Abstract
The mammalian gastrointestinal tract can harbor both beneficial commensal bacteria important for host health, but also pathogenic bacteria capable of intestinal damage. It is therefore important that the host immune system mount the appropriate immune response to these divergent groups of bacteria–promoting tolerance in response to commensal bacteria and sterilizing immunity in response to pathogenic bacteria. Failure to induce tolerance to commensal bacteria may underlie immune-mediated diseases such as human inflammatory bowel disease. At homeostasis, regulatory T (Treg) cells are a key component of the tolerogenic response by adaptive immunity. This review examines the mechanisms by which intestinal bacteria influence colonic T cells and B cell IgA induction, with an emphasis on Treg cells and the role of antigen-specificity in these processes. In addition to discussing key primary literature, this review highlights current controversies and important future directions.
Introduction
The mammalian gastrointestinal tract is adapted to harbor trillions of bacteria1, many of which provide beneficial functions to the host, including protection from infection by pathogenic organisms2, 3 and nutrient metabolism4, 5. However, the close physical proximity of these bacteria to the host represents a unique challenge to the immune system, as it must be able to discriminate pathogenic bacteria from those normally resident in the gut. Inappropriate tolerance to pathogens may facilitate infection6, 7, whereas immune reactivity against harmless commensal microbiota is thought to underlie the pathogenesis of human inflammatory bowel disease8, 9 (IBD). Thus, the generation of appropriate immune responses to bacteria is crucial for intestinal health.
Although intestinal homeostasis requires multiple arms of the immune system10, here we will focus on the role of T cell responses to commensal bacteria. The absence of adaptive immune T cells leads to a failure of gut homeostasis in murine models, with bacterial translocation and colitis11. Similarly, intestinal pathology without obvious pathogenic infection also happens in humans with HIV that is associated with decreased CD4+ T cell counts12, 13. Adaptive immune cells are therefore not only required to target pathogenic bacteria14, 15, but also recognize and control normal intestinal bacteria during homeostasis.
However, the predominant T cell responses to intestinal bacteria during homeostasis are likely inhibitory responses to limit inflammation and immune-mediated gut pathology, rather than effector responses to eliminate bacteria. It is now generally accepted that tolerance to intestinal bacteria requires CD4+ regulatory T (Treg) cells, as first suggested in adoptive transfer experiments in rodents16. Treg cells are defined by the transcription factor Foxp3, which is required for Treg cell function and development17. In humans, patients with Treg cell deficiency (IPEX; immune dysregulation, polyendocrinopathy, enteropathy, X-linked) exhibit diarrhea and at times colitis amongst other autoimmune disease manifestations17. Thus, Treg cell-mediated tolerance to gut bacteria is crucial for maintenance of immune homeostasis and prevention of IBD.
We will review our current understanding of the reciprocal interactions of T cells and intestinal bacteria during homeostasis. Specifically, we will discuss the roles of intestinal bacteria in shaping tolerogenic Treg cell responses via non-antigen-specific factors as well as address how antigen-specificity appears to be important for intestinal Treg cell development and function. Although other immunoregulatory T cell subsets will not be addressed in this review, it is important to note that they make important contributions to gut tolerance18-21. We will also explore the function of Treg and effector T cells during homeostasis and their roles in modulating antigen-specific IgA induction. Since bacteria-dependent immunopathology in the small intestine is less common in humans, we will focus on Treg cell:bacteria interactions in the colon. Throughout, we will discuss discrepancies in conclusions formulated from primary data as well as identify unanswered questions in the field.
Developmental Origin of Colonic Treg Cells
It is now well-established that the colonic Treg cell population is affected by intestinal bacteria10. For example, germ-free mice show a several-fold reduction in the frequency of Treg cells compared with conventionally housed specific pathogen free (SPF) mice22, 23. This process does not require a complex microbiota, as introduction of individual bacterial isolates or defined consortia into germ-free mice is sufficient to induce colonic Treg cells22, 23.
One mechanism by which intestinal bacteria can influence Treg cell numbers is by inducing expansion of pre-existing thymic Treg (tTreg) cells. Classically, tTreg cells are generated in response to antigen recognition at an immature stage of T cell development prior to their release into the periphery24. Although it may be possible that colonic bacterial antigens are transported and presented in the thymus to induce tTreg cells, there is currently no evidence that this occurs. A more compelling possibility is that some self-antigen reactive tTreg cells exhibit cross-reactivity with foreign antigens25, resulting in expansion of those bacterial-reactive clones in the intestines. Consistent with these possibilities, a marked overlap between the tTreg and colonic Treg TCR repertoires was observed in one study26.
Another mechanism is that intestinal bacteria can induce the peripheral differentiation of Treg (pTreg) cells from naïve T cells. There are several lines of data suggesting that pTreg cells comprise the majority of the colonic Treg population. First, the use of markers reported to identify tTreg versus pTreg cells suggest that gut bacteria induce pTreg cells. High expression of the transcription factor Helios and cell surface protein Nrp-1 have been associated with tTreg cells27-29. Although the utility of these markers remains controversial30-32, colon lamina propria (cLP) Treg cells contain a much lower proportion of pTreg cells in germ-free compared with SPF mice based on these markers22, 27-29, 33. In addition, treatment of SPF mice with broad-spectrum antibiotics results in the loss of cLP pTreg cells based on Helios levels22. Second, deletion of the conserved noncoding sequence 1 (CNS1) in the Foxp3 locus results in a marked decrease in pTreg, but not tTreg, cell induction34, 35. Notably, CNS1-deficient mice show fewer intestinal Treg cells at the time of weaning34, 35. Of the remaining intestinal Treg cells, there is a lower proportion of Nrp-1lo cells, consistent with loss of pTreg cells in CNS1-deficient mice28. Third, our analysis of colonic Treg TCRs showed that they were unable to induce tTreg cell selection36. The different conclusion from the aforementioned TCR repertoire analyses26 may relate to the different analytical approaches used to assess tTreg cell selection–intrathymic injection of TCR-expressing thymocytes36 vs comparison with thymic TCR repertoires26. The conclusions may also be affected by the different TCRβ chains used26, 36 or the use of a limited TCRα/fixed TCRβ model26, as effects on Treg cell selection has been reported for changes in a single TCR chain (TCRα37). The ability of two of these colonic TCRs (CT2/CT6) to facilitate pTreg cell induction from naïve T cells in the periphery in normal mice was confirmed via the generation of transgenic (Tg) lines33. Altogether, these data suggest that the majority of colonic Treg cells arise via pTreg cell differentiation from naïve T cells.
pTreg cells specific for intestinal bacteria appear to be important for colonic homeostasis. For example, treatment of experimental colitis with Treg cells was more effective using co-transfer of normal Treg cells plus naïve Foxp3- conventional T cells (Tconv) cells that became pTreg cells, than by co-transfer of Treg cells alone38. In conjunction with TCR repertoire analysis, it was suggested that the induced pTreg cells prevented colitis by providing additional TCR specificities not present in the transferred Treg cell population, which were mostly tTreg in origin39. Similarly, mice that have decreased induction of pTreg, but not tTreg, cells through genetic deletion of the CNS1 Foxp3 enhancer region, eventually develop spontaneous colitis characterized by plasmacytic enteritis and high CD4+ T cell expression of Th2 cytokines34. As in other tissues, tTreg cells specific for self-antigens such as those in intestinal epithelial cells are also involved in maintaining homeostasis in the gut16, 22, 40, and may expand specifically in the context of certain types of intestinal perturbations such as sterile injury41. However, the above data suggest that tTreg cells are insufficient to maintain intestinal tolerance and that pTreg cells with a unique set of bacterial antigen specificities are required for homeostasis.
Our group has used colon Treg TCR Tg lines to study the process of pTreg cell differentiation to commensal bacterial antigens33. Using adoptive transfer of peripheral naïve T cells from CT2/CT6 TCR Tg mice into wild type, lymphoreplete mice, it was found that pTreg cell generation was very efficient and resulted in over 80% of cells in the colon and mesenteric lymph nodes upregulating Foxp3 by three weeks after transfer. This was associated with induction of Foxp3 in the most proliferated T cell population, and is in stark contrast with previous studies in non-mucosal tissues showing pTreg cells in the least-divided population42, 43. These colonic TCR Tg data therefore demonstrate that pTreg cell generation can be the dominant outcome of naïve T cell activation.
TCR Tg cells from these two lines did not undergo Treg cell induction upon transfer in young mice, but only in mice around the age of weaning or thereafter33. This was due to the lack of the appropriate bacteria in young mice, rather than an immature immune compartment incapable of inducing Treg cell selection, as fecal microbiota transplant from three-, but not one-, week old donors into one-week old recipients was sufficient to induce Treg cell development. The time-period around weaning is associated with major changes in the composition of the intestinal microbiota due to the switch to solid food and acquisition of a more adult-like microbiota44. In fact, studies using Helios and Nrp-1 to identify Treg cell origin show a marked shift in the colon from tTreg to pTreg cells around weaning22, 23, 27, 33, 36. In summary, there is now a substantial body of work supporting the notion that the majority of colonic Treg cells are generated by pTreg cell selection in response to colonic bacteria acquired around weaning.
Mechanisms of Colonic pTreg Cell Generation
The molecular mechanisms involved in colon pTreg cell development have been recently reviewed45, which we will briefly summarize (Fig. 1). TGFβ, an important factor for the generation of pTreg cells46, can be induced by bacteria such as Clostridium from epithelial cells47. Retinoic acid, a metabolite of dietary vitamin A, has been shown to augment the Foxp3-inducing effects of TGFβ in vitro48-50. Indoleamine-2,3-dioxygenase (IDO) can be produced by dendritic cells and intestinal epithelial cells in response to gut bacteria to favor pTreg cell differentiation51, 52. Short chain fatty acids (SCFAs) produced by gut bacteria from dietary components increase the percentage of Treg cells in the CD4+ T cell subset as well as the expression of IL-10 in Treg cells53-56. SCFAs can inhibit histone deacetylase and via epigenetic modification confer greater stability to Foxp3 gene expression and increase pTreg differentiation54, 55. Polysaccharide A (PSA) from B. fragilis outer membrane vesicles (OMVs) was recently reported to tolerize dendritic cells through LC3-associated phagocytosis, which in turn increases IL-10 expression in gut Treg cells57. Thus, a number of signals originating directly or indirectly from intestinal bacteria or the diet have been described to facilitate Treg cell differentiation or expansion in the gut.
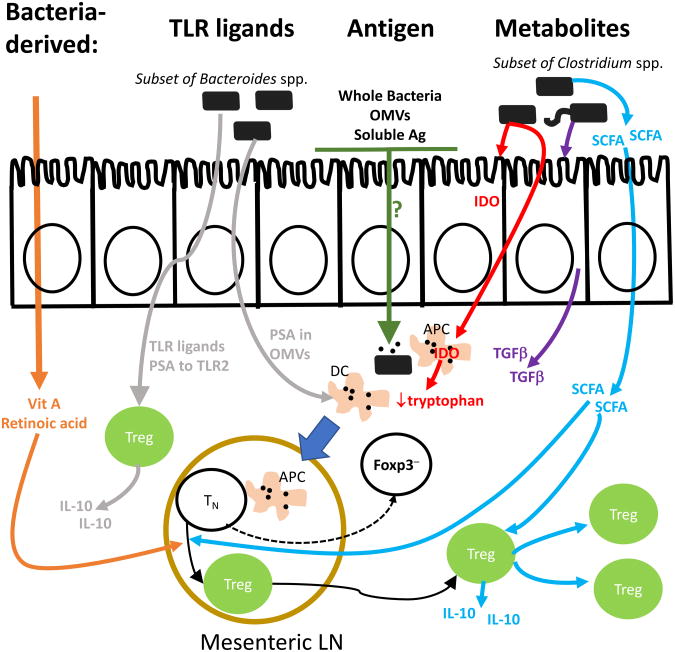
Figure 1. Antigen Specific and Non-Specific Mechanisms of Bacteria-Induced Treg Cell Generation and Function.
Clostridium spp. establish an environment favorable for pTreg cell generation via the induction of TGFβ secretion by intestinal epithelial cells (IECs) (purple arrow), IDO in IECs and APCs (red arrows), and the production of SCFAs from dietary components (blue). Bacteroides spp. such as B. fragilis have been described to increase Treg cell function through PSA activation of TLR2 ligation on T cells and DCs (grey arrows). PSA, as well as SCFAs (blue arrow), induce IL-10 secretion from LP Treg cells. SCFAs also act directly on T cells to induce Treg differentiation/expansion. Dietary vitamin A from the intestinal lumen (orange arrows) can enhance pTreg cell differentiation through its metabolite retinoic acid, which can affect pTreg cell selection by inducing transcription factor binding to a Foxp3 enhancer element in CNS148-50, or via blockade of effector cytokine production by effector T cells129. Bacterial antigens (green arrow), may gain access to immune cells by a variety of mechanisms that are currently unclear, including via goblet-associated passages, transcytosis through IECs, extension of APC processes into the intestinal lumen, or direct invasion of whole bacteria130, 131. Naïve T cells have been reported to encounter commensal antigens in the mesenteric lymph node and undergo pTreg cell selection33. The Peyer's patches and isolated lymphoid follicles may also be important sites of pTreg cell selection, but this requires further study.
While many of these mechanisms have been carefully studied on pTreg cell differentiation in vitro, their in vivo roles are not as well established. Recently, we have used bacteria-reactive colonic TCR Tg lines to begin to address these issues33. For example, colonic TCR Tg cells expressing a dominant negative TGFβRII (dnTGFβRII) transgene to inhibit TGFβ signaling showed only a 50% blockage of pTreg cell generation, whereas a previous study examining non-mucosal associated pTreg cell generation showed a 90% decrease43. As the dnTGFβRII transgene is a hypomorph58, a requirement for TGFβ cannot be excluded. However, these data suggest that pTreg cell generation in the colon is not very sensitive to the level of TGFβ signaling, contrary to predictions based on prior in vitro and in vivo data43. Although it has been suggested that the level of TGFβ is increased in the intestine59, there is little evidence to suggest that the normal level of TGFβ in vivo is high enough to overcome the effect of dnTGFβRII33, 60. Thus, one interpretation of these data is that TGFβ is not a singular or “master” factor that specifies pTreg cell selection, and that other signals in the colon are also important.
Another key mechanism involved in pTreg cell selection is reported to involve CNS1, a region in the Foxp3 locus34, 35 that includes transcription factor binding sites downstream of TGFβ and retinoic acid signaling61, 62. Analysis of naïve T cell differentiation using CNS1-deficient colonic TCR Tg cells revealed a strong inhibition of Foxp3-induction at seven days, but a gradual upregulation of Foxp3 by two to five weeks, which could also be observed in polyclonal cells using Helios or Nrp-133. Thus, CNS1 appears to be important, but not essential, for pTreg cell generation.
Many more studies are required before the factors that affect the colonic Treg cell population are fully understood. One important question is whether these factors affect pTreg cell differentiation, versus expansion or gut homing of pre-existing Treg cells. In one paper, tTreg cell expansion, and not pTreg cell differentiation, was proposed to be the mechanism by which SCFA via Gpr43 affect intestinal Treg cells53. However, this issue is not easily addressed via analysis of polyclonal Treg cell numbers, requiring an approach where the kinetics of Treg cell induction and expansion can be monitored33. Another major issue with experimental manipulations in the study of intestinal Treg cell generation are potential effects on the microbiota, both in terms of population composition but also bacterial gene expression, which is currently not often assessed. For example, NOD2-deficiency can lead to an expansion of B. vulgatus63, which could directly or indirectly affect pTreg cell generation to B. vulgatus or other intestinal species by altering synthesis of SCFA, TLR signals for innate and adaptive immune cells, antigen presentation, and so forth. We think that this issue can be minimized by restricting the experimental manipulation to transferred T cells, which constitute only a small fraction of T cells in the host. While the TCR transgenic transfer model is limited to the study of T cell intrinsic factors, it permits analysis of Foxp3 induction, proliferation, migration, and survival of T cells in a normal host during homeostasis.
Bacterial Species Specificity of Colonic T Cells
The above discussion suggests that tolerance to intestinal bacteria is dependent on induction of Treg cells by intestinal bacteria. In addition to mechanisms such as SCFA and TLR ligands that act through TCR-independent mechanisms, there is growing evidence that the development of colonic Treg cells is in response to species-specific bacterial antigens (Fig. 1). This notion is consistent with TCR repertoire analyses of colonic T cells during homeostasis. Our group found that colonic Treg cells utilize a different TCR repertoire than Treg cells from other secondary lymphoid organs36, suggesting recognition of local colonic antigens. Another study showed that the colonic Treg cell repertoire was markedly affected by broad-spectrum antibiotics26, consistent with bacterial antigens selecting the gut Treg cell population. Thus, these reports show that intestinal bacteria play an important role in shaping the TCR repertoire of colonic Treg cells during homeostasis. Another consideration is that the cLP may also contain pTreg cells with non-microbiota specificities and tTreg cells that are either specific to intestinal self-antigens or cross-react with bacterial antigens. Treg cells are observed in the cLP, albeit in reduced number, in germ free mice22, indicating that non-microbiota-dependent Treg cells can home to and survive in colonic tissue.
Studies of individual bacterial species transferred into germ-free mice support the concept that colonic Treg cells can be specific to bacteria. Both murine and human Clostridium species22, 47 (primarily from clusters IV, XIVa, and XVIII), but not all gut bacterial species, transferred to germ-free mice can induce increased frequencies of Treg cells. While the pro-Treg cell effects of Clostridium species may be both antigen specific and non-specific, in vitro suppression by Treg cells from colonized mice were enhanced by inclusion of Clostridium, but not germ-free isolates with CD11c+ antigen presenting cells47. While this is consistent with Clostridium TCR-specific Treg cell function, it is possible that Clostridium isolates enhanced Treg cell function via TCR-independent effects in this assay. In addition to Clostridium, a recent report identified several Bacteroides species that selectively induced increased percentages of Nrp1lo Treg cells in germ-free mice upon monocolonization64, although this may not be true of all Bacteroides species22. While these mono-colonization data in germ-free mice do not provide direct proof that Treg cells recognize these bacteria in the context of a complex microbiota, they are consistent with the notion that certain bacterial species preferentially induce bacteria-specific pTreg cells.
Our studies of colon TCR specificities also support the existence of bacteria-specific colonic Treg cells. We screened 10 colonic Treg TCRs against a panel of murine bacteria in vitro. One Treg TCR, CT6, showed reactivity to a mouse unclassified Clostridium spp36, which was unfortunately lost during cryopreservation. Subsequently, we have found that CT6 can recognize a human C. symbiosum isolate (unpublished). Another Treg TCR, CT7, was found to react to Parabacteroides distasonis and B. uniformis36. However, recognition of Clostridium or Bacteroides spp. by these TCRs have not yet been confirmed in vivo.
On the whole, the bacterial species recognized by colonic Tconv and Treg cells in vivo in the setting of the complex microbiota in normal SPF mice remain unclear, in contrast with studies showing direct Th17 cell recognition of segmented filamentous bacteria (SFB) in the small intestine65. The best characterized colonic TCR Tg model, CBir1, which recognizes a defined bacteria flagellin epitope, CBir1, is not reported to be activated during homeostasis66. This is not an issue with the TCR Tg line, as analysis of polyclonal T cells using a tetramer with the CBir1 epitope shows that the majority of T cells are in the phenotypically naïve CD44lo population66. Interestingly, CBir1 flagellin falls within the Clostridium subphylum XIVa, which is associated with induction of Treg cells22, 47. These Clostridium species are common constituents of mouse microbiota, and the epitope is clearly present in most microbiota67 (and unpublished observations). Thus, the availability of the epitope in the lumen does not always equate with antigen presentation to T cells.
The relative lack of conclusive data on T cell specificity to gut bacteria is due to several issues. First, in vivo analysis of total Treg cell numbers or function is not proof of TCR recognition, as non-TCR-specific bacterial signals may be important. For example, SCFA have been shown to expand Treg cells in germ-free mice53-55. Similarly, purified PSA from B. fragilis has been shown to induce IL-10 on polyclonal Treg cells68. Retinoic acid48-50 and Ahr ligands69 might be expected to act similarly in an antigen-independent fashion. Second, the study of individual bacteria introduced into germ-free mice may result in their abnormal exposure to the immune system due to the availability of niches which are normally filled by other bacterial species. T cell responses in mono-colonized mice would then need to be verified in more complex microbiota, requiring antigen-specific tools like transgenic mice or MHC tetramers. Third, culturing gut bacteria can be quite difficult. Most species require anaerobic techniques, and many remain unculturable. Fourth, the current protocols for in vitro culture may result in changes in bacterial antigen expression. Use of isolates with the same or similar taxonomy by 16S rDNA sequences may still miss isolate-specific changes in T cell epitopes. Finally, even if TCR recognition of in vitro grown bacteria is achieved, it remains possible that a different species actually provides the antigen to T cells in vivo. In our limited experience, we have observed overlap of TCR reactivity to bacteria of similar taxonomy. For example, CT7 TCR recognizes epitopes from B. uniformis and P. distasonis, whereas DP1 TCR reacts with B. thetaiotaomicron, B. acidifaciens, B. vulgatus, B. sp. TP5, and P. goldsteinii36. Thus, there are a number of hurdles in identifying the species responsible for induction of bacteria-specific Treg cells in vivo.
Dysregulation of pTreg Cell Differentiation in the Colon
The current data suggest that bacteria-reactive Treg cells are often generated from naïve T cells. Gut perturbations may therefore affect T cell differentiation. Models of intestinal inflammation such as IL-10-deficiency are associated with increases in Th1 and Th17 cell frequencies and a decrease in Treg cell frequencies70, 71. On a monoclonal level, CBir1 TCR Tg cells, specific to commensal-derived flagellin, largely remain naïve in healthy hosts but have been shown to develop into stable Th1 effector cells after Toxoplasma infection or into Th17 cells after DSS-mediated mucosal injury and colitis66. Adoption of effector, rather than regulatory, T cell fate may be particularly relevant to the induction of immune responses against commensal antigens, which has been implicated in the pathogenesis of IBD8, 72, 73. Consistent with this hypothesis, CT2/CT6 TCR sequences typically found in the Treg cell subset in normal mouse colons can be found in colonic effector Foxp3− T cell subsets in genetic backgrounds such as IL-10−/− and dnTGFβRII mice that lead to spontaneous colitis36. Treatment of mice with IL-10R antibodies and DSS to induce a transient colitis resulted in differentiation of a portion of the transferred naïve CT2/CT6 cells into RORγt+ Th17 T cells (unpublished, Chai, J.N. & Hsieh, C.S.). In addition, transfer of naïve CT2/CT6 T cells into a Rag1−/− lymphopenic mice resulted in Tbet+ Th1 differentiation33.
The specific signals that disrupt the normal process of pTreg cell development to gut bacteria have not been extensively studied. In a polyclonal setting, expansion of already existing effector cells is likely during inflammation. The inflammatory milieu with altered cytokine balance can also promote differentiation of naïve T-cells into effector phenotypes. Finally, intestinal changes that lead to bacterial antigens being acquired and presented by different APC subsets could affect pTreg vs effector T cell differentiation74. Further work in this area could have therapeutic implications for intestinal inflammatory diseases.
Treg Cells Co-expressing Canonical Effector T Cell Transcriptions Factors
While certain environments described above skew T cell development away from Treg towards effector subsets, normal Foxp3+ Treg cells can upregulate effector transcription factors such as Tbet, GATA3, and RORγt to provide additional functional capacity45. However, Treg expression of GATA3, the canonical transcription factor for Th2 cells, and Tbet for Th1 cells, appears to be independent of intestinal bacteria at homeostasis. GATA3 is expressed in about 15-20% of Treg cells in the colon and small intestine75. GATA3+ Treg cells are equally abundant in germ-free versus SPF mice76 and express Helios, suggestive of tTreg lineage76, 77. Tbet+ Treg cells are even less frequent at around 5% in the mesenteric and peripheral lymph nodes and small intestine lamina propria78. Thus, Treg cells co-expressing GATA3 or Tbet do not appear to play an integral part in the response to commensal bacteria during homeostasis.
By contrast, the colon is normally highly enriched in Treg cells that express the canonical Th17 transcription factor RORγt (30-80%), which is expressed in only 10-20% of Treg cells in the small intestine76, 77, 79, 80. These cells are likely to be bacteria-I nduced pTreg cells, as they are: Helioslo and Nrp-1lo, dramatically decreased in germ free and antibiotic-treated mice, and can be induced in germ-free mice to SPF percentages by several commensal species belonging to different phyla76, 77. RORγt+ Treg cells express high levels of IL-10 and CTLA-476, consistent with activated Treg cells with increased immunoregulatory capability80.
The process by which RORγt+ Treg cells are generated appears to be dependent on certain bacteria. A large study of single bacterial isolates from the human gastrointestinal tract revealed that only certain species induced RORγt expression in Treg cells77. Consistent with this observation, TCR repertoire analyses by our group using a fixed TCRβ model suggest that Foxp3+ RORγt+ Treg cells in the colon use TCRs largely distinct from RORγt− Treg cells and Th17 cells, although some overlap with Th17 cells was observed79. However, another report suggested that TCR usage could be shared between RORγt+ Treg and other subsets80. The different results obtained between these two reports may be due to fixed TCRβ-chain versus paired-chain TCR repertoire analysis in a fully polyclonal population, as well as sequencing of colon versus splenic and lymphoid organ T cells. Nonetheless, this latter analysis is consistent with the observation that non-bacterial ovalbumin in an oral tolerance model induced OT-II cells to become RORγt+ Treg cells in the small intestine76, suggesting that the developmental signals do not need to be directly linked to the T cell epitope. While additional studies are required to resolves these issues, the selectivity of certain gut bacteria to induce the RORγt+ Treg cell population77 favors, but does not prove, the hypothesis that generation of these cells occurs in an antigen-specific context.
The microbiota-derived signals that drive generation of RORγt+ Treg cells are not well established. It has been suggested that T cells first upregulate Foxp3 and then subsequently RORγt in both polyclonal and monoclonal T cell populations77, 79. The development of RORγt after pTreg cell generation suggests that this may occur upon interaction with microbial-induced factors in the colon. Such factors may include IL-6 and IL-23, although the studies were not entirely consistent76, 77. The microbial-derived signals that generate RORγt+ Treg cells in vivo therefore remain to be established. In addition, the temporal sequence of Foxp3 and then RORγt induction may only apply to a subset of cells. For example, it may be speculated that cells expressing TCRs that overlap with Th17 cells79 could arise from upregulating RORγt first and then Foxp3.
Although RORγt is the canonical Th17 transcription factor, it is not clear that RORγt+ Treg cells primarily exist to inhibit Th17-mediated inflammation. For example, the frequency of RORγt-expressing Treg cells76, 77, 79, 80 in a tissue is inverse that of Th17 cells, which are found predominantly in the small intestine and not the colon81. In addition, Treg cell-specific knockout of RORγt has been reported to result in Th2-mediated or Th1/Th17-mediated colitis76, 77, and not purely Th17-mediated disease. Future studies are therefore required to determine the specific mechanisms by which RORγt expression facilitates Treg cell suppression of specific subsets of T effector cells in the colon.
Function of Colonic Treg Cells
Intestinal Treg cells likely employ a number of mechanisms for regulating effector T cell responses to gut bacteria, including cell surface proteins and cytokines (reviewed in 17). One likely mechanism is the inhibition of antigen presenting cell (APC) function by CTLA-4, a transmembrane protein constitutively expressed in Treg cells82. CTLA-4 binding to CD80/86 can prevent CD28 on effector T cells from engaging and costimulating TCR activation83. Treg cell CTLA-4 may also directly remove CD80/CD86 from APC membranes84. Treg cell-mediated suppression via CTLA-4 appears to be important clinically, as an important side effect of CTLA-4 antibody therapy for human malignancy is the development of colitis85.
Treg cells also secrete pro-tolerogenic cytokines such as TGFβ86. Deletion of TGFβ1 from T cells results in spontaneous autoimmunity, albeit delayed in comparison with a germ-line knockout87. While one might have predicted that Treg cell-derived TGFβ1 was primarily affected by CD4Cre-mediated deletion, Foxp3Cre-mediated deletion of TGFβ1 did not result in development of spontaneous autoimmunity or colitis88. However, an effect of TGFβ1 was observed in the T cell transfer model of colitis into lymphopenic hosts87, suggesting that certain experimental conditions could reveal a role for Treg cell-derived TGFβ. Thus, these data demonstrate that while TGFβ production by Treg cells may play a role, it is not essential for preserving normal gut homeostasis.
IL-10 is another important immuno-regulatory cytokine in the gut. Genetic polymorphisms of IL-10 and IL-10R in humans are associated with development of IBD89, 90, and IL-10 deficient mice develop spontaneous colitis91. T cells are an important source of IL-1092, with important contributions from both Treg and non-Treg cells93. Notably, of the CD4+ T cell population, colonic Treg cells exhibit the greatest production of IL-10 based on the use of reporters94, and the expression of IL-10 is associated with co-expression of RORγt76. Induction of IL-10 in gut Treg cells has been reported to occur with microbiota-derived factors such as B. fragilis-derived PSA via a T cell intrinsic TLR2-dependent mechanism68. In summary, there is considerable evidence that gut bacteria induce IL-10 expressing pTreg cells in the colon, and that Treg cell production of IL-10 is necessary to preserve gut homeostasis.
However, none of the Treg cell-derived factors such as CTLA-4 or regulatory cytokines like IL-10 appear to be directed at effector T cells in an antigen-specific manner. Rather, Treg cell inhibition of antigen-specific effector cells could occur via effects on APCs. A recent study using multiplex quantitative imaging showed that Treg cells cluster with CD11c+MHCIIhi APCs in lymphoid tissue and that CTLA-4 expression is increased in clustered versus non-clustered Treg cells95. Expression of the TCR was required on Treg cells for Treg cell/APC clustering. Thus, these data provide evidence that TCR activation of gut bacteria-specific pTreg cells generate a local microenvironment that is anti-inflammatory via CTLA-4 and IL-10 interactions with dendritic cells, thereby inhibiting effector cells specific to bacterial-antigens that may also be presented on that APC.
Antigen-Specificity of IgA
The specificity of gut IgA responses may provide important clues for understanding T cell responses in the colon. IgA is known to be induced against toxins and pathogenic bacteria96, but it has become clear that IgA is also induced by normal intestinal bacteria during homeostasis97. Unexpectedly, this recognition may actually facilitate the uptake and presentation of bacteria or other antigens within the intestinal lumen to the adaptive immune system97. Thus, IgA responses may reflect adaptive immune responses to intestinal bacterial antigens.
One important issue, though, is that IgA specificity cannot be directly equated with T cell antigen-recognition, as both T-dependent (TD) and T-independent (TI) IgA responses occur98, 99. Some studies have suggested that the majority of IgA in the gut is TD, as the level of free IgA in T cell-deficient mice is 1/5th that of mice with T cells100. In addition, sequencing studies showed that a large portion of gut IgA has somatic hypermutation97, 101, supporting a substantial role for T follicular helper (Tfh) cells in gut IgA induction. By contrast, studies assessing IgA reactivity to bacteria using flow cytometry of T cell-deficient mice have varied in their interpretation as to whether TI IgA represents a major102 versus minor103 part of the B cell response to commensal bacteria. However, the flow cytometry assay does not assess reactivity to secreted bacterial antigens or outer-membrane vesicles (OMVs), which could be important targets of TD IgA. Thus, the relative contribution of TD versus TI IgA in response to commensal bacteria during homeostasis is incompletely understood.
TD IgA to surface bacterial antigens seen by bacterial flow cytometry may occur primarily to immunostimulatory bacteria strong enough to induce Tfh responses97. For example, IgA responses to bacteria obtained from co-housing with inflammasome-deficient (Asc−/−) mice occurred only in T cell-sufficient mice103 and were specific to Prevotellaceae, Helicobacter sp. flexspira, and SFB–all potential drivers of intestinal inflammation104-106. In other studies, two groups observed that SFB107 and Mucispirillum108, both bacteria that tightly adhere to the epithelium, induce TD IgA102, 103. In studies of fecal bacteria from IBD patients, species that were preferentially IgA+, and not IgA−, facilitated DSS-mediated colitis in mice103. Similarly, a study of human undernourished children with enteropathy revealed IgA-responses to Enterobacteriaceae, whose pathogenicity was confirmed in mice109. Finally, pathogenic secreted bacterial factors like cholera toxin specifically require Th17 cells for IgA induction110. Thus, bacteria with increased immunostimulatory potential such as pathobionts may be speculated to induce TD IgA responses.
In addition to these analyses, TCR Tg studies have supported a role for anti-bacterial TD IgA. CBir1 Tg Treg cells induce flagellin-specific IgA molecules when transferred into T-cell deficient mice111. Similarly, we have observed that DP1 transgenic T cells, which recognize Bacteroidaceae species in vitro36, can also induce IgA responses against those species when adoptively transferred into T cell-deficient mice (unpublished). One caveat of these studies is that they are done in lymphopenic mice, which may show altered T cell development as well as intestinal permeability to bacteria (Fig. 2). Nonetheless, these data directly demonstrate that antigen-specific T cell responses can induce bacteria-specific IgA. In summary, there has been significant progress in understanding the specificity of intestinal IgA. However, much remains to be learned about the characteristics of the bacteria that result in T and B cell responses during homeostasis and the T helper subset responses they elicit.
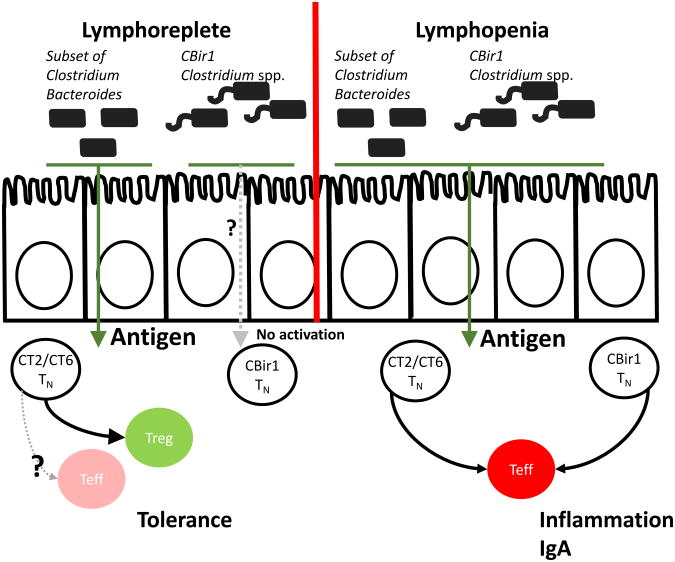
Figure 2. Alternative Regulation of Intestinal Bacterial Antigen Presentation and T Cell Fate in Lymphoreplete vs. Lymphopenic Mice.
In lymphoreplete mice, commensal spp. induce naïve colonic TCR transgenic CT2/CT6 cells into Treg cells. Based on in vitro and in vivo studies, it is predicted that select Bacteroides and Clostridium spp. induce antigen-specific Treg cells in lymphoreplete mice. However, naive CT2/CT6 T cells develop into effector T cells in Rag1−/− lymphopenic mice. In contrast to the behavior of of CT2/CT6, CBir1 antigen, a component of flagella present in many gut bacteria species including those in Clostridium subphylum XIVa, does not activate T cells or induce differentiation of Treg cells in lymphoreplete mice. However, these antigens are presented in lymphopenic mice leading to T cell activation and IgA production. Mucosal injury or infection will also lead to antigen presentation to CBir1 reactive T cells. In these environments, CBir1 reactive T cells become effector, and not Treg, cells.
Role for Treg cells in Induction or Suppression of IgA
With the exception of SFB, there is a relative dearth of studies that integrate both T and B cell reactivity to intestinal bacteria at homeostasis. For example, Clostridium spp. induce Treg cell responses22, but the IgA responses are not characterized in normal hosts. Several Lactobacillus species increase the percentage of Treg cells in the colon and spleen104, 112, but Lactobacillus-specific IgA appears to be TI102, 103. In contrast, Mucispirillum species induce TD IgA102, 103, but the T cell response to Mucispirillum during homeostasis has not been clearly established. One example in which data are available for both T and B cell responses is Bacteroides thetaiotamicron, which can induce IgA113 and expansion of colonic RORγt+ pTreg cells77 when monocolonized in germ-free hosts. By combining the results of these two different studies, it could be hypothesized that Treg cell responses drive IgA induction during homeostasis.
A role for Treg cells in IgA induction has been proposed. In a T cell transfer model into lymphopenic mice, co-transfer of Foxp3+ Treg cells is necessary to limit colitis, maintain intestinal microbial diversity, and induce IgA114. This process appeared to result from loss of Foxp3 in a portion of the transferred Treg cells, which then upregulate Tfh markers such as Bcl6, IL-21, CD40L, and CXCR5115. Another group used the CBir1 TCR Tg line to show that transfer of Treg cells into TCR-deficient mice preferentially induced CBir1-specific IgA111. In addition, depletion of Treg cells using anti-CD25 led to a marked drop in anti-CBir1 IgA, suggesting that Treg cells may directly induce IgA via TGFβ. However, the use of lymphopenic models may result in loss of Treg cell stability116, leading to the trans-differentiation of Treg into Foxp3– Tfh cells, which may not occur in normal hosts. In addition, the use of anti-CD25 antibodies may kill activated T cells or lead to gut inflammation, which may alter the range of antigens presented to the immune system66. Thus, while there is intriguing data suggesting that gut Treg cells induce IgA, either directly or serving as precursors for Foxp3− Tfh cells, this Treg cell function during normal homeostasis requires further investigation.
Moreover, it has been argued that Treg cells actually inhibit B cell germinal center responses via the T follicular regulatory (Tfr) cell subset. In the spleen, Tfr cells have been shown to develop from Foxp3+ tTreg cells, maintain expression of Foxp3, and upregulate expression of Tfh markers such as PD-1, CXCR5, and Bcl6117. However, Tfr cells do not express CD40L or the Tfh cytokines IL-21 and IL-4, and function to limit the number of Tfh cells in germinal centers as well as inhibit the non-antigen-specific selection of B cells to prevent autoantibody formation117-119. Consistent with these observations in non-mucosal sites, Peyer's patch (PP) Foxp3+ germinal center Tfr cells were necessary to produce more specific, affinity-matured IgA114. Defective Tfr function resulted in a larger percentage of fecal bacteria that stained IgA+, suggesting that Tfr responses are more important in the gut for limiting IgA induction, rather than directly inducing IgA responses themselves.
Interestingly, CNS1−/− Foxp3 mice, which show decreased induction of pTreg cells, develop plasmacytic enteritis characterized by increased serum antibodies against small intestine, large intestine, and chow antigens34, which could hint at the necessity of antigen-specific Tfr cells to maintain intestinal homeostasis. However, it can be difficult to dissect in this model whether this is due to Treg cells controlling effector T cell activation versus Tfh cells in the germinal center reaction. Thus, future studies are required to determine whether gut Treg cells promote and/or inhibit IgA responses.
In brief, the role of Treg cells in TD IgA remains unclear. A straightforward hypothesis is that bacteria induce Treg cells, which, via expression of TGFβ, facilitate IgA class switch. However, it may be possible that Treg cells do not induce TD IgA. In this case, it may be possible that bacteria could induce a small fraction of effector T cells that stimulate IgA production, which may be subsequently regulated by Treg cell-derived Tfr cells. Thus, new studies are required to understand the concomitant T and B cell response to intestinal bacteria at homeostasis.
Conclusions
In summary, there has been considerable progress recently in our understanding of adaptive immune responses to colonic bacteria during homeostasis. First, it is clear that intestinal bacteria play a crucial role in educating the colonic immune system, with distinct effects on the TCR repertoire. TD IgA responses are also induced, although it has not been established whether this occurs in the colon or elsewhere. Second, the T cell response to colonic bacteria can result in naïve T cell differentiation into pTreg cells. Commensal bacteria appear to facilitate this process via provision of antigens and generation of metabolites such as SCFA or inducing TGFβ production by epithelial cells. Third, pTreg cell responses can be the dominant outcome of naïve T cell differentiation in vivo, making it a bone fide peripheral T helper subset. Finally, bacteria-induced pTreg cells are necessary in vivo to prevent colitis.
However, these data also raise important issues. For example, what are the bacterial specificities of colonic Treg cells during homeostasis in the context of a normal microbiota (Fig. 1)? If IgA specificity is a guide, we might predict that there will be relatively few bacterial species that induce pTreg cell responses in a normal host. This would be consistent with the observation that CBir1 TCR Tg cells do not see their antigen under homeostatic conditions66, even though it is clear that the antigen is relatively abundant in the lumen. We speculate that this means that the mucous/mucosa layer functions as a “wall” to prevent many/most bacterial antigens access to immune cells (Fig. 2). This contrasts with the small intestine, where oral administration of ovalbumin results in antigen presentation to T cells and Treg cell induction120, 121. This is further supported by the recent observation that the majority of small intestinal pTreg cells are induced by dietary antigens122. Yet, it is evident that commensal bacteria have a tremendous impact on the colon TCR repertoire, suggesting that those bacteria that do get through are immunodominant over self-antigen presentation. One possible explanation is that OMVs123 or soluble antigens124 may have easier access to APCs than bacterial-associated antigens such as flagellin. Nonetheless, we predict that only a small fraction of intestinal bacterial antigens is presented to the adaptive immune system in the colon.
Another issue is the use of lymphopenic mice for the study of intestinal tolerance (Fig. 2). While it has been the classic model by which to study the role of Treg cells in the colon125, our study suggests that pTreg cell differentiation is markedly perturbed in this environment33. This may be due to abnormal barrier function, which results in bacterial translocation into the lamina propria, mesenteric lymph nodes, and spleen11. This may facilitate increased proliferation126 and induce an unbalanced cytokine environment that favors effector T cell differentiation of bacteria-specific T cells. Nonetheless, it may be possible in the future to establish conditions that “normalize” the lymphopenic model by adding in sub-therapeutic amounts of polyclonal Treg cells39 or other manipulations such as anti-cytokine antibody, thereby permitting the study of commensal-specific pTreg cell differentiation and function in a setting where the antigen-specificities can be experimentally controlled.
While most of this review has focused on Treg cells, the role of effector T cells during homeostasis is not well characterized. As noted above, the observed bacterial translocation from the intestinal lumen to secondary immune organs in lymphopenic mice suggests that T cells are required for full barrier function. However, other than Th17 cells reactive to SFB, identification of effector cells reactive to colonic bacteria during homeostasis has not been successful36. One possibility is that Treg cells, perhaps via RORγt, are capable of generating the signals required for epithelial homeostasis. Another is that commensal bacteria may induce, along with pTreg cells, some effector T cells that acts to improve barrier function and limit bacterial invasion. The small fraction of bacteria-specific effector T cells would be constrained by Treg cells. Like self-reactive effector T cells, their presence may only be recognized when Treg cells are depleted127. A combined effector:Treg cell response to commensal antigens would be analogous to concomitant immunity such as that observed with Treg cells in Leishmania infection128. Future studies will be required to determine the role of effector T cells for intestinal homeostasis to commensal bacteria.
Finally, the type and extent of antigen-specific T cell responses involved in IgA induction is poorly characterized during homeostasis. It is unknown whether induction of IgA to commensal bacteria even occurs in the colon, as it may occur in more proximal parts of the intestine which have a thinner mucous layer and more defined antigen uptake mechanisms. Another question is whether Treg, Th17, or another subset is involved in IgA induction to commensal bacteria. Thus, many questions remain regarding gut bacterial interactions with the adaptive immune system.
Acknowledgments
C.S.H. is supported by NIH grants R01 DK094995 and R21 AI097535, the CCFA, and the Burroughs Wellcome Fund. E.V.R.-G. is supported by NIH grant F30 DK111071-01. E.V.R.-G. and S.R. are supported by NIH grant T32 GM007200-42.
Footnotes
Conflict of Interest: The authors declared no conflict of interest.
Author Contributions: E.V.R.-G., S.R., and C.-S.H. contributed to the planning, writing, and editing of this review.
References
- 1.Gilbert JA, Quinn RA, Debelius J, Xu ZZ, Morton J, Garg N, et al. Microbiome-wide association studies link dynamic microbial consortia to disease. Nature. 2016;535(7610):94–103. doi: 10.1038/nature18850. [DOI] [PubMed] [Google Scholar]
- 2.Hand TW. The Role of the Microbiota in Shaping Infectious Immunity. Trends in immunology. 2016;37(10):647–658. doi: 10.1016/j.it.2016.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Leslie JL, Young VB. The rest of the story: the microbiome and gastrointestinal infections. Current opinion in microbiology. 2015;23:121–125. doi: 10.1016/j.mib.2014.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Flint HJ, Scott KP, Louis P, Duncan SH. The role of the gut microbiota in nutrition and health. Nat Rev Gastroenterol Hepatol. 2012;9(10):577–589. doi: 10.1038/nrgastro.2012.156. [DOI] [PubMed] [Google Scholar]
- 5.Blanton LV, Barratt MJ, Charbonneau MR, Ahmed T, Gordon JI. Childhood undernutrition, the gut microbiota, and microbiota-directed therapeutics. Science. 2016;352(6293):1533. doi: 10.1126/science.aad9359. [DOI] [PubMed] [Google Scholar]
- 6.Belkaid Y. Regulatory T cells and infection: a dangerous necessity. Nat Rev Immunol. 2007;7(11):875–888. doi: 10.1038/nri2189. [DOI] [PubMed] [Google Scholar]
- 7.Maizels RM, Smith KA. Regulatory T cells in infection. Adv Immunol. 2011;112:73–136. doi: 10.1016/B978-0-12-387827-4.00003-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Maloy KJ, Powrie F. Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature. 2011;474(7351):298–306. doi: 10.1038/nature10208. [DOI] [PubMed] [Google Scholar]
- 9.Alexander KL, Targan SR, Elson CO., 3rd Microbiota activation and regulation of innate and adaptive immunity. Immunol Rev. 2014;260(1):206–220. doi: 10.1111/imr.12180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Honda K, Littman DR. The microbiota in adaptive immune homeostasis and disease. Nature. 2016;535(7610):75–84. doi: 10.1038/nature18848. [DOI] [PubMed] [Google Scholar]
- 11.Mombaerts P, Mizoguchi E, Grusby MJ, Glimcher LH, Bhan AK, Tonegawa S. Spontaneous development of inflammatory bowel disease in T cell receptor mutant mice. Cell. 1993;75(2):274–282. doi: 10.1016/0092-8674(93)80069-q. [DOI] [PubMed] [Google Scholar]
- 12.Veazey RS, DeMaria M, Chalifoux LV, Shvetz DE, Pauley DR, Knight HL, et al. Gastrointestinal tract as a major site of CD4+ T cell depletion and viral replication in SIV infection. Science. 1998;280(5362):427–431. doi: 10.1126/science.280.5362.427. [DOI] [PubMed] [Google Scholar]
- 13.Brenchley JM, Douek DC. HIV infection and the gastrointestinal immune system. Mucosal Immunol. 2008;1(1):23–30. doi: 10.1038/mi.2007.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vallance BA, Deng W, Knodler LA, Finlay BB. Mice lacking T and B lymphocytes develop transient colitis and crypt hyperplasia yet suffer impaired bacterial clearance during Citrobacter rodentium infection. Infect Immun. 2002;70(4):2070–2081. doi: 10.1128/IAI.70.4.2070-2081.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Simmons CP, Clare S, Ghaem-Maghami M, Uren TK, Rankin J, Huett A, et al. Central role for B lymphocytes and CD4+ T cells in immunity to infection by the attaching and effacing pathogen Citrobacter rodentium. Infect Immun. 2003;71(9):5077–5086. doi: 10.1128/IAI.71.9.5077-5086.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maloy KJ, Powrie F. Regulatory T cells in the control of immune pathology. Nature Immunology. 2001;2(9):816–822. doi: 10.1038/ni0901-816. [DOI] [PubMed] [Google Scholar]
- 17.Josefowicz SZ, Lu LF, Rudensky AY. Regulatory T cells: mechanisms of differentiation and function. Annu Rev Immunol. 2012;30:531–564. doi: 10.1146/annurev.immunol.25.022106.141623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roncarolo MG, Gregori S, Bacchetta R, Battaglia M. Tr1 cells and the counter-regulation of immunity: natural mechanisms and therapeutic applications. Curr Top Microbiol Immunol. 2014;380:39–68. doi: 10.1007/978-3-662-43492-5_3. [DOI] [PubMed] [Google Scholar]
- 19.Sun M, He C, Cong Y, Liu Z. Regulatory immune cells in regulation of intestinal inflammatory response to microbiota. Mucosal Immunol. 2015;8(5):969–978. doi: 10.1038/mi.2015.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sarrabayrouse G, Alameddine J, Altare F, Jotereau F. Microbiota-Specific CD4CD8alphaalpha Tregs: Role in Intestinal Immune Homeostasis and Implications for IBD. Frontiers in immunology. 2015;6:522. doi: 10.3389/fimmu.2015.00522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sujino T, London M, Hoytema van Konijnenburg DP, Rendon T, Buch T, Silva HM, et al. Tissue adaptation of regulatory and intraepithelial CD4(+) T cells controls gut inflammation. Science. 2016;352(6293):1581–1586. doi: 10.1126/science.aaf3892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Atarashi K, Tanoue T, Shima T, Imaoka A, Kuwahara T, Momose Y, et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science. 2011;331(6015):337–341. doi: 10.1126/science.1198469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Geuking MB, Cahenzli J, Lawson MA, Ng DC, Slack E, Hapfelmeier S, et al. Intestinal bacterial colonization induces mutualistic regulatory T cell responses. Immunity. 2011;34(5):794–806. doi: 10.1016/j.immuni.2011.03.021. [DOI] [PubMed] [Google Scholar]
- 24.Klein L, Kyewski B, Allen PM, Hogquist KA. Positive and negative selection of the T cell repertoire: what thymocytes see (and don't see) Nat Rev Immunol. 2014;14(6):377–391. doi: 10.1038/nri3667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee HM, Bautista JL, Scott-Browne J, Mohan JF, Hsieh CS. A Broad Range of Self-Reactivity Drives Thymic Regulatory T Cell Selection to Limit Responses to Self. Immunity. 2012 doi: 10.1016/j.immuni.2012.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cebula A, Seweryn M, Rempala GA, Pabla SS, McIndoe RA, Denning TL, et al. Thymus-derived regulatory T cells contribute to tolerance to commensal microbiota. Nature. 2013;497(7448):258–262. doi: 10.1038/nature12079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thornton AM, Korty PE, Tran DQ, Wohlfert EA, Murray PE, Belkaid Y, et al. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J Immunol. 2010;184(7):3433–3441. doi: 10.4049/jimmunol.0904028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Weiss JM, Bilate AM, Gobert M, Ding Y, Curotto de Lafaille MA, Parkhurst CN, et al. Neuropilin 1 is expressed on thymus-derived natural regulatory T cells, but not mucosa-generated induced Foxp3+ T reg cells. J Exp Med. 2012;209(10):1723–1742. S1721. doi: 10.1084/jem.20120914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yadav M, Louvet C, Davini D, Gardner JM, Martinez-Llordella M, Bailey-Bucktrout S, et al. Neuropilin-1 distinguishes natural and inducible regulatory T cells among regulatory T cell subsets in vivo. J Exp Med. 2012;209(10):1713–1722. S1711–1719. doi: 10.1084/jem.20120822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Akimova T, Beier UH, Wang L, Levine MH, Hancock WW. Helios expression is a marker of T cell activation and proliferation. PLoS One. 2011;6(8):e24226. doi: 10.1371/journal.pone.0024226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gottschalk RA, Corse E, Allison JP. Expression of Helios in peripherally induced Foxp3+ regulatory T cells. J Immunol. 2012;188(3):976–980. doi: 10.4049/jimmunol.1102964. [DOI] [PubMed] [Google Scholar]
- 32.Szurek E, Cebula A, Wojciech L, Pietrzak M, Rempala G, Kisielow P, et al. Differences in Expression Level of Helios and Neuropilin-1 Do Not Distinguish Thymus-Derived from Extrathymically-Induced CD4+Foxp3+ Regulatory T Cells. PLoS One. 2015;10(10):e0141161. doi: 10.1371/journal.pone.0141161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nutsch K, Chai JN, Ai TL, Russler-Germain E, Feehley T, Nagler CR, et al. Rapid and Efficient Generation of Regulatory T Cells to Commensal Antigens in the Periphery. Cell Rep. 2016;17(1):206–220. doi: 10.1016/j.celrep.2016.08.092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Josefowicz SZ, Niec RE, Kim HY, Treuting P, Chinen T, Zheng Y, et al. Extrathymically generated regulatory T cells control mucosal TH2 inflammation. Nature. 2012;482(7385):395–399. doi: 10.1038/nature10772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zheng Y, Josefowicz S, Chaudhry A, Peng XP, Forbush K, Rudensky AY. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature. 2010;463(7282):808–812. doi: 10.1038/nature08750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lathrop SK, Bloom SM, Rao SM, Nutsch K, Lio CW, Santacruz N, et al. Peripheral education of the immune system by colonic commensal microbiota. Nature. 2011;478(7368):250–254. doi: 10.1038/nature10434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Stadinski BD, Shekhar K, Gomez-Tourino I, Jung J, Sasaki K, Sewell AK, et al. Hydrophobic CDR3 residues promote the development of self-reactive T cells. Nat Immunol. 2016;17(8):946–955. doi: 10.1038/ni.3491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Haribhai D, Lin W, Edwards B, Ziegelbauer J, Salzman NH, Carlson MR, et al. A central role for induced regulatory T cells in tolerance induction in experimental colitis. J Immunol. 2009;182(6):3461–3468. doi: 10.4049/jimmunol.0802535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Haribhai D, Williams JB, Jia S, Nickerson D, Schmitt EG, Edwards B, et al. A requisite role for induced regulatory T cells in tolerance based on expanding antigen receptor diversity. Immunity. 2011;35(1):109–122. doi: 10.1016/j.immuni.2011.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Legoux FP, Lim JB, Cauley AW, Dikiy S, Ertelt J, Mariani TJ, et al. CD4+ T Cell Tolerance to Tissue-Restricted Self Antigens Is Mediated by Antigen-Specific Regulatory T Cells Rather Than Deletion. Immunity. 2015;43(5):896–908. doi: 10.1016/j.immuni.2015.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Burzyn D, Kuswanto W, Kolodin D, Shadrach JL, Cerletti M, Jang Y, et al. A special population of regulatory T cells potentiates muscle repair. Cell. 2013;155(6):1282–1295. doi: 10.1016/j.cell.2013.10.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gottschalk RA, Corse E, Allison JP. TCR ligand density and affinity determine peripheral induction of Foxp3 in vivo. J Exp Med. 2010;207(8):1701–1711. doi: 10.1084/jem.20091999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kretschmer K, Apostolou I, Hawiger D, Khazaie K, Nussenzweig MC, von Boehmer H. Inducing and expanding regulatory T cell populations by foreign antigen. Nat Immunol. 2005;6(12):1219–1227. doi: 10.1038/ni1265. [DOI] [PubMed] [Google Scholar]
- 44.Pantoja-Feliciano IG, Clemente JC, Costello EK, Perez ME, Blaser MJ, Knight R, et al. Biphasic assembly of the murine intestinal microbiota during early development. The ISME journal. 2013;7(6):1112–1115. doi: 10.1038/ismej.2013.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tanoue T, Atarashi K, Honda K. Development and maintenance of intestinal regulatory T cells. Nat Rev Immunol. 2016;16(5):295–309. doi: 10.1038/nri.2016.36. [DOI] [PubMed] [Google Scholar]
- 46.Chen W, Jin W, Hardegen N, Lei KJ, Li L, Marinos N, et al. Conversion of peripheral CD4+ CD25- naive T cells to CD4+ CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. Journal of Experimental Medicine. 2003;198(12):1875–1886. doi: 10.1084/jem.20030152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Atarashi K, Tanoue T, Oshima K, Suda W, Nagano Y, Nishikawa H, et al. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature. 2013;500(7461):232–236. doi: 10.1038/nature12331. [DOI] [PubMed] [Google Scholar]
- 48.Benson MJ, Pino-Lagos K, Rosemblatt M, Noelle RJ. All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J Exp Med. 2007;204(8):1765–1774. doi: 10.1084/jem.20070719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mucida D, Pino-Lagos K, Kim G, Nowak E, Benson MJ, Kronenberg M, et al. Retinoic acid can directly promote TGF-beta-mediated Foxp3(+) Treg cell conversion of naive T cells. Immunity. 2009;30(4):471–472. doi: 10.1016/j.immuni.2009.03.008. author reply 472-473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nolting J, Daniel C, Reuter S, Stuelten C, Li P, Sucov H, et al. Retinoic acid can enhance conversion of naive into regulatory T cells independently of secreted cytokines. J Exp Med. 2009;206(10):2131–2139. doi: 10.1084/jem.20090639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Matteoli G, Mazzini E, Iliev ID, Mileti E, Fallarino F, Puccetti P, et al. Gut CD103+ dendritic cells express indoleamine 2,3-dioxygenase which influences T regulatory/T effector cell balance and oral tolerance induction. Gut. 2010;59(5):595–604. doi: 10.1136/gut.2009.185108. [DOI] [PubMed] [Google Scholar]
- 52.Munn DH, Mellor AL. Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends in immunology. 2013;34(3):137–143. doi: 10.1016/j.it.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Smith PM, Howitt MR, Panikov N, Michaud M, Gallini CA, Bohlooly YM, et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science. 2013;341(6145):569–573. doi: 10.1126/science.1241165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Furusawa Y, Obata Y, Fukuda S, Endo TA, Nakato G, Takahashi D, et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature. 2013 doi: 10.1038/nature12721. [DOI] [PubMed] [Google Scholar]
- 55.Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, Deroos P, et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature. 2013 doi: 10.1038/nature12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Singh N, Gurav A, Sivaprakasam S, Brady E, Padia R, Shi H, et al. Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity. 2014;40(1):128–139. doi: 10.1016/j.immuni.2013.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chu H, Khosravi A, Kusumawardhani IP, Kwon AH, Vasconcelos AC, Cunha LD, et al. Gene-microbiota interactions contribute to the pathogenesis of inflammatory bowel disease. Science. 2016;352(6289):1116–1120. doi: 10.1126/science.aad9948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ishigame H, Mosaheb MM, Sanjabi S, Flavell RA. Truncated form of TGF-betaRII, but not its absence, induces memory CD8+ T cell expansion and lymphoproliferative disorder in mice. J Immunol. 2013;190(12):6340–6350. doi: 10.4049/jimmunol.1300397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Konkel JE, Chen W. Balancing acts: the role of TGF-beta in the mucosal immune system. Trends in molecular medicine. 2011;17(11):668–676. doi: 10.1016/j.molmed.2011.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Feuerer M, Hill JA, Kretschmer K, von Boehmer H, Mathis D, Benoist C. Genomic definition of multiple ex vivo regulatory T cell subphenotypes. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(13):5919–5924. doi: 10.1073/pnas.1002006107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xu L, Kitani A, Stuelten C, McGrady G, Fuss I, Strober W. Positive and negative transcriptional regulation of the Foxp3 gene is mediated by access and binding of the Smad3 protein to enhancer I. Immunity. 2010;33(3):313–325. doi: 10.1016/j.immuni.2010.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Tone Y, Furuuchi K, Kojima Y, Tykocinski ML, Greene MI, Tone M. Smad3 and NFAT cooperate to induce Foxp3 expression through its enhancer. Nat Immunol. 2008;9(2):194–202. doi: 10.1038/ni1549. [DOI] [PubMed] [Google Scholar]
- 63.Ramanan D, Tang MS, Bowcutt R, Loke P, Cadwell K. Bacterial sensor Nod2 prevents inflammation of the small intestine by restricting the expansion of the commensal Bacteroides vulgatus. Immunity. 2014;41(2):311–324. doi: 10.1016/j.immuni.2014.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Faith JJ, Ahern PP, Ridaura VK, Cheng J, Gordon JI. Identifying gut microbe-host phenotype relationships using combinatorial communities in gnotobiotic mice. Sci Transl Med. 2014;6(220):220ra211. doi: 10.1126/scitranslmed.3008051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yang Y, Torchinsky MB, Gobert M, Xiong H, Xu M, Linehan JL, et al. Focused specificity of intestinal T17 cells towards commensal bacterial antigens. Nature. 2014 doi: 10.1038/nature13279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hand TW, Dos Santos LM, Bouladoux N, Molloy MJ, Pagan AJ, Pepper M, et al. Acute gastrointestinal infection induces long-lived microbiota-specific T cell responses. Science. 2012;337(6101):1553–1556. doi: 10.1126/science.1220961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pabst O, Hornef M. Gut microbiota: a natural adjuvant for vaccination. Immunity. 2014;41(3):349–351. doi: 10.1016/j.immuni.2014.09.002. [DOI] [PubMed] [Google Scholar]
- 68.Round JL, Mazmanian SK. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proceedings of the National Academy of Sciences of the United States of America. 2010;107(27):12204–12209. doi: 10.1073/pnas.0909122107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Quintana FJ, Basso AS, Iglesias AH, Korn T, Farez MF, Bettelli E, et al. Control of T(reg) and T(H)17 cell differentiation by the aryl hydrocarbon receptor. Nature. 2008;453(7191):65–71. doi: 10.1038/nature06880. [DOI] [PubMed] [Google Scholar]
- 70.Keubler LM, Buettner M, Hager C, Bleich A. A Multihit Model: Colitis Lessons from the Interleukin-10-deficient Mouse. Inflamm Bowel Dis. 2015;21(8):1967–1975. doi: 10.1097/MIB.0000000000000468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Chaudhry A, Samstein RM, Treuting P, Liang Y, Pils MC, Heinrich JM, et al. Interleukin-10 signaling in regulatory T cells is required for suppression of Th17 cell-mediated inflammation. Immunity. 2011;34(4):566–578. doi: 10.1016/j.immuni.2011.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Elson CO, Cong Y. Host-microbiota interactions in inflammatory bowel disease. Gut microbes. 2012;3(4):332–344. doi: 10.4161/gmic.20228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jostins L, Ripke S, Weersma RK, Duerr RH, McGovern DP, Hui KY, et al. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature. 2012;491(7422):119–124. doi: 10.1038/nature11582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Cummings RJ, Barbet G, Bongers G, Hartmann BM, Gettler K, Muniz L, et al. Different tissue phagocytes sample apoptotic cells to direct distinct homeostasis programs. Nature. 2016;539(7630):565–569. doi: 10.1038/nature20138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wohlfert EA, Grainger JR, Bouladoux N, Konkel JE, Oldenhove G, Ribeiro CH, et al. GATA3 controls Foxp3(+) regulatory T cell fate during inflammation in mice. The Journal of clinical investigation. 2011;121(11):4503–4515. doi: 10.1172/JCI57456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ohnmacht C, Park JH, Cording S, Wing JB, Atarashi K, Obata Y, et al. MUCOSAL IMMUNOLOGY. The microbiota regulates type 2 immunity through RORgammat(+) T cells. Science. 2015;349(6251):989–993. doi: 10.1126/science.aac4263. [DOI] [PubMed] [Google Scholar]
- 77.Sefik E, Geva-Zatorsky N, Oh S, Konnikova L, Zemmour D, McGuire AM, et al. MUCOSAL IMMUNOLOGY. Individual intestinal symbionts induce a distinct population of RORgamma(+) regulatory T cells. Science. 2015;349(6251):993–997. doi: 10.1126/science.aaa9420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yu F, Sharma S, Edwards J, Feigenbaum L, Zhu J. Dynamic expression of transcription factors T-bet and GATA-3 by regulatory T cells maintains immunotolerance. Nat Immunol. 2015;16(2):197–206. doi: 10.1038/ni.3053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Solomon BD, Hsieh CS. Antigen-Specific Development of Mucosal Foxp3+RORgammat+ T Cells from Regulatory T Cell Precursors. J Immunol. 2016 doi: 10.4049/jimmunol.1601217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Yang BH, Hagemann S, Mamareli P, Lauer U, Hoffmann U, Beckstette M, et al. Foxp3(+) T cells expressing RORgammat represent a stable regulatory T-cell effector lineage with enhanced suppressive capacity during intestinal inflammation. Mucosal Immunol. 2016;9(2):444–457. doi: 10.1038/mi.2015.74. [DOI] [PubMed] [Google Scholar]
- 81.Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, et al. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006;126(6):1121–1133. doi: 10.1016/j.cell.2006.07.035. [DOI] [PubMed] [Google Scholar]
- 82.Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008;322(5899):271–275. doi: 10.1126/science.1160062. [DOI] [PubMed] [Google Scholar]
- 83.Esensten JH, Helou YA, Chopra G, Weiss A, Bluestone JA. CD28 Costimulation: From Mechanism to Therapy. Immunity. 2016;44(5):973–988. doi: 10.1016/j.immuni.2016.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Qureshi OS, Zheng Y, Nakamura K, Attridge K, Manzotti C, Schmidt EM, et al. Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science. 2011;332(6029):600–603. doi: 10.1126/science.1202947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Scalapino KJ, Daikh DI. CTLA-4: a key regulatory point in the control of autoimmune disease. Immunol Rev. 2008;223:143–155. doi: 10.1111/j.1600-065X.2008.00639.x. [DOI] [PubMed] [Google Scholar]
- 86.Travis MA, Sheppard D. TGF-beta activation and function in immunity. Annu Rev Immunol. 2014;32:51–82. doi: 10.1146/annurev-immunol-032713-120257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Li MO, Wan YY, Flavell RA. T cell-produced transforming growth factor-beta1 controls T cell tolerance and regulates Th1- and Th17-cell differentiation. Immunity. 2007;26(5):579–591. doi: 10.1016/j.immuni.2007.03.014. [DOI] [PubMed] [Google Scholar]
- 88.Gutcher I, Donkor MK, Ma Q, Rudensky AY, Flavell RA, Li MO. Autocrine transforming growth factor-beta1 promotes in vivo Th17 cell differentiation. Immunity. 2011;34(3):396–408. doi: 10.1016/j.immuni.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Franke A, Balschun T, Karlsen TH, Sventoraityte J, Nikolaus S, Mayr G, et al. Sequence variants in IL10, ARPC2 and multiple other loci contribute to ulcerative colitis susceptibility. Nat Genet. 2008;40(11):1319–1323. doi: 10.1038/ng.221. [DOI] [PubMed] [Google Scholar]
- 90.Engelhardt KR, Shah N, Faizura-Yeop I, Kocacik Uygun DF, Frede N, Muise AM, et al. Clinical outcome in IL-10- and IL-10 receptor-deficient patients with or without hematopoietic stem cell transplantation. The Journal of allergy and clinical immunology. 2013;131(3):825–830. doi: 10.1016/j.jaci.2012.09.025. [DOI] [PubMed] [Google Scholar]
- 91.Kuhn R, Lohler J, Rennick D, Rajewsky K, Muller W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75(2):263–274. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- 92.Roers A, Siewe L, Strittmatter E, Deckert M, Schluter D, Stenzel W, et al. T cell-specific inactivation of the interleukin 10 gene in mice results in enhanced T cell responses but normal innate responses to lipopolysaccharide or skin irritation. J Exp Med. 2004;200(10):1289–1297. doi: 10.1084/jem.20041789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Rubtsov YP, Rasmussen JP, Chi EY, Fontenot J, Castelli L, Ye X, et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity. 2008;28(4):546–558. doi: 10.1016/j.immuni.2008.02.017. [DOI] [PubMed] [Google Scholar]
- 94.Maynard CL, Harrington LE, Janowski KM, Oliver JR, Zindl CL, Rudensky AY, et al. Regulatory T cells expressing interleukin 10 develop from Foxp3(+) and Foxp3(-) precursor cells in the absence of interleukin 10. Nat Immunol. 2007;8(9):931–941. doi: 10.1038/ni1504. [DOI] [PubMed] [Google Scholar]
- 95.Liu Z, Gerner MY, Van Panhuys N, Levine AG, Rudensky AY, Germain RN. Immune homeostasis enforced by co-localized effector and regulatory T cells. Nature. 2015;528(7581):225–230. doi: 10.1038/nature16169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Macpherson AJ, McCoy KD, Johansen FE, Brandtzaeg P. The immune geography of IgA induction and function. Mucosal Immunol. 2008;1(1):11–22. doi: 10.1038/mi.2007.6. [DOI] [PubMed] [Google Scholar]
- 97.Pabst O. New concepts in the generation and functions of IgA. Nat Rev Immunol. 2012;12(12):821–832. doi: 10.1038/nri3322. [DOI] [PubMed] [Google Scholar]
- 98.Fagarasan S, Kawamoto S, Kanagawa O, Suzuki K. Adaptive immune regulation in the gut: T cell-dependent and T cell-independent IgA synthesis. Annu Rev Immunol. 2010;28:243–273. doi: 10.1146/annurev-immunol-030409-101314. [DOI] [PubMed] [Google Scholar]
- 99.Macpherson AJ, Gatto D, Sainsbury E, Harriman GR, Hengartner H, Zinkernagel RM. A primitive T cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science. 2000;288(5474):2222–2226. doi: 10.1126/science.288.5474.2222. [DOI] [PubMed] [Google Scholar]
- 100.Bemark M, Boysen P, Lycke NY. Induction of gut IgA production through T cell-dependent and T cell-independent pathways. Ann N Y Acad Sci. 2012;1247:97–116. doi: 10.1111/j.1749-6632.2011.06378.x. [DOI] [PubMed] [Google Scholar]
- 101.Lindner C, Wahl B, Fohse L, Suerbaum S, Macpherson AJ, Prinz I, et al. Age, microbiota, and T cells shape diverse individual IgA repertoires in the intestine. J Exp Med. 2012;209(2):365–377. doi: 10.1084/jem.20111980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Bunker JJ, Flynn TM, Koval JC, Shaw DG, Meisel M, McDonald BD, et al. Innate and Adaptive Humoral Responses Coat Distinct Commensal Bacteria with Immunoglobulin A. Immunity. 2015;43(3):541–553. doi: 10.1016/j.immuni.2015.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Palm NW, de Zoete MR, Cullen TW, Barry NA, Stefanowski J, Hao L, et al. Immunoglobulin A coating identifies colitogenic bacteria in inflammatory bowel disease. Cell. 2014;158(5):1000–1010. doi: 10.1016/j.cell.2014.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Tang C, Kamiya T, Liu Y, Kadoki M, Kakuta S, Oshima K, et al. Inhibition of Dectin-1 Signaling Ameliorates Colitis by Inducing Lactobacillus-Mediated Regulatory T Cell Expansion in the Intestine. Cell Host Microbe. 2015;18(2):183–197. doi: 10.1016/j.chom.2015.07.003. [DOI] [PubMed] [Google Scholar]
- 105.Kullberg MC, Ward JM, Gorelick PL, Caspar P, Hieny S, Cheever A, et al. Helicobacter hepaticus triggers colitis in specific-pathogen-free interleukin-10 (IL-10)-deficient mice through an IL-12- and gamma interferon-dependent mechanism. Infect Immun. 1998;66(11):5157–5166. doi: 10.1128/iai.66.11.5157-5166.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Stepankova R, Powrie F, Kofronova O, Kozakova H, Hudcovic T, Hrncir T, et al. Segmented filamentous bacteria in a defined bacterial cocktail induce intestinal inflammation in SCID mice reconstituted with CD45RBhigh CD4+ T cells. Inflamm Bowel Dis. 2007;13(10):1202–1211. doi: 10.1002/ibd.20221. [DOI] [PubMed] [Google Scholar]
- 107.Ivanov, II, Atarashi K, Manel N, Brodie EL, Shima T, Karaoz U, et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139(3):485–498. doi: 10.1016/j.cell.2009.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Robertson BR, O'Rourke JL, Neilan BA, Vandamme P, On SL, Fox JG, et al. Mucispirillum schaedleri gen. nov., sp. nov., a spiral-shaped bacterium colonizing the mucus layer of the gastrointestinal tract of laboratory rodents. Int J Syst Evol Microbiol. 2005;55(Pt 3):1199–1204. doi: 10.1099/ijs.0.63472-0. [DOI] [PubMed] [Google Scholar]
- 109.Kau AL, Planer JD, Liu J, Rao S, Yatsunenko T, Trehan I, et al. Functional characterization of IgA-targeted bacterial taxa from undernourished Malawian children that produce diet-dependent enteropathy. Sci Transl Med. 2015;7(276):276ra224. doi: 10.1126/scitranslmed.aaa4877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Hirota K, Turner JE, Villa M, Duarte JH, Demengeot J, Steinmetz OM, et al. Plasticity of Th17 cells in Peyer's patches is responsible for the induction of T cell-dependent IgA responses. Nat Immunol. 2013;14(4):372–379. doi: 10.1038/ni.2552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Cong Y, Feng T, Fujihashi K, Schoeb TR, Elson CO. A dominant, coordinated T regulatory cell-IgA response to the intestinal microbiota. Proceedings of the National Academy of Sciences of the United States of America. 2009;106(46):19256–19261. doi: 10.1073/pnas.0812681106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Karimi K, Inman MD, Bienenstock J, Forsythe P. Lactobacillus reuteri-induced regulatory T cells protect against an allergic airway response in mice. Am J Respir Crit Care Med. 2009;179(3):186–193. doi: 10.1164/rccm.200806-951OC. [DOI] [PubMed] [Google Scholar]
- 113.Peterson DA, McNulty NP, Guruge JL, Gordon JI. IgA response to symbiotic bacteria as a mediator of gut homeostasis. Cell Host Microbe. 2007;2(5):328–339. doi: 10.1016/j.chom.2007.09.013. [DOI] [PubMed] [Google Scholar]
- 114.Kawamoto S, Maruya M, Kato LM, Suda W, Atarashi K, Doi Y, et al. Foxp3(+) T cells regulate immunoglobulin a selection and facilitate diversification of bacterial species responsible for immune homeostasis. Immunity. 2014;41(1):152–165. doi: 10.1016/j.immuni.2014.05.016. [DOI] [PubMed] [Google Scholar]
- 115.Tsuji M, Komatsu N, Kawamoto S, Suzuki K, Kanagawa O, Honjo T, et al. Preferential generation of follicular B helper T cells from Foxp3+ T cells in gut Peyer's patches. Science. 2009;323(5920):1488–1492. doi: 10.1126/science.1169152. [DOI] [PubMed] [Google Scholar]
- 116.Komatsu N, Mariotti-Ferrandiz ME, Wang Y, Malissen B, Waldmann H, Hori S. Heterogeneity of natural Foxp3+ T cells: a committed regulatory T-cell lineage and an uncommitted minor population retaining plasticity. Proceedings of the National Academy of Sciences of the United States of America. 2009;106(6):1903–1908. doi: 10.1073/pnas.0811556106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Chung Y, Tanaka S, Chu F, Nurieva RI, Martinez GJ, Rawal S, et al. Follicular regulatory T cells expressing Foxp3 and Bcl-6 suppress germinal center reactions. Nature medicine. 2011;17(8):983–988. doi: 10.1038/nm.2426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Linterman MA, Pierson W, Lee SK, Kallies A, Kawamoto S, Rayner TF, et al. Foxp3+ follicular regulatory T cells control the germinal center response. Nature medicine. 2011;17(8):975–982. doi: 10.1038/nm.2425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wollenberg I, Agua-Doce A, Hernandez A, Almeida C, Oliveira VG, Faro J, et al. Regulation of the germinal center reaction by Foxp3+ follicular regulatory T cells. J Immunol. 2011;187(9):4553–4560. doi: 10.4049/jimmunol.1101328. [DOI] [PubMed] [Google Scholar]
- 120.Thorstenson KM, Khoruts A. Generation of anergic and potentially immunoregulatory CD25+CD4 T cells in vivo after induction of peripheral tolerance with intravenous or oral antigen. J Immunol. 2001;167(1):188–195. doi: 10.4049/jimmunol.167.1.188. [DOI] [PubMed] [Google Scholar]
- 121.Zhang X, Izikson L, Liu L, Weiner HL. Activation of CD25(+)CD4(+) regulatory T cells by oral antigen administration. J Immunol. 2001;167(8):4245–4253. doi: 10.4049/jimmunol.167.8.4245. [DOI] [PubMed] [Google Scholar]
- 122.Kim KS, Hong SW, Han D, Yi J, Jung J, Yang BG, et al. Dietary antigens limit mucosal immunity by inducing regulatory T cells in the small intestine. Science. 2016;351(6275):858–863. doi: 10.1126/science.aac5560. [DOI] [PubMed] [Google Scholar]
- 123.Hickey CA, Kuhn KA, Donermeyer DL, Porter NT, Jin C, Cameron EA, et al. Colitogenic Bacteroides thetaiotaomicron Antigens Access Host Immune Cells in a Sulfatase-Dependent Manner via Outer Membrane Vesicles. Cell Host Microbe. 2015;17(5):672–680. doi: 10.1016/j.chom.2015.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Veenbergen S, van Berkel LA, du Pre MF, He J, Karrich JJ, Costes LM, et al. Colonic tolerance develops in the iliac lymph nodes and can be established independent of CD103 dendritic cells. Mucosal Immunol. 2015 doi: 10.1038/mi.2015.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Shale M, Schiering C, Powrie F. CD4(+) T-cell subsets in intestinal inflammation. Immunol Rev. 2013;252(1):164–182. doi: 10.1111/imr.12039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Kieper WC, Troy A, Burghardt JT, Ramsey C, Lee JY, Jiang HQ, et al. Recent immune status determines the source of antigens that drive homeostatic T cell expansion. J Immunol. 2005;174(6):3158–3163. doi: 10.4049/jimmunol.174.6.3158. [DOI] [PubMed] [Google Scholar]
- 127.Kim JM, Rasmussen JP, Rudensky AY. Regulatory T cells prevent catastrophic autoimmunity throughout the lifespan of mice. Nat Immunol. 2007;8(2):191–197. doi: 10.1038/ni1428. [DOI] [PubMed] [Google Scholar]
- 128.Belkaid Y, Piccirillo CA, Mendez S, Shevach EM, Sacks DL. CD4+ CD25+ regulatory T cells control Leishmania major persistence and immunity. Nature. 2002;420:502–507. doi: 10.1038/nature01152. [DOI] [PubMed] [Google Scholar]
- 129.Hill JA, Hall JA, Sun CM, Cai Q, Ghyselinck N, Chambon P, et al. Retinoic acid enhances Foxp3 induction indirectly by relieving inhibition from CD4+CD44hi Cells. Immunity. 2008;29(5):758–770. doi: 10.1016/j.immuni.2008.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Worbs T, Hammerschmidt SI, Forster R. Dendritic cell migration in health and disease. Nat Rev Immunol. 2017;17(1):30–48. doi: 10.1038/nri.2016.116. [DOI] [PubMed] [Google Scholar]
- 131.Knoop KA, Miller MJ, Newberry RD. Transepithelial antigen delivery in the small intestine: different paths, different outcomes. Current opinion in gastroenterology. 2013;29(2):112–118. doi: 10.1097/MOG.0b013e32835cf1cd. [DOI] [PMC free article] [PubMed] [Google Scholar]