Abstract
Since microglia possess both neuroprotective and neurotoxic potential, they play a crucial role in the central nervous system (CNS). Excessive microglial activation induces inflammation-mediated neuronal damage and degeneration. At present, numerous herbal compounds are able to suppress neurotoxicity via inhibiting microglial activation. Therefore, many researchers focus on pharmacological inhibitors of microglial activation to ameliorate neurodegenerative disorders. Further work should concentrate on the exploration of new herbal compounds, which characteristically inhibit microglial neurotoxicity, rather than modulating neuroprotection alone. In this review, we summarize these herbal compounds, which in the past several years have been shown to exert potential neuroprotective activity by inhibiting microglial activation. The therapeutic targets and pharmacological mechanisms of these compounds have also been discussed.
1. Introduction
A large amount of evidence has demonstrated that neuroinflammation plays a significant role in both acute and chronic neurodegenerative disorders including Parkinson's disease, Alzheimer's disease, multiple sclerosis, stroke, and traumatic brain injury (TBI) [1–3]. They are all related to microglial activation and are accompanied by high expression of proinflammatory mediators. Neuroinflammation is a defense mechanism with the purpose of preventing the CNS from being infected and damaged. Microglia, the resident macrophages of the CNS, act as the primary effector cells in mediating neuroinflammation, the activation of which is characteristic of several inflammatory and neurodegenerative disorders [4–7]. Microglia support the normal function of neurons and monitor the health of neurons in homeostasis, the resting state. Therefore, microglia display beneficial effects in normal conditions. Once brain injury or infection occurs, microglia turn into the activated state and secrete a series of proinflammatory and neurotoxic mediators, such as interleukin-1 beta (IL-1β), nitric oxide (NO), tumor necrosis factor alpha (TNF-α), and reactive oxygen species (ROS), which not only regulate neuronal function and synaptic transmission but also give rise to neuronal oxidative stress and degeneration associated with deficits in a variety of cognitive and memory tasks [8–10]. As mentioned above, activated microglia could cause and regulate the neuroinflammatory reaction by impairing neurons with a mass of proinflammatory mediators that may result in neuronal death in the end.
Thus, protecting neurons through suppressing microglial activation and neuroinflammation may be considered as a potential therapeutic method for improving neurodegenerative disorders. A number of studies proved that natural plants and their active ingredients could ameliorate neurodegenerative progression by suppressing microglial activation and neuronal damage [11, 12]. This article discusses the conclusions about natural compounds, which based on recent studies could prevent neurons from damage via inhibiting microglial activation and neuroinflammation.
2. Herbal Compounds
2.1. Resveratrol
Resveratrol is a polyphenolic phytochemical that is extracted from plants including grape, peanut, and berry with pharmacological effects on multiple pathological phenomena [13, 14]. Research has shown that resveratrol has bioactivity-containing antioxidative, anti-inflammatory, and anticancer properties and neuroprotection [15–17].
Myeloperoxidase (MPO) plays a very important role in the host defense system against many pathogens. Research has demonstrated that both overactivation and deficiency of MPO result in a pathological state in the brain. However, resveratrol remarkably decreases MPO levels and NO production, which obviously suppressed neuroinflammatory responses including phagocytosis and ROS production in rotenone-triggered microglia. Resveratrol could alleviate the rotenone-induced impaired responses of primary mixed glia from MPO−/− mice. In neuron-glia cocultures, the impairment of neurons could be relieved by resveratrol. The results displayed that resveratrol affected the microglial response to rotenone via modulating MPO and thus prevented neurons from rotenone-induced injury. As mentioned above, the regulation of MPO levels in microglia by resveratrol provide its neuroprotective ability [18]. Resveratrol remarkably improved trigeminal allodynia dose dependently and reduced the high levels of calcitonin gene-related peptide and c-Fos expression in the spinal trigeminal nucleus. In addition, resveratrol inhibited chronic constriction injury-provoked astrocyte and microglial activation and decreased the levels of proinflammatory mediators in the spinal trigeminal nucleus. Moreover, the effect of resveratrol on pain relief was partially regulated via suppressing the phosphorylation of mitogen-activated protein kinases (MAPKs) through the activation of adenosine monophosphate-activated protein kinase [19]. A study demonstrated that in BV2 microglial cell lines, resveratrol could inhibit NLR family pyrin domain containing 3 (NLRP3) activation and IL-1β cleavage caused by ATP. In summary, resveratrol could alleviate the deficit of spatial memory in mice with sepsis-associated encephalopathy by suppressing the NLRP3/IL-1β axis in microglia [20]. Resveratrol could not only reduce nicotinamide adenine dinucleotide phosphate (NADPH) oxidase-induced level of ROS generation but also alleviate the transposition of the subunit of NADPH oxidase to the cytomembrane caused by lipopolysaccharide (LPS). Furthermore, the effects of resveratrol on neuroprotection were also relevant to the suppression of the activation of MAPKs and nuclear factor-kappa B (NF-κB) signaling pathways in microglia. The study explicitly revealed that resveratrol could prevent dopaminergic neurons from being damaged by LPS, which depended on time and concentration by inhibiting microglial activation and the expression of proinflammatory mediators [21].
The other research has demonstrated that resveratrol treatment in rat with subarachnoid hemorrhage (SAH) could obviously decrease the expression of Toll-like receptor 4 (TLR4), high-mobility group box1 protein, myeloid differentiation factor (MyD88), and NF-κB. In addition, resveratrol remarkably inhibited microglial activation and proinflammatory mediators, which gave rise to the alleviation of neuronal apoptosis, cerebral edema, and behavior deficits at 24 h after SAH [22]. In conclusion, resveratrol is capable of exerting neuroprotection via suppressing microglial activation through the blockage of related pathways, such as the TLR4/MAPK and NF-κB pathways.
2.2. Gastrodin
Gastrodin is the primary bioactive component derived from the traditional Chinese herb Gastrodia elata Blume root and has been widely used as an anticonvulsant, analgesic, anti-inflammatory, antioxidative, and sedative agent [23, 24].
The research indicated that gastrodin remarkably decreased the levels of proinflammatory mediators such as cyclooxygenase-2 (COX-2), TNF-α, inducible nitric oxide synthase (iNOS), and IL-1β via blocking the activation of the NF-κB and MAPK pathways in microglia induced by LPS [25]. Li et al. revealed that gastrodin could protect dopaminergic neurons via obviously suppressing microglial activation and the level of IL-1β, COX-2, and iNOS in the substantia nigra of rotenone-induced rats with Parkinson's disease [26]. The other research also reported that gastrodin was capable of significantly improving chronic inflammatory pain and the accompanying anxiety-like behaviors in mice induced by complete Freund's adjuvant (CFA). Furthermore, gastrodin treatment could downregulate the increasing expression of glutamate receptor 1, N-methyl-d-aspartate receptor subunit 2A, N-methyl-d-aspartate receptor subunit 2B, and Ca2+/calmodulin-dependent protein kinase II-alpha by reducing microglial activation and proinflammatory mediators such as TNF-α and IL-6 in the anterior cingulate cortex of mice with CFA injection [24].
Recently, there was also significant evidence indicating that gastrodin elicited strong neuroprotective effects against loss of retinal ganglion cells in an acute glaucoma rat via inhibiting phosphorylated p38 MAPK and the production of proinflammatory mediators in activated retinal microglia. The results demonstrated that gastrodin possessed a potential therapeutic effect on acute glaucoma and other retinal neurodegenerative diseases by suppressing microglial activation [27]. In conclusion, gastrodin is a new drug that could protect neurons through inhibiting microglial activation.
2.3. Trans-Cinnamaldehyde
Trans-cinnamaldehyde (TCA) is a main component isolated from the stem bark of Cinnamomum cassia, which has been reported to have anti-inflammatory, antioxidative, antibacterial, antifungal, and antiapoptotic properties in a large amount of in vitro and in vivo models [28–30].
A study revealed that TCA could decrease viability loss and apoptosis in neuronal PC12 cells induced by oxygen and glucose deprivation/reperfusion. The effect indicated that TCA could reduce the production of NO. Additionally, using LY294002, the inhibitor of phosphoinositide 3-kinase (PI3K), could abolish the neuroprotection of TCA, demonstrating that the neuroprotection of TCA can be induced via provoking the PI3K pathway [31]. Other studies confirmed that TCA could inhibit LPS-induced inflammation in BV2 cells and reduce the infarction area and neurological deficit score in injured cerebral tissue of mice induced with ischemia/reperfusion. Furthermore, TCA could obviously alleviate neuronal damage by decreasing the levels of iNOS and COX-2 expression through blocking the NF-κB pathway in injured cerebral tissue of mice induced with ischemia/reperfusion. Therefore, TCA may recede neuroinflammation by suppressing microglial activation and play a key role in neuroprotection [32]. Recent research also showed that TCA could promote the degradation of iNOS mRNA in LPS-induced microglia, thus reducing NO production. Additionally, TCA could not only significantly decrease the expression of iNOS and phosphorylated extracellular signal-regulated kinase 1/2 (ERK1/2) in the hippocampus but also evidently alleviate memory deficits and synaptic plasticity damage in LPS-induced mice. They concluded that TCA improved neuronal damage by suppressing microglial activation via degrading the stability of iNOS mRNA [33]. In addition, TCA decreased the expression of iNOS and COX-2 in the LPS-stimulated BV2 cells and noticeably increased the number of tyrosine hydroxylase-positive dopaminergic neurons in the striatum and substantia nigra of mice with 6-hydroxydopamine challenge. These data indicated that TCA has a function of neuroprotection on dopaminergic neurons, which is associated with the suppression of neuroinflammatory responses induced by microglial activation [34]. As mentioned above, TCA has the potential to prevent neuronal damage via inhibiting microglial activation.
2.4. Salvianolic Acid B
Salvianolic acid B (Sal B) is the main active ingredient as a water-soluble component of Salvia miltiorrhiza roots (Danshen). The studies revealed that Sal B possessed anticancer activity [35, 36]. Other researches confirmed the therapeutic potential of Sal B on hepatic protection, cardiovascular protection, and neuroprotection [37, 38].
In recent research, Sal B could suppress neutrophil infiltration and microglial activation after TBI. Salvianolic acid B could not only reduce the productions of proinflammatory mediators such as TNF-α and IL-1β but could also upregulate the levels of anti-inflammatory mediators such as IL-10 and transforming growth factor beta 1. These results demonstrated that the neuroprotective role of Sal B on the TBI model may be related to its anti-inflammatory effects [38]. Research has shown that Sal B could reduce the mRNA levels of iNOS, TNF-α, and IL-1β in LPS-stimulated microglia by decreasing NF-κB activation. Moreover, Sal B could prevent neuronal damage via the inhibition of microglial activation in a coculture system including microglia and neurons [39]. A study demonstrated that Sal B treatment remarkably lessened the infarction volume and neuroinflammation in the middle cerebral artery occlusion rat model. The TLR4/NF-κB pathway could be significantly suppressed by Sal B treatment in the ischemic hemisphere via inhibiting the activation of microglia. Meanwhile, the secretion of IL-1β and IL-6 could be decreased by Sal B. This study confirmed that Sal B could significantly alleviate brain damage following cerebral ischemia by inhibiting inflammation in activated microglia [40]. In conclusion, Sal B is a potential herbal compound to improve neuronal damage through inhibiting microglial activation and neuroinflammation.
2.5. Tanshinone
Tanshinone is one of the constituents extracted from Salvia miltiorrhiza roots, containing tanshinone I and tanshinone IIA. Tanshinone I is one of the critical active ingredients and exhibits many bioactivities, including antioxidative and anti-inflammatory activities in several laboratorial models [41–43]. Research displayed that tanshinone I could destroy the biomembrane reactor in vitro and decrease the bacterial content in vivo [44]. Tanshinone I could protect mitochondria via the nuclear factor erythroid 2-related factor 2-dependent mechanism in SH-SY5Y cells induced by paraquat [45].
Further studies revealed that tanshinone I could significantly decrease the production of several proinflammatory mediators including TNF-α, NO, IL-1β, and IL-6 and also distinctly inhibit NF-κB activation in activated M1 microglia stimulated by LPS. Furthermore, tanshinone I had the ability to improve motor function, normalize striatal neurotransmitters, and protect dopaminergic neurons in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine- (MPTP-) intoxicated mice. The animal studies also revealed that tanshinone I might reduce the increase in TNF-α and IL-10 concentrations through modulating microglial activation in MPTP-intoxicated mice. Therefore, tanshinone I has the potential to protect nigrostriatal dopaminergic neurons by decreasing the level of proinflammatory mediators through the inhibition of NF-κB activation in microglia [46].
Tanshinone IIA is also an active constituent of Salvia miltiorrhiza and has been widely used for many years in Asia to treat various diseases for its observable organ protective activities [47, 48]. Research showed that MPTP could not only damage nigrostriatal dopaminergic neurons but also induce microglial activation. Western blot and immunohistochemistry revealed that MPTP could increase the expression of NADPH oxidase and iNOS in substantia nigra pars compacta. In addition, the impairment of nigrostriatal dopaminergic neurons and the high expression of NADPH oxidase and iNOS could be reversed by tanshinone IIA treatment. Thus, tanshinone IIA could protect nigrostriatal dopaminergic neurons through suppressing microglial activation and reducing the expression of NADPH oxidase and iNOS in the model of Parkinson's disease [49]. As mentioned above, tanshinones are likely to protect neurons via suppressing microglial activation and reducing neuroinflammation and oxidative stress.
2.6. Oxymatrine
Oxymatrine is a major active ingredient isolated from Sophora flavescens Ait (kushen), which has been used in China for thousands of years. It has been reported that oxymatrine could exhibit anticancer, antiapoptotic, and neuroprotective effects [50–52].
Oxymatrine could not only reduce the secretion of heat shock protein 60 (HSP60) in BV2 cells stimulated by LPS but also decrease the expression of heat shock factor 1, which is the transcription factor of HSP60. In addition, oxymatrine could alleviate the expression of MyD88, caspase-3, NF-κB, IL-6, iNOS, TNF-α, and IL-1β in LPS-stimulated BV2 cells. From the results mentioned above, oxymatrine plays a key role in protecting neurons by blocking microglial activation and HSP60/TLR-4/MyD88/NF-κB pathways. Therefore, oxymatrine, herbal compound, represents as a potent therapeutic agent against microglial activation for ameliorating neurodegenerative disorders [53].
2.7. Curcumin
Curcumin is a primary ingredient of turmeric, and studies have shown its anti-inflammatory and antioxidative effects, and it observably alleviates CFA-induced pain hypersensitivity [54].
Studies revealed that curcumin could decrease amyloid beta 42- (Aβ42-) induced expression of IL-1β, IL-6, and TNF-α in microglia, depending on its concentration. Moreover, curcumin showed an effect of inhibiting the levels of phosphor-ERK1/2 and p38 in Aβ42-activated microglia. These results demonstrated that curcumin alleviated proinflammatory mediators released by microglia via inhibiting ERK1/2 and p38 signaling pathways [55]. Curcumin could dramatically ameliorate the phagocytic abilities of prostaglandin E2- (PGE2-) stimulated N9 cells. Further, curcumin could reverse the decreased effect of PGE2 on Aβ42-induced microglial phagocytosis via inhibiting PGE2 receptor subtype 2 and protein kinase A signaling pathways [56].
Curcumin inhibited inflammatory response and microglial activation by decreasing the upregulated fractalkine/CX3C chemokine receptor 1, thus protecting neuronal injury in the hippocampal dentate gyrus of fructose-fed mice [57]. Furthermore, the other result demonstrated that using curcumin nanoparticles may be a feasible way of enhancing neurological function in early brain injury of rat following SAH [58]. In conclusion, curcumin is a promising herbal compound to protect neuronal damage in degenerative disorders via suppressing the inflammatory response in microglia.
2.8. Other Herbal Compounds
Isobavachalcone is the major constituent extracted from Fructus psoraleae, which presents versatile effects including antitumor [59, 60], antibacterial [61], and bone strengthening [62] effects. A study showed that isobavachalcone could recede the LPS-induced oxidative stress and inflammatory cytokine levels and that it possessed an effect of neuroprotection by inhibiting microglia-mediated inflammation [63]. Scutellarin, a member of flavone glucuronide, is considered the major active component of Erigeron breviscapus [64, 65]. Scutellarin was able to reduce the distribution of activated microglia and the levels of TNF-α, IL-1β, and iNOS in vivo. In vitro, it had the ability to prohibit the upregulated level of ROS, NO, and iNOS in LPS-induced BV2 cells [66]. Additionally, many studies demonstrated that scutellarin could regulate the activation of microglia and protection of neurons by the anti-inflammatory effect in primary microglia and BV2 cells [67]. Sophoraflavanone G decreased the cytotoxicity of conditioned medium prepared by activated BV2 cells induced by LPS to PC12 cells and increased cell viability. As mentioned above, sophoraflavanone G was able to suppress neuroinflammation via MAPKs, PI3K/protein kinase B, Janus kinase/signal transducers, and activators of transcription and nuclear factor erythroid 2-related factor 2/heme oxygenase-1 signaling pathways and might react as a potential constituent for various neuroinflammatory conditions [68]. A recent report revealed that four sesquiterpenoids isolated from Tussilago farfara also had neuroprotective effects by reducing the levels of NO, PGE2, TNF-α, and ROS in the LPS-stimulated BV2 cell and PC12 cell coculture system through the blockage of the NF-κB pathway [69]. The other study indicated that treatment with baicalein, a flavonoid from Scutellaria baicalensis Georgi, exerted neuroprotective effects on dopaminergic neurons by decreasing TNF-α, NO, and superoxide productions in the neuron-glia coculture system with LPS stimulation and blocking morphological change of microglial activation [70]. In addition, Wogonin, another flavonoid from the root of Scutellaria baicalensis Georgi, also had a potent neuroprotection by suppressing microglial activation through the blockage NF-κB pathway in vivo and in vitro experiment [71]. These results suggest that a number of natural compounds have the potential to protect neurons via inhibiting microglial activation. Further, it is possible for these natural compounds to be used as therapeutics for neurodegenerative disorders with neuroinflammation.
3. Conclusions
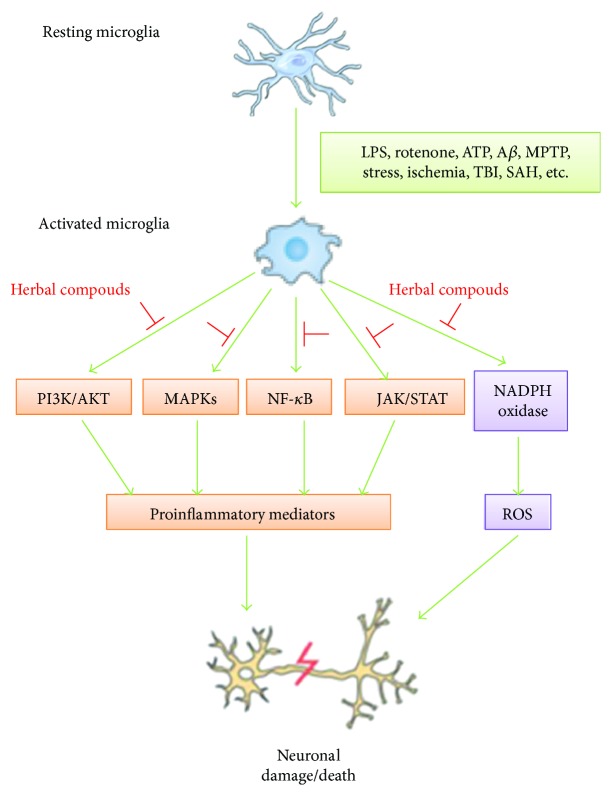
An increasing amount of findings has demonstrated that microglial activation and neuroinflammation play a crucial role in the pathogenesis of neurodegenerative disorders. Recent researches revealed that there are many compounds isolated from natural plants that can delay the neuronal damaged and degenerative progression by inhibiting microglial activation, so they have attracted considerable attention as pharmacological intervention against neurodegenerative discords with neuroinflammatory condition. As illustrated in the summary diagram (Figure 1), these literatures provide the evidences that herbal compounds can protect neuronal damage characterized by neuroinflammatory and oxidative stress condition and they accomplish their role by suppressing microglial activation and proinflammatory and neurotoxic mediator expression via blocking the related signaling pathway in activated microglia. Moreover, the natural products and compounds are inexpensive, easily accessible, and safe. Therefore, they can be widely observed in laboratory researches. If these activities of herbal compounds that are found in laboratory research are beneficial to delaying the development of neurodegenerative disorders, then large and well-designed studies in clinic are required to confirm whether or not their activity is also possible in humans. Further study of the pharmacological mechanisms of natural herbal compounds on the inhibition of microglial activation and neuroinflammation could not only benefit the discovery of effective neuroprotective components but also help researchers to learn more about the pathological mechanisms of neurodegenerative disorders.
Figure 1.
Schematic diagram represents prevention of neuronal damage/death by herbal compounds via inhibiting microglial activation through the blockage of related signaling pathway. LPS: lipopolysaccharide; Aβ: amyloid beta; MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; TBI: traumatic brain injury; SAH: subarachnoid hemorrhage; PI3k/AKT: phosphoinositide 3-kinase/protein kinase B; MAPKs: mitogen-activated protein kinases; NF-κB: nuclear factor-kappa B; JAK/STAT: Janus kinase/signal transducers and activators of transcription; NADPH oxidase: nicotinamide adenine dinucleotide phosphate oxidase; ROS: reactive oxygen species.
Acknowledgments
This research was financially supported by grants from General Program of National Natural Science Foundation of China (Grant 81274119 and 81773927 to Ying Xu and Grant 31571041 to Yongjun Chen) and Shanghai Municipal Health and Family Planning Commission (Grant 2016LP012 to Jianmei Yang).
Contributor Information
Yongjun Chen, Email: chyj417@126.com.
Ying Xu, Email: ying6122003@aliyun.com.
Conflicts of Interest
The authors declare no competing financial interests.
Authors' Contributions
Yan Fu, Jianmei Yang, and Xingyu Wang contributed equally to this work.
References
- 1.Grimmig B., Morganti J., Nash K., Bickford P. Immunomodulators as therapeutic agents in mitigating the progression of Parkinson’s disease. Brain Sciences. 2016;6(4) doi: 10.3390/brainsci6040041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hesse R., Wahler A., Gummert P., et al. Decreased IL-8 levels in CSF and serum of AD patients and negative correlation of MMSE and IL-1β. BMC Neurology. 2016;16(1):p. 185. doi: 10.1186/s12883-016-0707-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rissanen E., Tuisku J., Rokka J., et al. In vivo detection of diffuse inflammation in secondary progressive multiple sclerosis using PET imaging and the radioligand 11C-PK11195. Journal of Nuclear Medicine. 2014;55(6):939–944. doi: 10.2967/jnumed.113.131698. [DOI] [PubMed] [Google Scholar]
- 4.Kumar A., Chen S. H., Kadiiska M. B., et al. Inducible nitric oxide synthase is key to peroxynitrite-mediated, LPS-induced protein radical formation in murine microglial BV2 cells. Free Radical Biology & Medicine. 2014;73:51–59. doi: 10.1016/j.freeradbiomed.2014.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Reemst K., Noctor S. C., Lucassen P. J., Hol E. M. The indispensable roles of microglia and astrocytes during brain development. Frontiers in Human Neuroscience. 2016;10 doi: 10.3389/fnhum.2016.00566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Du L., Zhang Y., Chen Y., Zhu J., Yang Y., Zhang H. L. Role of microglia in neurological disorders and their potentials as a therapeutic target. Molecular Neurobiology. 2017;54(10):7567–7584. doi: 10.1007/s12035-016-0245-0. [DOI] [PubMed] [Google Scholar]
- 7.Chhor V., Moretti R., Le Charpentier T., et al. Role of microglia in a mouse model of paediatric traumatic brain injury. Brain, Behavior, and Immunity. 2017;63:197–209. doi: 10.1016/j.bbi.2016.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pascual O., Ben Achour S., Rostaing P., Triller A., Bessis A. Microglia activation triggers astrocyte-mediated modulation of excitatory neurotransmission. Proceedings of the National Academy of Sciences of the United States of America. 2012;109(4):E197–E205. doi: 10.1073/pnas.1111098109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Surace M. J., Block M. L. Targeting microglia-mediated neurotoxicity: the potential of NOX2 inhibitors. Cellular and Molecular Life Sciences. 2012;69(14):2409–2427. doi: 10.1007/s00018-012-1015-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Donzis E. J., Tronson N. C. Modulation of learning and memory by cytokines: signaling mechanisms and long term consequences. Neurobiology of Learning and Memory. 2014;115:68–77. doi: 10.1016/j.nlm.2014.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Choi D. K., Koppula S., Suk K. Inhibitors of microglial neurotoxicity: focus on natural products. Molecules. 2011;16(12):1021–1043. doi: 10.3390/molecules16021021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liu W., Ma H., DaSilva N. A., et al. Development of a neuroprotective potential algorithm for medicinal plants. Neurochemistry International. 2016;100:164–177. doi: 10.1016/j.neuint.2016.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Malhotra A., Bath S., Elbarbry F. An organ system approach to explore the antioxidative, anti-inflammatory, and cytoprotective actions of resveratrol. Oxidative Medicine and Cellular Longevity. 2015;2015:15. doi: 10.1155/2015/803971.803971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Singh N., Agrawal M., Dore S. Neuroprotective properties and mechanisms of resveratrol in in vitro and in vivo experimental cerebral stroke models. ACS Chemical Neuroscience. 2013;4(8):1151–1162. doi: 10.1021/cn400094w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ido Y., Duranton A., Lan F., Weikel K. A., Breton L., Ruderman N. B. Resveratrol prevents oxidative stress-induced senescence and proliferative dysfunction by activating the AMPK-FOXO3 cascade in cultured primary human keratinocytes. PLoS One. 2015;10(2, article e0115341) doi: 10.1371/journal.pone.0115341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sekiguchi K., Takehana S., Shibuya E., et al. Resveratrol attenuates inflammation-induced hyperexcitability of trigeminal spinal nucleus caudalis neurons associated with hyperalgesia in rats. Molecular Pain. 2016;12, article 174480691664308 doi: 10.1177/1744806916643082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chin Y.-T., Yang S.-H., Chang T.-C., et al. Mechanisms of dihydrotestosterone action on resveratrol-induced anti-proliferation in breast cancer cells with different ERα status. Oncotarget. 2015;6(34):35866–35879. doi: 10.18632/oncotarget.5482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chang C. Y., Choi D.-K., Lee D. K., Hong Y. J., Park E. J. Resveratrol confers protection against rotenone-induced neurotoxicity by modulating myeloperoxidase levels in glial cells. PLoS One. 2013;8(4, article e60654) doi: 10.1371/journal.pone.0060654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yang Y. J., Hu L., Xia Y. P., et al. Resveratrol suppresses glial activation and alleviates trigeminal neuralgia via activation of AMPK. Journal of Neuroinflammation. 2016;13(1):p. 84. doi: 10.1186/s12974-016-0550-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sui D. M., Xie Q., Yi W. J., et al. Resveratrol protects against sepsis-associated encephalopathy and inhibits the NLRP3/IL-1βaxis in microglia. Mediators of Inflammation. 2016;2016:10. doi: 10.1155/2016/1045657.1045657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang F., Shi J.-S., Zhou H., Wilson B., Hong J.-S., Gao H.-M. Resveratrol protects dopamine neurons against lipopolysaccharide-induced neurotoxicity through its anti-inflammatory actions. Molecular Pharmacology. 2010;78(3):466–477. doi: 10.1124/mol.110.064535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang X. S., Li W., Wu Q., et al. Resveratrol attenuates acute inflammatory injury in experimental subarachnoid hemorrhage in rats via inhibition of TLR4 pathway. International Journal of Molecular Sciences. 2016;17(12) doi: 10.3390/ijms17081331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kumar H., Kim I.-S., More S. V., Kim B.-W., Bahk Y.-Y., Choi D.-K. Gastrodin protects apoptotic dopaminergic neurons in a toxin-induced Parkinson’s disease model. Evidence-based Complementary and Alternative Medicine: Ecam. 2013;2013, article 514095:13. doi: 10.1155/2013/514095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sun T., Wang J., Li X., et al. Gastrodin relieved complete Freund’s adjuvant-induced spontaneous pain by inhibiting inflammatory response. International Immunopharmacology. 2016;41:66–73. doi: 10.1016/j.intimp.2016.10.020. [DOI] [PubMed] [Google Scholar]
- 25.Dai J.-N., Zong Y., Zhong L.-M., et al. Gastrodin inhibits expression of inducible NO synthase, cyclooxygenase-2 and proinflammatory cytokines in cultured LPS-stimulated microglia via MAPK pathways. PLoS One. 2011;6(7, article e21891) doi: 10.1371/journal.pone.0021891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li C., Chen X., Zhang N., Song Y., Mu Y. Gastrodin inhibits neuroinflammation in rotenone-induced Parkinson’s disease model rats. Neural Regeneration Research. 2012;7(5):325–331. doi: 10.3969/j.issn.1673-5374.2012.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang J. W., Liu Y. M., Zhao X. F., Zhang H. Gastrodin protects retinal ganglion cells through inhibiting microglial-mediated neuroinflammation in an acute ocular hypertension model. International Journal of Ophthalmology. 2017;10(10):1483–1489. doi: 10.18240/ijo.2017.10.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trinh N.-T.-T., Dumas E., Thanh M. L., et al. Effect of a Vietnamese Cinnamomum cassia essential oil and its major component trans-cinnamaldehyde on the cell viability, membrane integrity, membrane fluidity, and proton motive force of Listeria innocua. Canadian Journal of Microbiology. 2015;61(4):263–271. doi: 10.1139/cjm-2014-0481. [DOI] [PubMed] [Google Scholar]
- 29.Rajamani K., Lin Y.-C., Wen T.-C., et al. The antisenescence effect of trans-cinnamaldehyde on adipose-derived stem cells. Cell Transplantation. 2015;24(3):493–507. doi: 10.3727/096368915X686959. [DOI] [PubMed] [Google Scholar]
- 30.Andersen H. H., Gazerani P., Arendt-Nielsen L. High-concentration L-menthol exhibits counter-irritancy to neurogenic inflammation, thermal and mechanical hyperalgesia caused by trans-cinnamaldehyde. The Journal of Pain. 2016;17(8):919–929. doi: 10.1016/j.jpain.2016.05.004. [DOI] [PubMed] [Google Scholar]
- 31.Qi X., Zhou R., Liu Y., et al. Trans-cinnamaldehyde protected PC12 cells against oxygen and glucose deprivation/reperfusion (OGD/R)-induced injury via anti-apoptosis and anti-oxidative stress. Molecular and Cellular Biochemistry. 2016;421(1-2):67–74. doi: 10.1007/s11010-016-2785-z. [DOI] [PubMed] [Google Scholar]
- 32.Chen Y. F., Wang Y. W., Huang W. S., et al. Trans-cinnamaldehyde, an essential oil in cinnamon powder, ameliorates cerebral ischemia-induced brain injury via inhibition of neuroinflammation through attenuation of iNOS, COX-2 expression and NFκ-B signaling pathway. Neuromolecular Medicine. 2016;18(3):322–333. doi: 10.1007/s12017-016-8395-9. [DOI] [PubMed] [Google Scholar]
- 33.Zhang L., Zhang Z., Fu Y., et al. Trans-cinnamaldehyde improves memory impairment by blocking microglial activation through the destabilization of iNOS mRNA in mice challenged with lipopolysaccharide. Neuropharmacology. 2016;110(Part A):503–518. doi: 10.1016/j.neuropharm.2016.08.013. [DOI] [PubMed] [Google Scholar]
- 34.Pyo J.-H., Jeong Y.-K., Yeo S., et al. Neuroprotective effect of trans-cinnamaldehyde on the 6-hydroxydopamine-induced dopaminergic injury. Biological and Pharmaceutical Bulletin. 2013;36(12):1928–1935. doi: 10.1248/bpb.b13-00537. [DOI] [PubMed] [Google Scholar]
- 35.Zhao Y., Guo Y., Gu X. Salvianolic acid B, a potential chemopreventive agent, for head and neck squamous cell cancer. Journal of Oncology. 2011;2011:8. doi: 10.1155/2011/534548.534548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wang Z. S., Luo P., Dai S. H., Liu Z. B., Zheng X. R., Chen T. Salvianolic acid B induces apoptosis in human glioma U87 cells through p38-mediated ROS generation. Cellular and Molecular Neurobiology. 2013;33(7):921–928. doi: 10.1007/s10571-013-9958-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen S. C., Lin Y. L., Huang B., Wang D. L., Cheng J. J. Salvianolic acid B suppresses IFN-γ-induced JAK/STAT1 activation in endothelial cells. Thrombosis Research. 2011;128(6):560–564. doi: 10.1016/j.thromres.2011.08.032. [DOI] [PubMed] [Google Scholar]
- 38.Chen T., Liu W., Chao X., et al. Salvianolic acid B attenuates brain damage and inflammation after traumatic brain injury in mice. Brain Research Bulletin. 2011;84(2):163–168. doi: 10.1016/j.brainresbull.2010.11.015. [DOI] [PubMed] [Google Scholar]
- 39.Wang S. X., Hu L. M., Gao X. M., Guo H., Fan G. W. Anti-inflammatory activity of salvianolic acid B in microglia contributes to Its neuroprotective effect. Neurochemical Research. 2010;35(7):1029–1037. doi: 10.1007/s11064-010-0151-1. [DOI] [PubMed] [Google Scholar]
- 40.Zhuang P., Wan Y., Geng S., et al. Salvianolic acids for injection (SAFI) suppresses inflammatory responses in activated microglia to attenuate brain damage in focal cerebral ischemia. Journal of Ethnopharmacology. 2017;198:194–204. doi: 10.1016/j.jep.2016.11.052. [DOI] [PubMed] [Google Scholar]
- 41.Lee W. D., Liang Y. J., Chen B. H. Effects of tanshinone nanoemulsion and extract on inhibition of lung cancer cells A549. Nanotechnology. 2016;27(49, article 495101) doi: 10.1088/0957-4484/27/49/495101. [DOI] [PubMed] [Google Scholar]
- 42.Lee K.-W., Jung S. Y., Choi S.-M., Yang E. J. Effects of ginsenoside Re on LPS-induced inflammatory mediators in BV2 microglial cells. BMC Complementary and Alternative Medicine. 2012;12(1) doi: 10.1186/1472-6882-12-196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee J. C., Park J. H., Park O. K., et al. Neuroprotective effects of tanshinone I from Danshen extract in a mouse model of hypoxia-ischemia. Anatomy & Cell Biology. 2013;46(3):183–190. doi: 10.5115/acb.2013.46.3.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang D., Lu C., Sun F., et al. A tanshinone I derivative enhances the activities of antibiotics against Staphylococcus aureus in vitro and in vivo. Research in Microbiology. 2017;168(1):46–54. doi: 10.1016/j.resmic.2016.08.002. [DOI] [PubMed] [Google Scholar]
- 45.de Oliveira M. R., Schuck P. F., Bosco S. M. D. Tanshinone I induces mitochondrial protection through an Nrf2-dependent mechanism in paraquat-treated human neuroblastoma SH-SY5Y cells. Molecular Neurobiology. 2017;54(6):4597–4608. doi: 10.1007/s12035-016-0009-x. [DOI] [PubMed] [Google Scholar]
- 46.Wang S., Jing H., Yang H., et al. Tanshinone I selectively suppresses pro-inflammatory genes expression in activated microglia and prevents nigrostriatal dopaminergic neurodegeneration in a mouse model of Parkinson’s disease. Journal of Ethnopharmacology. 2015;164:247–255. doi: 10.1016/j.jep.2015.01.042. [DOI] [PubMed] [Google Scholar]
- 47.Zeng J., Xue Y., Shu P., et al. Diterpenoids with immunosuppressive activities from Cinnamomum cassia. Journal of Natural Products. 2014;77(8):1948–1954. doi: 10.1021/np500465g. [DOI] [PubMed] [Google Scholar]
- 48.Jia L. Q., Yang G. L., Ren L., et al. Tanshinone IIA reduces apoptosis induced by hydrogen peroxide in the human endothelium-derived EA.hy926 cells. Journal of Ethnopharmacology. 2012;143(1):100–108. doi: 10.1016/j.jep.2012.06.007. [DOI] [PubMed] [Google Scholar]
- 49.Ren B., Zhang Y. X., Zhou H. X., et al. Tanshinone IIA prevents the loss of nigrostriatal dopaminergic neurons by inhibiting NADPH oxidase and iNOS in the MPTP model of Parkinson’s disease. Journal of the Neurological Sciences. 2015;348(1-2):142–152. doi: 10.1016/j.jns.2014.11.026. [DOI] [PubMed] [Google Scholar]
- 50.Wu C., Huang W., Guo Y., et al. Oxymatrine inhibits the proliferation of prostate cancer cells in vitro and in vivo. Molecular Medicine Reports. 2015;11(6):4129–4134. doi: 10.3892/mmr.2015.3338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Liu F., Wang B., Wang J., et al. Oxymatrine inhibits proliferation and migration while inducing apoptosis in human glioblastoma cells. BioMed Research International. 2016;2016:7. doi: 10.1155/2016/1784161.1784161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Huang M., Hu Y. Y., Dong X. Q., Xu Q. P., Yu W. H., Zhang Z. Y. The protective role of oxymatrine on neuronal cell apoptosis in the hemorrhagic rat brain. Journal of Ethnopharmacology. 2012;143(1):228–235. doi: 10.1016/j.jep.2012.06.028. [DOI] [PubMed] [Google Scholar]
- 53.Ding F., Li Y., Hou X., Zhang R., Hu S., Wang Y. Oxymatrine inhibits microglia activation via HSP60-TLR4 signaling. Biomedical Reports. 2016;5(5):623–628. doi: 10.3892/br.2016.776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chen J. J., Dai L., Zhao L. X., Zhu X., Cao S., Gao Y. J. Intrathecal curcumin attenuates pain hypersensitivity and decreases spinal neuroinflammation in rat model of monoarthritis. Scientific Reports. 2015;5(1, article 10278) doi: 10.1038/srep10278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Shi X., Zheng Z., Li J., et al. Curcumin inhibits Aβ-induced microglial inflammatory responses in vitro: Involvement of ERK1/2 and p38 signaling pathways. Neuroscience Letters. 2015;594:105–110. doi: 10.1016/j.neulet.2015.03.045. [DOI] [PubMed] [Google Scholar]
- 56.He G. L., Luo Z., Yang J., Shen T. T., Chen Y., Yang X. S. Curcumin ameliorates the reduction effect of PGE2 on fibrillar β-amyloid peptide (1-42)-induced microglial phagocytosis through the inhibition of EP2-PKA signaling in N9 microglial cells. PLoS One. 2016;11(1, article e0147721) doi: 10.1371/journal.pone.0147721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Xu M. X., Yu R., Shao L. F., et al. Up-regulated fractalkine (FKN) and its receptor CX3CR1 are involved in fructose-induced neuroinflammation: suppression by curcumin. Brain, Behavior, and Immunity. 2016;58:69–81. doi: 10.1016/j.bbi.2016.01.001. [DOI] [PubMed] [Google Scholar]
- 58.Zhang Z. Y., Jiang M., Fang J., et al. Enhanced therapeutic potential of nano-curcumin against subarachnoid hemorrhage-induced blood-brain barrier disruption through inhibition of inflammatory response and oxidative stress. Molecular Neurobiology. 2017;54(1):1–14. doi: 10.1007/s12035-015-9635-y. [DOI] [PubMed] [Google Scholar]
- 59.Wang H. M., Zhang L., Liu J., et al. Synthesis and anti-cancer activity evaluation of novel prenylated and geranylated chalcone natural products and their analogs. European Journal of Medicinal Chemistry. 2015;92:439–448. doi: 10.1016/j.ejmech.2015.01.007. [DOI] [PubMed] [Google Scholar]
- 60.Ismail, Kuthati B., Thalari G., et al. Synthesis of novel spiro [pyrazolo [4,3- d ] pyrimidinones and spiro [benzo] [4, 5] thieno [2,3- d ] pyrimidine-2,3′-indoline]-2′,4(3H)-diones and their evaluation for anticancer activity. Bioorganic & Medicinal Chemistry Letters. 2017;27(6):1446–1450. doi: 10.1016/j.bmcl.2017.01.088. [DOI] [PubMed] [Google Scholar]
- 61.Cui P., Li X., Zhu M., Wang B., Liu J., Chen H. Design, synthesis and antimicrobial activities of thiouracil derivatives containing triazolo-thiadiazole as SecA inhibitors. European Journal of Medicinal Chemistry. 2017;127:159–165. doi: 10.1016/j.ejmech.2016.12.053. [DOI] [PubMed] [Google Scholar]
- 62.Li Y. G., Hou J., Li S. Y., et al. Fructus Psoraleae contains natural compounds with potent inhibitory effects towards human carboxylesterase 2. Fitoterapia. 2015;101:99–106. doi: 10.1016/j.fitote.2015.01.004. [DOI] [PubMed] [Google Scholar]
- 63.Jing H., Wang S., Wang M., Fu W., Zhang C., Xu D. Isobavachalcone attenuates MPTP-induced Parkinson’s disease in mice by inhibition of microglial activation through NF-κB pathway. PLoS One. 2017;12(1, article e0169560) doi: 10.1371/journal.pone.0169560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tang H., Tang Y., Li N., et al. Neuroprotective effects of scutellarin and scutellarein on repeatedly cerebral ischemia-reperfusion in rats. Pharmacology, Biochemistry, and Behavior. 2014;118:51–59. doi: 10.1016/j.pbb.2014.01.003. [DOI] [PubMed] [Google Scholar]
- 65.Qian L., Shen M., Tang H., et al. Synthesis and protective effect of scutellarein on focal cerebral ischemia/reperfusion in rats. Molecules. 2012;17(12):10667–10674. doi: 10.3390/molecules170910667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yuan Y., Rangarajan P., Kan E., Wu Y., Wu C., Ling E.-A. Scutellarin regulates the Notch pathway and affects the migration and morphological transformation of activated microglia in experimentally induced cerebral ischemia in rats and in activated BV-2 microglia. Journal of Neuroinflammation. 2015;12(1):p. 11. doi: 10.1186/s12974-014-0226-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Wang S., Wang H., Guo H., Kang L., Gao X., Hu L. Neuroprotection of scutellarin is mediated by inhibition of microglial inflammatory activation. Neuroscience. 2011;185:150–160. doi: 10.1016/j.neuroscience.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 68.Guo C., Yang L., Wan C. X., et al. Anti-neuroinflammatory effect of Sophoraflavanone G from Sophora alopecuroides in LPS-activated BV2 microglia by MAPK, JAK/STAT and Nrf2/HO-1 signaling pathways. Phytomedicine. 2016;23(13):1629–1637. doi: 10.1016/j.phymed.2016.10.007. [DOI] [PubMed] [Google Scholar]
- 69.Lim H. J., Dong G. Z., Lee H. J., Ryu J. H. In vitro neuroprotective activity of sesquiterpenoids from the flower buds of Tussilago farfara. Journal of Enzyme Inhibition and Medicinal Chemistry. 2014;30(5):852–856. doi: 10.3109/14756366.2014.965701. [DOI] [PubMed] [Google Scholar]
- 70.Li F. Q., Wang T., Pei Z., Liu B., Hong J. S. Inhibition of microglial activation by the herbal flavonoid baicalein attenuates inflammation-mediated degeneration of dopaminergic neurons. Journal of Neural Transmission. 2005;112(3):331–347. doi: 10.1007/s00702-004-0213-0. [DOI] [PubMed] [Google Scholar]
- 71.Lee H., Kim Y. O., Kim H., et al. Flavonoid wogonin from medicinal herb is neuroprotective by inhibiting inflammatory activation of microglia. FASEB Journal. 2003;17(13):1943–1944. doi: 10.1096/fj.03-0057fje. [DOI] [PubMed] [Google Scholar]