Abstract
This study investigated the relationship between atrial fibrillation (AF) and left ventricular (LV) geometric patterns in a hospital-based population in Japan. We retrospectively analyzed 4444 patients who had undergone simultaneous scheduled transthoracic echocardiography (TTE) and electrocardiography during 2013. A total of 430 patients who had findings of previous myocardial infarctions (n = 419) and without the data on body surface area (n = 11) were excluded from the study. We calculated the LV mass index (LVMI) and relative wall (RWT) and categorized 4014 patients into four groups as follows: normal geometry (n = 3046); concentric remodeling (normal LVMI and high RWT, n = 437); concentric hypertrophy (high LVMI and high RWT, n = 149); and eccentric remodeling (high LVMI and normal RWT, n = 382). The mean left atrial volume indices (LAVI) were 22.5, 23.8, 33.3, and 37.0 mm/m2 in patients with normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy, respectively. The mean LV ejection fractions (LVEF) were 62.7, 62.6, 60.8, and 53.8%, respectively, whereas the prevalence of AF was 10.4%, 10.5%, 14.8%, and 16.8% in patients with normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy, respectively. In conclusion, the prevalence of AF was increasing according to LV geometric remodeling patterns in association with LA size and LVEF.
Introduction
Left ventricular (LV) hypertrophy (LVH) may be considered a compensatory effect since increasing LV wall thickness reduces LV wall stress1. However, as LVH progresses, it is associated with considerable cardiovascular (CV) morbidity and mortality2,3. Recent studies have focused on the prognostic impact of more subtle LV geometric abnormalities. Patterns of LVH and geometric remodeling have previously been investigated extensively in patients with hypertension and valvular heart diseases4,5. Concentric LVH has a high mortality risk with preserved ejection fraction (EF)6,7 or a high mortality risk in patients without regression of abnormal LV geometry8; other studies have reported that relative wall thickness has less impact on prognosis in patients with coronary heart disease3.
LVH and LV dilatation cause an in increase in end-diastolic pressure, followed by enlargement of the left atrium9. Atrial fibrillation (AF) is more prevalent among older people with pronounced morbidity and mortality10–13. AF can cause heart failure, affect quality of life, lengthen the hospitalization period, and increase mortality14. Left atrial (LA) remodeling, LA enlargement, and LV remodeling are related to AF development15. However, the association between LV geometric patterns, LA enlargement, and AF remains unknown. Therefore, in this study, we investigated the association between LV geometric patterns, LA enlargement, and AF.
Methods
Study population
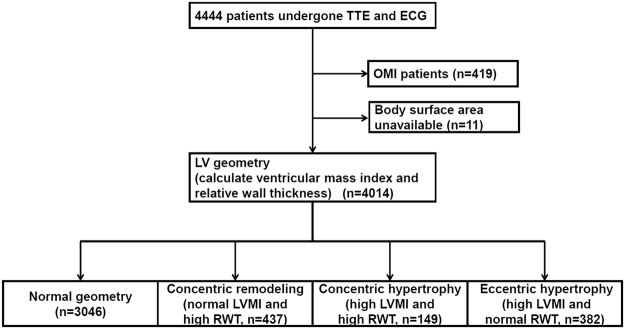
We retrospectively analyzed 4444 patients who had undergone simultaneous scheduled transthoracic echocardiography (TTE) and electrocardiography (ECG) at the Cardiovascular Center of Kitano Hospital during 2013. A flowchart of the study population is shown in Fig. 1. A total of 430 patients who had findings of previous myocardial infarctions (n = 419) and without the data on body surface area (n = 11) were excluded from the study, because the old myocardial infarction affected their wall thickness and other data from the TTE. On the basis of the data from the TTE and ECG examinations in addition to the catheter database, we identified patients who had a previous myocardial infarction. The ECG and TTE were ordered by each physician.
Figure 1.
Flowchart of the study population. Abbreviations: TTE = transthoracic echocardiography; ECG = electrocardiography; LV = left ventricular; LVMI = LV mass index; RWT = relative wall thickness; and OMI = old myocardial infarction.
The research protocol was approved by the institutional review board of Kitano Hospital (approval no.: P16-02-005). Informed consent was not obtained from each patient since this was a retrospective study. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee. Patients’ records and information were anonymized and de-identified before analysis.
Data collection
From the TTE database, we extracted data for patients’ wall thickness, LV diastolic diameter (LVDd), E/e’, LA diameter (LAD), LA volume index (LAVI), and LV ejection fraction (LVEF). We also extracted the body surface area from the TTE report. From the ECG database, we extracted cardiac rhythm and recorded it as it was documented. Therefore, we could not determine whether the AF was paroxysmal or persistent.
The LV mass was calculated using the formula recommended by the American Society of Echocardiography (ASE), and it was indexed to the body surface area as follows: LV mass = 0.8 × 1.04 [(LVDd + LVPWTd + IVSTd)3 − (LVDd)3] + 0.6, where LVDd was the LV diastolic diameter, IVSTd was the diastolic interventricular septal wall thickness, and LVPWTd was the diastolic LV posterior wall thickness. In line with the ASE recommendations, a high LV mass index (LVMI) was defined as >115 g/m2 for male patients and >95 g/m2 for female patients. Relative wall thickness (RWT) was calculated using the following formula: (2 × LVPWTd)/(LVDd), which permits physicians to categorize an increase in the LV mass as either concentric (RWT >0.42) or eccentric (RWT ≤0.42) hypertrophy and identify concentric remodeling (a normal LV mass with an increased RWT)16,17. The LAVI was calculated using the biplane area-length method and body surface area and defined high as a value >42 mL/m214,15. Data from two-dimensional TTE were analyzed at baseline. The LVEF was measured using the Teichholz method or the modified Simpson rule method and defined low as LVEF <50%.
We then categorized 4014 patients into four groups as follows (Fig. 1): normal geometry (n = 3046), concentric remodeling (normal LVMI and high RWT, n = 437), concentric hypertrophy (high LVMI and high RWT, n = 149), and eccentric hypertrophy (high LVMI and normal RWT, n = 382).
We extracted patients’ data from the electronic medical data at our institution, including age, sex, and type of disease, i.e., hypertension (International Statistical Classification of Diseases and Related Health Problems, Tenth Edition [ICD-10] codes I10, I11, I12, I13, I14, and I15), hyperlipidemia (ICD-10 code E78), diabetes mellitus (ICD-10 codes E10, E11, E12, E13, and E14), and chronic kidney disease (CKD) (ICD-10 code N18), from electronic medical data at our institution.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Statistical analysis
Categorical variables are presented as numbers and percentages, and were compared using the chi-square test or Fisher exact test. Continuous variables are expressed as a mean ± standard deviation or median (interquartile range). Based on their distributions, continuous variables were compared using the Student t-test or Wilcoxon rank-sum test. To analyze factors associated with AF, we used a multivariable logistic regression model (model 1) involving the following potential independent, clinically relevant variables: age >70 years; sex; LV geometric remodelings that was defined as ordered variables (normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy); and the presence of comorbidities such as ishemic heart disease, hypertension, diabetes mellitus, hyperlipidemia, CKD and overweight (body mass index >25 kg/m2). The adjusted odds ratios and 95% confidence intervals were calculated. To show the association of two variables (LAD and LVMI, LAD and RWT, LVEF and LVMI, and LVEF and RWT), we presented scatter plots, lines of best fit, and correlation coefficients. To visualize the effect size of each variable, the LogWorth (−log10(p-value)) scales were presented18. In LogWorth sclaes, a highly significant p-value had a large LogWorth value and a nonsignificant p-value had a low LogWorth value18. We generated the second multivariable regression model (model 2) including the LA size and LVEF to visualize the effect size of the LA size and LVEF as the LogWorth scales. In addition, we generated a multivariable logistic regression model using the same variables in AF for adjustment to analyze factors associated with a high LAVI and a low LVEF. Finally, from a logistic regression model with nominal responses (AF or not) using LVEF and LAVI, we generated a formula for linear combinations of the response levels (x = a + b x LVEF + c x LAVI) and prediction formulas for the response levels predicting the presence of AF (1/ (1 + Exp(x)) and the absence of AF (1/(1 + EXP(-x)), then we compared the two levels and predicted the presence or absence of AF according to the larger response level. We showed a receiver-operating curve with an area under the curve and provided the sensitivity and 1-specificity of this prediction formulas.
All statistical analyses were performed using JMP, version 13.2 (SAS Institute Inc., Chicago, IL, USA).
Results
Patients’ characteristics according to the LV geometric patterns
Baseline characteristics of patients are provided in Table 1. Among patients’ characteristics, patients with a normal geometry were significantly younger than who had abnormal geometry. The ratio of men were 52.4%, 58.3%, 48.3%, and 45.0% in patients with a normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy, respectively. Patients with concentric remodeling, concentric hypertrophy, and eccentric hypertrophy had high rates of hypertension, hyperlipidemia, diabetes mellitus, aortic valve stenosis, and CKD (Table 1).
Table 1.
Baseline characteristics of the study subjects.
| Total (n = 4,014) |
Normal geometry (n = 3,046) |
Concentric remodeling (n = 437) |
Concentric hypertrophy (n = 149) |
Eccentric hypertrophy (n = 382) |
p | |
|---|---|---|---|---|---|---|
| Age, yr, SD | 66.3, 15.9 | 64.5, 16.3 | 72.8, 12.1 | 71.9, 12.6 | 70.3, 13.6 | <0.0001 |
| Male, % | 52.2 | 52.4 | 58.3 | 48.3 | 45.0 | 0.0014 |
| Diabetes, % | 29.9 | 27.3 | 40.3 | 45.6 | 33.0 | <0.0001 |
| Hypertension, % | 57.0 | 50.6 | 74.4 | 89.3 | 76.7 | <0.0001 |
| Hyperlipidemia, % | 29.0 | 26.6 | 36.6 | 42.3 | 35.1 | <0.0001 |
| Aortic stenosis, % | 3.2 | 1.3 | 11.0 | 11.4 | 5.8 | <0.0001 |
| Aortic regurgitation, % | 3.1 | 2.0 | 1.1 | 9.4 | 11.5 | <0.0001 |
| Mitral stenosis, % | 0.2 | 0.2 | 0.2 | 0.7 | 0.5 | 0.340 |
| Mitral regurgitation, % | 3.6 | 2.5 | 1.8 | 4.7 | 13.0 | <0.0001 |
| Chronic kidney disease, % | 14 | 10.5 | 20.1 | 38.9 | 25.9 | <0.0001 |
| Overweight, % | 27.7 | 26.9 | 30.1 | 33.6 | 29.6 | 0.1419 |
Abbreviation: SD = standard deviation.
Baseline characteristics of echocardiography
Baseline characteristics of echocardiographic findings are provided in Table 2. The mean LVEFs were 62.7, 62.6, 60.8, and 53.8% in patients with a normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy, respectively. The mean LAVIs were 22.5, 23.8, 33.3, and 37.0 mm/m2 in patients with a normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy, respectively.
Table 2.
Baseline transthoracic echocardiography results.
| Total (n = 4,014) |
Normal geometry (n = 3,046) |
Concentric remodeling (n = 437) |
Concentric hypertrophy (n = 149) |
Eccentric hypertrophy (n = 382) |
p | |
|---|---|---|---|---|---|---|
| LVDd, mm | 46.7, 6.1 | 46.4, 4.8 | 40.7, 4.1 | 46.2, 4.9 | 55.6, 7.6 | <0.0001 |
| E/e’ | 11.6, 4.8 | 10.8, 4.1 | 13.3, 5.4 | 17.3, 7.0 | 14.3, 5.7 | <0.0001 |
| LAD, mm | 35.6, 7.0 | 34.7, 6.5 | 35.4, 6.8 | 40.0, 7.3 | 41.1, 7.7 | <0.0001 |
| LAVI, ml/m2 | 24.4, 14.8 | 22.5, 12.9 | 23.8, 12.5 | 33.3, 17.2 | 37.0, 21.6 | <0.0001 |
| LVEF, % | 61.7, 7.6 | 62.7, 6.0 | 62.6, 4.9 | 60.8, 7.5 | 53.8, 14.5 | <0.0001 |
Abbreviations: LVDd = left ventricular diastolic dimension; LAD = left atrial diameter; LAVI = left atrial volume index; LVEF = LV ejection fraction.
Prevalence of AF
Overall, 3460 patients had a normal sinus rhythm, 455 had AF, 95 had a pacemaker rhythm, and 15 had other rhythms. The prevalence rates of AF were 10.4, 10.5, 14.8, and 16.8% in patients with a normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy, respectively (p < 0.0001, Fig. 2).
Figure 2.
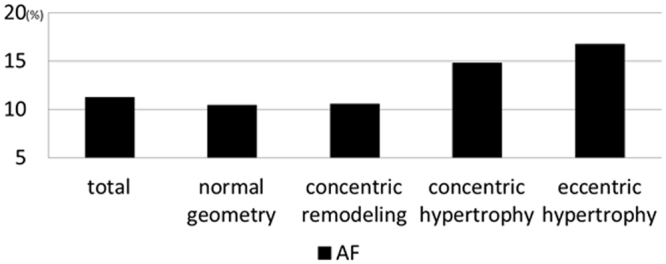
Prevalence of atrial fibrillation (AF) with each geometry patients.
Association among LV geometry, LA size, and LVEF
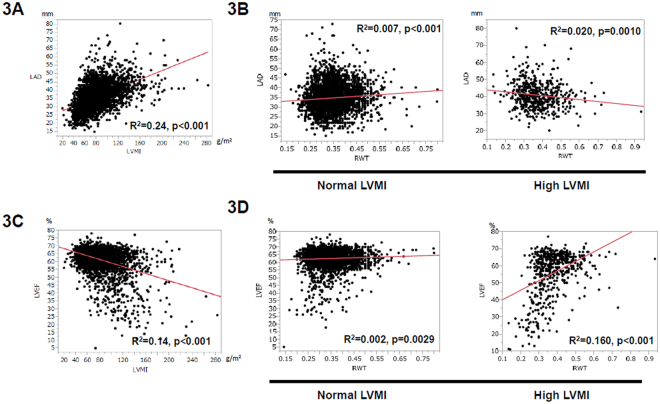
In order to investigate the underlying link between AF and LV geometry, we evaluated the relationship between LVMI, RWT, LVD, and LVEF. By using scatter plots, we visualized the association (Fig. 3). LAD was significantly related to the high LVMI (Fig. 3A). In contrast, LAD was small but positively related to high RWT when LVMI was normal (Fig. 3B), and negatively related to high RWT when LVMI was high (Fig. 3B). LVEF was significantly negatively correlated with LVMI (Fig. 3C). With regards to RWT, LVEF was only slightly correlated with RWT when LVMI was normal (Fig. 3D), but positively correlated with RWT when LVMI was high (Fig. 3D), showing that the relationship between LVEF and RWT was different for normal and high LVMI.
Figure 3.
Association with LVMI, RWT, LA diameter, and LVEF. (A) Association between LVMI and LA diameter (LAD). Scatter plots show the positive correlation between LVMI and LAD. R2 = 0.24, p < 0.001. R = correlation coefficient. (B) Association between RWT and LAD. In patients with normal LVMI (left panel), scattered plots showed small correlation between RWT and LA diameter. R2 = 0.007, p < 0.001. In high LVMI (right panel), scatter plots showed small but, negative correlation between RWT and LA diameter. R2 = 0.020, p = 0.0010. (C) Association between LVMI and LVEF. Scatter plots showed the negative correlation between LVMI and LVEF. R2 = 0.14, p < 0.001. (D) Association between RWT and LVEF. In normal LVMI (left panel), scatter plots showed small correlation between RWT and LVEF. R2 = 0.002, p = 0.0029. In high LVMI (right panel), scatter plots showed small but, positive correlation between RWT and LVEF. R2 = 0.160, p < 0.001.
Factors associated with AF and underlying LA enlargement and a low LVEF
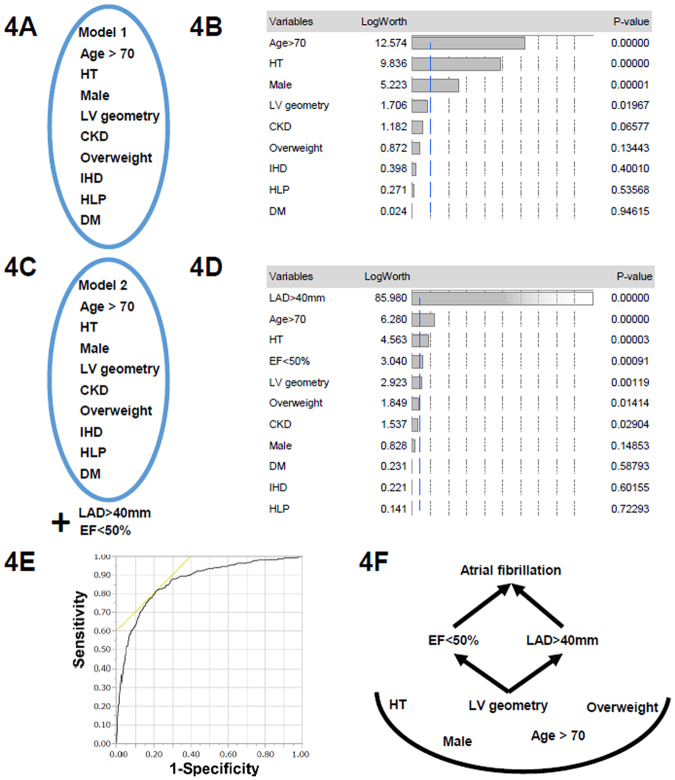
According to the results of the multivariate logistic regression analysis (model 1, Fig. 4A), the following variables were significantly associated with AF: age >70 years, male sex, hypertension, and LV geometric patterns (Table 3 and Fig. 4B). When we included two additional echocardiographic parameters, LA enlargement (LAD >40 mm) and a low LVEF (LVEF <50%), into a multivariate model for the presence of AF (model 2, Fig. 4C), LA enlargement was the echocardiographic parameter most associated with AF, and a low LVEF was the second most associated parameter (Fig. 4D), superior to the LV geometry. Finally, we sought the associated factors for LA enlargement and low LVEF. Table 4 shows the significant association with LV geometry and a high LAVI or LVEF. Finally, the presence of AF was well predicted by the values of LAVI and LVEF by receiver operating curve analysis (area under the curve 0.87; Fig. 4F). These indicated that the LV morphology was linked to the LV function and LA size (schematic in Fig. 4F).
Figure 4.
(A) Model 1 in Table 3 includes factors included age >70 years; sex; LV geometric remodelings that were defined as ordered variables (normal geometry, concentric remodeling, concentric hypertrophy, and eccentric hypertrophy); and the presence of comorbidities such as ischemic heart disease (IHD), hypertension (HT), diabetes mellitus (DM), hyperlipidemia (HLP), CKD and overweight (body mass index >25 kg/m2). (B) LogWorth scales of multivariate model 1 in Table 3. The p-values were transformed to the LogWorth (−log10(p-value)) scale. Hence, any LogWorth above 2 corresponds to a p-value below 0.01. A LogWorth of zero corresponds to a nonsignificant p-value of 1. (C) Model 2 includes factors included as factors for model 1, as well as left atrial enlargement [LAD (left atrial diameter) >40 mm] and low LVEF (EF <50%). (D) LogWorth scales of multivariate model 2. (E) Results of receiver operating characteristic (ROC) curve analysis. For predicting the presence of AF, the formula was generated from a linear regression (x = 3.55296 + 0.0208 × LVEF +− 0.09732 × LAVI). The prediction formulas for the response levels predicting the presence of AF and the absence of AF were (1/(1 + Exp(x)) and (1/(1 + EXP(−x)), respectively. Then we compared the two levels and predicted the presence or absence of AF according to the larger response level. The ROC curve shows an area under the curve of 0.871, and when x = 2.1433, the sensitivity is 0.8044 and 1-specificity is 0.6042. (F) A proposed schema for the relationship between LV geometry, LVEF, LA enlargement, and AF.
Table 3.
Factors associated with AF according to multivariate logistic regression analysis.
| AF | |||
|---|---|---|---|
| Multivariate OR | 95% CI | P | |
| Age >70 years | 2.16 | 1.75–2.68 | <0.0001 |
| Male sex | 1.67 | 1.35–2.06 | <0.0001 |
| HT | 2.22 | 1.74–2.84 | <0.0001 |
| DM | 1.00 | 0.80–1.24 | 0.987 |
| CKD | 0.77 | 0.57–1.02 | 0.078 |
| HLP | 0.94 | 0.75–1.18 | 0.615 |
| Overweight | 1.18 | 0.99–1.47 | 0.13 |
| LV geometry | |||
| Normal geometry | 1 (Reference) | ||
| Concentric remodeling | 0.75 | 0.53–1.03 | 0.085 |
| Concentric hypertrophy | 1.10 | 0.66–1.75 | 0.683 |
| Eccentric hypertrophy | 1.45 | 1.06–1.96 | 0.018 |
| Concentric remodeling | 1 (Reference) | ||
| Concentric hypertrophy | 1.47 | 0.83–2.54 | 0.179 |
| Eccentric hypertrophy | 1.96 | 1.28–2.94 | 0.001 |
| Concentric hypertrophy | 1 (Reference) | ||
| Eccentric hypertrophy | 1.31 | 0.78–2.28 | 0.309 |
Abbreviations: OR = odds ratio; CI = confidence interval; RWT = relative wall thickness; LVMI = left ventricular mass index; HT = hypertension; DM = diabetes mellitus; CKD = chronic kidney disease, HLP = Hyperlipidemia.
Table 4.
Factors associated with a high LAVI and a low LVEF according to multivariate logistic regression analysis.
| A high LAVI | A low LVEF | |||||
|---|---|---|---|---|---|---|
| Multivariate OR | 95% CI | P | Multivariate OR | 95% CI | p | |
| Age >70 years | 3.29 | 2.43–4.47 | <0.0001 | 0.85 | 0.63–1.14 | 0.288 |
| Male sex | 0.95 | 0.73–1.25 | 0.751 | 2.14 | 1.55–2.94 | <0.0001 |
| HT | 2.14 | 1.53–2.99 | <0.0001 | 4.60 | 2.93–7.23 | <0.0001 |
| DM | 1.19 | 0.89–1.60 | 0.242 | 1.08 | 0.78–1.49 | 0.623 |
| CKD | 0.95 | 0.67–1.35 | 0.789 | 1.07 | 0.74–1.53 | 0.705 |
| HLP | 0.658 | 0.47–0.89 | 0.007 | 1.24 | 0.91–1.70 | 0.166 |
| Overweight | 0.91 | 0.68–1.24 | 0.574 | 0.72 | 0.51–0.99 | 0.045 |
| LV geometry | ||||||
| Normal geometry | 1 (Reference) | 1 (Reference) | ||||
| Concentric remodeling | 1.03 | 0.66–1.61 | 0.872 | 0.41 | 0.19–0.86 | 0.018 |
| Concentric hypertrophy | 3.36 | 2.05–5.51 | <0.0001 | 1.48 | 0.74–2.96 | 0.259 |
| Eccentric hypertrophy | 5.77 | 4.20–7.98 | <0.0001 | 11.3 | 8.18–15.5 | <0.0001 |
| Concentric remodeling | 1 (Reference) | 1 (Reference) | ||||
| Concentric hypertrophy | 3.24 | 1.77–5.94 | 0.0001 | 3.59 | 1.37–9.37 | 0.008 |
| Eccentric hypertrophy | 5.56 | 3.44–8.99 | <0.0001 | 27.5 | 13.1–57.9 | <0.001 |
| Concentric hypertrophy | 1 (Reference) | 1 (Reference) | ||||
| Eccentric hypertrophy | 1.71 | 1.02–2.87 | 0.040 | 7.66 | 3.83–15.3 | <0.0001 |
Abbreviations: OR = odds ratio; CI = confidence interval; RWT = relative wall thickness; LVMI = left ventricular mass index; HT = hypertension; DM = diabetes mellitus; CKD = chronic kidney disease, HLP = Hyperlipidemia.
Discussion
The main finding of the present study was that the prevalence of AF increased per the geometric remodeling patterns. The mechanism for why the prevalence of AF increased is unclear; however, cardiac remodeling manifests as changes in the cardiac size, shape, and function in response to aging, cardiac impairment, or an increased load9. Cardiac remodeling causes an increase in end-diastolic pressure and expansion of end-diastolic volume with diastolic and systolic dysfunction9. In the present study, patients with concentric remodeling, concentric hypertrophy, and eccentric hypertrophy were older than those with a normal geometry. In addition, patients with concentric remodeling, concentric hypertrophy, and eccentric hypertrophy had a higher E/e’. E/e’ is highest in patients with concentric hypertrophy17.
Concentric remodeling and hypertrophy were often caused by pressure overload that increased the RWT. This was an adaptation to normalize the systolic wall stress. However, LV was shown to be a substrate for LV diastolic dysfunction in concentric remodeling and an incremental risk for diastolic heart failure in concentric hypertrophy. Direct underlying triggers of AF was mainly due to atrial remodeling. Atrial remodeling is caused by multiple factors19. The left atrium is a thin-walled structure that connects the pulmonary veins and LV in diastole and pumps the LV (atrial kick) in end-diastole during sinus rhythm15. Previous work has highlighted the complex mechanisms contributing to AF progression19–23, as the morphological, electrical, or neurohormonal remodeling along with the ventricular responses were related. Diastolic impairment and an increasing diastolic filling pressure are related to the LA size. With an increase in LA filling pressures, the atrial wall stretches and enlarges. In this study, overweight was not significantly associated with AF, which was inconsistent with the previous studies24. One of the reasons may be the differences in baseline characteristics in Japanese population and in Western population. Body weights in Japan are much lower than those in the US and Europe. Other possible reason may be due to small numbers studied in our study, considering that a marginally significance existed.
Eccentric hypertrophy is typically induced by volume overload, such as in valvular heart disease or systolic heart failure. The prevalence of systolic dysfunction is mostly noted in eccentric hypertrophy. Systolic dysfunction affects LV geometry through enlargement of the LV chamber. A large LV and low LVEF cause congestion of the left atrium21–23. Consequently, LA enlargement in eccentric hypertrophy is suggested to be highest among groups with pressure and volume overload. A larger LA volume is associated with a higher risk of AF15. In addition, patients with AF and patients with a larger LA showed a higher LVMI and a low LVEF in a sub-study of the AFFIRM trial25. In our study analysis of both AF and non-AF populations, LA enlargement and LVEF were significantly related to LV geometric remodeling. Therefore, the rate of AF was increasing per the LV remodeling patterns. This was true for patients with non-valvular AF. The prevalence of LVH was high in patients with non-valvular AF26. Patel et al. reported that LAVI was associated only with LVMI, not RWT21. This is consistent with our present study which showed that the relationship between LA size and RWT was different for normal and high LVMI.
In addition, LAVI and LVEF accurately predicted the presence of AF in our study; therefore, we performed multivariate analyses excluding these co-linear factors. A linear association with LVMI and LA enlargement was observed; however, the effect of RWT on a low LVEF was different between patients with a normal LVMI and those with a high LVMI. Because LV geometric remodeling was clearly associated with LA enlargement and a low LVEF, a schematic in which that LV morphology was linked to the LV function and LA size is provided (Fig. 4F). Furthermore, the LV morphology was linked to the prognosis7,26. Thromboembolic events may be one of the mechanisms for the higher mortality in LVH27. Although there are reportedly many factors related to AF and LA enlargement, such as age, the presence of hypertension, and obesity24, LV geometric remodeling has an impact on the presence of AF through the association with LA enlargement and a low LVEF. However, atrial fibrillation can develop without left atrial enlargement28. Atrial size increases with time in patients with atrial fibrillation, without any structural or functional abnormality of LV and the valve28. Since our study is cross-sectional, we could not identify the cause-effect relationship in each patient. Longitudinal follow-up could address this issue in each patient.
In clinical practice, LVH is a risk factor in hypertensive patients29 and severe AS patients30. In our study, we provided the link between LV geometric patterns and AF prevalence through the contribution of atrial enlargement and decreased LVEF. AF and LA enlargement is a risk factor for ischemic stroke and may contribute to doubling all-cause31 and CV deaths14, and, in a specific condition, non-CV deaths32. However, anticoagulants were sub-optimally prescribed in these patients. Attention should be paid to paroxysmal AF in the LVH population to improve our daily practice.
There are several limitations to the present study. First, the subjects are heterogeneous because of the TTE and ECG findings. The ordering criteria for ECG and TTE were not set. Second, patients’ data were extracted from electronic medical data. A lack of information regarding exercise was an important limitation24. Third, we did not consider the effect of valvular diseases such as mitral valve regurgitation. Fourth, patients with short-lasting paroxysmal AF were excluded, because we included patients with AF when they had an AF rhythm according to an ECG examination. Lastly, the data presented provide only hypothesis-generating associations between LV geometry and AF. Since this study had a cross-sectional design, a cause-effect relationship could not be assessed. Further prospective studies are needed to determine the association of LA remodeling, LV geometric changes, LV function, mortality, and the time-course for the development of AF.
Conclusions
The prevalence of AF was increasing according to LV geometric remodeling patterns in association with LA size and LVEF.
Author Contributions
Y.S. and T.K.: conceived the design, performed statistical analysis, and wrote manuscript. T.H., T.I., S.M., E.N., and M.I.: collected the data and made critical revision.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Zabalgoitia M, et al. Impact of coronary artery disease on left ventricular systolic function and geometry in hypertensive patients with left ventricular hypertrophy (the LIFE study) Am. J. Cardiol. 2001;88:646–650. doi: 10.1016/S0002-9149(01)01807-0. [DOI] [PubMed] [Google Scholar]
- 2.Casale PN, et al. Value of echocardiographic measurement of left ventricular mass in predicting cardiovascular morbid events in hypertensive men. Ann. Intern. Med. 1986;105:173–178. doi: 10.7326/0003-4819-105-2-173. [DOI] [PubMed] [Google Scholar]
- 3.Ghali JK, Liao Y, Cooper RS. Influence of left ventricular geometric patterns on prognosis in patients with or without coronary artery disease. J. Am. Coll. Cardiol. 1998;31:1635–1640. doi: 10.1016/S0735-1097(98)00131-4. [DOI] [PubMed] [Google Scholar]
- 4.Koren MJ, Devereux RB, Casale PN, Savage DD, Laragh JH. Relation of left ventricular mass and geometry, to morbidity and mortality in uncomplicated essential hypertension. Ann. Intern. Med. 1991;114:345–352. doi: 10.7326/0003-4819-114-5-345. [DOI] [PubMed] [Google Scholar]
- 5.Ganau A, et al. Patterns of left ventricular hypertrophy and geometric remodeling in essential hypertension. J. Am. Coll. Cardiol. 1992;19:1550–1558. doi: 10.1016/0735-1097(92)90617-V. [DOI] [PubMed] [Google Scholar]
- 6.Patel DA, Lavie CJ, Milani RV, Ventura HO. Left atrial volume index predictive of mortality independent of left ventricular geometry in a large clinical cohort with preserved ejection fraction. Mayo Clin. Proc. 2011;86:730–737. doi: 10.4065/mcp.2010.0682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Oktay AA, et al. Current Perspectives on Left Ventricular Geometry in Systemic Hypertension. Prog Cardiovasc Dis. 2016;59:235–246. doi: 10.1016/j.pcad.2016.09.001. [DOI] [PubMed] [Google Scholar]
- 8.Lavie CJ, et al. Impact of echocardiographic left ventricular geometry on clinical prognosis. Prog Cardiovasc Dis. 2014;57:3–9. doi: 10.1016/j.pcad.2014.05.003. [DOI] [PubMed] [Google Scholar]
- 9.Cohn JN, Ferrari R, Sharpe N. Cardiac remodeling–concepts and clinical implications: a consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J. Am. Coll. Cardiol. 2000;35:569–582. doi: 10.1016/S0735-1097(99)00630-0. [DOI] [PubMed] [Google Scholar]
- 10.Wolf PA, Abbott RD, Kannel WB. Atrial fibrillation as an independent risk factor for stroke: the Framingham Study. Stroke. 1991;22:983–988. doi: 10.1161/01.STR.22.8.983. [DOI] [PubMed] [Google Scholar]
- 11.Lin HJ, et al. Stroke severity in atrial fibrillation: the Framingham Study. Stroke. 1996;27:1760–1764. doi: 10.1161/01.STR.27.10.1760. [DOI] [PubMed] [Google Scholar]
- 12.Whisnant J,P, et al. Comparison of population-based models of risk factors for TIA and ischemic stroke. Neurology. 1999;53:532–536. doi: 10.1212/WNL.53.3.532. [DOI] [PubMed] [Google Scholar]
- 13.Benjamin EJ, et al. Impact of atrial fibrillation on the risk of death: the Framingham Heart Study. Circulation. 1998;98:946–952. doi: 10.1161/01.CIR.98.10.946. [DOI] [PubMed] [Google Scholar]
- 14.Wolf PA, Mitchell JB, Baker CS, Kannel WB, D’Agostino RB. Impact of atrial fibrillation on mortality, stroke, and medical costs. Arch. Intern. Med. 1998;158:229–234. doi: 10.1001/archinte.158.3.229. [DOI] [PubMed] [Google Scholar]
- 15.Tsang TS, et al. Left atrial volume: important risk marker of incident atrial fibrillation in 1655 older men and women. Mayo Clin. Proc. 2001;76:467–475. doi: 10.4065/76.5.467. [DOI] [PubMed] [Google Scholar]
- 16.Lang RM, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015;28:1–39.e14. doi: 10.1016/j.echo.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 17.Nagueh SF, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2016;29:277–314. doi: 10.1016/j.echo.2016.01.011. [DOI] [PubMed] [Google Scholar]
- 18.JMP 13.2 Online Documentation. https://www.jmp.com/support/help/13-2/Effect_Summary.shtml.
- 19.Kirchhof P, et al. 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace. 2016;18:1609–1678. doi: 10.1093/europace/euw295. [DOI] [PubMed] [Google Scholar]
- 20.El Aouar LM, et al. Relationship between left atrial volume and diastolic dysfunction in 500 Brazilian patients. Arq. Bras. Cardiol. 2013;101:52–58. doi: 10.5935/abc.20130109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Patel DA, et al. Association of left ventricular geometry with left atrial enlargement in patients with preserved ejection fraction. Congest. Heart Fail. 2012;8:4–8. doi: 10.1111/j.1751-7133.2011.00264.x. [DOI] [PubMed] [Google Scholar]
- 22.Cioffi G, Mureddu GF, Stefenelli C, de Simone G. Relationship between left ventricular geometry and left atrial size and function in patients with systemic hypertension. J. Hypertens. 2004;22:1589–1596. doi: 10.1097/01.hjh.0000125454.28861.76. [DOI] [PubMed] [Google Scholar]
- 23.Melenovsky V, et al. Left atrial remodeling and function in advanced heart failure with preserved or reduced ejection fraction. Circ. Heart Fail. 2015;8:295–303. doi: 10.1161/CIRCHEARTFAILURE.114.001667. [DOI] [PubMed] [Google Scholar]
- 24.Lavie CJ, et al. Obesity and Atrial Fibrillation Prevalence, Pathogenesis, and Prognosis: Effects of Weight Loss and Exercise. J Am Coll Cardiol. 2015;70:2022–2035. doi: 10.1016/j.jacc.2017.09.002. [DOI] [PubMed] [Google Scholar]
- 25.Proietti M, Raparelli V, Basili S, Olshansky B, Lip GY. Relation of female sex to left atrial diameter and cardiovascular death in atrial fibrillation: The AFFIRM Trial. Int J Cardiol. 2016;207:258–263. doi: 10.1016/j.ijcard.2016.01.169. [DOI] [PubMed] [Google Scholar]
- 26.Gerdts E, et al. Relation of Left Ventricular Mass to Prognosis in Initially Asymptomatic Mild to Moderate Aortic Valve Stenosis. Circ Cardiovasc Imaging. 2015;8:e003644. doi: 10.1161/CIRCIMAGING.115.003644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Proietti M, et al. Frequency of Left Ventricular Hypertrophy in Non-Valvular Atrial Fibrillation. Am J Cardiol. 2015;116:877–882. doi: 10.1016/j.amjcard.2015.05.060. [DOI] [PubMed] [Google Scholar]
- 28.Sanfilippo AJ. Atrial Enlargement as a Consequence of Atrial Fibrillation A Prospective Echocardiographic Study. Circulation. 1990;82:792–797. doi: 10.1161/01.CIR.82.3.792. [DOI] [PubMed] [Google Scholar]
- 29.Verdecchia P, et al. Left ventricular hypertrophy as an independent predictor of acute cerebrovascular events in essential hypertension. Circulation. 2001;109:2039–2044. doi: 10.1161/hc4201.097944. [DOI] [PubMed] [Google Scholar]
- 30.Minamino-Muta E, et al. Impact of the left ventricular mass index on the outcomes of severe aortic stenosis. Heart. 2017;103:1992–1999. doi: 10.1136/heartjnl-2016-311022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Patel DA, et al. Prediction of All-Cause Mortality by the Left Atrial Volume Index in Patients With Normal Left Ventricular Filling Pressure and Preserved Ejection Fraction. Mayo Clin Proc. 2015;90:1499–505. doi: 10.1016/j.mayocp.2015.07.021. [DOI] [PubMed] [Google Scholar]
- 32.Minamino-Muta E, et al. Causes of Death in Patients with Severe Aortic Stenosis: An Observational study. Sci Rep. 2017;7:14723. doi: 10.1038/s41598-017-15316-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.