Ascending aortic pseudoaneurysms are rare complications of cardiac surgery that present with variable timing and often occur at sites of cannulation, aortotomy, or anastomoses from previous grafting or repair.1 Historically reported only in small case series, they complicate fewer than 0.5% of cardiac surgical cases.2 Surgical approach and perioperative management can be particularly challenging when the pseudoaneurysm is large; infected; or has significant mass effect on the airway, right heart, or pulmonary vasculature. Anesthetic management of these patients necessitates significant planning and communication between the surgical and anesthesia teams to ensure patient safety. The ideal approach for surgical repair is debated in the setting of mediastinitis or infection, further complicating surgical planning. In some cases, endovascular surgery may be indicated to exclude the pseudoaneurysm as a bridge to definitive surgical repair or in lieu of traditional surgical repair in high-risk patients. Transesophageal echocardiography (TEE) is an important component of intraoperative management.
Case Report*
A 55-year-old female 8 months after coronary artery bypass grafting (CABG) presented with persistent left ear pain radiating to the neck and swelling and pain at the site of her sternotomy incision. She was started on antibiotics for presumed incisional cellulitis. Computed tomography angiography demonstrated a 4.5×3.2 cm aortic pseudoaneurysm (Fig 1) and mediastinitis; the origin was suspected to be the previous aortic cannulation site. Notably, her initial CABG procedure was complicated by methicillin-resistant Staphylococcus aureus sternal infection and mediastinitis, which required multiple courses of antibiotics and hyperbaric oxygen therapy.
Fig 1.
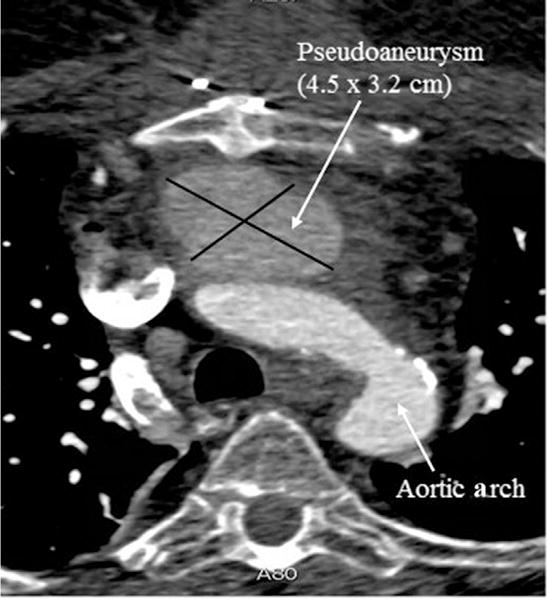
Preoperative computed tomography angiogram demonstrating a 4.5×3.2 cm aortic pseudoaneurysm effacing the right lateral wall of the main pulmonary artery and with presternal and periaortic fluid collections consistent with mediastinitis.
At the time of the first attempted repair, the patient denied experiencing orthopnea or symptoms of intrathoracic tracheal obstruction, and imaging did not indicate tracheal compression by the pseudoaneurysm. As such, her airway was secured with a standard intravenous induction and direct laryngoscopy without complication. Because this was a high-risk surgery with an expected long cardiopulmonary bypass (CPB) duration, a pulmonary artery catheter (PAC) was placed without difficulty for intraoperative and postoperative monitoring. Intraoperative TEE subsequently confirmed a large pseudoaneurysm in the proximal ascending aorta with flow from the aorta into the pseudoaneurysmal sac (Fig 2) and compression of the main pulmonary artery (PA) (Fig 3). PAC readings demonstrated right ventricular (RV) systolic pressures to be greater than the PA systolic pressures, suggestive of functional pulmonic stenosis (PS). Consistent with the suspicion for functional PS, precipitous hypotension occurred when the patient’s legs were raised during surgical preparation, suggesting intolerance of an acute, drastic increase in preload to the RV. The hypotension resolved after her legs were lowered and the administration of a modest dose of phenylephrine (Baxter, Deerfield, IL).
Fig 2.
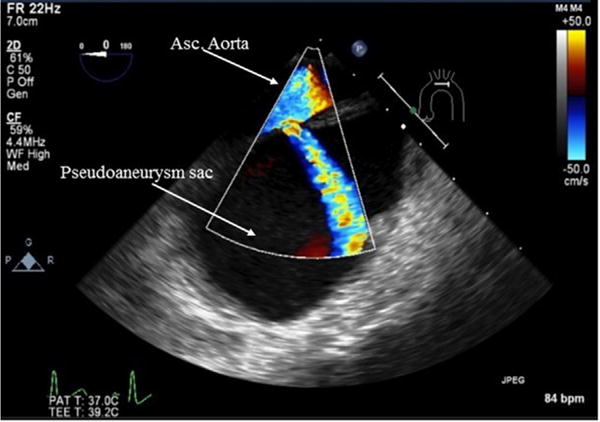
Midesophageal ascending aorta long-axis view with color-flow Doppler. Flow is visualized from the ascending aorta into the pseudoaneurysm sac.
Fig 3.
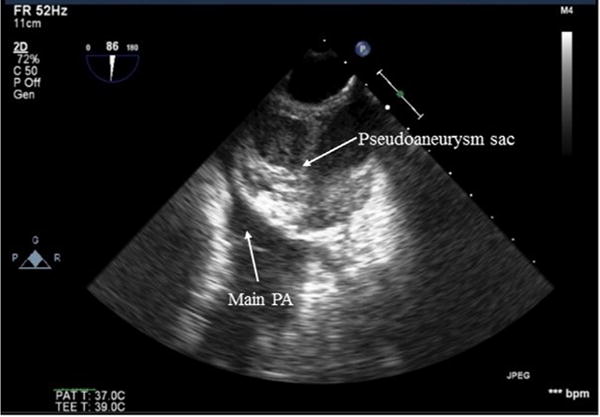
Upper esophageal aortic arch short-axis view. The pseudoaneurysm sac can be seen compressing the main PA just proximal to the pulmonic valve. PA, pulmonary artery.
Given the proximity of the pseudoaneurysm to the sternum, femoral arterial and venous CPB cannulas were placed, CPB was initiated, and the patient’s body temperature was cooled to 201C for potential circulatory arrest before sternotomy. The pseudoaneurysm was breached with sternotomy, but bleeding was contained with digital control. Inflamed mediastinal tissue and the pseudoaneurysm were debrided. With the patient under brief circulatory arrest, the pseudoaneurysm neck was closed with 2 pledgeted sutures. The location of the pseudoaneurysm neck corresponded with the presumed location of a previous aortic cannulation site from her CABG. Aortic grafting was considered but deferred given the risk of recurrent infection. After rewarming, the patient was weaned successfully and separated from CPB. Total CPB time was 170 minutes. Circulatory arrest time was 1 minute. The sternum was left open with a wound vacuum–assisted closing (VAC) device in place with the intention to return to the operating room for a repeat mediastinal washout and debridement before definitive sternal closure in order to minimize her risk of wound dehiscence and recurrent mediastinitis. She returned to the operating room on postoperative day (POD) 1 for delayed sternal closure and her airway was extubated on POD 2. The patient had an uneventful postoperative course, was discharged on POD 9, and completed 6 weeks of intravenous (IV) antibiotics.
Three months later, the patient returned to the emergency department with wound dehiscence, fevers, and hemoptysis. She was found to be in septic shock with methicillin-resistant S. aureus bacteremia. Computed tomography angiography revealed a 9.2 × 8.7 cm recurrent pseudoaneurysm, mediastinitis, and sternal osteitis. Preoperative TEE was negative for endocarditis and confirmed the presence of a large, recurrent aortic pseudoaneurysm with flow from the aorta to the pseudoaneurysm sac.
Given the large size and clinical concern for rupture, vascular surgery was consulted to assist in isolation and control of the pseudoaneurysm before definitive surgical repair. Options considered included thoracic endovascular aortic repair, endovascular plug, or septal occlusion device placement. Thoracic endovascular aortic repair would have been ideal; however, the patient’s aortic diameter and distances between the coronary vessels and the innominate artery were too large and short, respectively, for available endovascular devices. Even though investigational devices are available at the authors’ institution for use, the time required for approval would have delayed her repair, placing the patient at increased risk. An Amplatzer (St. Jude Medical, St. Paul, MN) plug would not have been effective due to the size of the opening of the pseudoaneurysm. Septal occlusion therefore was the only option. The patient was taken to the hybrid operating room with the intent of placing an endovascular septal plug at the neck of the pseudoaneurysm. Brachial arterial access was obtained, and a septal occluder was placed in the neck of the pseudoaneurysm with incomplete occlusion. Even though the plug was positioned correctly, it was unstable as a result of poor aortic integrity at the edges of the pseudoaneurysm, resulting in an incomplete exclusion. Attempts to place a second occluder to provide stability ultimately were unsuccessful and dislodged the first occluder into the pseudoaneurysm sac. Given the poor aortic wall integrity, additional attempts at exclusion were aborted given the risk of potential embolization. The first septal occluder remained in the pseudoaneurysm and was not at risk of embolization.
Five days later, the patient returned to the operating room again for surgical repair. Given continued stability from a respiratory standpoint with little concern for tracheal compression, her airway was intubated in standard fashion. Post-induction PAC placement was challenging; TEE-guided placement of the PAC into the right PA demonstrated a waveform resembling PA occlusion pressure, which suggested significant mass effect on the RV outflow tract by the pseudoaneurysm. TEE again demonstrated a large pseudoaneurysm off the ascending aorta (Fig 4) with near-complete compression of the main PA by the pseudoaneurysm sac, new moderate tricuspid regurgitation (TR), and hypokinesis/enlargement of the RV with a D-shaped, underfilled left ventricle (LV). CPB was initiated via femoral cannulation, and the patient’s body temperature was cooled for circulatory arrest before sternotomy. Upon incision, an RV outflow tract injury occurred and was repaired with bovine patch.
Fig 4.
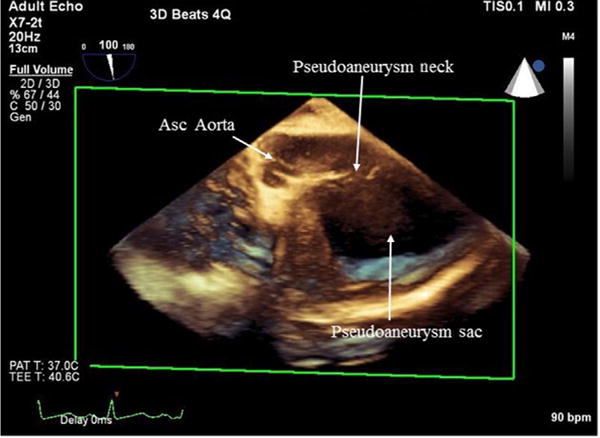
Three-dimensional full-volume midesophageal aortic valve long-axis view. The neck and sac of the pseudoaneurysm can be visualized coming off the ascending aorta.
The pseudoaneurysm sac again was debrided, and with the patient under circulatory arrest, the defective portion of the aorta, from the sinotubular junction to the bifurcation of the innominate artery, was replaced with a 30-mm tube graft. After rewarming, the patient was weaned uneventfully from CPB. Total CPB time was 173 minutes with a cross-clamp time of 38 minutes and circulatory arrest time of 32 minutes. Post-CPB TEE showed improved RV function and LV filling, trace TR, and resolution of the main PA compression. Given evidence of superinfection, a wound VAC dressing was placed, and she was transferred to the intensive care unit intubated and without vasoactive medications.
Postoperatively, the patient underwent multiple mediastinal washouts on PODs 2, 3, 10, and 14 for persistent contamination of the wound. She ultimately required a complex sternal closure with bipedicaled omental flap reconstruction and skin grafting by plastic surgery on POD 17. Her airway ultimately was extubated on POD 25, and the patient was discharged to a rehabilitation facility on POD 50 and then home on POD 61. As of her 1 year follow-up appointments, her wounds have healed completely without evidence of recurrent infection, although she did develop a subxiphoid hernia that was being managed nonoperatively. Her surgeries are summarized in Table 1.
Table 1.
Summary of Surgical Procedures
| Procedure 1: Sternotomy (8 months status post coronary artery bypass grafting) |
| • Functional pulmonic stenosis; hypotension with straight leg raise |
| • Femoral-femoral cannulation and cardiopulmonary bypass initiation before incision |
| • Aortic grafting deferred |
| • Pledgeted closure of pseudoaneurysm neck |
| Procedure 2: Attempted endovascular septal occluder placement (3 months status post procedure 1) |
| • Brachial endovascular access |
| • Occluder placement with incomplete pseudoaneurysm exclusion |
| • Attempted second occluder placement dislodged first occluder |
| • Procedure aborted |
| Procedure 3: Sternotomy (5 days status post procedure 2) |
| • Near complete main pulmonary artery occlusion, new moderate tricuspid regurgitation, and left ventricle underfilling |
| • Difficult pulmonary artery catheter placement |
| • Femoral-femoral cannulation and cardiopulmonary bypass initiation before incision |
| • Ascending aortic tube graft placed |
Discussion
Pseudoaneurysms form when the tunica intima and media of a vessel are compromised, forming a fibrous capsule around the defect and leading to instability and outpouching of the vessel wall.1 This is in contrast to a true aneurysm, in which all intimal layers remain intact.3 Most often, aortic pseudoaneurysms are associated with a defect from prior procedures or conditions that predispose the vessel to weakness, including connective tissue diseases, vasculitides, and infection.1 In 1 case series, 83% of patients with an ascending aortic pseudoaneurysm had undergone at least 1 previous cardiac surgery and 43% had evidence of infection.2 As with the patient presented here, multiple case series have identified the Staphylococcus species as the culprit organism.2,4
Even though extremely rare, cardiac surgery–related aortic pseudoaneurysms are associated with significant morbidity and mortality.3,4 Incidence of cardiac surgery–related aortic pseudoaneurysms is difficult to measure due to the rarity of these cases; several large single-institution case series report an average rate of 2 to 3 cases per year2,4–7 with an overall incidence of less than 0.5% of all cardiac surgical procedures.2 Some argue this may be underestimated, however, in patients with a history of ascending aortic procedures, for which the incidence may reach 12% to 15%.7,8 Given the frequency of previous surgery, a majority of these patients are by definition those with pre-existing vascular disease and multiple related comorbidities, including obesity, diabetes, hypertension, and coronary artery disease. Collectively, these increase the perioperative risk for morbidity and mortality in these patients. Of those patients described in the literature, in-hospital mortality has been reported to be 6.7% to 16.6% with 5-and 10-year survival rates of 62% to 79% and 60% to 68%, respectively.2,4–7
Given the significant perioperative risk, incidentally discovered or small and asymptomatic pseudoaneurysms may be clinically monitored.3 Surgical repair should be considered in patients with dissecting, enlarging, or symptomatic pseudoa-neurysms.3 Historically, nearly all cases with an acute, symptomatic presentation were surgically repaired because of the risk of expansion and rupture. A unique aspect of the case described here was the decision to perform primary closure of the pseudoaneurysm neck rather than perform aortic grafting during the patient’s first surgical intervention. At the time, the surgeons believed this was the appropriate approach because of the significant risk associated with graft infection, which yields a mortality rate between 24% and 75%.9,10 The ideal approach to pseudoaneurysm repair in the setting of infection remains controversial. Surgical exploration with extensive debridement of infected mediastinal tissues, ample intraoperative irrigation, open or VAC dressings with and without washouts or continuous irrigation, and primary or delayed soft tissue or omental flap reconstruction are common therapies across the literature, often in combination.10,11 Some literature advocates for omental wrapping of any prosthetics to minimize seeding of new grafts.9
Most contentious is whether to perform primary closure versus aortic grafting in patients without an existing prosthesis. Several large case series indicate that a primary or patch closure is performed in up to 63% of aortic pseudoaneurysm cases,12 with 1 large case series reporting preferential use of nonprosthetic material if infection is present in the absence of an existing prosthesis.7 This is supported by earlier studies indicating that the use of homografts may reduce the incidence of postoperative graft infection.9 Contrastingly, Atik et al indicated that primary closure or patch repair is only performed in patients with smaller defects with minimally infected tissue that is easily debrided (23%), whereas septic pseudoaneurysms are preferentially replaced or repaired with allografts at their institution.2 Even though the overall rates of redo surgery are as low as 13% at 1 year,2,7 the frequency of redo surgery is rarely described in terms of the type of repair undertaken; however, 1 series reported that all patients who experienced recurrent pseudoaneurysms had undergone primary sutured repair.13 Notably, Atik et al reported that with grafting in the setting of infection, lifelong antibiotics are indicated;2 however, whether patient compliance can be relied on long term if this approach were to be universally adopted is questionable.
Postoperative pseudoaneurysms unassociated with previous aortotomies (ie, aortic valve replacement, Bentall procedure) are comparatively rare (fewer than 10% of patients in major case series),12 which makes collective outcome data difficult to interpret for patients like the one presented here who do not have a graft in place at the time of repair. Nonetheless, given that mediastinitis may increase the risk for failure of the primary repair and a suspicious correlation between primary closure and recurrence, it is arguable that the literature appears to support grafting over primary repair despite the infectious risk. As such, in retrospect, grafting at the time of initial repair for the patient in the case presented here may have prevented pseudoaneurysm recurrence, although the extent of her infection may have increased her risk for recurrence regardless of the repair performed. Ultimately, in determining the best course of action for these patients, the risk of perioperative morbidity and mortality related to graft infection itself must be weighed against the risk associated with pseudoaneurysm recurrence and potentially multiple redo sternotomies.
Even though surgical repair consistently has been the standard approach for pseudoaneurysm control in these patients, recent advances in endovascular techniques and closure devices may hold promise in minimizing the need for surgical intervention or allowing for preoperative optimization.14 Even though unsuccessful in this case, occlusion of the pseudoaneurysm neck before sternotomy also may increase safety in subsequent surgical cases through minimization of the risk of hemorrhage. Percutaneous transcatheter closures and endovascular stent grafting of aortic pseudoaneurysms largely are limited to case series, with fewer than 25 published cases of successful endovascular closure, the majority of which were used to correct small neck pseudoaneurysms. This technique also has demonstrated promise for successful closures of wide neck pseudoaneurysms, for which historical methods of thrombin injection or pseudoaneurysm coiling are likely to fail.15 It also may pose a favorable treatment method as a definitive repair technique for those with prohibitively high surgical risk.13 Given the novelty of endovascular closure of aortic pseudoaneurysms, little follow-up data are available regarding long-term outcomes with this approach. Investigational aortic stent-graft devices are being tested for implantation in the ascending aorta. After approval, these devices likely are to be used in an “off-label” fashion to address high-risk ascending aortic conditions such as in this case. When implanted in the presence of infection, the intent is for these devices to be used as a bridge toward definitive repair. In some cases, however, the device may be left in place in conjunction with long-term antibiotic treatment.
Pseudoaneurysms of the aortic arch or ascending aorta are frequently located substernally, and as such, there is potential for massive hemorrhage upon sternotomy if the pseudoaneurysm is breached and significant mass effect on the mediastinum. As a result, preoperative planning for these cases may be quite complex. The risk of hemorrhage may necessitate alternative cannulation strategies such as axillary-femoral or femoral-femoral cannulation, with initiation of CPB before sternal incision.
Likewise, airway obstruction can occur with large pseudoaneurysms, which may manifest as orthopnea, difficult ventilation, or difficult intubation. Furthermore, introducing a TEE probe into a high-pressure mediastinal space has been reported to cause tracheal compression even after the airway has been secured.16 Unfortunately, because the primary literature regarding airway management in those with mediastinal masses is largely limited to case series in the pediatric population, there is little evidence-based guidance for risk stratification of patients to guide airway interventions.17 Nonetheless, in patients who are symptomatic or appear high risk for tracheal compression based on imaging, alternative airway management strategies should be considered. For patients with proximal compression, awake fiber optic intubation remains the gold standard. However, volatile mask induction or IV ketamine induction with maintenance of spontaneous respiratory effort until the airway is secured also can be considered. Rigid bronchoscopy and a thoracic surgeon or otolaryngologist should be immediately available in these cases. Regardless of approach, all patients should be adequately preoxygenated before attempted intubation. In cases of severe distal obstruction at or below the carina beyond which standard and even microlaryngeal tracheal tubes may extend, peripheral cannulation and potentially initiation of CPB before induction may be necessary. Should unanticipated airway obstruction occur, moving the patient to a lateral, semiprone, or sitting position may alleviate some compression and allow for oxygenation and ventilation to resume.17
Right heart or pulmonary vasculature compression may cause RV dysfunction or RV outflow tract obstruction, which can lead to pulmonary hypertension and make preoperative line placement challenging and dangerous. It is important to avoid exacerbating pulmonary hypertension in these patients by minimizing hypoxia and hypercarbia and sparing the use of alpha agonists. Although main and branch PA compression have been described previously, to the authors’ knowledge, this case represents only the third patient to present with severe functional PS,15,18 which manifested as difficult PAC placement due to the near-complete compression of the RV outflow tract and subsequent intolerance of sudden preload with straight leg raise. In retrospect, PAC placement carried increased risk in this setting, but the extent of the PA compression was not clinically appreciated until PAC placement. Given the degree of right heart dysfunction, the authors believe that the benefits ultimately outweighed the risk. TEE substantiated the clinical manifestations of functional PS because it demonstrated new TR, RV compression, and underfilling of the LV. Finally, both superior vena cava (SVC) syndrome16,19 and perioperative myocardial ischemia secondary to kinking or occlusion of coronary bypass grafts have been reported.1 In patients with concern for SVC compression, IV access in the upper extremities and neck may be compromised, necessitating peripheral or central IV access at the femoral veins or below.17 Careful planning, including review of the available imaging and preoperative discussion with the surgeon, is necessary to ensure patient safety.
Patients who have undergone a sternotomy for previous procedures also face challenges related to repeat cardiac surgery. This includes longer, more complicated dissections with higher estimated blood loss due to scarring and longer CPB times, especially if CPB is initiated before incision and dissection, as in this case. Patients with recent heparin exposure are at risk for acquired antithrombin III deficiency, which manifests as heparin resistance.20 Patients also may develop antibodies to blood products. Combined, these factors may complicate the perioperative planning for these patients. Specific considerations for the care of patients with aortic pseudoaneurysms are summarized in Table 2.
Table 2.
Anesthetic and Surgical Implications of Aortic Pseudoaneurysm
| Preoperative planning |
| • Review all available imaging with surgical and anesthesia teams for evidence of extrinsic compression of structures (trachea, right heart, major vessels) by the pseudoaneurysm |
| • Discuss repair (open v endovascular) versus conservative management |
| • Consult infectious disease specialists (if indicated) |
| Perioperative concerns |
| • Airway: If compression, consider awake fiber optic intubation versus induction with intravenous or inhalational agents to maintain spontaneous respiration |
| • Monitoring: Potential difficult pulmonary artery catheter placement; consider transesophageal echocardiography for advanced hemodynamic monitoring |
| • Hemodynamics: May have compromised right ventricle outflow, pulmonary hypertension, or functional pulmonic stenosis |
| • Revision sternotomy concerns: Risk of pseudoaneurysm rupture, heparin resistance, acquired antibodies from transfusion |
| • Massive blood loss: Large-bore central access, blood products in room before incision, cell salvage; consider alternative cardiopulmonary bypass cannulation |
| • Heparin resistance: Consider recombinant AntithrombinIII versus fresh frozen plasma |
In summary, aortic pseudoaneurysms are rare complications of cardiac surgery that pose unique perioperative anesthetic and surgical challenges with potentially catastrophic implications. As in this case, large aortic pseudoaneurysms can function as a mediastinal mass with significant distortion of critical structures, necessitating careful perioperative management and planning.
Commentary 1†
Since the introduction of endovascular devices to treat various aortic pathologies, the morbidity and mortality associated with treatment of these conditions have been altered drastically. This is true not only for primary aortic procedures, but also for secondary procedures to manage their complications. Endovascular therapies allow the vascular specialist to address various arterial problems from remote locations using fluoroscopic guidance in a percutaneous manner, thereby limiting the physiologic insult to the patient. However, the Achilles heel of these procedures is that they often require the implantation of a foreign body, which is a relative contraindication in patients with infectious pathologies.
Patients undergoing cardiac procedures can experience complications such as an aortic pseudoaneurysm associated with the cannulation site. Although once managed solely through redo surgery, endovascular treatment of aortic pseudoaneurysms has been shown to be quite effective, with a low morbidity and mortality in recent years.21 The determination of their effectiveness and success is related to the location of the pseudoaneurysm relative to vital structures such as bypass origins, the aortic valve, and the innominate artery. In addition, the diameter and length of the pseudoaneurysm’s neck are critical in choosing the type of device needed for repair. For narrow, long pseudoaneurysm necks, vascular plugs typically are the ideal device. When larger connections are present, septal occlusion plugs or aortic stent grafts can be used.22 Septal occlusion devices are slightly more cumbersome to deploy and occasionally can fail due to poor apposition to the aortic wall. Aortic stent grafts provide the largest areas of coverage but are limited by their size and potential need to cross the aortic valve for implantation in certain scenarios.
Infectious aortic complications present a difficult dilemma for the endovascular specialist. In order to eradicate the infection, it is advisable to debride the affected aorta and the surrounding tissues. Autologous tissue or homografts offer the lowest risk of re-infection and often are used.23 When these are unavailable, antibiotic-impregnated Dacron (Medline, Northfield, IL) grafts can be implanted. Because debridement is not possible with endovascular techniques, the decision of optimal approach should be made in consultation with all involved specialists so that the relative risks of open surgical repair versus endovascular treatment with life-long antibiotics can be determined.
In some cases, the risk of open repair is related to the difficulty in obtaining safe exposure of the aorta without life-threatening bleeding. In these circumstances, endovascular exclusion can be used as a bridge to safe surgical exposure and subsequent debridement and repair. This was the original intent in this particular case. The aortic diameter and length of the ascending aorta prevented the use of an aortic stent graft. In addition, the removal of such a device is more cumbersome and can be associated with an increased risk of procedural complications such as stroke. A septal occlusion plug was selected for attempted exclusion of the pseudoaneurysm due to the large neck diameter. The initial plug, even though deployed accurately, was unstable as a result of extremely poor aortic integrity due to the extensive infection. The placement of a second plug was attempted to provide complete exclusion but was not successful. In many cases, arterial access for the endovascular exclusion is difficult as result of vessel tortuosity; direct mediastinal access between the ribs from the anterior chest wall can circumvent this issue.
As endovascular solutions become more common, it should be remembered that appropriate imaging follow-up is imperative to ensure that the repair is successful. Secondary procedures sometimes are necessary to provide complete exclusion of these lesions and patients should be counseled about appropriate follow-up. As this technology advances, more innovative solutions likely will become available. However, the ability to manage patients with concomitant infectious issues may be the limiting factor.
Commentary 2‡
This is a very challenging case of ascending aortic pseudoaneurysm after a CABG procedure. These conditions are quite rare, but when they occur, they are catastrophic in nature and represent very high-risk surgical cases.24
Ascending aortic pseudoaneurysms typically occur at sites of previous anastomoses, graft suture lines, or cannulation sites. The 2 common causes are iatrogenic aortic wall disruption (or a chronic incomplete seal of the aorta) and infection of surrounding aortic tissue related to foreign material (sutures and pledgets), leading to loss of integrity of the aortic wall. Autoimmune causes and trauma are less common.5,6 An attractive, less-invasive approach to these conditions is embolization or exclusion using various devices, including stents, coils, vascular plugs, and septal occluders. These less invasive techniques provide an alternative treatment for patients determined to be unsuitable for surgery. However, there are specific challenges related to these procedures, including the presence of short landing zones for grafts, risk of damaging the aortic valve with guidewires and other devices, risk of occlusion of aortic branches, device migration, and limited established techniques for the treatment of ascending aortic pseudoaneurysms.13,14,25–27
Surgical repair of these conditions typically is advocated when possible but is associated with significant mortality and morbidity. There is some controversy regarding whether these conditions should be treated with complete aortic replacement or simple patching. Complete aortic replacement seems less likely to fail but is more technically challenging. Simple patching has been used with success,28 but there is a significant risk of local failure and recurrence of the pseudoa-neurysm.29 Whether a previous aortic graft was implanted can influence the surgical strategy because it is reasonable to assume that the entire prosthetic graft may be infected, and hence, should be removed. This was not the case with the present clinical scenario.
Infection of aortic grafts is a potentially devastating complication of aneurysm surgery. Even though rare, it may be possible to reduce the frequency of the prosthetic graft infections further by soaking the graft in antibiotic solution before implantation.30 Graft wrapping with vascularized tissue such as omentum is rarely performed in first-time, noninfected implants but has been used successfully in the case of infected graft material in an attempt to control the infection rather than explanting the graft.31,32
The sequence of events and surgeries performed in the present case were reasonable, but in this author’s opinion, not ideal. The timing of surgical intervention in this case was fortunate because many of these patients do not present until the pseudoaneurysm has eroded through the sternum and into the soft tissues.5,28 Nevertheless, in this case, the chances of entering the pseudoaneurysm with the initial repair were high, and the surgical team acted reasonably with the establishment of femoral-femoral bypass and profound cooling before opening the sternum. It is unclear whether the initial plan was to locally repair the pseudoaneurysm rather than to replace a section of the ascending aorta. If graft replacement were a possibility, then a better arterial cannulation strategy would have been via the right axillary artery, which would have allowed for selective antegrade cerebral perfusion should a prolonged circulatory arrest period been necessary. In general, this author prefers this approach to cases such as these over femoral arterial cannulation.
Antibiotic use in vascular surgery can have a significant benefit in reducing recurrence of aortic infections. Typically, broad-spectrum antibiotics are used at first, followed by culture-specific antibiotics later.33 The duration of antimicrobial treatment in patients with surgically treated infectious pseudoaneurysms or graft infections is controversial. There are no current guidelines that define evidence-based algorithms for this disease. Recommendations have varied from 6 weeks to lifelong therapy, either intravenously or orally.34 A reasonable recommendation is to use antibiotics for a minimum of 6 weeks after surgery, follow-up the patient closely in consultation with infectious disease physicians, and discontinue therapy when there are no clinical, radiologic, or laboratory signs of infection.35
In summary, this author believes that the appropriate first surgery should have been resection of the infected segment of ascending aorta and replacement with an ascending aortic homograft. Even though risky, this would have greatly reduced the chance of re-infection by eliminating both the infected tissue and the pseudoaneurysm neck. In addition, this approach would have avoided the inherent risks of placing a foreign body in an infected field. That being said, however, it is well understood that things are often much clearer when viewed retrospectively.
Acknowledgments
M.A. Farber has received clinical trial funding from and is a research consultant for Cook Medical, Bloomington, IN; WL Gore & Associates, Newark, DE; Endologix, Irvine, CA; Medtronic, Minneapolis, MN; and GE, Boston, MA, and has received research support from Cook Medical.
Footnotes
M.D. Henley, M.A. Farber, L.M. Kolarczyk, E.G. Teeter, B.P. Barrick, T.G. Caranasos, and S.M. Martinelli
M.A. Farber
J.S. Ikonomidis
References
- 1.Konia M, Uppington J, Moore P, et al. Ascending aortic pseudoaneurysm: A late complication of coronary artery bypass. Anesth Analg. 2008;106:767–8. doi: 10.1213/ane.0b013e3181608bbd. [DOI] [PubMed] [Google Scholar]
- 2.Atik FA, Navia JL, Svensson LG, et al. Surgical treatment of pseudoaneurysm of the thoracic aorta. J Thorac Cardiovasc Surg. 2006;132:379–85. doi: 10.1016/j.jtcvs.2006.03.052. [DOI] [PubMed] [Google Scholar]
- 3.Sabri MN, Henry D, Wechsler AS, et al. Late complications involving the ascending aorta after cardiac surgery: Recognition and management. Am Heart J. 1991;121:1779–83. doi: 10.1016/0002-8703(91)90026-e. [DOI] [PubMed] [Google Scholar]
- 4.Dhadwal AK, Abrol S, Zisbrod Z, et al. Pseudoaneurysms of the ascending aorta following coronary artery bypass surgery. J Card Surg. 2006;21:221–4. doi: 10.1111/j.1540-8191.2006.00220.x. [DOI] [PubMed] [Google Scholar]
- 5.Malvindi PG, Cappai A, Raffa GM, et al. Analysis of postsurgical aortic false aneurysm in 27 patients. Tex Heart Inst J. 2013;40:274–80. [PMC free article] [PubMed] [Google Scholar]
- 6.Malvindi PG, van Putte BP, Heijmen RH, et al. Reoperations for aortic false aneurysms after cardiac surgery. Ann Thoracic Surg. 2010;90:1437–43. doi: 10.1016/j.athoracsur.2010.06.103. [DOI] [PubMed] [Google Scholar]
- 7.Villavicencio MA, Orszulak TA, Sundt TM, III, et al. Thoracic aorta false aneurysm: What surgical strategy should be recommended? Ann Thorac Surg. 2006;82:81–9. doi: 10.1016/j.athoracsur.2006.02.081. [DOI] [PubMed] [Google Scholar]
- 8.Preisman S, Shinfield A, Raanani E. Safe approach for chest reentry in a patient with large pseudoaneurysm of ascending aorta facilitated by intraoperative transesophageal echocardiography. J Cardiothorac Vasc Anesth. 2014;28:709–13. doi: 10.1053/j.jvca.2013.03.028. [DOI] [PubMed] [Google Scholar]
- 9.Von Aspern K, Etz CD, Mohr FW, et al. Two-stage procedure for infected aortic graft pseudoaneurysm. Aorta. 2015;3:140–4. doi: 10.12945/j.aorta.2015.14.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Coselli JS, Köksoy C, LeMaire SA. Management of thoracic aorta graft infections. Ann Thorac Surg. 1999;67:1990–3. doi: 10.1016/s0003-4975(99)00355-0. [DOI] [PubMed] [Google Scholar]
- 11.Sjögren J, Malmsjö M, Gustaffson R. Poststernotomy medastinitis: A review of conventional surgical treatments, vacuum-assisted closure therapy and presentation of the Lund University Hospital mediastinitis algorithm. Eur J Cardiothorac Surg. 2016;30:898–905. doi: 10.1016/j.ejcts.2006.09.020. [DOI] [PubMed] [Google Scholar]
- 12.Quevedo HC, Santiago-Trinidad S, Castellanos J, et al. Systematic review of interventions to repair ascending aortic pseudoaneurysms. Ochsner J. 2014;14:576–85. [PMC free article] [PubMed] [Google Scholar]
- 13.Quevedo HC, Alonso A. Endovascular therapy for ascending aorta pseudoaneurysm. Cardiovasc Revasc Med. 2016;17:586–8. doi: 10.1016/j.carrev.2016.08.008. [DOI] [PubMed] [Google Scholar]
- 14.Alameddine AK, Alimov VK. Endovascular closure of an aortic pseudoaneurysm complicated by aorto-pulmonary fistula. J Card Surg. 2016;31:453–5. doi: 10.1111/jocs.12758. [DOI] [PubMed] [Google Scholar]
- 15.Hussain J, Strumpf R, Wheatley G, et al. Percutaneous closure of aortic pseudoaneurysm by Amplatzer occlude device – case series of six patients. Catheter Cardiovasc Interv. 2009;73:521–9. doi: 10.1002/ccd.21833. [DOI] [PubMed] [Google Scholar]
- 16.Arima H, Sobue K, Tanaka S, et al. Airway obstruction associated with transesophageal echocardiography in a patient with a giant aortic pseudoaneurysm. Anesth Analg. 2002;95:558–60. doi: 10.1097/00000539-200209000-00010. [DOI] [PubMed] [Google Scholar]
- 17.Blank RS, de Souza DG. Anesthetic management of patients with an anterior mediastinal mass: Continuing professional development. Can J Anesth. 2011;58:853–67. doi: 10.1007/s12630-011-9539-x. [DOI] [PubMed] [Google Scholar]
- 18.Van Camp G, De Mey J, Daenen W, et al. Pulmonary stenosis caused by extrinsic compression of an aortic pseudoaneurysm of a composite aortic graft. J Am Soc Echocardiogr. 1999;12:997–1000. doi: 10.1016/s0894-7317(99)70155-0. [DOI] [PubMed] [Google Scholar]
- 19.Taillandier S, Guillon A, Favelle O, et al. Airway obstruction by an aortic false aneurysm. Ann Fr Anesth Reanim. 2011;30:79–81. doi: 10.1016/j.annfar.2011.08.011. [DOI] [PubMed] [Google Scholar]
- 20.Finley A, Greenberg C. Heparin sensitivity and resistance: Management during cardiopulmonary bypass. Anesth Analg. 2013;116:1210–22. doi: 10.1213/ANE.0b013e31827e4e62. [DOI] [PubMed] [Google Scholar]
- 21.Piffaretti G, Galli M, Lomazzi C, et al. Endograft repair for pseudoaneurysms and penetrating ulcers of the ascending aorta. J Thorac Cardiovasc Surg. 2016;151:1606–14. doi: 10.1016/j.jtcvs.2015.12.055. [DOI] [PubMed] [Google Scholar]
- 22.Preventza O, Henry MJ, Cheong BY, et al. Endovascular repair of the ascending aorta: When and how to implement the current technology. Ann Thoracic Surg. 2014;97:1555–60. doi: 10.1016/j.athoracsur.2013.11.066. [DOI] [PubMed] [Google Scholar]
- 23.Vogt PR, von Segesser LK, Goffin Y, et al. Eradication of aortic infections with the use of cryopreserved arterial homografts. Ann Thorac Surg. 1996;62:640–5. doi: 10.1016/s0003-4975(96)0045-4. [DOI] [PubMed] [Google Scholar]
- 24.Mohammadi S, Bonnet N, Leprince P. Reoperation for false aneurysm of the ascending aorta after its prosthetic replacement: Surgical strategy. Ann Thorac Surg. 2005;79:147–52. doi: 10.1016/j.athoracsur.2004.06.032. [DOI] [PubMed] [Google Scholar]
- 25.Stasek J, Polansky P, Bis J, et al. The percutaneous closure of a large pseudoaneurysm of the ascending aorta with an atrial septal defect Amplatzer occluder: Two year follow up. Can J Cardiol. 2008;24:e99–101. doi: 10.1016/s0828-282x(08)70703-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bhindi R, Newton J, Wilson N, et al. Percutaneous plugging of an ascending aortic pseudoaneurysm. JACC Cardiovasc Interv. 2008;1:327–8. doi: 10.1016/j.jcin.2008.04.006. [DOI] [PubMed] [Google Scholar]
- 27.Hadjivassiliou A, Gagnon J, Janusz M, et al. Combined CT, fluoroscopic and IVUS guidance for percutaneous treatment of a postsurgical pseudoaneurysm following repair of ascending thoracic aortic aneurysm. Cardiovasc Intervent Radiol. 2017 Jun 5; doi: 10.1007/s00270-017-1710-5. [E-pub ahead of print] [DOI] [PubMed] [Google Scholar]
- 28.Hasan SB, Khan FW, Hashmi S, et al. Repair of ascending aortic pseudoaneurysm eroding through the sternum. Asian Cardiovasc Thorac Ann. 2017 Jan 1; doi: 10.1177/0218492317707626. [E-pub ahead of print] [DOI] [PubMed] [Google Scholar]
- 29.Tochii M, Amano K, Sakurai Y, et al. Recurrence of aneurysm of the ascending aorta after patch repair: The fate of an aortic patch. Ann Vasc Dis. 2016;9:322–5. doi: 10.3400/avd.cr.16-00043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Aboshady I, Raad I, Shah AS, et al. A pilot study of a triple antimicrobial-bonded Dacron graft for the prevention of aortic graft infection. J Vasc Surg. 2012;56:794–801. doi: 10.1016/j.jvs.2012.02.008. [DOI] [PubMed] [Google Scholar]
- 31.Jamieson RW, Burns PJ, Dawson AR, et al. Aortic graft preservation by debridement and omental wrapping. Ann Vasc Surg. 2012;26:423. doi: 10.1016/j.avsg.2011.07.021. [DOI] [PubMed] [Google Scholar]
- 32.Yamashiro S, Arakaki R, Kise Y, et al. Potential role of omental wrapping to prevent infection after treatment for infectious thoracic aortic aneurysms. Eur J Cardiothorac Surg. 2013;43:1177–82. doi: 10.1093/ejcts/ezs600. [DOI] [PubMed] [Google Scholar]
- 33.Chiesa R, Astore D, Frigerio S, et al. Vascular prosthetic graft infection: Epidemiology, bacteriology, pathogenesis and treatment. Acta Chir Belg. 2002;102:238–47. doi: 10.1080/00015458.2002.11679305. [DOI] [PubMed] [Google Scholar]
- 34.Robinson WP, Schuksz M. Surgical and antimicrobial management of a thoracic aortic aneurysm due to Q fever: A case report and brief review. Vasc Endovascular Surg. 2016;50:290–4. doi: 10.1177/1538574416642876. [DOI] [PubMed] [Google Scholar]
- 35.Treska V, Certik B, Molacek J. Management of aortic graft infections - the present strategy and future perspectives. Bratisl Lek Listy. 2016;117:125–32. doi: 10.4149/bll_2016_024. [DOI] [PubMed] [Google Scholar]


