Abstract
Weight regain after weight loss is one of the most significant challenges to successful obesity treatment. Regular exercise has long been touted as a strategy for weight loss maintenance, but the lack of clear evidence in clinical trials has caused some to question its effectiveness. In this review, we present the arguments both questioning and in support of exercise as an obesity therapeutic. Our purpose is to bring clarity to the literature, present a unified perspective, and identify the gaps in knowledge that need to be addressed in future studies. Critical questions remain including sex differences, individual variability and compensatory behaviors in response to exercise, exercise adherence, the role of energy flux and the molecular mechanisms mediating the beneficial effects of exercise after weight loss and during weight regain. Future research should focus on these critical questions to provide a more complete understanding of the potential benefits of exercise on weight loss maintenance.
Keywords: Obesity, Physical activity, Individual variability, Weight regain
1. Introduction
Obesity is a global health problem that affects individuals of every age, sex, ethnicity, race, and socioeconomic status [1]. As little as a 5% reduction in body weight can achieve clinically significant improvements in metabolic health markers, while decreasing health risks and disease comorbidities [2]. However, it is clear that current treatments for obesity are, for the most part, only transiently effective; less than one-fifth of individuals who achieve a 10% reduction in body weight successfully maintain that weight loss after one year [3]. These high recidivism rates have lead the National Institues of Health (NIH) to organize a working group, comprised of both basic and clinical scientists, to address the issues surrounding weight regain [4].
The NIH working group identified two primary reasons to explain the high recidivism rates [4]. First, weight loss achieved by dieting induces a number of physiological and behavioral adaptations that collectively result in an elevation of appetite and a suppression of energy expenditure [5,6]. This mismatch between appetite and metabolic requirements, termed the energy gap (see Fig. 1) [7], establishes a strong and persistent biological pressure to regain lost weight. Second, the behavioral and pharmaceutical strategies used to induce weight loss are commonly employed in a transient manner and adherence to these strategies generally wanes as weight loss plateaus [4]. These observations have reframed the perspective on obesity and its treatment strategies demonstrating that obesity is a chronic and complex condition that requires a comprehensive and sustained approach in clinical care. To address the obesity problem, researchers and clinicians must apply this perspective to the development and implementation of treatments for more effective long-term clinical outcomes.
Fig. 1.
The energy gap
The energy gap is represented with data from MacLean et al. Am J Physiol Regul Integr Comp Physiol, 2009. 297(3): p. R793–802 [11]. Energy intake and expenditure are displayed for obese-prone, male, Wistar rats under three different conditions. First, in the obese state following 16 weeks of ad libitum feeding on a high fat diet (Research Diets Inc. D12344). Second, after calorie-restricted weight loss on a low fat diet (Research Diets Inc. D11724) to yield a 10–15% body weight reduction. Third, upon ad libitum refeeding on a low fat diet during the first day of relapse. The energy gap, defined by the mismatch between appetite and energy requirements following weight loss, is apparent during this ad libitum refeeding phase. This mismatch between the calories desired and calories required establishes a strong and persistent biological pressure to regain lost weight.
Regular exercise has emerged as a strategy with great potential to improve outcomes in obesity treatment [8–11]. The benefits of exercise for general health are clear and unquestioned [12]. Regular exercise lowers all-cause mortality and can prevent the onset of obesity, type II diabetes, hypertension, and cardiovascular disease [13]. However, the clinical effectiveness of physical activity as a weight loss maintenance strategy, in the form of regimented exercise or increased activity of daily living, has been the subject of much debate [14].
In this review, we present the arguments on both sides of this debate. Our purpose is to bring clarity to the published literature, present a more unified perspective, and identify gaps in our knowledge that need to be addressed in future studies.
2. Arguments questioning exercise as a weight loss maintenance strategy
Four main arguments have emerged that question exercise as a successful weight loss maintenance (WLM) strategy, namely: (1) the lack of evidence from randomized controlled trials (RCTs); (2) behavioral compensation in response to exercise; (3) inter-individual variability in response to exercise; and (4) “it doesn’t matter, they won’t do it”.
2.1. Lack of evidence from RCTs
RCTs are the gold standard in determining the efficacy of any strategy in obesity therapeutics. The most compelling argument that discounts regular exercise as an effective WLM strategy is the lack of evidence from such trials. Although not all [15,16], the vast majority of RCTs that examine weight regain or WLM as the primary outcome fail to show a beneficial effect of exercise (Table 1 – Column 1) [17–24]. Despite the numerous health benefits that can be realized via physical activity, the failure of this intervention to meet the gold standard of RCT evidence has cast doubt on the usefulness of exercise in long-term weight control.
Table 1.
Results of Randomized Control Trials analyzed by randomized groups and by self- reported physical activity.
| Benefit of exercise on weight loss maintenance? (by randomized groups) | Exercise compliance at follow-up (participants completing study) | Retrospective analysis? | Association between actual exercise and weight loss maintenance? | ||||
|---|---|---|---|---|---|---|---|
|
|
|
|
|||||
| YES | NO | < 70% | < 50% | No difference between groups (final follow-up) | YES | YES | NO |
| Jeffery et al. [15] | Borg et al. [17] | Pavlou et al. [16] | Borg et al. [17] | Fogelholm et al. [18] | Borg et al. [17] | Borg et al. [17] | |
| Pavlou et al. [16] | Fogelholm et al. [18] | Jeffery et al. [15] | Jakicic et al. [19] | Jakicic et al. [50] | Jakicic et al. [50] | ||
| Jakicic et al. [19] | Perri et al. [21] | Leermakers et al. [20] | Jakicic et al. [19] | Jakicic et al. [19] | |||
| Leermakers et al. [20] | Skender et al. [22] | Tate et al. [23] | Pavlou et al. [16] | Pavlou et al. [16] | |||
| Perri et al. [21] | Wing et al. [24] | Tate et al. [23] | Tate et al. [23] | ||||
| Skender et al. [22] | Wadden et al. [51] | Wadden et al. [51] | |||||
| Tate et al. [23] | |||||||
| Wing et al. [24] | |||||||
2.2. Biological benefits of exercise can be masked by compensatory behaviors
Practically speaking, if everything else remains the same, exercise should make WLM easier by acutely increasing energy expenditure. However, increasing energy expenditure through a discrete exercise bout can result in compensatory behaviors that negate the exercise-induced negative energy balance [25–30]. These behaviors may be performed consciously or unconsciously and include decreases in non-exercise physical activity [27] and/or increases in energy intake (for a review see [30]). The magnitude of this compensation will vary between individuals and can summate to minimize the exercise-induced negative energy balance or, in some cases, even result in a positive energy balance. It remains unclear which psychosocial factors predict whether an individual will compensate during an exercise intervention although there is some evidence that disinhibition [31,32], hunger [31,32], dietary status [31,32] and binge eating [33] may confer a higher risk. In individuals susceptible to behavioral compensation, exercise may not improve weight loss maintenance unless it can be implemented in a manner that minimizes or eliminates these compensatory behaviors [34].
2.3. Inter-individual variability in the response to exercise
Clinical exercise studies report a broad range of responses in the measurement of appetite, energy expenditure, and energy balance [15–24]. This inconsistency in response has led many researchers to classify research participants as exercise “responders” or “non-responders” [35,36], with the intention of studying their distinguishing characteristics. Even so, the etiology of this inter-individual variability is poorly understood. It is likely very complex, with origins in the genetic or epigenetic predisposition, spontaneous compensatory behaviors, or other environmental, socioeconomic and psychological factors that affect eating and physical activity behaviors. Additionally, an individual’s metabolism, which is influenced by age, sex, adiposity, insulin sensitivity, intrinsic aerobic capacity, or maximal fat oxidation during exercise may also moderate how physical activity impacts the homeostatic and hedonic adaptations driving weight regain [4,30]. The individual variability in response is not unique to exercise interventions, but is observed in all types of obesity treatments [34,37,38]. Blanket statements suggesting exercise will improve the maintenance of lost weight in all individuals must be tempered because variability in response to exercise interventions will ultimately influence whether exercise is beneficial for an individual.
2.4. It Doesn’t matter; they Won’t do it
The clear beneficial effects of exercise on general health have led many agencies across the world, including the World Health Organization (WHO), to develop guidelines for physical activity [39]. The WHO guidelines recommend at least 150 min of moderate-intensity aerobic physical activity or 75 min of vigorous intensity physical activity per week. For additional benefits such as the maintenance of body weight, the amount of recommended physical activity is doubled [39]. Growing rates of obesity worldwide would suggest that either the guidelines are set at an insufficient level for the maintenance of body weight, individuals are not receiving a benefit due to compensatory behaviors, or that the public is not attaining the prescribed levels of physical activity. Results of the National Health and Nutrition Examination Survey (NHANES) suggest the latter. Sixty-five percent of the NHANES population self-reported meeting recommendations for physical activity; however, objective measurements of physical activity using accelerometry devices demonstrated that only 5% were achieving recommended levels of activity [40]. These results demonstrate that the public overall is not achieving the levels of physical activity recommended for the maintenance of body weight. This is mirrored in clinical exercise studies of WLM, where compliance is relatively poor even when substantial behavioral, psychological, environmental, and financial support is provided (see Table 1 – Column 2) [15–24]. Furthermore, approximately 50% of adults who initiate an exercise program drop out within the first 6 to 12 months [41]. Unless the barriers to exercise compliance and adherence in weight-reduced individuals are addressed, exercise will fail as an effective strategy for WLM.
3. Arguments supporting exercise as a WLM strategy
Despite the arguments that have been levied against the effective-ness of exercise for weight control, there remains a considerable body of evidence that supports the utility of physical activity for WLM programs. This evidence stems primarily from observational and correlative clinical studies, along with preclinical studies which provide additional supportive data based on the biological responses of exercise during weight loss and weight regain.
3.1. Observational evidence
The National Weight Control Registry (NWCR) is the largest prospective investigation of long-term successful WLM [8,9,42]. Included in this registry are the behavioral and psychological characteristics of > 10,000 people who have lost at least 30 pounds (~13.6 kg) and have kept the weight off for at least one year. Studies from the registry have identified a program of regular exercise as a key characteristic of those who have been successful with WLM [8,9,42]. Most NWCR subjects report physical activity levels corresponding to one hour of moderate-intensity physical activity per day [9]. This exceeds both levels of the WHO physical activity guidelines and suggests that perhaps the WHO recommendations are insufficient for WLM. Population studies on long-term WLM corroborate the findings of the NWCR. These studies demonstrate a positive correlation between the amount of exercise completed and the percentage of maintained weight loss [43–49]. Although these studies cannot definitively show that programed exercise is responsible for WLM, they do suggest that success is unlikely, unless regular exercise is incorporated into a WLM strategy.
3.2. Re-evaluation of RCTs
RCTs are plagued by issues of adherence. The lack of adherence yields considerable variation in the amount of exercise actually performed (Table 1 – Column 2), to the extent that, in many studies, intention-to-treat analyses fail to detect a benefit of exercise on WLM (Table 1 – Column 1) [17–24]. When the same RCTs are re-analyzed based on the self-reported amount of performed exercise, a relationship between exercise and WLM readily emerges (Table 1- Columns 3 & 4) [16,17,19,23,50,51]. This may be due to the RCT design, which randomly assigns participants without taking into account their proclivity to exercise regularly. As a result, participants that may have no desire or even an aversion to regular exercise may be assigned to an exercise arm of a trial. Conversely, those who are more inclined or highly motivated to exercise may be assigned to a sedentary arm of a trial. Given that adherence to exercise prescriptions continues to influence the results of RCTs, acquiring definitive proof of a beneficial effect of exercise on WLM, in this type of research paradigm, may prove to be cost prohibitive.
3.3. Countering the biological drive to regain weight
Arguably, one of the most significant barriers to successful WLM is the underlying physiological adaptations that occur in response to diet-induced weight loss [11,52]. A compilation of animal and human studies provides clear evidence that these adaptations occur in a number of key regulatory nodes in peripheral tissues and neural circuits in the brain (Reviewed in [6]; summarized in Fig. 2). The overarching effect of these adaptations is the creation of an energy gap, characterized by a mismatch between appetite and energy requirements (Fig. 1). There may also be additional neuroendocrine adaptations that enhance the rewarding aspects of food [53,54] and reduce the desire to be physically active [11,55–57], which further exacerbates the mismatch between appetite and energy expenditure. Collectively, these adaptations provide a physiological defense against further weight loss and increase the biological drive to regain lost weight. The majority of these adaptive responses do not appear to resolve once weight has stabilized at the reduced body weight [6]. In fact, data from our preclinical models would suggest the converse, that the collection of adaptive responses strengthen as time in the weight-reduced state increases [58].
Fig. 2.
Impact of Weight Loss and Exercise on the Energy Gap
The energy gap is a term used to describe the mismatch between appetite and energy requirements, expressed in energy equivalents. There are numerous adaptations to weight loss that culminate in an elevated appetite and suppressed energy expenditure, which drive weight regain. Some of the adaptations contributing to the energy gap are displayed in the left panel including: changes in hypothalamic neuropeptide expression and decreases in sympathetic nervous system (SNS) tone, resting energy expenditure, oxidative capacity, insulin, leptin, and circulating nutrients. Exercise counters many of the adaptations to weight loss and can attenuate the energy gap in a tissue specific manner including: increases in SNS tone, hepatic de novo lipogenic capacity, skeletal muscle dietary fat oxidation, circulating glucose and free fatty acids. An exercise signal has yet to determined, however, given the complexities of the exercise response it is unlikely we will find a single hormone or factor that is responsible for the effects of exercise on the energy gap.
Abbreviations: neuropeptide Y; NPY, pro-opiomelanocortin; POMC, sympathetic nervous system; SNS, resting energy expenditure; REE, gastrointestinal; GI, free fatty acids; FFA, triglycerides; TG, de novo lipogenesis DNL, calcium; Ca, brain-derived neurotrophic factor; BDNF, interleukin-6; IL-6.
Evidence derived primarily from preclinical models of refeeding and weight regain after energy restriction indicates that exercise counters numerous aspects of this adaptive response (Fig. 2). These responses to exercise culminate in a reduced drive to overfeed [59,60]. Exercise also increases energy expenditure beyond the cost of the exercise bout itself [60]. We have linked this increase in energy expenditure to changes in peripheral tissues (skeletal muscle [60] and adipose [61]) that together promote the preferential trafficking of dietary fat toward oxidative pathways rather than to storage depots. In relapsing exercised rats, excess nutrients are preferentially deposited via de novo lipogenesis. This makes the deposition of excess nutrients more expensive and results in an increased thermic effect of food [60]. Although the underlying mechanisms responsible for the attenuated energy gap are not fully understood, the impact of exercise on appetite in relapsing animals may involve changes in leptin and insulin sensitivity [62,63] or alterations to the concentrations of gut-released satiety signals [64]. Together, these effects of exercise counter the physiological response to weight loss by attenuating appetite, increasing expended energy, and making weight regain more energetically expensive.
4. A practical interpretation of the literature
4.1. Subtle, but distinct perspectives about exercise and WLM
When translating research findings into a message for the public, the manner in which the message is conveyed can have significant implications. Consider two statements that could be conveyed to the lay public:
Statement 1. Regular exercise is an effective strategy for keeping weight off.
Statement 2. Those who can maintain a program of regular exercise are more likely to keep weight off.
The first statement appears to step beyond the existing scientific evidence, as RCTs fail to provide definitive answers regarding the effectiveness of exercise for WLM. However, there is considerable correlative data, retrospective analyses, and evidence from preclinical models of weight regain that support this statement. Advocates who convey this message do so based on the collective strength of this supportive evidence.
The second statement conveys the state of the evidence in humans, based upon the traditional standards for establishing effectiveness. It does not preclude that exercise might impart a significant beneficial effect on WLM, but acknowledges that we simply do not have the evidence from clinical trials to establish causality. Beyond maintaining the standards for determining causality, there may be some value in holding to this distinction.
In this review, our objective is not to judge which of these perspectives is correct, but rather to distinguish between the two, clarify the arguments, and place them in the context of the evidence that supports them. We conclude that, in the extreme, both perspectives fail to acknowledge the strength of the evidence or the limitations of our science. Even so, these perspectives are not drastically different in their fundamental understanding of the issues or in the message conveyed to the lay public. With this debate occurring in the shadows of the overarching guidance that society should exercise and increase physical activity for the broader health benefits, one might conclude that it is hardly worth mentioning. What we would argue is that this debate spotlights a significant gap in knowledge – why do some people, particularly those with obesity or who have lost excess weight, fail to sustain their exercise program, when the potential benefits seem so apparent?
5. Moving forward to advance the science
5.1. An emerging theme: Lack of adherence
The inability to implement or sustain a regular exercise program emerges as a central theme in this discussion of exercise and WLM. It underlies the inability to acquire definitive data from RCTs. Noncompliance likely stems from psychological, environmental, and biological explanations that we currently do not understand. As such, an interdisciplinary approach will be necessary to elucidate what motivates an individual to be physically active, the metabolic or psychological barriers to adherence, and the environmental conditions that can facilitate a physically active lifestyle.
5.2. Impact of exercise on energy flux
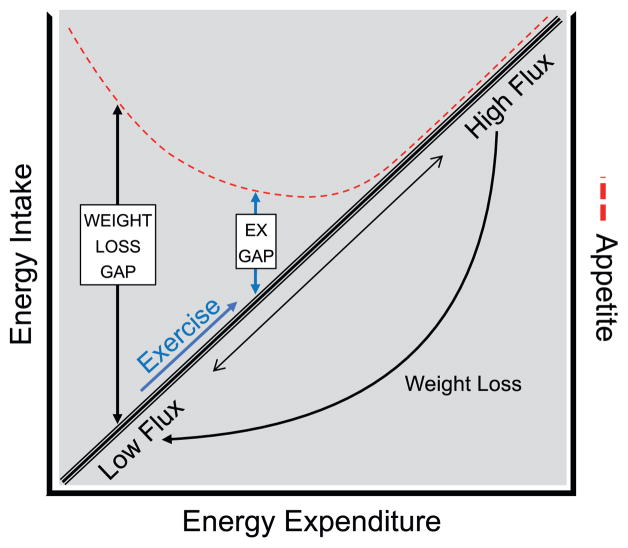
Mayer first observed in rodents [65] and later in humans [66] the relationship between energy intake, daily physical activity and the regulation of body weight. In 1956, he examined workers in West Bengal and observed that as daily physical activity decreased as a function of occupation, daily intake also decreased [66]. This led to the matching of energy intake and expenditure so that energy balance was maintained. This, however, was only effective to a certain point. Below a certain level of physical activity, sedentary workers failed to adjust their intake to match their very low level of daily expenditure. The mismatch between intake and expenditure resulted in a positive energy balance and weight gain. His findings suggest that daily physical activity, above a certain threshold of energy flux (turnover), allows an individual to better couple energy intake with energy expenditure. Placing this concept in the context of WLM (see Fig. 3), the low energy flux from the restriction on intake and the suppression of energy expenditure would be expected to fall well below this threshold. However, increasing levels of physical activity through regimented exercise and/or daily living physical activity could raise energy flux and improve weight loss maintenance by allowing for a better matching of appetite with energy requirements [67]. A recent pilot study tested this hypothesis, the authors took individuals with obesity through calorie-restricted weight loss to produce a 7% reduction in body weight and maintained that weight loss for 3 weeks prior to entering the experimental conditions [67]. In both 5-day low and high flux conditions, participants were carefully kept in energy balance; body weight and resting energy expenditure were measured daily and all food was provided for the participants. Low flux was characterized by low physical activity equivalent to a sedentary physical activity level of 1.35. High flux was achieved by a daily, supervised exercise bout (~500 kcal deficit) and through an increased daily step count. This study found that even while maintaining energy balance, increasing energy flux over 5 days decreased hunger, increased satiety and increased resting metabolic rate. These results suggest that high energy flux can attenuate both aspects (hunger and expenditure) of the energy gap produced during weight loss. Without environmental pressures that demand greater levels of physical activity or proactive behavioral changes to become more physically active, the biological response to weight loss becomes the default, and the result is weight regain.
Fig. 3.
The role of energy flux on appetite
Energy balance (black diagonal line) can be achieved at various levels of energy flux (turnover) so long as energy intake is match to energy expenditure. Weight loss maintenance following calorie restriction is often characterized by a low flux state (low energy intake and low energy expenditure). In this low flux state, there is a large mismatch between appetite (red dashed line) and energy requirements. This large energy gap commonly results in overfeeding and weight regain. Exercise can be used to shift a weight-reduced individual into a higher flux state. The high flux state increases energy expenditure and decreases appetite to minimizes the energy gap [67]. This closer matching of appetite with energy requierments can minimized the drive to regain lost weight and improve weight loss maintenance. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
While Mayer’s data are still employed today to conceptualize the benefit of exercise for body weight regulation, we have yet to understand the underlying mechanisms and complex interactions between the regulatory systems that coordinately influence appetite and physical activity. This is particularly true in the context of WLM. Fig. 2 provides an initial glimpse of what we have learned about these mechanisms in recent years. The complexity suggests that we are unlikely to find one hormonal signal, individual neural center, or single tissue factor that is responsible for the effect of exercise in total. An approach that integrates perspectives from different disciplines with well-designed in vitro and preclinical studies will undoubtedly be needed to gain a deeper understanding of the underlying molecular mechanisms and proactively pursue their translation and application to the human condition.
5.3. Individual variability with exercise and WLM
Individual variability must be expected in the ability and motivation to maintain an exercise program, in the response to exercise, and in the molecular and psychological mechanisms that underlie the impact of exercise [36]. The variability likely stems from the wide range of environmental conditions imposed and the genetic and epigenetic differences that influence the psychological and biological responses to weight loss. Variability in psychosocial factors and other behaviors such as dietary restraint [42], diet composition [42], and self-weighing frequency [68] may also affect individual weight loss maintenance success with exercise. This individual variability must be acknowledged as we pursue studies of WLM, and it is critical that we specifically design studies to understand this variability in order to leverage it for better outcomes.
5.4. Potential sex differences in the effects of exercise
One component of this individual variability may be rooted in the sex-specific differences of exercise in countering the biological drive to regain weight. We employ a preclinical model of weight loss and regain in which Wistar rats are fed a high fat diet (Research Diets Inc. D D12344) for 16 weeks prior to undergoing calorie restricted weight loss on a lower fat diet with or without forced treadmill exercise (15 m/min, 1 h/day, 6 days/week, equivalent to a 1-h slow jog in humans). While continuing the exercise protocol, we maintain this weight loss (10–15% of body weight) for 8 weeks then allow the rats to refeed ad libitum. We find that a regimen of regular exercise counters the biological drive to regain weight during relapse in the male rats (Fig. 4A) [11]. The exercising male rats successfully maintained a portion (~20%) of the original weight loss and defended a lower body weight and fat mass than the sedentary controls. We have seen this response in males on both a low [11] and high [59] fat diet as have others [69] including classical animal studies carried out by Mayer, et al. in which normal weight male rats decreased food intake and body weight when exercising 20, 40 or 60 min [65]. We recently put female rats through the same experimental paradigm and, in line with the work from Nance et al. [69], found that the same exercise intervention did not similarly attenuate female weight regain (Fig. 4B). Both exercising and sedentary females regained about 140% of their original body weight (Fig. 4B) and had similar fat mass percentages during weight regain.
Fig. 4.
Sex differences of exercise on weight regain
Female body weight in the weight-reduced state and during the early stages of weight regain (B) are compared to data from males (A) that are published in MacLean et al. Am J Physiol Regul Integr Comp Physiol, 2009. 297(3): p. R793–802 [11]. In both cases, obese-prone, Wistar rats were fed a high fat diet (Research Diets Inc. D12344) for 16 weeks prior to calorie-restricted weight loss on a low (males) or medium (females) fat diet (Research Diets Inc. D11724 & D07091301, respectively). Following a 15% reduction in body weight, rats were maintained at the lower body weight for 8 weeks to minimize the acute response to weight loss. At the end of 8 weeks, rats were allowed to refeed ad libitum for 8 weeks. Rats were randomized to forced treadmill exercise (EX) training (15 m/min, 1 h/day, 6 days/week) or sedentary conditions (SED) at the time of weight loss and continued EX or SED conditions for the remainder of the study. The red dashed line denotes the body weight of the males and females in the obese state (prior to exercise and weight loss). These studies were approved and carried out in accordance with the institutional review committee.
*indicates significant (p < 0.05) differences between EX and SED groups. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
When we examined the energy gap of weight reduced male and female rats, as they transitioned to ad libitum feeding, the sex-specific difference in the effects of exercise on appetite and expenditure become evident (Fig. 5A & B). In males, regular exercise reduced the magnitude of overfeeding, by attenuating food intake and increasing expended energy (Fig. 5A). The impact on energy expenditure was revealed when overfeeding in sedentary animals (Sedentary Gap Matched) was titrated down to that of the exercised group. Despite the same positive energy balance, these rats expended ~10 kcal less than those that exercised [60]. In contrast, exercise had little effect on female rats in the same experimental paradigm (Fig. 5B).
Fig. 5.
Sex Differences in the Energy Gap in Response to Exercise
The energy gap in respose to exercise in females is compared to data from males published in Steig et al. Am J Physiol Regul Integr Comp Physiol, 2011. 301(3): p. R656–67 [60]. Energy intake and expenditure in male (A) and female (B) Wistar rats in the obese state after 16 weeks of high fat diet (Research Diets Inc. D12344) feeding and during the first day of ad libitum refeeding on a medium fat diet (Research Diets Inc. D07091301). Refeeding took place following calorie restricted weight loss to achieve a 10–15% reduction in body weight and 6–8 weeks of weight loss maintenance. At the time of weight loss rats were randomized to forced treadmill exercise (EX) training (15 m/min, 1 h/day, 6 days/week) or sedentary conditions (SED). The energy gap identifies the difference between appetite (energy intake) and energy requirements (energy expenditure) during the first day of refeeding. A third group of sedentary males (sedentary gap matched) did not refeed ad libitum but were provided calories sufficient to achieve an equilavent positive energy balance to that of the exercised males. These studies were approved and carried out in accordance with the institutional review committee.
Abbreviations: EE; Energy Expenditure.
Inconsistencies in the clinical literature, containing both male and female participants, allude to the possibility that our findings may translate to the human condition; however, many of these clinical studies are unpowered to detect sex-differences in response to an exercise intervention. Although not in the weight-reduced state, one very well-controlled, randomized, clinical study by Donnelly et al. was powered to detect the differences between men and women (ages 17–35) in response to an exercise intervention [37]. Over the course of the 16-month, supervised intervention, exercising male participants lost an average of 5.2 kg compared to sedentary controls but exercising female participants displayed no change in body weight on average [37]. In addition to differences in the average weight loss between males and females, there was a striking difference in the individual variability in response to the exercise intervention. The change in body weight in the male participants ranged from −14 kg to +5 kg while the change in female body weight ranged from −5 kg to +11 kg [34]. This suggests that the women, much like our female rats, were more likely to compensate for the cost of the exercise bout by either increasing caloric intake and/or decreasing non-exercise physical activity. It is interesting to speculate that distinct evolutionary pressures of reproduction between males and females [70] have resulted in divergent feeding responses to exercise. Either way, important clinical implications emerge if these sex differences broadly translate to humans.
5.5. Unanswered questions
There are many additional questions that need to be addressed so that we can fully understand the utility of regular exercise for WLM. Are there sex differences in the biological, psychological or behavioral responses to exercise? How does aging affect these responses throughout the lifespan? How does changing the frequency, duration, intensity or type of structured exercise influence the response? What are the differences between the benefits of an exercise regimen and those that come via altering daily activities? Does increasing energy flux improve WLM and if so, by what mechanism? Why do some individuals enjoy exercise while others have an aversion to it? Finally, and likely most importantly, is there a way to increase the rewarding value of the exercise to render exercise more appealing to people after they have lost weight? Expanding our understanding of the mechanisms and answering these questions will be critical if we are to leverage this valuable strategy to better outcomes in obesity treatments.
6. Summary
Exercise is likely to be a very effective strategy for WLM for those individuals who are able to implement and maintain a regular regimen. Although the limitations of our science may prevent us from acquiring the critical pieces of data necessary for establishing causality, there is a substantial amount of supporting evidence from both animal and human studies that demonstrate the benefits of exercise for WLM if compensatory behaviors can be avoided. There are critical questions around the issues of adherence, the molecular mechanisms driving regain, and the individual variability that need to be answered, specifically in the context of WLM, if we are to leverage this powerful tool effectively in obesity therapeutics.
Acknowledgments
Funding
This work was supported by: F31 DK115238 (RMF), TL1 TR001081 (RMF, DMP), P50 HD073063 (PSM), R01 CA164166 (PSM), P30 48520 (JOH, PSM), T32 DK007658 (VDS), K99/R00 CA169430 (EDG), K99/R00 DK100465 (AB)
We thank Ginger C. Johnson and Julie A. Houck for their assistance in carrying out the preclinical studies.
References
- 1.Ogden CL, et al. Prevalence of childhood and adult obesity in the United States, 2011–2012. JAMA. 2014;311(8):806–814. doi: 10.1001/jama.2014.732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Magkos F, et al. Effects of moderate and subsequent progressive weight loss on metabolic function and adipose tissue biology in humans with obesity. Cell Metab. 2016;23(4):591–601. doi: 10.1016/j.cmet.2016.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kraschnewski JL, et al. Long-term weight loss maintenance in the United States. Int J Obes. 2010;34(11):1644–1654. doi: 10.1038/ijo.2010.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.MacLean PS, et al. NIH working group report: innovative research to improve maintenance of weight loss. Obesity (Silver Spring) 2015;23(1):7–15. doi: 10.1002/oby.20967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rosenbaum M, et al. Energy intake in weight-reduced humans. Brain Res. 2010;1350:95–102. doi: 10.1016/j.brainres.2010.05.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Maclean PS, et al. Biology’s response to dieting: the impetus for weight regain. Am J Phys Regul Integr Comp Phys. 2011;301(3):R581–600. doi: 10.1152/ajpregu.00755.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hill JO, et al. Obesity and the environment: where do we go from here? Science. 2003;299(5608):853–855. doi: 10.1126/science.1079857. [DOI] [PubMed] [Google Scholar]
- 8.Catenacci VA, et al. Physical activity patterns using accelerometry in the National Weight Control Registry. Obesity (Silver Spring) 2011;19(6):1163–1170. doi: 10.1038/oby.2010.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Catenacci VA, et al. Physical activity patterns in the National Weight Control Registry. Obesity (Silver Spring) 2008;16(1):153–161. doi: 10.1038/oby.2007.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Donnelly JE, et al. The role of exercise for weight loss and maintenance. Best Pract Res Clin Gastroenterol. 2004;18(6):1009–1029. doi: 10.1016/j.bpg.2004.06.022. [DOI] [PubMed] [Google Scholar]
- 11.MacLean PS, et al. Regular exercise attenuates the metabolic drive to regain weight after long-term weight loss. Am J Phys Regul Integr Comp Phys. 2009;297(3):R793–802. doi: 10.1152/ajpregu.00192.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garber CE, et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43(7):1334–1359. doi: 10.1249/MSS.0b013e318213fefb. [DOI] [PubMed] [Google Scholar]
- 13.King NA, et al. Beneficial effects of exercise: shifting the focus from body weight to other markers of health. Br J Sports Med. 2009;43(12):924–927. doi: 10.1136/bjsm.2009.065557. [DOI] [PubMed] [Google Scholar]
- 14.Doucet E, et al. Update on exercise and weight control. J Obes. 2011;2011:358205. doi: 10.1155/2011/358205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jeffery RW, et al. Physical activity and weight loss: does prescribing higher physical activity goals improve outcome? Am J Clin Nutr. 2003;78(4):684–689. doi: 10.1093/ajcn/78.4.684. [DOI] [PubMed] [Google Scholar]
- 16.Pavlou KN, Krey S, Steffee WP. Exercise as an adjunct to weight loss and maintenance in moderately obese subjects. Am J Clin Nutr. 1989;49(Suppl 5):1115–1123. doi: 10.1093/ajcn/49.5.1115. [DOI] [PubMed] [Google Scholar]
- 17.Borg P, et al. Effects of walking or resistance training on weight loss maintenance in obese, middle-aged men: a randomized trial. Int J Obes Relat Metab Disord. 2002;26(5):676–683. doi: 10.1038/sj.ijo.0801962. [DOI] [PubMed] [Google Scholar]
- 18.Fogelholm M, et al. Effects of walking training on weight maintenance after a very-low-energy diet in premenopausal obese women: a randomized controlled trial. Arch Intern Med. 2000;160(14):2177–2184. doi: 10.1001/archinte.160.14.2177. [DOI] [PubMed] [Google Scholar]
- 19.Jakicic JM, et al. Effect of exercise on 24-month weight loss maintenance in overweight women. Arch Intern Med. 2008;168(14):1550–1559. doi: 10.1001/archinte.168.14.1550. (discussion 1559–60) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Leermakers EA, et al. Effects of exercise-focused versus weight-focused maintenance programs on the management of obesity. Addict Behav. 1999;24(2):219–227. doi: 10.1016/s0306-4603(98)00090-2. [DOI] [PubMed] [Google Scholar]
- 21.Perri MG, et al. Enhancing the efficacy of behavior therapy for obesity: effects of aerobic exercise and a multicomponent maintenance program. J Consult Clin Psychol. 1986;54(5):670–675. doi: 10.1037//0022-006x.54.5.670. [DOI] [PubMed] [Google Scholar]
- 22.Skender ML, et al. Comparison of 2-year weight loss trends in behavioral treatments of obesity: diet, exercise, and combination interventions. J Am Diet Assoc. 1996;96(4):342–346. doi: 10.1016/S0002-8223(96)00096-X. [DOI] [PubMed] [Google Scholar]
- 23.Tate DF, et al. Long-term weight losses associated with prescription of higher physical activity goals. Are higher levels of physical activity protective against weight regain? Am J Clin Nutr. 2007;85(4):954–959. doi: 10.1093/ajcn/85.4.954. [DOI] [PubMed] [Google Scholar]
- 24.Wing RR, et al. Lifestyle intervention in overweight individuals with a family history of diabetes. Diabetes Care. 1998;21(3):350–359. doi: 10.2337/diacare.21.3.350. [DOI] [PubMed] [Google Scholar]
- 25.Blaak EE, et al. Total energy expenditure and spontaneous activity in relation to training in obese boys. Am J Clin Nutr. 1992;55(4):777–782. doi: 10.1093/ajcn/55.4.777. [DOI] [PubMed] [Google Scholar]
- 26.Donnelly JE, et al. Nutrition and physical activity program to attenuate obesity and promote physical and metabolic fitness in elementary school children. Obes Res. 1996;4(3):229–243. doi: 10.1002/j.1550-8528.1996.tb00541.x. [DOI] [PubMed] [Google Scholar]
- 27.Goran MI, Poehlman ET. Endurance training does not enhance total energy expenditure in healthy elderly persons. Am J Phys. 1992;263(5 Pt 1):E950–7. doi: 10.1152/ajpendo.1992.263.5.E950. [DOI] [PubMed] [Google Scholar]
- 28.Kempen KP, Saris WH, Westerterp KR. Energy balance during an 8-wk energy-restricted diet with and without exercise in obese women. Am J Clin Nutr. 1995;62(4):722–729. doi: 10.1093/ajcn/62.4.722. [DOI] [PubMed] [Google Scholar]
- 29.Meijer EP, Westerterp KR, Verstappen FT. Effect of exercise training on total daily physical activity in elderly humans. Eur J Appl Physiol Occup Physiol. 1999;80(1):16–21. doi: 10.1007/s004210050552. [DOI] [PubMed] [Google Scholar]
- 30.King NA, et al. Metabolic and behavioral compensatory responses to exercise interventions: barriers to weight loss. Obesity (Silver Spring) 2007;15(6):1373–1383. doi: 10.1038/oby.2007.164. [DOI] [PubMed] [Google Scholar]
- 31.Keim NL, et al. Effect of exercise and dietary restraint on energy intake of reduced-obese women. Appetite. 1996;26(1):55–70. doi: 10.1006/appe.1996.0005. [DOI] [PubMed] [Google Scholar]
- 32.Visona C, George VA. Impact of dieting status and dietary restraint on post-exercise energy intake in overweight women. Obes Res. 2002;10(12):1251–1258. doi: 10.1038/oby.2002.170. [DOI] [PubMed] [Google Scholar]
- 33.Emery RL, Levine MD, Jakicic JM. Examining the effect of binge eating and disinhibition on compensatory changes in energy balance following exercise among overweight and obese women. Eat Behav. 2016;22:10–15. doi: 10.1016/j.eatbeh.2016.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Donnelly JE, Smith BK. Is exercise effective for weight loss with ad libitum diet? Energy balance, compensation, and gender differences. Exerc Sport Sci Rev. 2005;33(4):169–174. doi: 10.1097/00003677-200510000-00004. [DOI] [PubMed] [Google Scholar]
- 35.King AC, et al. Diet vs exercise in weight maintenance. The effects of minimal intervention strategies on long-term outcomes in men. Arch Intern Med. 1989;149(12):2741–2746. doi: 10.1001/archinte.149.12.2741. [DOI] [PubMed] [Google Scholar]
- 36.King NA, et al. Individual variability following 12 weeks of supervised exercise: identification and characterization of compensation for exercise-induced weight loss. Int J Obes. 2008;32(1):177–184. doi: 10.1038/sj.ijo.0803712. [DOI] [PubMed] [Google Scholar]
- 37.Donnelly JE, et al. Effects of a 16-month randomized controlled exercise trial on body weight and composition in young, overweight men and women: the Midwest Exercise Trial. Arch Intern Med. 2003;163(11):1343–1350. doi: 10.1001/archinte.163.11.1343. [DOI] [PubMed] [Google Scholar]
- 38.Smith SR, et al. Lorcaserin (APD356), a selective 5-HT(2C) agonist, reduces body weight in obese men and women. Obesity (Silver Spring) 2009;17(3):494–503. doi: 10.1038/oby.2008.537. [DOI] [PubMed] [Google Scholar]
- 39.World, Health, and Organization. Global Recommendations on Physical Activity for Health. WHO; Geneva: 2010. [PubMed] [Google Scholar]
- 40.Troiano RP, et al. Physical activity in the United States measured by accelerometer. Med Sci Sports Exerc. 2008;40(1):181–188. doi: 10.1249/mss.0b013e31815a51b3. [DOI] [PubMed] [Google Scholar]
- 41.Dishman RK, Buckworth J. Increasing physical activity: a quantitative synthesis. Med Sci Sports Exerc. 1996;28(6):706–719. doi: 10.1097/00005768-199606000-00010. [DOI] [PubMed] [Google Scholar]
- 42.Catenacci VA, et al. Dietary habits and weight maintenance success in high versus low exercisers in the National Weight Control Registry. J Phys Act Health. 2014;11(8):1540–1548. doi: 10.1123/jpah.2012-0250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Di Pietro L, Dziura J, Blair SN. Estimated change in physical activity level (PAL) and prediction of 5-year weight change in men: the Aerobics Center Longitudinal Study. Int J Obes Relat Metab Disord. 2004;28(12):1541–1547. doi: 10.1038/sj.ijo.0802821. [DOI] [PubMed] [Google Scholar]
- 44.McGuire MT, et al. Behavioral strategies of individuals who have maintained long-term weight losses. Obes Res. 1999;7(4):334–341. doi: 10.1002/j.1550-8528.1999.tb00416.x. [DOI] [PubMed] [Google Scholar]
- 45.Donnelly JE, et al. American College of Sports Medicine Position Stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med Sci Sports Exerc. 2009;41(2):459–471. doi: 10.1249/MSS.0b013e3181949333. [DOI] [PubMed] [Google Scholar]
- 46.Fogelholm M, Kukkonen-Harjula K. Does physical activity prevent weight gain—a systematic review. Obes Rev. 2000;1(2):95–111. doi: 10.1046/j.1467-789x.2000.00016.x. [DOI] [PubMed] [Google Scholar]
- 47.Lee IM, et al. Physical activity and weight gain prevention. JAMA. 2010;303(12):1173–1179. doi: 10.1001/jama.2010.312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Saris WH, et al. How much physical activity is enough to prevent unhealthy weight gain? Outcome of the IASO 1st Stock Conference and consensus statement. Obes Rev. 2003;4(2):101–114. doi: 10.1046/j.1467-789x.2003.00101.x. [DOI] [PubMed] [Google Scholar]
- 49.Williamson DF, et al. Recreational physical activity and ten-year weight change in a US national cohort. Int J Obes Relat Metab Disord. 1993;17(5):279–286. [PubMed] [Google Scholar]
- 50.Jakicic JM, et al. Effects of intermittent exercise and use of home exercise equipment on adherence, weight loss, and fitness in overweight women: a randomized trial. JAMA. 1999;282(16):1554–1560. doi: 10.1001/jama.282.16.1554. [DOI] [PubMed] [Google Scholar]
- 51.Wadden TA, et al. Four-year weight losses in the Look AHEAD study: factors associated with long-term success. Obesity (Silver Spring) 2011;19(10):1987–1998. doi: 10.1038/oby.2011.230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.MacLean PS, et al. Peripheral metabolic responses to prolonged weight reduction that promote rapid, efficient regain in obesity-prone rats. Am J Phys Regul Integr Comp Phys. 2006;290(6):R1577–88. doi: 10.1152/ajpregu.00810.2005. [DOI] [PubMed] [Google Scholar]
- 53.Cornier MA, et al. The effects of overfeeding on the neuronal response to visual food cues in thin and reduced-obese individuals. PLoS One. 2009;4(7):e6310. doi: 10.1371/journal.pone.0006310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rosenbaum M, et al. Leptin reverses weight loss-induced changes in regional neural activity responses to visual food stimuli. J Clin Invest. 2008;118(7):2583–2591. doi: 10.1172/JCI35055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Levin BE, Dunn-Meynell AA. Chronic exercise lowers the defended body weight gain and adiposity in diet-induced obese rats. Am J Phys Regul Integr Comp Phys. 2004;286(4):R771–8. doi: 10.1152/ajpregu.00650.2003. [DOI] [PubMed] [Google Scholar]
- 56.Wang X, et al. Weight regain is related to decreases in physical activity during weight loss. Med Sci Sports Exerc. 2008;40(10):1781–1788. doi: 10.1249/MSS.0b013e31817d8176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Weigle DS. Contribution of decreased body mass to diminished thermic effect of exercise in reduced-obese men. Int J Obes. 1988;12(6):567–578. [PubMed] [Google Scholar]
- 58.MacLean PS, et al. Enhanced metabolic efficiency contributes to weight regain after weight loss in obesity-prone rats. Am J Phys Regul Integr Comp Phys. 2004;287(6):R1306–15. doi: 10.1152/ajpregu.00463.2004. [DOI] [PubMed] [Google Scholar]
- 59.Higgins JA, et al. Resistant starch and exercise independently attenuate weight regain on a high fat diet in a rat model of obesity. Nutr Metab Lond. 2011;8:49. doi: 10.1186/1743-7075-8-49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Steig AJ, et al. Exercise reduces appetite and traffics excess nutrients away from energetically efficient pathways of lipid deposition during the early stages of weight regain. Am J Phys Regul Integr Comp Phys. 2011;301(3):R656–67. doi: 10.1152/ajpregu.00212.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Giles ED, et al. Exercise decreases Lipogenic gene expression in adipose tissue and alters adipocyte cellularity during weight regain after weight loss. Front Physiol. 2016;7:32. doi: 10.3389/fphys.2016.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Duncan GE, et al. Exercise training, without weight loss, increases insulin sensitivity and postheparin plasma lipase activity in previously sedentary adults. Diabetes Care. 2003;26(3):557–562. doi: 10.2337/diacare.26.3.557. [DOI] [PubMed] [Google Scholar]
- 63.Zhao J, et al. Endurance exercise is a leptin signaling mimetic in hypothalamus of Wistar rats. Lipids Health Dis. 2011;10:225. doi: 10.1186/1476-511X-10-225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Martins C, et al. Effects of exercise on gut peptides, energy intake and appetite. J Endocrinol. 2007;193(2):251–258. doi: 10.1677/JOE-06-0030. [DOI] [PubMed] [Google Scholar]
- 65.Mayer J, et al. Exercise, food intake and body weight in normal rats and genetically obese adult mice. Am J Phys. 1954;177(3):544–548. doi: 10.1152/ajplegacy.1954.177.3.544. [DOI] [PubMed] [Google Scholar]
- 66.Mayer J, Roy P, Mitra KP. Relation between caloric intake, body weight, and physical work: studies in an industrial male population in West Bengal. Am J Clin Nutr. 1956;4(2):169–175. doi: 10.1093/ajcn/4.2.169. [DOI] [PubMed] [Google Scholar]
- 67.Paris HL, et al. Increasing energy flux to decrease the biological drive toward weight regain after weight loss - a proof-of-concept pilot study. Clin Nutr ESPEN. 2016;11:e12–e20. doi: 10.1016/j.clnesp.2015.11.005. [DOI] [PubMed] [Google Scholar]
- 68.Butryn ML, et al. Consistent self-monitoring of weight: a key component of successful weight loss maintenance. Obesity (Silver Spring) 2007;15(12):3091–3096. doi: 10.1038/oby.2007.368. [DOI] [PubMed] [Google Scholar]
- 69.Nance DM, et al. Sexually dimorphic effects of forced exercise on food intake and body weight in the rat. Physiol Behav. 1977;19(1):155–158. doi: 10.1016/0031-9384(77)90173-1. [DOI] [PubMed] [Google Scholar]
- 70.Frisch RE. Body fat, menarche, fitness and fertility. Hum Reprod. 1987;2(6):521–533. doi: 10.1093/oxfordjournals.humrep.a136582. [DOI] [PubMed] [Google Scholar]