Abstract
A mutation in an inbred line of petunia (Petunia hybrida) produces a reduction in the deep-purple corolla pigmentation and changes the anther color from yellow to white. In addition, the mutant, designated white anther (wha), is functionally male sterile. The inability of pollen from wha plants to germinate in vitro provides a physiological basis for the lack of seed set observed in self-crosses of the mutant. Biochemical complementation with nanomolar amounts of kaempferol, a flavonol aglycone, confirms that the inability of the wha pollen to germinate is due to a lack of this essential compound. Transgenic complementation with a functional ChsA (Chalcone synthase A) cDNA suggests that the genetic lesion responsible for the wha phenotype is in Chs, the gene for the first enzyme in the flavonol biosynthesis pathway. The genetic background of the parental line, as well as the pollen phenotype, allowed us to deduce that the wha mutation is in ChsA. To our knowledge, wha is the first induced, nontransgenic Chs mutant described in petunia, and analysis of the mutation confirms earlier molecular and genetic observations that only two Chs genes (A and J) are expressed in reproductive tissues and that they are differentially regulated in corolla and anther.
Flavonoids are plant-specific compounds that accumulate in virtually all tissues of plants, ranging from mosses to angiosperms (Koes et al., 1994). They are classified according to the oxidation state of the central pyran ring. A variety of functions have been attributed to the different classes of flavonoids, e. g. the red and blue anthocyanin pigments in combination with UV-absorbing flavonol copigments act as attractants for insect pollinators (Harborne, 1976; Stafford, 1990; Koes et al., 1994). More recently, flavonols, particularly kaempferol and quercetin (Vogt et al., 1995), have been shown to be essential for pollen germination and tube growth in petunia (Petunia hybrida) and maize (Mo et al., 1992; Taylor and Jorgensen, 1992; Pollak et al., 1995).
The flavonoid pathway in petunia, as in many other species, has been well characterized, and many structural and regulatory genes have been identified (Dooner et al., 1991; Martin, 1993; Forkmann, 1994). Sequences encoding Chs (Chalcone synthase), the gene encoding the first enzyme in flavonoid synthesis, have been isolated and characterized from a variety of plant sources (Dooner et al., 1991). In petunia 8 to 10 copies of Chs sequences have been identified in the genome of the inbred line V30 (Koes et al., 1989b). RNase protection analyses with gene-specific probes have shown that only two genes, ChsA and ChsJ, are expressed in petunia anthers and corollas (Koes et al., 1989a) and that ChsJ transcripts are from 5% to 20% of the ChsA levels. In addition, several regulatory loci have been identified, which transcriptionally control expression of the flavonoid structural genes in a temporal or tissue-specific manner. One of these genes, An4, controls flavonoid production in the anthers by regulating ChsJ but not ChsA (Quattrocchio et al., 1993). Results of RNase protection assays suggested that plants that are homozygous recessive at an4 express both ChsA and ChsJ transcripts in corollas, but anther tissue accumulates ChsA mRNA exclusively (Quattrocchio et al., 1993).
Because of the visible and nonessential nature of anthocyanins, mutations in flavonoid pathway genes are easily recognized and maintained. In contrast to maize and Arabidopsis, in which spontaneous and induced Chs mutations have been described (Dooner et al., 1991), no such Chs mutations have been identified in petunia. However, there are several examples of homology-dependent suppression of Chs transcription in petunia (Napoli et al., 1990; van der Krol et al., 1990). Transgenic plants suppressed for Chs in anthers were instrumental in identifying an essential role for flavonols in pollen function (Mo et al., 1992; Taylor and Jorgensen, 1992; Ylstra et al., 1994). The lack of CHS protein in both petunia and maize anthers results in white pollen that is devoid of all flavonols and is unable to germinate or produce a functional pollen tube in self-pollinations (Mo et al., 1992; Pollak et al., 1993). However, when this flavonol-deficient pollen was crossed to wild-type stigmas, the pollen germinated and seeds were produced, thereby demonstrating that the reproductive defect is conditional. Plants expressing this phenotype were designated CMF by Taylor and Jorgensen (1992). The bioactive compound from wild-type petunia stigmas was identified as kaempferol, a flavonol aglycone. Although the mechanism of flavonol induction of pollen germination is unknown, biochemical complementation of CMF pollen (pollen rescue) established that the response to exogenously added flavonols was sensitive (maximum germination at 0.4 μm) and specific for flavonol aglycones (Vogt et al., 1995).
Here we describe a novel male-sterile mutant that was found in an M2 population derived from EMS-mutagenized V26 petunia seeds. This mutant displays both a reduction in the purple color of the corolla and produces white, rather than wild-type yellow, pollen. Pollen from the mutant was nonfunctional in self-crosses. The similarity of this EMS-induced phenotype to the transgene-induced CMF phenotype suggested a mutation at a Chs gene. In this report we describe the genetic and biochemical characterization of the reproductive tissues of the wha (white anther) mutant and show that the mutation is complemented by a functional ChsA cDNA.
MATERIALS AND METHODS
Plant Materials and Plant Culture
The EMS mutagenesis, plant materials, and the greenhouse culture conditions were as described by Napoli and Ruehle (1996). Flower stages used for biochemical and molecular analysis are defined by bud length and morphology as described by Pollak et al. (1993). The mutant population used for the pollen rescue experiments was a segregating F2 population derived from a backcross of the mutant to V26. V26 is an inbred line, which is referred to as the wild type in this study.
Plant Transformation
Axenic explants of mutant plants were prepared from axillary buds by the following procedure: Axillary buds were removed from a greenhouse-grown wha plant and surface-disinfected for 20 min with 20% bleach. Buds were washed with five changes of sterile deionized water and then placed on the surface of half-strength Murashige and Skoog agar (Jorgensen et al., 1996) supplemented with 2 μm indole butyric acid for rooting. Young leaf pieces from these axenic plants were used for the transformation. The transformation binary vector, pDVS680, was kindly provided by Drs. Qiudeng Que and Richard Jorgensen (Que et al., 1997). The transformation vector has a neomycin phosphotransferase II selectable marker and a functional cDNA clone for chalcone synthase (ChsA) under the control of the CaMV 35S promoter.
Agrobacterium tumefaciens-mediated plant transformation was carried out as described by Jorgensen et al. (1996), except for the following changes: Explants were grown on one-half-strength Murashige and Skoog salts and 2% Suc; acetosyringone was omitted from the cocultivation medium; after removal of the plates from dark incubation, the explants were transferred to fresh selection/regeneration medium; the concentration of carbenicillin was decreased to 200 μg/mL; and adventitious shoots were transferred to magenta boxes, not plates, for rooting. The kanamycin assay to confirm the identity of transformed shoots was performed on 0.5 μm IAA and 0.5 μm benzoaminopurine.
In Vitro Pollen Germination and Rescue
In vitro pollen germination assays and flavonol-induced germination were performed as described by Mo et al. (1992) and Xu et al. (1997).
HPLC of Anther and Corolla Extracts
Extracts for HPLC were prepared by soaking 10 anthers or 50 mg of corolla tube (stage 9) in 250 μL of 100% (v/v) methanol for 2 h at room temperature on a rotary shaker at 100 rpm. To convert flavonol glycosides to flavonol aglycones, 200 μL of centrifuged extract was mixed with 200 μL of 4 n HCl and heated at 50°C for 45 min. An additional 200 μL of methanol was added to prevent precipitation of flavonol aglycones, the extract was centrifuged, and an aliquot of the supernatant was separated by HPLC.
HPLC analysis was performed using a Millennium 2.1 chromatographic workstation (Waters) equipped with a dual-pump system (model 600, Waters) and photodiode array detector (model 996, Waters). Chromatographic separations were accomplished with a 0- to 15-min linear gradient of 100% solvent A (10% [v/v] acetonitrile in water and 0.04% [v/v] trifluoroacetic acid) to 100% solvent B (100% [v/v] acetonitrile and 0.04% [v/v] trifluoroacetic acid). The flow rate was 1.5 mL min−1 on a C18 reversed-phase matrix (150- × 3.9-mm i.d., particle size 4 mm, Nova-pak, Waters). Flavonols were detected at 365 nm. Kaempferol and quercetin were identified by retention time and spectral comparisons with authentic standards.
Anthocyanin Quantitation
Anthocyanins were extracted by soaking 40 mg of corolla tube (stage 9) in 2.5 mL of 1% HCl. The A542 of the extract was measured and quantified by comparison with a delphinidin chloride standard.
Immunoblotting
Anther proteins (stage 4–6) were isolated by grinding 50 anthers in 500 μL of ice-cold 200 mm Tris, pH 7.8, 0.5 mm DTT, and 10 mg mL−1 polyvinylpolypyrrolidone and centrifuging to recover soluble proteins in the supernatant. Corolla proteins (stage 6) were isolated according to the method of Schuster and Davies (1983), and the final pellet was dissolved in 100 mm Tris, pH 7.8, and 0.5% SDS. The protein concentration was estimated by the method of Bradford (1976). Samples, containing 40 μg of protein, were separated by SDS-PAGE in a 12% gel. Purified maize CHS protein (0.1 μg) was used as a standard (Pollak et al., 1993). Proteins were electroblotted to nitrocellulose, and the blot was immunostained as described by Pollak et al. (1993).
RNA-Blot Analysis
Anthers (stage 4–6) and corolla tube tissue (stage 4–5) were ground to a fine powder in liquid N2, and total RNA was extracted using Trizol (GIBCO-BRL) according to the manufacturer's instructions. Northern analysis was performed on samples containing 5 to 10 μg of RNA, as described by Taylor and Briggs (1990). Hybridization probes were isolated from the following petunia (Petunia hybrida) sequences: pCP8, a Chs cDNA (kindly provided by Hans-Jörg Reif, Bayer AG, Cologne, Germany), a 945-bp EcoRI cDNA fragment of flavanone 3-O hydroxylase (F3h; Britsch et al., 1992), a cDNA for flavonol synthase, and pGAP, a cDNA for a cytoplasmic GAP. The latter two cDNAs were isolated from a CMF anther stage-6 library (λZiplox, GIBCO-BRL) by way of heterologous probing using a potato Fls (flavonol synthase) cDNA (van Eldik et al., 1997) and a maize cytoplasmic GAP cDNA (Brinkmann et al., 1987). The identity of each cDNA was confirmed by sequence identity to relevant sequences in the EMBL data bank (L.P. Taylor, unpublished data). Hybridization of the RNA blots to the GAP probe or to a 400-bp EcoRI fragment of the rDNA gene from Petunia inflata (Mu et al., 1994) was used to calculate loading differences. The autoradiographic images were scanned, and signal intensity was quantified using the NIH Image program (National Institutes of Health, Bethesda, MD).
RESULTS
Identification of the Mutant Phenotype
Mutagenesis with EMS was carried out using the P. hybrida genetic stock V26 (Napoli and Ruehle, 1996). Mutant plants with an alteration in corolla pigmentation, as well as a loss of anther pigmentation, were found in 1 of approximately 1200 M2 families. The loss of yellow pigmentation resulted in white anthers; therefore, the mutant was designated wha. The results of a genetic analysis using F2 progeny derived from a backcross to the wild type are consistent with wha being a single, recessive allele (232 wild type:74 mutant; χ2 = 0.11; P = 0.74). Whereas self-pollinations in a homozygous recessive wha background were not successful, reciprocal crosses of mutant (wha) and wild-type plants resulted in the production of viable seeds (Table I). The success of the reciprocal crosses demonstrates that the fertility problem associated with the wha mutation can be complemented. However, as shown in Table I, reciprocal crosses between the wild type and the mutant were not consistently successful, and capsules were produced only 55% of the time using wha plants as the male and 68% of the time in the case when the wha plant was the female.
Table I.
Results of reciprocal crosses with mutant and wild-type plants
| Female | Male | Pollination Attempts | Capsules Produced | Capsules Formed |
|---|---|---|---|---|
| % | ||||
| WTa | WT | 25 | 25 | 100 |
| wha | wha | 52 | 0 | 0 |
| WT | wha | 44 | 24 | 55 |
| wha | WT | 19 | 13 | 68 |
WT, inbred line V26.
Rescue of Pollen Germination
The similarity of pollen expressing the wha phenotype to the transgene-induced CMF phenotype reported by Taylor and Jorgensen (1992) suggested that the reproductive defect might be due to an inability of wha pollen to germinate because of an absence of flavonols. When wild-type pollen was placed in germination medium, up to 80% to 85% of the grains germinated and extruded a tube, which grew for several hours (Table IV, line 1). The observation that flavonol-deficient pollen such as CMF hydrates, but does not germinate unless provided with an exogenous source of flavonols (Mo et al., 1992), led us to propose that pollen from wha plants may be flavonol deficient. This hypothesis was tested by adding kaempferol, a flavonol aglycone, to an in vitro suspension of the wha pollen grains in germination medium or to the stigma at the time of wha pollination (Mo et al., 1992; Vogt et al., 1994). We found that in vitro pollen germination and seed production in self-crosses of plants expressing the wha phenotype required flavonol addition (Table II). However, within the population of 38 homozygous recessive mutant plants that were analyzed in depth, two groups were identified based on their response during the initial stages of hydration and germination. When placed in nonsupplemented germination medium, less than 5% of the pollen grains from wha plants designated as class 1 initiated tube outgrowth, whereas up to 20% of the grains from class 2 wha mutants germinated. A pollen grain was scored as germinated if the tube length exceeded the pollen grain diameter. However, after the initial outgrowth, tube elongation ceased in both class 1 and 2 plants and never exceeded 5 pollen-grain diameters in the absence of exogenous kaempferol. Although the initial response to in vitro conditions differed, both classes showed a similar in vitro pollen rescue frequency of about 75% when kaempferol was added to the germination medium at a concentration of 1 μm. Tube outgrowth continued throughout a 4-h observation period and was similar in extent to wild-type pollen.
Table IV.
Flavonoid content of anthers and corollas of mutant, wild-type, and complemented petunia
| Plant | Corolla
|
Anther
|
Pollen Analysis
|
||||
|---|---|---|---|---|---|---|---|
| Quercetin | Kaempferol | Anthocyanin | Quercetin | Kaempferol | Germination | Rescue | |
| pmol/mg fresh wt | pmol/anther | % | |||||
| Wild type | 1849 | 415 | 17.0 × 103 | 3514 | 1735 | 81 | 86 |
| wha 6.1 | 186 | 26 | 5.8 × 103 | 0 | 0 | 5 | 76 |
| CMF | 0 | 0 | 0 | 0 | 0 | 6 | 81 |
| Chs8705.3 | 390 | 85 | 14.4 × 103 | 0 | 0 | 8 | 61 |
| Chs8705.5 | 582 | 98 | 11.0 × 103 | 4670 | 2843 | 75 | 72 |
| Chs8705.6 | 1567 | 271 | 11.6 × 103 | 3190 | 1270 | 70 | 76 |
Table II.
Analysis of pollen function of 38 homozygous recessive wha plants
| Class | Germinationa
|
wha × wha
|
wha × V26 | ||
|---|---|---|---|---|---|
| −Flavonol | +Flavonolb | −Flavonol | +Flavonol | ||
| In vitro germination | % | ||||
| 1 | <5 | >75 | – | – | |
| 2 | 5–20 | >75 | – | – | |
| Seed setc | |||||
| 1 | – | – | 0 | 14 | 53 |
| 2 | – | – | 0 | 21 | 50 |
Data represent the means of at least 5 independent analyses.
1 μm kaempferol.
Plants producing full-seed capsules.
The lack of pollen tube growth in the absence of exogenous flavonols was confirmed in planta. In more than 75 self-crosses of plants homozygous for the wha mutation, no seed was set. However, when the mutants were self-crossed in the presence of added kaempferol, 14% of the crosses of class 1 plants produced seed capsules, and class 2 plants, which showed a higher frequency of early tube outgrowth, produced 21% successful self-crosses. More than 50% of the crosses of wild-type pollen to wha stigmas from both class 1 and class 2 were successful, confirming that the lack of seed set in the nonsupplemented self-crosses was due to nonfunctional pollen.
Complementation of the wha Mutation
The phenotypic similarities between the wha mutant and the transgenic CMF phenotype, as well as the genetic background of V26, suggested the possibility that wha was a mutation in a Chs gene. Despite the fact that two different Chs transcripts accumulate in petunia corolla tissue (Koes et al., 1989a), only one Chs gene should function in V26 pollen. This is because V26 is homozygous recessive for an4, a gene that is required for ChsJ, but not ChsA, expression in anther tissue. Thus, a Chs mutation in the V26 background should result in loss of pigmentation in anthers. However, we wanted to rule out the possibility that the wha mutation mapped to other genetic loci involved in flavonol biosynthesis, e.g. either the Fls locus, which controls the synthesis of flavonols (quercetin and kaempferol), or the An3 locus, which controls the synthesis of dihydroflavonols (Forkmann, 1994). Genetic allelism tests were carried out with EMS-induced mutations in the V26 background that had been previously characterized as fls recessive or an3 recessive (C. Napoli, unpublished data). The wha mutation complemented both fls and an3 mutant phenotypes, because the F1 progeny of each outcross produced phenotypically wild-type flowers, e.g. yellow, functional pollen and dark-violet corollas.
Confirmation that the wha phenotype resulted from a Chs mutation was obtained by transgenic complementation of the mutant using a ChsA cDNA expressed from a CaMV 35S promoter (Que et al., 1997). A total of 63 transgenic plants homozygous recessive for wha were produced from two independent transformation experiments. Corolla pigmentation was evaluated visually for the entire population. Three classes of phenotypes were observed and the data are reported in Table III. Three-quarters (47 plants) of the 63 transgenotes had corolla pigmentation complementation, as evidenced by either a phenotype indistinguishable from the wild type or one intermediate between the mutant and the wild type. Complementation in the anthers did not occur as frequently as corolla complementation; only 34 of the 47 transgenotes had a restoration of yellow anther pigmentation. A second class of plants comprised 8 transgenotes with white flowers and anthers, resulting most likely from transgene silencing that is common to introduced copies of Chs. The third class included 8 transgenotes that retained the wha plant phenotype, showing no complementation in either the corolla or anther pigmentation. The latter two classes were not analyzed.
Table III.
Results of transgenic complementation of the wha mutant with a ChsA cDNA
Percentages are based on two experiments with a total of 63 transformed plants.
The corolla pigmentation phenotype was visually the same as that of the wild type.
The corolla pigmentation phenotype was intermediate in color between the wha mutant and the wild type.
The corolla pigmentation phenotype was visually the same as that of the wha mutant.
Quantitative Flavonoid Analyses
To determine whether the levels of flavonols and anthocyanins, the major classes of flavonoids that accumulate in the anthers and the corollas of petunia, were restored in the complemented wha transgenotes, HPLC and spectrophotometric analyses were performed on methanol extracts of these tissues from three transformants, Chs8705.3, Chs 8705.5, and Chs8705.6. For a comparison the amount of these compounds was also determined for the wha progenitor, the wild type, and a cosuppressed CMF mutant (Taylor and Jorgensen, 1992). The data are shown in Table IV. Neither the EMS-induced chs mutant (wha) nor the cosuppressed chs mutant (CMF) accumulated flavonols in the anthers. This result shows that the inability of the pollen to germinate or set seed is strictly correlated with a lack of flavonols in pollen (Table II). On the other hand, flavonols and anthocyanins are produced in wha corollas, although the levels are reduced (5%–10% and approximately 30%, respectively) when compared with the wild type. This result suggests that altered substrate availability resulting from suppression of CHS activity in wha corollas limits flavonol production more than anthocyanin.
Flavonol levels in the anthers of two of the complemented transgenotes (Chs8705.5 and Chs8705.6) were virtually the same as wild-type levels. Accompanying this accumulation of flavonols was a restoration of pollen germination frequency to wild-type levels (Table IV). Flavonol and anthocyanin levels in the corollas of all three transgenotes increased but showed more variation than the increases in pollen flavonols. Although anthocyanin levels reached only 65% to 82% of wild-type levels, the complemented corollas were visually indistinguishable from the deep-purple wild-type corollas. This indicates that visual estimates of pigmentation are not reliable at high anthocyanin concentrations.
It is interesting that the third transgenote, Chs8705.3, exhibited complementation in the corolla but not in the anther. Phenotypically, anther color reflected the restoration of flavonol production; transgenotes Chs8705.5 and Chs8705.6 had yellow anthers and Chs8705.3 retained the mutant white anthers. Of these three transgenotes, Chs8705.6 produced one capsule in three self-pollinations, Chs8705.5 set seed in two of three self-pollinations, and Chs8705.3 produced no capsules from three self-pollination attempts.
mRNA Accumulation of Flavonoid Biosynthetic Genes in Anthers and Corollas
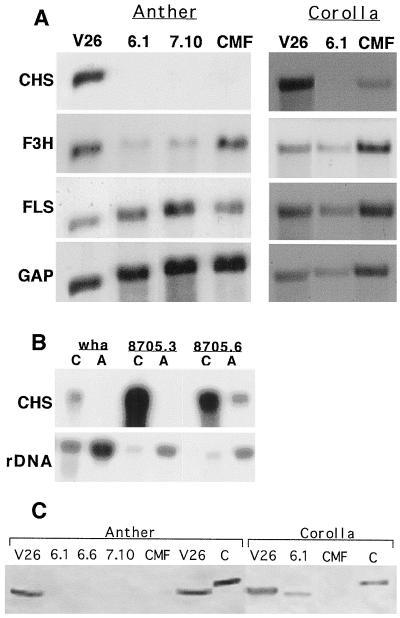
To confirm the biochemical analysis, steady-state levels of Chs mRNA were compared in reproductive tissues from mutant, wild-type, and cosuppressed CMF plants (Taylor and Jorgensen, 1992). The data are shown in Figure 1A with the relative intensities of the hybridization signal corrected for loading differences listed in the figure legend. No Chs-homologous transcripts were detected in the anthers of two different sibling plants homozygous recessive for wha (6.1 and 7.10, both class 1 plants) or the CMF mutant. Extremely low levels (about 3% of wild-type levels) accumulated in the corolla (Fig. 1A). This pattern reflects the accumulation of flavonols and anthocyanins in these tissues, suggesting that transcriptional control is chiefly responsible for flavonoid accumulation. The Chs transcript detected in the nonpigmented CMF corollas (approximately 7% of wild-type levels) was noted previously (Napoli et al., 1990).
Figure 1.
Expression of flavonoid transcripts and CHS protein in anthers and corollas of wha and transgenic complemented wha plants. A, Total RNA was extracted from stage 4-6 anthers or corolla tissue, electrophoresed in formaldehyde agarose gels, transferred to nylon filters, and hybridized to 32P-labeled inserts of Chs, F3h, and Fls cDNA clones from petunia. The correction for loading differences was calculated following hybridization with a GAP cDNA probe: B, Ten micrograms of anther total RNA from two complemented transgenotes and the parental wha line was hybridized to a Chs probe. The rDNA hybridization pattern was used to detect loading differences. C, Western analysis of CHS protein in corollas and anthers. A recombinant maize CHS protein (lane C) was included as a positive control (Pollak et al., 1993).
The expression of two other flavonoid biosynthesis genes was also determined. F3h acts downstream of Chs to convert flavanones to dihydroflavonols, which are precursors of both flavonols and anthocyanins. Fls converts dihydroflavonols to flavonols and is not involved in anthocyanin production. F3h expression in the corolla was not affected by the wha mutation but was decreased in anthers from both the wha and CMF mutant plants. The level of the F3h transcript in wha anthers was 10% that of the wild type, and the cosuppressed CMF mutant had a 50% reduction (relative to the wild type) of F3h mRNA levels. On the other hand, Fls expression was not affected in the corolla or anthers of either mutant.
Two transgenotes, Chs8705.3 and Chs8705.6, were examined by northern analysis to determine whether an increase in Chs transcripts correlated with complementation of pigmentation and germination of the pollen and pigmentation in the corolla. The wha mutant parent was included as a control (Fig. 1B). Based on the biochemical analysis and germination results, transgenote Chs8705.6 was complemented in both the anther and the corolla, which was confirmed by the detection of Chs-homologous transcripts in both organs. Transgenote Chs8705.3 showed differential complementation; it accumulated flavonols and anthocyanins in the corolla but the pollen remained devoid of flavonols and was nonfunctional (Table IV). As expected, no Chs-homologous transcripts were detected in anthers but high steady-state levels were present in the corolla.
CHS Protein Accumulation in wha Reproductive Tissues
The accumulation of CHS protein was determined in anthers and corollas by western analysis using an antibody raised against a maize recombinant CHS protein. This antibody was previously shown to cross-react with petunia CHS protein (Pollak et al., 1993). No CHS protein was detected in extracts from stage-4 to -6 anthers from three different wha sibling plants or the CMF control extract (Fig. 1C). On the other hand, a corolla extract from plants homozygous recessive for wha contained CHS protein but the level was reduced compared to the wild type. Thus, CHS protein levels in the mutants and wild-type extracts of anthers and corollas reflect the expression of Chs mRNA and the accumulation of flavonoid compounds in these tissues. It also confirms that both ChsJ and ChsA transcripts translate into functional protein.
DISCUSSION
A combination of molecular and biochemical data supports the hypothesis that the wha mutation is in ChsA. Furthermore, our results provide genetic confirmation that, in the absence of a functional An4 allele, ChsJ is not expressed in anthers. Koes et al. (1989a) suggested that the two Chs genes expressed in petunia anthers were differentially regulated: Expression of ChsJ, but not ChsA, was controlled by An4. Thus, our use of the V26 inbred as the progenitor line for the EMS mutagenesis was a fortuitous choice. In a recessive an4 genetic background such as V26, a chsA mutation should result in flavonoid-deficient anthers containing viable pollen that is unable to germinate unless supplemented with kaempferol, a flavonol aglycone. Furthermore, because ChsJ is not regulated by An4 in corollas, it would be expressed at its normal level (3% of that of ChsA), resulting in corollas with reduced anthocyanin pigmentation. Thus, the phenotype of the wha mutant reflects a mutation of ChsA and the differential expression of ChsJ in the relevant tissues of the wild type.
The mutation could have been mapped using restriction fragment-length polymorphism markers, but given that mapping indicates only linkage and petunia shows extensive regions of recombination suppression (Robbins et al., 1995), we believed a more straightforward approach would be to use transgenic complementation. Clearly, our results show that introduction and expression of a ChsA cDNA completely restores flavonoid production and pollen germination to the wha mutant. Additional evidence that wha plants require a functional Chs gene for restoration of wild-type pigmentation comes from Que et al. (1997), who engineered a frame-shift mutation in the ChsA cDNA with the intent to prematurely terminate transcription. When the wha mutant was transformed with the engineered frame shift, the altered cDNA failed to complement the mutation. The molecular and biochemical basis for differential complementation seen in 28% of the transgenotes with wild-type corolla pigmentation (e.g. Chs8705.3) bears investigation. It suggests that the CaMV 35S promoter may not be as active in anthers as corollas, a suggestion that is supported by the lack of anther transformation observed in numerous antisense experiments using the CaMV 35S promoter.
The characterization of the wha mutant confirmed previous molecular data that suggested that only two of the eight Chs genes of petunia were expressed in reproductive tissue. Using RNase protection assays to distinguish transcripts, Koes et al. (1989a) estimated that ChsJ contributed about 10% to the total Chs expression in anthers, tube, and corolla with ChsA providing the bulk. Analyzing wha plants, which express only ChsJ, we detected Chs transcripts in the corolla at levels approximately 3% of that found in wild-type corollas, which express both ChsA and ChsJ. This level of expression is limiting for both flavonol and anthocyanin production but is more limiting for the former class, as evidenced by the different effects on flavonol and anthocyanin accumulation shown in Table IV. This may indicate that the dihydroflavonol reductase enzyme involved in anthocyanin production is more efficient than flavonol synthase at competing for the common substrate dihydroflavonol. The wha mutant also provides an opportunity to determine the effect on pollen germination of reducing flavonol content in pollen. This is accomplished by introgressing a functional An4 allele into the wha homozygous recessive background and selecting for the appropriate corolla and anther pigmentation. This genotype would express ChsJ in anthers but at 3% of the levels of mRNA as the wild type. Whether the expected lower levels of pollen flavonols would be sufficient for the pollen to function awaits analysis of these plants.
An interesting observation was the decreased expression in anthers, but not corollas, of a flavonoid biosynthesis gene, F3h, in the absence of Chs activity. In maize Chs mutations can also have an effect on other genes in the flavonoid pathway; transcripts from the A1 (DFR) and Bz1 (UFGT) genes are altered in C2-Idf plants (U. Weinand, personal communication). F3h acts downstream of Chs to oxidize flavanones to dihydroflavonols, a class characterized by a hydroxyl group at the C3 position of the C-ring. This 3-hydroxyl group is the major site for glycosylation of pollen flavonols, converting them from potentially cytotoxic compounds to a water-soluble form, which can be sequestered (Vogt et al., 1994; Xu et al., 1997). F3h expression in anthers may be controlled by substrate availability. In maize anthers F3h has been shown to control flavonol production in a stage-specific manner (Deboo et al., 1995). All other flavonol biosynthesis genes are constitutively expressed or are transcriptionally activated following the appearance of the F3h transcript at the uninuclear stage.
Rescue of pollen germination is extremely sensitive to small amounts of flavonol; tube outgrowth can be detected after adding as little as 0.4 μm kaempferol (Mo et al., 1992; V. Guyon, unpublished data). The differences in the percentage of germination and rescue (Table II) suggest that there is variation in the expression of the wha phenotype in a population of the mutants. This may reflect differences in the penetrance of the mutation, since the EMS-induced change is most likely a point mutation that might be affected by environmental influences. However, the absolute amount of flavonols in any one plant is likely to be quite small because no flavonols were detected by HPLC (detection limit 5 pmol). In any event, the few pollen tubes that formed were nonfunctional, since no seed was ever produced from mutant self-crosses.
Although 70% to 80% of the pollen grains from wha mutants could be successfully rescued in vitro (Tables II and IV), seed set was often lower than expected in both kaempferol-supplemented self-crosses and reciprocal crosses to wild-type stigmas (Tables I and II). The basis of this reduced fecundity is not known, but it should be noted that the highly inbred parental line V26 exhibits variable fertility, which may respond to environmental fluctuations. Vogt et al. (1994) were able to increase seed set in self-crosses of V26 by more than 100% by wounding the flower 24 h before pollinating. Wounding was accompanied by a dramatic increase in stigmatic kaempferol levels.
CHS is the first enzyme in flavonoid biosynthesis, and an alteration in CHS activity would be expected to affect accumulation of all classes of these compounds. Although both spontaneous and chemically induced mutations in Chs have been characterized in maize and Arabidopsis, to our knowledge, this is the first nontransgenically induced Chs mutant described in petunia. Although cosuppressed plants (CMF) were useful in establishing the pollen germination requirement for flavonols, these plants are phenotypically unstable and revert to coexpression (restoration of pigmentation) in certain branches on older plants (Jorgensen et al., 1996). Thus, a nontransgenic Chs mutant in petunia is extremely useful as the progenitor of transgenic plants to test various gene functions during pollen development and germination in the absence of flavonols. They will be especially useful for testing whether flavonoid regulatory genes respond to product accumulation or for determining the role of flavonoid-modifying activities in cellular processes (Vogt and Taylor, 1995).
ACKNOWLEDGMENTS
L.P.T. is thankful for the valuable contribution of Michaela Witcher for the experiments presented in Table II as part of a Howard Hughes undergraduate research project at Washington State University, Pullman.
Abbreviations:
- CaMV
cauliflower mosaic virus
- CMF
conditionally male fertile
- EMS
ethane methanesulfonate
- GAP
glyceraldehyde phosphate dehydrogenase
Footnotes
This work was supported by grants from the National Science Foundation (no. IBN-9507082 to C.A.N. and no. IBN-9405361 to L.P.T.).
LITERATURE CITED
- Bradford M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Brinkmann H, Martinez P, Quigley F, Martin W, Cerff R. Endosymbiotic origin and codon bias of the nuclear gene for chloroplast glyceraldehyde-3-phosphate dehydrogenase from maize. J Mol Evol. 1987;26:320–328. doi: 10.1007/BF02101150. [DOI] [PubMed] [Google Scholar]
- Britsch L, Ruhnau-Britch B, Forkmann G. J Biol Chem. 1992;267:5380–5387. [PubMed] [Google Scholar]
- Deboo GB, Albertsen MC, Taylor LP. Flavanone 3-hydroxylase and flavonol accumulation are temporally coordinate in maize anthers. Plant J. 1995;7:703–713. doi: 10.1046/j.1365-313x.1995.07050703.x. [DOI] [PubMed] [Google Scholar]
- Dooner HK, Robbins TP, Jorgensen RA. Genetic and developmental control of anthocyanin biosynthesis. Annu Rev Genet. 1991;25:173–199. doi: 10.1146/annurev.ge.25.120191.001133. [DOI] [PubMed] [Google Scholar]
- Forkmann G. Biosynthesis of flavonoids. In: Harborne JB, editor. The Flavonoids: Advances in Research Since 1986. London: Chapman & Hall; 1994. pp. 499–536. [Google Scholar]
- Harborne JB. Functions of flavonoids in plants. In: Goodwin TW, editor. Chemistry and Biochemistry of Plant Pigments, Ed 2, Vol 1. London: Academic Press; 1976. pp. 736–774. [Google Scholar]
- Jorgensen RA, Cluster PD, English J, Que Q, Napoli CA. Chalcone synthase cosuppression phenotypes in petunia flowers: comparison of sense versus antisense constructs and single copy versus complex T-DNA sequences. Plant Mol Biol. 1996;31:957–973. doi: 10.1007/BF00040715. [DOI] [PubMed] [Google Scholar]
- Koes RE, Quattrocchio F, Mol JNM. The flavonoid biosynthetic pathway in plants: function and evolution. Bioessays. 1994;16:123–132. [Google Scholar]
- Koes RE, Spelt CE, Mol JNM. The chalcone synthase multigene family of Petunia hybrida (V30): differential, light-regulated expression during flower development and UV light induction. Plant Mol Biol. 1989a;12:213–225. doi: 10.1007/BF00020506. [DOI] [PubMed] [Google Scholar]
- Koes RE, Spelt CE, van den Elzen PJM, Mol JNM. Cloning and molecular characterization of the chalcone synthase multigene family of Petunia hybrida. Gene. 1989b;81:245–257. doi: 10.1016/0378-1119(89)90185-6. [DOI] [PubMed] [Google Scholar]
- Martin CR. Structure, function, and regulation of the chalcone synthase. Int Rev Cytol. 1993;147:233–284. doi: 10.1016/s0074-7696(08)60770-6. [DOI] [PubMed] [Google Scholar]
- Mo Y, Nagel C, Taylor LP. Biochemical complementation of chalcone synthase mutants defines a role for flavonols in functional pollen. Proc Natl Acad Sci USA. 1992;89:7213–7217. doi: 10.1073/pnas.89.15.7213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mu J-H, Stains JP, Kao T-H. Plant Mol Biol. 1994;25:539–544. doi: 10.1007/BF00043881. [DOI] [PubMed] [Google Scholar]
- Napoli C, Lemieux C, Jorgensen R. Introduction of a chimeric chalcone synthase gene into petunia results in reversible co-suppression of homologous genes intrans. Plant Cell. 1990;4:279–289. doi: 10.1105/tpc.2.4.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Napoli CA, Ruehle J. New mutations affecting meristem growth and potential in Petunia hybrida. J Hered. 1996;87:371–377. [Google Scholar]
- Pollak PE, Hansen K, Astwood JA, Taylor LP. Conditional male fertility in maize. Sex Plant Reprod. 1995;8:231–241. [Google Scholar]
- Pollak PE, Vogt T, Mo Y, Taylor LP. Chalcone synthase and flavonol accumulation in stigmas and anthers of Petunia hybrida. Plant Physiol. 1993;102:925–932. doi: 10.1104/pp.102.3.925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quattrocchio F, Wing JF, Leppen HTC, Mol JNM, Koes RE. Regulatory genes controlling anthocyanin pigmentation are functionally conserved among plant species and have distinct sets of target genes. Plant Cell. 1993;5:1497–1512. doi: 10.1105/tpc.5.11.1497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Que Q, Wang HY, English JJ, Jorgensen RA. The frequency and degree of cosuppression by sense chalcone synthase transgenes are dependent on transgene promoter strength and are reduced by premature nonsense codons in the transgene coding sequence. Plant Cell. 1997;9:1357–1368. doi: 10.1105/tpc.9.8.1357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robbins TP, Gerats AGM, Fiske H, Jorgensen RA. Suppression of recombination in wide hybrids of Petunia hybrida as revealed by genetic mapping of marker transgenes. Theor Appl Genet. 1995;90:957–968. doi: 10.1007/BF00222909. [DOI] [PubMed] [Google Scholar]
- Schuster AM, Davies E. Ribonucleic acid and protein metabolism in pea epicotyls. Plant Physiol. 1983;73:809–816. doi: 10.1104/pp.73.3.809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stafford HA. Flavonoid Metabolism. Boca Raton, FL: CRC Press; 1990. [Google Scholar]
- Taylor LP, Briggs WR. Genetic regulation and photocontrol of anthocyanin accumulation in maize seedlings. Plant Cell. 1990;2:115–127. doi: 10.1105/tpc.2.2.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor LP, Jorgensen R. Conditional male fertility in chalcone synthase-deficient petunia. J Hered. 1992;83:11–17. [Google Scholar]
- van der Krol AR, Mur LA, Beld M, Mol JNM, Stutje AR. Flavonoid genes in petunia: addition of a limited number of gene copies may lead to a suppression of gene expression. Plant Cell. 1990;4:291–299. doi: 10.1105/tpc.2.4.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Eldik GJ, Reijnen WH, Ruiter RK, van Herpen MMA, Schrauwen JAM, Wullems GJ. Plant J. 1997;11:105–113. doi: 10.1046/j.1365-313x.1997.11010105.x. [DOI] [PubMed] [Google Scholar]
- Vogt T, Pollak P, Tarlyn N, Taylor LP. Pollination- or wound-induced kaempferol accumulation in petunia stigmas enhances seed production. Plant Cell. 1994;6:11–23. doi: 10.1105/tpc.6.1.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogt T, Taylor LP. Flavonol 3-O-glycosyltransferases associated with petunia pollen produce gametophyte-specific flavonol diglycosides. Plant Physiol. 1995;108:903–911. doi: 10.1104/pp.108.3.903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogt T, Wollenweber E, Taylor LP. The structural requirements of flavonols that induce pollen germination of conditionally male fertile petunia. Phytochemistry. 1995;38:589–592. [Google Scholar]
- Xu P, Vogt T, Taylor LP. Uptake and metabolism of flavonols during in-vitro germination of Petunia hybrida (L.) pollen. Planta. 1997;202:257–265. [Google Scholar]
- Ylstra B, Busscher J, Franken J, Hollman PCH, Mol JNM, van Tunen AJ. Flavonols and fertilization in Petunia hybrida: localization and mode of action during pollen tube growth. Plant J. 1994;6:201–212. [Google Scholar]