Abstract
Lipoxygenase (LOX) activity has been identified consistently during pathogen-induced defense responses. Here we report the involvement of a specific leaf LOX gene of potato (Solanum tuberosum), designated POTLX-3 (GenBank/EMBL accession no. U60202), in defense responses against pathogens. The sequence of POTLX-3 does not match any other LOX genes of potato and has the greatest match to a tobacco LOX gene that contributes to a resistance mechanism against Phytophthora parasitica var nicotianae. POTLX-3 transcript accumulation was not detected in untreated, healthy potato organs or in wounded mature leaves. POTLX-3 mRNA accumulation was induced in potato leaves treated with ethylene or methyl jasmonate or infected with either virulent or avirulent strains of Phytophthora infestans, the causal agent of late blight. During the resistance response, POTLX-3 was induced within 6 hours, increased steadily through 24 hours, and its mRNA continued to accumulate for a week after inoculation. In contrast, when a plant was susceptible to P. infestans, induction of mRNA accumulation in response to inoculation was inconsistent and delayed. LOX activity assayed during an incompatible interaction in leaves peaked 3 days earlier than during a compatible interaction. POTLX-3 mRNA accumulation also was induced during hypersensitive response development caused by the incompatible pathogen Pseudomonas syringae pv phaseolicola. Our results show that POTLX-3 may be involved specifically in defense responses against pathogen infection.
Lipoxygenases (LOX; EC 1.13.11.12) are dioxygenases that catalyze the hydroperoxidation of polyunsaturated fatty acids or their esters that contain a cis,cis-1,4-pentadiene moiety. In higher plants, the natural substrates for these enzymes are linolenic and linoleic acids (Siedow, 1991; Conconi et al., 1996). The primary products are fatty acid hydroperoxides that are metabolized enzymatically into compounds like traumatin, jasmonic acid (JA), and methyl jasmonate (MJ) (Anderson, 1989; Koda, 1992; Creelman and Mullet, 1997). These compounds have physiological functions (Anderson, 1989; Siedow, 1991) in growth and development, senescence, and wound- and pathogen-induced defense responses (Siedow, 1991).
LOX protein and activity levels were induced in response to mechanical wounding and insect attack (Hildebrand et al., 1988; Saravitz and Siedow, 1995). An increase in LOX activity in response to infection has been reported for several plant-pathogen systems, and LOX activity has been correlated with plant resistance against pathogens (Slusarenko, 1996). Increased LOX activity occurred in rice leaves after infection with an incompatible race of rice blast fungus but not with a compatible race (Ohta et al., 1991). In several other host-pathogen combinations, LOX activity was induced more rapidly and to greater levels in an incompatible response than in a compatible one (Slusarenko, 1996; Veronesi et al., 1996). LOX activity also is induced when cell cultures or plants are treated with elicitors. When potato (Solanum tuberosum) tuber discs were treated with the fungal elicitor arachidonic acid, the activity of LOX increased almost 2-fold in 0.5 to 3 h after treatment (Bostock et al., 1992). In addition, LOX genes were activated transcriptionally by wounding, pathogens, or their elicitors (Bell and Mullet, 1991; Melan et al., 1993; Peng et al., 1994; Veronesi et al., 1996; Bohland et al., 1997; Fidantsef and Bostock, 1998).
It has been suggested that LOX is involved in the development of an active resistance mechanism known as the hypersensitive response (HR), a form of programmed cell death (Keppler and Novacky, 1987; Croft et al., 1990; Koch et al., 1992; Rusterucci et al., 1999). In the HR, an infection event is followed by rapid death of plant cells localized around the infection site, and this leads to necrotic lesion formation. This reaction limits pathogen spread and prevents further damage to the remainder of the plant organ. In several plant-pathogen systems, HR occurrence is linked tightly to increased activity, protein, or mRNA levels of LOXs (Vaughn and Lulai, 1992; Slusarenko, 1996; Rusterucci et al., 1999). Transgenic studies have shown that expression of a tobacco LOX gene is involved directly in the HR and resistance to Phytophthora parasitica var nicotianae (Rance et al., 1998). Products of the 9-LOX pathway contributed to the development of hypersensitive cell death induced by an elicitin on tobacco leaves (Rusterucci et al., 1999).
Identification of specific LOX genes involved in mechanisms of resistance is difficult, however, because multiple LOX isozymes are involved in wound- and pathogen-induced defense responses (Saravitz and Siedow, 1995, 1996; Bohland et al., 1997). For example, several potato cDNA clones that encode distinct LOX isoforms have been identified, and their expression is organ-specific and differentially regulated during tuber development and in response to wounding, pathogen infection, and MJ treatments (Geerts et al., 1994; Casey, 1995; Kolomiets et al., 1996a, 1996b; Royo et al., 1996; Fidantsef and Bostock, 1998). A recent study of LOX isozyme profiles in the wheat-rust fungus pathosystem revealed that several LOX species were induced differentially during the HR evoked by the pathogen, its specific glycopeptide elicitor, other elicitors like chitosan and chitin oligosaccharides, and MJ (Bohland et al., 1997). Several potato LOX genes have been identified and implicated in wound-induced defense responses and in tuber development (Geerts at al., 1994; Kolomiets et al., 1996a; Royo et al., 1996), but none are involved specifically in pathogen-induced defense responses. Here we report the specific induction of a novel LOX gene of potato, designated POTLX-3 (Kolomiets et al., 1996b), during an incompatible interaction with the pathogen Phytophthora infestans but not in response to wounding.
RESULTS
Characterization of POTLX-3
The full-length potato LOX cDNA clone, POTLX-3 (Kolomiets et al., 1996b), encodes a polypeptide of 862 amino acids with a calculated molecular mass of 97.8 kD. A comparison of POTLX-3 with other plant LOXs from potato and several other species revealed considerable amino acid sequence similarity with LOXs from solanaceous species (Table I). POTLX-3 showed the greatest match (85.5% identity and 89.7% similarity) to tobacco LOX1 (Veronesi et al., 1995), which was shown to be essential for resistance against pathogens (Rance et al., 1998). The identity match was somewhat lower (less than 80%) for the representatives of the Lox1 isozyme family from potato, POTLX-1 and POTLX-2 (Kolomiets et al., 1996a) and tomato, TomLoxA (Ferrie et al., 1994; Heitz et al., 1997). The two other known leaf LOXs of potato, H1 and H3, which contain chloroplast-targeting signal peptides (Royo et al., 1996), shared the least identity match (43.1% and 46.1%, respectively) with POTLX-3. In comparison, the sequence identity was greater than 59% for non-solanaceous LOXs from Arabidopsis and barley (Table I).
Table I.
Comparison of percentage identity and percentage similarity of deduced amino acid sequences of POTLX-3 gene with those of other plant LOX genes
| Plant LOX Genea | GenBank Accession No. | POTLX-3b
|
|
|---|---|---|---|
| Identity (%) | Similarity (%) | ||
| Tobacco, LOX1 | X84040 | 85.5 | 89.7 |
| Potato, POTLX-2 | U60201 | 79.1 | 85.3 |
| Tomato, tomloxA | U09026 | 78.9 | 84.7 |
| Potato, POTLX-1 | U60200 | 77.9 | 84.1 |
| Arabidopsis, AtLox1 | L04637 | 69.0 | 76.0 |
| Barley, LoxA | L35931 | 59.3 | 68.9 |
| Potato, H3 | X96406 | 46.1 | 57.1 |
| Potato, H1 | X96405 | 43.1 | 54.2 |
The predicted amino acid sequence of POTLX-3 (GenBank accession no. U60202) was compared with other plant LOX gene products available in the GenBank database: tobacco LOX1 (Veronesi et al. 1995); POTLX-1 and POTLX-2 (Kolomiets et al. 1996a); tomato tomloxA (Ferrie et al. 1994); Arabidopsis AtLox1 (Melan et al. 1993); barley LoxA (van Mechelen et al. 1995); and potato H1 and H3 (Royo et al. 1996).
Percentage identity and percentage similarity were calculated with the GAP program of the Genetics Computer Group of the University of Wisconsin (Madison, WI).
The predicted POTLX-3 polypeptide contained the conserved 39-amino acid motif, the three conserved His residues, and one Ile residue that are essential for iron binding and enzyme activity. It also contained the 11 highly conserved amino acids possibly involved in the accommodation of the fatty acid substrate (Steczko et al., 1992; Boyington et al., 1993). Like other plant 9-LOXs (Royo et al., 1996; Heitz et al., 1997), POTLX-3 contains no transit peptide for chloroplast targeting and has the conserved space-saving amino acid pairing at positions 580 to 581 (Hornung et al., 1999). The PSORT protein-sorting analysis (Nakai and Kanehisa, 1992), which predicts subcellular localization of proteins, indicated peroxisomes and/or the cytoplasm as the most probable sites (48% and 45% certainty, respectively) for POTLX-3 localization.
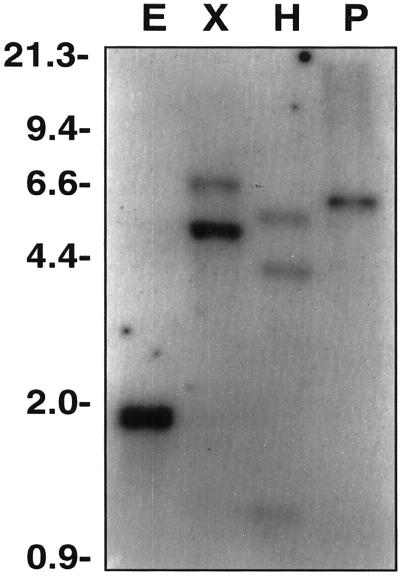
Southern hybridization of potato genomic DNA was performed to evaluate the complexity of POTLX-3-related genes in the potato genome. Under the hybridization conditions used, the POTLX-3 probe did not cross-hybridize with the sequences of two other potato LOXs, POTLX-1 and POTLX-2 (Kolomiets et al., 1996a), that share the greatest sequence identity (Table I). Southern analysis revealed one to two bands that hybridized to the POTLX-3 probe (Fig. 1), indicating the existence of 1 to 2 copies of POTLX-3 or closely related genes in the potato genome.
Figure 1.
Southern-blot analysis of potato genomic DNA with POTLX-3 probe. Genomic DNA (10 μg per lane) was digested with EcoRI (lane E), XbaI (lane X), HindIII (lane H), and PstI (lane P). The membrane was probed with the 32P-labeled 1.5-kb EcoRI fragment of the POTLX-3 cDNA clone. DNA size markers in kilobases are indicated on the left.
Effect of Wounding and Hormonal Treatments on POTLX-3 mRNA Levels
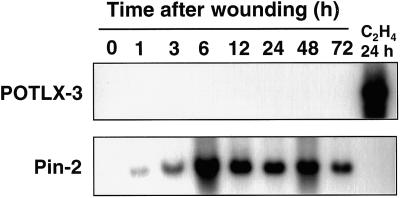
We studied organ-specific expression of POTLX-3 at the mRNA level in potato plants by performing northern-blot analysis on total and poly(A+) RNA isolated from leaves, stems, flowers, roots, tubers, and stolons. POTLX-3 transcripts were not detected in any of these organs during normal growth and development (data not shown). Although POTLX-1, POTLX-2, and other members of the potato Lox1 multigene family are regulated developmentally during tuber formation (Kolomiets et al., 1996a; Royo et al., 1996), no POTLX-3 mRNA accumulation was detected in tubers at several stages of development (data not shown). To determine if POTLX-3 can be induced by wounding, we examined POTLX-3 mRNA accumulation in mature leaves that had been wounded mechanically. No accumulation of POTLX-3 transcripts occurred in leaves for up to 72 h after wounding, even though Pin2 mRNA levels were detected as early as 1 h after wounding and persisted for 72 h (Fig. 2).
Figure 2.
Northern-blot analysis of POTLX-3 and Pin-2 mRNA accumulation after wounding potato leaves. Total RNA was isolated from wounded leaves of potato cv Superior plants harvested 0 to 72 h after wounding. The 0-h RNA sample was extracted from leaves harvested immediately after wounding. RNA samples (18 μg per lane) from wounded leaves were loaded on duplicate gels, and northern blots were probed with 32P-labeled inserts of either POTLX-3 cDNA or proteinase inhibitor II (Pin-2) cDNA. Total RNA (18 μg per lane) from leaves treated with ethylene for 24 h was included as a positive control. Hybridization to the Pin2 probe was conducted as a positive control for wounding. POTLX-3 transcript size is approximately 2.8 kb. Equal loading of RNA samples into each lane was confirmed by visualizing the RNA gel stained with ethidium bromide.
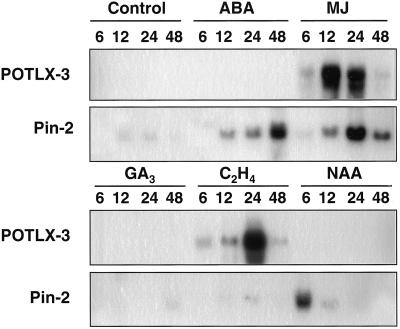
Northern analysis also was performed to determine whether POTLX-3 mRNA accumulation could be induced by phytohormones that affect expression of other plant LOX and defense-related genes. Steady-state levels of POTLX-3 mRNA were induced only by MJ and ethylene (Fig. 3). In leaves treated with MJ, mRNA accumulation was detected at 6 h, reached maximal levels at 12 h, and decreased drastically after 24 h. Induction by ethylene also occurred within 6 h, but maximal levels of POTLX-3 mRNA were attained only at 24 h and rapidly decreased at 48 h. Induction of proteinase inhibitor-II (Pin-2) transcript accumulation was used as a positive control for abscisic acid (ABA), MJ, and NAA treatments (Hildmann et al., 1992).
Figure 3.
Northern-blot analysis of POTLX-3 mRNA accumulation in potato leaves treated with various phytohormones. Leaf-petiole cuttings were incubated for 6, 12, 24, and 48 h in either water (control), 100 μm ABA, 100 μm MJ, 100 μm gibberellic acid (GA3), 50 μm auxin (NAA), or in hermetically sealed 4-L jars that contained 10 μL/L ethylene. An equal amount of total RNA (12 μg per lane) was loaded in each lane, and the consistency of loading was determined by visualizing RNA under UV light after staining with ethidium bromide. Hybridization was conducted with the 32P-labeled 1.5-kb EcoRI fragment of the POTLX-3 cDNA clone. Blots subsequently were stripped and rehybridized with probe derived from a proteinase inhibitor II (Pin-2) gene.
POTLX-3 mRNA Accumulation and LOX Activity in Response to Pathogen Attack
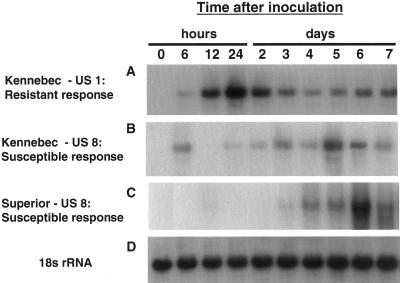
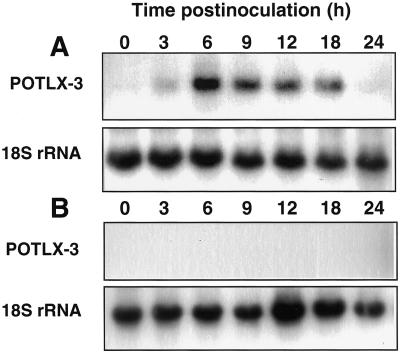
Because LOX activity is induced strongly by inoculation with P. infestans, we used this pathogen to study the dynamics of POTLX-3 expression during the resistance response in potato. To examine POTLX-3 expression during both compatible and incompatible potato-P. infestans interactions, two potato cultivars, cv Superior and cv Kennebec, were inoculated with sporangial suspensions of two P. infestans strains, US940507 (US1) or ME93-2A (US8), in a factorial combination. cv Kennebec harbors resistance gene R-1, and cv Kennebec is resistant to US940507 (incompatible interaction) and susceptible to ME93-2A (compatible interaction). Both strains are compatible on cv Superior. Northern analysis of total RNA extracted from infected leaves revealed that POTLX-3 transcript accumulation was induced by infection with both P. infestans strains in all combinations. POTLX-3 mRNA accumulation was induced rapidly and to high levels during the incompatible interaction of cv Kennebec leaves with P. infestans US940507 (Fig. 4A). POTLX-3 transcripts were detected at 6 h, increased in abundance at 12 h, and reached maximal levels at 24 h postinoculation. At d 2 and 3 postinoculation, transcript levels steadily declined but were detectable from 3 to 7 d. POTLX-3 transcripts were not detected in mock-inoculated control potato leaves incubated under identical conditions (data not shown). By d 2, when POTLX-3 transcript accumulation started to decrease, the first symptoms of necrotic lesion development characteristic of the HR were clearly visible. POTLX-3 mRNA accumulation was also induced early (12–48 h) in leaves during an incompatible interaction in several breeding lines of potato selected for resistance against the late blight US8 type (data not shown).
Figure 4.
Northern-blot analysis of POTLX-3 mRNA accumulation in response to P. infestans inoculation. Detached leaflets of cv Kennebec (resistance gene R-1) were inoculated with a sporangial suspension of P. infestans US940507 (US1), incompatible interaction resulting in resistance (A), or with ME93-2A (US8), compatible interaction (B). cv Superior leaflets were inoculated with P. infestans ME93-2A, compatible interaction that resulted in susceptibility (C). Total RNA was extracted from inoculated leaflets that were incubated in a growth chamber at 18°C for the indicated times. An equal amount of total RNA (18 μg per lane) was loaded in each lane and transferred onto a nylon membrane. The blots were hybridized with the 32P-labeled 1.5-kb EcoRI fragment of the POTLX-3 cDNA clone. The blots were stripped and rehybridized to a 1.2-kb wheat 18S ribosomal RNA probe (D) to confirm that similar quantities of total RNA were present in each lane.
During compatible interactions, large water-soaked lesions first were observed 4 d postinoculation, and leaves collapsed completely by d 7. In contrast to the resistance response, POTLX-3 transcripts accumulated more slowly or inconsistently during the compatible interaction between cv Kennebec or cv Superior and P. infestans ME93-2A (Fig. 4, B and C, respectively). For cv Kennebec, transcript accumulation was inconsistent and delayed with the highest levels detected at d 5 (Fig. 4). For cv Superior, transcript accumulation was detected first at 3 d, reached a maximum at 6 d, and declined at 7 d postinoculation. This overall delayed pattern of POTLX-3 transcript accumulation was consistent across all compatible interactions observed. Subsequent stripping and rehybridization of the blots to a wheat 18S rRNA gene probe confirmed that RNA loading between samples was comparable (Fig. 4D).
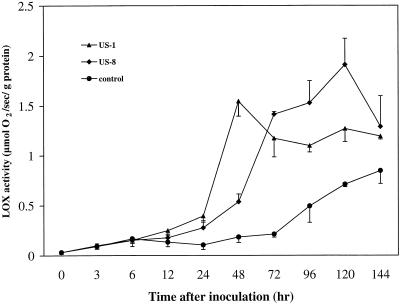
LOX activity was assayed in the same pathogen-infection system used for the RNA analysis. LOX activity in leaves of cv Kennebec inoculated with US1 (incompatible interaction) exhibited a peak at 48 h, whereas in response to inoculation with US8 (compatible interaction), LOX activity was relatively low at 48 h with peak activity occurring 3 d later at 120 h (Fig. 5). The LOX activity assay in cv Kennebec leaves was repeated several times with similar results. The increase in LOX activity during the compatible interaction and in control leaves after 72 h is probably the result of necrosis associated with infection and senescence. Transcript accumulation of two other LOXs of potato leaf, H1 and H3 (Royo et al., 1996), was not induced by pathogen infection (data not shown). Transcripts of POTLX-1 and -2 are only detected in tubers, stolons, and roots (data not shown).
Figure 5.
LOX activity in potato leaflets inoculated with P. infestans. Detached leaflets of cv Kennebec (resistance gene R-1) were inoculated with a sporangial suspension of P. infestans US940507 (US1, incompatible interaction resulting in resistance, ▴) or with ME93-2A (US8, compatible interaction resulting in susceptibility, ♦). Control cv Kennebec leaflets were inoculated with water (no interaction, ●). LOX activity (μmol O2 s−1 g−1 protein) in leaves was determined by measuring oxygen consumption in protein extracts from pooled samples by using a Clark electrode for detection (Royo et al., 1996). Linolenic acid was added as a substrate and the pH of the reaction mix was 6.8. Each data point represents the mean of three measurements.
To determine whether POTLX-3 gene induction is a specific response to P. infestans infection or a general host response associated with the HR, leaves of cv Superior plants were challenged with the incompatible bacterial pathogen Pseudomonas syringae pv phaseolicola. This pathogen causes halo blight disease on common bean (Phaseolus spp.), and it is non-pathogenic on potato in which it induces development of necrotic lesions typical of the HR. The leaflet areas infiltrated with P. syringae pv phaseolicola exhibited symptoms of incompatible-type tissue collapse after 6 h and were completely collapsed by 24 h. The timing of the visual appearance of HR lesions was preceded by pronounced induction of POTLX-3 mRNA accumulation (Fig. 6A). Transcripts were detected 3 h after inoculation and reached maximal levels by 6 h. After 9 h, when HR necrotic lesions were clearly visible, POTLX-3 mRNA levels started to decline steadily through 24 h postinoculation. POTLX-3 transcripts were not detected in control, mock-inoculated potato plants (Fig. 6B).
Figure 6.
Northern-blot analysis of POTLX-3 mRNA accumulation in response to P. syringae pv phaseolicola inoculation. cv Superior leaflets were inoculated with P. syringae pv phaseolicola bacterial suspension (1.2 × 108 colony forming units mL−1) (A) or with sterile water (mock-inoculated) (B). Plants with inoculated leaves were incubated for 0, 3, 6, 12, 18, or 24 h in the greenhouse, and total RNA was extracted from the entire leaflet at each time. Blots of total RNA (18 μg per lane) were hybridized with the 32P-labeled 1.5-kb EcoRI fragment of the POTLX-3 cDNA clone. Equal loading of RNA samples into each lane was confirmed by stripping and rehybridizing the blots with a 1.2-kb wheat 18S ribosomal RNA probe.
DISCUSSION
Correlative evidence exists that strongly implicates LOX activity in potato resistance mechanisms against pathogens (Bostock et al., 1992; Slusarenko, 1996; Weber et al., 1999). Specific pathogen-induced LOXs of leaves of potato and tobacco have been implicated in the production of antifungal compounds (Rance et al., 1998; Hamberg, 1999; Rusterucci et al., 1999; Weber et al., 1999), but no potato LOX genes involved specifically in defense against pathogens have been identified. Here we report the characterization of a novel leaf lipoxygenase of potato and examine its role in defense responses.
The polypeptide deduced from POTLX-3 showed high overall amino acid sequence similarity with plant LOXs from both dicots and monocots. These data and the presence of the highly conserved amino acid motifs and residues required for LOX enzyme activity established POTLX-3 as a lipoxygenase. To some extent, the involvement of individual LOX isozymes in physiological processes of plants depends on their subcellular compartmentalization (Siedow, 1991; Stephenson et al., 1998). Among all of the plant LOX sequences available from the databases, POTLX-3 shared the least amino acid sequence similarity with the chloroplast-targeted LOXs from potato leaves (H1 and H3 in Table I) (Royo et al., 1996), tomato (Heitz et al., 1997), rice (Peng et al., 1994), and Arabidopsis (Bell et al., 1995) (data not shown). The Arabidopsis AtLOX2 isozyme is required for wound-induced JA biosynthesis (Bell et al., 1995), and a similar role has been proposed for other wound- and pathogen-inducible chloroplast-localized LOXs (Peng et al., 1994; Royo et al., 1996; Heitz et al., 1997). Amino acid sequence analysis showed that POTLX-3 does not possess a chloroplast transit peptide found in these LOXs, and this indicates that POTLX-3 likely is not targeted to chloroplasts.
Potato LOX genes have been grouped into three classes on the basis of the similarity of their deduced amino acid sequences (Royo et al., 1996). The Lox1 class comprises several genes that share more than 95% sequence similarity, including two tuber-specific genes, POTLX-1 and POTLX-2 (Kolomiets et al., 1996a). The Lox2 and Lox 3 classes share limited similarity to each other and to the Lox1 isozymes (less than 65%) and are represented by single genes H1 and H3, respectively (Table I; Royo et al., 1996). A comparison of the sequence of POTLX-3 with the proteins from these three classes (Table I) indicates that POTLX-3 represents a novel potato LOX gene. The best match (89.7% similarity) to POTLX-3 is LOX1 from tobacco, a lipoxygenase involved in resistance against black shank via development of the HR (Rance et al., 1998). Southern analysis showed that, unlike potato Lox1 and Lox2 genes that exist as multigene families (Royo et al., 1996; Fidantsef and Bostock, 1998), POTLX-3 is most likely represented by only one or two genes in the potato genome.
Developmental and inducible regulation of POTLX-3 mRNA accumulation also was unique when compared with other potato LOXs. The known classes of potato genes showed clear organ-specific expression (Royo et al., 1996). Unlike these other potato LOX genes, POTLX-3 mRNA was not expressed constitutively in any healthy potato organ and was not induced during tuber development. Unlike Lox2 and Lox3 genes from potato (Royo et al., 1996) and many other plant LOXs (Melan et al., 1993; Heitz et al., 1997), mechanical wounding of mature potato leaves did not induce POTLX-3 gene expression. These results suggest that POTLX-3 probably is not involved in the octadecanoid wound-inducible signal transduction pathway that leads to the activation of proteinase inhibitors (Farmer and Ryan, 1992; Royo et al., 1999) or that POTLX-3 has a very low sensitivity to the wound signal. POTLX-3 mRNA accumulation was induced in potato leaves after treatment with MJ or ethylene. These results are consistent with the observation that MJ and ethylene synergistically induce expression of other pathogen-induced defense genes such as those encoding PR-1 and PR-5 proteins (Xu et al., 1994). Arabidopsis defensin genes, which are involved in defense against fungal pathogens, were also induced by MJ and ethylene but not wounding (Penninckx et al., 1996).
LOX activity and mRNA levels increased in response to pathogen challenge or elicitor treatments in a number of plant species (Ohta et al., 1991; Koch et al., 1992; Melan et al., 1993; Peng et al., 1994; Rusterucci et al., 1999). We demonstrated that POTLX-3 transcript accumulation was induced in leaves inoculated by both compatible and incompatible strains of P. infestans. POTLX-3 transcripts accumulated more rapidly, more consistently, and to greater levels during an incompatible interaction. The greatest levels of LOX activity coincided with the appearance of HR lesions 48 h after inoculation. In contrast, during compatible interactions, POTLX-3 transcript accumulation and LOX activity were delayed and coincided with the occurrence of massive tissue collapse 96 to 144 h postinoculation. In other studies that compared compatible and incompatible interactions, it was observed that LOX mRNA accumulation and activity reach a greater level more rapidly in resistant than susceptible plants (Koch et al., 1992; Melan et al., 1993; Veronesi et al., 1996). In the Veronesi et al. (1996) study with tobacco, during the resistance response, LOX activity peaked at 3 d, whereas in the susceptible response, activity peaked 1 d later. With the incompatible interactions of both P. infestans and P. syringae, the most abundant accumulation of POTLX-3 transcripts preceded the formation of necrotic lesions characteristic of the HR, whereas LOX activity coincided with it, suggesting that POTLX-3 may play a role in localized cell death associated with the HR in potato.
Several reports implicate 9-LOX activity in a causal relationship with the HR. The production of free polyunsaturated fatty acid hydroperoxides dependent on 9-LOX activity was identified during leaf necrosis in tobacco (Rusterucci et al., 1999). In this study, inhibition and activation of the 9-LOX pathway was shown to inhibit or to activate localized cell death. LOX involvement in the HR was further demonstrated in transgenic tobacco plants in which expression of a specific pathogen-inducible 9-lipoxygenase was suppressed by antisense sequences (Rance et al., 1998). The resulting decrease of LOX activity was sufficient to abolish the HR and reduce the resistance of tobacco against P. parasitica var nicotianae. Because POTLX-3 protein shared the greatest sequence similarity (approximately 90%, Table I) with this tobacco gene and the pattern of mRNA accumulation and LOX activity are similar (Veronesi et al., 1996), we propose that POTLX-3 may have a similar function in potato.
Because the timing of POTLX-3 transcript accumulation is correlated with the onset of HR lesions during incompatible interactions, it is plausible that POTLX-3 may have a causal relationship with the development of the HR. Similar to the 9-LOX of tobacco, the POTLX-3 isozyme may contribute to cell death by the massive production of free fatty acid hydroperoxides that are responsible for tissue necrosis (Deighton et al., 1999; Rusterucci et al., 1999). Free radicals and reactive oxygen species are produced from this pathway that can lead to cell membrane damage (Keppler and Novacky, 1987; Croft et al., 1990). Other potential functions of POTLX-3 in disease resistance could be associated with the production of antimicrobial substances (Kato et al., 1986; Croft et al., 1993; Hamberg, 1999; Weber et al., 1999) or signaling molecules such as JA and MJ capable of inducing defense-related genes. The POTLX-3 isozyme may have a specific function in potato-P. infestans interactions by mediating the elicitor activity of arachidonic and eicosapentaenoic acids. These two P. infestans-derived elicitors are LOX substrates, and LOX activity may induce the HR and phyto-alexin accumulation in this system (Preisig and Kuc, 1987; Bostock et al., 1992; Castoria et al., 1992). Consistent with this function, a potato LOX converted arachidonic acid into the highly reactive intermediate 5-S-hydroperoxyeicosatetraenoic acid (5-S-HPTE) that induced phytoalexin accumulation to much greater levels than the elicitor itself (Castoria et al., 1992).
Some LOX isozymes operate in both wound- and pathogen-induced defense signal transduction pathways (Melan et al., 1993; Royo et al., 1999), whereas others, like POTLX-3 and tobacco LOX1 (Veronesi et al., 1996; Rance et al., 1998), appear to have a specialized function in pathogen-induced defense responses only. There is clear evidence that products of the 9-LOX pathway have antimicrobial and cytotoxic properties (Vaughn and Gardner, 1993; Adams et al., 1999; Weber et al., 1999). Hamberg (1999) showed that approximately 95% of the LOX activity in potato leaves produced 9-hydroperoxides of linoleic acid that could be converted into antifungal compounds. Specific local and temporal expression of the LOXs that regulate this pathway would be an efficient means of targeting and controlling pathogen spread early in the development of the disease. Weber et al. (1999) showed that a completely different profile of oxylipins was produced in potato leaves depending on whether they were infected with P. infestans or were wounded. They propose the existence of a 9-LOX pathway in potato leaves, which produces antimicrobial compounds like the divinyl ether fatty acids they detected in infected leaves. It is quite plausible that POTLX-3 serves this function in the defense against pathogen attack in potato.
MATERIALS AND METHODS
Plants, Wounding, and Hormonal Treatments of Leaves
Potato (Solanum tuberosum) plants were grown vegetatively from seed tubers in a greenhouse at 20°C to 22°C under a 16-h daylength. Plants 5 to 6 weeks old were used for all experiments. The cv Superior was used for studies of organ-specific expression, wounding, and hormone treatments. The fourth, fifth, and sixth fully expanded leaves were used for wounding experiments, and leaflets were wounded by crushing the lamina between veins with a hemostat. Leaf-petiole cuttings were used for hormone treatments, and they were incubated with the cut end placed in 100 mL of either water, 100 μm ABA, 100 μm MJ, 100 μm gibberellic acid (GA3), 50 μm auxin (NAA), or 100 μm cytokinin (benzyladenine). For ethylene treatment, the cut ends of leaf-petiole cuttings were kept in water and incubated in hermetically sealed 4-L jars that contained 10 μL/L ethylene. Treatment with MJ was conducted in hermetically sealed 4-L jars. All treatments were incubated under constant light at 22°C, and treated leaves were harvested after 6, 12, 24, and 48 h, frozen in liquid N2, and stored at −80°C. Each treatment contained five leaf-petiole cuttings, and each part of the experiment was conducted at least two times. All chemicals were purchased from Sigma (St. Louis) except MJ, which was purchased from Bedoukian Industries (Danbury, CT).
Pathogens, Inoculum Production, and Inoculation Methods
cv Superior and cv Kennebec (resistance gene R-1) were used for inoculations with Phytophthora infestans. P. infestans strains ME93-2A (U.S. Genotype US8, Mating Type A2, compatible on cv Kennebec and cv Superior) and US940507 (U.S. Genotype US1, Mating Type A1, incompatible on cv Kennebec) were obtained from Dr. W.E. Fry (Department of Plant Pathology, Cornell University). The fungi were grown on Rye B Agar for 2 weeks at 22°C for sporangia production, and the sporangia were harvested by washing the plates with sterile water (Erwin and Ribeiro, 1996). Sporangial suspensions were diluted to approximately 10,000 sporangia per mL. Inoculations were performed by using a detached leaflet assay. Leaflets recently expanded from the fourth, fifth, and sixth leaves from the plant apex were placed in Petri dishes that contained water agar. Droplets (10 μL) of the sporangial suspension were distributed evenly over the abaxial surface of the leaflets. Inoculated leaflets were incubated in dark overnight in a growth chamber at 18°C and then maintained in light at 18°C for the remainder of the incubation period. Samples were taken after 6, 12, and 24 h, and at 24-h intervals for 7 d after inoculation, frozen in liquid N2, and stored at −80°C.
For infiltration with the incompatible bacterial pathogen, Pseudomonas syringae pv phaseolicola, the fourth, fifth, and sixth fully expanded leaves of cv Superior plants 5 weeks old were used. For inoculum production, cells were harvested from bacterial cultures grown on plate count agar (DIFCO Laboratories, Detroit) at 22°C for 48 h. Cells were suspended in sterile distilled water at a final concentration of 1.2 × 108 colony forming units mL−1. The abaxial surface of the leaflets was infiltrated with this bacterial suspension by using a hypodermic syringe with a short piece of latex tubing attached. Infiltration was conducted by forcing the liquid into the intercellular spaces through the stomates. Plants with infiltrated leaves were incubated for 1, 3, 6, 12, 18, or 24 h in the greenhouse. Control leaves were infiltrated with sterile water (mock inoculated). After each incubation time, entire leaflets were collected, frozen immediately in liquid N2, and stored at −80°C until used for RNA extractions.
Isolation and Analysis of POTLX-3 cDNA Clone
A λ-gt11 cDNA library constructed from mRNA extracted from ABA-treated potato leaves (Hildmann et al., 1992), a generous gift from Dr. Salomé Prat (Barcelona), was screened for LOX cDNAs by using a nick-translated 0.85-kb potato LOX fragment. To obtain a potato LOX fragment to use as a probe for screening this library, RT-PCR was conducted with primers based on regions of low degeneracy from known plant LOX sequences. Two PCR primers (5′-CAGCCATATCTCCCAAGTGAA and 5′-TCTCGAGGCATATGTTTT) were synthesized at the DNA Synthesis Facility (Iowa State University). Template cDNA for PCR was obtained by reverse transcription of total RNA extracted from wounded potato leaves. Wounded leaves were used because some plant LOXs can be induced by both wounding and pathogens (Melan et al., 1993). PCR amplification of this cDNA produced one band of approximately 850 bp, which is an expected size based on known plant LOX sequences. This product was cloned into a pCR1 TA cloning vector (Invitrogen, San Diego), sequenced, and identified as a potato LOX fragment due to high sequence identity with other plant LOX genes.
This fragment was used as a heterologous probe to screen the library under low-stringency conditions (40% [v/v] formamide hybridization solution, hybridization carried out at 40°C, and washing at 58°C). The remainder of the screening procedure has been described previously (Polking et al., 1995). Four putative LOX cDNA clones were isolated from more than 500,000 plaque forming units screened. Two of these were approximately 2.8 kb, the expected size of a full-length plant lipoxygenase cDNA. These longest two clones were subcloned into pGEM-11Zf(+) (Promega, Madison, WI). Partial sequencing and sequence analysis indicated that they were identical. Therefore, only one of these clones, pABA-1, was sequenced completely by using automated dideoxy sequencing at the Nucleic Acid Facility (Iowa State University). The clone was designated POTLX-3 (Kolomiets et al., 1996b). The amino acid sequence comparison was conducted by using the GAP and FASTA programs of the Genetics Computer Group of the University of Wisconsin (Madison, WI). The predicted subcellular localization of POTLX-3 protein was determined by the PSORT protein-sorting program (Nakai and Kanehisa, 1992) available on the Nakai server (http://psort.nibb.ac.jp).
Genomic Southern Analysis
Genomic DNA was extracted from cv Superior leaves according to the cetyl-trimethyl-ammonium bromide method (Rogers and Bendich, 1985). Ten micrograms of genomic DNA was digested with restriction endonucleases, electrophoretically separated on a 0.9% (v/v) agarose gel, denatured, and transferred to a MagnaGraph nylon membrane (Micron Separations, Westboro, MA). Membranes were hybridized at high stringency in a 50% (v/v) formamide hybridization buffer at 42°C for 48 h. The hybridization buffer was 50% (v/v) formamide, 6× SSC (1× SSC: 0.15 m NaCl, 0.015 m sodium citrate), 3.3× Denhardt's solution (1× Denhardt's: 0.02% [v/v] Ficoll, 0.02% [v/v] polyvinylpyrrolidone, and 0.02% [v/v] bovine serum albumin), 25 mm sodium phosphate buffer (pH 7.0), 0.115 mg/mL salmon sperm DNA. Membranes were washed in 2× SSC, 0.1% (v/v) SDS at 23°C for 10 min, 1× SSC, 0.1% (v/v) SDS at 65°C for 30 min, followed by 0.1× SSC, 0.1% (v/v) SDS at 65°C for 30 min. For autoradiography, membranes were dried and exposed to x-ray film with intensifying screens for 6 d at −80°C. We used a 32P-labeled 1.4-kb EcoRI fragment of the POTLX-3 cDNA clone as the hybridization probe for Southern and northern analyses, and we refer to it as the POTLX-3 probe. The POTLX-3 probe did not cross-hybridize to POTLX-1 or POTLX-2 sequences (data not shown), which are the two other LOX cDNA clones that we have isolated from potato tubers (Kolomiets et al., 1996a).
Northern Analysis
Total RNA was extracted from leaf tissue of cv Superior or cv Kennebec plants according to the phenol/chloroform extraction procedure (Dix and Rawson, 1983) or by using an RNA isolation kit (TRI reagent, Molecular Research Center, Cincinnati), according to the manufacturer's manual. Various amounts of RNA were size-fractionated via electrophoresis through a 1% (v/v) agarose gel that contained 5 mm methyl-mercury hydroxide and transferred onto a MagnaGraph nylon membrane (Micron Separations). Equal loading of RNA samples and uniform transfer onto a nylon membrane were confirmed by visualizing RNA stained with ethidium bromide under UV light. Membranes were hybridized with the POTLX-3 probe under hybridization conditions identical to those described for Southern analysis. Membranes were washed in 1× SSC, 0.1% (v/v) SDS at 23°C for 15 min, in 0.1× SSC, 0.1% (v/v) SDS at 23°C for 30 min, followed by a final wash in 0.1× SSC, 0.1% (v/v) SDS at 65°C for 30 min. For autoradiography, RNA blots were exposed to x-ray film by using intensifying screens for 2 to 4 d. In the wound-induction study, a duplicate blot was hybridized to a 32P-labeled p755 cDNA fragment that represented a potato proteinase inhibitor II (Pin2) gene isolated from a tuber cDNA library (D.J. Hannapel, unpublished data). This probe was used as a positive control for wounding and hormonal induction. A 1.2-kb wheat 18S ribosomal RNA probe was used to confirm uniform loading of RNA on blots in Figures 4 and 6. Blots presented are representative examples of at least two independent experiments.
LOX Activity Assays
LOX activity was determined as the consumption of oxygen in μmol s−1 g−1 protein by using linolenic acid as a substrate at pH 6.8 and a Clark electrode for detection (Royo et al., 1996). Optimum pH for the reaction mix was determined by monitoring LOX activity at a range of pH values (Royo et al., 1996). Data presented here were the average of three measurements per sample.
ACKNOWLEDGMENTS
We thank Dr. William E. Fry for P. infestans isolates and advice on the protocols that concern the detached leaf assay, sporangia production, and maintenance of the fungus, and Dr. Salomé Prat (Centro de Investigacion y Desarrollo-Consejo Superior de Investigaciones Científicas, Barcelona) for the generous gift of the ABA-induced potato leaf cDNA library.
Footnotes
This work was supported in part by the Iowa Agriculture and Home Economics Experiment Station (journal paper no. J–18164; project nos. 3703 and 3189).
LITERATURE CITED
- Adams LK, Benson EE, Staines HJ, Bremner DH, Milliam S, Deighton N. Effects of lipid peroxidation products 4-hydroxy-2-nonenal and malondialdehyde on the proliferation and morphogenetic development of in vitro plant cells. J Plant Physiol. 1999;155:376–386. [Google Scholar]
- Anderson JM. Membrane derived fatty acids as precursors to second messengers. In: Boss WF, Morre DJ, editors. Second Messengers in Plant Growth and Development. Vol. 6. New York: Alan R. Liss; 1989. pp. 181–212. [Google Scholar]
- Bell E, Creelman RA, Mullet JE. A chloroplast lipoxygenase is required for wound-induced jasmonic acid accumulation in Arabidopsis. Proc Natl Acad Sci USA. 1995;92:8675–8679. doi: 10.1073/pnas.92.19.8675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell E, Mullet JE. Lipoxygenase gene expression is modulated in plants by water deficit, wounding, and methyl jasmonate. Mol Gen Genet. 1991;230:456–462. doi: 10.1007/BF00280303. [DOI] [PubMed] [Google Scholar]
- Bohland C, Balkenhohl T, Loers G, Feussner I, Grambow HJ. Differential induction of lipoxygenase isoforms in wheat upon treatment with rust fungus elicitor, chitin oligosaccharades, chitosan, and methyl jasmonate. Plant Physiol. 1997;114:679–685. doi: 10.1104/pp.114.2.679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bostock RM, Yamamoto H, Choi D, Ricker KE, Ward BL. Rapid stimulation of 5-lipoxygenase activity in potato by the fungal elicitor arachidonic acid. Plant Physiol. 1992;100:1448–1456. doi: 10.1104/pp.100.3.1448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyington JC, Gaffney BJ, Amzel ML. The three-dimensional structure of an arachidonic acid 15-lipoxygenase. Science. 1993;260:1482–1486. doi: 10.1126/science.8502991. [DOI] [PubMed] [Google Scholar]
- Casey R. Sequence of a cDNA clone encoding a potato (Solanum tuberosum) tuber lipoxygenase. Plant Physiol. 1995;107:265–266. doi: 10.1104/pp.107.1.265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castoria R, Fanelli C, Fabbri AA, Passi S. Metabolism of arachidonic acid involved in its eliciting activity in potato tuber. Physiol Mol Plant Pathol. 1992;41:127–137. [Google Scholar]
- Conconi A, Miquel M, Browse JA, Ryan CA. Intracellular levels of free linolenic and linoleic acids increase in tomato leaves in response to wounding. Plant Physiol. 1996;111:797–803. doi: 10.1104/pp.111.3.797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Creelman RA, Mullet JE. Biosynthesis and action of jasmonates in plants. Annu Rev Plant Physiol Plant Mol Biol. 1997;48:355–381. doi: 10.1146/annurev.arplant.48.1.355. [DOI] [PubMed] [Google Scholar]
- Croft KPC, Juttner F, Slusarenko AJ. Volatile products of the lipoxygenase pathway evolved from Phaseolus vulgaris (L.) leaves inoculated with Pseudomonas syringaepv phaseolicola. Plant Physiol. 1993;101:13–24. doi: 10.1104/pp.101.1.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croft KPC, Voisey CR, Slusarenko AJ. Mechanism of hypersensitive cell collapse: correlation of increased lipoxygenase activity with membrane damage in leaves of Phaseolus vulgaris (L.) inoculated with an avirulent race of Pseudomonas syringaepv phaseolicola. Physiol Mol Plant Pathol. 1990;36:49–62. [Google Scholar]
- Deighton N, Muckenschnabel I, Goodman BA, Williamson B. Lipid peroxidation and the oxidative burst associated with infection of Capsicum annuum by Botrytis cinerea. Plant J. 1999;20:485–492. doi: 10.1046/j.1365-313x.1999.00622.x. [DOI] [PubMed] [Google Scholar]
- Dix KP, Rawson JRY. In vivo transcriptional products of the chloroplast DNA of Euglena gracilis. Curr Genet. 1983;7:265–272. doi: 10.1007/BF00376071. [DOI] [PubMed] [Google Scholar]
- Erwin DC, Ribeiro OK. Culture, physiology, and genetics of Phytophthoraspecies. In: Erwin DC, editor. Phytophthora Diseases Worldwide, Ch 3. St. Paul, MN: APS Press; 1996. pp. 42–95. [Google Scholar]
- Farmer EE, Ryan CA. Octadecanoid precursors of jasmonic acid activate the synthesis of wound-inducible proteinase inhibitors. Plant Cell. 1992;4:129–134. doi: 10.1105/tpc.4.2.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrie BJ, Beaudoin N, Burkhart W, Bowsher CG, Rohstein SJ. The cloning of two tomato lipoxygenase genes and their differential expression during fruit ripening. Plant Physiol. 1994;106:109–118. doi: 10.1104/pp.106.1.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fidantsef AL, Bostock RM. Characterization of potato tuber lipoxygenase cDNAs and lipoxygenase expression in potato tubers and leaves. Physiol Plant. 1998;102:257–271. [Google Scholar]
- Geerts A, Feltkamp D, Rosahl S. Expression of lipoxygenase in wounded tubers of Solanum tuberosumL. Plant Physiol. 1994;105:269–277. doi: 10.1104/pp.105.1.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamberg M. An epoxy alcohol synthase pathway in higher plants: biosynthesis of antifungal trihydroxy oxylipins in leaves of potato. Lipids. 1999;34:1131–1142. doi: 10.1007/s11745-999-0464-7. [DOI] [PubMed] [Google Scholar]
- Heitz T, Bergey DR, Ryan CA. A gene encoding a chloroplast-targeted lipoxygenase in tomato leaves is transiently induced by wounding, systemin, and methyl jasmonate. Plant Physiol. 1997;114:1085–1093. doi: 10.1104/pp.114.3.1085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hildebrand DF, Hamilton-Kemp TR, Legg CS, Bookjans G. Plant lipoxygenases: occurrence, properties and possible functions. Curr Top Plant Biochem Physiol. 1988;7:201–219. [Google Scholar]
- Hildmann T, Ebneth M, Peña-Cortés H, Sanchez-Serrano JJ, Willmitzer L, Prat S. General roles of abscisic and jasmonic acids in gene activation as a result of mechanical wounding. Plant Cell. 1992;4:1157–1170. doi: 10.1105/tpc.4.9.1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hornung E, Walther M, Kuhn H, Feussner I. Conversion of cucumber linoleate 13-lipoxygenase to a 9-lipoxygenating species by site-directed mutagenesis. Proc Natl Acad Sci USA. 1999;96:4192–4197. doi: 10.1073/pnas.96.7.4192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato T, Yamaguchi Y, Ohnuma S, Uyehara T, Namai T, Kodama M, Shiobara Y. Structure and synthesis of 11, 12, 13-9Z, 15Z-octadecadienoic acids from rice plants suffering from rice blast disease. Chem Lett. 1986;4:577–580. [Google Scholar]
- Keppler LD, Novacky A. The initiation of membrane lipid peroxidation during bacteria-induced hypersensitive reaction. Physiol Mol Plant Pathol. 1987;30:233–245. [Google Scholar]
- Koch E, Meier BM, Eiben HG, Slusarenko A. A lipoxygenase from leaves of tomato (Lycopersicon esculentumMill.) is induced in response to plant pathogenic pseudomonads. Plant Physiol. 1992;99:571–576. doi: 10.1104/pp.99.2.571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koda Y. The role of jasmonic acid and related compounds in the regulation of plant development. Int Rev Cytol. 1992;135:155–199. doi: 10.1016/s0074-7696(08)62040-9. [DOI] [PubMed] [Google Scholar]
- Kolomiets MV, Hannapel DJ, Gladon RJ. Potato lipoxygenase genes expressed during the early stages of tuberization (accession nos. U60200 and U60201) (PGR 96-065) Plant Physiol. 1996a;112:445. [Google Scholar]
- Kolomiets MV, Hannapel DJ, Gladon RJ. Nucleotide sequence of a cDNA clone for a lipoxygenase from abscisic acid-treated potato leaves (accession no. U60202) (PGR 96-069) Plant Physiol. 1996b;112:445. [Google Scholar]
- Melan MA, Dong X, Endara ME, Davis KR, Ausubel FM, Peterman TK. An Arabidopsis thalianalipoxygenase gene can be induced by pathogens, abscisic acid, and methyl jasmonate. Plant Physiol. 1993;101:441–450. doi: 10.1104/pp.101.2.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakai K, Kanehisa M. A knowledge base for predicting protein localization sites in eukaryotic cells. Genomics. 1992;14:897–911. doi: 10.1016/S0888-7543(05)80111-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohta H, Shida K, Peng Y-L, Furusawa I, Shishiyama J, Aibara S, Morita Y. A lipoxygenase pathway is activated in rice after infection with the rice blast fungus Magnaporthe grisea. Plant Physiol. 1991;97:94–98. doi: 10.1104/pp.97.1.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng YL, Shirano Y, Ohta H, Hibino T, Tanaka K, Shibata D. A novel lipoxygenase from rice: primary structure and specific expression upon incompatible infection with rice blast fungus. J Biol Chem. 1994;269:3755–3761. [PubMed] [Google Scholar]
- Penninckx IAMA, Eggermont K, Terras FRG, Thomma BPHJ, De Samblanx GW, Buchala A, Metraux J-P, Manners JM, Broekaert WF. Pathogen-induced systemic activation of a plant defensin gene in Arabidopsisfollows a salicylic acid-independent pathway. Plant Cell. 1996;8:2309–2323. doi: 10.1105/tpc.8.12.2309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polking GF, Hannapel DJ, Gladon RJ. Characterization of a cDNA encoding 5-aminolevulinic acid dehydratase in tomato (Lycopersicon esculentumMill.) Plant Cell Rep. 1995;14:366–369. doi: 10.1007/BF00238598. [DOI] [PubMed] [Google Scholar]
- Preisig CL, Kuc JA. Inhibition by salicylhydroxamic acid, BW755c, eicosatetraynoic acid, and disulfiram of hypersensitive resistance elicited by arachidonic acid or poly-l-lysine in potato tuber. Plant Physiol. 1987;84:891–894. doi: 10.1104/pp.84.3.891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rance I, Fournier J, Esquerre-Tugaye M-T. The incompatible interaction between Phytophthora parasitica var. nicotianaerace 0 and tobacco is suppressed in transgenic plants expressing antisense lipoxygenase sequences. Proc Natl Acad Sci USA. 1998;95:6554–6559. doi: 10.1073/pnas.95.11.6554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers SO, Bendich AJ. Extraction of DNA from milligram amounts of fresh, herbarium, and mummified plant tissues. Plant Mol Biol. 1985;5:69–76. doi: 10.1007/BF00020088. [DOI] [PubMed] [Google Scholar]
- Royo J, Leon J, Vancanneyt G, Albar JP, Rosahl S, Ortego F, Castanera P, Sanchez-Serrano JJ. Antisense-mediated depletion of a potato lipoxygenase reduces wound induction of proteinase inhibitors and increases weight gain of insect pests. Proc Natl Acad Sci USA. 1999;96:1146–1151. doi: 10.1073/pnas.96.3.1146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Royo J, Vancanneyt G, Perez AG, Sanz C, Stormann K, Rosahl S, Sanchez-Serrano JJ. Characterization of three potato lipoxygenases with distinct enzymatic activities and different organ-specific and wound-regulated expression patterns. J Biol Chem. 1996;271:21012–21019. doi: 10.1074/jbc.271.35.21012. [DOI] [PubMed] [Google Scholar]
- Rusterucci C, Montillet JL, Agnel JP, Battesti C, Alonso B, Knoll A, Bessoule JJ, Etienne P, Suty L, Blein JP, Triantaphylides C. Involvement of lipoxygenase-dependent production of fatty acid hydroperoxides in the development of the hypersensitive cell death induced by cryptogein on tobacco leaves. J Biol Chem. 1999;274:36446–36455. doi: 10.1074/jbc.274.51.36446. [DOI] [PubMed] [Google Scholar]
- Saravitz DM, Siedow JN. The lipoxygenase isozymes in soybean (Glycine max[L.] Merr.) leaves: changes during leaf development, after wounding, and following reproductive sink removal. Plant Physiol. 1995;107:535–543. doi: 10.1104/pp.107.2.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saravitz DM, Siedow JN. The differential expression of wound-inducible lipoxygenase genes in soybean leaves. Plant Physiol. 1996;110:287–299. doi: 10.1104/pp.110.1.287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siedow JN. Plant lipoxygenase: structure and function. Annu Rev Plant Physiol Plant Mol Biol. 1991;42:145–188. [Google Scholar]
- Slusarenko AJ. The role of lipoxygenase in plant resistance to infection. In: Piazza G, editor. Lipoxygenase and Lipoxygenase Pathway Enzymes. Champaign, IL: AOCS Press; 1996. pp. 176–197. [Google Scholar]
- Steczko J, Donoho GP, Clemens JC, Dixon JE, Axelrod B. Conserved histidine residues in soybean lipoxygenase: functional consequences of their replacement. Biochemistry. 1992;31:4053–4057. doi: 10.1021/bi00131a022. [DOI] [PubMed] [Google Scholar]
- Stephenson LC, Bunker TW, Dubbs WE, Grimes HD. Specific soybean lipoxygenases localize to discrete subcellular compartments and their mRNAs are differentially regulated by source-sink status. Plant Physiol. 1998;116:923–933. doi: 10.1104/pp.116.3.923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Mechelen JR, Smits M, Douma AC, Rouster J, Cameron-Mills V, Heidekamp F, Valk BE. Primary structure of a lipoxygenase from barley grain as deduced from its cDNA sequence. Biochem Biophys Acta. 1995;1224:221–225. doi: 10.1016/0005-2760(94)00231-m. [DOI] [PubMed] [Google Scholar]
- Vaughn SF, Gardner HW. Lipoxygenase-derived aldehydes inhibit fungi pathogenic on soybean. J Chem Ecol. 1993;19:2337–2345. doi: 10.1007/BF00979668. [DOI] [PubMed] [Google Scholar]
- Vaughn SF, Lulai EC. Further evidence that lipoxygenase activity is required for arachidonic acid-elicited hypersensitivity in potato callus cultures. Plant Sci. 1992;84:91–98. [Google Scholar]
- Veronesi C, Fournier J, Rickauer M, Marolda M, Esquerre-Tugaye M-T. Nucleotide sequence of an elicitor-induced tobacco lipoxygenase cDNA (GenBank X84040) Plant Physiol. 1995;108:1342. [Google Scholar]
- Veronesi C, Rickauer M, Fournier J, Pouenat M-L, Esquerre-Tugaye M-T. Lipoxygenase gene expression in the tobacco-Phytophthora parasitica nicotianaeinteraction. Plant Physiol. 1996;112:997–1004. doi: 10.1104/pp.112.3.997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber H, Chetelat A, Caldelari D, Farmer EF. Divinyl ether fatty acid synthesis in late blight-diseased potato leaves. Plant Cell. 1999;11:485–493. doi: 10.1105/tpc.11.3.485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Y, Chang P-FL, Liu D, Narasimhan ML, Raghothama KG, Hasegawa PM, Bressan RA. Plant defense genes are synergistically induced by ethylene and methyl jasmonate. Plant Cell. 1994;6:1077–1085. doi: 10.1105/tpc.6.8.1077. [DOI] [PMC free article] [PubMed] [Google Scholar]