Abstract
Collagen IV is a major component of the basement membrane in epithelial tissues. The NC1 domains of collagen IV protomers are covalently linked together through sulfilimine bonds, the formation of which is catalyzed by peroxidasin. Although hydrogen peroxide is essential for this reaction, the exact source of the oxidant remains elusive. Members of the NOX/DUOX NADPH oxidase family are specifically devoted to the production of superoxide and hydrogen peroxide. Our aim in this study was to find out if NADPH oxidases contribute in vivo to the formation of collagen IV sulfilimine crosslinks. We used multiple genetically modified in vivo model systems to provide a detailed assessment of this question. Our data indicate that in various peroxidasin-expressing tissues sulfilimine crosslinks between the NC1 domains of collagen IV can be readily detected in the absence of functioning NADPH oxidases. We also analyzed how subatmospheric oxygen levels influence the collagen IV network in collagen-producing cultured cells with rapid matrix turnover. We showed that collagen IV crosslinks remain intact even under strongly hypoxic conditions. Our hypothesis is that during collagen IV network formation PXDN cooperates with a NOX/DUOX-independent H2O2 source that is functional also at very low ambient oxygen levels.
Abbreviations: col4a, collagen type IV alpha; DUOX, Dual oxidase; DUOXA, Dual oxidase Activator; NC1, non-collagenous; HIF-1alpha, Hypoxia-inducible Factor 1-alpha, Nox: NADPH oxidase; PHG, phloroglucinol; PXDN, peroxidasin
Keywords: Peroxidasin, NADPH oxidase, Hydrogen peroxide, Collagen IV, Sulfilimine
1. Introduction
Basement membranes are extracellular structures that are essential in organizing individual cells into tissues and organs [39]. Besides providing mechanical support for different cell types, including epithelial and endothelial cells, basement membranes are also important in cellular signaling. An essential core component of basement membranes is collagen IV, which is organized into a sheet-like network through covalent binding between individual collagen IV triplexes [14]. Sulfilimine bond is a unique type of chemical link which is so far only found in collagen IV molecules in living organisms [34]. The formation of this recently identified chemical bond is catalyzed by peroxidasin (PXDN), a member of the animal heme peroxidase family [4], [26].
First, in the presence of hydrogen peroxide (H2O2), PXDN turns bromide into hypobromous acid (HOBr), which then reacts with a methionine in the NC1 domain of collagen IV forming a methionine halosulfonium intermediate [4], [22], [28]. Finally, this intermediate reacts with a hydroxylysine on the opposing protomers’ NC1 domain to create the sulfilimine crosslink between neighboring protomers.
While bromine is a ubiquitously accessible trace element in animal tissues, the exact source of H2O2 has not been identified yet. There are numerous metabolic and signaling pathways the activity of which are accompanied by the generation of reactive oxygen species (ROS) [35], [37]. There are also some extracellular enzymes which are also known to produce H2O2 while accomplishing their catalytic functions (e.g. lysyl oxidases and their homologs). The family of NADPH oxidases comprises enzymes that are specifically devoted to the generation of superoxide anion and H2O2 [2], [31]. There are seven members of the NOX/DUOX family of NADPH oxidases, including NOX1–5, DUOX1, and DUOX2. NOX/DUOX enzymes show unique expression pattern and biochemical regulation [2], [6]. When exerting its pleiotropic effects H2O2 can directly react with and modify target molecules. Some actions of H2O2, however, are mediated through animal heme peroxidases, which use H2O2 to oxidize substrates in a diverse array of reactions [9]. The group of mammalian heme peroxidases includes myeloperoxidase (MPO), eosinophil peroxidase (EPX), lactoperoxidase (LPO), thyroperoxidase (TPO) and peroxidasin (PXDN) [9], [32]. Peroxidases are often expressed in cells and tissues, where NOX/DUOX NADPH oxidases can also be found and there are a number of examples for the cooperation between NADPH oxidases and heme peroxidases. For example, in neutrophil granulocytes, MPO uses H2O2 produced by the NOX2 complex to oxidize chloride into hypochlorous acid, which is a potent antimicrobial compound [15]. NOX2 is also highly expressed in eosinophils, where EPX utilizes its product for the oxidation of bromide and thiocyanate [36]. The oxidation of iodide in the thyroid is also dependent on the cooperation between a peroxidase and an NADPH oxidase, namely TPO and DUOX2. While DUOXes were originally considered to be specific for the thyroid, subsequent research has uncovered they role in antimicrobial host defense where they supply H2O2 for lactoperoxidase, an enzyme expressed in different exocrine glands, including the salivary gland and submucosal glands of large airways [12].
For a complete understanding of collagen IV role in basement membrane biosynthesis, it would be crucial to determine the biochemical and mechanical importance of sulfilimine crosslinks. An early step towards this goal could be the characterization of the H2O2 source for PXDN-mediated crosslinks. Our primary goal in this study was to find out if members of the NOX/DUOX family participate in the formation of collagen IV sulfilimine crosslinks in mammalian tissues. Our results suggest that NADPH oxidases do not support collagen IV crosslinking and the molecular identity of the H2O2 source in collagen IV synthesis remains to be identified.
2. Materials and methods
2.1. Reagents
Collagenase type I was ordered from Worthington Biochemical Corporation. Isoform-specific rat monoclonal col4a1, col4a5 NC1 antibodies were purchased from Chondrex Inc. We used rabbit HIF1alpha antibody from Abcam (ab179483). The anti-actin antibody, phloroglucinol, diphenyliodonium were all purchased from Sigma. PFHR-9, a mouse embryonic carcinoma cell line was obtained from ATCC. For transfecting PFHR-9 cells, we used TurboFect from Thermo Scientific. AlamarBlue and calcein-AM were ordered from ThermoFisher Scientific.
Rabbit polyclonal anti-Nox4 antibody was a generous gift from Professor Ajay Shah (King's College, London) and polyclonal anti-Duox1 antibody was a kind gift from Dr. Xavier De Deken (Université Libre de Bruxelles).
2.2. Cell culture
PFHR-9 cells (obtained from ATCC) were grown in Dulbecco's Modified Eagles Medium with glutamine and 4,5 g/L glucose, supplemented with 50 U/ml penicillin and 50 µg/ml streptomycin (Lonza Group Ltd., Basel, Switzerland) and 10% fetal calf serum (Biowest SAS, France, S182P-500, French origin). Cells were grown in a humidified incubator with 5% CO2 in the air, at 37 °C.
2.3. PFHR-9 peroxidasin CRISPR
PFHR-9 cells were genetically mutated for peroxidasin, using a pSpCas9(BB)−2A-GFP (PX458) vector. The vector contained the 5′-caggcccgcttctctga-3′ guide sequence which is identical to the 408–424 bases of the 17th exon of the mouse Pxdn gene. The cells were transfected with Turbofect and GFP positive cells were sorted onto 96 well plates. Cell clones were screened by PCR of genomic DNA using 5′-acaactgctcagacatgtgc-3′ sense and 5′-acacattggaggcatcgatg-3′ antisense oligos. PCR products were analyzed by Surveyor mismatch analysis, then subcloned into a pcDNA cloning vector and sent for sequencing. One cell line was selected which contained frameshift mutations on both alleles. We confirmed by Western blot analysis that the selected cell line does not express PXDN and is not able to crosslink collagen IV NC1 domains.
2.4. Mouse strains used in the study
PXDN-deficient mice were generated by SAGE Laboratories using Compo Zr zinc finger endonuclease technology [18]. Detailed characterization of PXDN-deficient animals is subject to a different paper. The knockout line contains a 2 bp deletion at the beginning of the peroxidase domain coding region.
Duox1 knockout mice were purchased from Lexicon Pharmaceuticals, Inc. (TheWoodlands, TX, USA) and described earlier by Donkó et al. [11].
The p22phox mutant nmf333 mouse strain [23] was obtained from The Jackson Laboratory.
The Duoxa knockout mice were developed by Grasberger et al. [13].
The Nox4 knock-out animals were generated by the Transcription Activator-Like Effector Nuclease (TALEN) technique using plasmids ordered from Addgene (TALE Toolbox Kit # 1000000019 deposited by Feng Zhang). Mouse Nox4 specific TALEN recognition sites were designed for the genomic sequence prior to and to the first exon using the 5`-TN19 N14–20 N19A-3` formula, with the following sequences: left TALEN recognition site: 5′TCCCCGCGCCGGCGGCATGG3′, right TALEN recognition site: 5′GCTGGCCAACGAAGGGGTTA3′, and a 19 bp (5′CGGTGTCCTGGAGGAGCTG3′) FokI nuclease dimerization and cutting sequence in between. Mouse Nox4 recognizing TALEN plasmids were assembled according to the protocol of Sanjana et al. [29] TALEN mRNA was injected into the pronuclei of one-cell embryos of FVB/NJ mice. Pups were analyzed with Surveyor assay plus sequencing and a founder bearing a 518 bp deletion including the START and 18 bp additional nucleotide was chosen to establish colony.
Nox4 and Duox1 specific Western blot data of knockout animals are shown in Supplementary Fig. 2. Western blots for p22phox and Duox1 were already shown in [16] and (11) respectively.
All animal experiments were approved by the Animal Experimentation Review Board of the Semmelweis University.
2.5. Western blots
Laemmli sample buffer was added to the cell lysate samples and these were run on 8–10% SDS polyacrylamide gels and blotted onto nitrocellulose membranes. Membranes were blocked in phosphate buffered saline containing 0,1% Tween- 20% and 5% dry milk. The first antibodies were diluted in phosphate buffered saline containing 0,1% Tween- 20% and 5% bovine serum albumin and used either for 2 h at room temperature or overnight at 4 °C. After several washing steps in PBS-Tween-20, membranes were incubated with HRP-linked secondary antibodies (Santa Cruz Biotechnology, Inc., USA) diluted in blocking buffer. Antibody binding was detected using enhanced chemiluminescence and Fuji Super RX medical X-ray films.
For the detection of collagen IV NC1 monomers and dimers, cells or small tissue pieces were harvested in hypotonic lysis buffer (10 mM CaCl2, 50 mM Hepes, pH 7.4) completed with 0.1 mM benzamidine hydrochloride, 25 mM 6-aminocaproic acid, 1 mM Phenylmethylsulfonyl fluoride and 0.5 mg/ml type I collagenase at 37 °C for 12–18 h. For the lysis of hypoxia/anoxia treated and control cells, the lysis buffer was supplemented also with 150 µM phloroglucinol to inhibit PXDN activity during the lysis process.
2.6. PXDN antibody
Polyclonal mouse anti-PXDN antibody was raised against the full-length mouse PXDN in Pxdn knockout mice. The recombinant mouse PXDN-V5 antigen was produced in HEK293-FS cells and purified using monoclonal anti-V5 (Clone V5–10) antibody coupled to agarose beads (Sigma). Pxdn knockout animals (see above) were injected with Freund adjuvant containing the purified PXDN-V5 antigen.
2.7. Hypoxic treatments
Cells grown on 6-well plates were kept at 37 °C in a Billups-Rothenberg modular hypoxia chamber (Billups-Rothenberg, Inc., Del Mar CA, USA) which was previously ventilated with the appropriate gas mixture (anoxic mixture: 95% N2, 5% CO2; 1% O2 mixture: 1% O2, 5% CO2, 94% N2; 5% O2 mixture: 5% O2, 5% CO2, 90% N2). After 24 h cells from 1 well were lysed in ice-cold RIPA lysis buffer in the presence of protease inhibitors (for HIF1alpha detection) and content of another well was lysed in hypotonic lysis buffer containing collagenase I (see above) for collagen IV NC1 crosslink analysis.
2.8. Cell viability assays
A two-fold serial dilution of PFHR9 cells was plated on black 96-well plates and grown at normoxic or anoxic conditions. After 24 h cell culture medium was replaced with extracellular “H-medium” (containing 145 mmol/L NaCl, 5 mmol/L KCl, 1 mmol/L MgCl2, 0.8 mmol/L CaCl2, 5 mmol/L glucose, and 10 mmol/L HEPES). Alamar Blue (1:10 of stock solution) and calcein-AM (2 μM) was added to the cells and fluorescent intensities were measured every third minute for 15 min in a POLARstar OPTIMA fluorometer (BMG Labtech) at 37 °C. Excitation/Emission filters were 500/530 and 580/610 for calcein and alamarBlue respectively. The slope of fluorescence increase over time was determined for each cell dilution and displayed as the function of relative cell number.
2.9. Catalase constructs
Catalase sequence was cloned into the pmRFP-N1 vector to express RFP-catalase fusion protein in a cytosolic localization. As control, the F153V mutation was introduced to destroy catalase activity of the protein. For targeting catalase to the mitochondrial matrix, we used duplicate of the N-terminal localization signal of human COX8a [27].
3. Results
3.1. Disruption of the Pxdn gene inhibits collagen IV crosslinking in mice
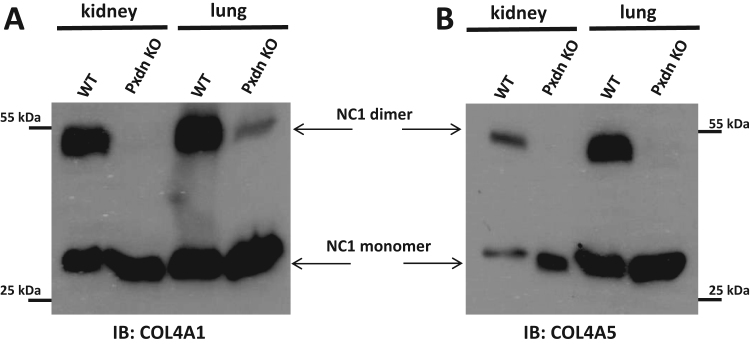
To investigate the enzymes which participate in collagen IV crosslinking in vivo, we first analyzed mice where the Pxdn gene was mutated by zinc finger endonucleases (ZFN) [18]. Pxdn knockout animals showed the ocular developmental defect and white belly spot, which were earlier described by Yan X. et al. in a different PXDN mutant strain [38]. The detailed characterization of the PXDN-deficient animals is subject of a separate study. Previously, we reported deficient collagen crosslinks in cultured embryonic fibroblasts of these ZFN-modified mice [18]. To analyze the effect of PXDN deficiency on in vivo collagen IV coupling, we prepared collagenase digests from different organs, including the kidneys and the lungs. There are six different collagen IV isoforms, designated as alpha1 to alpha6 [14]. In our experiments, we analyzed the crosslink formation between the col4a1 and col4a5 NC1 domains. The isoform-specific monoclonal antibodies consequently detected the ~ 54 kD dimeric and ~ ~28 kD monomeric form of NC1 domains in the collagenase-digested tissue lysates (Fig. 1). Importantly, a dramatic difference was observed between the tissue lysates of wild-type and Pxdn knockout mice. In the PXDN-deficient samples the ratio of NC1 dimers and monomers was substantially reduced (Fig. 1, Supplementary Fig. 2A). These observations suggested that mammalian PXDN has an indispensable, in vivo role in the generation of collagen IV crosslinks.
Fig. 1.
Collagen IV crosslinking in wild type and Pxdn knockout mouse kidney and lung tissues. Small tissue pieces of wild type (WT) and Pxdn knockout (PXDN KO) adult littermate animals were lysed in collagenase I containing hypotonic lysis buffer and digested O/N at 37 °C before processing for collagen IV NC1 western blot. Isoform specific monoclonal col4a1 (A) and col4a5 (B) antibodies were used to detect the crosslinked NC1 domain dimers and non-crosslinked NC1 monomers.
3.2. Assessment of the role of NADPH oxidases in collagen IV crosslinking
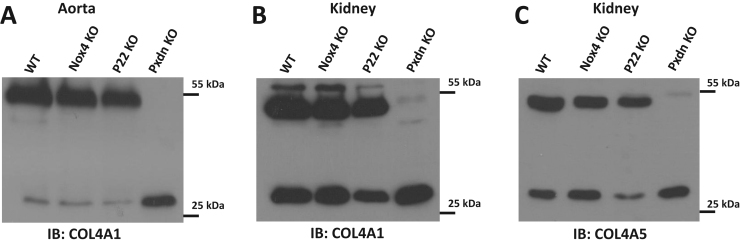
Animal heme peroxidases and members of the NOX/DUOX family of NADPH oxidases often cooperate in reactions where peroxidases use NOX-derived H2O2 to oxidize different substances. Halides and pseudohalides are well-recognized substrates of peroxidases [9], thus we wanted to know whether NOX/DUOX enzymes provide H2O2 for the reaction where bromide is oxidized into hypobromite by PXDN. Knockout models are now available for almost all NOX/DUOX isoforms (except NOX5, which is not present in mice) [31], therefore we decided to analyze the collagen IV crosslinking reaction in different NOX/DUOX knockout models. First, we tested whether NOX4 is involved in collagen IV crosslinking. This particular NOX isoform was especially interesting for us because NOX4 is expressed in cell types (including endothelial, epithelial cells and cardiomyocytes) which are in close contact with underlying or surrounding basement membranes [2], [5]. To investigate the role of NOX4, we created Nox4 knockout animals using TALEN-mediated gene modification. A detailed description of the knockout strategy is described in Materials & Methods. Fig. 2 (A-C, second lanes) show that the western blot picture of Nox4-deficient aorta and kidney samples is indistinguishable from that of wild-type collagenase digests (Fig. 2, Supplementary Fig. 2B). In these and subsequent Western blot experiments, we always included PXDN-deficient samples, where diminished collagen IV crosslinking was detected (Fig. 2A-C, fourth lanes). An important shared feature of NOX1, NOX2, NOX3, and NOX4 is that their activity is dependent on the co-expression of the p22phox protein [1], [21]. The nmf333 mouse strain carries a mutation in the p22phox gene, which leads to the destabilization of the protein with a consequent loss of Nox2 and Nox4 activity [23], [40]. Testing collagen IV crosslinking in this strain offered us the possibility to study the possible involvement of four different NOX isoforms simultaneously, although the uniquely limited expression pattern of NOX3 made unlikely that this particular isoform contributes to the formation of sulfilimine linkages. As shown in Fig. 2 (A-C, third lanes) the crosslinking of collagen IV was perfectly normal when tested in different organs of nmf333 animals. These experiments suggested that a p22phox-dependent NOX isoform is unlikely to provide H2O2 for the PXDN-catalyzed reaction.
Fig. 2.
Collagen IV crosslinking is not altered in vivo in the lack of functional NOX1, NOX2 or NOX4 NADPH oxidase complexes. Small pieces of the thoracic aorta (A) and of the kidney (B,C) from wild type (WT), NOX4-deficient (Nox4 KO), p22phox mutant (P22 KO) and Pxdn knock-out (Pxdn KO) adult animals were lysed in collagenase I containing hypotonic lysis buffer and digested O/N at 37 °C before processing for collagen IV NC1 western blot. Isoform specific monoclonal col4a1 (in A and B) and col4a5 (in C) antibodies were used to detect the crosslinked NC1 domain dimers and non-crosslinked NC1 monomers. For each genotype the tissues of two animals were analyzed with similar results.
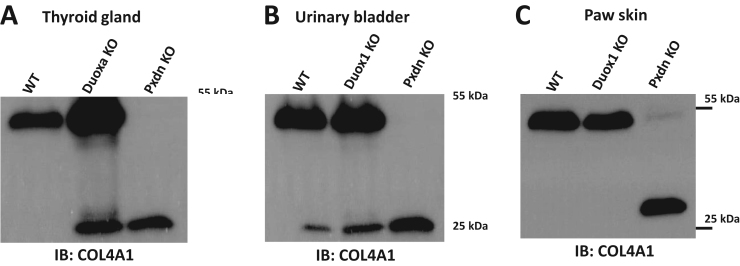
Dual oxidases, DUOX1 and DUOX2 are NADPH oxidase isoforms which operate independently of p22phox and show prominent expression in epithelial cells [10], [20]. We aimed to examine the possible contribution of DUOXes in collagen IV crosslinking. To this end, we analyzed thyroid tissue isolated from mice which are deficient for dual oxidase maturation factor 1 and dual oxidase maturation factor 2 (DUOXA1 and DUOXA2 respectively) [13]. In the absence of DUOX activators, DUOX1 and DUOX2 are not functional and mice without DUOXA proteins show the symptoms of severe hypothyroidism [13]. Collagen IV crosslinking, however, was undisturbed in Duoxa knockouts (Fig. 3A, Supplementary Fig. 2C) suggesting that Duox enzymes do not serve as an H2O2 source in the crosslinking reaction. Interestingly, the amount of NC1 monomers and dimers were consequently higher in the double-knockout thyroid samples, possibly explained by a more active basement membrane formation in the TSH-stimulated thyroid glands of knockout animals (Fig. 3A). Corroborating these results, we found that Duox1 knockout animals [11] also showed normal collagen IV NC1 crosslinks in the skin and urinary bladder samples where DUOX1 shows high expression levels in wild-type animals (Fig. 3 A, B).
Fig. 3.
In the absence of DUOX1 and DUOX2 activity collagen IV crosslinking is not inhibited in vivo. Thyroid gland (A) from Duoxa knockout (Duoxa KO), wild type (WT) and Pxdn knockout (Pxdn KO) mice, paw skin (B), urinary bladder (C) from Duox1 knockout (Duox1 KO), wild type (WT) and Pxdn knockout (Pxdn KO) animals were lysed in collagenase I containing hypotonic lysis buffer and digested O/N at 37 °C. Isoform specific monoclonal col4a1 antibody was used to detect the crosslinked NC1 domain dimers and non-crosslinked NC1 monomers. The thyroid gland of Duoxa knockout animals were significantly enlarged in accordance with the chronic TSH stimulus of the hypothyroid tissue. Note that the proliferative effect of TSH might explain the different Col4a1 picture compared to WT animals. For each genotype the tissues of two animals were analyzed with similar results.
3.3. Determination of the oxygen-dependence of collagen IV crosslinking
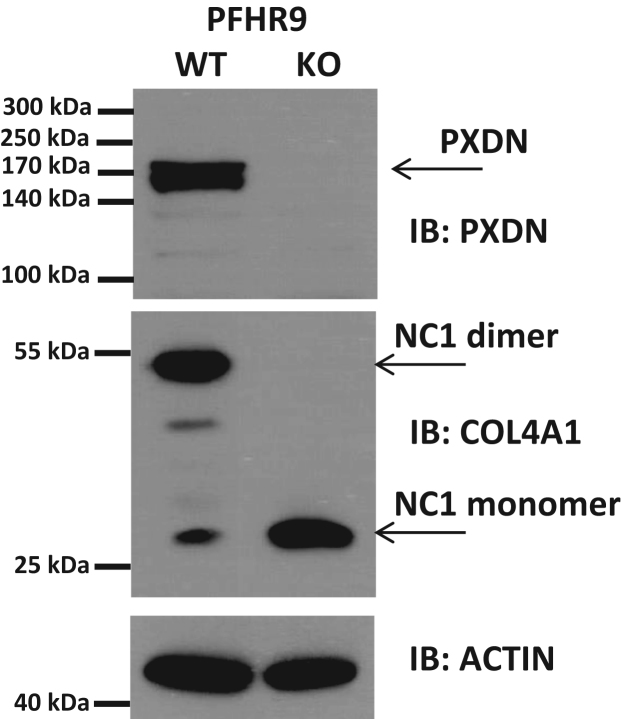
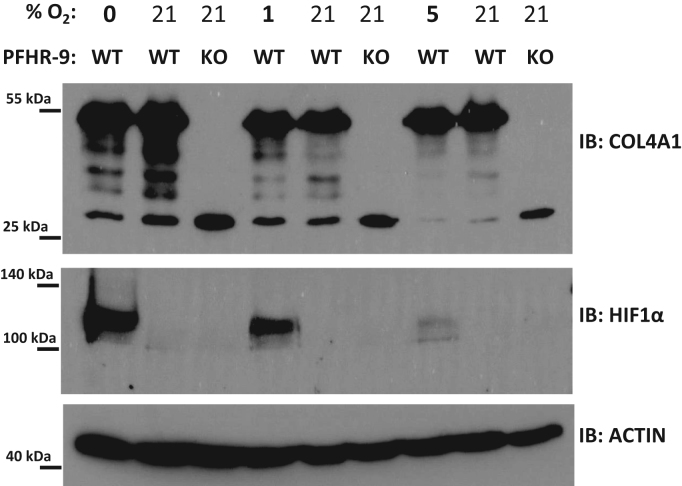
Animal heme peroxidases use H2O2 to oxidize a variety of substrates, which in the case of PXDN appears to be bromide. Although H2O2 can be produced through divergent biochemical pathways, oxygen is required for all of these biosynthetic mechanisms. We, therefore, wanted to determine the dependency of collagen IV crosslinking on oxygen concentration. We chose an embryonic cancer cell line, PFHR-9 cells as a model system in these experiments. Using this particular cell line was justified by previous findings describing intense collagen IV production and crosslinking in PFHR-9 cells [4]. We also established PXDN-deficient PFHR-9 cells making use of the CRISPR gene editing technique. To determine the crosslinking of collagen IV in wild-type and PXDN-deficient PFHR-9 cells we digested the cells with collagenase I and the amount of NC1 dimers and monomers were analyzed by western blot using isoform-specific anti-collagen IV antibodies (Fig. 4). Next, we measured the formation of NC1 dimers at four different oxygen concentrations, anoxia (0%), 1%, 5% and 21%, respectively.
Fig. 4.
Characterization of CRISPR modified PFHR-9 cells. Wild type (WT) or CRISPR modified PXDN knockout (KO) PFHR9 cells were grown on 6-well plates and confluent cultures were harvested in either a RIPA lysis buffer (for PXDN and actin western blots) or in a collagenase I containing hypotonic lysis buffer (for col4a1 NC1 western blot). The two bands around 170 kDa most probably represent the full length and the proprotein convertase- (e.g., furin) cleaved form of PXDN both recognized by our polyclonal anti-PXDN serum.
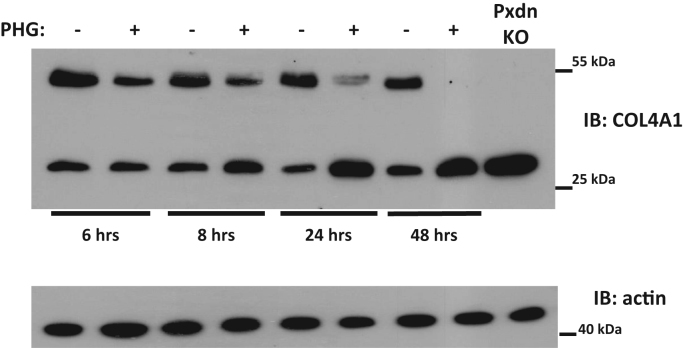
We wanted to reduce the potential toxicity of anoxia, therefore we determined the minimal time which was required to detect the effect of inhibiting the PXDN-catalyzed crosslinking pathway in PFHR-9 cells. To achieve this, we treated the cells with the drug phloroglucinol (PHG) for different times. Phloroglucinol was previously shown to effectively inhibit PXDN activity in PFHR-9 cells [4]. Interestingly, PHG treatment for as little as 6 h was enough to reduce the amount of NC1 dimers and parallelly increase the amount of NC1 monomers, indicating the rapid turnover of collagen IV matrix (Fig. 5). After 24 h of PHG treatment, we observed marked changes in the NC1 dimer/monomer ratio, therefore, we chose 24 h incubation times for the hypoxic treatments. The subatmospheric oxygen levels were applied in a sealed Billups-Rothenberg chamber. As displayed in Fig. 6 at different levels of hypoxia collagen IV crosslinking was essentially undisturbed. To test that oxygen levels were indeed reduced within the cells, we detected the accumulation of HIF-1alpha by western blot (Fig. 6).
Fig. 5.
Analysis of the turnover rate of crosslinked collagen IV in the extracellular matrix of PFHR-9 cells. One day after seeding the cells on culture plates 50 µM phloroglucinol (PHG) was added to the cells for the indicated times. Parallel to each PHG-treated well there was one non-treated control well (indicated by “-”). Cells were lysed in collagenase I containing hypotonic lysis buffer and digested O/N at 37 °C before processing for collagen IV NC1 western blot. This western blot is representative of three experiments with similar results.
Fig. 6.
Various levels of hypoxia do not disturb the collagen IV NC1 crosslinking in cultured PFHR-9 cells. Cells were plated on 6-well culture plates and one day later, nearly confluent cells were placed in a Billups-Rothenberg hypoxia chamber which was ventilated with a gas mixture containing the indicated oxygen levels for 40 min before closing the chamber. A control plate was incubated in a standard cell culture incubator at atmospheric oxygen concentrations. 24 h later cells were lysed either in RIPA buffer (for HIF1-alpha and actin western blot) or lysed in collagenase I containing hypotonic lysis buffer and digested O/N at 37 °C before processing for collagen IV NC1 western blot. The hypotonic lysis buffer was supplemented also with 150 µM phloroglucinol to inhibit PXDN activity during the lysis process. The anoxic treatment (0% O2) was repeated in 4, the 5% and 1% hypoxia in 3 separate experiments with similar results.
To assess the viability of cells after 24 h long anoxic conditions, we applied a fluorescent cell viability assay based on the parallel measurement of calcein-AM cleavage and AlamarBlue reduction. Although we observed reduced viability values with AlamarBlue under anoxic conditions, the result of the calcein-AM assay did not suggest significant toxicity. This indicates that the cells were still biochemically active and survived the 24 h long anoxic treatment (Supplementary Fig. 2).
3.4. Exploring the potential role of mitochondria-derived ROS in collagen IV crosslinking
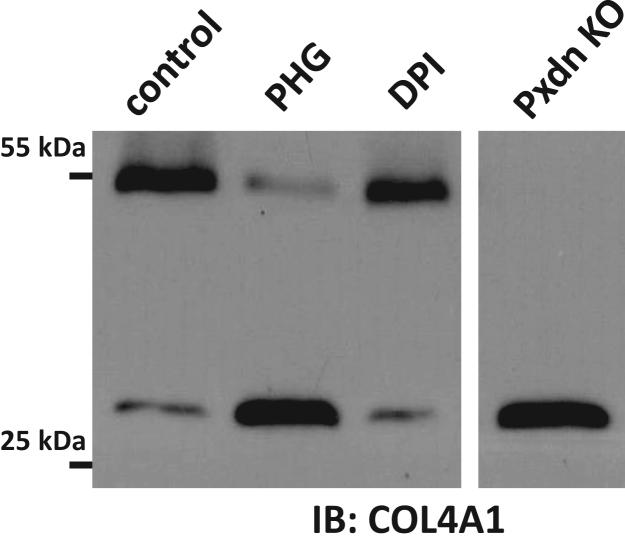
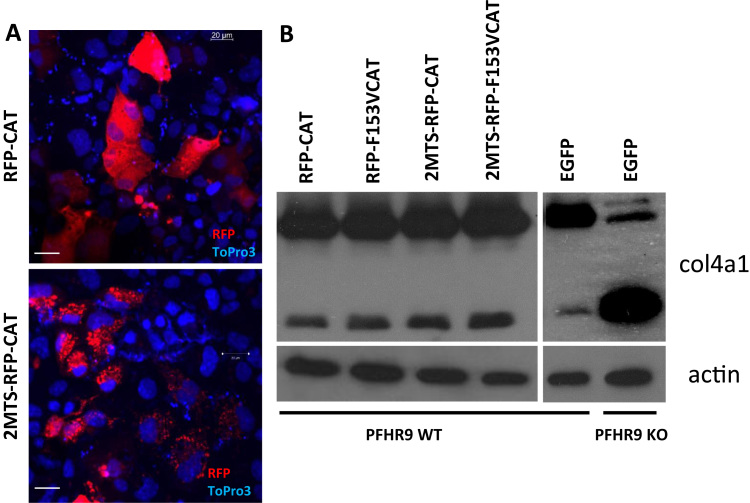
The previous experiments have essentially excluded NOX/DUOX NADPH oxidases as possible sources of H2O2 in collagen IV synthesis. In the following experiments, we tested the potential contribution of other H2O2 sources to NC1 crosslinking. During mitochondrial respiration, ROS are produced as byproducts by complexes I. and III. [17]. Diphenyliodonium (DPI), a potent inhibitor of numerous flavoproteins inhibits the mitochondrial respiratory chain [19]. However, DPI was unable to inhibit the collagen IV crosslinking activity of PXDN (Fig. 7, third lane). Next, we tried an approach where we targeted heterologously expressed catalase to different subcellular locations in PFHR-9 cells (cytosol, and mitochondrial matrix) and checked if this causes any changes in NC1 crosslinking. Fig. 8A shows the intracellular localization of the different catalase constructs. Catalase conjugated to RFP was evenly distributed in the cytosol (upper panel) and RFP-catalase fusion proteins targeted to the mitochondrial matrix (middle panel) showed characteristic mitochondrial localization. In previous experiments, we confirmed that fusing catalase to fluorescent proteins and different targeting sequences did not affect the catalytic activity of the enzyme (data not shown). When we analyzed collagen IV crosslinking in G418 selected, stably transfected cells we also included controls, where catalytically-inactive catalase proteins were introduced into the cells. As shown in Fig. 8B, the tested catalase constructs did not affect the amount of NC1 dimers synthesized by PFHR-9 cells.
Fig. 7.
Analysis of the role of mitochondria as potential ROS sources for PXDN-mediated collagen IV crosslinking. PFHR-9 cells were plated on 6-well plates and one day later 50 µM phloroglucinol (phg) or 10 µM DPI was added to the cells for 24 h. Cells were lysed in collagenase I containing hypotonic lysis buffer and digested O/N at 37 °C before processing for collagen IV NC1 western blot. This western blot is representative of 5 similar experiments.
Fig. 8.
Analysis of the effect of recombinant catalase-mediated decomposition of intracellular hydrogen peroxide on collagen IV crosslinking. Catalase-RFP fusion proteins were targeted to the cytosol (RFP-CAT) or to the mitochondrial matrix (2MTS-RFP-CAT). Confocal microscopic images of transiently transfected PFHR9 cell. Topro3 positive nuclei are displayed in blue (A). Transfected PFHR9 cells were selected with 250 μg/ml G418 for 7 days. During this time all non-transfected cells died and G418 cells reached confluency. Wild type and Pxdn KO cells were also transfected with an EGFP expressing plasmid and went through the same 7 day long G418 selection. Cells were lysed in collagenase I containing hypotonic lysis buffer and digested O/N at 37 °C before processing for collagen IV NC1 western blot. Before adding collagenase I to the lysis buffer a small fraction of the lysates were processed for actin western blot.
4. Discussion
A common feature of reactions catalyzed by animal heme peroxidases is that halides or pseudohalides are oxidized by the enzymes. A recent addition to this list is the reaction catalyzed by PXDN, which involves the oxidation of bromide and the subsequent hypobromite-mediated crosslinking of collagen IV NC1 domains via methionine and hydroxylysine residues [4], [22], [28]. The presence of bromide and peroxidasin is indispensable for the collagen IV NC1 crosslink formation [4], [22], however, the source of H2O2 in the PXDN-catalyzed reaction has not yet been identified. It is also an important question and subject of current research whether HOBr attacks other matrix components as well or it creates only the specific halosulfonium intermediate on Met93 of collagen IV NC1.
PXDN is a highly conserved protein that appears early in evolution and it can be found across the animal kingdom [32]. Based on the examples where NOX/DUOX enzymes act as a source of H2O2 in the peroxidase-catalyzed reactions we hypothesized that the activity of PXDN might be also dependent on NOX/DUOX isoforms. We tested this idea by assessing collagen IV crosslinking in mice which are deficient for either p22phox or for certain NOX/DUOX isoforms. We found collagen IV crosslinking to be normal in all tested Nox/Duox knockout models, indicating that PXDN uses H2O2 produced in a NOX-independent manner. By measuring collagen IV crosslinking in multiple organs of PXDN-deficient animals we demonstrated the essential role of PXDN in this biochemical pathway. These results extend earlier observations, where deficient collagen IV crosslinking was described in PXDN knockout embryonic fibroblasts and kidney [3], [18]. Although PXDN belongs to the family of heme peroxidases, several features make this protein unique. Most importantly, besides its peroxidase part, PXDN also contains domains, which are characteristic of protein components of the extracellular matrix [24]. This raises the possibility that extracellular enzymes might act as an H2O2 source for PXDN. However, in previous studies, lysyl oxidases, which are ROS-producing and extracellular collagen- modifying enzymes, were also excluded as a potential H2O2 source for NC1 crosslinking [4].
Another distinct feature of PXDN is that its expression is more widespread than that of other members of the animal heme peroxidase family since PXDN is expressed in virtually all tissues, where basement membranes are formed [7], [25]. The expression pattern of PXDN is actually consistent with the idea that H2O2 is supplied for the enzyme in a NOX/DUOX independent manner because no single NOX/DUOX enzyme shows similarly widespread expression. It is also important to note that currently no data obtained from in vivo experiments prove the essential role of H2O2 in the crosslinking reaction, thus the contribution of alternative oxidants cannot be excluded. For example, protein peroxides, which are formed in a wide variety of reactions between proteins and free radicals, may act as oxidants during extracellular matrix formation [8].
Since we know so little about the source of ROS in collagen IV crosslinking, we were interested in characterizing the oxygen dependence of the process. Interestingly, we found that the coupling of collagen IV is undisturbed even at very low oxygen levels, indicating that a ROS-producing pathway with low Kd for oxygen participates in the process. Another intriguing implication of the above observations might be that even relatively long hypoxia, per se, will not disturb the integrity of collagen IV crosslinks in the basal membrane of vessel walls in hypoxic tissues.
In polarized epithelial cells, mitochondria are often located and enriched at the basolateral side of the cells, in relative proximity to the underlying basement membrane. Moreover, the mitochondrial respiratory chain has a very high affinity for oxygen. It is not easy to assess the possible role of mitochondrial ROS in the crosslinking reaction. The flavoprotein inhibitor, DPI, a non-specific inhibitor of mitochondrial respiration and NADPH oxidases, did not affect NC1 coupling. However, it is possible, that inhibition of the respiratory chain by DPI or other inhibitors may actually increase the production of superoxide by the organelles [33].
To explore the role of mitochondrial ROS in a more specific way, we expressed catalase constructs in PFHR9 cells and checked NC1 dimer formation. However, targeting catalase to the cytosol or to the mitochondrial matrix did not cause any decrease in collagen IV crosslinking. Schriner et al. created a transgenic mouse model which overexpressed mitochondrially targeted catalase [30]. These mice showed delayed aging, had a longer lifespan but did not display any gross developmental abnormalities. This is in contrast with the eye development and skin pigmentation phenotype of peroxidasin mutant mice. As the affinity of catalase towards H2O2 can be significantly lower than that of animal heme peroxidases we have to admit that our approach to testing the involvement of mitochondria can be considered only preliminary and further experiments are needed to explore the potential contribution of mitochondria-produced H2O2 to the process.
In summary, our observations suggest that in collagen IV synthesis PXDN cooperates with a NOX/DUOX-independent H2O2 source that is functional also at low oxygen levels.
Acknowledgments
We are grateful to Beáta Molnár and Barbara Bodor-Kis for technical assistance. This work was supported by grants from the National Research, Development and Innovation Office (K106138, K119955, NVKP_16-1-2016-0039) and by “Lendület” grant from the Hungarian Academy of Sciences (LP2011-001/2011).
Acknowledgments
Author disclosure statements
The authors declare no competing financial interests.
Footnotes
Supplementary data associated with this article can be found in the online version at doi:10.1016/j.redox.2018.03.009.
Appendix A. Supplementary material
Supplementary material. Supplementary Figure 1. Nox4 and Duox1 specific western blots on tissue samples of knockout animals A) isolated kidney was mechanically dissected in ice cold RIPA lysis buffer using a Turrax disperser. Insoluble fraction was centrifuged and the soluble fraction was processed for Nox4 western blot on 10% SDS polyacrylamide gel. B) paw skin was isolated and processed in RIPA lysis buffer as above and processed for Duox1 western blot on 8% SDS polyacrylamide gel. Supplementary Figure 2. Statistical analysis is shown in A,B and C related to western blot data presented in Figs. 1, 2, and 3 respectively. (A) ImageJ analysis of collagen IV-a1 and −5 NC1 dimer / (dimer+monomer) ratios in WT and Pxdn KO kidney and lung tissues are displayed on a box-and-whisker plot. (B) ImageJ analysis of collagen IV-a1 and −5 NC1 dimer / (dimer+monomer) ratios in WT, Nox4 KO, p22phox mutant and Pxdn KO aorta and kidney tissues are displayed on a box-and-whisker plot. (C) ImageJ analysis of collagen IV-a1 NC1 dimer / (dimer+monomer) ratios in WT, Duox1 KO, Duoxa KO and PXDN KO thyroid, urinary bladder and paw skin tissues are displayed on a box-and-whisker plot. In the box area “x” indicates the mean value, the horizontal line indicates the median value. For each genotype the tissues of 2 animals were analyzed. Supplementary Figure 3. Cell viability measurements following 24 h of anoxic/normoxic conditions. Calcein and alamarBlue fluorescence of cells plated on 96 well plates in a two-fold serial dilution of 5 different cell densities (16x-8x-4x-2x-x). For each cell density the slope of fluorescence increase over a time period of 15 min was determined and displayed as the function of the relative cell number. Data points show the average of 7 parallel samples +/- SD values.
References
- 1.Ambasta R.K., Kumar P., Griendling K.K., Schmidt H.H., Busse R., Brandes R.P. Direct interaction of the novel Nox proteins with p22phox is required for the formation of a functionally active NADPH oxidase. J. Biol. Chem. 2004;279:45935–45941. doi: 10.1074/jbc.M406486200. [DOI] [PubMed] [Google Scholar]
- 2.Bedard K., Krause K.H. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol. Rev. 2007;87:245–313. doi: 10.1152/physrev.00044.2005. [DOI] [PubMed] [Google Scholar]
- 3.Bhave G., Colon S., Ferrell N. The sulfilimine cross-link of collagen IV contributes to kidney tubular basement membrane stiffness. Am. J. Physiol. Ren. Physiol. 2017;313:596–602. doi: 10.1152/ajprenal.00096.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bhave G., Cummings C.F., Vanacore R.M., Kumagai-Cresse C., Ero-Tolliver I.A., Rafi M., Kang J.S., Pedchenko V., Fessler L.I., Fessler J.H., Hudson B.G. Peroxidasin forms sulfilimine chemical bonds using hypohalous acids in tissue genesis. Nat. Chem. Biol. 2012;8:784–790. doi: 10.1038/nchembio.1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brandes R.P., Schroder K. Composition and functions of vascular nicotinamide adenine dinucleotide phosphate oxidases. Trends Cardiovasc. Med. 2008;18:15–19. doi: 10.1016/j.tcm.2007.11.001. [DOI] [PubMed] [Google Scholar]
- 6.Brandes R.P., Weissmann N., Schroder K. Nox family NADPH oxidases: molecular mechanisms of activation. Free Radic. Biol. Med. 2014;76C:208–226. doi: 10.1016/j.freeradbiomed.2014.07.046. [DOI] [PubMed] [Google Scholar]
- 7.Cheng G., Salerno J.C., Cao Z., Pagano P.J., Lambeth J.D. Identification and characterization of VPO1, a new animal heme-containing peroxidase. Free Radic. Biol. Med. 2008;45:1682–1694. doi: 10.1016/j.freeradbiomed.2008.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Davies M.J. Protein oxidation and peroxidation. Biochem. J. 2016;473:805–825. doi: 10.1042/BJ20151227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Davies M.J., Hawkins C.L., Pattison D.I., Rees M.D. Mammalian heme peroxidases: from molecular mechanisms to health implications. Antioxid. Redox Signal. 2008;10:1199–1234. doi: 10.1089/ars.2007.1927. [DOI] [PubMed] [Google Scholar]
- 10.Donko A., Peterfi Z., Sum A., Leto T., Geiszt M. Dual oxidases. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2005;360:2301–2308. doi: 10.1098/rstb.2005.1767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Donko A., Ruisanchez E., Orient A., Enyedi B., Kapui R., Peterfi Z., De D., X., Benyo Z., Geiszt M. Urothelial cells produce hydrogen peroxide through the activation of Duox1. Free Radic. Biol. Med. 2010;49:2040–2048. doi: 10.1016/j.freeradbiomed.2010.09.027. [DOI] [PubMed] [Google Scholar]
- 12.Geiszt M., Witta J., Baffi J., Lekstrom K., Leto T.L. Dual oxidases represent novel hydrogen peroxide sources supporting mucosal surface host defense. FASEB J. 2003;17:1502–1504. doi: 10.1096/fj.02-1104fje. [DOI] [PubMed] [Google Scholar]
- 13.Grasberger H., De D., X., Mayo O.B., Raad H., Weiss M., Liao X.H., Refetoff S. Mice deficient in dual oxidase maturation factors are severely hypothyroid. Mol. Endocrinol. 2012;26:481–492. doi: 10.1210/me.2011-1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Khoshnoodi J., Pedchenko V., Hudson B.G. Mammalian collagen IV. Microsc. Res. Technol. 2008;71:357–370. doi: 10.1002/jemt.20564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Klebanoff S.J., Kettle A.J., Rosen H., Winterbourn C.C., Nauseef W.M. Myeloperoxidase: a front-line defender against phagocytosed microorganisms. J. Leukoc. Biol. 2013;93:185–198. doi: 10.1189/jlb.0712349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kovacs I., Horvath M., Lanyi A., Petheo G.L., Geiszt M. Reactive oxygen species-mediated bacterial killing by B lymphocytes. J. Leukoc. Biol. 2015;97:1133–1137. doi: 10.1189/jlb.4AB1113-607RR. [DOI] [PubMed] [Google Scholar]
- 17.Kowaltowski A.J., Souza-Pinto N.C., Castilho R.F., Vercesi A.E. Mitochondria and reactive oxygen species. Free Radic. Biol. Med. 2009;47:333–343. doi: 10.1016/j.freeradbiomed.2009.05.004. [DOI] [PubMed] [Google Scholar]
- 18.Lazar E., Peterfi Z., Sirokmany G., Kovacs H.A., Klement E., Medzihradszky K.F., Geiszt M. Structure-function analysis of peroxidasin provides insight into the mechanism of collagen IV crosslinking. Free Radic. Biol. Med. 2015;83:273–282. doi: 10.1016/j.freeradbiomed.2015.02.015. [DOI] [PubMed] [Google Scholar]
- 19.Li Y., Trush M.A. Diphenyleneiodonium, an NAD(P)H oxidase inhibitor, also potently inhibits mitochondrial reactive oxygen species production. Biochem. Biophys. Res. Commun. 1998;253:295–299. doi: 10.1006/bbrc.1998.9729. [DOI] [PubMed] [Google Scholar]
- 20.Little A.C., Sulovari A., Danyal K., Heppner D.E., Seward D.J., Van D. V Paradoxical roles of dual oxidases in cancer biology. Free Radic. Biol. Med. 2017;110:117–132. doi: 10.1016/j.freeradbiomed.2017.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Martyn K.D., Frederick L.M., von Loehneysen K., Dinauer M.C., Knaus U.G. Functional analysis of Nox4 reveals unique characteristics compared to other NADPH oxidases. Cell Signal. 2006;18:69–82. doi: 10.1016/j.cellsig.2005.03.023. [DOI] [PubMed] [Google Scholar]
- 22.McCall A.S., Cummings C.F., Bhave G., Vanacore R., Page-McCaw A., Hudson B.G. Bromine is an essential trace element for assembly of collagen IV scaffolds in tissue development and architecture. Cell. 2014;157:1380–1392. doi: 10.1016/j.cell.2014.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nakano Y., Longo-Guess C.M., Bergstrom D.E., Nauseef W.M., Jones S.M., Banfi B. Mutation of the Cyba gene encoding p22phox causes vestibular and immune defects in mice. J. Clin. Invest. 2008;118:1176–1185. doi: 10.1172/JCI33835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nelson R.E., Fessler L.I., Takagi Y., Blumberg B., Keene D.R., Olson P.F., Parker C.G., Fessler J.H. Peroxidasin: a novel enzyme-matrix protein of Drosophila development. EMBO J. 1994;13:3438–3447. doi: 10.1002/j.1460-2075.1994.tb06649.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Peterfi Z., Donko A., Orient A., Sum A., Prokai A., Molnar B., Vereb Z., Rajnavolgyi E., Kovacs K.J., Muller V., Szabo A.J., Geiszt M. Peroxidasin is secreted and incorporated into the extracellular matrix of myofibroblasts and fibrotic kidney. Am. J. Pathol. 2009;175:725–735. doi: 10.2353/ajpath.2009.080693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Peterfi Z., Geiszt M. Peroxidasins: novel players in tissue genesis. Trends Biochem. Sci. 2014;39:305–307. doi: 10.1016/j.tibs.2014.05.005. [DOI] [PubMed] [Google Scholar]
- 27.Rizzuto R., Brini M., Pizzo P., Murgia M., Pozzan T. Chimeric green fluorescent protein as a tool for visualizing subcellular organelles in living cells. Curr. Biol. 1995;5:635–642. doi: 10.1016/s0960-9822(95)00128-x. [DOI] [PubMed] [Google Scholar]
- 28.Ronsein G.E., Winterbourn C.C., Di Mascio P., Kettle A.J. Cross-linking methionine and amine residues with reactive halogen species. Free Radic. Biol. Med. 2014;70:278–287. doi: 10.1016/j.freeradbiomed.2014.01.023. [DOI] [PubMed] [Google Scholar]
- 29.Sanjana N.E., Cong L., Zhou Y., Cunniff M.M., Feng G., Zhang F. A transcription activator-like effector toolbox for genome engineering. Nat. Protoc. 2012;7:171–192. doi: 10.1038/nprot.2011.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schriner S.E., Linford N.J., Martin G.M., Treuting P., Ogburn C.E., Emond M., Coskun P.E., Ladiges W., Wolf N., Van Remmen H., Wallace D.C., Rabinovitch P.S. Extension of murine life span by overexpression of catalase targeted to mitochondria. Science. 2005;308:1909–1911. doi: 10.1126/science.1106653. [DOI] [PubMed] [Google Scholar]
- 31.Sirokmany G., Donko A., Geiszt M. Nox/duox family of NADPH oxidases: lessons from knockout mouse models. Trends Pharmacol. Sci. 2016 doi: 10.1016/j.tips.2016.01.006. [DOI] [PubMed] [Google Scholar]
- 32.Soudi M., Zamocky M., Jakopitsch C., Furtmuller P.G., Obinger C. Molecular evolution, structure, and function of peroxidasins. Chem. Biodivers. 2012;9:1776–1793. doi: 10.1002/cbdv.201100438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Turrens J.F. Superoxide production by the mitochondrial respiratory chain. Biosci. Rep. 1997;17:3–8. doi: 10.1023/a:1027374931887. [DOI] [PubMed] [Google Scholar]
- 34.Vanacore R., Ham A.J., Voehler M., Sanders C.R., Conrads T.P., Veenstra T.D., Sharpless K.B., Dawson P.E., Hudson B.G. A sulfilimine bond identified in collagen IV. Science. 2009;325:1230–1234. doi: 10.1126/science.1176811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Veal E., Day A. Hydrogen peroxide as a signaling molecule. Antioxid. Redox Signal. 2011;15:147–151. doi: 10.1089/ars.2011.3968. [DOI] [PubMed] [Google Scholar]
- 36.Wang J., Slungaard A. Role of eosinophil peroxidase in host defense and disease pathology. Arch. Biochem. Biophys. 2006;445:256–260. doi: 10.1016/j.abb.2005.10.008. [DOI] [PubMed] [Google Scholar]
- 37.Winterbourn C.C. The biological chemistry of hydrogen peroxide. Methods Enzymol. 2013;528:3–25. doi: 10.1016/B978-0-12-405881-1.00001-X. [DOI] [PubMed] [Google Scholar]
- 38.Yan X., Sabrautzki S., Horsch M., Fuchs H., Gailus-Durner V., Beckers J., Hrabe d. A., Graw J. Peroxidasin is essential for eye development in the mouse. Hum. Mol. Genet. 2014;23:5597–5614. doi: 10.1093/hmg/ddu274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yurchenco P.D. Basement membranes: cell scaffoldings and signaling platforms. Cold Spring Harb. Perspect. Biol. 2011;3:a004911. doi: 10.1101/cshperspect.a004911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zana M., Peterfi Z., Kovacs H.A., Toth Z.E., Enyedi B., Morel F., Paclet M.H., Donko A., Morand S., Leto T.L., Geiszt M. Interaction betweenp22(phox) and Nox4 in the endoplasmic reticulum suggests a unique mechanism of NADPH oxidase complex formation. Free Radic. Biol. Med. 2018;116:41–49. doi: 10.1016/j.freeradbiomed.2017.12.031. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material. Supplementary Figure 1. Nox4 and Duox1 specific western blots on tissue samples of knockout animals A) isolated kidney was mechanically dissected in ice cold RIPA lysis buffer using a Turrax disperser. Insoluble fraction was centrifuged and the soluble fraction was processed for Nox4 western blot on 10% SDS polyacrylamide gel. B) paw skin was isolated and processed in RIPA lysis buffer as above and processed for Duox1 western blot on 8% SDS polyacrylamide gel. Supplementary Figure 2. Statistical analysis is shown in A,B and C related to western blot data presented in Figs. 1, 2, and 3 respectively. (A) ImageJ analysis of collagen IV-a1 and −5 NC1 dimer / (dimer+monomer) ratios in WT and Pxdn KO kidney and lung tissues are displayed on a box-and-whisker plot. (B) ImageJ analysis of collagen IV-a1 and −5 NC1 dimer / (dimer+monomer) ratios in WT, Nox4 KO, p22phox mutant and Pxdn KO aorta and kidney tissues are displayed on a box-and-whisker plot. (C) ImageJ analysis of collagen IV-a1 NC1 dimer / (dimer+monomer) ratios in WT, Duox1 KO, Duoxa KO and PXDN KO thyroid, urinary bladder and paw skin tissues are displayed on a box-and-whisker plot. In the box area “x” indicates the mean value, the horizontal line indicates the median value. For each genotype the tissues of 2 animals were analyzed. Supplementary Figure 3. Cell viability measurements following 24 h of anoxic/normoxic conditions. Calcein and alamarBlue fluorescence of cells plated on 96 well plates in a two-fold serial dilution of 5 different cell densities (16x-8x-4x-2x-x). For each cell density the slope of fluorescence increase over a time period of 15 min was determined and displayed as the function of the relative cell number. Data points show the average of 7 parallel samples +/- SD values.