Abstract
Chronic cardiac ischemia that impairs cardiac function, but does not result in infarct, is termed hibernating myocardium (HM). A large clinical subset of coronary artery disease (CAD) patients have HM, which in addition to causing impaired function, puts them at higher risk for arrhythmia and future cardiac events. The standard treatment for this condition is revascularization, but this has been shown to be an imperfect therapy. The majority of pre-clinical cardiac research focuses on infarct models of cardiac ischemia, leaving this subset of chronic ischemia patients largely underserved. To address this gap in research, we have developed a well-characterized and highly reproducible model of hibernating myocardium in swine, as swine are ideal translational models for human heart disease. In addition to creating this unique disease model, we have optimized a clinically relevant treatment model of coronary artery bypass surgery in swine. This allows us to accurately study the effects of bypass surgery on heart disease, as well as investigate additional or alternate therapies. This model surgically induces single vessel stenosis by implanting a constrictor on the left anterior descending (LAD) artery in a young pig. As the pig grows, the constrictor creates a gradual stenosis, resulting in chronic ischemia with impaired regional function, but preserving tissue viability. Following the establishment of the hibernating myocardium phenotype, we perform off-pump coronary artery bypass graft surgery to revascularize the ischemic region, mimicking the gold-standard treatment for patients in the clinic.
Keywords: Medicine, Issue 133, Ischemia, revascularization, coronary artery bypass graft, hibernating myocardium, surgical model, swine
Introduction
Coronary heart disease (CHD) affects an estimated 15.5 million people in the United States 1 and is one of the leading causes of death globally. While the mortality rate associated with CHD has gone down in recent years, the incidence and burden on patients and the health care system remain high 1. Primary treatment of severe CAD is revascularization, which improves survival and reduces angina2,3,4. However, cardiac function often remains depressed, especially under increased workload and can progress to heart failure5,6. Clinical trials of coronary artery bypass surgery (CABG) for chronic ischemia demonstrate improvement in survival and symptoms, but ejection fraction shows only modest improvements of 8-10% 7,8. Our innovative and well-characterized swine model of chronic myocardial ischemia is a model of clinical CAD with progressive vascular stenosis. We have demonstrated reduced myocardial contractility resulting from gradual reduction in blood flow 9. The myocardium does not infarct and can remain viable in this scenario. Recovery is possible, though outcomes are variable even with timely revascularization. Chronically ischemic myocardium that remains viable has been characterized by reduced blood flow and function at rest with retained contractile reserve has been termed HM and treatment requires CABG.Although the revascularization of HM should restore contractile function, experimental and clinical observations demonstrate that the recovery is incomplete8,10.
HM is characterized by the presence of viable yet dysfunctional myocardium in the presence of reduced regional blood flow11. Despite impaired contractility and metabolic activity at rest, HM is able to demonstrate functional and metabolic reserve under inotropic stimulation12. HM is suspected in a majority of patients with CAD, and encompasses a broad spectrum of disease. In this protocol, we demonstrate our established swine model of HM bypassed with left internal mammary artery (LIMA) to LAD artery that mimics the clinical scenario. The swine provide an excellent model of heart disease over other large animals as they do not have epicardial bridging collaterals. This allows the stenosis of the LAD alone to result in regional ischemia13.
Here, we describe the surgical method of inducing hibernating myocardium in swine by creating single-vessel stenosis at the LAD artery. Once chronic ischemia has been established (8 weeks post implant of LAD constrictor), we describe the method of recreating the clinical treatment for HM in our swine model: an off-pump coronary artery bypass graft. These surgical methods can be used to not only study a clinically relevant model of chronic cardiac ischemia, but also to investigate the effects of bypass surgery on cardiac ischemia as well as test potential alternate or adjunctive therapies for cardiac ischemia.
Protocol
All animal studies were approved by the Institutional Animal Care and Use Committees of the Minneapolis VA Medical Center and the University of Minnesota. Follow current National Institutes of Health guidelines for the use and care of laboratory animals.
1. Hibernation Surgery
- Animal Preparation
- To create a model of hibernating myocardium, use 5-week old, female, Yorkshire pigs, which weigh in the range of 8-10 kg with 9.2 kg as the average weight.
- For pre-surgical health monitoring, weigh the animal within 3 days prior to surgery.
- Fast the animal for 12 h before starting anesthesia. Ensure that water is available at all times for fasting animals.
- Give SR Buprenorphine 0.18 mg/kg IM 24 h prior to the surgery.
- Induction
- Sedate the animal with 6.6 mg/kg tiletamine-zolazepam and xylazine cocktail given intramuscularly. Allow 5-15 min for the drug cocktail to take effect.
- Prepare the ear vein aseptically by wiping three times with 70% isopropyl alcohol for IV catheter placement. NOTE: If an ear vein is not adequate, another peripheral vein may be used (i.e. cephalic).
- Apply ophthalmic ointment to both eyes to prevent corneal drying.
- If necessary, induce anesthesia by administering 1-2 mg/kg of propofol IV or masked with 3 L/min of oxygen and 3% isoflurane, to allow for tracheal intubation. NOTE: The animal is considered appropriately anesthetized when the blink reflex or jaw tone is absent.
- Intubate the animal with an appropriately sized endotracheal tube.
- Surgery
- Once the animal is anesthetized, shave the left thoracic area for the surgery.
- Initiate the mechanical ventilation at 10-15 breaths per min. Set oxygen at 1-4 L/min, and isoflurane between 1.0 and 3.0% as needed to maintain a surgical plane of anesthesia.
- Place the monitoring devices on the animal. Monitoring devices should include ECG, blood pressure, temperature, and pulse oximeter.
- Connect the IV catheter to an IV drip with 0.9% normal saline at a rate of 5-10 mL/kg/h.
- Aseptically prepare the left lateral chest with an FDA approved surgical scrub.
- Evaluate the depth of anesthesia every 5-15 min. Record the baseline vital signs (heart rate, ECG, SpO2, NIBP, ETCO2 and temperature) every 15 min. NOTE: The animal is considered appropriately anesthetized when the blink reflex or jaw tone is absent.
- With the animal laying on its right side, place sterile drapes the left side of the chest from the spine to the scapula centering the third rib space in the sterile field. NOTE: The sterile field involves left front shoulder including the axillary area continuing to the sternum to the sixth rib space cranially.
- Gently scrub the field with an FDA approved surgical method such as chlorhexidine gluconate or an iodine surgical scrub for 2 min using a target pattern.
- Rinse the field with water and wash it two more times with an FDA approved method as above for 2 min. Rinse the surgical field with water in between washes, then pat dry with sterile toweling or 4 x 4 cm gauze sponges.
- Spray the area with an iodine solution and drape the entire animal (except the surgical field) with sterile drapes. Use steam or gas sterilization to sterilize the instruments, gowns, and drapes for the procedure.
- Administer an intercostal nerve block using 0.3 mL of 0.5% bupivicaine at the 2nd, 3rd and 4th ribs.
- Give 125 mg of cefazolin IV before opening the chest. NOTE: 1.5 mg/kg succinylcholine IV may be given as a paralytic agent to reduce muscle movement per surgeon's request during the opening of the chest cavity.
- Expose the heart via a left thoracotomy. Prior to heart manipulation, give a 2 mg/kg IV bolus of lidocaine to prevent arrhythmia. Make the lateral 3rd space thoracotomy incision with monopolar electrocautery. The incision should extend from the cranial aspect of the scapula to the axillary area for approximately 10 cm. Use a cautery to incise the skin and underlying musculature to allow the entry into the chest cavity.
- Place a finochietto rib retractor and enter the pleural space at the third intercostal space to retract the lung.
- Sharply incise the pericardium and retract to the side using 2-0 polyglactin stay sutures, exposing the LAD artery and vein. Retract the left atrial appendage with a gauze-covered spatula retractor.
- Using iris scissors, open the adventitia over the LAD.
- Bluntly dissect an approximately 0.25 cm portion of the LAD artery approximately 1 cm distal to the LAD and circumflex artery junction. Use a right-angle clamp to encircle the LAD.
- Place two 4-0 polyester sutures around the artery without placing any tension.
- Place a plastic c-shaped constrictor with an internal diameter of 1.5 mm on the LAD proximal to the first diagonal without occluding the vessel.
- Secure it with two 4-0 polyester ties encircling the LAD.
- Loosely close the pericardium using 2-0 polyglactin suture in a simple interrupted pattern.
- Perform a breath hold to remove air from the chest while the intercostal muscle layer is closed. Carefully monitor the airway pressure manometer on the anesthesia machine to maintain the pressure in a proper range of 20-30 cm of water and release when complete. NOTE: This eliminates the need for a chest tube in the post-operative period.
- Close the muscle and skin layers in the standard fashion using 2-0 and 3-0 absorbable suture.
- Use skin adhesive over the incision for skin closure.
- Administer a total dose of 1 mg/kg bupivacaine intramuscularly at multiple sites along the length of the incision at the end of the procedure. Administer meloxicam 0.2 mg/kg SQ before transport to the recovery area.
- Wean the animal off the ventilator as the skin layer is closed.
- Leave the animal connected to the anesthesia machine until it can spontaneously breathe and protective reflexes begin to return.
- Do not remove the endotracheal tube until the animal can swallow to protect its airway.
- Apply a sterile, non-adherent dressing with triple-antibiotic ointment over the incision.
- Post-Surgery
- Record the heart rate, respiration rate, body temperature and mucous membrane color every 15 min until the animal can hold a sternal position unassisted.
- Do not leave the animal unattended until it can lift and hold its head up and can stand unassisted.
- Give meloxicam (0.2 mg/kg) subcutaneously.
- Administer sustained release buprenorphine 6 h following the initial pre-surgical dose.
- Leave the dressing on the incision for up to 3 days, if it remains clean and dry. Replace the dressing if it becomes soiled.
- Monitor the pain level, general well-being and the condition of the incision for 5 days following the procedure. NOTE: A piglet grimace scale is now available to assess the level of pain in these animals post-operatively14. Use half dose meloxicam as needed once per day for breakthrough pain.
- Single-house the animal during the recovery period of 5 day to limit activity and allow the wound to heal.
- Allow 8 weeks to fully develop the hibernating myocardium phenotype.
2. Revascularization or Off Pump Bypass
- Animal Preparation
- Weigh the animal within 3 days prior to surgery.
- Fast the animal for 12 h prior to surgery. Ensure that water is available at all times for fasting animals.
- Give sustained release buprenorphine 0.18 mg/kg intramuscularly 24 h prior to the surgery.
- Induction
- Sedate the animal with 6.6 mg/kg tiletamine-zolazepam/xylazine cocktail intramuscularly. 5-15 min after the sedation, prepare the ear vein aseptically for catheter placement. NOTE: If an ear vein is not adequate, another peripheral vein may be used (i.e. cephalic).
- Apply ophthalmic ointment to both eyes.
- Induce anesthesia with 1-2 mg/kg of propofol IV.
- Intubate the animal with an appropriately sized endotracheal tube.
- Surgery
- Shave the animal's sternum, left lateral chest and femoral triangles for the surgery.
- Set mechanical ventilation to 10-15 breaths per minute, oxygen at 1-4 L/min, and isoflurane at 1.0-3.0% as needed to maintain a surgical plane of anesthesia.
- Position the monitoring equipments (heart rate, ECG, SpO2, direct blood pressure, ETCO2 and temperature) on the animal.
- Connect the IV catheter to an IV drip with normal saline or LRS.
- Aseptically prepare the skin with povidone iodine scrub or other FDA approved method.
- Evaluate the animal for the depth of anesthesia. NOTE: The animal is considered deeply anesthetized when the eye or jaw reflex is absent.
- Position the animal dorsally, prepare the skin as previously described and drape the animal with sterile towels.
- Perform a cut down to the femoral artery and cannulation. Use monopolar electrocautery to create an approximately 3 cm skin incision over the femoral artery to expose it for cannulation.
- Perform a femoral nerve block after the incision is made, to allow precise placement of drug. Connect the femoral cannula to a transducer to monitor blood pressures during the surgery.
- Use electro-cautery to incise from the manubrium to the xiphoid process, as well as to incise the muscle, adipose, and connective tissue to the level of the sternum.
- Divide the sternum with an oscillating saw. NOTE: Using an oscillating saw, rather than a standard sternal saw avoids potential myocardial injury from pericardial adhesions as a result of the thoracotomy for constrictor placement on the LAD.
- Administer lidocaine (2 mg/kg IV loading dose, 50 µg/kg/min continuous IV infusion) to prevent arrhythmias. Dilute lidocaine in a 500 mL bag of normal saline at a concentration of 1 mg/mL.
- Divide the posterior sternal plate with straight Mayo scissors.
- Use a chest retractor to expose the thoracic cavity.
- Take down the adhesions with monopolar electro-cautery.
- Using the same retractor, with gentle elevation of the left sternal border by an assistant, dissect the left internal mammary artery free from the chest wall.
- Control the arterial branches with hemoclips.
- Heparinize the animal with 100-300 units/kg of heparin IV.
- After 3 min, divide the artery with the distal end over sewn. Prepare the proximal end for grafting.
- Open and retract the pericardium. Confirm that the length of the LIMA is adequate to reach the appropriate anastomotic site on the LAD.
- Perform the LIMA to LAD anastomosis with 7-0 polypropylene suture using an off pump technique with a cardiac stabilizer, spring form clip, and appropriately sized intraluminal devices.
- Place a chest tube between the ribs on the left lateral chest, externalized and attached to vacuum to remove air from the chest cavity for 15-30 min.
- Approximate the sternum with #5 polyester sutures using a figure eight pattern.
- Close the muscle, subcutaneous and skin layers in the usual fashion as described in the Hibernation surgery. Remove the chest tube while performing a Valsalva maneuver to remove any residual air from the chest cavity and close the incision with one skin suture.
- Apply skin adhesive over the skin to protect the sternal incision. Gently remove the femoral catheter and repair the artery using 7-0 polypropylene suture in a purse string pattern. Close the skin using 2-0 polyglactin suture in a subcuticular pattern.
- Administer a total dose of 1 mg/kg bupivacaine intramuscularly at multiple sites along the length of the incision at the end of the procedure.
- Administer meloxicam 0.2 mg/kg subcutaneously before transport to the recovery area.
- Follow the post-operative protocol in the hibernation procedure.
Representative Results
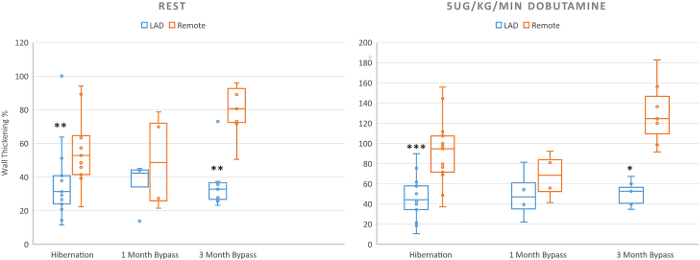
Following the initial hibernation surgery, stenosis greater than 70% should be able to be observed by clinical imaging techniques such as angiography or cardiac MRI (Figure 1A). 8 weeks following the surgical application of the LAD constrictor, analysis of regional function by ECHO or cardiac MRI reveals reduced function under increased workload (Figure 2). This can be tested by stimulating the heart with dobutamine (5mg/kg/min), and measuring circumferential strain and regional wall thickening. PET imaging demonstrates decreased blood flow and increased glucose uptake in the hibernating region as compared to the non-LAD territory within the same heart, indicating that the ischemic tissue remains viable. This enhancement in glucose uptake relative to blood perfusion is known as the "flow-metabolism mismatch" and mimics clinical findings of chronically hibernating myocardium4. There is no evidence of infarct by any imaging technique. If an infarct in the LAD region is present, the constrictor was too tight and created a full occlusion of the artery. If no regional abnormalities are evident, a hibernating phenotype has not been achieved.
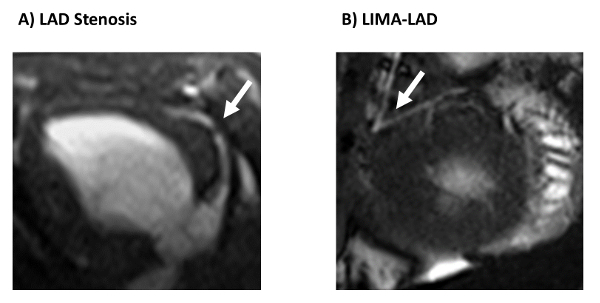
Following successful coronary artery bypass surgery, animals may show incremental improvements in regional cardiac function both at rest and under inotropic stimulation with dobutamine, though these improvements will not restore function back to normal levels (Figure 3). Successful bypass surgery will eliminate the mortality risk associated with HM (Figure 4). The patent graft can be visualized by either angiography or cardiac MRI (Figure 1B). At necropsy, the LAD stenosis and LIMA patency is confirmed using sized coronary dilators. Myocardium is inspected to confirm viable tissue is present in all regions with infarction.
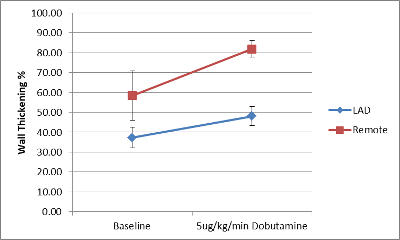
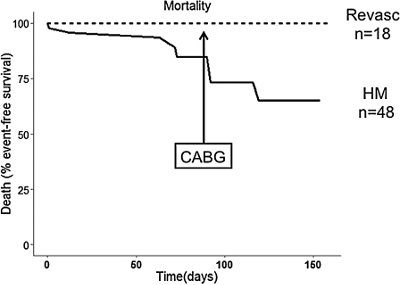
Figure 1. Cardiac MRI images of stenosis and bypass graft. Cardiac MRI visualizes A) the LAD stenosis following constrictor placement, and B) the LIMA-LAD graft following coronary artery bypass surgery. This figure has been adapted from Hocum Stone, et al.9. Please click here to view a larger version of this figure.
Figure 2. Cardiac MRI measurement of percent wall thickening. Measurement of wall thickening % by cardiac MRI shows impairment of regional function in hibernating regions of the left ventricle. Wall thickening % is significantly decreased in hibernating animals both at rest and under dobutamine infusion as compared to remote regions. Bypassed animals showed improvement in wall thickening % both at rest and under dobutamine infusion. (*=p< 0.05;**=p<0.01 ***=p<0.001) (Hibernation n=12; 1 month revasc n=4; 3 months revasvc n=5) This figure has been adapted from Hocum Stone, et al.9. Data are presented as mean ± SEM. Please click here to view a larger version of this figure.
Figure 3. Regional functional deficit remains present following bypass surgery. Echocardiogram performed 4 weeks following revascularization of hibernating myocardium shows regional functional deficit under inotropic stimulation following bypass surgery. (n=5) Data are presented as mean ± SEM. Please click here to view a larger version of this figure.
Figure 4. Effect of bypass surgery on mortality in HM. Surgical revascularization eliminates mortality risk associated with HM. (Bypass n=18; hibernation n=48). This figure has been adapted from Holley, et al.15 Please click here to view a larger version of this figure.
Discussion
Here, we show that our swine model of HM accurately mimics the clinical experience of patients with single vessel disease and preserved left ventricular function. Prior to the revascularization, animals with single vessel HM exhibit minimal impairment in global function as measured by ejection fraction, but show significant reduction in regional wall thickening. Following the revascularization, CMR imaging at one or three months of recovery demonstrates preserved viability and graft patency but persistent regional dysfunction, as noted by the estimates of contractile reserve with low dose dobutamine stress testing.
There are several critical steps for the initial hibernation operation. Entering the chest at the third intercostal space allows for the easiest access to the proximal LAD. The retraction of the left atrial appendage with a moist gauze facilitates visualization of the vessel without inducing arrhythmias. The assistance to expose the LAD minimizes the bleeding and secures the constrictor. Chest closure with the Valsalva maneuver to evacuate the air prevents pneumothorax.
There are several critical steps for a successful revascularization procedure. Appropriate depth of anesthesia and use of paralytic agent ensured no movement during the anastomosis portion of the procedure. Use of lidocaine and 200p units/kg Heparin eliminates arrhythmia and thrombosis events. Using a femoral arterial line to maintain appropriate blood pressure monitoring is critical to the hemodynamic stability of the animal. Use a flo-thru device to improve animal stability during the anastomosis and alleviate the need for retraction stabilizing tapes. While sewing the graft, an O2 blower is helpful to visualize the anastomosis.
During the revascularization procedure, if arrhythmia is observed, the animal may require a second dose of lidocaine. If the LAD is difficult to visualize, place the stabilizer after dissection of epicardial fat or fibrinous tissue. Anesthetically, once the stabilizer is placed and the heart is lifted, mild decreases in arterial blood pressure will be noted in addition to ST depression on the ECG. These abnormalities are usually tolerable and do not require intervention. If changes in cardiovascular stability are more dramatic, a dose of phenylephrine (5-20 µg/kg IV) may be given IV to increase arterial blood pressure. Epinephrine (0.1 µg/kg IV; diluted 1:10,000) would be used as an emergency rescue drug if changes are life threatening. Blood loss is replaced with a crystalloid solution IV. A bolus of 100-300 mL of normal saline is used for additional blood pressure support. The LIMA is most easily taken down as a near-skeletonized vessel, but it may be necessary to have papaverine available if a spasm occurs.
Our model uses off-pump surgery rather than on-pump for the revascularization, as this allows us to minimize the operative time and avoid the cannulation of the aorta and right atrium with full heparinization. It also reduces the risk of post-operative bleeding and/or cardiac tamponade, simplifying the animal's recovery. Of note, there is no similar model of coronary bypass surgery in a model of HM in an animal that is then allowed to recover for 30-120 days. These are presumed advantages based on clinical experience in patients undergoing CABG both on and off pump.
This technique can be expanded to involve multiple coronary artery disease by the placement of a constrictor on the circumflex artery at the same time as the LAD or as an alternative vessel. This two-vessel disease model would result in more rapid development of ischemic cardiomyopathy and greater understanding of resultant myocardial adaptions. It is a model that would still allow for ongoing adjunctive interventions including pharmacologic, cell based or mechanical options.
This complex model of the revascularization of HM reflects the clinical difficulty of managing patients with viable but chronically ischemic, dysfunctional myocardium. There is a high prevalence of patients with HM, often presenting with various comorbidities and cardiovascular structural diseases16, and are at risk for sudden cardiac death (SCD)6. The revascularization of viable, impaired myocardium is associated with a 79% reduction in annual death rate3. In fact, we have shown that revascularization of patients with viable hibernating myocardium, as defined by PET imaging, is associated with a greater degree of improvement in LV ejection fraction at 6 weeks following CABG17. In animals with HM, circumferential strain is impaired at baseline, but there is evidence of contractile reserve under inotropic stimulation with a low dose of dobutamine. The presence of contractile reserve is one of the most specific indicators of myocardial viability 18, and the presence of such viability is a predictor of the potential benefit of bypass surgery when present.
Our model is limited by the necessity of using young, healthy animals to create the model of HM. It is necessary to use a young animal to implant the constrictor on the LAD artery as juvenile animals have arteries that are small enough to place the constrictor around without creating immediate stenosis. This model cannot be achieved by beginning with adult pigs, though that would more closely simulate the clinical experience, due to size limitations of both the constrictor, as well as the size of standard surgical and MRI equipment.
An additional limitation is that this animal model of HM only allows the analysis of the effects of a single territory of chronic ischemia, whereas clinical cases are typically far more complex, and may respond differently to the revascularization.
Disclosures
The authors have nothing to disclose.
Acknowledgments
This work was supported by the VA Merit Review #I01 BX000760 (RFK) from the United States (U.S.) Department of Veterans Affairs BLR&D. The contents of this work do not represent the views of the U.S. Department of Veterans Affairs of the United States Government.
References
- Mozaffarian D, et al. Heart Disease and Stroke Statistics-2016 Update A Report From the American Heart Association. Circulation. 2015;133(4):350. doi: 10.1161/CIR.0000000000000350. [DOI] [PubMed] [Google Scholar]
- Di Carli MF, et al. Value of metabolic imaging with positron emission tomography for evaluating prognosis in patients with coronary artery disease and left ventricular dysfunction. Am J Cardiol. 1994;73(8):527–533. doi: 10.1016/0002-9149(94)90327-1. [DOI] [PubMed] [Google Scholar]
- Gerber BL, et al. Prognostic value of myocardial viability by delayed-enhanced magnetic resonance in patients with coronary artery disease and low ejection fraction: impact of revascularization therapy. J Am Coll Cardiol. 2012;59(9):825–835. doi: 10.1016/j.jacc.2011.09.073. [DOI] [PubMed] [Google Scholar]
- McFalls E, et al. Regional glucose uptake within hypoperfused swine myocardium as measured by positron emission tomography. Am J Physiol-Heart C. 1997;272(1):343–349. doi: 10.1152/ajpheart.1997.272.1.H343. [DOI] [PubMed] [Google Scholar]
- Velazquez EJ, et al. Coronary-artery bypass surgery in patients with left ventricular dysfunction. New Engl J Med. 2011;364(17):1607–1616. doi: 10.1056/NEJMoa1100356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canty JM, Fallavollita JA. Hibernating myocardium. J Nucl Cardiol. 2005;12(1):104–119. doi: 10.1016/j.nuclcard.2004.11.003. [DOI] [PubMed] [Google Scholar]
- Joyce D, et al. Revascularization and ventricular restoration in patients with ischemic heart failure: the STICH trial. Curr Opin Cardiol. 2003;18(6):454–457. doi: 10.1097/00001573-200311000-00006. [DOI] [PubMed] [Google Scholar]
- Shah BN, Khattar RS, Senior R. The hibernating myocardium: current concepts, diagnostic dilemmas, and clinical challenges in the post-STICH era. Eur Heart J. 2013;34(18):1323–1336. doi: 10.1093/eurheartj/eht018. [DOI] [PubMed] [Google Scholar]
- Stone LLH, et al. Magnetic resonance imaging assessment of cardiac function in a swine model of hibernating myocardium 3 months following bypass surgery. J Thorac Cardiov Sur. 2016. [DOI] [PubMed]
- Kukulski T, et al. Implication of right ventricular dysfunction on long-term outcome in patients with ischemic cardiomyopathy undergoing coronary artery bypass grafting with or without surgical ventricular reconstruction. J Thorac Cardiov Sur. 2014;149(5):1312–1321. doi: 10.1016/j.jtcvs.2014.09.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahimtoola SH. The hibernating myocardium. Am Heart J. 1989;117(1):211–221. doi: 10.1016/0002-8703(89)90685-6. [DOI] [PubMed] [Google Scholar]
- Fallavollita JA, Logue M, Canty JM. Stability of hibernating myocardium in pigs with a chronic left anterior descending coronary artery stenosis: absence of progressive fibrosis in the setting of stable reductions in flow, function and coronary flow reserve. J Am Coll Cardiol. 2001;37(7):1989–1995. doi: 10.1016/s0735-1097(01)01250-5. [DOI] [PubMed] [Google Scholar]
- White FC, Carroll SM, Magnet A, Bloor CM. Coronary collateral development in swine after coronary artery occlusion. Circ Res. 1992;71(6):1490–1500. doi: 10.1161/01.res.71.6.1490. [DOI] [PubMed] [Google Scholar]
- Viscardi AV, Hunniford M, Lawlis P, Leach M, Turner PV. Development of a Piglet Grimace Scale to Evaluate Piglet Pain Using Facial Expressions Following Castration and Tail Docking: A Pilot Study. Front Vet Sci. 2017;4:51. doi: 10.3389/fvets.2017.00051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holley CT, Long EK, Lindsey ME, McFalls EO, Kelly RF. Recovery of hibernating myocardium: what is the role of surgical revascularization. J Cardiac Surg. 2015;30(2):224–231. doi: 10.1111/jocs.12477. [DOI] [PubMed] [Google Scholar]
- Cooper HA, Braunwald E. Clinical importance of stunned and hibernating myocardium. Coronary Artery Dis. 2001;12(5):387–392. doi: 10.1097/00019501-200108000-00008. [DOI] [PubMed] [Google Scholar]
- McFalls EO, et al. Utility of positron emission tomography in predicting improved left ventricular ejection fraction after coronary artery bypass grafting among patients with ischemic cardiomyopathy. Cardiology. 2000;93(1-2):105–112. doi: 10.1159/000007010. [DOI] [PubMed] [Google Scholar]
- Pasquet A, et al. Prediction of global left ventricular function after bypass surgery in patients with severe left ventricular dysfunction. Impact of pre-operative myocardial function, perfusion, and metabolism. Eur Heart J. 2000;21(2):125–136. doi: 10.1053/euhj.1999.1663. [DOI] [PubMed] [Google Scholar]


