Abstract
Objective
To explore the current evidence concerning the effect of oral antioxidant supplementation on various male fertility outcomes, as antioxidants are widely available compounds that are commonly used for the treatment of male infertility.
Materials and methods
PubMed, Medline and Cochrane electronic databases were searched according to a modified Preferred Reporting Items for Systemic Reviews and Meta-Analyses (PRISMA) guidelines looking for studies investigating the effect of antioxidant therapy on infertile men. The studies were explored looking for antioxidants: (i) types and doses; (ii) mechanism of action and rationale for use; and (iii) effect on the different outcome measures reported.
Results
In all, 26 studies reported a significant positive effect of antioxidant therapy on basic semen parameters, advanced sperm function, outcomes of assisted reproductive therapy, and live-birth rate. Vitamin E, vitamin C, carnitines, N-acetyl cysteine, co-enzyme Q10, zinc, selenium, folic acid and lycopene were most commonly used. The vitamins’ mechanism of action and reported doses is presented in Table 1, Table 2.
Conclusion
Antioxidants generally have a favourable effect on male fertility. Further studies are needed to identify the optimal antioxidant regimen that can be used safely and efficiently in clinical practice.
Abbreviations: ART, assisted reproductive therapy; coQ10, co-enzyme Q10; DDS, DNA degraded sperm; ICSI, intracytoplasmic sperm injection; IVF, in vitro fertilisation; LAC, l-acetyl carnitine; LC, l-carnitine; MeSH, Medical Subject Heading; NAC, N-acetyl cysteine; OAT, oligoasthenozoospermia; OS, oxidative stress; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; ROS, reactive oxygen species; SDF, sperm DNA fragmentation
Keywords: Male infertility, Antioxidants, Reactive oxygen species, Semen analysis, Sperm DNA fragmentation
Introduction
Infertility is defined as the inability to conceive after at least 12 months of regular, unprotected intercourse with a prevalence of 15% of couples worldwide. Male factors contribute to almost half of the reported cases [1], [2] and are thought to occur secondary to derangement in testicular function or obliteration of the reproductive passages. Several causes of male factor infertility have been recognised; however, in most cases the exact cause remains unknown [1]. Efforts aimed to unravel the idiopathic causes of male infertility at the molecular level have highlighted the significant contribution of oxidative stress (OS), a term given to describe the imbalance of the bodies’ redox state caused either by too high levels of oxidants or too low amounts of antioxidants.
Reactive oxygen species (ROS) or ‘free radicals’ are highly reactive oxygen-derived molecules characterised by having unpaired electrons in their outer valence orbital. They include oxygen-centred radicals (hydroxyl radical, nitric oxide radical, and superoxide anion radical) and non-radical derivatives (hydrogen peroxide, peroxynitrite anion, and hypochlorous acid) [3]. ROS play an important role in cell signalling and homoeostasis. They are produced by the sperm cell in small quantities providing beneficial functional effects including initiation of sperm capacitation, regulation of sperm maturation, and enhancement of cellular signalling pathways [4]. However, high levels of ROS may have paradoxical effects on sperm function, ultimately resulting in infertility. Increased DNA damage and lipid peroxidation are noticeable effects of exaggerated ROS levels in seminal plasma [5]. Several endogenous (immature spermatozoa, leucocytes, varicocele) and exogenous (testicular hyperthermia, environmental and habitual exposures) conditions have been recognised as potential causes of increased ROS production.
ROS are counterbalanced by antioxidants that help maintain the equilibrium in the redox potential desired for optimal sperm function [6]. Seminal fluid is rich in antioxidants that nourish and protect the sperm. They exist in two forms; an enzymatic and a non-enzymatic antioxidant system [7]. The enzymatic system is comprised of glutathione peroxidase, superoxide dismutase, and catalase. These enzymes are naturally occurring in the sperm cell or seminal plasma and are thought to originate from the prostate. The non-enzymatic system, on the other hand, is composed of multiple compounds that are consumed through diet or as supplements.
When excessive amounts of ROS are produced, or when antioxidant activity fails, the equilibrium state between oxidation and reduction is disrupted, resulting in OS. Spermatozoa are particularly vulnerable to OS. They contain very low levels of enzymatic antioxidants, which are insufficient in protecting the sperm against high ROS levels. Furthermore, the exceptionally high amounts of polyunsaturated fatty acids, especially docosahexaenoic acid, in the sperm cell’s plasma membrane are appealing reactants for ROS-induced oxidation reactions.
Major advances have been witnessed in the field of male infertility over the past few decades. Tests of sperm function, such as sperm DNA fragmentation (SDF), and measures of OS have been added to the clinician’s armamentarium to provide a better understanding of the true male fertility potential [8]. Also, the breakthroughs perceived in assisted reproductive therapy (ART) allowed men who were deemed infertile to father their biological children. Nevertheless, OS remains a key factor that can potentially influence the reproductive outcome, whether naturally or with ART. When compared to fertile men, up to 25% of infertile men were found to have significant levels of ROS in their semen [9]. OS was also found to have a significant negative influence on semen parameters, fertilisation rate, embryonic development, and pregnancy rate [10], [11]. Therefore, identifying and treating OS through either ROS reduction or antioxidant therapy appear to be an appealing tactic in infertility management. However, having said that, a clear consensus about the clinical effectiveness of antioxidant therapy is still lacking [12]. This is principally because studies investigating different antioxidant forms vary considerably in the dosage or combinations used, as well as outcome measures. The aim of the present literature review was to investigate the most common antioxidants used for the treatment of male infertility and explore the doses that are associated with potential benefit on basic semen parameter results, advanced sperm function tests, outcomes of ART, and live-birth rate.
Material and methods
Research strategy
The search strategy was conducted according to a modified Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [11]. A literature search was performed using PubMed, Medline and Cochrane electronic databases to identify studies investigating the utility of antioxidants for male infertility. The search was executed using the following keywords; ‘antioxidants’, ‘semen parameters’, ‘male infertility’, ‘pregnancy rate’, ‘live birth rate’ and ‘sperm function’. Medical Subject Heading (MeSH) phrases included: (‘Antioxidants’[MeSH]) AND ‘Infertility, Male’[MeSH], (‘Antioxidants’[MeSH]) AND ‘Pregnancy Rate’[MeSH], (‘Antioxidants’[MeSH]) AND ‘Semen Analysis’[MeSH].
Study selection
The generated list of articles was screened by title and abstract by the two authors (A.M. and A.A.) and then relevant full papers were examined. Review articles were also explored to find additional appropriate papers. The exclusion criteria were based on gender (females), species (other animals), language (non-English) and study methods (retrospective, case report, editorial or commentary). Data were then extracted cross-checked and verified.
Outcome measures
The outcomes of interest were as follows: antioxidants type and dosage, their mechanism of action, their impact on basic semen parameters and advanced sperm function tests, and their influence on ART outcome and live-birth rate. Relevant results were tabulated.
Results
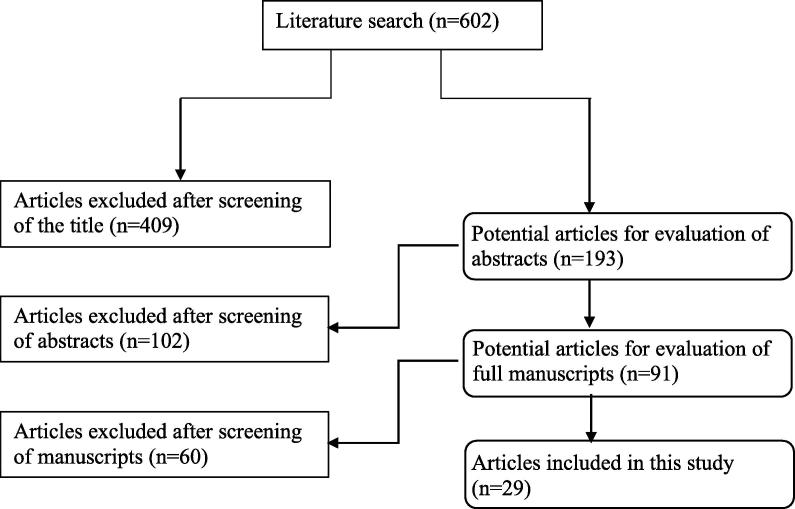
The search strategy yielded 602 articles, with 511 excluded on the basis of the title and/or abstract. The remaining 91 articles were screened, with 29 identified that passed the inclusion and exclusion criteria (Fig. 1). They included 19 randomised clinical trials [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31] and 10 prospective studies [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42]. In 26 studies [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], a significant positive effect was detected for antioxidant therapy on basic semen parameters, advanced sperm function tests, ART outcomes or live-birth rate. The most commonly investigated compounds and their doses were as follows: vitamin E (400 mg), vitamin C (500–1000 mg), carnitines (500–1000 mg), N-acetyl cysteine (NAC; 600 mg), co-enzyme Q10 (CoQ10; 100–300 mg), zinc (25–400 mg), selenium (Se) (200 µg), folic acid (0.5 mg), and lycopene (6–8 mg). Table 1 shows the mechanism of action of each proposed antioxidant. The list of antioxidants used in the specific searched outcome measures is presented in Table 2 [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42].
Fig. 1.
PRISMA flowchart of literature search.
Table 1.
Mechanism of action of commonly used antioxidants.
| Antioxidant compound | Mechanism of action |
|---|---|
| Ascorbic acid (vitamin C) | Neutralises free radicals |
| Tocopherol (vitamin E) | Neutralises free radicals |
| Folate (vitamin B9) | Scavenges free radicals |
| Selenium | Enhancement of enzymatic antioxidant activity |
| Zinc | Inhibition of NADPH oxidase |
| Carnitines | Neutralises free radicals and acts as an energy source |
| CoQ10 | In its reduced form, scavenges free radicals intermediate in mitochondrial electron transport system |
| NAC | Enhances enzymatic antioxidant activity |
| Lycopene | Quenches free radicals |
NADPH, nicotinamide adenine dinucleotide phosphate.
Table 2.
Proposed antioxidants in various clinical scenarios based on available evidence.
| Clinical circumstance | Antioxidant | Reference |
|---|---|---|
| Basic semen parameters | ||
| Oligozoospermia | Vitamin E, vitamin C, NAC, carnitines, CoQ10, lycopene, selenium and zinc | |
| Vitamin E (300 mg) | [14] | |
| Vitamin E (180 mg), vitamin A (30 mg) and essential fatty acids or NAC (600 mg) | [33] | |
| NAC (600 mg) + other vitamins/minerals | [15] | |
| LC (2 g) | [16] | |
| CoQ10 (300 mg) | [17] | |
| NAC (600 mg) and selenium (200 µg) | [18] | |
| Folic acid (5 mg) + zinc (66 mg) | [19] | |
| Lycopene (2 mg) | [34] | |
| Asthenozoospermia | Vitamin E, vitamin C, NAC, carnitines, CoQ10, lycopene, selenium and zinc | |
| Vitamin E (400 mg) + selenium (200 μg) | [35] | |
| Zinc (400 mg), vitamin E (20 mg) and vitamin C (10 mg) | [20] | |
| LC (2 g) and LAC (1 g) | [21] | |
| CoQ10 (300 mg) | [17] | |
| NAC (600 mg) | [22] | |
| NAC (600 mg) and selenium (200 µg) | [18] | |
| Lycopene (2 mg) | [34] | |
| Teratozoospermia | Vitamin E, NAC, lycopene, selenium and zinc | |
| Vitamin E (400 mg) + selenium (200 μg) | [35] | |
| NAC (600 mg) and selenium (200 µg) | [18] | |
| Zinc (400 mg), vitamin E (20 mg) and vitamin C (10 mg) | [20] | |
| Lycopene (8 mg) | [36] | |
| Advanced sperm function | ||
| High SDF | Vitamin E, vitamin C, zinc, selenium and folic acid | |
| Vitamin E (1 g) + vitamin C (1 g) | [23] | |
| Vitamin C (400 mg), vitamin E (400 mg), b-carotene (18 mg), zinc (500 µmol) and selenium (1 µmol) | [37] | |
| LC (1500 mg); vitamin C (60 mg); CoQ10 (20 mg); vitamin E (10 mg); zinc (10 mg); folic acid (200 μg), selenium (50 μg); vitamin B12 (1 μg) | [38], [39] | |
| OS | Vitamin E, vitamin C, NAC, selenium and zinc | |
| Vitamin E (300 mg) | [24] | |
| Vitamin E (180 mg) and β-carotene (30 mg) | [33] | |
| Vitamin E (20 mg), vitamin C (10 mg) and zinc (400 mg) | [20] | |
| Vitamin E (400 mg) and selenium (225 g) | [25] | |
| NAC (600 mg) | [22] | |
| Improving success rate of ART | Vitamin E, vitamin C, lycopene, CoQ10, folic acid, selenium, zinc | |
| Vitamin E (200 mg daily) | [40] | |
| Lycopene (6 mg), vitamin E (400 IU), vitamin C (100 mg), zinc (25 mg), selenium (26 g), folate (0.5 mg) and garlic (1 g) | [26] | |
| Vitamin E (600 mg) | [29] | |
| Vitamin C (1 g) + vitamin E (1 g) | [42] | |
| Live-birth rate | Vitamin E, vitamin C, carnitines, coQ10, and zinc | |
| CoQ10 (300 mg) | [17], [41] | |
| Vitamin E (300 mg) | [24] | |
| Zinc (5000 mg) | [27] | |
| Vitamin E (1 g) + vitamin C (1 g) | [23] | |
| Carnitines: LC (2 g) + LAC (1 g/day) | [28] |
Antioxidants
Antioxidants and the redox paradox
Antioxidants are biological or chemical compounds that scavenge free radicals, neutralise their effect and halt the chain reaction leading to OS in body tissues. To overcome OS in infertile patients, antioxidants have been a common prescription for men seeking fertility supported by their relatively inexpensive and easily accessible nature. However, several studies have shown conflicting results for the effect of the antioxidant therapy on male fertility. Whilst a number of studies conveyed a favourable effect on basic semen parameters, advanced sperm function tests, and pregnancy rates; others failed to confirm such an effect or even reported a negative influence on male fertility. This may be attributed to the extensive heterogeneity that characterises the studies conducted on this particular topic. Furthermore, the ideal balance of the redox system necessary for optimal sperm function is not known and overconsumption of antioxidants may result in reductive stress that could cause detrimental effects on human health and well-being. Impairment of mitochondrial activity [43], reduction in blood–brain barrier permeability [44], and attenuation of endothelial cell proliferation [45] are consequences that have been reported to occur secondary to reductive stress.
Antioxidants mechanism of action and rationale for use
A large number of compounds with antioxidant properties have been investigated for the treatment of infertile men (Table 1). An understanding of the antioxidant mechanism of action of commonly used compounds is important before embarking on the evidence associated with their use in clinical practice.
Vitamin E (α-tocopherol): this powerful chain breaking antioxidant is an organic fat soluble compound located mainly in cell membranes. It quenches free hydroxyl radicals and superoxide anions thereby reducing lipid peroxidation initiated by ROS at the level of plasma membranes. A direct relationship has been detected between the levels of vitamin E in seminal plasma and the percentage of motile spermatozoa in semen [46]. Furthermore, lower levels of vitamin E were observed in the semen of infertile men [47].
Vitamin C (ascorbic acid): this water-soluble compound exists at a concentration 10-times higher in seminal plasma than in blood serum [48]. It neutralises hydroxyl, superoxide and hydrogen peroxide radicals providing protection against endogenous oxidative damage [49]. Seminal fluid analyses from infertile men with asthenozoospermia were found to contain lower vitamin C levels and higher ROS levels than those obtained from fertile controls [50].
Carnitines [l-carnitine (LC) and l-acetyl carnitine (LAC)] are also water-soluble antioxidants involved in sperm metabolism fuelling important activities like sperm motility. Indeed, in vitro studies of sperm cultured in media containing carnitines had higher motility and viability in comparison with controls [51], [52]. They exhibit their antioxidant activities through scavenging superoxide anions and hydrogen peroxide radicals thereby inhibiting lipid peroxidation [53]. Significantly lower carnitine levels were observed in semen samples from infertile men with oligoasthenoteratozoospermia [54].
CoQ10 is a vital antioxidant omnipresent in almost all body tissues. It is particularly present at high concentrations in sperm mitochondria involved in cellular respiration and plays an integral role in energy production [55]. This contribution rationalises its use as a pro-motility and antioxidant molecule. Furthermore, CoQ10 inhibits superoxide formation delivering protection against OS-induced sperm dysfunction. A significant negative correlation between CoQ10 levels and hydrogen peroxide has been reported and a linear correlation between CoQ10 levels in seminal plasma sperm count and motility was detected [56].
NAC: an amino acid that is converted in body tissues to cysteine, a precursor of glutathione. The latter is an important naturally occurring antioxidant capable of neutralising various ROS preventing their detrimental effects. Additionally, NAC is also capable of directly reducing OS through scavenging hypochlorous acid and hydroxyl radicals [57]. These antioxidant properties of NAC were well documented via its favourable influence on germ cell survival [58]. In vitro studies showed significant reductions in ROS levels and improvement in sperm motility after incubation of semen samples with NAC [59].
Selenium: an essential trace element whose involvement in spermatogenesis is thought to stem from its ability to protect sperm DNA against OS damage in a mechanism that is not very well understood. As selenium is a major constituent of a specific group of proteins called selenoenzymes, its antioxidant properties are thought to stem from its ability to augment the function of glutathione. More than 25 selenoproteins exist, such as phospholipid hydroperoxide glutathione peroxidase (PHGPX) [60] and sperm capsular selenoprotein glutathione peroxidase [61], which help maintain sperm structural integrity [62]. Selenium deficiency has been most commonly associated with morphological sperm mid-piece abnormalities and impairment of sperm motility [63].
Zinc: another essential trace element with ubiquitous biological roles [64]. It plays a vital role in the metabolism of RNA and DNA, signal transduction, gene expression, and regulation of apoptosis. Its antioxidant properties are thought to result from its ability to decrease production of hydrogen peroxide and hydroxyl radicals through antagonising redox-active transition metals, such as iron and copper [65]. Zinc concentrations of seminal plasma were found to be significantly higher in fertile men in comparison with subfertile men [66]. Zinc is thought to deliver an important protective effect on sperm structure. Sperm flagellar abnormalities, such as hypertrophy and hyperplasia of the fibrous sheath, axonemal disruption, defects of the inner microtubular dynein arms, and abnormal or absent mid-piece have all be associated with zinc deficiency [19].
Folic acid (vitamin B9): involved in nucleic acid synthesis and amino acid metabolism. Its use in the treatment of male infertility is based on its free radical scavenging abilities. Folic acid intake was associated with an elevation in reduced:oxidised glutathione ratio [67].
Lycopene: a naturally synthesised carotenoid present in fruits and vegetables. Its powerful ROS quenching abilities make it a major contributor to the human redox defence system [68]. Lycopene is detected at high concentrations in human testes and seminal plasma with levels that tend to be lower in infertile men [69].
Antioxidant effect on basic semen parameters
Semen analysis remains the cornerstone test for male fertility assessment. Its relative simplicity and wide availability have made it the initial investigation of choice for evaluation of sperm quality and male fertility potential [70]. In addition to the well-known shortcomings of semen analysis, the continuous updates witnessed in its reference values further confound the evidence surrounding the potential effect of antioxidants. This drawback is most evident on the inclusion criteria used by studies as patients who were considered to have abnormal sperm quality before the 2010 update to the WHO reference values were labelled ‘normal’ after its implementation. Nonetheless, several studies have reported an improvement in basic semen parameters after the oral intake of antioxidants either alone or in combination.
Vitamin E has mainly been used in conjunction with other vitamins and minerals. One of the earlier studies investigating vitamin E alone (300 mg daily) on 380 infertile men reported significant improvement in sperm motility, at a mean (SD) of +25.2 (1.1)% (P < 0.001) [23]. A comparative prospective randomised study examined the effectiveness of vitamin E alone, clomiphene citrate alone or a combination of both treatments on semen parameters of 90 patients with idiopathic oligoasthenozoospermia (OAT). After a treatment duration of 6 months, the study revealed the presence of a significant improvement in sperm concentration (P = 0.001) and sperm motility (P < 0.001) with the combined regimen [13]. Another observational study investigated a daily regimen of vitamin E (400 mg) + selenium (200 μg), for a period of 100 days, on 690 infertile men with idiopathic asthenoteratospermia. Results revealed that 52.6% of patients showed a significant improvement in sperm motility, morphology, or both (P ≤ 0.001) [34]. Comhaire et al. [32] in an open prospective study of 27 infertile men, showed that the combination of vitamin E, vitamin A and essential fatty acids resulted in improvement in sperm concentration in oligozoospermic men from a mean (SD) of 7.4 (1.3) to 12.5 (1.9) × 106 sperm/mL. However, there was no change in sperm motility or morphology. On the other hand, a few other studies failed to reproduce any significant effect on semen parameters using vitamin E as a single treatment [28], [42] or in combination with other antioxidants [30].
Vitamin C as a single agent was investigated in an earlier study by Dawson et al. [71]. The authors treated 90 men who were heavy smokers with a daily dose of 200 mg or 1000 mg vitamin C or placebo for 1 month, reporting significant improvement in sperm quality only in the vitamin C groups in a dose-dependent manner. Another study examined vitamin C as an adjunct therapy for patients undergoing varicocelectomy. In all, 115 patients were randomised to either receive 500 mg daily vitamin C or placebo for a total of 3 months after undergoing varicocelectomy. The authors reported a statistically significant improvement in sperm motility (P = 0.041) and morphology (P < 0.001) in the treatment group compared with the placebo group [72]. Despite the lack of studies examining the effect of vitamin C (as a single agent) for the treatment of male subfertility, several antioxidant supplements containing vitamin C have been investigated. Omu et al. [19] reported a significant improvement in sperm motility (P < 0.001) with a combination of zinc, vitamin E and vitamin C. Another study by Galatioto et al. [14] examined the effect of an antioxidant regimen (containing NAC and vitamin C) in patients with varicocele with persistent oligozoospermia 6 months after radiographic embolisation. In all, 20 patients received treatment with antioxidants and were compared to 22 controls. A statistically significant increase in sperm count (P = 0.009) was recorded only in the treatment group, without a significant influence on sperm motility or morphology.
On the contrary, a randomised controlled double-blind study by Rolf et al. [30] failed to show an improvement in semen parameters in couples with male factor infertility after the administration of an oral dose of vitamin C (1000 mg) and E (800 mg) for 56 days.
Carnitines were shown to have a significant influence particularly on sperm motility [20], [27], [73]. A placebo-controlled double-blind randomised trial comparing a combined treatment of LC (2 g) and LAC (1 g) for 2 months’ duration to placebo in men with OAT showed significant improvement in all semen parameters; however, with the most significant increase was in sperm motility (both forward and total) [20].
Balercia et al. [73] confirmed such response to therapy specifically amongst patients with lower baseline values of motility. Cavallini et al. [27] investigated patients with low-grade varicocele and idiopathic infertility and reported a significant improvement in all semen parameters amongst patients treated with LC and LAC in comparison with placebo. A double-blind placebo controlled randomised cross-over study investigated the efficacy of LC in improving sperm quality in 30 infertile men. Patients were randomised to receive either LC or placebo for 2 months, after a washout period of similar duration the two groups switched treatment for an additional 2 months. The authors reported significant improvements in mean sperm concentration at the end of the study period (P < 0.05) [15]. On the contrary, Sigman et al. [31] treated 26 asthenozoospermic men with LC (1000 mg) and LAC (500 mg) daily for 12 weeks and failed to show any significant improvement in sperm motility.
CoQ10 has been shown to significantly improve sperm concentration and motility in comparison with placebo [16]. Safarinejad [16] randomly assigned 212 infertile men with idiopathic OAT to receive either 300 mg CoQ10 or a placebo orally for 26 weeks. He reported a significant increase in sperm density and motility with CoQ10 therapy (P = 0.01). A double-blind placebo-controlled clinical trial examining the effect of CoQ10 on semen parameters and plasma antioxidant enzyme levels showed a significant improvement in sperm morphology (P = 0.037), catalase (P = 0.041), and superoxide dismutase (P < 0.001) in the treatment group compared with the placebo group [74]. A systemic review of three randomised controlled clinical trials on 332 infertile men revealed that treatment with CoQ10 (200–300 mg daily) resulted in a significant increase in sperm concentration (mean difference [MD] 5.33 × 106 sperm/mL, P < 0.001) and motility (MD 4.5%, P < 0.001) [75].
NAC was investigated in a randomised placebo-controlled study of 120 patients with idiopathic infertility. Patients were randomised to receive a daily dose of 600 mg NAC (n = 60) or placebo (n = 60) for 3 months. Results revealed a significant improvement in volume, motility, and viscosity of semen with NAC therapy in comparison with placebo [21]. The combination of 600 mg NAC and 200 µg selenium resulted in a significant improvement in all semen parameters with a dose-dependent positive correlation between the sum of selenium and NAC concentrations, and mean sperm concentration, motility, and percentage of normal morphology [17].
Folic acid was investigated in a double-blind, placebo-controlled interventional study, where 108 fertile men and 103 subfertile men were randomised into four groups; folic acid only, zinc only, a combination of both folic acid and zinc, and placebo. After 26 weeks of treatment, a statistically significant increase (74%) in total normal sperm concentration was noted amongst the subfertile group receiving combined therapy [18].
Selenium has been less frequently investigated for the treatment of subfertile men. As previously noted, a randomised placebo-controlled clinical trial [17] included 468 infertile men with idiopathic OAT who were treated with either selenium (200 µg) alone, NAC (600 mg) alone, a combination of both supplements, or placebo for 26 weeks. Results showed a significant improvement in all semen parameters in the treatments groups in comparison with placebo with an additive beneficial effect. A strong correlation was seen between the sum of the selenium and NAC concentrations, and mean sperm concentration (r = 0.67, P = 0.01), sperm motility (r = 0.64, P = 0.01), and percentage normal morphology (r = 0.66, P = 0.01) [17]. Furthermore, the combination of selenium with vitamin E resulted in an increase in sperm motility [24], [76]. On the other hand, treatment with selenium (300 mg) daily for 48 weeks did not result in a significant influence on semen parameters of a group of normozoospermic men [29].
Zinc was compared to no therapy in a prospective trial in men with asthenozoospermia given for 3 months. Results revealed a significant improvement in sperm concentration (P < 0.02), progressive motility (P < 0.05), fertilising capacity (P < 0.01), and a reduction in the incidence of anti-sperm antibodies (P < 0.01) [19]. Oral zinc supplementation successfully restored seminal catalase-like activity and improved sperm concentration and progressive motility in a group of asthenozoospermic men [77].
Lycopene therapy was assessed in 30 men with idiopathic OAT. Patients were treated with 2 mg lycopene twice daily for 3 months [33]. The authors reported statistically significant improvements in sperm concentration and motility in 66% and 53% of patients, respectively. However, the improvements were only significant in patients who had baseline sperm concentrations of >5 × 106 sperm/mL. A similar dose of lycopene was also used for the treatment of 50 patients with idiopathic OAT by Mohanty et al. [35]. After a follow-up period of 1-year, sperm concentration, motility, and morphology improved in 70%, 54% and 38% of patients, respectively [35].
Antioxidant effect on advanced sperm function tests
Basic semen analysis has been criticised for being a poor predictor of fecundity. Whilst it provides useful information on the patency of sperm production, secretions of the accessory organs, as well as ejaculation and emission, it does not predict fertility [78], [79]. It provides no insights into the functional potential of the spermatozoon to fertilise an ovum or to undergo the subsequent maturation processes required to achieve fertilisation. As such, advanced tests of sperm function were developed to provide a better understanding of the true contribution of the human sperm to conception aiming to improve the predictive power of semen studies in this regard. Of the different advanced sperm function tests that have been developed, SDF and measures of OS have been most commonly investigated.
SDF
Research published in recent years has raised our understanding of the implications of SDF on male factor fertility [8]. Most DNA in human sperm is bound to protamine giving rise to a condensed form of chromatin that (i) is easy to transport via the sperm head and (ii) is more resistant to damage during sperm transport through the reproductive tracts [80]. However, SDF can occur secondary to errors in chromatin packaging or remodelling that may develop during spermatogenesis [81] or after exposure to seminal OS [82] during epididymal transit. Both in vitro and in vivo studies have confirmed that elevated SDF may negatively influence fertility through its effect on fertilisation, early embryo development, implantation and pregnancy [50], [83], [84], [85]. As such, treatment of OS seems to be a justified approach to minimise the incidence of SDF in semen samples.
The effect of dietary antioxidant supplementation on sperm DNA integrity has been investigated in few studies [22], [25], [36], [86]. In general, these reports assessed antioxidant effect on small-sized samples and for short treatment durations; however, they did report a beneficial effect on measures of SDF. Greco et al. [22] assessed the utility of 1 g vitamin C + 1 g vitamin E daily for 2 months vs placebo in 64 patients with unexplained infertility and elevated levels of SDF. Whilst no effect on semen parameters was reported, the authors identified significant reduction in the percentage of SDF (P < 0.001) in the treatment group. In another study a statistically significant decrease in SDF by 19% (P < 0.001) was detected with a combination of antioxidants containing zinc and selenium [36]. After various periods of sperm storage (0, 2, 6, 8 and 24 h) at 37 °C, Abad et al. [37] examined the effects of oral antioxidant therapy (LC, CoQ10, vitamins E and C, zinc, and selenium) on the dynamics of SDF, measured with the sperm chromatin dispersion test. This antioxidant combination regimen was used on 20 infertile men with asthenoteratozoospermia for 3 months. The authors revealed significant reductions in SDF measures at each experimental time-point (P < 0.05). Moreover, a significant reduction in the portion of highly DNA degraded sperm (DDS) was detected after antioxidant therapy (P = 0.04).
The utility of antioxidant therapy in patients with high SDF secondary to varicocele has been investigated. In one study, 20 infertile men with low-grade varicocele and high SDF levels, measured with the sperm chromatin dispersion test, were treated with a combined antioxidant regimen containing LC, vitamin C, CoQ10, vitamin E, zinc, folic acid, selenium, and vitamin B12 for 3 months [38]. The authors reported a significant decrease in SDF levels (−22.1%, P = 0.02) and a significant increase in sperm concentration (P = 0.04). After assessing the amount of highly DDS, the authors revealed that 31.3% fewer DDS were observed after antioxidant therapy (P = 0.07).
Measures of OS
In recent years, screening for OS has been incorporated into the evaluation of infertile men due to evidence confirming its utility in various clinical scenarios [87]. Assessing seminal OS levels over time would help in monitoring antioxidant therapies and define effective doses and durations. Nevertheless, test availability, complexity, cost-effectiveness, and lack of a universally accepted analysis method are some of the drawbacks that still prevent its routine use in clinical practice. Many different assays are available; they are classified into direct assays (e.g. chemiluminescence and flow cytometry assays) that measure the degree of oxidation within the sperm cell membrane or indirect assays (e.g. myeloperoxidase test, lipid peroxidation levels and measures of redox potential) that estimate the detrimental effects of OS on cells and tissues [88]. Each class of tests has its advantages and disadvantages. Direct assays can provide accurate measures of OS; however, they are expensive and require superior expertise to quantify the short-lived ROS [89]. On the other hand, indirect assays are relatively simple and less expensive; however, they assess an end state that could be secondary to other unknown pathological processes.
Several studies examining the effect of antioxidant therapy on male fertility have assessed their influence on OS as an outcome measure. Therapy with vitamin E (300 mg daily) for 6 months significantly reduced lipid peroxidation in semen samples obtained from 110 asthenozoospermic men [23]. Similarly, Comhaire et al. [32] reported a significant reduction in seminal ROS levels with the combined therapy of 180 mg vitamin E and 30 mg β-carotene in a group of 27 infertile men.
Omu et al. [19] assessed the utility of daily vitamin E (20 mg), vitamin C (10 mg) and zinc (400 mg) for 3 months on 45 asthenozoospermic men. The authors confirmed a reduction of OS measures through detecting a significant twofold decrease in malondialdehyde (P < 0.01), an indicator of lipid peroxidation, a significant decrease in pro-apoptosis markers (P < 0.05), and a significant increase in total antioxidant capacity (P < 0.01).
In another study, vitamin E (400 mg) and selenium (225 µg) consumption for 3 months resulted in significant reductions in the malondialdehyde level, together with significant improvements in sperm motility and viability [24].
A direct dose- and time-dependent reduction of seminal ROS has been observed by Oeda et al. [59] after incubating semen samples with NAC, suggesting that treatment of patients with NAC might be a useful option for reducing OS.
Clinically, a randomised placebo-controlled study of 120 patients with idiopathic infertility receiving either 600 mg of NAC daily or placebo showed significant improvement in sperm motility with NAC treatment in comparison with placebo [21]. Additionally, the authors reported higher serum total antioxidant capacity and lower total peroxide and OS index in the NAC-treated group compared with the placebo group.
A systemic review by Gharagozloo and Aitken [90] addressed the impact of oral antioxidants on measures of sperm OS and/or DNA damage. The authors selected 20 trials studying this outcome and showed that a significant reduction in OS or SDF after treatment with antioxidants was reported by 19 of them.
Antioxidant effect on ARTs and live-birth rate
OS from both male and female origins may affect the clinical outcome of ART. ROS in seminal plasma were found to have significant negative correlation with fertilisation rate in both in vitro fertilisation (IVF)/intracytoplasmic sperm injection (ICSI) programmes [91]. The toxic effects exhibited by OS characterised by the alteration of lipids, proteins, and nucleic acids may cause loss of membrane integrity, enzyme inactivation, and structural damage to DNA, all of which are necessary for embryo development and live birth. Few studies have looked into the effect of antioxidant therapy on the live-birth rate whether naturally of after ART.
Vitamin E supplementation had a beneficial effect on in vitro sperm function and fertilisation rates in IVF compared to placebo groups [23], [39]. Vitamin E (200 mg daily) was found to significantly reduce malondialdehyde levels and improve fertilisation rates with IVF, specifically in patients with prior history of IVF failure [39]. Similarly, Kessopoulou et al. [28] randomised 30 men with high seminal ROS levels into a treatment group receiving vitamin E (600 mg) or placebo for 3 months. After a 1 month washout phase, despite not reporting any significant influence on basic semen parameters, patients receiving vitamin E therapy showed significant enhancement in in vitro sperm function characterised by better zona pellucida binding.
Another randomised double-blind, placebo-controlled trial investigated a combined antioxidant regimen, including lycopene 6 mg, vitamin E 400 IU, vitamin C 100 mg, zinc 25 mg, selenium 26 µg, folic acid 0.5 mg and garlic 1 g in couples undergoing ICSI. A significant improvement in the viable pregnancy rate was identified in the treatment group (38.5%) compared with placebo (16%) (P = 0.03) [25].
An increase in spontaneous pregnancy rates along with improvement in sperm concentration and motility was reported by studies evaluating the oral use of CoQ10 on male infertility [16], [40]. CoQ10 therapy was also found to influence the outcome of ICSI [55]. In 17 patients with low fertilisation rates after a prior ICSI trial were treated with oral CoQ10 (60 mg/day) for a mean of 103 days before a subsequent ICSI trial. Whilst no significant differences were noted in most sperm parameters, a significant improvement in fertilisation rates from a mean (SD) of 10.3 (10.5) to 26.3 (22.8)% after CoQ10 therapy (P < 0.05) was reported.
Another study of similar design assessed the effect of oral antioxidant supplementation (1 g vitamin C + 1 g vitamin E) on males who had at least one unsuccessful previous ICSI trial. After treatment for 2 months, implantation rates increased from 2% to 19% and pregnancy rates increased from 7% to 48% [41].
A recent Cochrane review of 48 randomised controlled clinical trials examining the effects of antioxidant therapy in male infertility revealed that live-birth and pregnancy rates were reported in four and seven trials, respectively [92]. Despite a considerable variability in the reported antioxidant effect on semen parameters, a statistically significant improvement in live-birth rate (odds ratio [OR] 4.21, 95% CI 2.08–8.51, P < 0.001) and clinical pregnancy rate (OR 3.43, 95% CI 1.92–6.11, P < 0.001) were detected [92]. Another literature review by Ross et al. [93] analysed 17 randomised trials, including a total of 1665 infertile men in whom oral antioxidant(s) were compared to placebo or no treatment. Despite the methodological and clinical heterogeneity, improvement in semen parameters after antioxidant therapy was reported in 14 of the 17 trials. Pregnancy rate was measured in seven trials, six of which showed a significant improvement after antioxidant therapy. The authors concluded that the use of oral antioxidants in infertile men may have a beneficial effect on sperm quality and pregnancy rates.
Conclusion
Extensive research has been conducted to study the effect of antioxidant therapy in improving male fertility. A beneficial influence was generally observed for antioxidants in reversing OS-induced sperm dysfunction and in improving pregnancy rates. The most commonly used compounds either as monotherapy or in combination were as follows: vitamin E (400 mg), vitamin C (500–1000 mg), carnitines (500–1000 mg), NAC (600 mg), CoQ10 (100–300 mg), zinc (25–400 mg), selenium (200 µg), folic acid (0.5 mg) and lycopene (6–8 mg). However, the recognition of an ideal antioxidant treatment method is still hampered by the heterogeneous nature of the study designs, which include different antioxidants regimens at different concentrations. Furthermore, the normal physiological level of the fine redox balance is still unknown, making proper diagnosis and treatment difficult. Additional studies are needed to determine the optimal antioxidant preparation that can be used safely for the management of male infertility.
Conflict of interest
None.
Source of funding
None.
Management
Footnotes
Peer review under responsibility of Arab Association of Urology.
References
- 1.Sharlip I.D., Jarow J.P., Belker A.M., Lipshultz L.I., Sigman M., Thomas A.J. Best practice policies for male infertility. Fertil Steril. 2002;77:873–882. doi: 10.1016/s0015-0282(02)03105-9. [DOI] [PubMed] [Google Scholar]
- 2.Agarwal A., Mulgund A., Hamada A., Chyatte M.R. A unique view on male infertility around the globe. Reprod Biol Endocrinol. 2015;13:37. doi: 10.1186/s12958-015-0032-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Halliwell B. Free radicals and vascular disease: how much do we know? BMJ. 1993;307:885–886. doi: 10.1136/bmj.307.6909.885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ford C.E., Jones K.W., Miller O.J., Mittwoch U., Penrose L.S., Ridler M. The chromosomes in a patient showing both mongolism and the Klinefelter syndrome. Lancet. 1959;1:709–710. doi: 10.1016/s0140-6736(59)91891-4. [DOI] [PubMed] [Google Scholar]
- 5.Brooker R.J. 4th ed. McGraw-Hill Higher Education; Ohio, USA: 2011. Genetics: analysis and principles. [Google Scholar]
- 6.Aitken R.J., Clarkson J.S., Fishel S. Generation of reactive oxygen species, lipid peroxidation, and human sperm function. Biol Reprod. 1989;41:183–197. doi: 10.1095/biolreprod41.1.183. [DOI] [PubMed] [Google Scholar]
- 7.Sies H. Strategies of antioxidant defense. Eur J Biochem. 1993;215:213–219. doi: 10.1111/j.1432-1033.1993.tb18025.x. [DOI] [PubMed] [Google Scholar]
- 8.Agarwal A., Majzoub A., Esteves S.C., Ko E., Ramasamy R., Zini A. Clinical utility of sperm DNA fragmentation testing: practice recommendations based on clinical scenarios. Transl Androl Urol. 2016;5:935–950. doi: 10.21037/tau.2016.10.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Agarwal A., Sharma R.K., Nallella K.P., Thomas A.J., Jr, Alvarez J.G., Sikka S.C. Reactive oxygen species as an independent marker of male factor infertility. Fertil Steril. 2006;86:878–885. doi: 10.1016/j.fertnstert.2006.02.111. [DOI] [PubMed] [Google Scholar]
- 10.Tremellen K. Oxidative stress and male infertility–a clinical perspective. Hum Reprod Update. 2008;14:243–258. doi: 10.1093/humupd/dmn004. [DOI] [PubMed] [Google Scholar]
- 11.Agarwal A., Saleh R.A., Bedaiwy M.A. Role of reactive oxygen species in the pathophysiology of human reproduction. Fertil Steril. 2003;79:829–843. doi: 10.1016/s0015-0282(02)04948-8. [DOI] [PubMed] [Google Scholar]
- 12.Gharagozloo P., Gutiérrez-Adán A., Champroux A., Noblanc A., Kocer A., Calle A. A novel antioxidant formulation designed to treat male infertility associated with oxidative stress: promising preclinical evidence from animal models. Hum Reprod. 2016;31:252–2562. doi: 10.1093/humrep/dev302. [DOI] [PubMed] [Google Scholar]
- 13.ElSheikh M.G., Hosny M.B., Elshenoufy A., Elghamrawi H., Fayad A., Abdelrahman S. Combination of vitamin E and clomiphene citrate in treating patients with idiopathic oligoasthenozoospermia: a prospective, randomized trial. Andrology. 2015;3:864–867. doi: 10.1111/andr.12086. [DOI] [PubMed] [Google Scholar]
- 14.Paradiso Galatioto G., Gravina G.L., Angelozzi G., Sacchetti A., Innominato P.F., Pace G. May antioxidant therapy improve sperm parameters of men with persistent oligospermia after retrograde embolization for varicocele? World J Urol. 2008;26:97–102. doi: 10.1007/s00345-007-0218-z. [DOI] [PubMed] [Google Scholar]
- 15.Peivandi S., Karimpour A., Moslemizadeh N. Effects of l-carnitine on infertile men’s spermogram; a randomized clinical trial. J Reprod Infertil. 2010;10:245–251. [Google Scholar]
- 16.Safarinejad M.R. Efficacy of coenzyme Q10 on semen parameters, sperm function and reproductive hormones in infertile men. J Urol. 2009;182:237–248. doi: 10.1016/j.juro.2009.02.121. [DOI] [PubMed] [Google Scholar]
- 17.Safarinejad M.R., Safarinejad S. Efficacy of selenium and/or N-acetyl-cysteine for improving semen parameters in infertile men: a double-blind, placebo controlled, randomized study. J Urol. 2009;181:741–751. doi: 10.1016/j.juro.2008.10.015. [DOI] [PubMed] [Google Scholar]
- 18.Wong W.Y., Merkus H.M., Thomas C.M., Menkveld R., Zielhuis G.A., Steegers-Theunissen R.P. Effects of folic acid and zinc sulfate on male factor subfertility: a double-blind, randomized, placebo-controlled trial. Fertil Steril. 2002;77:491–498. doi: 10.1016/s0015-0282(01)03229-0. [DOI] [PubMed] [Google Scholar]
- 19.Omu A.E., Al-Azemi M.K., Kehinde E.O., Anim J.T., Oriowo M.A., Mathew T.C. Indications of the mechanisms involved in improved sperm parameters by zinc therapy. Med Princ Pract. 2008;17:108–116. doi: 10.1159/000112963. [DOI] [PubMed] [Google Scholar]
- 20.Lenzi A., Sgrò P., Salacone P., Paoli D., Gilio B., Lombardo F. A placebo-controlled double-blind randomized trial of the use of combined l-carnitine and l-acetyl-carnitine treatment in men with asthenozoospermia. Fertil Steril. 2004;81:1578–1584. doi: 10.1016/j.fertnstert.2003.10.034. [DOI] [PubMed] [Google Scholar]
- 21.Ciftci H., Verit A., Savas M., Yeni E., Erel O. Effects of N-acetylcysteine on semen parameters and oxidative/antioxidant status. Urology. 2009;74:73–76. doi: 10.1016/j.urology.2009.02.034. [DOI] [PubMed] [Google Scholar]
- 22.Greco E., Iacobelli M., Rienzi L., Ubaldi F., Ferrero S., Tesarik J. Reduction of the incidence of sperm DNA fragmentation by oral antioxidant treatment. J Androl. 2005;26:349–353. doi: 10.2164/jandrol.04146. [DOI] [PubMed] [Google Scholar]
- 23.Suleiman S.A., Ali M.E., Zaki Z.M., el-Malik E.M., Nasr M.A. Lipid peroxidation and human sperm motility: protective role of vitamin E. J Androl. 1996;17:530–537. [PubMed] [Google Scholar]
- 24.Keskes-Ammar L., Feki-Chakroun N., Rebai T., Sahnoun Z., Ghozzi H., Hammami S. Sperm oxidative stress and the effect of an oral vitamin E and selenium supplement on semen quality in infertile men. Arch Androl. 2003;49:83–94. doi: 10.1080/01485010390129269. [DOI] [PubMed] [Google Scholar]
- 25.Tremellen K., Miari G., Froiland D., Thompson J. A randomised control trial examining the effect of an antioxidant (Menevit) on pregnancy outcome during IVF-ICSI treatment. Aust N Z J Obstet Gynaecol. 2007;47:216–221. doi: 10.1111/j.1479-828X.2007.00723.x. [DOI] [PubMed] [Google Scholar]
- 26.Omu A.E., Dashti H., Al-Othman S. Treatment of asthenozoospermia with zinc sulphate: andrological, immunological and obstetric outcome. Eur J Obstet Gynecol Reprod Biol. 1998;79:179–184. doi: 10.1016/s0301-2115(97)00262-5. [DOI] [PubMed] [Google Scholar]
- 27.Cavallini G., Ferraretti A.P., Gianaroli L., Biagiotti G., Vitali G. Cinnoxicam and l-carnitine/acetyl-l-carnitine treatment for idiopathic and varicocele-associated oligoasthenospermia. J Androl. 2004;25:761–772. doi: 10.1002/j.1939-4640.2004.tb02853.x. [DOI] [PubMed] [Google Scholar]
- 28.Kessopoulou E., Powers H.J., Sharma K.K., Pearson M.J., Russell J.M., Cooke I.D. A double-blind randomized placebo cross-over controlled trial using the antioxidant vitamin E to treat reactive oxygen species associated male infertility. Fertil Steril. 1995;64:825–831. doi: 10.1016/s0015-0282(16)57861-3. [DOI] [PubMed] [Google Scholar]
- 29.Hawkes W.C., Alkan Z., Wong K. Selenium supplementation does not affect testicular selenium status or semen quality in North American men. J Androl. 2009;30:525–533. doi: 10.2164/jandrol.108.006940. [DOI] [PubMed] [Google Scholar]
- 30.Rolf C., Cooper T.G., Yeung C.H., Nieschlag E. Antioxidant treatment of patients with asthenozoospermia or moderate oligoasthenozoospermia with high-dose vitamin C and vitamin E: a randomized, placebo-controlled, double-blind study. Hum Reprod. 1999;14:1028–1033. doi: 10.1093/humrep/14.4.1028. [DOI] [PubMed] [Google Scholar]
- 31.Sigman M., Glass S., Campagnone J., Pryor J.L. Carnitine for the treatment of idiopathic asthenospermia: a randomized, double-blind, placebo-controlled trial. Fertil Steril. 2006;85:1409–1414. doi: 10.1016/j.fertnstert.2005.10.055. [DOI] [PubMed] [Google Scholar]
- 32.Comhaire F.H., Christophe A.B., Zalata A.A., Dhooge W.S., Mahmoud A.M., Depuydt C.E. The effects of combined conventional treatment, oral antioxidants and essential fatty acids on sperm biology in subfertile men. Prostaglandins Leukot Essent Fatty Acids. 2000;63:159–165. doi: 10.1054/plef.2000.0174. [DOI] [PubMed] [Google Scholar]
- 33.Gupta N.P., Kumar R. Lycopene therapy in idiopathic male infertility – a preliminary report. Int Urol Nephrol. 2002;34:369–372. doi: 10.1023/a:1024483520560. [DOI] [PubMed] [Google Scholar]
- 34.Moslemi M.K., Tavanbakhsh S. Selenium-vitamin E supplementation in infertile men: effects on semen parameters and pregnancy rate. Int J Gen Med. 2011;4:99–104. doi: 10.2147/IJGM.S16275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mohanty N.K., Kumar S., Jha A.K., Arora R.P. Management of idiopathic oligoasthenospermia with lycopene. Indian J Urol. 2001;18:57–61. [Google Scholar]
- 36.Ménézo Y.J., Hazout A., Panteix G., Robert F., Rollet J., Cohen-Bacrie P. Antioxidants to reduce sperm DNA fragmentation: an unexpected adverse effect. Reprod Biomed Online. 2007;14:418–421. doi: 10.1016/s1472-6483(10)60887-5. [DOI] [PubMed] [Google Scholar]
- 37.Abad C., Amengual M.J., Gosálvez J., Coward K., Hannaoui N., Benet J. Effects of oral antioxidant treatment upon the dynamics of human sperm DNA fragmentation and subpopulations of sperm with highly degraded DNA. Andrologia. 2013;45:211–216. doi: 10.1111/and.12003. [DOI] [PubMed] [Google Scholar]
- 38.Gual-Frau J., Abad C., Amengual M.J., Hannaoui N., Checa M.A., Ribas-Maynou J. Oral antioxidant treatment partly improves integrity of human sperm DNA in infertile grade I varicocele patients. Hum Fertil (Camb) 2015;18:225–229. doi: 10.3109/14647273.2015.1050462. [DOI] [PubMed] [Google Scholar]
- 39.Geva E., Bartoov B., Zabludovsky N., Lessing J.B., Lerner-Geva L., Amit A. The effect of antioxidant treatment on human spermatozoa and fertilization rate in an in vitro fertilization program. Fertil Steril. 1996;66:430–434. doi: 10.1016/s0015-0282(16)58514-8. [DOI] [PubMed] [Google Scholar]
- 40.Safarinejad M.R. The effect of coenzyme Q(1)(0) supplementation on partner pregnancy rate in infertile men with idiopathic oligoasthenoteratozoospermia: an open-label prospective study. Int Urol Nephrol. 2012;44:689–700. doi: 10.1007/s11255-011-0081-0. [DOI] [PubMed] [Google Scholar]
- 41.Greco E., Romano S., Iacobelli M., Ferrero S., Baroni E., Minasi M.G. ICSI in cases of sperm DNA damage: beneficial effect of oral antioxidant treatment. Hum Reprod. 2005;20:2590–2594. doi: 10.1093/humrep/dei091. [DOI] [PubMed] [Google Scholar]
- 42.Moilanen J., Hovatta O. Excretion of alpha-tocopherol into human seminal plasma after oral administration. Andrologia. 1995;27:133–136. doi: 10.1111/j.1439-0272.1995.tb01083.x. [DOI] [PubMed] [Google Scholar]
- 43.Singh F., Charles A.L., Schlagowski A.I., Bouitbir J., Bonifacio A., Piquard F. Reductive stress impairs myoblasts mitochondrial function and triggers mitochondrial hormesis. BBA. 2015;1853:1574–1585. doi: 10.1016/j.bbamcr.2015.03.006. [DOI] [PubMed] [Google Scholar]
- 44.Mentor S., Fisher D. Aggressive antioxidant reductive stress impairs brain endothelial cell angiogenesis and blood brain barrier function. Curr Neurovasc Res. 2017;14:71–81. doi: 10.2174/1567202613666161129113950. [DOI] [PubMed] [Google Scholar]
- 45.Lamosová D., Juráni M., Greksák M., Nakano M., Vaneková M. Effect of Rooibos tea (Aspalathus linearis) on chick skeletal muscle cell growth in culture. Comp Biochem Physiol C: Pharmacol Toxicol Endocrinol. 1997;116:39–45. doi: 10.1016/s0742-8413(96)00138-7. [DOI] [PubMed] [Google Scholar]
- 46.Thérond P., Auger J., Legrand A., Jouannet P. Alpha-tocopherol in human spermatozoa and seminal plasma: relationships with motility, antioxidant enzymes and leukocytes. Mol Hum Reprod. 1996;2:739–744. doi: 10.1093/molehr/2.10.739. [DOI] [PubMed] [Google Scholar]
- 47.Omu A.E., Fatinikun T., Mannazhath N., Abraham S. Significance of simultaneous determination of serum and seminal plasma alpha-tocopherol and retinol in infertile men by high-performance liquid chromatography. Andrologia. 1999;31:347–354. doi: 10.1046/j.1439-0272.1999.00296.x. [DOI] [PubMed] [Google Scholar]
- 48.Jacob R.A., Pianalto F.S., Agee R.E. Cellular ascorbate depletion in healthy men. J Nutr. 1992;122:1111–1118. doi: 10.1093/jn/122.5.1111. [DOI] [PubMed] [Google Scholar]
- 49.Fraga C.G., Motchnik P.A., Shigenaga M.K., Helbock H.J., Jacob R.A., Ames B.N. Ascorbic acid protects against endogenous oxidative DNA damage in human sperm. PNAS. 1991;88:11003–11006. doi: 10.1073/pnas.88.24.11003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lewis S.E., John Aitken R., Conner S.J., Iuliis G.D., Evenson D.P., Henkel R. The impact of sperm DNA damage in assisted conception and beyond: recent advances in diagnosis and treatment. Reprod Biomed Online. 2013;27:325–337. doi: 10.1016/j.rbmo.2013.06.014. [DOI] [PubMed] [Google Scholar]
- 51.Shi J.Z., Zhang S.S., Zhang Z., Liang Q., Shi Y., Hua J.L. Expressions of sperm-specific genes in carnitine-cultured testis sperm of obstructive azoospermia patients. Zhonghua Nan Ke Xue. 2010;16:504–509. [article in Chinese] [PubMed] [Google Scholar]
- 52.Banihani S., Agarwal A., Sharma R., Bayachou M. Cryoprotective effect of l-carnitine on motility, vitality and DNA oxidation of human spermatozoa. Andrologia. 2014;46:637–641. doi: 10.1111/and.12130. [DOI] [PubMed] [Google Scholar]
- 53.Gülçin I. Antioxidant and antiradical activities of L-carnitine. Life Sci. 2006;78:803–811. doi: 10.1016/j.lfs.2005.05.103. [DOI] [PubMed] [Google Scholar]
- 54.Ahmed S.D., Karira K.A., Jagdesh, Ahsan S. Role of l-carnitine in male infertility. J Pak Med Assoc. 2011;61:732–736. [PubMed] [Google Scholar]
- 55.Lewin A., Lavon H. The effect of coenzyme Q10 on sperm motility and function. Mol Aspects Med. 1997;18(Suppl.):S213–S219. doi: 10.1016/s0098-2997(97)00036-8. [DOI] [PubMed] [Google Scholar]
- 56.Mancini A., Conte B., De Marinis L., Hallgass M.E., Pozza D., Oradei A. Coenzyme Q10 levels in human seminal fluid: diagnostic and clinical implications. Mol Aspects Med. 1994;15(Suppl.):S249–S255. doi: 10.1016/0098-2997(94)90035-3. [DOI] [PubMed] [Google Scholar]
- 57.Gressier B., Cabanis A., Lebegue S., Brunet C., Dine T., Luyckx M. Decrease of hypochlorous acid and hydroxyl radical generated by stimulated human neutrophils: comparison in vitro of some thiol-containing drugs. Methods Find Exp Clin Pharmacol. 1994;16:9–13. [PubMed] [Google Scholar]
- 58.Erkkilä K., Hirvonen V., Wuokko E., Parvinen M., Dunkel L. N-acetyl-l-cysteine inhibits apoptosis in human male germ cells in vitro. J Clin Endocrinol Metab. 1998;83:2523–2531. doi: 10.1210/jcem.83.7.4949. [DOI] [PubMed] [Google Scholar]
- 59.Oeda T., Henkel R., Ohmori H., Schill W.B. Scavenging effect of N-acetyl-l-cysteine against reactive oxygen species in human semen: a possible therapeutic modality for male factor infertility? Andrologia. 1997;29:125–131. doi: 10.1111/j.1439-0272.1997.tb00305.x. [DOI] [PubMed] [Google Scholar]
- 60.Roveri A., Casasco A., Maiorino M., Dalan P., Calligaro A., Ursini F. Phospholipid hydroperoxide glutathione peroxidase of rat testis. Gonadotropin dependence and immunocytochemical identification. J Biol Chem. 1992;267:6142–6146. [PubMed] [Google Scholar]
- 61.Alvarez J.G., Storey B.T. Lipid peroxidation and the reactions of superoxide and hydrogen peroxide in mouse spermatozoa. Biol Reprod. 1984;30:833–841. doi: 10.1095/biolreprod30.4.833. [DOI] [PubMed] [Google Scholar]
- 62.Ursini F., Heim S., Kiess M., Maiorino M., Roveri A., Wissing J. Dual function of the selenoprotein PHGPx during sperm maturation. Science. 1999;285:1393–1396. doi: 10.1126/science.285.5432.1393. [DOI] [PubMed] [Google Scholar]
- 63.Noack-Fuller G., De Beer C., Seibert H. Cadmium, lead, selenium, and zinc in semen of occupationally unexposed men. Andrologia. 1993;25:7–12. doi: 10.1111/j.1439-0272.1993.tb02674.x. [DOI] [PubMed] [Google Scholar]
- 64.Hambidge K.M., Krebs N.F. Zinc deficiency: a special challenge. J Nutr. 2007;137:1101–1105. doi: 10.1093/jn/137.4.1101. [DOI] [PubMed] [Google Scholar]
- 65.Powell S.R. The antioxidant properties of zinc. J Nutr. 2000;130(Suppl.):1447S–1454S. doi: 10.1093/jn/130.5.1447S. [DOI] [PubMed] [Google Scholar]
- 66.Chia S.E., Ong C.N., Chua L.H., Ho L.M., Tay S.K. Comparison of zinc concentrations in blood and seminal plasma and the various sperm parameters between fertile and infertile men. J Androl. 2000;21:53–57. [PubMed] [Google Scholar]
- 67.Joshi R., Adhikari S., Patro B.S., Chattopadhyay S., Mukherjee T. Free radical scavenging behavior of folic acid: evidence for possible antioxidant activity. Free Radic Biol Med. 2001;30:1390–1399. doi: 10.1016/s0891-5849(01)00543-3. [DOI] [PubMed] [Google Scholar]
- 68.Kelkel M., Schumacher M., Dicato M., Diederich M. Antioxidant and anti-proliferative properties of lycopene. Free Radic Res. 2011;45:925–940. doi: 10.3109/10715762.2011.564168. [DOI] [PubMed] [Google Scholar]
- 69.Agarwal A., Sekhon L.H. Oxidative stress and antioxidants for idiopathic oligoasthenoteratospermia: Is it justified? Indian J Urol. 2011;27:74–85. doi: 10.4103/0970-1591.78437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Cooper T.G., Noonan E., von Eckardstein S., Auger J., Baker H.W., Behre H.M. World Health Organization reference values for human semen characteristics. Hum Reprod Update. 2010;16:231–245. doi: 10.1093/humupd/dmp048. [DOI] [PubMed] [Google Scholar]
- 71.Dawson E.B., Harris W.A., Teter M.C., Powell L.C. Effect of ascorbic acid supplementation on the sperm quality of smokers. Fertil Steril. 1992;58:1034–1039. [PubMed] [Google Scholar]
- 72.Cyrus A., Kabir A., Goodarzi D., Moghimi M. The effect of adjuvant vitamin C after varicocele surgery on sperm quality and quantity in infertile men: a double blind placebo controlled clinical trial. Int Braz J Urol. 2015;41:230–238. doi: 10.1590/S1677-5538.IBJU.2015.02.07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Balercia G., Regoli F., Armeni T., Koverech A., Mantero F., Boscaro M. Placebo-controlled double-blind randomized trial on the use of l-carnitine, l-acetylcarnitine, or combined l-carnitine and l-acetylcarnitine in men with idiopathic asthenozoospermia. Fertil Steril. 2005;84:662–671. doi: 10.1016/j.fertnstert.2005.03.064. [DOI] [PubMed] [Google Scholar]
- 74.Nadjarzadeh A., Shidfar F., Amirjannati N., Vafa M.R., Motevalian S.A., Gohari M.R. Effect of Coenzyme Q10 supplementation on antioxidant enzymes activity and oxidative stress of seminal plasma: a double-blind randomised clinical trial. Andrologia. 2014;46:177–183. doi: 10.1111/and.12062. [DOI] [PubMed] [Google Scholar]
- 75.Lafuente R., González-Comadrán M., Solà I., López G., Brassesco M., Carreras R. Coenzyme Q10 and male infertility: a meta-analysis. J Assist Reprod Genet. 2013;30:1147–1156. doi: 10.1007/s10815-013-0047-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Vézina D., Mauffette F., Roberts K.D., Bleau G. Selenium-vitamin E supplementation in infertile men. Effects on semen parameters and micronutrient levels and distribution. Biol Trace Elem Res. 1996;53:65–83. doi: 10.1007/BF02784546. [DOI] [PubMed] [Google Scholar]
- 77.Hadwa M.H., Almashhedy L.A., Alsalman A.R. Oral zinc supplementation restores superoxide radical scavengers to normal levels in spermatozoa of Iraqi asthenospermic patients. Int J Vitam Nutr Res. 2015;85:165–173. doi: 10.1024/0300-9831/a000235. [DOI] [PubMed] [Google Scholar]
- 78.Jequier A.M. Semen analysis: a new manual and its application to the understanding of semen and its pathology. Asian J Androl. 2010;12:11–13. doi: 10.1038/aja.2009.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Esteves S.C. Clinical relevance of routine semen analysis and controversies surrounding the 2010 World Health Organization criteria for semen examination. Int Braz J Urol. 2014;40:443–453. doi: 10.1590/S1677-5538.IBJU.2014.04.02. [DOI] [PubMed] [Google Scholar]
- 80.Erenpreiss J., Spano M., Erenpreisa J., Bungum M., Giwercman A. Sperm chromatin structure and male fertility: biological and clinical aspects. Asian J Androl. 2006;8:11–29. doi: 10.1111/j.1745-7262.2006.00112.x. [DOI] [PubMed] [Google Scholar]
- 81.Shamsi M.B., Kumar R., Dada R. Evaluation of nuclear DNA damage in human spermatozoa in men opting for assisted reproduction. Indian J Med Res. 2008;127:115–123. [PubMed] [Google Scholar]
- 82.Sharma R.K., Said T., Agarwal A. Sperm DNA damage and its clinical relevance in assessing reproductive outcome. Asian J Androl. 2004;6:139–148. [PubMed] [Google Scholar]
- 83.Saleh R.A., Agarwal A., Sharma R.K., Said T.M., Sikka S.C., Thomas A.J., Jr. Evaluation of nuclear DNA damage in spermatozoa from infertile men with varicocele. Fertil Steril. 2003;80:1431–1436. doi: 10.1016/s0015-0282(03)02211-8. [DOI] [PubMed] [Google Scholar]
- 84.Saleh R.A., Agarwal A., Nada E.A., El-Tonsy M.H., Sharma R.K., Meyer A. Negative effects of increased sperm DNA damage in relation to seminal oxidative stress in men with idiopathic and male factor infertility. Fertil Steril. 2003;79(Suppl. 3):1597–1605. doi: 10.1016/s0015-0282(03)00337-6. [DOI] [PubMed] [Google Scholar]
- 85.Agarwal A., Cho C.L., Esteves S.C. Should we evaluate and treat sperm DNA fragmentation? Curr Opin Obstet Gynecol. 2016;28:164–171. doi: 10.1097/GCO.0000000000000271. [DOI] [PubMed] [Google Scholar]
- 86.Gil-Villa A.M., Cardona-Maya W., Agarwal A., Sharma R., Cadavid A. Role of male factor in early recurrent embryo loss: do antioxidants have any effect? Fertil Steril. 2009;92:565–571. doi: 10.1016/j.fertnstert.2008.07.1715. [DOI] [PubMed] [Google Scholar]
- 87.Agarwal A., Makker K., Sharma R. Clinical relevance of oxidative stress in male factor infertility: an update. Am J Reprod Immunol. 2008;59:2–11. doi: 10.1111/j.1600-0897.2007.00559.x. [DOI] [PubMed] [Google Scholar]
- 88.Agarwal A., Tvrda E., Sharma R. Relationship amongst teratozoospermia, seminal oxidative stress and male infertility. Reprod Biol Endocrinol. 2014;12:45. doi: 10.1186/1477-7827-12-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cocuzza M., Sikka S.C., Athayde K.S., Agarwal A. Clinical relevance of oxidative stress and sperm chromatin damage in male infertility: an evidence based analysis. Int Braz J Urol. 2007;33:603–621. doi: 10.1590/s1677-55382007000500002. [DOI] [PubMed] [Google Scholar]
- 90.Gharagozloo P., Aitken R.J. The role of sperm oxidative stress in male infertility and the significance of oral antioxidant therapy. Hum Reprod. 2011;26:1628–1640. doi: 10.1093/humrep/der132. [DOI] [PubMed] [Google Scholar]
- 91.Hammadeh M.E., Al Hasani S., Rosenbaum P., Schmidt W., Fischer Hammadeh C. Reactive oxygen species, total antioxidant concentration of seminal plasma and their effect on sperm parameters and outcome of IVF/ICSI patients. Arch Gynecol Obstet. 2008;277:515–526. doi: 10.1007/s00404-007-0507-1. [DOI] [PubMed] [Google Scholar]
- 92.Showell M.G., Mackenzie-Proctor R., Brown J., Yazdani A., Stankiewicz M.T., Hart R.J. Antioxidants for male subfertility. Cochrane Database Syst Rev. 2014;12:CD007411. doi: 10.1002/14651858.CD007411.pub3. [DOI] [PubMed] [Google Scholar]
- 93.Ross C., Morriss A., Khairy M., Khalaf Y., Braude P., Coomarasamy A. A systematic review of the effect of oral antioxidants on male infertility. Reprod Biomed Online. 2010;20:711–723. doi: 10.1016/j.rbmo.2010.03.008. [DOI] [PubMed] [Google Scholar]