Abstract
Background
Brain injury (BI) is reported in 60% of newborns with critical congenital heart disease (CHD) as white matter injury (WMI) or stroke. Neurodevelopmental (ND) impairments are reported in these patients. The relationship between neonatal BI and ND outcome has not been established.
Objectives
To determine the association between peri-operative BI and ND outcomes in infants with single ventricle physiology (SVP) and d- Transposition of the great arteries (TGA).
Methods
Term newborns with TGA and SVP had pre- and post-operative brain MRIs and ND outcomes assessed at 12 and 30 months with Bayley Scales of Infant Development-II. BI was categorized by the Brain Injury Severity score and WMI was quantified by volumetric analysis.
Results
104 infants had follow-up at 12 months and 70 at 30 months. At 12 months, only clinical variables were associated with ND outcome. At 30 months, subjects with moderate-severe WMI had significantly lower psychomotor development index (PDI) scores (13 points lower) as compared to those with none/minimal WMI for TGA and SVP (p= 0.03 and 0.05) after adjusting for various factors. Quantitative WMI volume was likewise associated. Stroke was not associated with outcome. The Bland-Altman limits of agreement for PDI scores at 12 and 30 months were wide (−40.3 to 31.2) across the range of mean PDI values.
Conclusions
Increasing burden of WMI is associated with worse motor outcomes at 30 months for infants with critical CHD while no adverse association was seen between small strokes and outcome. These results support the utility of neonatal brain MRI in this population to aid in predicting later outcomes and the importance of ND follow-up beyond one year of age.
Keywords: Congenital heart disease, Neurodevelopmental outcomes, Brain Injury
Introduction
Advances in peri-operative care have led to improved survival of newborns with critical congenital heart disease (CHD) (1,2). Although there has been a decline in overt neurologic insults in these children, many experience behavioral, emotional, cognitive and motor impairment suggesting widespread brain dysfunction that continues into adulthood (3–5). Despite significant improvements over time in survival, surgical strategies and perioperative care, neurodevelopmental (ND) outcomes have only modestly improved (6). These findings suggest that patient specific risk factors and brain health may be key determinants of neurodevelopmental outcome.
Studies have documented a high prevalence of peri-operative brain injury and delayed brain development in neonates with CHD (7–10). However, few studies have identified a strong correlation between perioperative brain injury and ND outcome. The largest study in infants with CHD found no association between neonatal peri-operative brain injury and outcome (11) while other studies reported divergent results (12,13). In contrast, several important clinical variables are known to influence ND outcomes in children with CHD through mechanisms that don’t involve brain injury, including maternal education, socioeconomic status and presence of a genetic abnormality (14–16).
Given the high rates of brain injury in neonates with CHD, the use of peri-operative brain imaging in their clinical care is increasing. We hypothesize that the presence of peri-operative brain injury is associated with ND deficits in infancy. The aim of this study was to prospectively determine the association between neonatal brain injury and ND outcome at 12 and 30 months of age among patients with d- Transposition of the great arteries (d-TGA) and single ventricle physiology (SVP).
Methods
Between 2001 and 2013, newborns with critical CHD at the University of California San Francisco Benioff Children’s Hospital (UCSF) and University of British Columbia (UBC) were consecutively invited to participate in a prospective protocol obtaining pre- and post-operative MRI and ND follow-up at 12 and 30 months of age. Brain imaging findings from earlier versions of this cohort were previously reported (7,8). Patients who were born prior to 36 weeks gestation, had a suspected congenital infection, had clinical evidence of a congenital malformation or syndrome, and/or had a suspected or confirmed genetic or chromosomal anomaly were excluded. Informed consent was obtained. The institutional committee on human research approved the study protocol at each site.
Patients diagnosed as having d-TGA or SVP were included in the study. D-TGA was defined as great vessel malposition with the aorta arising from the right ventricle and pulmonary artery arising from the left ventricle with or without a ventricular septal defect. SVP was defined as the absence of one of two functioning ventricles requiring a palliative surgical intervention for survival in the newborn period.
MRI Study
Preoperative MRI studies were performed as soon as the baby could be safely transported to the MRI scanner as determined by the clinical team. Postoperative studies were performed after completion of perioperative care and prior to discharge from the hospital. Imaging time points were separated by an average of 15 days in the entire cohort. Detailed methods are listed in the Online Appendix. A neuroradiologist at each site reviewed each MRI for focal, multifocal or global changes (A.J.B. and K.P.), blinded to clinical variables. Brain injury was characterized as stroke, white matter injury (WMI), intraventricular hemorrhage (IVH), and/or global hypoxic ischemic injury as previously described(8). The description of post-operative brain injuries was limited to newly acquired lesions not evident on the pre-operative scan. WMI was further classified as mild (1–3 foci each < 2mm), moderate (>3 foci or any foci > 2mm), or severe (>5% of WM volume). IVH was characterized as grade I, II, III or periventricular hemorrhagic infarct using the system of Papile et al (17). No subjects were found to have IVH > grade II. Brain Injury Severity (BIS) was categorized for each subject as previously described in an ordinal scale (18): 0= none or minimal injury (mild WMI and IVH grade I–II; no stroke); 1= stroke (any size stroke without moderate to severe WMI); 2= moderate to severe injury (moderate and severe WMI). BIS score was assigned to the pre-operative MRI and to the post-operative MRI for newly acquired lesions. The BIS score was assigned based on the worst injury observed. For example, if a subject had stroke and moderate to severe WMI a BIS score of 2 was assigned. To account for multiple injuries in a single subject at both time points, a maximal BIS score was determined, which was the highest score between the pre-op and post-op BIS scores. The quantitative assessment of WMI was performed by a trained rater (TG) and reviewed by an experienced neonatal neurologist (SM). Punctate WMI was characterized by areas of T1 hyperintensity and was manually delineated on all available pre- and post-operative scans. The total WMI volume was determined by manual segmentation as previously described (19). If WMI was identified on both the pre- and post-operative T1-weighted images, the largest total WMI volume was included in the statistical analysis (maximal WMI volume).
Diffusion Tensor Imaging
Diffusion tensor imaging was performed using a sequence optimized at each site for neonatal brain imaging to measure microstructural brain development. For the purposes of this analysis, we focused on the regional directionality of water motion in the white matter, defined as fractional anisotropy (FA). With increasing microstructural brain development, the regional directionality of water motion increases thus FA increases (20). The FA was calculated for 5 voxels in the white matter bilaterally using prespecified anatomical references (7): 1) anterior white matter; 2) central white matter; 3) posterior white matter; 4) posterior limb of the internal capsule; 5) optic radiations. Correct region of interest placements were confirmed by neuroradiologists at each site (A.J.B. and K.P.). The values from the left and right hemispheres were averaged and a mean value was used for analysis.
Clinical Variables
Clinical data was prospectively collected from the medical records by a team of trained neonatal research nurses and reviewed by a pediatric intensivist and cardiologist (PM and SP) blinded to all neuroimaging findings. Maternal education was recorded as a surrogate of socioeconomic status using the Hollingshead scale of educational status (1–7) in the Four Factor Index of Social Status.
Neurodevelopmental Assessment
The Bayley Scales of Infant Development-II (BSID-II) was administered at 12 and 30 months of age to obtain the psychomotor development index (PDI) and mental development index (MDI). Testing was administered by a single qualified individual (licensed psychologist or experienced psychometrician) at each site, blinded to the subject’s cardiac diagnosis, clinical factors and brain imaging findings.
Statistical Analysis
Clinical and MRI characteristics were compared between subjects that had follow-up at 12 and 30 months and those that did not (due to death or loss to follow-up). Our primary outcome was prospectively defined as the cognitive (MDI) and motor (PDI) scores of the BSID-II at 12 and 30 months of age. Our primary exposure was peri-operative brain injury as measured by the BIS score. To assess the relationship between brain injury and neurodevelopmental outcome, linear regression was used with maximal BIS or maximal WMI volume as the independent variable and either the PDI or MDI score as the dependent variable at each time point (12 and 30 months). A univariable linear regression was performed assessing known or biologically plausible predictors of ND outcome or confounders in the relationship between brain injury and ND outcome. Variables with an association of p<0.1 were included in the final model. The final multivariable linear regression model was stratified by cardiac lesion and adjusted for all significant variables in the univariable analysis. Bland-Altman limits of agreement were calculated to assess the concordance of BSID-II scores at 12 and 30 months. All analyses were performed on Stata 14.0 software (Stata corporation, College, Texas, U.S.A.).
Results
A total of 165 neonates were enrolled at birth and obtained peri-operative (pre- and postoperative) brain MRIs: 98 at UCSF and 67 at UBC. By 12 months, 28 subjects had died and 33 subjects were lost to follow-up and by 30 months, 2 additional subjects had died and an additional 32 subjects were lost to follow-up (Figure 1). At each time point, baseline demographics did not differ between those that achieved follow-up versus those that did not, except for site and cardiac diagnosis (Online Tables 1A, 1B). At each time point, there was no difference in the prevalence of neonatal brain injury (either pre-or post-op) based on follow-up status (Online Tables 2A, 2B).
Figure 1. Study subject flow chart.
Subjects were consecutively invited to participate in the prospective protocol obtaining pre- and post-operative MRI’s and neurodevelopmental follow-up at 12 and 30 months of age.
104 infants had peri-operative imaging and ND follow-up at 12 months (d-TGA= 84; SVP= 20). At 30 months, 70 children had peri-operative imaging and ND follow-up (d-TGA= 54; SVP= 16). At each time point (12 and 30 months), baseline demographics were no different between those with peri-operative brain injury and those without (Tables 1A, 1B). The prevalence and type of brain injury among subjects with follow-up is demonstrated in Table 2.
Table 1a.
Baseline characteristics of subjects with 12 month outcome by injury status (n= 104)
| No Injury (n= 48) | Injury (n= 56) | p-value | |
|---|---|---|---|
| Male N(%) | 10 (20.8%) | 23 (41.1%) | 0.03 |
| Race/ethnicity | 0.29 | ||
| White non-Hispanic | 26 (39.4%) | 40 (60.6%) | |
| Hispanic | 11 (57.9%) | 8 (42.1%) | |
| Black | 0 | 1 (100%) | |
| Asian | 5 (62.5%) | 3 (37.5%) | |
| Other | 6 (60%) | 4 (40%) | |
| GA birth, weeks | 39.3 (38.9–39.6) | 38.8 (38.3–39.2) | 0.055 |
| Birthweight, kg | 3.3 (3.2–3.5) | 3.3 (3.2–3.5) | 0.93 |
| Birth HC, cm | 34.0 (33.6–34.4) | 34.0 (33.6–34.4) | 0.99 |
| Weight 12 m, kg | 10.9 (9.3–12.6) | 9.6 (9.3–10.0) | 0.12 |
| HC 12m, cm | 46.6 (46.1–47.0) | 46.0 (45.5–46.4) | 0.07 |
| Preop Sat (low) | 64.8 (58.6–71.0) | 65.9 (60.9–70.9) | 0.77 |
| Lesion N(%) | 0.60 | ||
| TGA (n= 84) | 40 (47.6%) | 44 (52.4%) | |
| SVP (n= 20) | 8 (40%) | 12 (60%) | |
| Site | |||
| UCSF | 24 (45.3%) | 29 (54.7%) | 0.85 |
| UBC | 24 (47.1%) | 27 (52.9%) |
GA= gestational age; HC= head circumference; preop sat= lowest pre-operative oxygen saturation; TGA= d-transposition of the great arteries; SVP= single ventricle physiology.
Table 1b.
Baseline characteristics of subjects with 30 month outcome by injury status (n= 70)
| No Injury (n= 30) | Injury (n= 40) | p-value | |
|---|---|---|---|
| Male N(%) | 9 (30%) | 16 (40%) | 0.38 |
| Race/Ethnicity | |||
| White non-Hispanic | 19 (39.6%) | 29 (60.4%) | 0.74 |
| Hispanic | 4 (50%) | 4 (50%) | |
| Black | 0 | 1 (100%) | |
| Asian | 5 (62.5%) | 3 (37.5%) | |
| Other | 2 (40%) | 3 (60%) | |
| GA birth, weeks | 39.0 (38.6–39.5) | 38.9 (38.5–39.3) | 0.67 |
| Birth HC, cm | 33.7 (33.1–34.3) | 34.1 (33.5–34.5) | 0.40 |
| Birthweight, kg | 3.3 (3.1–3.5) | 3.4 (3.2–3.6) | 0.52 |
| Weight 30m, kg | 13.7 (13.1–14.3) | 13.8 (13.1–14.5) | 0.75 |
| HC 30m, cm | 48.7 (48.0–49.3) | 47.5 (45.9–49.1) | 0.21 |
| Preop Sat (low) | 65.9 (57.9–73.9) | 68.2 (62.7–73.7) | 0.62 |
| Lesion N(%) | 0.10 | ||
| TGA (n= 54) | 26 (48.2%) | 28 (51.8%) | |
| SVP (n= 16) | 4 (25%) | 12 (75%) | |
| Site | 0.94 | ||
| UCSF | 11 (42.3%) | 15 (57.7%) | |
| UBC | 19 (43.2%) | 25 (56.8%) |
Table 2.
Prevalence of neonatal brain injury among subjects with 12 and 30 month follow-up data.
| 12 month follow-up* (n= 104) | 30m follow-up† (n= 70) | |
|---|---|---|
| Any Injury | ||
| All Subjects | 56/104 (53.8%) | 40/70 (57.1%) |
| TGA | 44/84 (52.4%) | 28/54 (51.9%) |
| SVP | 12/20 (60%) | 12/16 (75%) |
| WMI | ||
| TGA | 30/84 (35.7%) | 19/54 (35.2%) |
| SVP | 9/20 (45%) | 8/16 (50%) |
| Stroke | ||
| TGA | 24/84 (28.6%) | 14/54 (25.9%) |
| SVP | 3/20 (15%) | 6/16 (37.5%) |
TGA= d-transposition of the great arteries; SVP= single ventricle physiology; WMI= white matter injury
At 12 months, 10 subjects with d-TGA had both stroke and white matter injury noted on either the pre- or post-operative MRI
At 30 months, 5 subjects with d-TGA and 2 subjects with SVP had both stroke and white matter injury noted on either the pre- or post-operative MRI.
Approximately 55% of subjects had some type of peri-operative brain injury in the neonatal period, with WMI being the most common type of injury (Figure 2). Two subjects had hypoxic-ischemic brain injury, both of which primarily involved the white matter, thus they were characterized as having severe WMI. Although stroke was also highly prevalent, 25 of 29 subjects with ND outcome data (86%) had small strokes (<1/3 of the arterial territory). Four subjects had larger strokes (1/3–2/3 of the arterial territory), all of whom had MDI and PDI scores below 90. For this study, we assessed the maximal BIS score. Among the subjects with follow-up data, all BIS scores either stayed the same or were worse on the post-operative scan as compared to the pre-operative scan. As expected, mean PDI and MDI scores were lower than the normative mean (100±15) at each time point (Online Table 3).
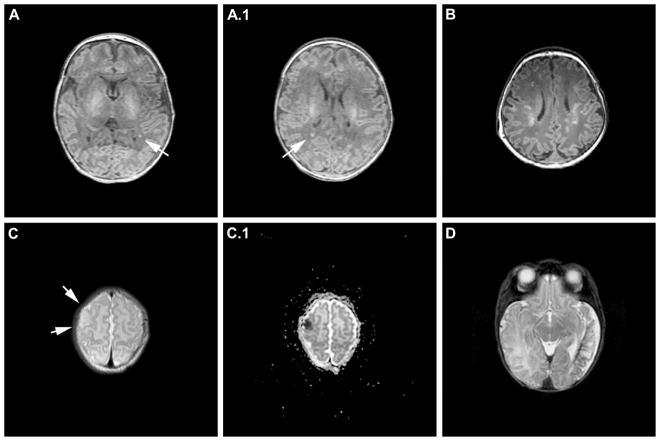
Figure 2. Brain MRI examples of subjects with varying degrees of white matter injury or stroke.
Panels A and A.1 are pre-operative T1-weighted images from a subject with hypoplastic left heart syndrome and moderate white matter injury (>/= 3 foci or any foci > 2mm). There are at least 3 (one seen in panel A and another in A.1, arrows) small foci of hyperintensities consistent with white matter injury. Panel B is a pre-operative T-1 weighted image from a subject with hypoplastic left heart syndrome and severe white matter injury (> 5% of white matter volume). Panel C is a T2 weighted image from a pre-operative scan on a subject with d-transposition of the great arteries. The arrows demonstrate a small stroke manifested as hyperintense cortical signal in the middle cerebral artery distribution (< 1/3 of the arterial distribution). Panel C.1 is the corresponding average diffusivity map demonstrating reduced water diffusivity (dark spot) in the same region. Panel D is a T2-weighted image from a postoperative scan on a subject with d-Transposition of the great arteries with a large subacute/chronic stroke in the middle cerebral artery distribution.
In the univariable analyses at 12 months, only clinical factors were significantly associated with MDI or PDI (Online Table 4). In particular, cardiac lesion, BAS and maternal education were associated with MDI. Similarly, only birth weight, pre-operative saturations, cardiac lesion and BAS were associated with PDI. Those with pre-operative or any stroke (BIS = 1) had significantly higher MDI and PDI values at 12 months respectively. No other relationship was noted between BIS and outcome at 12 months.
At 30 months, maximal BIS score was significantly associated with PDI. Those with BIS = 2 (moderate to severe WMI) had PDI scores on average 13 (95% CI: −22.52, −3.26) points lower as compared to those with none or minimal injury. Additional variables associated with PDI included maternal education, cardiac lesion, BAS and site. No association was seen between WMI and MDI (Online Table 5). Of note, on univariable analyses BAS was associated with significantly higher PDI and MDI scores at 30 months as compared to those that did not have a BAS. Those with stroke (BIS = 1) had significantly higher MDI scores as compared to those with none or minimal injury (similar findings were seen with PDI and MDI at 12 months). The associations between BAS, stroke and outcome were not significant in the multivariable analysis stratified by cardiac lesion.
In the final model, the relationship between maximal BIS and PDI was assessed at 30 months. The analysis was stratified by cardiac lesion. After adjustment for site, BAS and maternal education, those with moderate to severe WMI (BIS = 2) had significantly lower PDI scores as compared to those with none or minimal injury (BIS = 0) for both d-TGA and SVP (TGA: PDI scores were 14 (95% CI: −26.8, −1.1) points lower for those with BIS = 2 compared to those with BIS = 0, p= 0.03; SVP: PDI scores were 14 (95% CI: −27.7,0.13) points lower for those with BIS = 2 compared to those with BIS = 0, p= 0.05) (Central Illustration ,Table 3). The effects of other variables in the multivariable analysis are listed in Table 4. To address the possibility of misclassification of severity of WMI using the qualitative scale (21), the same analysis was performed using the maximal WMI volume as the predictor. In line with the qualitative scale, larger WMI volumes were associated with lower PDI scores at 30 months after adjusting for site, cardiac group, maternal education and BAS; for every mm3 increase in WMI volume, the PDI score was on average 0.06 points lower (95% CI: −0.11, −0.009, p= 0.02).
Central Illustration. The effect of peri-operative brain injury on motor outcome at 30 months of age.
Coefficients (round dots) and corresponding 95% confidence intervals for difference in psychomotor development index (PDI) at 30 months by BIS (brain injury severity) category as compared to BIS = 0 (none to minimal white matter injury) for d-TGA (d- transposition of the great arteries) and SVP (single ventricle physiology) after adjusting for site, maternal education and balloon atrial septostomy. BIS =1 (stroke); BIS=2 (moderate to severe white matter injury).
Table 3.
Mean PDI values for d-TGA and SVP subjects at 30 months by BIS category. The unadjusted and adjusted differences in PDI values for each BIS category are listed categorized by lesion. Subjects with BIS = 1 or BIS =2 were compared to those with BIS = 0 (reference category). At 30 months, d-TGA and SVP subjects with moderate to severe white matter injury (BIS = 2) had significantly lower PDI values as compared to those with none or minimal injury (BIS = 0) after adjusting for variables (listed below).
| 30 months | |||
|---|---|---|---|
| Mean PDI (SE) | Unadjusted Difference (95% CI) | Adjusted Difference* (95% CI) | |
| d-TGA (n= 54) | |||
| BIS† 0 (n= 36) | 92.4 (2.4) | Ref | Ref |
| 1 (n= 13) | 97.4 (2.9) | 5.0 (−3.7,13.8) | 4.9 (−4.5,14.5) |
| 2 (n= 5) | 76.7 (4.6) | −15.8 (−28.6,−2.8)‡ | −14.2 (−27.1,−1.2)‡ |
| SVP (n= 16) | |||
| BIS† 0 (n= 5) | 88 (6.8) | Ref | Ref |
| 1 (n= 6) | 73.3 (6.8) | −14.7 (−34.7,5.4) | −10.2 (−24.6,4.1) |
| 2 (n= 5) | 76.2 (6.0) | −8.4 (−29.4,12.6) | −13.8 (−27.8,0.1)‡ |
PDI= psychomotor development index; d-TGA= d-transposition of the great arteries; SVP= single ventricle physiology; BIS= brain injury severity score.
Adjusted model included the following variables: site, maternal education levels, and balloon atrial septostomy (for d-TGA only).
BIS score categorized as follows: BIS = 0: none or mild white matter injury; BIS = 1: Stroke (any size); BIS= 2: Moderate to severe white matter injury
Denotes significance at p ≤ 0.05.
Table 4.
Multivariable analysis demonstrating association between moderate to severe white matter injury and psychomotor development index and the effect of other adjusted variables at 30 months of age.
| TGA (n= 54) | SVP (n= 16) | |||
|---|---|---|---|---|
| Coefficient | P-value | Coefficient | P-value | |
| BIS* | ||||
| 0 (ref) | - | - | - | - |
| 1 | 5.0 (−4.5, 14.5) | 0.30 | −8.6 (−22.1, 4.8) | 0.18 |
| 2 | −14.2 (−27.1, −1.2) | 0.03 | −12.8 (−25.8, −0.2) | 0.05 |
| Site† | −9.9 (−19.2, −0.7) | 0.03 | −1.2 (−18.5, 16.1) | 0.87 |
| Maternal Education§ | 0.43 (−2.8, 3.6) | 0.78 | 8.2 (4.5, 12.0) | 0.001 |
| BAS | 4.1 (−4.6, 12.7) | 0.35 | - | - |
TGA= transposition of the great arteries; SVP= single ventricle physiology; BIS= brain injury severity score; BAS= balloon atrial septostomy
BIS: 0= none or minimal white matter injury; 1= all stroke; 2= moderate to severe white matter injury
UCSF subjects compared to UBC patients as the reference
Based on a scale from 1–7 from the Hollingshead four-factor index of social status
Given the lack of significant association between brain injury and 12 month ND testing, we examined the concordance of 12 month and 30 month testing. The Bland-Altman limits of agreement for PDI scores at 12 and 30 months were wide (−40.3 to 31.2) across the range of mean PDI values (Pitman’s test of difference in variance, p= 0.27) (Figure 3). To examine whether the limits of agreement were modified by site, we stratified this analysis by site finding no appreciable difference: UCSF limits of agreement −31.0 to 23.6 and UBC limits of agreement −44.5 to 34.6.
Figure 3. Bland-Altman plot of psychomotor development index (PDI) at 12 months and at 30 months by site.
The limits of agreement range from −40.3 (bottom line) to 31.2 (top line) suggesting wide variability between PDI scores at the two time points (the middle line represents no difference) for the entire cohort. Similar limits of agreement were obtained for each site (UCSF & UBC). Pitman’s test of variance (p= 0.28) suggests no difference in agreement across the range of PDI values.
Discussion
In this prospective, longitudinal study we demonstrate an association between brain injury in neonates with critical CHD and neurodevelopmental outcomes at 30 months of age. Specifically, those with moderate to severe WMI score on average one standard deviation below the normative mean for motor development at 30 months. In contrast, we find no significant association at 12 months. Finally, we see no deleterious association between small strokes (< 1/3 of the arterial territory) and outcome. Our results support the utility of neonatal brain MRI for predicting outcome in this high-risk population and underscore the importance of neurodevelopmental follow-up beyond one year of age.
The literature is rich with observational papers demonstrating a high prevalence of pre- and post-operative brain injury and delayed brain development in those with critical CHD (7–10, 22). However, there are few reports of the predictive value of neonatal brain imaging for subsequent ND outcomes in this population. In the largest study to date, Beca et al reported no association between neonatal WMI or combined brain injury and outcomes at 2 years of age in a large cohort of subjects with mixed CHD (11). Their analysis considered brain injury as a dichotomous predictor. Furthermore, very few subjects that had more severe brain injury survived until the ND assessment at 2 years of age, limiting the conclusions regarding different severities of injury and their impact on outcome. In a well characterized cohort of neonates with d-TGA, Andropolous et al demonstrated an association between pre-operative, but not postoperative brain injury and motor and language deficits at 12 months of age (12); however, in a follow-up paper with a larger cohort and varying types of CHD, the same group found only an association with post-operative brain injury and cognitive outcome at 2 years of age (13). Similar to Beca et al, the analysis was focused on injury as a dichotomous predictor, which may mask important associations between more severe types of brain injury and outcome. In our study, brain injury was assessed both with a clinically applicable categorical predictor (BIS score) and quantitative WMI volumes, with similar findings using both approaches. The BIS category prevents comparisons that group different types of brain injury (i.e. WMI vs. stroke) with uninjured subjects. Second, maximal BIS (taking into account both pre- and new post-operative injuries) was assessed to represent the total burden of injury rather than focusing on individual time points. At both 12 and 30 months, approximately 16% of subjects had brain injury on the severe end of the spectrum (i.e. moderate to severe WMI), allowing for an adequate representation of varying degrees of brain injury. Importantly, the relationship between peri-operative brain injury and ND outcome remains significant after adjustment for factors such as maternal education, site and BAS. Although the BIS score allows comparisons across different injury categories (i.e. stroke vs. WMI), it only crudely categorizes injury severity. To address the possibility of misclassification of WMI using this qualitative scale (21), we utilized a quantitative measure of brain injury, namely a volumetric assessment of white matter volume. Using this measurement, which has been found to have high inter-rater reliability (21), we found similar results in that larger WMI volumes were associated with worse PDI scores at 30 months, even after adjusting for the same variables. Given that we measured the maximal WMI volume on either the pre- or post-operative MRI, there is a small possibility of underestimating overall WMI for subjects in which the pre-operative WMI lesion disappears on the post-operative scan. However, this is a rare occurrence based on our prior publication (23).
Studies have shown that WMI is the most frequent pattern of injury in subjects with critical CHD (8,10,23). Various risk factors in the pre-, intra- and post-operative time period contribute to neonatal brain injury (8,24–28). Our findings increase the urgency to identify and minimize modifiable risk factors for WMI. The variable relationship between early and later neurodevelopmental testing may complicate neuroprotective trial design given the long interval between neonatal interventions and meaningful outcomes. However, the present findings demonstrate the predictive value of neonatal imaging for later outcomes and may facilitate design of such clinical trials by using perioperative imaging as a surrogate outcome variable (29).
One of the first associations we reported in this prospective cohort study was a tight relationship between the need for BAS and preoperative brain injury, especially stroke (24) in d-TGA. This relationship was not observed in other cohorts without preoperative stroke (26) or with a very low prevalence of pre-operative stroke (11). Despite the lack of consensus among imaging studies, the association of BAS with stroke was both biologically plausible and concerning. The present findings offer important information regarding this relationship. Consistent with our prior report, the vast majority of strokes (25/29, 86%) were small in size (< 1/3 of arterial territory) and remarkably, we demonstrate that BAS and stroke are associated with better ND outcomes on the univariable analysis. This does not suggest that stroke or BAS are protective per se since these associations were no longer seen on the multivariable analyses, but rather reflects the impact of BAS on cardiovascular physiology. Thus, although the procedure is associated with small embolic strokes, it improves oxygen delivery and hence may contribute to better ND outcomes, though this conjecture warrants further scrutiny with more sophisticated testing based upon stroke location and specific brain functions at later ages. Not surprisingly, larger strokes do seem to result in adverse outcomes, although our sample size for this injury precluded statistical testing.
Previous studies have suggested an association between measures of brain development and outcome in this population. The Total Maturation Score (TMS) at 3 months of age in a mixed cohort of subjects with critical CHD was reported to be with deficits in multiple ND domains at 2 years of age (11). We assessed brain development as measured by diffusion tensor imaging (FA in white matter) and found no association between isolated pre- or post-operative white matter FA values with outcomes at 30 months of age. Although this does not exclude the possibility of a relationship between brain development and ND outcomes in this patient population, it does not appear to impact the relationship between brain injury and outcome.
Neurodevelopmental outcomes for subjects with SVP were systematically lower at both 12 and 30 months as compared to those with d-TGA. Although both subjects with SVP and d-TGA demonstrate a similar prevalence of pre-operative brain injury and delays in brain development, they differ significantly from a physiologic perspective in addition to operative strategies and post-operative management all of which can influence ND outcomes. Recently, we demonstrated slower peri-operative brain growth in patients with hypoplastic left heart syndrome as compared to d-TGA, independent of brain injury severity (30). The distribution of PDI and MDI scores for SVP subjects is consistent with prior literature (3). Although moderate to severe WMI appears to predict even worse outcome for SVP subjects, all patients with SVP warrant frequent and ongoing surveillance for developmental impairments as recommended by current guidelines (31), with additional vigilance for those with moderate to severe WMI; early intervention should be offered to these patients routinely.
Although d-TGA subjects with minimal injury and/or stroke had PDI and MDI scores near the normative mean, these patients have ongoing deficits in childhood and adolescent years that may not be apparent in infancy (4,32). In addition, some studies have suggested that earlier measures of ND are only modestly associated with later measures of ND in childhood for the CHD population (33). Relevant to this concept, we found that PDI scores at 12 months did not reliably predict PDI scores at 30 months, even though the same testing tool was used. This finding has two important implications. First, given the evidence of ongoing developmental deficits in both subjects with SVP and TGA, surveillance should begin in infancy to establish interventions as soon as possible and must continue into teenage and adulthood years. Second, our results suggest that for the purposes of clinical trials in this area, outcome needs to be measured at least at 2.5 years or later.
There are some limitations to our study. There was a high percentage of loss to follow-up either due to death or lack of return to a study visit. Although this may potentially bias the results towards the healthier patients in the cohort (i.e. sicker children died or did not return for follow-up), we found that there was no difference in prevalence of neonatal brain injury between those that remained in the study and those that were lost to follow-up. There was a significant difference in neurodevelopmental outcome at both 12 and 30 months by site. The subjects evaluated at UCSF had significantly lower MDI and PDI scores as compared to the Canadian UBC cohort. Site remained a significant predictor of PDI at 30 months in the multivariable model in subjects with d-TGA. This may be secondary to multiple factors including differences in socioeconomic status, different testing operators, or environmental influences. Prior literature has suggested an attrition bias in the rate of neurodevelopmental impairments that varies by country in preterm-born children (34). In particular, when comparing Canada to the United States, attrition rates were much lower and the rates of neurodevelopmental impairments were lower at 18–24 months in Canada. This may be secondary to different healthcare systems and perhaps easier accessibility to screening and early intervention services in Canada. In our study the rate of follow-up at both 12 months and 30 months was lower in the UCSF cohort as compared to the Canadian cohort, which may have influenced the PDI and MDI scores. Similarly, differences in MRI technique, acquisition and interpretation can exist by site as each site optimized their acquisitions for their local scanner. Given this, we accounted for site in the analyses in both the quantitative and qualitative MRI measures and found that the association between moderate to severe WMI and PDI at 30 months remained even after this adjustment. Finally, maternal education remained a significant predictor of PDI at 30 months in the multivariable model for SVP subjects. Studies have shown that maternal education can predict both short- and long-term health outcomes in offspring (35). Higher maternal education may reflect increased advocacy and receipt of services for children resulting in better outcomes. This finding deserves further study as a potential environmental influence on ND outcome.
Conclusions
In conclusion, moderate to severe WMI is associated with worse motor outcomes at 30 months of age. In contrast, small strokes and the use of BAS are not associated with significant impairment. Although all patients with critical CHD such as d-TGA and SVP are at increased risk of neurodevelopmental impairments throughout the lifespan, extra vigilance is needed for those with moderate to severe WMI. These findings suggest that peri-operative neonatal brain MRI is a useful tool to help further risk stratify patients for neurodevelopmental impairments later in life and that ND follow-up should begin early with continued surveillance.
Supplementary Material
Clinical Perspectives.
Competency in Medical Knowledge
In patients with critical congenital heart disease, clinically silent peri-operative white matter brain injury detected by MRI is associated with neurodevelopmental impairment at 30 months of age.
Translational Outlook
Future studies should focus on the relationship between neonatal brain imaging findings and neurodevelopmental outcomes beyond infancy, in childhood and adolescence.
Acknowledgments
Sources of Funding: This work was supported by grants K23 NS099422, R01 NS40117, R01NS063876, R01EB009756, R01HD07274, and P01 NS082330 from the National Institutes of Health; grant MOP93780 from the Canadian Institutes of Health Research; grant 5-M01-RR-01271 from the National Center for Research Resources; grants 5-FY05-1231 and 6-FY2009-303 from the March of Dimes Foundation; grant 0365018Y from the American Heart Association; and grant 2002/3E from the Larry L. Hillblom Foundation. Dr. Miller is the Bloorview Children’s Hospital Chair in Pediatric Neuroscience.
We would like to thank the neonatal nurses of the Neonatal Clinical Research Center at the University of California at San Francisco and the study team at UBC whose skill and expertise made this study possible.
Abbreviations
- CHD
congenital heart disease
- ND
neurodevelopment
- d-TGA
d- Transposition of the great arteries
- SVP
single ventricle physiology
- WMI
white matter injury
- BIS
Brain Injury Severity
- FA
fractional anisotropy
- PDI
psychomotor development index
- MDI
mental development index
- BAS
balloon atrial septostomy
Footnotes
Disclosures: All authors have no disclosures relevant to this manuscript
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Karamlou T, Diggs BS, Ungerleider RM, Welke KF. Evolution of treatment options and outcomes for hypoplastic left heart syndrome over an 18-year period. J Thorac Cardiovasc Surg. 2010;139:119–26. doi: 10.1016/j.jtcvs.2009.04.061. [DOI] [PubMed] [Google Scholar]
- 2.Karamlou T, Jacobs ML, Pasquali S, et al. Surgeon and Center Volume Influence on Outcomes After Arterial Switch Operation: Analysis of the STS Congenital Heart Surgery Database. Ann Thorac Surg. 2014 doi: 10.1016/j.athoracsur.2014.04.093. [DOI] [PubMed] [Google Scholar]
- 3.Newburger JW, Sleeper LA, Bellinger DC, et al. Early developmental outcome in children with hypoplastic left heart syndrome and related anomalies: the single ventricle reconstruction trial. Circulation. 2012;125:2081–2091. doi: 10.1161/CIRCULATIONAHA.111.064113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marelli A, Miller SP, Marino BS, Jefferson AL, Newburger JW. Brain in Congenital Heart Disease Across the Lifespan: The Cumulative Burden of Injury. Circulation. 2016;133:1951–1962. doi: 10.1161/CIRCULATIONAHA.115.019881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bellinger DC, Wypij D, Rivkin MJ, et al. Adolescents with d-transposition of the great arteries corrected with the arterial switch procedure: neuropsychological assessment and structural brain imaging. Circulation. 2011;124:1361–1369. doi: 10.1161/CIRCULATIONAHA.111.026963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gaynor JW, Stopp C, Wypij D, et al. Neurodevelopmental outcomes after cardiac surgery in infancy. Pediatrics. 2015;135:816–825. doi: 10.1542/peds.2014-3825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Miller SP, McQuillen PS, Hamrick S, et al. Abnormal brain development in newborns with congenital heart disease. N Engl J Med. 2007;357:1928–1938. doi: 10.1056/NEJMoa067393. [DOI] [PubMed] [Google Scholar]
- 8.McQuillen PS, Barkovich AJ, Hamrick SEG, et al. Temporal and anatomic risk profile of brain injury with neonatal repair of congenital heart defects. Stroke. 2007;38:736–741. doi: 10.1161/01.STR.0000247941.41234.90. [DOI] [PubMed] [Google Scholar]
- 9.Licht DJ, Shera DM, Clancy RR, et al. Brain maturation is delayed in infants with complex congenital heart defects. J Thorac Cardiovasc Surg. 2009;137:529–36. doi: 10.1016/j.jtcvs.2008.10.025. discussion 536–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Licht DJ, Wang J, Silvestre DW, et al. Preoperative cerebral blood flow is diminished in neonates with severe congenital heart defects. J Thorac Cardiovasc Surg. 2004;128:841–849. doi: 10.1016/j.jtcvs.2004.07.022. [DOI] [PubMed] [Google Scholar]
- 11.Beca J, Gunn JK, Coleman L, et al. New white matter brain injury after infant heart surgery is associated with diagnostic group and the use of circulatory arrest. Circulation. 2013;127:971–979. doi: 10.1161/CIRCULATIONAHA.112.001089. [DOI] [PubMed] [Google Scholar]
- 12.Andropoulos DB, Easley RB, Brady K, et al. Changing expectations for neurological outcomes after the neonatal arterial switch operation. Ann Thorac Surg. 2012;94:1250–5. doi: 10.1016/j.athoracsur.2012.04.050. discussion 1255–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Andropoulos DB, Ahmad HB, Haq T, et al. The association between brain injury, perioperative anesthetic exposure, and 12-month neurodevelopmental outcomes after neonatal cardiac surgery: a retrospective cohort study. Pediatric Anesthesia. 2014;24:266–274. doi: 10.1111/pan.12350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goff DA, Luan X, Gerdes M, et al. Younger gestational age is associated with worse neurodevelopmental outcomes after cardiac surgery in infancy. J Thorac Cardiovasc Surg. 2012;143:535–542. doi: 10.1016/j.jtcvs.2011.11.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Calderon J, Angeard N, Moutier S, Plumet M-H, Jambaqué I, Bonnet D. Impact of prenatal diagnosis on neurocognitive outcomes in children with transposition of the great arteries. The Journal of Pediatrics. 2012;161:94–8. doi: 10.1016/j.jpeds.2011.12.036. [DOI] [PubMed] [Google Scholar]
- 16.Gaynor JW, Wernovsky G, Jarvik GP, et al. Patient characteristics are important determinants of neurodevelopmental outcome at one year of age after neonatal and infant cardiac surgery. J Thorac Cardiovasc Surg. 2007;133:1344–53. 1353.e1–3. doi: 10.1016/j.jtcvs.2006.10.087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. The Journal of Pediatrics. 1978;92:529–534. doi: 10.1016/s0022-3476(78)80282-0. [DOI] [PubMed] [Google Scholar]
- 18.Dimitropoulos A, McQuillen PS, Sethi V, et al. Brain injury and development in newborns with critical congenital heart disease. Neurology. 2013;81:241–248. doi: 10.1212/WNL.0b013e31829bfdcf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guo T, Duerden EG, Adams E, et al. Quantitative assessment of white matter injury in preterm neonates: Association with outcomes. Neurology. 2017;88:614–622. doi: 10.1212/WNL.0000000000003606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hüppi PS, Dubois J. Diffusion tensor imaging of brain development. Semin Fetal Neonatal Med. 2006;11:489–497. doi: 10.1016/j.siny.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 21.McCarthy AL, Winters ME, Busch DR, et al. Scoring system for periventricular leukomalacia in infants with congenital heart disease. Pediatr Res. 2015;78:304–309. doi: 10.1038/pr.2015.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Miller SP, McQuillen PS, Vigneron DB, et al. Preoperative brain injury in newborns with transposition of the great arteries. Ann Thorac Surg. 2004;77:1698–1706. doi: 10.1016/j.athoracsur.2003.10.084. [DOI] [PubMed] [Google Scholar]
- 23.Block AJ, McQuillen PS, Chau V, et al. Clinically silent preoperative brain injuries do not worsen with surgery in neonates with congenital heart disease. J Thorac Cardiovasc Surg. 2010;140:550–557. doi: 10.1016/j.jtcvs.2010.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.McQuillen PS, Hamrick SEG, Perez MJ, et al. Balloon atrial septostomy is associated with preoperative stroke in neonates with transposition of the great arteries. Circulation. 2006;113:280–285. doi: 10.1161/CIRCULATIONAHA.105.566752. [DOI] [PubMed] [Google Scholar]
- 25.Goff DA, Shera DM, Tang S, et al. Risk factors for preoperative periventricular leukomalacia in term neonates with hypoplastic left heart syndrome are patient related. J Thorac Cardiovasc Surg. 2013 doi: 10.1016/j.jtcvs.2013.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Petit CJ, Rome JJ, Wernovsky G, et al. Preoperative brain injury in transposition of the great arteries is associated with oxygenation and time to surgery, not balloon atrial septostomy. Circulation. 2009;119:709–716. doi: 10.1161/CIRCULATIONAHA.107.760819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Andropoulos DB, Hunter JV, Nelson DP, et al. Brain immaturity is associated with brain injury before and after neonatal cardiac surgery with high-flow bypass and cerebral oxygenation monitoring. J Thorac Cardiovasc Surg. 2010;139:543–556. doi: 10.1016/j.jtcvs.2009.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Peyvandi S, De Santiago V, Chakkarapani E, et al. Association of Prenatal Diagnosis of Critical Congenital Heart Disease With Postnatal Brain Development and the Risk of Brain Injury. JAMA Pediatr. 2016 doi: 10.1001/jamapediatrics.2015.4450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sherlock RL, McQuillen PS, Miller SP. aCCENT Preventing brain injury in newborns with congenital heart disease: brain imaging and innovative trial designs. Stroke. 2009;40:327–332. doi: 10.1161/STROKEAHA.108.522664. [DOI] [PubMed] [Google Scholar]
- 30.Peyvandi S, Kim H, Lau J, et al. The association between cardiac physiology, acquired brain injury, and postnatal brain growth in critical congenital heart disease. J Thorac Cardiovasc Surg. 2017 doi: 10.1016/j.jtcvs.2017.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Marino BS, Lipkin PH, Newburger JW, et al. Neurodevelopmental outcomes in children with congenital heart disease: evaluation and management: a scientific statement from the American Heart Association. Circulation. 2012;126:1143–1172. doi: 10.1161/CIR.0b013e318265ee8a. [DOI] [PubMed] [Google Scholar]
- 32.Rollins CK, Watson CG, Asaro LA, et al. White matter microstructure and cognition in adolescents with congenital heart disease. The Journal of Pediatrics. 2014;165:936–44. e1–2. doi: 10.1016/j.jpeds.2014.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McGrath E, Wypij D, Rappaport LA, Newburger JW, Bellinger DC. Prediction of IQ and achievement at age 8 years from neurodevelopmental status at age 1 year in children with D-transposition of the great arteries. Pediatrics. 2004;114:e572–6. doi: 10.1542/peds.2003-0983-L. [DOI] [PubMed] [Google Scholar]
- 34.Guillén U, DeMauro S, Ma L, et al. Relationship between attrition and neurodevelopmental impairment rates in extremely preterm infants at 18 to 24 months: a systematic review. Arch Pediatr Adolesc Med. 2012;166:178–184. doi: 10.1001/archpediatrics.2011.616. [DOI] [PubMed] [Google Scholar]
- 35.Braveman PA, Cubbin C, Egerter S, et al. Socioeconomic status in health research: one size does not fit all. JAMA. 2005;294:2879–2888. doi: 10.1001/jama.294.22.2879. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.