Abstract
It is not known whether pylorus-preserving pancreaticoduodenectomy (PPPD) is as effective as the standard pancreaticoduodenectomy (SPD) in the treatment of duodenal papilla carcinoma (DPC). A retrospective cohort trial was undertaken to compare the results of these two procedures. Clinical data, histological findings, short-term results, survival and quality of life of all patients who had undergone surgery for primary DPC between January 2003 and February 2010 were analyzed. According to the inclusion criteria and the surgical methods, 116 patients were divided into the PPPD group (n=43) and the SPD group (n=73). There were no significant differences in various indices, including surgery duration, extent of intraoperative hemorrhage and postoperative pathological indexes. The incidence of postoperative complications, including pancreatic fistula and delayed gastric emptying, were also similar between the two groups (20.9 vs. 21.9%; P=0.900 and 11.6 vs. 5.4%; P=0.402). Long-term survival and quality of life were identical following a median follow-up of 45.6 months (range, 4–144 months). Within 6 months, there was a decreased loss of appetite following the pylorus-preserving procedure (26.9 vs. 49.3; P=0.003). The procedures were equally effective for the treatment of DPC. PPPD offers minor advantages in the early postoperative period but not in the long term.
Keywords: duodenal papilla carcinoma, standard pancreaticoduodenectomy, pylorus-preserving pancreaticod-uodenectomy, survival, quality of life
Introduction
Duodenal papilla carcinoma (DPC) is a relatively uncommon type of cancer, with a prevalence of 0.01% among all clinically diagnosed malignant tumors (1). In total, ~60% of patients with primary malignant tumors of the duodenum are diagnosed with DPC (2). The majority of studies have demonstrated that, compared with other malignant periampullary carcinomas and malignant tumors of the pancreas, DPC has higher rates of excision and improved prognosis (3–5). Therefore, standard pancreaticoduodenectomy (SPD) remains the primary treatment for DPC.
SPD is a relatively complex surgery for the treatment of malignant tumors of the digestive system. The incidence of early complications following SPD surgery is as high as 16.7–34.1%, of which postoperative pancreatic fistula is the most common (6,7). Although the individual risk of complications following SPD has gradually decreased (8), surgeons are still required to improve the method of SPD in order to enhance efficacy.
The first pylorus-preserving pancreaticoduodenectomy (PPPD) was performed on a patient with ampullary carcinoma in 1944 (9). After almost 70 years of development, PPPD is now increasingly recognized as a treatment option by surgeons (10,11). Compared with SPD, PPPD preserves the storage and digestive functions of the stomach and reduces the postoperative complications of partial gastrectomy, thereby potentially improving the postoperative quality of life of the patients (12–16). However, the majority of reports in the literature focus on periampullary carcinoma and carcinoma of the pancreas head, making clinical contrast analyses of the two surgical methods for DPC unreliable.
Therefore, in the present study, a single-center prospective clinical trial was performed in order to validate the efficacy and safety of PPPD and SPD procedures for the treatment of patients with DPC through various therapeutic approaches at different postoperative treatment points.
Materials and methods
General information
Patients who had undergone SPD or PPPD for primary DPC at the People's Liberation Army (PLA) General Hospital (Beijing, China) between January 2003 and February 2010 were eligible for participation in the present study. Patients were excluded if they had locally advanced DPC or metastases, advanced liver cirrhosis (Child-Pugh class B or C) (17) with portal hypertension, malnutrition or coagulopathy. A total of 116 patients were divided into the PPPD group (n=43) and the SPD group (n=73). The cohort comprised 76 males and 40 females with an average age of 58.1 years (range, 36–77 years). Written informed consent for treatment was obtained from all patients and included a detailed explanation of the nature of the disease, possible treatment methods and potential postoperative complications. The present study was approved by the ethics committee of the PLA General Hospital and was performed in accordance with the ethical standards specified in The 1964 Declaration of Helsinki and its later amendments.
Surgical conditions
Prophylactic antibiotics were administered 0.5–1 h prior to surgery and, if the surgery duration exceeded 3 h, an additional dose was administered. The excision range of the SPD surgery included the following: 40% distal stomach, duodenum and 10 cm of the jejunum from the Treitz's ligament; the gallbladder and common bile duct (proximal end to the crotch of the left and right hepatic ducts); the head, neck and uncinate process of the pancreas; and the lymph nodes and adipose tissues in the excision area. The reconstruction of the digestive tract included the following: Pancreatico enteric anastomosis (anastomosis of the pancreatic duct and jejunal mucous membrane, with a drainage tube placed into the pancreatic duct); cholecysto-colonic anastomosis; gastrojejunostomy anastomosis; and, for all patients, jejunum-jejunum side-to-side anastomosis (Braun's anastomosis) was performed under the gastrojejunostomy anastomosis. PPPD is different from SPD in that the distal end of the stomach was preserved in PPPD. The excision began 2 cm below the pylorus, and the lymph nodes at the corresponding lesser curvature of the stomach and those above and under the pylorus were also preserved.
Postoperative treatment
For all patients, conventional postoperative treatment of the pancreas was performed. Prior to returning to their wards, patients were observed for ≥1 day in the intensive care unit. Following the surgery, 100 µg octreotide was subcutaneously administered to all patients three times daily for 7 days. On the second day after surgery, routine blood, liver and kidney function tests were performed. On the third day after surgery, an abdominal color Doppler ultrasound examination was performed. At 7 days following the surgery, an abdominal computed tomography (CT) examination was performed to observe the conditions of the abdomen. Prior to the end of the surgery, a drainage tube was inserted into the pancreatico enteric anastomosis, cholecystocolonic anastomosis and gastrojejunostomy anastomosis, and the volume, color and appearance of the drained fluid were recorded daily.
Following the surgery, the pancreatico enteric drainage tube was removed when the amylase level was <300 U/l inside the drainage tube (<2× the serum amylase level), the drainage amount was <50 ml each day, or the drainage duration was >10 days following surgery (15).
According to the diagnostic criteria defined by the International Study Group on Pancreatic Fistula (18), a pancreatic fistula was defined as follows: present ≥3 days following surgery, the draining liquid could be measured and the activity of amylase was 3× that of the serum amylase activity. Pancreatic fistulas were designated one of three grades (Grades A, B and C) according to the clinical events of the patient hospitalization. Grade A pancreatic fistulas required no change from the normal clinical approach, did not delay discharge and could usually be resolved by the removal of the retained surgical drainage tube. Grade B pancreatic fistulas required a change of treatment strategy or adjustment of the clinical approach (for instance, fasting, total parenteral nutritional support or the addition of antibiotics or somatostatin), delayed discharge or required readmission for treatment following discharge. If, according to the pathogenetic conditions of the patient, invasive procedures were required, the grade of the pancreatic fistula was upgraded to Grade C. Grade C pancreatic fistulas required a significant change in treatment strategy or adjustment of clinical approach. If clinical symptoms were aggravated and there were complications including sepsis and organ dysfunction, investigation through reoperation may be required. Grade C pancreatic fistulas were often accompanied by complications, leading to an increased probability of postoperative mortality.
Biliary fistula was diagnosed if there was persistent secretion of >50 ml/day bilirubin-rich drainage fluid or if secretion continued after the 10th postoperative day (15).
Postoperative bleeding was defined as the requirement for >2 units of red blood cells >2.4 h after surgery or relaparotomy for bleeding (15).
The nasogastric tube was removed when the drainage decreased to <200 ml/24 h (15). Delayed gastric emptying was defined as gastric stasis requiring nasogastric intubation for ≥10 days or the inability to tolerate a regular diet on the 14th postoperative day (19).
Pathological analysis
Primary DPC was confirmed by two independent pathologists (Pathology department, People's Liberation Army General Hospital, Beijing, China) through examination of all the pathological sections. Grading of tumors was determined according to the Union for International Cancer Control Tumor-Node-Metastasis (TNM) grading system (20). Strict pathological examination was performed on the margin of the tumors, including the neck of the pancreas, uncinate process, bile duct, duodenum/stomach, portal vein and mesenteric arteries, in order to ensure R0 excision. In addition, a detailed assessment regarding whether or not DPC involved the bile duct, pancreas, nerves or lymph nodes, was performed.
Follow-up
Telephone and outpatient follow-ups were performed. The patients were followed up once every 3 months for 1 year following the surgery, once every 6 months for the 2nd and 3rd years, and once every year for 4–6 years following the surgery. The final date for any follow-up was August 2016. Follow-up observations included the survival of the patients and whether there was progression or recurrence. Tumor recurrence was re-examined by CT and magnetic resonance imaging.
For the analysis of postoperative quality of life, the European Organization for Research and Treatment of Cancer (EORTC-QLQ-C30) (19) questionnaire was used to assess the quality of life of patients 3, 6, 12 and 24 months after surgery. The EORTC QLQ-C30 consists of a total of 30 items, including 15 domains, of which there were 5 function-based subscales (physical, role, cognitive, emotional and social functioning), 3 symptom subscales (fatigue, pain, and nausea and vomiting), a whole health condition subscale and 6 single items. Role functions, included daily activities, work, and whether hobbies were restricted or not. The answers were then translated into scores between 0 and 100, according to the scoring manual provided by the EORTC.
Statistical analysis
Statistical analysis was performed using SPSS software 16.0 (SPSS, Inc., Chicago, IL, USA). Continuous variables are presented as the mean values and ranges, and categorical variables are presented as percentages. Differences between continuous variables were determined using the Mann-Whitney U test or Student's t-test, and differences between categorical variables were determined using χ2 or Fisher's exact tests (where n<5). The Kaplan-Meier method was used to analyze survival and levels of significance were determined using the log-rank test. P<0.05 was considered to indicate a statistically significant difference.
Results
A total of 116 patients, including 43 patients in the PPPD group and 73 in the SPD group (comprising 76 males and 40 females with an average age of 58.1 years), were enrolled in the present study. No significant differences were identified in the various indices prior to surgery between the two groups of patients, including sex, age, symptoms, comorbidities (combination with other diseases), body mass index, serum albumin cancer antigen 19-9 and carcinoembryonic antigen (Table I).
Table I.
Patient characteristics.
| Characteristic | Total (%) | PPPD (%) | SPD (%) | P-value |
|---|---|---|---|---|
| Sex | ||||
| Male | 76 (66.5) | 25 (58.1) | 51 (69.8) | 0.199a |
| Female | 40 (34.5) | 18 (41.8) | 22 (30.1) | 0.199a |
| Age, years | 58.1 (36–77)c | 58.1 (41–77)c | 58.1 (36–75)c | 0.812b |
| Symptoms | ||||
| Jaundiced | 60 (51.7) | 19 (44.1) | 41 (56.1) | 0.212a |
| AP | 18 (15.5) | 7 (16.2) | 11 (15.1) | 0.861a |
| Jaundice with AP | 15 (12.9) | 6 (13.9) | 9 (12.3) | 0.801a |
| Fever | 6 (5.2) | 2 (4.6) | 4 (5.4) | 0.810a |
| Fever with AP | 4 (3.4) | 2 (4.6) | 2 (2.7) | 0.985a |
| Cholangitis | 4 (3.4) | 2 (4.6) | 2 (2.7) | 0.985a |
| Melena | 2 (1.7) | 1 (2.3) | 1 (1.3) | 0.721a |
| Identified in PE | 2 (1.7) | 2 (4.6) | 0 (0.0) | 0.262a |
| Emaciation | 2 (1.7) | 1 (2.3) | 1 (1.3) | 0.721a |
| Others | 3 (2.6) | 1 (2.3) | 2 (2.7) | 0.638a |
| Combined diseases | ||||
| Weight loss | 29 (25.0) | 10 (23.2) | 19 (26.0) | 0.739a |
| Hypertension | 20 (17.2) | 10 (23.2) | 10 (13.7) | 0.188a |
| CHD | 12 (10.3) | 4 (9.3) | 8 (10.9) | 0.973a |
| Diabetes | 9 (7.8) | 4 (9.3) | 5 (6.8) | 0.906a |
| BMI (kg/m2) | 24.4 (18.4–29.1)c | 24.5 (19.7–27.6)c | 24.3 (18.4–29.1)c | 0.804b |
| Serum albumin (<35 g/l) | 35 (30.2) | 14 (32.5) | 21 (28.7) | 0.667a |
| CA19-9 (>100 U/ml) | 39 (33.6) | 14 (32.5) | 25 (34.2) | 0.852a |
| CEA (>5 ng/ml) | 15 (12.9) | 4 (9.3) | 11(15.0) | 0.543a |
χ2 test or Fisher's exact test
Mann-Whitney U test.
Values are presented as the median (range), as opposed to the n (%) presentation used for all other data.
Defined as clinical jaundice in combination with a serum bilirubin level >100 µmol/l. AP, abdominal pain; PE, physical examination; CHD, coronary heart disease; BMI, body mass index; PPPD, pylorus-preserving pancreaticoduodenectomy; SPD, standard pancreaticoduodenectomy; CA19-9, cancer antigen 19-9; CEA, carcinoembryonic antigen.
The present study demonstrated that the average surgery duration in the PPPD group was shorter than that in the SPD group (319.2 vs. 331.4 min); however, no statistically significant difference was observed between the two groups (P=0.982). Furthermore, no statistically significant differences were observed in intraoperative blood loss (363.9 vs. 415.0 ml, P=0.893), the diameter of the pancreatic duct (3.5 vs. 3.2 mm, P=0.367) or duration of hospitalization (38.7 vs. 33.5 days, P=0.527) between the two groups of patients (Table II).
Table II.
Intraoperative observations and hospitalization duration.
| Observation | Total | PPPD | SPD | P-value |
|---|---|---|---|---|
| Duration of surgery, min | 326.8 (74.5) | 319.2 (67.9) | 331.4 (8.2) | 0.982 |
| Blood loss, ml | 389.1 (286.5) | 363.9 (196.7) | 415.0 (327.9) | 0.893 |
| Pancreatic duct diameter, mm | 3.3 (1.4) | 3.5 (1.4) | 3.2 (1.5) | 0.367 |
| Hospitalization duration, days | 35.4 (18.2) | 38.7 (25.0) | 33.5 (12.5) | 0.527 |
Data are presented as mean ± (standard deviation). PPPD, pylorus-preserving pancreaticoduodenectomy; SPD, standard pancreaticoduodenectomy.
No statistically significant differences in tumor size, depth of tumor invasion, involvement of bile duct, involvement of the pancreas or surrounding tissues of the pancreas, involvement of nerves, lymph node metastasis, differentiation or TNM stage of the tumor were observed between the two groups of patients (Table III).
Table III.
Pathology.
| Characteristic | Total (%) (n=116) | PPPD (%) (n=116) | SPD (%) (n=116) | P-value, χ2 test or Fisher's exact test |
|---|---|---|---|---|
| Size of mass (≥2.5 cm) | 49 (42.2) | 19 (44.2) | 30 (41.1) | 0.744 |
| Tumor invasion | 0.569 | |||
| Tis/T1 | 15 (12.9) | 8 (18.6) | 7 (9.6) | |
| T2 | 35 (30.2) | 12 (27.9) | 23 (31.5) | |
| T3 | 37 (31.9) | 11 (25.6) | 26 (35.6) | |
| T4 | 29 (25.0) | 12 (27.9) | 17 (23.2) | |
| Involvement of bile duct | 40 (34.5) | 15 (34.9) | 25 (34.2) | 0.944 |
| Involvement of pancreas | 50 (43.1) | 14 (32.5) | 36 (49.3) | 0.078 |
| Involvement of nerves | 5 (4.3) | 1 (2.3) | 4 (5.4) | 0.737 |
| Lymph node metastasis (yes) | 25 (21.6) | 9 (20.9) | 16 (21.9) | 0.900 |
| Numbers of lymph node metastasis | 0.419 | |||
| 1 | 13 (11.2) | 6 (13.9) | 7 (9.6) | |
| 2 | 7 (6.0) | 3 (6.9) | 4 (5.5) | |
| 3 | 2 (1.7) | 0 (0.0) | 2 (2.7) | |
| >3 | 3 (2.6) | 1 (2.3) | 2 (2.7) | |
| Differentiation | 0.699 | |||
| High | 30 (25.9) | 13 (30.2) | 17 (23.3) | |
| Middle-high | 18 (15.5) | 5 (11.6) | 13 (17.8) | |
| Middle | 38 (32.8) | 13 (30.2) | 25 (34.2) | |
| Middle-low | 28 (24.1) | 12 (27.9) | 16 (21.9) | |
| Low | 2 (1.7) | 0 (0.0) | 2 (2.7) | |
| TNM stage | 0.699 | |||
| I | 43 (37.1) | 17 (39.5) | 26 (35.6) | |
| II | 43 (37.1) | 14 (32.5) | 29 (39.7) | |
| III | 30 (25.9) | 12 (27.9) | 18 (24.6) | |
| IV | 0 (0.0) | 0 (0.0) | 0 (0.0) |
Data are presented as n (%). PPPD, pylorus-preserving pancreaticoduodenectomy; SPD, standard pancreaticoduodenectomy; TNM, Tumor-Node-Metastasis.
The most common postoperative complications in the patients receiving PPPD or SPD included pancreatic fistula (21.6%), biliary leakage (1.7%), postoperative bleeding (5.2%) and delayed gastric emptying (7.8%). In the PPPD group, there were 9 cases (9/43, 20.9%) of pancreatic fistula, including 2 cases with Grade A, 3 cases with Grade B and 4 cases with Grade C; while in the SPD group, there were 16 cases (16/73, 21.6%) of pancreatic fistula, including 7 cases with Grade A, 6 cases with Grade B and 3 cases with Grade C. There were no significant differences between the two groups (P=0.900). The incidence of delayed gastric emptying was similar between the two groups of patients, with 5 cases (5/43, 11.6%) in the PPPD group and 4 cases (4/73, 5.4%) in the SPD group (P=0.402). Similarly, there were no significant differences in biliary leakage (P=0.721), postoperative bleeding (P=0.810) or infection (wound or abscess) (P=0.938) between the two groups of patients. Three patients (2.6%) required relaparotomy for significant postoperative bleeding (one patient in each group) and bowel obstruction (one patient who had undergone SPD). The overall operative mortality rate was 0.9%; 1 patient in the PPPD group succumbed to acute cardiac failure within 30 days (Table IV).
Table IV.
Postoperative complications, relaparotomy and mortality.
| Total | PPPD | SPD | P-value | |
|---|---|---|---|---|
| Complications | ||||
| Pancreatic fistulaa | 25 (21.6) | 9 (20.9) | 16 (21.9) | 0.900 |
| A | 9 (7.8) | 2 (4.6) | 7 (9.5) | |
| B | 9 (7.8) | 3 (6.9) | 6 (8.2) | |
| C | 7 (6.0) | 4 (9.3) | 3 (4.1) | |
| Biliary leakage | 2 (1.7) | 0 (0.0) | 2 (2.7) | 0.721 |
| Postoperative bleeding | 6 (5.2) | 2 (4.6) | 4 (5.4) | 0.810 |
| Delayed gastric emptyingb | 9 (7.8) | 5 (11.6) | 4 (5.4) | 0.402 |
| Infection (wound or abscess) | 7 (6.0) | 2 (4.6) | 5 (6.8) | 0.938 |
| Other | 38 (32.8) | 15 (34.8) | 23 (31.5) | 0.708 |
| Relaparotomy | 3 (2.6) | 1 (2.3) | 2 (2.7) | 0.638 |
| Mortalityc | 1 (0.9) | 1 (2.3) | 0 (0.0) | 0.788 |
Defined as ≥3 days after surgery and amylase activity of the draining liquid 3× higher than that of the serum.
Defined as gastric stasis requiring nasogastric intubation for ≥10 days or the inability to tolerate a regular diet on the 14th postoperative day.
Mortality resulting from surgery within 30 days. PPPD, pylorus-preserving pancreaticoduodenectomy; SPD, standard pancreaticoduodenectomy.
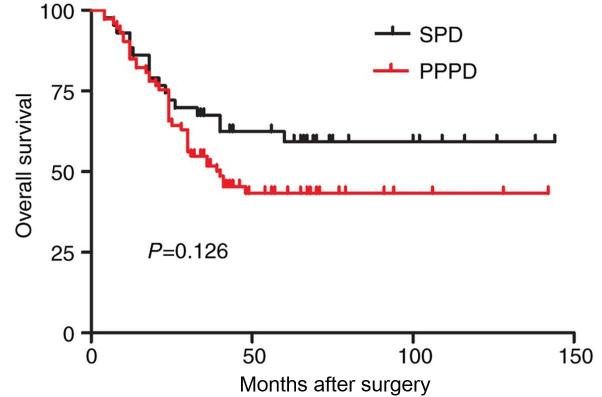
Between the time of surgery and August 2016, all 116 patients underwent follow up. In this cohort of patients, the postoperative survival time was between 4 and 144 months, with a median survival time of 45.6 months. The overall survival rate of the 116 patients following surgery was 49.1%. The median survival time was 55.1 months (range, 4–142 months) in the patients in the PPPD group and 40.1 months (range 4–144 months) in those in the SPD group. No statistically significant differences were observed in the total survival rate between the two groups (P=0.126; Table V; Fig. 1).
Table V.
Overall survival and quality of life analysis.
| Variable | Total | PPPD | SPD | P-value |
|---|---|---|---|---|
| OS | 45.6 (4–144)c | 55.1 (4–142)c | 40.1 (4–144)c | 0.126a |
| OS of positive lymph nodes (EORTC)-QLQ-C30 | 25.2 (4–56)c | 27.4 (4–56)c | 24.0 (4–54)c | 0.592a |
| Physical functioning | ||||
| 6 months | 49.8 (18.2) | 53.6 (16.2) | 47.7 (19.1) | 0.189b |
| 24 months | 49.2 (15.4) | 52.3 (15.5) | 47.5 (15.3) | 0.210b |
| Role functioning | ||||
| 6 months | 50.9 (21.8) | 50.6 (23.8) | 51.1 (20.9) | 0.934b |
| 24 months | 41.8 (21.8) | 44.8 (24.8) | 40.2 (20.0) | 0.389b |
| Emotional functioning | ||||
| 6 months | 44.6 (14.6) | 40.9 (16.0) | 45.1 (14.0) | 0.742b |
| 24 months | 42.9 (17.5) | 43.3 (18.6) | 42.7 (17.1) | 0.893b |
| Cognitive functioning | ||||
| 6 months | 55.5 (22.3) | 57.0 (23.6) | 54.7 (21.8) | 0.673b |
| 24 months | 46.9 (20.8) | 48.7 (19.9) | 46.0 (21.4) | 0.600b |
| Social functioning | ||||
| 6 months | 39.1 (21.3) | 41.6 (23.2) | 37.6 (20.3) | 0.450b |
| 24 months | 47.4 (21.2) | 44.2 (23.5) | 49.2 (19.8) | 0.336b |
| Overall quality of life | ||||
| 6 months | 46.8 (16.1) | 43.9 (16.7) | 48.5 (15.7) | 0.244b |
| 24 months | 47.1 (20.0) | 49.3 (21.4) | 45.8 (19.3) | 0.477b |
| Appetite loss | ||||
| 6 months | 41.2 (31.4) | 26.9 (24.9) | 49.3 (32.0) | 0.003b,d |
| 24 months | 45.3 (28.6) | 47.3 (31.5) | 44.2 (27.2) | 0.649b |
| Nausea and vomiting | ||||
| 6 months | 41.0 (24.1) | 40.3 (25.0) | 41.3 (23.9) | 0.868b |
| 24 months | 38.1 (22.1) | 42.3 (24.1) | 35.8 (20.7) | 0.237b |
| Diarrhea | ||||
| 6 months | 29.1 (26.7) | 28.2 (26.1) | 29.7 (27.4) | 0.820b |
| 24 months | 22.6 (21.5) | 21.7 (20.9) | 23.1 (22.0) | 0.794b |
Log-rank test
Student's t-test.
Values are presented as the median (range), as opposed to the mean (standard deviation) presentation used for all other data.
P<0.05. PPPD, pylorus-preserving pancreaticoduodenectomy; SPD, standard pancreaticoduodenectomy; OS, overall survival; EORTC, European Organization for Research and Treatment of Cancer.
Figure 1.
Kaplan-Meier analysis of the overall survival rates of 116 patients with duodenal papilla carcinoma who had undergone SPD or PPPD. P=0.126. SPD, standard pancreaticoduodenectomy; PPPD, pylorus-preserving pancreaticoduodenectomy.
The present study also identified that the presence or absence of lymph node metastasis is a key factor influencing the prognosis of the 116 patients. The median survival time of the patients with lymph node metastasis was significantly shorter than that of the patients without lymph node metastasis (25.2 vs. 51.3 months; P<0.001; Fig. 2). Therefore, the possible difference between the two surgical methods was investigated in patients with lymph node metastasis. The present study identified that in the 116 patients, there were 25 cases of lymph node metastasis, compared with 9 cases and an average survival time of 27.4 months (range, 4–56 months) in the PPPD group and 16 cases and an average survival time of 24.0 months (range, 4–54 months) in the SPD group, with no statistically significant differences (27.4 vs. 24.0 months; P=0.592; Table V; Fig. 3). The results revealed that the two surgical methods had no significant influence on the prognosis of patients with lymph node metastasis.
Figure 2.
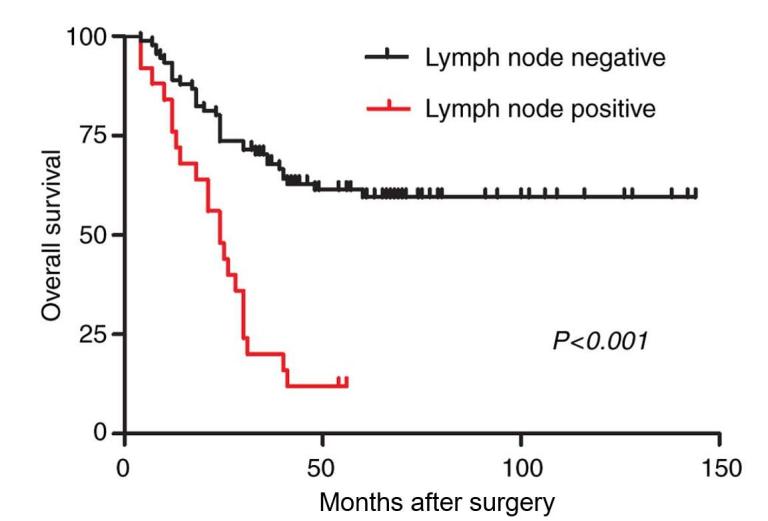
Kaplan-Meier analysis of the overall survival rates of 116 patients who had undergone pancreatoduodenectomy, with (positive) and without (negative) lymph node involvement. P<0.001.
Figure 3.
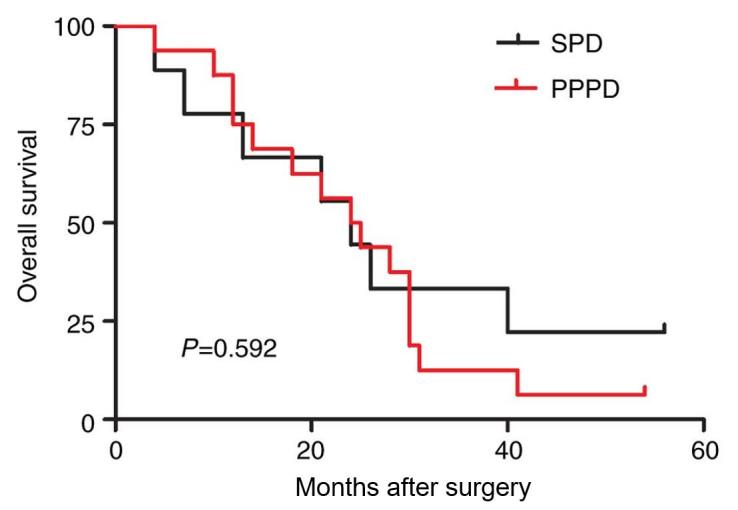
Kaplan-Meier analysis of the overall survival rates of 25 patients with positive lymph nodes who had undergone SPD or PPPD. P=0.592. SPD, standard pancreaticoduodenectomy; PPPD, pylorus-preserving pancreaticoduodenectomy.
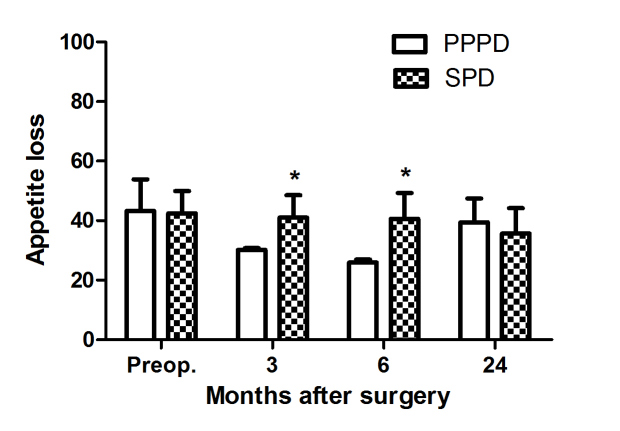
Statistical analysis was performed on the postoperative quality of life of the patients with EORTC-QLQ-C30 scale. The present study identified that there were no significant differences in the overall health level or 5 functional domains (physical, role, cognitive, emotional and social functioning) between the two groups of patients. Within 6 months, in terms of loss of appetite, patients in the PPPD had a minor advantage over those in the SPD group (26.9 vs. 49.3; P=0.003; Table V; Fig. 4), while in terms of nausea, vomiting, diarrhea or pain, no statistically significant differences were observed (Table V).
Figure 4.
Postoperative appetite loss in 116 patients with duodenal papilla carcinoma treated with SPD or PPPD. Values are presented as the mean ± standard deviation. No statistically significant differences were observed between the two procedures, with the exception of 3 and 6 months after surgery (Student's t-test). *P<0.05. SPD, standard pancreaticoduodenectomy; PPPD, pylorus-preserving pancreaticoduodenectomy.
Discussion
Compared with SPD, PPPD preserves the entire stomach and the lymph nodes at the lesser curvature of the stomach and above and below the pylorus. Therefore, there remain disputes regarding its influence on patient prognosis (14–16). Compared with carcinoma of the pancreas head and other periampullary carcinomas, primary DPC has a lower prevalence and improved prognosis, and may benefit from pylorus-preserving pancreaticoduodenectomy (21,22). Therefore, the present study investigated whether PPPD is as effective as SPD in the treatment of DPC.
Previous studies have reported that since PPPD is a simpler procedure than SPD, the duration of surgery is shorter (13–15). However, the present study identified no statistically significant differences in the durations of the two surgical methods. This may result from the improvements in the development of surgical devices in previous years or due to the fact that surgeries were performed by four different surgeons with considerable experience. Additionally, no statistically significant differences in the incidence of intraoperative hemorrhage were observed.
Pancreatic fistulas, biliary fistulas, postoperative intra-abdominal hemorrhage, intra-abdominal infection and gastric emptying disorders were the most common postoperative complications of the pancreas and duodenum; of which, the incidence of pancreatic fistula was the highest. A study by Lin and Lin (16) demonstrated that the incidence of postoperative pancreatic fistula in the patients in the SPD group was higher than in those in the PPPD group, while the results of a study by Seiler et al (14) revealed the opposite. However, there are also studies demonstrating that there was no significant difference in the incidence of pancreatic fistula between patients in the PPPD and SPD groups (12,15). The present study also identified that in terms of pancreatic fistula, there was no statistically significant differences between the PPPD and SPD groups.
Since the first reported gastric emptying disorder in 1978, there have been numerous studies reporting this symptom with various incidences, ranging between 25 and 70% (23–27). PPPD was once considered to be associated with the occurrence of gastric emptying disorder, and thus it was hypothesized that PPPD increased the incidence of gastric emptying disorder and prolonged the duration of hospitalization. Lytras et al (28) suggested that this may be associated with the intraoperative severance of the right gastric artery and the gastroduodenal artery during PPPD, which influences the blood supply to the pylorus. Tran et al (15) demonstrated that there was no statistically significant difference in the incidence of gastric emptying disorder between the two groups; however, gastric emptying disorder was revealed to be associated with intra-abdominal complications. The results of the present study were the same as those of a previous study by Tran et al (15), in that there was no significant difference in the incidence of gastric emptying disorder between the two groups.
The present study also demonstrated that there was no significant difference in the overall postoperative survival time between the two surgical methods. According to a long-term retrospective study in patients with periampullary carcinoma performed by Klein et al (29), the 15-year survival rate of patients without lymph node metastasis was significantly increased compared with that of patients with lymph node metastasis, an observation that was in line with the results of the present study. This indicates that lymph node metastasis was one of the factors influencing the postoperative long-term survival of patients with periampullary carcinoma (30,31). Therefore, the influence of the two surgical methods on the survival rate of patients with lymph node metastasis was investigated in the present study. The results demonstrated that, in patients with pathologically-confirmed post-operative lymph node metastasis confirmed, there was no significant difference in the overall survival rate between the patients in the PPPD and SPD groups. This demonstrated that the PPPD surgical method was not a contraindication for patients with lymph node metastasis. Of the patients with lymph node metastasis included in the present study, metastases were primarily to the lymph nodes near the pancreas and duodenum, and there were no patients with distant metastasis of the lymph nodes. The number of cases included in the present study was small and thus, large-scale retrospective studies on the PPPD surgical method are required in order to confirm the conclusions of the present study.
Wenger et al (32) identified in a survey on the quality of life of patients at 2, 4, 6, 24, 36, 48 and 60 weeks after pancreaticoduodenectomy that, in terms of loss of appetite, nausea and vomiting, diarrhea and weight gain, PPPD provided a significantly better outcome than SPD, while in terms of the overall health level, there were no significant differences. The present study also identified that the overall health level was similar between the two groups, and that there were no statistically significant differences in early nausea and vomiting, diarrhea and weight gain following surgery. However, 6 months after surgery, loss of appetite was less marked in the PPPD group than in the SPD group; while in terms of nausea and vomiting, diarrhea and pain, there were no statistically significant differences. These results indicate that, for patients at an early postoperative stage, the appetite loss of those treated with PPPD was improved compared with those treated with SPD. However, the number of cases included in the present study was small, patients were recruited from a single center, and the majority of the patients included had R0 excision.
To conclude, the incidence of postoperative pancreatic fistula and gastrointestinal dysfunction were similar in the PPPD and SPD groups. No statistically significant differences were observed prior to surgery in various indices, including the duration of surgery and intraoperative hemorrhage. According to the postoperative follow-up, the overall survival time was similar between the two groups and no statistically significant difference was observed in the prognosis of the patients with lymph node metastasis. In the survey of the quality of life, no statistically significant differences were observed in the overall health level or later postoperative symptoms; however, 6 months after surgery, PPPD of fered a minor advantage with regards to loss of appetite. These results indicated that the two surgical methods had similar effects in the treatment of PDC, but that PPPD may improve the early postoperative quality of life of the patients.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Natural Science Foundation of China (grant no. 81302123).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
RZ and YC designed the study, drafted and revised the paper; XW and PZ performed the research; CZ and PL searched the literature and analyzed the data. RZ revised the paper and approved the final version.
Ethics approval and consent to participate
Written informed consent for treatment was obtained from all patients and included a detailed explanation of the nature of the disease, possible treatment methods and potential postoperative complications. The present study was approved by the ethics committee of the PLA General Hospital and was performed in accordance with the ethical standards specified in The 1964 Declaration of Helsinki and its later amendments.
Consent for publication
Written informed consent for publication was obtained from all patients.
Competing interests
The authors declare that they have no competing interests.
References
- 1.DeOliveira ML, Trivino T, de Jesus Lopes Filho G. Carcinoma of the papilla of Vater: Are endoscopic appearance and endoscopic biopsy discordant? J Gastrointest Surg. 2006;10:1140–1143. doi: 10.1016/j.gassur.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 2.Kim MH, Lee SK, Seo DW, Won SY, Lee SS, Min YI. Tumors of the major duodenal papilla. Gastrointest Endosc. 2001;54:609–620. doi: 10.1067/mge.2001.119254. [DOI] [PubMed] [Google Scholar]
- 3.Di Giorgio A, Alfieri S, Rotondi F, Prete F, Di Miceli D, Ridolfini MP, Rosa F, Covino M, Doglietto GB. Pancreatoduodenectomy for tumors of Vater's ampulla: Report on 94 consecutive patients. World J Surg. 2005;29:513–518. doi: 10.1007/s00268-004-7498-x. [DOI] [PubMed] [Google Scholar]
- 4.Moriya T, Kimura W, Hirai I, Mizutani M, Ma J, Kamiga M, Fuse A. Nodal involvement as an indicator of postoperative liver metastasis in carcinoma of the papilla of Vater. J Hepatobiliary Pancreat Surg. 2006;13:549–555. doi: 10.1007/s00534-006-1113-5. [DOI] [PubMed] [Google Scholar]
- 5.Winter JM, Cameron JL, Olino K, Herman JM, de Jong MC, Hruban RH, Wolfgang CL, Eckhauser F, Edil BH, Choti MA, et al. Clinicopathologic analysis of ampullary neoplasms in 450 patients: Implications for surgical strategy and long-term prognosis. J Gastrointest Surg. 2010;14:379–387. doi: 10.1007/s11605-009-1080-7. [DOI] [PubMed] [Google Scholar]
- 6.Alexakis N, Halloran C, Raraty M, Ghaneh P, Sutton R, Neoptolemos JP. Current standards of surgery for pancreatic cancer. Br J Surg. 2004;91:1410–1427. doi: 10.1002/bjs.4794. [DOI] [PubMed] [Google Scholar]
- 7.Klaiber U, Probst P, Knebel P, Contin P, Diener MK, Büchler MW, Hackert T. Meta-analysis of complication rates for single-loop versus dual-loop (Roux-en-Y) with isolated pancreaticojejunostomy reconstruction after pancreaticoduodenectomy. Br J Surg. 2015;102:331–340. doi: 10.1002/bjs.9703. [DOI] [PubMed] [Google Scholar]
- 8.Huang W, Xiong JJ, Wan MH, Szatmary P, Bharucha S, Gomatos I, Nunes QM, Xia Q, Sutton R, Liu XB. Meta-analysis of subtotal stomach-preserving pancreaticoduodenectomy vs pylorus preserving pancreaticoduodenectomy. World J Gastroenterol. 2015;21:6361–6373. doi: 10.3748/wjg.v21.i20.6361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Traverso LW, Longmire WP., Jr Preservation of the pylorus in pancreaticoduodenectomy a follow-up evaluation. Ann Surg. 1980;192:306–310. doi: 10.1097/00000658-198009000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Alsaif F. Pylorus preserving pancreaticoduodenectomy for peri-ampullary carcinoma, is it a good option? Saudi J Gastroenterol. 2010;16:75–78. doi: 10.4103/1319-3767.61231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Song KB, Kim SC, Hwang DW, Lee JH, Lee DJ, Lee JW, Park KM, Lee YJ. Matched case-control analysis comparing laparoscopic and open pylorus-preserving pancreaticoduodenectomy in patients with periampullary tumors. Ann Surg. 2015;262:146–155. doi: 10.1097/SLA.0000000000001079. [DOI] [PubMed] [Google Scholar]
- 12.Diener MK, Fitzmaurice C, Schwarzer G, Seiler CM, Hüttner FJ, Antes G, Knaebel HP, Büchler MW. Pylorus-preserving pancreaticoduodenectomy (pp Whipple) versus pancreaticoduodenectomy (classic Whipple) for surgical treatment of periampullary and pancreatic carcinoma. Cochrane Database Syst Rev CD006053. 2014 doi: 10.1002/14651858.CD006053.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kawai M, Hirono S, Okada K, Sho M, Nakajima Y, Eguchi H, Nagano H, Ikoma H, Morimura R, Takeda Y, et al. Randomized controlled trial of pancreaticojejunostomy versus stapler closure of the pancreatic stump during distal pancreatectomy to reduce pancreatic fistula. Ann Surg. 2016;264:180–187. doi: 10.1097/SLA.0000000000001395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Seiler CA, Wagner M, Bachmann T, Redaelli CA, Schmied B, Uhl W, Friess H, Büchler MW. Randomized clinical trial of pylorus-preserving duodenopancreatectomy versus classical Whipple resection-long term results. Br J Surg. 2005;92:547–556. doi: 10.1002/bjs.4881. [DOI] [PubMed] [Google Scholar]
- 15.Tran KT, Smeenk HG, van Eijck CH, Kazemier G, Hop WC, Greve JW, Terpstra OT, Zijlstra JA, Klinkert P, Jeekel H. Pylorus preserving pancreaticoduodenectomy versus standard Whipple procedure: A prospective, randomized, multicenter analysis of 170 patients with pancreatic and periampullary tumors. Ann Surg. 2004;240:738–745. doi: 10.1097/01.sla.0000143248.71964.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lin PW, Lin YJ. Prospective randomized comparison between pylorus-preserving and standard pancreaticoduodenectomy. Br J Surg. 1999;86:603–607. doi: 10.1046/j.1365-2168.1999.01074.x. [DOI] [PubMed] [Google Scholar]
- 17.Federico A, Orditura M, Cotticelli G, DE Sio I, Romano M, Gravina AG, Dallio M, Fabozzi A, Ciardiello F, Loguercio C, DE Vita F. Safety and efficacy of sorafenib in patients with advanced hepatocellular carcinoma and Child-Pugh A or B cirrhosis. Oncol Lett. 2015;9:1628–1632. doi: 10.3892/ol.2015.2960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bassi C, Dervenis C, Butturini G, Fingerhut A, Yeo C, Izbicki J, Neoptolemos J, Sarr M, Traverso W, Buchler M, et al. Postoperative pancreatic fistula: An international study group (ISGPF) definition. Surgery. 2005;138:8–13. doi: 10.1016/j.surg.2005.05.001. [DOI] [PubMed] [Google Scholar]
- 19.Klinkenbijl JH, Jeekel J, Sahmoud T, van Pel R, Couvreur ML, Veenhof CH, Arnaud JP, Gonzalez DG, de Wit LT, Hennipman A, Wils J. Adjuvant radiotherapy and 5-fluorouracil after curative resection of cancer of the pancreas and periampullary region: Phase III trial of the EORTC gastrointestinal tract cancer cooperative group. Ann Surg. 1999;230:776–782. doi: 10.1097/00000658-199912000-00006. discussion 782–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hermanek P, Scheibe O, Spiessl B, Wagner G. TNM classification of malignant tumors: The new 1987 edition. Rontgenblatter. 1987;40:200. (In German) [PubMed] [Google Scholar]
- 21.Fitzsimmons D, Kahl S, Butturini G, van Wyk M, Bornman P, Bassi C, Malfertheiner P, George SL, Johnson CD. Symptoms and quality of life in chronic pancreatitis assessed by structured interview and the EORTC QLQ-C30 and QLQ-PAN26. Am J Gastroenterol. 2005;100:918–926. doi: 10.1111/j.1572-0241.2005.40859.x. [DOI] [PubMed] [Google Scholar]
- 22.Wellner UF, Küsters S, Sick O, Busch C, Bausch D, Bronsert P, Hopt UT, Karcz KW, Keck T. Hybrid laparoscopic versus open pylorus-preserving pancreatoduodenectomy: Retrospective matched case comparison in 80 patients. Langenbecks Arch Surg. 2014;399:849–856. doi: 10.1007/s00423-014-1236-0. [DOI] [PubMed] [Google Scholar]
- 23.Hanna MM, Tamariz L, Gadde R, Allen C, Sleeman D, Livingstone A, Yakoub D. Delayed gastric emptying after pylorus preserving pancreaticoduodenectomy-does gastrointestinal reconstruction technique matter? Am J Surg. 2016;211:810–819. doi: 10.1016/j.amjsurg.2015.10.015. [DOI] [PubMed] [Google Scholar]
- 24.Hanna MM, Gadde R, Allen CJ, Meizoso JP, Sleeman D, Livingstone AS, Merchant N, Yakoub D. Delayed gastric emptying after pancreaticoduodenectomy. J Surg Res. 2016;202:380–388. doi: 10.1016/j.jss.2015.12.053. [DOI] [PubMed] [Google Scholar]
- 25.Walters DM, Shada AL, LaPar DJ, Adams RB, Bauer TW. A long gastrojejunostomy is associated with decreased incidence and severity of delayed gastric emptying after pancreaticoduodenectomy. Pancreas. 2015;44:1273–1279. doi: 10.1097/MPA.0000000000000415. [DOI] [PubMed] [Google Scholar]
- 26.Sahora K, Morales-Oyarvide V, Thayer SP, Ferrone CR, Warshaw AL, Lillemoe KD, Fernández-Del Castillo C. The effect of antecolic versus retrocolic reconstruction on delayed gastric emptying after classic non-pylorus-preserving pancreaticoduodenectomy. Am J Surg. 2015;209:1028–1035. doi: 10.1016/j.amjsurg.2014.04.015. [DOI] [PubMed] [Google Scholar]
- 27.Sakamoto Y, Hori S, Oguro S, Arita J, Kishi Y, Nara S, Esaki M, Saiura A, Shimada K, Yamanaka T, Kosuge T. Delayed gastric emptying after stapled versus hand-sewn anastomosis of duodenojejunostomy in pylorus-preserving pancreaticoduodenectomy: A randomized controlled trial. J Gastrointest Surg. 2016;20:595–603. doi: 10.1007/s11605-015-2961-6. [DOI] [PubMed] [Google Scholar]
- 28.Lytras D, Paraskevas KI, Avgerinos C, Manes C, Touloumis Z, Paraskeva KD, Dervenis C. Therapeutic strategies for the management of delayed gastric emptying after pancreatic resection. Langenbecks Arch Surg. 2007;392:1–12. doi: 10.1007/s00423-006-0096-7. [DOI] [PubMed] [Google Scholar]
- 29.Klein F, Jacob D, Bahra M, Pelzer U, Puhl G, Krannich A, Andreou A, Gül S, Guckelberger O. Prognostic factors for long-term survival in patients with ampullary carcinoma: The results of a 15-year observation period after pancreaticoduodenectomy. HPB Surg. 2014;2014:970234. doi: 10.1155/2014/970234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tol JA, Brosens LA, van Dieren S, van Gulik TM, Busch OR, Besselink MG, Gouma DJ. Impact of lymph node ratio on survival in patients with pancreatic and periampullary cancer. Br J Surg. 2015;102:237–245. doi: 10.1002/bjs.9709. [DOI] [PubMed] [Google Scholar]
- 31.Bronsert P, Kohler I, Werner M, Makowiec F, Kuesters S, Hoeppner J, Hopt UT, Keck T, Bausch D, Wellner UF. Intestinal-type of differentiation predicts favourable overall survival: Confirmatory clinicopathological analysis of 198 periampullary adenocarcinomas of pancreatic, biliary, ampullary and duodenal origin. BMC Cancer. 2013;13:428. doi: 10.1186/1471-2407-13-428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wenger FA, Jacobi CA, Haubold K, Zieren HU, Muller JM. Gastrointestinal quality of life after duodenopancreatectomy in pancreatic carcinoma. Preliminary results of a prospective randomized study: Pancreatoduodenectomy or pylorus-preserving pancreatoduodenectomy. Chirurg. 1999;70:1454–1459. doi: 10.1007/PL00002580. (In German) [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.