Abstract
Purpose
The purpose of this systematic literature review was to assess the usefulness of the Thyroid Image Reporting and Data System (K-TIRADS) classification proposed by Kwak for differentiation of thyroid nodules.
Material and methods
Four literature databases were searched for relevant articles through early January 2017. A meta-analysis was performed to calculate pooled sensitivity, specificity, positive likelihood ratio (LR+), negative likelihood ratio (LR-) and diagnostic odds ratio (DOR). The area under the curve (AUC) from the pooled receiver operating characteristic (ROC) was used to assess the usefulness of this classification for differentiation of thyroid nodules. Meta-analysis was conducted by using meta-analysis software.
Results
We analysed six publications describing 10,926 nodules. Pooled sensitivity, specificity, LR+, LR-, DOR, and AUC for pooled ROC were 0.983 (95 % CI 0.976–0.989), 0.552 (95 % CI 0.542–0.562), 2.666 (95 % CI 1.692–4.198), 0.05 (95 % CI 0.035–0.072), 51.020 (95 % CI 15.241–170.79) and 0.938, respectively.
Conclusions
Kwak TIRADS has high sensitivity and low specificity. Thus, it is very useful to discard the benign cases and to reduce the number of biopsies.
Key Points
• Routine, adequate standardization of thyroid nodules ultrasound classification is mandatory.
• Kwak TIRADS parameters are accurate for differentiating focal thyroid lesions.
• Kwak TIRADS system is simple to apply.
• Kwak TIRADS system may become a useful diagnostic tool.
Keywords: Thyroid nodules, Thyroid neoplasm, Ultrasonography, Risk assessment, Meta-analysis
Introduction
Ultrasound imaging is a basic technique used in the visualization and characterization of focal thyroid lesions and to estimate the risk of malignancy. The suspicious nature of lesions is confirmed on the basis of cytological examination of specimens collected via ultrasound-guided fine-needle aspiration biopsy (FNAB) and further histological examination if necessary [1]. Currently, ultrasound and FNAB serve as essential tools for diagnosing thyroid nodules [1, 2]. Numerous studies have shown that ultrasonography has an important place in the diagnosis of malignant and benign thyroid lesions and is marked by high sensitivity and low specificity [3]. Many lesions without suspicious features may be observed conservatively in this manner without the necessity of biopsy [2, 4].
Currently, the fundamental technique used for clinical assessment of thyroid lesions is grey-scale imaging (B-mode). In B-mode, suspicious features include: solid nature; low or very low echogenicity; irregular, microlobular or blurred borders; vertical shape or an anteroposterior diameter greater than lateral; and microcalcifications [2, 5, 6].
There are numerous guidelines of many medical societies worldwide that describe the clinical and ultrasound features that necessitate FNAB [7–9]. However, there is a need for application of routine, adequate and common standardization system of thyroid nodules ultrasound classification. The system has already been proposed and is called Thyroid Imaging Reporting and Data System (TIRADS). It relies on B-mode imaging and represents an important step in standardization of ultrasound examination of the thyroid. TIRADS has its foundation in the Breast Imaging Reporting and Data System (BIRADS) classification [10–12], which is based on varying, increasing the risk of malignancy of focal lesions in different categories. Data relating to TIRADS classification were first published in 2009 by two independent teams led by Horvath [13] and Park [14]. The two different approaches proposed by the teams, in our opinion, proved to be complicated and difficult to use on a daily basis. The later study published by Kwak in 2011 [6] had a different approach to this classification. Whether the individual lesion belongs to a TIRADS category or not was determined based on the number of suspicious features, including solid structure, low or very low echogenicity, irregular or microlobular borders, microcalcifications and vertical shape (TIRADS 3 = no suspicious features; TIRADS 4a = 1 suspicious feature; TIRADS 4b = 2 suspicious features; TIRADS 4c = 3 or 4 suspicious features; TIRADS 5= 5 suspicious features).
Furthermore, other groups proposed different TIRADS interpretations [15–17].
The aim of this study was to conduct a systematic literature review and to assess the diagnostic utility of the Kwak’s TIRADS classification in the risk stratification of thyroid nodules in adults.
Materials and method
Eligibility criteria
The described systematic review of the literature and meta-analysis were carried out in accordance with the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) [18]. The studies were included in the analysis based on the following criteria (participants, interventions, comparators, outcomes, and study design (PICOS) questions): adults with focal thyroid changes; TIRADS classification as proposed by Kwak used in differential diagnosis of thyroid nodules; reference examinations were histological and/or cytological, ultrasound follow-up lasting more than 12 months; retrospective and prospective studies published in English or German. The full text of duplicated publications was used to obtain more precise data necessary for analysis.
Data sources
We searched four databases (PubMed, Cochrane database, ScienceDirect and EMBASE) from January 2009 to January 2017. The following terminology was adopted: ‘TIRADS’ OR ‘TI-RADS’ OR ‘thyroid imaging reporting and data system’ OR ‘reporting system for thyroid nodules’. Subsequently, the titles of studies and abstracts were validated for inclusion of the object of the analysis. Full versions of relevant articles were then downloaded for further analysis. Full text articles were examined for inclusion criteria by two independent reviewers (B.M. and M.S.M.). The reference list of obtained publications was then manually checked to identify other studies related to the topic.
Inclusion criteria
For inclusion in the present study, patients had to meet all of the following criteria: (1) adults with thyroid nodules, including patients with nodular goitre; (2) differential diagnosis used the TIRADS classification proposed by Kwak; (3) data contained in publications had to allow performing calculations in 2 x 2 pivot tables; (4) conclusive diagnosis was established on the basis of histopathological and/or cytological examination or ultrasound follow-up longer than 12 months in case of benign nodules; (5) the patient must not have been a group subject in previous studies. If samples were part of previously published material, the data from such studies were used to obtain more accurate information on the study group. The decision to classify the study was made independently by two authors (B.M. and M.M.). Discrepancies were resolved by consensus, which occurred in four cases of 42 full-text articles.
Data extraction
The extracted data included authors, country of origin, patient group data (size of group, sex distribution and average age/range of age), number of nodules, study design (prospective, retrospective), reference method and study results. To qualitatively assess the methodology of analysed publications, the widely used and recognized Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) tool was used [19].
Statistical analysis and data synthesis
A random effects model that assumes statistical heterogeneity of study results was used in the meta-analysis. Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), diagnostic accuracy (ACC) and odds ratio (OR) for each study was calculated using 2 x 2 pivot tables. The Spearman correlations coefficient was used to carry out the threshold analysis for the index test. Heterogeneity was assessed using probabilities of the Chi2 (χ2) test by reporting the I2 statistics, which is independent of the number of studies in the meta-analysis. The I2 value varies from 0 % to 100 %, where 0 % means no heterogeneity between studies and values greater than 50 % indicate a significant heterogeneity.
After the assessment of heterogeneity, the following pooled values were calculated: sensitivity, specificity, positive likelihood ratio (LR+), negative likelihood ratio (LR-) and diagnostic odds ratio (DOR). A forest plot was generated for each value. In addition, a funnel plot was used to assess the possibility of errors in publication and statistics using the Egger, Begg and Mazumdar tests to assess the significance of funnel plot asymmetry. The diagnostic usefulness of Kwak’s TIRADS classification was assessed using the summarized receiver operating characteristic (ROC) curve computed via the DerSimonian-Laird random effects model. The area under the curve (AUC), standard error (SE) for AUC, Q* statistics and its SE are reported.
In the case of high heterogeneity, meta-analyses were performed in subgroups and via univariate meta-regression. Grouping variables included the year of publication (before or after 2016), the number of nodules (more or less than 1,000), the country (Korea vs. other), the reference type (cytology and histology vs. cytology, histology and ultrasound follow-up), the cut-off (K-TIRADS category 3/4a vs 4a/4b) and the type of study (retrospective vs. prospective).
P-values less than 0.05 were considered significant. Statistical analyses were conducted using Statistica 13.1 (StatSoft Inc.) and MetaDisc (Freeware Software).
Results
Selection and data extraction
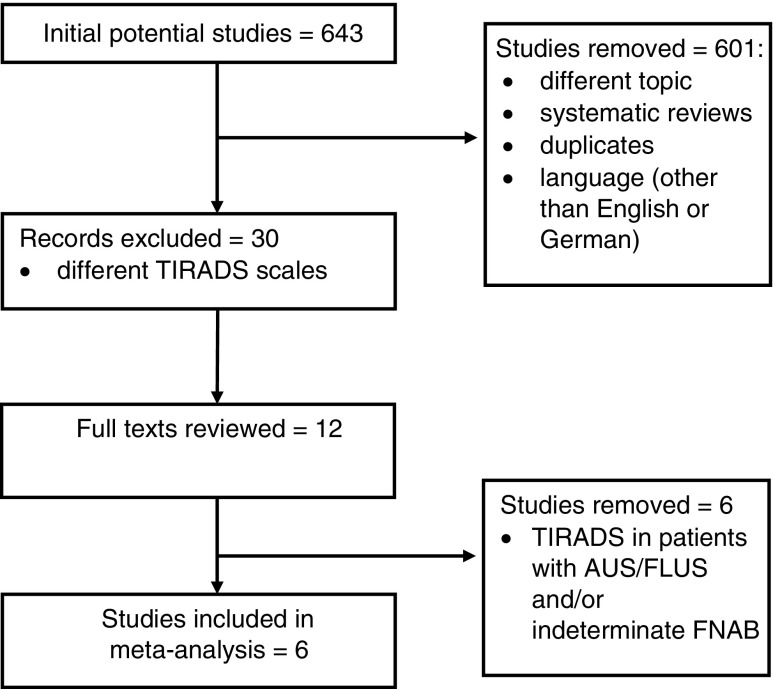
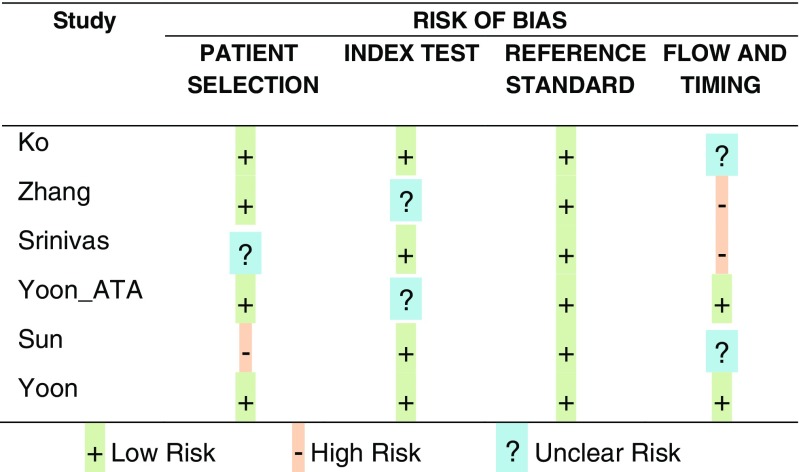
A total of 643 publications were identified upon systematic review of the literature, of which 42 remained after removing duplicates, literature reviews, studies not related to Kwak’s TIRADS and those written in a language other than English or German. Analysis of abstracts and full texts allowed isolation of six original studies that assessed the K-TIRADS scale proposed by Kwak, consistent with the meta-analysis objectives [20–25] (Fig. 1). The six studies that referred to K-TIRADS in patients with AUS/FLUS or indeterminate cytology were excluded [26–31]. A total of 10,926 nodules were included in the meta-analysis. QUADAS-2 classification (Fig. 2) [19], funnel graphs and tests verifying asymmetry were used to assess the quality of included publications.
Fig. 1.
Literature search and selection. TIRADS Thyroid Image Reporting and Data System, AUS/FLUS atypia of undetermined significance/follicular lesion of undetermined significance, FNAB fine needle aspiration biopsy
Fig. 2.
Results of QUADAS-2 assessment for risk of bias in individual studies
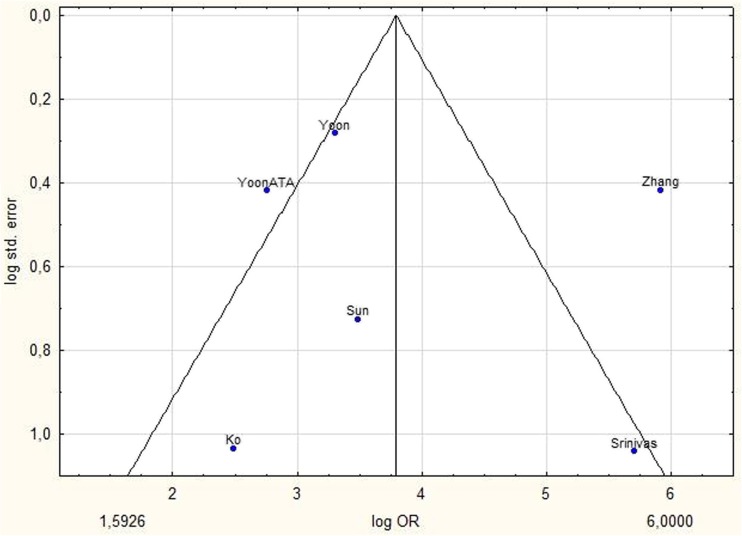
Analysed studies were published between 2014 and 2017. There were two prospective and four retrospective studies. The number of nodules in each publication varied between 204 and 4,696. The average patient age was 47.8 years. In four of six studies, cytological and histological examinations were used for verification. In the study by Zhang and Sun, ultrasound check was also taken into account for TIRADS 2 category lesions with initial benign cytology [21, 25] (Table 1). The cut-off value in four studies was category 4a/4b [20, 22–24] and in last two studies category 3/4a [21, 25]. The funnel graph for analysed studies was asymmetrical, which may suggest publication bias (Fig. 3). Begg, Mazumdar and Eggar tests were used to assess the significance of asymmetry. Finally, the magnitude of the calculated effect in each of the six studies was not strongly enough associated with standard error for tests to confirm the observed asymmetry. None of the tests showed a relationship between the values of effect and their precision (P = 0.6015 by Begg and Mazumdar test, P = 0.7783 by Egger test).
Table 1.
Baseline characteristics of included studies
| Study | Year of publication | Country | Patients | Nodules | Average age | Study design | Reference standard | Sensitivity | Specificity | PPV | NPV | ACC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ko et al. [20] | 2014 | Korea | 195 | 204 | 51 | retrospective | Cytology and histology | 0.985 | 0.158 | 0.354 | 0.957 | 0.422 |
| Zhang et al. [21] | 2015 | China | 2,921 | 3,980 | 51.6 | prospective | Cytology, histology and US follow-up | 0.974 | 0.91 | 0.396 | 0.998 | 0.913 |
| Srinivas et al. [22] | 2016 | India | 365 | 365 | 33.1 | prospective | Cytology and histology | 0.96 | 0.926 | 0.49 | 0.997 | 0.929 |
| Yoon ATA et al. [24] | 2016 | Korea | 1,241 | 1,293 | 50.8 | retrospective | Cytology and histology | 0.974 | 0.293 | 0.233 | 0.981 | 0.416 |
| Sun et al. [25] | 2017 | China | 1,293 | 388 | 49.5 | retrospective | Cytology, histology, and US follow-up | 0.987 | 0.307 | 0.473 | 0.973 | 0.57 |
| Yoon et al. [23] | 2016 | Korea | 4,585 | 4,696 | 50.9 | retrospective | Cytology and histology | 0.988 | 0.255 | 0.275 | 0.986 | 0.418 |
PPV positive predictive value, NPV negative predictive value, ACC diagnostic accuracy
Fig. 3.
Funnel plot shows log standard error by log OR for Kwak’s TIRADS. P-value calculated with Begg and Mazumdar test 0.6015 and Egger test 0.7783 indicates that there was no substantial publication bias. TIRADS Thyroid Image Reporting and Data System
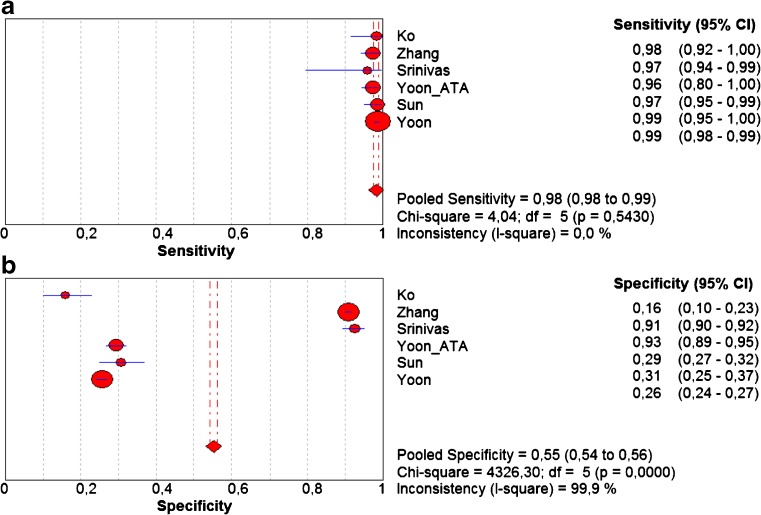
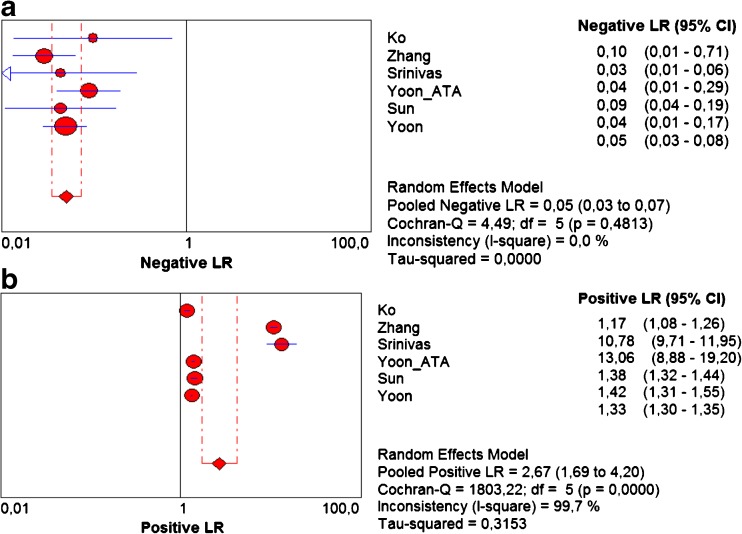
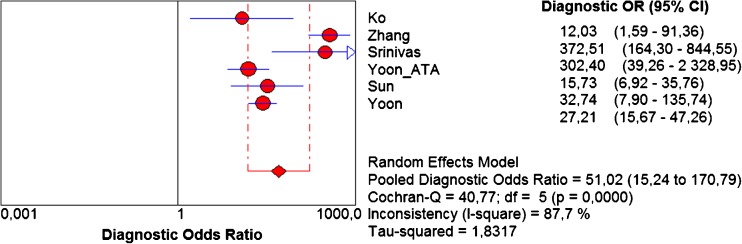
The pooled sensitivity and specificity for K-TIRADS classification were 0.983 (95 % CI 0.976–0.989) and 0.552 (95 % CI 0.542–0.562), respectively (Fig. 4 a,b). Sensitivity was very high in all studies ranging from 0.972 to 0.985. Specificity was more varied and ranged from 0.261 to 0.911. Pooled values of positive likelihood ratio (LR+) and negative likelihood ratio (–LR-) were 2.666 (95 % CI 1.692–4.198) and 0.05 (95 % CI 0.035–0.072), respectively (Fig. 5 a,b). Pooled diagnostic odds ratio (DOR) was 51.020 (95 % CI 15.241–170.79) (Fig. 6). The area under the ROC curve was equal to 0.938 and the Q* statistic was 0.8772, confirming a very good diagnostic accuracy of the analysed test.
Fig. 4.
Forest plots of eligible studies showing individual and pooled (A) sensitivities and (B) specificities of Kwak’s TIRADS in the differential diagnosis of thyroid nodules. Pooled values indicated by diamonds with 95 % confidence intervals (in brackets). TIRADS Thyroid Image Reporting and Data System
Fig. 5.
Forest plots of eligible studies showing individual and pooled (A) negative and (B) positive likelihood ratios (LRs) of Kwak’s TIRADS in the differential diagnosis of thyroid nodules. Pooled values indicated by diamonds with 95 % confidence intervals (in brackets). TIRADS Thyroid Image Reporting and Data System
Fig. 6.
Forest plot of eligible studies showing individual and pooled diagnostic odds ratio (DOR) of Kwak’s TIRADS in the differential diagnosis of thyroid nodules. Pooled value marked by diamonds with 95% confidence interval (in brackets). TIRADS Thyroid Image Reporting and Data System
Analysis in subgroups and meta-regression
Meta-regression was performed in four subgroups, of which heterogeneity was found in two (country of origin p = 0.0037 and study design p < 0.0001). Both subgroups had a significant impact on the heterogeneity of this meta-analysis. The relative DOR for the country of origin was 2.14, while for study design it was 78.61 (Table 2). These results demonstrate that the diagnostic accuracy of K-TIRADS classification in prospective studies was higher than in retrospective studies. Moreover, the studies from outside of Korea had a greater effect on pooled sensitivity and specificity (p < 0.05).
Table 2.
Subgroup analysis
| Subgroup | No. of studies | Pooled sensitivity (95 % CI) | Pooled specificity (95 % CI) | Pooled DOR | Meta-regression RDOR | Meta-regression (p-value) | |
|---|---|---|---|---|---|---|---|
| Total | 6 | 0.983 (0.976–0.989) | 0.552 (0.542–0.562) | 51.020 | |||
| Publication year | < 2016 | 2 | 0.976 (0.951–0.990) | 0.883 (0.872–0.893) | 75.907 | ||
| ≥ 2016 | 4 | 0.985 (0.977–0.990) | 0.308 (0.296–0.321) | 31.260 | 0.71 | 0.2701 | |
| No. of nodules | < 1000 | 4 | 0.983 (0.958–0.995) | 0.572 (0.535–0.608) | 47.350 | ||
| ≥ 1000 | 2 | 0.983 (0.976–0.989) | 0.550 (0.539–0.561) | 53.742 | 0.98 | 0.9301 | |
| Korea vs. other | Korea | 3 | 0.985 (0.977–0.991) | 0.261 (0.249–0.273) | 22.237 | ||
| Other | 3 | 0.978 (0.958–0.990) | 0.878 (0.868–0.887) | 156.13 | 2.14 | 0.0037 | |
| Reference standard | Cytology, histology | 4 | 0.971 (0.961–0.980) | 0.392 (0.379–0.405) | 49.665 | ||
| Cytology, histology and US follow-up | 2 | 0.979 (0.959–0.991) | 0.874 (0.863–0.884) | 118.31 | 0.36 | 0.4241 | |
| Cut–off | 3/4a | 2 | 0.979 (0.959–0.991) | 0.874 (0.863–0.884) | 118.31 | ||
| 4/4b | 4 | 0.971 (0.9610.980) | 0.392 (0.379–0.405) | 49.665 | 0.36 | 0.4241 | |
| Study design | Prospective | 2 | 0.972 (0.944–0.989) | 0.911 (0.902–0.920) | 361.91 | ||
| Retrospective | 4 | 0.985 (0.978–0.991) | 0.263 (0.251–0.275) | 23.023 | 78.61 | <0.0001 | |
CI confidence interval, DOR diagnostic odds ratio, RDOR relative diagnostic odds ratio
Analysed differences in cut-off point and reference standard did not revealed significant importance (p>0.05).
Discussion
Ultrasonography is considered to be the test of choice in preoperative diagnosis of thyroid nodules [6]. There is no doubt that the coexistence of a greater number of suspicious features in a focal lesion significantly increases the risk of malignancy compared to a single suspicious feature. At the same time, the lack of a unified system for categorizing nodules often caused misunderstandings between radiologists and clinicians. The proposed TIRADS classification was intended to be a response to these needs. A large number of studies have been published evaluating the discriminatory capabilities of TIRADS based on different classifications [13, 32–34] and recent WFUMB (World Federation for Ultrasound in Medicine and Biology) guidelines have suggested using TIRADS in order to improve characterization of thyroid nodules, and especially the communication between specialists and patients [35].
Previous meta-analyses evaluating the diagnostic utility of the TIRADS classification took into account its different variants, which could significantly affect the results obtained. In Wei's 2016 study, reported pooled sensitivity and specificity were 0.79 and 0.71, respectively, and for AUC was 0.92 for the summarized ROC. The results of the current meta-analysis including six studies and a total of 10,926 nodules show that the classification proposed by Kwak has a much higher pooled sensitivity of 0.983 and lower specificity of 0.552 in differentiation of thyroid nodules, compared to the previous meta-analyses [36, 37]. Other parameters, such as pooled positive likelihood ratio (2.67, 95 % Cl 1.69–4.2) and negative likelihood ratio (0.05, 95 % Cl 0.03–0.07), were slightly lower than those reported in Wei's study, while the diagnostic odds ratio of 51.02 (95 % CI 15.24–170.79) was considerably higher. Apart from these differences, the parameters also indicate good diagnostic value of the TIRADS classification proposed by Kwak.
The initial assessment of obtained studies suggested heterogeneity, which after more detailed analysis turned out to be statistically insignificant with p > 0.05. Exploration of possible causes revealed that important factors that may increase the heterogeneity of results include study type and country of origin. Analysis of subgroups showed a statistically significant difference between retrospective and prospective studies with p < 0.0001 and significantly higher values of diagnostic odds ratio in prospective studies (361.91 vs. 23.023 for retrospective studies). Further analysis demonstrated that in prospective studies, the pooled sensitivity was lower than in the retrospective study group (0.972 vs. 0.985). The pooled specificity was significantly higher (0.911 vs. 0.263). The results of prospective studies that verify nodules by means of cytological and histopathological evaluation better reflect everyday practice than retrospective studies, which classify material via histopathological verification.
Another essential feature impacting the heterogeneity of results was the country of origin. It is worth noting that three of the six studies originated from Korea, one was from India, and two were from China. Subgroup analysis demonstrated a statistically significant difference between the studies originating from Korea versus other countries (p = 0.0244). In China, cytological verification of focal lesions is rarely performed and thyroid cancer is confirmed mainly on the basis of histological examination [37]. The result is that in the case of patients with nodules classified as TIRADS 1–3 and first cytological, non-diagnostic, atypical or follicular lesion of undetermined significance (Bethesda categories I and III), the invasive diagnostics were not enhanced with another aspiration biopsy or surgery. As a consequence, this causes a lack of diagnosis of cancer in these TIRADS categories and negatively affects the assessment of specificity for this classification [37].
In analysed publications the cut-off point was different. In two cases the cut-off was 3/4a, while in the remaining four it was 4a/4b (Table 2). There is a high difference in specificity, which is higher for 3/4a 0.874 (95 % CI 0.863–0.884) compared to 4a/4b 0.392 (95 % CI 0.379–0.405), when sensitivity for 3/4a 0.979 (95 % CI 0.959–0.991) is almost parallel to 4a/4b 0.971 (95 % CI 0.961–0.980). Similar results were obtained for accepted reference methods. In two cases authors used cytology, histology and ultrasound follow-up for non-suspicious lesions in ultrasound examination or after initial benign cytology (Table 2). In both cases accepted cut-off differences and reference standards did not significantly influence homogeneity of pooled data p > 0.05 (Table 2).
In our results comparing French-TIRADS we report slightly lower specificity, 0.552 versus 0.61, but sensitivity was higher, 0.983 versus 0.957 [15]. The specificity difference could be the consequence of implementation of elastography in French-TIRADS. Obtained pooled data turned out to be lower compared to Horvath’s recent work from 2016 reporting sensitivity of 0.996 and specificity of 0.744. A recently published work on TI-RADS by ACR (American College of Radiology) proposed a different approach [38]. The study is based on a previously published ACR Thyroid Ultrasound Reporting Lexicon [39]. The authors proposed a five-grade scale of TR1 (benign) to TR5 (high suspicion of malignancy) based on scoring of five nodule characteristics (composition, echogenicity, shape, margin and echogenic foci). It was noted that due to the lack of elastography in each ultrasound scanner, it was not included in the ACR TI-RADS. This system needs to be verified as a tool in thyroid nodule stratification.
To minimise the erroneous selection of publications in the course of this systematic review, four databases were used: PubMed, Cochrane database, ScienceDirect and EMBASE. In addition, in order to assess the quality of publications the updated version of the QUADAS-2 tool was used [19].
This systematic review also has some limitations. First, in some studies it was not clearly established if the interpretation of ultrasound images of focal lesions on the basis of tested K-TIRADS classification was carried out without knowledge of the results of reference tests and vice versa. At this level, we cannot clearly determine whether this error resulted from incorrect planning of the original study methodology or from inadequate reporting. Currently, it is recommended that people studying diagnostic accuracy use the Standard for Reporting Diagnostic Accuracy check-list to minimize errors in publication of results [40]. Second, the final diagnosis was not always established on the basis of histopathological examination. Patients in whom lesions were classified as categories 1–3 were included into routine ultrasound follow-up similarly, and not in every case of a focal lesion with category 4–5 was histopathological verification performed; some diagnoses were established on the basis of cytological examination. Third, some articles found in the database searches were rejected as they were in a language other than those approved in the study protocol. Fourth, the disadvantage of previous studies is the small number of non-papillary carcinomas, which often present with a different appearance on the ultrasound examination. Particularly interesting is the group of follicular lesions of indeterminate cytology in which elastography may be useful in differentiating benign and malignant nodules [41]. The creation of a final, comprehensive, ultimate TIRADS classification in the future should include evaluation of a significant number of less prevalent non-papillary cancers as their proper diagnosis is a very important issue from a medical point of view.
Conclusions
K-TIRADS classification has high sensitivity and low specificity. Thus, it is very useful to discard the benign cases and to reduce the number of biopsies. Further prospective, multicentre studies are needed in order to systematize this classification.
Abbreviations and acronyms
- ACC
Accuracy
- ACR
American college of radiology
- AUS/FLUS
Atypia of undetermined significance/follicular lesion of undetermined significance
- B-mode
Grey-scale imaging
- DOR
Diagnostic odds ratio
- FNAB
Fine-needle aspiration biopsy
- LR-, LR+
Negative, positive likelihood ratio
- PICO
Patient, problem, population; intervention, cause, or prognosis; comparison or control; outcome
- PRISMA
Preferred reporting items for systematic reviews and meta-analysis
- QUADAS
Quality assessment of diagnostic accuracy studies
- SE
Standard error
- WFUMB
World federation for ultrasound in medicine and biology
Funding
The authors state that this work has not received any funding.
Compliance with ethical standards
Guarantor
The scientific guarantor of this publication is Ass. Prof. Rafal Zenon Slapa MD.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board for this meta-analysis.
Ethical approval
Institutional Review Board approval was not required because this meta-analysis was based on the results of previous reported studies.
Methodology
• Meta-analysis
• diagnostic or prognostic study
• performed at one institution
References
- 1.Sidoti M, Marino G, Resmini E, et al. The rational use of fine needle aspiration biopsy (FNAB) in diagnosing thyroid nodules. Minerva endocrinologica. 2006;31:159–172. [PubMed] [Google Scholar]
- 2.Kim EK, Park CS, Chung WY, et al. New sonographic criteria for recommending fine-needle aspiration biopsy of nonpalpable solid nodules of the thyroid. AJR Am J Roentgenol. 2002;178:687–691. doi: 10.2214/ajr.178.3.1780687. [DOI] [PubMed] [Google Scholar]
- 3.Morris LF, Ragavendra N, Yeh MW. Evidence-based assessment of the role of ultrasonography in the management of benign thyroid nodules. World J Surg. 2008;32:1253–1263. doi: 10.1007/s00268-008-9494-z. [DOI] [PubMed] [Google Scholar]
- 4.Iannuccilli JD, Cronan JJ, Monchik JM. Risk for malignancy of thyroid nodules as assessed by sonographic criteria: the need for biopsy. J Ultrasound Med. 2004;23:1455–1464. doi: 10.7863/jum.2004.23.11.1455. [DOI] [PubMed] [Google Scholar]
- 5.Moon WJ, Jung SL, Lee JH, et al. Benign and malignant thyroid nodules: US differentiation--multicenter retrospective study. Radiology. 2008;247:762–770. doi: 10.1148/radiol.2473070944. [DOI] [PubMed] [Google Scholar]
- 6.Kwak JY, Han KH, Yoon JH, et al. Thyroid imaging reporting and data system for US features of nodules: a step in establishing better stratification of cancer risk. Radiology. 2011;260:892–899. doi: 10.1148/radiol.11110206. [DOI] [PubMed] [Google Scholar]
- 7.Jarzab B, Dedecjus M, Handkiewicz-Junak D, et al. Diagnostics and Treatment of Thyroid Carcinoma. Endokrynol Pol. 2016;67:74–145. doi: 10.5603/EP.2016.0011. [DOI] [PubMed] [Google Scholar]
- 8.Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26:1–133. doi: 10.1089/thy.2015.0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Trzebinska A, Dobruch-Sobczak K, Jakubowski W, Jedrzejowski M. Standards of the Polish Ultrasound Society - update. Ultrasound examination of thyroid gland and ultrasound-guided thyroid biopsy. J Ultrason. 2014;14:49–60. doi: 10.15557/JoU.2014.0005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lazarus E, Mainiero MB, Schepps B, Koelliker SL, Livingston LS. BI-RADS lexicon for US and mammography: interobserver variability and positive predictive value. Radiology. 2006;239:385–391. doi: 10.1148/radiol.2392042127. [DOI] [PubMed] [Google Scholar]
- 11.Stavros AT, Thickman D, Rapp CL, Dennis MA, Parker SH, Sisney GA. Solid breast nodules: use of sonography to distinguish between benign and malignant lesions. Radiology. 1995;196:123–134. doi: 10.1148/radiology.196.1.7784555. [DOI] [PubMed] [Google Scholar]
- 12.Radiology ACo (2003) BI-RADS: ultrasound. Breast Imaging Reporting and Data System Altas (BI-RADS Atlas). Am Coll Radiol, Am Coll Radiol
- 13.Horvath E, Majlis S, Rossi R, et al. An ultrasonogram reporting system for thyroid nodules stratifying cancer risk for clinical management. J Clin Endocrinol Metab. 2009;94:1748–1751. doi: 10.1210/jc.2008-1724. [DOI] [PubMed] [Google Scholar]
- 14.Park JY, Lee HJ, Jang HW, et al. A proposal for a thyroid imaging reporting and data system for ultrasound features of thyroid carcinoma. Thyroid. 2009;19:1257–1264. doi: 10.1089/thy.2008.0021. [DOI] [PubMed] [Google Scholar]
- 15.Russ G, Royer B, Bigorgne C, Rouxel A, Bienvenu-Perrard M, Leenhardt L. Prospective evaluation of thyroid imaging reporting and data system on 4550 nodules with and without elastography. Eur J Endocrinol. 2013;168:649–655. doi: 10.1530/EJE-12-0936. [DOI] [PubMed] [Google Scholar]
- 16.Choi YJ, Baek JH, Baek SH, et al. Web-Based Malignancy Risk Estimation for Thyroid Nodules Using Ultrasonography Characteristics: Development and Validation of a Predictive Model. Thyroid. 2015;25:1306–1312. doi: 10.1089/thy.2015.0188. [DOI] [PubMed] [Google Scholar]
- 17.Maia FF, Matos PS, Pavin EJ, Zantut-Wittmann DE. Thyroid imaging reporting and data system score combined with Bethesda system for malignancy risk stratification in thyroid nodules with indeterminate results on cytology. Clin Endocrinol (Oxf) 2015;82:439–444. doi: 10.1111/cen.12525. [DOI] [PubMed] [Google Scholar]
- 18.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Whiting PF, Rutjes AW, Westwood ME, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155:529–536. doi: 10.7326/0003-4819-155-8-201110180-00009. [DOI] [PubMed] [Google Scholar]
- 20.Ko SY, Lee HS, Kim E-K, Kwak JY. Application of the Thyroid Imaging Reporting and Data System in thyroid ultrasonography interpretation by less experienced physicians. Ultrasonography (Seoul, Korea) 2014;33:49–57. doi: 10.14366/usg.13016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang J, Liu BJ, Xu HX, et al. Prospective validation of an ultrasound-based thyroid imaging reporting and data system (TI-RADS) on 3980 thyroid nodules. Int J Clin Exp Med. 2015;8:5911–5917. [PMC free article] [PubMed] [Google Scholar]
- 22.Srinivas MN, Amogh VN, Gautam MS, et al. A Prospective Study to Evaluate the Reliability of Thyroid Imaging Reporting and Data System in Differentiation between Benign and Malignant Thyroid Lesions. J Clin Imaging Sci. 2016;6:5. doi: 10.4103/2156-7514.177551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yoon JH, Han K, Kim E-K, Moon HJ, Kwak JY. Diagnosis and Management of Small Thyroid Nodules: A Comparative Study with Six Guidelines for Thyroid Nodules. Radiology. 2016;283:560–569. doi: 10.1148/radiol.2016160641. [DOI] [PubMed] [Google Scholar]
- 24.Yoon JH, Lee HS, Kim EK, Moon HJ, Kwak JY. Malignancy Risk Stratification of Thyroid Nodules: Comparison between the Thyroid Imaging Reporting and Data System and the 2014 American Thyroid Association Management Guidelines. Radiology. 2016;278:917–924. doi: 10.1148/radiol.2015150056. [DOI] [PubMed] [Google Scholar]
- 25.Sun CY, Lei KR, Liu BJ, et al. Virtual touch tissue imaging and quantification (VTIQ) in the evaluation of thyroid nodules: the associated factors leading to misdiagnosis. Sci Rep. 2017;7:41958. doi: 10.1038/srep41958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moon HJ, Kim EK, Yoon JH, Kwak JY. Malignancy risk stratification in thyroid nodules with nondiagnostic results at cytologic examination: combination of thyroid imaging reporting and data system and the Bethesda System. Radiology. 2015;274:287–295. doi: 10.1148/radiol.14140359. [DOI] [PubMed] [Google Scholar]
- 27.Yoon JH, Lee HS, Kim EK, Moon HJ, Kwak JY. Thyroid Nodules: Nondiagnostic Cytologic Results according to Thyroid Imaging Reporting and Data System before and after Application of the Bethesda System. Radiology. 2015;276:579–587. doi: 10.1148/radiol.15142308. [DOI] [PubMed] [Google Scholar]
- 28.Baser H, Cakir B, Topaloglu O et al (2016) Diagnostic accuracy of Thyroid Imaging Reporting and Data System in the prediction of malignancy in nodules with atypia and follicular lesion of undetermined significance cytologies. Clin Endocrinol (Oxf). 10.1111/cen.13274 [DOI] [PubMed]
- 29.Grani G, Lamartina L, Ascoli V et al (2016) Ultrasonography scoring systems can rule out malignancy in cytologically indeterminate thyroid nodules. Endocrine:1-6 [DOI] [PubMed]
- 30.Park VY, Kim EK, Kwak JY, Yoon JH, Kim MJ, Moon HJ. Thyroid Imaging Reporting and Data System and Ultrasound Elastography: Diagnostic Accuracy as a Tool in Recommending Repeat Fine-Needle Aspiration for Solid Thyroid Nodules with Non-Diagnostic Fine-Needle Aspiration Cytology. Ultrasound Med Biol. 2016;42:399–406. doi: 10.1016/j.ultrasmedbio.2015.10.011. [DOI] [PubMed] [Google Scholar]
- 31.Yoon JH, Kwon HJ, Kim EK, Moon HJ, Kwak JY. Subcategorization of atypia of undetermined significance/follicular lesion of undetermined significance (AUS/FLUS): a study applying Thyroid Imaging Reporting and Data System (TIRADS) Clin Endocrinol (Oxf) 2016;85:275–282. doi: 10.1111/cen.12987. [DOI] [PubMed] [Google Scholar]
- 32.Na DG, Baek JH, Sung JY, et al. Thyroid Imaging Reporting and Data System Risk Stratification of Thyroid Nodules: Categorization Based on Solidity and Echogenicity. Thyroid. 2016;26:562–572. doi: 10.1089/thy.2015.0460. [DOI] [PubMed] [Google Scholar]
- 33.Russ G. Risk stratification of thyroid nodules on ultrasonography with the French TI-RADS: description and reflections. Ultrasonography. 2016;35:25–38. doi: 10.14366/usg.15027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zayadeen AR, Abu-Yousef M, Berbaum K (2016) Retrospective Evaluation of Ultrasound Features of Thyroid Nodules to Assess Malignancy Risk: A Step Toward TIRADS. AJR Am J Roentgenol. 10.2214/AJR.15.15121:1-10 [DOI] [PubMed]
- 35.Cosgrove D, Barr R, Bojunga J, et al. WFUMB Guidelines and Recommendations on the Clinical Use of Ultrasound Elastography: Part 4. Thyroid. Ultrasound Med Biol. 2017;43:4–26. doi: 10.1016/j.ultrasmedbio.2016.06.022. [DOI] [PubMed] [Google Scholar]
- 36.Wei X, Li Y, Zhang S, Gao M. Thyroid imaging reporting and data system (TI-RADS) in the diagnostic value of thyroid nodules: a systematic review. Tumour Biol. 2014;35:6769–6776. doi: 10.1007/s13277-014-1837-9. [DOI] [PubMed] [Google Scholar]
- 37.Wei X, Li Y, Zhang S, Gao M. Meta-analysis of thyroid imaging reporting and data system in the ultrasonographic diagnosis of 10,437 thyroid nodules. Head Neck. 2016;38:309–315. doi: 10.1002/hed.23878. [DOI] [PubMed] [Google Scholar]
- 38.Tessler FN, Middleton WD, Grant EG et al (2017) ACR Thyroid Imaging, Reporting and Data System (TI-RADS): White Paper of the ACR TI-RADS Committee. J Am Coll Radiol. 10.1016/j.jacr.2017.01.046 [DOI] [PubMed]
- 39.Grant EG, Tessler FN, Hoang JK, et al. Thyroid Ultrasound Reporting Lexicon: White Paper of the ACR Thyroid Imaging, Reporting and Data System (TIRADS) Committee. J Am Coll Radiol. 2015;12:1272–1279. doi: 10.1016/j.jacr.2015.07.011. [DOI] [PubMed] [Google Scholar]
- 40.Bossuyt PM, Reitsma JB, Bruns DE, et al. Towards complete and accurate reporting of studies of diagnostic accuracy: The STARD Initiative. Radiology. 2003;226:24–28. doi: 10.1148/radiol.2261021292. [DOI] [PubMed] [Google Scholar]
- 41.Cantisani V, Maceroni P, D'Andrea V, et al. Strain ratio ultrasound elastography increases the accuracy of colour-Doppler ultrasound in the evaluation of Thy-3 nodules. A bi-centre university experience. Eur Radiol. 2016;26:1441–1449. doi: 10.1007/s00330-015-3956-0. [DOI] [PubMed] [Google Scholar]