.
Table 3.
Correlation between essential oils components in Thymus vulgaris
| Variables | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| 1. Aliphatic aldehydes | 1 | 0.960* | 0.678ns | 0.985* | 0.489ns | 0.484ns | 0.276ns | 0.484ns |
| 2. Monoterpene hydrocarbons | 1 | 0.778ns | 0.900* | 0.709ns | 0.704ns | 0.526ns | 0.704ns | |
| 3. Aliphatic alcohols | 1 | 0.601ns | 0.807* | 0.809* | 0.732ns | 0.810* | ||
| 4. Oxygenated monoterpenes | 1 | 0.341ns | 0.336ns | 0.119ns | 0.336ns | |||
| 5. Monoterpenes esters | 1 | 0.999** | 0.972* | 0.999** | ||||
| 6. Sesquiterpene hydrocarbons | 1 | 0.974* | 0.999** | |||||
| 7. Oxygenated sesquiterpenes | 1 | 0.974* | ||||||
| 8. Essential oil content | 1 |
*Significant at P ≤ 0.05
**Significant at P ≤ 0.01
nsNon-significant
The path coefficient analysis results are shown in Table 4. The highest direct effects on essential oil content were observed in monoterpene esters (3.465) and oxygenated sesquiterpenes (3.146). The direct effects of aliphatic aldehydes, monoterpene hydrocarbons, and sesquiterpene hydrocarbons were negative. Whereas, the highest positive indirect effect of aliphatic aldehydes, monoterpene hydrocarbons and sesquiterpene hydrocarbons via monoterpene esters were 1.677, 2.439 and 3.375, respectively. Path coefficient analysis helps in the dividing of correlation coefficient into indirect and direct effects of different traits on antioxidant or any other attributes (Khan et al. 2009). The results showed that monoterpenes esters, oxygenated sesquiterpenes, and oxygenated monoterpenes were effective major factors on essential oil content.
Table 4.
Path analysis of essential oil components in Thymus vulgaris
| Character | 1 | 2 | 3 | 4 | 5 | 6 | 7 | Pooled effects |
|---|---|---|---|---|---|---|---|---|
| 1. Aliphatic aldehydes | − 0.344 | − 2.147 | 0.040 | 0.916 | 1.538 | 1.677 | − 1.199 | 0.484 |
| 2. Monoterpene hydrocarbons | − 0.330 | − 2.237 | 0.046 | 0.837 | 2.230 | 2.439 | − 2.285 | 0.704 |
| 3. Aliphatic alcohols | − 0.233 | − 1.74 | 0.060 | 0.559 | 2.539 | 2.803 | − 3.180 | 0.810 |
| 4. Oxygenated monoterpenes | − 0.339 | − 2.013 | 0.036 | 0.930 | 1.072 | 1.164 | − 0.517 | 0.336 |
| 5. Oxygenated sesquiterpenes | − 0.169 | − 1.586 | 0.048 | 0.317 | 3.146 | 3.461 | − 4.231 | 0.999 |
| 6. Monoterpenes esters | − 0.167 | − 1.575 | 0.048 | 0.312 | 3.143 | 3.465 | − 4.231 | 0.999 |
| 7. Sesquiterpene hydrocarbons | − 0.095 | − 1.177 | 0.044 | 0110 | 3.075 | 3.375 | − 4.344 | 0.974 |
Bold values are direct effect
As shown in Table 5, the effect of humic acid on nutrients content of leaves was markedly significant. The highest N (2.42%), P (0.75%), K (2.63%), Mg (0.23%) and Fe (1436.58 ppm) were observed in medium supplemented with 100 g m−2 humic acid. These finding has been approved by previous researches (Atiyeh et al. 2002; Ayuso et al. 1996; Yazdani et al. 2014). Humic acid plays a role in the incorporation of nitrate uptake through an interaction with plasma membrane H+-ATPase (Pinton et al. 1999). In addition, it can increase the solubility P compounds in soil with its chelating capacity. Previous researches have shown that humic acid has been widely considered as natural chelates for cationic micronutrients (Varanini and Pinton 1995). Sanchez-Sanchez et al. (2006) reported that the addition of humic acid might ameliorate Fe uptake by chelating free Fe and so hamper its immobilization. Morard et al. (2010) also described that cationic micronutrients could be related to hydrogen bonds inward the structure of humic acid. Humic acid might upgrade plant development via the induction of carbon and N metabolism. Glutamate dehydrogenase, nitrate reductase and glutamine synthetase are enzymes connected to N assimilation pathways and were incited by humic acid (Canellas et al. 2013; Hernandez et al. 2015). Humic acid is considered to stimulate microbial activity. This biological activity increased the enhancement of water solvable P and total N content (Busato et al. 2012).
Table 5.
Effects of different levels of humic acid on N, P, K, Mg and Fe concentrations of the leaves of Thymus vulgaris
| Humic acid (g m−2) | N (%) | P (%) | K (%) | Mg (%) | Fe (ppm) |
|---|---|---|---|---|---|
| 0 | 0.76d | 0.14c | 0.80c | 0.10d | 399.06d |
| 50 | 1.25c | 0.31b | 2.10b | 0.13c | 781.35c |
| 75 | 1.78b | 0.64a | 2.22b | 0.20b | 1149.57b |
| 100 | 2.42a | 0.75a | 2.63a | 0.23a | 1436.58a |
Mean in each column followed by same letters are not significantly different at 5% level using Duncan’s multiple range test
Conclusion
The present study indicated that humic acid affected secondary metabolites (quantity and quality of chemical compositions) and nutrients uptake in T. vulgaris. Thus, the utilization of humic acid could be pondered as a strong biotechnological approach for plant growth development in sustainable agriculture systems. In addition, humic acid is constructed to positively change biological activities, essential oil content and its major constituents in T. vulgaris.
Acknowledgements
The authors gratefully acknowledge the Sayyed Jamaleddin Asadabadi University Research Council for financial support of this project.
Compliance with ethical standards
Conflict of interest
The authors declare there is no conflict of interest.
Contributor Information
Alireza Noroozisharaf, Phone: +98 9188121637, Email: noroozi2ar@yahoo.com, Email: noroozi@sjau.ac.ir.
Maryam Kaviani, Email: kavianimaryam170@yahoo.com.
References
- Adams R. Identification of essential oil components by gas chromatography/mass spectroscopy. J Am Soc Mass Spectrom. 1997;6:671–672. [Google Scholar]
- Alavi-Samani SM, Kachouei MA, Pirbalouti AG. Growth, yield, chemical composition, and antioxidant activity of essential oils from two thyme species under foliar application of jasmonic acid and water deficit conditions. Hortic Environ Biotechnol. 2015;56:411–420. doi: 10.1007/s13580-015-0117-y. [DOI] [Google Scholar]
- Al-Ramamneh EALDM. Plant growth strategies of Thymus vulgaris L. in response to population density. Ind Crop Prod. 2009;30:389–394. doi: 10.1016/j.indcrop.2009.07.008. [DOI] [Google Scholar]
- Amiri H. Essential oils composition and antioxidant properties of three thymus species. Evid Based Complement Altern Med. 2011;2012:1–8. doi: 10.1155/2012/728065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arancon NQ, Edwards CA, Lee S, Byrne R. Effects of humic acids from vermicomposts on plant growth. Eur J Soil Biol. 2006;42:S65–S69. doi: 10.1016/j.ejsobi.2006.06.004. [DOI] [Google Scholar]
- Atiyeh R, Lee S, Edwards C, Arancon N, Metzger J. The influence of humic acids derived from earthworm-processed organic wastes on plant growth. Bioresour Technol. 2002;84:7–14. doi: 10.1016/S0960-8524(02)00017-2. [DOI] [PubMed] [Google Scholar]
- Ayuso M, Hernández T, García C. Effect of humic fractions from urban wastes and other more evolved organic materials on seed germination. J Sci Food Agric. 1996;72:461–468. doi: 10.1002/(SICI)1097-0010(199612)72:4<461::AID-JSFA680>3.0.CO;2-#. [DOI] [Google Scholar]
- Badi HN, Yazdani D, Ali SM, Nazari F. Effects of spacing and harvesting time on herbage yield and quality/quantity of oil in thyme, Thymus vulgaris L. Ind Crops Prod. 2004;19:231–236. doi: 10.1016/j.indcrop.2003.10.005. [DOI] [Google Scholar]
- Bohlmann J, Meyer-Gauen G, Croteau R. Plant terpenoid synthases: molecular biology and phylogenetic analysis. Proc Natl Acad Sci. 1998;95:4126–4133. doi: 10.1073/pnas.95.8.4126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busato JG, Lima LS, Aguiar NO, Canellas LP, Olivares FL. Changes in labile phosphorus forms during maturation of vermicompost enriched with phosphorus-solubilizing and diazotrophic bacteria. BioresourTechnol. 2012;110:390–395. doi: 10.1016/j.biortech.2012.01.126. [DOI] [PubMed] [Google Scholar]
- Canellas LP, Balmori DM, Médici LO, Aguiar NO, Campostrini E, Rosa RC, Façanha AR, Olivares FL. A combination of humic substances and Herbaspirillum seropedicae inoculation enhances the growth of maize (Zea mays L.) Plant Soil. 2013;366:119–132. doi: 10.1007/s11104-012-1382-5. [DOI] [Google Scholar]
- Canellas LP, Olivares FL, Aguiar NO, Jones DL, Nebbioso A, Mazzei P, Piccolo A. Humic and fulvic acids as biostimulants in horticulture. Sci Hortic. 2015;196:15–27. doi: 10.1016/j.scienta.2015.09.013. [DOI] [Google Scholar]
- Chen Y, Clapp C, Magen H. Mechanisms of plant growth stimulation by humic substances: the role of organo-iron complexes. Soil Sci Plant Nutr. 2004;50:1089–1095. doi: 10.1080/00380768.2004.10408579. [DOI] [Google Scholar]
- Cronquist A. The evolution and classification of flowering plants. New York: The New York Botanical Garden; 1988. [Google Scholar]
- Delfine S, Tognetti R, Desiderio E, Alvino A. Effect of foliar application of N and humic acids on growth and yield of durum wheat. Agron Sustain Dev. 2005;25:183–191. doi: 10.1051/agro:2005017. [DOI] [Google Scholar]
- Dewey DR, Lu K. A correlation and path-coefficient analysis of components of crested wheatgrass seed production. Agron J. 1959;51:515–518. doi: 10.2134/agronj1959.00021962005100090002x. [DOI] [Google Scholar]
- Ghasemi Pirbalouti A. Medicinal plants used in Chaharmahal and Bakhtyari districts of Iran. Herba Pol. 2009;55:69–77. [Google Scholar]
- Ghasemi Pirbalouti A, Hashemi M, Ghahfarokhi FT. Essential oil and chemical compositions of wild and cultivated Thymus daenensis Celak and Thymus vulgaris L. Ind Crops Prod. 2013;48:43–48. doi: 10.1016/j.indcrop.2013.04.004. [DOI] [Google Scholar]
- Ghasemi Pirbalouti A, Samani MR, Hashemi M, Zeinali H. Salicylic acid affects growth, essential oil and chemical compositions of thyme (Thymus daenensis Celak.) under reduced irrigation. Plant Growth Regul. 2014;72:289–301. doi: 10.1007/s10725-013-9860-1. [DOI] [Google Scholar]
- Haghighi M, Nikbakht A, Xia YP, Pessarakli M. Influence of humic acid in diluted nutrient solution on growth, nutrient efficiency, and postharvest attributes of gerbera. Commun Soil Sci Plant Anal. 2014;45:177–188. doi: 10.1080/00103624.2013.848885. [DOI] [Google Scholar]
- Hernandez OL, Calderín A, Huelva R, Martínez-Balmori D, Guridi F, Aguiar NO, Olivares FL, Canellas LP. Humic substances from vermicompost enhance urban lettuce production. Agron Sustain Dev. 2015;35:225–232. doi: 10.1007/s13593-014-0221-x. [DOI] [Google Scholar]
- Hogue E, Wilcox G, Cantliffe D. Effect of soil phosphorus levels on phosphate fractions in tomato leaves. J Am Soc Hortic Sci. 1970;95:174–176. [Google Scholar]
- Jones CA, Jacobsen JS, Mugaas A. Effect of low-rate commercial humic acid on phosphorus availability, micronutrient uptake, and spring wheat yield. Commun Soil Sci Plant Anal. 2007;38:921–933. doi: 10.1080/00103620701277817. [DOI] [Google Scholar]
- Khan A, Kabir M, Alam M. Variability, correlation path analysis of yield and yield components of pointed gourd. J Agric Rural Dev. 2009;7:93–98. [Google Scholar]
- Lee C-H, Shin H-S, Kang K-H. Chemical and spectroscopic characterization of peat moss and its different humic fractions (Humin, Humic acid and fulvic acid) J Soil Groundw Environ. 2004;9:42–51. [Google Scholar]
- Letchamo W, Xu H, Gosselin A. Variations in photosynthesis and essential oil in thyme. J Plant Physiol. 1995;147:29–37. doi: 10.1016/S0176-1617(11)81408-2. [DOI] [Google Scholar]
- Loreto F, Ciccioli P, Cecinato A, Brancaleoni E, Frattoni M, Fabozzi C, Tricoli D. Evidence of the photosynthetic origin of monoterpenes emitted by Quercus ilex L. leaves by 13C labeling. Plant Physiol. 1996;110:1317–1322. doi: 10.1104/pp.110.4.1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mora V, Bacaicoa E, Zamarreño A-M, Aguirre E, Garnica M, Fuentes M, García-Mina J-M. Action of humic acid on promotion of cucumber shoot growth involves nitrate-related changes associated with the root-to-shoot distribution of cytokinins, polyamines and mineral nutrients. J Plant Physiol. 2010;167:633–642. doi: 10.1016/j.jplph.2009.11.018. [DOI] [PubMed] [Google Scholar]
- Morard P, Eyheraguibel B, Morard M, Silvestre J. Direct effects of humic-like substance on growth, water, and mineral nutrition of various species. J Plant Nutr. 2010;34:46–59. doi: 10.1080/01904167.2011.531358. [DOI] [Google Scholar]
- Naidu Y, Meon S, Siddiqui Y. Foliar application of microbial-enriched compost tea enhances growth, yield and quality of muskmelon (Cucumis melo L.) cultivated under fertigation system. Sci Hortic. 2013;159:33–40. doi: 10.1016/j.scienta.2013.04.024. [DOI] [Google Scholar]
- Nardi S, Pizzeghello D, Muscolo A, Vianello A. Physiological effects of humic substances on higher plants. Soil Biol Biochem. 2002;34:1527–1536. doi: 10.1016/S0038-0717(02)00174-8. [DOI] [Google Scholar]
- Nickavar B, Mojab F, Dolat-Abadi R. Analysis of the essential oils of two Thymus species from Iran. Food Chem. 2005;90:609–611. doi: 10.1016/j.foodchem.2004.04.020. [DOI] [Google Scholar]
- Nikbakht A, Kafi M, Babalar M, Xia YP, Luo A, N-a Etemadi. Effect of humic acid on plant growth, nutrient uptake, and postharvest life of gerbera. J Plant Nutr. 2008;31:2155–2167. doi: 10.1080/01904160802462819. [DOI] [Google Scholar]
- Olivares FL, Aguiar NO, Rosa RCC, Canellas LP. Substrate biofortification in combination with foliar sprays of plant growth promoting bacteria and humic substances boosts production of organic tomatoes. Sci Hortic. 2015;183:100–108. doi: 10.1016/j.scienta.2014.11.012. [DOI] [Google Scholar]
- Panuccio MR, Muscolo A, Nardi S. Effect of humic substances on nitrogen uptake and assimilation in two species of pinus. J Plant Nutr. 2001;24:693–704. doi: 10.1081/PLN-100103663. [DOI] [Google Scholar]
- Pilanal N, Kaplan M. Investigation of effects on nutrient uptake of humic acid applications of different forms to strawberry plant. J Plant Nutr. 2003;26:835–843. doi: 10.1081/PLN-120018568. [DOI] [Google Scholar]
- Pinton R, Cesco S, Iacolettig G, Astolfi S, Varanini Z. Modulation of NO−3 uptake by water-extractable humic substances: involvement of root plasma membrane H+ ATPase. Plant Soil. 1999;215:155–161. doi: 10.1023/A:1004752531903. [DOI] [Google Scholar]
- Pizzeghello D, Nicolini G, Nardi S. Hormone-like activity of humic substances in Fagus sylvaticae forests. New Phytol. 2001;151:647–657. doi: 10.1046/j.0028-646x.2001.00223.x. [DOI] [PubMed] [Google Scholar]
- Plaza BM, Soriano F, Jiménez-Becker S, Lao MT. Nutritional responses of Cordyline fruticosa var. ‘Red Edge’ to fertigation with leachates versus conventional fertigation: chloride, nitrogen, phosphorus and sulphate. Agric Water Manag. 2016;173:61–66. doi: 10.1016/j.agwat.2016.04.031. [DOI] [Google Scholar]
- Rahimmalek M, Bahreininejad B, Khorrami M, Tabatabaei BES. Genetic variability and geographic differentiation in Thymus daenensis subsp. daenensis, an endangered medicinal plant, as revealed by inter simple sequence repeat (ISSR) markers. Biochem Genet. 2009;47:831–842. doi: 10.1007/s10528-009-9281-z. [DOI] [PubMed] [Google Scholar]
- Sajjadi SE, Khatamsaz M. Composition of the essential oil of Thymus daenensis Celak. ssp. lancifolius (Celak.) Jalas. J Essent Oil Res. 2003;15:34–35. doi: 10.1080/10412905.2003.9712257. [DOI] [Google Scholar]
- Sanchez-Sanchez A, Sanchez-Andreu J, Juarez M, Jorda J, Bermudez D. Improvement of iron uptake in table grape by addition of humic substances. J Plant Nutr. 2006;29:259–272. doi: 10.1080/01904160500476087. [DOI] [Google Scholar]
- Savvas D, Gizas G. Response of hydroponically grown gerbera to nutrient solution recycling and different nutrient cation ratios. Sci Hortic. 2002;96:267–280. doi: 10.1016/S0304-4238(02)00054-7. [DOI] [Google Scholar]
- Schiavon M, Pizzeghello D, Muscolo A, Vaccaro S, Francioso O, Nardi S. High molecular size humic substances enhance phenylpropanoid metabolism in maize (Zea mays L.) J Chem Ecol. 2010;36:662–669. doi: 10.1007/s10886-010-9790-6. [DOI] [PubMed] [Google Scholar]
- Schuman G, Stanley M, Knudsen D. Automated total nitrogen analysis of soil and plant samples. Soil Sci Soc Am J. 1973;37:480–481. doi: 10.2136/sssaj1973.03615995003700030045x. [DOI] [Google Scholar]
- Singh R, Gupta R, Patil R, Sharma R, Asrey R, Kumar A, Jangra K. Sequential foliar application of vermicompost leachates improves marketable fruit yield and quality of strawberry (Fragaria × ananassa Duch.) Sci Hortic. 2010;124:34–39. doi: 10.1016/j.scienta.2009.12.002. [DOI] [Google Scholar]
- Stahl-Biskup E, Saez F. Thyme, the Genus Thymus. London: Taylor and Francis; 2003. p. 331. [Google Scholar]
- Suh HY, Yoo KS, Suh SG. Tuber growth and quality of potato (Solanum tuberosum L.) as affected by foliar or soil application of fulvic and humic acids. Hortic Environ Biotechnol. 2014;55:183–189. doi: 10.1007/s13580-014-0005-x. [DOI] [Google Scholar]
- Tuefenkci S, Türkmen Ö, Sönmez F, Erdinc C, Sensoy S. Effects of humic acid doses and aplication times on the plant growth, nutrient and heavy metal contents of lettuce grown on sewage sludge-applied soils. Fres Environ Bull. 2006;15:295–300. [Google Scholar]
- Varanini Z, Pinton R. Humic substances and plant nutrition, progress in botany. Berlin: Springer; 1995. pp. 97–117. [Google Scholar]
- Verlinden G, Coussens T, De Vliegher A, Baert G, Haesaert G. Effect of humic substances on nutrient uptake by herbage and on production and nutritive value of herbage from sown grass pastures. Grass Forage Sci. 2010;65:133–144. doi: 10.1111/j.1365-2494.2009.00726.x. [DOI] [Google Scholar]
- Yazdani B, Nikbakht A, Etemadi N. Physiological effects of different combinations of humic and fulvic acid on Gerbera. Commun Soil Sci Plant Anal. 2014;45:1357–1368. doi: 10.1080/00103624.2013.875200. [DOI] [Google Scholar]
- Yildirim E. Foliar and soil fertilization of humic acid affect productivity and quality of tomato. Acta Agric Scand Sect B Soil Plant Sci. 2007;57:182–186. [Google Scholar]