Abstract
The study objective was to investigate whether sex influences 3′-deoxy-3′-18F-fluorothymidine (18F-FLT) uptake and tissue distribution in mouse models of cancer. Methods: 18F-FLT biodistribution was measured in 3 strains of male and female mice (129S6/SvEv, athymic nude, and BALB/c). 18F-FDG biodistribution was measured for comparison. 18F-FLT uptake was also measured in female 129S6/SvEv mice bearing estrogen-dependent SSM3 mouse mammary tumors, male athymic nude mice bearing androgen-dependent CWR22 prostate cancer xenografts, and male and female athymic nude mice bearing estrogen-independent MDA-MB-231 human breast cancer xenografts. Ki-67 expression was assayed by immunohistochemistry. PET/CT imaging was performed to visualize 18F-FLT biodistribution and to determine pharmacokinetics. Results: Greater 18F-FLT activity was observed in blood, liver, muscle, heart, kidney, and bone in female than male mice. Pharmacokinetic analysis demonstrated higher early renal 18F-FLT activity and greater accumulation of 18F-FLT in the urinary bladder in male than female mice. The differential pattern of 18F-FLT biodistribution between the sexes seen with 18F-FLT was not observed with 18F-FDG. Increased tumoral 18F-FLT uptake compared with muscle was observed in both the SSM3 mammary tumors (2.4 ± 0.17 vs. 1.6 ± 0.14 percentage injected dose [%ID]/g at 2 h after injection, P = 0.006) and the CWR22 prostate cancer xenografts (0.34 ± 0.08 vs. 0.098 ± 0.033 %ID/g at 2 h after injection, P = 0.03). However, because of higher nonspecific muscle uptake in female mice, tumor-to-muscle uptake ratios were greater for CWR22 tumors than for SSM3 tumors (4.2 ± 0.78 vs. 1.5 ± 0.049 at 2 h after injection, P = 0.008). Sex-dependent differences in 18F-FLT uptake were also observed for MDA-MB-231 xenografts (tumor-to-muscle ratio, 7.2 ± 0.9 for female vs. 16.9 ± 8.6 for male, P = 0.039). Conversely, greater tumoral Ki-67 staining was observed in female mice (71% ± 3% for female vs. 54% ± 2% for male, P = 0.009), and this finding more closely matched the relative differences in absolute 18F-FLT tumor uptake values (4.5 ± 0.99 %ID/g for female vs. 1.9 ± 0.30 %ID/g for male, P = 0.03). Conclusion: Depending on whether female or male mice are used, differences in biodistribution and nonspecific tissue uptake can adversely affect quantitative measures of 18F-FLT uptake. Thus, sex is a potential variable to consider in defining quantitative imaging metrics using 18F-FLT to assess tumor proliferation.
Keywords: 3′-deoxy-3′-18F-fluorothymidine, cancer, sex differences, positron emission tomography, mice
A rising concern from the National Institutes of Health is the importance of including sex as a biologic variable in animal studies and human clinical trials (1). Women were not required to be included as subjects in clinical research until the Revitalization Act of 1993, and consideration of sex as an experimental variable did not extend to federally funded preclinical work until 2014. This policy is part of a broader goal to increase reproducibility and transparency between research studies and to ensure scientific rigor.
Oftentimes, the sex of cell lines is not considered in experimental design because the canonical thought was that, on the molecular level, basic cellular processes transcend sex differences. Although basic molecular mechanisms may be similar, male and female cells may respond differently to various stimuli. For example, it was shown that male neurons are more sensitive to oxidative stress than female neurons (2). Sex differences also extend to the organismal level, with sexual dimorphism identified in approximately 57% of phenotypes in mice (3). Thus, sex is an important biologic variable to consider in the design, analysis, and reporting of preclinical research. This concept is especially pertinent for understanding the pathophysiology and treatment of diseases that affect both sexes, such as many forms of cancer.
One of the hallmarks of cancer is abnormal sustained proliferation (4). As more therapies emerge and the field of precision medicine matures, imaging tools that can detect biologic changes before a change in anatomic tumor size occurs are becoming increasingly important. The most-studied radiopharmaceutical for in vivo imaging of cell proliferation is 3′-deoxy-3′-18F-fluorothymidine (18F-FLT) (5,6). It enters cells via membrane nucleoside transporters and undergoes phosphorylation by the cytosolic thymidine kinase-1 enzyme as part of the thymidine salvage pathway of DNA synthesis during the S-phase of the cell cycle. Unlike endogenous thymidine, phosphorylated 18F-FLT does not incorporate into DNA, and because of a relatively slow rate of dephosphorylation, it accumulates intracellularly (7).
PET imaging using 18F-FLT is being studied as a noninvasive method to measure the proliferative capacity of cancer and may provide an early indication of treatment response (5,6). Although not yet approved by the U.S. Food and Drug Administration, 18F-FLT has been studied through clinical trials in over 1,000 individuals and in patients with multiple cancer types (8). Biologic variables that can affect 18F-FLT uptake include plasma thymidine levels; the expression level and functional activity of thymidine kinase-1, thymidylate synthase, and nucleoside transporters; and the overall balance of de novo versus salvage pathways of thymidine use (9). This study aimed to determine whether sex influences 18F-FLT uptake and tissue biodistribution in preclinical oncology models.
MATERIALS AND METHODS
Mice
Athymic NCr-nu/nu mice were purchased from the Animal Production Program of the National Cancer Institute–Frederick National Laboratory for Cancer Research. BALB/c mice were also purchased from the National Cancer Institute. Male and female 129S6/SvEv mice were purchased from Taconic Farms, Inc.
Cell Lines and Tumor Implantation
Experiments were performed under an approved biosafety protocol. The human estrogen receptor–negative breast cancer cell line MDA-MB-231 was cultured at 37°C and 10% CO2 in Dulbecco modified Eagle medium (Corning) with high glucose containing 10% fetal bovine serum (Corning) with penicillin and streptomycin (Gibco). SSM1 and SSM3 cell lines were isolated from primary spontaneous mammary adenocarcinomas in female transgenic STAT1−/− mice and were maintained in culture as described previously (10). The prostate cancer xenograft model, CWR22, was created from primary prostate carcinoma from a man with metastatic prostate cancer and was maintained in vivo via serial transplantation (11,12). All cell lines were negative for murine pathogens and for Mycoplasma contamination, which can affect uptake of thymidine analogs (13). The MDA-MB-231 cell line was authenticated using short tandem repeat analysis.
Experiments were performed according to the guidelines of the American Association for Laboratory Animal Science under a protocol approved by the Animal Studies Committees in specific pathogen-free facilities accredited by the Association for Assessment and Accreditation of Laboratory Animal Care at Washington University School of Medicine and the University of Wisconsin–Madison. Mice aged 6–8 wk received a subcutaneous injection into the right thoracic mammary fat pad of 1 × 106 SSM1 or SSM3 cells in 100 μL of phosphate-buffered saline or 2 × 106 MDA-MB-231 cells in 50% Matrigel (BD Biosciences) by volume. Tumor growth was monitored by palpation and measured in 2 perpendicular dimensions with calipers. Tumor volumes were calculated using the formula a × b2/2, where a is the long diameter and b is the short diameter.
Radiopharmaceuticals, PET/CT Imaging, and Tissue Biodistribution Assay
18F-FLT and 18F-FDG were synthesized by the Cyclotron Facility at Washington University (14,15). 18F-FLT–specific activity at the end of synthesis ranged from 59 to 225 GBq/μmol (1,606–6,068 mCi/μmol). 18F-FLT was also provided by the University of Wisconsin–Madison Radiopharmaceutical Production Facility. Specific activity at the end of synthesis ranged from 333 to 629 GBq/μmol (9,000–17,000 mCi/μmol). Radiochemical purity was 100% for all preparations.
For PET/CT imaging, nonfasted mice were injected via the tail vein with approximately 0.93 MBq (25 μCi) of 18F-FLT. The mice were not anesthetized during the radiotracer uptake period. Mice anesthetized with 1.5%–2.0% isoflurane were scanned supine in a small-animal PET/CT scanner (Inveon; Siemens Preclinical Solutions) 1 h after injection. CT images were acquired for approximately 12 min, followed by PET images (40 million counts—typically less than 10 min). One male and one female mouse were positioned side by side and imaged simultaneously for each scan. Images were analyzed using Inveon Research Workplace, version 3.0 (Siemens Medical Solutions USA, Inc.). The reconstruction method was 3-dimensional ordered-subset expectation maximization/maximum a posteriori with attenuation correction. Dynamic emission imaging began immediately after injection and continued for 60 min with the following time bins: 1 × 3, 6 × 2, 9 × 5, 6 × 10, 4 × 30, 2 × 60, 2 × 120, and 10 × 300 s. PET and CT images were automatically coregistered and adjusted if needed via visual alignment in all 3 planes. Regions of interest were manually drawn around the tumor and within the triceps muscle to determine nontarget tissue uptake using CT images for visual anatomic localization. Additional regions of interest were drawn around the kidneys, urinary bladder, bone (femur), liver, and heart for blood activity. Data are expressed as mean percentage injected dose (%ID)/g. Tumor-to-muscle (T:M) ratio was calculated as the ratio of tumor %ID/g to muscle %ID/g. 18F-FLT clearance from the blood was calculated using a 1-phase exponential decay equation of the time–activity curve.
For biodistribution assays, tissues (blood, liver, muscle, heart, kidney, and whole bone) were harvested 1 or 2 h after tail vein injection of approximately 0.74–0.89 MBq (20–24 μCi) of 18F-FLT or 0.74 MBq (20 μCi) of 18F-FDG in nonfasted mice. Radioactivity was measured using a γ-counter (2480 Wizard2; Perkin Elmer) and decay-corrected to calculate %ID/g. To minimize differences due to variations in 18F-FLT preparations, the same batch of 18F-FLT was used for experimentation within the same strains of male and female mice.
Tissue Histology
Sections of formalin-fixed paraffin-embedded mammary tumors were deparaffinized and rehydrated followed by antigen retrieval in citrate buffer (pH 6) and stained for Ki-67 (VP-K452, 1:800; Vector Laboratories, Inc.). Positive signal was developed using SignalStain IHC Detection Reagent (horseradish peroxidase, mouse; Cell Signaling Technology) followed by diaminobenzidine chromogen. Hematoxylin and eosin staining was also performed. A whole-slide bright-field imaging system (Aperio Image Scope software; Leica Biosystems) was used to scan slides at ×40 magnification. The percentage of tumor cells staining positively for Ki-67 was visually scored by a pathologist with subspecialty training in breast pathology and 4 y of experience.
Statistical Analyses
Results are presented as mean ± SE unless indicated otherwise. Two-way ANOVA with a Šídák posttest was used to determine significance between male and female mice for biodistribution assays. The Mann–Whitney test was used to determine the statistical significance between control and experimental groups. The paired t test was used to compare across different time points within the same group of animals. All tests are 2-sided, with a P value of 0.05 or less considered significant. Analyses were performed using Prism, version 6.05 (GraphPad Software).
RESULTS
18F-FLT Tissue Biodistribution in Female Versus Male Mice
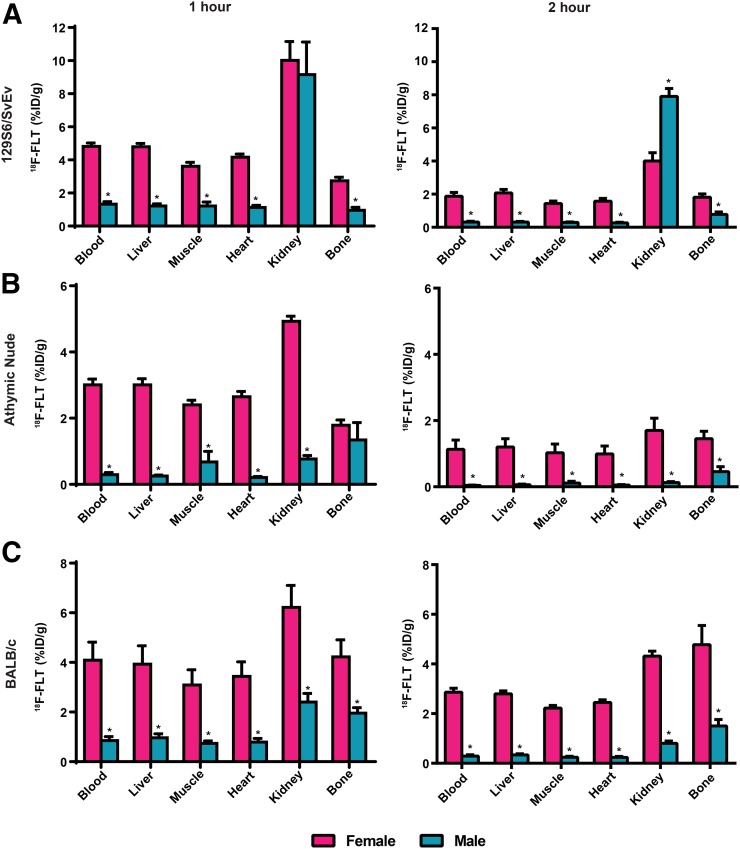
To determine whether sex is a potential biologic variable for 18F-FLT uptake in preclinical models of cancer, we performed tissue biodistribution experiments using 3 different mouse strains, 129S6/SvEv, athymic nude, and BALB/c (Fig. 1). 18F-FLT activity was higher at 1 and 2 h after injection in the blood, liver, muscle, heart, and bone in female than male 129S6/SvEv mice (Fig. 1A). At 1 h, there was similar renal accumulation of 18F-FLT in both sexes. At 2 h, 18F-FLT activity in the kidneys was less in female than male mice. For athymic nude mice, 18F-FLT uptake was greater in female than male mice for all tissues measured, with the exception of bone at 1 h, which was not significantly different (Fig. 1B). The third mouse strain tested, BALB/c, showed 18F-FLT uptake significantly higher in all female than male organs (Fig. 1C). Sex-dependent differences in 18F-FLT tissue biodistribution were accentuated when results were normalized to mouse weight (Supplemental Fig. 1; supplemental materials are available at http://jnm.snmjournals.org). Thus, we observed that 18F-FLT uptake in the blood, liver, muscle, heart, and bone was higher in female than male mice across all 3 mouse strains.
FIGURE 1.
18F-FLT biodistribution (%ID/g) in male and female 129S6/SvEv (A), athymic nude (B), and BALB/c (C) mice sacrificed 1 and 2 h after injection of 0.74–0.89 MBq (20–24 μCi) of 18F-FLT (n = 5 129S6/SvEv, 5 athymic nude, and 4 BALB/c mice per time point). *P < 0.05 (male vs. female for each tissue).
To test whether these differences in tissue biodistribution could be visualized with PET imaging, we chose female and male athymic nude mice as a representative strain to undergo PET/CT. Female mice had greater overall visual 18F-FLT signal than male mice, with the exception of the kidneys and urinary bladder, which appeared similar (Fig. 2). Therefore, PET/CT is sensitive enough to detect sex differences in 18F-FLT biodistribution and further confirmed our data obtained via γ-counting of excised tissues.
FIGURE 2.
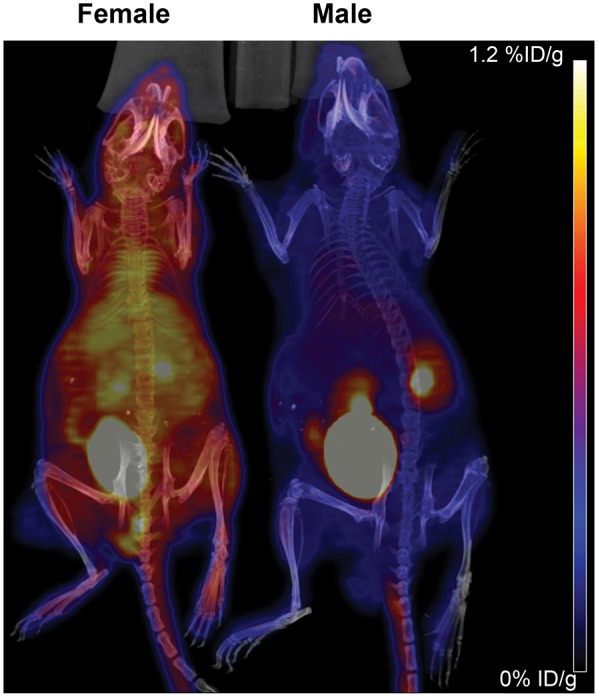
18F-FLT PET/CT maximal-intensity-projection images of representative female (n = 3) and male (n = 3) athymic nude mice scanned concurrently 1 h after injection of 0.93 MBq (25 μCi) of 18F-FLT.
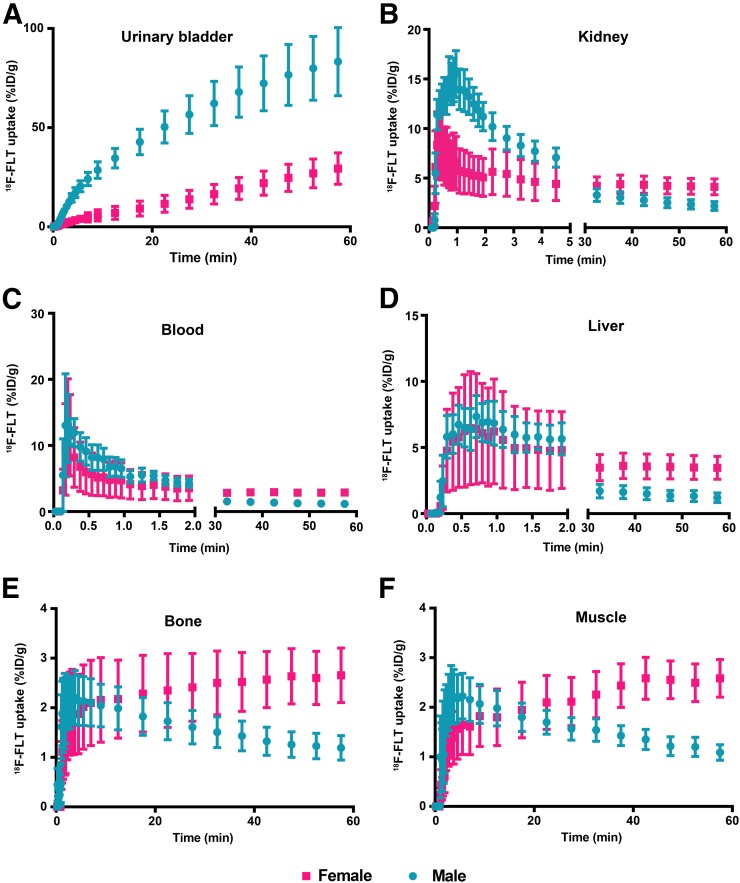
Dynamic 18F-FLT PET/CT Imaging in Female Versus Male Mice
To investigate 18F-FLT pharmacokinetics in male and female mice, we performed dynamic PET/CT imaging of athymic nude mice at multiple time points during the first hour after injection (Fig. 3). Time–activity curves demonstrated that 18F-FLT activity in the urinary bladder was higher in male than female mice at all time points (Fig. 3A). Male mice also had a greater spike in renal 18F-FLT uptake during the first 1–2 min after injection (Fig. 3B). For blood (Fig. 3C), liver (Fig. 3D), bone (Fig. 3E), and muscle (Fig. 3F), the time–activity curves appeared similar in male and female mice at early time points. The half-life of 18F-FLT in blood was 0.36 min (95% confidence interval, 0.29–0.47) for male mice and 0.20 min (95% confidence interval, 0.15–0.30) for female mice. However, the curves tended to diverge at around 30 min after injection, and by 60 min the activity in these tissues was higher in female than male mice. These data suggest that there is a difference in the physiologic processing of 18F-FLT during the first 30 min after injection.
FIGURE 3.
Time–activity curves for 18F-FLT in male and female athymic nude mice imaged using dynamic PET from 0 to 60 min after injection of approximately 0.93 MBq (25 μCi) of 18F-FLT. Regions of interest were drawn, and time–activity curves determined, for urinary bladder (A), kidney (B), blood (C), liver (D), bone (E), and muscle (F).
18F-FDG Tissue Biodistribution in Female Versus Male Mice
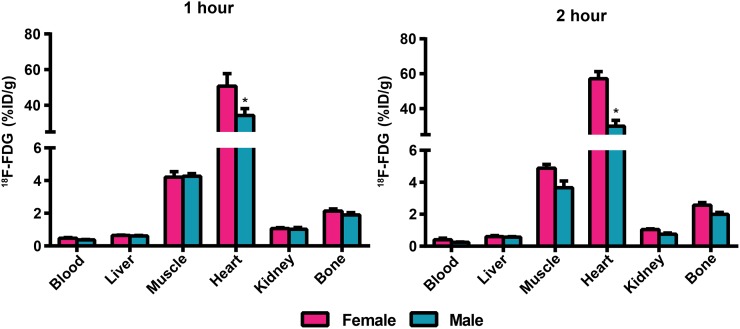
To determine whether sex variability is specific to 18F-FLT, we tested a more commonly used 18F-labeled radiopharmaceutical, 18F-FDG, for imaging glucose metabolism. A tissue biodistribution assay was performed on male and female 129S6/SvEv mice 1 and 2 h after injection using the same dose of 18F-FDG as was used previously for 18F-FLT (Fig. 4). The only significant difference in 18F-FDG tissue uptake was in the heart, with greater uptake in female than male mice at both time points. Thus, with this exception, differential sex-dependent tissue biodistribution may be specific to 18F-FLT and may not pertain to all 18F-labeled molecular imaging agents.
FIGURE 4.
18F-FDG biodistribution (%ID/g) in male and female 129S6/SvEv mice sacrificed 1 and 2 h after injection of 0.74 MBq (20 μCi) of 18F-FDG (n = 5 per group). *P < 0.05 (male vs. female for each tissue).
18F-FLT Uptake by Estrogen-Dependent Mouse Mammary Tumors Versus Androgen-Dependent Prostate Cancer Xenografts
We tested the potential impact of the sex-based physiologic differences in 18F-FLT biodistribution and kinetics on the imaging of proliferation in the hormone-dependent tumor models. We used estrogen receptor–positive SSM3 tumors as a representative estrogen-dependent mammary tumor model grown in female mice and CWR22 tumors as a representative androgen-dependent prostate tumor model in which tumoral 18F-FLT uptake can be reduced by androgen ablation in male mice (16). SSM3 and CWR22 tumors were similar in size (mean ± SD, 488 ± 494 mm3 and 272 ± 192 mm3, respectively; P = 0.21) at the time of assay. Tumor uptake of 18F-FLT was measured at 1 and 2 h after injection using tissue biodistribution assays. There was statistically significant higher 18F-FLT uptake in tumor than in muscle both for the SSM3 tumors (4.3 ± 0.27 vs. 3.2 ± 0.25 %ID/g at 1 h, P = 0.02; 2.4 ± 0.17 vs. 1.6 ± 0.14 %ID/g at 2 h, P = 0.006) and for the CWR22 xenografts (0.86 ± 0.06 vs. 0.41 ± 0.07 %ID/g at 1 h, P = 0.004; 0.34 ± 0.08 vs. 0.098 ± 0.033 %ID/g at 2 h, P = 0.03). However, because of higher nonspecific muscle uptake in female mice, T:M ratios were greater for CWR22 xenografts than for SSM3 tumors at both 1 and 2 h after injection (P = 0.03 and 0.0083, respectively) (Fig. 5). The highest T:M ratio for the CWR22 xenografts occurred at 2 h after injection and was 4.2 ± 0.78. The highest T:M ratio for the SSM3 tumors occurred at 2 h after injection and was only 1.5 ± 0.05. As a result, CWR22 xenografts display more tumor-specific uptake of 18F-FLT based on their higher T:M ratios, whereas SSM3 tumors exhibit higher absolute values of 18F-FLT uptake along with higher nonspecific muscle uptake. Another mouse mammary carcinoma cell line (SSM1) showed biodistribution results similar to those of the SSM3 tumors grown in female 129S6/SvEv mice (Supplemental Fig. 2).
FIGURE 5.
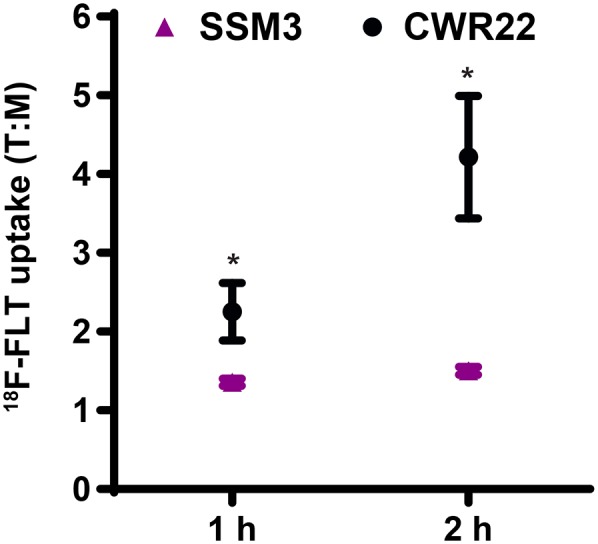
18F-FLT biodistribution (T:M %ID/g) in SSM3 mammary tumor–bearing and CWR22 prostate cancer xenograft–bearing mice sacrificed 1 h (n = 5 SSM3 and 4 CWR22) and 2 h (n = 5 SSM3 and 5 CWR22) after injection of 0.85 MBq (23 μCi) of 18F-FLT. *P < 0.05 (CWR22 vs. SSM3 tumors).
18F-FLT Uptake by Hormone-Independent Human Breast Cancer Xenografts in Female Versus Male Mice
The mouse-derived SSM3 tumors require estrogen for tumor growth and were grown in female immunocompetent mice. The human-derived CWR22 tumors depend on androgens for tumor growth and were grown in male immunocompromised mice. Thus, we chose to directly compare the 18F-FLT uptake of a hormone-independent breast cancer cell line that can be grown as xenografts in either female or male mice. MDA-MB-231 cells were grown as tumor xenografts in athymic nude female and male mice. The mice were injected with 18F-FLT and, 2 h later, sacrificed. Tissue was then harvested and counted for radioactivity. Tumor volumes were 499 ± 143 mm3 (mean ± SD) in the female mice and 352 ± 104 mm3 in the male mice at the time of the biodistribution experiment (P = 0.05). Both female and male mice had greater 18F-FLT uptake in MDA-MB-231 tumors than in muscle (female mice: 4.5 ± 0.99 %ID/g for tumor vs. 0.67 ± 0.18 %ID/g for muscle, P = 0.005; male mice: 1.9 ± 0.30 %ID/g for tumor vs. 0.16 ± 0.07 %ID/g for muscle, P = 0.0004) (Fig. 6A). However, female mice also had more nonspecific muscle uptake than male mice (0.67 ± 0.18 %ID/g vs. 0.16 ± 0.07 %ID/g, P = 0.0275). This disparity resulted in a lower T:M ratio for female than male mice (7.2 ± 0.9 vs. 16.9 ± 8.6, P = 0.039).
FIGURE 6.
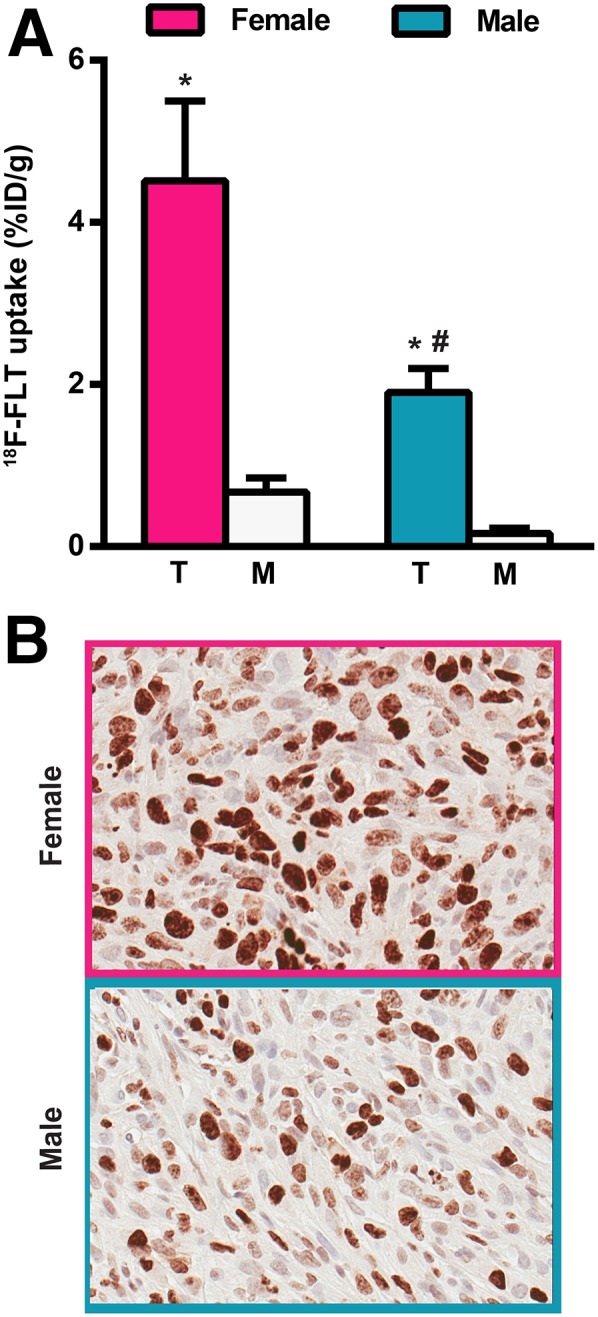
(A) 18F-FLT biodistribution (tumor [T] and muscle [M] %ID/g) in female (n = 5) and male (n = 5) mice bearing hormone-independent, estrogen receptor–negative MDA-MB-231 human breast cancer cells and sacrificed 2 h after injection of 0.67–0.78 MBq (18–21 μCi) of 18F-FLT. *P < 0.05 (tumor vs. muscle). #P < 0.05 (female tumor vs. male tumor). (B) Representative Ki-67 immunohistochemistry of MDA-MB-231 tumors (×40).
To determine whether absolute tumor uptake (%ID/g) or T:M ratio better reflects proliferation, tumor Ki-67 staining was performed on MDA-MB-231 tumors grown in female and male mice. A greater percentage of Ki-67–positive staining was observed in female than male tumors (71% ± 3% vs. 54% ± 2%; P = 0.009) (Fig. 6B).
DISCUSSION
The purpose of this study was to investigate whether sex is a potential biologic variable in preclinical studies of 18F-FLT PET imaging. We demonstrated that female mice have an overall greater 18F-FLT uptake than male mice in multiple tissues. We also observed higher 18F-FLT activity in the kidney at early time points and greater accumulation of 18F-FLT in the urinary bladder at all time points in male than female mice, suggesting sex-dependent differences in renal clearance. For the 2 hormone-dependent tumor models, differences in 18F-FLT biodistribution between the sexes resulted in altered interpretation of proliferative status on imaging, depending on whether absolute tumor uptake (%ID/g) or T:M ratio was used as the method of quantification. On images of the MDA-MB-231 xenograft (the hormone-independent breast cancer xenograft capable of being grown in both male and female mice), distinct patterns of 18F-FLT uptake were observed depending on the sex of the mouse. Furthermore, quantification of 18F-FLT uptake as absolute tumor uptake (%ID/g) better approximated the Ki-67 proliferative index than did T:M ratios. This work demonstrates that sex is an important consideration in defining quantitative imaging metrics using 18F-FLT as a noninvasive measure of tumor proliferation.
To the best of our knowledge, sex-dependent differences in 18F-FLT biodistribution and PET imaging of preclinical models have not been previously reported. In a systematic review of 174 primary publications using 18F-FLT for oncologic imaging, factors influencing 18F-FLT uptake in tumors were identified (9). The reviewed studies used tumor models grown in only male or female mice and lacked a direct comparison of 18F-FLT imaging between the sexes. Similarly, a comprehensive review of preclinical studies comparing 18F-FDG and 18F-FLT PET imaging for tumor-response monitoring lacked information on whether any differences between the sexes were investigated (17).
Our results add to a small, but growing, amount of literature determining how sex may affect preclinical imaging research. Some evidence points to differences due to the pharmacokinetic properties of the imaging agent, such as absorption, distribution, metabolism, and excretion. For example, sex differences in the metabolism and distribution of the opioid receptor ligand N-(3-18F-fluoropropyl)-N-nordiprenorphine in rats have been reported (18). Other evidence reflects the underlying sex-dependent differences in the expression level of the binding target or receptor of the imaging agent. In research using an 11C-labeled PET radioligand for imaging sphingosine-1-phosphate receptor 2 in a mouse model of multiple sclerosis, differences in cerebellar tracer uptake between male and female mice were identified, reflecting the underlying sex-dependent difference in receptor protein expression (19,20). Sex differences were also reported in σ2-receptor binding density in the brain of a transgenic mouse model of Alzheimer disease using a σ2-receptor–specific imaging agent (21).
There are comparatively more reports on sex differences in clinical imaging research, particularly for neurologic and cardiac imaging. For example, several studies have reported sex differences in regional brain glucose metabolism using 18F-FDG PET and neuroreceptor imaging with targeted radioligands for opioid, dopamine, and cannabinoid receptors (22–25). Sex differences in cardiac glucose metabolism have also been demonstrated using 18F-FDG PET/CT in healthy men and women (26).
This study had several limitations. First, this initial report included only a few breast and prostate cancer models. Subsequent work using additional cell lines will establish the generalizability of the results. In addition to sex, age is another potential biologic variable in preclinical studies (27). Although our results show sex differences in 18F-FLT biodistribution using only one age group (6–8 wk) for xenograft studies, results may differ in older mice used for transgenic or spontaneous tumor models. Last, our results may not translate to humans because of species-level pharmacokinetic differences. In humans, 18F-FLT undergoes hepatic metabolism via glucuronidation (28), but in mice, 18F-FLT is excreted mainly by the kidneys in its original form (29,30). Furthermore, plasma thymidine levels have been shown to be 320-fold higher in rodents than in humans (31).
CONCLUSION
Our results demonstrate that sex is an important variable to consider for preclinical imaging using 18F-FLT. Sex-dependent differences in 18F-FLT biodistribution and nonspecific tissue uptake can adversely affect quantitative measures of 18F-FLT uptake, as well as conclusions on relative tumor proliferation status in comparison with ex vivo reference standards, such as Ki-67 staining. These results have broader implications, as the National Cancer Institute and the imaging community have embarked on numerous initiatives to define imaging metrics of response to therapy. Thus, standardization between the sexes to yield sex-independent imaging metrics of tumor phenotype and therapy response is critical to advance the translational utility of molecular imaging.
DISCLOSURE
This work was supported by grants from the NCI (U01CA141541-01) and the Mallinckrodt Institute of Radiology at Washington University (MIR 11-037). Kooresh Shoghi is supported by grants U24CA209837 and U54CA199092 from the NCI. Funding was also provided to Amy Fowler through the University of Wisconsin Institute of Clinical and Translational Research KL2 Scholar Award (KL2TR000428). Facilities and services were provided by the University of Wisconsin Carbone Cancer Center (UWCCC) Small Animal Imaging Facility, Experimental Pathology Laboratory, and the Translational Research Initiatives in Pathology Laboratory (P30 CA014520). No other potential conflict of interest relevant to this article was reported.
Supplementary Material
Acknowledgments
We thank Dr. John Katzenellenbogen for thoughtful discussions and critical reading of the manuscript; Nicole Fettig and the staff of the Washington University Pre-Clinical Imaging Facility for excellent technical assistance; the Washington University Cyclotron Facility and Carmen Dence for expertise in 18F-FLT radiosynthesis; and the University of Wisconsin–Madison Cyclotron Laboratory for 18F production.
REFERENCES
- 1.Clayton JA, Collins FS. Policy: NIH to balance sex in cell and animal studies. Nature. 2014;509:282–283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Du L, Bayir H, Lai Y, et al. Innate gender-based proclivity in response to cytotoxicity and programmed cell death pathway. J Biol Chem. 2004;279:38563–38570. [DOI] [PubMed] [Google Scholar]
- 3.Karp NA, Mason J, Beaudet AL, et al. Prevalence of sexual dimorphism in mammalian phenotypic traits. Nat Commun. 2017;8:15475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. [DOI] [PubMed] [Google Scholar]
- 5.Bollineni VR, Kramer GM, Jansma EP, Liu Y, Oyen WJ. A systematic review on [18F]FLT-PET uptake as a measure of treatment response in cancer patients. Eur J Cancer. 2016;55:81–97. [DOI] [PubMed] [Google Scholar]
- 6.Tehrani OS, Shields AF. PET imaging of proliferation with pyrimidines. J Nucl Med. 2013;54:903–912. [DOI] [PubMed] [Google Scholar]
- 7.Grierson JR, Schwartz JL, Muzi M, Jordan R, Krohn KA. Metabolism of 3′-deoxy-3′-[F-18]fluorothymidine in proliferating A549 cells: validations for positron emission tomography. Nucl Med Biol. 2004;31:829–837. [DOI] [PubMed] [Google Scholar]
- 8.Investigator’s brochure for 3′-deoxy-3′-[F-18] fluorothymidine: [F-18]FLT: an investigational positron emission tomography (PET) radiopharmaceutical for injection intended for use as an in vivo diagnostic for imaging active cellular proliferation of malignant tumors. American College of Radiology Imaging Network website. https://www.acrin.org/portals/0/protocols/6688/flt_ib__final_v5-5-2011.pdf. Published May 11, 2011. Accessed January 22, 2018.
- 9.Schelhaas S, Heinzmann K, Bollineni VR, et al. Preclinical applications of 3′-deoxy-3′-[18F]fluorothymidine in oncology: a systematic review. Theranostics. 2017;7:40–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chan SR, Vermi W, Luo J, et al. STAT1-deficient mice spontaneously develop estrogen receptor alpha-positive luminal mammary carcinomas. Breast Cancer Res. 2012;14:R16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wainstein MA, He F, Robinson D, et al. CWR22: androgen-dependent xenograft model derived from a primary human prostatic carcinoma. Cancer Res. 1994;54:6049–6052. [PubMed] [Google Scholar]
- 12.Pretlow TG, Wolman SR, Micale MA, et al. Xenografts of primary human prostatic carcinoma. J Natl Cancer Inst. 1993;85:394–398. [DOI] [PubMed] [Google Scholar]
- 13.Sinigaglia F, Talmadge KW. Inhibition of [3H]thymidine incorporation by Mycoplasma arginini-infected cells due to enzymatic cleavage of the nucleoside. Eur J Immunol. 1985;15:692–696. [DOI] [PubMed] [Google Scholar]
- 14.Suehiro M, Vallabhajosula S, Goldsmith SJ, Ballon DJ. Investigation of the role of the base in the synthesis of [18F]FLT. Appl Radiat Isot. 2007;65:1350–1358. [DOI] [PubMed] [Google Scholar]
- 15.Yun M, Oh SJ, Ha HJ, Ryu JS, Moon DH. High radiochemical yield synthesis of 3′-deoxy-3′-[18F]fluorothymidine using (5′-O-dimethoxytrityl-2′-deoxy-3′-O-nosyl-beta-D-threo pentofuranosyl)thymine and its 3-N-BOC-protected analogue as a labeling precursor. Nucl Med Biol. 2003;30:151–157. [DOI] [PubMed] [Google Scholar]
- 16.Oyama N, Ponde DE, Dence C, Kim J, Tai YC, Welch MJ. Monitoring of therapy in androgen-dependent prostate tumor model by measuring tumor proliferation. J Nucl Med. 2004;45:519–525. [PubMed] [Google Scholar]
- 17.Jensen MM, Kjaer A. Monitoring of anti-cancer treatment with 18F-FDG and 18F-FLT PET: a comprehensive review of pre-clinical studies. Am J Nucl Med Mol Imaging. 2015;5:431–456. [PMC free article] [PubMed] [Google Scholar]
- 18.Chesis PL, Griffeth LK, Mathias CJ, Welch MJ. Sex-dependent differences in N-(3-[18F]fluoropropyl)-N-nordiprenorphine biodistribution and metabolism. J Nucl Med. 1990;31:192–201. [PubMed] [Google Scholar]
- 19.Yue X, Jin H, Liu H, Rosenberg AJ, Klein RS, Tu Z. A potent and selective C-11 labeled PET tracer for imaging sphingosine-1-phosphate receptor 2 in the CNS demonstrates sexually dimorphic expression. Org Biomol Chem. 2015;13:7928–7939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cruz-Orengo L, Daniels BP, Dorsey D, et al. Enhanced sphingosine-1-phosphate receptor 2 expression underlies female CNS autoimmunity susceptibility. J Clin Invest. 2014;124:2571–2584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sahlholm K, Liao F, Holtzman DM, Xu J, Mach RH. Sigma-2 receptor binding is decreased in female, but not male, APP/PS1 mice. Biochem Biophys Res Commun. 2015;460:439–445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hu Y, Xu Q, Li K, et al. Gender differences of brain glucose metabolic networks revealed by FDG-PET: evidence from a large cohort of 400 young adults. PLoS One. 2013;8:e83821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zubieta JK, Dannals RF, Frost JJ. Gender and age influences on human brain mu-opioid receptor binding measured by PET. Am J Psychiatry. 1999;156:842–848. [DOI] [PubMed] [Google Scholar]
- 24.Okita K, Petersen N, Robertson CL, Dean AC, Mandelkern MA, London ED. Sex differences in midbrain dopamine D2-type receptor availability and association with nicotine dependence. Neuropsychopharmacology. 2016;41:2913–2919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Normandin MD, Zheng MQ, Lin KS, et al. Imaging the cannabinoid CB1 receptor in humans with [11C]OMAR: assessment of kinetic analysis methods, test-retest reproducibility, and gender differences. J Cereb Blood Flow Metab. 2015;35:1313–1322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kakinuma Y, Okada S, Nogami M, Kumon Y. The human female heart incorporates glucose more efficiently than the male heart. Int J Cardiol. 2013;168:2518–2521. [DOI] [PubMed] [Google Scholar]
- 27.Jackson SJ, Andrews N, Ball D, et al. Does age matter? The impact of rodent age on study outcomes. Lab Anim. 2017;51:160–169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shields AF, Briston DA, Chandupatla S, et al. A simplified analysis of [18F]3′-deoxy-3′-fluorothymidine metabolism and retention. Eur J Nucl Med Mol Imaging. 2005;32:1269–1275. [DOI] [PubMed] [Google Scholar]
- 29.Kim SJ, Lee JS, Im KC, et al. Kinetic modeling of 3′-deoxy-3′-18F-fluorothymidine for quantitative cell proliferation imaging in subcutaneous tumor models in mice. J Nucl Med. 2008;49:2057–2066. [DOI] [PubMed] [Google Scholar]
- 30.Barthel H, Cleij MC, Collingridge DR, et al. 3′-deoxy-3′-[18F]fluorothymidine as a new marker for monitoring tumor response to antiproliferative therapy in vivo with positron emission tomography. Cancer Res. 2003;63:3791–3798. [PubMed] [Google Scholar]
- 31.Li KM, Clarke SM, Rivory LP. Quantitation of plasma thymidine by high-performance liquid chromatography-atmospheric pressure chemical ionization mass spectrometry and its application to pharmadynamic studies in cancer patients. Anal Chim Acta. 2003;486:51–61. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.