Abstract
There is some evidence to suggest that oxytocin promotes social behavior, especially for disorders characterized by social dysfunction, such as social anxiety disorder (SAD). The goal of this study was to examine the effect of oxytocin on reward motivation in SAD. We tested whether oxytocin promotes prosocial, or antisocial, self-directed decisions, and whether its effects depended on social anxiety severity and attachment. Fifty-two males with SAD received 24 international units of oxytocin or placebo, and completed a reward motivation task that measured willingness to work for self vs. other monetary rewards. Although there was no main drug effect, social anxiety severity moderated the effect of oxytocin. Less socially anxious individuals who received oxytocin worked harder for other vs. own rewards, compared to high socially anxious individuals. Attachment did not moderate this effect. Among people with SAD, oxytocin enhances prosocial behaviors in individuals with relatively lower levels of social anxiety.
National Institutes of Health ClinicalTrials.gov Registry #NCT01856530. https://clinicaltrials.gov/ct2/show/NCT01856530?term=oxytocin+pro-social&rank=2.
Keywords: Oxytocin, Self, Other, Reward, Motivation
1. Introduction
Oxytocin, a hypothalamic neuropeptide, is a promising pharmacologic target for modulating social cognition (Hurlemann & Scheele, 2016; Shahrestani, Kemp, & Guastella, 2013). Individuals with social anxiety disorder (SAD) display anxiety and self-consciousness in social situations (Hofmann, 2007), which may be modulated by oxytocin (Labuschagne et al., 2010; Shamay-Tsoory & Abu-Akel, 2016).
Several theories have been put forth to explain the effects of oxytocin: a prosocial, affiliative account (Kosfeld, Heinrichs, Zak, Fischbacher, & Fehr, 2005), a social salience enhancing account (De Dreu et al., 2010; Shamay-Tsoory et al., 2009; Shamay-Tsoory, 2010), a social approach/withdrawal account (Kemp & Guastella, 2011), and an anxiety reduction account (Bartz, Zaki, Bolger, & Ochsner 2011).
The prosocial theory of oxytocin proposes that oxytocin increases a wide range of “prosocial” behaviors, which are defined broadly as voluntary acts that benefit other people, and are driven by non-specific motives (Eisenberg & Miller, 1987). Reciprocal altruism is more narrowly defined as a prosocial behavior that benefits another even at personal cost, but with the expectation of being helped at a later point (Eisenberg & Miller, 1987). Reciprocal altruism has been linked behaviorally to empathic concern for unfamiliar others (De Waal, 2008), which are both oxytocin-dependent processes (Bartz et al., 2010; Hurlemann et al., 2010). The social salience account hypothesizes that oxytocin alters the perceptual salience of social information depending on the context of the situation itself (Shamay-Tsoory et al., 2009; Shamay-Tsoory, 2010). Oxytocin may either increase prosociality in cooperative social contexts, or promote envy and gloating (Shamary-Tsoory et al., 2009), and defense-motivated aggression (De Dreu et al., 2010), in competitive out-group interactions. The social approach/withdrawal hypothesis proposes that oxytocin may enhance approach-related emotions (including negative emotions, such as anger or jealousy) or inhibit social withdrawal-related emotions (such as anxiety and fear) (Kemp & Guastella, 2011). In patients with SAD, oxytocin led to reduced negative self-appraisals after exposure therapy despite having no changes on social anxiety symptom severity, relative to placebo, which supports this hypothesis that oxytocin alters cognitive biases involved in processing threat (Guastella et al., 2009). The anxiety reduction hypothesis proposes that oxytocin leads to beneficial social effects by reducing anxiety, especially social anxiety (Bartz et al., 2011; Heinrichs et al., 2003).
Each of these theories has different implications for how oxytocin may be potentially advantageous for individuals with SAD. The prosocial and anxiety reduction models hypothesize that oxytocin would facilitate social approach behavior, by reducing anxiety and fear in social situations. The social salience and social approach/withdrawal models hypothesize that oxytocin could be potentially harmful to patients with SAD by magnifying negative emotional or attentional tendencies. They also propose that oxytocin could be potentially beneficial by modulating emotional experiences and attentional processing to facilitate a more favorable self-view, and promote social engagement. In particular, some evidence suggests that oxytocin may induce a favorable self-bias, as studies in healthy male subjects have shown that oxytocin enhanced positive attitudes towards oneself, compared to placebo, in an adjective sorting task (Colonnello & Heinrichs, 2014), and enhanced the ability to recognize differences between self and other using a face morphing task (Colonello, Chen, Panksepp, & Heinrichs, 2013). However, some findings suggest that oxytocin may actually blur the self-other distinction and reduces medial prefrontal cortex responses and connectivity with other cortical midline regions involved in self-referential processing (Zhao et al., 2016). Thus, it remains unclear if oxytocin could be advantageous to individuals with SAD, who are excessively and negatively self-focused in social settings (Hofmann, 2007; Ingram, 1990; Spurr and Stopa, 2002).
In the current study, our objective was to test divergent accounts of oxytocin’s effects on self-other reward motivation among individuals with SAD. We operationalized reward motivation as one’s willingness to expend effort in exchange for monetary rewards for oneself vs. monetary rewards that would be given to a stranger. Given competing accounts of oxytocin’s effects, we hypothesized that in patients with SAD who display excessive social anxiety and negative self-focus, oxytocin would either (1) promote motivation to work harder for others’ rewards (pro-social, affiliative account (Kosfeld et al., 2005), and anxiety reduction account (Bartz et al., 2011; Heinrichs et al., 2003)), or (2) oxytocin would promote more self-oriented behavior to reward oneself (social salience account (Olff et al., 2013; Shamay-Tsoory, 2010) and social approach/withdrawal account (Kemp & Guastella, 2011)). Our previous work showed that in a sample of male SAD patients, oxytocin improved cooperative behavior toward a rejecting, but initially cooperative, player during a social ostracism paradigm called Cyberball, but only for those who were less severe in their avoidant attachment style (Fang, Hoge, Heinrichs, & Hofmann 2014). This is consistent with prior research demonstrating that individual difference factors moderate oxytocin’s effects (Olff et al., 2013), and that oxytocin may have the most potent effects for individuals only within a certain range of functioning (Scheele et al., 2014). Thus, we also hypothesized that the effects of oxytocin would be moderated by social anxiety symptom severity and attachment orientation.
2. Materials and methods
2.1. Participants
Participants were recruited through an outpatient specialty anxiety clinic and through advertisements in the community. All participants were adult men with a principal or co-principal diagnosis of SAD, who met a symptom severity cutoff score of ≥ 60 on the Liebowitz Social Anxiety Scale (LSAS; Liebowitz, 1987). Exclusion criteria included the following: significant nasal pathology; smoking ≥ 15 cigarettes per day; serious medical illnesses; active suicidal or homicidal ideation; current diagnosis of schizophrenia, psychotic disorder, bipolar disorder, or substance abuse or dependence; and, use of psychotropic medications, except for antidepressants taken at a stable dose for at least 2 weeks prior to study entry. Women were excluded from the study due to fluctuations in oxytocin during their menstrual cycles, as well as sex differences in responses to oxytocin (Dumais & Veenema, 2015). Please refer to the CONSORT diagram for a full description of participant recruitment and trial design. Our final sample consisted of 52 participants (mean age = 24.42 years, SD = 6.63, range = 18–45). See Table 1 for demographic and clinical data for the final sample. Of these 52 participants, 3 participants displayed unusual behavior on the reward motivation task: one chose all hard trials, one timed out on a larger percentage (10%) of trials, and one stopped responding halfway through the task. Removing these subjects from the analyses did not affect the main findings (effects of drug), so they were included in the analyses. There were no differences between groups on demographic or baseline clinical characteristics (all p’s > 0.05). The study was approved by the Boston University Medical Center Institutional Review Board.
Table 1.
Demographic and Clinical Data.
| Demographic variable | Oxytocin group (n = 26) |
Placebo group (n = 26) |
t/X2 | p |
|---|---|---|---|---|
| Age (in years), Mean (SD) | 24.65 (7.28) | 24.19 (6.05) | −0.25 | 0.81 |
| Ethnicity, n (%) | 0.75 | 0.39 | ||
| Hispanic or Latino | 4 (15.40) | 2 (7.70) | ||
| Not Hispanic or Latino | 22 (84.60) | 24 (92.30) | ||
| Race, n (%) | 1.27 | 0.74 | ||
| Caucasian | 18 (69.20) | 16 (61.50) | ||
| African American | 2 (7.70) | 2 (7.70) | ||
| Asian | 4 (15.40) | 7 (26.90) | ||
| Other | 2 (7.70) | 1 (3.80) | ||
| Marital Status, n (%) | 4.02 | 0.26 | ||
| Single | 22 (84.60) | 23 (88.50) | ||
| Living with partner | 1 (3.80) | 3 (11.50) | ||
| Married | 2 (7.70) | 0 | ||
| Divorced | 1 (3.80) | 0 | ||
| Highest Educational Level, n (%) | 6.29 | 0.10 | ||
| Graduate School | 2 (7.70) | 8 (30.80) | ||
| College Graduate | 9 (34.60) | 6 (23.10) | ||
| Partial College | 14 (53.80) | 9 (34.60) | ||
| High School Graduate | 1 (3.80) | 3 (11.50) | ||
| Occupational Status, n (%) | 0.60 | 0.90 | ||
| Full-time employment | 5 (19.2) | 5 (19.20) | ||
| Part-time employment | 6 (23.1) | 6 (23.1) | ||
| Dependent on spouse or is a student | 9 (34.6) | 11 (42.30) | ||
| Other | 6 (23.1) | 4 (15.4) | ||
| Clinical Variable | ||||
| LSAS Total, Mean (SD) | 82.00 (18.16) | 83.00 (16.39) | 0.21 | 0.84 |
| SIAS Totala, Mean (SD) | 50.40 (10.35) | 47.96 (11.93) | −0.77 | 0.44 |
| ECR Avoidant Attachment Subscaleb, Mean (SD) | 3.43 (.89) | 3.61 (.98) | 0.49 | 0.63 |
| ECR Anxious Attachment Subscaleb, Mean (SD) | 4.47 (1.02) | 3.90 (1.13) | −1.33 | 0.20 |
LSAS = Liebowitz Social Anxiety Scale; SIAS = Social Interaction Anxiety Scale; ECR = Experiences in Close Relationships Inventory.
n = 25 per group.
n = 12 (placebo), n = 13 (oxytocin).
2.2. Materials
Social Interaction and Anxiety Scale (SIAS) (Mattick and Clarke, 1998). The SIAS is a 20-item self-report measure that assesses anxiety in social interaction situations. Responses to items are given on a 5-point Likert scale, with 0 = not at all characteristic of me, and 4 = extremely characteristic of me. Total scores range from 0 to 80. The SIAS has been shown to be a valid measure of social interactional anxiety, and has also been demonstrated to have good internal consistency and reliability, in samples of patients with SAD (Clark et al., 1997). The SIAS was administered at baseline. In the current sample, the internal consistency was α = 0.88, and the SIAS scores were highly correlated with LSAS scores (r = 0.58, p < 0.001). The SIAS was selected as our measure of social anxiety to assess the moderating effect of oxytocin, rather than the LSAS, as it specifically measures anxiety related to social interactions without items assessing social avoidance.
Experiences in Close Relationships Inventory (ECR) (Brennan, Clark, & Shaver, 1998). The ECR is a 36-item self-report questionnaire assessing attachment behavior in romantic relationships. Participants were asked to rate how they usually experience relationships, regardless of their current relationship status, on a 1–7 Likert scale (1 = disagree strongly, 7 = agree strongly). The measure yields two subscale scores representing the average of items assessing anxious attachment (reflecting anxiety about being rejected, e.g., “I worry about being abandoned.”) and avoidant attachment (reflecting discomfort with closeness and intimacy, e.g., “I prefer not to show a partner how I feel deep down.”). The internal consistencies of the anxious attachment subscale (α = 0.87) and avoidant attachment subscale (α = 0.88) were excellent.
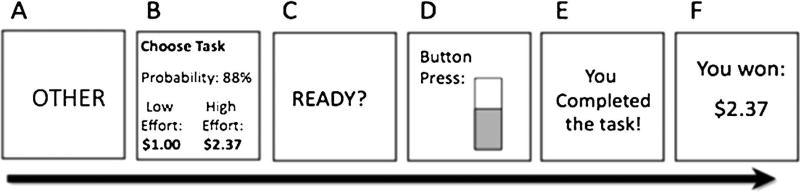
“Pay-it-forward” Effort Expenditure for Rewards Task (Treadway, Buckholtz, Schwartzman, Lambert, & Zald 2009). We used a modified version of the effort expenditure for rewards task to assess self- vs. other-directed reward motivation. The EEfRT is a multi-trial computer-based task that assesses an individual’s willingness to expend effort for the chance to earn monetary rewards. Trials were modified to include “self” vs. “other” conditions, which participants were told would reflect opportunities to earn rewards for themselves, or for the next participant (i.e., “pay it forward”), respectively. Participants were also told that at the end of the study they would receive an amount earned for them by the prior participant, which was included to increase the believability of self/other condition. In reality, all participants received $4.40–$12.40 in additional earnings from this task. See Fig. 1 for a schematic diagram of an example trial from this version of the EEfRT.
Fig. 1. Schematic diagram of an example “Other” trial from the “Pay-it-forward” Effort Expenditure for Rewards Task (EEfRT).
A) Trials begin with being told whether the upcoming trial is for “Self” or “Other”. B) Subjects are presented with information regarding the reward magnitude of the hard task for each trial, and the probability of receiving any reward for completing that trial, and have 5s to choose to play either the easy or hard task. C) 1s “Ready” screen. D) Subjects make rapid button presses to complete their chosen task for 7s (easy task) or 21s (hard task). E) Subjects receive feedback about whether they have completed the task. F) Subjects receive reward feedback about whether they received any money for that trial.
The EEfRT is a multi-trial game in which participants are given an opportunity on each trial to choose between two different task difficulty levels in order to obtain varying monetary rewards. A detailed description of the task has been published previously (Treadway et al., 2009). Briefly, each trial began by presenting the subject with a choice between, a ‘hard task’ (high effort/high reward option) and an ‘easy task’ (low effort/low reward option), which required different amounts of speeded manual button pressing. The easy task required the subject to make 30 button presses in 7 s with the dominant-hand index finger, while the hard task required 100 button presses in 21 s with the non-dominant pinky finger. Each time the subjects chose the ‘easy task’, they were eligible to win the same amount, $1.00, if they successfully completed the task. For hard task choices, subjects were eligible to win higher amounts that varied per trial within a range of $1.24–$4.30. Subjects were not guaranteed to win the reward if they completed the task; some trials were “win” trials, in which the subject received the stated reward amount for the chosen task, while others were “no win” trials, in which the subject received no money for that trial. At the beginning of each trial, subjects were presented with accurate probability cues indicating the likelihood of winning any money on that trial. Trials had three levels of probability: “high” 88% probability of being a win trial, “medium” 50%, and “low” 12%. Probability levels always applied to both the hard task and easy task, and there were equal proportions of each probability level across the experiment. Probability levels were made explicit at the outset of the task. Although the hard task took twice as long as the easy task, the hard task selections were overall more advantageous, as the rewards available for the hard task were up to 400% greater than those for the easy task. Participants had the opportunity to test their understanding of the reward structure in 4 practice trials prior to starting the experiment. Participants were explicitly made aware that the number of trials was not fixed and that they would need to play for 20 min regardless of how quickly or slowly they moved through the trials.
To examine effects of expending effort for self or another person, each trial began with a screen that stated whether the trial was for “Self” or “Other”. Self/other trials were interleaved and presented to all subjects in the same pseudo-randomized order. The EEfRT was performed continuously for 20 min. Because completion of high effort options takes approximately twice as long as low effort options, completion of more high effort options results in fewer overall trials. This means that making high-effort choices in the “Other” condition was costly to participants both in terms of the effort provided, as well as the opportunity cost of being able to complete fewer “Self” trials. Although we did not systematically measure the strategies participants employed during the task, based on participants’ individual data, they appeared to understand the inherent tradeoffs in the task by demonstrating a preference for high effort choices as rewards increased.
2.3. Oxytocin and placebo nasal sprays
The oxytocin and placebo nasal sprays were administered in a double-blind fashion. The sprays were compounded by a local pharmacy under Investigational New Drug #113827 (sponsor-investigator: Hofmann). The oxytocin sprays consisted of 24 international units (IU) of oxytocin, which reflects the most commonly used dosage in single-session intranasal oxytocin studies. Sprays were dispensed in metered-dose bottles to deliver exactly 4 IU per spray. The placebo sprays were identical to the oxytocin nasal sprays, except for the addition of 0.65% sodium chloride to the placebo nasal spray to minimize nasal irritation. The bottles were packaged and labeled identically to protect the experimental blind. Nasal sprays were associated with some minor adverse events (from most to least common): jitteriness/restlessness (17%), anxiety/nervousness (11%), sedation/drowsiness (7%), and dry mouth (7%). A majority (63%) of our sample reported no adverse events. There was no difference in the frequency and nature of reported adverse events between drug conditions. Our assessment of blind revealed that participants could not distinguish between drug conditions.
2.4. Procedure
Participants gave written informed consent and were assessed for eligibility through a diagnostic evaluation using the Mini Adult Diagnostic Interview Schedule for DSM-IV (DiNardo, Brown, & Barlow, 1994) and Liebowitz Social Anxiety Scale (Liebowitz, 1987). Participants then met with the study physician to complete a medical screen, which consisted of assessing concurrent psychotropic medications and significant nasal pathology, as well as measuring vital signs.
The study utilized a double-blind, placebo-controlled design. Participants were randomly assigned to receive a nasal spray containing either oxytocin or placebo using a computer-generated list of random numbers prepared by the study nurse. Using a standardized administration protocol, participants self-administered a nasal spray with three puffs per nostril in the presence of the study physician or nurse. Participants were reminded before their visit to avoid caffeine, alcohol, or nicotine for 24 h prior to the appointment. After nasal spray administration, participants’ vital signs were measured again. Participants were then asked to sit in a waiting room for 45 min before starting the Cyberball Task, a social rejection paradigm (William, Cheung, & Choi, 2000) and a modified Posner task (Posner, 1980). A cover story was provided prior to the Cyberball Task to enhance believability of playing the game with real players. Findings associated with these manipulations are reported elsewhere (Fang et al., 2014). At approximately 60 min post-administration, participants completed the EEfRT.
2.5. Analytic approach
The primary outcomes from the EEfRT were mean proportions of high effort choices across each level of reward probability for self and other conditions, as well as the difference in mean proportions in high effort choices between self and other conditions. A mixed factorial analysis of variance (ANOVA) was conducted with between- (drug: oxytocin vs. placebo) and within-subject factors (condition: self vs. other) to test the effect of oxytocin vs. placebo on proportion of high effort choices in self vs. other conditions. Hierarchical regressions were conducted to test interactions with continuous moderators (e.g. symptom severity, as measured by the SIAS, and attachment orientation, as measured by the ECR). For the moderator analyses, the group and corresponding moderator variables were mean-centered in the first step, and included the product term in the second step. Outcomes and moderating factors were checked for normality. Post-hoc analyses were conducted using t-tests. A median split on proposed moderators was conducted to examine post-hoc comparisons. Two subjects were missing between 5 and 15% of data on the baseline social anxiety measure (SIAS), and were thus removed from the analysis.
3. Results
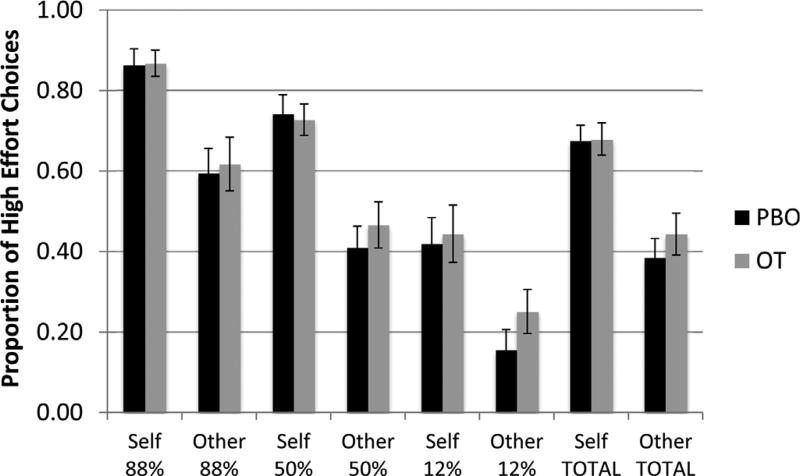
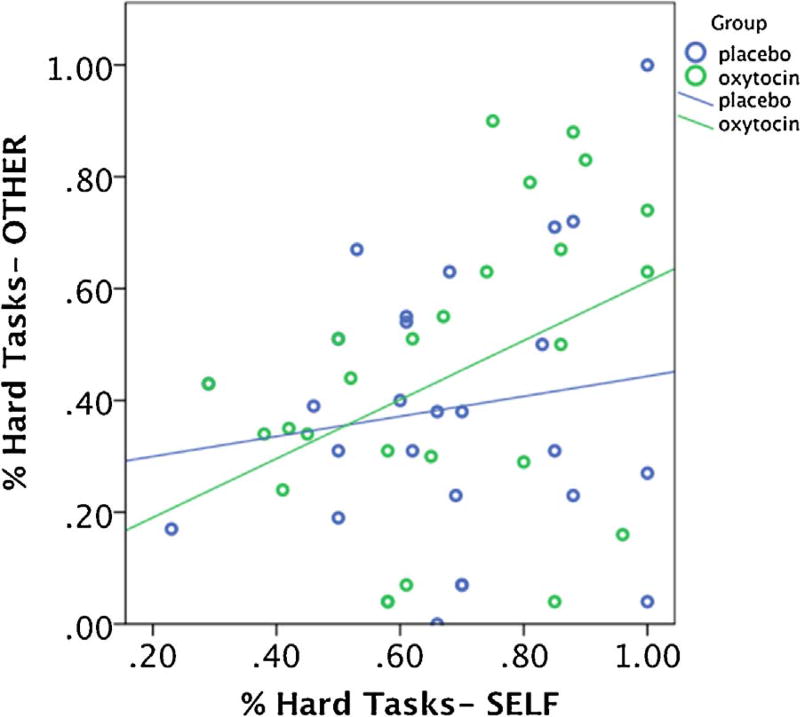
Using the “Pay-it-forward” EEfRT, there was a main effect for self vs. other choices, as everyone in the sample made more high effort choices for themselves rather than for others (pairwise comparisons, all p’s < 0.001; See Fig. 2). There was a significant correlation between self and other high effort choices for the oxytocin condition, but not placebo condition, and the difference between groups was not significant (oxytocin: r(26) = 0.41 p = 0.039; placebo: r(26) = 0.15, p = 0.465) (Fig. 3). Self and other high effort choices were not significantly correlated with social anxiety symptom severity.
Fig. 2.
Main effect of self vs. other condition across levels of reward probability. Error bars represent standard error of the mean. All condition comparisons of self vs. other p < 0.001.
Fig. 3.
Correlations between percentages of hard-task choices for self vs. other by treatment group.
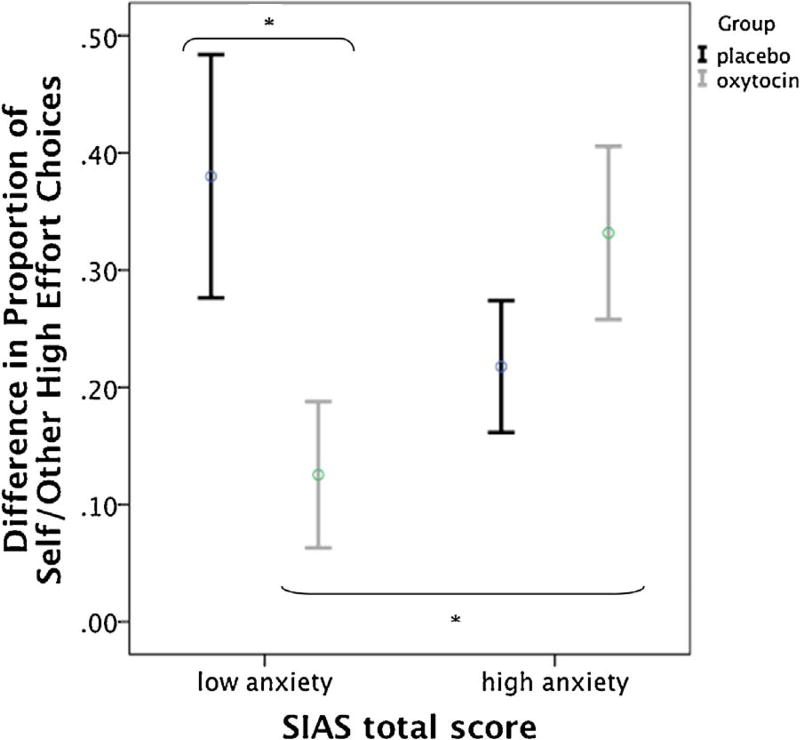
There were no differences between oxytocin and placebo groups on the motivation to work for one’s own vs. another’s rewards, as there was no drug × condition interaction effect (Wilks’ Lambda = 0.99, F (1,50) = 0.48, p = 0.490, partial ŋ2 = 0.01). Results revealed no main effect of drug (p = 0.532, partial ŋ2 = 0.008). However, there was a significant interaction between drug and baseline social anxiety severity (B = 0.02, t(50) = 3.00, p = 0.004, 95% CI: 0.007–0.034). The product term explained an additional 16.0% (ΔR2) of the variance in self-other high effort choices, ΔF(1.46) = 8.97, p = 0.004. Post-hoc comparisons revealed that within the low social anxiety group, there was a significant difference between treatment groups in self vs. other effort choices (t(23) = 2.14, p = 0.043, 95% CI: 0.009–0.501), whereas there was no difference between treatment groups within the high social anxiety group (t(23) = −1.24, p = 0.228, 95% CI: −0.304–0.076). Moreover, within the oxytocin group, there was a significant difference in self vs. other effort choices between the high and low social anxiety groups (t(23) = −2.14, p = 0.043, 95% CI: −0.405–−0.007), but there was no difference within the placebo group (t(23) = 1.41, p= 0.173, 95% CI: −0.077–0.401). Thus, for those with less severe social anxiety symptoms, receiving oxytocin led to smaller differences in self vs. other choices (less self-oriented behavior), compared to those with greater social anxiety symptoms (Fig. 4). Groups were comparable in levels of social anxiety between oxytocin and placebo-treated groups (randomized to oxytocin: low social anxiety M = 42.85, SD = 6.19, n = 13, high social anxiety M = 58.58, SD = 7.18, n = 12; randomized to placebo: low social anxiety M = 38.67, SD = 9.75, n = 12, high social anxiety M = 56.54, SD = 5.61, n = 13). The effect of oxytocin on motivation to work for one’s own rewards varied as a function of social anxiety severity, where oxytocin exerted the strongest effects on other-directed rewards for those within a lower range of social anxiety severity. Although this finding was not supported using the overall LSAS total score, the social interaction factor of the LSAS (as identified by Safren et al. (1999)) did significantly moderate the effect of oxytocin in the same direction as the SIAS, B = 0.05, t(52) = 2.08, p= 0.043, 95% CI: 0.002–0.088, thus supporting our SIAS result that social interaction anxiety moderates oxytocin’s effects on high effort choices in the EEfRT.
Fig. 4.
Significant interaction of drug × social anxiety symptom severity on difference in proportions of high effort choices for self vs. other conditions. Error bars represent 95% CI.
In addition, there was a significant interaction between drug and baseline attachment anxiety (B = 0.276, t(25) = 2.443, p = 0.025, 95% CI: 0.040–0.513). No interaction was found between drug and baseline attachment avoidance (p = 0.209, 95% CI: −0.103–0.442). The product terms (for anxious and avoidant attachment styles) explained an additional 23.9% (ΔR2) of the variance in self-other high effort choices, ΔF(2,19) = 3.155, p = 0.066. Post-hoc comparisons revealed that there were no significant differences between treatment groups in self vs. other effort choices within the low anxious attachment group (p = 0.115, 95% CI: −0.070–0.564) and within the high anxious attachment group (p = 0.125, 95% CI: −0.664–0.094). There were also no differences in self vs. other effort choices between high and low anxious attachment groups within the placebo condition (p = 0.067, 95% CI: −0.027–0.646) and within the oxytocin condition (p = 0.194, 95% CI: −0.576–0.131). Post-hoc comparisons showed that the moderating effect of attachment anxiety on self other reward motivation was not robust.
Given that 7 participants in our sample (4 assigned to placebo, 3 assigned to oxytocin) were taking antidepressant medication (at a stable dose for 2 weeks prior to study entry), we conducted separate post-hoc analyses to examine effect of medication status on reward choices. In addition, 25 participants had at least one comorbid disorder. We conducted our analysis controlling for both number of comorbidities and medication status, and our findings were the same. Social anxiety symptom severity still significantly moderated the effect of drug, whereas attachment orientation did not.
4. Discussion
Consistent with earlier studies, our findings point to meaningful differences within a sample of SAD individuals based on their response to oxytocin (Bartz et al., 2011; Olff et al., 2013). Although there was no main effect of oxytocin on social behavior, our moderator results suggest that oxytocin enhances other-oriented reward motivation only for individuals with less severe social interaction anxiety as compared to those with high levels of social interaction anxiety. It is possible that for individuals who were less socially anxious, oxytocin facilitated motivation to reward the other participant by reducing social withdrawal-related behaviors, which is consistent with some prior work showing that oxytocin increases generosity by 80% relative to a placebo in the ultimatum game (Zak et al., 2007). In keeping with the notion that oxytocin potentiates a self-processing bias (Hurlemann & Scheele, 2016; Quattrocki & Friston, 2014), it has been shown to sharpen the boundary between self and other (Colonell et al., 2013). Our data suggest that oxytocin may be potentially beneficial for promoting other-oriented behavior, only in some individuals with less severe symptoms. This is consistent with research showing a more pronounced effect of oxytocin in individuals with low autism traits (Scheele et al., 2014), but contrasts with other results showing that oxytocin selectively improves empathy for those who are less socially proficient than those who are more socially proficient (Bartz et al., 2010).
Our findings are also consistent with a body of literature demonstrating that individuals with SAD exhibit alterations in corticostriatal circuits known to support reward-related behavior. Compared to healthy controls, individuals with generalized SAD show decreased resting state functional connectivity between the nucleus accumbens and the ventromedial prefrontal cortex, regions associated with reward (Manning et al., 2015). Additionally, healthy controls demonstrate striatal responses that were modulated by partner reputations (for cooperative and uncooperative partners) in a social exchange game, whereas individuals with generalized SAD show no differences in striatal responses by partner reputation (Sripada, Angstadt, Liberzon, McCabe, & Phan, 2013).
Interestingly, our results indicated that insecure attachment orientation did not moderate the effects of oxytocin on self versus other reward choices. This contrasts with our previous finding in individuals with SAD that oxytocin’s effect on social cooperation was most potent for those who were low in attachment avoidance (Fang et al., 2014). Future research should examine the attachment-dependent effects of oxytocin on social cognition in SAD, as a recent study found a strong association between the oxytocin receptor genotype (rs53576) and insecure attachment among healthy individuals at risk for social anxiety (Notzon et al., 2016).
Some limitations of our study warrant mention. First, we did not include a healthy control group. However, diagnostic categories themselves are not meaningful with respect to underlying etiology and maintenance factors, and any differences between diagnostic and healthy groups would not reveal information about mechanisms associated with reward motivation. Future research should include a healthy control group to examine if the effects of oxytocin on self-other reward motivation are specific to SAD. Second, it is possible but unlikely that our findings could have been influenced by the Cyberball manipulation, which preceded the EEfRT paradigm. We examined correlations between ball tosses (from the Cyberball task) and high effort choices for self vs. other conditions (from the EEfRT task) by high vs. low anxiety levels, which revealed no correlation (all p’s > 0.05). It is also unlikely that the lack of effect in the high anxious group was due to the need to reciprocate, as subjects were not aware of whether previous subjects worked harder for themselves or others prior to completing the EEfRT. However, future research should assess whether trial-by-trial information on a running total of money for Self and Other trials may influence responses in a more equitable manner. Furthermore, it is unlikely that the lack of effect in the high anxious group was due to greater stress associated with the task because subjects were able to complete their trials and rarely timed out when deciding whether to play the Hard or Easy task. Lastly, there is ongoing controversy regarding the mechanisms by which intranasal delivery of oxytocin exerts behavioral effects, as evidence is still lacking that oxytocin (when delivered intranasally) actually reaches its presumed central targets (Quintana & Woolley, 2015). Related to this issue are the uncertainties that remain regarding the optimal dosing and optimal time point for testing oxytocin’s effects, as recent evidence has shown that peak CSF levels in humans actually occur 75 min post intranasal administration (Striepens et al., 2013).
In sum, the current study revealed a novel moderation effect of oxytocin on pro-social motivation in patients with different severity levels of SAD symptoms. These findings may lead to an improved psychiatric nosology in line with RDoC (Insel et al., 2010) and possibly enhanced treatments, given the moderate efficacy of existing interventions (Hofmann & Smits, 2008).
Supplementary Material
Acknowledgments
Funding sources and acknowledgments
This work was supported by the Clara Mayo Memorial Fellowship at Boston University, Weil Dissertation Award, and National Institute of Mental Health (K23 MH109593) to AF, and by the National Institute of Mental Health (R00MH102355) to MTT. Dr. Hofmann receives support from NIH/NCCIH (R01AT007257), NIH/NIMH (R01MH099021, R34MH099311, R34MH086668, R21MH102646, R21MH101567, K23MH100259)the James S. McDonnell Fundation 21st Century Science Initiative in Understanding Human Cognition – Special Initiative, and the Department of the Army. He receives compensation for his work as an advisor from the Palo Alto Health Sciences and Otsuka Digital Health, Inc., and for his work as a Subject Matter Expert from John Wiley & Sons, Inc. and SilverCloud Health, Inc. He also receives royalties and payments for his editorial work from various publishers. Dr. Treadway has served as a paid consultant to Avanir Pharmaceuticals and the Boston Consulting Group. No funding or sponsorship was provided by these companies for the current work, and all views expressed herein are solely those of the authors. The authors would like to thank Daniel Brager, M.A., Türküler Aka, Ph.D., Sheina Godovich, B.A., Danielle Cooper, M.A., Brian Gambardella, B.A., James Steinberg, B.A., Katherine Eisenmenger, M.D., and Bonnie Brown, R.N., at Boston University for their assistance in running the protocol.
Footnotes
The authors declare that they have no competing financial interests.
Appendix A. Supplementary data
Supplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.biopsycho.2017.05.015.
References
- Bartz JA, Zaki J, Bolger N, Hollander E, Ludwig NN, Kolevzon A, Ochsner KN. Oxytocin selectively improves empathic accuracy. Psychological Science. 2010;21:1426–1428. doi: 10.1177/0956797610383439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartz JA, Zaki J, Bolger N, Ochsner KN. Social effects of oxytocin in humans: Context and person matter. Trends in Cognitive Sciences. 2011;15:301–309. doi: 10.1016/j.tics.2011.05.002. [DOI] [PubMed] [Google Scholar]
- Brennan KA, Clark CL, Shaver PR. Self-report measurement of adult attachment: An integrative overview. In: Simpson JA, Rholes WS, editors. Attachment theory and close relationships. New York: Guilford Press; 1998. pp. 46–76. [Google Scholar]
- Clark DB, Feske U, Masia CL, Spaulding SA, Brown C, Mammen O, Shear MK. Systematic assessment of social phobia in clinical practice. Depression and Anxiety. 1997;6:47–61. doi: 10.1002/(sici)1520-6394(1997)6:2<47::aid-da1>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- Colonello V, Chen FS, Panksepp J, Heinrichs M. Oxytocin sharpens self-other perceptual boundary. Psychoneuroendocrinology. 2013;38:2996–3002. doi: 10.1016/j.psyneuen.2013.08.010. [DOI] [PubMed] [Google Scholar]
- Colonnello V, Heinrichs M. Intranasal oxytocin enhances positive self-attribution in healthy men. Journal of Psychosomatic Research. 2014;77:415–419. doi: 10.1016/j.jpsychores.2014.06.016. [DOI] [PubMed] [Google Scholar]
- De Dreu CKW, Greer LL, Handgraaf MJJ, Shalvi S, Van Kleef GA, Baas M, Feith SWW. The neuropeptide oxytocin regulates parochial altruism in intergroup conflict among humans. Science. 2010;328:1408–1411. doi: 10.1126/science.1189047. [DOI] [PubMed] [Google Scholar]
- De Waal FBM. Putting the altruism back into altruism: The evolution of empathy. Annual Review of Psychology. 2008;59:279–300. doi: 10.1146/annurev.psych.59.103006.093625. [DOI] [PubMed] [Google Scholar]
- DiNardo PA, Brown TA, Barlow DH. Anxiety disorders interview schedule for DSM-IV: Client interview schedule. New York: Oxford University Press; 1994. [Google Scholar]
- Dumais KM, Veenema AH. Vasopressin and oxytocin receptor systems in the brain: Sex differences and sex-specific regulation of social behavior. Frontiers in Neuroendocrinology. 2015;40:1–23. doi: 10.1016/j.yfrne.2015.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg N, Miller PA. The relation of empathy to prosocial and related behaviors. Psychological Bulletin. 1987;101:91–119. [PubMed] [Google Scholar]
- Fang A, Hoge EA, Heinrichs M, Hofmann SG. Attachment style moderates the effects of oxytocin on social behaviors and cognitions during social rejection: Applying a research domain criteria framework to social anxiety. Clinical Psychological Science. 2014;2:740–747. doi: 10.1177/2167702614527948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guastella AJ, Einfeld SL, Gray KM, Rinehart NJ, Tonge BJ, Lambert TJ, Hickie IB. A randomized controlled trial of intranasal oxytocin as an adjunct to exposure therapy for social anxiety disorder. Psychoneuroendocrinology. 2009;34:917–923. doi: 10.1016/j.psyneuen.2009.01.005. [DOI] [PubMed] [Google Scholar]
- Heinrichs M, Baumgartner T, Kirschbaum C, Ehlert U. Social support and oxytocin interact to suppress cortisol and subjective responses to psychosocial stress. Biological Psychiatry. 2003;54:1389–1398. doi: 10.1016/s0006-3223(03)00465-7. [DOI] [PubMed] [Google Scholar]
- Hofmann SG, Smits JA. Cognitive-behavioral therapy for adult anxiety disorders: A meta-analysis of randomized placebo-controlled trials. Journal of Clinical Psychiatry. 2008;69:621–632. doi: 10.4088/jcp.v69n0415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofmann SG. Cognitive factors that maintain social anxiety disorder: A comprehensive model and its treatment implications. Cognitive Behaviour Therapy. 2007;36:193–209. doi: 10.1080/16506070701421313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurlemann R, Scheele D. Dissecting the role of oxytocin in the formation and loss of social relationships. Biological Psychiatry. 2016;79:185–193. doi: 10.1016/j.biopsych.2015.05.013. [DOI] [PubMed] [Google Scholar]
- Hurlemann R, Patin A, Onur OA, Cohen MX, Baumgartner T, Metzler S, Kendrick KM. Oxytocin enhances amygdala-dependent:socially reinforced learning and emotional empathy in humans. Journal of Neuroscience. 2010;30:4999–5007. doi: 10.1523/JNEUROSCI.5538-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingram RE. Self-focused attention in clinical disorders: Review and a conceptual model. Psychological Bulletin. 1990;107(2):156–176. doi: 10.1037/0033-2909.107.2.156. [DOI] [PubMed] [Google Scholar]
- Insel T, Cuthbert B, Garvey M, Heinssen R, Pine DS, Quinn K, Wang P. Research domain criteria (RDoC): Toward a new classification framework for research on mental disorders. American Journal of Psychiatry. 2010;167:748–751. doi: 10.1176/appi.ajp.2010.09091379. [DOI] [PubMed] [Google Scholar]
- Kemp AH, Guastella AJ. The role of oxytocin in human affect: A novel hypothesis. Current Directions in Psychological Science. 2011;20:222–231. [Google Scholar]
- Kosfeld M, Heinrichs M, Zak PJ, Fischbacher U, Fehr E. Oxytocin increases trust in humans. Nature. 2005;435:673–676. doi: 10.1038/nature03701. [DOI] [PubMed] [Google Scholar]
- Labuschagne I, Phan KL, Wood A, Angstadt M, Chua P, Heinrichs M, Nathan PJ. Oxytocin attenuates amygdala reactivity to fear in generalized social anxiety disorder. Neuropsychopharmacology. 2010;35:2403–2413. doi: 10.1038/npp.2010.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liebowitz MR. Social phobia. Modern Problems in Pharmacopsychiatry. 1987;22:141–173. doi: 10.1159/000414022. [DOI] [PubMed] [Google Scholar]
- Manning J, Reynolds G, Saygin ZM, Hofmann SG, Pollack M, Gabrieli JDE, Whitfield-Gabrieli S. Altered resting-state functional connectivity of the frontal-striatal reward system in social anxiety disorder. PLoS ONE. 2015;10:e0125286. doi: 10.1371/journal.pone.0125286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattick RP, Clarke JC. Development and validation of measures of social phobia scrutiny fear and social interaction anxiety. Behaviour Research and Therapy. 1998;36:455–470. doi: 10.1016/s0005-7967(97)10031-6. [DOI] [PubMed] [Google Scholar]
- Notzon S, Domschke K, Holitschke K, Ziegler C, Arolt V, Pauli P, Zwanzger P. Attachment style and oxytocin receptor gene variation interact in influencing social anxiety. World Journal of Biological Psychiatry. 2016;17:76–83. doi: 10.3109/15622975.2015.1091502. [DOI] [PubMed] [Google Scholar]
- Olff M, Frijling JL, Kubzansky LD, Bradley B, Ellenbogen MA, Cardoso C, van Zuiden M. The role of oxytocin in social bonding, stress regulation and mental health: An update on the moderating effects of context and interindividual differences. Psychoneuroendocrinology. 2013;38:1863–1894. doi: 10.1016/j.psyneuen.2013.06.019. [DOI] [PubMed] [Google Scholar]
- Posner MI. Orienting of attention. Quarterly Journal of Experimental Psychology. 1980;32:3–25. doi: 10.1080/00335558008248231. [DOI] [PubMed] [Google Scholar]
- Quattrocki E, Friston K. Autism, oxytocin and interoception. Neuroscience and Biobehavioral Reviews. 2014;47:410–430. doi: 10.1016/j.neubiorev.2014.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quintana DS, Woolley JD. Intranasal oxytocin mechanisms can be better understood, but its effects on social cognition and behavior are not to be sniffed at. Biological Psychiatry. 2015;79:e49–50. doi: 10.1016/j.biopsych.2015.06.021. [DOI] [PubMed] [Google Scholar]
- Safren SA, Heimberg RG, Horner KJ, Juster HR, Schneier FR, Liebowitz MR. Factor structure of social fears: The liebowitz social anxiety scale. Journal of Anxiety Disorders. 1999;13:253–270. doi: 10.1016/s0887-6185(99)00003-1. [DOI] [PubMed] [Google Scholar]
- Scheele D, Kendrick KM, Khouri C, Kretzer E, Schläpfer TE, Stoffel-Wagner B, Hurlemann R. An oxytocin-induced facilitation of neural and emotional responses to social touch correlates inversely with autism traits. Neuropsychopharmacology. 2014;39:2078–2085. doi: 10.1038/npp.2014.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahrestani S, Kemp AH, Guastella AJ. The impact of a single administration of intranasal oxytocin on the recognition of basic emotions in humans: A meta-analysis. Neuropsychopharmacology. 2013;38:1929–1936. doi: 10.1038/npp.2013.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shamay-Tsoory SG, Abu-Akel A. The social salience hypothesis of oxytocin. Biological Psychiatry. 2016;79:194–202. doi: 10.1016/j.biopsych.2015.07.020. [DOI] [PubMed] [Google Scholar]
- Shamay-Tsoory SG, Fischer M, Dvash J, Harari H, Perach-Bloom N, Levkovitz Y. Intranasal administration of oxytocin increases envy and schadenfreude (gloating) Biological Psychiatry. 2009;66:864–870. doi: 10.1016/j.biopsych.2009.06.009. [DOI] [PubMed] [Google Scholar]
- Shamay-Tsoory SG. Oxytocin, social salience, and social approach. Biological Psychiatry. 2010;67:e35. doi: 10.1016/j.biopsych.2015.07.020. [DOI] [PubMed] [Google Scholar]
- Spurr JM, Stopa L. Self-focused attention in social phobia and social anxiety. Clinical Psychology Review. 2002;22:947–975. doi: 10.1016/s0272-7358(02)00107-1. [DOI] [PubMed] [Google Scholar]
- Sripada C, Angstadt M, Liberzon I, McCabe K, Phan KL. Aberrant reward center response to partner reputation during a social exchange game in generalized social phobia. Depression and Anxiety. 2013;30:353–361. doi: 10.1002/da.22091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Striepens N, Kendrick KM, Hanking V, Landgraf R, Wüllner U, Maier W, Hurlemann R. Elevated cerebrospinal fluid and blood concentrations of oxytocin following its intranasal administration in humans. Scientific Reports. 2013;3:3440. doi: 10.1038/srep03440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Treadway MT, Buckholtz JW, Schwartzman AN, Lambert WE, Zald DH. Worth the ‘EEfRT’? The effort expenditure for rewards task as an objective measure of motivation and anhedonia. PLoS ONE. 2009;4:1–9. doi: 10.1371/journal.pone.0006598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams KD, Cheung CKT, Choi W. Cyberostracism: Effects of being ignored over the Internet. Journal of Personality and Social Psychology. 2000;79:748–762. doi: 10.1037//0022-3514.79.5.748. [DOI] [PubMed] [Google Scholar]
- Zak PJ, Stanton AA, Ahmadi S. Oxytocin increases generosity in humans. PloS One. 2007;2:e1128. doi: 10.1371/journal.pone.0001128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao W, Yao S, Li Q, Geng Y, Ma X, Luo L, Kendrick KM. Oxytocin blurs the self-other distinction during trait judgments and reduces medial prefrontal cortex responses. Human Brain Mapping. 2016;37:2512–2527. doi: 10.1002/hbm.23190. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.