Abstract
Objectives
To assess neurodevelopmental (ND) outcomes among biliary atresia (BA) participants with their native liver at ages 12 months (Age 1) and 24 months (Age 2) years, and to evaluate variables predictive of ND impairment.
Study design
Participants enrolled in a prospective longitudinal multi-center study underwent ND testing with either the Bayley Scales of Infant Development, 2nd edition (BSID-II) or Bayley Scales of Infant and Toddler Development, 3rd edition (Bayley-III). Scores (normative mean=100±15) were categorized as ≥100, 85-99, and <85 for chi-square analysis. Risk for ND impairment (defined as at least one score <85 on BSID-II or Bayley-III scales) was analyzed using logistic regression.
Results
148 children completed 217 Bayley examinations (Age 1 n=132; Age 2 n=85). ND score distributions significantly shifted downward compared with test norms at Ages 1 and 2. Multivariate analysis identified ascites (OR=3.17, P = .01) and low length z-scores at time of testing (OR=0.70, p<0.04) as risk factors for Physical/Motor Impairment; low weight z-score (OR=0.57, p=0.001) and ascites (OR=2.89, p=0.01) for Mental/Cognitive/Language Impairment at Age 1. An unsuccessful hepatoportoenterostomy was predictive of both Physical/Motor (OR=4.88, p<0.02) and Mental/Cognitive/Language Impairment (OR=4.76, p=0.02) at Age 2.
Conclusion
Participants with biliary atresia surviving with native livers after HPE are at increased risk for ND delays at 12 and 24 months of age. Those with unsuccessful HPE are over 4 times more likely to have ND impairment compared with those with successful HPE. Growth delays and/or complications indicating advanced liver disease should alert clinicians to risk for ND delays, and expedite appropriate interventions.
Trial Registration
Keywords: chronic liver disease, Kasai, cognitive, motor
Biliary atresia is the most common cause of chronic progressive liver disease in children, manifesting as cholestatic jaundice shortly after birth. It results from an inflammatory and fibrosing obstruction of extrahepatic bile ducts and abnormalities of the intrahepatic bile ducts 1. The primary treatment is surgical hepatoportoenterostomy (HPE) performed as early as possible in an attempt to re-establish biliary flow 2. Despite surgical treatment, most patients require liver transplant in childhood 3, but survival with native liver into school age 4 and adulthood 5-8 is achievable, and understanding the challenges these patients may encounter at the earliest phase possible is key. A recent prospective study of infants with biliary atresia demonstrated that failure to achieve a serum total bilirubin level of <2.0 mg/dL in the first 3 months after HPE is associated with increased risk of various medical complications including ascites, coagulopathy, and failure to thrive 9. However, neurodevelopmental outcomes were not evaluated. Prior single-center reports describing ND functioning in BA infants before LT 5-8 were limited by small samples. ND data from a large, contemporaneous, multi-center cohort of young BA patients with native liver followed longitudinally can better inform intervention strategies aimed at improving overall outcomes for this population.
The Biliary Atresia Research Consortium (BARC) was established in 2002 by the National Institutes of Health to improve understanding of the etiology of BA, its natural history, and clinical outcomes, and to develop interventional strategies to improve outcomes. BARC was expanded into the Childhood Liver Disease Research Network (ChiLDReN) in 2009. In this prospective multi-center study, ND outcomes at 1- and 2- years of age were assessed in a cohort of BA children alive with native liver followed longitudinally in ChiLDReN. We hypothesized that BA patients with their native livers at ages 1 and 2 years would exhibit significant ND delays and that specific demographic and clinical variables would be predictive of worse ND outcomes.
Methods
Study participants were infants with BA enrolled in the ChiLDReN's Prospective Study of Infants and Children with Cholestasis (PROBE) Protocol (Clinicaltrials.gov: NCT00061828) between July 1, 2004, and November 1, 2012, who underwent HPE at a ChiLDReN site. Informed consent was obtained from parents or guardians and the protocol was carried out under IRB approval. A subset of these infants with biliary atresia with birth weight >2 kg was co-enrolled in the Steroids in Biliary Atresia Randomized Trial (START), a prospective, randomized double-blinded, placebo-controlled trial of corticosteroids as adjunctive therapy following HPE for BA (Clinicaltrials.gov: NCT00294684)10. Exclusion criteria for this present study included birth weight ≤2000 grams, acute liver failure, previous hepatobiliary surgery (other than HPE), sepsis, hypoxia, shock, malignancy, primary hemolytic disease, parenteral nutrition-associated cholestasis, extracorporeal membrane oxygenation (ECMO)-associated cholestasis, or LT.
Data Collection
Data were prospectively collected and entered into a centralized database. Baseline data included patient demographics, medical history (age at HPE, the presence of BA splenic malformation syndrome (BASM: asplenia or polysplenia), family history, and laboratory studies9. START randomization group (START steroid or START placebo) or PROBE only assignment were also recorded. Research visits were scheduled 1, 2, 3, and 6 months after HPE, and then at 12, 18, and 24 months of age. Longitudinal data included anthropometry measures, liver biochemical values, and interval history of ascites, cholangitis, spontaneous bacterial peritonitis, gastrointestinal bleeding, and nasogastric feeding within the preceding year. Response to HPE was dichotomized into two groups based upon serum total bilirubin levels (TB) in the first 3 months after HPE 9. “Unsuccessful HPE” was defined for those patients never achieving a TB < 2.0 mg/dL (34.2 μmol/L) in the first 3 months post-HPE. “Successful HPE” was defined for those patients having any TB < 2.0 mg/dL (34.2 uM) within the first 3 months post-HPE. ND testing was performed at 1 year and 2 years of age (±2 months).
Neurodevelopmental Assessment Measures
The Bayley Scales of Infant Development, second edition (BSID-II) was used to assess ND outcomes through January 2010 11. Starting in February 2010, all participants who were new to the study completed the updated Bayley Scales of Infant and Toddler Development, third edition (Bayley-III) 12. However, participants previously tested with the BSID-II at Age 1, were re-tested with the BSID-II version at Age 2 to be consistent. Both versions are normed for use between patient ages 1 – 42 months of age, and involve a standard series of developmental play tasks which are organized into scales yielding standardized scores (mean = 100 ±15). The Bayley-III improved upon the BSID-II with significant changes in test structure, updated norms, and improved psychometrics. Participants assessed with the BSID-II completed the “Mental” and “Physical” scales; those assessed with the Bayley-III completed “Cognitive”, “Language”, and “Motor” scales.
Statistical Analyses
We used descriptive statistics to report continuous and categorical ND outcomes, and all demographic and other clinical characteristics. One-sample t-test was used to determine whether the mean Bayley scores in our sample were lower than the test norms (100 ± 15). Domain scores for each Bayley version (BSID-II – Physical/Mental; or Bayley-III – Motor/Cognitive/Language) were categorized as ≥100 (z-score≥0), 85-99 (z-score between 0 and -1), and <85 (z-score < -1). Chi-square test was used to compare the distribution of Bayley scores in the study cohort in these 3 bins to the normal population. P values ≤ 0.05 were considered statistically significant.
Because of the significant changes to the construction and content of the scales, BSID-II and Bayley-III scores are only modestly correlated and are not equivalent. We combined results from the two Bayley versions to derive ND outcomes reflecting the entire sample for modeling. The two primary ND outcomes were “At Risk for Physical/Motor Delay”, defined as either a BSID-II Physical score <85 or Bayley-III Motor score <85, and “At Risk for Mental/Cognitive/Language Delay”, defined as BSID-II Mental score <85 or Bayley-III Cognitive and/or Language score <85. This approach resulted in characterization of below average scores on either Bayley version as “at risk” (capturing those performing relatively worse regardless of measure).
Bowker test of symmetry was used to assess agreement between the three categorical responses for each domain between Age 1 and Age 2. Associations between patient characteristics and the two ND outcomes were determined using logistic regression models. In univariate analyses, risk factors were included one at a time. In multivariate analysis, we first included all risk factors of interest and a backward stepwise selection procedure was used. A p-value <0.1 was used as both inclusion and exclusion criteria in the stepwise selection procedure. Acknowledging the widely reported systematic differences between BSID-II and Bayley-III scores, Bayley version was adjusted in both univariate and multivariate models 13. Due to missing data, multiple imputations were used for multivariate analysis using IVEware 14. All other analyses were performed using SAS.
Results
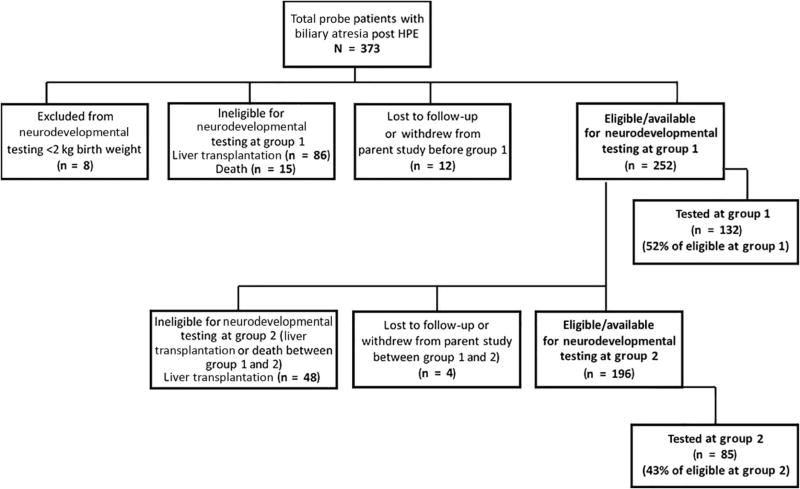
A total of 373 PROBE participants with BA were identified. Participants who underwent LT (n=86), died (n=15), were withdrawn prior to 1 year of age (n=12), or were <2 kg at birth (n=8) did not undergo ND assessment (Figure 1; available at www.jpeds.com); 132 (52%) of remaining 252 eligible participants completed ND testing at Age 1, and 85 (43%) of 196 eligible had ND testing at Age 2. Eligible participants with ND testing at Age 1 (N=132) were more likely than those who were eligible but not tested (N=120) to be white, non-Hispanic, have college-educated parents, and have a successful HPE (p<0.04). Similarly, Age 2 tested participants (n=85) were more likely than those who were eligible but not tested (n=111) to be white and have college-educated parents (p<0.04) [data not shown]. Figure 1 provides details of sample ascertainment. A total of 217 completed Bayley assessments from 148 participants were available for the final analysis.
Figure 1.
(online): Consort diagram describing ascertainment of sample with ND testing at Age 1 and Age 2.
Table I provides demographic, clinical and medical characteristics for Age 1 and Age 2 participants. Participants at Age 1 had more medical events in the preceding year than Age 2 participants, including ascites (29% vs 14%) and cholangitis (51% vs. 18%). At Age 1, among 150 participants with successful HPE who were eligible for ND testing, 91 (61%) completed ND testing, compared with 29 (37%) of 78 with unsuccessful HPE (p=0.0006). This was seen again at Age 2, with 67 (50%) of 133 eligible participants with successful HPE completed testing, whereas only 10 (26%) of 39 eligible participants with unsuccessful HPE completed testing.
Table 1.
Participant demographic and clinical characteristics at Age 1 and Age 2.
| Age 1 (N=132) | Age 2 (N=85) | |||
|---|---|---|---|---|
| Demographics and medical history at baseline | ||||
| Gender – no. (%) | 132 | 85 | ||
| Male | 65(49.2%) | 44 (51.8%) | ||
| Female | 67 (50.8%) | 41 (48.2%) | ||
| Race– no. (%) | 126 | 84 | ||
| White | 81 (64.3%) | 61 (72.6%) | ||
| Black | 20 (15.9%) | 10 (11.9%) | ||
| Other | 25 (19.8%) | 13 (15.5%) | ||
| Ethnicity– no. (%) | 130 | 84 | ||
| Hispanic | 23 (17.4%) | 16 (19.0%) | ||
| Non-Hispanic | 107 (81.1%) | 68 (81.0%) | ||
| Highest household education– no. (%) | 124 | 80 | ||
| Some high school or less | 4 (3.0%) | 1 (1.3%) | ||
| High school diploma/GED | 13 (10.5%) | 11 (13.8%) | ||
| Some college or more | 107 (86.2%) | 68 (85.0%) | ||
| BASM syndrome– no. (%) | 131 | 9 (6.9%) | 85 | 5 (5.9%) |
| Study group– no. (%) | 132 | 85 | ||
| PROBE only | 64 (48.5%) | 39 (45.9%) | ||
| Placebo in START trial | 34 (25.8%) | 18 (21.1%) | ||
| Steroid in START trial | 34 (25.8%) | 28 (32.9%) | ||
| Age at HPE (month) – mean (SD) | 132 | 2.0 (0.9) | 85 | 2.0 (0.7) |
| Successful HPE*– no. (%) | 120 | 91 (76.8%) | 77 | 67 (87.0%) |
| Medical history in year prior to testing | ||||
| Ascites – no. (%) | 132 | 38 (28.8%) | 85 | 12 (14.1%) |
| Cholangitis – no. (%) | 132 | 68 (51.5%) | 85 | 15 (17.7%) |
| Spontaneous bacterial peritonitis– no. (%) | 132 | 2 (1.5%) | 85 | 4 (4.7%) |
| Gastrointestinal bleeding– no. (%) | 132 | 4 (3.0%) | 85 | 4 (4.7%) |
| Measures taken at testing (+/- 2 mos.) | ||||
| Age at testing (month)– mean (SD) | 132 | 12.4 (0.8) | 85 | 24.2 (0.8) |
| Growth parameters | ||||
| Weight Z-score– mean (SD) | 128 | -1.1 (1.3) | 79 | -0.1 (1.4) |
| Height Z-score– mean (SD) | 124 | -0.9 (1.2) | 78 | -0.4 (1.1) |
| Liver biochemical values | ||||
| Serum total bilirubin (mg/dl)– mean (SD) | 126 | 2.2 (4.9) | 76 | 1.3 (2.4) |
| INR– mean (SD) | 107 | 1.0 (0.2) | 66 | 1.0 (0.1) |
| Platelets (×109)–mean (SD) | 112 | 223.4 (116.7) | 70 | 214.1(106.2) |
| Thrombocytopenia (i.e. platelet count <150)at testing | 132 | 19 (14.4%) | 85 | 14 (16.5%) |
Unsuccessful HPE = never achieving a serum total bilirubin < 2 mg/dL within 3 months after HPE BASM=biliary atresia splenic malformation; HPE=hepatoportoenterostomy; INR=international normalized ratio.
Bayley Scores in Age 1 and Age 2 participants
Participants completing the 2 Bayley versions did not differ significantly in terms of sex, race, ethnicity, and/or highest household education. Table 2 (available at www.jpeds.com) provides mean Bayley scores by measure, scale, and age. At Age 1, mean participant scores on all BSID-II and all but one Bayley-III scale (Cognitive) were significantly lower than test norms with medium to large effect sizes (0.50 to 1.15). The Bayley-III Cognitive scale mean trended towards significance (effect size 0.21). At age 2, mean scores for those tested with BSID-II continued to show robust differences from test norms (medium effect sizes 0.65-0.72). Those tested with Bayley-III showed more modest differences from test norms on Cognitive and Motor scales (small effect sizes 0.30-0.43), and no significant difference on the Language scale.
Table 2.
(online)- Bayley scores compared with test norms.
| Age 1 participants | |||||
|---|---|---|---|---|---|
| Measure/Scale | N | Mean (SD) | t-test P value | Chi square P value | Effect Size |
| BSID-II | |||||
| Mental | 87 | 89.2 (14.5) | <0.0001 | <0.0001 | 0.72 |
| Physical | 83 | 82.7 (17.6) | <0.0001 | <0.0001 | 1.15 |
| Bayley-III | |||||
| Cognitive | 42 | 96.9 (10.8) | 0.07 | NS | 0.21 |
| Language | 42 | 92.5 (14.4) | <0.002 | 0.01 | 0.50 |
| Motor | 41 | 89.0 (13.6) | <0.0001 | <0.001 | 0.73 |
| Age 2 participants | |||||
| BSID-II | |||||
| Mental | 53 | 89.1 (15.7) | <0.0001 | <0.0001 | 0.72 |
| Physical | 52 | 90.3 (16.0) | <0.0001 | <0.0001 | 0.65 |
| Bayley-III | |||||
| Cognitive | 30 | 95.5 (9.6) | 0.02 | NS | 0.30 |
| Language | 27 | 96.5(11.6) | NS | NS | 0.23 |
| Motor | 29 | 93.6 (10.3) | 0.002 | NS | 0.43 |
One sample t-test (h0: μ=100). Test norms are Mean = 100 (SD 15). Chi-square test comparing distribution of scores in 3 bins with the normal population (≥100 [z-score ≥ 0]; 85-99 [z-score between 0 and -1]; and <85 [z-score < -1]). Effect sizes are designated as small (0.20), medium (0.50), and large (0.80).
As seen in Figure 2, the distribution of scores at Age 1 was shifted downward, with a higher proportion of below-average scores than expected compared with the normal population, for all BSID-II and Bayley-III scales except Cognitive (p<0.05). At Age 1, participants' scores fell below 85 at least twice the expected rate on BSID-II Physical and Bayley-III Motor (59% and 32% vs. 16% expected), and BSID-II Mental and Bayley-III Language scales (38% and 33% vs. 16% expected). At Age 2, scores were still shifted downward for those completing the BSID-II, but differences from the expected normal distribution were not significant on Bayley-III.
Figure 2.
Distribution of Bayley scores at age one year (Age 1) and 2 years (Age 2) compared with the normal population. Chi-square test comparing distribution of scores in 3 bins with the normal population (≥100 [z score ≥0]; 85-99 [-1 ≤ z-score <0]; <85 [z< -1].
Sixty-nine participants completed ND testing (45 BSID-II and 21 Bayley-III) at both Age 1 and Age 2, and used the same Bayley version each time. The distribution of motor and cognitive subdomain scores did not change significantly over time in these subjects.
Predictors for being “At Risk for Physical/Motor Delay”
Results of univariate and multivariate analyses identifying “At risk for Physical/Motor Delay” predictors are reported in Table 3. Univariate analysis identified older age at HPE, the presence of one or more medical complications (ascites or GI bleed in prior year or thrombocytopenia at testing), ascites alone in the prior year, and decreased length z-scores at time of testing as risk factors for Physical/Motor impairment at 1 year of age. Unsuccessful HPE was also identified a risk factor for Physical/Motor delay at 1 and 2 years of age. In multivariate analysis, only ascites (OR 3.17) and lower length z-score (OR 0.70) were independently associated with higher risk of physical/motor impairment in participants at one year of age, and unsuccessful HPE remained the unique predictor of higher risk at Age 2 (OR 4.88).
Table 3.
Variables included in univariate and multivariate logistic regression results for “At Risk for Physical/Motor Delay”.
| UNIVARIATE | Age 1 (N=132) | Age 2 (N=85) | ||
|---|---|---|---|---|
| Odds Ratio (95% CI) | P value | Odds Ratio (95% CI) | P value | |
| Demographic variables | ||||
| Male vs. Female | 0.84 (0.41, 1.73) | 0.63 | 1.16 (0.43, 3.12) | 0.76 |
| Race | ||||
| Black vs. White | 0.34 (0.12, 0.95) | 0.10 | 2.04 (0.49, 8.41) | 0.30 |
| Other vs. White | 0.62 (0.24, 1.61) | 2.39 (0.70, 8.21) | ||
| Non-Hispanic vs. Hispanic | 0.37 (0.13, 1.06) | 0.064 | 0.49(0.15, 1.62) | 0.24 |
| Highest Household Education: College and above vs. High school or less | 0.98 (0.35, 2.76) | 0.98 | 0.45 (0.13, 1.54) | 0.20 |
| Baseline medical variables | ||||
| BASM syndrome | NE | n/a | 4.31 (0.65, 28.66) | 0.13 |
| Study Group | ||||
| START Steroid vs. START placebo | 2.03 (0.69, 5.93) | 0.20 | 0.75 (0.19, 2.99) | 0.68 |
| START Steroid vs. PROBE only | 0.86 (0.34, 2.22) | 0.76 | 0.43 (0.13, 1.41) | 0.17 |
| START Placebo vs. PROBE only | 0.43 (0.17, 1.09) | 0.076 | 0.67 (0.19, 2.34) | 0.53 |
| Age at HPE | 1.67 (1.05, 2.65) | 0.030 | 1.56 (0.81, 2.99) | 0.18 |
| Unsuccessful HPE (total bilirubin >2 at 3 mos.) | 3.22 (1.19, 9.09) | 0.022 | 0.21 (0.06, 0.76) | 0.018 |
| Medical variables since HPE | ||||
| Ascites | 3.35 (1.35, 8.27) | 0.009 | 1.99 (0.49, 8.12) | 0.34 |
| Cholangitis | 1.14 (0.55, 2.34) | 0.73 | 2.55 (0.74, 8.79) | 0.14 |
| Gastrointestinal bleeding | 0.55 (0.03, 9.08) | 0.67 | 2.93 (0.37, 23.16) | 0.31 |
| Thrombocytopenia at testing (platelet <150 103/microliter) | 2.09 (0.69, 6.34) | 0.19 | 1.83 (0.52, 6.49) | 0.35 |
| Combined medical risk** | 2.59 (1.17, 5.73) | 0.0187 | 2.95 (0.96, 9.03) | 0.06 |
| Variables on day of testing (+/- 2mos) | ||||
| Growth parameters | ||||
| Weight z-score | 0.78 (0.59, 1.03) | 0.080 | 0.93 (0.66, 1.32) | 0.70 |
| Height z-score | 0.68 (0.49, 0.95) | 0.020 | 0.87 (0.54, 1.40) | 0.57 |
| Liver biochemical values | ||||
| Serum total bilirubin (mg/dl) | 1.09 (0.98, 1.21) | 0.13 | 1.20 (0.96, 1.49) | 0.11 |
| INR | 2.70 (0.21, 35.21) | 0.45 | 5.78 (0.14, 235.00) | 0.35 |
| MULTIVARIATE*** | Age 1 (N=132) | Age 2 (N=85) | ||
| Odds Ratio (95 % CI) | P value | Odds Ratio (95 % CI) | P value | |
| Height Z Score at Testing | 0.70 (0.50, 0.98) | 0.035 | -- | -- |
| Ascites | 3.17 (1.27, 7.93) | 0.014 | -- | -- |
| Unsuccessful HPE (TB > 2 ug/dL at 3 months) | 4.88 (1.31, 16.7) | 0.018 | ||
one or more of the following: History of ascites, or gastrointestinal bleed in preceding year; platelets <150 at time of testing.
Stepwise selection process was used to arrive at final models. Only variables with p≤0.05 remain in the final models.
OR >1.0 indicates increased risk for ND delay.
All models adjusted for Bayley version.
NE: not estimable; n/a: not applicable.
Predictors for being “At Risk for Mental, Cognitive or Language Delay”
Univariate and multivariate analyses of risk factors associated with Mental/Cognitive/Language delay are provided in Table 4. At Age 1, univariate analysis identified five significant predictors of Mental/Cognitive/Language delays: low weight and length z-scores, higher recent serum total bilirubin level, ascites, and older age at HPE. Only race, low weight at time of ND testing (OR 0.57) and ascites within the prior year (OR 2.89) retained significance by multivariate analysis. Univariate analysis of participants at Age 2 revealed higher level of serum total bilirubin at testing, older age at HPE, unsuccessful HPE, and presence of medical complications as risk factors for impairments in Mental/Cognitive/Language development; whereas household education of college or greater was protective. On multivariate analysis, only two variables retained significance: unsuccessful HPE (OR 4.76) and household educational level (OR 0.26).
Table 4.
Variables included in univariate and multivariate logistic regression results for “At Risk for Mental/Cognitive/Language Delay”.
| UNIVARIATE | Age 1 (N=132) | Age 2 (N=85) | ||
|---|---|---|---|---|
| Odds Ratio (95% CI) | P value | Odds Ratio (95% CI) | P value | |
| Demographics | ||||
| Male vs. Female | 0.84 (0.41, 1.73) | 0.63 | 0.99 (0.40, 2.46) | 0.99 |
| Race | 0.09 | 0.35 | ||
| Black vs. White | 0.92 (0.34, 2.50) | 3.45 (0.87, 13.77) | ||
| Other vs. White | 2.84 (1.09, 7.42) | 3.12 (0.94, 10.33) | ||
| Highest Household Education College and above vs. High school or less | 1.55 (0.51, 4.73) | 0.44 | 0.29 (0.09, 0.96) | 0.043 |
| Medical history at baseline | ||||
| BASM syndrome | 3.64 (0.86, 15.34) | 0.0784 | 7.51 (0.79, 71.10) | 0.079 |
| Study Group | ||||
| START Steroid vs. START placebo | 0.81 (0.30, 2.17) | 0.67 | 2.28 (0.62, 8.41) | 0.21 |
| START Steroid vs. PROBE only | 1.01 (0.41, 2.50) | 0.99 | 1.29 (0.48, 3.50) | 0.62 |
| START Placebo vs. PROBE only | 1.32 (0.54, 3.20) | 0.54 | 0.65 (0.19, 2.25) | 0.50 |
| Age at HPE | 1.54 (1.02, 2.32) | 0.038 | 1.86 (0.997, 3.46) | 0.051 |
| Unsuccessful HPE (total bilirubin>2 at 3 mos) | 2.13 (0.94, 5) | 0.068 | 4.17 (1.12, 14.29) | 0.0326 |
| Medical history in year prior to testing | ||||
| Ascites | 2.82 (1.28, 6.22) | 0.010 | 3.80 (0.98, 14.81) | 0.054 |
| Cholangitis | 1.26 (0.62, 2.57) | 0.53 | 0.61 (0.17, 2.16) | 0.45 |
| Gastrointestinal bleeding | NE | n/a | 1.76 (0.23, 13.33) | 0.58 |
| Thrombocytopenia at testing (platelet <150 103/microliter) | 1.24 (0.46, 3.40) | 0.67 | 1.94 (0.60, 6.28) | 0.27 |
| Combined medical risk** | 1.98 (0.95, 4.13) | 0.068 | 2.98 (1.04, 8.56) | 0.0420 |
| Measures taken at testing (+/- 2 mos) | ||||
| Growth parameters | ||||
| Weight z-score | 0.69 (0.52, 0.92) | 0.012 | 0.96 (0.69, 1.34) | 0.83 |
| Height z-score | 0.66 (0.48, 0.92) | 0.015 | 1.01 (0.66, 1.56) | 0.95 |
| Liver biochemical values | ||||
| Serum total bilirubin (mg/dl) | 1.11 (1.02, 1.21) | 0.020 | 1.80 (1.17, 2.75) | 0.0071 |
| INR | 4.09 (0.49, 33.87) | 0.19 | 1.72 (0.07, 57.21) | 0.69 |
| MULTIVARIATE | Age 1 (N=132) | Age 2 (N=85) | ||
| Odds Ratio (95 % CI) | P value | Odds Ratio (95 % CI) | P value | |
| Race | 0.010 | |||
| Black vs white | 0.70 (0.22, 2.21) | |||
| Other vs white | 4.68 (1.56, 14.08) | |||
| Highest Household Education | 0.26 (0.08, 0.90) | 0.03 | ||
| Weight z-score at testing | 0.57 (0.41, 0.80) | 0.0013 | -- | -- |
| Ascites in last year | 2.89 (1.23, 6.72) | 0.014 | -- | -- |
| Unsuccessful HPE (total bilirubin >2 at 3 mos) | 4.76 (1.20, 16.67) | 0.025 | ||
One or more of the following: History of ascites, or gastrointestinal bleed in preceding year; platelets<150 at time of testing.
Stepwise selection process was used to arrive at final models. Only variables with p≤0.05 remain in the final models.
OR >1.0 indicates increased risk for ND delay.
All models adjusted for Bayley version.
NE: not estimable; n/a: not applicable
Successful vs Unsuccessful HPE and “At risk for ND delay” Outcomes
At Age 1, 53% (46/87) of participants with successful HPE were identified as being at risk for Physical/Motor delay compared with 77% (20/26) of participants with unsuccessful HPE. At Age 2, 25% (16/64) of participants with successful HPE were identified as at risk in this domain, compared with 66% (6/9) of patients with unsuccessful HPE; 38% (33/88) of Age 1 and 33% (22/66) of Age 2 participants with successful HPE were at risk for Mental/Cognitive/Language delay, compared with 55% (16/29) Age 1 participants and 67% (6/9) Age 2 participants with unsuccessful HPE.
Discussion
In this multicenter study, we demonstrate that young children with BA surviving with native livers after HPE are at increased risk for ND delays at 1 and 2 years of age compared with test norms. Markers of advanced liver disease such as ascites and growth deficits were associated with worse ND outcomes. ND scores remained stable across categories (≥100, 85-99, <85) from Age 1 to 2 in those tested at both times. Children with BA living with an unsuccessful HPE have a 2 to 5 times higher incidence of delays in both Physical/Motor skills as well as Mental/Cognitive/Language development during the first 2 years of life.
Recent research initiatives have focused largely on neurocognitive outcomes in cohorts of pediatric LT recipients, comprised largely of BA participants and show that developmental delays are prominent at the time of listing and post-transplant 15, 16. Only a handful of small, single-center studies have examined ND outcomes specifically in infants with BA and their native livers, with cognitive and motor delays most consistently reported in both pre-transplant 5, 6, 8, 17 as well as one-year post-LT patient populations 17.
In our study, unsuccessful HPE was associated with greatly increased risk of ND delays with 75% at risk in the Physical/Motor domain, and 50% at risk for Mental/Cognitive/Language delays at Age 1. Nevertheless, even BA patients with evidence of a successful HPE remain vulnerable, with 1/4 at risk for Physical/Motor delays, and 1/3 at risk for Mental/Cognitive/Language delays at 2 years of age. Given that the median age at LT is less than 2 years, our results underscore the importance of identifying and addressing those at risk for ND delays swiftly even when they do not meet indications for LT. Previous detailed characterization of children with biliary atresia with their native liver 5 or more years after HPE (ie, who are not listed for LT) revealed progressive clinical or biochemical evidence of chronic liver disease and its consequences 18. Strategies are needed to identify those most at risk for ND delays and enable interventions at the earliest age possible to ensure optimal outcomes for patients with this chronic childhood liver condition.
Infancy and early childhood is a time of critical brain development, and an important period for ND surveillance, especially in children with chronic illnesses. Infants diagnosed with BA are potentially more vulnerable given the absence of alternate treatments and the need for timely surgical intervention (HPE) requiring prolonged general anesthesia (GA) within the first months of life. However, a recent study examining 336 infants found few significant effects of GA in the first year of life on ND outcomes compared with controls 19. Additionally, interim results from two long-term studies – one a randomized trial comparing GA with regional anesthesia20, and another looking at sibling pairs21 – noted no significant effect of GA on ND outcomes using standardized testing after a single GA exposure. Given the essential role of GA in order for surgical procedures (like the HPE) to be performed safely in infants, concern for GA on the developing brain should not result in a delay in the timely completion of the HPE procedure, as it is likely that the ND issues are related to the primary conditions and comorbidities, and not to the use of GA20, 21.
Our study found that BA children with ascites at 1 year of age were 3 times more likely to have Physical/Motor delays. The presence of ascites, a known marker of advanced liver disease, may impair motor development due to abdominal distension that could potentially inhibit normal infant movement and exploration 6, 7, 17, 22. Advanced liver disease may also affect motor development through its effect on the brain and its association with poor nutritional status and loss of muscle mass. Other markers of advanced liver disease, such as international normalized ratio (INR), have also been reported to correlate negatively with both gross and fine motor performance 6, although, this was not found in our cohort. Motor deficits have been found to persist through school age in patients with chronic liver disease 23. An association has been found between motor deficits and long-term outcomes in pediatric acute liver failure 24, and in children with extrahepatic portal vein obstruction with signs of hyperammonemia and cerebral edema on imaging 25, 26. Ascites was also a significant predictor of Mental/Cognitive/Language attainment at 1 and 2 years of age. Diseased liver with a reduced capacity to metabolize a variety of potentially cerebrotoxic substances in the blood may also be contributing to cognitive deficits in infants with advanced liver disease and portal hypertension7.
Failure to thrive is a serious consequence of chronic liver disease and is associated with increased morbidity and mortality 27. Our study found that lower weight and length z-scores at testing were significant predictors of Mental/Cognitive/Language delay at 1 year of age and lower length z-scores predicted risk for Physical/Motor delay at 1 year, but neither length nor weight predicted outcome at 2 years of age. In our cohort, participants surviving to age 2 with their native livers had mean length and weight z-scores much closer to the average than participants tested at Age 1, who underwent LT or died prior to Age 2 testing. Unfortunately, growth delays related to early malnutrition have consistently predicted long-term neurocognitive outcomes in chronic liver disease patients, well after LT27, suggesting an early possibly irreversible impact on brain development. These findings suggest that optimizing early nutrition and growth in infants with BA could potentially have important ramifications for ND outcomes.
In our study, a small sub-sample was simultaneously enrolled in a blinded, randomized, placebo-controlled study of corticosteroids as adjunctive therapy post-HPE 28. Steroid use was not associated with ND outcomes in these patients. The limited research available on cognitive effects of corticosteroids in children suggests potential acute worsening in areas such as attention, memory, and executive functioning 29, 30 that are not measurable in infants. Thus, prospective assessment of relevant functional domains later in development is needed before concluding that corticosteroids have no impact on ND outcomes in BA patients.
Despite examining ND outcomes in the largest prospective cohort of infants with biliary atresia to date, we acknowledge potential limitations and biases of this study. First, our sample reflects at best 50% attainment of eligible participants. Although specific reasons for non-participation were not available, ND data collection in infants (particularly those with a serious chronic disease or those who are extremely ill) is challenging. Secondly, a much larger proportion of participants with successful HPE underwent ND testing at both time points compared with eligible participants with unsuccessful HPE, indicating that our sample was likely healthier than the general BA population. Indeed, our study did not capture children with unsuccessful HPE who were transplanted (or died) prior to Age 1, and participants with ND testing at Age 2 were healthier than those with testing at Age 1 for the same reason (drop-out due to death or LT after Age 1). This bias towards recruitment of healthier participants is common in research studies, and actually, underscores the significance of these findings. Finally, a particular challenge to the interpretation of this database was the mid-study shift in Bayley version. The test structure changed significantly with major revision and redistribution of items from 2 core scales on the BSID-II to 3 on the Bayley-III, thereby eliminating the possibility of simply combining scores across versions for analysis. Validity studies show only modest correlations between the two versions, with significantly lower scores on BSID-II compared with Bayley-III editions 12, 13. Indeed, this pattern held in our own sample. Other studies have also faced this challenge, and used various strategies to attempt to equate the two versions by conversion algorithm, regression analysis, or setting a higher cut point on Bayley-III Cognitive (and Language) scales compared with BSID-II Mental scale 31-34. Although these strategies have all assumed that the Bayley-III underestimates ND delay, there is now accumulating evidence that BSID-II overestimates ND delay, and Bayley-III scores are actually more valid 11. Some studies are reporting high correlations of 0.81 and 0.78 between Bayley-III Cognitive and Language scores at 2 years of age with Wechsler Preschool and Primary Scale of Intelligence-III (WPPSI-III) Full Scale IQ at 4 years of age in preterm children, with 81% of studied participants maintaining the same developmental classification from Bayley-III to WPPSI-III 35, 36. In our study, we addressed this dilemma with the definition of “at risk” groups for modeling, which allowed capture of all patients with scores at the lower end of the distribution regardless of version or scale. We also ensured that our univariate multivariate analyses adjusted for Bayley version. Only longitudinal assessment of this BA population will determine if the risk for ND delays found within the first 2 years will persist at older ages; these studies are in progress to document development and evaluate the impact of interventions.
In conclusion, this study provides benchmarks for ND outcomes expected in 1- and 2-year-old children with BA with their native livers. These young children remain at significant risk for ND impairments including both Physical/Motor and Mental/Cognitive/Language domains even in those patients with a successful HPE and not listed for pediatric LT. Stunted growth and complications indicative of advanced liver disease are associated with worse ND outcomes. These findings suggest that strategies to promote successful HPE surgery (such as earlier diagnosis and intervention) and optimize growth and nutrition may improve ND outcomes in this vulnerable population.
Acknowledgments
Financial Support: Supported by U01 grants from the National Institute of Diabetes and Digestive and Kidney Diseases (DK 62445 [Mount Sinai School of Medicine], DK 62497 [Cincinnati Children's Hospital Medical Center], DK 62470 [Children's Healthcare of Atlanta], DK 62481 [The Children's Hospital of Philadelphia], DK 62456 [The University of Michigan], DK 84536 [Riley Hospital for Children], DK 84575 [Seattle Children's Hospital], DK 62500 [UCSF Children's Hospital], DK 62503 [Johns Hopkins School of Medicine], DK 62466 [Children's Hospital of Pittsburgh of UPMC], DK 62453 [Children's Hospital Colorado], DK 62452 [Washington University School of Medicine], DK 84538 [Children's Hospital Los Angeles], DK 62436 [Ann & Robert H Lurie Children's Hospital of Chicago], DK103149 [Texas Children's Hospital], DK103135 [The Hospital for Sick Children], DK103140 [University of Utah]), and by CTSA grants from the National Center for Advancing Translational Sciences: UL1TR000003 [The Children's Hospital of Philadelphia, Philadelphia, PA], UL1TR000077 [Cincinnati Children's Hospital Medical Center, Cincinnati, OH], UL1TR000130 [Children's Hospital Los Angeles, Keck School of Medicine University of Southern California, Los Angeles, CA], UL1TR000067 [Mount Sinai School of Medicine, New York, NY], U01DK062500 and UL1TR000004 [UCSF Benioff Children's Hospital, University of California, San Francisco, CA], UL 1TR000424 [Johns Hopkins School of Medicine, Baltimore, MD], UL1TR000005 [Children's Hospital Pittsburgh of UPMC, Pittsburgh, PA], UL1TR000448 [Washington University, St. Louis, MO], UL1TR000150 [Ann and Robert H. Lurie Children's Hospital and Northwestern University, Chicago, IL], and T32DK67009, UL1TR001082 [University of Colorado School of Medicine, Children's Hospital Colorado, Aurora, CO]. E.A. holds stock in ≪ ≫. K.M. holds stock in ≪ ≫ and received research support from ≪ ≫. B.H. holds stock in ≪ ≫ and is employed by Merck. V.N. is a consultant for ≪ ≫. B.S. is a consultant for ≪ ≫. P.R. is a consultant for ≪ ≫, is on the speaker's bureau of ≪ ≫, and receives research support from ≪ ≫. K.S. is a consultant for ≪ ≫, and receives research support from ≪ ≫. R.S. is a consultant for ≪ ≫ and receives research support from ≪ ≫. S.K. is a consultant for ≪ ≫. J.M. serves on the advisory board for ≪ ≫ and receives research support from ≪ ≫. J.B. receives research support from ≪ ≫.
List of Abbreviations
- BARC
Biliary Atresia Research Consortium
- Bayley-III
Bayley Scales of Infant and Toddler Development, 3rd edition
- BSID-II
Bayley Scales of Infant Development, 2nd edition
- ChiLDReN
Childhood Liver Diseases Research Network
- HPE
Hepatoportoenterostomy
- IRB/REB
Institutional Review Board or Research Ethics Board
- PROBE
Prospective Database of Infants with Cholestasis
- TB
Total Bilirubin
Appendix
Additional members of the Childhood Liver Disease Research Network (ChiLDReN)
Houston
Dr. Paula M. Hertel, MD, Baylor College of Medicine, Co-Investigator, Texas Children's Hospital, Houston, TX.
Dr. Sanjiv Harpavat, MD, PhD, Baylor College of Medicine, Co-Investigator, Texas Children's Hospital, Houston, TX.
Dr. Mary L. Brandt, MD, FACS, Baylor College of Medicine, Co-Investigator, Texas Children's Hospital, Houston, TX.
Dr. Daniel H. Leung, MD, Baylor College of Medicine, Co-Investigator, Texas Children's Hospital, Houston, TX.
Baltimore
Dr. Wikrom Karnsakul, MD, Johns Hopkins School of Medicine, Co-investigator, Baltimore, MD.
Bethesda
Rebecca Torrance, RN, Liver Diseases Research Branch, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, MD
Sherry Hall, MS, Liver Diseases Research Branch, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, MD
Dr. Edward Doo, MD, Liver Diseases Research Branch, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, MD
Dr. Jay H. Hoofnagle, MD, Liver Diseases Research Branch, National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, MD
Chicago
Dr. Peter Whitington, MD, Ann and Robert H. Lurie Children's Hospital of Chicago, Principal Investigator, Pediatrics Division of Gastroenterology, Hepatology, and Nutrition, Chicago, IL.
Dr. Lee Bass, MD, Ann & Robert H. Lurie Children's Hospital, Co-Investigator, Division ofPediatric Gastroenterology, Hepatology and Nutrition Chicago, IL
Cincinnati
Dr. Alexander G. Miethke, MD, Cincinnati Children's Hospital, Co-Investigator, Division of Pediatric Gastroenterology, Hepatology and Nutrition, Cincinnati, OH.
Dr. James E. Heubi, MD, Cincinnati Children's Hospital, Co-Director, Bile Acid Core, Gastroenterology, Hepatology and Nutrition, Cincinnati, OH.
Dr. Kenneth Setchell, PhD, Cincinnati Children's Hospital, Co-Director, Bile Acid Core, Department of Pediatrics – Pathology, Cincinnati, OH.
Dr. Kevin E. Bove, MD, Cincinnati Children's Hospital, Co-Investigator, Pathology, Division of Pediatric Pathology and Laboratory Medicine, Cincinnati, OH.
Dr. Greg Tiao, MD, Cincinnati Children's Hospital, Co-Investigator, Surgeon Division of Pediatric and Thoracic Surgery, Cincinnati, OH.
Colorado
Dr. Cara L. Mack, MD, Children's Hospital Colorado, Multiple Principle Investigator, Section of Pediatric Gastroenterology, Hepatology, Nutrition, Aurora, CO.
Dr. Michael R. Narkewicz, MD, Children's Hospital Colorado, Co-Investigator, Section of Pediatric Gastroenterology, Hepatology, Nutrition, Aurora, CO.
Dr. Amy G. Feldman, MD, Children's Hospital Colorado, Co-Investigator, Section of Pediatric Gastroenterology, Hepatology, Nutrition, Aurora, CO.
Dr. Shikha S. Sundaram, MD, Children's Hospital Colorado, Co-Investigator, Section of Pediatric Gastroenterology, Hepatology, Nutrition, Aurora, CO.
Dr. Frederick J. Suchy, MD, Children's Hospital Colorado, Co-Investigator, Director, Children's Hospital Research Institute, Aurora, CO.
Dr. Frederick M. Karrer, MD, Children's Hospital Colorado, Co-Investigator, Pediatric Surgery, Aurora, CO.
Dr. Mark Lovell, MD, Children's Hospital Colorado, Co-Investigator, Pediatric Pathology, Clinical Genetics, Aurora, CO.
Dr. Johan L. Van Hove, MD, Children's Hospital Colorado, Co-Investigator, Clinical Genetics, Aurora, CO.
Philadelphia
Dr. Elizabeth B. Rand, MD, Children's Hospital of Philadelphia, Co-Investigator, Philadelphia, PA.
Pittsburgh
Dr. James E. Squires, MD, Children's Hospital of Pittsburgh of UPMC, Co-Investigator, Department of Pediatric Gastroenterology, Hepatology and Nutrition, Pittsburgh PA.
Dr. Veena L. Venkat, MD, Children's Hospital of Pittsburgh of UPMC, Co- Investigator, Department of Pediatric Gastroenterology, Hepatology and Nutrition, Pittsburgh PA.
Dr. Rakesh Sindhi, MD, Children's Hospital of Pittsburgh of UPMC, Co- Investigator, Hillman Center for Pediatric Transplantation, Pittsburgh PA.
Dr. Sarangarajan Ranganathan, MD, Children's Hospital of Pittsburgh of UPMC, Co-Investigator, Department of Pathology, Pittsburgh, PA.
San Francisco
Dr. Laura Bull, PhD, UCSF Benioff Children's Hospital, Genetics Core Lab, UCSF Institute for Human Genetics, San Francisco, CA
St. Louis
Dr. Jeffrey Teckman, MD, Saint Louis University School of Medicine, Principal Investigator, Saint Louis University/Cardinal, Glennon Children's Medical Center, St. Louis, MO.
Indianapolis
Dr. Molly Bozic, MD, Riley Hospital for Children, Co-Investigator, Pediatric Gastroenterology, Indianapolis, IN.
Dr. Girish Subbarao, MD, Riley Hospital for Children, Co-Investigator, Pediatric Gastroenterology, Hepatology and Nutrition, Indianapolis, IN.
Seattle
Dr. Simon Horslen, MD, Seattle Children's Hospital, Co-Investigator, Division of Gastroenterology and Hepatology, Seattle, WA.
Dr. Evelyn Hsu, MD, Seattle Children's Hospital, Co-Investigator, Division of Gastroenterology and Hepatology, Seattle, WA.
Dr. Laura Finn, MD, Seattle Children's Hospital, Co-Investigator, Division of Gastroenterology and Hepatology, Seattle, WA.
Dr. Patrick Healey, MD, Seattle Children's Hospital, Co-Investigator, Division of Gastroenterology and Hepatology, Seattle, WA.
Los Angeles
Dr. Rohit Kohli, MD, Children's Hospital Los Angeles, Co-Investigator, Division of Gastroenterology, Hepatology, and Nutrition, Los Angeles, CA.
Dr. Danny Thomas, MD, Children's Hospital Los Angeles, Co-Investigator, Division of Gastroenterology, Hepatology, and Nutrition, Los Angeles, CA.
Dr. Nisreen Soufi, MD, Children's Hospital Los Angeles, Co-Investigator, Division of Gastroenterology, Hepatology, and Nutrition, Los Angeles, CA.
Dr. Sonia Michail, MD, FAAP, CPE, AGAF, Children's Hospital Los Angeles, Co-Investigator, Division of Gastroenterology, Hepatology, and Nutrition, Los Angeles, CA.
Atlanta
Dr. Matt Clifton, MD, Emory University School of Medicine, Co-Investigator, Atlanta, GA.
Dr. Nitika Gupta, MD, Emory University School of Medicine/Children's Healthcare of Atlanta, Co-Investigator, Atlanta, GA.
Dr. Rene Romero, MD, Emory University School of Medicine/Children's Healthcare of Atlanta, Co-Investigator, Atlanta, GA.
Dr. Miriam Vos, MD, Emory University School of Medicine/Children's Healthcare of Atlanta, Co-Investigator, Atlanta, GA.
Dr. Shelley Caltharp, MD, Children's Healthcare of Atlanta, Co-Investigator, Atlanta, GA.
Toronto
Dr. Binita M. Kamath, MD, MBBChir, MRCP, the Hospital for Sick Children, Principal Investigator, Division of Pediatric Gastroenterology, Hepatology and Nutrition, Toronto, ON.
Dr. Simon C. Ling, MB, ChB, MRCP(UK), the Hospital for Sick Children, Co-Investigator, Division of Pediatric Gastroenterology, Hepatology and Nutrition, Toronto, ON.
Dr. Anna Gold, PhD, the Hospital for Sick Children, Co-Investigator, Division of Psychology, Toronto, ON.
Dr. Annie Fecteau, MD CM MSc, FRCS(C), FACS, the Hospital for Sick Children, Co-Investigator, Division of Surgery, Toronto, ON.
Utah
Dr. Stephen L. Guthery, MD, University of Utah, Principal Investigator, Division of Pediatric Gastroenterology, Hepatology and Nutrition, Salt Lake City, UT.
Dr. Kyle Jensen, MD, University of Utah, Co- Investigator, Division of Pediatric Gastroenterology, Hepatology and Nutrition, Salt Lake City, UT.
Dr. Rebecka Meyers, MD, University of Utah, Co-Investigator, Division of Pediatric Surgery, Salt Lake City, UT.
Dr. Amy Lowichik, MD, PhD, University of Utah, Co-Investigator, Division of Pediatric Pathology, Salt Lake City, UT.
Dr. Linda Book, MD, University of Utah, Co-Investigator, Division of Pediatric Gastroenterology Hepatology and Nutrition, Salt Lake City, UT.
Ann Arbor (Data Coordinating Center)
Dr. Robert M. Merion, MD, University of Michigan, Principal Investigator, Department of Transplantation, Ann Arbor, USA
Dr. Cathie Spino, Sc.D., University of Michigan, Co-Investigator, Department of Biostatistics, Ann Arbor, MI, USA
Karen Jones, MS, Department of Biostatistics, University of Michigan, Ann Arbor, MI, USA
Footnotes
The other authors declare no conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Hartley JL, Davenport M, Kelly DA. Biliary Atresia. Lancet. 2009;374:1704–13. doi: 10.1016/S0140-6736(09)60946-6. [DOI] [PubMed] [Google Scholar]
- 2.Kasai MSS. A new operation for non-correctable biliary atresia-hepatic portoenterostomy. Shujitsu. 1959;13:733–9. [Google Scholar]
- 3.Squires RH, Ng V, Romero R, Ekong U, Hardikar W, Emre S, et al. Evaluation of the pediatric patient for liver transplantation: 2014 practice guideline by the American Association for the Study of Liver Diseases, American Society of Transplantation and the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr Gastroenterol Nutr. 2014;59:112–31. doi: 10.1097/MPG.0000000000000431. [DOI] [PubMed] [Google Scholar]
- 4.Erlichman J, Hohlweg K, Haber BA. Biliary atresia: how medical complications and therapies impact outcome. Expert Rev Gastroenterol Hepatol. 2009;3:425–34. doi: 10.1586/egh.09.30. [DOI] [PubMed] [Google Scholar]
- 5.Caudle SE, Katzenstein JM, Karpen S, McLin V. Developmental assessment of infants with biliary atresia: differences between boys and girls. J Pediatr Gastroenterol Nutr. 2012;55:384–9. doi: 10.1097/MPG.0b013e318259ed20. [DOI] [PubMed] [Google Scholar]
- 6.Caudle SE, Katzenstein JM, Karpen SJ, McLin VA. Language and motor skills are impaired in infants with biliary atresia before transplantation. J Pediatr. 2010;156:936–40. doi: 10.1016/j.jpeds.2009.12.014. , 40.e1. [DOI] [PubMed] [Google Scholar]
- 7.Stewart SM, Campbell RA, McCallon D, Waller DA, Andrews WS. Cognitive patterns in school-age children with end-stage liver disease. J Dev Behav Pediatr. 1992;13:331–8. [PubMed] [Google Scholar]
- 8.Stewart SM, Uauy R, Waller DA, Kennard BD, Andrews WS. Mental and motor development correlates in patients with end-stage biliary atresia awaiting liver transplantation. Pediatrics. 1987;79:882–8. [PubMed] [Google Scholar]
- 9.Shneider BL, Magee JC, Karpen SJ, Rand EB, Narkewicz MR, Bass LM, et al. Total Serum Bilirubin within 3 Months of Hepatoportoenterostomy Predicts Short-Term Outcomes in Biliary Atresia. J Pediatr. 2016;170:211–7.e2. doi: 10.1016/j.jpeds.2015.11.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bezerra JA, Spino C, Magee JC, Shneider BL, Rosenthal P, Wang KS, et al. Use of corticosteroids after hepatoportoenterostomy for bile drainage in infants with biliary atresia: the START randomized clinical trial. Jama. 2014;311:1750–9. doi: 10.1001/jama.2014.2623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bayley N. Bayley Scales of Infant Development, Second Edition: Manual. San Antonio, Texas: The Psychological Corporation; 1993. [Google Scholar]
- 12.Bayley N. Bayley scales of infant development—Third edition. Third. San Antonio, Texas, USA: PsychCorp; 2006. [Google Scholar]
- 13.Bayley–III Technical Report 2: Factors Contributing to Differences Between Bayley–III and BSID–II Scores. Pearson Education Inc. or its affiliate(s); 2007. [Google Scholar]
- 14.Raghunathan TE, Lepkowski J, Van Hoewyk J, Solenberger Survey Methodology. A Multivariate Technique for Multiply Imputing Missing Values Using a Sequence of Regression Models. 2001 [Google Scholar]
- 15.Sorensen LG, Neighbors K, Martz K, Zelko F, Bucuvalas JC, Alonso EM. Cognitive and academic outcomes after pediatric liver transplantation: Functional Outcomes Group (FOG) results. Am J Transplant. 2011;11:303–11. doi: 10.1111/j.1600-6143.2010.03363.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Reed-Knight B, Lee JL, Cousins LA, Mee LL. Intellectual and academic performance in children undergoing solid organ pretransplant evaluation. Pediatr Transplant. 2015;19:229–34. doi: 10.1111/petr.12389. [DOI] [PubMed] [Google Scholar]
- 17.Wayman KI, Cox KL, Esquivel CO. Neurodevelopmental outcome of young children with extrahepatic biliary atresia 1 year after liver transplantation. J Pediatr. 1997;131:894–8. doi: 10.1016/s0022-3476(97)70039-8. [DOI] [PubMed] [Google Scholar]
- 18.Hadzic N, Davenport M, Tizzard S, Singer J, Howard ER, Mieli-Vergani G. Long-term survival following Kasai portoenterostomy: is chronic liver disease inevitable? J Pediatr Gastroenterol Nutr. 2003;37:430–3. doi: 10.1097/00005176-200310000-00006. [DOI] [PubMed] [Google Scholar]
- 19.Nestor KA, Zeidan M, Boncore E, Richardson A, Alex G, Weiss M, et al. Neurodevelopmental outcomes in infants undergoing general anesthesia. J Pediatr Surg. 2017;52:895–900. doi: 10.1016/j.jpedsurg.2017.03.008. [DOI] [PubMed] [Google Scholar]
- 20.Davidson AJ, Disma N, de Graaff JC, Withington DE, Dorris L, Bell G, et al. Neurodevelopmental outcome at 2 years of age after general anaesthesia and awake-regional anaesthesia in infancy (GAS): an international multicentre, randomised controlled trial. Lancet. 2016;387:239–50. doi: 10.1016/S0140-6736(15)00608-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sun LS, Li G, Miller TL, Salorio C, Byrne MW, Bellinger DC, et al. Association Between a Single General Anesthesia Exposure Before Age 36 Months and Neurocognitive Outcomes in Later Childhood. Jama. 2016;315:2312–20. doi: 10.1001/jama.2016.6967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stewart SM, Uauy R, Waller DA, Kennard BD, Benser M, Andrews WS. Mental and motor development, social competence, and growth one year after successful pediatric liver transplantation. J Pediatr. 1989;114:574–81. doi: 10.1016/s0022-3476(89)80696-1. [DOI] [PubMed] [Google Scholar]
- 23.Almaas R, Jensen U, Loennecken MC, Tveter AT, Sanengen T, Scholz T, et al. Impaired motor competence in children with transplanted liver. J Pediatr Gastroenterol Nutr. 2015;60:723–8. doi: 10.1097/MPG.0000000000000757. [DOI] [PubMed] [Google Scholar]
- 24.Sorensen LG, Neighbors K, Zhang S, Limbers CA, Varni JW, Ng VL, et al. Neuropsychological functioning and health-related quality of life: pediatric acute liver failure study group results. J Pediatr Gastroenterol Nutr. 2015;60:75–83. doi: 10.1097/MPG.0000000000000575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yadav SK, Saksena S, Srivastava A, Srivastava A, Saraswat VA, Thomas MA, et al. Brain MR imaging and 1H-MR spectroscopy changes in patients with extrahepatic portal vein obstruction from early childhood to adulthood. AJNR American journal of neuroradiology. 2010;31:1337–42. doi: 10.3174/ajnr.A2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yadav SK, Srivastava A, Srivastava A, Thomas MA, Agarwal J, Pandey CM, et al. Encephalopathy assessment in children with extra-hepatic portal vein obstruction with MR, psychometry and critical flicker frequency. J Hepatol. 2010;52:348–54. doi: 10.1016/j.jhep.2009.12.012. [DOI] [PubMed] [Google Scholar]
- 27.Rodriguez-Baez N, Wayman Karen I, Cox Kenneth L. Cox Growth and Development in Chronic Liver Disease. NeoReviews. 2011;2 [Google Scholar]
- 28.Venkat VL, Shneider BL, Magee JC, Turmelle Y, Arnon R, Bezerra JA, et al. Total serum bilirubin predicts fat-soluble vitamin deficiency better than serum bile acids in infants with biliary atresia. Journal of pediatric gastroenterology and nutrition. 2014;59:702–7. doi: 10.1097/MPG.0000000000000547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Klein-Gitelman MS, Pachman LM. Intravenous corticosteroids: adverse reactions are more variable than expected in children. The Journal of rheumatology. 1998;25:1995–2002. [PubMed] [Google Scholar]
- 30.Mrakotsky C, Forbes PW, Bernstein JH, Grand RJ, Bousvaros A, Szigethy E, et al. Acute cognitive and behavioral effects of systemic corticosteroids in children treated for inflammatory bowel disease. Journal of the International Neuropsychological Society : JINS. 2013;19:96–109. doi: 10.1017/S1355617712001014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Moore T, Johnson S, Haider S, Hennessy E, Marlow N. Relationship between test scores using the second and third editions of the Bayley Scales in extremely preterm children. J Pediatr. 2012;160:553–8. doi: 10.1016/j.jpeds.2011.09.047. [DOI] [PubMed] [Google Scholar]
- 32.Jary S, Whitelaw A, Walloe L, Thoresen M. Comparison of Bayley-2 and Bayley-3 scores at 18 months in term infants following neonatal encephalopathy and therapeutic hypothermia. Dev Med Child Neurol. 2013;55:1053–9. doi: 10.1111/dmcn.12208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Younge N, Goldstein RF, Bann CM, Hintz SR, Patel RM, Smith PB, et al. Survival and Neurodevelopmental Outcomes among Periviable Infants. N Engl J Med. 2017;376:617–28. doi: 10.1056/NEJMoa1605566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Johnson S, Moore T, Marlow N. Using the Bayley-III to assess neurodevelopmental delay: which cutoff should be used? Pediatric research. 2014;75:670–4. doi: 10.1038/pr.2014.10. [DOI] [PubMed] [Google Scholar]
- 35.Aylward GP. Continuing issues with the Bayley-III: where to go from here. J Dev Behav Pediatr. 2013;34:697–701. doi: 10.1097/DBP.0000000000000000. [DOI] [PubMed] [Google Scholar]
- 36.Bode MM, D'Eugenio DB, Mettelman BB, Gross SJ. Predictive validity of the Bayley, Third Edition at 2 years for intelligence quotient at 4 years in preterm infants. J Dev Behav Pediatr. 2014;35:570–5. doi: 10.1097/DBP.0000000000000110. [DOI] [PubMed] [Google Scholar]