Abstract
Human stem cells hold significant potential for the treatment of various diseases. However, their use as a therapy is hampered because of limited understanding of the mechanisms by which they respond to environmental stimuli. Efforts to understand extracellular biophysical cues have demonstrated the critical roles of geometrical and mechanical signals in determining the fate of stem cells. The goal of this study was to explore the interplay between cell polarity and matrix stiffness in stem cell lineage specification. We hypothesize that confining cells to asymmetric extracellular matrix (ECM) islands will impart polarity at a single cell-level and will interact with mechanical signals to define the lineage of stem cells. To test these hypotheses, we employed microcontact printing to create patterned symmetric and asymmetric hydrogel islands of soft and hard surface stiffness. Human mesenchymal stem cells (hMSCs) were confined to these islands at the single-cell level and given the ability to differentiate along adipogenic or osteogenic routes. Our results demonstrated that cell polarity defines the lineage specification of hMSCs only on islands with low stiffness. Insight gained from this study provides a rational basis for designing stem cell cultures to enhance tissue engineering and regenerative medicine strategies.
Keywords: Cell polarity, matrix elasticity, micropatterning, mesenchymal stem cells
1. Introduction
Asymmetric cell division is critical in the normal development of organisms, the maintenance of homeostasis, wound healing, and cancer.[10, 18, 46, 47] A balance between symmetric and asymmetric divisions allows organisms to preserve stemness, replenish the population of differentiated cells, and prevent unregulated growth that leads to tumor formation.[29] Studies to understand these divisions during development have mainly focused on C. elegans and Drosophila, demonstrating both intrinsic and extrinsic mechanisms of control. In the intrinsic mechanism, regulators within the cell are unequally distributed during mitosis, effectively polarizing the cell and resulting in daughter cells with different internal signals and fate specifications.[30] In contrast, the extrinsic mechanism of cell division control involves cues from the surrounding niche, in which asymmetric division occurs when the cell divides perpendicular to the niche resulting in one cell that is proximal and the other that is distal.[19] The extrinsic mechanism can also depend on matrix contact when one daughter cell remains bound to the matrix while the other cell is no longer attached and can migrate to distant areas.[33] Therefore, asymmetric division is controlled by cell polarity (defined as asymmetry in cell shape and cytoskeleton and location of proteins and organelles) and cues from the ECM.
Morphological changes occur during cell differentiation, indicating a link between cell shape and function. Ingber et al. showed that disruption of the cellular native morphology using micropatterns of decreasing size prevented the cells from spreading as well as caused apoptosis in endothelial cells.[3, 4] Cell shape can also influence other cellular functions, such as migration, proliferation, and differentiation.[36, 50] Early studies looking at the density of the ECM and how it affects hepatocyte cell shape and spreading concluded that a low density inhibited cell spreading and growth while a high density increased spreading and proliferation and decreased differentiation.[28] Further manipulation of the ECM by Zhang and Kilian showed that confining human mesenchymal stem cells (hMSCs) to small microislands hindered differentiation due to lower actomyosin contractility.[49] In addition, previous work by Kilian had demonstrated a marked effect of shape, and especially curvature, in driving differentiation of hMSCs. In this study, it was shown that shapes that induce high cell contractility promote osteogenesis and low contractility favors adipogenesis.[15] Similar results were found by McBeath et al. who found that shape of cells influenced Ras homolog gene family, member A (RhoA) activity and that this was responsible for lineage commitment.[27] These studies began to unravel the complexities of the extrinsic cues provided by the extracellular matrix, which influence cell shape and contractility, and the mechanisms by which they regulate lineage specification.
The elasticity of the ECM also plays a large role in stem cell fate. Matrices with soft stiffness, similar to bone marrow, cause cells to have small, rounded morphology and be quiescent.[42] In a pioneering study, the link between physiological stiffness and lineage commitment was established by Engler et al., in which he subjected MSCs and myoblasts to gels of different stiffness and found that cell fate specification was based on how well the gel’s stiffness matched that of the physiological tissue.[6, 7] Very soft matrices of up to 1 kPa that mimic the brain microenvironment result in MSC differentiation to neurons. Meanwhile, slightly stiffer matrices of 8–17 kPa cause myogenesis, and stiff matrices of 25–40 kPa lead to osteogenesis.[6] Matrix stiffness can directly influence cell shape by allowing the cell to go from a round morphology to a spread and branched shape as stiffness increases.[9, 24, 34] The interplay between matrix elasticity and shape provide biophysical cues that drive cell division, cell fate, and differentiation.
While matrix elasticity and shape are extrinsic cues for asymmetric division, polarization is an intrinsic control. Polarization is a key factor in normal development, cell differentiation, and tumor suppression.[5, 25] A lack of polarization promotes pluripotency in embryonic stem cells, disrupting normal development.[21] Studies in organisms such as C. elegans and Drosophila have been able to pinpoint the molecules involved in polarization and subsequent asymmetric divisions, and these molecules appear to be conserved in mammals as well.[19] There are various types of polarities (planar, epithelial, apical-basal, immunological, etc.) and each is regulated by different proteins. For example, differentiation and stratification of mammalian skin is caused by the apical localization of aPKC, Par3-LGN-Inscutable complex, and NuMA-dynactin,[22] but in the mammalian hematopoietic system, Notch signaling is responsible for polarity.[19] These polarity cues organize the cytoskeleton and determine the axis of division.[37] In a seminal study, Théry et al. was able to demonstrate that by changing the ECM geometry, polarity was induced in the cell influencing the cell division axis orientation and the organization of organelles within the cell.[39] A different study showed that ECM also helps to establish polarity by signaling through cellular integrin and receptor contacts.[48] These findings suggest that extrinsic cues from the microenvironment can control intrinsic factors associated with cell division and fate.
Asymmetric division is not solely controlled by any of the above, but rather the interplay between all aspects determines the type of cell division or lineage commitment. To deconstruct the interplay between matrix elasticity and geometry, our lab previously used ultraviolet (UV) lithography to create three shapes (circle, square and rectangle) in three different sizes (1000, 2500, and 5000 μm2) featuring three different elasticities (7, 47 and 105 kPa). We found that at the smallest size, elasticity and shape did not play a role in lineage commitment and cells underwent adipogenesis. On the larger sizes, an interplay between shape and elasticity was identified, with shape appearing to play a larger role in fate specification.[8] Lee et al. also showed a connection between shape and matrix stiffness with osteogenesis, demonstrating that shape could enhance the amount of osteogenesis observed as the matrix stiffness increased.[23] Previous work has also shown MSCs can modulate their lineage commitment when there is a shift in their matrix stiffness. The study found that switching stem cells from soft to stiff matrix changed the expression of lineage markers from neurogenic to osteogenic. Furthermore, a shift from an unpatterned matrix to a patterned matrix could enhance the change in lineage marker expression depending on the shape, indicating that cell geometry provides important cues for lineage specification.[24] While multiple studies have found a connection between matrix stiffness and cell shape, there have been a lack of studies on the interplay between polarization and matrix stiffness and their effect on cell differentiation.
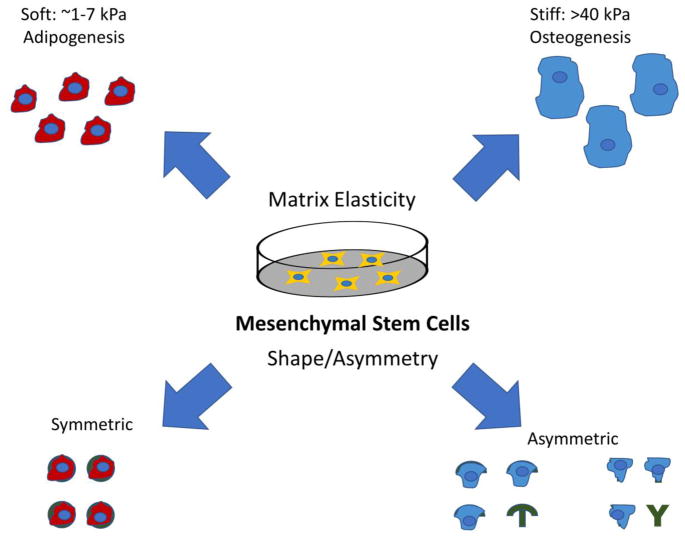
In this study, we aim to elucidate the dynamics between polarity, matrix stiffness, and lineage commitment of hMSCs. Micropatterning techniques were used to create polyethylene glycol (PEG) hydrogels of soft (~5 kPa) and hard (~230 kPa) stiffness and patterns featuring different shapes (O, Y and T) to induce cell polarity, Figure 1. By exposing hMSCs to the different combinations of matrix stiffness and ECM shape, we were able to test two central hypotheses: (1) extrinsic cues from the ECM geometry can induce internal cell polarity and (2) the sensitivity of cells to geometric polarity signals is dependent on the stiffness of ECM. The hydrogel stiffness chosen span ranges known to induce adipogenesis and osteogenesis and the shapes range from nonpolar circles with multiaxial symmetry to more polarizing shapes such as T and Y with only one axis of symmetry, subsequently referred to as “asymmetric”. Our work shows that cell polarity induced by ECM geometry provides osteogenic inductive signals at low matrix stiffness.
Figure 1.
Schematic of the effects of matrix elasticity and cell asymmetry on mesenchymal stem cell lineage.
2. Materials and Methods
Surface preparation
Glass slides (22 × 22 mm, VWR) were cleaned with 70% ethanol for 10 minutes and dried. The slides were then sputter coated with a 5 nm layer of titanium (Denton Desk II TSC, Moorestown, NJ) followed by a 40 nm layer of gold (Denton Desk II, Moorestown, NJ). The coated slides were stored at room temperature until further use.
Micropattern fabrication
Micropatterns were designed using Autocad software (Autodesk, San Rafael, CA) and printed on transparencies (CAD/Art Services, Inc. Bandon, OR) to create photomasks. A hydrogel precursor, consisting of 700 MW PEG-DA (Sigma-Aldrich, St. Louis, MO), 2,000 MW 4-arm PEG-SH (CreativePEGWorks, Chapel Hill, NC), 1% 2-hydroxy-2-methylpropriophenone (Sigma-Aldrich, St. Louis, MO), and water, was placed on the gold slides followed by the photomask and allowed to polymerize under UV light (UVP, Upland, CA). The unpatterned regions were passivated with triethylene glycol mono-11-mercaptoundecyl ether (Sigma-Aldrich, St. Louis, MO) to prevent protein binding. The slides were then rinsed with 70% ethanol followed by phosphate-buffered saline (PBS) before protein incubation. Fibronectin was allowed to conjugate with heterogeneous maleimide/N-hydroxysuccinimide crosslinker (Thermo Fisher, Waltham, MA) for one hour at room temperature before using a ZebaSpin desalting column (Thermo Fisher, Waltham, MA) to separate the functionalized protein from unreacted crosslinker. Conjugated fibronectin was then incubated on the patterned slides for 4 hours at room temperature and then overnight at 4 °C.
Hydrogel mechanical characterization
Hydrogel discs of approximately 6.5 mm in diameter and 2 mm in height were created using the same formulation as mentioned previously. The discs were incubated at 37 °C for 48 hours prior to testing under unconfined compression at 0.05 mm/s using an Electroforce 3200 (Bose, New Castle, DE). The Young’s modulus was then calculated based on the force applied and displacement measured.
Cell culture
Human bone-marrow derived mesenchymal stem cells were purchased from Lonza (Walkersville, MD). The hMSCs were grown in mesenchymal stem cell basal medium (Lonza, Walkersville, MD) supplemented with MSCGM™ SingleQuots™ (mesenchymal cell growth supplement, L-glutamine, and GA-1000) in T-75 culture flasks. The cells were allowed to reach 80% confluence before passaging with 0.25% trypsin-EDTA (Corning, Manassas, VA). Cells up to passage 6 were used and seeded on substrates at 5000 cells/cm2. Cells were given up to a day to attach before switching to differentiation media consisting of 1:1 adipogenic to osteogenic medium. Adipogenic medium was made using Dulbecco’s Modified Eagle Medium (DMEM) (Invitrogen, Carlsbad, CA), 10% fetal bovine serum (FBS) (FB Essence, VWR, Radnor, PA), 1 μM dexamethasone (Sigma-Aldrich, St. Louis, MO), 10 μM insulin (Sigma-Aldrich, St. Louis, MO), 200 μM indomethacin (Sigma-Aldrich, St. Louis, MO), 0.5 mM isobutyl-methylxanthine (Sigma-Aldrich, St. Louis, MO), and 1% penicillin/streptomycin (Hyclone, Chicago, IL). The osteogenic medium contained DMEM/F-12 (Hyclone, Chicago, IL), 10% FBS (FB Essence, VWR, Radnor, PA), 10 mM β-glycerophosphate (Sigma-Aldrich, St. Louis, MO), 50 μg/mL ascorbic acid (Sigma-Aldrich, St. Louis, MO), 1 μM dexamethasone, and 1% penicillin/streptomycin (Hyclone, Chicago, IL). Inhibition media was made by adding 1 μM nocodazole (Sigma-Aldrich, St. Louis, MO) or 2 μM Y-27632 (Calbiochem CAS, San Diego, CA) to the differentiation media and changed daily.
Immunocytochemistry and histological staining
Cells were fixed with 4% paraformaldehyde (Sigma-Aldrich, St. Louis, MO), permeabilized with 0.1% Triton X-100 in PBS, and blocked using 1% BSA. The actin cytoskeleton, nucleus, and LGN were stained with phalloidin-rhodamine (Sigma-Aldrich, St. Louis, MO), DAPI (Sigma-Aldrich, St. Louis, MO), and anti-GPSM2 (Sigma-Aldrich, St. Louis, MO), respectively. Fluorescent images were captured using a Nikon eclipse 80i microscope with CoolSnap HQ camera. Fate specification was determined with dual staining of alkaline phosphatase and Oil Red O for osteogenesis and adipogenesis, respectively, and imaged using a Nikon E600 microscope with a color camera. Cells with lipid vacuoles stained red and were considered adipocytes. Cells that stained deep purple were determined to be osteoblasts.
Statistics
Values graphed represent mean ±SEM. Statistical analysis was performed using Microsoft Excel. One factor ANOVA and two-tailed student t-test were used to calculate p-values. Data was from at least two independent experiments with at least 50 cells for each condition. Significance was set at p < 0.05.
3. Results
3.1 Cell density and matrix elasticity both influence hMSC differentiation
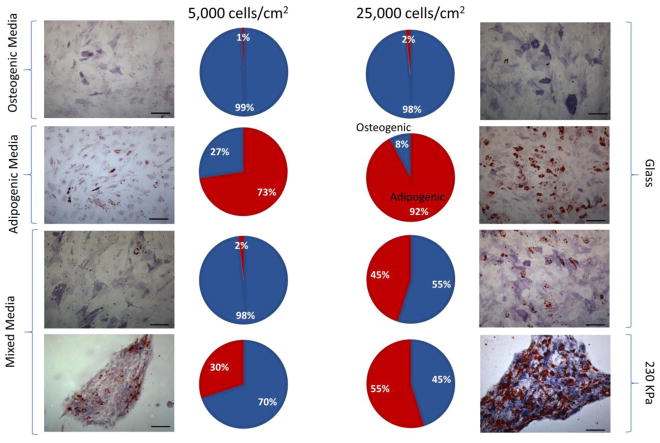
Researchers have shown that MSC cell fate is influenced by seeding density and the stiffness of the environment.[8, 32] When hMSCs were seeded onto glass coverslips and exposed to lineage specific medium, a difference was observed in the percent of cells that differentiated to adipocytes and osteoblasts based on whether the cells were seeded at low (5,000 cells/cm2) or high densities (25,000 cells/cm2) (Figure 2). In strictly adipogenic medium, an increase in seeding density increased adipogenesis from 72.6±7.4% at low density to 92.1±0.9% at high density. Seeding density had less of an effect on osteogenesis in osteogenic medium, with 99.4±1.0% of differentiated cells identified as osteoblasts under low seeding density and 98.4±0.8% at high seeding density. When exposed to a 1:1 ratio of adipogenic to osteogenic medium, the seeding density caused a decrease in osteogenesis, from 98.2±0.9% at low density to 54.9±2.2% at high density (Figure 2). These results indicate high density seeding favors adipogenic differentiation, which is consistent with previous findings.[32] To determine the effects of matrix elasticity, 20% PEG hydrogels (230 kPa) were made and the cells were exposed to the mixed adipogenic/osteogenic medium. This resulted in 69.9±3.4% of the cells differentiating to osteoblasts at low seeding density and 45.2±3.4% at high seeding density, demonstrating that softer substrates favor adipogenesis (Figure 2). These results indicate that matrix stiffness and seeding density, which affect cell-cell contact, cell shape, and cell size, are all important cues in hMSC differentiation.
Figure 2.
Human mesenchymal stem cells (hMSCs) are capable of osteogenic and adipogenic lineage specification when exposed to medium containing growth factors for both. Fate specification capabilities were tested after 10 days in osteogenic medium alone, adipogenic medium alone, mixed medium on glass, and mixed medium on 20% PEG hydrogel, as demonstrated by dual staining of alkaline phosphatase (blue/purple) and Oil Red O (red lipid deposits). All conditions were tested with two seeding densities. The pie graphs show the percent osteogenesis (blue) and adipogenesis (red) for each condition. Data was collected from two independent experiments counting four arbitrary regions of the coverslip. Scale bars are 200 μm.
3.2 Micropatterns induce polarization of LGN protein and cytoskeleton in hMSCs
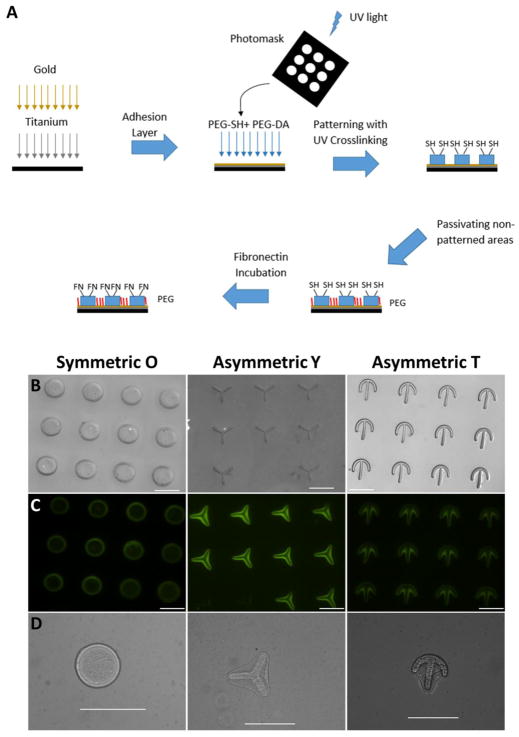
To control individual hMSC cell polarity and shape while preventing cell-cell contact, we used UV lithography to micropattern three different configurations (O, T, and Y) on a hydrogel surface (Figure 3A–B). The patterning process begins by creating a gold adhesion layer on a glass slide, followed by deposition of the hydrogel precursor, which was composed of polyethylene glycol diacrylate (PEG-DA) and PEG-SH. Subsequent UV exposure through a photomask enabled crosslinking polymer in the desired regions. The non-patterned areas were then passivated to prevent protein or cells from binding. Functionalized fibronectin was then incubated with the polymer, selectively binding to the exposed thiol groups, to render the patterned regions cell adhesive. To ensure preferential binding of the protein to the patterned regions, fluorescein isothiocyanate (FITC) conjugated bovine serum albumin was functionalized and incubated on the patterned hydrogels. As shown in Figure 3C, only the patterned regions were fluorescent, indicating a non-adhesive background. Stem cells were seeded onto the patterned substrate and allowed to adhere. Individual cells were then able to spread and assume the shape of the underlying pattern (Figure 3D).
Figure 3.
Schematic of the patterning process. (A) UV lithography was used to create consistent patterns in the shape of the letters O, Y, and T. (B) Thiol groups allow selective binding of the adhesive fibronectin protein to patterned areas, as confirmed using fluorescent bovine serum albumin (C). Individual hMSCs adhered and conformed to the patterned regions. Scale is 100 μm.
To confirm polarization, the nucleus was localized by staining with 4′,6-Diamidino-2-Phenylindole (DAPI), the organization of the cytoskeleton was examined by staining for F-actin and the localization of polarity protein G protein signaling modulator 2 (GPSM2), was determined by staining with anti-LGN antibody (Figure 4). LGN localization was of interest because it has been shown to be important in establishing cellular polarity with lack of polarization resulting in disease.[14, 22, 41] Furthermore, LGN levels and localization dictate cell migration, focal adhesions and differentiation.[13, 20, 45] On the symmetric patterns (O), the stained images showed the actin cytoskeleton was evenly distributed throughout and the nucleus was close to the center of the cell. Additionally, the polarity protein, LGN, was distributed around the nucleus, indicating the hMSCs were non-polarized. In contrast, we observed polarization of the hMSCs that had spread on the T and Y shapes. On the asymmetric T shape, the nucleus was no longer localized in the center of the cell, but rather was distal to the adhesive regions. The actin cytoskeleton was aligned along the non-adhesive sides in agreement with a previous study by Théry.[39] The LGN protein localized near the nucleus, resulting in an asymmetric distribution towards the non-adhesive region. Similarly, the actin cytoskeleton of the cells on Y shaped patterns aligned along the non-adhesive regions, with the nucleus polarized to one side and the LGN protein concentrated around the nucleus. Thus, we were able to establish cytoskeletal polarization and internal polarization of the organelles and proteins.
Figure 4.
Human mesenchymal stem cells (hMSCs) adopt the shape of the underlying hydrogel patterns, with asymmetric configurations resulting in internal polarization. LGN polarity protein (green) is localized near the nucleus, and the actin cytoskeleton aligns along the non-adhesive regions. Scale bars are 100 μm.
3.3 Polarization and matrix stiffness both contribute to hMSC lineage commitment
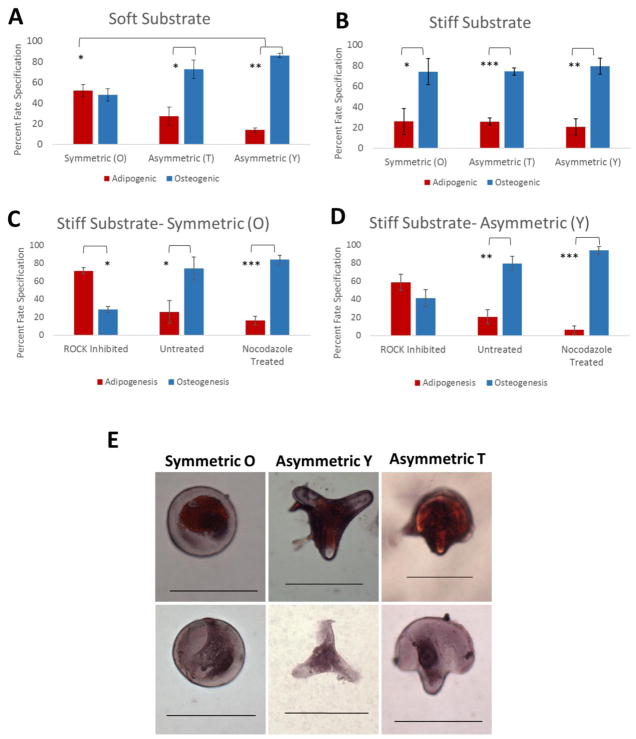
To understand the combined roles of matrix elasticity and cell polarization on hMSC lineage commitment, we cultured asymmetric and symmetric patterned cells on hydrogels featuring soft (~5 kPa) and hard (~230 kPa) stiffness for 10 days in mixed adipogenic/osteogenic differentiation media. Cells were fixed and stained for alkaline phosphatase and lipid deposits using Oil Red O and counted. Cells which stained positive for alkaline phosphatase were purple and counted as osteoblasts, while cells with lipid deposits were stained red and counted as adipocytes (Figure 5E). On the soft matrix, the symmetric cell shape (O) resulted in a mixed population of adipocytes (52.1%) and osteoblasts (47.9%) (Figure 5A). In contrast, confinement to asymmetric hydrogel microislands shifted fate specification toward osteogenesis. Cells on the asymmetric T shape microislands had a decrease of adipogenesis to 27.4% and an increase of osteogenesis to 72.6%. Cells on the Y shape had an even greater bias toward osteogenesis, with 86.0% of cells identified as osteoblasts and only 14.0% as adipocytes. The significant increase in osteogenesis on asymmetric patterns in the soft matrix shows that cell polarity signals have the capability to tune cell fate specification and differentiation under these conditions.
Figure 5.
Asymmetry and matrix elasticity both play a role in hMSC lineage commitment. (A) Asymmetry has a significant role in osteogenesis in the soft matrix (n=4) (B) High matrix stiffness overrides asymmetry signals resulting in similar osteogenesis in all three shapes (n=4) (C–D) Cytoskeletal inhibitor Y-27632 (ROCK inhibitor) can override matrix stiffness, favors adipogenesis in the O and disrupts the osteogenic bias of the Y shape. Nocodazole treatment enhances osteogenesis in both shapes. (E) Representative images of adipocytes and osteoblasts on O, T, and Y shapes. Bars represent mean ± standard error with more than 50 cells per condition. Error bars are the standard error from at least two separate experiments with more than 50 cells per condition. Statistical significance was evaluated using one-way ANOVA, * = p < 0.05, ** = p < 0.01, *** = p < 0.001. Scale bars are 100 μm.
However, on the stiff matrix, we observed no significant difference in terms of lineage specification between cells cultured on symmetric and asymmetric ECM islands (Figure 5B). Osteogenesis for all shapes was similar, with cells on the symmetric shape having the least osteogenesis at 74.1%. MSCs on asymmetric patterns resulted in a slight increase of osteogenesis to 74.2% in cells cultured on T and 79.4% of cells on the Y shape patterns. Clearly, at a high enough matrix stiffness, cells are biased toward osteogenesis and are not influenced by geometric asymmetry signals.
3.4 Cytoskeletal inhibitors dampen asymmetric signaling
To confirm the role of matrix mechanics in cell fate decisions, we used pharmaceuticals Y-27632 and nocodazole to disrupt the cytoskeleton and make the cell insensitive to the matrix stiffness. Y-27632 is a ROCK inhibitor that has been shown to diminish stress fibers in cells and decrease cytoskeletal tension.[11, 17] On the other hand, nocodazole stabilizes microtubules and increases the cell contractility by activating RhoA and ROCK.[2, 40] We seeded the hMSCs onto the representative symmetric (O) and asymmetric (Y) PEG hydrogels and allowed them to adhere and spread in growth medium before switching to mixed adipogenic/osteogenic differentiation medium containing either inhibitor. After 10 days, the percent of osteogenic cells on the symmetric patterns treated with Y-27632 decreased from 74.1% to 28.6% (Figure 5C). In contrast, after 10 days of nocodazole treatment osteogenesis increased from 74.1% to 83.8%. Similarly, Y-27632 decreased the percent of osteogenesis in the asymmetric Y shape from 79.4% to 41.4% and nocodazole treatment increased osteogenesis from 79.4% to 93.8% (Figure 5D). These results indicate that ROCK inhibition was capable of switching the differentiation trend from osteogenesis to adipogenesis, while nocodazole treatment enhanced osteogenesis. The difference in osteogenesis and adipogenesis in the ROCK inhibited group was significant in the symmetric shape but not significant for the asymmetric shapes, implying that asymmetry in the cell can still partially drive osteogenesis when matrix stiffness is no longer a factor.
4. Discussion
In this study, we used micropatterning to determine the combinational roles of cell polarity and matrix stiffness on hMSC differentiation. By creating symmetric and asymmetric shapes on patterned hydrogels with tunable mechanical properties, we were able to isolate each effect at the individual cell level. Our patterned hydrogel microislands were able to control environmental cues, such as cell size, cell-cell contact, and ligand density, which have been shown to influence stem cell behavior.[16, 27, 31] hMSCs were able to survive on the hydrogel patterns for 10 days and differentiate into adipogenic or osteogenic lineages. The cells were able to integrate signals resulting from polarization due to ECM geometry and matrix stiffness to determine lineage specification.
While the role of biophysical cues in differentiation have been well studied, the effect of polarization is still poorly understood. Studies have shown that a lack of polarization can lead to improper development of the cochlea (resulting in deafness), polycystic kidney disease, and cancer. [35, 43] A better understanding of how to induce polarity in cells could help to prevent such diseases. Théry et al. showed that micropatterns were capable of polarizing the internal organization of the cell including the nucleus, Golgi apparatus, centrosome, and microtubules, concluding that matrix geometry had the ability to induce internal cell polarity in endothelial cells.[39] Our study showed that polarization of stem cells could be achieved using similar micropatterns. The patterns not only polarized the actin cytoskeleton, but also affected the internal organization of the organelles and polarity protein distribution (Figure 4). This proved that matrix geometry provides the cell with cues that can change its internal organization, including mitotic spindle orientation, and can drive asymmetric divisions and fate specification.[1, 38] This platform could be used to study effects of polarization at the single cell level in other biological systems such epithelial-mesenchymal transition.
Our results demonstrate an interplay between polarity and matrix elasticity in regulation of cell processes. On a soft matrix (~5 kPa), polarity had the greatest impact on fate specification. According to our results, symmetry does not bias specification to adipocytes or osteoblasts, while induction of polarity with asymmetric micropatterns favored osteogenesis (Figure 5A). This bias toward osteogenesis suggests polarity activates signaling pathways that either inhibit adipogenesis or promote osteogenesis. Our findings suggest that at low matrix stiffness, the cell receives signals from polarity cues that drive cells toward an osteogenic fate.
Polarity signaling, however, has its limitations. At the higher matrix stiffness (~230 kPa), we observed the role of polarity signaling to disappear. All shapes, polar and non-polar, were biased toward osteogenesis and resulted in similar proportions of osteogenic cells. This shows that matrix stiffness can override polarity signaling. High matrix stiffness has been shown to activate ROCK/RhoA signaling, which is responsible for osteogenesis and mechanotransduction.[12, 26, 44] Thus, matrix stiffness and polarity could work through the same ROCK/RhoA signaling pathway, with high matrix stiffness playing a larger role in activating it and thus resulting in regions where polarity does not provide the cell with differentiation cues.
This study used the Young’s modulus to characterize the mechanics of the hydrogel. While this measurement does not provide full details of the mechanical properties of the hydrogel or give insight into ECM and cell dynamic interactions, its purpose was to create two different environments that could bias osteogenic and adipogenic differentiation. Further characterization of the hydrogels after cell differentiation could help to understand how the cells interact with their matrix. To develop better stem cell therapies or regenerative medicine strategies, it would be important to understand how these cells behave in situ and create hydrogels that mimic these matrix properties. Nonetheless, the difference in cell behavior based on matrix stiffness was evident and showed that a better understanding of ECM mechanics are required to further explain cell differentiation.
Biophysical cues have been known to play a major role in stem cell differentiation. Here we show that the interplay between cell polarity and matrix stiffness can drive osteogenesis in hMSCs. By beginning to unravel this relationship, we can start to understand the intricacies of development and regenerative medicine. We can further test the behavior of other cells of interest (e.g., embryonic stem cells) to see how perturbations in polarity or matrix stiffness can cause developmental defects or disease.
Acknowledgments
Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number AR063338. We would like to thank Dr. Tarek Shazly and Will Torres for assisting us with the use of the Electroforce 3200.
Contributor Information
Maria E. Piroli, Biomedical Engineering Program, University of South Carolina, SC 29208
Ehsan Jabbarzadeh, Department of Chemical Engineering, Biomedical Engineering Program, University of South Carolina, SC 29208.
References
- 1.Ben-Yair R, Kahane N, Kalcheim C. Lgn-dependent orientation of cell divisions in the dermomyotome controls lineage segregation into muscle and dermis. Development. 2011;138:4155–66. doi: 10.1242/dev.065169. [DOI] [PubMed] [Google Scholar]
- 2.Chang YC, Nalbant P, Birkenfeld J, Chang ZF, Bokoch GM. Gef-h1 couples nocodazole-induced microtubule disassembly to cell contractility via rhoa. Mol Biol Cell. 2008;19:2147–53. doi: 10.1091/mbc.E07-12-1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chen CS, Mrksich M, Huang S, Whitesides GM, Ingber DE. Geometric control of cell life and death. Science. 1997;276:1425–8. doi: 10.1126/science.276.5317.1425. [DOI] [PubMed] [Google Scholar]
- 4.Chen CS, Mrksich M, Huang S, Whitesides GM, Ingber DE. Micropatterned surfaces for control of cell shape, position, and function. Biotechnol Prog. 1998;14:356–63. doi: 10.1021/bp980031m. [DOI] [PubMed] [Google Scholar]
- 5.Drubin DG, Nelson WJ. Origins of cell polarity. Cell. 1996;84:335–44. doi: 10.1016/s0092-8674(00)81278-7. [DOI] [PubMed] [Google Scholar]
- 6.Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126:677–89. doi: 10.1016/j.cell.2006.06.044. [DOI] [PubMed] [Google Scholar]
- 7.Guilak F, Cohen DM, Estes BT, Gimble JM, Liedtke W, Chen CS. Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell. 2009;5:17–26. doi: 10.1016/j.stem.2009.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Harris GM, Piroli ME, Jabbarzadeh E. Deconstructing the effects of matrix elasticity and geometry in mesenchymal stem cell lineage commitment. Adv Funct Mater. 2014;24:2396–2403. doi: 10.1002/adfm.201303400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Harris GM, Shazly T, Jabbarzadeh E. Deciphering the combinatorial roles of geometric, mechanical, and adhesion cues in regulation of cell spreading. PLoS ONE. 2013;8:e81113. doi: 10.1371/journal.pone.0081113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Horvitz HR, Herskowitz I. Mechanisms of asymmetric cell division: Two bs or not two bs, that is the question. Cell. 1992;68:237–55. doi: 10.1016/0092-8674(92)90468-r. [DOI] [PubMed] [Google Scholar]
- 11.Ishizaki T, Uehata M, Tamechika I, Keel J, Nonomura K, Maekawa M, Narumiya S. Pharmacological properties of y-27632, a specific inhibitor of rho-associated kinases. Mol Pharmacol. 2000;57:976–83. [PubMed] [Google Scholar]
- 12.James AW. Review of signaling pathways governing msc osteogenic and adipogenic differentiation. Scientifica (Cairo) 2013;2013:684736. doi: 10.1155/2013/684736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kamakura S, Nomura M, Hayase J, Iwakiri Y, Nishikimi A, Takayanagi R, Fukui Y, Sumimoto H. The cell polarity protein minsc regulates neutrophil chemotaxis via a noncanonical g protein signaling pathway. Dev Cell. 2013;26:292–302. doi: 10.1016/j.devcel.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 14.Kaushik R, Yu F, Chia W, Yang X, Bahri S. Subcellular localization of lgn during mitosis: Evidence for its cortical localization in mitotic cell culture systems and its requirement for normal cell cycle progression. Molecular Biology of the Cell. 2003;14:3144–3155. doi: 10.1091/mbc.E03-04-0212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kilian KA, Bugarija B, Lahn BT, Mrksich M. Geometric cues for directing the differentiation of mesenchymal stem cells. Proc Natl Acad Sci U S A. 2010;107:4872–7. doi: 10.1073/pnas.0903269107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kilian KA, Mrksich M. Directing stem cell fate by controlling the affinity and density of ligand-receptor interactions at the biomaterials interface. Angew Chem Int Ed Engl. 2012;51:4891–5. doi: 10.1002/anie.201108746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kimura K, Ito M, Amano M, Chihara K, Fukata Y, Nakafuku M, Yamamori B, Feng J, Nakano T, Okawa K, Iwamatsu A, Kaibuchi K. Regulation of myosin phosphatase by rho and rho-associated kinase (rho-kinase) Science. 1996;273:245–8. doi: 10.1126/science.273.5272.245. [DOI] [PubMed] [Google Scholar]
- 18.Knoblich JA. Asymmetric cell division: Recent developments and their implications for tumour biology. Nat Rev Mol Cell Biol. 2010;11:849–60. doi: 10.1038/nrm3010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Knoblich JA. Mechanisms of asymmetric stem cell division. Cell. 2008;132:583–597. doi: 10.1016/j.cell.2008.02.007. [DOI] [PubMed] [Google Scholar]
- 20.Konno D, Shioi G, Shitamukai A, Mori A, Kiyonari H, Miyata T, Matsuzaki F. Neuroepithelial progenitors undergo lgn-dependent planar divisions to maintain self-renewability during mammalian neurogenesis. Nat Cell Biol. 2008;10:93–101. doi: 10.1038/ncb1673. [DOI] [PubMed] [Google Scholar]
- 21.Krtolica A, Genbacev O, Escobedo C, Zdravkovic T, Nordstrom A, Vabuena D, Nath A, Simon C, Mostov K, Fisher SJ. Disruption of apical-basal polarity of human embryonic stem cells enhances hematoendothelial differentiation. Stem Cells. 2007;25:2215–23. doi: 10.1634/stemcells.2007-0230. [DOI] [PubMed] [Google Scholar]
- 22.Lechler T, Fuchs E. Asymmetric cell divisions promote stratification and differentiation of mammalian skin. Nature. 2005;437:275–80. doi: 10.1038/nature03922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee J, Abdeen AA, Huang TH, Kilian KA. Controlling cell geometry on substrates of variable stiffness can tune the degree of osteogenesis in human mesenchymal stem cells. Journal of the Mechanical Behavior of Biomedical Materials. 2014;38:209–218. doi: 10.1016/j.jmbbm.2014.01.009. [DOI] [PubMed] [Google Scholar]
- 24.Lee J, Abdeen AA, Kilian KA. Rewiring mesenchymal stem cell lineage specification by switching the biophysical microenvironment. Scientific reports. 2014;4 doi: 10.1038/srep05188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee M, Vasioukhin V. Cell polarity and cancer--cell and tissue polarity as a non-canonical tumor suppressor. J Cell Sci. 2008;121:1141–50. doi: 10.1242/jcs.016634. [DOI] [PubMed] [Google Scholar]
- 26.Ling L, Nurcombe V, Cool SM. Wnt signaling controls the fate of mesenchymal stem cells. Gene. 2009;433:1–7. doi: 10.1016/j.gene.2008.12.008. [DOI] [PubMed] [Google Scholar]
- 27.McBeath R, Pirone DM, Nelson CM, Bhadriraju K, Chen CS. Cell shape, cytoskeletal tension, and rhoa regulate stem cell lineage commitment. Developmental cell. 2004;6:483–495. doi: 10.1016/s1534-5807(04)00075-9. [DOI] [PubMed] [Google Scholar]
- 28.Mooney D, Hansen L, Vacanti J, Langer R, Farmer S, Ingber D. Switching from differentiation to growth in hepatocytes: Control by extracellular matrix. J Cell Physiol. 1992;151:497–505. doi: 10.1002/jcp.1041510308. [DOI] [PubMed] [Google Scholar]
- 29.Morrison SJ, Kimble J. Asymmetric and symmetric stem-cell divisions in development and cancer. Nature. 2006;441:1068–74. doi: 10.1038/nature04956. [DOI] [PubMed] [Google Scholar]
- 30.Mukherjee S, Kong J, Brat DJ. Cancer stem cell division: When the rules of asymmetry are broken. Stem Cells Dev. 2015;24:405–16. doi: 10.1089/scd.2014.0442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nelson CM, Chen CS. Cell-cell signaling by direct contact increases cell proliferation via a pi3k-dependent signal. FEBS Lett. 2002;514:238–42. doi: 10.1016/s0014-5793(02)02370-0. [DOI] [PubMed] [Google Scholar]
- 32.Peng R, Yao X, Cao B, Tang J, Ding J. The effect of culture conditions on the adipogenic and osteogenic inductions of mesenchymal stem cells on micropatterned surfaces. Biomaterials. 2012;33:6008–19. doi: 10.1016/j.biomaterials.2012.05.010. [DOI] [PubMed] [Google Scholar]
- 33.Pham K, Sacirbegovic F, Russell SM. Polarized cells, polarized views: Asymmetric cell division in hematopoietic cells. Front Immunol. 2014;5:26. doi: 10.3389/fimmu.2014.00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rowlands AS, George PA, Cooper-White JJ. Directing osteogenic and myogenic differentiation of mscs: Interplay of stiffness and adhesive ligand presentation. Am J Physiol Cell Physiol. 2008;295:C1037–44. doi: 10.1152/ajpcell.67.2008. [DOI] [PubMed] [Google Scholar]
- 35.Simons M, Mlodzik M. Planar cell polarity signaling: From fly development to human disease. Annual review of genetics. 2008;42:517. doi: 10.1146/annurev.genet.42.110807.091432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Thery M. Micropatterning as a tool to decipher cell morphogenesis and functions. J Cell Sci. 2010;123:4201–13. doi: 10.1242/jcs.075150. [DOI] [PubMed] [Google Scholar]
- 37.Thery M, Bornens M. Cell shape and cell division. Curr Opin Cell Biol. 2006;18:648–57. doi: 10.1016/j.ceb.2006.10.001. [DOI] [PubMed] [Google Scholar]
- 38.Thery M, Jimenez-Dalmaroni A, Racine V, Bornens M, Julicher F. Experimental and theoretical study of mitotic spindle orientation. Nature. 2007;447:493–496. doi: 10.1038/nature05786. [DOI] [PubMed] [Google Scholar]
- 39.Thery M, Racine V, Piel M, Pepin A, Dimitrov A, Chen Y, Sibarita JB, Bornens M. Anisotropy of cell adhesive microenvironment governs cell internal organization and orientation of polarity. Proc Natl Acad Sci U S A. 2006;103:19771–6. doi: 10.1073/pnas.0609267103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vasquez RJ, Howell B, Yvon AM, Wadsworth P, Cassimeris L. Nanomolar concentrations of nocodazole alter microtubule dynamic instability in vivo and in vitro. Mol Biol Cell. 1997;8:973–85. doi: 10.1091/mbc.8.6.973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Walsh T, Shahin H, Elkan-Miller T, Lee MK, Thornton AM, Roeb W, Abu Rayyan A, Loulus S, Avraham KB, King M-C, Kanaan M. Whole exome sequencing and homozygosity mapping identify mutation in the cell polarity protein gpsm2 as the cause of nonsyndromic hearing loss dfnb82. The American Journal of Human Genetics. 2010;87:90–94. doi: 10.1016/j.ajhg.2010.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Winer JP, Janmey PA, McCormick ME, Funaki M. Bone marrow-derived human mesenchymal stem cells become quiescent on soft substrates but remain responsive to chemical or mechanical stimuli. Tissue Eng Part A. 2009;15:147–54. doi: 10.1089/ten.tea.2007.0388. [DOI] [PubMed] [Google Scholar]
- 43.Wodarz A, Näthke I. Cell polarity in development and cancer. Nature Cell Biology. 2007;9:1016. doi: 10.1038/ncb433. [DOI] [PubMed] [Google Scholar]
- 44.Wozniak MA, Chen CS. Mechanotransduction in development: A growing role for contractility. Nat Rev Mol Cell Biol. 2009;10:34–43. doi: 10.1038/nrm2592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wright CE, Kushner EJ, Du Q, Bautch VL. Lgn directs interphase endothelial cell behavior via the microtubule network. PLoS One. 2015;10:e0138763. doi: 10.1371/journal.pone.0138763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yamashita YM, Yuan H, Cheng J, Hunt AJ. Polarity in stem cell division: Asymmetric stem cell division in tissue homeostasis. Cold Spring Harb Perspect Biol. 2010;2:a001313. doi: 10.1101/cshperspect.a001313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yang S, Ma K, Geng Z, Sun X, Fu X. Oriented cell division: New roles in guiding skin wound repair and regeneration. Biosci Rep. 2015;35 doi: 10.1042/BSR20150225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yeaman C, Grindstaff KK, Nelson WJ. New perspectives on mechanisms involved in generating epithelial cell polarity. Physiol Rev. 1999;79:73–98. doi: 10.1152/physrev.1999.79.1.73. [DOI] [PubMed] [Google Scholar]
- 49.Zhang D, Kilian KA. The effect of mesenchymal stem cell shape on the maintenance of multipotency. Biomaterials. 2013;34:3962–9. doi: 10.1016/j.biomaterials.2013.02.029. [DOI] [PubMed] [Google Scholar]
- 50.Zhong Y, Ji B. Impact of cell shape on cell migration behavior on elastic substrate. Biofabrication. 2013;5:015011. doi: 10.1088/1758-5082/5/1/015011. [DOI] [PubMed] [Google Scholar]