Abstract
[17-14C]-Labeled GA15, GA24, GA25, GA7, and 2,3-dehydro-GA9 were separately injected into normal, dwarf-1 (d1), and dwarf-5 (d5) seedlings of maize (Zea mays L.). Purified radioactive metabolites from the plant tissues were identified by full-scan gas chromatography-mass spectrometry and Kovats retention index data. The metabolites from GA15 were GA44, GA19, GA20, GA113, and GA15-15,16-ene (artifact?). GA24 was metabolized to GA19, GA20, and GA17. The metabolites from GA25 were GA17, GA25 16α,17-H2-17-OH, and HO-GA25 (hydroxyl position not determined). GA7 was metabolized to GA30, GA3, isoGA3 (artifact?), and trace amounts of GA7-diene-diacid (artifact?). 2,3-Dehydro-GA9 was metabolized to GA5, GA7 (trace amounts), 2,3-dehydro-GA10 (artifact?), GA31, and GA62. Our results provide additional in vivo evidence of a metabolic grid in maize (i.e. pathway convergence). The grid connects members of a putative, non-early 3,13-hydroxylation branch pathway to the corresponding members of the previously documented early 13-hydroxylation branch pathway. The inability to detect the sequence GA12 → GA15 → GA24 → GA9 indicates that the non-early 3,13-hydroxylation pathway probably plays a minor role in the origin of bioactive gibberellins in maize.
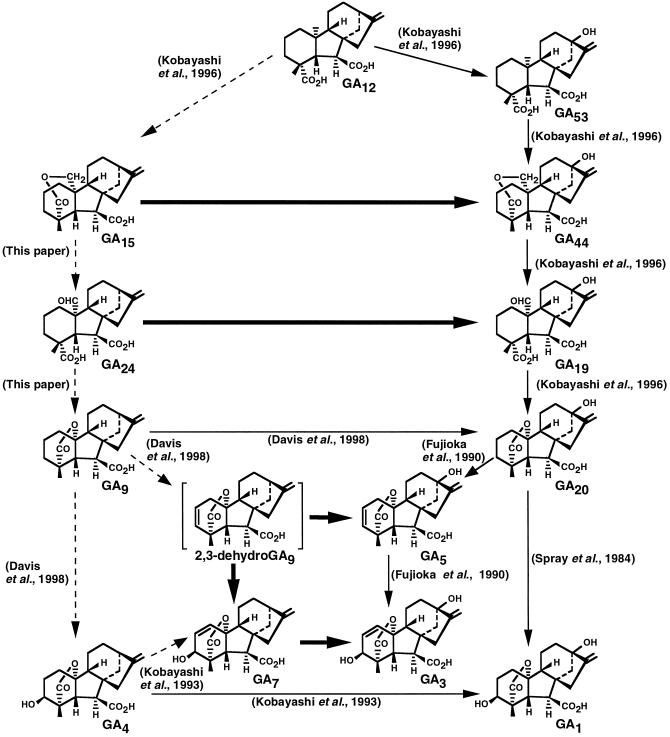
The biosynthesis of the gibberellins (GAs) has been recently reviewed (MacMillan, 1997). In all systems studied, the pathway has been shown to proceed from the cyclic diterpene ent-kaurene to GA12 aldehyde then to GA12. Depending on the sequence of hydroxylation at the 3β- and 13-positions, parallel pathways branch from GA12 to the C19-GAs, the number of these branch pathways varying from species to species. For maize (Zea mays L.) we previously demonstrated (see Fig. 1) the presence of the early 13-hydroxylation branch pathway, a pathway originating from GA12 and leading to the hydroxylated C19-GAs, GA1, GA3, and GA5 (Kobayashi et al., 1996 and refs. therein; Spray et al., 1996). As shown in Figure 1, the steps from GA12 to bioactive GA1, GA3, and GA5, the early 13-hydroxylation branch pathway, have been established by feeding studies using labeled substrates; the immediate metabolites were identified by full-scan gas chromatography-mass spectrometry (GC-MS) and Kovats retention index (KRI) data (Fujioka et al., 1990; Kobayashi et al., 1996). All members of this branch pathway are native to maize (Fujioka et al., 1988a, 1988b).
Figure 1.
Maize branch pathways from GA12: right vertical row, the early 13-hydroxylation branch pathway; left vertical row, the presumptive non-early 3,13-hydroxylation branch pathway. All of the GAs are endogenous to maize except 2,3-dehydroGA9, shown in brackets. ➞, Steps established in this paper; →, steps previously established; - - ➛, steps tested, not observed.
There is indirect evidence for the presence of a second pathway from GA12, the non-early 3,13-hydroxylation branch pathway. The pathway originates from GA12 and leads via GA9 to the 3β-hydroxylated C19-GAs GA4, and GA7 (see Fig. 1). While the pathway has been shown to be present in a number of plant species (for review, see MacMillan, 1997), its presence in maize is based solely on the identification from maize of the five members GA15, GA24, GA9, GA4, and GA7. Moreover, in vivo feeding studies have provided no evidence for the metabolism of GA12 to GA15 (Kobayashi et al., 1996), GA9 to GA4 (Davis et al., 1998), and GA4 to GA7 (Kobayashi et al., 1993).
In the present study, we describe the metabolism of [17-14C]GA15, [17-14C]GA24, [17-14C]GA25, and [17-14C]GA7 in seedlings of tall, dwarf-1 (d1), and dwarf-5 (d5) maize. Given the previous demonstration of the sequence GA20 → GA5 (2, 3-dehydro-GA20) → GA3 in maize (Fujioka et al., 1990), the possible existence of a parallel sequence of GA9 → 2,3-dehydro-GA9 → GA7 was tested by feeding 2,3-dehydro-[17-14C]GA9, a GA not reported to be present in maize (Fujioka et al., 1988b). The data obtained, together with the previous results from the metabolism of [17-13C,3H]GA9 and [17-13C,3H]GA4, are discussed in terms of the biosynthesis of GAs in maize.
MATERIALS AND METHODS
Plant Material
Normal (tall), dwarf-1 (d1), and dwarf-5 (d5) maize (Zea mays L.) seedlings came from seed stocks of known genotype (Spray et al., 1996). The seeds were pre-soaked in water for 12 h and planted in vermiculite:soil (1:1). The seedlings were then grown in the greenhouse at the University of California, Los Angeles. Three- to four-week-old seedlings (three- to four-leaf stage) were used for feeds.
Labeled Substrates
[17-14C]GA15 (2.07 TBq mol−1), [17-14C]GA24 (2.07 TBq mol−1), and [17-14C]GA7 (2.07 TBq mol−1) were purchased from Prof. L.N. Mander (Australian National University, Canberra). [17-14C]GA25 (2.07 TBq mol−1) was prepared from [17-14C]GA24 (300 kBq; a gift from Prof. L.N. Mander) with cell lysates (3.5 mL) from Escherichia coli NM522 containing clone E5 by methods detailed by Lange (1997) and purified as described by Lange and Graebe (1993). 2,3-Dehydro-[17-14C]GA9 (1.75 TBq mol−1) was prepared as described in MacMillan et al. (1997).
Treatment, Purification, and Analysis
Each of the five labeled GAs, [17-14C]GA15, [17-14C]GA24, [17-14C]GA25, [17-14C]GA7, and 2,3-dehydro-[17-14C]GA9, was dissolved in 90 μL of ethanol:water (1:1). Two microliters of the [17-14C]GA15 solution (1,570 Bq; 250 ng) were individually injected into the coleoptile nodes of three sets of 10 plants (normal, d1, and d5). Similar injections were made with [17-14C]GA24 (1,490 Bq; 250 ng), [17-14C]GA25 (1,420 Bq; 250 ng), and [17-14C]GA7 (1,550 Bq; 250 ng). One set of 10 d5 seedlings was used for the 2,3-dehydro-[17-14C]GA9 injections (485 Bq; 88 ng).
The seedlings were incubated in the greenhouse for 24 h, harvested as sets of 10, frozen with dry ice, and stored at −80°C. Each set of frozen seedlings was homogenized, extracted, and solvent-fractionated to give an acidic ethyl acetate-soluble (AE) fraction. Each fraction was concentrated and purified using Bond Elut (Varian, Harbor City, CA) columns and two steps of HPLC (Davis et al., 1998). All samples were methylated and the GAs in each sample were identifed by full-scan GC-MS and KRI (Gaskin and MacMillan, 1991; Spray et al., 1996).
Isotopic Dilution
To determine whether 2,3-dehydro-GA9 is endogenous to maize, [17-14C]2,3-dehydro-GA9 (1.75 TBq mol−1) was used in an isotopic dilution experiment. Fifteen nanograms (3 Bq) was dissolved in 100 μL of 50% (v/v) aqueous ethanol and added to a homogenate from 50 normal maize seedlings (200 g fresh weight). The homogenate was extracted immediately and solvent fractionated to give an AE fraction. The fraction was processed for the determination of isotopic dilution using the isotope dilution fit program described by Croker et al. (1994).
RESULTS AND DISCUSSION
Metabolism [17-14C]GA15
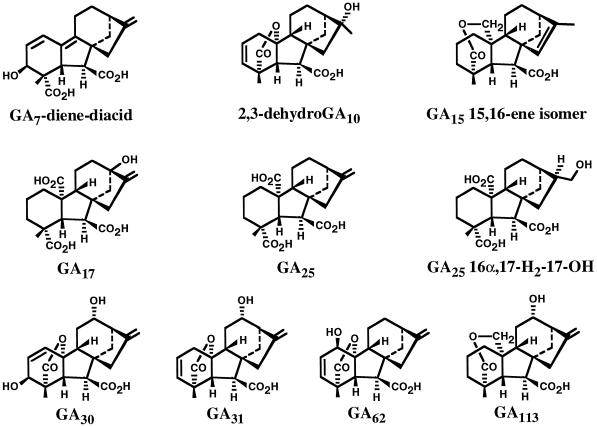
The recovered [14C]labeled metabolites GA44, GA19, GA20, GA113, and GA15-15,16-ene (artifact?) are shown in Table I, and are based on identification by the full-scan GC-MS and KRI data presented in Table II. The step from GA15 to GA44 (Fig. 1) is a direct 13-hydroxylation that is new for maize. The observed 13-hydroxylation of GA15 to GA44 in maize (Fig. 1) has also been reported in a cell-free preparation from germinating barley (Grosslindemann et al., 1992). In addition, the opened lactone of GA15 is metabolized to GA44 from in vitro studies using seeds of pea (Kamiya and Graebe, 1983) and bean (Takahashi et al., 1986). The steps GA44 → GA19 and GA19 → GA20 have been previously demonstrated in maize seedlings (Kobayashi et al., 1996). The step from GA15 to GA113 (Fig. 2) is a direct 12α-hydroxylation, which is new for maize and for higher plants. GA113 has not been found to occur naturally in maize but has been recently isolated from the seeds and shoots of the Japanese radish (Nakayama et al., 1998). The relatively high levels of endogenous GA44 and GA19 present in the normal and d1 seedlings compared with the d5 seedlings (Fujioka et al., 1988a) may create feedback inhibition and thus account for the absence of the labeled metabolite GA19, in the normal and d1 seedlings, in contrast to the recovery of [14C]GA19 from d5 seedlings.
Table I.
Analysis of metabolites from feeds of [17-14C]GA15 (250 ng, 1.57 × 103 Bq each) to normal, d1, and d5 seedlings of maize
| Plant Material | ODS-HPLC Fraction | N(CH3)2-HPLC Fraction | Radioactivity | [14C]Producta |
|---|---|---|---|---|
| Bq | ||||
| Normal (12.0 g) | 18–21 | 31–34 | 128 | GA113 |
| 22–25 | 31–34 | 49 | GA44 | |
| 30–34 | 26–29 | 34 | GA15-15,16-ene | |
| 30–34 | 30–33 | 628 | GA15 (feed) | |
| d1 (7.2 g) | 22–25 | 31–32 | 109 | GA44 |
| 30–34 | 28–31 | 452 | GA15 (feed) | |
| d5 (7.0 g) | 18–21 | 31–34 | 228 | GA20, GA113 |
| 22–25 | 28–30 | 102 | GA44 | |
| 22–25 | 45–47 | 94 | GA19 | |
| 30–34 | 28–31 | 726 | GA15 (feed) |
Identified by data shown in Table II.
Table II.
Representative GC-MS and KRI data used for the identification of GA metabolites (listed in Table I) from the feeds of [17-14C]GA15 to maize
| [14C]GA Metabolite/Ref. Compound | KRIa | Diagnostic Ions | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [14C]GA15 | 2,587 | m/z | 346 | 314 | 300 | 286 | 241 | 213 | 195 | |||
| intensity | 19 | 19 | 13 | 54 | 100 | 9 | 30 | |||||
| GA15 ref. | 2,605 | m/z | 344 | 312 | 298 | 284 | 239 | 211 | 193 | |||
| intensity | 25 | 27 | 18 | 70 | 100 | 13 | 33 | |||||
| [14C]GA15-15,16-ene | 2,542 | m/z | 346 | 314 | 288 | 286 | 243 | 229 | 217 | 199 | 185 | 159 |
| intensity | 30 | 59 | 53 | 62 | 100 | 42 | 66 | 24 | 28 | 54 | ||
| GA15-15,16-ene ref. | 2,551 | m/z | 344 | 312 | 286 | 284 | 243 | 227 | 217 | 197 | 183 | 159 |
| intensity | 18 | 60 | 39 | 69 | 100 | 30 | 36 | 19 | 23 | 41 | ||
| [14C]GA19 | 2,584 | m/z | 464 | 436 | 404 | 376 | 347 | 317 | 287 | 258 | 241 | 210 |
| intensity | 17 | 100 | 24 | 52 | 18 | 18 | 23 | 28 | 46 | 40 | ||
| GA19 ref. | 2,596 | m/z | 462 | 434 | 402 | 374 | 345 | 315 | 285 | 258 | 239 | 208 |
| intensity | 4 | 100 | 7 | 4 | 24 | 5 | 21 | 30 | 33 | 32 | ||
| [14C]GA20 | 2,473 | m/z | 420 | 405 | 377 | 303 | 237 | 209 | 207 | |||
| intensity | 100 | 3 | 47 | 19 | 15 | 52 | 58 | |||||
| GA20 ref. | 2,482 | m/z | 418 | 403 | 375 | 301 | 235 | 207 | ||||
| intensity | 100 | 16 | 46 | 12 | 8 | 30 | ||||||
| [14C]GA44 | 2,768 | m/z | 434 | 419 | 375 | 240 | 209 | 182 | ||||
| intensity | 60 | 7 | 11 | 32 | 100 | 12 | ||||||
| GA44 ref. | 2,786 | m/z | 432 | 417 | 373 | 238 | 207 | 180 | ||||
| intensity | 46 | 6 | 14 | 33 | 100 | 11 | ||||||
| [14C]GA113 | 2,801 | m/z | 434 | 402 | 374 | 312 | 298 | 284 | 239 | 227 | ||
| intensity | 75 | 26 | 18 | 49 | 35 | 86 | 100 | 30 | ||||
| GA113 ref. | 2,801 | m/z | 432 | 400 | 372 | 310 | 296 | 282 | 237 | 225 | ||
| intensity | 100 | 31 | 27 | 56 | 36 | 83 | 82 | 38 | ||||
The discrepancies between the KRI values for the metabolites and for the standards (ref.) are due to batch-to-batch variations in the GC columns used.
Figure 2.
Structures of metabolites not shown in Figure 1.
[17-14C]GA24
The recovered [14C]labeled metabolites, GA19, GA20, and GA17 are shown in Table III, and are based on identification by the full-scan GC-MS and KRI data presented in Table IV. The step from GA24 to GA19 (Fig. 1) is a direct 13-hydroxylation and is new for maize seedlings. The step from GA19 to GA20 has been previously established for maize (Kobayashi et al., 1996) with no evidence for the conversion of GA19 to GA17. However, the conversion of GA19 to GA17 has been demonstrated using GA 20-oxidases from spinach (Wu et al., 1996) and pumpkin (Lange et al., 1994), which have been cloned and expressed in E. coli.
Table III.
Analysis of metabolites from feeds of [17-14C]GA24 (250 ng, 1.49 × 103 Bq each) to normal, d1, and d5 seedlings of maize
| Plant Material | ODS-HPLC Fraction | N(CH3)2-HPLC Fraction | Radioactivity | [14C]Productsa |
|---|---|---|---|---|
| Bq | ||||
| Normal (20.8 g) | 19–21 | 33–36 | 117 | GA20 |
| 22–23 | 42–45 | 4,290 | GA19 | |
| 29–30 | 40–43 | 663 | GA24 (feed) | |
| d1 (9.5 g) | 19–21 | 33–36 | 452 | GA20 |
| 29–30 | 40–43 | 852 | GA24 (feed) | |
| d5 (10.8 g) | 19–21 | 33–36 | 710 | GA20 |
| 24–25 | 33–35 | 218 | GA17 | |
| 29–30 | 40–43 | 347 | GA24 (feed) |
Identified by data shown in Table IV.
Table IV.
Representative GC-MS and KRI data used for the identification of GA metabolites (listed in Table III) from the feeds of [17-14C]GA24 to maize
| [14C]GA Metabolite/Ref. Compound | KRIa | Diagnostic Ions | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [14C]GA17 | 2,563 | m/z | 494 | 462 | 435 | 434 | 403 | 375 | 374 | 253 | 210 | 195 | ||
| intensity | 64 | 37 | 28 | 37 | 19 | 26 | 23 | 27 | 100 | 21 | ||||
| GA17 ref. | 2,575 | m/z | 492 | 460 | 433 | 432 | 401 | 373 | 372 | 251 | 208 | 193 | ||
| intensity | 43 | 23 | 26 | 15 | 11 | 23 | 14 | 24 | 100 | 22 | ||||
| [14C]GA19 | 2,584 | m/z | 464 | 436 | 404 | 376 | 347 | 317 | 287 | 258 | 241 | 210 | ||
| intensity | 17 | 100 | 24 | 52 | 18 | 18 | 23 | 28 | 46 | 40 | ||||
| GA19 ref. | 2,596 | m/z | 462 | 434 | 402 | 374 | 345 | 315 | 285 | 258 | 239 | 208 | ||
| intensity | 4 | 100 | 7 | 4 | 24 | 5 | 21 | 30 | 33 | 32 | ||||
| [14C]GA20 | 2,473 | m/z | 420 | 405 | 377 | 361 | 303 | 237 | 209 | 194 | 182 | 169 | ||
| intensity | 100 | 12 | 50 | 14 | 14 | 6 | 32 | 8 | 8 | 8 | ||||
| GA20 ref. | 2,482 | m/z | 418 | 403 | 375 | 359 | 301 | 235 | 207 | 192 | 180 | 167 | ||
| intensity | 100 | 16 | 6 | 2 | 2 | 8 | 30 | 8 | 6 | 7 | ||||
| [14C]GA24 | 2,426 | m/z | 376 | 348 | 344 | 316 | 312 | 288 | 287 | 284 | 256 | 229 | 228 | 227 |
| intensity | 3 | 8 | 33 | 91 | 42 | 72 | 50 | 34 | 58 | 58 | 83 | 100 | ||
| GA24 ref. | 2,442 | m/z | 374 | 346 | 342 | 314 | 310 | 286 | 285 | 282 | 254 | 227 | 226 | 225 |
| intensity | 4 | 8 | 30 | 80 | 26 | 79 | 72 | 42 | 29 | 70 | 100 | 78 | ||
The discrepancies between the KRI values for the metabolites and for the standards (ref.) are due to batch-to-batch variations in the GC columns used.
[17-14C]GA25
The recovered [14C]labeled metabolites GA17, GA25 16α,17-H2-17-OH, and HO-GA25 (hydroxyl position not determined) are shown in Table V, based on identification by the full-scan GC-MS and KRI data presented in Table VI. The metabolism of GA25 to GA17 (Fig. 2) is a result of direct 13-hydroxylation. This step is new for plants.
Table V.
Analysis of metabolites from feeds of [17-14C]GA25 (250 ng, 1.42 × 103 Bq each) to normal, d1, and d5 seedlings of maize
| Plant Material | ODS-HPLC Fraction | N(CH3)2-HPLC Fraction | Radioactivity | [14C]Producta |
|---|---|---|---|---|
| Bq | ||||
| Normal (17.1 g) | 19–21 | 23–25 | 88 | GA25 16α, 17-H2 17-OH |
| 19–21 | 26–28 | 101 | HO-GA25, unknown position of hydroxyl | |
| 23–25 | 27–29 | 412 | GA17 | |
| 29–31 | 26–28 | 498 | GA25 (feed) | |
| d1 (6.0 g) | 19–21 | 26–28 | 35 | HO-GA25, unknown position of hydroxyl |
| 23–25 | 27–29 | 560 | GA17 | |
| 29–31 | 26–28 | 365 | GA25 (feed) | |
| d5 (5.2 g) | 19–21 | 23–25 | 41 | GA25 16α, 17-H2 17-OH |
| 19–21 | 26–28 | 78 | HO-GA25, unknown position of hydroxyl | |
| 23–25 | 27–29 | 563 | GA17 | |
| 29–31 | 26–28 | 595 | GA25 (feed) |
Identified by data shown in Table VI.
Table VI.
Representative GC-MS and KRI data used for the identification of GA metabolites (listed in Table V) from the feeds of [17-14C]GA25 to maize
| [14C]GA Metabolite/Ref. Compound | KRIa | Diagnostic Ions | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [14C]GA17 | 2,539 | m/z | 494 | 462 | 435 | 434 | 403 | 375 | 374 | 253 | 210 | 195 |
| intensity | 77 | 94 | 67 | 94 | 33 | 41 | 41 | 11 | 100 | 11 | ||
| GA17 ref. | 2,575 | m/z | 492 | 460 | 433 | 432 | 401 | 373 | 372 | 251 | 208 | 193 |
| intensity | 43 | 23 | 26 | 15 | 11 | 23 | 14 | 24 | 100 | 22 | ||
| [14C]GA25 | 2,455 | m/z | 406 | 374 | 314 | 255 | 286 | 227 | 199 | |||
| intensity | 0 | 19 | 63 | 9 | 100 | 37 | 6 | |||||
| GA25 ref. | 2,440 | m/z | 404 | 372 | 312 | 253 | 284 | 225 | 197 | |||
| intensity | 0 | 13 | 82 | 8 | 100 | 41 | 4 | |||||
| [14C]HO-GA25, unknown position of hydroxylb | 2,667 | m/z | 494 | 462 | 460 | 434 | 432 | 402 | 400 | 374 | 372 | |
| intensity | 3 | 77 | 19 | 36 | 14 | 41 | 17 | 100 | 31 | |||
| [14C]GA25 16α,17-H2 17-OH | 2,738 | m/z | 496 | 464 | 436 | 404 | 376 | 342 | 314 | 286 | 227 | |
| intensity | 0 | 69 | 22 | 100 | 66 | 28 | 36 | 57 | 37 | |||
| [14C]GA25 16α,17-H2 17-OH ref. | 2,760 | m/z | 494 | 462 | 434 | 402 | 374 | 340 | 312 | 284 | 225 | |
| intensity | 0 | 86 | 26 | 100 | 63 | 30 | 78 | 97 | 93 | |||
The discrepancies between the KRI and ion abundance values for the metabolites and for the standards (ref.) are due to the change in the GC-MS instrument from a DANI-3800 GC-VG Analytical 70–250 (Micromass, Beverly, MA) mass spectrometer to a Thermoquest GCQ (Thermoquest, San Jose, CA) gas chromatograph with a WCOT BPX5 capillary column (25-m × 0.22-mm × 0.25-μm film thickness; Scientific Glass Engineering).
No reference data available; identification by analogy with known HO-GA25 examples.
[17-14C]GA7
The [14C]labeled metabolites GA30, GA3, isoGA3, and GA7-diene-diacid (trace amounts) are shown in Table VII, and are based on identification by the full-scan GC-MS and KRI data shown in Table VIII. However, in each case, most of the radioactivity was recovered in fractions (Table VII) that contained products not analyzable by GC-MS. The products are presumed to be conjugates.
Table VII.
Analysis of metabolites from feeds of [17-14C]GA7 (250 ng, 1.55 × 103 Bq each) to seedlings of normal, d1, and d5 seedlings of maize
| Plant Material | ODS-HPLC Fraction | N(CH3)2-HPLC Fraction | Radioactivity | [14C]Productb |
|---|---|---|---|---|
| Bq | ||||
| Normal (14.2 g) | 8–9 | 34–35 | 17a | GA30 |
| 10–11 | 33–34 | 23a | GA3, isoGA3 | |
| 19–22 | 33–35 | 18a | GA7-diene-diacid | |
| 24–26 | 31–34 | 216a | c | |
| 24–26 | 44–47 | 265a | c | |
| d1 (8.3 g) | 8–9 | 34–35 | 33 | GA30 |
| 10–11 | 33–34 | 54 | GA3, isoGA3 | |
| 19–22 | 33–35 | 24 | GA7-diene-diacid | |
| 24–26 | 31–34 | 432 | c | |
| 24–26 | 44–47 | 723 | c | |
| d5 (8.1 g) | 8–9 | 34–35 | 53 | GA30 |
| 10–11 | 33–34 | 98 | GA3, isoGA3 | |
| 19–22 | 33–35 | 15 | GA7-diene-diacid | |
| 24–26 | 31–34 | 470 | c | |
| 24–26 | 44–47 | 393 | c |
One-half of the original feed.
Identified by data shown in Table VIII.
No 14C-labeled compounds were identified by GC-MS.
Table VIII.
Representative GC-MS and KRI data used for the identification of GA metabolites (listed in Table VII) from the feeds of [17-14C]GA7 to maize
| [14C]GA Metabolite/Ref. Compound | KRIa | Diagnostic Ions | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [14C]GA3 | 2,685 | m/z | 506 | 491 | 447 | 433 | 372 | 349 | 313 | 240 | 210 | |
| intensity | 100 | 6 | 8 | 5 | 12 | 6 | 8 | 12 | 19 | |||
| GA3 ref. | 2,692 | m/z | 504 | 489 | 445 | 431 | 370 | 347 | 311 | 238 | 208 | |
| intensity | 100 | 7 | 12 | 9 | 24 | 9 | 14 | 21 | 37 | |||
| iso[14C]GA3 | 2,625 | m/z | 506 | 501 | 477 | 447 | 372 | 240 | 223 | |||
| intensity | 100 | 9 | 20 | 17 | 16 | 24 | 22 | |||||
| isoGA3 ref. | 2,633 | m/z | 504 | 499 | 475 | 445 | 370 | 238 | 221 | |||
| intensity | 100 | 10 | 12 | 9 | 12 | 28 | 12 | |||||
| [14C]GA7b (feed) | 2,520 | m/z | 418 | 386 | 358 | 300 | 284 | 225 | 224 | 195 | 181 | 155 |
| intensity | 11 | 12 | 17 | 21 | 25 | 60 | 100 | 28 | 28 | 31 | ||
| GA7 ref.b | 2,525 | m/z | 416 | 384 | 356 | 298 | 282 | 223 | 222 | 193 | 179 | 155 |
| intensity | 9 | 22 | 22 | 19 | 35 | 73 | 100 | 43 | 42 | 48 | ||
| [14C]GA7 di-acid 9,10-ene | 2,399 | m/z | 432 | 372 | 313 | 283 | 223 | 195 | ||||
| intensity | 14 | 45 | 59 | 100 | 100 | 60 | ||||||
| GA7 di-acid 9,10-ene ref. | 2,405 | m/z | 430 | 370 | 311 | 281 | 221 | 193 | ||||
| intensity | 17 | 23 | 80 | 100 | 77 | 24 | ||||||
| [14C]GA30 | 2,754 | m/z | 506 | 446 | 416 | 384 | 371 | 315 | 282 | 223 | 195 | |
| intensity | 11 | 6 | 13 | 18 | 38 | 44 | 34 | 100 | 42 | |||
| GA30 ref. | 2,759 | m/z | 504 | 444 | 414 | 382 | 369 | 315 | 280 | 221 | 193 | |
| intensity | 30 | 10 | 26 | 21 | 50 | 17 | 37 | 100 | 47 | |||
The discrepancies between the KRI values for the metabolites and for the standards (ref.) are due to batch-to-batch variations in the GC columns used.
Data for [14C]GA7 is reported, although not recovered from feed.
2,3-Dehydro-[17-14C]GA9
The recovered [14C]labeled metabolites, GA5, GA7 (trace amounts), 2,3 dehydro-GA10 (artifact), GA31, and GA62 are shown in Table IX, based on identification by the full-scan GC-MS and KRI data shown in Table X. Four of the metabolites are formed by hydroxylation at C-1β (GA62, Fig. 2), at C-3β (GA7, Fig. 1), at C-12α (GA31, Fig. 2), and at C-13 (GA5, Fig. 1). 2,3-Dehydro-GA10 (Fig. 2) is the product of hydration of the 16,17-double bond and this step may be non-enzymatic. The metabolism of 2,3-dehydro-[17-2H2]GA9 to [2H2]GA7 has been previously reported from cell-free systems from seeds of wild cucumber and apple (Albone et al., 1990). The metabolism of 2,3-dehydro-GA9 to GA62, to GA31, and to GA5 are the first examples of these conversions in plants.
Table IX.
Analysis of metabolites from feeds of 2,3-dehydro-[17-14C]GA9 (88 ng, 485 Bq each) to d5 maize (10.0 g of plant material)
| ODS-HPLC Fraction | N(CH3)2-HPLC Fraction | Radioactivity | [14C]Producta | Specific Radioactivity |
|---|---|---|---|---|
| Bq | TBq mol−1 | |||
| 14–15 | 13 | 77 | GA31 | 1.81 |
| 16–18 | 9–10 | 136 | 2,3-Dehydro-GA10 | 1.74 |
| 16–18 | 14–15 | 162 | GA5 | 1.76 |
| 19–21 | 10–13 | 124 | GA62 | Not determined |
| 22–24 | 9–10 | 14 | GA7 (trace) | Not determined |
| 26–27 | 12 | 381 | 2,3-Dehydro-GA9 (feed) | 1.76 |
Identified by data shown in Table X.
Table X.
Representative GC-MS and KRI data used for the identification of GA metabolites (listed in Table IX) from the feeds of 2,3-dehydro-[17-14C]GA9 to d5 maize
| [14C]GA Metabolite/Ref. Compound | KRIa | Diagnostic Ions | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [14C]GA5 | 2,475 | m/z | 418 | 403 | 359 | 345 | 315 | 301 | 209 | |||||
| intensity | 100 | 17 | 24 | 9 | 31 | 402 | 32 | |||||||
| GA5 ref. | 2,479 | m/z | 416 | 401 | 357 | 343 | 313 | 299 | 207 | |||||
| intensity | 100 | 21 | 24 | 22 | 9 | 58 | 55 | |||||||
| [14C]GA7 | 2,522 | m/z | 418 | 386 | 358 | 300 | 284 | 225 | 224 | |||||
| intensity | 8 | 46 | 8 | 18 | 20 | 60 | 100 | |||||||
| GA7 ref. | 2,525 | m/z | 416 | 384 | 356 | 298 | 282 | 223 | 222 | |||||
| intensity | 9 | 14 | 22 | 19 | 35 | 30 | 100 | |||||||
| 2,3-Dehydro-[14C]GA9 | 2,298 | m/z | 299 | 286 | 284 | 227 | 226 | 156 | ||||||
| intensity | 7 | 41 | 12 | 51 | 100 | 45 | ||||||||
| 2,3-Dehydro-GA9 ref. | 2,301 | m/z | 297 | 284 | 282 | 225 | 224 | 156 | ||||||
| intensity | 10 | 46 | 2 | 62 | 100 | 36 | ||||||||
| 2,3-Dehydro-[14C]GA10b | 2,563 | m/z | 420 | 361 | 329 | 286 | 227 | 226 | 143 | 132 | ||||
| intensity | 24 | 24 | 46 | 52 | 58 | 79 | 52 | 100 | ||||||
| [14C]GA31 | 2,546 | m/z | 418 | 386 | 371 | 296 | 284 | 268 | 251 | 241 | 225 | 224 | 223 | 195 |
| intensity | 7 | 7 | 10 | 14 | 47 | 33 | 15 | 10 | 58 | 100 | 77 | 44 | ||
| GA31 ref. | 2,550 | m/z | 416 | 384 | 369 | 294 | 282 | 266 | 269 | 239 | 223 | 222 | 221 | 193 |
| intensity | 6 | 8 | 10 | 19 | 49 | 33 | 23 | 7 | 71 | 100 | 72 | 36 | ||
| [14C]GA62 | 2,424 | m/z | 418 | 403 | 374 | 315 | 284 | 225 | 224 | 209 | ||||
| intensity | 2 | 7 | 13 | 18 | 36 | 100 | 95 | 26 | ||||||
| GA62 ref. | 2,424 | m/z | 416 | 401 | 372 | 313 | 282 | 223 | 222 | 207 | ||||
| intensity | 0 | 4 | 8 | 10 | 24 | 100 | 93 | 10 | ||||||
The discrepancies between the KRI values for the metabolites and for the standards (ref.) are due to batch-to-batch variations in the GC columns used.
No reference data available; identification by analogy with known GA-15,16-enes.
Isotopic Dilution of 2,3-Dehydro-GA9
In view of the observed conversion of 2,3-dehydro-GA9 to GA7, we investigated the possible natural occurrence of 2,3-dehydro-GA9 in maize. Thus, we determined the level of isotopic dilution of 2,3-dehydro-[17-14C]GA9 added to a homogenate of normal maize seedlings. No dilution of label was observed based on a full-scan GC-MS analysis of the recovered 2,3-dehydro-[17-14C]GA9 (data not shown), thus indicating that 2,3-dehydro-GA9 is not endogenous to maize.
General
The structures of the substrates and metabolites presented in this report are shown in Figures 1 and 2, with the exception of the HO-GA25 metabolite for which the hydroxylation site was not determined. In maize, the 13-hydroxylation of GA15 to GA44, GA24 to GA19, GA9 to GA20, and GA4 to GA1 results in the formation of a grid connecting members of the (presumptive) non-early 3,13-hydroxylation pathway to members of the early 13-hydroxylation pathway (Fig. 1). The two steps, GA15 → GA44 and GA24 → GA19, represent the first demonstration of in vivo crossovers between C20-GAs. A similar grid connecting the two branch pathways has been demonstrated from in vitro studies from a number of plant species (Kamiya and Graebe, 1983; Takahashi et al., 1986; Grosslindemann et al., 1992). The 13-hydroxylation of GA15, GA24, GA9, and GA4 in maize may reside in a single 13-hydroxylase with low substrate specificity or with the presence of separate substrate-specific enzymes. The failure to detect the sequence GA12 → GA15 → GA24 → GA9 → GA4 → GA7 could be because the Km values for these substrates are much lower for the 13-hydroxylase(s) than for the 20-oxidase(s).
The two labeled metabolites GA15-15,16-ene and GA7-diene-diacid were probably formed by the non-enzymatic rearrangement of a double bond. Additionally, 2,3-dehydro-GA10 was probably formed as a result of a non-enzymatic hydration of the 16,17-double bond in the substrate 2,3-dehydro-GA9.
Based on the previous demonstration that GA5 is an intermediate between GA20 and GA3 in maize shoots (Fujioka et al., 1990; Spray et al., 1996), we examined the possibility that 2,3-dehydro-GA9 is an intermediate between GA9 and GA7. Our results show that 2,3-dehydro-GA9 is predominantly 13-hydroxylated to GA5, 12α-hydroxylated to GA31, and 1β-hydroxylated to GA62, and converted into GA7 in trace amounts. However, isotope dilution studies gave no evidence for the natural occurrence of 2,3-dehydro-GA9 in maize shoots (data not shown). The metabolic origin of GA15, GA24, GA9, GA4, and GA7 in maize remains unresolved.
Footnotes
This work was supported by the National Science Foundation (grant nos. MCB–9604460 and MCB–9306597) and by the U.S. Department of Energy (grant no. DE–FG03–90ER20016). The IACR receives grant-aided support from the Biotechnological and Biological Science Research Council of the United Kingdom.
LITERATURE CITED
- Albone KS, Gaskin P, MacMillan J, Phinney BO, Willis CL. Biosynthetic origin of gibberellins A3 and A7 in cell-free preparations from seeds of Marah macrocarpus and Malus domestica. Plant Physiol. 1990;94:132–142. doi: 10.1104/pp.94.1.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croker SJ, Gaskin P, Hedden P, MacMillan J, MacNeil KAG. Quantitative analysis of gibberellins by isotope dilution mass spectrometry: a comparison of the use of calibration curves, an isotope dilution fit program and arithmetical correction of isotope ratios. Phytochem Anal. 1994;5:74–80. [Google Scholar]
- Davis G, Kobayashi M, Phinney BO, MacMillan J, Gaskin P. The metabolism of GA9 in maize (Zea mays) Phytochemistry. 1998;47:635–639. [Google Scholar]
- Fujioka S, Yamane H, Spray CR, Gaskin P, MacMillan J, Phinney BO, Takahashi N. Qualitative and quantitative analyses of gibberellins in vegetative shoots of normal, dwarf-1, dwarf-2, dwarf-3, and dwarf-5 seedlings of Zea mays L. Plant Physiol. 1988a;88:1367–1372. doi: 10.1104/pp.88.4.1367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujioka S, Yamane H, Spray CR, Katsumi M, Phinney BO, Gaskin P, MacMillan J, Takahashi N. The dominant non-gibberellin-responding dwarf mutant (D8) of maize accumulates native gibberellins. Proc Natl Acad Sci USA. 1988b;85:9031–9035. doi: 10.1073/pnas.85.23.9031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujioka S, Yamane H, Spray CR, Phinney BO, Gaskin P, MacMillan J, Takahashi N. Gibberellin A3 is biosynthesized from gibberellin A20 via gibberellin A5 in shoots of Zea mays L. Plant Physiol. 1990;94:127–131. doi: 10.1104/pp.94.1.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaskin P, MacMillan J. GC-MS of Gibberellins and Related Compounds: Methodology and a Library of Reference Spectra. Bristol, UK: Cantocks Enterprises; 1991. [Google Scholar]
- Grosslindemann E, Lewis MJ, Hedden P, Graebe JE. Gibberellin biosynthesis from gibberellin A12-aldehyde in a cell-free system from germinating barley (Hordeum vulgare L., cv Himalaya) embryos. Planta. 1992;188:252–257. doi: 10.1007/BF00216821. [DOI] [PubMed] [Google Scholar]
- Kamiya Y, Graebe JE. The biosynthesis of all major pea gibberellins in a cell-free system from Pisum sativum. Phytochemistry. 1983;22:681–689. [Google Scholar]
- Kobayashi M, Gaskin P, Spray CR, Suzuki Y, Phinney BO, MacMillan J. Metabolism and biological activity of gibberellin A4 in vegetative shoots of Zea mays, Oryza sativa, and Arabidopsis thaliana. Plant Physiol. 1993;102:379–386. doi: 10.1104/pp.102.2.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi M, Spray CR, Phinney BO, Gaskin P, MacMillan J. Gibberellin metabolism in maize: the stepwise conversion of gibberellin A12-aldehyde to gibberellin A20. Plant Physiol. 1996;110:413–418. doi: 10.1104/pp.110.2.413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lange T. Cloning gibberellin dioxygenase genes from pumpkin endosperm by heterologous expression of enzyme activities in Escherichia coli. Proc Natl Acad Sci USA. 1997;94:6553–6558. doi: 10.1073/pnas.94.12.6553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lange T, Graebe JE. Enzymes of gibberellin synthesis. In: Lea PJ, editor. Methods in Plant Biochemistry. Vol. 9. London: Academic Press; 1993. pp. 403–430. [Google Scholar]
- Lange T, Hedden P, Graebe JE. Expression cloning of a gibberellin 20-oxidase, a multifunctional enzyme involved in gibberellin biosynthesis. Proc Natl Acad Sci USA. 1994;91:8552–8556. doi: 10.1073/pnas.91.18.8552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacMillan J. Biosynthesis of the gibberellin plant hormones. Nat Prod Rep. 1997;14:221–243. [Google Scholar]
- MacMillan J, Ward DA, Phillips AL, Sanchez-Beltran MJ, Gaskin P, Lange T, Hedden P. Gibberellin biosynthesis from gibberellin A12-aldehyde in endosperm and embryos of Marah macrocarpus. Plant Physiol. 1997;113:1369–1377. doi: 10.1104/pp.113.4.1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakayama M, Nishijama T, Koshioka M, Yamane H, Owen DJ, Mander LN. Identification of GA113, GA114, GA115 and GA116 and further novel gibberellins in Raphanus sativus. Phytochemistry. 1998;48:587–593. [Google Scholar]
- Spray CR, Kobayashi M, Suzuki Y, Phinney BO, Gaskin P, MacMillan J. The dwarf-1 (d1) mutant of Zea mays blocks three steps in the gibberellin-biosynthetic pathway. Proc Nat Acad Sci USA. 1996;93:10515–10518. doi: 10.1073/pnas.93.19.10515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi M, Kamiya Y, Takahashi N, Graebe JE. Metabolism of gibberellins in a cell-free system from immature seeds of Phaseolus vulgaris L. Planta. 1986;168:190–199. doi: 10.1007/BF00402963. [DOI] [PubMed] [Google Scholar]
- Wu K, Li L, Gage DA, Zeevaart JAD. Molecular cloning and photoperiod-regulated expression of gibberellin 20-oxidase from the long-day plant spinach. Plant Physiol. 1996;110:547–554. doi: 10.1104/pp.110.2.547. [DOI] [PMC free article] [PubMed] [Google Scholar]