Abstract
Purpose
CPI-613, a lipoate analog that inhibits pyruvate dehydrogenase (PDH) and α-ketogluterate dehydrogenase (KGDH) has activity in patients with myeloid malignancies. This study explored the role of mitochondrial metabolism in chemotherapy response and determined the maximally tolerated dose, efficacy and safety of CPI-613 combined with high dose cytarabine and mitoxantrone in patients with relapsed or refractory acute myeloid leukemia.
Methods
The role of mitochondrial response to chemotherapy was assessed in cell lines and animal models. A phase I study of CPI-613 plus cytarabine and mitoxantrone was conducted in patients with relapsed or refractory AML.
Results
Exposure to chemotherapy induced mitochondrial oxygen consumption that depended on PDH. CPI-613 sensitized AML cells to chemotherapy indicating that mitochondrial metabolism is a source of resistance. Loss of p53 did not alter response to CPI-613. The phase I study enrolled 67 patients and 62 were evaluable for response. The overall response rate was 50% (26CR+5CRi/62). Median survival was 6.7 months. In patients over 60 years old, the CR/CRi rate was 47% (15/32) with a median survival of 6.9 months. The response rate for patients with poor risk cytogenetics also was encouraging with 46% (11 of 24 patients) achieving a CR or CRi. RNA sequence analysis of a subset of baseline bone marrow samples revealed a gene expression signature consistent with the presence of B cells in the pretreatment marrow of responders.
Conclusion
The addition of CPI-613 to chemotherapy is a promising approach in older patients and those with poor risk cytogenetics.
Introduction
Acute myeloid leukemia (AML) is characterized by poor outcomes and resistance to therapy. It is the most common acute leukemia in adults affecting approximately 20,000 people per year in the United States; despite decades of research, the overall 5-year survival remains 30–40% (1). Current therapy is a combination of cytarabine and daunorubicin, first reported in 1973 and not substantially changed since then (2). Therapy results in a 60 to 80% remission rate; but over 50% of patients will relapse, and once relapsed most will die from AML within a year (3). Despite the central role of relapse and resistance to therapy, little is known about the mechanisms involved.
With the discovery of the role of mutant isocitrate dehydrogenase in the leukemogenesis of some types of AML it has become clear that mitochondrial metabolism can have a profound effect (reviewed by Medeiros e. at. (4)). Despite these studies it is not well understood to what degree AML cells can alter their mitochondrial metabolism in response to environmental stressors, including those induced by chemotherapy. Fatty acid oxidation may support myeloid leukemic stem cell persistence (5), but the role of mitochondrial metabolism in resistance to chemotherapy in AML is poorly understood. A recent elegant study demonstrated that residual, cytarabine resistant AML cells in the marrow of treated animals had increased oxidative metabolism and mitochondrial mass suggesting a direct role of mitochondrial metabolism in resistance to therapy (6).
CPI-613 is a novel lipoic acid analogue with anticancer activity that inhibits PDH and KGDH (7, 8). CPI-613 inhibits mitochondrial respiration, causing hyper-phosphorylation of PDH and activation of adenosine monophosphate activated kinase (AMPK) in AML cell lines (9). In a single-agent phase I study, CPI-613 was well tolerated. The current study sought to establish the role of PDH in resistance to chemotherapy in preclinical models of AML and conducted a phase I clinical trial to determine the safety of CPI-613 plus high-dose cytarabine (HiDAC) and mitoxantrone in patients with relapsed or refractory AML. The role of p53 in response to CPI-613 was also assessed. Finally, RNA sequence analysis on pretreatment bone marrow samples from a subset of responders and non-responders was performed to define a gene expression signature predictor of response.
Materials and Methods
Study Design
The study was approved by the Wake Forest School of Medicine Institutional Review Board (IRB) and conducted under the supervision of the Safety and Toxicity Review Committee. All patients provided written informed consent. The trial design was an open-label, single-arm, dose escalation study using a 1-3-6 accelerated titration design (see supplemental figure 1 and (10)).
Eligibility
Patients must have histologically or cytological documented relapsed and/or refractory Acute Myeloid Leukemia, be ≥18 years old, fit for intensive salvage therapy, Eastern Cooperative Oncology Group (ECOG) performance status of ≤3 and an expected survival of >3 months. Patients could not have received radiotherapy, treatment with cytotoxic agents, treatment with biologic agents or any anti-cancer therapy within the 2 weeks prior to enrolment on study. Additionally, patients were required to have adequate hepatic function (aspartate aminotransferase [AST/SGOT] ≤3x upper normal limit [UNL], alanine aminotransferase [ALT/SGPT] ≤3x UNL (≤5x ULN if leukemic liver infiltration was present), bilirubin ≤1.5x UNL) and renal function (serum creatinine ≤1.5 mg/dL or 133 μmol/L).
Study Treatment
CPI-613 was given over 2 hours on days 1–5. Cytarabine was given at 3 gm/m2 for age <60 or 1.5 gm/m2 for age ≥60 in 500 mL normal saline (NS), over 3 hours every 12 hours for 5 doses, starting on day 3. Mitoxantrone was given at 6 mg/m2 daily for 3 doses in 50 mL NS over 15 minutes after the 1st, 3rd, and 5th doses of cytarabine. A treatment cycle was defined as CPI-613, cytarabine, and mitoxantrone, given on days 1–5 of Week 1, followed by 1 week of rest (14 days total). Each patient received 1 cycle of treatment; residual disease was assessed on day 14. Any patient with significant residual disease (defined as blasts ≥5%) could, at the discretion of the treating physician, receive a second full course or a 3-day course with CPI-613 given on days 1–3, cytarabine given for 3 doses starting on day 2, and mitoxantrone given after the first and third cytarabine doses. Any patient with significant residual disease after 2 cycles was considered refractory and removed from the study. Responding patients could receive up to two consolidation cycles of the 3-day course.
Dose Limiting Toxicity
A dose limiting toxicity was defined as the occurrence of any clinically relevant, grade ≥ 3, non-hematologic, non-infectious toxicity attributed as probable or definitely related to CPI-613. The following toxicities were excluded from defining a DLT: grade 3 nausea and vomiting responsive to anti-emetics, grade 3 diarrhea responsive to anti-diarrheal therapy, grade 3 tumor lysis syndrome, grade 3 or 4 metabolic derangements attributed to tumor lysis syndrome or antimicrobial medications that correct with oral or IV supplementation.
Assessments
Bone marrow biopsies were performed on day 14 following each salvage induction cycle to assess residual disease. Biopsies were also performed to assess remission status when peripheral blood counts recovered to an absolute neutrophil count of >1000/mcL and a platelet count of >100,000/mcL (or at the physician’s discretion).
Mouse Studies
The Wake Forest University Institutional Animal Care and Use Committee approved all mouse experiments. Luciferase-tagged leukemia cells were transplanted into 6- to 8-wk-old sub-lethally irradiated recipient mice (4 Gy) by tail-vein injection. Mice were monitored by bioluminescent imaging performed using an IVIS100 imaging system (Caliper LifeSciences, Hopkinton, MA). Mice were injected with 150 mg/kg D-Luciferin (Gold Biotechnology, St. Louis, MO), anesthetized with isoflurane, and imaged for 2 min. Chemotherapy was initiated upon detection of clear signals. Mice were treated with 100 mg/kg cytarabine and 3 mg/kg doxorubicin (both from Bedford Laboratories, Bedford, OH) by intraperitoneal injection. Control animals were injected with phosphate buffered saline. Following completion of therapy mice were followed for survival.
Western blots
Cultured cells were pelleted and washed in cold PBS. Pellets were lysed in Laemmli buffer, separated by SDS-PAGE, and transferred to an Immobilon PVDF membrane (Millipore). Antibodies against PDH e1α subunit (Abcam #177461, 1:1000), p53 (Leica, product #NCL-p53-505), AceCS1 (Cell Signaling, #3658S, 1:1000), AMPK (Cell Signaling, #2532S, 1:1000), phosphorylated AMPK (Cell Signaling, #2532S, 1:1000), SOD2 (Cell Signaling #13141, 1:1000) and actin (Abcam #AC-15, 1:5000) were used.
Cell culture/Cas9 Crisper gene deletion
MFL2 cells were grown in stem cell media at 37° C with 5% CO2 as in Pardee et. al. (11). MFL2 cells were infected with the Cas9 expressing vector, MSCV_Cas9_puro (gift from Christopher Vakoc) (Addgene plasmid # 65655) and infected cells selected with puromycin. Resistant cells were then transfected with sgRNA expressing vector LRG (Lenti_sgRNA_EFS_GFP) also a gift from Christopher Vakoc (Addgene plasmid # 65656) tagged with GFP targeting the indicated gene. Gene deletion was confirmed by Western blot. For competition assays mixed GFP+/− cell populations were exposed to the indicated therapy for 72 hours and GFP+ percentage of the viable cell population determined as in Pardee et al (12). Clonal populations of deleted cells were obtained by serial dilution. All human cell lines were maintained in RPMI media (Gibco) supplemented with 20% FBS, penicillin and streptomycin. Cells were grown at 37°C with 5% CO2. Viability assays were done using the EZQuant assay (ALSTEM, Richmond, CA) according to the manufacturer’s protocol.
Mitochondrial Respiration and Extracellular Acidification Assays
All oxygen consumption and acidification rate assays were performed using the XF24 Extracellular Flux Analyzer (Seahorse Bioscience) as per the manufacturer’s instructions. Cells were plated at a density of 400,000 viable cells per well for oxygen consumption assays and 600,000 viable cells per well for the acidification rate assay. Data was normalized to viable cell number and viability was routinely between 96% and 99% as determined by trypan blue exclusion assay (Countess automated cell counter, ThermoFisher). The media used for oxygen consumption assays was the mitochondrial stress test media, specifically: DMEM with 5.5mM glucose, 2.0mM Sodium Pyruvate, and 2mM L-glutamine – warmed to 37C and pH 7.35. The media used for the extracellular acidification rate assay was the glycolysis stress test media, specifically: DMEM with 2mM L-glutamine – warmed to 37C and pH 7.35. Cells were placed in assay media and incubated for 2 hours prior to being placed in the assay. For the oxygen consumption experiments oligomycin at a final concentration of 1 μM, FCCP at 0.5 μM, and Actinomycin A and Rotenone each at 1 μM were used. For the extracellular acidification rate experiments glucose was added at a final concentration of 10 mM, oligomycin at 1 μM and 2-deoxyglucose at 100 mM. All assays were done in triplicate (3 wells per condition each measured in triplicate) and repeated in three independent experiments. For the extracellular acidification rate assays in order to limit variability the results from the single well with the highest and lowest mean rates for each condition were omitted.
Electron Microscopy
Cultured cells were fixed with 2.5% glutaraldehyde in 0.1M Millonig’s phosphate buffer pH 7.3 for a minimum of one hour. Subsequently, the samples were washed 3x in Millonig’s buffer and post-fixed with 1% osmium tetroxide in phosphate buffer for one hour. After 3x washes in buffer the samples were dehydrated through a graded series of ethanols for 10 minutes each. The cultured cells were scraped to form a pellet once they were in 50% ethanol and subsequently dehydrated to 100%.The samples were prepared for resin infiltration by incubation in propylene oxide for two changes of 15 minutes each. Finally, the samples were gradually infiltrated with 1:1, 1:2 and pure solutions of Spurr’s resin after which they were allowed to cure in a 70 C oven overnight. 90 nm sections were obtained with a Reichert-Jung Ultracut E ultramicrotome, stained with lead citrate and uranyl acetate and viewed with a FEI Tecnai Spirit TEM operating at 80 kV. Images were obtained with an AMT 2Vu CCD camera.
Metabolite Extraction
Each cell pellet was extracted with 950 μL of ice cold methanol to which 50 μL of internal standard solution (0.5 ng/μL MES) was added and then mixed by vortexing. Extracts were incubated on ice for 1 hour before being centrifuged at 18,000 x g. The supernatant was removed and then stored at −20C until LC-MS/MS analysis.
LC-MS/MS
LC-MS/MS analysis was performed with a Shimadzu UHPLC-MS/MS equipped with 2 LC-30AD pumps, a SIL-30AC autosampler, a CBM-20A communications bus module, a DGU-20A5R degassing unit, a CTO-30A column oven, and an 8050 triple-quadrupole mass spectrometer operated with a DUIS source. The analytical separation was conducted with a Restek Ultra PFPP column (150 x 2.1 mm, 3 μm; Restek, Bellefonte, PA) under gradient conditions with a mobile phase A consisting of 0.1% formic acid in H2O and a mobile phase B consisting of 0.1% formic acid in 100% acetonitrile. The gradient began with 2 minutes at 0% B followed by a ramp to 25% B between 2 and 5 minutes, another ramp to 35% B between 5 and 11 minutes, a final increase from 35% to 95% B between 11 and 15 minutes, a hold at 95% B from 15 to 20 minutes, then a decrease to 0% B between 20 and 20.10 minutes, and a final hold at 0% B from 20.10 to 25 minutes. A flow rate of 0.25 mL/minute was used with a sample injection volume of 10 μL. Ionization occurred in the DUIS source switching between the positive and negative ESI ion mode with the following conditions: nebulizing gas flow of 2 L/min, heating gas flow of 5 L/min, interface temperature of 350 °C, DL temperature of 250 °C, heat block temperature of 400 °C, and a drying gas flow of 15 L/min. Shimadzu LabSolutions software (version 5.72) was used to quantify the analytes of interest.
RNA Sequencing analysis
Patient samples: Mononuclear cells from bone marrow biopsies were isolated by Ficoll gradient separation and stored in liquid nitrogen. Total RNA was isolated from bone marrow-derived mononuclear cell samples using the Qiagen RNeasy RNA isolation kit per the manufacturer’s instructions. RNA integrity was verified by electrophoretic tracing using an Agilent Bioanalyzer. Nineteen samples (68%) yielded sufficient RNA quantity and integrity for further analysis. RNA-seq libraries were constructed from ~500 ng of total RNA using the Illumina TruSeq Stranded Total RNA LT kit with Ribo-Zero rRNA depletion. Indexed libraries were sequenced on an Illumina NextSeq 500 DNA sequencer with 150-nt paired end reads generating, on average, >64 million reads per sample (>45 million uniquely mapped reads) with >75% of sequences achieving Q30 Phred quality scores. Quality control of raw sequence reads was conducted using FASTQC (http://www.bioinformatics.babraham.ac.uk/projects/fastqc/). Reads were aligned against human reference assembly GRCh37 (hg19) using the STAR sequence aligner (13) version 2.4.0j, and reads per gene determined using feature Counts software (14). Two samples were excluded from further analysis on the basis of having <50% of their sequence reads mapping to unique genomic loci. Differential gene expression analysis among responders (n=10) and non-responders (n=7) was conducted using DESEQ2 software (15). Genes differentially expressed between responders and non-responders were identified according to the following selection criteria: 1) FDR <20% (Benjamini and Hochberg), 2) base-mean average expression >20 reads, and 3) absolute average log2 fold-change > 1.0. Significant enrichment of gene ontologies and biological pathways was assessed using the DAVID Bioinformatics resource (16) version 6.8 with updated Knowledgebase (Oct. 2016).
Somatic mutation calling from RNA-sequencing data
The pipeline developed by Broad institute (https://gatkforums.broadinstitute.org/gatk/discussion/3891/calling-variants-in-rnaseq) was utilized. For alignment each sample had four fastq files generated by four different lanes from an Illumina NextSeq. Each of the 4 fastq files was aligned to hg19 reference by using a two-pass method with STAR2 (13). For adding group and marking duplicates, Picard (https://broadinstitute.github.io/picard/) added read group information of sample id and lane number into the bam and marked the duplicates in the bam. The four bam files of one sample were merged into one bam file and be sorted by samtools. For Split ‘N’ Trim and reassign mapping qualities a GATK (https://software.broadinstitute.org/gatk/) tool called SplitNCigarReads was used to split reads into exon segments by getting rid of Ns but maintaining groups and hard-clip any sequences overhanging into the intronic regions. To rescue some indels and adjust the qualities that have systematic errors, another two preprocessing steps were included, indel realignment by GATK 3.6 (https://software.broadinstitute.org/gatk/), and base recalibration by GATK 3.6. Mutation calling was done utilizing the HaplotypeCaller of GATK to call the indels and SNVs from the cleaned bams. Two different hard filters were utilized for SNVs and indels, respectively. The filter of “”QD < 2.0 || FS > 60.0 || MQ < 40.0 || MQRankSum < −12.5 || ReadPosRankSum < −8.0” was applied the VCF files of SNVs from the caller and “QD < 2.0 || FS > 200.0 || ReadPosRankSum < −20.0” was applied the VCF files of INDELs.
SNPs in dbSNP database (https://www.ncbi.nlm.nih.gov/projects/SNP/) and 1000 genome project (http://www.internationalgenome.org/) were excluded. Thus, the final somatic mutation list included 640 variants that are known to be AML somatic mutations in COSMIC (http://cancer.sanger.ac.uk/cosmic) and another two TP53 variants found in two patients who did not respond to CPI-613.
Visualization of the somatic mutations
The visualization was enabled by waterfalls plot in the R package of GenVisR (Skidmore et al. 2016). The genes shown in the figure were selected by setting a threshold to “percentage of samples” at y axis (>0.05).
Statistical Analysis
All means were compared by two-tailed student’s T test. Survival curves were estimated by the Kaplan-Meier method and p values were determined by the log rank test. P values below 0.05 were considered significant. Evaluable patients (who completed at least one cycle of study treatment and had a bone marrow sample obtained at day 14) were followed and analyzed for response rate and overall survival. Response, overall survival, and early mortality (death within 30 and 60 days of beginning of treatment) were compared to rates in a previous cohort treated with an identical dose and schedule of HiDAC, mitoxantrone but with the addition of L-asparaginase (17). Analysis was performed using Graph Pad Prism version 6.05 (Graph Pad Software Inc).
Results
Chemotherapy induces mitochondrial oxygen consumption
To determine the effects of chemotherapy on mitochondrial metabolism, K562 (CML in myeloid blast crisis), MFL2 (murine AML), and OCI-AML3 (monocytic AML) cells were exposed to cytarabine and their mitochondrial oxygen consumption rate (OCR) assessed. Baseline mitochondrial OCR increased after cytarabine exposure, in a dose-dependent fashion (Figure 1A). Proton leakage (assessed by OCR in the presence of the ATP synthase inhibitor oligomycin) was not significantly different following treatment, ruling out chemotherapy- induced changes in mitochondrial membrane permeability (data not shown). To determine the contribution of PDH and KGDH (the targets of CPI-613) to the increased OCR, a Cas9 expressing murine AML cell line (Cas9-MFL2) was generated. These cells were infected with a vector expressing a sgRNA targeting the E1α subunit of PDH, the E2 subunit of KGDH or the ROSA26 locus as a control. No viable clones could be isolated that expressed the sgRNA targeting KGDH suggesting its activity is essential in these cells. Clones expressing the sgRNA targeting PDH were isolated and loss of expression of PDH was confirmed by Western blot (Figure 1B). PDH- or ROSA-deleted cells were exposed to cytarabine and assessed for mitochondrial OCR. Baseline OCR and maximal respiratory capacity was severely compromised in PDH-deleted cells and did not increase following chemotherapy exposure. In contrast, ROSA-deleted cells maintained the dose-dependent increase in OCR (Figure 1C). To assess the role of glycolysis the extracellular acidification rate (ECAR), a surrogate for glycolysis, was determined in in ROSA- and PDH- deleted cells. In control cells basal ECAR was near maximal under all conditions assayed and did not significantly change following exposure to chemotherapy (Figure 1D and data not shown). In contrast, PDH deleted cells had lower basal ECAR that significantly increased following exposure to chemotherapy (Figure 1D). To determine if the observed diminished OCR was due to loss of mitochondria PDH-deleted cells were stained with a mitochondrial-specific dye and compared to controls. PDH-deleted cells displayed a significantly higher uptake of mitotracker dye when compared to control cells (Figure 2A). To corroborate this finding, electron micrographs of ROSA- and PDH- deleted cells were obtained. Strikingly, PDH-deleted cells contained mitochondria that were larger but less electron dense and with less cristae when compared with control cells suggesting impaired function (Figure 2B). To further characterize these mitochondria, cells were stained with the mitochondrial dye Tetramethylrhodamine (TMRM). Consistent with the micrographs the mean fluorescence intensity was significantly lower for PDH-deleted cells when compared to controls suggesting a lower mitochondrial membrane potential (Figure 2C). To further examine this steady-state levels of citrate and glutamate, a proxy for α-ketogluterate, in ROSA- and PDH- deleted cells were measured following exposure to chemotherapy. Consistent with the data above, citrate and glutamate levels were higher in the ROSA cells at baseline and fell following exposure to chemotherapy. In contrast, PDH-deleted cells had low levels of citrate and glutamate at baseline that did not change after treatment (Figure 2D+E). These results suggest that PDH activity is necessary for the chemotherapy induced mitochondrial OCR in this model. Consistent with this concept, glucose-6-phosphate levels fell in ROSA cells in response to chemotherapy exposure (Figure 2F). These data suggest that AML cells require PDH function for optimal glycolytic rates and that following exposure to chemotherapy glucose-6-phosphate levels fall consistent with increased glycolysis while ECAR remains stable and OCR increases suggesting more pyruvate is being shunted to the mitochondria to enter the TCA cycle. In the absence of PDH function glycolysis is increased following chemotherapy however the pyruvate produced is at least in part converted to lactate.
Figure 1. Chemotherapy induces mitochondrial oxygen consumption.
(A) Basal oxygen consumption rates (OCR) following exposure to cytarabine. AML cells were exposed to the indicated amount of cytarabine for 8 (MFL2) or 16 hours (OCI-AML3 and K562) and basal mitochondrial OCR determined. Shown are results of 3 independent experiments, each done in triplicate. P values by one-way ANOVA analysis. (B) Cells expressing sgRNAs against either the ROSA26 locus or PDH E1α were lysed and subjected to western blotting. (C) ROSA control cells or PDH deleted cells were exposed to cytarabine for 16 hours and assessed for basal mitochondrial OCR. Shown are results of 3 independent experiments, each done in triplicate. (D) ROSA control cells or PDH deleted cells were exposed to cytarabine for 16 hours and assessed for basal extracellular acidification rates (ECAR). Shown are results of 4 independent experiments, each done in triplicate. * indicates statistically significant change by Sidak’s multiple comparisons test.
Figure 2. Mitochondrial content and metabolite levels in ROSA- and PDH-deleted AML cells.
(A) ROSA- (ROSA KO) or PDH-deleted (PHD KO) cells were analyzed by flow cytometry with mitotracker red staining. Median fluorescence intensity of mitotracker red stained cells from 3 independent experiments were quantitated and plotted. (B) Electron micrographs of ROSA- or PDH-deleted cells. Cells were cultured in the absence of chemotherapy and electron micrographs were obtained. Representative images are shown. (C) TMRM staining. ROSA- or PDH-deleted cells were stained with TMRM and assessed by flow cytometry. Median fluorescence intensity of TMRM stained cells from 3 independent experiments were quantitated and plotted. (D+E) Citrate and Glutamate levels in ROSA- or PDH-deleted cells were determined following a 16 hour exposure to the indicated amount of cytarabine (Ara). (F) Glucose-6-phosphate levels in ROSA-deleted cells following a 16 hour exposure to the indicated amount of cytarabine (Ara). * indicates a statistically significantly different comparison by Sidak’s multiple comparison test. NS= a non-significant difference by Sidak’s multiple comparison test.
Loss of PDH activity confers sensitivity to chemotherapy in vivo
To determine if there was a functional consequence to the loss of PDH, cells were assessed for sensitivity to chemotherapy. PDH-deleted cells were more sensitive to both doxorubicin and cytarabine compared to control cells (Figure 3A). Thus, in AML cells, chemotherapy exposure leads to increased glycolysis and mitochondrial oxidation of pyruvate via PDH activity, which is a source of resistance. AML cells alter mitochondrial metabolism when co-cultured with supporting stromal cells, leading to resistance (5). To determine if AML cells in vivo still require PDH for optimal chemotherapy response, 1x106 ROSA26 or 1.5x106 PDH-deleted cells were injected into C57Bl/6 mice. Different cell numbers were injected, as PDH-deleted cells took longer to engraft, suggesting that PDH activity is needed for optimal engraftment. When engraftment was confirmed to similar levels by bioluminescent imaging (Figure 3B), mice were randomly assigned to saline (control) or combined cytarabine and doxorubicin treatment, as previously published (18). After treatment, mice injected with PDH-deleted cells had a significantly longer survival compared to the ROSA- injected mice (p=0.0034, Figure 3C). In contrast, the control animals from each group did not have significantly different survival (p= 0.266). These data suggest that PDH activity is a source of resistance for AML cells, even within the appropriate microenvironment.
Figure 3. Loss of PDH function induces sensitivity to chemotherapy.
(A) ROSA- (ROSA KO) or PDH-deleted (PHD KO) cells were exposed to the indicated chemotherapy for 72 hours and assessed for viability. Shown are results of 3 independent experiments, each done in triplicate. (B) C57Bl/6 mice were injected with either ROSA- or PDH-deleted cells and imaged after 9 days for ROSA-deleted and 15 days for PDH-deleted cells. Luminescence was quantitated and compared. (C) Kaplan-Meier curves of mice with either control or PDH-deleted leukemia. Survival was assessed from completion of therapy. (D) OCI or MFL2 cells were incubated with the indicated chemotherapy, alone or with CPI-613 for 72 hours, and viability assessed. *= indicates statistically significant change by Sidak’s multiple comparisons test.
Pharmacologic inhibition of the TCA with CPI-613 increases sensitivity to chemotherapy in AML cell lines
To determine if CPI-613 increased sensitivity of AML cells to chemotherapy OCI-AML3 and MFL2 AML cell lines were exposed to chemotherapy with and without CPI-613 at 25 or 50 μM (clinically achievable doses (9)) and assessed viability. Cells exposed to chemotherapy and CPI-613 were significantly more sensitive compared to cells treated with chemotherapy alone (Figure 3D). These data suggest that pharmacological inhibition of PDH and KGDH with clinically achievable doses of CPI-613 sensitizes AML to chemotherapy. To determine if PDH or KGDH inhibition played the more prominent role in our model systems we attempted to rescue the effects of CPI-613 on viability by co-treating cells with either acetate (to replace lost acetyl-CoA) or methyl-succinate (to replace lost succinyl-CoA). Conversion of acetate to acetyl-CoA is accomplished mainly via the enzyme acetyl-CoA synthetase (AceCS1). Both cell lines used expressed AceCS1 as demonstrated by Western blot (supplemental figure 2) indicating an ability to convert acetate to acetyl-CoA. Co-treatment with acetate had a modest but significant rescue of the growth inhibitory effects of CPI-613. In contrast, methyl-succinate alone or in combination with acetate had no significant effect on reversing CPI-613 toxicity (supplemental figure 3 and data not shown). These data suggest that PDH production of acetyl-CoA may play more of a role in CPI-613 mediated cytotoxicity than production of succinate. However, the modest effects seen suggest that coordinated production of acetyl-CoA and consumption of pyruvate may be needed for optimal cell survival and/or that additional non-TCA cycle effects may play a role in response to CPI-613.
Loss of p53 does not lead to resistance to CPI-613
To determine the effect of loss of p53 function on response to CPI-613, a competition assay was utilized where a portion of MFL2-Cas9 cells have deleted p53. Cytarabine treatment significantly enriched p53-deleted cells, but when treated with CPI-613, no enrichment was seen (Figure 4A). To extend these results, pure populations were compared. Knockout of p53 was confirmed by Western blot (Figure 4B). As expected, loss of p53 resulted in significant resistance to doxorubicin (Figure 4C) but had no significant effect in response to CPI-613 (Figure 4D). Importantly, when chemotherapy was combined with CPI-613, p53-deleted cells had increased sensitivity (Figure 4E). These data suggest that CPI-613 induces a p53 independent mode of cell death and that AML cells without p53 function could be sensitized to chemotherapy by CPI-613.
Figure 4. Loss of p53 does not alter response to CPI-613.
(A) MFL2 cells expressing Cas9 were partially infected with GFP labeled vector that expressed an sgRNA targeting p53. The mixed population was treated with the indicated therapy for 72 hours and the percentage of GFP+ cells was assessed and normalized to controls. (B) Western blot analysis of control or p53-deleted cells. Actin was used as a loading control. (C+D) Control ROSA26 KO cells and p53 (p53 KO) knocked out cells were incubated with the indicated therapy for 72 hours and viability assessed. (E) Control ROSA26 KO cells and p53 (p53 KO) knocked out cells were incubated with the indicated amounts of CPI-613 or doxorubicin (Dox) for 72 hours and viability assessed. Shown are the averaged results of at least 3 independent experiments, each done in triplicate. * indicates a statistically significantly different comparison by Sidak’s multiple comparison test. NS= a non-significant difference by Sidak’s multiple comparison test.
Phase I clinical trial of CPI-613 plus HiDAC and mitoxantrone in relapsed or refractory AML
To determine the safety and efficacy of adding CPI-613 to HiDAC and mitoxantrone for patients with relapsed or refractory AML a phase I trial was conducted (see treatment schema in supplemental figure 4).
Study Patients
Patient characteristics are summarized in Table 1. Briefly, 66 patients with relapsed or refractory AML were enrolled, one patient with advanced phase CML was enrolled in error. The median marrow blast involvement at time of enrolment was 43%. For 72% of patients, the study treatment was their first salvage. Disease status was relapsed in 69% of patients with a median duration of first remission of 10 months. Of the 21 patients with refractory AML, 2 did not respond to clofarabine induction, and 5 showed clear resistance or recovered with disease following a single course of standard-dose cytarabine based induction. All others received either 2 cycles of standard dose cytarabine-based induction or 1 or more HiDAC-based inductions without achieving remission.
Table 1.
Patient Characteristics
| Median age (range) | 60 (21–79) |
| Age ≥ 60 years old (%) | 36/67 (54%) |
| Male (%) | 37/67 (55%) |
| No. Caucasian (%) | 60/67 (90%) |
| ECOG Performance Status | |
| 0–1 | 45/63 (71%) |
| 2 | 17/63 (27%) |
| 3 | 1/63 (2%) |
| Median Duration of CR1 in months (Range) | 11.2 (2–94) |
| Refractory disease (%) | 21/67 (31%) |
| Previous Lines of Salvage | |
| 0 | 48 (72%) |
| 1 | 9/67 (13%) |
| 2 | 7/67 (10%) |
| >2 | 3/67 (4%) |
| Therapy-related AML | 6/67 (9%) |
| Antecedent hematologic disorder | 5/67 (7%) |
| Prior Allogeneic Transplant | 5/67 (7%) |
| Prior HIDAC+Mitoxantrone | 5/67 (7%) |
| Previous HIDAC or IDAC based salvage | 17/67 (25%) |
| Cytogenetic Risk Score | |
| Not applicable (CML in accelerated phase) | 1/67 (1%) |
| Good risk | 6/67 (9%) |
| Intermediate risk | 33/67 (49%) |
| Poor risk | 27/67 (40%) |
| FLT3-ITD (Tested in 37) | 11/37 (30%) |
| FLT3-TKD (Tested in 36) | 4/36 (11%) |
| NPM1 (Tested in 34) | 11/34 (32%) |
| CEBPα (Tested in 30) | 4/30 (13%) |
| >1 cycle of CPI-HIDAC-Mito | 41/67 (61%) |
| Median %Blasts in Marrow (Range) | 43% (4–97) |
Safety and Dose Escalation
The starting dose of CPI-613 was 500 mg/m2. The second patient (treated at 1,000 mg/m2) experienced a toxicity that triggered the expansion to the traditional 3+3 dose escalation cohorts (see supplemental figure 1). Once the dose reached 2250 mg/m2 without a DLT, an extended cohort dosing phase was initiated to ensure safety as the single agent maximally tolerated dose (MTD) of CPI-613 was previously determined to be 2940 mg/m2. In this phase cohorts of 6 patients each were treated starting at a dose of 1750 mg/m2 and escalating by 250 mg/m2 through 2500 mg/m2. Upon completion of these cohorts without reaching an MTD the dose was then escalated to 2750 mg/m2. At this dose level, one patient experienced a DLT in the form of grade 3 diarrhea not responsive to anti-diarrheals and another experienced a second DLT in the form of grade 3 nausea not responsive to anti-emetics. This established the MTD as 2,500 mg/m2. Once the MTD was determined to be 2,500 mg/m2 two additional cohorts of 6 patients each were treated at 1500 and 2000 mg/m2 to better assess dose response relationships. Supplemental table 1 shows the dose escalation and response of each patient. All patients who received even one dose of CPI-613 were included in the safety analysis (n=67). Overall, CPI-613 given with HiDAC and mitoxantrone was well tolerated. The most common toxicities (defined as experienced by at least 30% of patients) are listed in Table 2. Thirty-day mortality was 12% (8 patients) and 60-day mortality was 19% (13 patients). This was similar to the historical experience with a regimen of identical dose and schedule of HiDAC and mitoxantrone, but with the addition of asparaginase (HAMA) (30-day and 60-day mortalities of 13% and 22%, respectively) (17). The recommended phase 2 dose was established as 2,000 mg/m2, since there was a similar response rate and decreased toxicity compared to 2,500 mg/m2.
Table 2.
Summary of Toxicities (N=67 participants)
| Number of Subjects, by CTC Toxicity Grade | ||||||
|---|---|---|---|---|---|---|
| Toxicity | Grade 1 | Grade 2 | Grade 3 | Grade 4 | Grade 5 | Total |
| Hemoglobin | 0 | 5 | 53 | 9 | 0 | 67 |
| Hyperglycemia | 29 | 19 | 18 | 1 | 0 | 67 |
| Neutropenia | 0 | 0 | 1 | 66 | 0 | 67 |
| Thrombocytopenia | 0 | 0 | 1 | 66 | 0 | 67 |
| Hypomagnesemia | 61 | 4 | 1 | 0 | 0 | 66 |
| Leukopenia | 0 | 0 | 0 | 66 | 0 | 66 |
| Lymphopenia | 0 | 0 | 0 | 66 | 0 | 66 |
| Hypoalbuminemia | 17 | 44 | 4 | 0 | 0 | 65 |
| Hypokalemia | 37 | 0 | 23 | 0 | 0 | 60 |
| Hypocalcemia | 18 | 29 | 9 | 1 | 0 | 57 |
| Diarrhea | 10 | 32 | 13 | 0 | 0 | 55 |
| Hyponatremia | 38 | 0 | 8 | 1 | 0 | 47 |
| Hypophosphatemia | 0 | 22 | 24 | 0 | 0 | 46 |
| DIC | 0 | 32 | 13 | 1 | 0 | 46 |
| AST, SGOT elevation | 28 | 6 | 7 | 0 | 0 | 41 |
| Nausea | 21 | 16 | 1 | 0 | 0 | 38 |
| Hyperbilirubinemia | 21 | 10 | 5 | 1 | 0 | 37 |
| ALT, SGPT elevation | 23 | 9 | 4 | 0 | 0 | 36 |
| Fatigue | 10 | 20 | 3 | 0 | 0 | 33 |
| Proteinuria | 18 | 15 | 0 | 0 | 0 | 33 |
| PTT prolongation | 25 | 1 | 5 | 0 | 0 | 31 |
| Alkaline phosphatase elevation | 24 | 6 | 0 | 0 | 0 | 30 |
| Febrile neutropenia | 0 | 0 | 27 | 1 | 0 | 28 |
| Cardiac troponin T | 6 | 7 | 8 | 6 | 0 | 27 |
| LV systolic dysfunction | 16 | 6 | 4 | 0 | 0 | 26 |
| Head/headache | 16 | 9 | 1 | 0 | 0 | 26 |
| Prolonged QTc interval | 9 | 6 | 9 | 1 | 0 | 25 |
| Cough | 22 | 3 | 0 | 0 | 0 | 25 |
| Dyspnea | 4 | 5 | 13 | 2 | 0 | 24 |
| Bicarbonate, serum-low | 19 | 5 | 0 | 0 | 0 | 24 |
| Edema: limb | 17 | 7 | 0 | 0 | 0 | 24 |
| Anorexia | 9 | 10 | 4 | 0 | 0 | 23 |
| Vomiting | 15 | 8 | 0 | 0 | 0 | 23 |
| Hypotension | 3 | 13 | 3 | 2 | 1 | 22 |
| Pleural effusion | 5 | 12 | 3 | 2 | 0 | 22 |
Efficacy
Of the 67 patients enrolled, 1 did not complete the first cycle, 3 died before the day 14 bone marrow biopsy, and 1 with accelerated-phase CML was enrolled in error. Responses by dosing cohort are presented in table 3. Median survival for all evaluable patients was 6.7 months (supplemental figure 5A). Patients achieving a CR/CRi had a median survival of 13.2 months. In patients ≥60 years of age, the CR/CRi rate was 47% (15/32) with a median survival of 6.9 months. Interesting, this survival was not different from patients under the age of 60 (supplemental figure 5B). The response rate for patients with poor risk cytogenetics also was encouraging with 46% (11 of 24 patients) achieving a CR or CRi. These data suggest this approach may be especially effective in older patients or those with poor risk cytogenetics.
Table 3.
Efficacy by Dose Level Among 62 Evaluable Subjects
| Response | |
|---|---|
| 1000 mg/m2 | Evaluable n=5 |
| CR | 2/5 (40%) |
| CRi | 1/5 (20%) |
| MFLS | 2/5 (40%) |
| ORR (CR+CRi+MLFS) | 5/5 (100%) |
| 1500 mg/m2 | Evaluable n=9 |
| CR | 6/9 (67%) |
| CRi | 0/9 (0%) |
| MFLS | 0/9 (0%) |
| ORR (CR+CRi+MLFS) | 6/9 (67%) |
| 1750 mg/m2 | Evaluable n=9 |
| CR | 4/9 (44%) |
| CRi | 0/9 (0%) |
| MFLS | 0/9 (0%) |
| ORR (CR+CRi+MLFS) | 4/9 (44%) |
| 2000 mg/m2 | Evaluable n=15 |
| CR | 7/15 (47%) |
| CRi | 1/15 (7%) |
| MFLS | 0 (0%) |
| ORR (CR+CRi+MLFS) | 8/15 (53%) |
| 2250 mg/m2 | Evaluable n=10 |
| CR | 2/10 (20%) |
| CRi | 2/10 (20%) |
| MFLS | 0/10 (0%) |
| ORR (CR+CRi+MLFS) | 4/10 (40%) |
| 2500 mg/m2 | Evaluable n=8 |
| CR | 2/8 (25%) |
| CRi | 0/8 (0%) |
| MFLS | 0/8 (0%) |
| ORR (CR+CRi+MLFS) | 2/8 (25%) |
Potential Biomarker Analysis
Transcriptomes of mononuclear cells isolated from baseline bone marrow biopsies taken from 10 responders and 7 non-responders were analyzed. Using RNAseq and bioinformatics analysis, 228 and 63 gene sequences (Ensemble Gene IDs) significantly overexpressed in responders and non-responders, respectively, were identified. Analysis for enrichment of gene ontologies revealed multiple statistically significant ontological terms among the genes overexpressed in responders, while none were identified among the genes overexpressed in non-responders. Genes overexpressed in responders were highly significantly enriched in GO categories related to immune system activation and B-cell biology (supplemental table 2). CD79A, MS4A1 (CD20), FCRL2, TCL1A, and BANK1 were among the top 20 most differentially expressed genes (false discovery adjusted p-values <0.01) (supplemental figure 6). All are in the upper 99th percentile of genes most significantly enriched in CD19+ B cells (compared to a diverse compendium of FACs-sorted immune cell types (19)). Baseline blast percentage in the marrow was not significantly different between responders and non-responders (50 vs 64%, p=0.3467). This suggests that the presence of certain immune cells in the marrow at baseline may influence response to combined CPI-613 and chemotherapy.
In addition to the unbiased approach taken above the expression data was also analyzed for genes involved in mitochondrial metabolism. No clear differences were seen in expression levels of the target enzymes of CPI-613 or the regulating kinases PDK1-4 between responders and non-responders. SOD2 is a key enzyme that protects mitochondria from oxidative damage including in cancer (reviewed by Soo Kim et al (20)). Higher levels of SOD2 would allow for increased oxidative capacity of leukemia cell mitochondria and possible resistance to the combination of CPI-613 and chemotherapy. Consistent with this there was a significant increase in SOD2 expression levels in patients who did not respond (supplemental figure 7). Both OCI-AML3 and MFL2 cell lines expressed SOD2 (supplemental figure 8A) and this may contribute to the micromolar IC50 seen with CPI-613 as a single agent. Consistent with this when SOD2 was deleted in Cas-9 expressing MFL2 AML cells there was significantly increased sensitivity to CPI-613 (supplemental figure 8B). Additionally, SOD2 expression was not altered in PDH deleted cells (supplemental figure 8A) arguing that the sensitivity seen in these cells does not stem from a decrease in SOD2.
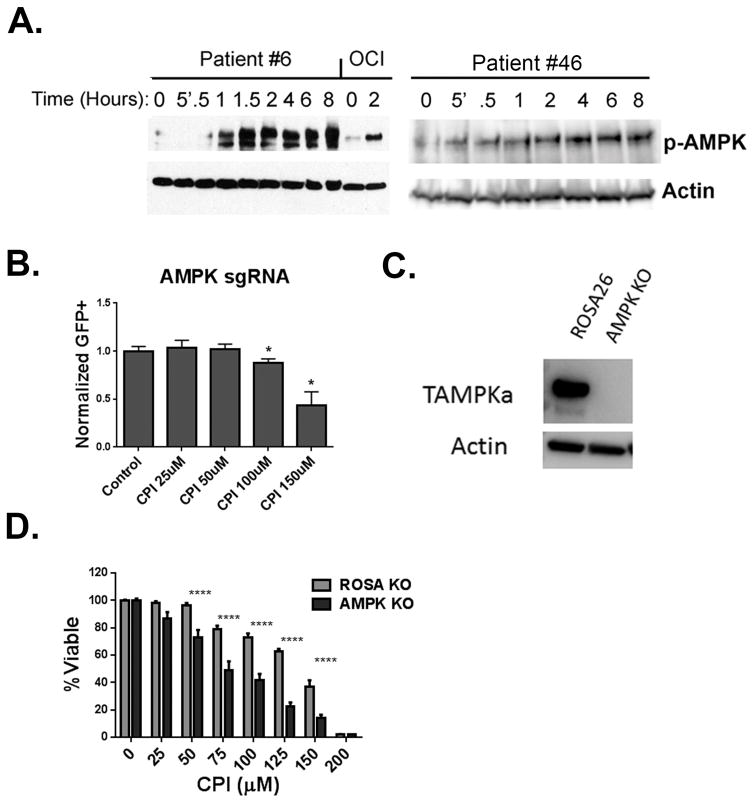
Previous studies have shown that Adenosine Monophosphate Activated Kinase (AMPK) is activated by CPI-613 in AML cells (9). To determine if this activation could be detected in patients, blood samples taken prior to and following administration of CPI-613 on day 1 were subjected to Western blotting for phosphorylated AMPK. Blood samples from two of the five patients who had circulating leukemia cells and sufficient quality samples to assay demonstrated a robust phosphorylation of AMPK (Figure 5A). Of note neither patient achieved a remission. While no conclusions can be drawn on the basis of two patients the known role of AMPK in metabolic stress response (21) suggests that it’s activation by CPI-613 may represent an adaptive response leading to resistance. The role of AMPK in response to CPI-613 was assessed by competition assays using a mixed population of Cas-9 expressing AML cells some of which express a sgRNA that targets AMPK resulting in its deletion. Normal cells out-competed AMPK deleted cells when treated with CPI-613 suggesting AMPK is a resistance factor against TCA cycle inhibition (Figure 5B). This was confirmed by performing viability assays of pure populations of AMPK deleted and ROSA26 targeted control cells. Knockout of AMPK was confirmed by Western blot (Figure 5C). Consistent with the competition assays AMPK deleted cells were more sensitive to CPI-613 (Figure 5D). These data show that the AMPK response protects cells from the metabolic stress induced by CPI-613.
Figure 5. AMPK is a source of resistance to CPI-613.
(A) Western blot analysis of patient samples taken on cycle 1 day 1 at the indicated time points before and following infusion of CPI-613 for phosphorylation of AMPK. OCI lanes refer to the positive control, OCI-AML3 cells treated with CPI-613 for the indicated time. Actin was used as a loading control. (B) Competition assay. MFL2 cells expressing Cas9 were partially infected with GFP labeled vector that expressed an sgRNA targeting AMPK in conjunction with a GFP reporter. The mixed population was treated with the indicated therapy for 72 hours and the percentage of GFP+ cells was assessed and normalized to controls. (C) Western blot analysis of control or AMPK-deleted cells. Actin was used as a loading control. (D) Viability assays. Control or AMPK deleted cells were incubated with the indicated therapy for 72 hours in viability was assessed.
Finally, the RNA sequence data was also analyzed for mutation data with the following criteria: sufficient data to make a mutation call, not previously reported as a SNP in dbSNP or the 1,000 genome database and appeared in COSMIC in AML patients. Given the small dataset there were no significant differences in the non-synonymous mutation rates in the genes examined between responders and non-responders. Despite this fact several genes showed an uneven distribution. For example AXSL2 was mutated in 3/7 non-responders but only 1/10 responders and conversely NSD1 was mutated in 4/10 responders but only 1/7 non-responders (supplemental figure 9) among others. The significance of these mutations is the subject of ongoing work.
Discussion
The contribution of mitochondrial metabolism to resistance in AML remains unclear. In this study, it was observed that following chemotherapy exposure, AML cells increase mitochondrial OCR, which was related to ATP production, as differences in OCR were abrogated when the F0F1 ATP synthase inhibitor oligomycin was added. The increased OCR was a consequence of glucose-derived carbon entry into the TCA cycle, since cells that had PDH deleted by Cas9 had a minimal OCR at baseline that did not increase following chemotherapy exposure. Additionally, citrate and glutamate levels (a proxy for α-ketogluterate) were higher in control cells at baseline and fell in response to chemotherapy, while PDH-deleted cells had low levels of citrate and glutamate that did not vary following chemotherapy exposure. Additionally, glycolytic rates as measured by ECAR were higher in control cells and did not increase following therapy. Control cells also exhibited a significant decrease in glucose-6-phosphate levels following therapy. In contrast, PDH-deleted cells showed a significant increase ECAR following chemotherapy treatment. These data are consistent with a model where AML cells following exposure to chemotherapy increase glycolysis but shunt the additional pyruvate into the TCA cycle to be oxidized and increase ATP levels, presumably to drive costly DNA damage repair processes. This idea is further supported by the fact that PDH-deficient cells were significantly more sensitive to chemotherapy in vitro and in vivo. These results corroborate a recent study that found cytarabine resistant AML cells are enriched in mitochondrial mass and have a high oxidative phosphorylation status (6). The importance of mitochondrial function in AML was further supported by a recent study that showed that inhibiting mitochondrial DNA replication was cytotoxic in several preclinical AML models (22). Additionally, studies in chronic myeloid leukemia have revealed that TKI resistant cells rely on mitochondrial oxidative phosphorylation and the addition of an antibiotic that inhibits mitochondrial translation improved response of a xenotransplantation model of human CML (23). Thus the data in the current study fit into a larger, emerging picture of the importance of mitochondrial metabolism in resistance to therapy (reviewed by Bosc et al. (24)).
CPI-613 is a novel lipoate analog that targets PDH and KGDH, blocking the entry of either glucose or glutamine-derived carbons into the TCA cycle (7, 8). CPI-613 inhibits mitochondrial respiration and stimulates pyruvate dehydrogenase kinases, resulting in an inhibitory phosphorylation of PDH in AML cell lines (9). Consistent with the PDH knock out studies, inhibition of PDH/KGDH by CPI-613 sensitized AML cells to chemotherapy at clinically relevant concentrations. As a result, a phase I study of CPI-613 plus HiDAC and mitoxantrone in patients with relapsed or refractory AML was conducted. This chemotherapy combination is the standard salvage regimen at the Comprehensive Cancer Center of Wake Forest Baptist Health and historic data using a similar regimen were recently published (17). The current regimen was identical to the historic regimen, except that Native E. Coli L-asparaginase was not included, as it was no longer commercially available when the study began. Importantly, addition of CPI-613 did not increase 30- or 60-day mortality compared to historic data. Dose-limiting toxicities were nausea and diarrhea (Table 2), similar to the single-agent trial (9).
Treatment with the combined regimen resulted in a CR/CRi rate of 50% of evaluable patients. The response rate in patients 60 years of age or older was 47%; historically, only 33% of older patients achieved a CR/CRi. In contrast to most studies of cytotoxic chemotherapy in AML, there was no difference in median survival between younger patients and those 60 years of age or older (supplemental figure 5B). One possible explanation for this result is the decreased mitochondrial quality associated with aging (25), producing a mitochondrial pool with low spare respiratory capacity (26). AML arising in an older host may lack the necessary reserve capacity to overcome inhibitory effects of CPI-613, resulting in increased response rates. If so, CPI-613 may be the first agent that specifically targets an aging-derived vulnerability of cancer. When compared to historical data increased response rate in patients with poor risk cytogenetics was also observed. This was especially apparent in older patients, with a response rate of 40% versus 15% in the historic cohort (although there were few patients). Much of the prognostic power of poor risk karyotype is conveyed by loss or mutation of p53 (27). Preclinical models of AML with p53 loss did not show altered responses to CPI-613, suggesting that TCA cycle inhibition may induce p53-independent cell death. While this data is encouraging historical comparisons must be interpreted with caution, and these data will need to be replicated in a randomized setting.
Given the heterogeneity in AML, selecting patients most likely to respond to TCA cycle inhibition plus chemotherapy is critical to further develop this approach. Gene expression profiles of baseline marrow samples suggested a possible role of immune cells, particularly B lymphocytes, in response to CPI-613 and chemotherapy. While regulatory B cells can suppress infiltrating T cell response to tumor, (28) tumor-infiltrating B cells may also assist in that response (29). Thus, the B cell signature in responders may reflect a B cell population important for generating humoral responses to leukemia, where CPI-613 then enabled an immune response. There was not a significant difference in the expression levels of several TCA cycle enzymes including the targets of CPI-613 between responders and non-responders. This maybe because the study treated patients with the combination of chemotherapy and CPI-613 and the levels of TCA cycle enzymes themselves may not predict response to the combination. In contrast, levels of SOD2 were significantly higher in non-responders and its genetic deletion in AML cells rendered them more sensitive to CPI-613 (supplemental figure 8B). SOD2 is the main reactive oxygen species scavenger in the mitochondrial matrix and increased levels likely contribute to increased capacity for oxidative metabolism. Indeed, SOD2 suppression has been implicated in chemo-sensitivity in ovarian cancer (30) and its increased expression is associated with metastatic prostate, colon and lung cancers (31). AMPK contributes to resistance to CPI-613 in preclinical AML models. This is consistent with the role of AMPK as a master metabolic regulator (21). This finding suggests that tumors that have lost functional AMPK may be particularly susceptible to metabolically directed attacks like TCA cycle inhibition. Consistent with this are reports that non-small cell lung tumors which have deleted the AMPK activating kinase LKB1 showed enhanced sensitivity to electron transport chain inhibition (32). Further studies will be required to fully assess the roles of SOD2 and AMPK as possible predictive markers for this combined approach. Finally, mutational data from the RNA sequence analysis did not show a statistically significant difference between responders and non-responders. However, the analysis was limited but the small number of samples and the limited set of genes with sufficient quality data to call a mutation with confidence. Several genes did show an increased, though not significant, mutation rate in either group however additional study is required to determine the significance of these mutations.
In summary, the novel PDH/KGDH inhibitor CPI-613 can be safely combined with HiDAC and mitoxantrone in relapsed or refractory AML patients. Pharmacologic inhibition of PDH and KGDH by CPI-613 appeared to sensitize AML cells to chemotherapy and response rates of CPI-613 combined with HiDAC and mitoxantrone compared favorably with historical cohorts of similarly treated patients. To our knowledge, this is the first report documenting feasibility of adding a TCA cycle inhibitor to chemotherapy for AML. We believe these results justify a randomized trial to further explore this approach.
Supplementary Material
Translational Relevance.
AML is an aggressive malignancy of the bone marrow with poor outcomes. Recent studies have shown a general role for mitochondrial metabolism in resistance to therapy in AML. The specific roles of PDH and KGDH in therapy response are unknown and no previous clinical studies have looked at inhibitors of these enzymes in combination with chemotherapy. When leukemia cells were exposed to chemotherapy they increased their mitochondrial oxygen consumption consistent with increased tricarboxylic acid (TCA) cycle activity. This effect was dependent on PDH activity and genetic deletion or inhibition with CPI-613 induced sensitivity to chemotherapy. In a phase I trial of CPI-613 in combination with chemotherapy for relapsed or refractory AML patients response rates and survival of older patients or those with poor risk cytogenetics was encouraging. This suggests that inhibition of mitochondrial metabolism with CPI-613 sensitizes AML to chemotherapy, especially in the elderly.
Acknowledgments
Support was provided by the Proteomics and Metabolomics Shared Resource Facility Director Cristina M. Furdui, PhD, and facility personnel, Mr. Anderson O’Brien Cox. Support was also provided by the Bioinformatics Shared Resource. We also thank Brian Mullaney and Chi Van Dang for their critical reading of the manuscript and Howard Jonas, Sanjeev Luther, Paul Bingham and Rob Shor from Rafael Pharmaceuticals for insightful comments and CPI-613 for both preclinical and clinical work. We acknowledge the editorial assistance of Karen Klein, MA, in the Wake Forest Clinical and Translational Science Institute (UL1 TR001420; PI: McClain).
Funding: TSP, RGA, LPG and KMP are supported by 1R01CA197991-01A1. The Cancer Genomics Shared Resource, Bioinformatics Shared Resources, Proteomics and Metabolomics Shared Resource, and the Biostatistics and Bioinformatics Shared Resource contributed to this work and are supported by P30CA012197.
Footnotes
Conflicts of Interest: TSP and BLP are paid consultants of Rafael Pharmaceuticals and TSP is their Chief Medical Officer. Rafael Pharmaceuticals owns the licensing rights to CPI-613 and is currently developing it for use in oncology patients. Rafael Pharmaceuticals had no input over the design of the protocol as it was initiated as an investigator sponsored trial by TSP prior to his consultancy and role as Chief Medical Officer. Rafael Pharmaceuticals had no approval rights over the manuscript. All other authors have nothing to disclose.
Authorship: TSP conceived of experiments, interpreted data, conducted the clinical trial, and wrote the manuscript. RGA, LPG and KMP conducted experiments and revised the manuscript. LDM, JWC and SS analyzed data and edited the manuscript. LRE, DB, DSH, DDH, MM, SD, SL and BLP enrolled patients and edited the manuscript.
Data and materials availability: RNA seq data will be deposited in the Gene Expression Omnibus (GEO).
References
- 1.Dohner H, Estey EH, Amadori S, Appelbaum FR, Buchner T, Burnett AK, et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood. 2010;115:453–74. doi: 10.1182/blood-2009-07-235358. [DOI] [PubMed] [Google Scholar]
- 2.Yates JW, Wallace HJ, Jr, Ellison RR, Holland JF. Cytosine arabinoside (NSC-63878) and daunorubicin (NSC-83142) therapy in acute nonlymphocytic leukemia. Cancer chemotherapy reports Part 1. 1973;57:485–8. [PubMed] [Google Scholar]
- 3.Bennett JM, Young ML, Andersen JW, Cassileth PA, Tallman MS, Paietta E, et al. Long-term survival in acute myeloid leukemia: the Eastern Cooperative Oncology Group experience. Cancer. 1997;80:2205–9. [PubMed] [Google Scholar]
- 4.Medeiros BC, Fathi AT, DiNardo CD, Pollyea DA, Chan SM, Swords R. Isocitrate dehydrogenase mutations in myeloid malignancies. Leukemia. 2017;31:272–81. doi: 10.1038/leu.2016.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Samudio I, Harmancey R, Fiegl M, Kantarjian H, Konopleva M, Korchin B, et al. Pharmacologic inhibition of fatty acid oxidation sensitizes human leukemia cells to apoptosis induction. The Journal of clinical investigation. 2010;120:142–56. doi: 10.1172/JCI38942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Farge T, Saland E, de Toni F, Aroua N, Hosseini M, Perry R, et al. Chemotherapy-Resistant Human Acute Myeloid Leukemia Cells Are Not Enriched for Leukemic Stem Cells but Require Oxidative Metabolism. Cancer discovery. 2017;7:716–35. doi: 10.1158/2159-8290.CD-16-0441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stuart SD, Schauble A, Gupta S, Kennedy AD, Keppler BR, Bingham PM, et al. A strategically designed small molecule attacks alpha-ketoglutarate dehydrogenase in tumor cells through a redox process. Cancer & metabolism. 2014;2:4. doi: 10.1186/2049-3002-2-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zachar Z, Marecek J, Maturo C, Gupta S, Stuart SD, Howell K, et al. Non-redox-active lipoate derivates disrupt cancer cell mitochondrial metabolism and are potent anticancer agents in vivo. Journal of molecular medicine. 2011;89:1137–48. doi: 10.1007/s00109-011-0785-8. [DOI] [PubMed] [Google Scholar]
- 9.Pardee TS, Lee K, Luddy J, Maturo C, Rodriguez RJ, Isom S, et al. A Phase I Study of the First-in-Class Anti-Mitochondrial Metabolism Agent, CPI-613, in Patients with Advanced Hematologic Malignancies. Clinical cancer research : an official journal of the American Association for Cancer Research. 2014 doi: 10.1158/1078-0432.CCR-14-1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Le Tourneau C, Lee JJ, Siu LL. Dose escalation methods in phase I cancer clinical trials. J Natl Cancer Inst. 2009;101:708–20. doi: 10.1093/jnci/djp079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pardee TS, Gomes E, Jennings-Gee J, Caudell D, Gmeiner WH. Unique dual targeting of thymidylate synthase and topoisomerase1 by FdUMP[10] results in high efficacy against AML and low toxicity. Blood. 2012;119:3561–70. doi: 10.1182/blood-2011-06-362442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pardee TS, Zuber J, Lowe SW. F1t3-ITD alters chemotherapy response in vitro and in vivo in a p53-dependent manner. Experimental hematology. 2011;39:473–85. doi: 10.1016/j.exphem.2011.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dobin A, Davis CA, Schlesinger F, Drenkow J, Zaleski C, Jha S, et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics. 2013;29:15–21. doi: 10.1093/bioinformatics/bts635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liao Y, Smyth GK, Shi W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics. 2014;30:923–30. doi: 10.1093/bioinformatics/btt656. [DOI] [PubMed] [Google Scholar]
- 15.Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome biology. 2014;15:550. doi: 10.1186/s13059-014-0550-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huang DW, Sherman BT, Tan Q, Kir J, Liu D, Bryant D, et al. DAVID Bioinformatics Resources: expanded annotation database and novel algorithms to better extract biology from large gene lists. Nucleic acids research. 2007;35:W169–75. doi: 10.1093/nar/gkm415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ahmed T, Holwerda S, Klepin HD, Isom S, Ellis LR, Lyerly S, et al. High dose cytarabine, mitoxantrone and l-asparaginase (HAMA) salvage for relapsed or refractory acute myeloid leukemia (AML) in the elderly. Leukemia research. 2015;39:945–9. doi: 10.1016/j.leukres.2015.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zuber J, Radtke I, Pardee TS, Zhao Z, Rappaport AR, Luo WJ, et al. Mouse models of human AML accurately predict chemotherapy response. Genes & development. 2009;23:877–89. doi: 10.1101/gad.1771409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Abbas AR, Baldwin D, Ma Y, Ouyang W, Gurney A, Martin F, et al. Immune response in silico (IRIS): immune-specific genes identified from a compendium of microarray expression data. Genes Immun. 2005;6:319–31. doi: 10.1038/sj.gene.6364173. [DOI] [PubMed] [Google Scholar]
- 20.Kim YS, Gupta Vallur P, Phaeton R, Mythreye K, Hempel N. Insights into the Dichotomous Regulation of SOD2 in Cancer. Antioxidants (Basel) 2017:6. doi: 10.3390/antiox6040086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jeon SM. Regulation and function of AMPK in physiology and diseases. Exp Mol Med. 2016;48:e245. doi: 10.1038/emm.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liyanage SU, Hurren R, Voisin V, Bridon G, Wang X, Xu C, et al. Leveraging increased cytoplasmic nucleoside kinase activity to target mtDNA and oxidative phosphorylation in AML. Blood. 2017;129:2657–66. doi: 10.1182/blood-2016-10-741207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kuntz EM, Baquero P, Michie AM, Dunn K, Tardito S, Holyoake TL, et al. Targeting mitochondrial oxidative phosphorylation eradicates therapy-resistant chronic myeloid leukemia stem cells. Nat Med. 2017;23:1234–40. doi: 10.1038/nm.4399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bosc C, Selak MA, Sarry JE. Resistance Is Futile: Targeting Mitochondrial Energetics and Metabolism to Overcome Drug Resistance in Cancer Treatment. Cell metabolism. 2017;26:705–7. doi: 10.1016/j.cmet.2017.10.013. [DOI] [PubMed] [Google Scholar]
- 25.Kauppila TE, Kauppila JH, Larsson NG. Mammalian Mitochondria and Aging: An Update. Cell metabolism. 2017;25:57–71. doi: 10.1016/j.cmet.2016.09.017. [DOI] [PubMed] [Google Scholar]
- 26.Tyrrell DJ, Bharadwaj MS, Van Horn CG, Marsh AP, Nicklas BJ, Molina AJ. Blood-cell bioenergetics are associated with physical function and inflammation in overweight/obese older adults. Exp Gerontol. 2015;70:84–91. doi: 10.1016/j.exger.2015.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rucker FG, Schlenk RF, Bullinger L, Kayser S, Teleanu V, Kett H, et al. TP53 alterations in acute myeloid leukemia with complex karyotype correlate with specific copy number alterations, monosomal karyotype, and dismal outcome. Blood. 2012;119:2114–21. doi: 10.1182/blood-2011-08-375758. [DOI] [PubMed] [Google Scholar]
- 28.Zhang Y, Eliav Y, Shin SU, Schreiber TH, Podack ER, Tadmor T, et al. B lymphocyte inhibition of anti-tumor response depends on expansion of Treg but is independent of B-cell IL-10 secretion. Cancer Immunol Immunother. 2013;62:87–99. doi: 10.1007/s00262-012-1313-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Haro MA, Littrell CA, Yin Z, Huang X, Haas KM. PD-1 Suppresses Development of Humoral Responses That Protect against Tn-Bearing Tumors. Cancer Immunol Res. 2016;4:1027–37. doi: 10.1158/2326-6066.CIR-16-0184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cui Y, She K, Tian D, Zhang P, Xin X. miR-146a Inhibits Proliferation and Enhances Chemosensitivity in Epithelial Ovarian Cancer via Reduction of SOD2. Oncol Res. 2016;23:275–82. doi: 10.3727/096504016X14562725373798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Miar A, Hevia D, Munoz-Cimadevilla H, Astudillo A, Velasco J, Sainz RM, et al. Manganese superoxide dismutase (SOD2/MnSOD)/catalase and SOD2/GPx1 ratios as biomarkers for tumor progression and metastasis in prostate, colon, and lung cancer. Free Radic Biol Med. 2015;85:45–55. doi: 10.1016/j.freeradbiomed.2015.04.001. [DOI] [PubMed] [Google Scholar]
- 32.Shackelford DB, Abt E, Gerken L, Vasquez DS, Seki A, Leblanc M, et al. LKB1 inactivation dictates therapeutic response of non-small cell lung cancer to the metabolism drug phenformin. Cancer cell. 2013;23:143–58. doi: 10.1016/j.ccr.2012.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.