Abstract
Objective
Human papillomavirus (HPV) 16 is the most carcinogenic HPV genotype. We investigated if HPV16 L1 capsid protein and E2/E6 ratio, evaluated by cervical cytology, may be used as biomarkers of ≥cervical intraepithelial neoplasia (CIN) 2 lesions.
Methods
Cervical specimens were obtained from 226 patients with HPV16 single infection. Using cytology specimen, L1 capsid protein and E2/E6 ratio were detected and the results were compared with those of the conventional histologic analysis of cervical tissues (CIN1–3 and squamous cell carcinoma [SCC]) to evaluate the association.
Results
The L1 positivity of CIN2/3 was significantly lower than that of normal cervical tissue (p<0.001) and SCC demonstrated significantly lower L1 positivity than CIN1 (p<0.001). The mean E2/E6 ratios of specimens graded as SCC (0.356) and CIN2/3 (0.483) were significantly lower than those of specimens graded as CIN1 (0.786) and normal (0.793) (p<0.05). We observed that area under the receiver operating characteristic curve (AUC) for E2/E6 ratio (0.844; 95% confidence interval [CI]=0.793–0.895) was higher than that for L1 immunochemistry (0.636; 95% CI=0.562–0.711). A combination of E2/E6 ratio and L1 immunocytochemistry analyses showed the highest AUC (0.871; 95% CI=0.826–0.917) for the prediction of ≥CIN2 lesions.
Conclusion
To our knowledge, this is the first study to validate HPV L1 capsid protein expression and decreased HPV E2/E6 ratio as valuable predictive markers of ≥CIN2 cervical lesions. Cervical cytology may be analyzed longitudinally on an outpatient basis with noninvasive procedures as against invasive conventional histologic analysis.
Keywords: Cervical Intraepithelial Neoplasia, Uterine Cervical Neoplasms, Human Papillomavirus Type 16 L1 Protein, Immunocytochemistry, Viral Integration
INTRODUCTION
It is known that 99.7% of cervical cancer cases are attributed to the infection with human papillomavirus (HPV) [1]. HPV16 is the most carcinogenic HPV genotype and HPV integration into the host genome is regarded as one of the most important steps for cervical intraepithelial neoplasia (CIN) and cervical cancer [2]. While the low-grade disease, CIN1, shows spontaneous regression, CIN2 and CIN3 have substantial potency to progress into cancer [3]. Therefore, the use of biomarkers is crucial to distinguish cervical lesions based on their progression/regression ability. The results of p16 and Ki-67 immunohistochemistry were recently used to predict high-grade CIN. However, these analyses require biopsy of cervical tissues [4,5]. Invasive procedures such as biopsy may cause discomfort, pain, and even bleeding from biopsy site. In this direction, we have established a less invasive and more convenient method using cervical cytology for the prediction of high-grade CIN.
Disruption of E2 gene and overexpression of E6 or E7 during HPV integration into the host genome have been utilized as surrogate markers to discriminate CIN1 from CIN2, CIN3, and carcinoma [6,7]. We have recently reported that the absence of HPV L1 immunocytochemistry in HPV16-positive atypical squamous cells of undetermined significance (ASCUS) and low-grade squamous intraepithelial lesion (LSIL) is significantly related with higher-grade CIN [8,9]. Furthermore, the absence of the HPV L1 protein is suggestive of HPV integration into the host genome and is associated with disease progression [10,11].
Here, we investigate whether the detection of HPV16 L1 capsid protein and measurement of E2/E6 ratio are predictive of CIN2/3 and cancer and if analyses of cervical cytological specimen noninvasively collected yield sufficiently conclusive results.
MATERIALS AND METHODS
1. Patients and study design
We analyzed 226 cervical specimens tested positive for HPV16 single infection. The specimens were collected from Seoul St. Mary's Hospital, The Catholic University of Korea (Seoul, Korea) from January 2010 to August 2017. This study was approved by the Institutional Review Board (IRB) of The Catholic University of Korea College of Medicine (IRB number: KC13SISI0901). Cervical cytology was performed from the endocervix and exocervix, and specimens were assigned to standard categories (i.e., ASCUS, LSIL, high-grade squamous intraepithelial lesion [HSIL], and squamous cell carcinoma [SCC]). HPV types were determined by a DNA genotyping polymerase chain reaction (PCR)-based DNA microarray system (Mygene, Seoul, Korea) that enabled detection of 16 types of high-risk HPV (16, 18, 31, 33, 35, 39, 45, 51, 52, 53, 54, 56, 58, 59, 66, and 68) and eight types of low-risk HPV (6, 11, 34, 40, 42, 43, 44, and 70). Cervical tissues were collected after loop electrosurgical excision procedure (LEEP) or hysterectomy and categorized into four groups such as normal, CIN1, CIN2/3, and SCC.
2. Immunocytochemistry
ThinPrep slides (Hologic, Bedford, MA, USA) were immunochemically stained as part of a Cytoactiv® HPV L1 screening set (Cytoimmun Diagnostics, Pirmasens, Germany) to detect L1 capsid protein of all known HPV types, as previously described [8,9]. Staining was performed according to the manufacturer's protocol. Briefly, slides were unmounted without prior destaining and sections were microwaved to achieve antigen unmasking. Cytoactiv® HPV L1 capsid antibody was applied and slides were incubated for 30 minutes at 20°C. Following incubation, slides were treated with the detection reagents for 10 minutes and 3-amino-9-ethylcarbazole chromogen for 5 minutes. After counterstaining with hematoxylin, slides were mounted with Aquatex (Merck, Darmstadt, Germany) and a cover slip. Slides with a distinct nuclear staining for at least one epithelial cell were scored as positive. The slides were reviewed by 2 pathologists for final diagnosis.
3. DNA extraction from cervical cells and analysis of HPV16 E2/E6
For the HPV DNA extraction, the remaining cervical cells were pelleted by centrifugation and resuspended in digestion solution (10 mmol/L Tris, 1 mmol/L ethylenediaminetetraacetic acid, 200 μg/mL proteinase K) for 3 hours at 56°C. It was incubated for 10 minutes at 95°C to inactivate proteinase K. Real-time quantitative PCR was carried out with modifications in the previously described protocol to determine E2/E6 ratio of HPV16 DNA (GenBank Accession number: K02718) [6]. E2 and E6 sequences were amplified by PCR. E2 PCR was performed with primer pairs 5′-TGT GTT TAG CAG CAA CGA AG-3′ (3349–3368) and 5′-GCT GGA TAG TCG TCT GTG TT-3′ (3454–3473), while E6 PCR was performed with primer pairs 5′-CGA CCC AGA AAG TTA CCA-3′ (125–142) and 5′-AGC AAA GTC ATA TAC CTC ACG-3 (220–241). β-globin was used as an experimental control for sample quality and adequacy during PCR. β-globin PCR was with primer pairs 5′-ACA CAA CTG TGT TCA CTA GC and 5′-CAA CTT CAT CCA CGT TCA CC. Based on the PCR results, E2/E6 ratios of cervical cells were calculated to investigate HPV integration status. E2/E6 ratio of 1 was regarded as “episome state,” whereas that ranging from >0 to <1 indicated “mixed state”; a ratio of 0 was considered as “fully integrated state.” The “episomal state” represents that HPV is not integrated into the host genome and characterized by an identical amount of E2 and E6. In contrast, “fully integrated state” induces loss of E2 and shows E2/E6 ratio as nearly 0 [6].
4. Statistical analysis
Statistical analyses were carried out with SAS software package (version 8.0; SAS Institute Inc., Cary, NC, USA). Analysis of variance (ANOVA) was applied for the analysis of the differences of means among 4 groups. Tukey's test was used to compare characteristics of multiple subgroups during post hoc analysis. To evaluate linear association between severity of cervical pathology and physical status of HPV integration, linear by linear analysis was performed. Diagnostic accuracy of HPV L1 immunocytochemistry and E2/E6 ratio for ≥CIN2 lesions was expressed as the area under the receiver operating characteristic (ROC) curve (AUC). The p values <0.05 were considered statistically significant.
RESULTS
1. The percentage of adequately matching cytology and tissue pathology
Analysis of the 226 cervical tissue samples with HPV16 single infection indicated that 57 (25.2%) were normal, 42 (18.6%) were classified as CIN1, 69 (35.0%) were categorized as CIN2/3, and 48 (21.2%) had evidence of SCC. Clinicopathologic features of the study population are summarized in Table 1. The indications of hysterectomy for 21 patients with normal cytology were abnormal uterine bleeding or uncontrolled dysmenorrhea related with uterine fibroids. The percentage of adequately matching cytology and tissue pathology varied from 18.9% to 100.0% (i.e., normal cytology to normal pathology, ASCUS/LSIL to CIN1, HSIL to CIN2/3, and SCC to SCC). The accordance rates of ASCUS and LSIL (18.9% and 45.5%, respectively) were lower than those of HSIL and SCC (75.5% and 89.2%, respectively).
Table 1. Clinicopathologic features of the study population (n=226).
| Tissue pathology | No. of patients (%) | Mean age (yr) | Cervical cytology | ||||
|---|---|---|---|---|---|---|---|
| Normal (n = 21) | ASCUS (n = 53) | LSIL (n = 66) | HSIL (n = 49) | SCC (n = 37) | |||
| Normal | 57 (25.2%) | 37.8 | 21 (100%) | 17 (32.1%) | 19 (28.8%) | 0 (0%) | 0 (0%) |
| CIN1 | 42 (18.6%) | 37.3 | 0 (0%) | 10 (18.9%) | 30 (45.5%) | 2 (4.1%) | 0 (0%) |
| CIN2/3 | 79 (35.0%) | 38.6 | 0 (0%) | 21 (39.6%) | 17 (25.8%) | 37 (75.5%) | 4 (10.8%) |
| SCC | 48 (23.2%) | 52.6 | 0 (0%) | 5 (9.4%) | 0 (0%) | 10 (20.4%) | 33 (89.2%) |
ASCUS, atypical cells of undetermined significance; CIN, cervical intraepithelial neoplasia; HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion; SCC, squamous cell carcinoma.
2. Immunocytochemistry of HPV L1 protein
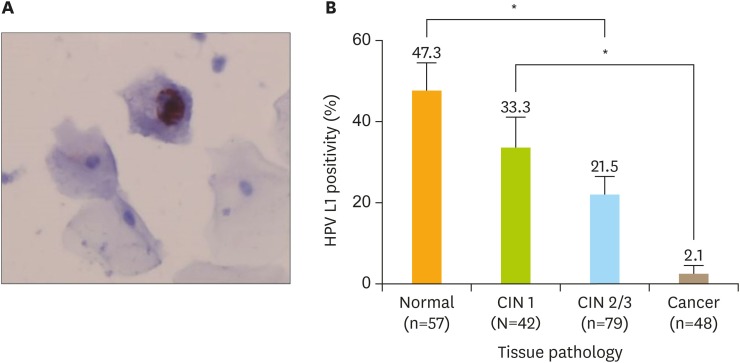
Fig. 1 illustrates HPV L1 immunoreactivity of HPV16-positive cervical lesions. The red nucleus in Fig. 1A indicates the synthesis of L1 structural protein as a result of the productive infection. We found that cervical lesions with higher grades were associated with lower percentages of L1 positivity (Fig. 1B). Of 57 cervical tissue specimens with normal pathology, 27 (47.3%) were L1-positive and 33.3% (14 of 42) of CIN1-graded samples were L1-positive. Moreover, 21.5% (17 of 79) of CIN2/3 and 2.1% (1 of 48) of SCC-graded specimens showed L1 positivity (Fig. 1B). A significant difference was observed between four groups in terms of HPV L1 positivity (p<0.001, ANOVA test). Tukey's multiple comparison test showed that L1 positivity of CIN2/3 was significantly lower than that of normal cervical tissue (p<0.001; 95% confidence interval [CI]=0.071–0.445) and SCC demonstrated significantly lower L1 positivity than CIN1 (p<0.001; 95% CI=0.085–0.539). The immunocytochemistry of HPV L1 protein favorably discriminated CIN2/3 from SCC; however, no statistical significance was observed for the distinction power (p=0.052). Furthermore, HPV L1 immunostaining failed to effectively differentiate CIN2/3 from CIN1 (p=0.440).
Fig. 1.
HPV L1 immunoreactivity in HPV16-positive cervical cytology. (A) Positivity of L1 capsid protein in a LSIL; note the presence of strong nuclear staining. The red nucleus represents L1 positivity (original magnification, ×400). B) HPV L1 positivity by cervical lesion.
CIN, cervical intraepithelial neoplasia; HPV, human papillomavirus; LSIL, low-grade squamous intraepithelial lesion.
*Asterisks indicate cervical lesions with significantly different percentages of L1 positivity.
3. The physical status of HPV integration in cytological samples
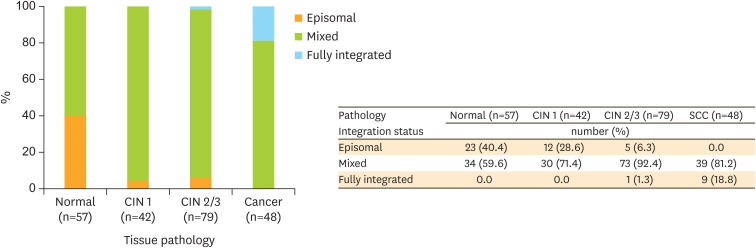
The analysis of the physical status of HPV integration in 226 samples revealed that the proportion of episome, mixed, and fully integrated state was 17.7%, 77.9%, and 4.4%, respectively (Fig. 2). The episomal form of HPV was found in 40.4% (23 of 57) of normal pathology but undetected in SCC, whereas the fully integrated form of HPV was noted in 18.8% (9 of 48) of SCC but undetected in normal pathology and CIN1. The categories of the physical status of HPV exhibited a significant linear association with the degrees of cervical pathology (linear by linear association analysis, p<0.01).
Fig. 2.
The physical status of HPV integration in HPV16-positive cervical lesions (normal, CIN1, CIN2/3, and SCC). A linear association was observed between the status of HPV integration and degrees of cervical lesions.
CIN, cervical intraepithelial neoplasia; HPV, human papillomavirus; SCC, squamous cell carcinoma.
4. Characteristics of E2/E6 ratios by cervical lesions
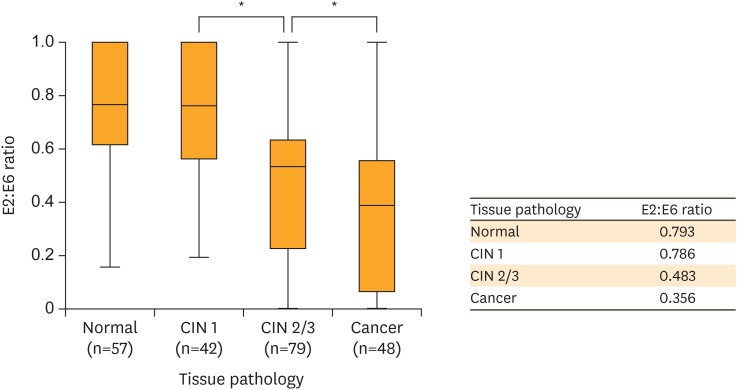
We observed a significant difference in means of E2/E6 ratios in four groups (p<0.001, ANOVA test) (Fig. 3). No significant difference was observed between normal pathology and CIN1 in terms of means of E2/E6 ratio (p=0.999). In particular, E2/E6 ratios of CIN2/3-graded (mean, 0.483) and SCC-graded (mean, 0.356) specimens were significantly lower than those of specimens determined to be normal (mean, 0.793) or graded as CIN1 (mean, 0.786) (p<0.05). Furthermore, E2/E6 ratios of CIN2/3 and SCC specimens were significantly different (p<0.05).
Fig. 3.
E2/E6 ratio in HPV16-positive cervical lesions (normal, CIN1, CIN2/3, and SCC).
CIN, cervical intraepithelial neoplasia; HPV, human papillomavirus; SCC, squamous cell carcinoma.
*Asterisks indicate cervical lesions with significantly different E2/E6 ratios.
5. Prediction of E2/E6 ratio and L1 immunocytochemistry for ≥CIN2 lesions
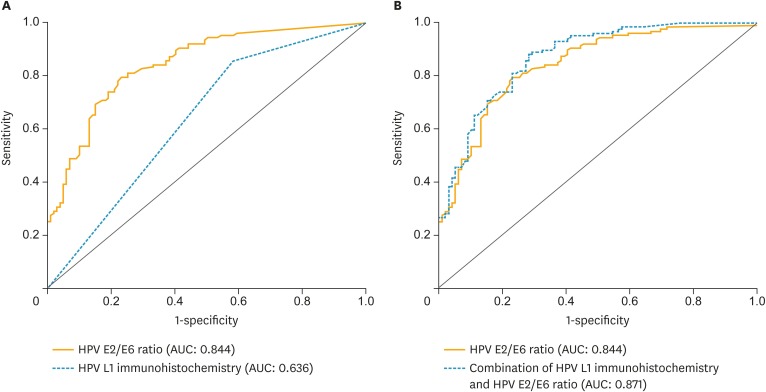
To predict ≥CIN2 lesions, ROC analyses of E2/E6 ratios and presence of HPV L1 were performed; immunocytochemistry and HPV integration tests were evaluated individually and in combination (Fig. 4). We observed that AUC for E2/E6 ratio (0.844; 95% CI=0.793–0.895) was higher than that for L1 immunocytochemistry (0.636; 95% CI=0.562–0.711) (Fig. 4A). A combination of E2/E6 ratio and L1 immunocytochemistry analyses showed the highest AUC (0.871; 95% CI=0.826–0.917) for the prediction of ≥CIN2 lesions (Fig. 4B). The sensitivity of HPV E2/E6 ratio for women with ≥CIN2 cervical lesions was 69.3% and specificity was 84.8%. The sensitivity and specificity of combination of HPV E2/E6 ratio and HPV L1 immunocytochemistry in the differential diagnosis of ≥CIN2 cervical lesions was 99.2% and 31.3%, respectively (Supplementary Table 1).
Fig. 4.
(A) E2/E6 ratio and immunocytochemical staining of HPV L1 and (B) the combination 287 of both analyses; results are expressed as AUC.
AUC, area under the receiver operating characteristic curve; HPV, human papillomavirus.
DISCUSSION
This study demonstrated that cervical cytology samples negative for HPV L1 immunocytochemistry in combination with a decreased E2/E6 ratio showed significant association with CIN2/3 and cancer. Hence, our results may provide information of CIN2/3 and cancer with less invasive methods.
Cervical cytology has been routinely practiced as a screening tool for the detection of abnormal cervical lesion [12]. However, results of cervical cytology may not always match with those of cervical tissue analyses [13]. For instance, patients with low-grade cervical cytology may exhibit unexpectedly higher grade CIN. In ASCUS-LSIL Triage Study (ALTS) trial, 15% LSIL and 9% ASCUS showed higher grade CIN [14]. In this study, 25.8% LSIL showed ≥CIN2 cervical lesions and this was higher than that reported in the ALTS trial. Higher progression for ≥CIN2 among LSIL may be explained by the effect of specific HPV genotypes such as HPV16 during infection. Moreover, of all high-risk HPV types, HPV16 showed the strongest oncogenic capacity for development of cervical cancer. In concordance with our results, Khan et al. [15] reported that HPV type 16 was significantly associated with a higher incidence of CIN3 in LSIL. Various biomarkers were developed to reduce the discrepancy between cervical cytology and pathology and identify patients at the risk of developing higher grade pathology (Table 2) [6,8,9,10,16,17,18,19,20,21,22].
Table 2. Biomarkers in order to demonstrate biologic function of cervical disease.
| Methods | Authors | Number | Markers | Diagnosis | Key findings |
|---|---|---|---|---|---|
| Immunocytochemistry | Lee et al. [8] | 475 | HPV L1 | ASCUS, LSIL, HSIL, cancer | HPV L1 negativity is detected more frequently in HSIL and cancer than ASCUS and LSIL. |
| Melsheimer et al. [10] | 74 | HPV L1 | LSIL, HSIL | HPV L1 negativity is observed more frequently in HSIL than LSIL. | |
| Immunohistochemistry | Waxman et al. [16] | N/A | p16 | CIN1–3 | P16 is useful to discriminate HSILs from benign lesions. |
| Cao et al. [17] | 72 | HPV L1, CK7 | CIN 1 | HPV L1 negativity and CK7 positivity show higher progression rate to CIN2. | |
| Chen et al. [18] | 75 | HPV L1, p16, Ki-67 | CIN 1–3 | HPV L1 negativity was related with high-grade CIN. | |
| Negri et al. [19] | 66 | HPV L1, p16 | CIN1, CIN3 | HPV L1 negativity and p16 positivity shows poor prognosis | |
| Yemelyanova et al. [20] | 212 | HPV L1, L2 | CIN1–3 | HPV L1 and L2 negativity are found more frequently in high grade CIN. | |
| HPV genome PCR | Cricca et al. [6] | 166 | HPV E2/E6 ratio | CIN1–3, Cancer | No full integration status is found in CIN1 and no episomal status is found in cancer. |
| Peitsaro et al. [21] | 31 | HPV E2/E6 ratio | Normal, CIN1–3 | HPV integration status shows progression to high-grade CIN. | |
| Cheung et al. [22] | 78 | HPV E2/E7 ratio | Normal, CIN1–3, Cancer | HPV integration is frequently detected in normal cervix and CIN1. |
ASCUS, atypical cells of undetermined significance; CIN, cervical intraepithelial neoplasia; HSIL, high-grade squamous intraepithelial lesion; HPV, human papillomavirus; LSIL, low-grade squamous intraepithelial lesion; N/A, Not applicable; PCR, polymerase chain reaction.
The shell of HPV particle comprises 2 coated proteins (L1 and L2) that are crucial in mediating efficient viral infectivity. L1 capsid protein constitutes the primary structural element of infectious virions; 360 copies of L1 are organized into 72 capsomeres [23]. L1 represents approximately 90% of the total protein on the viral surface and is typically detectable during productive life cycle. Of all HPV proteins, L1 is most frequently targeted by the humoral immune response [10,24] and detected by neutralizing antibodies produced by HPV preventive vaccine [25]. The production of HPV L1 protein is halted after the integration of HPV into the host's genome, owing to the loss of HPV L1 gene in the infected cell [24]. It is interesting that HPV L1 capsid protein is predominantly detected in mild cervical dysplasia and rarely found in severe cervical dysplasia; L1 is undetectable in cervical carcinoma [9,10]. These results support the hypothesis that HPV L1 negativity is associated with immunologic evasion in high-grade cervical lesions and that L1 positivity may be an indicator of defense status in HPV infections [9,11].
We determined that 78.5% of specimens graded as CIN2/3 and 97.9% of SCC were negative for L1 protein (Fig. 1B). The expression rate of L1 immunocytochemistry was apparently lower in CIN2/3 and SCC than normal pathology and CIN1. Our results were consistent with those of the previous HPV L1 immunohistochemistry studies that showed an association between HPV-L1 negativity and high-grade CIN [18,19]. In the present study, HPV L1 immunostaining could effectively differentiate CIN2/3 from the normal tissue and SCC from CIN1; however, it failed to clearly discriminate CIN2/3 from CIN1. Immunocytochemistry alone was considered insufficient to predict ≥CIN2 cervical lesions. Therefore, an alternative adjuvant method was necessary to enhance the differentiation accuracy of CIN2/3 from CIN1.
Human papillomavirus infects cells of the cervical epithelium, and lesions progress to cervical pre-malignancy (e.g., CIN1 and CIN2/3) and cancer through epigenetic and genetic changes [26,27]. In benign HPV lesions and low-grade cervical lesions, viral sequences appear to be present only as episomes [24]. In contrast, viral DNA is integrated into the host genome in virtually all cases of cervical carcinoma [28]. The integration of HPV DNA into that of the host cell usually results in the disruption of E2 open reading frame (ORF) of HPV, which is important in the regulation of viral transcription. In addition, methylation of E2 was suggested to be related with the dysfunction of E2 and HPV integration into the host genome [29]. The elimination of E2 expression leads to augmented transcription of E6 and E7 oncogenes and is thought to confer a selective growth advantage to the infected cell. E6 and E7 interfere with the normal cell cycle by targeting p53 and pRb tumor suppressors, respectively [30]. Consistent expression of E6 and E7 is required for the progression of CIN and cervical cancer [31].
The physical status of HPV integration was reported to be different as per the severity of the cervical disease; episomal state was more frequently found in CIN, whereas full integration of HPV occurred more frequently in SCC [32]. However, there exists discrepancy in the degrees of the physical status of HPV integration in CIN [33]. Although the mixed form of HPV integration was most commonly observed in all groups in our study, increased prevalence of full integration was observed in CIN2/3 and SCC. In addition, a significant linear association was observed between the physical status of HPV and severity of cervical disease. Therefore, our findings support the association between full integration and carcinogenesis to SCC.
Human papillomavirus integration has been determined with the aid of RNA in situ hybridization (ISH), DNA ISH, Southern blotting, and quantitative real-time PCR [33]. In the present study, the physical status of HPV integration was detected with real-time PCR. This technique could efficiently interpret the status of HPV integration irrespective of the transcriptional activity, which may be variable in LSIL and HSIL [33]. PCR analysis showed that E1/E6 or E2/E6 ratio of HPV16 was significantly distinct according to the severity of cervical lesions and is regarded as a surrogate marker for HPV integration in cervical tissues; full integration is thought to be related to the progression of cervical cancer [34]. We determined that E2/E6 ratio is predictive of ≥CIN2 lesions and allows discrimination of cancer from pre-malignancy (Fig. 3). We also found that E2/E6 ratio is similar for specimens graded as normal and CIN1. Thus, the measurement of E2/E6 ratio, which reflects the integration status of HPV16 in cervical lesions, may be valuable for discriminating normal and low-grade (CIN1) lesions from high-grade (≥CIN2) lesions. In contrast to our data, Boulet et al. [35] suggested that HPV viral marker, HPV viral load or E2/E6 ratio, alone was insufficient to exhibit good diagnostic accuracy for ≥CIN2 lesions (ROC of E2/E6 ratio, 0.58). We agree with this opinion because E2/E6 ratio of HPV integration acts as the marker only for the early phase of carcinogenesis of cervical cancer. Hence, an additional marker may be necessary for the evaluation of later stages of HPV integration. Herein, we combined immunocytochemistry of HPV L1 capsid protein and E2/E6 ratio using cervical cytology sample.
To the best of our knowledge, this is the first study to validate the utility of the combination of cervical cytology and viral marker of HPV integration. We utilized HPV L1 protein expression and integration method such as E2/E6 ratio for the diagnosis of ≥CIN2 cervical lesions (Fig. 4). The accuracy of the identification of ≥CIN2 cervical lesions was increased in the combined analysis versus measurement of E2/E6 ratio or L1 immunocytochemistry alone (AUC=0.871, 0.844, and 0.636, respectively).
As a result of the steady efforts to develop cervical markers [5,16,18,19], recommendation at Lower Anogenital Squamous Terminology (LAST) project in 2012 suggested that p16 immunostaining positivity may specifically diagnose CIN2 as high-grade squamous intraepithelial lesions [16]. In addition, immunohistochemical positivity of p16 may predict higher risk for progression in low-grade lesions. An increase in Ki-67 expression was found in CIN, suggestive of its potential as the proliferation maker for the discrimination between LSIL and normal tissue. However, the indirect correlation between the combined markers has a limitation to explain the progression or regression phenotype of cervical diseases. To overcome this limitation, we utilized negativity in HPV L1 immunocytochemistry, which is considered to be the direct consequence of HPV integration into the host genome.
This study has two key findings. First, we examined the association of L1 expression and HPV integration status (E2/E6 ratio) with grades of cervical lesions using noninvasive cervical cytology. Our results may provide the opportunity for the early diagnosis of ≥CIN2 cervical lesions at the stage of cervical cytology. Second, we attempted, for the first time, to evaluate the feasibility of a combined analysis of L1 detection and E2/E6 measurement as a predictive combined biomarker of ≥CIN2. Our findings indicate that cervical cytology was suitable for analyses of HPV L1 protein and E2/E6 ratio and that the combined analysis serves as a better platform for the prediction of cervical lesions as compared with the individual analysis.
In conclusion, our results indicate that the lack of HPV L1 expression and decreased E2/E6 ratio in HPV DNA are strongly associated with a higher grade of cervical disease. We advocate cervical cytology from noninvasive procedures and combined measurement of E2/E6 ratio and HPV L1 immunocytochemistry as a suitable means to predict ≥CIN2 cervical lesions. Our findings may provide a useful basis for the accurate cytological diagnosis of cervical pre-malignancy and cancer in the future translational research.
ACKNOWLEDGMENTS
The authors would like to thank Min-Jin Jung, Eun-Young Ki, Jin-Hwi Kim, and Soo-Young Hur for their excellent assistance in this work.
Footnotes
Funding: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI15C2915) and by the St. Vincent's Hospital, research institute of medical science (SVHR-2015-9).
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
- Conceptualization: C.Y.J., K.T.J.
- Data curation: C.Y.J., L.A.
- Investigation: C.Y.J., J.H.T., S.Y.B.
- Methodology: C.Y.J., L.A., K.T.J., J.H.T., S.Y.B.
- Project administration: C.Y.J., L.S.J.
- Supervision: P.J.S.
- Visualization: C.Y.J., L.S.J.
- Writing - original draft: C.Y.J.
- Writing - review & editing: C.Y.J.
SUPPLEMENTARY MATERIAL
Diagnostic accuracy of E2/E6 ratio and HPV L1 immunocytochemistry in the differential diagnosis of ≥CIN2 cervical lesions
References
- 1.Walboomers JM, Jacobs MV, Manos MM, Bosch FX, Kummer JA, Shah KV, et al. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J Pathol. 1999;189:12–19. doi: 10.1002/(SICI)1096-9896(199909)189:1<12::AID-PATH431>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- 2.Hu Z, Zhu D, Wang W, Li W, Jia W, Zeng X, et al. Genome-wide profiling of HPV integration in cervical cancer identifies clustered genomic hot spots and a potential microhomology-mediated integration mechanism. Nat Genet. 2015;47:158–163. doi: 10.1038/ng.3178. [DOI] [PubMed] [Google Scholar]
- 3.Hosaka M, Fujita H, Hanley SJ, Sasaki T, Shirakawa Y, Abiko M, et al. Incidence risk of cervical intraepithelial neoplasia 3 or more severe lesions is a function of human papillomavirus genotypes and severity of cytological and histological abnormalities in adult Japanese women. Int J Cancer. 2013;132:327–334. doi: 10.1002/ijc.27680. [DOI] [PubMed] [Google Scholar]
- 4.Ebisch RM, van der Horst J, Hermsen M, Rijstenberg LL, Vedder JE, Bulten J, et al. Evaluation of p16/Ki-67 dual-stained cytology as triage test for high-risk human papillomavirus-positive women. Mod Pathol. 2017;30:1021–1031. doi: 10.1038/modpathol.2017.16. [DOI] [PubMed] [Google Scholar]
- 5.Nam EJ, Kim JW, Hong JW, Jang HS, Lee SY, Jang SY, et al. Expression of the p16 and Ki-67 in relation to the grade of cervical intraepithelial neoplasia and high-risk human papillomavirus infection. J Gynecol Oncol. 2008;19:162–168. doi: 10.3802/jgo.2008.19.3.162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cricca M, Morselli-Labate AM, Venturoli S, Ambretti S, Gentilomi GA, Gallinella G, et al. Viral DNA load, physical status and E2/E6 ratio as markers to grade HPV16 positive women for high-grade cervical lesions. Gynecol Oncol. 2007;106:549–557. doi: 10.1016/j.ygyno.2007.05.004. [DOI] [PubMed] [Google Scholar]
- 7.Lukaszuk K, Liss J, Wozniak I, Emerich J, Wójcikowski C. Human papillomavirus type 16 status in cervical carcinoma cell DNA assayed by multiplex PCR. J Clin Microbiol. 2003;41:608–612. doi: 10.1128/JCM.41.2.608-612.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee SJ, Lee AW, Kang CS, Park JS, Park DC, Ki EY, et al. Clinicopathological implications of human papilloma virus (HPV) L1 capsid protein immunoreactivity in HPV16-positive cervical cytology. Int J Med Sci. 2013;11:80–86. doi: 10.7150/ijms.5585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee SJ, Lee AW, Kim TJ, Kim JH, Bae JH, Lee CW, et al. Correlation between immunocytochemistry of human papilloma virus L1 capsid protein and behavior of low-grade cervical cytology in Korean women. J Obstet Gynaecol Res. 2011;37:1222–1228. doi: 10.1111/j.1447-0756.2010.01506.x. [DOI] [PubMed] [Google Scholar]
- 10.Melsheimer P, Kaul S, Dobeck S, Bastert G. Immunocytochemical detection of HPV high-risk type L1 capsid proteins in LSIL and HSIL as compared with detection of HPV L1 DNA. Acta Cytol. 2003;47:124–128. doi: 10.1159/000326491. [DOI] [PubMed] [Google Scholar]
- 11.Steele JC, Roberts S, Rookes SM, Gallimore PH. Detection of CD4(+)- and CD8(+)-T-cell responses to human papillomavirus type 1 antigens expressed at various stages of the virus life cycle by using an enzyme-linked immunospot assay of gamma interferon release. J Virol. 2002;76:6027–6036. doi: 10.1128/JVI.76.12.6027-6036.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Badaracco G, Venuti A, Sedati A, Marcante ML. HPV16 and HPV18 in genital tumors: significantly different levels of viral integration and correlation to tumor invasiveness. J Med Virol. 2002;67:574–582. doi: 10.1002/jmv.10141. [DOI] [PubMed] [Google Scholar]
- 13.Chacho MS, Mattie ME, Schwartz PE. Cytohistologic correlation rates between conventional Papanicolaou smears and ThinPrep cervical cytology: a comparison. Cancer. 2003;99:135–140. doi: 10.1002/cncr.11102. [DOI] [PubMed] [Google Scholar]
- 14.Schiffman M, Solomon D. Findings to date from the ASCUS-LSIL Triage Study (ALTS) Arch Pathol Lab Med. 2003;127:946–949. doi: 10.5858/2003-127-946-FTDFTA. [DOI] [PubMed] [Google Scholar]
- 15.Khan MJ, Castle PE, Lorincz AT, Wacholder S, Sherman M, Scott DR, et al. The elevated 10-year risk of cervical precancer and cancer in women with human papillomavirus (HPV) type 16 or 18 and the possible utility of type-specific HPV testing in clinical practice. J Natl Cancer Inst. 2005;97:1072–1079. doi: 10.1093/jnci/dji187. [DOI] [PubMed] [Google Scholar]
- 16.Waxman AG, Chelmow D, Darragh TM, Lawson H, Moscicki AB. Revised terminology for cervical histopathology and its implications for management of high-grade squamous intraepithelial lesions of the cervix. Obstet Gynecol. 2012;120:1465–1471. doi: 10.1097/aog.0b013e31827001d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cao L, Sun PL, Yao M, Chen S, Gao H. Clinical significance of CK7, HPV-L1, and koilocytosis for patients with cervical low-grade squamous intraepithelial lesions: a retrospective analysis. Hum Pathol. 2017;65:194–200. doi: 10.1016/j.humpath.2017.05.017. [DOI] [PubMed] [Google Scholar]
- 18.Chen L, Sun MJ, Shi HY, He Q, Liu DG. Association of human papillomavirus L1 capsid protein with koilocytosis, expression of p16, and Ki-67, and its potential as a prognostic marker for cervical intraepithelial neoplasia. Anal Quant Cytopathol Histpathol. 2013;35:139–145. [PubMed] [Google Scholar]
- 19.Negri G, Bellisano G, Zannoni GF, Rivasi F, Kasal A, Vittadello F, et al. p16 ink4a and HPV L1 immunohistochemistry is helpful for estimating the behavior of low-grade dysplastic lesions of the cervix uteri. Am J Surg Pathol. 2008;32:1715–1720. doi: 10.1097/PAS.0b013e3181709fbf. [DOI] [PubMed] [Google Scholar]
- 20.Yemelyanova A, Gravitt PE, Ronnett BM, Rositch AF, Ogurtsova A, Seidman J, et al. Immunohistochemical detection of human papillomavirus capsid proteins L1 and L2 in squamous intraepithelial lesions: potential utility in diagnosis and management. Mod Pathol. 2013;26:268–274. doi: 10.1038/modpathol.2012.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Peitsaro P, Johansson B, Syrjänen S. Integrated human papillomavirus type 16 is frequently found in cervical cancer precursors as demonstrated by a novel quantitative real-time PCR technique. J Clin Microbiol. 2002;40:886–891. doi: 10.1128/JCM.40.3.886-891.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cheung JL, Cheung TH, Ng CW, Yu MY, Wong MC, Siu SS, et al. Analysis of human papillomavirus type 18 load and integration status from low-grade cervical lesion to invasive cervical cancer. J Clin Microbiol. 2009;47:287–293. doi: 10.1128/JCM.01531-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Modis Y, Trus BL, Harrison SC. Atomic model of the papillomavirus capsid. EMBO J. 2002;21:4754–4762. doi: 10.1093/emboj/cdf494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Doorbar J. Molecular biology of human papillomavirus infection and cervical cancer. Clin Sci (Lond) 2006;110:525–541. doi: 10.1042/CS20050369. [DOI] [PubMed] [Google Scholar]
- 25.Lee SJ, Yang A, Wu TC, Hung CF. Immunotherapy for human papillomavirus-associated disease and cervical cancer: review of clinical and translational research. J Gynecol Oncol. 2016;27:e51. doi: 10.3802/jgo.2016.27.e51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Steenbergen RD, Snijders PJ, Heideman DA, Meijer CJ. Clinical implications of (epi)genetic changes in HPV-induced cervical precancerous lesions. Nat Rev Cancer. 2014;14:395–405. doi: 10.1038/nrc3728. [DOI] [PubMed] [Google Scholar]
- 27.Jung SH, Choi YJ, Kim MS, Baek IP, Lee SH, Lee AW, et al. Progression of naive intraepithelial neoplasia genome to aggressive squamous cell carcinoma genome of uterine cervix. Oncotarget. 2015;6:4385–4393. doi: 10.18632/oncotarget.2981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cullen AP, Reid R, Campion M, Lörincz AT. Analysis of the physical state of different human papillomavirus DNAs in intraepithelial and invasive cervical neoplasm. J Virol. 1991;65:606–612. doi: 10.1128/jvi.65.2.606-612.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chaiwongkot A, Vinokurova S, Pientong C, Ekalaksananan T, Kongyingyoes B, Kleebkaow P, et al. Differential methylation of E2 binding sites in episomal and integrated HPV 16 genomes in preinvasive and invasive cervical lesions. Int J Cancer. 2013;132:2087–2094. doi: 10.1002/ijc.27906. [DOI] [PubMed] [Google Scholar]
- 30.Vousden K. Interactions of human papillomavirus transforming proteins with the products of tumor suppressor genes. FASEB J. 1993;7:872–879. doi: 10.1096/fasebj.7.10.8393818. [DOI] [PubMed] [Google Scholar]
- 31.Kessis TD, Connolly DC, Hedrick L, Cho KR. Expression of HPV16 E6 or E7 increases integration of foreign DNA. Oncogene. 1996;13:427–431. [PubMed] [Google Scholar]
- 32.Hudelist G, Manavi M, Pischinger KI, Watkins-Riedel T, Singer CF, Kubista E, et al. Physical state and expression of HPV DNA in benign and dysplastic cervical tissue: different levels of viral integration are correlated with lesion grade. Gynecol Oncol. 2004;92:873–880. doi: 10.1016/j.ygyno.2003.11.035. [DOI] [PubMed] [Google Scholar]
- 33.Pett M, Coleman N. Integration of high-risk human papillomavirus: a key event in cervical carcinogenesis? J Pathol. 2007;212:356–367. doi: 10.1002/path.2192. [DOI] [PubMed] [Google Scholar]
- 34.Tsakogiannis D, Kyriakopoulou Z, Ruether IG, Amoutzias GD, Dimitriou TG, Diamantidou V, et al. Determination of human papillomavirus 16 physical status through E1/E6 and E2/E6 ratio analysis. J Med Microbiol. 2014;63:1716–1723. doi: 10.1099/jmm.0.076810-0. [DOI] [PubMed] [Google Scholar]
- 35.Boulet GA, Benoy IH, Depuydt CE, Horvath CA, Aerts M, Hens N, et al. Human papillomavirus 16 load and E2/E6 ratio in HPV16-positive women: biomarkers for cervical intraepithelial neoplasia >or=2 in a liquid-based cytology setting? Cancer Epidemiol Biomarkers Prev. 2009;18:2992–2999. doi: 10.1158/1055-9965.EPI-09-0025. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Diagnostic accuracy of E2/E6 ratio and HPV L1 immunocytochemistry in the differential diagnosis of ≥CIN2 cervical lesions