Abstract
The Ts65Dn mouse model of Down syndrome recapitulates the hallmark areas of dysfunction that characterize the human disorder, including impaired performance in tasks designed to tap hippocampus-dependent learning and memory. Unfortunately, performance in the water maze tasks most commonly used for this purpose can be affected by behavioral and/or physiological abnormalities characteristic of Ts65Dn mice (e.g., thigmotaxis, susceptibility to hypothermia, stress reactivity), which complicates interpretation of impaired performance. The current study assessed hippocampal function in Ts65Dn mice using the social transmission of food preference (STFP) paradigm, which does not entail water escape or aversive reinforcement, and thus avoids these interpretive confounds. We tested Ts65Dn mice and disomic controls on this task using 1- and 7-day retention intervals. The Ts65Dn mice exhibited normal learning and memory following the 1-day retention interval, but rapid forgetting of the socially acquired information, evidenced by impaired performance following the 7-day retention interval. The STFP paradigm can be a valuable tool for studies using the Ts65Dn mouse model to evaluate potential therapies that may ameliorate hippocampal dysfunction and aging-related cognitive decline in Down syndrome.
Keywords: Down syndrome, Ts65Dn mice, hippocampus, social transmission of food preference
Introduction
Down syndrome (DS) affects roughly 1 in 700 live births in the United States (Parker et al., 2010), and is the most common genetic cause of intellectual disability. The extensively studied Ts65Dn mouse model of DS (Davisson et al., 1990) has been reported to recapitulate many of the hallmark areas of dysfunction that characterize the human disorder, including impairment of memory and attention, as well as atrophy of cholinergic neurons in early adulthood. Ts65Dn mice, like humans with DS, perform more poorly than controls on tests designed to tap hippocampus-dependent learning and memory (Galdzicki & Siarey, 2003; Nadel, 2003), including the Morris water maze, the radial arm water maze, novel object recognition, and context discrimination (Ash et al., 2014; Bimonte-Nelson et al., 2003; Hyde et al., 2001b; Holtzman et al., 1996; Reeves et al., 1995; Sago et al., 2000; Sansevero et al., 2016; Velazquez et al., 2013). Functional deficits in Ts65Dn mice coincide with an early onset degeneration of basal forebrain cholinergic neurons, which project to the hippocampus (Granholm et al., 2000; Holtzman et al., 1996).
Numerous studies have utilized Ts65Dn mice to test potential therapies for DS, and have relied on the Morris water maze or radial arm water maze to assess spatial learning and memory (De la Torre et al., 2014; Lockrow et al., 2011; Sansevero et al., 2016; Stagni et al., 2015; Stringer et al., 2015, 2017). However, certain behavioral and physiological abnormalities characteristic of Ts65Dn mice complicate the interpretation of their impaired performance in these tasks. For example, Ts65Dn mice exhibit thigmotaxis (swimming along the walls of the pool) when tested in the Morris water maze (Costa et al., 1999; Escorihuela et al., 1995). Another factor complicating interpretation of group differences in water mazes is the fact that Ts65Dn mice are more susceptible than controls to swimming-induced hypothermia (Stasko & Costa, 2004). Additionally, Ts65Dn mice can display motor dysfunction and hyperactivity (Costa et al., 1999; Coussons-Read & Crnic, 1996), which may affect performance in tasks such as the radial arm maze and novel object recognition. Finally, there is evidence that Ts65Dn mice exhibit heightened reactivity to a variety of stressors (Crnic & Pennington, 2000; Driscoll et al., 2004; Hyde et al., 2001a; Martínez-Cué et al., 2006; Turner et al., 2001), further complicating interpretation of impaired performance in water maze tasks, and more generally in tasks which use stressors to motivate learning. The current study was designed to identify a test of hippocampus-dependent memory that is free of these confounds. A test which has been shown to be sensitive to hippocampal dysfunction but does not entail water escape or aversive motivation is the social transmission of food preference (STFP) paradigm. This task has been effectively used by many groups to study the neuroanatomical, neurochemical and genetic substrates of memory (see Galef, 2012).
The STFP is a social learning paradigm, initially developed as a means of assessing intrinsically motivated learning — learning in which animals would spontaneously engage without the need for either appetitive or aversive reinforcement (Strupp and Levitsky, 1983; 1984). The development of this paradigm was based on anecdotal reports that Norway rats in the wild learn about the safety of novel foods through social interaction with other rats (Strupp and Levitsky, 1984). The general procedure consists of a three-stage process. First, a demonstrator rodent is given a distinctly scented food to eat. Second, the demonstrator interacts with a conspecific observer, during which time the observer can smell the scented food on the breath of the demonstrator, which is a critical component of the learning (Galef et al., 1988). Third, after a retention interval, the observer is presented with two novel scented foods, one of which had previously been consumed by the paired demonstrator. Prior experiments have provided evidence that both mice and rats show a strong preference for consuming the food that the paired demonstrator had previously consumed. Following elucidation of the key elements that underlie this social learning phenomenon in rats, the paradigm was later modified and streamlined for both rats (Galef, 2003) and mice (Wrenn, 2004). Recall of the information acquired in the STFP paradigm following lengthy retention intervals has been shown to be impaired by hippocampal damage (Bunsey & Eichenbaum, 1995; Winocur, 1990).
We tested Ts65Dn and disomic (2N) control mice in this STFP procedure, using a 1-day retention interval (Experiment 1) and a 7-day retention interval (Experiment 2). We hypothesized that Ts65Dn mice would exhibit impaired memory relative to 2N controls.
Methods
Subjects
Subjects in Experiment 1 were from our breeding colony at Cornell University (Ithaca, NY). Breeder pairs were female Ts65Dn and male C57Bl/6J Eicher × C3H/HeSnJ F1 mice, purchased from Jackson Laboratories (Bar Harbor, ME). All subjects were nulliparous female Ts65Dn or control (2N) mice. All demonstrators were 2N (n = 16); observers were Ts65Dn (n = 16) and 2N (n = 13). Mice were tested at approximately 12 months of age.
Subjects in Experiment 2 were purchased directly from Jackson Laboratories. All subjects were nulliparous female Ts65Dn or 2N mice. All demonstrators were 2N (n = 6); observers were Ts65Dn (n = 12) and 2N (n = 12). Mice were 3–4 months of age upon arrival and were tested at 6–7 months of age.
Mice were housed in microisolator cages, in a temperature- and humidity-controlled room (72°C, 45% humidity) on a 12:12 reverse light-dark cycle. Cages contained paper bedding, a plastic house, t-tube, ceramic cup, and plastic-gel bone. All mice were maintained on AIN-76A purified rodent diet (Dyets, Inc., Bethlehem, PA) and had ad libitum access to food and water.
Mice were group-housed by genotype until two weeks prior to testing. At that time, all subjects were housed singly and ration fed to keep them at roughly 90% of their ad libitum body weight. Subjects were weighed daily before and after testing, and were fed 20 minutes after being returned to their home cage. With this regimen, mice consumed their rations within ~1 hour, resulting in ~23-hour food restriction prior to testing, which ensured motivation to consume food during the test sessions.
Social Transmission of Food Preference (STFP)
Our STFP protocol was adapted from Wrenn (2004), which describes the basic protocol for STFP in mice, adapted from prior research with rats. Demonstrator feeding and STFP testing, respectively, were conducted in two separate rooms adjacent to the colony room, during the dark phase of the cycle, in red light. Test chambers were standard rat cages (9.5 in. × 18 in. × 8 in.) with opaque walls, empty except for two glass food jar assemblies, one placed at each end of the cage.
Half of the demonstrators were assigned to eat powdered chow adulterated with cocoa (2% w/w), and the other half were given powdered chow adulterated with cinnamon (1% w/w). These two chow mixtures have been used extensively in prior research using the STFP paradigm, and in general, are consumed in approximately equal amounts by naïve rodents (Strupp and Levitsky, 1983; 1984; Galef, 2003). In addition, a pilot study in our lab involving six demonstrators from Experiment 1 confirmed that these two diets were relatively equally preferred by this strain of mouse. When presented with the two adulterated diets simultaneously, these mice consumed roughly equal amounts of each (within 43% to 57%).
Beginning one week prior to the experiment, each demonstrator was placed for 1 hour per day into a test chamber containing two food jars, both filled with the diet assigned to that demonstrator mouse. Food jars were weighed at start and finish to determine amount consumed.
On test days 1–3, observers were habituated to the test chambers and plain (unadulterated) powdered chow. Each observer was transferred to a test chamber for 1 hour on each of these days. Both food jars contained unadulterated, powdered chow. Food jars were weighed at start and finish to determine amount consumed.
Test day 4 was a food neophobia test, to provide an assessment of olfactory acuity. Each observer was transferred to a test chamber for 1 hour. One food jar contained unadulterated, powdered chow and the other food jar contained powdered chow adulterated with ginger (2% w/w). Food jars were weighed at the start and end of the 1 hour period to determine the amount consumed. In this test, most rodents will exhibit a preference for the unadulterated chow, due to neophobia for novel foods; in this case, the diet adulterated with ginger. Thus, if a given rodent (or group) does not exhibit an avoidance of the ginger-scented chow, olfactory acuity may be questioned.
On test day 5 the food jars were removed from the test chambers. Each observer was placed into a chamber along with a demonstrator which had just previously consumed either the cocoa-scented or cinnamon-scented chow. Within each group (Ts65Dn and 2N), half of the mice were paired with a demonstrator who had previously consumed cocoa-scented chow, whereas the other half were paired with a demonstrator who had previously consumed cinnamon-scented chow. Each observer-demonstrator pair could freely interact for 30 minutes. A wide-angle, infrared video camera was used to record the interaction.
Test day 6 was the STFP test, and was conducted either 1 day after observer-demonstrator interaction (Experiment 1) or 7 days after the interaction (Experiment 2). Each observer was transferred to a test chamber for 1 hour. One food jar contained cocoa-scented chow and the other food jar contained cinnamon-scented chow. Food jars were weighed at start and finish to determine amount consumed.
Demonstrators were paired only with one observer per test day, but were re-used to interact with multiple observers. The pairings were counterbalanced across observer genotype. The spatial locations of the food jars during the food neophobia test and the STFP test were counterbalanced across genotype and food type. Demonstrators were required to consume a minimum of 0.2 g of food prior to interaction with observers. Likewise, observers were required to consume a minimum of 0.2 g of food during the food neophobia test and the STFP test to permit a reliable assessment of the food preference, and hence a valid assessment of the construct being assessed (neophobia, and learning/memory, respectively). All mice consumed this minimum amount of food in the allotted time.
All protocols were approved by the Institutional Animal Care and Use Committees of Cornell University and were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Statistical Analysis
For each experiment, body weight (g) was analyzed using an unpaired t-test. For the food neophobia test, the percent of the normal chow consumed ([grams of unadulterated chow consumed]/[grams of total chow consumed] × 100) was analyzed using an unpaired t-test. Group sizes for all t-tests were — Experiment 1: Ts65Dn = 16, 2N = 13; Experiment 2: Ts65Dn = 12, 2N = 12.
For the STFP data, the dependent measure was the percent cocoa-scented chow consumed ([grams of cocoa-scented chow consumed]/[grams of total chow consumed] × 100). Subgroups were characterized by genotype (Ts65Dn, 2N), retention interval (1-day, 7-day), and food consumed by the paired demonstrator (cocoa, cinnamon). For each genotype, at each delay, a significant social learning effect was indicated by a significant difference in the mean percent cocoa chow consumed between the observers paired with a cocoa demonstrator vs those paired with a cinnamon demonstrator. Due to the relatively small sample sizes within each of these subgroups (n = 6–8, see Figure 1), the data were analyzed using the nonparametric Wilcoxon Rank Sum Test. The alpha level for all analyses was set at 0.05. Statistical analysis was performed using the Statistical Analysis System (version 9.3; SAS Institute, Cary, NC).
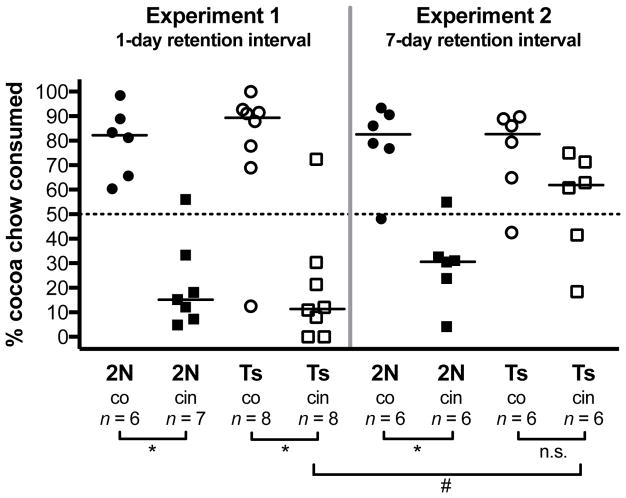
Figure 1. Percentage of cocoa-adulterated chow consumed during the STFP test, as a function of genotype, retention interval, and the diet consumed by the paired demonstrator (cocoa- or cinnamon-adulterated chow).
A significant social learning effect was observed for the 2N mice tested at a 1-day retention interval, the 2N mice tested at a 7-day retention interval, and the Ts65Dn mice tested at a 1-day retention interval (*p < 0.05). There was no significant social learning effect for the Ts65Dn mice tested at a 7-day retention interval (n.s.). Significant social learning was defined as a significant difference in the percent cocoa-adulterated chow consumed between the subgroup paired with a demonstrator fed cocoa chow versus the subgroup paired with a demonstrator who had consumed cinnamon chow. In addition, there was a significant effect of delay for the Ts65Dn mice paired with cinnamon demonstrators (#p < 0.05). Solid symbols = 2N mice, open symbols = Ts65Dn mice. Circles = mice paired with cocoa demonstrators, squares = mice paired with cinnamon demonstrators. Solid lines represent the median within each group.
Results
In both experiments, body weight (g) of the Ts65Dn mice was significantly less than their 2N counterparts, consistent with prior studies (Ash et al., 2014; Bianchi et al., 2010; Fuchs et al., 2012; Roper et al., 2006; Velazquez et al., 2013). For Experiment 1, the means (± SEM) were 27.32 ± 0.76 vs 23.73 ± 0.61 [t(27) = 3.73, p = 0.0009]. For Experiment 2, the means were 27.93 ± 0.76 vs 23.58 ± 0.99 [t(22) = 3.46, p = 0.002].
In Experiment 1, the food neophobia test did not reveal a significant difference in the percentage of unadulterated chow consumed by the 2N and Ts65Dn mice [t(27) = 1.173, p = 0.25]; both groups exhibited a preference for the unadulterated chow (i.e., an avoidance of the ginger-scented chow): (2N: 75.93 ± 3.1; Ts65Dn: 81.11 ± 3.1). Similarly, in Experiment 2 there was no significant difference between the two genotypes in terms of the percentage of unadulterated chow consumed [t(22) = 0.8798, p = 0.39]; both groups exhibited a preference for the unadulterated chow (2N: 78.83 ± 3.7; Ts65Dn: 72.01 ± 6.8).
Figure 1 depicts the percent cocoa-scented chow consumed by each observer, and indicates the median within each group. The Wilcoxon Rank Sum Test revealed a significant social learning effect for the 2N mice tested at a 1-day retention interval (p = 0.003), the 2N mice tested at a 7-day retention interval (p = 0.008), and the Ts65Dn mice tested at a 1-day retention interval (p = 0.004). In these groups, observer mice paired with cocoa demonstrators exhibited a preference for the cocoa-scented chow, whereas the observer mice paired with cinnamon demonstrators exhibited a preference for the cinnamon-scented chow. However, for the Ts65Dn mice tested at a 7-day retention interval, the social learning effect was not significant (p > 0.05). Following this longer retention interval, the Ts65Dn observers paired with cocoa demonstrators consumed mostly cocoa-scented chow, and even those paired with cinnamon demonstrators exhibited a slight preference for the cocoa-scented chow. Consequently, for the Ts65Dn mice cued to cinnamon, there was a significant effect of delay (p = 0.03), reflecting the fact that a preference for the cinnamon-scented chow was evident at the 1-day interval, but not at the 7-day interval. In contrast, there was no significant effect of delay for 2N mice cued to cocoa (p = 0.99), 2N mice cued to cinnamon (p = 0.43), or Ts65Dn mice cued to cocoa (p = 0.33), as strong social learning was evident for these subgroups at both retention intervals.
Discussion
The Ts65Dn mouse model has been extensively used by various labs to test potential therapies to lessen the cognitive dysfunction associated with DS (Begenisic et al., 2015; Colas et al., 2017; De la Torre et al., 2014; Lockrow et al., 2011; Sansevero et al., 2016; Stagni et al., 2015; Stringer et al., 2015). Our group, for example, has shown that supplementing the maternal diet with additional choline can ameliorate spatial learning and attention deficits in adult Ts65Dn offspring (Ash et al., 2014; Moon et al., 2010; Powers et al., 2017; Velazquez et al., 2013). However, behavioral and physiological abnormalities characteristic of Ts65Dn mice complicate interpretation of the impaired performance of these mice in tasks which entail water escape or other stressors. Therefore, the current study assessed hippocampus-dependent memory in Ts65Dn mice using the STFP paradigm, which does not entail water escape or aversive motivation. The results demonstrate that Ts65Dn mice exhibit normal social learning and memory following a 1-day retention interval, but rapid forgetting of the socially acquired information, as evidenced by performance after the 7-day retention interval.
When tested following a 1-day retention interval, Ts65Dn mice performed indistinguishably from 2N controls. Social learning was evident in both genotypes, i.e. observers showed a strong preference for the scented chow that their paired demonstrators had previously consumed. Those observers paired with demonstrators who had consumed cinnamon-scented chow exhibited a preference for the cinnamon diet, whereas those paired with cocoa demonstrators preferred the cocoa-scented chow. In contrast, when tested following a 7-day retention interval, the 2N mice showed a strong social learning effect but the Ts65Dn mice did not, indicative of rapid forgetting. As can be seen on the right-hand side of Figure 1, the trisomic mice demonstrated an overall preference for the cocoa-scented chow, regardless of demonstrator diet. The Ts65Dn mice paired with demonstrators who had consumed cocoa-scented chow, like their 2N counterparts, consumed a high percentage of cocoa-scented chow. However, the Ts65Dn mice paired with demonstrators who had previously consumed cinnamon-scented chow also showed a slight preference for the cocoa-scented chow (median = 59.2%), and a majority of these subjects exhibited a cocoa preference.
The pattern of results suggests that Ts65Dn (and possibly 2N) mice have a natural preference for the cocoa-scented chow, but that this preference is not seen if the animal has access to the memory of the scent information acquired from the demonstrator. The STFP paradigm is based on the idea that, when confronted with two novel foods, rodents will use available information about their relative safety to guide food choice. Thus, if the mouse remembers that the scent on the demonstrator’s breath matches one of the two novel foods, then it will preferentially eat that diet; but if the memory of that social interaction has eroded, then the animal will sample both diets, and exhibit a preference for whichever food is more palatable, which in this case, appears to be the cocoa-adulterated chow. It should be noted that although it is theoretically possible that, if the memory has faded, the mouse will eat neither of the two novel foods (due to neophobia), our prior data (and those of others) indicate that the desire to eat overrides the neophobia for scented chow in rodents under mild food restriction (as used here), and they do sample unfamiliar diets.
Although we had conducted a pilot study to verify that the cocoa- and cinnamon-adulterated diets were equally preferred by this strain, it is notable that we had used only six 2N mice for this pilot test. There may be genotype differences, or the small sample size may have precluded the detection of a preference in these 2N mice.
Scrutiny of individual performance supports our interpretation of an inherent preference for the cocoa chow. As can be seen in Figure 1, all groups paired with cocoa demonstrators showed a very strong preference for the cocoa-scented chow, whereas those paired with cinnamon demonstrators ranged more widely in the percentage consumed. Even at the 1-day retention interval, where both genotypes exhibited highly significant effects of the demonstrator’s diet, there were individuals paired with cinnamon demonstrators who exhibited a preference for the cocoa chow. Thus, the most parsimonious explanation is that Ts65Dn mice had experienced forgetting of the socially-acquired information following the 7-day retention interval, which manifested as an unmasking of a cocoa preference.
We can exclude alternative explanations for the impaired performance of the trisomic mice at the 7-day retention test. Importantly, the fact that the Ts65Dn mice exhibited strong social learning at the 1-day test (equal in magnitude to that of the 2N mice) demonstrates that the impaired performance at the 7-day test could not be attributed to an impaired ability to acquire or demonstrate this type of social learning. The strong social learning seen at the 1-day retention test also allows us to exclude impaired olfactory acuity as the basis of the impaired performance at the 7-day test, consistent with the results of the neophobia test.
In summary, Ts65Dn mice exhibited normal social learning but rapid forgetting on the STFP test, indicative of impaired hippocampal function. These data demonstrate that the STFP paradigm can be used as a test of hippocampus-dependent memory in Ts65Dn mice. This task is ethologically based, and has the advantage that it does not entail water escape or aversive motivation, which may differentially affect trisomic and 2N mice, and therefore complicate interpretation of altered performance in tasks which use these motivators. As such, it provides a useful tool to evaluate potential therapies that may lessen hippocampal dysfunction and aging-related cognitive decline in DS.
Acknowledgments
This work was supported by National Institute of Child Health and Human Development, grant number HD057564 (to BJS, EJM, SDG).
References
- Ash JA, Velazquez R, Kelley CM, Powers BE, Ginsberg SD, Mufson EJ, Strupp BJ. Perinatal choline supplementation improves spatial mapping and increases cholinergic basal forebrain cholinergic neuron number and size in aged Ts65Dn mice. Neurobiol Dis. 2014;70:32–42. doi: 10.1016/j.nbd.2014.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Begenisic T, Sansevero G, Baroncelli L, Cioni G, Sale A. Early environmental therapy rescues brain development in a mouse model of Down syndrome. Neurobiol Dis. 2015;82:409–419. doi: 10.1016/j.nbd.2015.07.014. [DOI] [PubMed] [Google Scholar]
- Bianchi P, Ciani E, Guidi S, Trazzi S, Felice D, Grossi G, Fernandez M, Giuliani A, Calzà L, Bartesaghi R. Early pharmacotherapy restores neurogenesis and cognitive performance in the Ts65Dn mouse model for Down syndrome. J Neurosci. 2010;30(26):8769–8779. doi: 10.1523/JNEUROSCI.0534-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bimonte-Nelson HA, Hunter CL, Nelson ME, Granholm AC. Frontal cortex BDNF levels correlate with working memory in an animal model of Down syndrome. Behav Brain Res. 2003;139(1–2):47–57. doi: 10.1016/s0166-4328(02)00082-7. [DOI] [PubMed] [Google Scholar]
- Bunsey M, Eichenbaum H. Selective damage to the hippocampal region blocks long-term retention of a natural and nonspatial stimulus-stimulus association. Hippocampus. 1995;5(6):546–556. doi: 10.1002/hipo.450050606. [DOI] [PubMed] [Google Scholar]
- Colas D, Chuluun B, Garner CC, Heller HC. Short-term treatment with flumazenil restores long-term object memory in a mouse model of Down syndrome. Neurobiol Learn Mem. 2017;140:11–16. doi: 10.1016/j.nlm.2017.02.006. [DOI] [PubMed] [Google Scholar]
- Costa AC, Walsh K, Davisson MT. Motor dysfunction in a mouse model for Down syndrome. Physiol Behav. 1999;68(1–2):211–220. doi: 10.1016/s0031-9384(99)00178-x. [DOI] [PubMed] [Google Scholar]
- Coussons-Read ME, Crnic LS. Behavioral assessment of the Ts65Dn mouse, a model for Down syndrome: altered behavior in the elevated plus maze and open field. Behav Genet. 1996;26(1):7–13. doi: 10.1007/BF02361154. [DOI] [PubMed] [Google Scholar]
- Crnic LS, Pennington BF. Down syndrome: Neuropsychology and animal models. Progress in Infancy Research. 2000;1:69–111. [Google Scholar]
- Davisson MT, Schmidt C, Akeson EC. Segmental trisomy of murine chromosome 16: a new model system for studying Down syndrome. Prog Clin Biol Res. 1990;360:263–280. [PubMed] [Google Scholar]
- De la Torre R1, De Sola S, Pons M, Duchon A, de Lagran MM, Farré M, Fitó M, Benejam B, Langohr K, Rodriguez J, Pujadas M, Bizot JC, Cuenca A, Janel N, Catuara S, Covas MI, Blehaut H, Herault Y, Delabar JM, Dierssen M. Epigallocatechin-3-gallate, a DYRK1A inhibitor, rescues cognitive deficits in Down syndrome mouse models and in humans. Mol Nutr Food Res. 2014;58(2):278–288. doi: 10.1002/mnfr.201300325. [DOI] [PubMed] [Google Scholar]
- Driscoll LL, Carroll JC, Moon J, Crnic LS, Levitsky DA, Strupp BJ. Impaired sustained attention and error-induced stereotypy in the aged Ts65Dn mouse: a mouse model of Down syndrome and Alzheimer’s disease. Behav Neurosci. 2004;118(6):1196–1205. doi: 10.1037/0735-7044.118.6.1196. [DOI] [PubMed] [Google Scholar]
- Escorihuela RM, Fernández-Teruel A, Vallina IF, Baamonde C, Lumbreras MA, Dierssen M, Tobeña A, Flórez J. A behavioral assessment of Ts65Dn mice: a putative Down syndrome model. Neurosci Lett. 1995;199(2):143–146. doi: 10.1016/0304-3940(95)12052-6. [DOI] [PubMed] [Google Scholar]
- Fuchs C, Ciani E, Guidi S, Trazzi S, Bartesaghi R. Early-occurring proliferation defects in peripheral tissues of the Ts65Dn mouse model of Down syndrome are associated with patched1 over expression. Lab Invest. 2012;92(11):1648–1660. doi: 10.1038/labinvest.2012.117. [DOI] [PubMed] [Google Scholar]
- Galdzicki Z, Siarey RJ. Understanding mental retardation in Down’s syndrome using trisomy 16 mouse models. Genes Brain Behav. 2003;2(3):167–178. doi: 10.1034/j.1601-183x.2003.00024.x. [DOI] [PubMed] [Google Scholar]
- Galef BG. Social learning of food preferences in rodents: rapid appetitive learning. Curr Protoc Neurosci. 2003;Chapter 8(Unit 8.5D) doi: 10.1002/0471142301.ns0805ds21. [DOI] [PubMed] [Google Scholar]
- Galef BG. A case study in behavioral analysis, synthesis and attention to detail: social learning of food preferences. Behav Brain Res. 2012;231(2):266–271. doi: 10.1016/j.bbr.2011.07.021. [DOI] [PubMed] [Google Scholar]
- Galef BG, Mason JR, Preti G, Bean NJ. Carbon disulfide: a semiochemical mediating socially-induced diet choice in rats. Physiol Behav. 1988;42(2):119–124. doi: 10.1016/0031-9384(88)90285-5. [DOI] [PubMed] [Google Scholar]
- Granholm AC, Sanders LA, Crnic LS. Loss of cholinergic phenotype in basal forebrain coincides with cognitive decline in a mouse model of Down’s syndrome. Exp Neurol. 2000;161:647–663. doi: 10.1006/exnr.1999.7289. [DOI] [PubMed] [Google Scholar]
- Holtzman DM, Santucci D, Kilbridge J, Chua-Couzens J, Fontana DJ, Daniels SE, Johnson RM, Chen K, Sun Y, Carlson E, Alleva E, Epstein CJ, Mobley WC. Developmental abnormalities and age-related neurodegeneration in a mouse model of Down syndrome. Proc Natl Acad Sci USA. 1996;93:13333–13338. doi: 10.1073/pnas.93.23.13333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyde LA, Crnic LS, Pollock A, Bickford PC. Motor learning in Ts65Dn mice, a model for Down syndrome. Dev Psychobiol. 2001a;38(1):33–45. doi: 10.1002/1098-2302(2001)38:1<33::aid-dev3>3.0.co;2-0. [DOI] [PubMed] [Google Scholar]
- Hyde LA, Frisone DF, Crnic LS. Ts65Dn mice, a model for Down syndrome, have deficits in context discrimination learning suggesting impaired hippocampal function. Behav Brain Res. 2001b;118:53–60. doi: 10.1016/s0166-4328(00)00313-2. [DOI] [PubMed] [Google Scholar]
- Lockrow J, Boger H, Gerhardt G, Aston-Jones G, Bachman D, Granholm AC. A noradrenergic lesion exacerbates neurodegeneration in a Down syndrome mouse model. J Alzheimers Dis. 2011;23(3):471–489. doi: 10.3233/JAD-2010-101218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez-Cué C, Rueda N, García E, Flórez J. Anxiety and panic responses to a predator in male and female Ts65Dn mice, a model for Down syndrome. Genes Brain Behav. 2006;5(5):413–22. doi: 10.1111/j.1601-183X.2005.00175.x. [DOI] [PubMed] [Google Scholar]
- Moon J, Chen M, Gandhy SU, Strawderman M, Levitsky DA, Maclean KN, Strupp BJ. Perinatal choline supplementation improves cognitive functioning and emotion regulation in the Ts65Dn mouse model of Down syndrome. Behav Neurosci. 2010;124(3):346–361. doi: 10.1037/a0019590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nadel L. Down’s syndrome: a genetic disorder in biobehavioral perspective. Genes Brain Behav. 2003;2(3):156–166. doi: 10.1034/j.1601-183x.2003.00026.x. [DOI] [PubMed] [Google Scholar]
- Parker SE, Mai CT, Canfield MA, Rickard R, Wang Y, Meyer RE, Anderson P, Mason CA, Collins JS, Kirby RS, Correa A National Birth Defects Prevention Network. Updated National Birth Prevalence estimates for selected birth defects in the United States, 2004–2006. Birth Defects Res A Clin Mol Teratol. 2010;88(12):1008–1016. doi: 10.1002/bdra.20735. [DOI] [PubMed] [Google Scholar]
- Powers BE, Kelley CM, Velazquez R, Ash JA, Strawderman MS, Alldred MJ, Ginsberg SD, Mufson EJ, Strupp BJ. Maternal choline supplementation in a mouse model of Down syndrome: Effects on attention and nucleus basalis/substantia innominata neuron morphology in adult offspring. Neuroscience. 2017;340:501–514. doi: 10.1016/j.neuroscience.2016.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves RH, Irving NG, Moran TH, Wohn A, Kitt C, Sisodia SS, Schmidt C, Bronson RT, Davisson MT. A mouse model for Down syndrome exhibits learning and behaviour deficits. Nat Genet. 1995;11:177–184. doi: 10.1038/ng1095-177. [DOI] [PubMed] [Google Scholar]
- Roper RJ, St John HK, Philip J, Lawler A, Reeves RH. Perinatal loss of Ts65Dn Down syndrome mice. Genetics. 2006;172(1):437–443. doi: 10.1534/genetics.105.050898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sago H, Carlson EJ, Smith DJ, Rubin EM, Crnic LS, Huang TT, Epstein CJ. Genetic dissection of region associated with behavioral abnormalities in mouse models for Down syndrome. Pediatr Res. 2000;48(5):606–613. doi: 10.1203/00006450-200011000-00009. [DOI] [PubMed] [Google Scholar]
- Sansevero G, Begenisic T, Mainardi M, Sale A. Experience-dependent reduction of soluble β-amyloid oligomers and rescue of cognitive abilities in middle-age Ts65Dn mice, a model of Down syndrome. Exp Neurol. 2016;283(Pt A):49–56. doi: 10.1016/j.expneurol.2016.06.006. [DOI] [PubMed] [Google Scholar]
- Stagni F, Giacomini A, Guidi S, Ciani E, Ragazzi E, Filonzi M, De Iasio R, Rimondini R, Bartesaghi R. Long-term effects of neonatal treatment with fluoxetine on cognitive performance in Ts65Dn mice. Neurobiol Dis. 2015;74:204–218. doi: 10.1016/j.nbd.2014.12.005. [DOI] [PubMed] [Google Scholar]
- Stasko MR, Costa AC. Experimental parameters affecting the Morris water maze performance of a mouse model of Down syndrome. Behav Brain Res. 2004;154(1):1–17. doi: 10.1016/j.bbr.2004.01.012. [DOI] [PubMed] [Google Scholar]
- Stringer M, Abeysekera I, Dria KJ, Roper RJ, Goodlett CR. Low dose EGCG treatment beginning in adolescence does not improve cognitive impairment in a Down syndrome mouse model. Pharmacol Biochem Behav. 2015;138:70–79. doi: 10.1016/j.pbb.2015.09.002. [DOI] [PubMed] [Google Scholar]
- Stringer M, Abeysekera I, Thomas J, LaCombe J, Stancombe K, Stewart RJ, Dria KJ, Wallace JM, Goodlett CR, Roper RJ. Epigallocatechin-3-gallate (EGCG) consumption in the Ts65Dn model of Down syndrome fails to improve behavioral deficits and is detrimental to skeletal phenotypes. Physiol Behav. 2017;177:230–241. doi: 10.1016/j.physbeh.2017.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strupp BJ, Levitsky DA. Early brain insult and cognition: a comparison of malnutrition and hypothyroidism. Dev Psychobiol. 1983;16(6):535–549. doi: 10.1002/dev.420160609. [DOI] [PubMed] [Google Scholar]
- Strupp BJ, Levitsky DE. Social transmission of food preferences in adult hooded rats (Rattus norvegicus) J Comp Psychol. 1984;98:257–266. [Google Scholar]
- Turner CA, Presti MF, Newman HA, Bugenhagen P, Crnic L, Lewis MH. Spontaneous stereotypy in an animal model of Down syndrome: Ts65Dn mice. Behav Genet. 2001;31(4):393–400. doi: 10.1023/a:1012226603255. [DOI] [PubMed] [Google Scholar]
- Velazquez R, Ash JA, Powers BE, Kelley CM, Strawderman M, Luscher ZI, Ginsberg SD, Mufson EJ, Strupp BJ. Maternal choline supplementation improves spatial learning and adult hippocampal neurogenesis in the Ts65Dn mouse model of Down syndrome. Neurobiol Dis. 2013;58:92–101. doi: 10.1016/j.nbd.2013.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winocur G. Anterograde and retrograde amnesia in rats with dorsal hippocampal or dorsomedial thalamic lesions. Behav Brain Res. 1990;16:145–154. doi: 10.1016/0166-4328(90)90012-4. [DOI] [PubMed] [Google Scholar]
- Wrenn CC. Social transmission of food preference in mice. Curr Protoc Neurosci. 2004;Chapter 8(Unit 8.5G) doi: 10.1002/0471142301.ns0805gs28. [DOI] [PubMed] [Google Scholar]