Abstract
Taurine is an abundant, β-amino acid with diverse cytoprotective activity. In some species, taurine is an essential nutrient but in man it is considered a semi-essential nutrient, although cells lacking taurine show major pathology. These findings have spurred interest in the potential use of taurine as a therapeutic agent. The discovery that taurine is an effective therapy against congestive heart failure led to the study of taurine as a therapeutic agent against other disease conditions. Today, taurine has been approved for the treatment of congestive heart failure in Japan and shows promise in the treatment of several other diseases. The present review summarizes studies supporting a role of taurine in the treatment of diseases of muscle, the central nervous system, and the cardiovascular system. In addition, taurine is extremely effective in the treatment of the mitochondrial disease, mitochondrial encephalopathy, lactic acidosis, and stroke-like episodes (MELAS), and offers a new approach for the treatment of metabolic diseases, such as diabetes, and inflammatory diseases, such as arthritis. The review also addresses the functions of taurine (regulation of antioxidation, energy metabolism, gene expression, ER stress, neuromodulation, quality control and calcium homeostasis) underlying these therapeutic actions.
Keywords: Taurine, Cytoprotection, Neurodegenerative diseases, Antioxidation, ER stress, MELAS
INTRODUCTION
Taurine is a β-amino acid found in very high concentration in most cells, with levels particularly high in excitable tissues. Although taurine possesses many functions in mammals, its cytoprotective actions have attracted the most attention, as they dramatically alter the health and nutritional status of various species. Because taurine regulates fundamental events in the cell, while altering the balance between life and death, interest in taurine’s physiological functions has grown. These findings have provided impetus for the use of taurine in infant formula, nutritional supplements and energy drinks. It also has stimulated research into its potential therapeutic uses. Although most of those studies have focused on taurine-mediated reversal of pathology in animals, there have been attempts to translate the basic science findings into clinical applications. Results of many clinical studies have been encouraging, suggesting a promising future for taurine therapy (Ginguay et al., 2016). Equally promising are studies showing the nutritional value of taurine (McCarty, 2013). In some species, such as the cat and fox, taurine is an essential nutrient (Schmidt et al., 1976; Novotny et al., 1991; Ito et al., 2008; Ripps and Shen, 2012). Not only does taurine deficiency cause pathology in those animals but it also shortens their lifespan (Ito et al., 2014a; Park et al., 2014). By contrast, taurine is classified as a conditionally essential nutrient or a functional nutrient in man (Gaull, 1986, 1989; Bouckenooghe et al., 2006). Although humans are incapable of synthesizing large amounts of taurine, the retention of taurine by human tissues is greater than that of cats or fox. Thus, unlike cats, humans do not readily develop overt signs of taurine deficiency although parenteral feeding can be associated with taurine deficiency (Arrieta et al., 2014). Nonetheless, human studies have revealed the nutritional value of taurine. Particularly noteworthy is a World Health Association study involving 50 population groups in 25 different countries throughout the world, which reports that elevated dietary taurine consumption is associated with decreased risk of hypertension and hypercholesterolemia (Yamori et al., 2004; Sagara et al., 2015). Taurine supplementation is also linked to diminished body mass index (Yamori et al., 2010) and reduced levels of inflammation markers in obese women (Rosa et al., 2014). Thus, the cytoprotective actions of taurine contribute to an improvement in the clinical and nutritional health of humans. The present review discusses the mechanisms underlying the cytoprotective activity of taurine, the influence of taurine on a wide range of diseases and the nutritional value of taurine supplementation.
MECHANISMS UNDERLYING THE CYTOPROTECTIVE ACTIVITY OF TAURINE
Antioxidant activity
Recent studies have uncovered novel mechanisms responsible for taurine-mediated cytoprotection (Table 1). One of the primary mechanisms of taurine cytoprotection appears to involve its antioxidant activity, which is mediated by three distinct events. First, taurine is a proven anti-inflammatory agent that neutralizes the neutrophil oxidant, hypochlorous acid. The product of the reaction between taurine and hypochlorous acid, taurine chloramine, also interferes with the inflammatory process (Kim and Cha, 2014; Marcinkiewicz and Kontny 2014). Second, taurine diminishes the generation of superoxide by the mitochondria (Jong et al., 2012; Schaffer et al., 2014a). In normal mitochondria, taurine forms a conjugate with a uridine residue of tRNALeu(UUR). Because the modified uridine residue is located in the Wobble position of the anticodon, the conjugation reaction is capable of enhancing the interaction of the AAU anticodon of tRNALeu(UUR) with the UUG codon of mitochondrial mRNAs. However, in certain mitochondrial diseases, the formation of the taurine conjugate is diminished, an effect that suppresses the expression of specific mitochondria encoded proteins, such as NADH-ubiquinone oxidoreductase chain 6 (ND6) (Schaffer et al., 2014a). ND6 is a subunit of complex I that is required for maximal complex I activity and the proper assembly of the complex. Therefore, a reduction in ND6 biosynthesis decreases complex I activity, the utilization of NADH by the respiratory chain and mitochondrial ATP generation but it increases the generation of superoxide by the respiratory chain (Jong et al., 2012; Schaffer et al., 2016; Shetewy et al., 2016). It is widely accepted that mitochondrial oxidative stress damages macromolecules within the mitochondria, but more importantly it is capable of triggering the mitochondrial permeability transition (permeabilization of the inner mitochondrial membrane) and mitochondria-dependent apoptosis (Ricci et al., 2008, Shetewy et al., 2016). This sequence of events can be disrupted by taurine treatment. In the mitochondrial disease, mitochondrial encephalopathy, lactic acidosis, and stroke-like episodes (MELAS), the formation of the taurine conjugate is impaired. Taurine therapy provides a source of substrate for the taurine conjugation reaction, thereby restoring mitochondrial protein biosynthesis, improving mitochondrial function and reducing superoxide generation (Rikimaru et al., 2012; Schaffer et al., 2014b). In support of this theory, it has been shown that promoters of mitochondrial oxidative stress, including ozone, nitrogen dioxide, bleomycin, amiodarone, arsenic, iron, Adriamycin and catecholamines, to name a few, respond favorably to taurine therapy, (Schaffer et al., 2009). Third, reactive oxygen species (ROS) generated by the mitochondria can damage antioxidant enzymes that are capable of preventing oxidative stress. Because the activity of some of the antioxidant enzymes is sensitive to oxidative damage, taurine may limit oxidative stress by preventing damage to those sensitive enzymes.
Table 1.
Mechanisms underlying cytoprotective actions of taurine to improve clinical and nutritional health of humans
| Cytoprotection | Functions of Taurine |
|---|---|
| Antioxidation | Anti-inflammation by neutralization of hypochlorous to produce taurine chloramine (Kim and Cha, 2014; Marcinkiewicz and Kontny, 2014) |
| Diminishes superoxide by conjugating with uridine of tRNALeu(UUR) in mitochondria (Jong et al., 2012; Schaffer et al., 2014a) | |
| Generates ATP by encoding mitochondrial ND6 protein (Jong et al., 2012; Schaffer et al., 2016; Shetewy et al., 2016) Prevents mitochondrial membrane permeability and apoptosis (Ricci et al., 2008, Shetewy et al., 2016) | |
| Benefits mitochondrial disease, MELAS by providing substrate for taurine conjugation (Rikimaru et al., 2012; Schaffer et al., 2014b) | |
| Energy metabolism | Activates complex I and NADH sensitive enzymes by reducing NADH/NAD+ ratio during glycolysis (Schaffer et al., 2016) |
| Restores fatty acid oxidation by increasing PPARalpha levels (Schaffer et al., 2016) | |
| Conjugates bile acids to facilitate lipid absorption by intestines (Schaffer et al., 2016) | |
| Gene expression | Changes transcription profile of metabolism-related genes (Park et al., 2006) |
| Modulates genes to induce longevity (Ito et al., 2014a) | |
| Changes transcription factors (Schaffer et al., 2016) | |
| Modulates protein phosphorylation and cell signalling (Lombardini, 1996; Ramila et al., 2015) | |
| ER stress | Attenuates ER stress by improving protein folding (Ito et al., 2015a) |
| Ameliorates stroke brain injury by inhibiting ER stress (Gharibani et al., 2015) | |
| Protects neurons in stroke and Alzheimer’s disease (Prentice et al., 2015) | |
| Neuromodulation | Protects CNS by agonizing GABAA, glycine and NMDA receptors (El Idrissi and L’Amoreaux, 2008; Chan et al., 2013) |
| Decreases seizures by binding with GABAA receptor (L’Amoreaux et al., 2010) | |
| Protects against seizures by elevating glutamic acid decarboxylase (El Idrissi and L’Amoreaux, 2008) | |
| Quality control | Protects cardiomyocytes by activating ubiquitin-proteasome system and autophagy (Jong et al., 2015) |
| Attenuates toxin-mediated autophagy (Li et al., 2012; Bai et al., 2016) | |
| Ca2+ homeostasis | Protects heart and brain during myocardial infarction and stroke by diminishing Ca2+ overload (Li et al., 2012; Bai et al., 2016) |
| Taurine loss during ischemia-reperfusion protects heart by reducing hypoxia-induced Ca2+ overload (Schaffer et al., 2002) | |
| Taurine depletion leads to cardiomyopathy due to reduced activity of SR Ca2+ ATPase (Ramila et al., 2015) | |
| Protects brain neurons during epilepsy by inducing Ca2+ binding proteins (Junyent et al., 2010) | |
| Protects neurons against glutamate excitotoxicity by reducing glutamate-induced elevation of [Ca2+]i (Wu et al., 2005) | |
| Osmoregulation | Serves as an organic osmolyte (Schaffer et al., 2002) |
Effect on energy metabolism
Taurine deficiency-mediated impairment of complex I activity also affects energy metabolism, largely through elevations in the NADH/NAD+ ratio, which regulate energy metabolism by feedback inhibiting key dehydrogenases. The citric acid cycle is very sensitive to increases in the NADH/NAD+ ratio, as three NADH sensitive enzymes (α-ketoglutarate dehydrogenase, isocitrate dehydrogenase and citrate synthase) are subject to inhibition by elevations in the NADH/NAD+ ratio. For example, oxidation of pyruvate by the taurine deficient heart falls, as elevations in the NADH/NAD+ ratio inhibits pyruvate dehydrogenase activity and causes a deficiency in pyruvate, arising from the massive conversion of pyruvate to lactate (Schaffer et al., 2016). Thus, despite stimulation of glycolysis, glucose oxidation is significantly reduced in the taurine deficient heart, dramatically decreasing the contribution of glucose metabolism toward overall ATP biosynthesis. The rate of taurine biosynthesis by the liver is low in humans, therefore, the major source of taurine in humans is the diet. For many years, diets rich in seafood were considered an excellent source of taurine, with meat containing some but less taurine than seafood. However, the recent introduction of taurine-containing supplements, such as Bacchus-D and Red Bull, provides an alternative source of taurine. The supplements have proven to be effective therapeutic agents, at least in the case of heart failure. According to Jeejeebhoy et al. (2002), taurine is deficient in hearts of patients suffering from heart failure. Restoration of taurine levels in these patients through supplementation leads to improved contractile function. This study reinforces the view that taurine supplements are important therapeutic agents. However, the largest decline in energy metabolism occurs in fatty acid oxidation, which falls in part because of a decrease in citric acid cycle flux. Also suppressing fatty acid oxidation in taurine deficiency are the low levels of the transcription factor, PPARα (Schaffer et al., 2016). PPARα regulates several proteins and enzymes involved in fatty acid metabolism, with the most important being the long chain fatty acyl carnitine transporter complex (Schaffer et al., 2016). Another factor affecting lipid metabolism is taurine deficiency-mediated reductions in bile acid biosynthesis, as bile acids facilitate the absorption of lipids by the intestines (see section on atherosclerosis).
Regulation of gene expression
Park et al. (2006) were the first investigators to recognize that taurine treatment triggers genetic changes. More recently, Ito et al. (2014a) have identified several taurine sensitive genes that contribute to a wide range of cellular functions (cell cycle progression, cell signaling, death and survival, amino acid metabolism, protein biosynthesis, protein folding and aging). Taurine-mediated changes in transcription factor content have also been reported (Schaffer et al., 2016). Although taurine is known to modulate protein phosphorylation and cell signaling (Lombardini, 1996; Ramila et al., 2015), it remains to be determined if alterations in protein phosphorylation are involved in taurine-mediated genetic changes.
Modulation of ER stress
Another important mechanism of taurine cytoprotection is attenuation of endoplasmic reticular (ER) stress. ER stress is an important regulatory mechanism designed to restore ER function and re-establish a balance between protein degradation and protein biosynthesis/folding. When a cell experiences excessive ER stress, pathways are stimulated that can kill the cell. A common initiator of ER stress is the accumulation of defective proteins, whose levels increase as a result of improper protein folding, inadequate protein degradation or ER dysfunction. To restore ER function and the balance between protein degradation and protein biosynthesis/folding, unfolded or misfolded proteins activate three stress sensors (PERK, ATF6 and IRE1) that initiate distinct pathways known as the unfolded protein response (UPR) pathways. Together, the UPR pathways are capable of suppressing protein biosynthesis, enhancing protein degradation, generating chaperones to improve protein folding and initiating either autophagy or apoptosis. During a stroke, taurine decreases glutamate toxicity, thereby reducing both oxidative stress and calcium overload. However, taurine also suppresses two of the three UPR pathways. Although the mechanisms underlying the actions of taurine against ER stress and the UPR pathways remain to be determined, it is relevant that taurine deficiency is associated with ER stress (Ito et al., 2015a). It has been proposed that taurine might alter protein folding, either by reducing oxidative stress or providing a better osmotic environment for protein folding (Ito et al., 2015a). The initial studies describing the effect of taurine on ER stress used cellular and animal models of stroke (Gharibani et al., 2013, 2015). According to the authors of those studies, ER stress, along with oxidative stress and mitochondrial dysfunction, are characteristic features of stroke and neurodegenerative diseases, including Alzheimer’s, Huntington’s and Parkinson’s diseases (Prentice et al., 2015). During stroke, massive amounts of the neurotransmitter, glutamate, are released which overstimulates postsynaptic neurons leading to a neuroexcitotoxic response, characterized by oxidative stress, calcium overload, ER stress and in some cases cell death (Prentice et al., 2015).
Taurine as an inhibitory neuromodulator
Although ER stress assumes an important role in the cytoprotective actions of taurine in the central nervous system (CNS), another important mechanism affecting the CNS is the neuromodulatory activity of taurine. Toxicity in the CNS commonly occurs when an imbalance develops between excitatory and inhibitory neurotransmitters. GABA is one of the dominant inhibitory neurotransmitters, therefore, reductions in either the CNS levels of GABA or the activity of the GABA receptors can favor neuronal hyperexcitability. Taurine serves as a weak agonist of the GABAA, glycine and NMDA receptors (El Idrissi and L’Amoreaux, 2008; Chan et al., 2013). Therefore, taurine can partially substitute for GABA by causing inhibition of neuronal excitability. However, the regulation of the GABAA receptor by taurine is complex. While acute taurine administration activates the GABAA receptor, chronic taurine feeding promotes the downregulation of the GABAA receptor (L’Amoreaux et al., 2010) and the upregulation of glutamate decarboxylase, the rate-limiting step in GABA biosynthesis (El Idrissi and L’Amoreaux, 2008). Therefore, complex interactions within the GABAeric system, as well as in the glycine and NMDA receptors, largely define the actions of taurine in the CNS.
Regulation of quality control processes
Taurine also regulates quality control processes, such as the ubiquitin-proteasome system and autophagy. These processes either rejuvenate damaged cells and subcellular organelles or eliminate them through degradation or cell death. In taurine deficient cells, a reduction in the activity of the proteasome leads to an accumulation of ubiquitinated proteins, an effect abolished by the mitochondrial specific antioxidant, mitoTEMPO (Jong et al., 2015). Taurine deficiency is also associated with diminished autophagy, a condition that allows damaged cells and organelles to accumulate (Jong et al., 2015). Inactivation of these quality control processes is extremely damaging to cells and tissue. However, excessive autophagy is also damaging because it can elevate cell death. Although there have only been a few studies examining the effect of taurine treatment on autophagy, the actions of taurine are compatible with its cytoprotective activity, as taurine attenuates toxin-mediated autophagy (Li et al., 2012; Bai et al., 2016).
Modulation of Ca2+ homeostasis
Excessive accumulation of Ca2+ by the heart and brain during a myocardial infarction or stroke, is also cytotoxic. Not only does high [Ca2+]i activate proteases and lipases, but it also initiates the mitochondrial permeability transition, an event that permeabilizes the inner mitochondrial membrane and provokes the release of pro-apoptotic factors from the mitochondria that kill the cell (Rasola and Bernadi, 2011, Shetewy et al., 2016). Taurine protects the cell by diminishing Ca2+ overload through three mechanisms (Schaffer et al., 2014b; Prentice et al., 2015). First, the loss of taurine from cells during an ischemia-reperfusion insult appears to be mediated by the taurine transporter, as taurine loss is also accompanied by the loss of Na+ from the cell. Consequently, upon taurine release, less Na+ is available for Ca2+ entry via the Na+/Ca2+ exchanger, which minimizes the degree of Ca2+ overload (Schaffer et al., 2002). Second, taurine indirectly regulates the activity of the sarcoplasmic reticular Ca2+ ATPase, which is responsible for maintaining cytosolic Ca2+ homeostasis through the removal of Ca2+ from the cytosol (Ramila et al., 2015). This action of taurine involves alterations of protein phosphorylation, however, the mechanism by which taurine modulates protein phosphorylation has not been ascertained and warrants further study. Third, taurine treatment is associated with changes in the presence of calbindin D28k, calretinin and parvalbumin (Junyent et al., 2010). Fourth, taurine inhibits glutamateinduced Ca2+ influx through the L-, P/Q- and N-type voltage gated Ca2+ channels, as well as the NMDA receptor channel (Wu et al., 2005).
Osmoregulation
The concentration of taurine within most cells is quite high. In response to elevations in an osmotic load, intracellular taurine levels increase while they decrease in response to hypo-osmotic stress. These are important mechanisms to protect the cell from excessive stretching in response to osmotic imbalances. Because taurine serves as an organic osmolyte, it also modulates the levels of other osmolytes, such as Na+, which not only carries a charge (unlike taurine which is a neutral zwitterion) but is also involved in many important cellular functions, such as transport and membrane potential (Schaffer et al., 2002). In the kidney, taurine serves as a weak diuretic and natriuretic agent, important properties for normal renal function.
TAURINE-MEDIATED PROTECTION AGAINST PATHOLOGY AND DISEASE
Effect of taurine on the central nervous system
Stroke: Stroke, whose incidence is on the rise, is presently a leading cause of death and disability. Under pathological conditions, such as ischemic stroke and hypo-osmotic stress, taurine is released from various cells in the central nervous system (CNS) and function as a neuroprotective agent (Albrecht and Schousboe, 2005). For many brain areas, concentrations of taurine are below 1 mM, which are adequate to activate glycine receptors but not most GABA receptors; the GABA receptors of the ventrobasal thalamus are an exception, as they exhibit especially high affinity for taurine. Taurine plays an important role in neuronal development in the cerebral cortex (Furukawa et al., 2014). Although concentrations of taurine below 1 mM do not activate the GABA receptor in many regions of the CNS, taurine deficiency leads to impaired GABAergic inhibition in the striatum, a condition associated with the development of a disease mimicking hepatic encephalopathy (Sergeeva et al., 2007).
A major cause of cellular damage and death during stroke is the release of large amounts of glutamate, which overstimulate glutamate receptors, resulting in hyper-excitability (Prentice et al., 2015). Among the damaging events associated with glutamate toxicity are calcium overload, oxidative stress, ATP depletion and mitochondrial dysfunction (Abramov and Duchan, 2008; Prentice et al., 2015). The combination of calcium excess and oxidative stress is capable of triggering the mitochondrial permeability transition and the release of pro-apoptotic factors that initiate mitochondrial apoptosis. Apoptosis can also be initiated by caspase 12, CHOP (C/EBP homologous protein) and JNK, which are end products of unfolded protein response (UPR) pathways that arise from ER stress. Interestingly, taurine is also released from brain slices in response to glutamate receptor stimulation (Saransaari and Oja, 2010). The release of taurine helps counteract the adverse effects of glutamate toxicity by reducing [Ca2+]i, increasing the Bcl-2/Bad ratio and suppressing ER stress (Wu and Prentice, 2010). Moreover, taurine reduces cell swelling in rat brain cortical slices subjected to a hypoxic-reoxygenation insult (Ricci et al., 2009). However, it remains to be determined whether taurine-mediated increases in ATP generation and suppression of mitochondrial ROS generation might also contribute to the reductions in the severity of stroke. There is reason to believe that the mitochondrial actions of taurine should reverse stroke-mediated suppression of complex I activity. Taurine might also attenuate another source of ROS in stroke, namely those from NADPH oxidase (Abramov et al., 2007), as taurine treatment is known to reduce the levels of the substrate, NADPH (Schaffer et al., 2016) and mediate the downregulation of Nox2/Nox4 (Han et al., 2016). Using an animal model of focal cerebral ischemia, Gharibani et al. (2015) found that the combination of taurine and a NMDA partial antagonist (5-methyl-N,N-diethylthiolcarbamate-sulfoxide) reduced apoptosis and ER stress while the partial antagonist alone exhibited no neuroprotection.
While there is abundant evidence that taurine is effective in treating stroke in animals (Menzie et al., 2013), only a few studies have addressed its effect on the risk of stroke in humans. In a prospective-case study based on the New York University Women’s Health Study, which examined 14,274 women, no association was observed between serum taurine levels and stroke risk (Wu et al., 2016). However, among non-smokers there may be a link that deserves further consideration, particularly in light of evidence that the incidence of stroke was reduced 90% in a genetic model of stroke (stroke-prone spontaneously hypertensive rats) fed a diet rich in taurine (Yamori et al., 2009). Clearly, clinical studies examining the effectiveness of taurine against stroke in humans is warranted (Fig. 1).
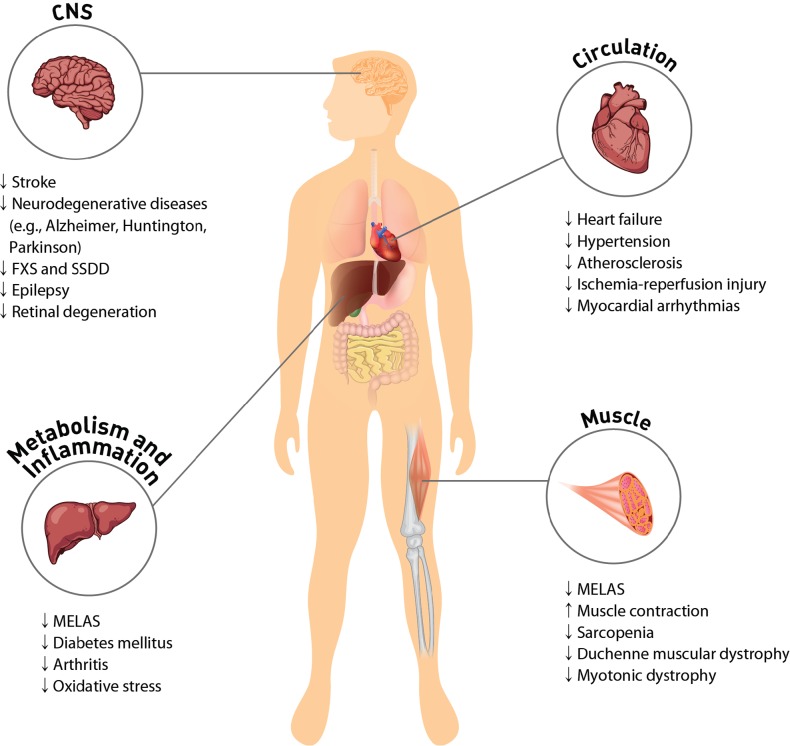
Fig. 1.
Taurine-mediated protection against pathology and disease. High concentrations of taurine in most cells regulate physiological function of excitable tissues and mitochondria. Taurine protects CNS by decreasing ER stress and antagonizing neurotransmitter receptors of GABAA, glycine and NMDA. Protection of the cardiovascular system by taurine occurs through regulation of cell signaling, such as Ca2+ transport, ROS generation and protein phosphorylation. Supplementation of taurine ameliorates symptoms of MELAS and diabetes mellitus. The anti-inflammatory activity of taurine involves either the formation of taurochloramine in neutrophils or the attenuation of nitric oxide and prostaglandin E2 in inflammatory diseases, such as rheumatoid arthritis and osteoarthritis. Taurine depletion or taurine transporter KO leads to cardiac and skeletal muscle dysfunction. Taurine prevents sarcopenia in aged person by minimizing gradual muscle loss. CNS: central nervous system; FXS: fragile X syndrome; SSDD: succinic semialdehyde dehydrogenase deficiency; MELAS: mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes.
Neurodegenerative diseases, such as Alzheimer’s, Huntington’s and Parkinson’s disease: The neurodegenerative diseases share many of the pathological features of stroke caused by glutamate-mediated activation of N-methyl-D-aspartate (NMDA) receptors, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) inotropic glutamate receptors and metabotropic glutamate receptors (Hara and Snyder, 2007). A major cause of cell death from glutamate-mediated hyperexcitability in neurodegenerative diseases appears to involve the collapse of the mitochondrial membrane potential (Abramov and Duchen, 2008). Activation of glutamate receptors leads to calcium overload and an increase in ROS production. Because mitochondrial function is adversely affected by neurodegeneration, one would expect that mitochondrial ROS generation should be a major cause of cellular damage. Indeed, respiratory chain defects have been detected in degenerative diseases, although the site of the defect is disease specific, with Parkinson’s disease affecting complex I, Huntington’s disease affecting complex II and Alzheimer’s disease affecting complex IV (Damiano et al., 2010; Alleyne et al., 2011). Interestingly, one of the animal models of Parkinson’s disease is generated by treating rodents with the complex I inhibitor, rotenone (Cannon et al., 2009). Taurine deficiency and rotenone actions are similar, as both lead to reductions in complex I activity, inhibition of NADH dehydrogenase activity, reductions in respiratory activity and elevations in NADH. Because a primary physiological function of taurine is the maintenance of complex I activity, there is reason to believe that taurine therapy should reduce the severity of Parkinson’s disease (Alkholifi et al., 2015). Indeed, it has recently been reported that reduced plasma taurine content is associated with motor severity in Parkinson’s disease (Zhang et al., 2016). Moreover, a single blind, randomized, controlled study of 47 patients with Parkinson’s disease revealed effectiveness in reducing excessive sleepiness upon treatment with the taurine analog, homotaurine (Ricciardi et al., 2015).
Fragile X Syndrome and succinic semialdehyde dehydrogenase deficiency: Fragile X syndrome is a genetic disease characterized by behavioral disorders and moderate to severe intellectual disabilities. The learning deficit (memory retention) of the fragile X mouse responds favorably to chronic oral administration of taurine (0.05% w/v) for 4 weeks, an effect seemingly linked to taurine’s GABAergic activity (Neuwirth et al., 2015).
Succinic semialdehyde dehydrogenase (SSADH) deficiency is a rare autosomal genetic disease involving a key enzyme in GABA catabolism. Patients with SSADH deficiency showed symptoms such as ataxia, hypotonia, language deficits, and intellectual disability. Nearly half of the SSADH patients suffer from seizures. Because the disease is associated with disruption of GABA homeostasis, the effect of taurine therapy on symptoms of the disease before and after treatment have been examined. In a single case study of a 2-year old boy, taurine therapy (200 mg/kg/day) for 12 months improved social behavior, coordination and activity (Saronwala et al., 2008). However, in a subsequent open-label study of 18 SSADH deficient subjects administered taurine (50-200 mg/kg/day) for a period up to one year there was no major improvement in adaptive behavior with taurine (Pearl et al., 2014). Clearly, a controlled, randomized double-blind study of a larger number of patients receiving either placebo or taurine therapy is warranted.
Epilepsy: Imbalances between excitatory and inhibitory neurotransmitters underlie the mechanism of seizures. Taurine is an abundant amino acid in the brain, where it serves as an inhibitory neuromodulator (Oja and Saransaari, 2013). Although taurine levels can suppress specific types of seizures, taurine deficiency is not required for initiation of seizures. In animal studies, taurine administration has been found to abolish seizures evoked by a wide range of stimulants, including [D-Ala,Met ]-enkphalinamide, opioids, kainite, isoniazid, picrotoxin, penicillin and hypoxia. Nonetheless, clinical trials examining the effect of taurine treatment on human epilepsy have been mixed, with only about 1/3 of the patients responding favorably to taurine therapy (Barbeau and Donaldson, 1974; Bergamini et al., 1974; Konig et al., 1977; Rumpl et al., 1977; Mantovani and DeVivo 1979; Airaksinen et al., 1980).
The dominant inhibitory neurotransmitter in the brain is GABA, therefore, regulation of neuroexcitability by GABA plays a prominent role in preventing neuronal hyperexcitability and seizures (L’Amoreaux et al., 2010). Taurine serves as an agonist of the GABAA receptor, an action that enhances chloride influx into postsynaptic neurons, which causes hyperpolarization that inhibits hyperexcitability. Suppression of kainic acid, isoniazid and picrotoxin-mediated seizures has been attributed to the actions of taurine on the GABAeric system (El Idrissi et al., 2003; El Idrissi and L’Amoreaux, 2008; Junyent et al., 2009; L’Amoreaux et al., 2010). Like GABA, glycine is a major inhibitory neurotransmitter that activates chloride conductance and hyperpolarizes neurons. The inhibitory neuromodulator activity of taurine also extends to the glycine receptor, as the binding of taurine to the glycine receptor evokes chloride current and suppresses neuronal firing (Wu et al., 2008).
Retinal degeneration: The landmark studies showing that taurine is an essential nutrient for cats focused on the link between taurine deficiency and the development of photoreceptor loss and retinal degeneration (Schmidt et al., 1976; Hayes et al., 1975). More recently, Hadj-Said et al. (2016) found that taurine deficiency also causes nuclear ganglion cell degeneration and loss. Moreover, the anti-epileptic agent, vigabatrin, can provoke retinal degeneration, including both photoreceptor and ganglion cell loss, accompanied by taurine deficiency (Heim and Gidal, 2012; Froger et al., 2014). In two patients with succinic semialdehyde dehydrogenase deficiency exhibiting abnormal ERGs at baseline and after 6 months of vigabatrin treatment, taurine therapy partially prevents retinal damage triggered by vigabatrin treatment (Horvath et al., 2016). Because taurine is required for normal retinal ganglion cell survival (Froger et al., 2012), it has been proposed that taurine therapy may serve an important role in the prevention of retinal degeneration (Froger et al., 2014).
Effect of taurine on the cardiovascular system
Congestive heart failure: Taurine has been approved for the treatment of congestive heart failure in Japan (Azuma et al., 1992). Like other heart failure medication, taurine not only diminishes the common symptoms of congestive heart failure (breathlessness on exertion and edema) but also eliminates or decreases the need for administering other heart failure medication, such as digoxin (Azuma et al., 1992). Although taurine exerts a mild positive inotropic effect on the hypodynamic heart and promotes natriuresis and diuresis, the major therapeutic effect of chronic taurine administration appears to involve a reduction in the actions of norepinephrine and angiotensin II, which are known to decrease myocardial performance through elevations in afterload pressure, ventricular remodeling and fluid remodeling (Ito et al., 2014b). Taurine is effective in reducing the adverse actions of norepinephrine through its ability to both decrease catecholamine overflow (through alterations in Ca2+ transport) and diminish cell signaling (through changes in Ca2+ transport, ROS content and protein phosphorylation). Although recent studies have shown that taurine therapy improves exercise capacity of patients with heart failure (Ahmadian et al., 2017), it remains to be determined whether taurine supplementation also reduces the risk of developing overt heart failure in the general population. Moreover, the possibility that taurine supplementation might lower the mortality rate of heart failure patients has not been examined. There is reason to believe that taurine might prolong lifespan of heart failure patients because it elevates high energy phosphate content of the heart, which is an important determinant of mortality among patients suffering from congestive heart failure (Schaffer et al., 2016).
Hypertension: Supplementation with taurine prevents the development of hypertension in several animal models (Fujita and Sato, 1986; Ideishi et al., 1994; Dawson et al., 2000; Harada et al., 2004; Hagar et al., 2006; Hu et al., 2009). In those models, taurine-mediated reductions in blood pressure appear to be mediated by a combination of diminished [Ca2+]i, oxidative stress, sympathetic activity and inflammatory activity, as well as an improvement in renal function (Sato et al., 1991; Dawson et al., 2000; Harada et al., 2004; Hagar et al., 2006; Mozaffari et al., 2006; Hu et al., 2009; Han and Chesney, 2012; Maia et al., 2014; Katakawa et al., 2016).
Two recent clinical studies support the view that taurine therapy reduces blood pressure in hypertensive subjects (Katakawa et al., 2016; Sun et al., 2016). Katakawa attributed the beneficial effects of taurine therapy against hypertension in humans to improved endothelial function secondary to a decline in oxidative stress. By comparison, Sun et al. (2016) focused on the vasodilatory effects of pre-hypertensive patients receiving taurine therapy. The most important feature of the study reported by Sun et al. (2016) was its size and design, as it is a single-center, double-blind, randomized, placebo-controlled trial of 120 pre-hypertensive subjects aged 18–75 with systolic pressure varying from 120–139 mm Hg and diastolic pressure from 80–89 mm Hg. After administration of taurine (1.6 g/day) for 12 weeks, systolic pressure of the pre-hypertensive subjects was reduced 7.2 mm Hg and diastolic pressure fell 4.7 mm Hg while pre-hypertensive subjects treated with placebo exhibited no significant reduction in blood pressure over the same treatment period (Sun et al., 2016). The taurine effect was greater in pre-hypertensive subjects with higher blood pressure than with the subjects with lower blood pressure at the time of initial taurine administration. The administration of taurine resulted in a 1.5-fold increase in plasma taurine concentration, an effect correlated with the improvement in blood pressure. This observation was consistent with an earlier epidemiological study by Yamori et al. (2010), who found that individuals with elevated amounts of taurine intake exhibit lower blood pressure values than individuals consuming less taurine. Moreover, Ogawa et al. (1985) had previously observed that plasma taurine content is reduced in essential hypertension. The human studies are consistent with animal studies showing that taurine deficiency accelerates the onset of hypertension in rats lacking a kidney and maintained on a high salt diet (Mozaffari et al., 2006). Moreover, a negative correlation has been detected between plasma taurine content and blood pressure in spontaneously hypertensive rats (Nara et al., 1978). In their clinical study, Sun et al. (2016) attributed the taurine-mediated reduction in blood pressure to improved flow- and nitroglycerin-mediated dilation, a change not observed in the placebo treated group. In addition to elevating plasma taurine content, the taurine treated group exhibited elevated H2S content, the latter that promotes hypotension by inhibiting transient receptor potential channel 3 (TRPC3)-induced signaling in the vasculature. Further studies are warranted to compare the importance of H2S content on blood pressure control relative to the other common regulators of vascular function, such as Ca2+, neurohumoral factors and nitric oxide.
Atherosclerosis: Atherosclerosis is the primary cause of pathology in stroke, myocardial infarction and peripheral artery disease. The process of atherosclerosis is complex, involving multiple factors and steps (Moore and Tabas, 2011). One of the key steps in the initiation of atherosclerosis is the uptake of the cholesterol-enriched lipoprotein, LDL, by the intima of the arterial wall. Also recruited in the arterial wall are monocytes, which normally exhibit little adhesion to endothelial cells and are not readily accumulated by smooth muscle. However, upon exposure to inflammatory factors and chemoattractants (chemokines), monocytes begin to adhere to endothelial cells, where they are taken up by the intima of the arterial wall. Upon exposure to factors, such as macrophage-colony factor, most of the monocytes in the early atheromata are differentiated into macrophages. Within the intima, LDL can undergo oxidation and glycation, which individually enhance the uptake of the lipoprotein by macrophages. While the uptake and handling of LDL by macrophages is considered an early event in the formation of foam cells, it is now apparent that there are multiple pathways involved in foam cell formation and atherosclerosis (Moore and Tabas, 2011).
Taurine treatment diminishes atherogenesis through several possible mechanisms. First, in most, but not all studies, taurine supplementation accelerates the regression in serum cholesterol levels in atherogenic animals (Petty et al., 1990; Murakami et al., 1996, 2010). During the regression period, hepatic cholesterol levels fall more rapidly in taurine treated animals, largely because of an increase in 7α-hydroxylase activity, which accelerates the degradation of cholesterol. Because there is a correlation between lower serum cholesterol and the dose of taurine used during treatment, the upregulation of the CYP7A1 gene in the liver is thought to regulate serum cholesterol levels (Yokogoshi et al., 1999; Lam et al., 2006; Murakami et al., 2010). At the same time, taurine treatment is associated with diminished activity of 3-hydroxy-3-methylglutaryl CoA reductase, the rate-limiting step of cholesterol biosynthesis (Bellentani et al., 1987). Second, exposure of hepatic cells for 24 hrs with medium containing taurine leads to a decline in the biosynthesis of cholesterol esters and triglycerides. Because the hepatic content of triglycerides and cholesterol esters is a determinant of lipoprotein assembly in the endoplasmic reticulum of the liver, taurine specifically decreases the assembly and secretion of lipoproteins containing the structural protein, apolipoprotein B100 (Yanagita et al., 2008; Murakami et al., 2010). Apolipoprotein B100 is the primary structural protein of both LDL and its precursor, VLDL. Third, taurine protects endothelial cells of vascular tissue from glucose-induced and oxidized LDL-induced toxicity, which is an early step in the development of atherosclerosis (Ulrich-Merzenich et al., 2007). It has also been suggested that taurine protects endothelial cells from homocysteine-induced ER stress and apoptosis by reducing hyperhomocysteinemia (Zulli et al., 2009). While oxidative stress is a primary adverse response to hyperglycemia and homocysteinemia, oxidized LDL-induced toxicity has been attributed to the accumulation of asymmetric dimethylarginine, an inhibitor of nitric oxide synthase (Tan et al., 2007). Fourth, taurine suppresses platelet-derived growth factor-BB (PDGF-BB) induced vascular smooth muscle cell proliferation, which plays an important role in atherosclerosis (Yoshimura et al., 2005). Taurine appears to alter the activity of a phosphatase that dephosphorylates the PDGF-β receptor, which is a potent chemoattractant and proliferative factor for vascular smooth muscle cells. However, taurine does not suppress phorbol ester-induced activation of the Raf/MEK/ERK pathway of vascular smooth muscle proliferation, leading to the suggestion that taurine’s actions are specific for the PDGF-β pathway. Nonetheless, the field remains controversial. According to Terashima et al. (2003), taurine suppresses mesenchymal cell proliferation by inhibiting ERK activity and immediate early gene expression. Clearly, studies clarifying the effect of taurine on smooth muscle cell proliferation are warranted. Fifth, taurine diminishes the expression of LOX-1, which mediates the uptake of oxidized LDL by endothelial cells and decreases the rate of stenosis in oxidatively stressed rabbits subjected to balloon injury of the iliac artery (Gokce et al., 2011). The authors also recognized the important role of taurine-mediated attenuation of oxidative stress in their findings. Finally, the possibility that taurine-mediated inhibition of atherosclerosis may involve its anti-inflammatory activity deserves consideration.
The epidemiological WHO-CARDIAC study showed that dietary taurine intake is correlated with reduced mortality of ischemic heart disease patients (Yamori et al., 2001). In support of that study, Elvevoll et al. (2008) found that taurine enhances the beneficial effects of n-3 fatty acid supplementation on total cholesterol, LDL cholesterol and triglycerides. Recently, Katakawa et al. (2016) attributed the diminished risk of atherogenesis in humans maintained on a diet supplemented with taurine and magnesium to reductions in oxidative stress and improvement in endothelial function. These studies have revealed the importance of taurine supplementation in human health.
Ischemia-reperfusion injury: Several actions of taurine (antioxidant, modulation of [Ca2+]i. osmoregulation, protein phosphorylation and high energy phosphate regulation) alter the outcome of an ischemia-reperfusion insult. A discussion of the differing effects of these factors in various models of ischemia-reperfusion injury and of the importance of taurine treatment and loss during an ischemia-reperfusion insult, appeared in a recent review (Schaffer et al., 2014b). Because of variable effects, it is unlikely that taurine would be adopted as an acute cardioprotective agent to diminish infarct size and minimize injury during a myocardial infarction. Rather, taurine’s use may be restricted to cardiac transplantation and bypass surgery. Several investigators have reported a benefit of taurine as a component of cardioplegic solutions (Oriyanhan et al., 2005) or of loading hearts with taurine prior to their use as donor hearts (Venturini et al., 2009; Sahin et al., 2011). Besides reducing oxidative stress and swelling, taurine loss during an ischemia-reperfusion insult leads to a decrease in [Na+]i, which not only diminishes osmotic stress but also Ca2+ overload (Schaffer et al., 2002; Modi and Suleiman, 2004). In addition, rapid intravenous infusion of taurine before bypass surgery protects against oxidative stress and cellular necrosis (Milei et al., 1992).
Myocardial arrhythmias: One of the earliest reported cardiovascular actions of taurine is its antiarrhythmic actions against a broad range of pro-arrhythmic agents (digoxin or related cardiac glycoside, epinephrine, ouabain, CsCl, hypokalemia)(Read and Welty, 1963; Chazov et al., 1974). This effect is likely related to the modulation of [K+]i, [Na+]i and [Ca2+]i. According to a clinical report, oral administration of taurine and L-arginine dramatically reduced cardiac arrhythmias of three subjects. Thus, under appropriate conditions, taurine is a very effective antiarrhythmic agent (Eby and Halcomb, 2006). Nonetheless, taurine is presently not used in the treatment of cardiac arrhythmias.
Role of taurine in metabolic diseases
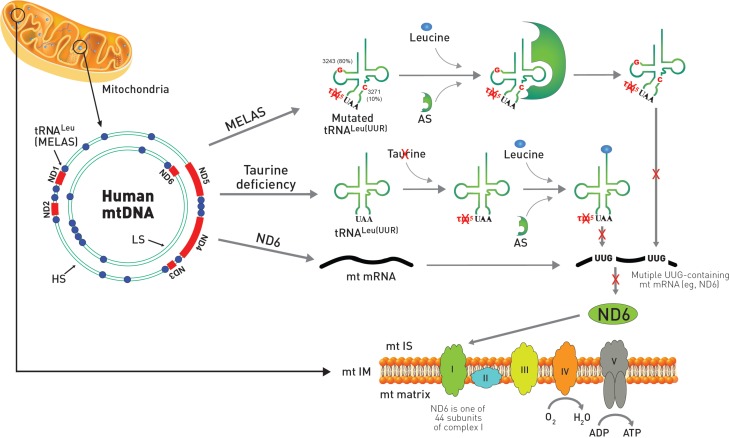
Mitochondrial disease, MELAS: A remarkable similarity exists between the symptoms of taurine deficiency and that of the mitochondrial disease, MELAS (Schaffer et al., 2013). Indeed, the characteristic symptoms of MELAS (myopathy, encephalopathy, lactic acidosis and stroke-like episodes) are also present in taurine deficiency. This is not surprising, as the pathophysiology of the two conditions is similar. MELAS is caused by specific point mutations in the region of DNA that codes for tRNALeu(UUR) (Schaffer et al., 2014a). The mutations appear to alter the structure of the tRNA, preventing the conjugation of taurine with the uridine base of tRNALeu(UUR) (Kirino et al., 2005). Modification of the uridine base alters the interaction of the UUG codon with the AAU anticodon of tRNALeu(UUR), thereby altering UUG decoding (Kurata et al., 2003). Taurine deficiency also appears to reduce the formation of the taurine conjugate, 5-taurinomethyluridine-tRNALeu(UUR), but the effect is related to a reduction in mitochondrial taurine content (Schaffer et al., 2014a). Taurine deficiency reduces the expression of UUG-dependent proteins, with one of the most UUG-dependent mitochondria encoded protein being ND6, a subunit of complex I. Because ND6 plays a prominent role in the assembly of complex I, taurine-mediated reductions in ND6 levels lead to several features seen in MELAS, including lactic acidosis, reduced complex I activity and diminished oxygen consumption. Impaired respiratory chain function causes elevations in superoxide generation and reduced ATP generation, effects that likely play a central role in the development of the myopathy and encephalopathy of MELAS (Fig. 2). Seizures, which are another symptom of MELAS, could depend on the inhibitory neuromodulatory activity of taurine although the mitochondrial activity of taurine might also be a determinant of seizures.
Fig. 2.
Comparison of MELAS and taurine deficiency in mitochondria. The mitochondrial disease, MELAS, is caused by specific point mutations in mitochondrial DNA (mtDNA) that codes for tRNALeu(UUR). Most of the point mutations of MELAS with 80% frequency occur at A3243G while mutations at T3271C exist with 10% frequency. In mtDNA, ND genes are shown in red color and tRNA genes are depicted as blue circles. The gene of tRNALeu(UUR) responsible for MELAS is located adjacent to ND1. The mutation in MELAS alters the structure of the tRNALeu(UUR) preventing the conjugation of taurine with the uridine base of the UAA anti-codon from forming 5-taurinomethyluridine (τm5U). MELAS patients also show reduced aminoacylation of taurine deficient tRNALeu(UUR) by leucine catalyzed by aminoacyl-tRNA synthetase (AS). Both reduced aminoacylation of tRNALeu(UUR) by leucine and formation of the taurine conjugate of τm5UAA-tRNALeu(UUR) prevent decoding of mitochondrial UUG-dependent proteins, including ND6, which is one of 44 protein subunits of complex I of the electron transport chain located in the mitochondria inner membrane. On the other hand, taurine deficiency has normal aminoacylation of tRNALeu(UUR) by leucine, but exhibits reduced formation of the taurine conjugate of τm5UAA-tRNALeu(UUR), which also prevents decoding of mitochondrial ND6 mRNA, resulting in increased superoxide generation and reduced ATP generation. MELAS: mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes; ND6: NADH-ubiquinone oxidoreductase chain 6; mt IS: motochodrial intermembrane space; mtIM: mitochondrial inner membrane; LS: light strand; HS: heavy strand.
Because of the direct link between taurine content and the development of MELAS, it is not surprising that patients with MELAS respond favorably to taurine therapy (Rikimaru et al., 2012). In one such case study, laboratory tests revealed that a 29-year-old woman presented at the hospital with stroke-like episodes had a MELAS-like A3243G point mutation in a left biceps brachii muscle biopsy. Anticonvulsants, both phenytoin and valproate, were administered over the next 7 months but failed to halt the epileptic and stroke-like episodes. Shortly after beginning oral taurine supplementation, which increased blood taurine content about 5-fold, the epileptic and stroke-like episodes completely ceased. Also corrected by taurine therapy was lactic acidosis. In a related case study, a 21-year-old male was admitted to a hospital with right homonymous hemianopsia. He was diagnosed with A3243G-linked MELAS and lactic acidosis. Anticonvulsion therapy was used over the next several years, but the patient still experienced several stroke-like episodes, including sensory aphasia and visual impairment. After taurine supplementation, which elevated blood levels approximately 10-fold, the stroke-like episodes ceased. The same group examined the effect of taurine treatment on cytoplasmic hybrids (cybrids) harboring a 3243G-MELAS mutation; incubation of the cybrids for 4 days with medium containing 40 mM taurine partially normalized oxygen consumption and mitochondrial membrane potential, which were suppressed in the A3243G containing cybrids. Taurine treatment also attenuated the degree of oxidative stress in cybrids harboring the 3243 mutant. Thus, taurine therapy not only abolishes stroke-like episodes in MELAS patients but also restores normal mitochondrial respiratory function. These data reveal promise for the use of taurine as a therapeutic agent against MELAS.
Diabetes mellitus: Diabetes mellitus is a disease characterized by elevated blood glucose and either a decrease in plasma insulin (type 1 diabetes) or resistance to the actions of insulin (type 2 diabetes). Type 1 diabetes is an autoimmune disease that is caused by T-cell-mediated destruction of pancreatic β-cells that dramatically reduces insulin biosynthesis. Thus, the disease is characterized by hypoinsulinemia and hyperglycemia. By contrast, type 2 diabetes is a progressive disorder characterized initially by insulin resistance, in which the actions of insulin at target organs are reduced, although in later stages of the disease insulin secretion is also impaired. Diabetes is associated with multiple complications, including cardiovascular disease, neuropathy, nephropathy, retinopathy, foot ulcers, skin lesions and hearing impairment. Although restrictions in the degree of hyperglycemia are central to the control of these complications, several downstream factors can contribute to the severity of the complications. One of the most important of these factors is ROS. According to seminal studies by Brownlee (2005), it was proposed that glucose oxidation, which involves pyruvate metabolism by the mitochondria, produces ROS, which in turn enhances flux through several damaging pathways (advanced glycosylation end product generation, protein kinase C activation, polyol formation and hexosamine pathway stimulation) involved in the development of diabetic complications.
Two important concepts arose from the work of Brownlee (2005). First, his unifying hypothesis provided a logical link between glucose-mediated oxidative damage and several pathways implicated in diabetic complications. Moreover, it introduced the insightful idea that mitochondria-derived ROS play an important role in the development of certain diabetic complications. However, it did not take into consideration the effect of diabetes-mediated oxidative stress on mitochondrial function itself (Higgins and Coughlan, 2014; Malik et al., 2015; Coughlan et al., 2016; Czajka and Malik, 2016).
The major source of ROS in most diabetic tissues is complexes I and III of the respiratory chain. Interestingly, ROS generated by complex I remain within the matrix of the mitochondria while complex III-derived ROS are distributed between the matrix and extra-mitochondrial locations. In the matrix, ROS provokes damage, as evidenced by decreased activity of the oxidant-sensitive enzyme, aconitase, increased mitochondrial DNA oxidation and if severe enough, death via apoptosis (Ricci et al., 2008; Lindblom et al., 2015). Although complex III-mediated ROS generation can affect targets outside of the mitochondria, the intra-mitochondrial effects appear most important in the development of diabetic complications. In leptin deficient, obese, type 2 diabetic db/db mice, elevations in mitochondrial ROS production of the heart lead to reduced ATP production, as respiratory function and energy metabolism are impaired, which in turn diminishes myocardial performance (Boudini et al., 2007). Respiration and ATP generation are also defective in diabetic renal mesangial and tubular cells. Because ATP is required for reabsorption in the proximal tubules while energy deficiency leads to renal failure, hyperglycemia-mediated mitochondrial damage and ATP deficiency appear to play a prominent roles in the development of the diabetic nephropathy (Higgins and Coughlan, 2014; Czajka and Malik, 2016; Hallan and Sharma, 2016).
Mitochondrial DNA damage, which has been detected in humans suffering from diabetes melllitus (Malik et al., 2015), invariably alters the expression of mitochondria encoded proteins, leading to diminished respiratory chain activity. As a result, more ROS is produced, which triggers further mitochondrial DNA damage, ultimately leading to a vicious cycle of damage. Evidence of severe mitochondrial structural changes and dysfunction (morphology, biogenesis, respiratory chain function, fatty acid and citric acid cycle metabolism, oxidative stress, apoptosis and uncoupling activity) support a role of mitochondrial damage in the development of diabetic complications (Sivitz and Yorek, 2010; Aon et al., 2015; Lindblom et al., 2015; Hallan and Sharma, 2016).
Plasma and platelet taurine levels are reduced in subjects with type 1 diabetes (Franconi et al., 1995). Based on 711 overweight, diabetic subjects, plasma taurine levels are related to the decline in insulin sensitivity (Zheng et al., 2016). In fact, most of the major targets of diabetes (kidney, retina and neurons) undergo hyperglycemic-mediated reductions in taurine content (Schaffer et al., 2009). Nonetheless, at an early age, the metabolic pattern of subjects with diabetes is very different from that of the taurine deficient mouse (Ito et al., 2015b; Schaffer et al., 2016). The initial defect in type 1 diabetes is insulinopenia, which reduces glucose metabolism and elevates fatty acid metabolism, while taurine deficient mice exhibit impaired respiratory chain function, increased glycolysis and decreased glucose and fatty acid oxidation (Schaffer et al., 2016). Nonetheless, taurine is required for normal β-cell viability, therefore, the number of pancreatic β-cells is reduced in 12-month-old TauTKO mice (Ito et al., 2015b). Moreover, the properties of the diabetic animal are altered with age, as mitochondrial oxidative injury leads to impaired respiratory chain function in the late phases of the disease. Also affecting mitochondrial function and biogenesis is diet and physical activity (Santos et al., 2014). Therefore, upon aging and dietary modification, the diabetic and taurine phenotypes adopt similar features. In this regard it is interesting that Han et al. (2015) claim that taurine deficiency is a necessary requirement for the development of an appropriate model of diabetic nephropathy.
There is overwhelming evidence that taurine therapy reduces pathology associated with diabetes, obesity and the metabolic syndrome (Schaffer et al., 2009; Ito et al., 2012; Imai et al., 2014; Murakami, 2015; Chen et al., 2016). In many animal studies, particularly of type II diabetes, taurine treatment diminishes the degree of hypoglycemia, an effect that in turn attenuates diabetic complications (Nakaya et al., 2000; Winiarska et al., 2009; Das and Sil, 2012; Kim et al., 2012; Chiang et al., 2014; Koh et al., 2014). Several mechanisms may contribute to the regulation of hyperglycemia in diabetic animals treated with taurine. First, taurine improves respiratory function and increases ATP production, effects that should improve pancreatic β-cell function and insulin secretion (Sivitz and Yorek, 2010; Schaffer et al., 2016). Second, hyperglycemia and lipidemia are associated with elevations in mitochondrial ROS generation. In pancreatic β-cells, fatty acid-mediated ROS generation appears to decrease insulin secretion, an effect attenuated by taurine treatment (Oprescu et al., 2007). Third, mitochondrial dysfunction can provoke insulin resistance (Sivitz and Yorek, 2010). Haber et al. (2003) found that taurine treatment prevents hyperglycemia-induced insulin resistance and oxidative stress. Together, these findings indicate that taurine protects against type 2 diabetes-mediated complications, but the mechanism by which taurine diminishes the development of the complications of type 2 diabetes remain unclear, largely because it is virtually impossible to separate the mitochondrial actions of taurine from its effects on insulin secretion and action.
On the other hand, in the streptozotocin-induced model of type 1 diabetes, plasma glucose levels remain unaltered by taurine treatment while the severity of the diabetic complications are diminished. Because diabetic status in the streptozotocin model of type 1 diabetes is unaffected by taurine, one can readily establish the mechanism underlying taurine’s effectiveness against the development of diabetic complications. According to several investigators, taurine-mediated reductions in the severity of type 2 diabetic complications are more closely linked to improvements in cellular stresses (ER, oxidative and inflammatory) and mitochondrial dysfunction (Schaffer et al., 2009; Ito et al., 2012; Imai et al., 2014). Trachtman et al. (1995) were the first group to recognize the benefit of taurine treatment against the development of diabetic complications. They reported that male rats administered streptozotocin developed a diabetic nephropathy characterized by elevated glomerular filtration rate, glomerular hypertrophy and proteinuria and albuminuria. Administration of taurine (1% in the drinking water) reduced proteinuria by 50% and dramatically suppress glomerular hypertrophy and tubointerstitial fibrosis without affecting blood glucose. Because the amino acid also abolished the elevation in renal cortical malondialdehyde, a marker of oxidative stress, and of advanced glycoloxidation products, a marker of advanced glycosylation end products, the protective effects of taurine were attributed to suppression of oxidative stress and advanced glycosylation. Recently, the effectiveness of taurine therapy against the development of diabetic nephropathy has been confirmed (Pandya et al., 2013; Koh et al., 2014). In a related study, Ikubo et al. (2011) found that streptozotocin-treated diabetic rats developed vascular defects that were associated with oxidative stress without a change in blood glucose. Interestingly, taurine also protects against apoptosis in cellular models of glucose toxicity (Ulrich-Merzenich et al., 2007).
Obesity is a disorder characterized by insulin resistance, hyperlipidemia, hyperglycemia and inflammatory responses related to enlarged adipocytes. Taurine has been effective in decreasing body weight of obese animals and in suppressing inflammatory responses. These effects and their mechanism are not reviewed here but are covered in a complete review by Murakami (2015).
Role of taurine in inflammatory diseases: Arthritis is a term used to describe over 100 diseases, the most common being rheumatoid arthritis and osteoarthritis. The major symptoms of arthritis are joint stiffness and pain with inflammation of the joints, the tissues surrounding the joint and connective tissue. Rheumatoid arthritis is characterized by synovial inflammation and proliferation, bone erosions and thinning of articular cartilage.
The distinctive features of the acute inflammatory phase, which can last up to several days, are vascular dilation, microvascular leakage and leukocyte recruitment, the latter mediated by adhesion factors that facilitate the interaction of leukocytes with activated endothelium. Acute inflammation, which is triggered by damaged or diseased tissue, as well as irritants and pathogens, is an early phase in the repair of damage and the removal of microorganisms and harmful agents by the innate immune system. During acute inflammation, leukocytes adhere to the endothelium prior to transmigrating across the endothelium into the interstitium. Chemotactic factors and cytokines recruit the neutrophils to the site of inflammation. Activation of the leukocytes, in particular neutrophils and mononuclear phagocytes, leads to the secretion of a host of proinflammatory mediators, including microbicidal peptides, cationic microbial proteins, lytic enzymes, ROS and lysosomal granule constituents. When these proinflammatory mediators are released into phagolysosomes, they destroy engulfed microbes and other pathogens. However, when they are released into the extracellular millieu, they can cause tissue damage. In rheumatoid arthritis, activation of the inflammatory process is part of the autoimmune disorder contributing to joint deformation, erosion of bone and disruption of the cartilagebone interface.
The content of taurine in the neutrophil is high, representing about 50% of the total free amino acid pool. The two primary functions of taurine in the neutrophil are anti-inflammatory and antioxidant actions. ROS are produced by the neutrophil as a weapon to kill pathogens, with one of those ROS being hypochlorous acid (HOCl). Myeloperoxidase-catalyzes the formation of taurine chloramine (TauCl) from taurine and HOCl. Because TauCl is a less potent oxidant than HOCl, the neutralization of HOCl represents one of the important antioxidant mechanisms of taurine. The myelperoxidase-catalyzed reaction is also responsible for the anti-inflammatory activity of taurine, as TauCl inhibits the production of proinflammatory cytokines (Marcinkiewicz et al., 1995; Park et al., 1997; Barua et al., 2001), attenuates elevations in nitric oxide and prostaglandin E2 (Park et al., 2000; Chorazy-Massalska et al., 2004; Kim et al., 2007), decreases the activity of matrix metalloproteinases and initiates leukocyte apoptosis to terminate acute inflammation (Klamt and Shacter, 2005). For a detailed discussion of the anti-inflammatory and anti-arthritic actions of taurine please refer to the extensive review by Marcinkiewicz and Kontny (2014).
Effect of taurine on muscle
Modulation of muscle contraction: Taurine deficiency leads to impaired contractile function of both cardiac and skeletal muscle (Cuisinier et al., 2000). Hamilton et al. (2006) found that exposure to the taurine transport inhibitor, guanidinoethanesulfonate, led to a 60% reduction in extensor digitorum longus taurine content, which was associated with a decrease in peak force contraction. More severe muscular dysfunction is seen in taurine transporter knockout mice, whose muscle taurine content is reduced by over 90%, which leads to a decline in muscle mass and muscular dysfunction (Warskulat et al., 2004; Ito et al., 2008; Ito et al., 2014b). Also associated with severe taurine deficiency are histological changes, including disruption of the myofibrils.
There are abundant reports that taurine administration enhances exercise performance of both humans (Ishikura et al., 2011; Balshaw et al., 2013; Ra et al., 2013, 2016) and animals (Goodman et al., 1985; Dawson et al., 2002; Yatabe et al., 2003; Sugiura et al., 2013; da Silva et al., 2014). Besides improving contractile function of rodents, taurine administration was found to increase the time until exhaustion, reduce exercise-induced fatigue and diminish damage from intense exercise. When taurine is administered prior to heavy exercise, the levels of pro-inflammatory factors are reduced and exercising muscle is protected (Kato et al., 2015). Also noteworthy is a trial of 36 male subjects that were administered either a branched-chain amino acid supplement, a taurine supplement, placebo or the combination of taurine and branched-chain amino acid supplements (Ra et al., 2013). The least muscle damage after eccentric exercise was observed in the combination group, as muscle soreness was diminished two days after exercise and there was less lactate dehydrogenase and aldolase released into the blood. Interestingly, serum levels of 8-hydroxydeoxyguanosine, a measure of DNA oxidative damage, were also reduced in the combination group, implicating the antioxidant activity of taurine in the beneficial effects of the combination supplement. The authors suggested that taurine potentiates the beneficial effect of the branched-chain amino acid supplement. The likely candidates for the protective activity of taurine are suppression of oxidative stress and inflammation. In another study by the same laboratory, the end point of the study was arterial stiffness after eccentric exercise (Ra et al., 2016). Serum malondialdehyde remained elevated for 4 days after exercise in the control placebo group while oxidative stress was decreased in the taurine treated group. There was a parallel between the increase in malondialdehyde and arterial stiffness, which led the authors to suggest that taurine-mediated reductions in oxidative stress helped attenuate the degree of arterial stiffness. For a more complete review of the role of taurine in muscle function and disorders, please refer to DeLuca et al. (2015).
Sarcopenia: Sarcopenia, which is the gradual loss of skeletal muscle tissue related to an imbalance between protein biosynthesis and degradation. Because sarcopenia occurs with aging and leads to physical impairment among the elderly it is a serious health problem. Interestingly, taurine deficiency is associated with a reduction in cell size (Schaffer et al., 1998; Ito et al., 2008). In a recent review, Scicchitano and Sica (2016) raised the possibility that taurine might counteract the adverse effects of sarcopenia. This is an interesting condition that could extend the utilization of taurine to another application.
Duchenne muscular dystrophy: Encouraging results have been reported using taurine therapy for the treatment of mdx mice, a model for Duchenne muscular dystrophy, which is a fatal muscle wasting disease characterized by oxidative stress and inflammation (Terrill et al., 2016b). In humans suffering from the disease, there are no symptoms at birth, however, symptoms in the form of waddling gait and difficulty climbing steps, begin to appear at a young age. The primary cause of the disease in humans are mutations in dystrophin, a cytoskeletal protein that connects the cytoskeleton and the extracellular matrix, while in the mdx mouse model of Duchenne muscular dystrophy the mutations are replaced by inadequate expression of dystrophin.
In the disease, structural changes within the sarcolemma increase the permeability of the cell membrane and facilitate the accumulation of Ca2+, contributing to the development of Ca2+ overload in which the rate of [Ca2+]i rise after depolarization is enhanced, peak [Ca2+]i is increased and the relaxation phase is prolonged (Blake et al., 2002). Because the protease inhibitor, leupeptin, abolishes the exaggerated increase in [Ca2+]i, it has been proposed that proteases contribute to the severity of the disorder. Another factor that contributes to the development of the disorder is a lack of nNOS, which normally produces nitric oxide to ensure adequate vasodilation. Because nitric oxide levels fall in an oxidatively stressed environment, it has been proposed that oxidative stress and acute inflammation may also contribute to pathology of the disease.
Taurine has a potential to disrupt several of the steps in the development of Duchenne’s muscular dystrophy, in part by restoring taurine levels that decline in the muscle disorder (Terrill et al., 2015, 2016b). ln fact, addition of 4% taurine to mouse chow of mdx mice after 14 days of age increases muscle taurine content, an effect correlated with reductions in inflammation (neutrophil infiltration) and mitigation of severe bouts of myocyte necrosis after exercise (Terrill et al., 2016a). According to DeLuca et al. (2003) taurine treatment also significantly enhances forelimb strength of exercising dmx mice. This effect of taurine is largely attributed to an improvement in Ca2+ homeostasis, although taurine supplementation has no effect on the expression of key E-C coupling proteins (Horvath et al., 2016). Creatine treatment is nearly as effective as taurine in enhancing muscle strength, indicating that delivery of more energy to the muscle prevents the decline in muscle function. In this regard, the effect of taurine on improvement in high-energy phosphate metabolism and respiratory chain function may also contribute to its beneficial actions (Schaffer et al., 2016).
Myotonic dystrophy: Myotonia is a condition characterized by delayed relaxation following skeletal muscle contraction. Conte-Camerino et al. (1989) found that acute administration of taurine was effective in attenuating myotonic discharges by 20,25 diazacholesterol-treated rats but not by rats treated with anthracene-9-carboxylic acid. The positive effect of taurine against the 20,25 diazacholesterol model of myotonia was attributed to an improvement in flux through voltage-gated chloride channels of extensor digitorum longus myofibers. It has been suggested that the actions of anthracene-9-carboxylate preclude an effect of taurine. Taurine requires functional chloride channels, but anthracene-9-carboxylate inhibits those channels.
Several clinical trials examining the effect of chronic taurine therapy on the severity of myotonia have been evaluated. In a double-blind, single crossover study of 9 patients with dystrophia myotonica, chronic taurine therapy diminished the severity of myotonia while decreasing potassium-induced hyperexcitability (Durelli et al., 1983). The possibility that taurine might also be effective against sodium channel myotonia and paramyotonia congenita has also been proposed, as taurine modulates sodium transients of the Nav1.4 channel (DeLuca et al., 2015).
Taurine also increases the sensitivity of myofibrils to Ca2+ (Dutka et al., 1985), an effect also seen in the TauTKO heart, where it has been attributed to enhanced phosphorylation of troponin (Ramila et al., 2015). It is also possible that taurine affects muscular activity of the sarcoplasmic reticular Ca2+ pump, an effect in the heart caused by alterations in the phosphorylation state of the regulator protein, phospholamban (Ramila et al., 2015).
CONCLUSION
Taurine, 2-aminoethanesulfonic acid, is an endogenous end metabolite that is distributed in various tissues at high concentration. It is a sulfur-containing amino acid synthesized from cysteine and is excreted without any further metabolism. Since the first discovery of taurine in 1827, many of its functions have been elucidated in experiments focusing on skeletal muscle, the retina and the central nervous and cardiovascular systems. The cytoprotective actions of taurine contribute to the improvement in the clinical and nutritional health of humans through various mechanisms, including antioxidation, energy production, neuromodulation, Ca2+ homeostasis and osmoregulation. The combination of one or more of these cytoprotective effects of taurine act to diminish the pathology and symptoms of a host of diseases ranging from those of the CNS, cardiovascular system, skeletal muscle and defective metabolism. Many CNS-related diseases, including stroke, neurodegeneration, FXS, SSDD and epilepsy, respond favorably to taurine therapy. Taurine treatment also diminishes the severity of inflammatory diseases and abnormalities of the circulatory system, such as heart failure, hypertension, atherosclerosis, ischemia-reperfusion injury, myocardial arrhythmias, diabetes mellitus and arthritis. The phenotype of taurine depletion resembles that of the mitochondrial disease, MELAS, as both conditions affect the activity of the respiratory chain. Also characteristic of the taurine transporter KO mouse is impaired skeletal muscle contraction and sarcopenia. Therefore, taurine therapy is likely to improve cardiac and skeletal muscular dysfunction in conditions, such as MELAS, diminished muscle contraction, sarcopenia, Duchenne muscular dystrophy and myotonic dystrophy. Because taurine is a naturally occurring substance that exhibits few adverse side effects and plays a fundamental role in the function of most mammalian cells, the future of taurine as an effective therapeutic agent and a nutritional supplement is seemingly bright. Although clinical evaluation of taurine has been limited to a few diseases, it has already been approved for use in congestive heart failure. Therefore, taurine is an essential neutraceutical with diverse cytoprotective and therapeutic actions.
Acknowledgments
The year 2017 represents the 190th anniversary since the discovery of taurine by the German scientists, Friedrich Tiedemann and Leopold Gmelin in 1827. This article was prepared in honor of the 190th anniversary of taurine’s discovery. The authors of this article, Stephen Schaffer, president of the International Taurine Society, and Ha Won Kim, president of the Korean Taurine Society, would like to express our appreciation for the support from Dong-A Pharmaceutical Company, Ltd, Korea.
REFERENCES
- Abramov AY, Duchen MR. Mechanisms underlying the loss of mitochondrial membrane potential in glutamate excitotoxicity. Biochim Biophys Acta. 2008;1777:953–964. doi: 10.1016/j.bbabio.2008.04.017. [DOI] [PubMed] [Google Scholar]
- Abramov AY, Scorziello A, Duchen MR. Three distinct mechanisms generate oxygen free radicals in neurons and contribute to cell death during anoxia and reoxygenation. J Neurosci. 2007;27:1129–1138. doi: 10.1523/JNEUROSCI.4468-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmadian M, Roshan D, Ashourpore E. Taurine supplementation improves functional capacity, myocardial oxygen consumption and electrical activity in heart failure. J Diet Suppl. 2017;14:422–432. doi: 10.1080/19390211.2016.1267059. [DOI] [PubMed] [Google Scholar]
- Airaksinen EM, Oja SS, Marnela K-M, Leino E, Paakkonen L. Effects of taurine treatment on epileptic patients. Prog Clin Biol. 1980;39:157–166. [PubMed] [Google Scholar]
- Albrecht J, Schousboe A. Taurine interaction with neurotransmitter receptors in the CNS: an update. Neurochem Res. 2005;30:1615–1621. doi: 10.1007/s11064-005-8986-6. [DOI] [PubMed] [Google Scholar]
- Alkholifi FK, Albers DS. Attenuation of rotenone toxicity in SY5Y cells by taurine and N-acetyl cysteine alone or in combination. Brain Res. 2015;1622:409–413. doi: 10.1016/j.brainres.2015.06.041. [DOI] [PubMed] [Google Scholar]
- Alleyne T, Mohan N, Joseph J, Adogwa A. Unraveling the role of metal ions and low catalytic activity of cytochrome C oxidase in Alzheimer’s disease. J Mol Neurosci. 2011;43:284–289. doi: 10.1007/s12031-010-9436-8. [DOI] [PubMed] [Google Scholar]
- Aon MKA, Tocchetti CG, Bhatt N, Paolocci N, Cortassa S. Protective mechanisms of mitochondria and heart function in diabetes. Antioxid Redox Signal. 2015;22:1563–1586. doi: 10.1089/ars.2014.6123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arrieta F, Balsa JA, de la Puerta C, Botella JI, Zamarron I, Elias E, Del Rio JI, Alonso P, Candela A, Blanco-Colio LM, Eqido J, Navarro P, Vazquez C. Phase IV prospective clinical study to evaluate the effect of taurine on liver function in postsurgical adult patients requiring parenteral nutrition. Nutr Clin Pract. 2014;29:672–680. doi: 10.1177/0884533614533610. [DOI] [PubMed] [Google Scholar]
- Azuma J, Sawamura A, Awata N. Usefulness of taurine in chronic congestive heart failure. Jpn Circ J. 1992;56:95–99. doi: 10.1253/jcj.56.95. [DOI] [PubMed] [Google Scholar]
- Bai J, Yao X, Jiang L, Zhang Q, Guan H, Liu S, Wu W, Qiu T, Gao N, Yang L, Yang G, Sun X. Taurine protects against As2O3-induced autophagy in livers of rat offsprings through PPARY pathway. Sci Rep. 2016;6:27733. doi: 10.1038/srep27733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balshaw TG, Bampouras TM, Barry TJ, Sparks SA. The effect of acute taurine ingestion on 3-km running performance in trained middle-distance runners. Amino Acids. 2013;44:555–561. doi: 10.1007/s00726-012-1372-1. [DOI] [PubMed] [Google Scholar]
- Barbeau A, Donaldson J. Zinc, taurine and epilepsy. Arch Neurol. 1974;30:52–58. doi: 10.1001/archneur.1974.00490310054009. [DOI] [PubMed] [Google Scholar]
- Barua M, Liu Y, Quinn MR. Taurine chloramine inhibits inducible nitric oxide synthase and TNF-alpha gene expression in activated alveolar macrophages decreased NF-KappaB activation and IkappaB kinase activity. J Immunol. 2001;167:2275–2281. doi: 10.4049/jimmunol.167.4.2275. [DOI] [PubMed] [Google Scholar]
- Bellentani S, Pecorari M, Cordonna P, Marchegiano P, Manenti F, Basisio E, Defabiani E, Galli G. Taurine increases bild acid poll size and reduces bile saturation index in the hamster. J Lipid Res. 1987;28:1021–1027. [PubMed] [Google Scholar]
- Bergamini L, Mutani R, Delsedime M, Durelli L. First clinical experience on the antiepileptic action of taurine. Eur Neurol. 1974;11:261–269. doi: 10.1159/000114324. [DOI] [PubMed] [Google Scholar]
- Blake DJ, Weir A, Newey SE, Davies KE. Function and genetics of dystrophin and dystrophin-related proteins in muscle. Physiol Rev. 2002;82:291–329. doi: 10.1152/physrev.00028.2001. [DOI] [PubMed] [Google Scholar]
- Bouckenooghe T, Remacle C, Reusens B. Is taurine a functional nutrient? Curr Opin Clin Nutr Metab Care. 2006;9:728–733. doi: 10.1097/01.mco.0000247469.26414.55. [DOI] [PubMed] [Google Scholar]
- Boudini S, Sena S, Theoblad H, Sheng X, Wright JJ, Hu XX, Aziz S, Johnson JI, Bugger H, Zaha VG, Abel ED. Mitochondrial energetics in the heart in obesity-related diabetes: direct evidence for increased uncoupled respiration and activation of uncoupling proteins. Diabetes. 2007;56:2457–2466. doi: 10.2337/db07-0481. [DOI] [PubMed] [Google Scholar]
- Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54:1615–1625. doi: 10.2337/diabetes.54.6.1615. [DOI] [PubMed] [Google Scholar]
- Cannon JR, Tapias V, Na HM, Honick AS, Drolet RE, Greenamyre JT. A highly reproducible rotenone model of Parkinson’s disease. Neurobiol Dis. 2009;34:279–290. doi: 10.1016/j.nbd.2009.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan CY, Sun HS, Shah SM, Agovic MS, Ho I, Friedman E, Banerjee SP. Direct interaction of taurine with the NMDA glutamate receptor subtype via multiple mechanisms. Adv Exp Med Biol. 2013;775:45–52. doi: 10.1007/978-1-4614-6130-2_4. [DOI] [PubMed] [Google Scholar]
- Chazov EI, Malchikova LS, Lipina NV, Asafov GB, Smirnov VN. Taurine and electrical activity of the heart. Circ. Res. 1974;35(Suppl 3):11–21. [PubMed] [Google Scholar]
- Chen W, Guo J, Zhang Y, Zhang J. The beneficial effects of taurine in preventing metabolic syndrome. Food Funct. 2016;7:1849–1863. doi: 10.1039/C5FO01295C. [DOI] [PubMed] [Google Scholar]
- Chiang STH, Yeh SM, Chen YC, Lin SL, Tseng JK. Investigation of the protective effects of taurine against alloxan-induced diabetic retinal changes via electroretinogram and retinal histology with New Zealand White rabbits. Int J Endocrinol. 2014;2014:631549. doi: 10.1155/2014/631549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chorazy-Massalska M, Kontny E, Kornatka A, Rell-Bakalarska M, Marcinkiewicz J, Maslinski W. The effect of taurine chloramine on pro-inflammatory cytokine production by peripheral blood mononuclear cells isolated from rheuatoid arthritis and osteoarthritis patients. Clin Exp Rheumatol. 2004;22:692–698. [PubMed] [Google Scholar]
- Conte-Camerino D, DeLuca A, Mambrini M, Ferrannini E, Franconi F, Giotta A, Bryant SH. The effects of taurine on pharmacologically induced myotonia. Muscle Nerve. 1989;12:898–904. doi: 10.1002/mus.880121105. [DOI] [PubMed] [Google Scholar]
- Coughlan MT, Higgins GC, Nguyen TV, Penfold SA, Thallas-Bonke V, Tan SM, et al. Deficiency in apoptosis-inducing factor recapitulates chronic kidney disease via aberrant mitochondrial homeostasis. Diabetes. 2016;65:1085–1098. doi: 10.2337/db15-0864. [DOI] [PubMed] [Google Scholar]
- Cuisinier C, Gailly P, Francaux M, Lebacq J. Effects of guanidinoethane sulfonate on contraction of skeletal muscle. Adv Exp Med Biol. 2000;483:403–409. doi: 10.1007/0-306-46838-7_46. [DOI] [PubMed] [Google Scholar]
- Czajka A, Malik AN. Hyperglycemia damage to mitochondrial respiration in renal mesangial and tubular cells: implication for diabetic nephropathy. Redox Biol. 2016;10:100–107. doi: 10.1016/j.redox.2016.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Silva LA, Tromm CB, Bom KF, Mariano I, Po0zzi B, da Rosa GL, Tuon T, da Luz G, Vuolo F, Petronilho F, Cassiano W, De Souza CT, Pinho RA. Effects of taurine supplementation following eccentric exercise in young adults. Appl Physiol Nutr Metab. 2014;39:101–104. doi: 10.1139/apnm-2012-0229. [DOI] [PubMed] [Google Scholar]
- Damiano M, Galvan L, Deglon N, Brouillet E. Mitochondria in Huntington’s disease. Biochim Biophys Acta. 2010;1802:52–61. doi: 10.1016/j.bbadis.2009.07.012. [DOI] [PubMed] [Google Scholar]
- Das J, Sil PC. Taurine ameliorates alloxan-induced renal injury, oxidative stress-related signaling pathways and apoptosis in rats. Amino Acids. 2012;43:1509–1523. doi: 10.1007/s00726-012-1225-y. [DOI] [PubMed] [Google Scholar]
- Dawson R, Jr, Biasetti M, Messina S, Dominy J. The cytoprotective role of taurine in exercise-induced muscle injury. Amino Acids. 2002;22:309–324. doi: 10.1007/s007260200017. [DOI] [PubMed] [Google Scholar]
- Dawson R, Jr, Liu S, Jung B, Messina S, Eppler B. Effects of high salt diets and taurine on the development of hypertension in the stroke-prone spontaneously hypertensive rat. Amino Acids. 2000;19:643–664. doi: 10.1007/s007260070014. [DOI] [PubMed] [Google Scholar]
- DeLuca A, Pierno S, Conte Camerino D. Taurine: the appeal of a safe amino acid for skeletal muscle disorders. J Transl Med. 2015;13:243. doi: 10.1186/s12967-015-0610-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeLuca A, Piernon S, Liantonio A, Cetrone M, Camerion C, Bodvael F, Mirabella M, Servidei S, Ruegg UT, Conte Camerino D. Enhanced dystrophic progression in mdx mice by exercise and beneficial effects of taurine and insulin-like growth factor-1. J Pharmacol Exp Therap. 2003;304:453–463. doi: 10.1124/jpet.102.041343. [DOI] [PubMed] [Google Scholar]
- Durelli L, Mutani R, Fassio F. The treatment of myotonia: evaluation of chronic oral taurine therapy. Neurology. 1983;33:599–603. doi: 10.1212/WNL.33.5.599-a. [DOI] [PubMed] [Google Scholar]
- Dutka TL, Lamboley CRl, Murphy RM, Lamb GD. Acute effects of taurine on sarcoplasmic reticulum Ca2+ accumulation and contractility in human type I and type II skeletal muscle fibers. J Appl Physiol. 1985;117:797–805. doi: 10.1152/japplphysiol.00494.2014. [DOI] [PubMed] [Google Scholar]
- Eby G, Halcomb WW. Elimination of cardiac arrhythmias using oral taurine with L-arginine with case histories: hypothesis for nitric oxide stabilization of the sinus node. Med Hypotheses. 2006;67:1200–1204. doi: 10.1016/j.mehy.2006.04.055. [DOI] [PubMed] [Google Scholar]
- El Idrissi A, L’Amoreaux WJ. Selective resistance of taurine-fed mice to isoniazide-potentiated seizures: in vivo functional test for the activity of glutamic acid decarboxylase. Neuroscience. 2008;156:693–699. doi: 10.1016/j.neuroscience.2008.07.055. [DOI] [PubMed] [Google Scholar]
- El Idrissi A, Messing J, Scalia J, Trenkner E. Prevention of epileptic seizures by taurine. Adv Exp Med Biol. 2003;526:515–525. doi: 10.1007/978-1-4615-0077-3_62. [DOI] [PubMed] [Google Scholar]
- Elvevoll EO, Eilertsen KE, Brox J, Dragnes BT, Falkenberg P, Olsen JO, Kirkhus B, Lamglait A, Osterud B. Seafood diets: and antiatherogenic effects of taurine and n-3 fatty acids. Atherosclerosis. 2008;200:396–402. doi: 10.1016/j.atherosclerosis.2007.12.021. [DOI] [PubMed] [Google Scholar]
- Franconi F, Bennardini F, Mattana A, Miceli M, Ciuti M, Mian M, Gironi A, Anichini R, Seghieri G. Plasma and platelet taurine are related in subjects with insulin-dependent diabetes mellitus: effect of taurine supplementation. Am J Clin Nutr. 1995;61:1115–1119. doi: 10.1093/ajcn/61.5.1115. [DOI] [PubMed] [Google Scholar]
- Froger N, Cadetti L, Lorach H, Martins J, Bemelmans AP, Dubus E, Degardin J, Pain D, Forster V, Chicaud L, Ivokovic I, Simonutti M, Fouquet S, Jammoul F, Leveillard T, Benosman R, Sahel JA, Picaud S. Taurine provides neuroprotection against retinal ganglion cell degradation. PLoS ONR. 2012;7:e42017. doi: 10.1371/journal.pone.0042017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Froger N, Moutsimilli L, Cadetti L, Jammoul F, Wang QP, Fan Y, Gaucher D, Rosolen SG, Neveux N, Cynober L, Sahel JA, Picaud S. Taurine: the comeback of a neutraceutical in the prevention of retinal degenerations. Prog Retin Eye Res. 2014;41:44–63. doi: 10.1016/j.preteyeres.2014.03.001. [DOI] [PubMed] [Google Scholar]
- Fujita T, Sato Y. Changes in blood pressure and extracellular fluid with taurine in DOCA-salt rats. Am J Physiol. 1986;250:R1014–R1020. doi: 10.1152/ajpregu.1986.250.6.R1014. [DOI] [PubMed] [Google Scholar]
- Furukawa T, Yamada J, Akita T, Matsushima Y, Yanagawa Y, Fukuda A. Role of taurine-mediated tonic GABAA receptor activation in the radial migration of neurons in the fetal mouse cerebral cortex. Front Cell Neurosci. 2014;8:88. doi: 10.3389/fncel.2014.00088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaull GE. Taurine as a conditionally essential nutrient in man. J Am Coll Nutr. 1986;5:121–125. doi: 10.1080/07315724.1986.10720119. [DOI] [PubMed] [Google Scholar]
- Gaull GE. Taurine in pediatric nutrition: review and update. Pediatrics. 1989;83:433–442. [PubMed] [Google Scholar]
- Gharibani PM, Modi J, Pan C, Menzie J, Ma Z, Chen PC, Tao R, Prentice H, Wu JY. The mechanism of taurine protection against endoplasmic reticulum stress in an animal stroke model of cerebral artery occlusion and stroke-related conditions in primary neuronal cell culture. Adv Exp Med Biol. 2013;776:241–258. doi: 10.1007/978-1-4614-6093-0_23. [DOI] [PubMed] [Google Scholar]
- Gharibani P, Modi J, Menzie J, Alexandrescu A, Ma Z, Tao R, Prentice H, Wu JY. Comparison between single and combined post-treatment with S-Methyl-N, N-diethylthiolcarbamate sulfoxide and taurine following transient focal cerebral ischemia in rat brain. Neuroscience. 2015;300:460–473. doi: 10.1016/j.neuroscience.2015.05.042. [DOI] [PubMed] [Google Scholar]
- Ginguay A, De Bandt JP, Cynober L. Indications and contraindications for infusing specific amino acids. (leucine, glutamine, arginine, citrulline and taurine) in critical illness. Curr Opin Clin Nutr Metab Care. 2016;19:161–169. doi: 10.1097/MCO.0000000000000255. [DOI] [PubMed] [Google Scholar]
- Gokce G, Ozsarlak-Sozer G, Oran I, Oktay G, Ozkal S, Kerry Z. Taurine suppresses oxidative stress-potentiated expression of lectin-like oxidized low-density lipoprotein receptor and restensosis in balloon-injured rabbit iliac artery. Clin Expt Pharmacol Physiol. 2011;38:811–818. doi: 10.1111/j.1440-1681.2011.05612.x. [DOI] [PubMed] [Google Scholar]
- Goodman CA, Horvath D, Stathis C, Mori T, Croft K, Murphy RM, Hayes A. Taurine supplementation increases skeletal muscle force production and protects muscle function during and after high-frequency in vitro stimulation. J Appl Physiol. 1985;107:144–154. doi: 10.1152/japplphysiol.00040.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haber CA, Lam TK, Yun Z, Gupta N, Goh T, Bogdanovic E, Giacca A, Fantus IG. N-acetylcysteine and taurine prevent hyperglycemia-induced insulin resistance in vivo: possible role of oxidative stress. Am J Physiol. 2003;285:E744–E753. doi: 10.1152/ajpendo.00355.2002. [DOI] [PubMed] [Google Scholar]
- Hadj-Saïd W, Froger N, Ivkovic I, Jiménez-López M, Dubus É, Dégardin-Chicaud J, Simonutti M, Quénol C, Neveux N, Villegas-Pérez MP, Agudo-Barriuso M, Vidal-Sanz M, Sahel JA, Picaud S, García-Ayuso D. Quantitative and topographical analysis of the losses of cone photoreceptors and retinal ganglion cells under taurine depletion. Invest Ophthalmol Vis Sci. 2016;57:4692–4703. doi: 10.1167/iovs.16-19535. [DOI] [PubMed] [Google Scholar]
- Hagar HH, El Etter E, Arafa M. Taurine attenuates hypertension and renal dysfunction induced by cyclosporine A in rats. Clin Exp Pharmacol Physiol. 2006;33:189–196. doi: 10.1111/j.1440-1681.2006.04345.x. [DOI] [PubMed] [Google Scholar]
- Hallan S, Sharma K. The role of mitochondria in diabetic kidney disease. Curr Diab Rep. 2016;16:61. doi: 10.1007/s11892-016-0748-0. [DOI] [PubMed] [Google Scholar]
- Hamilton EJ, Berg HM, Easton CJ, Bakker AJ. The effect of taurine depletion on the contractile properties and fatigue in fast-twitch skeletal muscle of the mouse. Amino Acids. 2006;31:273–278. doi: 10.1007/s00726-006-0291-4. [DOI] [PubMed] [Google Scholar]
- Han X, Chesney RW. The role of taurine in renal disorders. Amino Acids. 2012;43:2249–2263. doi: 10.1007/s00726-012-1314-y. [DOI] [PubMed] [Google Scholar]
- Han X, Patters AB, Ito T, Azuma J, Schaffer SW, Chesney RW. Knockout of the TauT gene predisposes C57BL/6 mice to streptozotocin-induced diabetic nephropathy. PLoS ONE. 2015;10:e0117718. doi: 10.1371/journal.pone.0117718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han Z, Gao LY, Lin YH, Chang L, Wu HY, Luo CX, Zhu DY. Neuroprotection of taurine against reactive oxygen species is associated with inhibiting NADPH oxidases. Eur J Pharmacol. 2016;777:129–135. doi: 10.1016/j.ejphar.2016.03.006. [DOI] [PubMed] [Google Scholar]
- Hara MR, Snyder SH. Cell signaling and neuronal death. Ann Rev Pharmacol Toxicol. 2007;47:117–141. doi: 10.1146/annurev.pharmtox.47.120505.105311. [DOI] [PubMed] [Google Scholar]
- Harada H, Isujino T, Watari Y, Nonaka H, Emoto N, Yokoyama M. Oral taurine supplementation prevents fructose-induced hypertension in rats. Heart Vessels. 2004;19:132–136. doi: 10.1007/s00380-003-0757-1. [DOI] [PubMed] [Google Scholar]
- Hayes KC, Carey RE, Schmidt SY. Retinal degeneration associated with taurine deficiency in the cat. Science. 1975;188:949–951. doi: 10.1126/science.1138364. [DOI] [PubMed] [Google Scholar]
- Heim MK, Gidal BE. Vigabatrin-associated retinal damage: potential biochemical mechanisms. Acta Neurol Scand. 2012;126:219–228. doi: 10.1111/j.1600-0404.2012.01684.x. [DOI] [PubMed] [Google Scholar]
- Higgins GC, Coughlan MT. Mitochondrial dysfunction and mitophagy: the beginning and end to diabetic nephropathy? Br J Pharmacol. 2014;171:1917–1942. doi: 10.1111/bph.12503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horvath DM, Murphy RM, Mollica JP, Hayes A, Goodman CA. The effect of taurine and β-alanine supplementation on taurine transporter protein and fatigue resistance in skeletal muscle from mdx mice. Amino Acids. 2016;48:2635–2645. doi: 10.1007/s00726-016-2292-2. [DOI] [PubMed] [Google Scholar]
- Horvath GA, Hukin J, Stockler-Ipsiroglu SG, Aroichane M. Eye findings on vigabatrin and taurine treatment in two patients with succinic semialdehyde dehydrogenase deficiency. Neuropediatrics. 2016;47:263–267. doi: 10.1055/s-0036-1583183. [DOI] [PubMed] [Google Scholar]
- Hu J, Xu X, Yang J, Wu G, Sun C, Lv Q. Antihypertensive effect of taurine in rat. Adv Exp Med Biol. 2009;643:75–84. doi: 10.1007/978-0-387-75681-3_8. [DOI] [PubMed] [Google Scholar]
- Ideishi M, Miura S, Sakai T, Sasaguri M, Misumi Y, Arakawa K. Taurine amplifies renal kallikrein and prevents salt-induced hypertension in Dahl rats. J Hypertens. 1994;12:653–661. doi: 10.1097/00004872-199406000-00005. [DOI] [PubMed] [Google Scholar]
- Ikubo N, Saito M, Tsounapi P, Dimitriadis F, Ohmasa F, Inoue S, Shimizu S, Kinoshita Y, Satoh K. Protective effect of taurine on diabetic rat endothelial dysfunction. Biomed Res. 2011;32:187–193. doi: 10.2220/biomedres.32.187. [DOI] [PubMed] [Google Scholar]
- Imai M, Asano T, Murakami S. Potential role of taurine in prevention of diabetes and metabolic syndrome. Amino Acids. 2014;46:81–88. doi: 10.1007/s00726-012-1434-4. [DOI] [PubMed] [Google Scholar]
- Ishikura K, Miyazaki T, Ra SG, Endo S, Nakamura Y, Matsuzaka T, Miyakawa S, Ohmori H. Effect of supplementation on the alterations in amino acid content in skeletal muscle with exercise in rat. J Sports Sci Med. 2011;10:306–314. [PMC free article] [PubMed] [Google Scholar]
- Ito T, Kimura Y, Uozumi Y, Takai M, Muraoka S, Matsuda T, Ueki K, Yoshiyama M, Ikawa M, Okabe M, Schaffer SW, Fujio Y, Azuma J. Taurine depletion caused by knocking out the taurine transporter gene leads to cardiomyopathy with cardiac atrophy. J Mol Cell Cardiol. 2008;44:927–937. doi: 10.1016/j.yjmcc.2008.03.001. [DOI] [PubMed] [Google Scholar]
- Ito T, Miyazaki N, Schaffer S, Azuma J. Potential antiaging role of taurine via proper protein folding: a study from taurine transporter knockout mouse. Adv Exp Med Biol. 2015a;803:481–487. doi: 10.1007/978-3-319-15126-7_38. [DOI] [PubMed] [Google Scholar]
- Ito T, Schaffer SW, Azuma J. The potential usefulness of taurine on diabetes mellitus and its complications. Amino Acids. 2012;42:1529–1539. doi: 10.1007/s00726-011-0883-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito T, Yoshikawa N, Inui T, Miyazaki N, Schaffer SW, Azuma J. Tissue depletion of taurine accelerates skeletal muscle senescence and leads to early death in mice. PLoS ONE. 2014a;9:e107409. doi: 10.1371/journal.pone.0107409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito T, Yoshikawa N, Ito H, Schaffer SW. Impact of taurine depletion on glucose control and insulin secretion in mice. J Pharmacol Sci. 2015b;129:59–64. doi: 10.1016/j.jphs.2015.08.007. [DOI] [PubMed] [Google Scholar]
- Ito T, Yoshikawa N, Schaffer SW, Azuma J. Tissue taurine depletion alters metabolic response to exercise and reduces running capacity in mice. J Amino Acids. 2014b;2014:964680. doi: 10.1155/2014/964680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeejeebhoy F, Keith M, Freeman M, Barr A, McCall M, Kurian R, Mazer D, Errett L. Nutritional supplementation with MyoVive repletes essential cardiac myocyte nutrients and reduces left ventricular size in patients with left ventricular dysfunction. Am Heart J. 2002;143:1092–1100. doi: 10.1067/mhj.2002.121927. [DOI] [PubMed] [Google Scholar]
- Jong CJ, Azuma J, Schaffer SW. Mechanism underlying the antioxidant activity of taurine: prevention of mitochondrial oxidant production. Amino Acids. 2012;42:2223–2232. doi: 10.1007/s00726-011-0962-7. [DOI] [PubMed] [Google Scholar]
- Jong CJ, Ito T, Schaffer SW. The ubiquitin-proteasome system and autophagy are defective in the taurine-deficient heart. Amino Acids. 2015;47:2609–2622. doi: 10.1007/s00726-015-2053-7. [DOI] [PubMed] [Google Scholar]
- Junyent F, Romero R, De Lemos L, Ultrera J, Camins A, Pallas M, Auladell C. Taurine treatment inhibits CaMKII activity and modulates the presence of calbindin D28k, calretinin and parvalbumin in the brain. J Neurosci Res. 2010;88:136–142. doi: 10.1002/jnr.22192. [DOI] [PubMed] [Google Scholar]
- Junyent F, Ultrera J, Romero R, Pallas M, Camins A, Duque D, Auladell C. Prevention of epilepsy by taurine treatments in mice experimental model. J Neurosci Res. 2009;87:1500–1508. doi: 10.1002/jnr.21950. [DOI] [PubMed] [Google Scholar]
- Katakawa M, Fukuda N, Tsunemi A, Mori M, Maruyama T, Matsumoto T, Abe M, Yamori Y. Taurine and magnesium supplementation enhances the function of endothelial progenitor cells through antioxidation in healthy men and spontaneously hypertensive rats. Hypertens Res. 2016;39:848–856. doi: 10.1038/hr.2016.86. [DOI] [PubMed] [Google Scholar]
- Kato T, Okita S, Wang S, Tsunekawa M, Ma N. The effects of taurine administraiton against inflammation in heavily exercised skeletal muscle of rats. Adv Exp Med Biol. 2015;803:773–784. doi: 10.1007/978-3-319-15126-7_62. [DOI] [PubMed] [Google Scholar]
- Kim C, Cha YN. Taurine chloramine produced from taurine under inflammation provides anti-inflammatory and cytoprotective effects. Amino Acids. 2014;46:89–100. doi: 10.1007/s00726-013-1545-6. [DOI] [PubMed] [Google Scholar]
- Kim KS, Oh DH, Kim JY, Lee BG, You JS, Chang KJ, Chung HJ, Yoo MC, Yang HI, Kang JH, Hwang YC, Ahn KJ, Chung HY, Jeong IK. Taurine ameliorates hyperglycemia and dyslipidemia by reducing insulin resistance and leptin level in Otsuka Long-Evans Tokushima fatty. (OLETF) rats with long-term diabetes. Exp Mol Med. 2012;44:665–673. doi: 10.3858/emm.2012.44.11.075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim KS, Park EK, Ju SM, Jung HS, Bang JS, Kim C, Lee YA, Hong SJ, Lee SH, Yang HI, Yoo MC. Taurine chloramine differentially inhibits matrix metalloproteinase 1 and 13 synthesis in interleukin-1β stimulated fibroblast-like synoviocytes. Arthritis Res Ther. 2007;9:R80. doi: 10.1186/ar2279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirino Y, Goto Y, Campos Y, Arenas J, Suzuki T. Specific correlation between the wobble modification deficiency in mutant tRNAs and the clinical features of a human mitochondrial disease. Proc Natl Acad Sci USA. 2005;102:7127–7132. doi: 10.1073/pnas.0500563102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klamt F, Shacter E. Taurine chloramine: an oxidant derived from neutrophils induces apoptosis in human B lymphoma cells through mitochondrial damage. J Biol Chem. 2005;280:21346–21352. doi: 10.1074/jbc.M501170200. [DOI] [PubMed] [Google Scholar]
- Koh JH, Lee ES, Hyun M, Kim HM, Choi YJ, Lee EY, Yadav D, Chung CH. Taurine alleviates the progression of diabetic nephropathy in type 2 diabetic rat model. Int J Endocrinol. 2014;2014:397307. doi: 10.1155/2014/397307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konig P, Kriechbaum G, Presslich O, Schubert H, Schuster P, Sieghart W. Orally-administered taurine in therapy-resistant epilepsy. Wien Klin Wochenschr. 1977;89:111–113. [PubMed] [Google Scholar]
- Kurata S, Ohtsuki T, Wada T, Kirino Y, Takai K, Saigo K, Watanabe K, Suzuki T. Decoding property of C5 uridine modification at the wobble position of tRNA anticodon. Nucleic Acids Res. 2003;3:245–246. doi: 10.1093/nass/3.1.245. [DOI] [PubMed] [Google Scholar]
- L’Amoreaux WJ, Marsillo A, El Idrissi A. Pharmacological characterization of GABAA receptors in taurine-fed mice. J Biomed Sci. 2010;17:S14. doi: 10.1186/1423-0127-17-S1-S14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam NV, Chen W, Suruga K, Nishimura N, Goda T, Yokogoshi H. Enhancing effect of taurine on CYP7A1 mRNA expression in Hep G2 cells. Amino Acids. 2006;30:43–48. doi: 10.1007/s00726-005-0244-3. [DOI] [PubMed] [Google Scholar]
- Li Y, Hu Z, Chen B, Bu Q, Lu W, Deng Y, Zhu R, Shao X, Hou J, Zaho J, Li H, Zhang B, Huang Y, Lv L, Zhao Y, Cen X. Taurine attenuates methamphetamine-induced autophagy and apoptosis in PC12 cells through mTOR signaling pathway. Toxicol Lett. 2012;215:1–7. doi: 10.1016/j.toxlet.2012.09.019. [DOI] [PubMed] [Google Scholar]
- Lindblom R, Higgins G, Coughlan M, de Haan JB. Targeting mitochondria and reactive oxygen species-driven pathogenesis in diabetic nephropathy. Rev Diabet Stud. 2015;12:134–156. doi: 10.1900/RDS.2015.12.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lombardini JB. Taurine depletion in the intact animal stimulates in vitro phosphorylation of an approximately 44-kDa protein present in the mitochondrial fraction of the rat heart. J Mol Cell Cardiol. 1996;28:1957–1961. doi: 10.1006/jmcc.1996.0188. [DOI] [PubMed] [Google Scholar]
- Maia AR, Batista TM, Victorio JA, Clerici SP, Delbin MA, Carneiro EM, Davel AP. Taurine supplementation reduces blood pressure and prevents endothelial dysfunction and oxidative stress in post-weaning protein restricted rats. PLoS ONE. 2014;9:e105851. doi: 10.1371/journal.pone.0105851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik AN, Parsade CK, Ajaz S, Crosby-Nwaobi RC, Gnudi L, Czajka A, Sivaprasad S. Altered circulating mitochondrial DNA and increased inflammation in patients with diabetic retinopathy. Diabetes Res Clin Pract. 2015;110:257–265. doi: 10.1016/j.diabres.2015.10.006. [DOI] [PubMed] [Google Scholar]
- Mantovani J, DeVivo DC. Effects of taurine on seizures and growth hormone release in epileptic patients. Arch Neurol. 1979;36:672–674. doi: 10.1001/archneur.1979.00500470042006. [DOI] [PubMed] [Google Scholar]
- Marcinkiewicz J, Kontny E. Taurine and inflammatory diseases. Amino Acids. 2014;46:7–20. doi: 10.1007/s00726-012-1361-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcinkiewicz J, Grabowska A, Bereta J, Stelmaszynska T. Taurine chloramine a product of activated neutrophils inhibits in vitro the generation of nitric oxide and other macrophage inflammatory mediators. J Leukoc Biol. 1995;58:667–674. doi: 10.1002/jlb.58.6.667. [DOI] [PubMed] [Google Scholar]
- McCarty MF. Nutraceutical strategies for ameliorating the toxic effects of alcohol. Med Hypotheses. 2013;80:456–462. doi: 10.1016/j.mehy.2012.12.040. [DOI] [PubMed] [Google Scholar]
- Menzie J, Prentice H, Wu JY. Neuroprotective mechanisms of taurine against ischemic stroke. Brain Sci. 2013;3:877–907. doi: 10.3390/brainsci3020877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milei J, Ferreira R, Llesuy S, Forcada P, Covarrubias J, Boveris A. Reduction of reperfusion injury with preoperative rapid intravenous infusion of taurine during myocardial revascularization. Am Heart J. 1992;123:339–345. doi: 10.1016/0002-8703(92)90644-B. [DOI] [PubMed] [Google Scholar]
- Modi P, Suleiman MS. Myocardial taurine, development and vulnerability to ischemia. Amino Acids. 2004;26:65–70. doi: 10.1007/s00726-003-0031-y. [DOI] [PubMed] [Google Scholar]
- Moore KJ, Tabas I. The cellular biology of macrophages in atherosclerosis. Cell. 2011;145:341–355. doi: 10.1016/j.cell.2011.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mozaffari MS, Patel C, Abdelsayed R, Schaffer SW. Accelerated NaCl-induced hypertension in taurine-deficient rat: role of renal function. Kidney Int. 2006;70:329–337. doi: 10.1038/sj.ki.5001503. [DOI] [PubMed] [Google Scholar]
- Murakami S. Role of taurine in the pathogenesis of obesity. Mol Nutr Food Res. 2015;59:1353–1363. doi: 10.1002/mnfr.201500067. [DOI] [PubMed] [Google Scholar]
- Murakami S, Nara Y, Yamori Y. Taurine accelerates the regression of hypercholesterolemia in stroke-prone spontaneously hypertensive rats. Life Sci. 1996;58:1643–1651. doi: 10.1016/0024-3205(96)00139-7. [DOI] [PubMed] [Google Scholar]
- Murakami S, Sakurai T, Tomoike H, Sakono M, Nasu T, Fukuda N. Prevention of hypercholesterolemia and atherosclerosis in the hyperlipidemia- and atherosclerosis-prone Japanese (LAP) quail by taurine supplementation. Amino Acids. 2010;38:271–278. doi: 10.1007/s00726-009-0247-6. [DOI] [PubMed] [Google Scholar]
- Nakaya Y, Minami A, Harada N, Sakamoto S, Niwa Y, Ohnaka M. Taurine improves insulin sensitivity in the Otsuka Long-Evans Tokushima Fatty rat, a model of spontaneous type 2 diabetes. Am J Clin Nutr. 2000;71:54–58. doi: 10.1093/ajcn/71.1.54. [DOI] [PubMed] [Google Scholar]
- Nara Y, Yamori Y, Lovenberg W. Effect of dietary taurine on blood pressure in spontaneously hypertensive rats. Biochem Pharmacol. 1978;27:2689–2692. doi: 10.1016/0006-2952(78)90043-6. [DOI] [PubMed] [Google Scholar]
- Neuwirth LS, Volpe NP, Ng S, Marsillo A, Corwin C, Madan N, Ferraro AM, El Idrissi A. Taurine recovers mice emotional learning and memory disruptions associated with fragile X syndrome in context gear and auditory-cued conditioning. Adv Exp Med Biol. 2015;803:425–438. doi: 10.1007/978-3-319-15126-7_33. [DOI] [PubMed] [Google Scholar]
- Novotny MJ, Hogan PM, Paley DM, Adams HR. Systolic and diastolic dysfunction of the left ventricle induced by dietary taurine deficiency in cats. Am J Physiol. 1991;261:H121–H127. doi: 10.1152/ajpheart.1991.261.1.H121. [DOI] [PubMed] [Google Scholar]
- Ogawa M, Takahara A, Ishijima M, Tazaki S. Decrease of plasma sulfur amino acids in essential hypertension. Jpn Circ J. 1985;49:1217–1224. doi: 10.1253/jcj.49.1217. [DOI] [PubMed] [Google Scholar]
- Oja SS, Saransaari P. Taurine and epilepsy. Epilepsy Res. 2013;104:187–194. doi: 10.1016/j.eplepsyres.2013.01.010. [DOI] [PubMed] [Google Scholar]
- Oprescu AI, Bikopoulos G, Naassan A, Allister EM, Tang C, Park E, Uchino H, Lewis GF, Fantus IG, Rozakis-Adcock M, Wheeler MB, Giacca A. Free fatty acid-induced reduction in glucose-stimulated insulin secretion: evidence for a role of oxidative stress in vitro and in vivo. Diabetes. 2007;56:2927–2937. doi: 10.2337/db07-0075. [DOI] [PubMed] [Google Scholar]
- Oriyanhan W, Yamazaki K, Miwa S, Takaba K, Ikeda T, Komeda M. Taurine prevents myocardial ischemia/reperfusion-induced oxidative stress and apoptosis in prolonged hypothermic rat heart preservation. Heart Vessels. 2005;20:278–285. doi: 10.1007/s00380-005-0841-9. [DOI] [PubMed] [Google Scholar]
- Pandya KG, Budhram R, Clark G, Lau-Cam CA. Comparative evaluation of taurine and thiotaurine as protectants against diabetes-induced nephropathy in a rat model. Adv Exp Med Biol. 2013;775:371–394. doi: 10.1007/978-1-4614-6130-2_29. [DOI] [PubMed] [Google Scholar]
- Park E, Park SY, Dobkin C, Schuller-Levis G. Development of a novel cysteine sulfinic acid decarboxylase knockout mouse: dietary taurine reduces neonatal mortality. J Amino Acids. 2014;2014:346809. doi: 10.1155/2014/346809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park E, Quinn MR, Schuller-Levis G. Taurine chloramine attenuates the hydrolytic activity of matrix metalloproteinase-9 in LPS-activated murine peritoneal macrophages. Adv Exp Med Biol. 2000;483:389–398. doi: 10.1007/0-306-46838-7_44. [DOI] [PubMed] [Google Scholar]
- Park E, Schuller-Levis Gl, Jia JH, Quinn MR. Preactivation exposure of RAW 264.7 cells to taurine chloramine attenuates subsequent production of nitric oxide and expression of iNOS mRNA. J Leukoc Biol. 1997;61:161–166. doi: 10.1002/jlb.61.2.161. [DOI] [PubMed] [Google Scholar]
- Park SH, Lee H, Park KK, Kim HW, Lee DH, Park T. Taurine-induced changes in transcription profiling of metabolism-related genes in human hepatoma cells HepG2. Adv Exp Med Biol. 2006;583:119–128. doi: 10.1007/978-0-387-33504-9_12. [DOI] [PubMed] [Google Scholar]
- Pearl PL, Schreiber J, Theodore WH, McCarter R, Barrios ES, Yu J, Wiggs E, Jianping H, Gibson KM. Taurine trial in succinic semialdehyde dehydrogenase deficiency and elevated CNS GABA. Neurology. 2014;82:940–944. doi: 10.1212/WNL.0000000000000210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petty MA, Kintz J, DiFrancesco GF. The effects of taurine on atherosclerosis development in cholesterol-fed rabbits. Eur J Pharmacol. 1990;180:119–127. doi: 10.1016/0014-2999(90)90599-2. [DOI] [PubMed] [Google Scholar]
- Prentice H, Modi JP, Wu JY. Mechanisms of neuronal protection against excitotoxicity, endoplasmic reticulum stress, and mitochondrial dysfunction in stroke and neurodegenerative diseases. Oxid Med Cell Longev. 2015;2015:964518. doi: 10.1155/2015/964518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ra SG, Choi Y, Akazawa N, Ohmori H, Maeda S. Taurine supplementation attenuates delayed increase in exercise-induced arterial stiffness. Appl Physiol Nutr Metab. 2016;41:618–623. doi: 10.1139/apnm-2015-0560. [DOI] [PubMed] [Google Scholar]
- Ra SG, Miyazaki T, Ishikura K, Nagayama H, Komine S, Nakata Y, Maeda S, Matsuzaki Y, Ohmori H. Combined effect of branched-chain amino acids and taurine supplementation on delayed onset muscle soreness and muscle damage in highintensity eccentric exercise. J Int Soc Sports Nutr. 2013;10:51. doi: 10.1186/1550-2783-10-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramila KC, Jong CJ, Pastukh V, Ito T, Azuma J, Schaffer SW. Role of protein phosphorylation in excitation-contraction coupling in taurine deficient hearts. Am J Physiol. 2015;308:H232–H239. doi: 10.1152/ajpheart.00497.2014. [DOI] [PubMed] [Google Scholar]
- Rasola A, Bernardi P. Mitochondrial permeability transition in Ca2+-dependent apoptosis and necrosis. Cell Calcium. 2011;50:222–233. doi: 10.1016/j.ceca.2011.04.007. [DOI] [PubMed] [Google Scholar]
- Read WO, Welty JD. Effect of taurine on epinephrine and digoxin induced irregularities of the dog heart. J Pharmacol Exp Ther. 1963;139:283–289. [PubMed] [Google Scholar]
- Ricci C, Pastukh V, Leonard J, Turrens J, Wilson G, Schaffer D, Schaffer SW. Mitochondrial DNA damage triggers mitochondrial superoxide generation and apoptosis. Am J Physiol. 2008;294:C413–C422. doi: 10.1152/ajpcell.00362.2007. [DOI] [PubMed] [Google Scholar]
- Ricci L, Valoti M, Sgaragli G, Frosini M. Protection by taurine of rat brain cortical slices against oxygen glucose deprivation- and reoxygenation-induced damage. Eur J Pharmacol. 2009;621:26–32. doi: 10.1016/j.ejphar.2009.08.017. [DOI] [PubMed] [Google Scholar]
- Ricciardi L, De Nigris F, Specchia A, Fasano A. Homotaurine in Parkinson’s disease. Neurosci Res. 2015;36:1581–1587. doi: 10.1007/s10072-015-2201-6. [DOI] [PubMed] [Google Scholar]
- Rikimaru M, Ohsawa Y, Wolf AM, Ichimiya H, Kamimura N, Nishimatsu S, Ohta S, Sunada Y. Taurine ameliorates impaired mitochondrial function and prevents stroke-like episodes in patients with MELAS. Intern Med. 2012;51:3351–3357. doi: 10.2169/internalmedicine.51.7529. [DOI] [PubMed] [Google Scholar]
- Ripps H, Shen W. Review: taurine: a “very essential” amino acid. Mol Vis. 2012;18:2673–2686. [PMC free article] [PubMed] [Google Scholar]
- Rosa FT, Freitas EC, Deminice R, Jordao AA, Marchini JS. Oxidative stress and inflammation in obesity after taurine supplementation: a double-blind placebo-controlled study. Eur J Nutr. 2014;53:823–830. doi: 10.1007/s00394-013-0586-7. [DOI] [PubMed] [Google Scholar]
- Rumpl E, Gerstenbrand F, Hengl W, Binder H. Electrophy siogical and neuropharmacological studies in a patient with progressive myoclonus-epilepsy. Elektromyograph Verwandte Geb. 1977;8:77–81. [PubMed] [Google Scholar]
- Sagara M, Murakami S, Mizushima S, Liu L, Mori M, Ikeda K, Nara Y, Yamori Y. Taurine in 24-h urine samples is inversely related to cardiovascular risks of middle aged subjects in 50 populations of the world. Adv Exp Med Biol. 2015;803:623–636. doi: 10.1007/978-3-319-15126-7_50. [DOI] [PubMed] [Google Scholar]
- Sahin MA, Yucel O, Guler A, Doganci S, Jaholliari A, Cinqoz F, Arslan S, Gamsizkan M, Yaman H, Demirkilic U. Is there any cardioprotective role for taurine during cold ischemic period following global myocardial ischemia? J Cardiothorac Surg. 2011;6:31. doi: 10.1186/1749-8090-6-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos JM, Mishra M, Kowluru RA. Posttranslational modification of mitochondrial transcription factor A in impaired mitochondria biogenesis. Implications in diabetic retinopathy and metabolic memory phenomenon. Exp Eye Res. 2014;121:168–177. doi: 10.1016/j.exer.2014.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saransaari P, Oja SS. Modulation of taurine release by glutamate receptors and nitric oxide. Prog Neurobiol. 2010;62:407–425. doi: 10.1016/s0301-0082(00)00005-8. [DOI] [PubMed] [Google Scholar]
- Saronwala A, Tournay A, Garguss JJ. Taurine treatment of succinate semialdehyde dehydrogenase (SSADH) deficiency reverses MRI-documented globus lesions and clinical syndrome.. Proc. Am. Coll. Med. Genet., 15th Ann. Clinical Genet Meeting; March 12–16 2008; Phoenix, AZ, USA. 2008. [Google Scholar]
- Sato Y, Ogata E, Fujita T. Hypotensive action of taurine in DOCA-salt rats--involvement of sympathoadrenal inhibition and endogenous opiate. Jpn Circ J. 1991;55:500–508. doi: 10.1253/jcj.55.500. [DOI] [PubMed] [Google Scholar]
- Schaffer SW, Azuma J, Mozaffari M. Role of antioxidant activity of taurine in diabetes. Can J Physiol Pharmacol. 2009;87:91–99. doi: 10.1139/Y08-110. [DOI] [PubMed] [Google Scholar]
- Schaffer SW, Ballard-Croft C, Azuma J, Takahashi K, Kakhniashvili DG, Jenkins TE. Shape and size changes induced by taurine depletion in neonatal cardiomyocytes. Amino Acids. 1998;15:135–142. doi: 10.1007/BF01345286. [DOI] [PubMed] [Google Scholar]
- Schaffer SW, Jong CJ, Ito T, Azuma J. Role of taurine in the pathogenesis of MELAS and MERRF. Amino Acids. 2014a;46:47–56. doi: 10.1007/s00726-012-1414-8. [DOI] [PubMed] [Google Scholar]
- Schaffer SW, Jong CJ, Ito T, Azuma J. Effect of taurine on ischemia-reperfusion injury. Amino Acids. 2014b;46:21–30. doi: 10.1007/s00726-012-1378-8. [DOI] [PubMed] [Google Scholar]
- Schaffer SW, Jong CJ, Warner D, Ito T, Azuma J. Taurine deficiency and MELAS are closely related syndromes. Adv Exp Med Biol. 2013;776:153–165. doi: 10.1007/978-1-4614-6093-0_16. [DOI] [PubMed] [Google Scholar]
- Schaffer SW, Shimada-Takaura K, Jong CJ, Ito T, Takahashi K. Impaired energy metabolism of the taurine-deficient heart. Amino Acids. 2016;48:549–558. doi: 10.1007/s00726-015-2110-2. [DOI] [PubMed] [Google Scholar]
- Schaffer SW, Solodushko V, Kakhniashvili D. Beneficial effect of taurine depletion on osmotic sodium and calcium loading during chemical hypoxia. Am J Physiol. 2002;282:C1113–C1120. doi: 10.1152/ajpcell.00485.2001. [DOI] [PubMed] [Google Scholar]
- Schmidt SY, Berson EL, Hayes KC. Retinal degeneration in cats fed casein. I. Taurine deficiency. Invest Ophthalmol. 1976;15:47–52. [PubMed] [Google Scholar]
- Scicchitano BM, Sica G. The beneficial effects of taurine to counteract sarcopenia. Curr Protein Pept Sci. 2016 doi: 10.2174/1389203718666161122113609. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sergeeva OA, Fleischer W, Chepkova AN, Warskulat U, Haussinger D. GABAA receptor modification in taurine transporter knockout mice causes striatal disinhibition. J Physiol. 2007;585:539–548. doi: 10.1113/jphysiol.2007.141432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shetewy A, Shimada-Takaura K, Warner D, Jong CJ, Mehdi AB, Alexeyev M, Takahashi K, Schaffer SW. Mitochondrial defects associated with B-alanine toxicity: relevance to hyper-beta-alaninemia. Mol Cell Biochem. 2016;416:11–22. doi: 10.1007/s11010-016-2688-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sivitz WI, Yorek MA. Mitochondrial dysfunction in diabetes: from molecular mechanisms to functional significance and therapeutic opportunitiees. Antioxid Redox Signal. 2010;12:537–577. doi: 10.1089/ars.2009.2531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiura H, Okita S, Kato T, Naka T, Kawanishi Sl, Ohnishi Sl, Oshida Y, Ma N. Protection by taurnine against iNOS-dependent DNA damage in heavily exercised skeletal muscle by inhibition of the NF-kappaB signalling pathway. Adv Exp Med Biol. 2013;775:237–246. doi: 10.1007/978-1-4614-6130-2_20. [DOI] [PubMed] [Google Scholar]
- Sun Q, Wang B, Li Y, Sun F, Li P, Xia W, Zhou X, Li Q, Wang X, Chen J, Zeng X, Zhao Z, He H, Liu D, Zhu Z. Taurine supplementation lowers blood pressure and improves vascular function in prehypertension: randomized, double-blind, placebo controlled study. Hypertension. 2016;67:541–549. doi: 10.1161/HYPERTENSIONAHA.115.06624. [DOI] [PubMed] [Google Scholar]
- Tan B, Jiang DJ, Huang H, Jia SJ, Hu CP, Li YJ. Taurine protects against low-density lipoprotein-induced endothelial dysfunction by the DDAH/ADMA pathway. Vascul Pharmacol. 2007;46:338–345. doi: 10.1016/j.vph.2006.11.006. [DOI] [PubMed] [Google Scholar]
- Terashima M, Mitani T, Hosokawa Y, Nariai Y, Imada K, Kageyama E, Tanigawa Y. Suppressive effect of taurine on platelet-derived growth factor (PDGF) BB-induced c-fos and c-jun mRNA expressions through extracellular signal-regulated kinase (ERK) in mesenchymal cell lines. J Nutr Sci Vitaminol (Tokyo) 2003;49:187–194. doi: 10.3177/jnsv.49.187. [DOI] [PubMed] [Google Scholar]
- Terrill JR, Grounds MD, Arthur PG. Taurine deficiency, synthesis and transport in the mdx mouse model for Duchenne Muscular Dystrophy. Int J Biochem Cell Biol. 2015;66:141–148. doi: 10.1016/j.biocel.2015.07.016. [DOI] [PubMed] [Google Scholar]
- Terrill JR, Grounds MD, Arthur PG. Increased taurine in pre-weaned juvenile mdx mice greatly reduces the acute onset of myofibre necrosis and dystropathology and prevents inflammation. PLoS Curr. 2016a;8 doi: 10.1371/currents.md.77be6ec30e8caf19529a00417614a072. ecurrents.md.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terrill JR, Pinniger GJ, Graves JA, Grounds MD, Arthur PG. Increasing taurine intake and taurine synthesis improves skeletal muscle function in the mdx mouse for Duchenne muscular dystrophy. J Physiol. 2016b;594:2095–2110. doi: 10.1113/JP271418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trachtman H, Futterweit S, Maesaka J, Ma C, Valderrama E, Fuchs A, Tarectetecan A, Rao PS, Sturman JA, Boles TH, et al. Taurine ameliorates chronic streptozocin-induced diabetic nephropathy in rats. Am J Physiol. 1995;269:F429–F438. doi: 10.1152/ajprenal.1995.269.3.F429. [DOI] [PubMed] [Google Scholar]
- Ulrich-Merzenich G, Zeitler H, Vetter H, Bhonde RR. Protective effects of taurine on endothelial cells impaired by high glucose and oxidized low density lipoproteins. Eur J Nutr. 2007;46:431–438. doi: 10.1007/s00394-007-0682-7. [DOI] [PubMed] [Google Scholar]
- Venturini A, Ascione R, Lin H, Polesel E, Angelini GD, Suleiman MS. The importance of myocardial amino acids during ischemia and reperfusion in dilated left ventricle of patients with degenerative mitral valve disease. Mol Cell Biochem. 2009;330:63–70. doi: 10.1007/s11010-009-0101-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warskulat U, Flogel U, Jacoby C, Hartwig HG, Thewissen M, Merx MW, Molojavyl A, Heller-Stilb B, Schrader J, Haussinger D. Taurine transporter knockout depletes muscle taurine levels and results in severe skeletal muscle impairment but leaves cardiac function uncompromised. FASEB J. 2004;18:577–579. doi: 10.1096/fj.03-0496fje. [DOI] [PubMed] [Google Scholar]
- Winiarska K, Szmanski K, Gorniak P, Dudziak M, Bryla J. Hypoglycaemic, antioxidative and nephroprotective effects of taurine in alloxan diabetic rabbits. Biochimie. 2009;91:261–270. doi: 10.1016/j.biochi.2008.09.006. [DOI] [PubMed] [Google Scholar]
- Wu F, Koenig KL, Zeleniuch-Jacquotte A, Jonas S, Afanasiyeva Y, Wojcik OP, Costa M, Chen Y. Serum taurine and stroke risk in women: A prospective, nested case-control study. PLoS ONE. 2016;11:e0149348. doi: 10.1371/journal.pone.0149348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H, Jin Y, Wei J, Jin H, Sha D, Wu JY. Mode of action of taurine as a neuroprotector. Brain Res. 2005;1038:123–131. doi: 10.1016/j.brainres.2005.01.058. [DOI] [PubMed] [Google Scholar]
- Wu J, Kohno T, Georgiev SK, Ikoma M, Ishii H, Petrenko AB, Baba H. Taurine activates glycine and gamma-amino-butyric acid A receptors in rat substantia gelatinosa neurons. Neuroreport. 2008;19:333–337. doi: 10.1097/WNR.0b013e3282f50c90. [DOI] [PubMed] [Google Scholar]
- Wu JY, Prentice H. Role of taurine in the central nervous system. J Biomed Sci. 2010;17:S1. doi: 10.1186/1423-0127-17-S1-S1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamori Y, Liu L, Ikeda K, Miura A, Mizushima S, Miki T, Nara Y. Distribution of twenty-four hour urinary taurine excretion and association with ischemic heart disease mortality in 24 populations of 16 countries: results from the WHO-CARDIAC study. Hypertens Res. 2001;24:453–457. doi: 10.1291/hypres.24.453. [DOI] [PubMed] [Google Scholar]
- Yamori Y, Liu L, Mori M, Sagara M, Murakami S, Nara Y, Mizushima S. Taurine as the nutritional factor for the longevity of the Japanese revealed by a world-wide epidemiological survey. Adv Exp Med Biol. 2009;643:13–25. doi: 10.1007/978-0-387-75681-3_2. [DOI] [PubMed] [Google Scholar]
- Yamori Y, Murakami S, Ikeda K, Nara Y. Fish and life-style-related disease prevention: experimental and epidemiological evidence for anti-atherogenic potential of taurine. Clin Expt Pharmacol Physiol. 2004;31:S20–S23. doi: 10.1111/j.1440-1681.2004.04122.x. [DOI] [PubMed] [Google Scholar]
- Yamori Y, Taguchi T, Hamada A, Kunimasa K, Mori H, Mori M. Taurine in health and diseases: consistent evidence from experimental and epidemiological studies. J Biomed Sci. 2010;17:S6. doi: 10.1186/1423-0127-17-S1-S6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagita T, Han SY, Hu Y, Nagao K, Kitajima H, Murakami S. Taurine reduces the secretion of apolipoprotein B100 and lipids in HepG2 cells. Lipids Health Dis. 2008;7:38. doi: 10.1186/1476-511X-7-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yatabe Y, Miyakawa S, Miyazaki T, Matsuzaki Y, Ochiai N. Effects of taurine administration in rat skeletal muscles on exercise. J Orthop Sci. 2003;8:415–419. doi: 10.1007/s10776-002-0636-1. [DOI] [PubMed] [Google Scholar]
- Yokogoshi H, Mochizuki H, Nanami K, Hida Y, Miyachi F, Oda H. Dietary taurine enhances cholesterol degradation and reduces serum and liver cholesterol concentrations in rats fed a high-cholesterol diet. J Nutr. 1999;129:1705–1712. doi: 10.1093/jn/129.9.1705. [DOI] [PubMed] [Google Scholar]
- Yoshimura H, Nariai Y, Etshima M, Mitani T, Tanigawa Y. Taurine suppresses platelet-derived growth factor (PDGF) BB-induced PDGF-beta receptor phosphorylation by protein tyrosine phosphatase-mediated dephosphorylation in vascular smooth muscle cells. Biochim Biophys Acta. 2005;1745:350–360. doi: 10.1016/j.bbamcr.2005.07.005. [DOI] [PubMed] [Google Scholar]
- Zhang L, Yuan Y, Tong Q, Jiang S, Xu Q, Ding J, Zhang L, Zhang R, Zhang K. Reduced plasma taurine level in Parkinson’s disease: association with motor severity and levodopa treatment. Int J Neurosci. 2016;126:630–636. doi: 10.3109/00207454.2015.1051046. [DOI] [PubMed] [Google Scholar]
- Zheng Y, Ceglarek U, Huang T, Wang T, Heianza Y, Ma W, Bray GA, Thiery J, Sacks FM, Qi L. Plasma taurine, diabetes genetic predisposition, and changes of insulin sensitivity in response to weight-loss diets. J Clin Endocrinol Metabol. 2016;101:3820–3826. doi: 10.1210/jc.2016-1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zulli A, Lau E, Wijaya BPP, Jin X, Sutarga K, Schwartz GD, Leamont J, Wookey PJ, Zinellu A, Carru C, Hare DL. High dietary taurine reduces apoptosis and atherosclerosis in the left main coronary artery. Hypertension. 2009;53:1017–1022. doi: 10.1161/HYPERTENSIONAHA.109.129924. [DOI] [PubMed] [Google Scholar]