Abstract
Idiopathic portal hypertension is a disorder that has various clinical features. It is mostly characterized by bleeding oesophageal varices, obvious splenomegaly, anaemia and, occasionally, jaundice and ascites. Here we described an interesting case of idiopathic portal hypertension caused by coeliac disease in a 38-year-old woman. By putting this patient on a gluten-free diet, liver function tests became normal and portal vein diameter returned to normal range. This report indicates that, in coeliac disease, repetitive stimulation by antigens along the portal vein—and immune responses to them—can result in the development of idiopathic portal hypertension.
Keywords: idiopathic portal hypertension, coeliac disease
Introduction
Coeliac disease is also known as coeliac sprue or gluten-sensitive enteropathy. Immune responses to gluten cause mucosal damage. Stimulation of the immune system is involved in pathogenesis of the disease. Non-cirrhotic (i.e. idiopathic) portal hypertension is a heterogeneous disorder with varying clinical features, and it is mainly demonstrated by signs and symptoms of portal hypertension in the absence of obvious hepatic disorder [1]. The aetiology of the disease is still unknown. There are various theories about the cause of this disease, such as autoimmunity disorder, chemical elements effect, infections, thrombosis, and hereditary causes [1, 2]. There are few cases of the co-existence of coeliac and hepatic disorders [3, 4]; here we report the interesting case of a 38-year-old woman with both idiopathic portal hypertension and coeliac disease.
Case presentation
The patient was a 38-year-old married woman who had for several years been under observation because of ascites, splenomegaly and general weakness. She had been under treatment for possible cryptogenic cirrhosis and had received medication. When she was referred to our centre, we began our evaluation with a complete history of her existing and past problems and she mentioned several previous admissions for the purpose of evaluating her current problems. She had been taking Aldactone (spironolactone) for years. No history of hepatic or other gastrointestinal problems was found in her first-degree relatives. Her vital signs were normal and stable. She was mildly pale, with no sign of jaundice. Auscultation found lungs and heart normal. An obvious splenomegaly was detected in her abdomen. She had a mild oedema in the upper and lower limbs. We then requested laboratory tests: the results of autoimmunity tests, antinuclear antibody (ANA), anti-smooth muscle antibody (ASMA), and serum protein electrophoresis were reported to be normal. None of the laboratory findings indicated Wilson’s disease.
Ultrasonography of the abdomen showed heterogeneous changes of liver parenchyma, hepatosplenomegaly and high portal vein pressure (15 mmHg). Doppler ultrasonography of the portal vein then found no signs of thrombosis in the portal system. Ultrasonography had previously been carried out at other medical centres and backed up these findings. We carried out an upper gastrointestinal (GI) endoscopy and found grade 2 oesophageal varices. In accordance with our findings—and also according to clinical suspicion—we asked for a liver biopsy. The pathologic findings indicated mild chronic hepatitis, stage zero, grade 2, a hepatic activity index (HAI) score of 2 and no cirrhosis or malignancy.
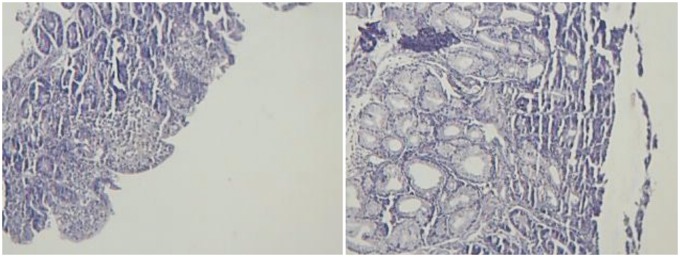
As our next step, serology tests for coeliac disease were requested. Anti-tissue transglutaminase (ATTG) was 18.8 (normal range <15), anti-endomesial antibody (AEA) was 27.8 (normal range <20). Histological findings of biopsy taken from duodenum showed flattening of mucosal villi with secretion of chronic inflammatory cells and neutrophils, equal to a Marsh score of 3 (Figure 1) and confirmed the presence of coeliac disease.
Figure 1.
Duodenal biopsy shows flattening of mucosal villi with secretion of chronic inflammatory cells and neutrophils.
In accordance with these findings, a gluten-free diet was prescribed for the patient and after 3 months her general health was improved and the results of laboratory tests results returned to normal ranges. Also in ultrasonography work-ups, signs of portal hypertension were relieved and the diameters of the portal vein and common bile duct were 8 mm and 4 mm, respectively. Spleen size was normal and no ascites were detected. A serology test result showed a 50% decrease in AEA titration, although still not in normal range.
Discussion
This has been a report of a case of coeliac disease presented by portal hypertension. Coeliac disease is intestinal dysfunction caused by gluten intolerance; the condition has various clinical presentations [5, 6]: in adults, the common symptoms are gastrointestinal disorders such as diarrhoea, fatty stool and weight loss, which are dependent on the severity of enteropathy. Other than the typical gastrointestinal presentations, coeliac disease has several extra-gastrointestinal presentations, e.g. neurological symptoms, ataxia [7], epilepsy [8], hepatobiliary disorders (PSC, autoimmune hepatitis) [3, 4, 9] and cirrhosis, dermopathy, infertility and anaemia [4, 10]; it is a little more common in women than in men. The prevalence of coeliac disease in the Middle East is similar to that in western countries [3, 11]. Work-up starts with clinical suspicion of the existence of coeliac disease [5, 6], after which serological tests will be requested, e.g. anti-endomesial antibody (AEA) and anti-tissue transglutaminase (ATTG). If the test results are positive, a biopsy of the small intestine is necessary to verify the diagnosis.
Idiopathic portal hypertension occurs secondary to microvascular occlusion of intra-hepatic small portal vein radicles. Multiple hypotheses regarding its pathogenesis have been proposed; one such entails the linking of gut disorders and subsequent hyper-coagulability in portal circulation. The role of a deficiency of ADAMTS 13 (a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13), a von Willebrand factor-cleaving protease, is also being actively explored [12]. Ordinarily, in the first stages of disease, the liver function remains normal (normal albumin level and normal prothrombin time) but bleeding from oesophageal varices (92%), splenomegaly (96.7%), anaemia (26.2%) and hematemesis (23.7%) then occur [13]. Hepatic failure and its symptoms occur in the last stages of the disease. Other symptoms include ascites, hepatic encephalopathy and jaundice. The prognosis for the disease relies on its progression and is often good. A study made in Japan showed that, among persons affected by portal hypertension, the cause of 30% of deaths was bleeding from oesophageal varices, and that 25% were from hepatic failure [14].
The relationships of both primary biliary cirrhosis and autoimmune hepatitis to coeliac disease have been emphasized [15–17], but the relationship of coeliac disease to idiopathic portal hypertension has very rarely been pointed out in case reports [18–20]. In 2008, the Gastroenterology Center of Firuzgar Hospital (Tehran, Iran) also reported a case of coeliac disease with portal hypertension presentation, which was cured by gluten-free diet [20]. Some other reports have mentioned the concurrence of coeliac disease, splenic vein thrombosis and Budd-Chiari syndrome [21, 22]. The probable causes of non-cirrhotic portal hypertension in coeliacs are immune mechanisms stimulating immune mediators in the portal system [18, 19].
The concurrence of immune disorders and coeliac disease may account for the production of some immune complexes in the intestines of coeliac patients and would answer questions about variable presentations of this malabsorption syndrome. There are similar findings in idiopathic portal hypertension and coeliac disease which are caused by abnormal immune mediators (T-cells and some antibodies) [2]. In some past studies, repetitive antigen stimulations along the portal vein have been recognised as the possible cause of idiopathic portal hypertension [23].
Portal hypertension has traditionally been viewed as a progressive process, involving ultrastructural changes including fibrosis, nodule formation, and vascular thrombosis, leading to increased intrahepatic resistance to flow; however, it is increasingly recognized that a significant component of this vascular resistance results from a dynamic process, regulated by complex interactions between the injured hepatocyte, the sinusoidal endothelial cell, the Kupffer cell and the hepatic stellate cell, which impact on sinusoidal calibre [24].
The treatment of coeliac disease is based on eliminating gluten from the patient’s diet. The main sources of gluten are wheat, oats and secale. Gluten is also found in the capsules of medications [5, 25]. An untreated coeliac disorder results in lactase deficiency and intestinal epithelial cell damages so, to treat the disease, the patient should avoid using dairy and milk products [25]: many symptoms of the disease will be relieved by this dietary regimen. Our patient improved to a normal and healthy state after 3 months on a gluten-free diet. Laboratory findings and ultrasonographic studies showed that the portal hypertension disorder had been relieved.
The reporting of similar cases will increasingly clarify the relationship between coeliac disease and portal hypertension, leading to an aetiological approach to hepatic disorders and also coeliac disease in clinical work-ups.
Acknowledgements
We wish to thank Dr. M. R. Mosadeq and Dr. Javaherian for their great help in laboratory studies.
Conflict of interest statement: none declared.
References
- 1. Sarin SK. Non-cirrhotic portal fibrosis. Gut 1989;30:406–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Sarin SK, Aggarwal SR. Idiopathic portal hypertension. Digestion 1998;59:420–3. [DOI] [PubMed] [Google Scholar]
- 3. Shahbazkhani B, Malekzadeh R, Sotoudeh M, et al. High prevalence of coeliac disease in apparently healthy Iranian blood donors. Eur J Gastroenterol Hepatol 2003;15:475–8. [DOI] [PubMed] [Google Scholar]
- 4. Meloni GF, Dessole S, Vargiu N, et al. The prevalence of coeliac disease in infertility. Hum Reprod 1999;14:2759–61. [DOI] [PubMed] [Google Scholar]
- 5. James MW, Scott BB. Coeliac disease: the cause of the various associated disorders? Eur J Gastroenterol Hepatol 2001;13:1119–21. [DOI] [PubMed] [Google Scholar]
- 6. Scott BB, Losowski MS. Celiac disease: a cause of various associated disease? Lancet 1975;2:956–7. [DOI] [PubMed] [Google Scholar]
- 7. Luostarinen L, Himanen SL, Luostarinen M, et al. Neuromuscular and sensory disturbances in patients with well treated coeliac disease. J Neurol Neurosurg Psychiatry 2003;74:490–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gobbi G, Bouquet F, Greco L, et al. Coeliac disease, epilepsy, and cerebral calcifications. The Italian Working Group on Coeliac Disease and Epilepsy. Lancet 1992;340:439–43. [DOI] [PubMed] [Google Scholar]
- 9. Kaukinen K, Halme L, Collin P, et al. Celiac disease in patients with severe liver disease: gluten-free diet may reverse hepatic failure. Gastroenterology 2002;122:881–8. [DOI] [PubMed] [Google Scholar]
- 10. Collin P, Vilska S, Heinonen PK, et al. Infertility and coeliac disease. Gut 1996;39:382–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Akbari MR, Mohammadkhani A, Fakheri H, et al. Screening of the adult population in Iran for coeliac disease: comparison of the tissue-transglutaminase antibody and anti-endomysial antibody tests. Eur J Gastroenterol Hepatol 2006;18:1181–6. [DOI] [PubMed] [Google Scholar]
- 12. Goel A, Madhu K, Zachariah U, et al. A study of aetiology of portal hypertension in adults (including the elderly) at a tertiary centre in southern India. Indian J Med Res 2013;137:922–7. [PMC free article] [PubMed] [Google Scholar]
- 13. Dhiman RK, Chawla Y, Vasishta RK, et al. Non-cirrhotic portal fibrosis (idiopathic portal hypertension): experience with 151 patients and a review of the literature. J Gastroenterol Hepatol 2002;17:6–16. [DOI] [PubMed] [Google Scholar]
- 14. Ichimura S, Sasaki R, Takemura Y, et al. The prognosis of idiopathic portal hypertension in Japan. Intern Med 1993;32:441–4. [DOI] [PubMed] [Google Scholar]
- 15. Kingham JG, Parker DR. The association between primery biliary cirrhosis and celiac disease: a study of relative prevalences. Gut 1998;42:120–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Caiulo VA, Averbene C, Olmi S, et al. Celiac disease associated with chronic autoimmune hepatitis. Description of a Case.. Minerve Pediatr 1993;45:511–13. [PubMed] [Google Scholar]
- 17. Abdo A, Meddings J, Swain M. Liver abnormalities in celiac disease. Clin Gastroenterol Hepatol 2004;2:107–12. [DOI] [PubMed] [Google Scholar]
- 18. M'saddek F, Gaha K, Ben Hammouda R, et al. Idiopathic portal hypertension associated with celiac disease: one case. Gastroenterol Clin Biol 2007;31:869–71. [DOI] [PubMed] [Google Scholar]
- 19. Sharma BC, Bhasin DK, Nada R. Association of celiac disease with non-cirrhotic portal fibrosis. J Gastroenterol Hepatol 2006;21:332–4. [DOI] [PubMed] [Google Scholar]
- 20. Zamani F, Amiri A, Shakeri R, et al. Celiac disease as a potential cause of idiopathic portal hypertension: a case report. J Medical Case Report 2009;3:68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ghoshal UC, Saraswat VA, Yachha SK. Association of splenic vein obstruction and celiac disease in an Indian patient. J Hepatol 1995;23:358. [PubMed] [Google Scholar]
- 22. Hamdi A, Ayachi R, Saad H, et al. Hemiplegia revealing Budd-Chiari syndrome associated with celiac disease in an infant. Presse Med 1990;19:1011–12. [PubMed] [Google Scholar]
- 23. Okabayashi A. Experimental production of non-cirrhotic portal hypertension and splenomegaly with prolonged antigenic stimulation. Tokyo Press 1983;175–93. [Google Scholar]
- 24. Mehta G, Gustot T, Mookerjee RP, et al. Inflammation and portal hypertension: the undiscovered country. J Hepatol 2014;61:155–63. [DOI] [PubMed] [Google Scholar]
- 25. Aletaha N, Poorshams A, Shahbazkhani B. Celiac Disease. J Med Coucil of Islamic Republic of Iran 2008;25:213–24. [Google Scholar]