Abstract
Objectives
To review the available literature on medication-related osteonecrosis of the jaw (MRONJ) associated with antiangiogenics in antiresorptive-naïve individuals.
Methods
A literature search was performed using MEDLINE via PubMed, EMBASE, and Web of Science in December 2017.
Results
We identified reports describing a total of 35 antiresorptive drugs-naïve patients who developed antiangiogenic-related MRONJ. The mean age of these patients was 59.06 years and the F : M ratio was 4 : 5. The most common underlying disease was metastatic renal cell cancer. Pain to the mandible was the most common complaint (34.29%) and the majority of patients presented with bone exposure. The mean duration of intravenous and oral antiangiogenics before MRONJ development was 6.5 and 16.72 months, respectively. The most common additional risk factor was dental extraction (37.14%). Almost half of the MRONJ patients (48.57%) received surgical treatment. 18 patients (62.06%) were reported to have disease resolution within an average time of 6.75 months.
Conclusion
MRONJ associated with antiangiogenic therapy in antiresorptive-naïve patients is a rare but potentially serious adverse effect. Available data suggests that there might be notable differences between MRONJ associated with antiangiogenics and antiresorptives; however, further prospective well-designed studies are required.
1. Introduction
Medication-related osteonecrosis of the jaw (MRONJ) is an uncommon and potentially serious adverse side effect of antiresorptive and antiangiogenic agents [1]. It can cause chronic pain, infection, dysfunction, and disfigurement and can affect the quality of life of affected individuals [2, 3]. The vast majority of cases of MRONJ are associated with antiresorptive agents including bisphosphonates, denosumab, and more recently romosozumab [4–7]. A notably smaller number of cases are associated with the use of antiangiogenic agents, both in individuals who also take antiresorptive drugs and in those who are antiresorptive drugs-naïve [8]. MRONJ can develop in approximately 7% of cancer patients taking high-potency bisphosphonates or high-dose denosumab and about 0.01–0.1% of those with osteoporosis using low-potency oral bisphosphonates or low-dosage denosumab [1, 9–12]. The use of antiangiogenic agents in combination with antiresorptive drugs is known to increase the risk of MRONJ development [13]; however, little is known regarding the incidence and prevalence of antiangiogenic-related MRONJ in antiresorptive drugs-naïve individuals. Antiangiogenic inhibitors have been increasingly used in the management of a range of malignancies including ovarian cancer, metastatic renal cell cancer, breast cancer, colorectal cancer, non-small-cell lung cancer (NSCLC), and glioblastoma multiforme [14]. Antiangiogenic inhibitors can be categorised into three major groups based on their mechanism of action: anti-VEGF monoclonal antibody (e.g., bevacizumab), VEGF decoy receptors or VEGF-Trap (e.g., aflibercept), and small molecule tyrosine kinase inhibitors (TKI) that block the VEGF receptors downstream signaling pathways (e.g., sunitinib, cabozantinib, and sorafenib) [15] (Table 1). Additionally, the mammalian target of rapamycin (mTOR) inhibitors also seems to have antiangiogenic effects by inhibiting the production of VEGF and platelet-derived growth factors (PDGF) [16–18].
Table 1.
| Approved antiangiogenic drugs | |
|---|---|
| Anti-VEGF monoclonal antibody | Indications for use |
| Bevacizumab | Metastatic colorectal cancer |
| Non-small-cell lung cancer | |
| Glioblastoma multiforme | |
| Metastatic renal cell cancer | |
| Macular degeneration | |
| Metastatic HER2 negative breast cancer | |
| Persistent, recurrent, and metastatic cervical cancer | |
| Platinum-resistant recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer | |
|
| |
| VEGF decoy receptor (VEGF-Trap) | Indications for use |
| Aflibercept | Metastatic colorectal cancer |
|
| |
| Tyrosine kinase inhibitors | Indications for use |
| Sorafenib | Metastatic renal cell cancer |
| Hepatic cancer (hepatocellular carcinoma) | |
| Sunitinib | Metastatic renal cell carcinoma |
| Gastrointestinal stromal tumour | |
| Pancreatic neuroendocrine tumour | |
| Cabozantinib | Medullary thyroid cancer |
| Erlotinib | Non-small-cell lung cancer |
| Pancreatic cancer | |
| Axitinib | Metastatic renal cell cancer |
| Pegaptanib | Macular degeneration |
| Ranibizumab | Macular degeneration |
| Pazopanib | Metastatic renal cell cancer |
| Soft tissue sarcoma | |
| Vandetanib | Medullary thyroid cancer |
| Regorafenib | Metastatic colorectal cancer |
| Gastrointestinal stromal tumour | |
| Imatinib | Chronic myeloid leukemia |
| Renal cell cancer | |
| Gastrointestinal stromal tumour | |
| Dasatinib | Philadelphia chromosome-positive (Ph+) chronic |
| myeloid leukemia (CML) | |
| Chronic phase Ph+ CML | |
| Philadelphia chromosome-positive (Ph+) acute lymphoblastic leukemia (Ph+ ALL) | |
|
| |
| Mammalian target of rapamycin inhibitors (mTOR inhibitors) | Indications for use |
| Temsirolimus | Renal cell cancer |
| Everolimus | Advanced breast cancer |
| Advanced renal cell cancer | |
| Pancreatic neuroendocrine tumour | |
| Tuberous sclerosis complex | |
| Subependymal giant cell astrocytoma | |
The number of patients developing MRONJ associated with antiangiogenic inhibitors or a combination of antiangiogenics and antiresorptive drugs has been growing over the last few years [8, 13, 19, 20]. The purpose of the present study is to provide a comprehensive review of the published reports of MRONJ associated with antiangiogenic agents in patients with no history of antiresorptive therapies.
2. Materials and Methods
2.1. Literature Search Strategy
A literature search was conducted to identify clinical trials, case reports, and case series on MRONJ associated with antiangiogenic treatment in antiresorptive drugs-naïve individuals using MEDLINE via PubMed (up to December 2017), EMBASE (from 1980 to December 2017), and Web of Science (from 1900 to December 2017). The search strategy used the following keywords: “osteonecrosis,” “jaw osteonecrosis,” “jaw bone necrosis,” “oral osteonecrosis,” “antiangiogenic,” “angiogenesis inhibitors,” “antineoplastic agents,” “antiangiogenic activity,” “antiangiogenic therapy,” “chemotherapy,” and “targeted therapy.” The references of retrieved articles were manually searched in order to identify additional relevant articles and abstracts. The search included articles published in English and other languages. Inclusion criteria were patients developing MRONJ associated with antiangiogenic agents based on the definition of MRONJ proposed by the special committee on MRONJ of the American Association of Oral and Maxillofacial Surgeons (AAOMS) in 2014 [1]. Patients with history of radiotherapy involving the jaw bones and patients having previous history or concurrent use of antiresorptive therapy were excluded.
3. Results
3.1. Search Results
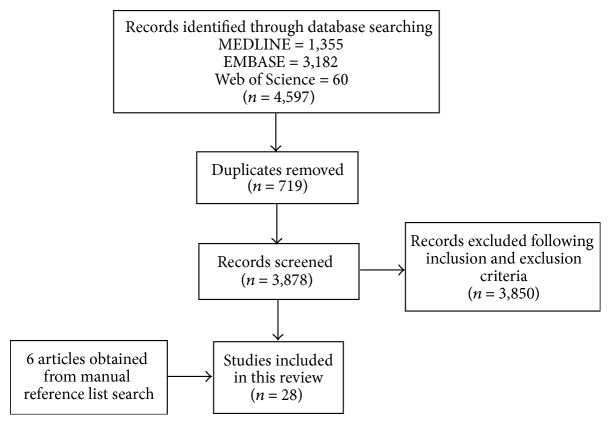
A total of 4,597 articles were retrieved by the initial search, including literature reviews, duplicate articles, clinical trials, and case reports with bisphosphonates and antiangiogenic treatment. The flow chart of review process to identify studies included and excluded is shown in Figure 1. Following screening the articles, we identified 28 articles describing 35 cases of MRONJ meeting the aforementioned inclusion criteria. Of these 28 publications, 26 papers were published in English, one was published in Italian, and one was published in Japanese. These 35 reported MRONJ cases were related to previous history of treatment with bevacizumab (14 cases), aflibercept (5 cases), sunitinib (3 cases), cabozantinib (2 cases), sorafenib (1 case), temsirolimus (1 case), everolimus (1 case), dasatinib (1 case), and multiple antiangiogenic agents (7 cases) (Table 2).
Figure 1.
Flow chart of the study selection process.
Table 2.
Previously reported cases of MRONJ associated with antiangiogenic medications (n = 35).
| Number | Authors | Age | Sex | Diagnosis of cancer | Treatment and concurrent medications | Antiangiogenic agents | Symptoms | Clinical presentation | Site of MRONJ | Time to MRONJ | Predisposing factors | Management of MRONJ | Outcomes | Definition of disease resolution |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (1) | Estilo et al. [23] | 51 | F | Metastatic breast cancer | Mastectomy Chest wall resection Chest wall radiation Doxorubicin Cyclophosphamide Letrozole Paclitaxel Capecitabine |
Bevacizumab 15 mg/kg every 3 weeks (total 8 doses) |
Jaw discomfort | Bone exposure | Mandible | 18 weeks after starting bevacizumab |
None | Surgical treatment (smoothen exposed bone) Chlorhexidine mouthwash 0.12% Interruption of bevacizumab |
Disease resolution (few weeks) Developed new MRONJ lesion (right mandible) |
Complete mucosal coverage |
|
| ||||||||||||||
| (2) | Estilo et al. [23] | 33 | F | Glioblastoma multiforme | Surgical resection Radiotherapy Temozolomide |
Bevacizumab 15 mg/kg every 2 weeks |
Jaw pain (gingival pain) |
Bone exposure | Mandible | 11 weeks after starting bevacizumab |
None | None | Persistent bone exposure (3 months) |
— |
|
| ||||||||||||||
| (3) | Greuter et al. [24] | 63 | F | Metastatic breast cancer | Liposomal-doxorubicin | Bevacizumab | Maxillary pain | Bone exposure | Maxilla | 2 months after starting bevacizumab |
Extraction due to dental infection (1 month) |
Surgical treatment | Disease resolution | Not specified |
|
| ||||||||||||||
| (4) | Serra et al. [25] | 64 | M | Metastatic non-small-cell lung cancer | Pneumectomy Lymph node ablation Cisplatin Gemcitabine |
Bevacizumab 7.5 mg/kg |
Jaw pain | Bone exposure | Mandible | 1 week after starting bevacizumab |
Extraction (1 week) |
Surgical treatment (local flap coverage) Amoxicillin with clavulanate Chlorhexidine mouthwash 0.2% |
Persistent bone exposure (3.5 months) |
— |
|
| ||||||||||||||
| (5) | Guarneri et al. [19] | NA | NA | Metastatic breast cancer | Docetaxel | Bevacizumab 7.5 mg/kg or 15 mg/kg every 3 weeks |
NA | Bone exposure | Mandible | 7 months after starting bevacizumab |
None | Surgical treatment (mandible decortication, tooth extraction) Interruption of bevacizumab |
Disease resolution (6 months) |
Complete mucosal coverage |
|
| ||||||||||||||
| (6) | Guarneri et al. [19] | NA | NA | Metastatic breast cancer | Docetaxel | Bevacizumab 7.5 mg/kg or 15 mg/kg every 3 weeks |
NA | Bone exposure | NA | 2 months after starting bevacizumab |
None | NA | NA | — |
|
| ||||||||||||||
| (7) | Brunamonti Binello et al. [26] |
47 | M | Adenocarcinoma of parotid gland | Surgical treatment Epirubicin Cisplatin |
Bevacizumab 15 mg/kg (total 8 doses in 6 months) |
Jaw pain, paraesthesia | Bone exposure | Mandible | 16 months after starting bevacizumab |
Symptomatic eruption of lower third molar | Surgical treatment (removed necrotic bone) Amoxicillin with clavulanate Metronidazole |
Persistent bone exposure (7 months) |
— |
|
| ||||||||||||||
| (8) | Bettini et al. [27] | 57 | F | Metastatic non-small-cell lung cancer | Gemcitabine Cisplatin Corticosteroid |
Bevacizumab 945 mg/21 days 4 cycles |
Jaw pain, halitosis, tooth loss |
Bone exposure | Mandible | 2 months after starting bevacizumab |
Periodontal infection | Amoxicillin with clavulanate Lincomycin (for 7 days) |
Disease resolution | Complete mucosal coverage |
|
| ||||||||||||||
| (9) | Dişel et al. [28] | 51 | M | Metastatic colon cancer | 5-Fluorouracil Leucovorin Oxaliplatin |
Bevacizumab 5 mg/kg every 2 weeks |
Jaw pain, ulcer, difficulty in chewing | Bone exposure | Mandible | NA | None | Surgical treatment (bone curettage) |
NA | — |
|
| ||||||||||||||
| (10) | Sato et al. [29] |
67 | M | Metastatic sigmoid colon cancer | Surgical treatment Oxaliplatin Leucovorin Irinotecan 5-Fluorouracil |
Bevacizumab | Jaw pain | Nonexposed MRONJ | Maxilla | 3 months after starting bevacizumab |
Extraction (1 month) |
Surgical treatment (removal of necrotic tissue) Antibiotics |
Disease resolution | Complete mucosal coverage |
|
| ||||||||||||||
| (11) | Fusco et al. [30] | 60 | M | Metastatic rectal cancer | Surgical treatment Radiotherapy 5-Fluorouracil Leucovorin Irinotecan Oxaliplatin |
Bevacizumab | Jaw pain | Bone exposure | Mandible | 9 months after starting bevacizumab |
Extraction (8 months) |
Antibiotics Chlorhexidine mouthwash |
NA | — |
|
| ||||||||||||||
| (12) | Tzermpos et al. [31] | 69 | M | Metastatic non-small-cell lung cancer | Carboplatin Docetaxel Cortisone |
Bevacizumab 15 mg/kg every 3 weeks |
Jaw pain, discomfort, paraesthesia | Bone exposure | Mandible | 3 years after starting bevacizumab |
Denture | Surgical treatment (surgical debridement) Amoxicillin Metronidazole Chlorhexidine mouthwash 0.12% Interruption of bevacizumab |
Disease resolution (8 weeks) | Complete mucosal coverage |
|
| ||||||||||||||
| (13) | Abel Mahedi Mohamed et al. [32] |
55 | F | Non-small-cell lung cancer | Corticosteroids | Bevacizumab | Asymptomatic | Bone exposure | Maxilla | 1.5 months after starting bevacizumab | Extraction | Conservative treatment | Disease resolution | Not specified |
|
| ||||||||||||||
| (14) | Abel Mahedi Mohamed et al. [32] |
66 | M | Glioblastoma multiforme | Corticosteroids | Bevacizumab | Pain | Nonexposed MRONJ | Mandible | 1.5 months after starting bevacizumab | Trauma | Conservative treatment (antibiotic treatment) |
Disease resolution | Not specified |
|
| ||||||||||||||
| (15) | Ponzetti et al. [33] | 64 | F | Metastatic colorectal cancer | 5-Fluorouracil Irinotecan |
Aflibercept | Spontaneous teeth loss with purulent discharge | Bone exposure | Mandible | 22 weeks after starting aflibercept |
Periodontal infection | Laser treatment | Persistent bone exposure | — |
|
| ||||||||||||||
| (16) | Mawardi et al. [34] | 43 | M | Metastatic colorectal cancer | 5-Fluorouracil Leucovorin Irinotecan |
Aflibercept | Jaw pain | Bone exposure | Mandible | 32 weeks after starting aflibercept | None | Amoxicillin Chlorhexidine mouthwash |
Persistent bone exposure (1.5 months) |
— |
|
| ||||||||||||||
| (17) | Mawardi et al. [34] | 63 | M | Metastatic carcinoid cancer | NA | Aflibercept | Asymptomatic | Bone exposure | Mandible | 46 weeks after starting aflibercept | None | Amoxicillin with clavulanate Chlorhexidine mouthwash |
Persistent bone exposure (2.5 months) | — |
|
| ||||||||||||||
| (18) | Mawardi et al. [34] | 51 | M | Metastatic esophageal cancer | 5-Fluorouracil Leucovorin Oxaliplatin |
Aflibercept | Jaw pain | Bone exposure | Mandible | 14 weeks after starting aflibercept | Extraction (2 weeks) |
Amoxicillin Chlorhexidine mouthwash Nonsurgical sequestrectomy |
Persistent bone exposure (2 months) | - |
|
| ||||||||||||||
| (19) | Zarringhalam et al. [35] | 47 | M | Metastatic colorectal, peritoneum, liver, and pelvic cancer | None | Aflibercept | Asymptomatic | Bone exposure | Mandible | 4 weeks after starting aflibercept | None | Surgical treatment (smoothen sharp exposed bone) |
Persistent bone exposure (12 weeks) | — |
|
| ||||||||||||||
| (20) | Nicolatou-Galitis et al. [36] |
64 | F | Metastatic renal cell cancer | Nephrectomy T4 replacement therapy Prednisolone (50 mg/day) |
Sunitinib 50 mg/day for 4 weeks on and 2 weeks off (for 4 years) |
Jaw pain | Bone exposure | Mandible | 4 years after starting sunitinib |
Denture | Amoxicillin Chlorhexidine mouthwash Azithromycin Interruption of sunitinib |
Disease resolution (3 months) | Complete mucosal coverage |
|
| ||||||||||||||
| (21) | Fleissig et al. [37] | 58 | F | Metastatic renal cell cancer | Nephrectomy Thyroxin sodium |
Sunitinib 50 mg/day for 4 weeks on and 2 weeks off |
Limited mouth opening, submandibular swelling, pain | Bone exposure | Mandible | 10 months after starting sunitinib |
Extraction (8 months) |
Amoxicillin with clavulanate (IV) PenG (IV) for 6 weeks and oral amoxicillin for 6 weeks Interruption of sunitinib |
Disease resolution (18 weeks) | Complete mucosal coverage |
|
| ||||||||||||||
| (22) | Melloni et al. [38] | 62 | M | Metastatic renal cell cancer | NA | Sunitinib 50 mg/day for 4 weeks on and 2 weeks off |
Jaw pain and infected lesion to the cutaneous side of the jaw | Bone exposure | Mandible | 5 years after starting sunitinib |
None | Surgical treatment (surgical sequestrectomy, ablation of necrotic bone, and local flap coverage) Amoxicillin with clavulanate Ofloxacin Interruption of sunitinib |
Disease resolution (12 months) | Complete mucosal coverage |
|
| ||||||||||||||
| (23) | Tempia Valenta et al. [39] |
51 | F | Medullary thyroid cancer | NA | Cabozantinib | NA | Bone exposure | Mandible | 6 months after starting cabozantinib |
Extraction | Surgical treatment (surgical debridement) Amoxicillin and clavulanate Chlorhexidine mouthwash 0.2% |
Disease resolution (22 months) | Not specified |
|
| ||||||||||||||
| (24) | Marino et al. [40] | 51 | F | Medullary thyroid cancer | Thyroidectomy 5-Fluorouracil Dacarbazine Radiotherapy Levothyroxine Calcitriol Vitamin D3 Duloxetine Propranolol Lansoprazole Loperamide |
Cabozantinib (175 mg/day) |
Asymptomatic | Bone exposure | Mandible | 3 months after starting cabozantinib |
Extraction due to dental infection (3 months) |
Surgical treatment (segmental ostectomy and tooth extraction) Amoxicillin and clavulanate Chlorhexidine mouthwash 0.2% |
Disease resolution | Complete mucosal coverage |
|
| ||||||||||||||
| (25) | Garuti et al. [41] | 74 | M | Metastatic hepatocellular carcinoma | Furosemide Potassium canrenoate Bisoprolol Allopurinol Tamsulosin Hydroxychloroquine Vitamin D Sertraline |
Sorafenib 400 mg/day |
Asymptomatic | Nonexposed MRONJ | Mandible | 3 months after starting sorafenib | None | Interruption of sorafenib | Persistent bone exposure (3 months) |
— |
|
| ||||||||||||||
| (26) | Abel Mahedi Mohamed et al. [32] |
53 | F | Acute lymphoblastic leukemia |
Corticosteroids | Dasatinib | Pain | Bone exposure | Mandible | 5 months after starting dasatinib | Extraction | Surgical treatment (block resection) | Disease resolution | Not specified |
|
| ||||||||||||||
| (27) | Parti et al. [42] | 60 | M | Metastatic renal cell cancer | Nephrectomy Prostatectomy |
Temsirolimus 25 mg every week |
NA | Bone exposure | Mandible | 3 months after starting temsirolimus |
Extraction (3 months) |
Interruption of temsirolimus | NA | — |
|
| ||||||||||||||
| (28) | Yamamoto et al. [43] |
80 | F | Metastatic breast cancer | Capecitabine Tamoxifen Fulvestrant Exemestane |
Everolimus | Jaw pain, localised heat, tenderness | Bone exposure | Mandible | 2 months after starting everolimus | None | Interruption of everolimus | Persistent bone exposure (2 months) | — |
|
| ||||||||||||||
| (29) | Agostino et al. [44] | 73 | M | Metastatic renal cell cancer | Nephrectomy | (1) Sunitinib 50 mg/day for 4 weeks of 6-week cycle (2) Temsirolimus 25 mg every week (3) Bevacizumab 10 mg/kg every two weeks |
NA | NA | NA | 12 months after starting bevacizumab |
NA | Interruption of bevacizumab | NA | — |
|
| ||||||||||||||
| (30) | Koch et al. [45] | 59 | M | Metastatic renal cell cancer | Nephrectomy Interferon Vinblastine Ramipril Hydrochlorothiazide Metoprolol I-Thyroxin |
(1) Sorafenib (2) Sunitinib 50 mg/day for 4 weeks and then sunitinib 37.5 mg/day |
Asymptomatic | Bone exposure | Mandible | 51 months after starting sunitinib |
Extraction (2 months) |
Surgical treatment (ablation of necrosis and local flap coverage) |
Disease resolution | Complete mucosal coverage |
|
| ||||||||||||||
| (31) | Santos-Silva et al. [46] | 61 | M | Metastatic renal cell cancer | Nephrectomy Hydrochlorothiazide Captopril |
(1) Bevacizumab 10 mg/kg every 2 weeks (2) Temsirolimus 25 mg every week |
Jaw pain | Bone exposure | Mandible | 55 weeks after starting bevacizumab and temsirolimus |
None | Chlorhexidine mouthwash 0.12% Interruption of bevacizumab and temsirolimus |
Disease resolution (3 months) | The absence of exposed necrotic bone |
|
| ||||||||||||||
| (32) | Pakosch et al. [47] | 53 | F | Pancreatic cancer | Surgical treatment Gemcitabine Leucovorin 5-Fluorouracil Oxaliplatin Paclitaxel Erlotinib |
(1) Bevacizumab (2) Sorafenib |
Jaw pain | Bone exposure | Mandible | 4 months after starting bevacizumab and sorafenib |
Denture | Surgical treatment (decortication), Amoxicillin with clavulanate Chlorhexidine mouthwash Solcoseryl Interruption of bevacizumab and chemotherapy |
Disease resolution (2 months) | Complete mucosal coverage |
|
| ||||||||||||||
| (33) | Jung [48] | 62 | F | Renal cell cancer | Nephrectomy | (1) Pazopanib (2) Everolimus |
Gingival bleeding and sore gum | Bone exposure | Mandible | 7 weeks after starting everolimus | Dental implant | Cephalosporin Surgical treatment (sequestrectomy and internal fixation) |
Disease resolution | Complete mucosal coverage |
|
| ||||||||||||||
| (34) | Patel et al. [49] | 67 | M | Metastatic renal cell cancer | Nivolumab Amlodipine Ramipril Levetiracetam Dexamethasone Lansoprazole Morphine Metoclopramide Amiodarone Cholecalciferol |
(1) Pazopanib (2) Axitinib |
Asymptomatic | Bone exposure | Maxilla | 1 months after starting axitinib | None | Hydrogen peroxide mouthwash | NA | — |
|
| ||||||||||||||
| (35) | Abel Mahedi Mohamed et al. [32] |
70 | M | Renal cell cancer | Corticosteroids | (1) Sunitinib (2) Everolimus |
Asymptomatic | Bone exposure | Mandible | 10 months after starting sunitinib, everolimus was commenced | Extraction | Conservative treatment | Persistent bone exposure | — |
NA: not available.
All 35 patients were reported to have developed MRONJ associated with at least one antiangiogenic agent and without a history of treatment with antiresorptive drugs. There were 19 males (54.29%) and 14 males (40%). The mean age of patients was 59.06 years (range: 33–80 years). The underlying diseases that required treatment with antiangiogenic agents included metastatic renal cell cancer (10 patients, 28.57%) followed by metastatic colorectal cancer (6 patients, 17.14%), metastatic breast cancer (5 patients, 14.29%), and other cancers (14 patients, 40%).
The most common presenting symptom was pain to the mandible/maxilla (12 patients, 34.29%) whereas 8 individuals (22.86%) reported no notable symptoms. The remaining patients had a variety of presenting complaints including mild discomfort to the mandible (1 patient, 2.86%), spontaneous teeth loss (1 patient, 2.86%), gingival bleeding (1 patient, 2.86%), and limited mouth opening together with submandibular swelling (1 patient, 2.86%). Moreover, there were 6 patients (17.14%) presenting with multiple symptoms including pain to the jaw, halitosis, spontaneous tooth loss, ulceration, difficulty in chewing, and paraesthesia. Regarding clinical characteristics of MRONJ, 32 patients (91.43%) had intraoral frank bone exposure, while the other three patients had nonexposed MRONJ. Mandible was the most common area of MRONJ development (29 patients, 82.86%), whereas four patients (11.43%) developed MRONJ in the maxilla.
Fourteen patients (40%) were exposed to bevacizumab, followed by aflibercept (5 patients, 14.29%), sunitinib (3 patients, 8.57%), cabozantinib (2 patients, 5.71%), sorafenib (1 patient, 2.86%), temsirolimus (1 patient, 2.86%), everolimus (1 patient, 2.86%), dasatinib (1 patient, 2.86%), and multiple antiangiogenic agents (7 patients, 20%). Regarding the routes of drug administration, antiangiogenic medications were administered intravenously in 21 patients (60.00%), while 12 patients (34.29%) were given antiangiogenic therapy orally. Two patients (5.71%) were given the combination of intravenous administration and oral administration. The mean duration of intravenous and oral antiangiogenic therapy before MRONJ development was 6.49 months (range: 0.23–36; SD = 1.82; 95% CI: 2.67–10.30) and 16.72 months (range: 1–60; SD = 6.42; 95% CI: 2.59–30.84), respectively. Patients with MRONJ also received a variety of concomitant medications including chemotherapy, hormone therapy, corticosteroids, antihypertensive drugs, antidepressants, and gastrointestinal medications.
Additional risk factors for MRONJ were reported in 21 patients, with dental extraction being the most prominent factor (13 patients, 37.14%). Other factors included history of mucosal trauma from dentures, chronic infection/inflammation to the tooth-bearing alveolar bone (periodontal disease), and insertion of osteointegrated dental implants (8 patients, 22.86%). The mean time to MRONJ diagnosis after tooth extraction was 3.09 months (range: 0.23–8; SD = 1.13; 95% CI: 0.40–5.77).
Regarding the management of MRONJ, seventeen patients (48.57%) were managed with surgical procedures alone or combined with medications (antibiotic therapy, antimicrobial mouthwash) and interruption of antiangiogenic agents. 16 patients did not receive surgery (45.71%), with antiangiogenic agents being discontinued in 7 cases. There was no active intervention reported in one patient. Most surgical interventions (11 patients) were minimally invasive procedures including smoothening of exposed bone, local flap coverage, removal of superficial necrotic bone, soft tissue debridement, and bone curettage, whereas 6 patients underwent major surgery such as bone decortication, resection of necrotic bone with local flap coverage, segmental osteotomy, and block resection. The outcomes of therapy were reported for 29 patients (82.8%), whereas no information was provided for the other 6 cases. 18 patients out of these 29 (62%) were reported to have disease resolution, while 11 patients showed persistent bone exposure. Disease resolution was described as complete mucosal coverage/no evidence of exposed bone in 13 cases, whereas no clear description was provided for the remaining 5 cases. Of note, one patient who experienced disease resolution to the left side of the mandible eventually developed a new area of MRONJ to the right mandible. The mean time from MRONJ diagnosis to complete healing was 6.75 months (range months: 1.84–22; SD = 2.47; 95% CI: 0.90–12.59). The data of 35 reported cases with MRONJ associated with antiangiogenics are summarised in Table 3.
Table 3.
Summary of data of reported cases of antiangiogenic-related MRONJ (n = 35).
| Age (years, range) | |
| Mean | 59.06 (33–80) |
| Gender (n, %) | |
| Male | 19 (54.29%) |
| Female | 14 (40.00%) |
| NA | 2 (5.71%) |
| Diagnosis of cancers (n, %) | |
| Metastatic renal cell cancer | 10 (28.57%) |
| Metastatic colorectal cancer | 6 (17.14%) |
| Metastatic breast cancer | 5 (14.29%) |
| Other cancers | 14 (40.00%) |
| Metastatic non-small-cell lung cancer | 4 |
| Glioblastoma multiforme | 2 |
| Medullary thyroid cancer | 2 |
| Malignant parotid tumour | 1 |
| Pancreatic cancer | 1 |
| Metastatic hepatocellular carcinoma | 1 |
| Metastatic carcinoid cancer | 1 |
| Metastatic oesophageal cancer | 1 |
| Presenting complaints (n, %) | |
| Jaw pain | 12 (34.29%) |
| Jaw pain with other complaints | 6 (17.14%) |
| Asymptomatic | 8 (22.86%) |
| Jaw discomfort | 1 (2.86%) |
| Spontaneous teeth loss | 1 (2.86%) |
| Limited mouth opening and submandibular area swelling | 1 (2.86%) |
| Gingival bleeding | 1 (2.86%) |
| NA | 5 (14.29%) |
| Clinical presentation (n, %) | |
| Bone exposure MRONJ | 32 (91.43%) |
| Nonexposed MRONJ | 3 (8.57%) |
| Location | |
| Mandible | 29 (82.86%) |
| Maxilla | 4 (11.43%) |
| NA | 2 (6.67%) |
| Types of antiangiogenic agents (n, %) | |
| Bevacizumab | 14 (40%) |
| Aflibercept | 5 (14.29%) |
| Sunitinib | 3 (8.57%) |
| Cabozantinib | 2 (5.71%) |
| Sorafenib | 1 (2.86%) |
| Temsirolimus | 1 (2.86%) |
| Everolimus | 1 (2.86%) |
| Dasatinib | 1 (2.86%) |
| Multiple antiangiogenic agents | 7 (20.00%) |
| Route of antiangiogenic administrations (n, %) | |
| Intravenous administration | 21 (60.00%) |
| Oral administration | 12 (34.29%) |
| Combination of intravenous administration and oral administration | 2 (5.71%) |
| Time to MRONJ (months, 95% CI) | |
| Intravenous antiangiogenics | 6.49 (2.67–10.30) |
| Oral antiangiogenics | 16.72 (2.59–30.84) |
| Predisposing factors (n, %) | |
| Extraction | 13 (37.14%) |
| Periodontal disease | 3 (8.57%) |
| Minor trauma from use of denture | 4 (11.43%) |
| Dental implant | 1 (2.86%) |
| Mean time to MRONJ after extraction (months, 95% CI) | 3.09 (0.40–5.77) |
| Management of MRONJ (n, %) | |
| Surgical treatment | 17 (48.57%) |
| Minimally invasive surgical procedures | 11 |
| Major surgical procedures | 6 |
| Nonsurgical treatment | 16 (45.71%) |
| No treatment | 1 (2.86%) |
| NA | 1 (2.86%) |
| Treatment outcomes (n, %) | |
| Disease resolution | 18 (62.06%) |
| Mean time to resolution (months, 95% CI) | 6.75 (0.90–12.59) |
| Incomplete resolution | 11 (37.93%) |
| NA | 6 |
NA: not available.
4. Discussion
The present study is the first comprehensive review upon MRONJ in patients treated with antiangiogenics in the absence of bone-modulating therapy.
We present data from 35 patients with different metastatic cancers who developed MRONJ following antiangiogenic treatments. All individuals were antiresorptive drugs-naïve. We have identified a number of differences between MRONJ associated with antiangiogenic agents and MRONJ associated with antiresorptive drugs. Our data showed a sex ratio of 4 : 5 (F : M) and an age range of 33–80 years (mean: 59.06 years), compared to sex ratio of 3 : 2 and age range of 42–90 years (mean 66 years) reported for antiresorptive drugs-associated MRONJ [50–52]. There also seem to be differences in the prevalence of MRONJ in these two populations. The reported prevalence of MRONJ in patients who had been treated with intravenous bevacizumab alone for the treatment of advanced breast cancer was 0.2%, which was lower than that of MRONJ associated with intravenous antiresorptive agents (7%) [11, 19]. However, it is important to note that the prevalence of MRONJ related to antiangiogenic agents may also depend on the epidemiology of underlying malignancies that require antiangiogenic therapy.
The clinical presentations of MRONJ associated with antiangiogenics also seem to be different from MRONJ due to antiresorptive agents. Approximately up to 25% of MRONJ cases related to antiresorptive medications can present without frank bone exposure [53], whereas most of the patients in the present review had clear evidence of bone exposure (91.43%). However, the number of patients with nonexposed MRONJ might be underestimated, since until 2014 MRONJ could only be diagnosed in individuals with clinical evidence of exposed bone as per AAOMS definition [53, 54].
With respect to the presenting complaints and location, they appear to be similar in two populations. The majority of patients with antiangiogenic-related MRONJ in this study experienced pain to the jaw, which is also the most common complaint in patients with MRONJ associated with antiresorptive agents [51, 55]. In the present study, most MRONJ cases associated with antiangiogenic therapy tended to occur in the mandible more frequently than in the maxilla, similar to those with antiresorptive drugs-induced MRONJ [50, 52].
A number of additional risk factors were identified in the present review including dental extraction, the use of denture, periodontal infection, and dental implant. Almost 40% of reported cases in this study were predominantly preceded by tooth extraction, which is similar to those with antiresorptive drugs-related MRONJ [50, 56].
There is a slight difference with respect to underlying malignancies between two populations. Patients with antiresorptive drugs-associated MRONJ showed the previous history of multiple myeloma, metastatic breast cancer, and metastatic prostate cancer, whereas those with MRONJ related to antiangiogenic medications in our review were mainly diagnosed with metastatic renal cell cancer, followed by metastatic colorectal cancer and metastatic breast cancer as demonstrated in Table 3 [52].
Although there was no consistent pattern in the time to MRONJ development in this review, the average time for developing MRONJ among patients with either intravenous or oral antiangiogenics was shorter than the average time to MRONJ onset in those treated with antiresorptive drugs. The mean time to event for intravenous and oral antiangiogenic agents in this study was 6.5 and 16.71 months, respectively, while it was reported to be approximately 1.8 and 3 years for bisphosphonate therapy [12, 57].
Patients with metastatic malignancy may receive a number of anticancer drugs simultaneously. In this review, we found that seven of the reported cases received more than one antiangiogenic agent in their treatment history [32, 44–49]. Of these patients, some were given different antiangiogenics at the same time, while others received these agents at different time points. The development of MRONJ is usually associated with the latest antiangiogenic agent used by the patient; however, one cannot exclude the fact that the antiangiogenic agents previously used by these patients might have contributed to it.
We included in this review two cases of MRONJ associated with new TKIs, namely, pazopanib in combination with axitinib (n = 1) and dasatinib (n = 1) [32, 49]. According to the Food and Drug Administration's Adverse Event Reporting System (FAERS), pazopanib and axitinib have been associated with the development of MRONJ in 10 and 9 individuals, respectively; however, as data regarding concurrent or previous medication were not available in FAERS documentation [58], it is difficult to conclude whether these individuals were indeed antiresorptive drugs-naïve. Therefore, we decided not to include these 19 cases in our review.
With regard to the management of MRONJ, approximately half of the individuals with MRONJ associated with antiangiogenics (48.57%) were managed surgically, which is similar to those with bisphosphonate-related MRONJ [11, 52]. However, the prognosis of antiangiogenic-related MRONJ appears to be better than that of individuals developing MRONJ associated with antiresorptive agents. We observed a 62% rate of disease resolution in those where outcomes were reported as opposed to approximately 50% reported in the literature for MRONJ associated with antiresorptive agents [56, 59, 60]. It is possible that the higher rate of disease resolution might be related to the shorter half-life of antiangiogenics [61, 62], as well as the lower cumulative dosages [63]. Moreover, the average time to resolution for MRONJ associated with antiangiogenics appears to be shorter than antiresorptive drugs-induced MRONJ (6.75 months, range: 1.84–22 months versus 8.2 months, range: 0.2–25.6 months) [55].
In this comprehensive review, we excluded a number of potential antiangiogenic-related MRONJ cases due to a lack of adequate clinical information. For example, the 2012 report on aflibercept by the US Food and Drug Administration (FDA) described 3 aflibercept-treated bisphosphonate-naïve patients who developed MRONJ; however, none of these patients were added to the present review as one had jaw bone exposure for less than 8 weeks and no information was provided for the other two cases [64]. Furthermore, in a pivotal BOLERO-2 trial, MRONJ has been described in 2 patients in the experimental arm (everolimus-exemestane) and 1 patient in the control arm (exemestane), with one of three patients to receive bisphosphonate treatment [65]. However, there was no evidence to show whether the patient with a history of bisphosphonate treatment was in the experimental arm or control arm. More recently, Antonuzzo et al. [66] reported the first case with MRONJ potentially associated with regorafenib, one of the tyrosine kinase inhibitors, in an antiresorptive drugs-naïve individual. Although MRONJ appeared 22 months after regorafenib treatment, Fusco et al. [67] have noted that some details such as the use of other medications prior to regorafenib treatment, dosing, and the time on medication are still missing. This medication is usually used as a third or further line of treatment of metastatic colorectal cancer. Therefore, it is also worth knowing whether this patient has received other well-documented antiangiogenic medications such as bevacizumab and aflibercept prior to regorafenib. If this is the case, bevacizumab or aflibercept possibly might contribute to the development of MRONJ rather than regorafenib alone. Another patient with gastrointestinal stromal tumours (GISTs) receiving imatinib monotherapy presented with pain and exposed bone at lower right mandible after having the tooth removed for 5 weeks. The patient was treated with debridement of necrotic bone and antibiotic and then was discharged. Unfortunately, there was no further information about this patient [68]. The duration of persistent bone exposure in this case was not mentioned if it was longer than 8 weeks. Therefore, available data seems not to be enough to classify this case as MRONJ according to the definition of MRONJ [1] and to confirm the association between MRONJ and imatinib. In addition to the above reported cases, Hopp et al. [69] reported one patient with necrotic bone exposure after the 2-year intravitreal injections of bevacizumab for treatment of retinal vascular thrombosis without notable dental risk factors or use of bisphosphonates. After the patient experienced pain to the mandible, the lesion was completely healed by antibiotics treatment within 8 weeks. Therefore, this case seems not to be correlated with the definition of MRONJ formulated by the AAOMS in 2014 [1].
It is important to highlight that another case of oral soft tissues complication associated with bevacizumab was also reported by Magremanne et al. [70]. Although this case was included in previous reviews regarding cases of MRONJ associated with antiangiogenic agents, there was no evidence of osteonecrosis of the jaw and also the necrotic area seemed to be limited only to oral soft tissues. This reported case does not meet the definition of MRONJ and hence it was excluded from this review.
5. Conclusion
There remains incomplete information regarding the cases of antiangiogenic-related MRONJ in antiresorptive-naïve individuals reported in the literature. It is therefore difficult to draw any conclusion regarding the epidemiology and the characteristics of MRONJ in this patient population. Within the limitation of available data, we have identified a number of differences between MRONJ associated with antiangiogenics and MRONJ related to antiresorptive drugs including demographic characteristics, prevalence, the underlying malignant disease, time to the onset, and time to resolution. Considering that the list of antiangiogenic inhibitors that have potential to increase the risk of MRONJ development is increasing, further prospective and well-designed research is warranted to confirm our findings and increase knowledge and understanding of the disease.
Acknowledgments
Professor Stefano Fedele received salary support from the NIHR UCLH Biomedical Research Centre.
Conflicts of Interest
The authors have no conflicts of interest to declare.
References
- 1.Ruggiero S. L., Dodson T. B., Fantasia J. American association of oral and maxillofacial surgeons position paper on medication-related osteonecrosis of the jaw—2014 update. Journal of Oral and Maxillofacial Surgery. 2014;72(10):1938–1956. doi: 10.1016/j.joms.2014.04.031. [DOI] [PubMed] [Google Scholar]
- 2.Hinchy N. V., Jayaprakash V., Rossitto R. A., et al. Osteonecrosis of the jaw - Prevention and treatment strategies for oral health professionals. Oral Oncology. 2013;49(9):878–886. doi: 10.1016/j.oraloncology.2013.06.008. [DOI] [PubMed] [Google Scholar]
- 3.Capocci M., Romeo U., Guerra F., et al. Medication-related osteonecrosis of the jaws (MRONJ) and quality of life evaluation: A pilot study. La Clinica Terapeutica. 2017;168(4):e253–e257. doi: 10.7417/T.2017.2015. [DOI] [PubMed] [Google Scholar]
- 4.Marx R. E. Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. Journal of Oral and Maxillofacial Surgery. 2003;61(9):1115–1117. doi: 10.1016/S0278-2391(03)00720-1. [DOI] [PubMed] [Google Scholar]
- 5.Stopeck A. T., Lipton A., Body J., et al. Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: a randomized, double-blind study. Journal of Clinical Oncology. 2010;28(35):5132–5139. doi: 10.1200/JCO.2010.29.7101. [DOI] [PubMed] [Google Scholar]
- 6.Uyanne J., Calhoun C. C., Le A. D. Antiresorptive drug-related osteonecrosis of the jaw. Dental Clinics of North America. 2014;58(2):369–384. doi: 10.1016/j.cden.2013.12.006. [DOI] [PubMed] [Google Scholar]
- 7.Cosman F., Crittenden D. B., Adachi J. D., et al. Romosozumab treatment in postmenopausal women with osteoporosis. The New England Journal of Medicine. 2016;375(16):1532–1543. doi: 10.1056/NEJMoa1607948. [DOI] [PubMed] [Google Scholar]
- 8.Hamadeh I. S., Ngwa B. A., Gong Y. Drug induced osteonecrosis of the jaw. Cancer Treatment Reviews. 2015;41(5):455–464. doi: 10.1016/j.ctrv.2015.04.007. [DOI] [PubMed] [Google Scholar]
- 9.Bagan J., Scully C., Sabater V., Jimenez Y. Osteonecrosis of the jaws in patients treated with intravenous bisphosphonates (BRONJ): A concise update. Oral Oncology. 2009;45(7):551–554. doi: 10.1016/j.oraloncology.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 10.Reid I. R. Osteonecrosis of the jaw—who gets it, and why? Bone. 2009;44(1):4–10. doi: 10.1016/j.bone.2008.09.012. [DOI] [PubMed] [Google Scholar]
- 11.Kühl S., Walter C., Acham S., Pfeffer R., Lambrecht J. T. Bisphosphonate-related osteonecrosis of the jaws—a review. Oral Oncology. 2012;48(10):938–947. doi: 10.1016/j.oraloncology.2012.03.028. [DOI] [PubMed] [Google Scholar]
- 12.Malden N., Lopes V. An epidemiological study of alendronate-related osteonecrosis of the jaws. A case series from the south-east of Scotland with attention given to case definition and prevalence. Journal of Bone and Mineral Metabolism. 2012;30(2):171–182. doi: 10.1007/s00774-011-0299-z. [DOI] [PubMed] [Google Scholar]
- 13.Fusco V., Porta C., Saia G., et al. Osteonecrosis of the Jaw in Patients with Metastatic Renal Cell Cancer Treated with Bisphosphonates and Targeted Agents: Results of an Italian Multicenter Study and Review of the Literature. Clinical Genitourinary Cancer. 2015;13(4):287–294. doi: 10.1016/j.clgc.2014.12.002. [DOI] [PubMed] [Google Scholar]
- 14.De Falco S. Antiangiogenesis therapy: an update after the first decade. Korean Journal of Internal Medicine. 2014;29(1):1–11. doi: 10.3904/kjim.2014.29.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Al-Husein B., Abdalla M., Trepte M., DeRemer D. L., Somanath P. R. Antiangiogenic therapy for cancer: an update. Pharmacotherapy. 2012;32(12):1095–1111. doi: 10.1002/phar.1147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Del Bufalo D., Ciuffreda L., Trisciuoglio D., et al. Antiangiogenic potential of the mammalian target of rapamycin inhibitor temsirolimus. Cancer Research. 2006;66(11):5549–5554. doi: 10.1158/0008-5472.CAN-05-2825. [DOI] [PubMed] [Google Scholar]
- 17.Yuan R., Kay A., Berg W. J., Lebwohl D. Targeting tumorigenesis: development and use of mTOR inhibitors in cancer therapy. Journal of Hematology & Oncology. 2009;2, article 45 doi: 10.1186/1756-8722-2-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Moriya M., Yamada T., Tamura M., et al. Antitumor effect and antiangiogenic potential of the mTOR inhibitor temsirolimus against malignant pleural mesothelioma. Oncology Reports. 2014;31(3):1109–1115. doi: 10.3892/or.2013.2948. [DOI] [PubMed] [Google Scholar]
- 19.Guarneri V., Miles D., Robert N., et al. Bevacizumab and osteonecrosis of the jaw: Incidence and association with bisphosphonate therapy in three large prospective trials in advanced breast cancer. Breast Cancer Research and Treatment. 2010;122(1):181–188. doi: 10.1007/s10549-010-0866-3. [DOI] [PubMed] [Google Scholar]
- 20.Wynn R. L. Bevacizumab (Avastin): An anti-angiogenic drug associated with osteonecrosis of the jaw. General dentistry. 2011;59(6):410–413. [PubMed] [Google Scholar]
- 21.Yoo S. Y., Kwon S. M. Angiogenesis and its therapeutic opportunities. Mediators of Inflammation. 2013;2013:11. doi: 10.1155/2013/127170.127170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.The U.S. Food and Drug Administration. FDA approved drug products. 2017, https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=021986.
- 23.Estilo C. L., Fornier M., Farooki A., Carlson D., Bohle G., III, Huryn J. M. Osteonecrosis of the jaw related to bevacizumab. Journal of Clinical Oncology. 2008;26(24):4037–4038. doi: 10.1200/JCO.2007.15.5424. [DOI] [PubMed] [Google Scholar]
- 24.Greuter S., Schmid F., Ruhstaller T., Thuerlimann B. Bevacizumab-associated osteonecrosis of the jaw. Annals of Oncology. 2008;19(12):2091–2092. doi: 10.1093/annonc/mdn653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Serra E., Paolantonio M., Spoto G., Mastrangelo F., Tetè S., Dolci M. Bevacizumab-related osteneocrosis of the jaw. International Journal of Immunopathology and Pharmacology. 2009;22(4):1121–1123. doi: 10.1177/039463200902200429. [DOI] [PubMed] [Google Scholar]
- 26.Brunamonti Binello P., Bandelloni R., Labanca M., Buffoli B., Rezzani R., Rodella L. F. Osteonecrosis of the jaws and bevacizumab therapy: A case report. International Journal of Immunopathology and Pharmacology. 2012;25(3):789–791. doi: 10.1177/039463201202500328. [DOI] [PubMed] [Google Scholar]
- 27.Bettini G., Blandamura S., Saia G., Bedogni A. Bevacizumab-related osteonecrosis of the mandible is a self-limiting disease process. BMJ Case Reports. 2012;2012 doi: 10.1136/bcr-2012-007284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dişel U., Beşen A. A., Özyilkan Ö., Er E., Canpolat T. A case report of bevacizumab-related osteonecrosis of the jaw: Old problem, new culprit. Oral Oncology. 2012;48(2):p. -e3. doi: 10.1016/j.oraloncology.2011.07.030. [DOI] [PubMed] [Google Scholar]
- 29.Sato M., Ono F., Yamamura A., Onochi S. A case of osteonecrosis of the jaw during treatment by bevacizumab for sigmoid colon cancer. Journal of Japanese Society of Gastroenterology. 2013;110(4):655–659. [PubMed] [Google Scholar]
- 30.Fusco V., Fasciolo A., Gaino R., et al. Bevacizumab-related osteonecrosis of jaw in a rectal cancer patient never treated with bisphosphonates. Ann Stomatol. 2014;5(2):p. 31. [Google Scholar]
- 31.Tzermpos F., Ismail A., Pavli M., Tosios K. I. Osteonecrosis of the mandible in a patient with lung adenocarcinoma undergoing anti-angiogenic therapy with bevacizumab. Oral Surgery. 2016;9(1):40–46. doi: 10.1111/ors.12153. [DOI] [Google Scholar]
- 32.Abel Mahedi Mohamed H., Nielsen C. E., Schiodt M. Medication related osteonecrosis of the jaws associated with targeted therapy as monotherapy and in combination with antiresorptives. A report of 7 cases from the Copenhagen Cohort. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2018;125(2):157–163. doi: 10.1016/j.oooo.2017.10.010. [DOI] [PubMed] [Google Scholar]
- 33.Ponzetti A., Pinta F., Spadi R., et al. Jaw osteonecrosis associated with aflibercept, irinotecan and fluorouracil: Attention to oral district. TUMORI. 2016;102:S74–S77. doi: 10.5301/tj.5000405. [DOI] [PubMed] [Google Scholar]
- 34.Mawardi H., Enzinger P., McCleary N., et al. Osteonecrosis of the jaw associated with ziv-aflibercept. Journal of Gastrointestinal Oncology. 2016;7(6):E81–E87. doi: 10.21037/jgo.2016.05.07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zarringhalam P., Brizman E., Shakib K. Medication-related osteonecrosis of the jaw associated with aflibercept. British Journal of Oral and Maxillofacial Surgery. 2017;55(3):314–315. doi: 10.1016/j.bjoms.2016.11.315. [DOI] [PubMed] [Google Scholar]
- 36.Nicolatou-Galitis O., Migkou M., Psyrri A., et al. Gingival bleeding and jaw bone necrosis in patients with metastatic renal cell carcinoma receiving sunitinib: Report of 2 cases with clinical implications. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2012;113(2):234–238. doi: 10.1016/j.tripleo.2011.08.024. [DOI] [PubMed] [Google Scholar]
- 37.Fleissig Y., Regev E., Lehman H. Sunitinib related osteonecrosis of jaw: a case report. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2012;113(3):e1–e3. doi: 10.1016/j.tripleo.2011.06.023. [DOI] [PubMed] [Google Scholar]
- 38.Melloni C., Tuttolomondo A., Anfosso A., Calamia C., Clemente F. D., Cordova A. Sunitinib related osteonecrosis of the jaw (SURONJ): a rare occurrence? European Journal of Plastic Surgery. 2016;39(2):161–162. doi: 10.1007/s00238-015-1112-3. [DOI] [Google Scholar]
- 39.Tempia Valenta G., Marino R., Erovigni F., Pentenero M., Gandolfo S. Mandibular osteonecrosis related to cabozantinib. 1:40. 2012. (Abstracts of the 11th Biennial Congress of the European Association of Oral Medicine). [Google Scholar]
- 40.Marino R., Orlandi F., Arecco F., Gandolfo S., Pentenero M. Osteonecrosis of the jaw in a patient receiving cabozantinib. Australian Dental Journal. 2015;60(4):528–531. doi: 10.1111/adj.12254. [DOI] [PubMed] [Google Scholar]
- 41.Garuti F., Camelli V., Spinardi L., Bucci L., Trevisani F. Osteonecrosis of the jaw during sorafenib therapy for hepatocellular carcinoma. TUMORI. 2016;102:S69–S70. doi: 10.5301/tj.5000504. [DOI] [PubMed] [Google Scholar]
- 42.Parti V., Ortega C., Aglietta M. Caso clinico di Osteonecrosi da inibitore di m-TOR. (Abstract Convegno Osteonecrosi dei mascellari (ONJ): ruolo della Rete Oncologica del Piemonte e della Valle d'Aosta). [DOI] [Google Scholar]
- 43.Yamamoto D., Tsubota Y., Utsunomiya T., et al. Osteonecrosis of the jaw associated with everolimus: A case report. Molecular and Clinical Oncology. 2017;6(2):255–257. doi: 10.3892/mco.2016.1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Agostino N. M., Gingrich R., Drabick J. J. Bevacizumab demonstrates prolonged disease stabilization in patients with heavily pretreated metastatic renal cell carcinoma: A case series and review of the literature. Advances in Urology. 2010 doi: 10.1155/2010/687043.687043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Koch F. P., Walter C., Hansen T., Jäger E., Wagner W. Osteonecrosis of the jaw related to sunitinib. Journal of Oral and Maxillofacial Surgery. 2011;15(1):63–66. doi: 10.1007/s10006-010-0224-y. [DOI] [PubMed] [Google Scholar]
- 46.Santos-Silva A. R., Belizário Rosa G. A., Castro Júnior G. D., Dias R. B., Prado Ribeiro A. C., Brandão T. B. Osteonecrosis of the mandible associated with bevacizumab therapy. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2013;115(6):e32–e36. doi: 10.1016/j.oooo.2013.02.001. [DOI] [PubMed] [Google Scholar]
- 47.Pakosch D., Papadimas D., Munding J., Kawa D., Kriwalsky M. S. Osteonecrosis of the mandible due to anti-angiogenic agent, bevacizumab. Journal of Oral and Maxillofacial Surgery. 2013;17(4):303–306. doi: 10.1007/s10006-012-0379-9. [DOI] [PubMed] [Google Scholar]
- 48.Jung T.-Y. Osteonecrosis of jaw after antiangiogenic agent administration in a renal cell carcinoma patient. Oral and Maxillofacial Surgery Cases. 2017;3(2):27–33. doi: 10.1016/j.omsc.2017.03.001. [DOI] [Google Scholar]
- 49.Patel V., Sproat C., Kwok J., Tanna N. Axitinib-related osteonecrosis of the jaw. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2017 doi: 10.1016/j.oooo.2017.08.003. [DOI] [PubMed] [Google Scholar]
- 50.Woo S. B., Hellstein J. W., Kalmar J. R. Systematic review: bisphosphonates and osteonecrosis of the jaws. Annals of Internal Medicine. 2006;144(10):753–756. doi: 10.7326/0003-4819-144-10-200605160-00009. [DOI] [PubMed] [Google Scholar]
- 51.Otto S., Schreyer C., Hafner S., et al. Bisphosphonate-related osteonecrosis of the jaws - Characteristics, risk factors, clinical features, localization and impact on oncological treatment. Journal of Cranio-Maxillo-Facial Surgery. 2012;40(4):303–309. doi: 10.1016/j.jcms.2011.05.003. [DOI] [PubMed] [Google Scholar]
- 52.Filleul O., Crompot E., Saussez S. Bisphosphonate-induced osteonecrosis of the jaw: a review of 2,400 patient cases. Journal of Cancer Research and Clinical Oncology. 2010;136(8):1117–1124. doi: 10.1007/s00432-010-0907-7. [DOI] [PubMed] [Google Scholar]
- 53.Fedele S., Bedogni G., Scoletta M. Up to a quarter of patients with osteonecrosis of the jaw associated with antiresorptive agents remain undiagnosed. British Journal of Oral and Maxillofacial Surgery. 2014 doi: 10.1016/j.bjoms.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 54.Ruggiero S. L., Dodson T. B., Assael L. A., Landesberg R., Marx R. E., Mehrotra B. American association of oral and maxillofacial surgeons postion paper on bisphosphonate-related osteonecrosis of the jaws. Journal of Oral and Maxillofacial Surgery. 2009;67(5, supplement 1):2–12. doi: 10.1016/j.joms.2009.01.009. [DOI] [PubMed] [Google Scholar]
- 55.Saad F., Brown J. E., Van Poznak C., et al. Incidence, risk factors, and outcomes of osteonecrosis of the jaw: integrated analysis from three blinded active-controlled phase III trials in cancer patients with bone metastases. Annals of Oncology. 2012;23(5):1341–1347. doi: 10.1093/annonc/mdr435. [DOI] [PubMed] [Google Scholar]
- 56.Campisi G., Fedele S., Fusco V., Pizzo G., Di Fede O., Bedogni A. Epidemiology, clinical manifestations, risk reduction and treatment strategies of jaw osteonecrosis in cancer patients exposed to antiresorptive agents. Future Oncology. 2014;10(2):257–275. doi: 10.2217/fon.13.211. [DOI] [PubMed] [Google Scholar]
- 57.Palaska P. K., Cartsos V., Zavras A. I. Bisphosphonates and time to osteonecrosis development. The Oncologist. 2009;14(11):1154–1166. doi: 10.1634/theoncologist.2009-0115. [DOI] [PubMed] [Google Scholar]
- 58.Zhang X., Hamadeh I. S., Song S., et al. Osteonecrosis of the Jaw in the United States Food and Drug Administration's Adverse Event Reporting System (FAERS) Journal of Bone and Mineral Research. 2016;31(2):336–340. doi: 10.1002/jbmr.2693. [DOI] [PubMed] [Google Scholar]
- 59.van den Wyngaert T., Claeys T., Huizing M. T., Vermorken J. B., Fossion E. Initial experience with conservative treatment in cancer patients with osteonecrosis of the jaw (ONJ) and predictors of outcome. Annals of Oncology. 2009;20(2):331–336. doi: 10.1093/annonc/mdn630. [DOI] [PubMed] [Google Scholar]
- 60.Lazarovici T. S., Yahalom R., Taicher S., Elad S., Hardan I., Yarom N. Bisphosphonate-realted osteonecrosis of the jaws: a single-center study of 101 patients. Journal of Oral and Maxillofacial Surgery. 2009;67(4):850–855. doi: 10.1016/j.joms.2008.11.015. [DOI] [PubMed] [Google Scholar]
- 61.Gordon M. S., Margolin K., Talpaz M., et al. Phase I safety and pharmacokinetic study of recombinant human anti-vascular endothelial growth factor in patients with advanced cancer. Journal of Clinical Oncology. 2001;19(3):843–850. doi: 10.1200/JCO.2001.19.3.843. [DOI] [PubMed] [Google Scholar]
- 62.Oudard S., Beuselinck B., Decoene J., Albers P. Sunitinib for the treatment of metastatic renal cell carcinoma. Cancer Treatment Reviews. 2011;37(3):178–184. doi: 10.1016/j.ctrv.2010.08.005. [DOI] [PubMed] [Google Scholar]
- 63.Fusco V., Santini D., Armento G., Tonini G., Campisi G. Osteonecrosis of jaw beyond antiresorptive (bone-targeted) agents: new horizons in oncology. Expert Opinion on Drug Safety. 2016;15(7):925–935. doi: 10.1080/14740338.2016.1177021. [DOI] [PubMed] [Google Scholar]
- 64.FDA report. 100. 141: Clin Rev. Aflibercept/Zaltrap.; 2012. FDA report on aflibercept: Center for drug evaluation and research. Application number 125418Orig1s000, p. 99. [Google Scholar]
- 65.Gnant M., Baselga J., Rugo H. S., et al. Effect of everolimus on bone marker levels and progressive disease in bone in BOLERO-2. Journal of the National Cancer Institute. 2013;105(9):654–663. doi: 10.1093/jnci/djt026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Antonuzzo L., Lunghi A., Giommoni E., Brugia M., Di Costanzo F. Regorafenib also can cause osteonecrosis of the jaw. Journal of the National Cancer Institute. 2016;108(4) doi: 10.1093/jnci/djw002.djw002 [DOI] [PubMed] [Google Scholar]
- 67.Fusco V., Campisi G., Numico G., Migliorati C. A., Santini D., Bedogni A. RE: Regorafenib Also Can Cause Osteonecrosis of the Jaw. Journal of the National Cancer Institute. 2016;108(9) doi: 10.1093/jnci/djw155.djw155 [DOI] [PubMed] [Google Scholar]
- 68.Viviano M., Rossi M., Cocca S. A rare case of osteonecrosis of the jaw related to imatinib. Journal of the Korean Association of Oral and Maxillofacial Surgeons. 2017;43(2):p. 120. doi: 10.5125/jkaoms.2017.43.2.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hopp R. N., Pucci J., Santos-Silva A. R., Jorge J. Osteonecrosis after administration of intravitreous bevacizumab. Journal of Oral and Maxillofacial Surgery. 2012;70(3):632–635. doi: 10.1016/j.joms.2011.02.104. [DOI] [PubMed] [Google Scholar]
- 70.Magremanne M., Lahon M., De Ceulaer J., Reychler H. Unusual bevacizumab-related complication of an oral infection. Journal of Oral and Maxillofacial Surgery. 2013;71(1):53–55. doi: 10.1016/j.joms.2012.03.022. [DOI] [PubMed] [Google Scholar]