In this review, we will provide pediatricians with an educational toolbox to improve their diagnosis and evaluation of antibiotic allergy in children on the basis of current literature.
Abstract
The overlabeling of pediatric antibiotic allergy represents a huge burden in society. Given that up to 10% of the US population is labeled as penicillin allergic, it can be estimated that at least 5 million children in this country are labeled with penicillin allergy. We now understand that most of the cutaneous symptoms that are interpreted as drug allergy are likely viral induced or due to a drug–virus interaction, and they usually do not represent a long-lasting, drug-specific, adaptive immune response to the antibiotic that a child received. Because most antibiotic allergy labels acquired in childhood are carried into adulthood, the overlabeling of antibiotic allergy is a liability that leads to unnecessary long-term health care risks, costs, and antibiotic resistance. Fortunately, awareness of this growing burden is increasing and leading to more emphasis on antibiotic allergy delabeling strategies in the adult population. There is growing literature that is used to support the safe and efficacious use of tools such as skin testing and drug challenge to evaluate and manage children with antibiotic allergy labels. In addition, there is an increasing understanding of antibiotic reactivity within classes and side-chain reactions. In summary, a better overall understanding of the current tools available for the diagnosis and management of adverse drug reactions is likely to change how pediatric primary care providers evaluate and treat patients with such diagnoses and prevent the unnecessary avoidance of antibiotics, particularly penicillins.
Antibiotic allergy labeling leads to significant individual and public health consequences. Unlike vaccination, there is no systematic approach to address antibiotic allergy during routine office visits, and allergy labels persist into adulthood. Antibiotic allergy usually comes to light when treatment is imminent, and physicians often find themselves choosing more expensive and time-intensive procedures, such as desensitization, or using higher-cost alternative antibiotics with potentially more side effects. These measures may satisfy the immediate need for treatment but do not address the primary problem.
Antibiotic allergy labels are often acquired because of rashes reported by parents, and most children never undergo an allergy evaluation to address the diagnosis. In a recent study, 75% of children diagnosed with penicillin allergy were labeled before their third birthday.1 The prevalent carriage of these childhood allergy labels into adulthood perpetuates the use of alternative antibiotics, which are often more expensive, less effective, and contribute to an increase in antibiotic-resistant bacteria.2–4 However, studies reveal that when children are tested and/or undergo drug challenging, >90% are able to tolerate the antibiotic.5–7 Unfortunately, even when the diagnosis of drug allergy is excluded by such procedures, not only parents but many providers are still resistant to drug allergy delabeling.8,9
Prescription costs are 30% to 40% higher in patients with suspected penicillin allergy.10 If just half of the children who visit a physician for acute otitis media annually were to receive amoxicillin instead of cefdinir (a common alternative prescribed for treating patients with a history of penicillin allergy), the estimated annual savings would exceed $34 million.11 Researchers in a recent cohort study were able to match 51 582 subjects with and without penicillin allergy at hospital admission. It confirmed that patients who require alternative drugs, such as fluoroquinolones, clindamycin, and vancomycin, because of a penicillin allergy have 23.4% more Clostridium difficile, 14.1% more methicillin-resistant Staphylococcus aureus, and 30.1% more vancomycin-resistant enterococci infections compared with controls.4 The accumulation of adverse drug labels is more limiting in populations that are susceptible to frequent infections, such as cystic fibrosis, particularly when drug resistance develops.12–14
In this state-of-the-art review, we aim to provide clinicians with an evidence-based toolbox for the diagnostic workup of children with antibiotic allergy. The ultimate goal is to improve patient and provider education to address and reconcile allergy labels early to prevent children from carrying these potentially false antibiotic allergy labels into adulthood.
Epidemiology of Antibiotic Allergy
Epidemiologic studies in children with antibiotic allergy are scarce and fraught with inconsistencies. It is challenging to accurately assess the incidence of true allergy in the United States, particularly because the term “allergic” has been used frequently without definition, which allows one to conclude that nonallergic reactions were included in many epidemiologic studies.15 Antibiotics are responsible for up to one-third of reported adverse drug reactions (ADRs), and ∼35% of ADRs seen in the emergency department are reported as allergic.16–18 In addition, as many as 10% of parents report that their children are allergic to 1 or more medications.19–23 Researchers in 1 large study in the United States evaluating 411 543 adult and pediatric medical records found that the overall incidence of self-reported antibiotic allergy was as high as 15.3%.24 Despite the high number of reported cases, <10% of cases are confirmed to be allergic after testing and/or challenge,25–31 indicating that true allergy to antibiotics is rare and overdiagnosed.32,33
The drug allergy box is the major place in most medical records where ADRs are documented, often without reference to the immunologic basis of the reaction. This label does not typically discriminate between pharmacological effects, side effects, temporally associated observations, or true drug allergies, making the drug allergy box subject to overestimation of true allergy risk.34
This overestimation has been demonstrated in multiple studies in which the initial drug allergy label was based on questionnaires and/or the opinions of experienced physicians, but subsequent drug challenges were used to disprove the majority of them.35–37 In a large study of consecutive patients with or without a history of penicillin allergy, the rate of positive skin testing results in those who were labeled as penicillin allergic with vague histories was 1.7%, which is the same as in those without a history of penicillin allergy.23
In 1 study, Erkoçoğlu et al33 found that of the 10 096 questionnaires returned, in 792 (7.87%), parents reported a history of drug allergy, but only 117 (1.1%) of these were consistent with an immunoglobulin E (IgE)–mediated reaction by history. There were 101 children for whom further workup was done, and only 7 (0.11%) of those with suggestive histories had positive testing results.
Penicillin allergy, which is the most common reported drug allergy, has a prevalence rate of 5% to 10% in community populations of adults and children and is as high as 20% in those linked to ongoing medical care.18,38–41 An allergy to amoxicillin is the most common drug allergy in children.32 Although the epidemiology in the United States is currently unknown, hypersensitivity to clavulanic acid appears prevalent in southern Europe and has been described in children.26,42,43
Of ADRs in pediatric patients, 23% are reported to be caused by non–β-lactam antibiotics. Although rarely confirmed in pediatric studies, macrolides are reported to cause drug allergy, mostly benign cutaneous reactions.44 Among macrolides, the 15-membered ring azalide (azithromycin) may be more allergenic than clarithromycin and without consistent cross-reactivity with clarithromycin, erythromycin, and other 14-membered ring traditional macrolides.45,46 Sulfonamide antimicrobial agents infrequently cause IgE-mediated symptoms in children but are known to cause a wide array of T-cell–mediated symptoms, most commonly mild cutaneous exanthems, but more severe reactions such as drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, fixed drug eruption, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug-induced liver disease, and cytopenia have been reported as well, especially in patients with HIV.12,47–50 Allergic reactions to quinolones, vancomycin, aminoglycosides, and tetracyclines are rare except in certain patient populations with chronic diseases, such as cystic fibrosis, likely because of repeated exposure to antibiotics.44,51
Classification of ADRs
ADRs are clinically classified as type A and type B reactions. Type A (on-target) reactions are dose dependent and pharmacologically predictable on the basis of the accentuation of the drug’s on-target therapeutic effect. They comprise >80% of ADRs, including drug–drug interactions, and may be subject to genetic variation.52,53 Common examples include bleeding with warfarin or tremor associated with albuterol. Antibiotic-associated type A reactions in pediatric practice include antibiotic-associated diarrhea because of an on-target effect dependent on dose and duration secondary to the alteration of the bacterial microbiome or dose-dependent adrenal suppression associated with azole antifungal agents.47,54
Type B (off-target) reactions are not predictable on the basis of the known target of therapeutic effect but are often dose dependent and subject to host genetic variation. A minority are dose independent, including antibody- and IgE-mediated reactions.52,53,55,56 The off-target mechanism of non–IgE-mediated mast cell activation for many drugs (such as opiates, neuromuscular blocking agents, fluoroquinolones, and potentially vancomycin) entails the dose-dependent activation of a specific mas-related G protein–coupled receptor on mast cells.57,58 Red man syndrome because of non–IgE-mediated mast cell activation secondary to vancomycin administration is a clinical example of a type B reaction in children.
Drug allergies comprise <15% of all ADRs; however, patients and physicians often erroneously refer to all ADRs as allergic.19,51,56 The Gell and Coombs mechanism–based system (Table 1) classifies ADRs into 4 types (I, II, III, and IV) and, more recently, subtypes IVa, IVb, IVc, and IVd on the basis of their immune mediators.57,59–63
TABLE 1.
Classification, Clinical Features, and Management of Classic Hypersensitivity Reactions to Antibiotics
| Gell and Coombs Classification | Immune Mechanism | Clinical Features | Timing |
|---|---|---|---|
| Type I: immediate-type hypersensitivity | Mast cell and/or basophil mediator release directed by drug interaction and/or cross-linking of drug-specific IgE bound to these cells | Anaphylaxis, urticaria, angioedema, gastrointestinal, respiratory, cardiovascular, and neurologic symptoms | Immediate: <1 h after drug exposure |
| Type II: cytotoxic or antibody dependent, hypersensitivity | Natural killer cells and macrophages kill IgG- or IgM-coated cells that are directed against the drug or drug metabolite on the patient’s cells | Drug-induced hemolytic anemia, thrombocytopenia | 1–2 wk after exposurea |
| Type III: immune complex–mediated hypersensitivity | Antibody (IgG>IgM) binds to soluble antigen (often a drug or drug metabolite), forming a circulating immune complex | Serum sickness, vasculitis | 1–2 wk after exposurea |
| Type IV: delayed-type hypersensitivities | An antigen-presenting cell expressing HLA bound to a peptide interacts with a T-cell receptor in the presence of a drug or drug metaboliteb,61 | Benign, delayed skin rashes | Nonimmediate: differs according to the specific phenotype (Table 3) but generally 24 h to 1 wk after first exposure and can be quicker (h) on rechallenge exposure |
| Type IVa | Allergic contact dermatitis, maculopapular exanthema, FDE, EM (typically infection, not drug) | ||
| Type IVb | More severe cutaneous skin rashes | ||
| Type IVc | Acute generalized exanthematous pustulosis, DRESS syndrome, SJS and/or TEN, generalized-bullous FDE | ||
| Type IVd |
EM, erythema multiforme; FDE, fixed drug eruption; HLA, human leukocyte antigen; IgG, immunoglobulin G; IgM, immunoglobulin M.
May be sooner if preformed antibodies.
Multiple models have been proposed including hapten/prohapten model, pharmacological interaction (p-i) model and altered peptide repertoire model. In the hapten/prohapten model, a drug/drug metabolite covalently bound to larger protein undergoes intracellular processing to generate modified peptides that are incorporated onto HLA proteins for presentation to T cells.61
Other proposed classification systems are used to establish biomarkers depending on the patient phenotype and endotype. Phenotype is determined on the basis of timing (immediate or delayed onset) and associated symptoms. Endotypes include IgE-mediated reactions, T-cell–mediated reactions, pharmacologic interactions, and genetic predisposition. Biomarkers include in vivo, in vitro and/or ex vivo testing, mediators, and genetic markers (such as human leukocyte antigen typing).64
Cross-Reactivity
Cross-reactivity is a clinically relevant topic because clinicians are often faced with alternative antibiotic choices when a patient develops a rash during an antibiotic course. Most studies have been focused on β-lactam cross-reactivity; however, other antibiotics, such as macrolides and quinolones, are also known to cross-react within their group.46,65–68
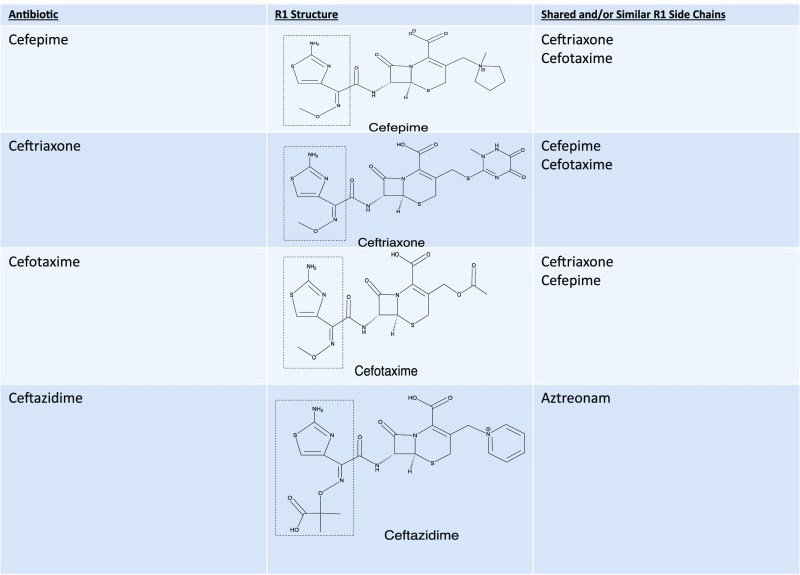
β-lactams (penicillins, cephalosporins, carbapenems, monobactams, oxacephems, and β-lactamase inhibitors) are a group of drugs that share a 4-membered β-lactam ring. The β-lactam ring opens spontaneously in vivo into benzylpenicillin without active metabolism, resulting in the formation of the major determinant, benzylpenicilloyl. Additionally, benzylpenicillin (the native penicillin drug) and the minor determinants (penicilloate and penilloate) can be immunogenic. There is a side chain that arises from the β-lactam ring (R1). Cephalosporins additionally have a 6-membered ring and another side chain (R2).47,69
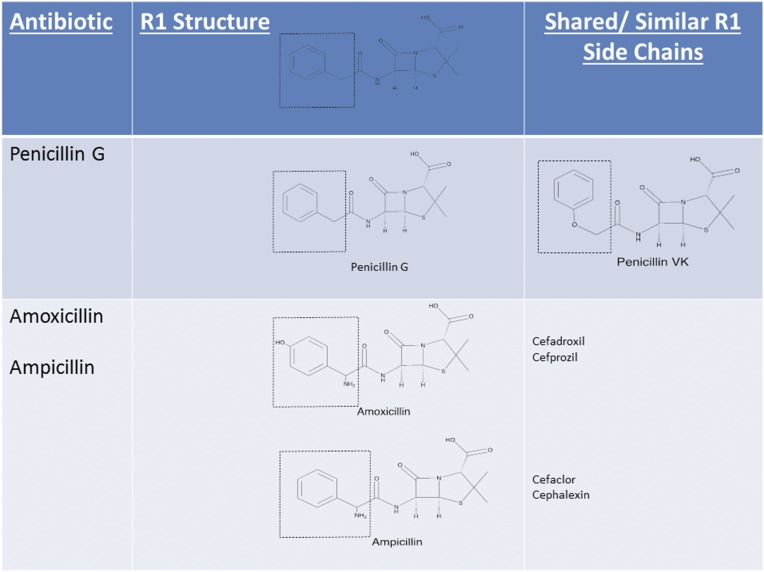
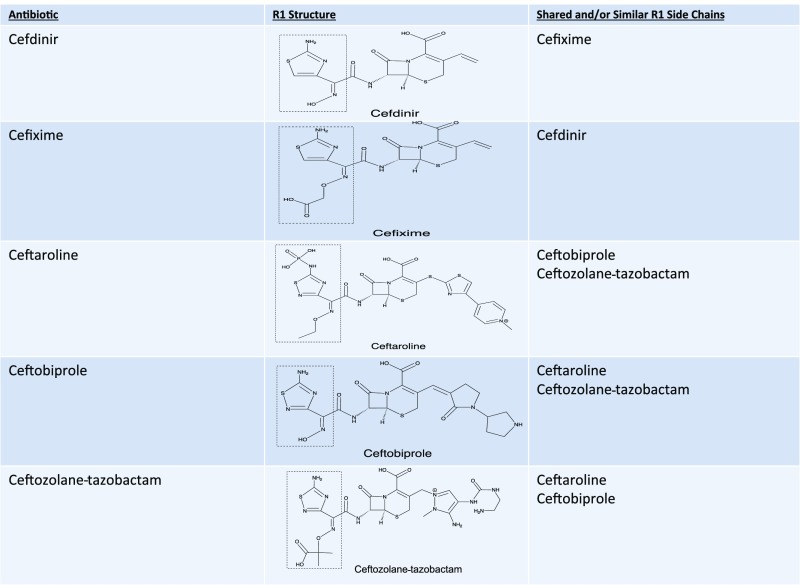
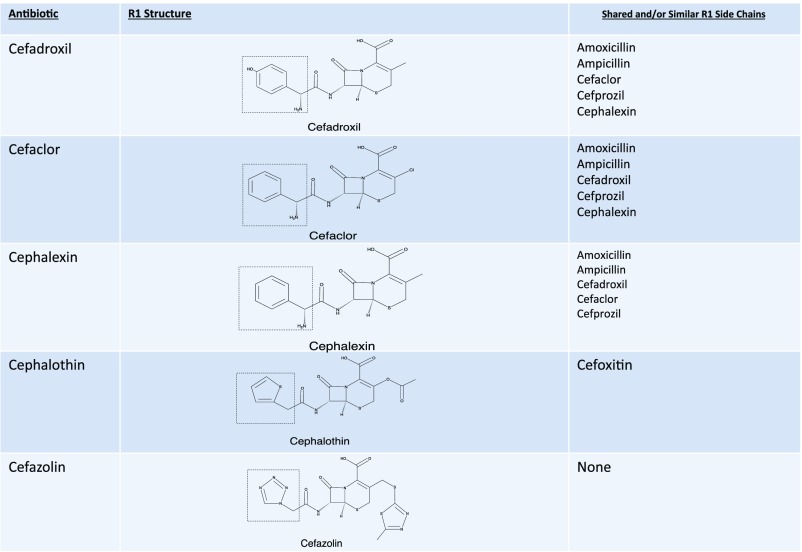
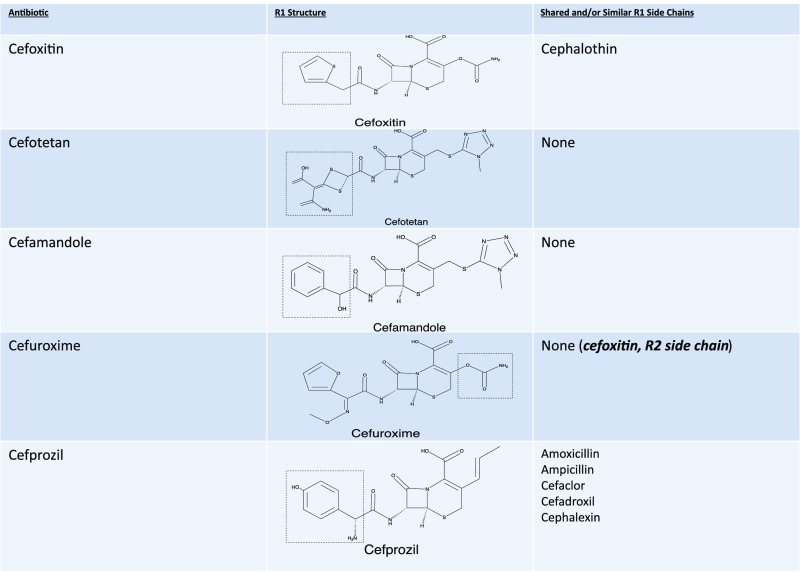
The β-lactam ring was initially believed to be the predominant cause of cross-reactivity between cephalosporins and penicillins.69 However, the R1 side chain and, less frequently, the R2 side chain have been demonstrated to contribute significantly to cross-reactivity within the penicillin class itself and also between penicillins and cephalosporins in vitro, in vivo through skin testing and challenge, and clinically through selective tolerance or reactivity (Figs 1–5).63,69–72 Approximately 2% of patients with penicillin allergy would be expected to react to a cephalosporin; however, this number may exceed 30% when administered cephalosporins have identical R1 side chains.47,71–73 The 2% figure is based on the statistic that only 10% of individuals with a history of penicillin allergy are expected to test positive to penicillins, and of those, ∼2% will develop a reaction to cephalosporins. The mechanism behind this is unknown but may be secondary to coexisting allergy. In the Practice Parameter on drug allergy, the Joint Task Force recommends skin testing individuals with penicillin allergy to penicillin and its major and minor determinants (although the percentage is low) before administering a cephalosporin because fatal anaphylaxis has been reported.47 When skin testing is not available, there is clinical evidence to support choosing a cephalosporin with a different R1 side chain to reduce reaction risk.69–76
FIGURE 1.
Penicillin-cephalosporin cross-reactivity based on side-chain similarity. Penicillin G, benzylpenicillin; penicillin VK, phenoxymethylpenicillin potassium. Approximately 2% of penicillin allergic patients would be expected to react to a cephalosporin, however this number may exceed 30% when administered cephalosporins with identical R1 side chains. Cefditoren and cefpodoxime are oral cephalosporins with the same side chain.
FIGURE 5.
Third-, fourth-, and fifth-generation cephalosporin cross-reactivity (continued). Approximately 2% of penicillin allergic patients would be expected to react to a cephalosporin, however this number may exceed 30% when administered cephalosporins with identical R1 side chains. Cefditoren and cefpodoxime are oral cephalosporins with the same side chain.
FIGURE 2.
First-generation cephalosporin cross-reactivity. Approximately 2% of penicillin allergic patients would be expected to react to a cephalosporin, however this number may exceed 30% when administered cephalosporins with identical R1 side chains. Cefditoren and cefpodoxime are oral cephalosporins with the same side chain.
FIGURE 3.
Second-generation cephalosporin cross-reactivity. Approximately 2% of penicillin allergic patients would be expected to react to a cephalosporin, however this number may exceed 30% when administered cephalosporins with identical R1 side chains. Cefditoren and cefpodoxime are oral cephalosporins with the same side chain.
FIGURE 4.
Third-, fourth-, and fifth-generation cephalosporin cross-reactivity. Approximately 2% of penicillin allergic patients would be expected to react to a cephalosporin, however this number may exceed 30% when administered cephalosporins with identical R1 side chains. Cefditoren and cefpodoxime are oral cephalosporins with the same side chain.
Cefazolin, which is a common cause of perioperative anaphylaxis, does not share R1 or R2 groups with other cephalosporins, and accumulating evidence supports that patients with IgE-mediated reactions to cefazolin may tolerate other cephalosporins and penicillins.77–80 Researchers in some studies have reported on subjects who are allergic >1 β-lactam drug, although their sensitivity cannot be explained by side-chain similarity.70,75 Ideally, testing to cephalosporins should be pursued along with penicillin reagents in children with a strong history of penicillin allergy or positive penicillin skin testing results.47,70 The rate of cross-reactivity between penicillins and both carbapenems and aztreonam in children has been determined to be <1%, so it is safe for a patient with penicillin allergy to receive these drugs.75,81–84 The tolerability of carbapenems in individuals with penicillin allergy has also recently been demonstrated in delayed-type drug allergy.83
Clinical Phenotyping
An accurate and detailed history helps identify the nature of the adverse reaction and the most appropriate management.85 Urticaria is the most common clinical symptom of a drug reaction, drug–viral interaction, as well as non–IgE-mediated mast cell activation.15,32,86 When it occurs within an hour of exposure to a drug, particularly if it’s reproducible on drug challenge, it can represent an immediate reaction potentially associated with anaphylaxis.6,33,87 Pseudoallergic reactions, also known as “anaphylactoid” reactions or non–IgE-mediated mast cell activation, can resemble type I hypersensitivity reactions (Table 2).47,57,88,89 Delayed or nonimmediate reactions range from benign to severe and can be classified by timing and clinical features (Table 3).47,88,89 Delayed urticaria that occurs several hours to days after drug exposure is often non-IgE mediated. The underlying cause of cutaneous drug reactions during viral infections may involve a viral-induced polyclonal activation of lymphocytes, an enhancement of cellular immunity, or changes in drug metabolism.90
TABLE 2.
Immediate Reactions: Clinical Features and Management
| Type of Reaction | Timing of Reaction | Symptoms of Reaction | Possible Laboratories in Acute Setting | Differential Diagnosis | Commonly Involved Antibiotics | Testing | Management |
|---|---|---|---|---|---|---|---|
| Non–IgE-mediated reaction, mast cell activation (anaphylactoid, pseudoallergic) | Immediate or during infusion | Urticaria | Elevated mast cell tryptase | IgE-mediated reaction | Quinolones | Serum total tryptase leveal | Pretreatment |
| Can occur on first course of drug | Flushing | Anaphylaxis | Vancomycin (Red man syndrome) | Serum histamineb | Slow infusion speed (with Red man syndrome) | ||
| Angioedema (occasionally) | Antihistamines | ||||||
| Hypotension | Desensitize in some cases | ||||||
| Mild fever | |||||||
| Muscle pain in severe instances | |||||||
| IgE-mediated, anaphylactic-producing, immediate reaction | 1–6 h after administration of the drug | Angioedema | Elevated mast cell tryptase | Non–IgE-mediated mast cell activation | β-lactams (ie, aminopenicillins) | Acute testing as above | Avoid drugs and/or cross-reacting drugs |
| Urticaria | Food allergy | Rarely non-β-lactams (macrolides) | Allergy referral testing and possible tests | Desensitization | |||
| Rhinitis | Venom allergy | Skin prick testing | |||||
| Conjunctivitis | Idiopathic urticaria and anaphylaxis | Serum-specific IgE | |||||
| Hypotension | Drug provocation testing | ||||||
| Laryngeal edema | |||||||
| Gastrointestinal symptoms | |||||||
| Bronchospasm | |||||||
| Cardiovascular shock and hypotension |
Obtain serum total tryptase 15 minutes to 3 hours after reaction.
Obtain 2-15 minutes after reaction.
TABLE 3.
Nonimmediate Reactions: Clinical Features and Management
| Type of Reaction | Timing | Cutaneous Symptoms | Systemic Symptoms | Possible Laboratories in Acute Setting | Differential Diagnosis | Commonly Involved Antibiotics | Testing | Management |
|---|---|---|---|---|---|---|---|---|
| Delayed drug reaction | 7–14 d | Maculopapular exanthema | Low-grade fever | Eosinophilia (mild) | Viral exanthem | Aminopenicillins | Delayed intradermal testing | Avoidance drugs and/or cross-reactive drugs |
| Urticaria | Pruritus | IgE-mediated drug allergy | Sulfonamides | Drug provocation testing | ||||
| Early DRESS syndrome or SJS and/or TEN | Clindamycin | |||||||
| SJS and/or TEN | 4–28 d | Painful erythematous macules with purpuric or dusky centers | Prodrome | Anemia | EM | Sulfonamides | LTT and/or ELISPOT | Avoidance drug and/or drug class |
| Superficial sloughing | High fever | Lymphopenia | Bullous pemphigoid | Minocycline (used for acne) | HLA screening | |||
| Mucositis in ≥2 surfaces | Malaise | Staphylococcal scalded skin syndrome | β-lactams | |||||
| Pneumonitis (occasionally) | FDE | |||||||
| DRESS syndrome | ||||||||
| DRESS syndrome | 2–8 wk | Morbilliform eruption >50% BSA | Fever | Eosinophilia | Viral or drug exanthem | Vancomycin | Patch testing | Avoidance of drugs and/or cross-reacting drugs |
| Nonerosive mucositis | Lymphadenopathy | Atypical lymphocytes | Early SJS and/or TEN | β-lactams | LTT and/or ELISPOT | |||
| ≥2 facial edema, infiltrated lesions, scaling, and purpura | Hepatitis | Severe eczema or psoriasis | Tetracyclines | HLA screening | ||||
| Renal impairment | Rifampin | |||||||
| Antituberculosis drugs | ||||||||
| Sulfonamides and/or sulfonesa,91 | ||||||||
| FDE | 1–14 db | 1 or more well-demarcated, round, dusky-to-violaceous macules or plaques | None | None | EM | Sulfonamides | Patch testing | Drug provocation testingc |
| Blistering may occur | Bullous pemphigoid | Tetracyclines | Avoidance of drug and/or cross-reactive drug | |||||
| Mucosal predilection but limited mucositis | SJS and/or TEN | β lactams | ||||||
| Postinflammatory hyperpigmentation | Quinolones | |||||||
| Fluconazole | ||||||||
| Acute generalized exanthematous pustulosis | 24–48 h | Dozens to hundreds of pustules on erythematous background | High fever | Neutrophilia | Pustular psoriasis | Aminopenicillins | Patch testing | Avoidance of drugs and/or cross-reactive drugs |
| Flexural accentuation | Edema | Eosinophilia | Bullous impetigo | Clindamycin | ||||
| Subcorneal pustular dermatosis | Sulfonamides | |||||||
| DRESS syndrome | Quinolones | |||||||
| Serum-sicknesslike reaction | 1–2 wk | Pruritic urticarial or serpiginous plaques (usually at injection site) | High fever | Neutropenia | Vasculitis | β lactams (especially cefaclor) | None | Avoidance of specific drugd |
| Malaise | Mild eosinophilia | Rheumatic fever | Macrolides | |||||
| Polyarthralgia or polyarthritis | Mild proteinuria | Other drug reactions | Sulfonamides |
BSA, Body Surface Area; EM, erythema multiforme; FDE, fixed drug eruption; HLA, human leukocyte antigen.
Sulfonamides include trimethoprim-sulfamethazole and sulfones include dapsone. There is no cross reactivity between sulfa antimicrobials and non-antibiotic sulfonamides (such as acetazolamide, bumetanide, celecoxib, chlorothiazide, diazoxide, dorzolamide, furosemide, glyburide, hydrochlorothiazide, indapamide, metolazone, sumatriptan, torsemide and zonisamide).
Rapid recurrence on drug re-exposure.
Contraindicated in generalized FDE.
Controversial. Some sources recommend avoiding drug and drug class while others (ie cefaclor) just recommend avoiding culprit drug.
In children, rashes during antibiotic treatment can be difficult to assess because they often result from a variety of triggers that are common in the pediatric population. Maculopapular rashes have been observed in 3% to 7% of children who are on ampicillin.92 In fact, researchers in recent studies have attempted to reveal the underlying viral causes of rashes by performing viral diagnostic studies with simultaneous allergy workup.31,35 In a 2011 study by Caubet et al,31 of 88 children with a history of nonimmediate drug allergy, only 6 had positive challenge results, and 5 of these were confirmed to have an underlying infection known to cause rash. Delayed-onset urticarial or maculopapular rashes are also frequently observed in children who are treated with β-lactam, with an estimated frequency of 1% to 5% experiencing rashes per prescription.39
Available Guidelines and Consensus Statements
Most information on pediatric drug allergy is tailored for specialists and extrapolated from guidelines for adults. Specialists refer to both American and European guidelines and consensus statements.44,47,89,93–98
Diagnosis and Treatment of Antibiotic Allergy in Children
An accurate history that combines all subjective and objective information available is key in the diagnostic evaluation of children with antibiotic allergy.38,44,55,56,89,99–101
The accurate documentation of medications taken at the time of the reaction is crucial because the presence of cofactors or coprescribed drugs may change the onset or progression of a reaction47,89 and could also be causal. The mechanism could be a true allergic reaction associated with immunologic memory or an off-target effect, such as non–IgE-mediated mast cell activation exacerbated by multiple inciting drugs administered concurrently (eg, opiates and vancomycin).52 Previous exposure to the same antibiotic or structurally similar antibiotics is important in determining the immunologic mechanism. The physician should gather particular signs and symptoms as precisely as possible and consider, on the basis of these symptoms, whether the reaction should be considered severe, benign, immediate, or nonimmediate. The provider should determine if treatment was required for the reaction as well as the response to treatment.32,55,102,103
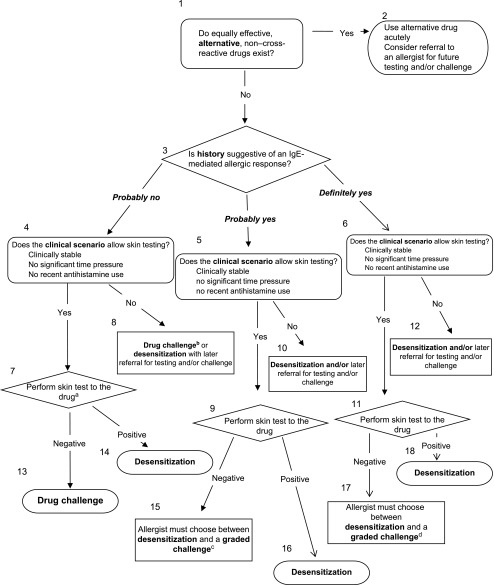
If a provider is suspicious that an IgE-mediated allergic reaction occurred (Table 1), workup should be considered (Fig 6). Immediate reactions that typically occur within 1 hour of exposure to oral drugs or within 15 to 20 minutes for parenteral drugs should prompt referral to an allergist for further workup. In reality, the immunologic mechanisms of the reactions may be accelerated in nature (1–72 hours after dosing), and these overlap considerably in time or may not be clearly differentiated by the medical history, which is why if there is any suspicion of drug allergy, referral should be considered.80
FIGURE 6.
Stepwise approach to the evaluation and treatment of patients with type I IgE-mediated drug allergy (see Table 1). This approach cannot be used in the case of severe reactions, including SJS, TEN, DRESS syndrome, nephritis, hepatitis, and hemolysis. Adapted from Turvey SE, Cronin B, Arnold AD, Dioun AF. Antibiotic desensitization for the allergic patient: 5 years of experience and practice. Ann Allergy Asthma Immunol. 2004;92(4): p. 430 and Dioun AF. Management of multiple drug allergies in children. Curr Allergy Asthma Rep. 2012;12(1): p. 81.
See Figs 1–5 for cross reactivity. aPursuing skin testing is dependent on negative predictive value of testing and reagent dependent. Consider going straight to challenge if reaction was mild and inconsistent with IgE mediated drug allergy. b Consider graded challenge for milder reactions or desensitization for more severe reactions. cConsider graded challenge for milder reactions. dConsider desensitization regardless of skin test, particularly if index reaction was severe.
In Vivo Testing: Immediate Reactions
When performed by trained professionals, skin prick and intradermal skin testing are safe and efficacious procedures to aid in the diagnosis of immediate reactions to antibiotics, particularly in β-lactams.26,102,104,105
If possible, skin testing should be delayed for 2 to 3 weeks after an inciting reaction because of the potential depletion of mediators, which may temporarily lead to false-negative results.55 Most guidelines suggest waiting 4 to 6 weeks after the complete resolution of all clinical symptoms and signs of a suspected delayed hypersensitivity reaction before testing.44,47,89,106
Standardized antibiotic skin testing protocols exist for penicillin, although the only labeled skin testing reagent currently available in the United States is penicillin major determinant (Pre-Pen). There are also published data regarding nonirritating concentrations and test specificity to other antibiotics that allergists may choose to use before challenging.107,108
In Vivo Testing: Nonimmediate Reactions
Some researchers suggest delayed intradermal testing reads at 24 to 48 hours or patch testing with reads at 48 hours, 72 hours, 96 hours, and 1 week for nonimmediate reactions. However, sensitivity has been reported to be <50% in many studies and is likely to be dependent on the specific antibiotic and the pretest clinical probability.35,98
Feasibility of Skin Testing in Children
Skin prick testing is performed easily in children of any age, even in infancy.109 Although intradermal skin testing is less well tolerated because of the discomfort from injections, when indicated, it is possible to perform this test in young children with adequate preparation. In routine clinical practice, the risk of resensitization to a drug after a negative testing result is extremely uncommon.27,110
In Vitro and/or Ex Vivo Testing
Serum-mature tryptase (β tryptase), a mast cell mediator, can be used as an adjunct for suspected IgE-mediated reactions during which peak levels will be reached within 30 minutes to 2 hours of the onset of symptoms and typically return to normal within 24 hours. Non–IgE-mediated mast cell activation can produce a positive mast cell tryptase in ∼10% of cases.111 Although a normal tryptase level does not rule out anaphylaxis and/or IgE-mediated drug allergy, an elevated level compared with baseline can be helpful in the diagnosis of such reactions.47
Drug-Specific Tests
There are currently no validated in vitro or ex vivo tests that have adequate sensitivity and/or specificity to be widely applied in clinical practice. The in vitro–specific IgE testing that is commercially available for penicillin has a low sensitivity, ranging from 0% to 25%, and in some studies, false-positive testing results have been found.112,113 Researchers in multiple studies have reported false-negatives when testing was done close to the acute reaction and when testing is delayed beyond 6 months despite a lack of tolerance.114–116
There are other ex vivo and in vitro assays that are potentially clinically useful but are currently only used in research settings. The basophil activation test is an in vitro test for antibiotics using flow cytometry to detect basophil surface (CD63 and/or CD203c) and intracellular (phospho-p38 mitogen-activated protein kinases) markers.85,112,117–120 Lymphocyte transformation testing (LTT) is used to measure T-cell proliferation to a drug in vitro and is possibly useful for the diagnosis of drug-induced type IV (delayed) hypersensitivities, although both false-negative and false-positive testing results have been reported.95,121–123
Enzyme-linked immunospot (ELISPOT) assays are used to analyze low-frequency, antigen-specific, cytokine-producing cells in the peripheral blood of patients with a type IV hypersensitivity reaction after stimulation with pharmacological drug concentrations. ELISPOT can be used to measure cytokine responses, including interleukin (IL)-13, interferon γ, IL-10, IL-5, granzyme B, granulysin, and tumor necrosis factor α. ELISPOT has been reported to have better sensitivity than LTT in detecting drug-specific T-cell responses and a specificity ranging from 95% to 100%.112,124–131 Intracellular cytokine staining is used to measure the production of targeted cytokines by T cells in response to drug stimulation.122
Tests such as the ELISPOT assay and intracellular cytokine staining potentially could be of great utility, particularly because many children are on multiple antibiotics at the time they develop a severe reaction such as DRESS syndrome or SJS and/or TEN, when delayed intradermal skin testing or patch testing may be riskier or lack sensitivity. The extent to which these tests remain positive over time is not known, and differences may exist between drugs and classes of drugs.132
Drug Challenge
The drug challenge, also referred to as a graded challenge or drug provocation test, is considered to be a gold standard for drug allergy diagnosis. It can be administered as a single dose or in multiple doses. Drug challenge strategies to reduce the risk of severe reaction, when the pretest probability of an IgE-mediated reaction is high and the negative predictive value of skin testing is low, include a 2-step graded challenge in which 10% of a weight-based dose is given and then the remaining 90% is given after a specified observation time of 30 to 60 minutes with an additional observation time of 60 minutes. For instance, if a penicillin testing result is negative, then it is reasonable to proceed with a single-dose challenge because the negative predictive value of penicillin testing has been well established at 97% to 99%. For all other antibiotics, the predictive values have not been determined in large population studies; therefore, if a testing result is negative, a graded challenge is the safest way to proceed.23,31,47 If >3 doses are administered, a graded challenge can be used to downregulate mast cells and runs the risk of desensitizing patients.47
The majority of studies in which researchers evaluate drug challenges reveal that they are safe and well tolerated in the pediatric population. Researchers in several studies report that if reactions were to occur as a result of drug challenge, they are similar or less severe than the original reaction.* Researchers support the use of allergy testing and challenge in special populations, such as oncologic and immune-compromised patients, and have comparable positive and negative predictive values to the general population.133,134
Drug Desensitization
Drug desensitization is described as a temporary induction of drug tolerance by the administration of incremental doses of the drug.47 It is important to realize that drug desensitization is a therapeutic measure for the safe administration of a drug to a patient who has either a proven or is highly likely to have a drug allergy as opposed to drug challenge, which is a diagnostic procedure performed in cases of low probability of drug allergy.23,47 In addition, drug challenge and desensitization should be done in a monitored setting and are contraindicated in patients with severe non–IgE-mediated reactions, such as SJS, TEN, DRESS syndrome, interstitial nephritis, hepatitis, or hemolytic anemia.47
Drug desensitization procedures vary depending on several factors, such as the drug itself, the route of administration, and the patient’s reaction and its severity. The starting dose is typically in fractions of a milligram, doubling every 15 to 30 minutes until a cumulative therapeutic dose has been achieved. The goal of desensitization is to render the individual nonreactive to the drug as long as he or she is receiving treatment with the drug. Once the drug is no longer present in the serum, the individual loses the tolerance to the drug, and repeat desensitization is usually indicated if there is a delay of >2 half-lives.47,55
There are few studies on antibiotic desensitization in children, so most of our knowledge of this procedure is extrapolated from adult studies.135,136 A high efficacy and safety rate has been reported in both β-lactam and non–β-lactam drugs.47,50,137–140 Case reports of successful desensitization in children to other β-lactam drugs (including meropenem, cefotaxime, ceftriaxone, and ceftazidime) as well as non–β-lactam drugs (such as macrolides or sulfa antimicrobial agents) have been reported.50,138–140
Desensitization is most effective in IgE-mediated reactions; however, there is evidence for its use in some non–IgE-mediated reactions, particularly with sulfa antimicrobial agents.141–144 Desensitization is also frequently employed and effective in the setting of suspected non–IgE-mediated reactions in children with cystic fibrosis.14,135,145
Antibiotic Stewardship Programs
Patients requiring frequent medical care or hospitalization are at high risk to be labeled as allergic to multiple antibiotics.3,146 In addition, labels frequently stick despite negative testing results and challenge.9 In fact, it is estimated that 36% to 49% of patients with negative penicillin testing results may have a persistence or redocumentation of their allergy despite proven negative testing results.147–149
Current evidence reveals that an interactive and electronically accessible drug allergy box in a medical record that is regularly reconciled improves the management of patients labeled with drug allergies. In 1 study, the electronic medical record was used proactively to identify patients for testing, which was then performed in the inpatient unit by a trained pharmacist. Researchers in this study reduced the use of second-line antibiotics during hospitalization and discharge.150 Researchers in another small pilot study prevented redocumentation with several interventions, including an electronic alert notifying providers when a penicillin allergy is added back for a patient with documented negative testing results.148
Targeting prospective antibiotic allergy management in adults has led to a positive impact on antibiotic use and appropriateness.151 Blumenthal et al152 used a quasi-experimental design to measure the impact of different strategies over discreet time periods among an internal medicine service and showed that inpatient skin testing to β-lactam drugs directed by an allergist and the use of previously adopted, computerized guidelines resulted in an increase in penicillin and cephalosporin use.153 Trubiano et al146 measured the impact of an integrated and responsive outpatient antibiotic allergy testing and antimicrobial stewardship program at 2 Australian centers and determined that after testing, appropriate antibiotics were more likely to be prescribed. To date studies using antibiotic allergy management as an antimicrobial stewardship tool have focused on adults. Programs in which researchers prioritize pediatric populations, in which the majority of antibiotic allergy labels are first realized, are warranted.
Future Directions
Educating the public and health care providers about the differences in ADRs and drug allergies could reduce overdiagnosis and promote appropriate referrals and procedures, such as skin testing and drug challenging, that will prevent the overlabeling of drug allergy. Education directed at community providers to make a more accurate diagnosis of drug allergy could potentially improve global health. An interactive electronic medical record that is regularly reconciled could help improve the management of patients with drug allergy. In the future, accessibility to preventive genetic testing and more sensitive diagnostic tests for both immediate and delayed antibiotic allergy could be invaluable. These tests would ideally aid in preventing reactions, unraveling the diagnostic complexity of multiple antibiotic allergies, or determining the underlying cause of a reaction and whether it is the drug, a virus, or a drug–virus interaction. If sufficiently sensitive and/or specific and widely available, such testing would also greatly reduce the risks in drug challenges, decrease the use of the more time-intensive and costly procedure of desensitization, and increase the use of first-line antibiotics.
Acknowledgments
Thank you to Zohaib Lakhani for contributing to Figs 1–5. Thank you also to Dr Melissa Fuller for reviewing the article and steering the content toward general pediatric providers.
Glossary
- ADR
adverse drug reaction
- DRESS
drug reaction with eosinophilia and systemic symptoms
- ELISPOT
enzyme-linked immunospot
- IgE
immunoglobulin E
- IL
interleukin
- LTT
lymphocyte transformation testing
- R1
side chain that arises from the β-lactam ring
- R2
side chain that arises from the dihydrothiazine ring on cephalosporins
- SJS
Stevens-Johnson syndrome
- TEN
toxic epidermal necrolysis
Footnotes
Dr Norton conceptualized and outlined the article, drafted the initial manuscript, and reviewed and revised the manuscript; Ms Konvinse aided in the conception and outline of the article, drafted sections of the article, and reviewed and revised the manuscript; Drs Phillips and Broyles aided in conceptualizing and outlining the article, and critically reviewed and revised the manuscript; and all authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
FINANCIAL DISCLOSURE: Dr Phillips has received consultancy fees from Xcovery and BioCryst; Drs Norton, Konvinse, and Broyles have indicated they have no financial relationships relevant to this article to disclose.
FUNDING: Ms Konvinse is supported by the National Institutes of Health (1P50GM115305, 2T32GM7347, and F30 AI131780). Dr Phillips is supported by 1P50GM115305-01, 1R01AI103348-01, 1P30AI110527-01A1, 5T32AI007474-20, and 1 R13AR71267-01 from Australia’s National Health and Medical Research Council and the Australian Centre for HIV and Hepatitis Virology Research. Funded by the National Institutes of Health (NIH).
POTENTIAL CONFLICT OF INTEREST: The authors have indicated they have no potential conflicts of interest to disclose.
References
- 1.Vyles D, Chiu A, Simpson P, Nimmer M, Adams J, Brousseau DC. Parent-reported penicillin allergy symptoms in the pediatric emergency department. Acad Pediatr. 2017;17(3):251–255 [DOI] [PubMed] [Google Scholar]
- 2.Chiriac AM; Demoly P . Drug Allergy. In: Leung DY, Sampson HA, Bonilla FA, Akdis CA, Szefler SJ, eds. Pediatric Allergy: Principles and Practice, 3rd ed . Edinburgh: Elsevier; 2016:498–504 [Google Scholar]
- 3.MacFadden DR, LaDelfa A, Leen J, et al. Impact of reported beta-lactam allergy on inpatient outcomes: a multicenter prospective cohort study. Clin Infect Dis. 2016;63(7):904–910 [DOI] [PubMed] [Google Scholar]
- 4.Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. J Allergy Clin Immunol. 2014;133(3):790–796 [DOI] [PubMed] [Google Scholar]
- 5.Vyles D, Adams J, Chiu A, Simpson P, Nimmer M, Brousseau DC. Allergy testing in children with low-risk penicillin allergy symptoms. Pediatrics. 2017;140(2):e20170471. [DOI] [PubMed] [Google Scholar]
- 6.Rebelo Gomes E, Fonseca J, Araujo L, Demoly P. Drug allergy claims in children: from self-reporting to confirmed diagnosis. Clin Exp Allergy. 2008;38(1):191–198 [DOI] [PubMed] [Google Scholar]
- 7.Vezir E, Erkocoglu M, Civelek E, et al. The evaluation of drug provocation tests in pediatric allergy clinic: a single center experience. Allergy Asthma Proc. 2014;35(2):156–162 [DOI] [PubMed] [Google Scholar]
- 8.Picard M, Paradis L, Nguyen M, Bégin P, Paradis J, Des Roches A. Outpatient penicillin use after negative skin testing and drug challenge in a pediatric population. Allergy Asthma Proc. 2012;33(2):160–164 [DOI] [PubMed] [Google Scholar]
- 9.Gerace KS, Phillips E. Penicillin allergy label persists despite negative testing. J Allergy Clin Immunol Pract. 2015;3(5):815–816 [DOI] [PubMed] [Google Scholar]
- 10.MacLaughlin EJ, Saseen JJ, Malone DC. Costs of beta-lactam allergies: selection and costs of antibiotics for patients with a reported beta-lactam allergy. Arch Fam Med. 2000;9(8):722–726 [DOI] [PubMed] [Google Scholar]
- 11.Coker TR, Chan LS, Newberry SJ, et al. Diagnosis, microbial epidemiology, and antibiotic treatment of acute otitis media in children: a systematic review. JAMA. 2010;304(19):2161–2169 [DOI] [PubMed] [Google Scholar]
- 12.Phillips E, Mallal S. Drug hypersensitivity in HIV. Curr Opin Allergy Clin Immunol. 2007;7(4):324–330 [DOI] [PubMed] [Google Scholar]
- 13.Caimmi S, Sanfiorenzo C, Caimmi D, Bousquet PJ, Chiron R, Demoly P. Comprehensive allergy work-up is mandatory in cystic fibrosis patients who report a history suggestive of drug allergy to beta-lactam antibiotics. Clin Transl Allergy. 2012;2(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Petroni DH, Aitken ML, Ham E, et al. Approach to the evaluation of adverse antibiotic reactions in patients with cystic fibrosis. Ann Allergy Asthma Immunol. 2016;117(4):378–381 [DOI] [PubMed] [Google Scholar]
- 15.Pichichero ME. A review of evidence supporting the American Academy of Pediatrics recommendation for prescribing cephalosporin antibiotics for penicillin-allergic patients. Pediatrics. 2005;115(4):1048–1057 [DOI] [PubMed] [Google Scholar]
- 16.Cohen AL, Budnitz DS, Weidenbach KN, et al. National surveillance of emergency department visits for outpatient adverse drug events in children and adolescents. J Pediatr. 2008;152(3):416–421 [DOI] [PubMed] [Google Scholar]
- 17.Star K, Norén GN, Nordin K, Edwards IR. Suspected adverse drug reactions reported for children worldwide: an exploratory study using VigiBase. Drug Saf. 2011;34(5):415–428 [DOI] [PubMed] [Google Scholar]
- 18.Le J, Nguyen T, Law AV, Hodding J. Adverse drug reactions among children over a 10-year period. Pediatrics. 2006;118(2):555–562 [DOI] [PubMed] [Google Scholar]
- 19.Gomes ER, Demoly P. Epidemiology of hypersensitivity drug reactions. Curr Opin Allergy Clin Immunol. 2005;5(4):309–316 [DOI] [PubMed] [Google Scholar]
- 20.Chan DS, Callahan CW, Moreno C. Multidisciplinary education and management program for children with asthma. Am J Health Syst Pharm. 2001;58(15):1413–1417 [DOI] [PubMed] [Google Scholar]
- 21.Kerr JR. Penicillin allergy: a study of incidence as reported by patients. Br J Clin Pract. 1994;48(1):5–7 [PubMed] [Google Scholar]
- 22.Smyth RM, Gargon E, Kirkham J, et al. Adverse drug reactions in children—a systematic review. PLoS One. 2012;7(3):e24061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gadde J, Spence M, Wheeler B, Adkinson NF Jr. Clinical experience with penicillin skin testing in a large inner-city STD clinic. JAMA. 1993;270(20):2456–2463 [PubMed] [Google Scholar]
- 24.Macy E, Poon K-Y T. Self-reported antibiotic allergy incidence and prevalence: age and sex effects. Am J Med. 2009;122(8):778.e1–778.e7 [DOI] [PubMed] [Google Scholar]
- 25.Vezir E, Erkocoglu M, Civelek E, et al. The evaluation of drug provocation tests in pediatric allergy clinic: a single center experience. Allergy Asthma Proc. 2014;35(2):156–162 [DOI] [PubMed] [Google Scholar]
- 26.Ponvert C, Perrin Y, Bados-Albiero A, et al. Allergy to beta-lactam antibiotics in children: results of a 20-year study based on clinical history, skin and challenge tests. Pediatr Allergy Immunol. 2011;22(4):411–418 [DOI] [PubMed] [Google Scholar]
- 27.Iglesias-Souto J, González R, Poza P, Sanchez-Machín I, Matheu V. Evaluating the usefulness of retesting for beta-lactam allergy in children. Pediatr Infect Dis J. 2012;31(10):1091–1093 [DOI] [PubMed] [Google Scholar]
- 28.Mattheij M, de Vries E. A suspicion of antibiotic allergy in children is often incorrect. J Allergy Clin Immunol. 2012;129(2):583–; author reply 583–584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Seitz CS, Bröcker EB, Trautmann A. Diagnosis of drug hypersensitivity in children and adolescents: discrepancy between physician-based assessment and results of testing. Pediatr Allergy Immunol. 2011;22(4):405–410 [DOI] [PubMed] [Google Scholar]
- 30.Messaad D, Sahla H, Benahmed S, Godard P, Bousquet J, Demoly P. Drug provocation tests in patients with a history suggesting an immediate drug hypersensitivity reaction. Ann Intern Med. 2004;140(12):1001–1006 [DOI] [PubMed] [Google Scholar]
- 31.Caubet JC, Kaiser L, Lemaître B, Fellay B, Gervaix A, Eigenmann PA. The role of penicillin in benign skin rashes in childhood: a prospective study based on drug rechallenge. J Allergy Clin Immunol. 2011;127(1):218–222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Caubet JC, Eigenmann PA. Managing possible antibiotic allergy in children. Curr Opin Infect Dis. 2012;25(3):279–285 [DOI] [PubMed] [Google Scholar]
- 33.Erkoçoğlu M, Kaya A, Civelek E, et al. Prevalence of confirmed immediate type drug hypersensitivity reactions among school children. Pediatr Allergy Immunol. 2013;24(2):160–167 [DOI] [PubMed] [Google Scholar]
- 34.Trubiano JA, Adkinson NF, Phillips EJ. Penicillin allergy is not necessarily forever. JAMA. 2017;318(1):82–83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Atanaskovic-Markovic M, Gaeta F, Medjo B, et al. Non-immediate hypersensitivity reactions to beta-lactam antibiotics in children - our 10-year experience in allergy work-up. Pediatr Allergy Immunol. 2016;27(5):533–538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Arikoglu T, Aslan G, Batmaz SB, Eskandari G, Helvaci I, Kuyucu S. Diagnostic evaluation and risk factors for drug allergies in children: from clinical history to skin and challenge tests. Int J Clin Pharm. 2015;37(4):583–591 [DOI] [PubMed] [Google Scholar]
- 37.Chambel M, Martins P, Silva I, Palma-Carlos S, Romeira AM, Leiria Pinto P. Drug provocation tests to betalactam antibiotics: experience in a paediatric setting. Allergol Immunopathol (Madr). 2010;38(6):300–306 [DOI] [PubMed] [Google Scholar]
- 38.Romano A, Caubet J-C. Antibiotic allergies in children and adults: from clinical symptoms to skin testing diagnosis. J Allergy Clin Immunol Pract. 2014;2(1):3–12 [DOI] [PubMed] [Google Scholar]
- 39.Ibia EO, Schwartz RH, Wiedermann BL. Antibiotic rashes in children: a survey in a private practice setting. Arch Dermatol. 2000;136(7):849–854 [DOI] [PubMed] [Google Scholar]
- 40.Solensky R. Allergy to β-lactam antibiotics. J Allergy Clin Immunol. 2012;130(6):1442– 1442.e5 [DOI] [PubMed] [Google Scholar]
- 41.Pawankar R, Canonica GW, Holgate ST, Lockey RF. WAO White Book on Allergy 2011-2012 Executive Summary. Milwaukee, WI: World Allergy Association; 2011 [Google Scholar]
- 42.Torres MJ, Ariza A, Mayorga C, et al. Clavulanic acid can be the component in amoxicillin-clavulanic acid responsible for immediate hypersensitivity reactions. J Allergy Clin Immunol. 2010;125(2):502–505.e2 [DOI] [PubMed] [Google Scholar]
- 43.Zambonino MA, Corzo JL, Muñoz C, et al. Diagnostic evaluation of hypersensitivity reactions to beta-lactam antibiotics in a large population of children. Pediatr Allergy Immunol. 2014;25(1):80–87 [DOI] [PubMed] [Google Scholar]
- 44.Gomes ER, Brockow K, Kuyucu S, et al. ; ENDA/EAACI Drug Allergy Interest Group . Drug hypersensitivity in children: report from the pediatric task force of the EAACI Drug Allergy Interest Group. Allergy. 2016;71(2):149–161 [DOI] [PubMed] [Google Scholar]
- 45.Barni S, Butti D, Mori F, et al. Azithromycin is more allergenic than clarithromycin in children with suspected hypersensitivity reaction to macrolides. J Investig Allergol Clin Immunol. 2015;25(2):128–132 [PubMed] [Google Scholar]
- 46.Mori F, Pecorari L, Pantano S, et al. Azithromycin anaphylaxis in children. Int J Immunopathol Pharmacol. 2014;27(1):121–126 [DOI] [PubMed] [Google Scholar]
- 47.Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma, and Immunology; Joint Council of Allergy, Asthma, and Immunology . Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105(4):259–273 [DOI] [PubMed] [Google Scholar]
- 48.Cacoub P, Musette P, Descamps V, et al. The DRESS syndrome: a literature review. Am J Med. 2011;124(7):588–597 [DOI] [PubMed] [Google Scholar]
- 49.Roujeau JC, Stern RS. Severe adverse cutaneous reactions to drugs. N Engl J Med. 1994;331(19):1272–1285 [DOI] [PubMed] [Google Scholar]
- 50.Gómez-Traseira C, Boyano-Martínez T, Escosa-García L, Pedrosa M, Martín-Muñoz F, Quirce S. Trimethoprim-sulfamethoxazole (cotrimoxazole) desensitization in an HIV-infected 5-yr-old girl. Pediatr Allergy Immunol. 2015;26(3):287–289 [DOI] [PubMed] [Google Scholar]
- 51.Thong BY, Tan TC. Epidemiology and risk factors for drug allergy. Br J Clin Pharmacol. 2011;71(5):684–700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Garon SL, Pavlos RK, White KD, Brown NJ, Stone CA Jr, Phillips EJ. Pharmacogenomics of off-target adverse drug reactions. Br J Clin Pharmacol. 2017;83(9):1896–1911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.White KD, Chung WH, Hung SI, Mallal S, Phillips EJ. Evolving models of the immunopathogenesis of T cell-mediated drug allergy: the role of host, pathogens, and drug response. J Allergy Clin Immunol. 2015;136(2):219–234; quiz 235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Phillips EJ. Classifying ADRs—does dose matter? Br J Clin Pharmacol. 2016;81(1):10–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dioun AF. Management of multiple drug allergies in children. Curr Allergy Asthma Rep. 2012;12(1):79–84 [DOI] [PubMed] [Google Scholar]
- 56.Caubet JC, Eigenmann PA. Diagnostic issues in pediatric drug allergy. Curr Opin Allergy Clin Immunol. 2012;12(4):341–347 [DOI] [PubMed] [Google Scholar]
- 57.McNeil BD, Pundir P, Meeker S, et al. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature. 2015;519(7542):237–241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Azimi E, Reddy VB, Lerner EA. Brief communication: MRGPRX2, atopic dermatitis and red man syndrome. Itch (Phila). 2017;2(1):e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gell P, Coombs R, Lachmann P. Classification of allergic reactions for clinical hypersensitivity disease In: Gell PGH, Coombs RPA, eds. Clinical Aspects of Immunology. Oxford, United Kingdom: Blackwell Scientific Publications; 1968:575–579 [Google Scholar]
- 60.Pichler WJ. Delayed drug hypersensitivity reactions. Ann Intern Med. 2003;139(8):683–693 [DOI] [PubMed] [Google Scholar]
- 61.Pichler WJ, Beeler A, Keller M, et al. Pharmacological interaction of drugs with immune receptors: the p-i concept. Allergol Int. 2006;55(1):17–25 [DOI] [PubMed] [Google Scholar]
- 62.Aung AK, Haas DW, Hulgan T, Phillips EJ. Pharmacogenomics of antimicrobial agents. Pharmacogenomics. 2014;15(15):1903–1930 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Trubiano J, Phillips E. Antimicrobial stewardship’s new weapon? A review of antibiotic allergy and pathways to ‘de-labeling’. Curr Opin Infect Dis. 2013;26(6):526–537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Muraro A, Lemanske RF Jr, Castells M, et al. Precision medicine in allergic disease-food allergy, drug allergy, and anaphylaxis-PRACTALL document of the European Academy of Allergy and Clinical Immunology and the American Academy of Allergy, Asthma and Immunology. Allergy. 2017;72(7):1006–1021 [DOI] [PubMed] [Google Scholar]
- 65.Blanca-Lopez N, Perez-Alzate D, Ruano F, et al. Selective immediate responders to amoxicillin and clavulanic acid tolerate penicillin derivative administration after confirming the diagnosis. Allergy. 2015;70(8):1013–1019 [DOI] [PubMed] [Google Scholar]
- 66.Esposito S, Castellazzi L, Tagliabue C, Principi N. Allergy to antibiotics in children: an overestimated problem. Int J Antimicrob Agents. 2016;48(4):361–366 [DOI] [PubMed] [Google Scholar]
- 67.Blanca-López N, Ariza A, Doña I, et al. Hypersensitivity reactions to fluoroquinolones: analysis of the factors involved. Clin Exp Allergy. 2013;43(5):560–567 [DOI] [PubMed] [Google Scholar]
- 68.Venturini Díaz M, Lobera Labairu T, del Pozo Gil MD, Blasco Sarramián A, González Mahave I. In vivo diagnostic tests in adverse reactions to quinolones. J Investig Allergol Clin Immunol. 2007;17(6):393–398 [PubMed] [Google Scholar]
- 69.Pichichero ME, Zagursky R. Penicillin and cephalosporin allergy. Ann Allergy Asthma Immunol. 2014;112(5):404–412 [DOI] [PubMed] [Google Scholar]
- 70.Romano A, Gaeta F, Valluzzi RL, et al. IgE-mediated hypersensitivity to cephalosporins: cross-reactivity and tolerability of alternative cephalosporins. J Allergy Clin Immunol. 2015;136(3):685–691.e3 [DOI] [PubMed] [Google Scholar]
- 71.Romano A, Gaeta F, Arribas Poves MF, Valluzzi RL. Cross-reactivity among beta-lactams. Curr Allergy Asthma Rep. 2016;16(3):24. [DOI] [PubMed] [Google Scholar]
- 72.Audicana M, Bernaola G, Urrutia I, et al. Allergic reactions to betalactams: studies in a group of patients allergic to penicillin and evaluation of cross-reactivity with cephalosporin. Allergy. 1994;49(2):108–113 [DOI] [PubMed] [Google Scholar]
- 73.Novalbos A, Sastre J, Cuesta J, et al. Lack of allergic cross-reactivity to cephalosporins among patients allergic to penicillins. Clin Exp Allergy. 2001;31(3):438–443 [DOI] [PubMed] [Google Scholar]
- 74.Antunez C, Blanca-Lopez N, Torres MJ, et al. Immediate allergic reactions to cephalosporins: evaluation of cross-reactivity with a panel of penicillins and cephalosporins. J Allergy Clin Immunol. 2006;117(2):404–410 [DOI] [PubMed] [Google Scholar]
- 75.Romano A, Gaeta F, Valluzzi RL, Caruso C, Rumi G, Bousquet PJ. IgE-mediated hypersensitivity to cephalosporins: cross-reactivity and tolerability of penicillins, monobactams, and carbapenems. J Allergy Clin Immunol. 2010;126(5):994–999 [DOI] [PubMed] [Google Scholar]
- 76.Mill C, Primeau MN, Medoff E, et al. Assessing the diagnostic properties of a graded oral provocation challenge for the diagnosis of immediate and nonimmediate reactions to amoxicillin in children. JAMA Pediatr. 2016;170(6):e160033. [DOI] [PubMed] [Google Scholar]
- 77.Kuhlen JL Jr, Camargo CA Jr, Balekian DS, et al. Antibiotics are the most commonly identified cause of perioperative hypersensitivity reactions. J Allergy Clin Immunol Pract. 2016;4(4):697–704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gonzalez-Estrada A, Silvers SK, Klein A, Zell K, Wang XF, Lang DM. Epidemiology of anaphylaxis at a tertiary care center: a report of 730 cases. Ann Allergy Asthma Immunol. 2017;118(1):80–85 [DOI] [PubMed] [Google Scholar]
- 79.Gonzalez-Estrada A, Pien LC, Zell K, Wang XF, Lang DM. Antibiotics are an important identifiable cause of perioperative anaphylaxis in the United States. J Allergy Clin Immunol Pract. 2015;3(1):101–105.e1 [DOI] [PubMed] [Google Scholar]
- 80.Hierro Santurino B, Mateos Conde J, Cabero Morán MT, Mirón Canelo JA, Armentia Medina A. A predictive model for the diagnosis of allergic drug reactions according to the medical history. J Allergy Clin Immunol Pract. 2016;4(2):292–300.e3 [DOI] [PubMed] [Google Scholar]
- 81.Atanasković-Marković M, Gaeta F, Gavrović-Jankulović M, Velicković TC, Valluzzi RL, Romano A. Tolerability of imipenem in children with IgE-mediated hypersensitivity to penicillins. J Allergy Clin Immunol. 2009;124(1):167–169 [DOI] [PubMed] [Google Scholar]
- 82.Romano A, Gaeta F, Valluzzi RL, et al. Absence of cross-reactivity to carbapenems in patients with delayed hypersensitivity to penicillins. Allergy. 2013;68(12):1618–1621 [DOI] [PubMed] [Google Scholar]
- 83.Romano A, Gaeta F, Valluzzi RL, Maggioletti M, Caruso C, Quaratino D. Cross-reactivity and tolerability of aztreonam and cephalosporins in subjects with a T cell-mediated hypersensitivity to penicillins. J Allergy Clin Immunol. 2016;138(1):179–186 [DOI] [PubMed] [Google Scholar]
- 84.Atanasković-Marković M, Gaeta F, Medjo B, Viola M, Nestorović B, Romano A. Tolerability of meropenem in children with IgE-mediated hypersensitivity to penicillins. Allergy. 2008;63(2):237–240 [DOI] [PubMed] [Google Scholar]
- 85.Romano A, Torres MJ, Castells M, Sanz ML, Blanca M. Diagnosis and management of drug hypersensitivity reactions. J Allergy Clin Immunol. 2011;127(suppl 3):S67–S73 [DOI] [PubMed] [Google Scholar]
- 86.Rubio M, Bousquet PJ, Gomes E, Romano A, Demoly P. Results of drug hypersensitivity evaluations in a large group of children and adults. Clin Exp Allergy. 2012;42(1):123–130 [DOI] [PubMed] [Google Scholar]
- 87.Segal AR, Doherty KM, Leggott J, Zlotoff B. Cutaneous reactions to drugs in children. Pediatrics. 2007;120(4). Available at: www.pediatrics.org/cgi/content/full/120/4/e1082 [DOI] [PubMed] [Google Scholar]
- 88.Peter JG, Lehloenya R, Dlamini S, et al. Severe delayed cutaneous and systemic reactions to drugs: a global perspective on the science and art of current practice. J Allergy Clin Immunol Pract. 2017;5(3):547–563 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Demoly P, Adkinson NF, Brockow K, et al. International consensus on drug allergy. Allergy. 2014;69(4):420–437 [DOI] [PubMed] [Google Scholar]
- 90.Cohen AD, Friger M, Sarov B, Halevy S. Which intercurrent infections are associated with maculopapular cutaneous drug reactions? A case-control study. Int J Dermatol. 2001;40(1):41–44 [DOI] [PubMed] [Google Scholar]
- 91.Strom BL, Schinnar R, Apter AJ, et al. Absence of cross-reactivity between sulfonamide antibiotics and sulfonamide nonantibiotics. N Engl J Med. 2003;349(17):1628–1635 [DOI] [PubMed] [Google Scholar]
- 92.Bass JW, Crowley DM, Steele RW, Young FS, Harden LB. Adverse effects of orally administered ampicillin. J Pediatr. 1973;83(1):106–108 [DOI] [PubMed] [Google Scholar]
- 93.Kuyucu S, Mori F, Atanaskovic-Markovic M, et al. ; Pediatric Task Force of EAACI Drug Allergy Interest Group . Hypersensitivity reactions to non-betalactam antibiotics in children: an extensive review. Pediatr Allergy Immunol. 2014;25(6):534–543 [DOI] [PubMed] [Google Scholar]
- 94.Mayorga C, Celik G, Rouzaire P, et al. ; In Vitro Tests for Drug Allergy Task Force of EAACI Drug Interest Group . In vitro tests for drug hypersensitivity reactions: an ENDA/EAACI Drug Allergy Interest Group position paper. Allergy. 2016;71(8):1103–1134 [DOI] [PubMed] [Google Scholar]
- 95.Romano A, Blanca M, Torres MJ, et al. ; ENDA; EAACI . Diagnosis of nonimmediate reactions to β-lactam antibiotics. Allergy. 2004;59(11):1153–1160 [DOI] [PubMed] [Google Scholar]
- 96.Torres MJ, Blanca M, Fernandez J, et al. ; ENDA; EAACI Interest Group on Drug Hypersensitivity . Diagnosis of immediate allergic reactions to beta-lactam antibiotics. Allergy. 2003;58(10):961–972 [DOI] [PubMed] [Google Scholar]
- 97.Greenberger PA. Chapter 30: drug allergy. Allergy Asthma Proc. 2012;33(suppl 1):S103–S107 [DOI] [PubMed] [Google Scholar]
- 98.Mirakian R, Ewan PW, Durham SR, et al. BSACI guidelines for the management of drug allergy. Clin Exp Allergy. 2009;39(1):43–61 [DOI] [PubMed] [Google Scholar]
- 99.Demoly P, Kropf R, Bircher A, Pichler WJ. Drug hypersensitivity: questionnaire. EAACI interest group on drug hypersensitivity. Allergy. 1999;54(9):999–1003 [DOI] [PubMed] [Google Scholar]
- 100.Atanaskovic-Markovic M, Caubet JC. Management of drug hypersensitivity in the pediatric population [published online ahead of print July 28, 2016]. Expert Rev Clin Pharmacol. doi: [DOI] [PubMed] [Google Scholar]
- 101.Fernandez TD, Mayorga C, Ariza A, Corzo JL, Torres MJ. Allergic reactions to antibiotics in children. Curr Opin Allergy Clin Immunol. 2014;14(4):278–285 [DOI] [PubMed] [Google Scholar]
- 102.Pichichero ME, Pichichero DM. Diagnosis of penicillin, amoxicillin, and cephalosporin allergy: reliability of examination assessed by skin testing and oral challenge. J Pediatr. 1998;132(1):137–143 [DOI] [PubMed] [Google Scholar]
- 103.Khan DA, Solensky R. Drug allergy. J Allergy Clin Immunol. 2010;125(2, suppl 2):S126–S137 [DOI] [PubMed] [Google Scholar]
- 104.Atanasković-Marković M, Velicković TC, Gavrović-Jankulović M, Vucković O, Nestorović B. Immediate allergic reactions to cephalosporins and penicillins and their cross-reactivity in children. Pediatr Allergy Immunol. 2005;16(4):341–347 [DOI] [PubMed] [Google Scholar]
- 105.Ponvert C, Le Clainche L, de Blic J, Le Bourgeois M, Scheinmann P, Paupe J. Allergy to beta-lactam antibiotics in children. Pediatrics. 1999;104(4). Available at: www.pediatrics.org/cgi/content/full/104/4/e45 [DOI] [PubMed] [Google Scholar]
- 106.Blanca M, Romano A, Torres MJ, et al. Update on the evaluation of hypersensitivity reactions to betalactams. Allergy. 2009;64(2):183–193 [DOI] [PubMed] [Google Scholar]
- 107.Brockow K, Garvey LH, Aberer W, et al. ; ENDA/EAACI Drug Allergy Interest Group . Skin test concentrations for systemically administered drugs – an ENDA/EAACI Drug Allergy Interest Group position paper. Allergy. 2013;68(6):702–712 [DOI] [PubMed] [Google Scholar]
- 108.Empedrad R, Darter AL, Earl HS, Gruchalla RS. Nonirritating intradermal skin test concentrations for commonly prescribed antibiotics. J Allergy Clin Immunol. 2003;112(3):629–630 [DOI] [PubMed] [Google Scholar]
- 109.Cantani A. Diagnosis of pediatric allergy In: Cantani A, ed. Pediatric Allergy, Asthma and Immunology. 1st ed. Berlin, Germany: Springer-Verlag Berlin Heidelberg; 2008:421–440 [Google Scholar]
- 110.Ponvert C, Weilenmann C, Wassenberg J, et al. Allergy to betalactam antibiotics in children: a prospective follow-up study in retreated children after negative responses in skin and challenge tests. Allergy. 2007;62(1):42–46 [DOI] [PubMed] [Google Scholar]
- 111.Dong SW, Mertes PM, Petitpain N, Hasdenteufel F, Malinovsky JM; GERAP . Hypersensitivity reactions during anesthesia. Results from the ninth French survey (2005-2007). Minerva Anestesiol. 2012;78(8):868–878 [PubMed] [Google Scholar]
- 112.Haw WY, Polak ME, McGuire C, Erlewyn-Lajeunesse M, Ardern-Jones MR. In vitro rapid diagnostic tests for severe drug hypersensitivity reactions in children. Ann Allergy Asthma Immunol. 2016;117(1):61–66 [DOI] [PubMed] [Google Scholar]
- 113.Johansson SG, Adédoyin J, van Hage M, Grönneberg R, Nopp A. False-positive penicillin immunoassay: an unnoticed common problem. J Allergy Clin Immunol. 2013;132(1):235–237 [DOI] [PubMed] [Google Scholar]
- 114.Hjortlund J, Mortz CG, Stage TB, Skov PS, Dahl R, Bindslev-Jensen C. Positive serum specific IgE has a short half-life in patients with penicillin allergy and reversal does not always indicate tolerance. Clin Transl Allergy. 2014;4:34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Opstrup MS, Poulsen LK, Malling HJ, Jensen BM, Garvey LH. Dynamics of plasma levels of specific IgE in chlorhexidine allergic patients with and without accidental re-exposure. Clin Exp Allergy. 2016;46(8):1090–1098 [DOI] [PubMed] [Google Scholar]
- 116.Poulsen LK, Hummelshoj L. Triggers of IgE class switching and allergy development. Ann Med. 2007;39(6):440–456 [DOI] [PubMed] [Google Scholar]
- 117.Aranda A, Mayorga C, Ariza A, et al. In vitro evaluation of IgE-mediated hypersensitivity reactions to quinolones. Allergy. 2011;66(2):247–254 [DOI] [PubMed] [Google Scholar]
- 118.Gamboa PM, García-Avilés MC, Urrutia I, Antépara I, Esparza R, Sanz ML. Basophil activation and sulfidoleukotriene production in patients with immediate allergy to betalactam antibiotics and negative skin tests. J Investig Allergol Clin Immunol. 2004;14(4):278–283 [PubMed] [Google Scholar]
- 119.Hausmann OV, Gentinetta T, Bridts CH, Ebo DG. The basophil activation test in immediate-type drug allergy. Immunol Allergy Clin North Am. 2009;29(3):555–566 [DOI] [PubMed] [Google Scholar]
- 120.Torres MJ, Padial A, Mayorga C, et al. The diagnostic interpretation of basophil activation test in immediate allergic reactions to betalactams. Clin Exp Allergy. 2004;34(11):1768–1775 [DOI] [PubMed] [Google Scholar]
- 121.Porebski G, Pecaric-Petkovic T, Groux-Keller M, Bosak M, Kawabata TT, Pichler WJ. In vitro drug causality assessment in Stevens-Johnson syndrome - alternatives for lymphocyte transformation test. Clin Exp Allergy. 2013;43(9):1027–1037 [DOI] [PubMed] [Google Scholar]
- 122.Rive CM, Bourke J, Phillips EJ. Testing for drug hypersensitivity syndromes. Clin Biochem Rev. 2013;34(1):15–38 [PMC free article] [PubMed] [Google Scholar]
- 123.Trautmann A, Seitz CS, Stoevesandt J, Kerstan A. Aminopenicillin-associated exanthem: lymphocyte transformation testing revisited. Clin Exp Allergy. 2014;44(12):1531–1538 [DOI] [PubMed] [Google Scholar]
- 124.Bensaid B, Rozieres A, Nosbaum A, Nicolas JF, Berard F. Amikacin-induced drug reaction with eosinophilia and systemic symptoms syndrome: delayed skin test and ELISPOT assay results allow the identification of the culprit drug. J Allergy Clin Immunol. 2012;130(6):1413–1414 [DOI] [PubMed] [Google Scholar]
- 125.El-Ghaiesh S, Monshi MM, Whitaker P, et al. Characterization of the antigen specificity of T-cell clones from piperacillin-hypersensitive patients with cystic fibrosis. J Pharmacol Exp Ther. 2012;341(3):597–610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Jenkins RE, Yaseen FS, Monshi MM, et al. β-Lactam antibiotics form distinct haptenic structures on albumin and activate drug-specific T-lymphocyte responses in multiallergic patients with cystic fibrosis. Chem Res Toxicol. 2013;26(6):963–975 [DOI] [PubMed] [Google Scholar]
- 127.Khalil G, El-Sabban M, Al-Ghadban S, et al. Cytokine expression profile of sensitized human T lymphocytes following in vitro stimulation with amoxicillin. Eur Cytokine Netw. 2008;19(3):131–141 [DOI] [PubMed] [Google Scholar]
- 128.Phatharacharukul P, Klaewsongkram J. A case of sulfasalazine-induced hypersensitivity syndrome confirmed by enzyme-linked immunospot assay. Allergy Asthma Immunol Res. 2013;5(6):415–417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Rozieres A, Hennino A, Rodet K, et al. Detection and quantification of drug-specific T cells in penicillin allergy. Allergy. 2009;64(4):534–542 [DOI] [PubMed] [Google Scholar]
- 130.Tanvarasethee B, Buranapraditkun S, Klaewsongkram J. The potential of using enzyme-linked immunospot to diagnose cephalosporin-induced maculopapular exanthems. Acta Derm Venereol. 2013;93(1):66–69 [DOI] [PubMed] [Google Scholar]
- 131.Tassignon J, Burny W, Dahmani S, et al. Monitoring of cellular responses after vaccination against tetanus toxoid: comparison of the measurement of IFN-gamma production by ELISA, ELISPOT, flow cytometry and real-time PCR. J Immunol Methods. 2005;305(2):188–198 [DOI] [PubMed] [Google Scholar]
- 132.Trubiano JA, Redwood A, Strautins K, et al. Drug-specific upregulation of CD137 on CD8+ T cells aids in the diagnosis of multiple antibiotic toxic epidermal necrolysis. J Allergy Clin Immunol Pract. 2017;5(3):823–826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Khumra S, Chan J, Urbancic K, et al. Antibiotic allergy labels in a liver transplant recipient study. Antimicrob Agents Chemother. 2017;61(5):e00078– e00117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Trubiano JA, Leung VK, Chu MY, Worth LJ, Slavin MA, Thursky KA. The impact of antimicrobial allergy labels on antimicrobial usage in cancer patients. Antimicrob Resist Infect Control. 2015;4:23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Turvey SE, Cronin B, Arnold AD, Dioun AF. Antibiotic desensitization for the allergic patient: 5 years of experience and practice. Ann Allergy Asthma Immunol. 2004;92(4):426–432 [DOI] [PubMed] [Google Scholar]
- 136.Castells M. Rapid desensitization for hypersensitivity reactions to medications. Immunol Allergy Clin North Am. 2009;29(3):585–606 [DOI] [PubMed] [Google Scholar]
- 137.Cernadas JR. Desensitization to antibiotics in children. Pediatr Allergy Immunol. 2013;24(1):3–9 [DOI] [PubMed] [Google Scholar]
- 138.D’Amelio CM, Del Pozo JL, Vega O, Madamba R, Gastaminza G. Successful desensitization in a child with delayed cotrimoxazole hypersensitivity: a case report. Pediatr Allergy Immunol. 2016;27(3):320–321 [DOI] [PubMed] [Google Scholar]
- 139.Petitto J, Chervinskiy SK, Scurlock AM, Perry TT, Jones SM, Pesek RD. Successful clarithromycin desensitization in a macrolide-sensitive pediatric patient. J Allergy Clin Immunol Pract. 2013;1(3):307–308 [DOI] [PubMed] [Google Scholar]
- 140.Logsdon S, Ramirez-Avila L, Castells M, Dioun A. Successful rifampin desensitization in a pediatric patient with latent tuberculosis. Pediatr Allergy Immunol. 2014;25(4):404–405 [DOI] [PubMed] [Google Scholar]
- 141.Berges-Gimeno MP, Stevenson DD. Nonsteroidal anti-inflammatory drug-induced reactions and desensitization. J Asthma. 2004;41(4):375–384 [DOI] [PubMed] [Google Scholar]
- 142.Demoly P, Messaad D, Sahla H, et al. Six-hour trimethoprim-sulfamethoxazole-graded challenge in HIV-infected patients. J Allergy Clin Immunol. 1998;102(6, pt 1):1033–1036 [DOI] [PubMed] [Google Scholar]
- 143.Dilley MA, Lee JP, Platt CD, Broyles AD. Rituximab desensitization in pediatric patients: results of a case series. Pediatr Allergy Immunol Pulmonol. 2016;29(2):91–94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Brennan PJ, Rodriguez Bouza T, Hsu FI, Sloane DE, Castells MC. Hypersensitivity reactions to mAbs: 105 desensitizations in 23 patients, from evaluation to treatment. J Allergy Clin Immunol. 2009;124(6):1259–1266 [DOI] [PubMed] [Google Scholar]
- 145.Scherer K, Brockow K, Aberer W, et al. ; European Network on Drug Allergy and the EAACI Drug Allergy Interest Group . Desensitization in delayed drug hypersensitivity reactions – an EAACI position paper of the Drug Allergy Interest Group. Allergy. 2013;68(7):844–852 [DOI] [PubMed] [Google Scholar]
- 146.Trubiano JA, Thursky KA, Stewardson AJ, et al. Impact of an integrated antibiotic allergy testing program on antimicrobial stewardship: a multicenter evaluation. Clin Infect Dis. 2017;65(1):166–174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Rimawi RH, Shah KB, Cook PP. Risk of redocumenting penicillin allergy in a cohort of patients with negative penicillin skin tests. J Hosp Med. 2013;8(11):615–618 [DOI] [PubMed] [Google Scholar]
- 148.Patel SV, Tarver SA, Alvarez KS, Lutek KE, Schlebus J, Khan DA. Effectiveness of interventions to maintain penicillin allergy label removal as part of an inpatient penicillin allergy testing protocol. J Allergy Clin Immunol. 2017;139(suppl 2):AB183 [Google Scholar]
- 149.Chen JR, Khan DA. Evaluation of penicillin allergy in the hospitalized patient: opportunities for antimicrobial stewardship. Curr Allergy Asthma Rep. 2017;17(6):40. [DOI] [PubMed] [Google Scholar]
- 150.Chen JR, Tarver SA, Alvarez KS, Tran T, Khan DA. A proactive approach to penicillin allergy testing in hospitalized patients. J Allergy Clin Immunol Pract. 2017;5(3):686–693 [DOI] [PubMed] [Google Scholar]
- 151.Blumenthal KG, Shenoy ES, Varughese CA, Hurwitz S, Hooper DC, Banerji A. Impact of a clinical guideline for prescribing antibiotics to inpatients reporting penicillin or cephalosporin allergy. Ann Allergy Asthma Immunol. 2015;115(4):294–300.e2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Blumenthal KG, Shenoy ES, Wolfson AR, et al. Addressing inpatient beta-lactam allergies: a multihospital implementation. J Allergy Clin Immunol Pract. 2017;5(3):616–625.e7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Blumenthal KG, Wickner PG, Hurwitz S, et al. Tackling inpatient penicillin allergies: assessing tools for antimicrobial stewardship. J Allergy Clin Immunol. 2017;140(1):154–161.e6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Moral L, Garde J, Toral T, Fuentes MJ, Marco N. Short protocol for the study of paediatric patients with suspected betalactam antibiotic hypersensitivity and low risk criteria. Allergol Immunopathol (Madr). 2011;39(6):337–341 [DOI] [PubMed] [Google Scholar]
- 155.Mori F, Cianferoni A, Barni S, Pucci N, Rossi ME, Novembre E. Amoxicillin allergy in children: five-day drug provocation test in the diagnosis of nonimmediate reactions. J Allergy Clin Immunol Pract. 2015;3(3):375–380.e1 [DOI] [PubMed] [Google Scholar]