Abstract
Domoic acid (DA) is an algal neurotoxin that accumulates in marine fish and shellfish. DA can move across the placenta and concentrate in amniotic fluid, which can be swallowed during late gestation. DA also transfers to infants via milk. Preclinical studies to determine effects of developmental DA expose have primarily involved DA exposure during the postnatal period and little is known about late CNS effects following prenatal DA. In the present study, we tested the hypothesis that prenatal exposure of FVB mice to low levels of DA would result in diminished social interaction and sensory motor gating associated with alterations in parvalbumin immunoreactivity in relevant brain regions undergoing development during and following DA exposure. In addition to parvalbumin, we stained with NeuN for a neuronal specific nuclear protein to determine if neuronal loss followed prenatal DA exposure. A single moderate dose of DA administered during gestation produces diminishes social investigation and alters sensorimotor gating, behavioral effects more pronounced in males than females. These behavioral changes were associated with discrete alterations in the parvalbumin-positive subtype of GABAergic neurons in the dentate gyrus and lateral amygdala.
Keywords: social investigation, prepulse inhibition, parvalbumin
1. Introduction
Domoic acid (DA) is an algal neurotoxin that accumulates in marine fish and shellfish (1). Human consumption of DA-contaminated seafood can result in Amnestic Shellfish Poisoning, with symptoms that include memory loss, seizures, and death (2). The regulatory limit for DA in seafood (20 mg/kg tissue) is designed to protect adults from the effects of acute exposure to DA (3). In the Pacific Northwest, Chesapeake Bay and along the coasts of western Europe, peak annual levels of DA in shellfish can exceed the regulatory limit, resulting in intermittent restrictions on shellfish harvests (4).
Rodent studies indicate that newborn animals are substantially more sensitive (5) than adults (6, 7) to the toxicity of DA exposure. DA can move across the placenta and concentrate in amniotic fluid, which can be swallowed during late gestation (8). DA also transfers to infants via milk (9). After crossing the blood-brain barrier (8). DA binds to kainate and AMPA receptors (10, 11) within several regions of the developing brain, including cortical and limbic areas that regulate social behavior (12, 13). Thus, infant rats that are exposed to DA can express social withdrawal at doses 100-fold lower than levels that cause adult mortality (14).
The effects of exposure to DA bear resemblance to social behaviors and neuropathology associated with Autism Spectrum Disorder (ASD). Perinatal rats exposed to low levels of DA, levels 100-fold lower than those that cause mortality in adult rodents, express long-term decreased social interaction that are more pronounced in males than females (14), resembling the sex bias in ASD. Exposure to DA results in other pathologies that are similar to features of ASD (15–17). These features include repetitive behaviors (15), impaired sensory motor gating (18, 19), heightened seizure susceptibility (20–29), increased brain excitatory tone (23, 25, 26, 30–33), altered axon growth and dendritic arborization (17, 34), aberrant interneuron structure and function (35–45), and anatomical changes in the cortex, hippocampus, and amygdala (13, 34, 46–56).
To further examine the extent that fetal exposure to DA might influence autistic-like features of brain anatomy, we chose to count neurons that express parvalbumin. Genes identified as part of genome-wide association studies (57, 58) and transcriptome co-expression network analyses (59) are highly expressed in parvalbumin-positive cortical interneurons. Two of those genes, PVALB and DLX1, are downregulated in brains of individuals with ASD (59). In addition, decreased parvalbumin mRNA and protein expression were seen in ASD patients (60). In contrast to the mRNA, autism has been associated with an elevated number of parvalbumin-ir neurons in the CA1 and CA3 hippocampal layers (61). Downregulation of parvalbumin (62–64) rather than loss of parvalbumin-positive neurons (60) has been proposed as an adaptive mechanism. Consistent with these human data, genetic mouse models suggested as relevant to autism can show alterations in the number of parvalbumin-immunoreactive (ir) cells in the cortex and hippocampus (65, 66) and abnormalities in parvalbumin circuit function (67). Furthermore, sex-dependent changes in parvalbumin-ir have been reported in the hippocampus in a rat model of postnatal DA exposure (32). Finally, parvalbumin knockout mice show altered social interactions, ultrasonic vocalizations, and a reduced startle response but intact pre-pulse inhibition (68). These effects might not be limited to DA exposure. Prenatal cocaine exposure in mice affects social development in mice and this is associated with molecular alterations in the medial prefrontal cortex (69). Consistent with a role of the medial prefrontal cortex, reduced cognitive flexibility was seen in rats neonatally treated with DA (70). Postnatal DA exposure in rats affected open field behavior, novelty-related behavior, and nicotine-induced conditioned place preference as well, indicating that other brain areas are likely involved as well (71). In addition to the hippocampus and cortex, the amygdala might also play an important role in the detrimental effects of DA exposure on the brain. The amygdala plays an important role in social behavior (72–76) and amygdala dysfunction has been proposed to account for impairments in social behavior in ASD (77), (78).
We chose to examine paravalbumin positive neurons in the amygdala for two reasons. First, there is ample evidence from studies of California sea lions exposed to DA that necrosis is most prominent in the hippocampus and, to a lesser extent, the amygdala (47), consistent with experimental data (79). Lesions in the hippocampus and amygdala were also reported following DA intoxication in humans (80). The second reason we chose to study neurons in the amygdala is that the amygdala plays a pivotal role in the development and maintenance of social interactions and amygdala function can be impaired in ASD.
Preclinical studies to determine late central nervous system (CNS) effects of developmental DA expose have primarily involved DA exposure during the postnatal period. Less is known about late CNS effects following prenatal DA. A better understanding of DA toxicity during prenatal development will help us assess whether current exposure levels are a risk factor for impaired psychosocial functioning. Therefore, in the present study we tested the hypothesis that prenatal exposure of FVB mice to low levels of DA would result in diminished social interaction and sensory motor gating associated with alterations in parvalbumin immunoreactivity in brain regions involved in the regulation of social behavior and undergoing development during and following DA exposure. In addition to parvalbumin, we used NeuN, a neuronal specific nuclear protein (81), to determine whether there was neuronal loss following prenatal DA exposure.
2. Materials and Methods
2.1. Mouse husbandry
FVB/NJ (FVB) mice, purchased from Jackson Laboratories (Bar Harbor, ME, USA), were bred and housed at Oregon Health and Science University (OHSU) under controlled temperature (21 ± 1°C) and humidity (40–60%). Lighting was maintained on a 12:12 h light/dark (dark period 0900–2100 hours) cycle. Mice were housed in standard polypropylene cages (290 × 180 × 130 mm) lined with pelleted paper bedding (ECOfresh, Absorption Corp.) and had ad libitum access to chow (Lab Rodent Diet 5001, Purina Mills) and water. Animal care and experimental protocols were conducted in accordance with the regulations of the institutional care and use committee at the Oregon Health and Science University and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Our own laboratory personnel carried out all aspects of the mouse husbandry under strict guidelines to insure gentle and consistent handling of the mice.
2.2. Maternal Domoic Acid Exposure
Pregnant mice were weighed daily. When they reached 30 grams (M = 30.159 grams, SD = 1.613), approximately six days prior to parturition (M = 5.86 days, SD = 1.7 days), either 1.5 mg/kg of DA or saline was injected s.c. Weaning occurred on postnatal day (PD) 20–22; pups were separated by sex and placed in separate clean colony cages. Housing groups of siblings were made up of two males and two females. During sexing, mice were randomly assigned as the test or stimulus mouse role for social behavior testing (see below). Test mice were marked on the tail with a black Sharpie and stimulus mice were ear-punched on the top left ear. To avoid euthanizing litters with uneven sex distributions, on rare occasions, two male and female housing groups were combined across litters if both litters had the same age and treatment condition.
For the social interaction testing, 62 same sex FVB pairs were analyzed. There were 22 DA exposed pairs (14 male pairs, 19 female pairs), and 30 saline pairs (15 male, 14 female). One DA treated female was excluded for climbing on the equipment during SI testing and vocalizations for four saline treated animals were unavailable due to a vocalization equipment malfunction (three saline males and one saline female).
2.3. Social interaction testing
Three social interaction (SI) tests were given, one five minute SI test on PD21, one 15-minute test on PD25, and a final 15-minute test was given on PD35. All testing was conducted during the dark period of the LD cycle (1400–1800) under dim red illumination. At each test, the age of the mice was accurate within one PD. Prior to testing on PD21 (immediately following weaning), mice were isolated in a clean new cage for 30 minutes on the housing rack before transfer to the experimental room where they were habituated to the environment for an additional five minutes in separate cages. The stimulus mouse was then placed into the opposite side of the test mouse's cage. Ultrasonic vocalizations (USVs) and social behaviors of the test mouse were recorded for five minutes. Mice were then removed from the experimental room and kept under the hood with soft food and water until placement into their social housing group for subsequent testing.
For SI testing on PD 25 and 35, all mice were isolated 24 hours prior to testing in a clean cage containing fresh bedding without nesting material and ad libitum access to food and water. Test and stimulus mice were then transported to the experimental room for 5 minutes of additional habituation. The behavior and vocalizations of the isolated test mouse were then recorded for an additional 5 minutes before the stimulus mouse was introduced to the opposite side of the cage. Social interactions and USVs were recorded for 15 additional minutes. The behaviors were video recorded (Sony, DCR-VX2100, Japan) and stored on a desktop computer (Precision T3400, Dell, Round Rock, TX, USA). With the aid of computer assisted analysis software (ButtonBox v.5.0, Behavioral Research Solutions, Madison, WI, USA), all test mouse behaviors were scored off-line by two different observers blind to the age and treatment group of the interacting mice. At the conclusion of behavioral testing on PD35, animals were euthanized by CO2 in accordance with institutional animal care and use committee (IACUC) protocols.
Social investigation (SI) was scored as several coded behaviors that each test mouse directed towards each stimulus mouse and included: [i] sniffing or snout contact with the head/ neck/mouth area, [ii] sniffing or snout contact with the flank area, [iii] direct contact with the anogenital area, [iv] social pursuit within one body-length as the stimulus mouse moved continuously throughout the cage and [v] social grooming. These variables were highly correlated and combined into a composite measure of SI (82) SI testing was conducted during the dark phase (4.5 to 9.5 hours after lights-off.), under dim red illumination, in a sounddampened room. SI was recoded for each experiment until the interrater reliability was greater than r = .90. The presentation of all SI data and statistical outcomes in this study are based on an average of these two independent measurements.
2.4. Measurement and characterization of ultrasonic vocalizations during social interaction
Ultrasonic vocalizations (USVs) were recorded during SI tests with an ultrasound microphone (10–180 kHz flat-frequency range) lowered to the plane of a 1/8”thick transparent Plexiglas cage top with a small central opening (30-mm diameter). USVs were collected with an UltraSoundGate 116 acquisition system (UltraSoundGate model CM16, Avisoft Bioacoustics, Berlin, Germany) and Avisoft-Recorder v.2.97 (Avisoft Bioacoustics), and stored as wav files for subsequent analysis. A 40-kHz band-pass filter was used to minimize background noise during recordings. Using SASLab Pro v.4.39 (Avisoft Bioacoustics), each sonogram was automatically tallied for the total number of USVs produced with during the SI period. However, some wav files still contained some ‘non-USV’ signal that compromised the precision of the automated parameter-measurements. Therefore, extraneous noise was manually identified and removed. When a researcher encountered a signal that was difficult to interpret, the USV was evaluated by a minimum of one additional trained observer. For the final count, two blinded researchers manually removed noise from the sonograms, and the total USVs were averaged across the two raters (inter-rater reliability, r > .9). Technological constraints do not allow us to record vocalizations from only one mouse in a dyad. However, while vocalizations of both mice were recorded, general consensus is that the resident (test) mouse vocalizes and the introduced (stimulus) mouse produces relatively few USVs.
2.5. Prepulse Inhibition
Mice were placed in an enclosure within a startle monitor sound-attenuated chamber and startle response amplitudes were measured with a force transducer (Kinder Scientific, Poway, CA). Following a 5 min acclimation period, mice were exposed to three acoustic stimuli (40 ms, 110 db). The testing phase consisted of 20 ms pre-pulses (70–80 db) followed by 50 ms delays and 40 ms acoustic stimuli (110 db). The same pattern of acoustic stimuli and testing with pre-pulses then occurred with a 120 db stimulus. Random inter-trial intervals were used between trials (15–30 sec). Pre-Pulse inhibition (PPI) was calculated using the following formula: % response = 100 × ((SPS)/S), where S was the mean startle amplitude without a pre-pulse and PS was the mean startle response following a pre-pulse. Thus, a 100% response in the PPI test indicates complete inhibition of the startle response during pre-pulse trials.
2.6. Immunohistochemistry
Following PPI testing, mice were intracardially perfused with 20 ml phosphate buffered saline (PBS) followed by 40 ml 4% paraformaldehyde. Brains were removed, stored in 4% paraformaldehyde overnight, and then transferred to 30% sucrose. Twenty brains were randomly chosen for immunohistochemistry (N=5 per treatment/sex). Brains were coronally sectioned at 40 µm into three series using a cryostat (Microm HM505E, MICROM international GmbH, Walldorf, Germany) and processed for immunohistochemical detection of NeuN and parvalbumin immunoreactivity. Parvalbumin immunoreactivity was performed using fluorescent immunohistochemistry. Briefly, one series of sections were rinsed with PBS 3 times, then blocked with 4% donkey serum in PBS-TX for 90 min. Sections were then incubated in antiparvalbumin (1:1500, mouse, Sigma, P3088) overnight. Sections were then incubated in 1:200 donkey anti-goat Dylight 594 (Abcam, Cambridge, MA, USA) for 3 hours at room temperature. Next, sections were rinsed in PBS 4 times. Sections were then slide mounted and coverslipped with antifade reagent to preserve fluorescent signal (Vectashield with DAPI, Vector) and light protected.
For NeuN immunohistochemistry, one series of sections was rinsed in phosphate buffered saline (PBS), incubated in 1% hydrogen peroxide and 0.3% Triton-X in PBS (PBS-TX) for 10 minutes, again rinsed in PBS, then incubated in 4% normal goat serum (NGS) in PBS-TX for 1 hour. After rinsing in PBS, sections were incubated in primary antisera (anti-NeuN, monoclonal: 1:10,000, mouse, Millipore, MAB377, Billerica, MA, USA) in 4% NGS and PBS-TX overnight at room temperature. Sections were then rinsed in PBS and incubated for 1 hour in biotinylated goat-anti rabbit antibody in PBS-TX (1:500, Vector Laboratories, Burlingame, CA) followed by rinses in PBS and a 1 hour incubation in avidin-biotin peroxidase complex (ABC Elite kit, Vector Laboratories). Following rinses in Tris buffered saline (TBS), sections were developed for visualization of c-Fos positive cells in a hydrogen peroxide/diaminobenzidine/TBS solution for 10 minutes, after which sections were rinsed in PBS and immediately mounted on slides. The following day, sections were dehydrated in ethanol, defatted in xylene, and coverslipped with Permount (Sigma Chemical Co., St. Louis MO, USA).
2.7. Microscopy
Quantification of parvalbumin-positive cells was performed using an Olympus IX81 microscope (Olympus, Center Valley, PA, USA) equipped with Slidebook software (Intelligent Imaging Innovations, Inc., Denver, CO, USA). Brain regions were identified using the mouse brain atlas of Franklin and Paxinos (2004)(83). Images of the lateral amygdala, basolateral amygdala, infralimbic cortex, prelimbic cortex, dentate gyrus, and CA1 and CA3 hippocampal regions were captured bilaterally within 3 sections using a 10× objective. Parvalbumin immunoreactive cells were quantified bilaterally within fixed area frames; lateral amygdala (box, 275 × 450 µm), basolateral amygdala (box, 790 × 410 µm each), infralimbic cortex (circle, 575 µm diameter), prelimbic cortex (box, 425 × 400 µm), dentate gyrus (box, 850 × 420 µm), CA1 (box, 850 × 420 µm), and CA3 (box, 850 × 420 µm). Quantification of NeuN was performed in the prelimbic cortex using a 20× objective. Images were captured bilaterally within 3 sections and NeuN positive cells were counted within a 365 × 280 µm rectangle area. For all cell quantifications, cells were counted in both hemispheres of a given region in each section and summed. Cell counts for two sections were averaged and are presented as cells per section.
2.8. Statistical Analysis
Effects of DA exposure on social behavior and USVs were analyzed using a full factorial repeated measures analysis of variance (ANOVA) and JMP version 6.0 (SAS Institute Inc., Cary, North Carolina). Treatment group and sex of each mouse pair were between-group factors and age at the social interaction test (postnatal day (PD) 25 and 35) were repeated measures. Trend level interactions and pre-planned post hoc analyses comparing the effect of DA for males and females, were followed up with t-tests when appropriate. Prepulse inhibition and immunohistochemical data were analyzed using GraphPad Prism v.6. Prepulse inhibition data were analyzed using two-way ANOVA separately by prepulse. Immunohistochemical data (NeuN, parvalbumin) were analyzed separately for each brain region using two-way ANOVA with sex and treatment as factors. Sidak’s corrected post hoc comparisons were performed when statistically appropriate. All data are reported as means ± standard error of the mean (SEM). p < 0.05 was considered statistically significant.
3. Results
3.1. Social interaction tests
Prenatal DA exposure significantly reduced time spent in social interaction (F(1,58)=5.9305, p = .018) during the 15 minute SI tests. Although no main effect of age at SI test was found (p = .78), the interaction between age and group (F(1,52) = 25.50, p < .0001) shows that the effect of DA is driven by the test at PD35. No significant effects of sex (p = .32), sex by group interaction (p = .32) or age by sex interactions (p = .39) were found. During the five minute SI test, a 2×2 ANOVA showed that DA exposed animals spent less time interacting than their saline exposed peers (F(1,47) = 4.11, p = .0482). No effect of sex (p=.55) or sex by treatment status (p = .08) was found.
Prenatal DA exposure significantly decreased time spent in social interaction (F(1,49)=4.22, p = .045) across each of the 15 minute SI tests. We also found a significant sex by group interaction (F(1,49)=6.69, p = .0127) suggesting that diminished SI was only expressed by males prenatally exposed to DA. Post hoc analyses comparing saline to DA at each SI test for each sex found that males had significantly less SI on PD25 (p = .041; Figure 1a) as well as PD35 (p = .043; Figure 2a). Similar tests showed no effect of DA on female mice. We also found an interaction between sex and time (F(1,49)=13.3881, p = .0006) where females increased and males decreased in SI between tests at day PD25 and 35 (Figure 1c). No other significant main effects or interactions were found.
Fig. 1.
Prenatal DA exposure impairs social interactions in adolescent male, but not female, mice at PD25 (A) and PD35 (B). *p < 0.05. All data are presented as the mean ± SEM.
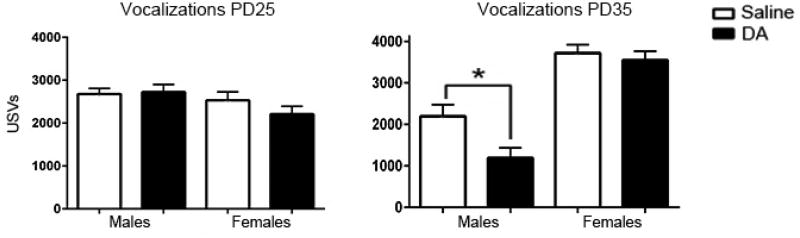
Fig. 2.
Prenatal DA exposure reduces USVs in male, but not female, adolescent mice at PD35 (B) but not at PD25 (A). *p < 0.05. All data are presented as the mean ± SEM.
3.2. Ultrasonic Vocalizations
Ultrasonic vocalizations were quantified across each of the 15-minute social interaction tests. Prenatal DA exposure significantly reduced the number of calls (F(1,50)=37.84, p < .0001) during the 15 minute SI tests. Although no main effect of age at SI test was found (p = .78), the interaction between age and group (F(1,50) = 57.18, p < .0001) shows that, similar to the SI data, the effect of DA is driven by the test at PD35. No significant effect of sex (p = .20), sex by group (p = .21), or age by sex interactions (p = .68) were found. Prenatal DA exposure significantly reduced the number of calls (F(1,43)=4.27, p < .044) across genders. We also found that overall, females had more USVs than did males (F(1,43)=22.52, p < .0001; Figure 2a,b), and females increased and males slightly decreased their USVs from PD25 to 35 (F(1,43)=59.94, p < .0001; Figure 2). Post hoc tests show that only in males at PD35 USVs are significantly lower after DA exposure (p = .0039; Figure 2b).
3.3. Prepulse Inhibition
For the 70db prepulse followed by a 110 db pulse a 2-way ANOVA revealed a significant interaction between sex and treatment ((F(1,16)= 5.34, p<0.05) but no main effects of sex or treatment. Sidak’s multiple comparisons test revealed a significant decrease in PPI in DA compared to SA treated males (Figure 3a). A significant sex × treatment interaction was also found for the 70 db prepulse/110 db pulse with no main effects of treatment or sex (Figure 3b). Sidak’s multiple comparisons analysis revealed no further effects of treatment within sex. No significant differences were found for the 80 db prepulse/110 db pulse (Figure 3c) or for 70, 75, or 80 db prepulse followed by a 120 db pulse (data not shown).
Figure 3. Pre-Pulse inhibition (PPI) of the startle response in prenatal saline- and domoic acid-treated mice.
Acoustic startle responses following a 110 decibel pulse preceded by pre-pulses of (A) 70, (B) 75, and (C) 80 decibels. *p < 0.05 indicates a significant decrease () in PPI in DA-treated male mice compared to SA-treated male mice.
3.4. Parvalbumin and NeuN Quantification
There was a significant main effect of treatment for the number of parvalbumin-immunoreactive cells in the lateral amygdala ((F(1,16)= 5.58, p<0.05). Domoic acid-treated mice showed an increase in parvalbumin-positive cells compared to saline-treated mice (Figure 4). No significant effects of sex or interaction were found in the lateral amygdala. In the dentate gyrus, a 2-Way ANOVA revealed a significant effect of sex with males showing a greater number of parvalbumin-immunoreactive cells compared to females ((F(1,16)= 5.34, p<0.05). This main effect of sex seemed primarily driven by a greater number of parvalbumin expressing cells in DA-treated males compared to DA-treated females. Sidak’s multiple comparisons test revealed a significant decrease in parvalbumin cells in domoic acid-treated female than male mice (p<.05; Figure 4b). No significant effects of sex, treatment, or interactions were found for parvalbumin-positive cells in the basolateral amygdala, infralimbic cortex, prelimbic cortex, CA1, or CA3 (Table 1). Furthermore, no group differences were found for the number of NeuN-positive cells in the prelimbic cortex (Figure 5).
Figure 4. Parvalbumin-positive cells in the lateral amygdala and dentate gyrus.
(A) There was an overall increase in the number of parvalbumin-positive cells in DA- (males and females) compared to SA- (males and females) treated mice. (B) In the dentate gyrus, males showed a greater number of parvalbumin positive cells compared to females. This main effect of sex was primarily driven by a greater number of parvalbumin expressing cells in DA-treated males compared to DA-treated females. Representative images of parvalbumin positive cells in the lateral amygdala (C) and dentate gyrus (D). ^ indicates an overall main effect of prenatal treatment (p < 0.05). * indicates significant difference between DA females and DA males.
Table 1.
Parvalbumin-immunoreactive cells after in utero treatment1
| Region | Saline | Domoic Acid |
|---|---|---|
| Basolateral Amygdala | 30.24 ± 1.73 | 31.40 ± 1.82 |
| Infralimbic Cortex | 50.75 ± 2.81 | 49.70 ± 3.17 |
| Prelimbic Cortex | 55.14 ± 3.66 | 56.41 ± 3.25 |
| CA1 | 10.93 ± 1.04 | 11.68 ± 1.04 |
| CA3 | 14.56 ± 1.48 | 15.22 ± 1.26 |
Averages and SEM values are shown. No significance difference was found between the groups. N = 10 mice/treatment.
Figure 5. NeuN-positive cells in the prelimbic cortex.
No differences were found for the number of NeuN-positive cells in the prelimbic cortex between male and female DA- or SA-treated mice.
4. Discussion
The results of this study indicate that a single prenatal exposure to DA leads to diminished social investigation (SI test) and ultrasonic vocalizations (USVs), and altered sensorimotor gating (PPI). Previous studies have indicated that repeated exposure to DA during postnatal development leads to impairments in social functioning (14) and sensorimotor gating (18). Though perinatal exposure is known to modulate social behavior (14), this is the first report indicating that a prenatal exposure to DA can alter both social behavior and sensory motor gating. Furthermore, we find that USVs emitted during social interaction are also influenced by DA exposure.
Male behaviors appear profoundly more sensitive to DA exposure than those of females. These data are consistent with a previous report that male rats exposed to low levels of DA between postnatal day 8–14 show more pronounced long-term decreases in social interaction than females (84). This sex-dependent effect of DA also extends beyond social behaviors and includes effects on attentional processing as assessed by latent inhibition. In this task, males treated neonatally with DA showed decreased attentional processing while females were unaffected by DA treatment (85). Like with the sex ratio in susceptibility to ASD and to exposures to various toxic chemicals, males are more susceptible to in utero DA exposure than females. In humans, males are more highly associated with DA susceptibility than females (86). Following DA exposure, social interaction was decreased in males at PD25 and PD35, whereas the rate of USV emission during these social interactions was decreased only among the older adolescent males (i.e., PD35). Previous studies have shown that the rate of USV emission by juvenile mice is highly correlated with the degree of social approach behavior (82). However, discrepancies between the vigor of SI response and the rate of USV production have been reported elsewhere. Of particular relevance are findings of a similar discrepancy. For instance, juveniles exposed prenatally to cocaine express increased SI and USV production at PD 25, but only increased SI at PD35 (69). Since these social functions require different brain regions, perhaps these differences reflect variations in their susceptibility.
In the present study, prenatal DA exposure was associated with an increased number of parvalbumin-containing cells in the lateral amygdala (both sexes) and in the dentate gyrus, specifically in males. In humans, autism has been associated with an elevated number of parvalbumin-ir neurons in the CA1 and CA3 hippocampal layers (61). These sex-dependent effects of DA may be timing- and dose-dependent. In rats, daily DA administration between postnatal day 8–14 resulted in decreased levels of hippocampal GABA synthesizing glutamic acid decarboxylase (GAD) and sex-dependent reductions in hippocampal parvalbumin (87). Specifically, DA reduced parvalbumin immunoreactivity in the dorsal dentate gyrus in females and in the middle dentate gyrus in males. This indicates that either repeated dosing has different effects on parvalbumin immunoreactivity than does a single injection or that neonatal and prenatal dosing regimens can produce opposing effects on parvalbumin-positive neurons in the dentate gyrus.
Alterations in the function of the amygdala and particularly the lateral amygdala has been associated with autism in humans (77, 88) (50, 78, 89) as well as in rodent models that have been considered relevant to ASD (90–94). Postmortem examination of human brains revealed a decrease in neuronal density specifically within the lateral region of the amygdala (95). While we did not see alterations in neuronal density, we did see an increase in parvalbumin immnoreactivity in the lateral amygdala in prenatal DA-treated mice, indicating that dysregulation of excitatory/inhibitory tone in the amygdala might be critical in impairing amygdala function in ASD. In a valproate-induced rat model considered relevant to ASD, neurons of the lateral amygdala show disrupted excitatory/inhibitory balance (96). Less is known specifically about parvalbumin-positive neurons in the lateral amygdala and their potential role in regulating social interaction and altering PPI. In genetic mouse models, abnormal numbers of parvalbumin-immunoreactive neurons in regions including the amygdala have been linked to reduced social interaction and altered PPI (97). In general, these genetically altered mouse models show decreased numbers of parvalbumin-containing cells which contrasts with the current findings following a prenatal DA exposure. This suggests that diminished social interaction and sensorimotor gating may be linked to both elevated and decreased numbers of parvalbumin-expressing cells and may reflect dysregulation of excitatory/inhibitory tone in the amygdala. The results of our study better parallel finding in humans in which autism has been associated with an elevated number of parvalbumin-immunoreactive neurons in hippocampal regions (61). While sex differences were seen showing increased susceptibility of male mice to effects of DA exposure on SI and rates of USV emission during the SI test, no sex differences were seen in the effects of DA exposure of parvalbumin-positive cells in the dentate gyrus and lateral amygdala. These data suggest that the male mice might be more susceptible to the consequences of altered parvalbumin-related measures than female mice. This might be especially pertinent to ASD which is more often seen in males than females.
In summary, the findings of the current study demonstrate that a single moderate dose of DA administered during gestation can diminish social interaction and sensorimotor gating, behavioral effects that were more pronounced in males than females. These behavioral changes bear resemblance to features associated with the ASD diagnosis and are accompanied by discrete alterations in the parvalbumin-positive subtype of GABAergic neurons in the dentate gyrus and lateral amygdala. These findings, along with evidence for a high prevalence of ASD in coastal communities adjacent to marine environments with high levels of DA (98–101), suggest that further study regarding the neurobiology of this potent algal toxin is warranted.
Highlights.
In utero DA exposure diminishes social investigation and alters sensorimotor gating.
This is more pronounced in males than females.
This is associated with alterations in the parvalbumin-positive subtype of GABAergic neurons.
Acknowledgments
This work was supported by T32DA007262 and the development accounts of Drs. Raber and Lahvis.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
None of the authors has competing financial interests or other conflicts of interest.
References
- 1.Lefebvre K, Robertson A. Domoic acid and human exposure risk: A review. Toxicon. 2010;56:218–230. doi: 10.1016/j.toxicon.2009.05.034. [DOI] [PubMed] [Google Scholar]
- 2.Grant K, Burbacher T, Fuastman E, Grattan L. Domoic acid: neurobehavioral consequences of exposure to a prevalent biotoxin. Neurotoxicol Teratol. 2010;32:132–141. doi: 10.1016/j.ntt.2009.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Todd E. Domoic acid and amnesic shelfish poisoning- a review. J Food Protection. 1993;56:69–83. doi: 10.4315/0362-028X-56.1.69. [DOI] [PubMed] [Google Scholar]
- 4.Wekell JC, Gauglitz EJJ, Barnett HJ, Hatfield CL, Simons D, Ayres D. Occurrence of domoic acid in Washington state razor clams (Siliqua patula) during 1991–1993. Natural Toxins. 1994;2:197–205. doi: 10.1002/nt.2620020408. [DOI] [PubMed] [Google Scholar]
- 5.Levin ED, Pang WG, Harrison J, Williams P, Petro A, Ramsdell JS. Persistent neurobehavioral effects of early postnatal domoic acid exposure in rats. Neurotoxicology and Teratology. 2006;28(6):673–680. doi: 10.1016/j.ntt.2006.08.005. [DOI] [PubMed] [Google Scholar]
- 6.Iverson F, Truelove J, Nera E, Tryphonas L, Campbell J, Lok E. Domoic acid poisoning and mussel-associated intoxication: Preliminary investigations into the response of mice and rats to toxic mussel extract. Food and Chemical Toxicology. 1989;27(6):377–384. doi: 10.1016/0278-6915(89)90143-9. [DOI] [PubMed] [Google Scholar]
- 7.Grimmelt B, Nijjar MS, Brown J, Macnair N, Wagner S, Johnson GR, Amend JF. Relationship between domoic acid levels in the blue mussel (Mytilus edulis) and toxicity in mice. Toxicon. 1990;28(5):501–508. doi: 10.1016/0041-0101(90)90294-h. [DOI] [PubMed] [Google Scholar]
- 8.Maucher J, Ramsdell J. Maternal-fetal transfer of domoic acid in rats at two gestational time points. Enviromental Health Perspec. 2007;115:1743–1746. doi: 10.1289/ehp.10446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maucher J, Ramsdell J. Domoic acid transfer to mil: evaluation of a potential route of neonatal exposure. Enviromental Health Perspec. 2005;113:461–464. doi: 10.1289/ehp.7649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hampson DR, Huang X-p, Wells JW, Walter JA, Wright JLC. Interaction of domoic acid and several derivatives with kainic acid and AMPA binding sites in rat brain. European Journal of Pharmacology. 1992;218(1):1–8. doi: 10.1016/0014-2999(92)90140-y. [DOI] [PubMed] [Google Scholar]
- 11.Nanao MHMH, Green TT, Stern-Bach YY, Heinemann SFSF, Choe SS. Structure of the kainate receptor subunit GluR6 agonist-binding domain complexed with domoic acid. Proceedings of the National Academy of Sciences of the United States of America. 2005;102(5):1708–1713. doi: 10.1073/pnas.0409573102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ritter LM, Vazquez DM, Meador-Woodruff JH. Ontogeny of ionotropic glutamate receptor subunit expression in the rat hippocampus. Developmental Brain Research. 2002;139(2):227–236. doi: 10.1016/s0165-3806(02)00572-2. [DOI] [PubMed] [Google Scholar]
- 13.Bahn S, Volk B, Wisden W. Kainate receptor gene expression in the developing rat brain. The Journal of Neuroscience. 1994;14(9):5525–5547. doi: 10.1523/JNEUROSCI.14-09-05525.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ryan C, Robbins M, Smith M, Gallant I, Adams-Marriott A, Doucette T. Altered social interaction in adult rats following neonatal treatment with domoic acid. Physio Behav. 2011;102:291–295. doi: 10.1016/j.physbeh.2010.11.020. [DOI] [PubMed] [Google Scholar]
- 15.Peng YG, Ramsdell JS. Brain Fos induction is a sensitive biomarker for the lowest observed neuroexcitatory effects of domoic acid. Fundamental & Applied Toxicology. 1996;31(2):162–168. doi: 10.1006/faat.1996.0087. [DOI] [PubMed] [Google Scholar]
- 16.Watanabe KH, Andersen ME, Basu N, Carvan MJ, 3rd, Crofton KM, King KA, Sunol C, Tiffany-Castiglioni E, Schultz IR, Levin ED, Pang WG, Harrison J, Williams P, Petro A, Ramsdell JS, Appel NM, Rapoport SI, O'Callaghan JP, Trudeau LE, Castellucci VF. Defining and modeling known adverse outcome pathways: Domoic acid and neuronal signaling as a case study. Environ Toxicol Chem. 2011;30(1):9–21. doi: 10.1002/etc.373. [DOI] [PubMed] [Google Scholar]
- 17.Colman JR, Nowocin KJ, Switzer RC, Trusk TC, Ramsdell JS. Mapping and reconstruction of domoic acid-induced neurodegeneration in the mouse brain. Neurotoxicology & Teratology. 2005;27(5):753–767. doi: 10.1016/j.ntt.2005.06.009. [DOI] [PubMed] [Google Scholar]
- 18.Adams AL, Doucette TA, Ryan CL. Altered pre-pulse inhibition in adult rats treated neonatally with domoic acid. Amino Acids. 2008;35(1):157–160. doi: 10.1007/s00726-007-0603-3. [DOI] [PubMed] [Google Scholar]
- 19.Perry W, Miinasian A, Lopez B, Maron L, Lincoln A. Sensorimotor gating deficits in adults with autism. Biol Psychiatry. 2007;61:482–486. doi: 10.1016/j.biopsych.2005.09.025. [DOI] [PubMed] [Google Scholar]
- 20.Canitano R. Epilepsy in autism spectrum disorders. European Child & Adolescent Psychiatry. 2007;16(1):61–66. doi: 10.1007/s00787-006-0563-2. [DOI] [PubMed] [Google Scholar]
- 21.Malow BA. Sleep disorders, epilepsy, and autism. Mental Retardation & Developmental Disabilities Research Reviews. 2004;10(2):122–125. doi: 10.1002/mrdd.20023. [DOI] [PubMed] [Google Scholar]
- 22.Tryphonas L, Truelove J, Iverson F. Acute parenteral neurotoxicity of domoic acid in cynomolgus monkeys (M. fascicularis). [Erratum appears in Toxicol Pathol 1990; 18(3):431] Toxicologic Pathology. 1990;18(2):297–303. doi: 10.1177/019262339001800208. [DOI] [PubMed] [Google Scholar]
- 23.Dakshinamurti KK, Sharma SKS, Sundaram MM. Domoic acid induced seizure activity in rats. Neuroscience Letters. 1991;127(2):193–197. doi: 10.1016/0304-3940(91)90792-r. [DOI] [PubMed] [Google Scholar]
- 24.Ben-Ari Y, Cossart R. Kainate, a double agent that generates seizures: two decades of progress. Trends in Neuroscience. 2000;23(11):580–587. doi: 10.1016/s0166-2236(00)01659-3. [DOI] [PubMed] [Google Scholar]
- 25.Rubenstein JL, Merzenich MM. Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes, Brain, & Behavior. 2003;2(5):255–267. doi: 10.1034/j.1601-183x.2003.00037.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hussman J. Letters to the editor: Suppressed GABAergic inhibition as a common factor in suspected etiologies of Autism. Journal of Autism and Developmental Disorders. 2001;31(2):247–248. doi: 10.1023/a:1010715619091. [DOI] [PubMed] [Google Scholar]
- 27.Bourgeron T. A synaptic trek to autism. Curr Opin Neurobiol. 2009;19(2):231–234. doi: 10.1016/j.conb.2009.06.003. [DOI] [PubMed] [Google Scholar]
- 28.Jones PB, Kerwin RW. Left temporal lobe damage in Asperger's syndrome. The British Journal of Psychiatry. 1990;156(4):570–572. doi: 10.1192/bjp.156.4.570. [DOI] [PubMed] [Google Scholar]
- 29.Marin JCM, Moura PJ, Cysneiros RM, Colugnati DB, Cavalheiro EA, Scorza FA, Xavier GF, Zilbovicius M, Mercadante MT. Temporal lobe epilepsy and social behavior: An animal model for autism? Epilepsy and Behavior. 2008;13(1):43–46. doi: 10.1016/j.yebeh.2008.03.004. [DOI] [PubMed] [Google Scholar]
- 30.Yizhar O, Fenno LE, Prigge M, Schneider F, Davidson TJ, O/'Shea DJ, Sohal VS, Goshen I, Finkelstein J, Paz JT, Stehfest K, Fudim R, Ramakrishnan C, Huguenard JR, Hegemann P, Deisseroth K. Neocortical excitation/inhibition balance in information processing and social dysfunction. Nature. 2011;477(7363):171–178. doi: 10.1038/nature10360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Esclapez M, Hirsch JC, Ben-Ari Y, Bernard C. Newly formed excitatory pathways provide a substrate for hyperexcitability in experimental temporal lobe epilepsy. J Comp Neurol. 1999;408(4):449–460. doi: 10.1002/(sici)1096-9861(19990614)408:4<449::aid-cne1>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- 32.Gill D, Ramsay S, Tasker R. Selective reductions in subpopulations of GABAergic neurons in a developmental rat model of epilepsy. Brain Res. 2010;1331:114–123. doi: 10.1016/j.brainres.2010.03.054. [DOI] [PubMed] [Google Scholar]
- 33.Dakshinamurti K, Sharma SK, Sundaram M, Watanabe T. Hippocampal changes in developing postnatal mice following intrauterine exposure to domoic acid. Journal of Neuroscience. 1993;13(10):4486–4495. doi: 10.1523/JNEUROSCI.13-10-04486.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Scallet AC, Binienda Z, Caputo FA, Hall S, Paule MG, Rountree RL, Schmued L, Sobotka T, Slikker W., Jr Domoic acid-treated cynomolgus monkeys (M. fascicularis): effects of dose on hippocampal neuronal and terminal degeneration. Brain Research. 1993;627(2):307–313. doi: 10.1016/0006-8993(93)90335-k. [DOI] [PubMed] [Google Scholar]
- 35.Levitt P. Disruption of Interneuron Development. Epilepsia. 2005;46(Suppl7):22–28. doi: 10.1111/j.1528-1167.2005.00305.x. [DOI] [PubMed] [Google Scholar]
- 36.Levitt P, Eagleson KL, Powell EM. Regulation of neocortical interneuron development and the implications for neurodevelopmental disorders. Trends in Neurosciences. 2004;27(7):400–406. doi: 10.1016/j.tins.2004.05.008. [DOI] [PubMed] [Google Scholar]
- 37.Blatt GJ, Soghomonian J-J, Yip J. Glutamic Acid Decarboxylase (GAD) as a Biomarker of GABAergic Activity in Autism: Impact on Cerebellar Circuitry and Function. In: Blatt GJ, editor. The Neurochemical Basis of Autism. Springer US; 2010. pp. 95–111. [Google Scholar]
- 38.Rodriguez-Moreno A, Herreras O, Lerma J. Kainate receptors presynaptically downregulate GABAergic Inhibition in the rat hippocampus. Neuron. 1997;19(4):893–901. doi: 10.1016/s0896-6273(00)80970-8. [DOI] [PubMed] [Google Scholar]
- 39.Nitecka L, Tremblay E, Charton G, Bouillot JP, Berger ML, Ben-Ari Y. Maturation of kainic acid seizure-brain damage syndrome in the rat. II. Histopathological sequelae. Neuroscience. 1984;13(4):1073–1094. doi: 10.1016/0306-4522(84)90289-6. [DOI] [PubMed] [Google Scholar]
- 40.Cossart R, Dinocourt C, Hirsch JC, Merchan-Perez A, De Felipe J, Ben-Ari Y, Esclapez M, Bernard C. Dendritic but not somatic GABAergic inhibition is decreased in experimental epilepsy. Nat Neurosci. 2001;4(1):52–62. doi: 10.1038/82900. [DOI] [PubMed] [Google Scholar]
- 41.Morin F, Beaulieu C, Lacaille J-C. Cell-specific alterations in synaptic properties of hippocampal CA1 interneurons after kainate treatment. Journal of Neurophysiology. 1998;80(6):2836–2847. doi: 10.1152/jn.1998.80.6.2836. [DOI] [PubMed] [Google Scholar]
- 42.Best N, Mitchell J, Baimbridge KG, Wheal HV. Changes in parvalbumin-immunoreactive neurons in the rat hippocampus following a kainic acid lesion. Neuroscience Letters. 1993;155(1):1–6. doi: 10.1016/0304-3940(93)90660-d. [DOI] [PubMed] [Google Scholar]
- 43.Esclapez M, Houser CR. somatostatin neurons are a subpopulation of GABA neurons in the rat dentate gyrus: evidence from colocalization of pre-prosomatostatin and glutamate decarboxylase messenger RNAs. Neuroscience. 1995;64:339–355. doi: 10.1016/0306-4522(94)00406-u. [DOI] [PubMed] [Google Scholar]
- 44.Sanon N, Carmant L, Emond M, Congar P, Lacaille J-C. Short-term effects of kainic acid on CA1 hippocampal interneurons differentially vulnerable to excitotoxicity. Epilepsia. 2005;46(6):837–848. doi: 10.1111/j.1528-1167.2005.21404.x. [DOI] [PubMed] [Google Scholar]
- 45.Casanova MF, Buxhoeveden D, Gomez J. Disruption in the inhibitory architecture of the cell minicolumn: implications for autisim. Neuroscientist. 2003;9(6):496–507. doi: 10.1177/1073858403253552. [DOI] [PubMed] [Google Scholar]
- 46.Strain SM, Tasker RA. Hippocampal damage produced by systemic injections of domoic acid in mice. Neuroscience. 1991;44(2):343–352. doi: 10.1016/0306-4522(91)90059-w. [DOI] [PubMed] [Google Scholar]
- 47.Silvagni PA, Lowenstine LJ, Spraker T, Lipscomb TP, Gulland FMD. Pathology of domoic acid toxicity in California sea lions (Zalophus californianus) Veterinary Pathology. 2005;42(2):184–191. doi: 10.1354/vp.42-2-184. [DOI] [PubMed] [Google Scholar]
- 48.Aylward EH, Minshew NJ, Goldstein G, Honeycutt NA, Augustine AM, Yates KO, Barta PE, Pearlson GD. MRI volumes of amygdala and hippocampus in non-mentally retarded autistic adolescents and adults. Neurology. 1999;53(9):2145–2150. doi: 10.1212/wnl.53.9.2145. [DOI] [PubMed] [Google Scholar]
- 49.Dager SR, Wang L, Friedman SD, Shaw DW, Constantino JN, Artru AA, Dawson G, Csernansky JG. Shape mapping of the hippocampus in young children with Autism Spectrum Disorder. American Journal of Neuroradiology. 2007;28(4):672–677. [PMC free article] [PubMed] [Google Scholar]
- 50.Schumann CM, Hamstra J, Goodlin-Jones BL, Lotspeich LJ, Kwon H, Buonocore MH, Lammers CR, Reiss AL, Amaral DG. The amygdala is enlarged in children but not adolescents with autism; the hippocampus is enlarged at all ages. Journal of Neuroscience. 2004;24(28):6392–6401. doi: 10.1523/JNEUROSCI.1297-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Haznedar M, Buchsbaum MS, Wei T-C, Hof PR, Cartwright C, Bienstock CA, Hollander E. Limbic circuitry in patients with autism spectrum disorders studied with positron emission tomography and magnetic resonance imaging. The American Journal of Psychiatry. 2000;157(12):1994–2001. doi: 10.1176/appi.ajp.157.12.1994. [DOI] [PubMed] [Google Scholar]
- 52.Ohnishi T, Matsuda H, Hashimoto T, Kunihiro T, Nishikawa M, Uema T, Sasaki M. Abnormal regional cerebral blood flow in childhood autism. Brain. 2000;123(Pt 9):1838–1844. doi: 10.1093/brain/123.9.1838. [DOI] [PubMed] [Google Scholar]
- 53.Singer T, Seymour B, O'Doherty J, Kaube H, Dolan RJ, Frith CD. Empathy for pain involves the affective but not sensory components of pain. Science. 2004;303(5661):1157–1162. doi: 10.1126/science.1093535. [DOI] [PubMed] [Google Scholar]
- 54.Hansman-Wijnands M, Hummelen J. Differential diagnosis of psychopathy and autism spectrum disorders in adults. Empathie deficit as a core symptom. Tijdschrift voor Psychiatrie. 2006;48(8):627–636. [PubMed] [Google Scholar]
- 55.Ball M, Carmody M, Wynne F, Dockery P, Aigner A, Cameron I, Higgins J, Smith SD, Aplin JD, Moore T. Expression of pleiotrophin and its receptors in human placenta suggests roles in trophoblast life cycle and angiogenesis. Placenta. 2009;30(7):649–653. doi: 10.1016/j.placenta.2009.05.001. [DOI] [PubMed] [Google Scholar]
- 56.Tasker RA, Tracy D. In: Domoic Acid. Seafood and Freshwater Toxins, editor. CRC Press; 2008. pp. 397–429. [Google Scholar]
- 57.Weiss L, Arking D, Daly M, Chakravarti A. A genome-wide linkage and association scan reveals novel loci for autism. Nature. 2009;461:802–808. doi: 10.1038/nature08490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ben-David E, Shifman S. Networks of neuronal genes affected by common and rare variants in autism spectrum disorders. PLoS Genet. 2012:e1002556. doi: 10.1371/journal.pgen.1002556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Voineagu I, Wang X, Johnston P, Lowe J, Tian Y, Horvath S. Transcriptomic analysis of autistic brain reveals convergent molecular pathology. Nature. 2011;474:380–384. doi: 10.1038/nature10110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zikopoulos B, Barbas H. Altered neural connectivity in excitatory and inhibitory cortical circuits in autism. Front Hum Neurosci. 2013;7:609. doi: 10.3389/fnhum.2013.00609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lawrence Y, Kemper T, Bauman M, Blatt G. Parvalbumin-, calbindin-, and calretininimmunoreactive hippocampal interneuron density in autism. Acta Neurol Scand. 2010;121:99–108. doi: 10.1111/j.1600-0404.2009.01234.x. [DOI] [PubMed] [Google Scholar]
- 62.Stoner R, Chow M, Boyle M, Sunkin S, Mouton P, Roy S. Patches of disorganization in the neocortex of children with autism. N Engl J Med. 2014;370:1209–1219. doi: 10.1056/NEJMoa1307491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Berridge M. Dysregulation of neural calcium signaling in Alzheimer disease, bipolar disorder and schizophrenia. Prion. 2013;7:2–13. doi: 10.4161/pri.21767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lewis D, Hashimoto T, Volk D. Cortical inhibitory neurons and schizophrenia. Nat Rev Neurosci. 2005;6:312–324. doi: 10.1038/nrn1648. [DOI] [PubMed] [Google Scholar]
- 65.Allegra M, Genovesi S, Maggia M, Cenni M, Zunino G, Sgado P, Caleo M, Bozzi Y. Altered GABAergic markers, increased binocularity and reduced plasticity in the visual cortex of Engrailed-2 knockout mice. Front Cell Neurosci. 2014;8:163. doi: 10.3389/fncel.2014.00163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Nakai T, Nagai T, Wang R, Yamada S, Kuroda K, Kaibuchi K, Yamada K. Alterations of GABAergic and dopaminergic systems in mutant mice with disruption of exons 2 and 3 of the Disc1 gene. Neurochemistry International. 2014;74:74–83. doi: 10.1016/j.neuint.2014.06.009. [DOI] [PubMed] [Google Scholar]
- 67.Gogolla N, Leblanc J, Quast K, Sudhof T, Fagiolini M, Hensch T. Common circuit defect of excitatory-inhibitory balance in mouse models of autism. J Neurodev Dirsord. 2009;1:172–181. doi: 10.1007/s11689-009-9023-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wohr M, Orduz D, Gregory P, Moreno H, Khan U, Vorckel K, Wolfer D, Welzl H, Gall D, Schiffmann SN, Schwaller B. Lack of parvalbumin in mice leads to behavioral deficits relevant to all human autism core symptoms and related neural morphofunctional abnormalities. Transl Psychatr. 2015;5:e525. doi: 10.1038/tp.2015.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kabir ZD, Kennedy B, Katzman A, Lahvis GP, Kosofsky BE. Effects of prenatal cocaine exposure on social development in mice. Developmental neuroscience. 2014;36(3–4):338–346. doi: 10.1159/000360524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Adams AL, Doucette TA, James R, Ryan CL. Persistent changes in learning and memory in rats following neonatal treatment with domoic acid. Physiology & Behavior. 2009;96(4/5):505–512. doi: 10.1016/j.physbeh.2008.11.019. [DOI] [PubMed] [Google Scholar]
- 71.Burt MA, Ryan CL, Doucette TA. Altered responses to novelty and drug reinforcement in adult rats treated neonatally with domoic acid. Physiology & Behavior. 2008;93(1–2):327–336. doi: 10.1016/j.physbeh.2007.09.003. [DOI] [PubMed] [Google Scholar]
- 72.Todd R, Anderson A. Six degrees of separation: the amygdala regulates social behavior and perception. Nature Neurosci. 2009;12:1217–1218. doi: 10.1038/nn1009-1217. [DOI] [PubMed] [Google Scholar]
- 73.Amaral D. The amygdala, social behavior, and danger detection. Ann NY Acad Sci. 2003;1000:337–347. doi: 10.1196/annals.1280.015. [DOI] [PubMed] [Google Scholar]
- 74.Felix-Ortiz A, Tye K. Amygdala inputs to the ventral hippocampus bidirectionally modulate social behavior. J Neurosci. 2014;34:586–595. doi: 10.1523/JNEUROSCI.4257-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bickart K, Wright C, Dautoff R, Dickerson B, Barrett L. Amygdala volume and social network size in humans. 2011;14:163–165. doi: 10.1038/nn.2724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Moadab G, Bliss-Moreau E, Amaral D. Adult social behavior with familiar partners following neonatal amygdala or hippocampus damage. Behav Neurosci. 2015;129:339–350. doi: 10.1037/bne0000062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zalla T, Sperduti M. The amygdala and the relevance detection theory of autism: an evolutionary perspective. Front Hum Neurosci. 2013;7:894. doi: 10.3389/fnhum.2013.00894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Baron-Cohen S, Ring H, Bullmore E, Wheelwright S, Ashwin C, Williams S. The amygdala theory of autism. Neurosc Biobeh Rev. 2000;24:355–364. doi: 10.1016/s0149-7634(00)00011-7. [DOI] [PubMed] [Google Scholar]
- 79.Tryphonas LL, Iverson FF. Neuropathology of excitatory neurotoxins: the domoic acid model. Toxicologic Pathology. 1990;18(1 Pt 2):165–169. doi: 10.1177/019262339001800122. [DOI] [PubMed] [Google Scholar]
- 80.Teitelbaum J, Zatorre R, Carpenter S, Gendron D, Evans AC, Gjedde A, Cashman N. Neurologic sequelae of domoic acid intoxication due to the ingestion of contaminated mussels. N Engl J Med. 1990;322:1781–1787. doi: 10.1056/NEJM199006213222505. [DOI] [PubMed] [Google Scholar]
- 81.Mullen R, Buck C, Smith A. NeuN, a neuronal specific nuclear protein in vertebrates. Development. 1992;116:201–211. doi: 10.1242/dev.116.1.201. [DOI] [PubMed] [Google Scholar]
- 82.Panksepp J, Jochman K, Jim J, Koy J, Wilson E, Chn Q, Wilson C, Lahvis G. Affiliative behavior, ultrasonic communication and social reward are influenced by genetic variation in adolescent mice. PLoS ONE. 2007;2 doi: 10.1371/journal.pone.0000351. doi10.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Paxinos G, Franklin KBJ. The Mouse Brain in Sterotactic Coordinates. Amsterdam: Elsevier; 2004. [Google Scholar]
- 84.Adams ALA, Doucette TAT, Ryan CLC. Altered pre-pulse inhibition in adult rats treated neonatally with domoic acid. Amino Acids. 2008;35(1):157–160. doi: 10.1007/s00726-007-0603-3. [DOI] [PubMed] [Google Scholar]
- 85.Marriott A, Tasker R, Ryan C, Doucette T. Neonatal domoic acid abolishes latent inhibition in male but not female rats and has differental interactions with social isolations. Neurosci Lett. 2014;578:22–26. doi: 10.1016/j.neulet.2014.06.025. [DOI] [PubMed] [Google Scholar]
- 86.Perl TM, Bédard L, Kosatsky T, Hockin JC, Todd ECD, Remis RS. An Outbreak of Toxic Encephalopathy Caused by Eating Mussels Contaminated with Domoic Acid. New England Journal of Medicine. 1990;322(25):1775–1780. doi: 10.1056/NEJM199006213222504. [DOI] [PubMed] [Google Scholar]
- 87.Gill DA, Bastlund JF, Watson WP, Ryan CL, Reynolds DS, Tasker RA. Neonatal exposure to low-dose domoic acid lowers seizure threshold in adult rats. Neuroscience. 2010;169(4):1789–1799. doi: 10.1016/j.neuroscience.2010.06.045. [DOI] [PubMed] [Google Scholar]
- 88.Ashwin C, Baron-Cohen S, Wheelwright S, O'Riordan M, Bullmore ET. Differential activation of the amygdala and the 'social brain' during fearful face-processing in Asperger Syndrome. Neuropsychologia. 2007;45(1):2–14. doi: 10.1016/j.neuropsychologia.2006.04.014. [DOI] [PubMed] [Google Scholar]
- 89.Sweeten TL, Posey DJ, Shekhar A, McDougle CJ. The amygdala and related structures in the pathophysiology of autism. Pharmacology Biochemistry and Behavior. 2002;71(3):449–455. doi: 10.1016/s0091-3057(01)00697-9. [DOI] [PubMed] [Google Scholar]
- 90.Howard MA, Cowell PE, Boucher J, Broks P, Mayes A, Farrant A, Roberts N. Convergent neuroanatomical and behavioural evidence of an amygdala hypothesis of autism. Neuroreport. 2000;11(13):2931–2935. doi: 10.1097/00001756-200009110-00020. [DOI] [PubMed] [Google Scholar]
- 91.Endo T, Shioiri T, Kitamura H, Kimura T, Endo S, Masuzawa N, Someya T. Altered chemical metabolites in the amygdala-hippocampus region contribute to autistic symptoms of autism spectrum disorders. Biological Psychiatry. 1030;62(9):1030–1037. doi: 10.1016/j.biopsych.2007.05.015. [DOI] [PubMed] [Google Scholar]
- 92.Wolterink G, Daenen LE, Dubbeldam S, Gerrits MA, van Rijn R, Kruse CG, Van Der Heijden JA, Van Ree JM. Early amygdala damage in the rat as a model for neurodevelopmental psychopathological disorders. Eur Neuropsychopharmacol. 2001;11(1):51–59. doi: 10.1016/s0924-977x(00)00138-3. [DOI] [PubMed] [Google Scholar]
- 93.Prather MD, Lavenex P, Mauldin-Jourdain ML, Mason WA, Capitanio JP, Mendoza SP, Amaral DG. Increased social fear and decreased fear of objects in monkeys with neonatal amygdala lesions. Neuroscience. 2001;106(4):653–658. doi: 10.1016/s0306-4522(01)00445-6. [DOI] [PubMed] [Google Scholar]
- 94.Diergaarde L, Gerrits MA, Stuy A, Spruijt BM, van Ree JM. Neonatal amygdala lesions and juvenile isolation in the rat: differential effects on locomotor and social behavior later in life. Behavioral Neuroscience. 2004;118(2):298–305. doi: 10.1037/0735-7044.118.2.298. [DOI] [PubMed] [Google Scholar]
- 95.Wegiel J, Flory M, Kuchna I, Nowicki K, Ma S, Imaki H, Wegiel J, Cohen I, London E, Brown W, Wisniewsky T. Brain-region-specific alterations of the trajectories of neuronal volume growth throughout the lifespan of autism. Acta Neuropathol Commun. 2014;2:28. doi: 10.1186/2051-5960-2-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lin H-C, Gean P-W, Wang C-C, Chan Y-H, Chen P. The amygdala excitatory/inhibitory balance in a valproate-induced rat autism model. PLOSOne. 2013;8:e55248. doi: 10.1371/journal.pone.0055248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Nakamura T, Matsumoto J, Takamura Y, Ishii Y, Sasahara M, Ono T, Nishijo H. Relationships among parvalbumin-immunoreactive neuron density, phase-locked gamma oscillations, and autistic/schizophrenic symptoms in PDGFR-b knock-out and control mice. PLOSOne. 2015;10:e0119258. doi: 10.1371/journal.pone.0119258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ouellette-Kuntz H, Coo H, Yu CT, Chudley AE, Noonan A, Breitenbach M, Ramji N, Prosick T, Bedard A, Holden JJA. Prevalence of pervasive developmental disorders in two Canadian provinces. Journal of Policy and Practice in Intellectual Disabilities. 2006;3(3):164–172. [Google Scholar]
- 99.Kim YS, Leventhal BL, Koh YJ, Fombonne E, Laska E, Lim EC, Cheon KA, Kim SJ, Kim YK, Lee H, Song DH, Grinker RR. Prevalence of autism spectrum disorders in a total population sample. American Journal of Psychiatry. 2011;168(9):904–912. doi: 10.1176/appi.ajp.2011.10101532. [DOI] [PubMed] [Google Scholar]
- 100.Choi KD, Lee JS, Lee JO, Oh KS, Shin IS. Investigation of domoic acid in shellfish collected from Korean fish retail outlets. Food Science and Biotechnology. 2009;18(4):842–848. [Google Scholar]
- 101.Waldman M, Nicholson S, Adilov N, Williams J. Autism Prevalence and Precipitation Rates in California, Oregon, and Washington Counties. Archives of Pediatrics Adolescent Medicine. 2008;162(11):1026–1034. doi: 10.1001/archpedi.162.11.1026. [DOI] [PubMed] [Google Scholar]