Abstract
Objective
Conflicting data exist regarding the impact of in utero exposure to maternal combination antiretrovirals. We compared neurodevelopmental outcomes between HIV-exposed/uninfected (HEU) children exposed in utero to 3-drug combination antiretroviral therapy (ART) versus zidovudine (ZDV) monotherapy.
Design
Prospective study of child neurodevelopment, nested within two cohorts of HIV-infected mothers and their children in Botswana (one observational, one interventional).
Methods
The Tshipidi and Mma Bana studies enrolled HIV-infected women during pregnancy and followed their HEU children for 24 months. Mothers took 3-drug ART or ZDV during pregnancy. ART-exposed babies were mostly breastfed, and ZDV-exposed were formula-fed. Neurodevelopmental outcomes, measured at 24 months using Bayley Scales of Infant and Toddler Development Third Edition (Bayley-III) and Development Milestones Checklist (DMC), were compared in adjusted linear regression according to antiretroviral exposure.
Results
Of 598 HEU children with valid neurodevelopment assessments, 382 were ART-exposed and 210 were ZDV-exposed. Adjusted mean Bayley-III scores were similar among ART-exposed versus ZDV-exposed, with adjusted mean differences (95% CI): Bayley-III Cognitive: −0.3 (−1.4, 0.9); Gross Motor: 0.8 (−0.1, 1.7); Fine Motor: 0.5 (−0.2, 1.3); Expressive Language: 0.7 (−0.3, 1.7); Receptive Language: 0.1 (−0.7, 0.8); and DMC Locomotor: 0.0 (−0.5, 0.6); Fine Motor: 0.3 (−0.3, 0.8); Language: −0.1 (−0.5, 0.4); Personal-Social: 0.2 (−0.7, 1.1). Similarly, when restricted to formula-fed children in one cohort (Tshipidi), there were no differences in adjusted mean scores.
Conclusions
Neurodevelopmental outcomes at 24 months of age were similar in ART-exposed versus ZDV-exposed HEU children. Maternal ART with breastfeeding does not appear to have an adverse effect on neurodevelopment.
Keywords: HIV-exposed/uninfected, antiretroviral therapy, pregnancy, breastfeeding, children, neurodevelopment, Botswana
Introduction
Globally, an increasing number of children are exposed in utero to maternal 3-drug antiretroviral therapy (ART) [1]. Despite marked reductions in perinatal HIV transmission with maternal ART, important concerns remain regarding the impact of in utero antiretroviral (ARV) exposure on child health and neurodevelopment [2–4]. Exploration of potential effects of in utero ART exposure on neurodevelopment in HIV-uninfected children born to HIV-infected mothers (HIV exposed-uninfected [HEU] children) remains an area of special interest [1–4].
The southern African nation of Botswana has the second highest HIV-1 prevalence in the world, with a severe, generalized HIV epidemic affecting 22% of adults aged 15–49 years [5]. HIV infection is predominantly transmitted heterosexually in Botswana, affects all socio-economic strata, and is not particularly associated with alcohol or substance abuse [6, 7]. Botswana emerged as a regional leader in the provision of mother to child HIV transmission (MTCT) prevention services and ART to its citizens [8]. Starting in 2012, the CD4 threshold for ART initiation was raised from ≤200 cells/mm3 to ≤350 cells/mm3, and universal ART in pregnancy regardless of CD4 count (PMTCT "Option B") was offered [9].
The benefits of antiretroviral medicines to prevent mother to child transmission (MTCT) of HIV outweigh their potential for harm [10]. However, data regarding the effects of in utero ARV exposure on child neurodevelopment have been somewhat conflicting. Prior analyses from populations in developed countries have been fraught with challenges in providing adequate control for potential confounders associated with both maternal HIV infection/ARV use and child neurodevelopment [11–17]. Limited data capture neurodevelopment in HEU children in resource-limited settings [18–24], particularly in Africa, the region most affected by HIV [19–24]. We sought to compare neurodevelopment in HEU children who were exposed in utero to 3-drug ART versus zidovudine (ZDV) monotherapy, in the context of the generalized HIV epidemic in Botswana. Worse neurodevelopmental outcomes in 3-drug ART-exposed children would warrant investigation of safer ART regimens, while similar neurodevelopmental outcomes provide appropriate reassurance regarding the ongoing widespread use of 3-drug ART in pregnancy.
Methods
Study design and populations
We conducted a combined analysis of two prospective cohort studies in Botswana (the Tshipidi and Mma Bana studies) in which neurodevelopmental assessments were conducted at 24 months of age among HEU children. In these two studies, children were exposed in utero to ART or to ZDV monotherapy for at least two weeks prior to delivery. ART is defined here as receipt of three or more antiretroviral drugs simultaneously.
The Tshipidi study was an observational study which enrolled consenting HIV-infected women and HIV-uninfected women (all Botswana citizens age 18 years or older) during pregnancy or within 1 week of delivery between May 2010 and February 2012 at two sites in Botswana: Gaborone (the capital city) and Mochudi (a nearby village). Only HIV-infected women and their HIV-uninfected children were included in this analysis. Mothers and infants were followed for 24 months postpartum, and received routine clinical care at Government facilities (details of study visits/evaluations are below). As Tshipidi was an observational study, maternal antiretroviral regimens were consistent with Botswana government guidelines in use at that time of enrollment [9]. Mothers were promptly evaluated and referred for treatment if they were not already receiving appropriate antiretrovirals at the time of recruitment. Mothers chose feeding methods with counseling per Botswana guidelines [9]. HIV-infected mothers who chose replacement feeding received free formula. HIV-exposed children generally received a single dose of nevirapine and one month of ZDV. At study entry, maternal demographic, socioeconomic, and health predictors were collected, including maternal CD4 count and HIV-1 RNA. Infant evaluations included HIV-1 DNA PCR at birth, 1, 6, 12 months and HIV-1 ELISA at 18 months postpartum. At each interval (and at 24 months), infant height, weight, head circumference, clinical history, medication history, feeding history and physical examination were documented.
The Mma Bana study was a randomized MTCT prevention trial, which enrolled pregnant HIV-infected women between July 2006 and May 2008 in four sites in Botswana (the same two as the Tshipidi study, plus the village of Molepolole and the town of Lobatse. The Mma Bana trial design and primary results have been previously published [27]. As part of the current study we added neurodevelopmental testing of children at 24 months of age to the Mma Bana trial cohort (thus only a subset of Mma Bana participants had the opportunity to take part in this sub-study of neurodevelopment). In Mma Bana, HIV-infected mothers with CD4 counts of ≥200cells/mm3 were randomized to receive either Trizivir (abacavir/ZDV/ lamivudine– “triple-nucleoside reverse-transcriptase inhibitor [NRTI] group”) or Kaletra/Combivir (lopinavir-ritonavir/ZDV/lamivudine – “protease-inhibitor [PI] group”), taken during pregnancy through ≥6 months post-partum (during the period of breastfeeding). Women with baseline CD4 <200 cells/mm3 received nevirapine, zidovudine and lamivudine antepartum and indefinitely postpartum (the “observational group”) [27]. Mma Bana participants were counseled to exclusively breastfeed and to wean at 6 months. Infants received a single dose of nevirapine and one month of zidovudine. Very similar baseline factors and follow-up evaluations were conducted in the Mma Bana study as within Tshipidi, following a similar visit schedule.
HIV tests and CD4+ cell counts were performed at the approved Botswana Harvard HIV Reference Laboratory in Gaborone. For the Mma Bana and Tshipidi studies, maternal HIV infection was confirmed during the identified pregnancy or within 7 days of delivery [25]. The first positive HIV test consisted of a positive rapid HIV test or licensed ELISA test result that was performed by trained staff. The first documented positive result was confirmed by a second positive HIV test result obtained by trained study staff at the clinic or reference laboratory (by rapid HIV test, any licensed ELISA test kit, a Western blot, or detectable HIV-1 RNA). Discordant confirmatory results were followed by plasma HIV-1 RNA. DNA PCR ascertained child HIV status at birth, 4–6 weeks of age, and after the cessation of breastfeeding. Positive DNA PCR tests were confirmed with a second DNA PCR, and further confirmed by ELISA at 18 months of age. HIV-infected children, of which there were only very few, were excluded from this analysis, as their neurodevelopment profiles were expected to be markedly different from that of HEU children.
All mothers provided written informed consent for study participation. The Botswana Health Research Development Committee and the Harvard TH Chan School of Public Health, Office of Human Research Administration IRBs approved all protocols.
Neurodevelopmental assessment
Neurodevelopmental outcomes were measured for all children at approximately two years of age (up to 30 months of age) using an adapted version of the Bayley Scales of Infant and Toddler Development Third Edition (Bayley-III). Bayley-III scores were recorded for each child for Cognitive, Receptive Language, Expressive Language, Fine Motor and Gross Motor domains [26]. The neurodevelopmental assessments were translated into Setswana and back translated. All instruments were reviewed by local personnel, and modifications or adaptations were made as needed. All tests were then piloted. The Bayley-III required further modifications and was re-piloted. Trained study personnel, research nurses, administered the Bayley-III, the DMC and all study questionnaires. All study questionnaires were completed by direct interview. The Social-Emotional and Adaptive Behavior sections of the Bayley-III were not administered, as they were not culturally appropriate; these were replaced with parent report measures of typical behaviors of the child in the home setting, including the Development Milestones Checklist (DMC), which was developed and normed in Kenya [27–28]. Data were entered into standardized CRFs at the study sites. An invalid score was assigned to children who were unable to complete neurodevelopmental testing or whose physical or behavioral problems were suggestive of potential clinical impairment. Invalid scores were noted by clinical assessors and confirmed by the lead neuropsychologist (BK). Assessor performance was monitored periodically through video and/or direct observation by study coordinators and lead neuropsychologist (BK). Reliability was checked by observation of testing tapes and monitoring by the study coordinator.
Statistical analyses
The primary comparison of interest in this analysis was between neurodevelopmental outcomes in HEU children exposed in utero either to ART (from either study) or ZDV (from the Tshipidi study only), to determine potential risk of either ARV exposure.
The primary outcomes of interest were the 24-month Bayley-III subscale raw scores (Cognitive, Gross Motor, Fine Motor, Expressive Language and Receptive Language) and the DMC subscale scores (Loco-motor, Fine motor, Language and Personal-Social). Unadjusted and adjusted linear regression models compared differences in HEU child mean neurodevelopment exposed to ART or ZDV. Cohen’s D standardized effect sizes for mean differences were reported [29]. Only valid assessments were included in primary analyses. Infants with an invalid score for a specific neurodevelopmental domain were excluded from analyses for that outcome, but included in any analyses of outcomes for which they had valid scores. Demographic and maternal health characteristics were summarized and compared according to cohort (Mma Bana and Tshipidi) and ARV exposure, using means and standard deviations, or medians for non-normally distributed characteristics, and t tests or ANOVA tests for categorical measures.
We assessed differences in baseline characteristics between those who did and did not complete a neurodevelopment assessment. We also compared baseline characteristics for those with invalid or low scores (1 SD below the domain-specific mean) versus higher valid scores. Univariate and multivariate logistic regression models were used to assess predictors of a composite adverse outcome (either a low score or an invalid score).
Potential confounders were identified using a priori knowledge of predictors of poor neurodevelopmental outcomes from other studies. Directed acyclic graphs were used to delineate assumptions regarding the causal pathway of interest. Potential confounders included maternal health-related factors (such as antenatal HIV-1 RNA, maternal CD4 counts, and maternal age), socio-economic factors (including education, income, toilet facilities, and availability of electricity and water in the household), and the year of neurodevelopmental testing. Further measures within the Tshipidi cohort included food security, type of cooking method employed within the household, and maternal depression, alcohol and substance use. All characteristics with unadjusted p<0.20 for association with a specific neurodevelopmental domain score were initially included in adjusted models. Covariates with p>0.20 in multivariate models were subsequently excluded.
Around 12% of eligible study participants had missing results for one or more Bayley-III domains (Supplemental Table 2), with less than <0.5% having invalid test results. Restricting our analysis to individuals with complete test results could introduce bias if exposure to ART were associated with completing the Bayley-III and neurodevelopment differed between those who completed the assessment and those who did not. To adjust for this potential selection bias, we computed inverse probability of censoring weights (IPCW). Each child who completed the domain of interest received a weight inversely proportional to the estimated probability of not being censored (i.e. completing the Bayley-III). Weights were computed using logistic regression models including ART exposure and other baseline covariates. The weights were stabilized and used in sensitivity analyses evaluating ART exposure and Bayley- III domain scores, adjusted for the same covariates.
Low birth-weight (<2500g) and preterm birth (<37 weeks of gestation) were considered to be potential mediators, as they have been identified in prior literature to be on the causal pathway between ARV exposure and neurodevelopment [30, 31]. Primary models excluded these factors, but in sensitivity analyses, models were further adjusted for these birth characteristics to evaluate the impact on findings.
Nearly all children in Mma Bana were both ART-exposed and breastfed, while nearly all children in Tshipidi were formula-fed. Therefore, the reference group for the primary comparison of interest was predominantly formula-fed (i.e. the reference group being formed by the ZDV-exposed children enrolled in Tshipidi). Decisions regarding feeding were generally made prior to exposure (in accordance with study recruitment of breastfeeding mothers within Mma Bana [25] or national policy recommendations regarding formula feeding during Tshipidi [9]). To address the potential effect of feeding strategy, we restricted a sub-analysis to only formula-fed children within Tshipidi. This approach also accounted for differences in conditions under which the two studies were conducted. Finally, the Tshipidi-only analysis also offered opportunity to control for a wider array of potential confounders (such as maternal depression and food insecurity) for which data were collected in only this study.
Results
Enrollment and neurodevelopmental testing completeness
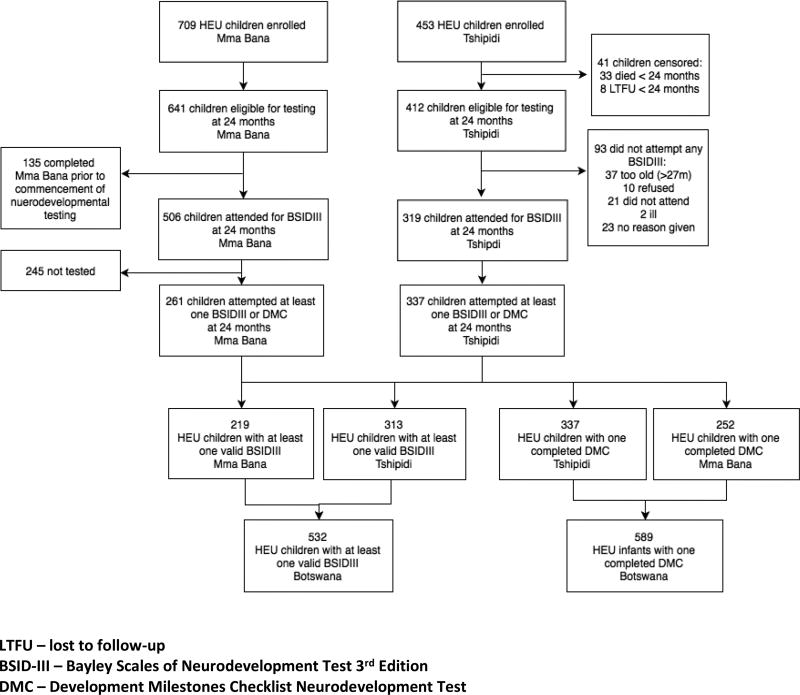
Among the 910 live-born children enrolled in Tshipidi, 453 were HEU. Of these, 412 children were alive and attended visits at 24 months, of which 313 completed at least one valid Bayley-III test and the parents of 357 completed the DMC (Figure 1). Among 709 live-born infants enrolled in the two randomized arms and observational cohort of the Mma Bana study, 219 completed at least one valid Bayley-III domain and 252 had a completed DMC at two years. The majority of Mma Bana participants were not approached for testing, as they had already exceeded the age for neurodevelopmental evaluation by the time this sub-study was initiated. Additionally, limitations on study staff time at the onset of the neurodevelopmental assessments meant that many eligible children were not tested (n=334, Supplemental Table 1). Among infants eligible but not tested, more were premature (22% vs. 10%), of low birthweight (24% vs. 16%) and had mothers of a younger age (Supplemental Table 1). Among children with at least 1 valid neurodevelopment test result, children who were missing one or more of the other neurodevelopment outcomes were more likely to have been formula-fed (Supplemental Table 2). A total of 532 children completed at least one valid Bayley-III domain and 589 completed at least one DMC domain at approximately 24 months of age; 598 HEU infants had at least one valid Bayley-III or one DMC domain score (Figure 1).
Figure 1.
Flow diagram of HIV-Exposed/Uninfected (HEU) children undergoing neurodevelopment testing in the Tshipidi and Mma Bana studies
Maternal and infant clinical and socio-demographic characteristics of these 598 HEU participants are summarized in Table 1, according to ART exposure status and study cohort. The majority (65%) of HEU children were ART-exposed, with 68% of the ART-exposed children enrolled in Mma Bana and 32% in Tshipidi. All ZDV-exposed HEU children were in the Tshipidi study. Almost all of children in Mma Bana were both breastfed and ART-exposed, whereas most (90%) Tshipidi infants were formula-fed. Entry median viral load was higher among mothers in Mma Bana than in Tshipidi (likely because many women enrolling in Tshipidi were taking antiretrovirals at enrollment, while Mma Bana participants were not). Entry median maternal CD4 count was lower among Mma Bana mothers than in Tshipidi. Mothers were similar with regards to age, education and income across the cohorts and according to ART exposure.
Table 1.
Baseline characteristics of HEU children and mothers by in utero ART exposure and study cohort (Mma Bana and Tshipidi) with at least one valid Bayley-III or at least one valid DMC outcome, Botswana 2009 – 2012
| ARV exposure | Study cohort | Overall | |||
|---|---|---|---|---|---|
| Characteristic | ZDV only (N=210) (N, % total) |
3-drug ART (N=382) (N, % total) |
Mma Bana (N=261) (N, % total) |
Tshipidi (N=331) (N, % total) |
All (N=592) (N, % total) |
| Study cohort | |||||
| Mma Bana | --- | 261 (68.3%) | 261 (100%) | --- | 261 (44.1%) |
| Tshipidi | 210 (100%) | 121 (31.7%) | --- | 331 (100%) | 331 (55.9%) |
| Testing site | |||||
| Gaborone | 121 (57.6%) | 340 (89.0%) | 110 (42.1%) | 203 (61.3%) | 461 (77.8%) |
| Mochudi | 89 (42.4%) | 42 (11.0%) | 151 (57.9%) | 134 (40.4%) | 131 (22.3%) |
| ARV exposure* | |||||
| ZDV | 210 (100%) | --- | --- | 210 (63.4%) | 210 (35.5%) |
| 3-drug ART | --- | 382 (100%) | 261 (100%) | 121 (36.6%) | 382 (64.5%) |
| ARV started during pregnancy | 210 (100%) | 319 (83.5%) | 261 (100%) | 267 (80.1%) | 528 (89.2%) |
| Gestational age at ARV start** | 28.4 (27.7–29.6) | 26.7 (26.1–29.3) | 26.7 (26.1–29) | 28.4 (27.4–29.7) | 27.9 (26.3–29.4) |
| Duration of ARV during pregnancy*** | 11.0 (8.9–12.3) | 12.3 (9.7–14.3) | 12.1 (9.9–13.9) | 11.3 (8.7–13.0) | 11.7 (9.4–13.4) |
| ART + feeding strategy**** | |||||
| ART + BF | --- | 273 (71.5%) | 260 (99.6%) | 13 (3.9%) | 273 (46.1%) |
| ART + FF | --- | 109 (28.5%) | 1 (0.4%) | 108 (32.6%) | 109 (18.4%) |
| ZDV + BF | 17 (8.1%) | --- | --- | 17 (5.1%) | 17 (2.9%) |
| ZDV + FF | 193 (91.9%) | --- | --- | 193 (58.3%) | 193 (3.3%) |
| Breast-fed | 17 (8.1%) | 273 (71.5%) | 260 (99.6%) | 30 (9.1%) | 290 (49.0%) |
| Female | 102 (48.6%) | 198 (51.8%) | 136 (52.1%) | 168 (50.1%) | 304 (51.4%) |
| Preterm birth | 26 (12.4%) | 29 (7.7%) | 12 (4.6%) | 46 (12.1%) | 58 (9.8%) |
| Low birthweight (<2,500g) | 24 (11.4%) | 71 (18.5%) | 38 (14.5%) | 58 (17.5%) | 96 (16.2%) |
| Maternal viral load no/mL† | 4786 | 5710 | 15400 | 1445 | 5481 |
| <=400 | 36 (17.2%) | 112 (29.6%) | 25 (9.6%) | 124 (37.2%) | 149 (25.2%) |
| >400–999 | 23 (11.0%) | 20 (5.3%) | 14 (5.4%) | 29 (8.7%) | 43 (7.3%) |
| >1000–9999 | 72 (34.5%) | 79 (20.8%) | 73 (28.0%) | 79 (23.7%) | 150 (25.3%) |
| >=10,000 | 78 (37.3%) | 168 (44.3%) | 149 (57.1%) | 101 (30.3%) | 250 (42.2%) |
| Maternal CD4 cells/mm3† | 449 | 362 | 343 | 429 | 394 |
| <200 | 45 (21.4%) | 113 (29.8%) | 84 (32.4%) | 74 (22.4%) | 158 (26.8%) |
| >=200–500 | 80 (38.1%) | 113 (29.8%) | 69 (26.6%) | 124 (37.6%) | 193 (32.8%) |
| >=500 | 78 (37.1%) | 90 (23.8%) | 58 (22.4%) | 111 (33.3%) | 168 (28.5%) |
| Maternal age | |||||
| 18–24 | 72 (34.3%) | 98 (25.7%) | 83 (31.8%) | 87 (26.3%) | 170 (28.7%) |
| 25–29 | 65 (31.0%) | 107 (28.0%) | 76 (29.1%) | 96 (29.0%) | 172 (29.1%) |
| 30–34 | 42 (20.0%) | 86 (22.5%) | 54 (20.7%) | 74 (22.4%) | 128 (21.6%) |
| 35–39 | 27 (12.9%) | 79 (20.7%) | 41 (15.7%) | 65 (19.6%) | 106 (17.9%) |
| 40–44 | 4 (1.9%) | 12 (3.1%) | 7 (2.7%) | 9 (2.7%) | 16 (2.7%) |
| Maternal education | |||||
| None | 5 (2.4%) | 8 (2.1%) | 6 (2.3%) | 7 (2.1%) | 13 (2.2%) |
| Primary | 27 (12.9%) | 72 (18.9%) | 56 (21.5%) | 43 (12.8%) | 99 (16.8%) |
| Secondary junior | 127 (60.8%) | 217 (56.8%) | 143 (54.8%) | 201 (60.9%) | 344 (58.2%) |
| Secondary senior | 50 (23.9%) | 85 (22.3%) | 56 (21.5%) | 79 (23.9%) | 135 (22.8%) |
| Maternal income | |||||
| None | 108 (51.9%) | 197 (51.7%) | 122 (46.9%) | 183 (55.6%) | 305 (51.8%) |
| <P500 | 16 (7.7%) | 76 (20.0%) | 66 (25.4%) | 26 (7.9%) | 92 (15.6%) |
| P501-P1000 | 48 (23.1%) | 57 (15.0%) | 41 (15.8%) | 64 (19.5%) | 105 (17.8%) |
| >P1000 | 36 (17.3%) | 51 (13.4%) | 31 (11.9%) | 56 (17.0%) | 87 (14.8%) |
| Access to electricity | 101 (48.3%) | 169 (44.2%) | 108 (41.4%) | 164 (48.8%) | 272 (45.6%) |
| Type of toilet facility | |||||
| Indoor toilet | 29 (15.1%) | 21 (19.4%) | --- | 52 (17.0%) | --- |
| Private pit latrine | 126 (65.6%) | 78 (72.2%) | --- | 208 (68.0%) | --- |
| Shared pit latrine | 37 (19.3%) | 9 (8.3%) | --- | 46 (15.0%) | --- |
| Access to water | |||||
| In home | 29 (15.1%) | 18 (16.7%) | --- | 49 (16.0%) | --- |
| Standpipe in yard | 133 (69.3%) | 73 (67.6%) | --- | 209 (68.3%) | --- |
| Communal | 27 (14.1%) | 13 (12.0%) | --- | 41 (13.4%) | --- |
| Other | 3 (1.6%) | 4 (3.7%) | --- | 7 (2.3%) | --- |
| Cooking method | |||||
| Gas or electric | 134 (69.8%) | 73 (67.6%) | --- | 212 (69.3%) | --- |
| Wood | 49 (25.5%) | 33 (30.6%) | --- | 83 (27.1%) | --- |
| Kerosene | 5 (2.6%) | 1 (0.9%) | --- | 6 (2.0%) | --- |
| None | 4 (2.1%) | 1 (0.9%) | --- | 5 (1.6%) | --- |
| Housing | |||||
| Formal | 189 (98.4%) | 106 (98.2%) | --- | 301 (98.4%) | --- |
| Informal | 3 (1.6%) | 1 (0.9%) | --- | 4 (1.3%) | --- |
| None | 0 (0%) | 1 (0.9%) | --- | 1 (0.3%) | --- |
| 1Food insecurity | |||||
| None | 58 (30.1%) | 29 (26.9%) | --- | 88 (28.7%) | --- |
| Mild | 40 (20.7%) | 19 (17.6%) | --- | 62 (20.2%) | --- |
| Moderate | 55 (28.5%) | 31 (28.7%) | --- | 86 (28.0%) | --- |
| Severe | 40 (20.7%) | 29 (26.9%) | --- | 71 (23.1%) | --- |
| Highest score | |||||
| 2Depression | 3.1 (2.8) | 2.4 (2.6) | --- | 2.7 (2.8) | --- |
| 3Social support | 32.0 (6.3) | 32.2 (8.2) | --- | 32.1 (7.0) | --- |
| 4Stigma | 2.9 (2.4) | 2.3 (2.1) | --- | 2.7 (2.3) | --- |
| Alcohol use‡ | 17 (8.8%) | 4 (3.7%) | --- | 24 (7.1%) | --- |
| Tobacco use‡ | 4 (2.1%) | 0 (0%) | --- | 5 (1.5%) | --- |
| Year of testing | |||||
| 2009 | --- | 75 (19.6%) | 75 (28.7%) | --- | 75 (12.7%) |
| 2010 | --- | 186 (48.7%) | 186 (71.3%) | --- | 186 (31.4%) |
| 2012 | 66 (31.4%) | 8 (2.1%) | --- | 74 (22.4%) | 74 (12.5%) |
| 2013 | 126 (60.0%) | 54 (14.1%) | --- | 180 (54.4%) | 180 (30.4%) |
| 2014 | 18 (8.6%) | 59 (15.5%) | --- | 77 (23.3%) | 77 (13.0%) |
ARV exposure: ART–3-drug antiretroviral therapy or ZDV- Zidovudine.
Median (IQR) gestational age in weeks at ARV start (among those women commencing ARV during current pregnancy).
Median (IQR) duration of ARV in weeks (among those women commencing ARV during current pregnancy).
Feeding strategy: BF-Breastfed FF-Formula-fed. Percentage of participants missing values for: Breastfeeding 0% Prematurity 1.2% Birthweight 0% Maternal viral load 0.7% Maternal CD4 count 12% Maternal education 0.2% Maternal income 0.5% Access to electricity 0.2% Access to sanitation 0.4% Source of water 0.1% Cooking method 0.1% Housing 0.1% Food insecurity 0.1% Depression 0% Social support 5.5% Alcohol use <1% Tobacco use <1%.
1Food insecurity as measured by an adapted Household Food Insecurity Access Scale (34). Means reported reflect highest value during study follow-up prior to neurodevelopmental testing for each mother e.g. Mean score using highest score on the 2Beck Depression Scale (35) (i.e. worse depression). Mean score using highest score for a given mother’s assessment of her 3social support (36) (i.e. worse support). 3Mean score using the highest score for a given mother’s assessment of her perceived experience of 4stigma (37) (i.e. worse stigma).
Any alcohol use or tobacco use during current pregnancy
Neurodevelopmental outcomes among HEU children participating in both the Mma Bana and Tshipidi cohorts (by ARV exposure group, 3-drug ART vs. ZDV only)
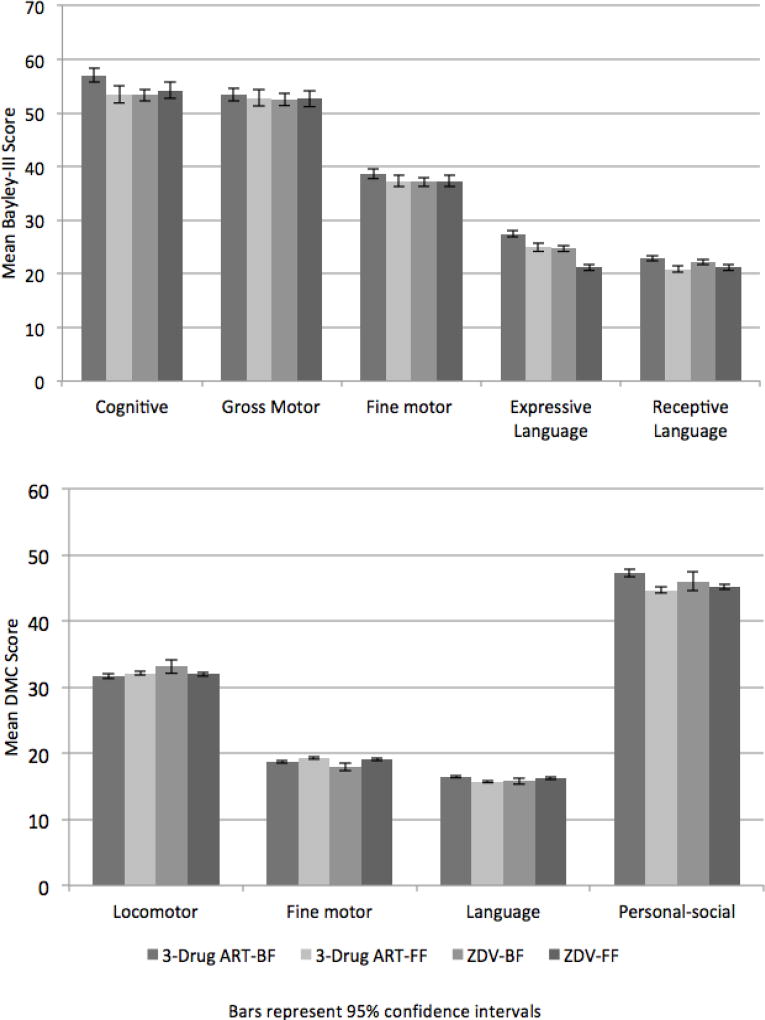
Figures 2a and 2b show crude unadjusted mean Bayley-III and DMC scores by antiretroviral exposure- and feeding-groups. Each neurodevelopment domain was significantly associated with at least one previously described clinical, socioeconomic or environmental predictor (Supplemental Tables 3–6). Crude unadjusted Bayley-III means scores were highest for all domains in the ART-exposed breastfed infants (Figure 2a). This finding was not, however, seen with the DMC crude mean scores (Figure 2b). Mean differences in Bayley-III and DMC outcomes between ART-exposed and ZDV-exposed children are summarized in Table 2, both with and without adjustment for calendar year and other confounders. ART-exposed children had similar adjusted neurodevelopment scores across all domains of the Bayley-III and DMC scale. When accounting for missing assessments using IP weighted regression, results were similar to those shown in Table 2 (data not shown).
Figure 2.
Unadjusted mean Bayley-III and DMC neurodevelopment scores at age 24 months according to ART + feeding strategy among 598 HEU children from the Mma Bana and Tshipidi studies, Botswana 2009–2012
Table 2.
Crude means, unadjusted and adjusted mean differences, and effect sizes in Bayley-III and DMC neurodevelopment domain scores at age 24 months for HEU children in the Tshipidi and Mma Bana studies, Botswana 2009 – 2012
| Neurodevelopment assessment |
N | Mean scores (SD) | Unadjusted difference | Adjusted difference | Effect-size | |||
|---|---|---|---|---|---|---|---|---|
| ART | ZDV | (95% CI) | P-value | (95% CI) | P-value | Cohen’s d (95% CI) | ||
| Bayley-III domain | ||||||||
|
| ||||||||
| Cognitive | 524 | 55.8 (6.9) | 54.2 (3.4) | 1.6 (0.6, 2.7) | <0.01 | −0.3 (−1.4, 0.9) | 0.64 | −0.05 (−0.55, 0.46) |
| Gross Motor | 519 | 53.2 (5.7) | 52.6 (2.9) | 0.6 (−0.3, 1.5) | 0.19 | 0.8 (−0.1, 1.7) | 0.08 | 0.12 (−0.30, 0.54) |
| Fine Motor | 531 | 38.2 (4.4) | 37.3 (1.8) | 0.9 (0.3, 1.6) | <0.01 | 0.5 (−0.2, 1.3) | 0.16 | 0.15 (−0.16, 0.46) |
| Expressive Language | 517 | 26.6 (5.4) | 25.0 (4.5) | 1.6 (0.7, 2.5) | <0.01 | 0.7 (−0.3, 1.7) | 0.19 | 0.14 (−0.31, 0.58) |
| Receptive Language | 518 | 22.2 (3.7) | 21.2 (3.8) | 1.0 (0.3, 1.7) | <0.01 | 0.1 (−0.7, 0.8) | 0.88 | 0.02 (−0.31, 0.34) |
|
| ||||||||
| DMC domain | ||||||||
|
| ||||||||
| Locomotor | 593 | 31.8 (2.9) | 32.1 (1.9) | −0.3 (−0.8, 0.1) | 0.15 | 0.0 (−0.5, 0.6) | 0.86 | 0.00 (−0.21, 0.21) |
| Fine Motor | 590 | 18.9 (2.5) | 18.9 (2.4) | 0.0 (−0.4, 0.4) | 0.95 | 0.3 (−0.3, 0.8) | 0.32 | 0.10 (−0.10, 0.30) |
| Language | 593 | 16.4 (3.0) | 16.5 (2.5) | −0.1 (−0.6, 0.3) | 0.53 | −0.1 (−0.5, 0.4) | 0.82 | −0.05 (−0.28, 0.17) |
| Personal-Social | 590 | 46.5 (5.1) | 45.3 (3.2) | 1.2 (0.4, 2.0) | <0.01 | 0.2 (−0.7, 1.1) | 0.69 | 0.04 (−0.33, 0.41) |
Bayley-III: Cognitive adjusted for year of neurodevelopmental testing. Gross Motor adjusted for maternal income, Fine Motor adjusted for maternal CD4 count and year of neurodevelopmental testing, Expressive Language adjusted for maternal education and year of neurodevelopmental testing, Receptive Language adjusted for year of neurodevelopmental testing. DMC: Locomotor adjusted for maternal income and year of neurodevelopmental testing, Fine Motor adjusted for maternal CD4, maternal age and year of neurodevelopmental testing, language adjusted for maternal CD4 count and maternal education, Personal-Social adjusted for year of neurodevelopmental testing.
Neurodevelopmental outcomes among formula-fed HEU children who participated in the Tshipidi cohort by ARV exposure group (3-drug ART vs. ZDV only)
This secondary analysis was performed to evaluate the potential effect of 3-drug ART vs. ZDV on neurodevelopment, independent of feeding-method or study cohort. In addition, more extensive baseline characteristics were collected within Tshipidi than was possible for Mma Bana (Supplemental Tables 3–6). Similar to our main analysis, no significant differences were observed among those exposed to ART when compared to ZDV monotherapy across all domains of the Bayley-III and DMC scale (Table 3). Sensitivity analyses additionally adjusting for birthweight and preterm birth produced similar findings to the above results (Supplemental Tables 7–8). Similar results were observed when applying IP weighted regression to account for eligible participants without assessments (data not shown).
Table 3.
Crude means, unadjusted and adjusted mean differences, and effect sizes in Bayley-III and DMC neurodevelopment domain scores at age 24 months, restricted to only HEU children in Tshipidi cohort who were formula-fed, Botswana 2009 – 2012
| Neurodevelopment assessment |
N | Mean scores (SD) | Unadjusted difference | Adjusted difference | Effect-size | |||
|---|---|---|---|---|---|---|---|---|
| ART | ZDV | (95% CI) | P-value | (95% CI) | P-value | Cohen’s d (95% CI) | ||
| Bayley-III domain | ||||||||
|
| ||||||||
| Cognitive | 282 | 53.0 (3.1) | 54.2 (3.3) | −1.2 (−2.0, −0.4) | <0.01 | −0.2 (−1.1, 0.7) | 0.66 | −0.38 (−0.76, 0.02) |
| Gross Motor | 278 | 52.8 (2.6) | 52.6 (2.8) | 0.2 (−0.5, 0.9) | 0.60 | 0.0 (−0.7, 0.7) | 0.95 | 0.07 (−0.26, 0.39) |
| Fine Motor | 282 | 37.3 (1.8) | 37.3 (1.8) | 0.0 (−0.5, 0.4) | 0.94 | −0.1 (−0.6, 0.4) | 0.55 | −0.01 (−0.22, 0.20) |
| Expressive Language | 279 | 24.9 (4.1) | 25.0 (4.5) | −0.1 (−1.2, 0.9) | 0.80 | −0.2 (−1.3, 0.9) | 0.71 | −0.03 (−0.55, 0.48) |
| Receptive Language | 278 | 20.8 (3.0) | 21.2 (3.8) | −0.4 (−1.2, 0.5) | 0.41 | 0.2 (−0.8, 1.2) | 0.70 | 0.06 (−0.36, 0.48) |
|
| ||||||||
| DMC Domain | ||||||||
|
| ||||||||
| Locomotor | 307 | 32.1 (1.7) | 32.0 (2.0) | 0.1 (−0.4, 0.5) | 0.36 | 0.1 (−0.3, 0.6) | 0.66 | 0.04 (−0.17, 0.25) |
| Fine Motor | 306 | 19.3 (2.0) | 19.0 (2.4) | 0.3 (−0.2, 0.8) | 0.29 | 0.0 (−0.6, 0.6) | 0.88 | 0.02 (−0.23, 0.27) |
| Language | 307 | 16.2 (2.9) | 16.5 (2.5) | −0.3 (−0.9, 0.3) | 0.37 | −0.6 (−1.3, 0.1) | 0.10 | −0.24 (−0.53, 0.06) |
| Personal-Social | 305 | 44.7 (5.1) | 45.2 (3.3) | −0.6 (−1.5, 0.4) | 0.26 | −0.7 (−1.6, 0.3) | 0.16 | −0.14 (−0.59, 0.32) |
Bayley-III: Cognitive adjusted for maternal education, depression and year of neurodevelopmental testing. Gross Motor adjusted for source of water and social score, Fine Motor adjusted for maternal education, access to sanitation and social support, Expressive Language is adjusted for maternal education and income, Receptive Language is adjusted for stigma and year of neurodevelopmental testing. DMC: Locomotor adjusted for maternal income and social support, Fine Motor adjusted for maternal education, use of alcohol during pregnancy and year of neurodevelopmental testing, Language adjusted for maternal education and year of neurodevelopmental testing. Personal-Social adjusted for depression.
Sensitivity analysis: predictors of composite adverse neurodevelopmental outcome (low or invalid score)
Across the Bayley-III tests, 5.4% to 17.3% of the domain-specific outcomes were considered to have an adverse outcome (see Supplemental Table 9). Among those infants who had an adverse Bayley-III neurodevelopment outcome, more were of low birthweight (27% vs. 15%) and formula fed (68% vs. 53%) for the Cognitive domain, more were premature for the Gross Motor (19% vs. 9%) and Fine Motor (17% vs. 9%) domains, and more were formula fed (71% vs. 51%), with lower maternal education and income for the Expressive Language domain and higher proportions formula-fed (74% vs. 50%) or premature (16% vs. 8%) for the Receptive Language domain (Supplemental Table 9). In multivariable logistic regression, low birthweight was associated with adverse Bayley-III cognitive outcomes and prematurity with adverse Bayley-III Gross Motor, Fine Motor and Receptive Language outcomes. Breastfeeding showed protective associations against composite adverse outcomes for Bayley-III Cognitive, Expressive and Receptive Language domains. Additionally, lack of maternal income was associated with composite adverse outcomes for Bayley-III Cognitive and Receptive Language domains (Supplemental Tables 10–11).
Discussion
Neurodevelopmental outcomes among 24-month-old HEU children who were exposed in utero to 3-drug ART versus ZDV monotherapy were similar across all domains/subscales tested, including after controlling for potential confounders such as maternal viral load, CD4 count and age, education and income. Findings provide reassurance that HEU child neurodevelopment, at two years of age, is not adversely affected by in utero combination ART exposure [13, 15]. Furthermore, maternal ART during pregnancy while breastfeeding is likely to lead to 24-month neurodevelopmental outcomes comparable to those of formula-fed children whose mothers took ZDV monotherapy. The sub-analysis within the Tshipidi cohort within formula-fed children only, further affirmed that ART-exposure in-utero was not associated with any neurodevelopmental deficit.
The strengths of our study include the relatively high participation rate among eligible cohort members, large sample size, collection of data on a broad range of potential confounders relevant to sub-Saharan Africa, and context of a generalized epidemic where HIV-infected mothers were more representative of the general population, when compared with Western epidemics. The study had 80% power to detect a difference of 0.27–0.38 standard deviations between mean scores of children exposed in utero to ART vs. ZDV, with sufficient power to detect a range of neurodevelopmental differences.
Our study also has several limitations. Children not tested differed from those who were tested (Supplemental Table 9), according to prematurity and birthweight. Sensitivity analyses incorporating prematurity and birthweight did not alter adjusted estimates. Our secondary analysis restricted to formula-fed children in Tshipidi only, yielded very similar results. Approximately 85% of the ART-exposed children’s mothers were ART-naïve at baseline. Given the small proportion (around 15%) of prevalent users we did not expect prevalent user bias to significantly affect our results. The majority of women commenced ART during pregnancy, between 18 and 34 weeks’ gestation and had a similar duration of use to the comparison group (median 12.3 weeks of ART or 11 weeks of ZDV). Little is known regarding the exact timing and duration of the critical window of exposure, however outstanding concerns of potential toxicity, following any in utero exposure to ARV medicines remain. This study was underpowered to further discern differences between trimester of exposure and outcomes.
Our study tested children at two years of age and found no differences according to ARV exposure, however findings cannot be generalized to older children. A small number of studies have followed children into later ages to negate negative neurodevelopmental outcomes [12, 32]. ART-exposed children in our study were exposed to abacavir/ZDV/lamivudine or lopinavir/ritonavir/ZDV/lamivudine within Mma Bana, and a range of ART regimens within Tshipidi (ZDV/lamivudine or tenofovir disoproxil/emtricitabine, in combination with nevirapine, or lopinavir/ritonavir, or atazanavir/ritonavir). Sirois et al (2013) reported on the neurodevelopment of 374 infants with Bayley-III evaluations in the Pediatric HIV/AIDS Cohort Study (PHACS) Surveillance Monitoring for Antiretroviral Therapy Toxicities (SMARTT), a US based multisite cohort study, at a median age of 12.7 months, and found no association with overall ART, specific ARV regimen, or ARV prophylaxis exposure in utero. In this study, the adjusted mean score for language was within age-expected ranges but significantly lower for infants exposed to atazanavir in utero [11], confirmed in a follow-up analysis within SMARTT by Caniglia et al (2016) [33].
Studies within resource-limited settings have not examined the potential effects of in utero exposure to maternal antiretroviral use on neurodevelopment among HEU children [18–24], particularly within Africa. Given the large number of ARV-exposed HEU infants that continue to be born globally, and the evolving treatment and landscape, surveillance of potential effects through standardized approaches, will be of continued importance, to examine differing ARV exposures, across a range of ages and neurophysiological functions [38].
Conclusions
HEU child neurodevelopment at 24 months of age does not differ according to exposure in utero to 3-drug ART versus ZDV alone. Our data provide additional support that benefits of 3-drug ART use during pregnancy, particularly when combined with breastfeeding, outweigh potential risks for HEU children.
Supplementary Material
Acknowledgments
S.C. performed the analyses in this manuscript and was the primary author of the manuscript. P.W. was the senior statistician and co-investigator on the Tshipidi study. J.L. provided data management and statistical support for the conduct and analysis of the Tshipidi study. G.M. provided oversight to the conduct of the Tshipidi study, and conducted some of the participant assessments. S.L and B.K. were co-principal investigators of the Tshipidi study. S.L. was co-investigator of the Mma Bana Study. R.S. was the principal investigator of the Mma Bana study. G.S., with expertise in pediatric HIV observational studies, assisted with interpretation of findings and manuscript drafting. All authors reviewed and approved the final version of this manuscript.
We would also like to acknowledge Matt McKenna for assisting with statistical programming. We would like to thank Penny Holding for designing the neurodevelopmental tests and both Penny Holding and Vicki Tepper for conducting the training for administration of the testing. We are grateful to the women and children who participated in the study. We would like to acknowledge all the Tshipidi and Mma Bana study staff in Botswana.
Funding: The Tshipidi study was supported by a grant from the National Institute of Mental Health (RO1MH087344). The Mma Bana study was supported by a grant from the National Institute of Allergy and Infectious Diseases (U01-AI066454). This work was also supported by Oak Foundation.
Footnotes
The authors declare that they have no conflicts of interest.
References
- 1.Filteau S. The HIV-exposed, uninfected African child. Trop Med Int Health. 2009;14:276–287. doi: 10.1111/j.1365-3156.2009.02220.x. [DOI] [PubMed] [Google Scholar]
- 2.Brogly SB, Ylitalo N, Mofenson LM, Oleske J, Van Dyke R, Crain MJ, et al. In utero nucleoside reverse transcriptase inhibitor exposure and signs of possible mitochondrial dysfunction in HIV-uninfected children. Aids. 2007;21:929–938. doi: 10.1097/QAD.0b013e3280d5a786. [DOI] [PubMed] [Google Scholar]
- 3.Le Doare K, Bland R, Newell ML. Neurodevelopment in children born to HIV-infected mothers by infection and treatment status. Pediatrics. 2012;130:e1326–1344. doi: 10.1542/peds.2012-0405. [DOI] [PubMed] [Google Scholar]
- 4.Heidari S, Mofenson L, Cotton MF, Marlink R, Cahn P, Katabira E. Antiretroviral drugs for preventing mother-to-child transmission of HIV: a review of potential effects on HIV-exposed but uninfected children. J Acquir Immune Defic Syndr. 2011;57:290–296. doi: 10.1097/QAI.0b013e318221c56a. [DOI] [PubMed] [Google Scholar]
- 5.UNAIDS. HIV and AIDS estimates. 2015 http://www.unaids.org/en/regionscountries/countries/botswana.
- 6.Wojcicki JM. Socioeconomic status as a risk factor for HIV infection in women in East, Central and Southern Africa: a systematic review. J Biosoc Sci. 2005;37:1–36. doi: 10.1017/s0021932004006534. [DOI] [PubMed] [Google Scholar]
- 7.Weiser SD, Leiter K, Heisler M, et al. A population-based study on alcohol and high-risk sexual behaviors in Botswana. PLoS Med. 2006;3:e392. doi: 10.1371/journal.pmed.0030392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Molomo BC. National operational plan for scaling up HIV prevention in Botswana: 2008–2010 [Google Scholar]
- 9.Ministry of Health. Republic of Bostwana. Botswana national HIV and AIDS treatment guidelines 2012. [Google Scholar]
- 10.Ciaranello AL, Seage GR, 3rd, Freedberg KA, Weinstein MC, Lockman S, Walensky RP. Antiretroviral drugs for preventing mother-to-child transmission of HIV in sub-Saharan Africa: balancing efficacy and infant toxicity. Aids. 2008;22:2359–69. doi: 10.1097/QAD.0b013e3283189bd7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sirois PA, Huo Y, Williams PL, Malee K, Garvie PA, Kammerer B, et al. Safety of perinatal exposure to antiretroviral medications: developmental outcomes in infants. Pediatr Infect Dis J. 2013;32:648–655. doi: 10.1097/INF.0b013e318284129a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nozyce ML, Huo Y, Williams PL, Kapetanovic S, Hazra R, Nichols S, et al. Safety of in utero and neonatal antiretroviral exposure: cognitive and academic outcomes in HIV-exposed, uninfected children 5–13 years of age. Pediatr Infect Dis J. 2014;33:1128–1133. doi: 10.1097/INF.0000000000000410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lindsey JC, Malee KM, Brouwers P, Hughes MD. Neurodevelopmental functioning in HIV-infected infants and young children before and after the introduction of protease inhibitor-based highly active antiretroviral therapy. Pediatrics. 2007;119:e681–693. doi: 10.1542/peds.2006-1145. [DOI] [PubMed] [Google Scholar]
- 14.Alimenti A, Forbes JC, Oberlander TF, Money DM, Grunau RE, Papsdorf MP, et al. A prospective controlled study of neurodevelopment in HIV-uninfected children exposed to combination antiretroviral drugs in pregnancy. Pediatrics. 2006;118:e1139–1145. doi: 10.1542/peds.2006-0525. [DOI] [PubMed] [Google Scholar]
- 15.Williams PL, Marino M, Malee K, Brogly S, Hughes MD, Mofenson LM. Neurodevelopment and in utero antiretroviral exposure of HIV-exposed uninfected infants. Pediatrics. 2010;125:e250–260. doi: 10.1542/peds.2009-1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Griner R, Williams PL, Read JS, Seage GR, 3rd, Crain M, Yogev R, et al. In utero and postnatal exposure to antiretrovirals among HIV-exposed but uninfected children in the United States. AIDS Patient Care STDS. 2011;25:385–394. doi: 10.1089/apc.2011.0068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Garvie PA, Zeldow B, Malee K, Nichols SL, Smith RA, Wilkins ML, et al. Discordance of cognitive and academic achievement outcomes in youth with perinatal HIV exposure. Pediatr Infect Dis J. 2014;33:e232–238. doi: 10.1097/INF.0000000000000314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kerr SJ, Puthanakit T, Vibol U, Aurpibul L, Vonthanak S, Kosalaraksa P, et al. Neurodevelopmental outcomes in HIV-exposed-uninfected children versus those not exposed to HIV. AIDS Care. 2014;26:1327–1335. doi: 10.1080/09540121.2014.920949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Msellati P, Lepage P, Hitimana DG, Van Goethem C, Van de Perre P, Dabis F. Neurodevelopmental testing of children born to human immunodeficiency virus type 1 seropositive and seronegative mothers: a prospective cohort study in Kigali, Rwanda. Pediatrics. 1993;92:843–8. [PubMed] [Google Scholar]
- 20.Abubakar A, Van Baar A, Van de Vijver FJ, Holding P, Newton CR. Paediatric HIV and neurodevelopment in sub-Saharan Africa: a systematic review. Trop Med Int Health. 2008;13:880–7. doi: 10.1111/j.1365-3156.2008.02079.x. [DOI] [PubMed] [Google Scholar]
- 21.Boivin MJ, Green SD, Davies AG, Giordani B, Mokili JK, Cutting WA. A preliminary evaluation of the cognitive and motor effects of pediatric HIV infection in Zairian children. Health Psychol. 1995;14:13–21. doi: 10.1037//0278-6133.14.1.13. [DOI] [PubMed] [Google Scholar]
- 22.Drotar D, Olness K, Wiznitzer M, et al. Neurodevelopmental outcomes of Ugandan infants with human immunodeficiency virus type 1 infection. Pediatrics. 1997;100:E5. doi: 10.1542/peds.100.1.e5. [DOI] [PubMed] [Google Scholar]
- 23.Van Rie A, Mupuala A, Dow A. Impact of the HIV/AIDS epidemic on the neurodevelopment of preschool-aged children in Kinshasa, Democratic Republic of the Congo. Pediatrics. 2008;122:e123–8. doi: 10.1542/peds.2007-2558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kandawasvika GQ, Ogundipe E, Gumbo FZ, Kurewa EN, Mapingure MP, Stray-Pedersen B. Neurodevelopmental impairment among infants born to mothers infected with human immunodeficiency virus and uninfected mothers from three peri-urban primary care clinics in Harare, Zimbabwe. Dev Med Child Neurol. 2011;53:1046–1052. doi: 10.1111/j.1469-8749.2011.04126.x. [DOI] [PubMed] [Google Scholar]
- 25.Shapiro R, Hughes M, Ogwu A, Kitch D, Lockman S, Moffat C, et al. Antiretroviral regimens in pregnancy and breast-feeding in Botswana. New England Journal of Medicine. 2010;362:2282–2294. doi: 10.1056/NEJMoa0907736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bayley N. Technical Manual. 3. Harcourt Assessment; San Antonio, TX: 2006. Bayley scales of infant and Toddler development. [Google Scholar]
- 27.Abubakar A, Holding P, van de Vijver FJ, Bomu G, Van Baar A. Developmental monitoring using caregiver reports in a resource-limited setting: the case of Kilifi, Kenya. Acta Paediatr. 2010;99:291–7. doi: 10.1111/j.1651-2227.2009.01561.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Prado EL, Abubakar AA, Abbeddou S, Jimenez EY, Some JW, Ouedraogo JB. Extending the Developmental Milestones Checklist for use in a different context in Sub-Saharan Africa. Acta paediatrica. 2014;103:447–54. doi: 10.1111/apa.12540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cohen J. Statistical Power Analysis for the Behavioral Sciences. Hillsdale, NJ: Lawrence Erlbaum Associates; 1988. [Google Scholar]
- 30.Vohr BR, Wright LL, Poole WK, McDonald SA. Neurodevelopmental outcomes of extremely low birth weight infants <32 weeks' gestation between 1993 and 1998. Pediatrics. 2005;116:635–643. doi: 10.1542/peds.2004-2247. [DOI] [PubMed] [Google Scholar]
- 31.Blencowe H, Lee AC, Cousens S, Bahalim A, Narwal R, Zhong N, et al. Preterm birth-associated neurodevelopmental impairment estimates at regional and global levels for 2010. Pediatr Res. 2013;74(Suppl 1):17–34. doi: 10.1038/pr.2013.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Smith R, Chernoff M, Williams PL, Malee KM, Sirois PA, Kammerer B, Wilkins M, Nichols S, Mellins C, Usitalo A, Garvie P, Rutstein R Pediatric HIV/AIDS Cohort Study Team. Impact of HIV severity on cognitive and adaptive functioning during childhood and adolescence. Pediatr Infect Dis J. 2012;31:592–8. doi: 10.1097/INF.0b013e318253844b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Caniglia EC, Patel K, Huo Y, Williams PL, Kapetanovic S, Rich KC, et al. Atazanavir exposure in utero and neurodevelopment in infants: a comparative safety study. Aids. 2016;30:1267–1278. doi: 10.1097/QAD.0000000000001052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jennifer CJ, Swindale A, Bilinsky P. Household Food Insecurity Access Scale (HFIAS) for Measurement of Household Food Access: Indicator Guide (v. 3) Washington, D.C: Food and Nutrition Technical Assistance Project, Academy for Educational Development; 2007. [Google Scholar]
- 35.Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J. An inventory for measuring depression. Archives of General Psychiatry. 1961;4:561–571. doi: 10.1001/archpsyc.1961.01710120031004. [DOI] [PubMed] [Google Scholar]
- 36.Broadhead WE, Gehlbach SH, de Gruy FV, Kaplan BH. The Duke-UNC Functional Social Support Questionnaire. Measurement of social support in family medicine patients. Med Care. 1988;26(7):709–23. doi: 10.1097/00005650-198807000-00006. [DOI] [PubMed] [Google Scholar]
- 37.Berger BE, Ferrans CE, Lashley FR. Measuring stigma in people with HIV: psychometric assessment of the HIV stigma scale. Res Nurs Health. 2001;24:518–529. doi: 10.1002/nur.10011. [DOI] [PubMed] [Google Scholar]
- 38.Zash RM, Williams PL, Sibiude J, Lyall H, Kakkar F. Surveillance monitoring for safety of in utero antiretroviral therapy exposures: current strategies and challenges. Expert Opin Drug Safety. 2016;15(11):1501–13. doi: 10.1080/14740338.2016.1226281. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.