Abstract
Background
The study was conducted to determine the impact of chronic obstructive pulmonary disease (COPD) in association with obstructive sleep apnea syndrome (OSAS) on cardiac autonomic control and functional capacity.
Subjects and methods
The study was a cross-sectional prospective controlled clinical study. Heart rate variability indices of 24 COPD (n = 12) and COPD+OSAS (n = 12) patients were evaluated and compared by electrocardiographic recordings acquired during rest, active postural maneuver (APM), respiratory sinus arrhythmia maneuver (RSA-m), and the 6-minute walk test (6MWT).
Results
The COPD group presented higher parasympathetic modulation during APM when compared to the COPD+OSAS group (P = 0.02). The COPD+OSAS group presented higher sympathetic modulation during RSA-m when compared to the COPD group (P = 0.00). The performance during 6MWT was similarly impaired in both groups, despite the greater severity of the COPD group.
Conclusion
Subjects with COPD+OSAS present marked sympathetic modulation, and the presence of OSAS in COPD subjects has a negative impact on functional capacity regardless of the severity of lung disease.
Keywords: COPD, OSAS, COPD+OSAS, functional capacity
Introduction
Chronic obstructive pulmonary disease (COPD) is a syndrome characterized by chronic airway obstruction, which is not completely reversible; it determines significant losses in pulmonary function, has systemic effects, and is associated with important comorbidities.1 Among them, obstructive sleep apnea syndrome (OSAS), characterized by recurrent events of upper airway obstruction during sleep, affects most COPD patients.2 COPD and OSAS are two of the most prevalent chronic respiratory diseases worldwide.3,4
According to Taranto-Montemurro et al,5 the association of the two diseases is present in about 0.5%–1% of the general population; the prevalence of COPD in individuals with OSAS exceeds its prevalence in individuals who do not present with sleep disorders.6 OSAS appears to be more commonly seen in subjects with COPD perhaps as a result of shared risk factors such as obesity, smoking, increased airway resistance, local and systemic inflammation, as well as anti-inflammatory therapy.6
It is known that COPD+OSAS has a great impact on the systemic manifestations of affected patients, such as an increase in daytime sympathetic activity, with an increase in rest heart rate (HR) and a decrease in heart rate variability (HRV), which may lead to greater morbidity and mortality from other diseases.5 On the other hand, other authors have demonstrated that COPD+OSAS determines marked parasympathetic hyperactivity in the airways and hypoxemia-related bronchoconstriction/vasoconstriction, among other neurohumoral effects.6 These still contradictory autonomic imbalances could have a negative impact on static postural adjustments and responses to exercise. To our knowledge, however, no previous study has evaluated and compared such adjustments in these patients.
Thus, the main objective of this study was to determine the impact of the coexistence of OSAS and COPD on cardiac autonomic control in response to changes in breathing pattern, active postural maneuver (APM), and during submaximal exercise (6-minute walk test [6MWT]). We hypothesized that the coexistence of OSAS and COPD would be accompanied by greater cardiovascular, autonomic, and functional impairments than COPD alone.
Subjects and methods
Subjects
Twenty-four individuals of both sexes were screened at the Pulmonary Ambulatory of the School Health Unit (USE) of Federal University of São Carlos (UFSCar) and at the Municipal Health Specialty Center of São Carlos (CEME) with clinical diagnosis of COPD according to the criteria defined by the GOLD scientific committee7 with suspected diagnosis of OSAS.
Inclusion criteria
COPD group: clinical diagnosis of COPD according to the criteria defined by the GOLD scientific committee,7 in regular treatment with optimized medication, current nonsmokers, people who were not in exacerbations period, and people whose after-home sleep examination did not confirm the suspicion of OSAS.
COPD+OSAS group: the same criteria for inclusion in COPD group were followed; however, the individuals presented a confirmed diagnosis of OSAS.
Subjects should not present clinical exacerbations or be on oral steroids for at least 3 months.
Exclusion criteria
Exclusion criteria included presence of diabetes mellitus, systemic arterial hypertension and uncontrolled pulmonary hypertension, use of medications capable of altering cardiac autonomic control, thyroid hormone alterations, use of home oxygen therapy or continuous positive airway pressure (CPAP) for the treatment of OSAS, pacemaker, cardiac arrhythmias interfering with >95% of heart beats, and neurological and orthopedic alterations that would prevent the performance of the proposed tests.
The study was approved by the Ethics and Research Committee of Federal University of São Carlos, São Paulo, Brazil (number 1.406.894/2016), and all the volunteers signed a written informed consent agreement in compliance with resolution number 466/2012 of the National Health Council.
Study protocol
All volunteers were subjected to two stages of evaluation: 1) investigation of the presence of OSAS in subjects with COPD and 2) evaluation of the autonomic function through HRV analysis and of the functional capacity through 6MWT application.
Measurements
In order to control the influence of circadian variations on the results, all tests were performed at the same time of the day in a laboratory with a mean temperature maintained between 20°C and 22°C and relative humidity in the range from 40% to 60%. All subjects were instructed to abstain from alcohol or stimulants on the day of the tests and not to practice physical or sports activities the day before and on the day of the tests.
Pulmonary function
Spirometric evaluation was performed with a CPFS/D® spirometer (Medgraphics, MGC Diagnostics Corporation, St Paul, MN, USA), according to the criteria of the Brazilian Society of Pulmonology and Tisiology,8 and were staged according to the GOLD criteria.7
Home sleep test
The apnea-hypopnea index (AHI) and the oxygen desaturation index (ODI) were obtained by an ApneaLink Plus™ equipment (ResMed Corporation, Poway, CA, USA), which is a portable sleep monitor validated to assess the presence of OSAS.9 Subjects with AHI ≥15 were included in the COPD+OSAS group, while those with AHI <15 were included in the COPD group.
After group stratification, autonomic evaluation was initiated and included.
Recording of R-R intervals
The electrocardiographic signal was recorded through a BioAmp FE132 device (ADInstruments, Sydney, Australia), with electrodes placed in the MC5 lead configuration. Signals were recorded during the conditions shown in the following sections.
Active postural maneuver
The subjects remained in supine position for 10 minutes and were instructed not to talk, sleep, or move during this time. After this rest period, the subjects were instructed to position themselves in orthostatic position (10 minutes) and then in sitting position (10 minutes), following the instructions given for the rest period.
Respiratory sinus arrhythmia maneuver
After APM, the subjects, still seated, were subjected to respiratory sinus arrhythmia maneuver (RSA-m; controlled breathing with cycles of 10 seconds, divided into 5 seconds of inspiration and 5 seconds of expiration) during 4 minutes.
For HRV analysis during MPA and m-ASR, the R-R intervals (iRR) were collected at a rate of 500 samples/s. All signals were visually inspected to identify artifacts or noises where narrow peaks (<100 ms) were removed by linear interpolation. All signals were filtered with low pass filters with a cutoff frequency of 20 Hz.
Data were analyzed using Kubios HRV® software (Version 2.1, Matlab, Kuopio, Finland). The data collected during each MPA posture had a total period of iRR examined and, for analysis, the most stable and noise-free segment that contains 300 points was selected. In addition, the data collected during m-ASR had all the iRR contained within the 4 minutes of the maneuver used for the analysis.
Submaximal exercise using the 6-minute walk test
General test principles, criteria for discontinuation, as well as verbal stimuli were standardized based on the recommendations of the American Thoracic Society and the American College of Chest Physicians.10 After a 4-minute rest period (2 minutes in sitting position and 2 minutes in orthostatic position), the volunteers were instructed to walk continuously, as far as possible, in a 30-meter walk for 6 minutes, and allowed to slow down and even interrupt the test if necessary. At the end, the distance traveled in meters as well as percentage in relation to the predicted was recorded.
HR was recorded by a Polar HR monitor (RX810, Kempele, Finland) during 4 minutes before the test (rest), throughout the test (exercise), and during 6 minutes after the test (recovery). For HRV analysis during the 6MWT, the data collected by the HR monitor were visually inspected to identify artifacts or noises where narrow peaks (<100 ms) were removed by linear interpolation. All signals were filtered with low pass filters with a cutoff frequency of 20 Hz.
Data were analyzed using Kubios HRV® software (Version 2.1, Matlab). The data were analyzed as follows:
Rest in the seated position: The 2 minutes of the seated rest period were analyzed.
Rest in the orthostatic position: iRR were analyzed from the 2 minutes of the standing rest period.
Walk: For analysis, we selected the most stable and noise-free segment that should have 3 minutes.
Recovery: The last 5 minutes of collected iRR were selected for analysis.
Selected sections were analyzed in the time and frequency domains and by means of non-linear analysis. In the time domain, the following indices were calculated: 1) SDNN: standard deviation of all iRR; 2) RMSSD: square root of the mean squared differences between adjacent iRR; 3) pnn50: percentage of adjacent iRR with duration difference greater than 50 ms; 4) TINN: iRR variability.11 SDNN and TINN represent global autonomic activity.11 RMSSD and pnn50 are representative of vagal modulation.11 In the frequency domain, the following indices were calculated: 1) high frequency component (HF); frequency band between 0.15 Hz and 0.4 Hz; 2) low frequency component (LF); frequency band between 0.04 Hz and 0.15 Hz; and 3) LF/HF ratio.11 The HF index represents cardiac parasympathetic modulation; the LF index represents the joint action of the sympathetic and parasympathetic components, with sympathetic predominance; the LF/HF ratio represents the sympathovagal balance.11 In addition, the following non-linear indices were calculated: 1) SD1: standard deviation of instantaneous beat-to-beat variability; 2) SD2: long-term standard deviation of the continuous iRR.11 SD1 index represents cardiac parasympathetic modulation and SD2 index represents global cardiac autonomic activity.11
Statistical analysis
The results were presented as mean ± standard deviation, with a level of significance of 95%. Categorical variables were compared using the chi-square test. Two-way ANOVA (with Bonferroni post hoc test) was used for comparative analysis of the differences between the HRV indexes. Linear associations were evaluated with Pearson product-moment coefficients. Statistical analysis was performed using SPSS 19.0 (IBM, Armonk, NY, USA) and GraphPad Prism 6.0 (MacKiev Software, Boston, MA, USA).
Results
Population characteristics
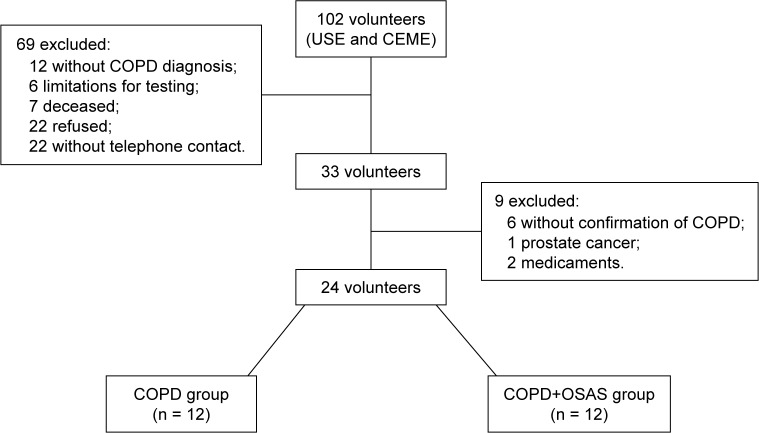
One hundred and two subjects from the Pulmonary Ambulatory of the School Health Unit (USE) of Federal University of São Carlos (UFSCar) and from the Municipal Health Specialty Center of São Carlos (CEME) were screened. The groups were screened as described in Figure 1.
Figure 1.
Volunteer flowchart.
Abbreviations: CEME, Municipal Health Specialty Center of São Carlos; COPD, chronic obstructive pulmonary disease; OSAS, obstructive sleep apnea syndrome; USE, Pulmonary Ambulatory of the School Health Unit.
The baseline characteristics of the volunteers are shown in Table 1. The data contained in Table 1 show that the lung function of the COPD group was worse when compared to the COPD+OSAS group, that is, the degree of respiratory commitment was higher for the COPD group.
Table 1.
Volunteers’ baseline characteristics
| Demographic data | COPD (n = 12) | COPD+OSAS (n = 12) | P-value |
|---|---|---|---|
| Age (years) | 64.83 ± 1.69 | 64.17 ± 2.48 | 0.82 |
| Sex female/male, n (%) | 2 (16.7)/10 (83.3) | 2 (16.7)/10 (83.3) | 1.00 |
| Weight (kg) | 73.32 ± 5.61 | 75.53 ± 3.58 | 0.74 |
| BMI (kg/m2) | 25.49 ± 1.70 | 27.13 ± 1.15 | 0.43 |
| mMRC, 0/1/2/3/4, n (%) | 1 (8.3)/1 (8.3)/2 (16.6)/4 (33.3)/4 (33.3) | 1 (8.3)/4 (33.3)/2 (16.6)/1 (8.3)/4 (33.3) | 0.46 |
| Smoking habits | |||
| Never/ex/current | 0.12.0 | 0.12.0 | 0.35 |
| Pack years | 53.33 ± 29.57 | 67.17 ± 41.09 | |
| Pulmonary function | |||
| FEV1, %pred | 42.70 ± 16.56 | 61.32 ± 18.79 | 0.01* |
| FEV1/FVC, %pred | 41.84 ± 12.22 | 56.29 ± 12.07 | 0.00* |
| COPD 1 and 2, n (%) | 4 (33) | 9 (75) | 0.04* |
| COPD 3 and 4, n (%) | 8 (67) | 3 (25) | 0.04* |
| OSA | |||
| AHI | 5.81 ± 4.44 | 26.72 ± 13.42 | 0.00* |
| ODI | 10.27 ± 7.10 | 46.45 ± 90.82 | 0.01* |
| SaO2 <90%, min | 193.81 ± 166.72 | 64.48 ± 48.57 | 0.04* |
| SaO2 <80%, min | 1.90 ± 3.78 | 1.50 ± 2.71 | 0.94 |
| Therapy, n (%) | |||
| SABA | 4 (33.3) | 5 (41.7) | 0.84 |
| SAMA | Not applicable | 1 (8.3) | 0.84 |
| LABA | 9 (75) | 10 (83.3) | 0.84 |
| LAMA | 6 (50) | 6 (50) | 0.84 |
| ICS | 6 (50) | 6 (50) | 0.84 |
| OCS | 1 (8.33) | Not applicable | 0.84 |
| Theophylline | 2 (16.6) | 1 (8.33) | 0.84 |
| Antihistamine | Not applicable | 1 (8.33) | 0.84 |
Notes:
Significant difference between COPD group and the COPD+OSAS group. The variables are presented as mean ± SD.
Abbreviations: COPD, chronic obstructive pulmonary disease; OSAS, obstructive sleep apnea syndrome; BMI, body mass index; mMRC, modified Medical Research Council dyspnea scale; FEV1, forced expiratory volume in 1 second; pred, predicted; FVC, forced vital capacity; AHI, apnea-hypopnea index; ODI, oxygen desaturation index; SaO2 <90%, saturation time <90%; SaO2 <80%, saturation time <80%; SABA, short-acting beta2-agonist; SAMA, short-acting muscarinic agonist; LABA, long-acting beta2-agonist; LAMA, long-acting muscarinic agonist; ICS, inhaled corticosteroids; OCS, oral corticosteroids.
Although the COPD+OSAS group presented higher levels of AHI and ODI, as expected due to the presence of OSAS, interestingly, the time spent with saturation below 90% (SAT < 90%) and below 80% (SAT < 80%) was much longer in the COPD group, confirming the great impact of the disease on nocturnal hypoxemia in these patients.
HRV indices at rest
The HRV indices during APM and RSA-m are represented in Tables 2 and 3, respectively. The COPD+OSAS group presented higher absolute and normalized LF values when compared to the COPD group (Table 2, P < 0.05). On the other hand, the COPD+OSAS group had lower normalized HF values when compared to the COPD group; taken together, data suggest greater sympathetic modulation in the COPD+OSAS group.
Table 2.
HRV indices before and after active postural change from supine position to orthostatic position
| COPD (n = 12)
|
COPD+OSAS (n = 12)
|
Group | Position | Interaction | |||
|---|---|---|---|---|---|---|---|
| Supine | Orthostasis | Supine | Orthostasis | ||||
| Time | – | – | – | – | |||
| iRR | 794 ± 103 | 719 ± 114* | 818 ± 123 | 764 ± 97* | 0.27 | 0.04 | 0.74 |
| SDNN | 13.9 ± 5.3 | 11.7 ± 6.6 | 16.1 ± 5.8 | 15.2 ± 7.3 | 0.12 | 0.38 | 0.73 |
| RMSSD | 14.3 ± 7.2 | 11.5 ± 6.5 | 15.8 ± 7.9 | 13.7 ± 6.9 | 0.42 | 0.21 | 0.78 |
| pnn50 | 1.7 ± 2.8 | 0.6 ± 1.1 | 1.8 ± 3.7 | 0.5 ± 0.9 | 0.98 | 0.11 | 0.85 |
| TINN | 67.0 ± 22.2 | 55.4 ± 30.4 | 77.9 ± 28.3 | 77.0 ± 34.6 | 0.06 | 0.46 | 0.52 |
| Frequency | – | – | – | – | |||
| LF_abs | 79.8 ± 84.8 | 69.2 ± 66.6 | 156.7 ± 107.7+ | 161.3 ± 144.2+ | ,0.01 | 0.92 | 0.80 |
| HF_abs | 67.5 ± 47.8 | 60.9 ± 101.2 | 106.7 ± 115.1 | 70.6 ± 61.3 | 0.39 | 0.33 | 0.55 |
| LF_nu | 50.1 ± 19.9 | 58.3 ± 19.5 | 62.9 ± 14.8+ | 68.6 ± 14.7+ | 0.02 | 0.17 | 0.80 |
| HF_nu | 49.7 ± 19.8 | 41.2 ± 19.4 | 36.7 ± 14.8 | 31.0 ± 14.6 | 0.02 | 0.16 | 0.78 |
| LF/HF | 1.3 ± 1.0 | 3.0 ± 4.7 | 2.1 ± 1.2 | 4.6 ± 7.4 | 0.37 | 0.11 | 0.75 |
| Non-linear | – | – | – | – | |||
| SD1 | 10.4 ± 5.1 | 8.1 ± 4.6 | 11.2 ± 5.6 | 9.7 ± 4.9 | 0.43 | 0.21 | 0.78 |
| SD2 | 16.3 ± 6.6 | 14.3 ± 8.4 | 19.7 ± 6.7 | 18.9 ± 9.5 | 0.08 | 0.53 | 0.78 |
Notes:
Significant difference between supine position and orthostatic position.
Significant difference between the COPD group and the COPD+OSAS group. The variables are presented as mean ± SD.
Abbreviations: HRV, heart rate variability; COPD, chronic obstructive pulmonary disease; OSAS, obstructive sleep apnea syndrome; iRR, interval between consecutive heart beats; SDNN, standard deviation of all R-R intervals; RMSSD, square root of the mean squared differences between adjacent R-R intervals; pnn50, percentage of adjacent R-R intervals with duration difference greater than 50 ms; TINN, triangular interpolation of the iRR interval histogram; LF_abs, absolute power in the low frequency band; HF_abs, absolute power in the high frequency band; LF_nu, normalized power in the low frequency band; HF_nu, normalized power in the high frequency band; LF/HF, low frequency to high frequency ratio; SD1, standard deviation of instantaneous beat-to-beat variability; SD2, long-term standard deviation of the continuous R-R interval.
Table 3.
HRV indices in sitting position and during RSA-m
| Time | COPD (n = 12)
|
COPD+OSAS (n = 12)
|
Group | Maneuver | Interaction | ||
|---|---|---|---|---|---|---|---|
| Sitting | RSA-m | Sitting | RSA-m | ||||
| iRR | 773 ± 113 | 757 ± 89 | 815 ± 116 | 802 ± 98 | 0.15 | 0.62 | 0.95 |
| SDNN | 14.4 ± 6.1 | 26 ± 16.2* | 17.3 ± 7.1 | 34.8 ± 20.3* | 0.15 | 0.00 | 0.46 |
| RMSSD | 13.2 ± 5.1 | 19.9 ± 19.5 | 17.1 ± 8.1 | 22.7 ± 13.6 | 0.37 | 0.10 | 0.87 |
| pnn50 | 0.4 ± 0.9 | 4.1 ± 11.9 | 1.9 ± 3.4 | 4.6 ± 8.5 | 0.64 | 0.15 | 0.81 |
| TINN | 70 ± 29.1 | 123.7 ± 83.0* | 87.5 ± 35.3 | 160 ± 87.2* | 0.15 | 0.00 | 0.61 |
Notes:
Significant difference between sitting position and RSA-m. The variables are presented as mean ± SD.
Abbreviations: HRV, heart rate variability; RSA-m, respiratory sinus arrhythmia maneuver; COPD, chronic obstructive pulmonary disease; OSAS, obstructive sleep apnea syndrome; iRR, interval between consecutive heart beats; SDNN, standard deviation of all R-R intervals; RMSSD, square root of the mean squared differences between adjacent R-R intervals; pnn50, percentage of adjacent R-R intervals with duration difference greater than 50 ms; TINN, triangular interpolation of the iRR interval histogram.
Table 3 shows that both groups had higher SDNN and TINN values during RSA-m when compared to the sitting position, suggesting a greater global autonomic modulation for both groups during the maneuver (P < 0.05). However, there was no interaction between APM values for the two groups.
6MWT variables
The 6MWT parameters are shown in Table 4. Table 4 shows that the COPD+OSAS group had higher SBP than the COPD group (P < 0.05). In addition, symptoms of dyspnea and lower limbs fatigue were worse in the COPD group than in the COPD+OSAS group (P < 0.05), even though we did not observe a worse performance in this group. Although there were no differences in SpO2 between groups during 6MWT (P = 0.21), the COPD group presented an average value below 90%, which indicates that desaturation was clinically more pronounced in this group.
Table 4.
6MWT parameters
| COPD (n = 12) |
COPD+OSAS (n = 12) |
P-value | |
|---|---|---|---|
| Rest | |||
| SBP (mmHg) | 118 ± 10 | 129 ± 9 | 0.01* |
| HR (bpm) | 82 ± 11 | 79 ± 13 | 0.56 |
| SpO2 | 93 ± 3 | 95 ± 3 | 0.13 |
| BORG D | 0 ± 1 | 0 ± 0 | 0.10 |
| BORG P | 0 ± 1 | 0 ± 1 | 0.75 |
| Peaks | |||
| SBP (mmHg) | 147 ± 15 | 159 ± 25 | 0.17 |
| HR (bpm) | 104 ± 17 | 106 ± 13 | 0.72 |
| SpO2 | 87 ± 9 | 91 ± 4 | 0.21 |
| BORG D | 3 ± 2 | 2 ± 1 | 0.04* |
| BORG P | 4 ± 3 | 1 ± 1 | 0.01* |
| Covered distance | |||
| CD predicted (m) | 557.17 ± 38.48 | 571.58 ± 41.42 | 0.38 |
| CD reached (m) | 362.12 ± 123.14 | 419.04 ± 77.66 | 0.18 |
| % predicted | 64.58 ± 20.03 | 73.45 ± 13.54 | 0.21 |
| CDR COPD 1 and 2/CDR COPD+OSAS 3 and 4 | 320 ± 137 | 448 ± 33.05 | 0.18 |
| CDR COPD 3 and 4/CDR COPD+OSAS 1 and 2 | 394 ± 114 | 409 ± 87 | 0.76 |
Notes:
Significant difference between groups (unpaired t-test). The variables are presented as mean ± SD.
Abbreviations: 6MWT, 6-minute walk test; COPD, chronic obstructive pulmonary disease; OSAS, obstructive sleep apnea syndrome; SBP, systolic blood pressure; HR, heart rate; SpO2, peripheral oxygen saturation; BORG D, dyspnea Borg Scale; BORG P, leg discomfort Borg Scale; CD, covered distance; CDR, covered distance reach.
Strong correlations were observed between the HRV indices during 6MWT and nocturnal desaturation in these patients. We observed that the longer the time spent with saturation below 90%, the greater the parasympathetic modulation during walking, represented by the RMSSD and SD1 indices (R = 0.80, P < 0.05).
Discussion
To our knowledge, this is the first cross-sectional study comparing autonomic responses to APM, RSA-m, and walking, as well as submaximal exercise performance between subjects with COPD+OSAS versus subjects with COPD. The main findings of this study showed that individuals in the COPD+OSAS group presented less severe lung disease and shorter nocturnal hypoxemia time when compared to the COPD group.
In addition, the autonomic modulation during APM was impaired in both groups; however, the severity of COPD appears to lead to a greater parasympathetic modulation for the COPD group and the association of OSAS to a greater sympathetic activity for the COPD+OSAS group. During 6MWT, the groups presented similar performance impairments despite the greater severity of the COPD group; the time spent with saturation below 90% was associated with marked parasympathetic modulation during sub-maximal exercise.
Effects of autonomic tests in subjects with COPD and in the coexistence of COPD+OSAS
In our study, the COPD group presented higher parasympathetic modulation during APM when compared to the COPD+OSAS group. In contrast to findings from this study, some authors have reported reduced sympathetic and parasympathetic activity for individuals with COPD.12,13 By contrast, Volterrani et al14 reported an increase in parasympathetic tone for this population, which is in accordance with the results obtained in this study, where higher normalized HF values were found during supine rest and after postural change to the orthostatic position. Stein et al15 related the changes in the parasympathetic modulation of individuals with COPD to the severity of the disease, which also seems to happen in the present study, where autonomic alteration is present in the group with more severe pulmonary disease.
The COPD+OSAS subjects presented higher sympathetic modulation during APM when compared to the COPD group.16 Taranto-Montemurro et al5 observed increased sympathetic modulation in subjects with heart failure + OSAS during sleep. Our results are relevant, as we do not know any other study that has evaluated the coexistence of COPD+OSAS during postural change. Although marked sympathetic modulation was observed in both supine and orthostatic positions for the mixed group, none of the groups presented satisfactory postural adjustments (position effect: P > 0.05), demonstrating that postural adjustment is impaired in the presence of OSAS, even with COPD severity being lower for the COPD+OSAS group. Previous findings have shown that early neuropathy is common in both COPD and OSA subjects and is closely related to the disease severity.17 Previous findings showed that hypoxemia contributes to autonomic impairment in COPD patients.18 In our study, the COPD group presented longer desaturation time during sleep and this observation may help explain impaired postural adjustment in this group.
Effects of COPD and coexistence of COPD+OSAS during RSA-m and 6MWT
In the present study, only RSA-m and 6MWT produced autonomic responses in the subgroups, but without significant differences between the groups. We observed that in both groups RSA-m increased total HRV, as demonstrated by SDNN and TINN (P < 0.05). Previous findings from our group showed greater responses to the RSA-m in subjects with COPD and that altered responses were associated with greater COPD severity and poor diffusion capacity.19 Similarly, in subjects with OSAS, one study showed that the component of respiratory sinus arrhythmia that is mediated by pulmonary vagal feedback remained intact, indicating that subjects presented a response to RSA-m.20 Our findings showed that both the COPD group and the COPD+OSAS group presented an increase in total HRV; however, the indices representative of parasympathetic modulation did not change with the maneuver, suggesting possible mismatches of the coexistence of the diseases and the severity of COPD in front of maneuver.
When comparing the two groups, performance during 6MWT was similarly impaired, despite the greater COPD severity of the COPD group; this leads us to suggest that both the coexistence of OSAS and COPD and severe COPD can cause functional losses. In this context, it is known that the severity of COPD, as well as the severity of OSAS, is closely associated with impairments in functional capacity.21 In the present study, only SDNN and SD2 (both representative of total HRV) were sensitive to HRV reduction during exercise. As for the other indexes, we did not observe any differences during walking. Exercise was shown to increase sympathetic modulation and reduce parasympathetic modulation and global HRV.22 In subjects with COPD, physical training may improve exercise capacity, increase independence for activities of daily living, and improve cardiac autonomic control.23 Results showed that in subjects with OSAS, sympathetic hyperactivity contributes to impairment in autonomic response during exercise.24 However, we did not find any studies investigating the coexistence of COPD and OSAS. We believe, therefore, that both parasympathetic hyperactivity (present in COPD) and sympathetic hyperactivity (present in OSAS) contribute to impaired autonomic adjustment during exercise. These aspects are physiologically grounded because the integrity of the parasympathetic modulation is fundamental for fast HR adjustments (which occur in the first 30 seconds of exercise, as well as during the first minute of recovery) to allow for greater pulmonary blood flow at this early stage of exercise (cardiodynamic phase). At the same time, the increase of sympathetic modulation during exercise continuation is necessary to maintain cardiac work efficiency and meet exercise demands.
Therefore, the lack of responses during the autonomic tests applied in the present study may indicate great functional impairment. Both severity and coexistence of the diseases have a negative impact on cardiac autonomic control. These results are important, because they can lead to the development of important non-pharmacological rehabilitation strategies in these subjects with the objective to recover the integrity of the autonomic control or to minimize the negative impact of the diseases.25–27
Another interesting result of the present study was the strong correlation found between the time that subjects spent with saturation lower than 90% and the HRV indexes during the 6MWT (RMSSD and SD1, R = 0.80, P < 0.05), both representative of parasympathetic modulation. Our data reinforce that higher nocturnal hypoxemia is closely related to greater parasympathetic modulation during walking in both groups. During exercise, normal subjects are expected to present decreased parasympathetic modulation. It is known that cardiac sympathetic hyperactivity is related to airway vagal hyperactivity in subjects with COPD.14 Therefore, we believe that the marked desaturation that occurred in COPD subjects in our study was related to greater parasympathetic modulation during exercise. These findings suggest that the use of nocturnal oxygen therapy may be appropriate to help minimize cardiac autonomic maladjustments in these patients.28–30
Study limitations
The present study has some limitations. In relation to the subjects involved in the study, it would be important to have groups with a greater number of individuals, which was not possible due to our rigid exclusion criteria and the difficulty in finding subjects with the coexistence of the diseases. Therefore, future clinical trials with a larger number of individuals are needed to confirm the findings of the present study.
Conclusion
In conclusion, the results of this study indicate that subjects with coexistence of COPD and OSAS have marked sympathetic hyperactivity. In addition, the results suggest that regardless of the degree of lung involvement caused by COPD, the presence of coexistence leads to a greater functional impairment than in the presence of pathologies alone. Therefore, it is suggested that COPD+OSA leads to greater cardiovascular, autonomic, and functional deficiencies.
Acknowledgments
Katiany Thays Lopes Zangrando was supported by a research grant from Capes (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brasília, Brazil) and a research grant from CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico – Brasil) (Process Number: 163789/2015-0), and a research grant from FAPESP (Fundação de Amparo a Pesquisa do Estado de São Paulo, São Paulo, Brazil) (Process Number: 2015/26501-1). Luiz Carlos Soares de Carvalho Jr has a CNPq fellowship.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Andreas S, Anker SD, Scanlon PD, Somers VK. Neurohumoral activation as a link to systemic manifestations of chronic lung disease. Chest. 2005;128(5):3618–3624. doi: 10.1378/chest.128.5.3618. [DOI] [PubMed] [Google Scholar]
- 2.Park JG, Ramar K, Olson EJ. Updates on definition, consequences, and management of obstructive sleep apnea. Mayo Clin Proc. 2011;86(6):549–554. doi: 10.4065/mcp.2010.0810. quiz 554–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McNicholas WT, Bonsigore MR, Management Committee of EU COST ACTION B26 Sleep apnoea as an independent risk factor for cardiovascular disease: current evidence, basic mechanisms and research priorities. Eur Respir J. 2007;29(1):156–178. doi: 10.1183/09031936.00027406. [DOI] [PubMed] [Google Scholar]
- 4.Sociedade Brasileira de Pneumologia e Tisiologia II Consenso Brasileiro sobre Doença Pulmonar Obstrutiva Crônica – DPOC [II Brazilian consensus on chronic obstructive pulmonary disease – COPD] J Bras Pneumol. 2004;30(5):1–52. Portuguese [with English abstract] [Google Scholar]
- 5.Taranto-Montemurro L, Messineo L, Perger E, et al. Cardiac sympathetic hyperactivity in patients with chronic obstructive pulmonary disease and obstructive sleep apnea. COPD. 2016;13(6):706–711. doi: 10.1080/15412555.2016.1199668. [DOI] [PubMed] [Google Scholar]
- 6.Ioachimescu OC, Teodorescu M. Integrating the overlap of obstructive lung disease and obstructive sleep apnoea: OLDOSA syndrome. Respirology. 2013;18(3):421–431. doi: 10.1111/resp.12062. [DOI] [PubMed] [Google Scholar]
- 7.Global Initiative for Chronic Obstructive Lung Disease GOLD 2017; Global Strategy for Diagnosis, Management, and Prevention of COPD. [Accessed April 11, 2018]. Available from: htttp://goldcopd.org/gold-2017-global-strategy-diagnosis-management-prevention-copd/
- 8.Pereira CA. Testes de função pulmonar [Pulmonary function tests] Proj Diretrizes Assoc Médica Bras e Cons Fed Med. 2001:1–12. Portuguese [with English abstract] [Google Scholar]
- 9.Erman MK, Stewart D, Einhorn D, Gordon N, Casal E. Validation of the ApneaLink for the screening of sleep apnea: a novel and simple single-channel recording device. J Clin Sleep Med. 2007;3(4):387–392. [PMC free article] [PubMed] [Google Scholar]
- 10.Crapo RO, Enright PL, Zeballos RJ. (Writing Committee Members) ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166(1):111–117. doi: 10.1164/ajrccm.166.1.at1102. [DOI] [PubMed] [Google Scholar]
- 11.Vanderlei LCM, Pastre CM, Hoshi RA, Carvalho TD, Godoy MF. Noções básicas de variabilidade da frequência cardíaca e sua aplicabilidade clínica. [Basics of heart rate variability and its clinical applicability] Rev Bras Cir Cardiovasc. 2009;24(2):205–217. doi: 10.1590/s0102-76382009000200018. Portuguese. [DOI] [PubMed] [Google Scholar]
- 12.Bédard ME, Marquis K, Poirier P, Provencher S. Reduced heart rate variability in patients with chronic obstructive pulmonary disease independent of anticholinergic or β-agonist medications. COPD. 2010;7(6):391–397. doi: 10.3109/15412555.2010.528083. [DOI] [PubMed] [Google Scholar]
- 13.Pantoni CB, Reis MS, Martins LE, Catai AM, Costa D, Borghi-Silva A. Estudo da modulação autonômica da freqüência cardíaca em repouso de pacientes idosos com doença pulmonar obstrutiva crônica [Study of the autonomic modulation of resting heart rate in elderly patients with chronic obstructive pulmonary disease] Rev Bras Fisioter. 2007;11(1):35–41. Portuguese. [Google Scholar]
- 14.Volterrani M, Scalvini S, Mazzuero G, et al. Decreased heart rate variability in patients with chronic obstructive pulmonary disease. Chest. 1994;106(5):1432–1437. doi: 10.1378/chest.106.5.1432. [DOI] [PubMed] [Google Scholar]
- 15.Stein PK, Nelson P, Rottman JN, et al. Heart rate variability reflects severity of COPD in PiZ alpha1-antitrypsin deficiency. Chest. 1998;113(2):327–333. doi: 10.1378/chest.113.2.327. [DOI] [PubMed] [Google Scholar]
- 16.Chhabra SK, De S. Cardiovascular autonomic neuropathy in chronic obstructive pulmonary disease. Respir Med. 2005;99(1):126–133. doi: 10.1016/j.rmed.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 17.Resta O, Rana L, Procacci V, Guido P, Picca V, Scarpelli F. Autonomic dysfunction in normotensive awake subjects with obstructive sleep apnoea syndrome. Monaldi Arch Chest Dis. 1998;53(1):23–29. [PubMed] [Google Scholar]
- 18.Stewart AG, Waterhouse JC, Howard P. Cardiovascular autonomic nerve function in patients with hypoxaemic chronic obstructive pulmonary disease. Eur Respir J. 1991;4(10):1207–1214. [PubMed] [Google Scholar]
- 19.Mazzuco A, Medeiros WM, Sperling MP, et al. Relationship between linear and nonlinear dynamics of heart rate and impairment of lung function in COPD patients. Int J Chron Obstruct Pulmon Dis. 2015;10:1651–1661. doi: 10.2147/COPD.S81736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jo JA, Blasi A, Valladares E, Juarez R, Baydur A, Khoo MC. Determinants of heart rate variability in obstructive sleep apnea syndrome during wakefulness and sleep. Am J Physiol Heart Circ Physiol. 2005;288(3):H1103–H1112. doi: 10.1152/ajpheart.01065.2003. [DOI] [PubMed] [Google Scholar]
- 21.Cholidou KG, Manali ED, Kapsimalis F, et al. Heart rate recovery post 6-minute walking test in obstructive sleep apnea: cycle ergometry versus 6-minute walking test in OSA patients. Clin Res Cardiol. 2014;103(10):805–815. doi: 10.1007/s00392-014-0721-3. [DOI] [PubMed] [Google Scholar]
- 22.Roque AL, Valenti VE, Massetti T, et al. Chronic obstructive pulmonary disease and heart rate variability: a literature update. Int Arch Med. 2014;7:43. doi: 10.1186/1755-7682-7-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Borghi-Silva A, Mendes RG, Trimer R, et al. Potential effect of 6 versus 12-weeks of physical training on cardiac autonomic function and exercise capacity in chronic obstructive pulmonary disease. Eur J Phys Rehabil Med. 2015;51(2):211–221. [PubMed] [Google Scholar]
- 24.Cepeda FX, Toschi-Dias E, Maki-Nunes C, et al. Obstructive sleep apnea impairs postexercise sympathovagal balance in patients with metabolic syndrome. Sleep. 2015;38(7):1059–1066. doi: 10.5665/sleep.4812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Camillo CA, Laburu Vde M, Gonçalves NS, et al. Improvement of heart rate variability after exercise training and its predictors in COPD. Respir Med. 2011;105(7):1054–1062. doi: 10.1016/j.rmed.2011.01.014. [DOI] [PubMed] [Google Scholar]
- 26.Borghi-Silva A, Reis MS, Mendes RG, et al. Noninvasive ventilation acutely modifies heart rate variability in chronic obstructive pulmonary disease patients. Respir Med. 2008;102(8):1117–1123. doi: 10.1016/j.rmed.2008.03.016. [DOI] [PubMed] [Google Scholar]
- 27.Sin DD, Wong E, Mayers I, et al. Effects of nocturnal noninvasive mechanical ventilation on heart rate variability of patients with advanced COPD. Chest. 2007;131(1):156–163. doi: 10.1378/chest.06-1423. [DOI] [PubMed] [Google Scholar]
- 28.Gong X, Huang L, Liu X, et al. Correlation analysis between polysomnography diagnostic indices and heart rate variability parameters among patients with obstructive sleep apnea hypopnea syndrome. PLoS One. 2016;11(6):e0156628. doi: 10.1371/journal.pone.0156628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Aeschbacher S, Bossard M, Schoen T, et al. Heart rate variability and sleep-related breathing disorders in the general population. Am J Cardiol. 2016;118(6):912–917. doi: 10.1016/j.amjcard.2016.06.032. [DOI] [PubMed] [Google Scholar]
- 30.Lewis MJ, Annandale J, Lewis KE. Influence of long-term oxygen therapy on heart rate and QT time-series in hypoxic patients with chronic obstructive pulmonary disease. Clin Physiol Funct Imaging. 2009;29(6):431–439. doi: 10.1111/j.1475-097X.2009.00891.x. [DOI] [PubMed] [Google Scholar]