Abstract
Background
The aim of this study was to explore the differences in the clinical characteristics and diagnostic rates of diabetes mellitus (DM) according to various criteria in different age groups and to evaluate the efficacy of each criterion for screening older patients.
Methods
We studied 515 patients and measured the fasting plasma glucose level (FPG), 2-hour plasma glucose level after the 75 g oral glucose tolerance test (2-hour postload glucose [2-h PG]), and glycosylated hemoglobin (HbA1c) for re-evaluation of hyperglycemia without a history of diabetes. Patients with newly diagnosed DM were grouped by age as younger (<65 years) or older (≥65 years).
Results
Older patients had significantly lower HbA1c, FPG, and 2-h PG levels and a higher homeostatic level of pancreatic β-cell function compared with younger patients (P<0.001). The older group had the lowest diagnostic rate when using the FPG level (45.5%) and the highest diagnostic rate when using the 2-h PG level (84.6%). These results were mostly due to the higher frequency of isolated post-challenge hyperglycemia in the older patients than in the younger group (28.8% vs. 9.2%). The use of both the FPG and HbA1c levels significantly enhanced the low diagnostic power when employing only the FPG levels in the older group (71.2% vs. 45.5%).
Conclusion
In the older patients, the 2-h PG level was the most accurate diagnostic criterion. When we consider the costs and convenience, a combination of the FPG and HbA1c criteria may be recommended as a screening test for DM in older people.
Keywords: Age, Diabetes mellitus, Postprandial glucose
INTRODUCTION
Type 2 diabetes mellitus (T2DM) has become a major medical concern because of continuing increases in prevalence, associated mortality, and medical costs. According to the 2017 National Diabetes Statistics report, diabetes affects 30.3 million people, or 9.4% of the population in the United States, and its prevalence increases with age [1]. It is common for T2DM to remain undiagnosed for at least 4 to 7 years, and chronic complications are already evident in >30% of newly diagnosed patients, even in those exhibiting impaired glucose tolerance [2,3,4,5]. Several studies have shown that early T2DM detection using optimal screening tests not only lowers the risk of chronic complications but also reduces the economic burden [6,7]. Thus, diabetes is increasingly screened for in populations with high risk factors, such as race, age, and a high body mass index (BMI).
Since an international expert committee suggested that the 2-hour plasma glucose level after the 75 g oral glucose tolerance test (OGTT) (2-hour postload glucose [2-h PG] level) be replaced by measurement of glycosylated hemoglobin (HbA1c) levels [8], the fasting plasma glucose (FPG) and/or the HbA1c level have been used to screen for diabetes because of convenience and low cost. However, both approaches have certain limitations. A recent study showed that the FPG is less sensitive than the 2-h PG level, especially in older adults [9]. Some studies indicated that the HbA1c level cutoff values should differ according to ethnicity and age [10,11,12]. A recent survey performed in the United States reported that the prevalence of diabetes varied depending on the diagnostic criteria employed [13]. Screening test limitations can create diagnostic errors; indeed, approximately 30% of individuals with diabetes are, in fact, undiagnosed. Because the peak prevalence of diabetes occurs in adults aged ≥65 years [1,14], the assumption might be that most cases of undiagnosed diabetes among those ≥18 years of age occur in adults aged ≥65 years. However, no optimal age-specific screening methods, or age-specific cutoff values have yet been defined.
The present study sought to define the clinical characteristics and the diagnostic rates of T2DM produced by various criteria in different age groups to determine the efficacy of each criterion for screening. We also aimed to define cutoff values that would reduce false-negative diagnoses.
METHODS
Study population
We retrospectively selected 62,248 adults aged 18 years or older without typical hyperglycemic symptoms who returned for re-evaluation of hyperglycemia at the Diabetes Center at Chungnam National University Hospital from March 2011 to February 2016. Among these patients, we only included 783 who had undertaken the OGTT and measurement of the HbA1c level. We excluded 40 patients previously diagnosed with diabetes and/or those treated with hypoglycemic agents, 214 with anemia, six with end-stage renal disease, and eight treated with anticancer chemotherapies or hyperglycemia-inducing agents, such as glucocorticoids, interferon γ, and thiazides. According to the inclusion and exclusion criteria, finally, 515 patients were included in this study. A diagnosis of diabetes mellitus (DM) was made by reference to one or more of the three 2015 criteria of the American Diabetes Association [15]. To identify whether the efficacy of the diagnostic criteria differed for younger (<65 years of age) and older (≥65 years of age) adults, we separated all patients into younger (n=274) and older (n=241) groups. Baseline age, sex, weight, height, blood pressure (BP), smoking status, alcohol consumption, and clinical histories (including medical treatment) were acquired by chart review or telephone inquiry. This work was supported by research fund of Chungnam National University. The protocol for this research was approved by the Institutional Review Board (2016-05-040-001) of Chungnam National University Hospital and written informed consent was provided to all of the participants.
Biochemical analyses
After an overnight fast (≥8 hours), blood samples were collected to estimate the FPG, fasting insulin, C-peptide, HbA1c, triglyceride, total cholesterol, high-density lipoprotein cholesterol (HDL-C) and low density lipoprotein cholesterol (LDL-C), blood urea nitrogen, and creatinine levels. At 2 hours after ingestion of the 75 g glucose, blood samples were collected to estimate the glucose, insulin, and C-peptide levels. All measurements were performed with the aid of an automated blood chemistry analyzer (Hitachi 747; Hitachi, Tokyo, Japan). The HbA1c levels were measured using an accredited automated analyzer (the HLC-723G7 hemoglobin analyzer; Tosoh Corp., Tokyo, Japan) employing high-performance liquid chromatography [16].
Calculations
BMI (kg/m2) was calculated as weight (kg)/[height (m)]2. The homeostasis model was used to assess insulin resistance (HOMA-IR) and β-cell function (HOMA-β) [17]. The HOMA-IR was calculated as the FPG level (mg/dL)×the insulin level (µIU/mL)/405. The HOMA-β was calculated as 360×the insulin level (µIU/mL)/[the FPG level (mg/dL)–63]. The estimated glomerular filtration rate (eGFR, mL/min/1.73 m2) was calculated using the abbreviated Modification of Diet in Renal Disease algorithm of the National Institutes of Health (NIH).
Statistical analyses
All statistical analyses were performed using SPSS version 22.0 (IBM Co., Armonk, NY, USA) and Medcalc version 16.4.3 (MedCalc, Ostend, Belgium) software. Categorical variables are presented as percentages and were compared using the chi-square test. Continuous variables are presented as mean±standard deviation (SD) and were compared with the aid of the independent two-sample Student t-tests. A two-sided P<0.05 was considered to reflect statistical significance. Receiver operating characteristic (ROC) curves were plotted for older patients diagnosed with diabetes; we evaluated HbA1c, FPG, and 2-h PG levels. In general, an area under the curve (AUC) was considered to be high (AUC >0.9), moderate (0.7< AUC ≤0.9), or low (0.5< AUC ≤0.7) in terms of diagnostic utility. Newly proposed cutoff values were based on the highest Youden indices of the various cutoff points, as described previously [18].
RESULTS
Clinical characteristics
Of the 515 patients meeting the inclusion criteria, we enrolled 74 patients exhibiting normal glucose tolerances, 132 diagnosed with prediabetes, and 309 diagnosed with DM; we grouped all patients by age as younger (<65 years) or older (≥65 years). Table 1 lists the clinical and laboratory features of patients with DM by age group. To explore whether these characteristics varied by age, we compared younger and older patients. We found no difference in the sex ratio, smoking history, systolic BP, HOMA-IR score, or HDL-C level between the groups. Older patients tended to have medical histories of cardiovascular and cerebrovascular disease, dyslipidemia, and hypertension but were less likely to have a family history of diabetes. Additionally, older patients had a significantly lower BMI and diastolic BP, reduced total and LDL-C and triglyceride levels, a lower eGFR, and reduced HbA1c, FPG, and 2-h PG levels. Interestingly, older patients showed significantly higher fasting and 2-hour insulin and C-peptide levels and better β-cell function (HOMA-β scores), than younger patients.
Table 1. Comparison of clinical and laboratory characteristics of study patients stratified by age.
| Characteristic | <65 Years (n=153) | ≥65 Years (n=156) | P value |
|---|---|---|---|
| Age, yr | 47.2±11.9 | 71.0±49.4 | <0.001 |
| Male sex | 83 (54.2) | 77 (49.4) | NS |
| Familial history of diabetes | 73 (47.7) | 18 (11.5) | <0.001 |
| Cardiovascular event | 2 (1.3) | 22 (14.1) | <0.001 |
| Cerebrovascular event | 4 (2.6) | 16 (10.3) | 0.009 |
| Smoking | 51 (33.3) | 47 (30.1) | NS |
| Dyslipidemia medication | 16 (10.5) | 49 (31.4) | <0.001 |
| Hypertension medication | 38 (24.8) | 92 (59.0) | <0.001 |
| SBP, mm Hg | 132±16 | 130±14 | NS |
| DBP, mm Hg | 80±12 | 75±8 | <0.001 |
| BMI, kg/m2 | 26.3±4.4 | 25.3±3.4 | 0.042 |
| HbA1c, % | 8.2±2.1 | 6.8±1.2 | <0.001 |
| FPG, mg/dL | 170±56 | 130±43 | <0.001 |
| 2-h PG, mg/dL | 311±102 | 257±81 | <0.001 |
| Fasting insulin, µIU/mL | 10.9±5.1 | 13.8±15.8 | 0.047 |
| 2-h insulin, µIU/mL | 44.5±42.3 | 64.9±48.8 | 0.003 |
| Fasting C-peptide, pmol/mL | 0.9±0.4 | 1.2±0.7 | 0.006 |
| 2-h C-peptide, pmol/mL | 2.5±1.4 | 3.7±1.9 | <0.001 |
| HOMA-IR | 1.7±0.9 | 2.0±1.7 | NS |
| HOMA-β | 45.7±30.6 | 76.0±54.4 | <0.001 |
| Total cholesterol, mg/dL | 202±45 | 178±40 | <0.001 |
| HDL-C, mg/dL | 48±12 | 49±12 | NS |
| LDL-C, mg/dL | 129±41 | 112±38 | 0.001 |
| Triglycerides, mg/dL | 202±155 | 159±90 | 0.004 |
| eGFR, mL/min/1.73 m2 | 113.7±28.8 | 95.6±28.3 | <0.001 |
Values are presented as mean±standard deviation or number (%). P values were calculated by independent Student t-test or the chi-square test.
NS, not significant; SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index; HbA1c, glycosylated hemoglobin; FPG, fasting plasma glucose; 2-h PG, 2-hour postload glucose; HOMA-IR, homeostasis model assessment-insulin resistance; HOMA-β, homeostasis model assessment-β-cell function; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate.
Utility of HbA1c, FPG, and 2-h PG tests for the diagnosis of DM in younger and older adults
To explore whether the efficacy of the diagnostic DM criteria varied by age, we measured the diagnostic rate according to various criteria (Table 2) and the number of criteria met by younger and older patients (Table 3). With regard to younger patients 81% were diagnosed by HbA1c and FPG and 91.5% by 2-h PG; while 61.5% of older patients were diagnosed by HbA1c, 45.5% by FPG, and 84.6% by 2-h PG (Table 2). Table 3 showed the subanalysis of these results according to the number of criteria met by younger and older patients. In the former group, 70.8% met all three diagnostic criteria, but this figure fell to 32.7% among older patients. Of the younger group, 12.4% met two of the diagnostic criteria; this proportion rose to 26.3% in the older group. A total of 17% of younger DM patients but 41% of older patients met only one DM diagnostic criterion. Because the FPG test was used to screen for DM, we explored the numbers of DM cases missed using this criterion. We found that this approach to screening was associated with a 19% under-diagnosis rate (HbA1c+2-h PG, 6.5%; HbA1c, 3.3%; 2-h PG, 9.2%) among younger patients but a 54.4% rate (HbA1c+2-h PG, 17.3%; HbA1c, 8.3%; 2-h PG, 28.8%) in older patients. The use of both the HbA1c and FPG levels to screen for DM missed 9.2% of younger and 28.8% of older patients. In other words, the prevalence of isolated post-challenge hyperglycemia (IPH) was much higher in the older than the younger group. Thus, older patients were often incorrectly diagnosed even when screened using both the FPG and HbA1c levels.
Table 2. The prevalence of diabetes using HbA1c, FPG, and 2-h PG criteria according to age group.
| Diagnostic criterion | <65 Years (n=153) | ≥65 Years (n=156) | P value |
|---|---|---|---|
| HbA1c | 124 (81) | 96 (61.5) | <0.001 |
| FPG | 124 (81) | 71 (45.5) | <0.001 |
| 2-h PG | 140 (91.5) | 132 (84.6 ) | NS |
Values are presented as number (%). P values were calculated by the chi-square test.
HbA1c, glycosylated hemoglobin; FPG, fasting plasma glucose; 2-h PG, 2-hour postload glucose; NS, not significant.
Table 3. Percentage of patients stratified by number of diagnostic criteria satisfied according to age group.
| No. of satisfied DM diagnostic criteria | <65 Years (n=153) | ≥65 Years (n=156) | P value |
|---|---|---|---|
| 3 | 108 (70.6) | 51 (32.7) | <0.001 |
| 2 | 19 (12.4) | 41 (26.3) | 0.002 |
| HbA1c+FPG | 1 (0.7) | 5 (3.2) | NS |
| HbA1c+2-h PG | 10 (6.5) | 27 (17.3) | 0.005 |
| FPG+2-h PG | 8 (5.2) | 9 (5.8) | NS |
| 1 | 26 (17.0) | 64 (41.0) | <0.001 |
| HbA1c | 5 (3.3) | 13 (8.3) | NS |
| FPG | 7 (4.6) | 6 (3.8) | NS |
| 2-h PG | 14 (9.2) | 45 (28.8) | <0.001 |
Values are presented as number (%). P values were calculated by the chi-square test.
DM, diabetes mellitus; HbA1c, glycosylated hemoglobin; FPG, fasting plasma glucose; NS, not significant; 2-h PG, 2-hour postload glucose.
Problems associated with the use of the FPG test to screen for DM in older adults
Because the FPG test is commonly employed to screen for DM in Korea, we determined the false-negative rate associated with such an approach (Table 4). Diagnosis of DM was missed in 24 of 106 younger patients (22.6%) with normal (<100 mg/dL) or impaired fasting glucose levels (≥100 and <126 mg/dL) and 72 of 114 older patients (63.2%) with normal or impaired fasting glucose levels. According to the subgroup analysis, DM in patients with normal FPG levels was missed in 1.89% of younger and 9.65% of older patients, and DM in patients with impaired fasting glucose levels was missed in 20.75% of younger and 53.51% of older patients. In other words, we may misdiagnose almost two in 10 younger patients and six in 10 older patients with normal or impaired fasting glucose level. These results suggested that recent FPG levels for DM diagnosis had very low diagnostic power, especially in older patients.
Table 4. Percentage of patients who met 2-h PG criteria among patients with undiagnosed diabetes using FPG levels.
| FPG, mg/dL | 2-h PG, mg/dL | |||
|---|---|---|---|---|
| <65 Years (n=106) | ≥65 Years (n=114) | |||
| <140 | ≥200 | <140 | ≥200 | |
| <100 | 58 (54.72) | 2 (1.89) | 19 (16.67) | 11 (9.65) |
| 100–125 | 24 (22.64) | 22 (20.75) | 23 (20.17) | 61 (53.51) |
| Subtotal | 82 (77.36) | 24 (22.64) | 42 (36.84) | 72 (63.16) |
Values are presented as number (%).
2-h PG, 2-hour postload glucose; FPG, fasting plasma glucose.
Ideal diagnostic methods for DM diagnosis in older adults
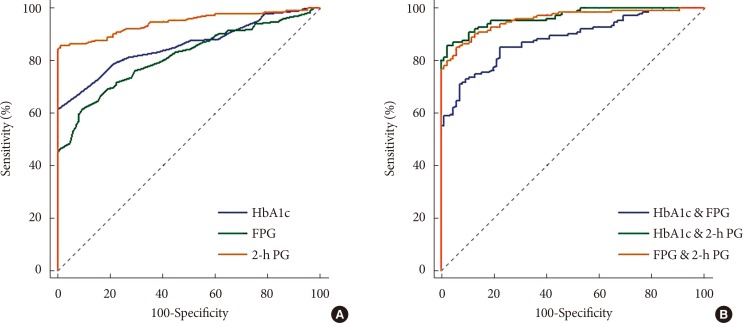
We sought to define ideal diagnostic methods which would reduce false-negative DM diagnoses; we recalculated the sensitivities, specificities, and the AUCs of the DM diagnostic criteria using different thresholds for older patients (Table 5, Fig. 1). The ROC curves revealed the diagnostic efficacy of the HbA1c, FPG, and 2-h PG levels used alone for DM diagnosis; we also determined those of HbA1c+FPG, HbA1c+2-h PG, and FPG+2-h PG when two criteria were employed. The AUCs were 0.859 (95% confidence interval [CI], 0.809 to 0.901) for the HbA1c level; 0.817 (95% CI, 0.762 to 0.869) for the FPG level; 0.946 (95% CI, 0.910 to 0.971) for the 2-h PG level; 0.884 (95% CI, 0.836 to 0.921) for HbA1c+FPG; 0.966 (95% CI, 0.935 to 0.985) for HbA1c+2-h PG; and 0.959 (95% CI, 0.925 to 0.980) for FPG+2-h PG. Thus, when only one criterion was used, the best result was from the 2-h PG AUC, and the HbA1c+2-h PG AUC was best when two criteria were employed. Reducing the FPG cutoff from 126 to 118 mg/dL increased the DM diagnostic sensitivity from 45.51% to 61.54% and reduced the specificity from 100% to 90.59%.
Table 5. Sensitivity and specificity of diagnostic criteria for diabetes mellitus in the older group.
| Variable | Sensitivity, % | Specificity, % |
|---|---|---|
| HbA1c >6.4 % | 61.54 | 100 |
| FPG, mg/dL | ||
| >117 | 61.54 | 90.59 |
| >125 | 45.51 | 100 |
| 2-h PG >198 mg/dL | 84.62 | 100 |
| HbA1c & FPG | 71.15 | 92.94 |
| HbA1c & 2-h PG | 85.90 | 97.65 |
| FPG & 2-h PG | 85.26 | 94.12 |
HbA1c, glycosylated hemoglobin; FPG, fasting plasma glucose; 2-h PG, 2-hour postload glucose.
Fig. 1. Receiver operating characteristic curves. (A) Single diagnostic criterion and (B) combination of two diagnostic criteria for screening of diabetes mellitus in the older group. (A) When the single criterion was used, area under the curve (AUC) of glycosylated hemoglobin (HbA1c) was 0.859 (95% confidence interval [CI], 0.809 to 0.901), the AUC of fasting plasma glucose (FPG) was 0.817 (95% CI, 0.762 to 0.863), and the AUC of 2-hour plasma glucose (2-h PG) after the 75 g oral glucose tolerance test, which was the largest, was 0.946 (95% CI, 0.910 to 0.971). (B) When the combination of two criteria was used, the AUC of HbA1c and FPG was 0.884 (95% CI, 0.836 to 0.921), the AUC of HbA1c and 2-h PG, which was the largest, was 0.966 (95% CI, 0.935 to 0.985), and the AUC of FPG and 2-h PG was 0.959 (95% CI, 0.925 to 0.980).
DISCUSSION
The prevalence of DM using various diagnostic criteria varied more in adults aged ≥65 years (the older group) than in adults aged <65 years (the younger group). Of the 156 older patients, DM was diagnosed in 132 (84.6%) using the 2-h PG criterion but in only 71 (45.5%) and 96 (61.5%) using the FPG and HbA1c criteria, respectively. These results are important because 64 older patients (41%) met only one criterion, usually the 2-h PG level (28.8%). Because most of the older patients were diagnosed using this criterion, the 2-h PG level appeared to be more sensitive (84.6%) than the FPG (45.5%) or HbA1c (61.54%) level. The 2-h PG level exhibited a sensitivity similar to that of combinations of the 2-h PG and HbA1c (85.9%) or FPG (85.3%) levels and a higher sensitivity than the combination of the FPG and HbA1c levels (71.2%). The low diagnostic power of FPG could be enhanced by reducing the FPG cutoff values from 126 to 118 mg/dL which provided the best combination of sensitivity and specificity.
One study found that patients who met only the 2-h PG criterion were significantly older and had lower BMIs than those who met only the FPG criterion [9]. Another study found that use of the FPG criterion in Asian populations missed approximately 45% of all diabetes patients, and those with IPH, which was defined as a 2-h PG level ≥200 mg/dL after OGTT but a fasting glucose level <126 mg/dL [19]. Such patients tended to be older than those diagnosed by the FPG criterion (60 years vs. 53 years). However, as all included patients were relatively young, no subgroup analysis by age was performed. Although the prevalence of diabetes increases with age, very few authors have researched elderly-onset diabetes. Aging is associated with a progressive decline in glucose tolerance caused by many factors, including reduced physical activity, abdominal obesity, sarcopenia, and dysfunctional insulin secretion; these factors increase the prevalence of T2DM and impair glucose tolerance in older adults [13,20,21,22]. Some studies have found that dysfunctional insulin secretion may be an important contributor to the IPH, which is common in those aged >60 years [23,24,25]. However, we found that although IPH was more common in older patients than the younger group (28.8% vs. 9.2%), the HOMA-IR (the insulin resistance), did not differ between the two age groups and the HOMA-β (the islet β-cell function), was much more efficient in older than younger patients. In our study, all the HbA1c, FPG, and 2-h PG levels were significantly lower in the older group than in the younger group. These results indicate that the older group may have less glucose intoxication than the younger group, which preserves their islet β-cell function.
We reduced the FPG cutoff value from 140 to 126 mg/dL in an effort to resolve the discordance between the 2-h PG and FPG criteria [26]. However, the diagnostic power of the FPG level did not increase significantly because of the high incidence of IPH in all diabetes patients [19]. Recent studies found that a high 1-hour PG in people with normal glucose tolerance predicted islet β-cell dysfunction and future DM [27,28]. These results suggested that the postprandial glucose level may change more rapidly than the FPG level. Therefore, some researchers recommend that patients with impaired fasting glucose levels and a high FPG level should proceed with the OGTT to detect more patients with DM [29]. We found that the FPG test produced more false-negative diagnoses of diabetes in older than in younger diabetic patients (54.5% vs. 19%, respectively). As the prevalence of diabetes peaks at >60 years of age, missed diagnoses will be much more common among older patients than among patients of all ages. This suggests that the 126 mg/dL FPG level is less useful when screening older than younger adults.
HbA1c, generated by a non-enzymatic reaction between glucose and hemoglobin, was discovered in 1968 [30]. Elevated HbA1c levels in diabetic patients were first reported in 1969 [31] and were shown to indicate aggravated glucose levels over a 3-month period [32]. Since the HbA1c level was included as a diagnostic criterion by the American Diabetes Association in 2010, many researchers have explored its diagnostic efficacy and have examined the optimal cutoff values [10,33,34,35,36]. These studies suggested that the HbA1c level was of low sensitivity when used to screen for diabetes, and the authors concluded that the cutoff values had to be reduced to increase sensitivity. Additionally, the optimal cutoff values varied slightly with the characteristics of particular study populations. In a Korean population, the HbA1c level was higher in older than in younger patients with similar glucose profiles, and it was more diagnostically accurate than the FPG level in older patients [10,37]. We obtained similar results, as the HbA1c level was of greater diagnostic efficacy than the FPG level in older patients (61.5% vs. 45.5%, respectively), although these patients had lower HbA1c levels than younger patients because of reduced glucose profiles.
Although it is known that the 2-h PG test, followed by the 75 g OGTT, afford the best sensitivity and specificity of all current diabetic criteria, the FPG test is most commonly used for screening because it is very convenient. Measurement of HbA1c levels in the absence of fasting is easier than employing the FPG method. Therefore, a combination of two criteria, the FPG and the HbA1c levels, would lower the false-negative rate of diabetic diagnosis. One study sought to verify this hypothesis, but found that a combination of FPG and HbA1c levels did not seem to afford any additional benefit when screening for diabetes [34]. On the other hand, another study found that a combination of FPG and HbA1c levels did increase the diagnostic power for diabetes [38]. We found that a combination of the FPG and HbA1c levels increased the accuracy of diabetic diagnosis. In younger patients, the prevalence of diabetes rose from 81% when the FPG or HbA1c level was used alone for diagnosis, to 90.8% when the two criteria were combined. In older patients, the prevalence of diabetes rose from 45.5% and 61.5%, respectively, when the FPG and HbA1c criteria were used alone, to 71.2% when the two criteria were combined. The diagnostic rate afforded by a combination of the FPG and HbA1c levels attained a level similar to that afforded by application of the 2-h PG criterion (90.8% vs. 91.5%) in younger patients, but did not achieve the diagnostic rate produced by the 2-h PG criterion in older patients (71.2% vs. 84.6%).
In summary, older patients had a higher prevalence of IPH, but nevertheless, exhibited preserved insulin secretion and did not progress to insulin resistance. Older patients were diagnosed least often using the FPG criterion and most often employing the 2-h PG criterion. To enhance DM diagnosis in elderly individuals, the FPG and 2-h PG test may be recommended. As cost and convenience must always be considered, a combination of the FPG and HbA1c criteria, or a reduction in the FPG criterion, could also be used to screen for DM in older patients. The limitations of our study include our small study population and selection bias attributable to the fact that most of patients with suspected hyperglycemia were recruited in a tertiary teaching hospital located in an urban region. Therefore, large longitudinal studies are needed to establish age-specific diagnostic criteria to enable individualized diagnoses, especially in adults aged ≥65 years, who have the highest prevalence of diabetes.
Footnotes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
References
- 1.The Centers for Disease Control and Prevention (CDC) National diabetes statistics report 2017. [updated 2017 Jul 17]. Available from: https://www.cdc.gov/diabetes/data/statistics/statistics-report.html.
- 2.Harris MI, Klein R, Welborn TA, Knuiman MW. Onset of NIDDM occurs at least 4–7 yr before clinical diagnosis. Diabetes Care. 1992;15:815–819. doi: 10.2337/diacare.15.7.815. [DOI] [PubMed] [Google Scholar]
- 3.Harris MI. Undiagnosed NIDDM: clinical and public health issues. Diabetes Care. 1993;16:642–652. doi: 10.2337/diacare.16.4.642. [DOI] [PubMed] [Google Scholar]
- 4.Wang FH, Liang YB, Zhang F, Wang JJ, Wei WB, Tao QS, Sun LP, Friedman DS, Wang NL, Wong TY. Prevalence of diabetic retinopathy in rural China: the Handan Eye Study. Ophthalmology. 2009;116:461–467. doi: 10.1016/j.ophtha.2008.10.003. [DOI] [PubMed] [Google Scholar]
- 5.Barrett-Connor E, Ferrara A. Isolated postchallenge hyperglycemia and the risk of fatal cardiovascular disease in older women and men. The Rancho Bernardo Study. Diabetes Care. 1998;21:1236–1239. doi: 10.2337/diacare.21.8.1236. [DOI] [PubMed] [Google Scholar]
- 6.Gillies CL, Lambert PC, Abrams KR, Sutton AJ, Cooper NJ, Hsu RT, Davies MJ, Khunti K. Different strategies for screening and prevention of type 2 diabetes in adults: cost effectiveness analysis. BMJ. 2008;336:1180–1185. doi: 10.1136/bmj.39545.585289.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.CDC Diabetes Cost-Effectiveness Study Group; Centers for Disease Control and Prevention. The cost-effectiveness of screening for type 2 diabetes. JAMA. 1998;280:1757–1763. [PubMed] [Google Scholar]
- 8.International Expert Committee. International Expert Committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care. 2009;32:1327–1334. doi: 10.2337/dc09-9033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang J, Ou HY, Karnchanasorn R, Samoa R, Chuang LM, Chiu KC, Feng W. Clinical implication of fasting and post-challenged plasma glucose in diagnosis of diabetes mellitus. Endocrine. 2015;48:511–518. doi: 10.1007/s12020-014-0301-3. [DOI] [PubMed] [Google Scholar]
- 10.Lee H, Oh JY, Sung YA, Kim DJ, Kim SH, Kim SG, Moon S, Park IeB, Rhee EJ, Chung CH, Kim BJ, Ku BJ. Optimal hemoglobin A1C cutoff value for diagnosing type 2 diabetes mellitus in Korean adults. Diabetes Res Clin Pract. 2013;99:231–236. doi: 10.1016/j.diabres.2012.09.030. [DOI] [PubMed] [Google Scholar]
- 11.Lu ZX, Walker KZ, O'Dea K, Sikaris KA, Shaw JE. A1C for screening and diagnosis of type 2 diabetes in routine clinical practice. Diabetes Care. 2010;33:817–819. doi: 10.2337/dc09-1763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kumar PR, Bhansali A, Ravikiran M, Bhansali S, Dutta P, Thakur JS, Sachdeva N, Bhadada SK, Walia R. Utility of glycated hemoglobin in diagnosing type 2 diabetes mellitus: a community-based study. J Clin Endocrinol Metab. 2010;95:2832–2835. doi: 10.1210/jc.2009-2433. [DOI] [PubMed] [Google Scholar]
- 13.Kirkman MS, Briscoe VJ, Clark N, Florez H, Haas LB, Halter JB, Huang ES, Korytkowski MT, Munshi MN, Odegard PS, Pratley RE, Swift CS. Diabetes in older adults. Diabetes Care. 2012;35:2650–2664. doi: 10.2337/dc12-1801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Korea Centers for Disease Control and Prevention (KCDC) and the Korean Ministry of Health and Welfare. Diabetes Fact Sheet in Korea 2016. [cited 2017 Dec 18]. Available from: http://www.diabetes.or.kr/temp/KDA_fact_sheet%202016.pdf.
- 15.American Diabetes Association. (2) Classification and diagnosis of diabetes. Diabetes Care. 2015;38:S8–16. doi: 10.2337/dc15-S005. [DOI] [PubMed] [Google Scholar]
- 16.International Federation of Clinical Chemistry and Laboratory Medicine; IFCC Scientific Division. Mosca A, Goodall I, Hoshino T, Jeppsson JO, John WG, Little RR, Miedema K, Myers GL, Reinauer H, Sacks DB, Weykamp CW. Global standardization of glycated hemoglobin measurement: the position of the IFCC Working Group. Clin Chem Lab Med. 2007;45:1077–1080. doi: 10.1515/CCLM.2007.246. [DOI] [PubMed] [Google Scholar]
- 17.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 18.Hajian-Tilaki K. Receiver operating characteristic (ROC) curve analysis for medical diagnostic test evaluation. Caspian J Intern Med. 2013;4:627–635. [PMC free article] [PubMed] [Google Scholar]
- 19.Qiao Q, Nakagami T, Tuomilehto J, Borch-Johnsen K, Balkau B, Iwamoto Y, Tajima N International Diabetes Epidemiology Group; DECODA Study Group. Comparison of the fasting and the 2-h glucose criteria for diabetes in different Asian cohorts. Diabetologia. 2000;43:1470–1475. doi: 10.1007/s001250051557. [DOI] [PubMed] [Google Scholar]
- 20.Chang AM, Halter JB. Aging and insulin secretion. Am J Physiol Endocrinol Metab. 2003;284:E7–E12. doi: 10.1152/ajpendo.00366.2002. [DOI] [PubMed] [Google Scholar]
- 21.Barzilai N, Huffman DM, Muzumdar RH, Bartke A. The critical role of metabolic pathways in aging. Diabetes. 2012;61:1315–1322. doi: 10.2337/db11-1300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ohn JH, Kwak SH, Cho YM, Lim S, Jang HC, Park KS, Cho NH. 10-Year trajectory of β-cell function and insulin sensitivity in the development of type 2 diabetes: a community-based prospective cohort study. Lancet Diabetes Endocrinol. 2016;4:27–34. doi: 10.1016/S2213-8587(15)00336-8. [DOI] [PubMed] [Google Scholar]
- 23.Chen M, Halter JB, Porte D., Jr The role of dietary carbohydrate in the decreased glucose tolerance of the elderly. J Am Geriatr Soc. 1987;35:417–424. doi: 10.1111/j.1532-5415.1987.tb04663.x. [DOI] [PubMed] [Google Scholar]
- 24.Garcia GV, Freeman RV, Supiano MA, Smith MJ, Galecki AT, Halter JB. Glucose metabolism in older adults: a study including subjects more than 80 years of age. J Am Geriatr Soc. 1997;45:813–817. doi: 10.1111/j.1532-5415.1997.tb01507.x. [DOI] [PubMed] [Google Scholar]
- 25.Chen M, Bergman RN, Pacini G, Porte D., Jr Pathogenesis of age-related glucose intolerance in man: insulin resistance and decreased beta-cell function. J Clin Endocrinol Metab. 1985;60:13–20. doi: 10.1210/jcem-60-1-13. [DOI] [PubMed] [Google Scholar]
- 26.Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 1997;20:1183–1197. doi: 10.2337/diacare.20.7.1183. [DOI] [PubMed] [Google Scholar]
- 27.Oh TJ, Lim S, Kim KM, Moon JH, Choi SH, Cho YM, Park KS, Jang H, Cho NH. One-hour postload plasma glucose concentration in people with normal glucose homeostasis predicts future diabetes mellitus: a 12-year community-based cohort study. Clin Endocrinol (Oxf) 2017;86:513–519. doi: 10.1111/cen.13280. [DOI] [PubMed] [Google Scholar]
- 28.Oh TJ, Min SH, Ahn CH, Kim EK, Kwak SH, Jung HS, Park KS, Cho YM. Normal glucose tolerance with a high 1-hour postload plasma glucose level exhibits decreased β-cell function similar to impaired glucose tolerance. Diabetes Metab J. 2015;39:147–153. doi: 10.4093/dmj.2015.39.2.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Oh JY, Lim S, Kim DJ, Kim NH, Kim DJ, Moon SD, Jang HC, Cho YM, Song KH, Ahn CW, Sung YA, Park JY, Shin C, Lee HK, Park KS Committee of the Korean Diabetes Association on the Diagnosis and Classification of Diabetes Mellitus. A report on the diagnosis of intermediate hyperglycemia in Korea: a pooled analysis of four community-based cohort studies. Diabetes Res Clin Pract. 2008;80:463–468. doi: 10.1016/j.diabres.2008.01.022. [DOI] [PubMed] [Google Scholar]
- 30.Bookchin RM, Gallop PM. Structure of hemoglobin AIc: nature of the N-terminal beta chain blocking group. Biochem Biophys Res Commun. 1968;32:86–93. doi: 10.1016/0006-291x(68)90430-0. [DOI] [PubMed] [Google Scholar]
- 31.Rahbar S, Blumenfeld O, Ranney HM. Studies of an unusual hemoglobin in patients with diabetes mellitus. Biochem Biophys Res Commun. 1969;36:838–843. doi: 10.1016/0006-291x(69)90685-8. [DOI] [PubMed] [Google Scholar]
- 32.Koenig RJ, Peterson CM, Jones RL, Saudek C, Lehrman M, Cerami A. Correlation of glucose regulation and hemoglobin AIc in diabetes mellitus. N Engl J Med. 1976;295:417–420. doi: 10.1056/NEJM197608192950804. [DOI] [PubMed] [Google Scholar]
- 33.Kramer CK, Araneta MR, Barrett-Connor E. A1C and diabetes diagnosis: The Rancho Bernardo Study. Diabetes Care. 2010;33:101–103. doi: 10.2337/dc09-1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mannucci E, Ognibene A, Sposato I, Brogi M, Gallori G, Bardini G, Cremasco F, Messeri G, Rotella CM. Fasting plasma glucose and glycated haemoglobin in the screening of diabetes and impaired glucose tolerance. Acta Diabetol. 2003;40:181–186. doi: 10.1007/s00592-003-0109-8. [DOI] [PubMed] [Google Scholar]
- 35.Karnchanasorn R, Huang J, Ou HY, Feng W, Chuang LM, Chiu KC, Samoa R. Comparison of the current diagnostic criterion of hba1c with fasting and 2-hour plasma glucose concentration. J Diabetes Res. 2016;2016:6195494. doi: 10.1155/2016/6195494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu Y, Xiao X, Sun C, Tian S, Sun Z, Gao Y, Li Y, Cheng J, Lv Y, Li M, Li Z, Zhang Y, Wang G, Liu Y, Gao Y, Zhu L, Liu Y, Wang G. Ideal glycated hemoglobin cut-off points for screening diabetes and prediabetes in a Chinese population. J Diabetes Investig. 2016;7:695–702. doi: 10.1111/jdi.12498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kim JH, Shin JH, Lee HJ, Kim SY, Bae HY. Discordance between HbA1c and fasting plasma glucose criteria for diabetes screening is associated with obesity and old age in Korean individuals. Diabetes Res Clin Pract. 2011;94:e27–e29. doi: 10.1016/j.diabres.2011.07.013. [DOI] [PubMed] [Google Scholar]
- 38.Ahn CH, Yoon JW, Hahn S, Moon MK, Park KS, Cho YM. Evaluation of non-laboratory and laboratory prediction models for current and future diabetes mellitus: a cross-sectional and retrospective cohort study. PLoS One. 2016;11:e0156155. doi: 10.1371/journal.pone.0156155. [DOI] [PMC free article] [PubMed] [Google Scholar]