Abstract
Purpose
Elevated intraocular pressure (IOP) is the major modifiable risk factor for the sight-threatening eye disease, glaucoma. We investigated whether oral omega-3 supplements affect IOP in normotensive adults.
Methods
We undertook a pooled analysis of data from two double-masked, placebo-controlled randomized trials (Australian New Zealand Clinical Trials Registry, ACTRN12614001019695, ACTRN12615000173594) that investigated the efficacy and safety of oral omega-3 supplementation for treating ocular surface inflammation. Recruitment involved adults (n = 105) with IOP <21 mm Hg, and without a current or prior glaucoma diagnosis. Participants were randomly allocated to either an oral omega-3 (∼1000 mg/day eicosapentaenoic acid + ∼500 mg/day docosahexaenoic acid ± 900 mg/day α-linolenic acid) or placebo (olive oil, 1500 mg/day) supplement. IOP was quantified at baseline and after 3 months of supplementation (day 90). Change in IOP, relative to baseline, was compared between groups.
Results
At baseline, participants were of similar age (omega-3/placebo groups: mean ± SEM, 33.7 ± 1.7, n = 72/35.6 ± 3.0 years, n = 33), sex (65%/79% female), and had similar IOP (14.3 ± 0.3/13.8 ± 0.5 mm Hg). At day 90, IOP was reduced to 13.6 ± 0.3 mm Hg in the omega-3 group; controls had a slight IOP increase to 14.2 ± 0.4 mm Hg (P < 0.05).
Conclusions
Oral omega-3 supplementation for 3 months significantly reduced IOP in normotensive adults. To our knowledge, this is the first study to report that omega-3 fatty acids lower IOP in humans.
Translational Relevance
These findings justify further investigation into the therapeutic potential of omega-3 supplementation for reducing IOP, to prevent and/or treat conditions with IOP elevation, including ocular hypertension and glaucoma.
Keywords: omega-3, intraocular pressure, fatty acids, glaucoma
Introduction
Increasing scientific evidence supports an association between dietary factors and long-term health outcomes.1 Of particular interest has been whether dietary omega-3 essential fatty acids (EFAs) can prevent and/or provide therapeutic management in a range of general health conditions, including cardiovascular disease, hyperlipidemia, and systemic inflammatory disease. Humans cannot form EFAs in vivo and must ingest them from dietary sources. Omega-3 fatty acids exist as short- (α-linolenic acid [ALA]) and long-chain (eicosapentaenoic acid [EPA] and docosahexaenoic acid [DHA]) subtypes. The omega-6 fatty acids are the other major class, which derive from the diet in the form of linolenic acid. As omega-3 and omega-6 fatty acids compete in vivo for enzymes regulating their metabolism, the ratio of consumed omega-3 to omega-6 EFAs determines the inflammatory status of the body, with omega-3s biasing prostaglandin metabolism towards the production of anti-inflammatory eicosanoids.2
Diets rich in omega-3 EFAs are associated with a reduced risk of coronary heart disease mortality,3 attenuated age-related cognitive decline,4 and a decreased risk of late-stage age-related macular degeneration.5 In addition, significant literature has considered the association between omega-3 fatty acid intake and eye health. Omega-3 EFA deprivation may predispose individuals to ocular disease in later life.6 It follows that the potential benefit of omega-3 fatty acid supplementation has undergone evaluation in clinical trials in eye diseases that show an increased prevalence with age, particularly age-related macular degeneration7 and dry eye disease.8
Another significant eye disease that shows an age-dependent increase in prevalence is glaucoma, the second major cause of blindness worldwide.9 Currently, the major modifiable risk factor for glaucoma is elevated intraocular pressure (IOP).10 Reducing IOP is the mainstay of glaucoma treatment, and is achieved typically by medical management using topical IOP-lowering agents as first-line therapy. Identification of other targetable risk factors has the potential to enhance intervention options for managing the disease.
It is noteworthy that IOP tends to increase with age in Western populations,11 but the opposite trend occurs in traditional Japanese populations, who have higher dietary intakes of polyunsaturated fats.12 A large prospective cohort study, undertaken in Europe and involving >17,000 participants, showed an association between the ratio of consumed omega-3 to omega-6 fatty acids and incident glaucoma, especially among older individuals.13 Most recently, data from the National Health and Nutrition Examination Survey (NHANES, 2005–2008), a cross-sectional survey involving 3865 participants from the United States, showed that increased daily consumption of the long-chain, polyunsaturated omega-3 fatty acids, EPA, and DHA, was associated with a lower likelihood of glaucomatous optic neuropathy.14 In that study, the odds of an individual having glaucoma were nearly three times as high in participants whose daily dietary total long-chain omega-3 consumption level was in the second and third quartiles, compared to those in the first quartile.14 Furthermore, evidence from animal studies indicates the potential to modify IOP by up to 23% through omega-3 dietary manipulation.15 Taken together, these findings suggest that omega-3 intake may modulate IOP in humans. However, to our knowledge the effect of omega-3 supplementation on IOP in normotensive adults has not been investigated previously.
We analyzed pooled data from two double-masked, randomized, placebo-controlled trials (RCTs) to investigate whether a 3-month period of oral omega-3 supplementation significantly alters IOP in a population of normotensive adults consuming a typical Western diet.
Methods
This paper describes the IOP data (being a predefined safety outcome parameter) from a pooled analysis of data obtained from two double-masked, placebo-controlled RCTs that investigated the efficacy of oral omega-3 supplementation for treating ocular surface inflammation, with a 3-month follow-up period. The RCTs evaluated the efficacy and safety of different formulations of omega-3 EFA supplements for treating dry eye disease and contact lens discomfort, respectively. The methodology for the RCT involving the dry eye disease population, including participant inclusion and exclusion criteria, has been described previously in detail.16,17 Methodological details for the RCT relating to contact lens discomfort are provided on the Australian New Zealand Clinical Trials Registry (ANZCTR; ACTRN12615000173594). Both RCTs were prospectively registered on the ANZCTR, approved by the University of Melbourne Human Research Ethics Committee, and adhered to the tenets of the Declaration of Helsinki.
In brief, adult participants (≥18 years) were recruited from the University of Melbourne eye care clinic (Parkville, Victoria, Australia). All participants provided written informed consent to participate, had bilateral IOP < 21 mm Hg, did not have a current or prior diagnosis of glaucoma, and did not use any medications with known effects on IOP over the study duration.
Participants were randomized to receive either an oral omega-3 supplement (n = 72) consisting of krill oil (945 mg/day EPA + 510 mg/day DHA), fish oil (1000 mg/day EPA + 500 mg/day DHA or 900 mg/day EPA + 600 mg/day DHA), or fish plus flaxseed oils (900 mg/day EPA + 600 mg/day DHA + 900 mg/day ALA); or an oral placebo supplement (n = 33, 1500 mg/day olive oil) for 3 months.
Participants were masked to treatment allocation, as achieved by the investigational products being dispensed in identical opaque containers. Study personnel also were masked to treatment allocations. Following completion of all participant visits, data were analyzed only with knowledge of the simple randomization code (e.g., groups A, B, or C allocation). Full unmasking of treatment allocations, by the independent data manager, occurred after statistical analyses were complete.
Participants were instructed to maintain their current dietary habits throughout the study. The approximate dietary intake of omega-3 EFAs was determined by asking participants about their consumption of omega-3 rich foods over the preceding month. Participants were asked to quantify the approximate serving size (25, 50, 100, 150 g) and frequency of consumption of foods (including fish, oils, nuts, seeds, and spreads) containing greater than 1000 mg of combined EPA, DHA, docosapentaenoic acid (DPA), and ALA per 100 g edible portion (Australia New Zealand Food Authority, 2011; Nutrient Data Laboratory and Beltsville Human Nutrition Research Centre, 2011), as described previously.17 At the 3-month evaluation visit, participants were questioned about changes to their diet or medications and about compliance with taking the study supplements.
Compliance was assessed by having participants return supplement containers with unused capsules; these were counted by an independent researcher. Acceptable participant compliance (for data inclusion) was defined as ≥75% capsule consumption, based upon capsule counts.
IOP measures were performed as a safety measure and secondary analyses have revealed our novel and unexpected finding. There was sufficient homogeneity of the study populations and methodologies to pool data from the two studies. IOP was measured using applanation tonometry at baseline (day 1) and at the study endpoints (3-month follow-up visit). To minimize any potential diurnal effects on outcome measures in both RCTs, the 3-month study visit occurred at a similar time of day (±2 hours) to the baseline visit. For each individual, the average of right and left eye measures was used to represent IOP at each time point.
Intergroup comparisons were achieved by calculating the change in IOP from baseline (day 1) at day 90. The variance in change in IOP between the two study groups was assessed using Bartlett's test. Data normality was confirmed using the D'Agostino and Pearson Omnibus tests, and intergroup comparisons were performed using a t-test. A χ2 test was used to compare data consisting of proportions. An α of 0.05 was considered statistically significant. An effect size (Cohen's d) was calculated using Equation 1: Cohen's d = (M2−M1)/SDpooled where M1 and M2 are the means of the samples and SDpooled is the pooled standard deviation of the samples.
Results
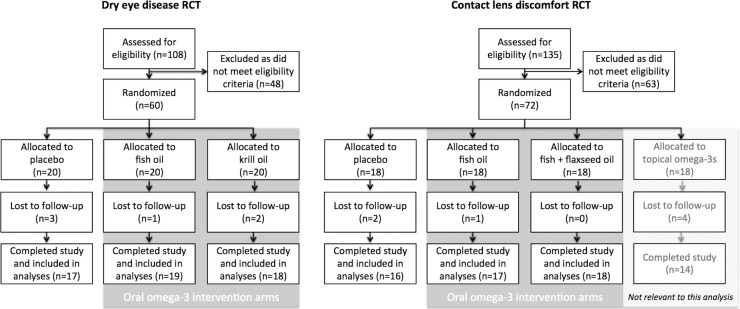
Figure 1 shows a Consolidated Standards of Reporting Trials (CONSORT) flow diagram,18 summarizing participant flow in the two RCTs. In both trials, participant retention was ≥90%, with similar dropout between groups. There was a high level of treatment fidelity, as measured by returned capsule counts. All participants exceeded the prespecified minimum level of compliance; no data were excluded from the analysis due to inadequate compliance.
Figure 1.
CONSORT flow diagram summarizing the key stages (enrollment, allocation, follow-up, and analysis) for the two clinical trials.
Our analysis includes 105 participants randomized to either an oral omega-3 supplement (n = 72) or placebo (n = 33). At baseline, the demographic and IOP characteristics of the participants were similar between the intervention groups (Table 1, P > 0.05 for all comparisons). The variance in change from baseline in IOP between study groups, assessed using Bartlett's test, also was similar (P = 0.14).
Table 1.
Baseline Participant Characteristics
Overall, participants consumed relatively low levels of dietary omega-3 fatty acids, consistent with a Western diet. At baseline, the combined EPA + DHA dietary intake of participants was similar between the treatment groups (mean ± SEM, 154.0 ± 22.7 vs. 172.4 ± 22.3 mg/day for placebo and omega-3 groups, respectively; P = 0.61).
No participant reported major changes to their intake of foods containing high levels of omega-3 fatty acids throughout the studies.
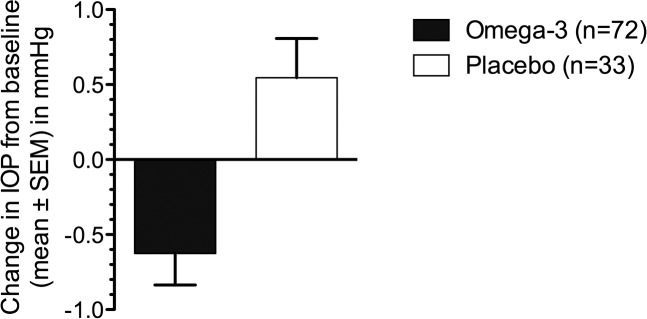
Relative to placebo, participants who received an oral omega-3 supplement for 3 months showed a significant reduction in IOP from baseline (−0.6 ± 0.2 vs. 0.5 ± 0.3 mm Hg for omega-3 versus placebo groups, respectively; P = 0.002, Fig. 2). The Cohen's effect size (d) for this change is 3.2, representing a ‘very large' experimental effect with substantial clinical significance.
Figure 2.
Change in IOP from baseline in individuals given omega-3 fatty acid (n = 72) and placebo (n = 33) supplements. Participants treated with oral omega-3 supplements had a significant reduction in IOP from baseline (omega-3 versus placebo groups, −0.6 ± 0.2 vs. 0.5 ± 0.3 mm Hg; P = 0.002) after 3 months of intervention.
Discussion
Our data demonstrated that 3 months of systemic omega-3 supplementation significantly reduces IOP in young normotensive adults consuming a typical Western diet. To our knowledge, this is the first prospective clinical study to report that omega-3 fatty acids directly modulate IOP in humans. Our findings indicated a modest (8%) reduction in IOP; however, the Cohen's effect size (d = 3.2) implied a substantial effect of major clinical significance. These data were consistent with previous experimental investigations, in animal models, showing that increasing dietary omega-3 fatty acid intake can lower IOP.15,19 As discussed herein, additional potential benefit(s) of omega-3 fatty acid dietary enhancement in modifying glaucoma risk may relate to neuroprotection and/or vascular regulation.
IOP is regulated by the balance between aqueous humor secretion and outflow. In a rat model of aging, dietary enrichment with omega-3 fatty acids reduced IOP by approximately 23%, via a mechanism involving increased aqueous outflow facility.15 The effect was proposed to derive from an increase in docosanoids, metabolic products of omega-3 fatty acid metabolism, which were upregulated in the ciliary body.15 An additional factor may be the direct anti-inflammatory effects of omega-3 fatty acids.6 Omega-3 supplementation can modify the expression of tear inflammatory cytokines in dry eye patients17 and suppress retinal inflammation in experimental models of autoimmune uveitis.20 Therefore, systemic anti-inflammatory modulation may contribute to the observed IOP-lowering effects, possibly through effects on outflow resistance through the trabecular meshwork and/or modulation of uveoscleral outflow; further investigation is required to explore these hypotheses.
An additional potential benefit is the neuroprotection that omega-3 fatty acid intake provides. Increasing dietary omega-3 fatty acid intake is beneficial for retinal function, particularly ganglion cell function,21 which is selectively vulnerable in glaucoma. In humans, 3-month systemic intervention with omega-3 fatty acids significantly improved blue-on-yellow perimetric indices in individuals with ocular hypertension.22 In addition, omega-3 supplementation has been shown to impart neuroprotective effects to the sensory peripheral nerves in the cornea of patients with dry eye disease.16 Furthermore, in experimental models, omega-3 long-chain fatty acids can enhance neurite outgrowth in cell cultures of primary sensory neurons23 and improve functional recovery of peripheral nerve injury.24 With respect to the mechanism underpinning these effects, the metabolism of DHA within the body results in the production of neuroprotectins and the D-series resolvins, which promote neuroprotection, the resolution of inflammation and tissue repair.25 DHA has been shown to have therapeutic potential for a broad range of acute and chronic neurodegenerative conditions.26
Another relevant consideration relates to the contribution of vascular insufficiency, in particular reduced blood flow to the optic nerve, which has a key pathogenic role in glaucoma.27 It has been shown that, through effects on red blood cell membrane composition,28 omega-3 fatty acids reduce blood viscosity,29 which may yield beneficial effects in terms of modulating optic nerve blood flow. In a cross-sectional, clinical study, Ren et al.30 reported that people with primary open angle glaucoma had lower serum long-chain omega-3 fatty acid levels, compared to age-matched controls. They proposed that sufficient dietary intake of EPA and DHA may enable more effective modulation of the ocular microcirculation, being physiologically necessary to prevent development of glaucoma.30 Consistent with this proposal, it has been reported that decreased red blood cell DHA levels are evident in individuals with glaucoma, with the reduction occurring before the onset of clinical signs of glaucomatous optic neuropathy.31
While the magnitude of the IOP-lowering effect that we observed was relatively modest in absolute terms (∼8%), notably this represents a large experimental effect and the change occurred in a young normotensive population (mean baseline IOP ∼14 mm Hg) who were supplemented with a moderate daily dose of omega-3 fatty acids (∼1500 mg/day combined EPA+DHA) for a relatively short time period (3 months). Past investigations in second-generation rats fed omega-3 fatty acids for their lifetime found a 23% change in IOP,15 which may represent the upper limit of potential change. Indeed, this level of IOP reduction is not dissimilar to some of the mainstays of topical glaucoma therapy, such as beta-blocker antagonists. Whether an IOP change in excess of 8% can be induced in humans, especially those with ocular hypertension or glaucoma, and/or with a higher daily dose of supplementation warrants further study, particular randomized controlled trials to extend our findings.
In conclusion, our study showed that 3 months of oral omega-3 fatty acid supplementation significantly reduces IOP by 8% in young normotensive adults. Our findings justify further clinical investigation into the therapeutic potential of omega-3 fatty acid supplements for reducing IOP, to prevent and/or treat conditions that are associated with an elevation in IOP, such as glaucoma.
Acknowledgments
We thank Laura Deinema and Anne Gad, who were involved in the clinical examination of participants.
The dry eye clinical trial was funded by a University of Melbourne Early Career Researcher Grant (LED) and the contact lens discomfort clinical trial was funded by CooperVision Pty Ltd (LED). The authors report no conflicts of interest in relation to this submission.
Disclosure: L.E. Downie, received funding from Allergan and Alcon, for research outside of this work; A.J. Vingrys, received funding from Allergan and Alcon, for research outside of this work.
References
- 1. Micha R, Penalvo JL, Cudhea F, Imamura F, Rehm CD, Mozaffarian D. . Association Between dietary factors and mortality from heart disease, stroke, and type 2 diabetes in the United States. JAMA. 2017; 317: 912– 924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Calder PC. . Omega-3 polyunsaturated fatty acids and inflammation: from molecular biology to the clinic. Lipids. 2003; 383: 343– 352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Whelton SP, He J, Whelton PK, Muntner P. . Meta-analysis of observational studies on fish intake and coronary heart disease. Am J Cardiol. 2004; 93: 1119– 1123. [DOI] [PubMed] [Google Scholar]
- 4. Fotuhi M, Mohassel P, Yaffe K. . Fish consumption, long-chain omega-3 fatty acids and risk of cognitive decline or Alzheimer disease: a complex association. Nat Clin Pract Neurol. 2009; 5: 140– 152. [DOI] [PubMed] [Google Scholar]
- 5. Seddon JM, Rosner B, Sperduto RD,et al. . Dietary fat and risk for advanced age-related macular degeneration. Arch Ophthalmol. 2001; 119: 1191– 1199. [DOI] [PubMed] [Google Scholar]
- 6. SanGiovanni JP, Chew EY. . The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog Ret Eye Res. 2005; 24: 87– 138. [DOI] [PubMed] [Google Scholar]
- 7. Downie LE, Keller PR. . Nutrition and age-related macular degeneration: research evidence in practice. Optom Vis Sci. 2014; 91: 821– 831. [DOI] [PubMed] [Google Scholar]
- 8. Hom MM, Asbell P, Barry B. . Omegas and dry eye: more knowledge, more questions. Optom Vis Sci. 2015; 92: 948– 956. [DOI] [PubMed] [Google Scholar]
- 9. Bourne RR, Taylor HR, Flaxman SR,et al. . Number of people blind or visually impaired by glaucoma worldwide and in world regions 1990 – 2010: a meta-analysis. PLoS One. 2016; 11: e0162229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Friedman DS, Wilson MR, Liebmann JM, Fechtner RD, Weinreb RN. . An evidence-based assessment of risk factors for the progression of ocular hypertension and glaucoma. Am J Ophthalmol. 2004; 138: S19– 31. [DOI] [PubMed] [Google Scholar]
- 11. Brubaker RF, Nagataki S, Townsend DJ, Burns RR, Higgins RG, Wentworth W. . The effect of age on aqueous humor formation in man. Ophthalmology. 1981; 88: 283– 288. [DOI] [PubMed] [Google Scholar]
- 12. Nomura H, Ando F, Niino N, Shimokata H, Miyake Y. . The relationship between age and intraocular pressure in a Japanese population: the influence of central corneal thickness. Curr Eye Res. 2002; 24: 81– 85. [DOI] [PubMed] [Google Scholar]
- 13. Perez de Arcelus M, Toledo E, Martinez-Gonzalez MA, Sayon-Orea C, Gea A, Moreno-Montanes J. . Omega 3:6 ratio intake and incidence of glaucoma: the SUN cohort. Clin Nutr. 2014; 33: 1041– 1045. [DOI] [PubMed] [Google Scholar]
- 14. Wang YE, Tseng VL, Yu F, Caprioli J, Coleman A. . Association of dietary fatty acid intake with glaucoma in the United States. JAMA Ophthalmol. 2017; 136: 141– 147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Nguyen CT, Bui BV, Sinclair AJ, Vingrys AJ. . Dietary omega 3 fatty acids decrease intraocular pressure with age by increasing aqueous outflow. Invest Ophthalmol Vis Sci. 2007; 48: 756– 762. [DOI] [PubMed] [Google Scholar]
- 16. Chinnery HR, Naranjo Golborne C, Downie LE. . Omega-3 supplementation is neuroprotective to corneal nerves in dry eye disease: a pilot study. Ophthalmic Physiol Opt. 2017; 37: 473– 481. [DOI] [PubMed] [Google Scholar]
- 17. Deinema LA, Vingrys AJ, Wong C-Y, Jackson DC, Chinnery HR, Downie LE. . A randomized, double-masked, placebo-controlled clinical trial of two forms of omega-3 supplements for treating dry eye disease. Ophthalmology. 2017; 124: 43– 52. [DOI] [PubMed] [Google Scholar]
- 18. Schulz KF, Altman DG, Moher D. . CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials. BMJ. 2010; 340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Mancino M, Ohia E, Kulkarni P. . A comparative study between cod liver oil and liquid lard intake on intraocular pressure on rabbits. Prostaglandins Leukot Essent Fatty Acids. 1992; 45: 239– 243. [DOI] [PubMed] [Google Scholar]
- 20. Shoda H, Yanai R, Yoshimura T,et al. . Dietary omega-3 Fatty acids suppress experimental autoimmune uveitis in association with inhibition of Th1 and Th17 cell function. PLoS One. 2015; 10: e0138241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Nguyen CT, Vingrys AJ, Bui BV. . Dietary omega-3 fatty acids and ganglion cell function. Invest Ophthalmol Vis Sci. 2008; 49: 3586– 3594. [DOI] [PubMed] [Google Scholar]
- 22. Cellini M, Rossi A, Moretti M. . The use of polyunsaturated fatty acids in ocular hypertension: a study with blue-on-yellow perimetry. Acta Ophthalmol Scand. 1999; 77: 54– 55. [Google Scholar]
- 23. Robson LG, Dyall S, Sidloff D, Michael-Titus AT. . Omega-3 polyunsaturated fatty acids increase the neurite outgrowth of rat sensory neurones throughout development and in aged animals. Neurobiol Aging. 2010; 31: 678– 687. [DOI] [PubMed] [Google Scholar]
- 24. Gladman SJ, Huang W, Lim SN,et al. . Improved outcome after peripheral nerve injury in mice with increased levels of endogenous omega-3 polyunsaturated fatty acids. J Neurosci. 2012; 32: 563– 571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Gordon WC, Bazan NG. . Mediator lipidomics in ophthalmology: targets for modulation in inflammation, neuroprotection and nerve regeneration. Curr Eye Res. 2013; 38: 995– 1005. [DOI] [PubMed] [Google Scholar]
- 26. Dyall SC, Michael-Titus AT. . Neurological benefits of omega-3 fatty acids. Neuromol Med. 2008; 10: 219– 235. [DOI] [PubMed] [Google Scholar]
- 27. Yanagi M, Kawasaki R, Wang JJ, Wong TY, Crowston J, Kiuchi Y. . Vascular risk factors in glaucoma: a review. Clin Exp Ophthalmol. 2011; 39: 252– 258. [DOI] [PubMed] [Google Scholar]
- 28. Ernst E. . Effects of n-3 fatty acids on blood rheology. J Int Med Suppl. 1989; 731: 129– 132. [DOI] [PubMed] [Google Scholar]
- 29. Mueller BA, Talbert RL. . Biological mechanisms and cardiovascular effects of omega-3 fatty acids. Clin Pharm. 1988; 7: 795– 807. [PubMed] [Google Scholar]
- 30. Ren H, Magulike N, Ghebremeskel K, Crawford M. . Primary open-angle glaucoma patients have reduced levels of blood docosahexaenoic and eicosapentaenoic acids. Prostaglandins Leukot Essent Fatty Acids. 2006; 74: 157– 163. [DOI] [PubMed] [Google Scholar]
- 31. Acar N, Berdeaux O, Gregoire S,et al. . Lipid composition of the human eye: are red blood cells a good mirror of retinal and optic nerve fatty acids? PLoS One. 2012; 7: e35102. [DOI] [PMC free article] [PubMed] [Google Scholar]