Abstract
Although it is well established that the olfactory epithelium of teleost fish detects at least 6 classes of biologically relevant odorants using 5 types of olfactory sensory neurons (OSNs), little is understood about the specificity of individual OSNs and thus how they encode identity of natural odors. In this study, we used in vivo extracellular single-unit recording to examine the odor responsiveness and physiological characteristics of 109 individual OSNs in mature male goldfish to a broad range of biological odorants including feeding stimuli (amino acids, polyamines, nucleotides), sex pheromones (sex steroids, prostaglandins [PGs]), and a putative social cue (bile acids). Sixty-one OSNs were chemosensitive, with over half of these (36) responding to amino acids, 7 to polyamines, 7 to nucleotides, 5 to bile acids, 9 to PGs, and 7 to sex steroids. Approximately a quarter of the amino acid-sensitive units also responded to polyamines or nucleotides. Three of 6 amino acid-sensitive units responded to more than 1 amino acid compound, and 5 sex pheromone-sensitive units detected just 1 sex pheromone. While pheromone-sensitive OSNs also responded to the adenylyl cyclase activator, forskolin, amino acid-sensitive OSNs responded to either forskolin or a phospholipase C activator, imipramine. Most OSNs responded to odorants and activators with excitation. Our results suggest that pheromone information is encoded by OSNs specifically tuned to single sex pheromones and employ adenylyl cyclase, suggestive of a labeled-line organization, while food information is encoded by a combination of OSNs that use both adenylyl cyclase and phospholipase C and are often less specifically tuned.
Keywords: olfactory receptor neurons, signal transduction, sex pheromone, amino acid, goldfish, electrophysiology
Introduction
Aquatic organisms including teleost (bony) fish use their olfactory sense to detect a wide variety of chemical cues to find mates, synchronize mating, find food, locate habitats, and avoid predators. A particularly large number of biologically relevant odorants have been identified in the goldfish, Carassius auratus, and their biological function clearly elucidated, making it one of the best understood vertebrate models of natural odor identity and function. In the goldfish, and seemingly the few other bony fishes that have also been tested, 6 distinctly different chemical classes of odorants with biological function have now been identified. In particular, dozens of essential l-amino acids and polyamines are known from both EOG recording to be detected with specificity and to drive feeding behaviors in the goldfish (Rolen et al. 2003; Hara 2006). Additionally in the goldfish, 2 sex steroids (androstenedione [AD], 17,20β-dihydroxy-4-pregnen-3-one [17,20βP]) and 1 sex steroid metabolite (17,20β-dihydroxy-4-pregnen-3-one-sulphate [17,20βP-s]) as well as 2 prostaglandins (PGs) (prostaglandin F2α [PGF], 15-keto prostaglandin F2α [15kPGF]) have been identified and shown to be both detected with great sensitivity and specificity, and to function as different types of sex pheromones (Sorensen et al. 1988, 1989, 2005). Unlike the situation for amino acids, the goldfish peripheral olfactory system is narrowly tuned to these 3 sex steroids. Of dozens of sex steroids tested, only these 3 are detected with notable sensitivity (Sorensen et al. 1990). Response specificity to both PG pheromones also appear similarly narrow (Sorensen et al. 1988). In addition to these 4 classes of chemicals, several nucleotides have also been shown to serve as feeding cues in the zebrafish, Danio rerio, (Wakisaka et al. 2017) and likely have a similar role in goldfish based on their essential roles in all aquatic animals. Finally, half a dozen bile acids (a class of biliary steroid) are also known to be detected by the goldfish with high sensitivity and specificity (Sorensen et al. 1987; Levesque et al. 2011; Buchinger et al. 2014) and may function as nonsexual social cues, as they do in the sea lamprey, Petromyzon marinus (Li et al. 1995). However, how this impressive variety of biological cues is encoded by the peripheral olfactory system of fish is poorly understood as little information is presently available on olfactory receptors (ORs) and how they are expressed and function in individual olfactory sensory neurons (OSNs).
Teleost fish, including the goldfish, have a single olfactory organ which contains many types of OSNs which are intermingled and detect all odorants. Five types of OSNs (ciliated OSNs, microvillous OSNs, pear-shaped OSNs, crypt neurons, and kappe neurons) have been described to date in the fish nose (Hansen et al 1999; Ahuja et al. 2014; Yoshihara 2014; Wakisaka et al. 2017). This scenario differs from rodents in which different chemosensory neurons are anatomically segregated with ciliated OSNs detecting general as well as food odors in the main olfactory systems while microvillous vomeronasal sensory neurons (VSNs) detect pheromones (Touhara and Vosshall 2009). Further, while ORs and trace amine-associated receptors (TAARs) are found in fish ciliated OSNs (Sato et al. 2005; Hussain et al. 2013), V2R receptors are commonly found in microvillous OSNs (Speca et al. 1999; Sato et al. 2005; Koide et al. 2009; DeMaria et al. 2013; Hussain et al. 2013). Additionally fish crypt neurons express a V1R (Oka et al. 2012) while kappe neurons express an ancestral V1R, termed ORA1 (Behrens et al. 2014). The binding of these receptors with their ligands activates specific G-protein α-subunits, which in turn activate a downstream signaling cascade, resulting in the generation of depolarizing receptor currents via the activation of cyclic nucleotide-gated (CNG) or transient receptor potential C2 (TRPC2) ion channels (Speca et al. 1999; Sorensen and Sato 2005; DeMaria et al. 2013; Hussain et al. 2013). These 2 transduction pathways are found in the different types of OSNs. While ciliated OSNs employ a Golf/adenylyl cyclase (AC) signaling cascade to activate CNG channels, microvillous OSNs employ a Gq/phospholipase C (PLC) pathway together with TRPC2 (Speca et al. 1999; Hansen et al. 2003; Sato et al. 2005). Although gene expression profiles demonstrate much functional similarity between rodent OSNs and fish ciliated OSNs, as well as rodent VSNs and fish microvillous OSNs, it appears based on a single molecular study in zebrafish that microvillous OSNs may not be involved in pheromonal communication in fish (Yabuki et al. 2016). The question of how functionally different odors are encoded in the over 25 000 species of bony fish, approximately half of vertebrates, and the sensitivities of individual fish OSNs to the broad array of biological odorants they discern is still unclear. Because of the relatively poor genomic information available for the vast majority of fishes including the goldfish, studies of olfactory encoding presently require physiological approaches to describe the chemosensitivies and activity of individual OSNs to the wide variety of known biological odorants.
The first step in neural coding and recognition of odors in fish and other vertebrates is activation of specific subsets of OSNs which project to specific glomeruli in the olfactory bulb to create unique spatial odor maps. Two ancient (non-teleost) fishes, the lampreys, and sharks, appear to primarily possess ciliated or microvillous OSNs respectively, yet the olfactory epithelia of both species responds to amino acids as measured by electroolfactogram (EOG) (Li et al. 1995; Laframboise et al. 2007; Tricas et al. 2009). Patch-clamp recordings of dissociated ciliated and microvillous OSNs of the rainbow trout Oncorhynchus mykiss, and Cabinza grunt, Isacia conceptionis, also have shown that both types of OSNs respond to amino acids (Sato and Suzuki 2001; Schmachtenberg and Bacigalupo 2004) while crypt neurons also respond to amino acids as well as bile acids (Vielma et al. 2008; Bazáes and Schmachtenberg 2012). Electrophysiological recording from the olfactory bulb of the channel catfish Ictalurus punctatus, also shows that while its ciliated OSNs detect amino acids and bile acids, its microvillous OSNs also detect amino acids (Hansen et al. 2003). In the zebrafish, certain microvillous OSNs express a V2R receptor that binds 2 hydrophobic amino acids (DeMaria et al. 2013), while some ciliated OSNs express ORs for PGF (Yabuki et al. 2016). In the goldfish, another V2R (GPRC) receptor found on microvillous OSNs responds to diverse amino acids and uses a PLC pathway (Speca et al. 1999). Together, these studies clearly show that a few different types of ORs and OSNs respond to food but provide little information on pheromones and the overall molecular receptive range of individual OSNs.
In this study, we performed in vivo extracellular single-unit recording to describe the sensitivity, the tuning and response characteristics of individual goldfish OSNs to 21 representative food and pheromonal odorants from all 6 known chemical classes of biologically relevant odorants. Representative odorants were chosen to reflect all known types (or subclasses) of odorant in each class known to have distinct olfactory and biological properties from prior EOG studies. Our study had 2 components. First, olfactory responsiveness of individual OSNs to the 6 odorant classes (classes were tested as mixtures of the representative odorants from that class) were determined, and then, each component of each mixture was tested to determine the active compound(s) in that odorant class/mixture. Second, the olfactory signal transduction mechanisms of these OSNs was characterized by testing enzyme activators and inhibitors of G-protein signaling cascades on OSNs using pharmacological agents. Our study showed that goldfish OSNs that detect known sex pheromones are highly selective, employ AC and are likely ciliated OSNs, whereas neurons that discern foods have overlapping molecular chemoreceptive ranges to multiple odorants and likely reflect both ciliated and microvillous cell types.
Materials and methods
Experimental animals
Sexually mature male goldfish (body weight: 12–30 g) were obtained from Hunting Creek Fisheries and maintained on a 16L:8D light/dark cycle in 1000 L tanks with flowing, aerated 15–17°C well water. We used males because the goldfish olfactory sense is sexually dimorphic to PGs (Ghosal et al. 2016). Fish were fed daily with tetramin-flaked food (Tetra GMBH). All fish were spermiated, possessed secondary sexual characteristics (tubercles), and spawned when offered the opportunity. All procedures were performed with the approval of the University of Minnesota’s Institutional Animal Care and Use Committee.
Recording procedures for in vivo extracellular single-unit activity and EOG
In vivo extracellular single-unit action potentials of OSNs were recorded using an electrophysiological technique that employs metal-filled glass electrodes developed by Gesteland (1975) (see below). Although extracellular electrophysiology does not permit identification of neuron cell type, it can reliably record the electrical activity of OSNs under natural conditions. EOG, a measure of integrated extracellular receptor potentials was recorded simultaneously to confirm preparation viability while serving as a marker of odor arrival time in the olfactory epithelium. Because we could not simultaneously optimize EOG electrode placement while recording with single-unit electrodes, the absolute size of EOG responses were not deemed biologically relevant and were not quantified. For recordings, individual goldfish were anesthetized with 2-phenoxyethanol (0.05%) and then immobilized with an intramuscular injection of Flaxedil (gallamine triethiodide; 0.6 µg/body weight g; DX53, Davis+Geck). Goldfish were then placed into a custom-built recording chamber and fixed in place using orbital ridge clamps. Their gills were perfused through their mouth with well water (15–17 °C, spawning temperature) while their body was covered with wet tissue paper to keep them moist throughout the experiment. An olfactory rosette was exposed using a fine scalpel to remove both the skin and connective tissue superficial to the olfactory epithelium. The exposed olfactory epithelium was flushed with flowing well water (9 ml/min) using an odor administrator (see below) for at least 30 min before recording commenced and throughout each experiment.
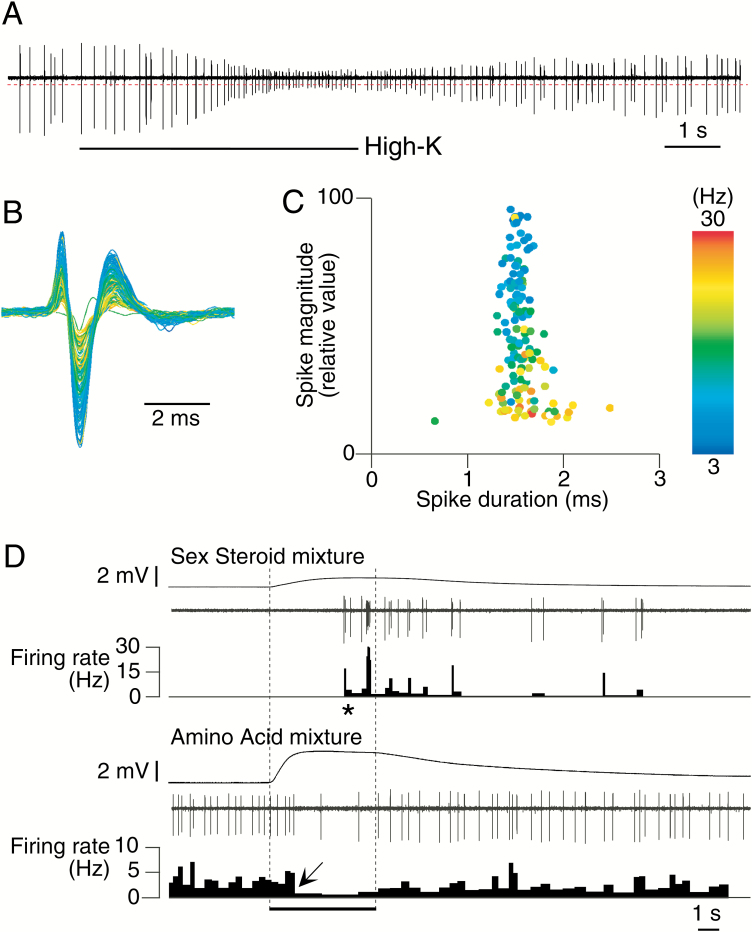
Single-unit action potentials were recorded extracellularly using metal-filled glass electrodes whose tips had been plated with platinum-black (tip opening: 1 µm inner diameter; ball diameter ranging from 5 to 8 µm). The impedance of these electrodes was 0.5–2 MΩ at DC. Spike signals were amplified by an AC amplifier (P511, Grass Instruments, West Warwick, RI; 10–1000 Hz bandpass). To monitor signals, the AC-output was connected to an audio speaker. To archive stable single-unit recording, the metal electrode was carefully placed on the dorsal-medial surface of an olfactory lamella, where the distribution and density of OSNs is the highest (Hansen et al. 1999). Then, the position of the electrode was carefully adjusted to locate spontaneous single-unit activity, using a hydraulic micromanipulator (MX610, Siskiyou Design Instruments). Extracellular spike waveforms were displayed on a storage oscilloscope (TDS210, Tektronix) and their magnitudes and durations carefully compared over time to verify consistent single-unit recording. We monitored spike duration because the time constant of individual action potential depends on the products of constant membrane resistance and capacitance at the individual recording sites should be constant, while spike size can (and often did) diminish with firing rate, presumably due to a progressive decrease of voltage-dependent sodium channel conductance (Figure 1A,B). Both before and after odor tests, unit identity (spike duration during depolarization) was confirmed by stimulating the olfactory epithelium with high-K solution (30 mM) dissolved in well water.
Figure 1.
Electrical characteristics of representative single goldfish OSNs. (A) A high-K-induced single-unit response of an OSN. The bottom bar indicates 5-s application of 30 mM high-K solution. (B) Waveforms of each spike event extracted from a trace shown in (A). The isolation threshold is indicated by a dotted line in (A). A bar indicates 2 ms. (C) Scatter plot illustrating the spike amplitudes (y-axis) and the spike duration at the 50% level of maximal spike amplitude (x-axis). In (B) and (C), pseudocolors indicate the firing rate, with blue and red corresponding to 3 and 30 Hz, respectively. (D) Typical excitatory (top) or inhibitory (bottom) single-unit responses. Each of the top traces in each set of the 3 describes the EOG, while the middle traces describe single-unit activity, and the bottom traces describe the firing rate at each spike event. The spike activity of the top single-unit was excited by the 10–9 M sex steroid mixture; the other was suppressed by the 10–4 M amino acid mixture. The bottom bar indicates the timing of 5-s odor stimulation. A vertical bar indicates voltage amplitude in EOG responses. Asterisks denote significant increases in firing rate (P < 0.05). Arrow indicate the significant decreases in firing rate (P < 0.05).
The EOG was simultaneously recorded using Ag/AgCl electrodes bridged to glass micropipettes, whose inner diameter were 100 µm, filled with goldfish ringer’s solution (in mM: 125 NaCl, 4 KCl, 0.3 MgSO4-7H2O, 0.9 MgCl2-6H2O, and 3.2 CaCl2-2H2O; pH 7.4) and 2% gelatin (Sorensen et al. 1987). EOG electrodes were positioned to provide a reasonable assessment of recording viability without interfering with single-unit electrode placement. EOG signals were amplified using a DC-amplifier (7P1G, Grass Instruments) with a low-pass-filter at 35 Hz. Both single-unit and EOG signals were stored on a PC-AT compatible computer using LabView software (National Instruments) at a 20-kHz sampling speed for subsequent offline analyses (see below).
During recording, odorants (stimuli) were applied directly to the surface of the olfactory epithelium as 5-s pulses through a Teflon tube via a custom-built gravity flow odor applicator using a pneumatic switcher (5300, Rheodyne) that reduced pressure and temperature fluctuations. This system continuously passed fresh well water (15–17 °C). We tested all stimuli at least twice with a 2-min break to confirm reproducibility of olfactory responsiveness.
Odorant solutions and pharmacological agents
We made up stock solutions of 21 representative odorants from 6 odorant classes (Table 1) every 4 weeks along with appropriate well water and methanol controls. Later, these stocks were also used to make up the 6 odorant mixtures which we tested first (see Experimental Design). Stocks were held at 4 °C and diluted later at the time of testing with well water to create biologically relevant concentrations which represented all types of odorants known from behavioral studies to be biologically relevant to the goldfish (Table 1). Concentrations were selected based on biological relevancy established in previous studies. We tested 5 amino acids, including each of the 4 different and discernable subclasses of amino acids based on sidechain structure (L-glutamic acid [Glu: acidic], L-arginine [Arg: basic], L-alanine [Ala: short chain-neutral], L-serine [Ser: short chain neutral], and L-methionine [Met: long chain neutral]) thought to activate independent EOG responses in the channel catfish (Caprio and Byrd 1984). By using a mixture of 4 amino acids representing each type of side chain, we sought to increase the chance of encountering amino acid sensitive single units. A fifth amino acid, 10–4 M L-serine, was tested alone as a standard amino acid as we have used it in previous studies (e.g. Sorensen et al. 1988, 1989). We also tested 3 polyamines, each of which appear to be discriminated by different combinations of TAARs and known to function as feeding cues (cadaverine, putrescine, and spermine) in the goldfish (Rolen et al. 2003; Li et al. 2015). Notably, polyamines have been found to be aversive in the zebrafish whose feeding habits differ from the goldfish (Hussain et al. 2013). Three nucleotides (adenosine 5ʹ-triphosphate [ATP], inosine 5ʹ-monophosphate [IMP] and inosine 5ʹ-triphosphate [ITP]), known to function as feeding cues in the related zebrafish (Wakisaka et al. 2017), were also tested. Four bile acids (cyprinol sulfate [CS], lithocholic acid [LCA], taurochenodeoxycholic acid [TCDC], and taurocholic acid [TCA])—each representing different chemical types of bile acid and putative social pheromonal cues, were tested as well (Buchinger et al. 2014). Additionally, 2 PGs (PGF and 15 kPGF) (Cayman Chemical), releaser pheromones, were also tested; these 2 compounds are known from EOG recording to be specifically discriminated by the goldfish olfactory system and to serve as a binary mixture with strong pheromonal activity. A PG metabolite, 13,14-dihydro-15keto-prostaglandin F2α (13dPGF) (Cayman Chemical) was tested as a negative control because it does not have pheromonal function in this species. Finally, we tested 3 sex steroids, AD, 17,20βP and 17,20βP-s, 3 components of a priming pheromone that have distinct chemical attributes and which we also knew to be discriminated by the goldfish olfactory system from EOG studies (Sorensen et al. 1990). L-amino acid, nucleotide, and polyamine stock solutions were made in well water at concentrations of 10–2 M, while bile acids, PGs, and sex steroids were first dissolved in a trace amount of methanol (1 mg/ml which is not detectable by EOG) and then dissolved in well water at 10–3 or 10–4 M. All compounds were of the highly possible purity (>99%). The 17,20βP-s was custom-synthesized by AP Scott (Lowestoft). We isolated and purified CS ourselves. All other compounds came from Sigma-Aldrich.
Table 1.
Odorants tested
| Chemical name | EOG detection threshold in goldfish | Concentration tested | Notes and Biological function |
|---|---|---|---|
| L-amino acid mixture | (10–4 M) | Common feeding stimulants in fisha,b | |
| Alanine (Ala) | 10–8 Mc | 10–4 M | Short side chain neutral |
| Arginine (Arg) | 10–7 Mb | 10–4 M | Basic side chain |
| Glutamic acid (Glu) | 10–8 Mc | 10–4 M | Acidic side chain |
| Methionine (Met) | 10–8 Mc | 10–4 M | Long side chain neutral |
| Nucleotide mixture | (10–5 M) | Putative feeding stimulantsd | |
| Adenosine 5ʹ-triphosphate | N.T. | 10–5 M | |
| Inosine 5ʹ-monophosphate | N.T. | 10–5 M | |
| Inosine 5ʹ-triphosphate | N.T. | 10–5 M | |
| Polyamine mixture | (10–5 M) | Goldfish feeding stimulantsb | |
| Cadaverine | 10–8 Mb | 10–5 M | |
| Putrescine | 10–8 Mb | 10–5 M | |
| Spermine | 10−8 Mb | 5 × 10-6 M | |
| Bile acid mixture | (10–7 M) | Putative aggregation cuese | |
| Cyprinol sulphate | 10−11 Mc | 10–7 M | Novel steroid only found in goldfish and relatives |
| Lithocholic acid | 10−11 Mc | 10–7 M | |
| Taurochenodeoxycholic acid | 10−11 Mc | 10–7 M | |
| Taurocholic acid | 10−9 Mf | 10–7 M | |
| F Prostaglandin mixture | (10–8 M) | ||
| Prostaglandin F2α (PGF) | 10−10 Mg | 10–8 M | Behavioral sex pheromoneg,h |
| 15-keto prostaglandin F2α (15KPGF) | 10–12 Mg | 10–8 M | Behavioral sex pheromone, more potent than prostaglandinF2α with which it is released, part of mixtureg,h |
| 13,14-dihydro-15-keto-prostaglandin F2α | 10–9 Mg | 10–8 M | PGF metabolite, non-pheromonal compound |
| Sex steroid mixture | (10–8 M) | ||
| Androstenedione (AD) | 10–11 Mi | 10–8 M | Male sex pheromone, and as part of female steroidal mixturei,,j |
| 17α,20β-dihydroxy-4-pregnen-3-one (17,20βP) | 5 × 10–12 Mk | 10–9 M | Female primer pheromone, typically as part of a steroidal mixturek,l |
| 17α,20β-dihydroxy-4-pregnen-3-one-sulfate (17,20βP-s) | 10–11 Ml | 10–8 M | Sulfated 17α,20β-dihydroxy-4-pregnen-3-one, behavioral female pheromone and as part of a steroid mixturel |
a(Hara 2006); b(Rolen et al. 2003); c(Sorensen and colleagues, unpublished); d(in zebrafish, Wakisaka et al. 2017); e(in lamprey, Li et al. 1995); f(Sorensen et al. 1987); g(Sorensen et al. 1988); h(Sorensen et al. 1989); i(Sorensen et al. 2005). j(Poling et al. 2001); k(Sorensen et al. 1990); l(Sorensen et al. 1995); N.T. not tested in goldfish.
To examine the role of AC and PLC signaling pathways in experiment #2 (see below), we used pharmacological inhibitors and activators for G-protein mediated signaling transduction machineries which were known to have these properties in goldfish (Sorensen and Sato 2005). A stock solution of 1 mM AC activator forskolin (7β-acetoxy-8,13-epoxt-1α,6β,9α-trihydroxylabd-14-en-11-one; Sigma-Aldrich) (Sorensen and Sato 2005) was prepared in 50% DMSO (v/v) and stored at −20 °C. A PLC activator imipramine (401840, CalBiochem) (Fukuda et al. 1994; Sorensen and Sato 2005) was prepared in 50% DMSO (v/v) at a concentration of 10 mM and stored at −20 °C. A stock solution of 1 mM PLC inhibitor (Sorensen and Sato 2005), U73122 (Sigma-Aldrich), was prepared in 50% DMSO (v/v) and stored at −20 °C. Well water was used to dilute these stock solutions during experiments to yield final DMSO concentration of <1 % (v/v). DMSO control (1% v/v in well water) was also tested.
Experimental design
In our initial experiment (#1), we first sought to determine the tuning properties of individual OSNs to mixtures of each of 6 classes of odorants and then each of the representative types of food and pheromone odorants they each contained. Because extracellular single-unit recordings rarely last longer than 20 min, we first screened the responsiveness of each unit using each of the 6 odorant mixtures, each of which was comprised of 3–4 representative odorants (Table 1), along with well water and methanol controls, while including high-K solution as a stimulus to confirm neuron identity/viability. If after testing responses to all 6 odorant mixtures, the recording was still stable, we tested the active mixtures at different concentrations to determine dose–response relationships and then tested each of their components to determine chemospecificity.
In experiment #2, we sought to characterize second messenger systems associated with single-unit responsiveness to either food (amino acids) or pheromonal odors (PGs, sex steroids). Because ciliated OSNs employ AC and CNG channels (Schmachtenberg and Bacigalupo 2004; Sato et al. 2005), whereas microvillous OSNs use PLC and TRPC2 as part of their olfactory signal transduction machineries (Speca et al. 1999; Sato et al. 2005), the effect of forskolin and imipramine addresses possible cell type. In this experiment, another group of OSNs were tested with controls, forskolin, imipramine, the amino acid mixture, the PG mixtures, and the sex steroid mixture. Procedures were identical to experiment #1.
Data analysis and statistics
All raw unit and EOG data were stored on a computer for offline analysis. For spike sorting, the consistency of extracted waveforms was confirmed using AutoCut 3.0 software (Datawave Technologies) in several steps. First, each spike event was extracted from the voltage trace using positive and negative going waveforms which crossed the baseline (0 mV). We excluded all those recordings which clearly included more than 2 OSNs because of difficulties in reliably separating units. Because spike frequency data are ordinal and not normally distributed, we used a non-parametric analysis called cumulative spike analysis (CSA) (Blejec 2000). This analytical technique accurately measures change in slope of cumulative spike time distribution across time and that can thus measure both excitation (increases in slope) and inhibition (decreases in slope) without making assumptions about data distribution while correcting for multiple comparisons. This analysis was conducted using a program based on S-Plus (Math Soft) on a PC-AT compatible computer. Using this program, we statistically compared each spike event for 15-s prior to each odorant application with the 20-s long odor application which followed it. The detection threshold was defined as the minimum concentration of odorant(s) which induced a significant increase in spike events. Raw data files were plotted by using Igor Pro (WaveMetrics) along with EOG. Finally, we also calculated a Response Index (RI) to quantify changes in firing rate (response magnitude) as follows:
RI = (the average firing rate of 5 (for excitatory responses) or 3 (for inhibition) spikes around the peak response – average firing rate of the neuron prior to odor exposure (i.e. spontaneous activity)/average firing rate of that neuron prior to odor exposure.
Results
Response profiles of goldfish ORNs to the 6 odorant mixtures
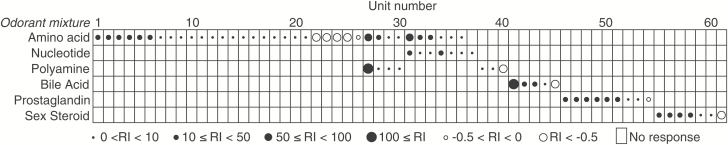
To examine whether the information of biologically relevant odorants is encoded by specific OSNs, or by the combinations of OSNs, we recorded olfactory responsiveness from 109 spontaneously active OSNs to all 6 odorant mixtures. All units responded to high-K and exhibited constant spike durations while showing typical action potentials (Figure 1A–C). Significant increases (excitation) or decreases (inhibition) of action potential firing rate were observed within approximately 6 s after the onset of EOG response (Figure 1D). Firing rates did not appear to be related to EOG amplitude. Spontaneous firing rates were generally less than 10 Hz, but there was great variation between units with some having a rate of less than 1 Hz (Supplementary Figure 1). Of the 109 units we recorded from, 61 (56%) were chemosensitive and responded to either one (n = 51; 84%) or 2 odorant mixtures (n = 10; 16%) (Figure 2). Forty-eight units did not respond to any odorant. None responded to 3 odorant classes. Most (n = 52) units responded with excitation to odorants although inhibition (n = 9) was also observed. Considerable variation was noted in the intensity of observed responses (RI), with some units responding with nearly 100-fold increases in firing rate and others merely doubling. No instances of units responding in an excitatory manner to one mixture and an inhibitory fashion to another were noted. No units responded to either methanol control or water control.
Figure 2.
Response profiles of the 61 chemosensitive single-units tested with all 6 odorant classes (mixtures) (48 insensitive units not shown). The Response Index (RI, relative change in response rate) of each olfactory response is shown by box drawings.
Amino acid-sensitive units were the most common type of unit (n = 36; 33% of 109 units) with 26 of these responding to just amino acid mixture and 10 responding to nucleotides or polyamine mixture as well (details below). Seven single units (6.4%) responded to nucleotides with 6 also responding to amino acids (see also Figure 3) and one of these just responding to nucleotides. Seven units (6.4%) responded to polyamines with 4 also responding to amino acids and 3 of these being specific to polyamines. A small subset of units (n = 5; 4.6%) responded to bile acids only. There were 9 PG-sensitive units (8.3%) and all responded to PGs only. Finally, 6.4% of all units (n = 7) responded to sex steroids, with all responding to just sex steroids. The chemoreceptive range of single-units to odorant mixtures did not change with repeated odor application, strongly suggesting that our single-unit recordings reflected the activities of single OSNs.
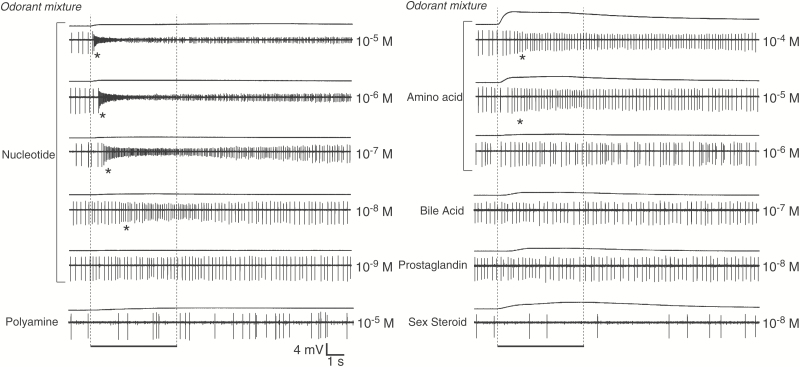
Figure 3.
An example of responses of a single goldfish OSN that responded to both the nucleotide and the amino acid mixture (unit #34 in Figure 2) and no other stimuli other than forskolin. Traces show EOG recordings (top traces of each pair of traces) and single-unit activity (bottom traces) elicited by the nucleotide, amino acid, polyamine, bile acid, prostaglandin, sex steroid mixtures, and 10–5 M forskolin. The mixture concentration of nucleotide and amino acid is indicated to the right. The vertical bar indicates amplitude of EOG amplitude. The bar at the bottom indicates the 5-s odorant stimulation period. Asterisks denote a significant increase in firing rate (P < 0.05).
We found that increasing concentrations of the amino acid mixture elicited increased firing rates (Figure 3; Supplementary Figure 2) as did nucleotide mixture with no change of specificity. Amino acid-sensitive units exhibited a variety of response types with about 14% (n = 5) showing inhibition and the rest, excitation (Figure 2). Polyamine-sensitive units also exhibited a variety of responses as did the bile acid, PG, and sex steroid sensitive units (Figure 2).
Detection thresholds were measured for several units that responded to food odorant mixtures. Sensitivity varied between units as did their dose-response relationships. For example, unit #27 (this unit number corresponds with that reported in Figure 2) showed a 10–6 M detection threshold for the polyamine mixture and 10–4 M for the amino acid mixture, while unit #28 showed a detection threshold of 10–5 M for both the polyamine mixture and the amino acid mixture, while the detection threshold in unit #34 to the nucleotide mixture and the amino acid mixture was 10–8 M and 10–5 M, respectively (Supplementary Figure 2). Unit #29 exhibited a 10–6 M and 10–5 M detection threshold to the polyamine mixture and amino acid mixture, respectively. Thus, the molecular receptive range of unit #28 was seemingly not specifically tuned to either amino acids or polyamines, whereas unit #27, #29, and #34 were rather selective to polyamines and nucleotides, respectively.
Response profiles of goldfish OSNs to individual amino acids and sex pheromones
Because amino acid-sensitive OSNs in channel catfish have been classified into 4 amino acid subclasses associated with different chemical attributes of their side chains: acidic, basic, short-chain neutral, and long-chain neutral (Caprio and Byrd 1984; Nikonov and Caprio 2007), we examined this possibility in the goldfish. We were able to examine the olfactory responsiveness of 6 amino acid-sensitive OSNs to the 4 individual amino acids contained in the amino acid mixture as well as L-Ser (Supplementary Figure 3). Units #1, #6, and #7 responded to arginine (basic), methionine (long-chain neutral), or glutamic acid (acidic), respectively, suggesting the involvement of independent receptor sites which recognized amino acid side chains. Further, 3 units responded to 3 or 4 amino acids with different side chains and with similar intensities (RIs), suggesting that amino acid side chains may be the key determinant of olfactory responsiveness (binding) in these instances and that amino acid detection maybe more complex than the catfish. We found that one OSN that responded to the amino acid mixture also detected 2 polyamines (Supplementary Figure 3).
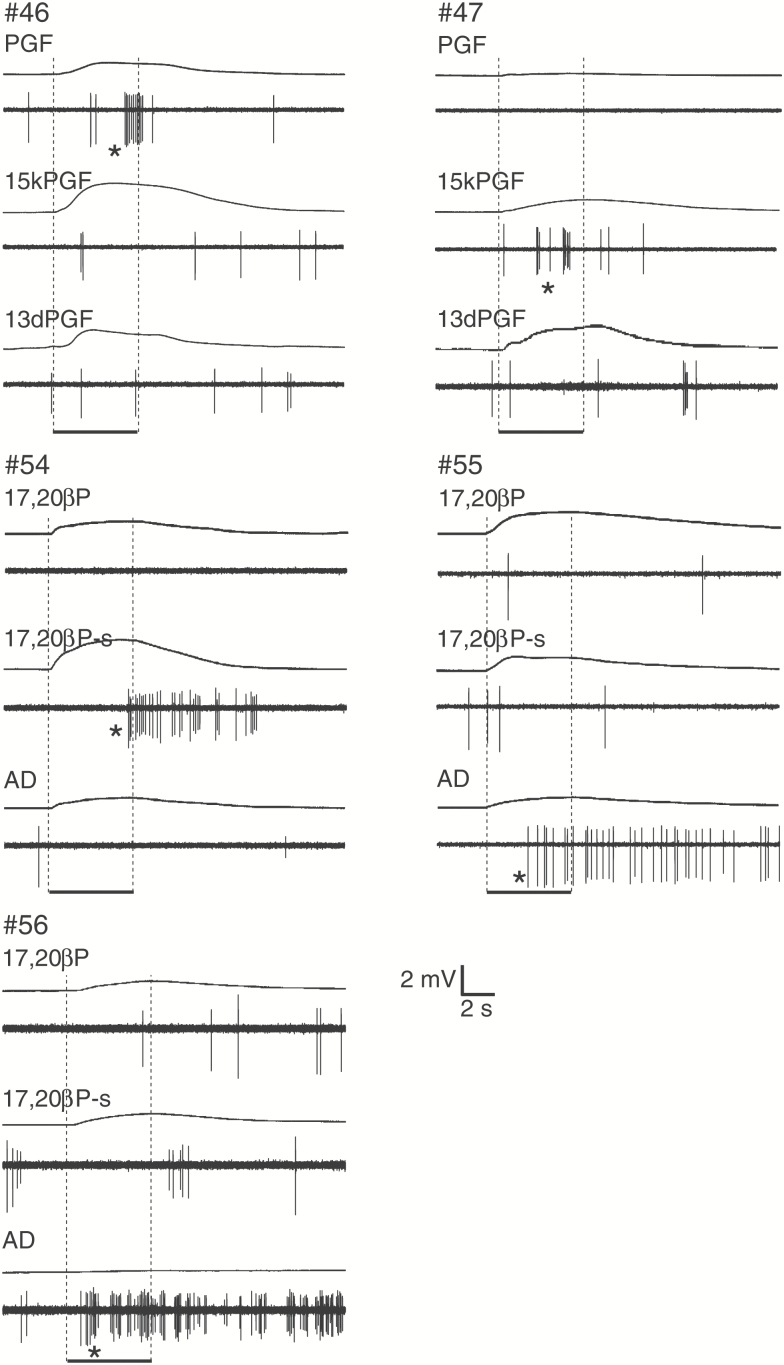
We also examined the tuning of 5 pheromone-sensitive OSNs to the individual components these mixtures contained. Two PG-sensitive units were found to respond to only a single PG, and with excitation. Three sex steroid-sensitive units were also tested; one of these responded to only 17,20βP-s, another 2 to AD only (Figure 4).
Figure 4.
Action potentials of 5 sex pheromone-sensitive single-units to single compounds. In each pair of traces, the top traces indicate EOGs, while the bottom traces indicate single-unit activity, respectively. Bottom bars indicate the timing of odorant stimulation for 5-s. A vertical bar indicates voltage amplitude in EOG responses. #46, #47, and #54 responded specifically to PGF, 15kPGF, 17,20βP-s, respectively. Both #55 and #56 was AD-specific single-units. Asterisks denote significant increase of firing rate (P < 0.05).
Pharmacological tests of olfactory signal transduction cascades
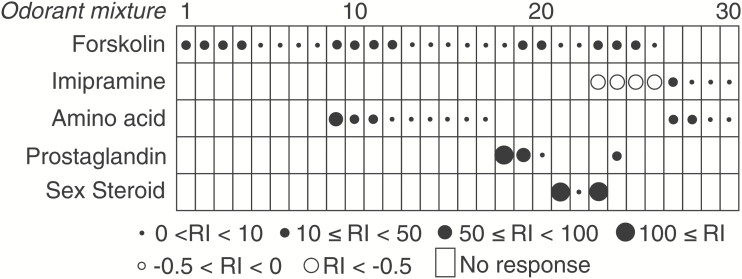
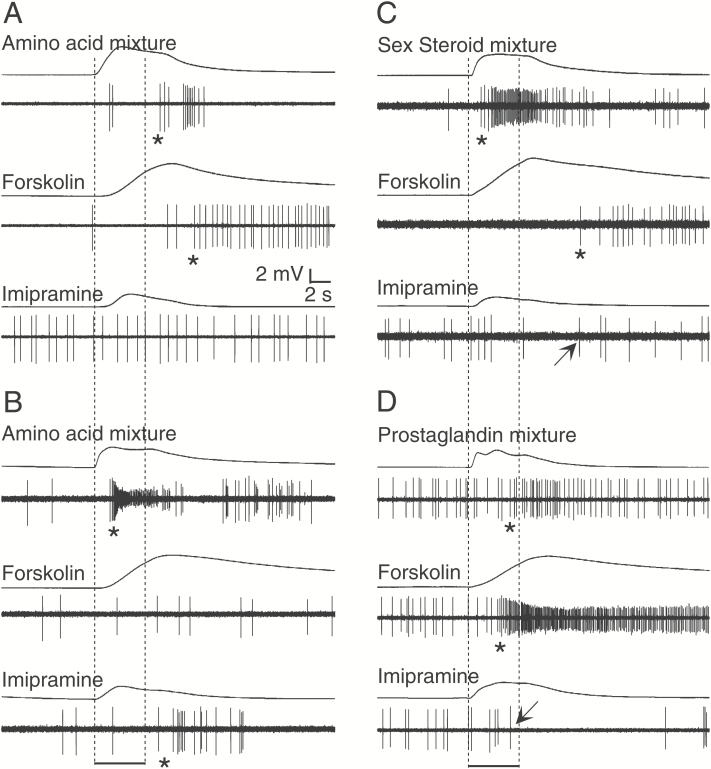
To examine the roles of AC and PLC signaling pathways in individual food and pheromone-sensitive OSNs, we tested the effect of an AC activator, forskolin, and a PLC activator, imipramine, along with mixture of amino acids, PGs, and sex steroids in additional 44 units. Twenty-six single-units (59%) of the 44 units responded to forskolin in an excitatory manner with 4 of these also responding to imipramine with a reduced firing rate, and 4 additional units responding to imipramine with excitation (and 14 not responding to either agents and odorants) (Figure 5). Although imipramine is one of the primary amines, it did not stimulate forskolin-sensitive single-units, indicating that imipramine did not activate AC and that the target of imipramine was not TAARs, but a PLC. Twenty-two of the forskolin and imipramine sensitive units also responded to an odorant mixture, and then in an excitatory fashion. Of the 13 units that responded to the amino acid mixture, 9 responded to forskolin, while 4 responded to imipramine with excitation (Figure 6A,B). All 3 sex steroid-sensitive units responded to forskolin in an excitatory fashion with one of these being suppressed by imipramine (Figure 6C). All 4 units that responded to PG mixture were also stimulated with forskolin and one of these being inhibited by imipramine as well (Figure 6D). None of the units responded to any controls including DMSO and no unit in this experiment responded to more than one odorant class. We also found differences in electrical activities between forskolin-sensitive and -insensitive single-units which responded to amino acid mixture: forskolin-insensitive single-units exhibited significantly higher action potential activity than forskolin-sensitive single-units (Supplementary Figure 1). Finally, when we tested the effect of a PLC inhibitor, U73122, to examine the contribution of PLC to action potential generation, we found that the responsiveness of 3 amino acid-sensitive neurons was blocked by U73122 but recovered when it was no longer being applied (Supplementary Figure 4).
Figure 5.
Response profiles of the 30 chemosensitive single-units, which were tested with 10–5 M forskolin, 10–4 M imipramine,10–4 M amino acid mixture, 10–8 M prostaglandin mixture, and 10–9 M sex steroid mixture. The RIs of each olfactory response and insensitiveness (no response) are shown as noted by box drawings.
Figure 6.
Four examples of the electrophysiological activity of single goldfish OSNs to pharmacological agents. (A) Activation of activity by exposure to 10–5 M forskolin in an amino acid-sensitive single-unit (#9 in Figure 5). (B) Activation of activity by 10–4 M imipramine in an amino acid-sensitive single-unit (#27 in Figure 5). (C) Activation of activity by 10–5 M forskolin in a sex steroid-sensitive single-unit (#23 in Figure 5). (D) Modulation of the spike activity by 10–4 M imipramine in a prostaglandin and forskolin-sensitive single units (#24 in Figure 5). In each pair of traces, the top trace shows the EOG while bottom trace indicates single-unit activity. Bottom bars indicate the timing of 5-s odorant stimulation. Asterisks denote significant increase of firing rate (P < 0.05). Arrows indicate a decrease in firing rate (P < 0.05).
Discussion
This study used single-unit electrophysiological recording to examine the specificities of over one hundred individual goldfish OSNs to a broad array of biologically relevant food and pheromonal odors and to determine their pharmacological characteristics. Our findings demonstrate that goldfish OSNs are specialized to detect odorants associated with specific biological functions including feeding, reproduction, and aggregation. Further, while sex pheromone information in the goldfish olfactory system is conveyed by narrowly tuned OSNs in a manner resembling labeled-lines, food information is encoded in a more complex manner. Remarkably, both the steroidal priming and PG-based releasing sex pheromones in the goldfish appear to be distinctly discerned by OSNs that detect only a single pheromone component and employ AC signaling pathways, as has been previously observed in ciliated OSNs in teleost fish (Schmachtenberg and Bacgalupo 2004; Sato et al. 2005; Yabuki et al. 2016). In contrast, amino acids and other feeding cues (polyamines, nucleotides) appear to be discerned by a large number of OSNs (about half) which exhibit a variety of molecular receptive ranges ranging from highly specific to quite broad and employ either AC or PLC pathways. Our finding that OSNs express either AC or PLC, but never both, suggests that both ciliated and microvillous OSNs detect food cues in the goldfish, as suggested by other reports (Speca et al. 1999; DeMaria et al. 2013; Wakisaka et al. 2017). Together, our results clearly demonstrate that specific neural pathways that encode sex pheromone and food odor information originate in the peripheral olfactory system of the goldfish, use distinctly different cellular processes, and that sex pheromone information is encoded by labeled-lines while food odor is not.
Our study found that a subset of forskolin-sensitive OSNs in the goldfish, a well established model of sex pheromone function in fish, exhibit highly specific and narrowly tuned responses to single F-type PG pheromones and sex steroidal pheromones. This level of specialization is reasonable because PGs and sex steroids are known to function as different pheromones in this species, with the former stimulating male pre-spawning hormonal surges (Dulka et al. 1987; Sorensen et al. 1995, 2005) and the latter, male spawning behavior (Sorensen et al. 1988, 1989). Consistent with a number of studies linking forskolin to AC, and AC to ciliated OSNs, these pheromone-sensitive neurons likely are ciliated OSNs (Schmachtenberg and Bacgalupo 2004; Sato et al. 2005; Yabuki et al. 2016). A similar conclusion was reached in the zebrafish using a genetic neuronal activation maker (Yabuki et al. 2016). Highly selective and sensitive detection of a single sex pheromone compound at near picomolar concentrations by goldfish OSNs is consistent with both EOG recording and behavioral tests in this species (Sorensen et al. 1988, 1989, 2005). One interesting difference between the zebrafish and goldfish is that goldfish OSNs appear to express 2 independent receptors for PGF and 15kPGF on different OSNs, while OR114-1, a zebrafish OR for PGF, detects both PGF, and 15kPGF (Yabuki et al. 2016). Given that sex pheromones are often species-specific, and that the goldfish appears to employ a mixture of PGF and 15KPGF as pheromone (Sorensen et al. 1988; Lim and Sorensen 2011), this difference may not be surprising. Our study also found evidence that sex steroid-sensitive OSNs in the goldfish are highly specialized which is notable because several studies have established that free-swimming goldfish naturally discern these particular steroids as distinct components in complex changing mixtures whose function changes with mixture composition (Sorensen et al. 1989, 2005; Derby and Sorensen 2008). Our results strongly suggest that labelled-line neural pathways are organized in the periphery for functional coding of different types of sex pheromones in the goldfish with mixture information possibly being recognized by the higher nervous system (Sorensen et al. 2005; Derby and Sorensen 2008; Levesque et al. 2011).
In contrast to sex pheromone-sensitive OSNs, we found very clear evidence that those goldfish OSNs which detect amino acids, an important class of food odor, use either an AC or a PLC second messenger system, indicating that multiple olfactory signaling pathways and neuron types are involved in food odor recognition in this species. Although ciliated OSNs have been reported to detect amino acids in several other fish species (Sato and Suzuki 2001; Vielma et al. 2008), zebrafish ciliated OSNs do not appear to respond to mixtures of amino acids although they do detect a food extract, amines and bile acids (DeMaria et al. 2013). Fish represent the largest and most diverse group of vertebrates and it is reasonable for there to be interspecific divergence of the ligand repertoire of ORs expressed in ciliated OSNs. The sub-millimolar detection thresholds we measured to the amino acid mixture in forskolin-sensitive goldfish OSNs are biological relevant (Valentinčič et al. 2000; Rolen et al. 2003). Interestingly, we found a subset of amino acid-sensitive OSNs responded to nucleotides, as well as polyamines; because zebrafish TARRs and an adenosine receptor are not known to be sensitive to amino acids, this observation suggests that multiple ORs might be expressed in some goldfish OSNs (Sato et al. 2005; DeMaria et al. 2013). Our observation that a subset of amino acid-sensitive goldfish OSNs detect polyamines is also intriguing, because polyamines function as feeding cues in the goldfish (Rolen et al. 2003) but as repellents in zebrafish (Hussain et al. 2013). The overlapping molecular receptive ranges of goldfish OSNs to amino acids and polyamines is biologically reasonable because this species feeds on detritus which contains all of these compounds. It is also possible that these compounds and amino acids in particular, may have more than a single role in the goldfish as they do in salmonids (Yambe et al. 2006; Yamamoto et al. 2010), but this possibility requires further study.
Consistent with studies of OSN activity in the channel catfish (Kang and Caprio 1995; Nikonov and Caprio 2007), goldfish OSNs showed both the excitatory and inhibitory responses. In Caenorhabditis elegans, ASK chemosensory neurons exhibit inhibitory olfactory responses to a subset of dauer pheromone components, which induce dauer formation, by the inhibition of guanylyl cyclase via 2 chemosenory receptors (Kim et al. 2009). Although antagonistic effects (Oka et al. 2004), CNG channel inhibition (Chen et al. 2006) by certain odorants, and presynaptic inhibition of OSNs by periglomerular interneurons (McGann et al. 2005) has been reported as molecular mechanisms of inhibitory responses of vertebrate OSNs, the molecular mechanism(s) underlying inhibition observed in our study is not clear. The precise biological importance of inhibition of goldfish OSN firing by certain odorants is interesting, although some argue it has little role in the rodent (McGann et al. 2005). We also found evidence that OSN action potentials were inhibited by imipramine, a PLC activator; and while we did not observe this in amino acid-sensitive OSNs, it has been reported that amino acid responses of salamander OSNs can be potentiated by pharmacological inhibition of a PLC (Delay and Dionne 2002). While it is tempting to consider the possibility that PLC might modulate the AC signaling cascade, other studies suggest that PLC is absent from ciliated OSNs along with its upstream G-protein subunit, Gαq/11 (Hansen et al. 2003; Elsaesser et al. 2005). Although at least 80% of imipramine-elicited electrical activity of goldfish OSNs requires activation of PLC (Sorensen and Sato 2005), imipramine is capable of activating olfactory inositol 1,4,5-trisphosphate-gated channels in common carp, Cyprinus carpio, OSNs (Cadiou and Molle 2003). Therefore, it is possible that other phospholipases and ion channels might be involved in the inhibitory response to imipramine. Possibly, there may be crosstalk between these signaling pathways.
Our study also speaks to the etiology of the EOG, a topic that has been of long standing interest in fish. It is notable that while amino acid-sensitive units are the most common, their number did not correlate with the size of the EOG they evoked which was smaller than that elicited by PGs and sex steroids as well as polyamines (Sorensen et al. 1987, 1988; Rolen et al. 2003). In the goldfish, calcium influx through the CNG or TRPC2 channels trigger Ca2+-activated Cl− channels resulting in the efflux of intracellular Cl−, which has the pivotal role in the generation of EOG response (Sorensen and Sato 2005). Smaller EOG responses to amino acids than sex pheromones could be attributable to low calcium conductance of TRPC2 channels expressed in microvillous OSNs relative to CNG channels in ciliated OSNs (Kaupp and Seifert 2002; Lucas et al. 2003).
Although a subset of odorants that we tested are nonvolatile odorants, other vertebrates including terrestrial animals can recognize these compounds via olfaction. Both amino acids and nucleotides are olfactory stimuli for Xenopus laevis tadpoles, and calcium responses to both odorants and forskolin have also been reported, similar to the goldfish (Czesnik et al. 2006). Although olfactory organ of Xenopus tadpoles also has the sensitivity to sulfated steroids, their detection thresholds are much higher than the tested concentration in our study (Sansone et al. 2015). The detection of a likely pheromonal steroid, androstenone, by a mammalian OR expressed in the main olfactory organ (Keller et al. 2007) and olfactory sensitivity of rainbow trout ciliated ORNs to a steroid metabolite (Sato and Suzuki 2001) have also been reported. The rodent vomeronasal organ also detects sulfated steroids using a TRPC2 signaling cascade (Nodari et al. 2008) although their biological function is unclear. The molecular basis of teleost chemical communication may employ orthologous receptors for the recognition of non-olfactory compounds in terrestrial vertebrates.
Taken together, our study provides compelling evidence that individual OSNs in the peripheral olfactory system of the goldfish detects and encodes different biologically relevant odorants and serves as the neural basis of natural food and pheromone recognition in this species. Food recognition appears especially complex and associated with a variety of OSNs with overlapping molecular receptive ranges. In contrast, both priming and releaser sex pheromone information appears to be encoded by highly specialized subsets of ciliated forskolin-sensitive OSNs whose function resembles labeled lines. Identification of sex pheromone receptors and their neural connectome should eventually shed additional light on the regulatory mechanism governing vertebrate chemical communication. Although we tested all known key representatives of all odorant classes and their components in the most comprehensive sampling of natural odors yet conducted, our inability to stimulate about a third of all spontaneously active OSNs, indicates that many odorants have not yet been identified in the goldfish, and likely other fishes. In addition, the insensitivity of about a third of all OSNs to either forskolin or imipramine, suggests the presence of unknown signaling pathway in fish OSNs, perhaps associated with crypt cells and kappe neurons. Screening of other biologically relevant odorants and receptor deorphanization will be required to fully elucidate the molecular basis of how the goldfish olfactory system discriminates and encodes natural odors, and then how this might pertain to other species.
Supplementary data
Supplementary data can be found at Chemical Senses online.
Funding
This study was supported by a grant from the National Institutes of Health - DC03792 (to P.W.S.) and the Okazaki ORION Project, JSPS KAKENHI Grant Number 16H04226 and Takeda Science Foundation (to K.S.).
Conflict of Interest
The authors do not have any conflicts of interest.
Acknowledgements
We thank Dr. John Caprio and Shane Rolen for their advice on single-unit recording and John for his helpful editorial advice. We also thank Dr. Andrej Blejec for help with statistical analysis and Asuka Sumita for help with single-unit data analysis.
References
- Ahuja G, Bozorg Nia S, Zapilko V, Shiriagin V, Kowatschew D, Oka Y, Korsching SI. 2014. Kappe neurons, a novel population of olfactory sensory neurons. Sci Rep. 4:4037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bazáes A, Schmachtenberg O. 2012. Odorant tuning of olfactory crypt cells from juvenile and adult rainbow trout. J Exp Biol. 215:1740–1748. [DOI] [PubMed] [Google Scholar]
- Behrens M, Frank O, Rawel H, Ahuja G, Potting C, Hofmann T, Meyerhof W, Korsching S. 2014. ORA1, a zebrafish olfactory receptor ancestral to all mammalian V1R genes, recognizes 4-hydroxyphenylacetic acid, a putative reproductive pheromone. J Biol Chem. 289:19778–19788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blejec A. 2000. Alternative statistical method for detecting changes in spontaneous activity of single olfactory receptor cells. Pflugers Arch. 439:R161–R162. [DOI] [PubMed] [Google Scholar]
- Buchinger TJ, Li W, Johnson NS. 2014. Bile salts as semiochemicals in fish. Chem Senses. 39:647–654. [DOI] [PubMed] [Google Scholar]
- Cadiou H, Molle G. 2003. Adenophostin A and imipramine are two activators of the olfactory inositol 1,4,5-trisphosphate-gated channel in fish olfatory cilia. Eur Biophys J. 32:106–112. [DOI] [PubMed] [Google Scholar]
- Caprio J, Byrd RP Jr. 1984. Electrophysiological evidence for acidic, basic, and neutral amino acid olfactory receptor sites in the catfish. J Gen Physiol. 84:403–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen TY, Takeuchi H, Kurahashi T. 2006. Odorant inhibition of the olfactory cyclic nucleotide-gated channel with a native molecular assembly. J Gen Physiol. 128:365–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czesnik D, Kuduz J, Schild D, Manzini I. 2006. ATP activates both receptor and sustentacular supporting cells in the olfactory epithelium of Xenopus laevis tadpoles. Eur J Neurosci. 23:119–128. [DOI] [PubMed] [Google Scholar]
- Delay RJ, Dionne VE. 2002. Two second messengers mediate amino acid responses in olfactory sensory neurons of the salamander, Necturus maculosus. Chem Senses. 27:673–680. [DOI] [PubMed] [Google Scholar]
- DeMaria S, Berke AP, Van Name E, Heravian A, Ferreira T, Ngai J. 2013. Role of a ubiquitously expressed receptor in the vertebrate olfactory system. J Neurosci. 33:15235–15247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derby CD, Sorensen PW. 2008. Neural processing, perception, and behavioral responses to natural chemical stimuli by fish and crustaceans. J Chem Ecol. 34:898–914. [DOI] [PubMed] [Google Scholar]
- Dulka JG, Stacey NE, Sorensen PW, Van Der Kraak GJ. 1987. A sex steroid pheromone synchronizes male-female spawning readiness in goldfish. Nature. 325: 251–253. [Google Scholar]
- Elsaesser R, Montani G, Tirindelli R, Paysan J. 2005. Phosphatidyl-inositide signalling proteins in a novel class of sensory cells in the mammalian olfactory epithelium. Eur J Neurosci. 21:2692–2700. [DOI] [PubMed] [Google Scholar]
- Fukuda H, Nishida A, Saito H, Shimizu M, Yamawaki S. 1994. Imipramine stimulates phospholipase C activity in rat brain. Neurochem Int. 25:567–571. [DOI] [PubMed] [Google Scholar]
- Gesteland RC. 1975. Techniques for investigating single unit activity in the vertebrate olfactory epithelium. In: Moulton DG, Turk A, Johnson W Jr, editors. Methods in olfactory research. London: Academic Press; p. 269–322. [Google Scholar]
- Ghosal R, Sorensen PW. 2016. Male-typical courtship, spawning behavior, and olfactory sensitivity are induced to different extents by androgens in the goldfish suggesting they are controlled by different neuroendocrine mechanisms. Gen Comp Endocrinol. 232:160–173. [DOI] [PubMed] [Google Scholar]
- Hansen A, Rolen SH, Anderson K, Morita Y, Caprio J, Finger TE. 2003. Correlation between olfactory receptor cell type and function in the channel catfish. J Neurosci. 23:9328–9339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen A, Zippel HP, Sorensen PW, Caprio J. 1999. Ultrastructure of the olfactory epithelium in intact, axotomized, and bulbectomized goldfish, Carassius auratus. Microsc Res Tech. 45:325–338. [DOI] [PubMed] [Google Scholar]
- Hara TJ. 2006. Feeding behaviour in some teleosts is triggered by single amino acids primarily through olfaction. J Fish Biol. 68:810–825. [Google Scholar]
- Hussain A, Saraiva LR, Ferrero DM, Ahuja G, Krishna VS, Liberles SD, Korsching SI. 2013. High-affinity olfactory receptor for the death-associated odor cadaverine. Proc Natl Acad Sci USA. 110:19579–19584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang J, Caprio J. 1995. In vivo responses of single olfactory receptor neurons in the channel catfish, Ictalurus punctatus. J Neurophysiol. 73:172–177. [DOI] [PubMed] [Google Scholar]
- Kaupp UB, Seifert R. 2002. Cyclic nucleotide-gated ion channels. Physiol Rev. 82:769–824. [DOI] [PubMed] [Google Scholar]
- Keller A, Zhuang H, Chi Q, Vosshall LB, Matsunami H. 2007. Genetic variation in a human odorant receptor alters odour perception. Nature. 449:468–472. [DOI] [PubMed] [Google Scholar]
- Kim K, Sato K, Shibuya M, Zeiger DM, Butcher RA, Ragains JR, Clardy J, Touhara K, Sengupta P. 2009. Two chemoreceptors mediate developmental effects of dauer pheromone in C. elegans. Science. 326:994–998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koide T, Miyasaka N, Morimoto K, Asakawa K, Urasaki A, Kawakami K, Yoshihara Y. 2009. Olfactory neural circuitry for attraction to amino acids revealed by transposon-mediated gene trap approach in zebrafish. Proc Natl Acad Sci USA. 106:9884–9889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laframboise AJ, Ren X, Chang S, Dubuc R, Zielinski BS. 2007. Olfactory sensory neurons in the sea lamprey display polymorphisms. Neurosci Lett. 414:277–281. [DOI] [PubMed] [Google Scholar]
- Levesque HM, Scaffidi D, Polkinghorne CN, Sorensen PW. 2011. A multi-component species identifying pheromone in the goldfish. J Chem Ecol. 37:219–227. [DOI] [PubMed] [Google Scholar]
- Li W, Sorensen PW, Gallaher DD. 1995. The olfactory system of migratory adult sea lamprey (Petromyzon marinus) is specifically and acutely sensitive to unique bile acids released by conspecific larvae. J Gen Physiol. 105:569–587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Q, Tachie-Baffour Y, Liu Z, Baldwin MW, Kruse AC, Liberles SD. 2015. Non-classical amine recognition evolved in a large clade of olfactory receptors. Elife. 4:e10441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim H, Sorenen PW. 2011. Polar metabolites synergize the activity of prostaglandin F2α in a species-specific hormonal sex pheromone released by ovulated common carp. J Chem Ecol. 37:695–704. [DOI] [PubMed] [Google Scholar]
- Lucas P, Ukhanov K, Leinders-Zufall T, Zufall F. 2003. A diacylglycerol-gated cation channel in vomeronasal neuron dendrites is impaired in TRPC2 mutant mice: mechanism of pheromone transduction. Neuron. 40:551–561. [DOI] [PubMed] [Google Scholar]
- McGann JP, Pírez N, Gainey MA, Muratore C, Elias AS, Wachowiak M. 2005. Odorant representations are modulated by intra- but not interglomerular presynaptic inhibition of olfactory sensory neurons. Neuron. 48:1039–1053. [DOI] [PubMed] [Google Scholar]
- Nikonov AA, Caprio J. 2007. Highly specific olfactory receptor neurons for types of amino acids in the channel catfish. J Neurophysiol. 98:1909–1918. [DOI] [PubMed] [Google Scholar]
- Nodari F, Hsu FF, Fu X, Holekamp TF, Kao LF, Turk J, Holy TE. 2008. Sulfated steroids as natural ligands of mouse pheromone-sensing neurons. J Neurosci. 28:6407–6418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka Y, Omura M, Kataoka H, Touhara K. 2004. Olfactory receptor antagonism between odorants. EMBO J. 23:120–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka Y, Saraiva LR, Korsching SI. 2012. Crypt neurons express a single V1R-related ora gene. Chem Senses. 37:219–227. [DOI] [PubMed] [Google Scholar]
- Poling KR, Fraser EJ, Sorensen PW. 2001. The three steroidal components of the goldfish preovulatory pheromone signal evoke different behaviors in males. Comp Biochem Physiol B Biochem Mol Biol. 129:645–651. [DOI] [PubMed] [Google Scholar]
- Rolen SH, Sorensen PW, Mattson D, Caprio J. 2003. Polyamines as olfactory stimuli in the goldfish Carassius auratus. J Exp Biol. 206:1683–1696. [DOI] [PubMed] [Google Scholar]
- Sansone A, Hassenklöver T, Offner T, Fu X, Holy TE, Manzini I. 2015. Dual processing of sulfated steroids in the olfactory system of an anuran amphibian. Front Cell Neurosci. 9:373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato Y, Miyasaka N, Yoshihara Y. 2005. Mutually exclusive glomerular innervation by two distinct types of olfactory sensory neurons revealed in transgenic zebrafish. J Neurosci. 25:4889–4897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato K, Suzuki N. 2001. Whole-cell response characteristics of ciliated and microvillous olfactory receptor neurons to amino acids, pheromone candidates and urine in rainbow trout. Chem Senses. 26:1145–1156. [DOI] [PubMed] [Google Scholar]
- Schmachtenberg O, Bacigalupo J. 2004. Olfactory transduction in ciliated receptor neurons of the Cabinza grunt, Isacia conceptionis (Teleostei: Haemulidae). Eur J Neurosci. 20:3378–3386. [DOI] [PubMed] [Google Scholar]
- Sorensen PW, Hara TJ, Stacey NE. 1987. Extreme olfactory sensitivity of mature and gonadally-regressed goldfish to 17α20β-dihydroxy-4-pregnen-3-one, a potent steroidal pheromone. J Comp Physiol A. 160:305–313. [Google Scholar]
- Sorensen PW, Hara TJ, Stacey NE, Goetz FW. 1988. F prostaglandins function as potent olfactory stimulants that comprise the postovulatory female sex pheromone in goldfish. Biol Reprod. 39:1039–1050. [DOI] [PubMed] [Google Scholar]
- Sorensen PW, Hara TJ, Stacey NE, Dulka JG. 1990. Extreme olfactory specificity of male goldfish to the preovulatory pheromone 17α, 20β-dihydroxy-4-pregnen-3-one. J Comp Physiol A. 166:373–385. [Google Scholar]
- Sorensen PW, Pinillos M, Scott AP. 2005. Sexually mature male goldfish release large quantities of androstenedione into the water where it functions as a pheromone. Gen Comp Endocrinol. 140:164–175. [DOI] [PubMed] [Google Scholar]
- Sorensen PW, Sato K. 2005. Second messenger systems mediating sex pheromone and amino acid sensitivity in goldfish olfactory receptor neurons. Chem Senses. 30Suppl 1:i315–i316. [DOI] [PubMed] [Google Scholar]
- Sorensen PW, Scott AP, Stacey NE, Bowdin L. 1995. Sulfated 17,20 beta-dihydroxy-4-pregnen-3-one functions as a potent and specific olfactory stimulant with pheromonal actions in the goldfish. Gen Comp Endocrinol. 100:128–142. [DOI] [PubMed] [Google Scholar]
- Sorensen PW, Stacey NE, Chamberlain KJ. 1989. Differing behavioral and endocrinological effects of two female sex pheromones on male goldfish. Horm Behav. 23:317–332. [DOI] [PubMed] [Google Scholar]
- Speca DJ, Lin DM, Sorensen PW, Isacoff EY, Ngai J, Dittman AH. 1999. Functional identification of a goldfish odorant receptor. Neuron. 23:487–498. [DOI] [PubMed] [Google Scholar]
- Touhara K, Vosshall LB. 2009. Sensing odorants and pheromones with chemosensory receptors. Annu Rev Physiol. 71:307–332. [DOI] [PubMed] [Google Scholar]
- Tricas TC, Kajiura SM, Summers AP. 2009. Response of the hammerhead shark olfactory epithelium to amino acid stimuli. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 195:947–954. [DOI] [PubMed] [Google Scholar]
- Valentincic T, Kralj J, Stenovec M, Koce A, Caprio J. 2000. The behavioral detection of binary mixtures of amino acids and their individual components by catfish. J Exp Biol. 203:3307–3317. [DOI] [PubMed] [Google Scholar]
- Vielma A, Ardiles A, Delgado L, Schmachtenberg O. 2008. The elusive crypt olfactory receptor neuron: evidence for its stimulation by amino acids and cAMP pathway agonists. J Exp Biol. 211:2417–2422. [DOI] [PubMed] [Google Scholar]
- Wakisaka N, Miyasaka N, Koide T, Masuda M, Hiraki-Kajiyama T, Yoshihara Y. 2017. An Adenosine receptor for Olfaction in fish. Curr Biol. 27:1437–1447.e4. [DOI] [PubMed] [Google Scholar]
- Yabuki Y, Koide T, Miyasaka N, Wakisaka N, Masuda M, Ohkura M, Nakai J, Tsuge K, Tsuchiya S, Sugimoto Y et al. 2016. Olfactory receptor for prostaglandin F2α mediates male fish courtship behavior. Nat Neurosci. 19:897–904. [DOI] [PubMed] [Google Scholar]
- Yamamoto Y, Hino H, Ueda H. 2010. Olfactory imprinting of amino acids in lacustrine sockeye salmon. PLoS One. 5:e8633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yambe H, Kitamura S, Kamio M, Yamada M, Matsunaga S, Fusetani N, Yamazaki F. 2006. L-Kynurenine, an amino acid identified as a sex pheromone in the urine of ovulated female masu salmon. Proc Natl Acad Sci USA. 103:15370–15374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshihara Y. 2014. Zebrafish olfactory system. In: Mori K, editor. The olfactory system: from odor molecules to motivational behaviors. Tokyo: Springer; p. 71–96. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.