Abstract
The epidemiology of tuberculosis (TB) in high-income countries is increasingly dictated by immigration. The influence of this trend on paediatric TB and TB elimination are not well defined.
We undertook a 25-year conventional and molecular epidemiologic study of paediatric TB in Alberta, one of four major immigrant-receiving provinces in Canada. All isolates of Mycobacterium tuberculosis were DNA fingerprinted using standard methodology.
Between 1990 and 2014, 176 children aged 0–14 years were diagnosed with TB. Foreign-born children or Canadian-born children of foreign-born parents accounted for an increasingly large proportion of total cases during the study period (from 32.1% to 89.5%). Of the 78 culture-positive cases, 35 (44.9%) had a putative source case identified by conventional epidemiology, with 34 (97.1%) having a concordant molecular profile. Of the remaining 43 culture-positive cases, molecular profiling identified spatially and temporally related sources in six cases (14.0%). These six children, along with four other children whose source cases were discovered through reverse-contact tracing, had a high morbidity and mortality.
The increasing burden of paediatric TB in both foreign-born children and Canadian-born children of foreign-born parents calls for more timely diagnosis of source cases and more targeted screening for latent TB infection.
Short abstract
Conventional and molecular epidemiology can inform paediatric TB elimination strategy in high-income countries http://ow.ly/mwbn30iY1WF
Introduction
As public health programmes focus on strategies to eliminate tuberculosis (TB), childhood TB is an increasingly important indicator of the ability of a programme to interrupt transmission and prevent TB among those already infected [1]. In many high-income low-incidence countries these strategies are in a state of transition largely due to the impact that immigration from high-incidence countries is having upon the epidemiology of the disease [2–9]. This is especially true of Canada which accepts approximately 250 000 new immigrants each year, most of whom settle in one of four provinces: Quebec, Ontario, British Columbia and Alberta. In such settings, childhood TB may be a sentinel event reflecting recent local transmission, or may result from reactivation of an infection that was acquired abroad under circumstances where screening of contacts and treatment of latent TB infection (LTBI) may not be performed. In recent years a deeper understanding of childhood TB in the context of immigration has emerged by combining conventional epidemiology (CE) and molecular epidemiology (ME). However, studies to date have either been limited in time and space, or have not been delimited by age [10–14]. For purposes of reporting childhood TB the World Health Organization (WHO) recommends two age bands: 0–4 and 5–14 years [15].
To overcome the aforementioned limitations we undertook a 25-year programme-wide paediatric TB study in the Province of Alberta, Canada. During the study period, Alberta received 523 473 new permanent residents and many more temporary residents and visitors (Statistics Canada). To standardise the definition of intrathoracic TB, chest radiographs were re-read by an experienced paediatric chest radiologist [16]. All isolates of Mycobacterium tuberculosis in children and greater than 95% of those in adults were DNA fingerprinted. Rates of disease were reported in two age bands: 0–4 and 5–14 years. Our purpose was to describe trends in paediatric TB as defined by the WHO and to identify groups of children at risk of TB in the context of immigration. In addition, we sought to identify those strategies most likely to achieve the elimination of paediatric TB in Canada and other high-income, low-incidence, immigrant-receiving countries.
Methods
In Canada, each province and territory has its own TB prevention and care program. This retrospective cohort study was performed in the Province of Alberta, population 4 252 900 in 2016 (Statistics Canada). In Alberta, all TB services are provided via three public health TB clinics [17]. All incident cases are treated with directly-observed therapy.
Population
Individuals born in Canada or born outside of Canada to Canadian-born parents were considered Canadian-born; all others were foreign-born. The Canadian-born population was divided into registered First Nations (RFN; North American Indians registered with Indian and Northern Affairs Canada (INAC)) and Canadian-born “others” because of known differences in the TB case rates of these groups. Canadian-born “others” (CBO) were further grouped as those with Canadian-born parents (CBO-CB) and those with at least one foreign-born parent (CBO-FB). The population estimates used in the analysis were derived from Canadian censuses and RFN population estimates provided by INAC [17].
Paediatric cases
All TB cases in Alberta between January 01, 1990 and December 31, 2014 (the “study period”) were eligible for study inclusion, as determined from the provincial TB Registry. Data from the TB Registry were combined with data from the Provincial Laboratory for Public Health (ProvLab), where all mycobacteriology was performed. Individuals under 15 years of age at diagnosis were defined as paediatric cases and all others as adults [15, 16].
Each paediatric case in the TB Registry was verified as being a “true” paediatric case through a comprehensive review process. First, the disease status of each notified paediatric TB case was validated through a review of the diagnostic data in their individual medical and laboratory records. Secondly, the chest radiographs of each “pulmonary” case were re-read by an experienced paediatric chest radiologist. Only children with a positive culture for M. tuberculosis complex or those meeting a strict clinical case definition were considered to be paediatric TB cases and included in the final dataset. The clinical case definition required that four criteria be met: 1) a history of contact with a known source case and/or a positive tuberculin skin test (TST); 2) a chest radiographic abnormality consistent with disease if pulmonary, or a clinical or radiologic picture consistent with extrapulmonary disease if extrapulmonary (in some instances clinical and/or radiologic findings were supported by pathologic findings); 3) a satisfactory clinical and radiographic response to treatment; and 4) a completed diagnostic work-up [16]. The Mantoux method was used for TSTs and results were interpreted in accordance with the Canadian TB Standards [16, 18].
Conventional and molecular epidemiology
Conventional contact tracing and molecular typing were used to reveal and/or verify linkages between paediatric TB case-patients and other active TB cases notified in Alberta. The ProvLab completed routine DNA fingerprinting as previously described using the IS6110 restriction fragment-length polymorphism (RFLP) method and, for isolates with fewer than six copies of IS6110, spoligotyping [19, 20]. Clusters were defined as groups of two or more isolates with identical RFLP patterns and, for low IS6110 copy number isolates, identical spoligotype patterns as well. Conventional contact tracing was completed by TB program nursing staff using the concentric circle approach [21]. A putative source case was defined as a culture-positive pulmonary case in an adult contact of the paediatric case patient. Putative source cases were considered to be verified if their M. tuberculosis molecular profile matched that of the paediatric case. Further reviews were undertaken to establish linkages between paediatric cases and other cases with matching DNA fingerprint patterns if no putative source case had initially been identified, or if the putative source case was refuted by genotyping. In such instances, the case with the matching DNA fingerprint was only considered to be the source case if temporal (diagnosed within 6 months of the paediatric case) and spatial (first three digits of the postal code) linkages could be established with the paediatric case patient. Culture-positive cases diagnosed in 2014 were followed in our DNA fingerprint database to the end of June, 2015.
Statistical analysis
Data were analysed using Stata/IC 11 (StataCorp LP, College Station, TX, USA). Incidence rates per 100 000 person-years were calculated overall, for each age band (0–4 years and 5–14 years) and for each population group. The calculation of 95% confidence intervals for rates assumed a Poisson distribution for case counts. Standardised incidence rates were calculated using the direct method, with the age and sex distribution of the total paediatric population in Alberta (1990–2014) serving as the standard population. Standardised rates were compared with incidence rate ratios (RRs) and 95% confidence intervals. Measures of association were assessed with bivariate and multivariate logistic regression at a 5% level of significance. Pearson's Chi-squared test or Fisher's exact test were used as appropriate for comparisons in bivariate analyses. Significance levels were reported at 5% and confidence intervals were reported at 95%.
Ethics approval was received from the University of Alberta Health Research Ethics Board and the London School of Hygiene and Tropical Medicine.
Results
The TB Registry was notified of 3817 TB cases during the study period and 225 (5.9%) of these cases were children. Review of the 225 child-aged cases found that 176 (78.2%) met the inclusion criteria for a paediatric TB case as defined in this study. Of the 49 (21.8%) cases that were excluded, 36 culture-negative “primary pulmonary” cases had, in retrospect, either normal chest radiographs (n=34) or abnormal chest radiographs not indicative of TB (n=2) and five culture-negative extrapulmonary cases had inconclusive biopsies and/or other investigations. These cases could be considered “provider diagnosed” [9]. Eight other children were from a jurisdiction outside Alberta. Of the “true” paediatric cases 78 (44.3%) had a positive culture. In comparison, M. tuberculosis was isolated from 3020 (84.1%) of the 3592 adult cases of TB.
Canadian-born children accounted for 117 (66.5%) of the 176 paediatric TB cases, with RFN and CBO children comprising similar proportions of the total cohort (34.7% and 31.8%, respectively). Of the 61 CBO cases, 43 (70.5%) had at least one foreign-born parent. In addition, 57 (96.6%) of the 59 foreign-born TB case-patients were born in a high TB incidence country (≥15 smear-positive pulmonary cases per 100 000 population) and 21 (36.8%) of these children were <5 years of age when they immigrated to Canada [18]. The leading country of birth of foreign-born children was the Philippines with 14 children, followed by Ethiopia, Sudan, Kenya, Pakistan and Haiti with four each, and Vietnam with three. Furthermore, 36 (61.0%) of the foreign-born case-patients were diagnosed with TB within two calendar years of arrival.
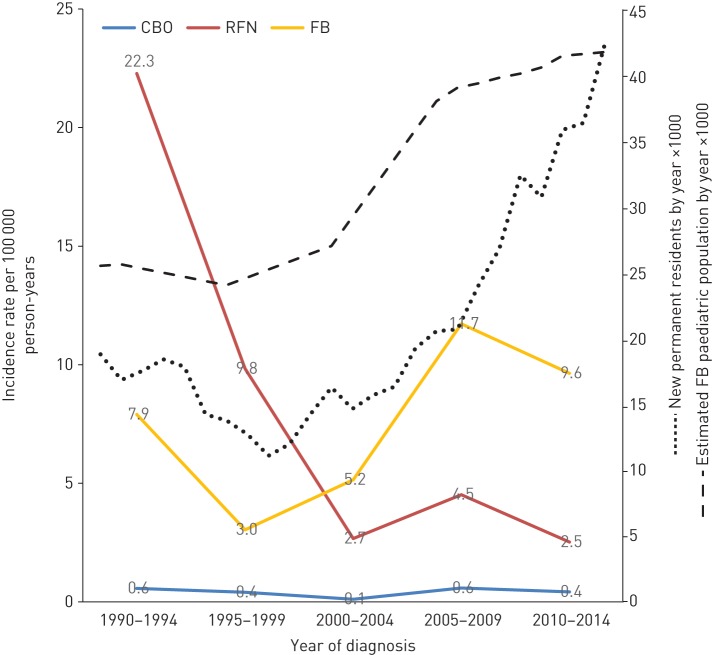
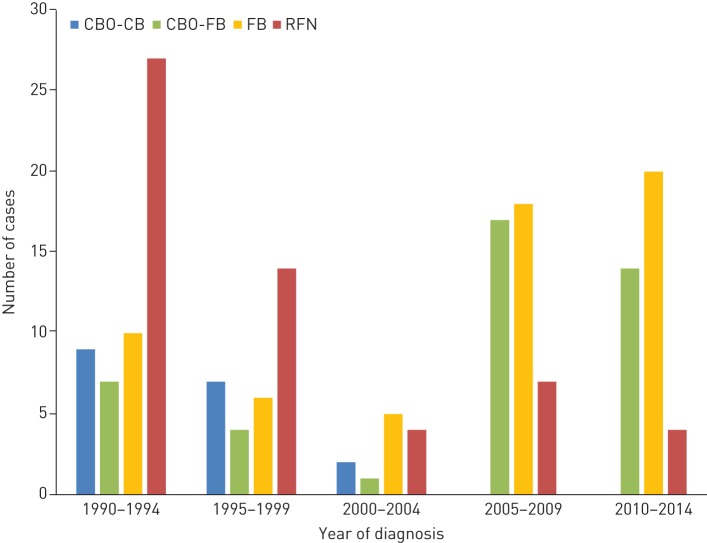
RFN and foreign-born children had age- and sex-adjusted TB rates that were, respectively, 16- and 18-times that of “other” Canadian-born children (table 1). Children in the 0–4 years age group had the highest rates regardless of population group. Between 1990–1994 and 2010–2014 there was a drop in the overall rate from 1.78 to 1.07 per 100 000 person-years; however, this relatively small change masked a remarkable decrease in the RFN rate (22.3 to 2.5 per 100 000 person-years) and an increase in the foreign-born rate (7.9 to 9.6 per 100 000 person-years) (figure 1). Foreign-born children accounted for an increasingly large proportion of total paediatric cases during the study period (from 18.9% in 1990–1994 to 52.6% in 2010–2014). Another trend was that all CBO cases in 2005–2014 were CBO-FB (figure 2).
TABLE 1.
The incidence of paediatric tuberculosis (TB) in Alberta by population group and age, 1990–2014
| Grouping | Cases n (%) | Observation person-years (×100 000)# | Incidence rate | RRadj (95% CI) | |
| Unadjusted | Adjusted (95% CI)¶ | ||||
| By population group | |||||
| Overall | 176 (100) | 159.37 | 1.10 | 1.20 (1.01–1.20) | |
| CBO | 61 (36) | 143.85 | 0.42 | 0.54 (0.39–0.66) | 1.00 |
| RFN | 56 (34) | 7.50 | 7.46 | 8.34 (6.13–11.07) | 16.37 (10.91–24.48) |
| FB | 59 (30) | 8.01 | 7.36 | 9.29 (6.43–12.95) | 18.25 (11.59–28.30) |
| By age and population group | |||||
| 0–4 years | |||||
| Overall | 78 (100) | 5.31 | 1.47 | 1.47 (1.16–1.83) | |
| CBO | 39 (50) | 4.95 | 0.79 | 0.79 (0.56–1.08) | 1.00 |
| RFN | 23 (29) | 0.22 | 10.48 | 10.48 (6.64–15.72) | 13.31 (7.59–22.85) |
| FB | 16 (21) | 0.13 | 12.22 | 12.22 (6.99–19.85) | 15.53 (8.10–28.44) |
| 5–14 years | |||||
| Overall | 98 (100) | 10.63 | 0.92 | 0.92 (0.75–1.12) | |
| CBO | 22 (22) | 9.43 | 0.23 | 0.23 (0.15–0.35) | 1.00 |
| RFN | 33 (34) | 0.53 | 6.21 | 6.22 (4.29–8.75) | 26.70 (15.10–48.10) |
| FB | 43 (44) | 0.67 | 6.41 | 6.41 (4.64–8.64) | 27.50 (16.09–48.28) |
RRadj: age-sex adjusted incidence rate ratio; CBO: Canadian-born “other”; RFN: registered First Nations; FB: foreign born. #: based upon Canadian censuses conducted in 1986, 1991, 1996, 2001, 2006 and 2011, as well as registered First Nations population estimates provided by Indian and Northern Affairs Canada; ¶: adjusted rates by population group were standardised by age and sex, while adjusted rates by age and population group were standardised by sex.
FIGURE 1.
Age- and sex-adjusted incidence of paediatric tuberculosis (TB) by population group and time period in Alberta, 1990–2014. Population figures are from Statistics Canada. CBO: Canadian-born “other”; RFN: registered First Nations; FB: foreign born.
FIGURE 2.
Paediatric tuberculosis (TB) cases by 5-year period and population group in Alberta, 1990–2014. CBO-CB: Canadian-born “other” with Canadian-born parents; CBO-FB: Canadian-born “other” with one or more foreign-born parents; RFN: registered First Nations; FB: foreign born.
The age at diagnosis varied significantly by population group (table 2). The odds of TB before 5 years of age were higher for the CBO-FB group than for RFN or foreign-born children (2.97-times (95% CI 1.29–6.82) and 5.57-times (95% CI 2.36–13.13), respectively). The proportion of female paediatric case-patients was similar in each population group. HIV co-infection was rare and, from 2003 when “opt-out” HIV testing was introduced, only one of 73 children tested was HIV positive [22].
TABLE 2.
Demographic and clinical characteristics of paediatric tuberculosis (TB) cases by population group in Alberta, 1990–2014
| Characteristics | Population group | Total | p-value# | |||
| RFN | CBO-CB | CBO-FB | FB | |||
| Cases | 56 | 18 | 43 | 59 | 176 | |
| Age years | 0.001 | |||||
| <5 | 23 (41) | 10 (56) | 29 (67) | 16 (27) | 78 (44) | |
| 5–14 | 33 (59) | 8 (44) | 14 (33) | 43 (73) | 98 (56) | |
| Sex | 0.09 | |||||
| Female | 25 (45) | 10 (56) | 30 (70) | 35 (59) | 100 (57) | |
| Male | 31 (55) | 8 (44) | 13 (30) | 24 (41) | 76 (43) | |
| Disease site | 0.03 | |||||
| Pulmonary | 51 (91) | 14 (78) | 36 (84) | 41 (70) | 142 (81) | |
| Extrapulmonary¶ | 5 (9) | 4 (22) | 7 (16) | 18 (31) | 34 (19) | |
| Method of detection | <0.001 | |||||
| Contact tracing | 52 (93) | 14 (78) | 27 (63) | 15 (25) | 108 (61) | |
| Symptoms | 4 (7) | 4 (22) | 14 (33) | 37 (63) | 59 (34) | |
| Other screening+ | 0 (0) | 0 (0) | 2 (5) | 7 (12) | 9 (5) | |
| Culture status | 0.47 | |||||
| Positive | 22 (39) | 6 (33) | 16 (37) | 34 (58) | 78 (44) | |
| Negative | 32 (57) | 9 (50) | 24 (56) | 25 (42) | 90 (51) | |
| Unknown§ | 2 (4) | 3 (17) | 3 (7) | 0 (0) | 8 (5) | |
Data are presented as n or n (%), unless otherwise stated. CBO-CB: Canadian-born “other” with Canadian-born parents; CBO-FB: Canadian-born “other” with foreign-born parents; RFN: registered First Nations; FB: foreign born. #: Pearson's Chi-squared test or Fisher's exact test if cells contain values <5; ¶: Extrapulmonary includes all cases with an International Classification of Diseases Ninth Revision (ICD-9) code of 013 (TB of meninges and the central nervous system) and/or 018 (miliary TB) with or without disease in other sites. It also includes the single ICD-9 codes 014 (TB of the intestines, peritoneum and mesenteric glands), 015 (TB of the bones and joints), 016 (TB of the genito-urinary system) and 017 (TB of other organs); +: other screening includes immigration medical evaluations, international adoptee screening and incidental findings; §: unknown means that no diagnostic specimens were submitted.
Contact tracing was the most frequent means of detecting paediatric TB cases overall (table 2). CBO-FB cases were 6.28-times (95% CI 1.89–20.85) more likely to have disease detected due to symptoms than RFN children. In addition to disease detection through contact tracing and symptoms, nine cases were detected by other screening methods including immigration medical evaluations, international adoptee screening and incidental findings [23].
Of the 168 cases from whom culture specimens were collected, 78 (46.4%) were culture-positive (table 3). Children ≥5 years of age, children with extrapulmonary TB and children whose method of detection was symptoms were more likely than children <5 years of age, children with pulmonary TB, or children whose method of detection was contact tracing to be culture-positive. Of the 78 M. tuberculosis isolates, seven were resistant to either streptomycin, isoniazid, or both. All strains with resistance were from foreign-born children or CBO-FB children. No isolates were multidrug-resistant (MDR).
TABLE 3.
Culture status of paediatric tuberculosis (TB) cases in Alberta by demographic and clinical characteristics, 1990–2014
| Characteristics | Total | Culture specimen | p-value# | OR (95% CI) | |
| Negative | Positive | ||||
| Cases | 168 | 90 (54) | 78 (46) | ||
| Age years | 0.01 | ||||
| <5 | 71 | 46 (65) | 25 (35) | 1 | |
| 5–14 | 97 | 44 (45) | 53 (55) | 2.21 (1.16–4.21) | |
| Sex | 0.23 | ||||
| Female | 93 | 46 (50) | 47 (51) | 1 | |
| Male | 75 | 44 (59) | 31 (41) | 0.69 (0.37–1.27) | |
| Population group | 0.20 | ||||
| CBO-CB | 15 | 9 (60) | 6 (40) | 1 | |
| CBO-FB | 40 | 24 (60) | 16 (40) | 1.00(0.30–3.36) | |
| RFN | 54 | 32 (59) | 22 (41) | 0.97(0.30–3.11) | |
| FB | 59 | 25(42) | 34(58) | 0.49(0.15–1.56) | |
| Disease site | 0.02 | ||||
| Pulmonary | 136 | 79 (58) | 57 (42) | 1 | |
| Extrapulmonary | 32 | 11 (34) | 21 (66) | 0.38 (0.17–0.85) | |
| Method of detection | <0.001 | ||||
| Symptoms | 58 | 19 (33) | 39 (67) | 1 | |
| Contact tracing¶ | 101 | 69 (68) | 32 (32) | 0.22 (0.11–0.45) | |
| Other screening+ | 9 | 2 (22) | 7 (78) | 1.71 (0.32–9.01) | |
Data are presented as n or n (%), unless otherwise stated. CBO-CB: Canadian-born “other” with Canadian-born parents; CBO-FB: Canadian-born “other” with foreign-born parents; RFN: registered First Nations; FB: foreign born; OR: odds ratio (crude and unadjusted for any other variables in the table). #: Pearson's Chi-squared test or Fisher's exact test if cells contain values <5. ¶: of the 101 children whose method of detection was contact tracing and who had one or more specimens submitted for culture, 85 had primary pulmonary TB complex alone (see figure 3). Of these only 22 (25.9%) were culture-positive. +: other screening includes immigration medical evaluations, international adoptee screening and incidental findings.
CE identified a putative source case for 111 (63.1%) paediatric case-patients (table 4). In 107 of these the putative source case was the index case; however, in four cases the child was the index case, detected through symptoms, and the putative source case was discovered through reverse-contact tracing. Of these latter cases, one had congenital TB and two had central nervous system (CNS)-TB. All three were <2 years of age and all three died. Population group was independently associated with the ability to identify a putative source case, although age or sex at diagnosis were not.
TABLE 4.
Utility of conventional epidemiology (CE) for the identification of paediatric tuberculosis (TB) source cases
| Characteristic | Total | Source case identified by CE | OR (95% CI) | |
| Yes | No | |||
| Cases | 176 | 111 (63) | 65 (37) | |
| Age years | ||||
| <5 | 78 | 54 (69) | 24 (31) | 1.0 |
| 5–14 | 98 | 57 (58) | 41 (42) | 1.62 (0.87–3.03) |
| Sex | ||||
| Female | 100 | 59 (59) | 41 (41) | 1.0 |
| Male | 76 | 52 (68) | 24 (32) | 0.66 (0.35–1.24) |
| Population group | ||||
| CBO | 61 | 43 (71) | 18 (30) | 1.0 |
| RFN | 56 | 53 (95) | 3 (5) | 0.14 (0.04–0.50) |
| FB | 59 | 15 (25) | 44 (75) | 7.01 (3.14–15.66) |
| Method of detection | ||||
| Contact tracing | 108 | 107 (99) | 1 (1)# | |
| Symptoms | 59 | 4 (4)¶ | 55 (97) | |
| Other screening+ | 9 | 0 (0) | 9 (100) | |
Data is presented as n or n (%), unless otherwise stated. CBO: Canadian-born “other”; RFN: registered First Nations; FB: foreign born; OR: odds ratio. #: this child was a close contact of a relative who lived in a neighbouring province-there was no source case in Alberta; ¶: four children were index cases with symptoms and their source cases were identified through reverse contact tracing; +: other screening includes immigration medical evaluations, international adoptee screening and incidental findings.
DNA fingerprints of isolates from the 78 culture-positive paediatric case-patients were compared to the entire database of DNA fingerprints obtained during the study period, which included the molecular profiles of 2897 (95.9%) of the 3020 culture-positive adult case-patients. Of the 78 DNA fingerprinted paediatric cases, 34 (97.1%) had a source case identified by CE with a concordant molecular profile (table 5). Only one paediatric case had a different molecular profile than their putative source case identified via contact tracing. This child was an older CBO-FB case with no travel/visitor history and no alternate source case by ME. Molecular profiling also identified spatially- and temporally-related source cases for six (14.0%) of the 43 culture-positive children for whom putative source cases were not identified by CE. Four of these child–source case-pairs were subsequently co-located to an apartment complex (n=2), a taxi (n=1) and a hotel room (n=1). Three of these four children, all aged <2 years, had CNS-TB and one died. Source cases for the remaining 37 (21.0%) culture-positive paediatric cases, the majority of whom were over 4 years of age (73.0%) and/or foreign-born (81.8%) could not be identified through either CE or ME investigations.
TABLE 5.
Utility of molecular epidemiology (ME) for the identification of paediatric tuberculosis (TB) source cases
| Characteristic | Total# | Source case identified by CE | No source case identified by CE | ||
| Subtotal | Identified by ME | Subtotal | Identified by ME¶ | ||
| Cases | 78 | 35 | 34 (97) | 43 | 6 (14) |
| Age years | |||||
| <5 | 25 | 11 | 11 (100) | 14 | 4 (29) |
| 5–14 | 53 | 24 | 23 (96) | 29 | 2 (7) |
| Sex | |||||
| Female | 47 | 20 | 19 (95) | 27 | 3 (11) |
| Male | 31 | 15 | 15 (100) | 16 | 3 (19) |
| Population group | |||||
| CBO | 22 | 12 | 11 (92) | 10 | 4 (40) |
| RFN | 22 | 20 | 20 (100) | 2 | 1 (50) |
| FB | 34 | 3 | 3 (100) | 31 | 1 (3) |
| Method of detection | |||||
| Symptoms | 39 | 4 | 4 (100) | 35 | 6 (17) |
| Contact tracing | 32 | 31 | 30 (97) | 1 | 0 (0) |
| Other screening+ | 7 | 0 | 0 (0) | 7 | 0 (0) |
Data are presented as n or n (%), unless otherwise stated. CE: conventional epidemiology; CBO: Canadian-born “other”; RFN: registered First Nations; FB: foreign born. #: total culture-positive cases; ¶: odds ratios of paediatric TB cases with source cases identified by ME only versus paediatric TB cases without source cases identified by ME or CE for various characteristics are as follows: age 5–14 years (0.1852 (95% CI 0.03–1.17)); male sex (1.8462 (95% CI 0.33–10.49)); FB population group (0.0467 (95% CI 0.005–0.47)); detection by symptoms (3.7458 (95% CI 0.19–73.45)). +: other screening includes immigration medical evaluations, international adoptee screening and incidental findings.
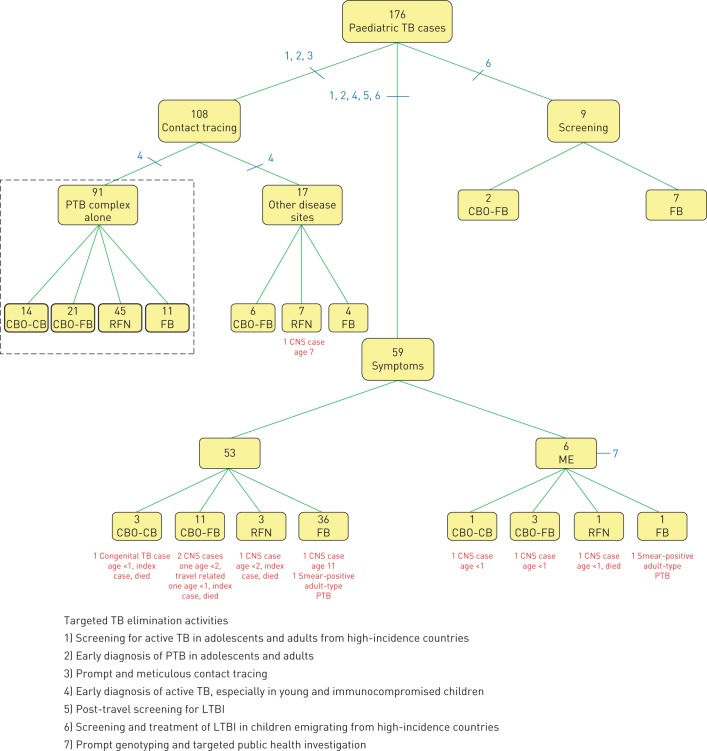
All 176 paediatric TB cases are described in figure 3 according to method of detection. Of the 108 cases identified by contact tracing, 91 (84.3%) had a primary pulmonary TB (PTB) complex alone and might arguably be said to have LTBI. Compared to children whose method of detection was contact tracing or screening, children whose method of detection was symptoms were more likely to have a major public health or clinical consequence (ten cases (16.9%) comprising two children with smear-positive adult-type PTB (both age 14 years), four CNS-TB survivors (three aged <2 years and one aged 11 years) and all of the TB deaths (three CNS-TB cases and one congenital TB case, all aged <2 years) versus one case (0.9%), a CNS-TB survivor aged 7 years). Among Canadian-born children <2 years of age, those whose method of detection was symptoms (n=13) were much more likely than those whose method of detection was contact tracing or screening (n=20) to die or be a CNS-TB survivor (7 cases (53.8%) versus zero cases (0%)). As noted, two of the CNS-TB survivors and all four of the TB deaths were among the ten symptomatic children whose source cases were discovered through either reverse contact tracing or ME. Only three CNS-TB cases and no TB deaths occurred among the other 166 cases in the cohort.
FIGURE 3.
Paediatric tuberculosis (TB) in Alberta by method of detection and strategically placed TB elimination activities. The dotted lines highlight contacts with a single International Classification of Diseases Ninth Revision (ICD-9) code of 010.0 (primary tuberculous complex). Of the CBO-FB children without a source case in Alberta (n=9), five could, on the basis of their travel history or their unique Mycobacterium tuberculosis DNA fingerprints, be reasonably said to have acquired their infection overseas. PTB: pulmonary TB; CBO-CB: Canadian-born “other” with Canadian-born parents; CBO-FB: Canadian-born “other” with one or more foreign-born parents; RFN: registered First Nations; FB: foreign born; ME: molecular epidemiology; CNS: central nervous system; LTBI: latent TB infection.
Discussion
Over the 25 year period from 1990 to 2014 there was substantial migration into Alberta. This migration, together with a known decline in the incidence of TB in Canadian-born adults [24], had a major impact on paediatric TB in the province. From the first to the last 5 years of the study the proportion of cases who were either foreign-born or CBO-FB children increased from 32.1% to 89.5%. In contrast, the number of cases in the CBO-CB group fell to zero over the first 15 years of the study if they were not RFN and from 27 to four cases from the first to the last 5 years if they were RFN. In a similar fashion to Montreal (Quebec) and Toronto (Ontario) [12, 14], almost all of the paediatric TB in the province came to be imported, either directly in the child or indirectly through the child's parents or extended family. Source case identification by CE was very good and 97.5% of paediatric case/source case pairs of M. tuberculosis isolates had identical DNA fingerprints. Children whose in-Alberta source case was diagnosed through reverse contact tracing or ME (n=10) had tragic outcomes and six (60%) had either CNS-TB or congenital TB and of these four died.
While the age- and sex-adjusted incidence of paediatric TB over the study period was 16- and 18-times higher in RFN and foreign-born children, respectively, compared to Canadian-born “other” children, it decreased in the former and increased in the latter over time. RFN outbreaks in the first ten but not the last 15 years of the study and the introduction in 1999 of a “virtual clinic” to serve rural Alberta are possible explanations for the former [17, 24–26]. By the end of the study period, rates in the RFN group were comparable to those recently reported in indigenous children in Australia (1.70 per 100 000 population) [7]. Increased rates in foreign-born children were assumed to reflect increased numbers of recently arrived children. Recent arrivals are known to be at higher risk of disease if infected [7, 27]. Unsurprisingly, given the well-known age-related risk stratification of paediatric TB and reports from the USA [3, 9], the incidence of TB was highest in the 0–4 years age group in all population groups [28–31]. However, it is of note that the likelihood of TB before 5 years of age was 3.0-times and 5.6-times higher in CBO-FB children than in RFN or foreign-born children, respectively. That 20.9% of the CBO-FB cases had no source case in Alberta by CE or ME suggested undocumented exposure in connection with travel or visits to the home by friends or family from abroad [32, 33].
A positive culture for M. tuberculosis was associated with older age, an extrapulmonary site of disease and symptoms as a method of detection. Drug resistance was uncommon despite 64.1% of culture-positive cases being foreign-born or from the CBO-FB group. While high rates of MDR-TB have been reported in children in other contexts and recently arrived immigrants to Alberta are more likely to have MDR-TB than historical arrivals [34], no MDR-TB was reported in this cohort [3, 35]. The high concordance of DNA fingerprints of M. tuberculosis isolates in paediatric case/source case pairs (97.1%) is reassuring given that when a child is culture-negative one must base treatment on the drug susceptibility test results in the putative source case. Among culture-positive cases whose in-Alberta source was discovered through ME alone, rapid genotyping methods may have accelerated the time to linkage [12]. The source cases of six of the seven children with very unsatisfactory outcomes (85.7%) were only discovered through reverse contact tracing or ME. When source case diagnosis is delayed or contact tracing is incomplete, paediatric TB morbidity and mortality are indeed high, a lesson that resource-rich countries must take to resource-poor countries.
The strengths of this study include its complete, programme-wide nature, its spatial and temporal breadth and it's use of WHO-defined paediatric age groups. Earlier studies were limited in these regards and, by including adolescents aged 15–17 years who were much more likely to be culture-positive [10–14], were potentially biased with respect to their ME analyses. Weaknesses include the inability to calculate TB rates in Canadian-born children by parental nativity and, given the retrospective nature of the study, to further refine the case definition. In the only study of its kind, Pang et al. [8] found that the rate of TB in US-born children of foreign-born parents aged 0–4 years was six-times higher than the rate in US-born children of US-born parents aged 0–4 years. Although we made certain that culture-negative pulmonary cases had chest radiographic abnormalities consistent with TB, we could not further sort intrathoracic cases according to the revised classification of such cases for diagnostic evaluation studies [36]. The notification of contacts with “primary TB complex”, most of whom are asymptomatic, is considered acceptable in resource-rich settings that are moving towards TB elimination, although data from the pre-chemotherapy era and outcomes after monotherapy suggest that the natural history of disease in these children, even those who may be culture-positive, is benign [28, 29].
For high-income, low-incidence countries to achieve the elimination of paediatric TB they must develop strategies that relate to the immigration medical examination, such as screening for LTBI in children emigrating from high-incidence countries [37], as well as to local programming (see figure 3). However, given our global interdependence, more must ultimately be done to help low and middle income countries rid themselves of this scourge [38].
Acknowledgement
The authors thank the staff of the Public Health TB clinics in Alberta, the Provincial Laboratory for Public Health and the TB Program Evaluation and Research Unit of the University of Alberta for their generous support of this project.
Footnotes
Conflict of interest: R. Long reports receiving grants from the Canadian Institutes of Health Research (CIHR) during the conduct of the study.
Support statement: This study was supported by a grant from the Canadian Institutes of Health Research (CIHR) (number 125961). D. Langlois-Klassen was a recipient of a CIHR Postdoctoral Fellowship. J. Bown was a Masters student at the London School of Hygiene and Tropical Medicine. Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Lobato MN, Mohle-Boetani JC, Royce SE. Missed opportunities for preventing tuberculosis among children younger than five years of age. Pedatrics 2000; 106: e75. [DOI] [PubMed] [Google Scholar]
- 2.Nelson LJ, Schneider E, Wells CD, et al. Epidemiology of childhood tuberculosis in the United States, 1993-2001: the need for continued vigilance. Pediatrics 2004; 114: 333–341. [DOI] [PubMed] [Google Scholar]
- 3.del Rosal T, Baquero-Artigao F, Garcia-Miguel MJ, et al. Impact of immigration on pulmonary tuberculosis in Spanish children: a three decade review. Pediatr Infect Dis J 2010; 29: 648–651. [DOI] [PubMed] [Google Scholar]
- 4.Hatleberg CI, Prahl JB, Rasmussen JN, et al. A review of paediatric tuberculosis in Denmark: a 10-year trend, 2000-2009. Eur Respir J 2014; 43: 863–871. [DOI] [PubMed] [Google Scholar]
- 5.Marais BJ, Tadolini M, Zignol M, et al. Paediatric tuberculosis in Europe: lessons from Denmark and inclusive strategies to consider. Eur Respir J 2014; 43: 678–684. [DOI] [PubMed] [Google Scholar]
- 6.Bennet R, Eriksson M. Paediatric tuberculosis cases increased in Stockholm from 1971 to 2015 following the rising number of children with immigrant backgrounds. Acta Paediatr 2016; 105: 1480–1486. [DOI] [PubMed] [Google Scholar]
- 7.Teo SS, Tay EL, Douglas P, et al. The epidemiology of tuberculosis in children in Australia, 2003-2012. Med J Aust 2015; 203: 440. [DOI] [PubMed] [Google Scholar]
- 8.Pang J, Teeter LD, Katz DJ, et al. Epidemiology of tuberculosis in young children in the United States. Pediatrics. 2014; 133: e494–e504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Epidemiology of pediatric tuberculosis in the United States, 1993-2015. Atlanta, Centers for Disease Control and Prevention (CDC), 2016. www.cdc.gov/tb/publications/slidesets/pediatrictb/ Date last accessed: August 10, 2017. Date last updated: December 08, 2016. [Google Scholar]
- 10.Sun SJ, Bennett DE, Flood F, et al. Identifying the sources of tuberculosis in young children: a multistate investigation. Emerg Infect Dis 2002; 8: 1216–1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wootton SH, Gonzalez BE, Pawlak R, et al. Epidemiology of pediatric tuberculosis using traditional and molecular techniques: Houston, Texas. Pediatrics 2005; 116: 1141–1147. [DOI] [PubMed] [Google Scholar]
- 12.Yeo IKT, Tannenbaum T, Scott AN, et al. Contact investigation and genotyping to identify tuberculosis transmission to children. Pediatr Infect Dis J 2006; 25: 1037–1043. [DOI] [PubMed] [Google Scholar]
- 13.Marquez L, Feske ML, Teeter LD, et al. Pediatric tuberculosis: the litmus test for tuberculosis control. Pediatr Infect Dis J 2012; 31: 1144–1147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rayment JH, Guthrie JL, Lam K, et al. Culture-positive pediatric tuberculosis in Toronto, Ontario. Sources of infection and relationship of birthplace and mycobacterial lineage to phenotype. Pediatr Infect Dis J 2016; 35: 13–18. [DOI] [PubMed] [Google Scholar]
- 15.Guidance for national tuberculosis programmes on the management of tuberculosis in children. 2nd edn Geneva, World Health Organization (WHO), 2014. www.who.int/tb/publications/childtb_guidelines/en/ Date last accessed: August 10, 2017. [PubMed] [Google Scholar]
- 16.Yip D, Bhargava R, Yao Y, et al. Pediatric tuberculosis in Alberta: epidemiology and case characteristics (1990-2004). Can J Public Health 2007; 98: 276–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Long R, Heffernan C, Gao Z, et al. Do “virtual” and “outpatient” public health tuberculosis clinics perform equally well? A program-wide evaluation in Alberta, Canada. PLoS One 2015; 10: e0144784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Public Health Agency of Canada, Canadian Lung Association/Canadian Thoracic Society. Canadian tuberculosis standards 6th edn www.collectionscanada.gc.ca/webarchives/20071223172131/http://www.phac-aspc.gc.ca///tbpc-latb/pubs/tbstand07-eng.php Date last accessed: August 10, 2017. Date last updated: December 13, 2007. [Google Scholar]
- 19.van Embden JDA, Cave MD, Crawford JT, et al. Strain identification of Mycobacterium tuberculosis by DNA fingerprinting: recommendations for a standardized methodology. J Clin Microbiol 1993; 31: 406–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dale JW, Brittain D, Cataldi AA, et al. Spacer oligonucleotide typing of bacteria of the Mycobacterium tuberculosis complex: recommendations for standardised nomenclature. Int J Tuberc Lung Dis 2001; 5: 216–219. [PubMed] [Google Scholar]
- 21.Veen J. Microepidemics of tuberculosis: the stone in the pond principle. Tuber Lung Dis 1992; 73: 73–76. [DOI] [PubMed] [Google Scholar]
- 22.Long R, Niruban S, Heffernan C, et al. A 10-year population-based study of ‘opt-out’ HIV testing of tuberculosis patients in Alberta, Canada: national implications. PLoS One 2014; 9: e98933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Long R, Boffa J. Why internationally adopted children should be screened for tuberculosis. CMAJ 2007; 177: 172–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jensen M, Lau A, Langlois-Klassen D, et al. A population-based study of tuberculosis epidemiology and innovative service delivery in Canada. Int J Tuberc Lung Dis 2012; 16: 43–49. [DOI] [PubMed] [Google Scholar]
- 25.Long R, Sutherland K, Kunimoto D, et al. The epidemiology of tuberculosis among foreign-born persons in Alberta, Canada, 1989-1998: identification of high risk groups. Int J Tuberc Lung Dis 2002; 6: 615–621. [PubMed] [Google Scholar]
- 26.Long R, Whittaker D, Russell K, et al. Pediatric tuberculosis in Alberta First Nations (1991-2000). Outbreaks and the protective effect of bacille Calmette-Guérin (BCG) vaccine. Can J Public Health 2004; 95: 249–255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Langlois-Klassen D, Wooldrage K, Manfreda J, et al. Piecing the puzzle together: foreign-born tuberculosis in an immigrant-receiving country. Eur Respir J 2011; 38: 895–902. [DOI] [PubMed] [Google Scholar]
- 28.Marais BJ, Gie RR, Schaaf HS, et al. The natural history of childhood intra-thoracic tuberculosis: a critical review of literature from the pre-chemotherapy era. Int J Tuberc Lung Dis 2004; 8: 392–402. [PubMed] [Google Scholar]
- 29.Marais BJ, Gie RP, Schaaf HS, et al. Childhood pulmonary tuberculosis: old wisdom and new challenges. Am J Respir Crit Care Med 2006; 173: 1078–1090. [DOI] [PubMed] [Google Scholar]
- 30.Newton SM, Brent AJ, Anderson S, et al. Pediatric tuberculosis. Lancet Infect Dis 2008; 8: 498–510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Perez-Velez CM, Marais BJ. Current concepts: tuberculosis in children. N Engl J Med 2012; 367: 348–361. [DOI] [PubMed] [Google Scholar]
- 32.Cobelens FG, van Deutekom H, Draayer-Jansen IW, et al. Risk of infection with Mycobacterium tuberculosis in travellers to areas of high tuberculosis endemicity. Lancet 2000; 356: 461–465. [DOI] [PubMed] [Google Scholar]
- 33.Kik SV, Mensen M, Beltman M, et al. Risk of travelling to the country of origin for tuberculosis among immigrants living in a low-incidence country. Int J Tuberc Lung Dis 2011; 15: 38–43. [PubMed] [Google Scholar]
- 34.Long R, Langlois-Klassen D. Increase in multidrug-resistant tuberculosis (MDR-TB) in Alberta among foreign-born persons: implications for tuberculosis management. Can J Public Health 2013; 104: e22–e27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Donnan EJ, Coulter C, Simpson G, et al. Paediatric tuberculosis in Queensland, Australia: overrepresentation of cross-border and indigenous children. Int J Tuberc Lung Dis 2017; 21: 263–269. [DOI] [PubMed] [Google Scholar]
- 36.Graham SM, Cuevas LE, Jean-Philippe P, et al. Clinical case definitions for classification of intrathoracic tuberculosis in children: an update. Clin Infect Dis 2015; 61: Suppl. 3, S179–S187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.CDC immigration requirements: technical instructions for tuberculosis screening and treatment. Atlanta, Centers for Disease Control and Prevention (CDC), 2009. www.cdc.gov/immigrantrefugeehealth/pdf/tuberculosis-ti-2009.pdf Date last accessed: August 11, 2017. Date last updated: October 01, 2009. [Google Scholar]
- 38.Schwartzman K, Oxlade O, Barr RG, et al. Domestic returns from investment in the control of tuberculosis in other countries. N Engl J Med 2005; 353: 1008–1020. [DOI] [PubMed] [Google Scholar]