Abstract
The TGR5 receptor (or GP-BAR1, or M-BAR) was characterized ten years ago as the first identified G-coupled protein receptor specific for bile acids. TGR5 gene expression is widely distributed, including endocrine glands, adipocytes, muscles, immune organs, spinal cord, and the enteric nervous system. The effect of TGR5 activation depends on the tissue where it is expressed and the signalling cascade that it induces. Animal studies suggest that TGR5 activation influences energy production and thereby may be involved in obesity and diabetes. TGR5 activation also influences intestinal motility. This review provides an overview of TGR5-bile acid interactions in health as well as the possible involvement of TGR5 in human disease.
Keywords: Bile acids, Diabetes, Inflammation, Motility, TGR5 receptor
1. Introduction
The TGR5 receptor is the first known G-protein coupled receptor specific for bile acids. Since its identification in 2002, TGR5 has been found to be ubiquitously expressed in humans and animals, and to activate various intracellular signalling pathways upon interaction with bile acids. In animals, initial experimental studies revealed that TRG5 activation plays a role in the regulation of basal metabolism and energy expenditure; TGR5 is now recognized as a potential target for the treatment of metabolic disorders, such as type 2 diabetes. In addition, the ability of TGR5 to lower the levels of proinflammatory cytokines in monocytes has opened new insights into the modulatory role of bile acids in pathology where inflammatory processes play a central role, including colitis and atheroma development. TGR5 activation has also been shown to influence experimental cholesterol gallstone formation in mice and found to stimulate bicarbonate secretion by cholangiocytes. Several recent reviews have addressed conditions in which TGR5 has emerged as a new target for pharmaceutical agents ([1.:p. 5,2–4]). The intent of the present review is to provide a summary of the currently available knowledge on TGR5 biology and to relate this information to the possible involvement of TGR5 in human diseases.
2. TGR5 discovery
In 1999, the field of bile acids experienced a change in thinking when bile acid function moved beyond that of digestive surfactants. Bile acids were found to be signalling molecules, acting as hormones and regulatory ligands. This paradigm shift was spurred by the discovery of the dominant nuclear receptor for bile acids, the farnesoid X receptor (FXR) [5]. Since then, convergent studies have established that FXR is involved in the regulation of genes acting in multiple metabolic pathways [6,7]. Three years after the discovery of FXR, Maruyama et al. described a cell membrane G-protein coupled receptor (GPCR) that was activated by bile acids. This receptor is generally named the membrane-bile acid receptor (M-BAR) [8], or TGR5 [9], rather than the Gprotein bile acid-activated receptor (GP-BAR1) [10,11]. It is important to note that GP-BAR1 remains the official designation for Homo sapiens in the NIH gene database and in the recommendations of the HUGO Gene Nomenclature Committee [12].
The existence of a cell surface receptor activated by bile acids was previously suspected, given the differential immunosuppressive effects of bile acids in immune cells assessed in vitro. Chenodeoxycholic and deoxycholic acids were found to decrease the production of interleukin 6 (IL-6) and tumour necrosis factor alpha (TNF-α) from macrophages, whereas ursodeoxycholic acid, a more hydrophilic bile acid, had a much smaller effect [13,14]. Based on the sequence of a probable GPCR identified in the Human Genome database (GenBank), Maruyama et al. [8] cloned the TGR5 receptor in human embryonic kidney HEK 293 cells, and characterized the variations in binding affinity of individual bile acids to TGR5. These findings were confirmed by studies in Chinese Hamster Ovarian cells (CHO) co-transfected with a reporter gene (cyclic Adenosine Monophospate (cAMP) responsive element fused to the luciferase gene), and with an expression vector of the human hypothetical GPCR sequence [9]. Among thousands of compounds screened, bile acids were the only ones that triggered a specific increase in luciferase activity due to cAMP production. Similarly, a co-transfection assay of CHO cells with the rat TGR5 cDNA sequence demonstrated that rat TGR5 emerged as the specific bile acid receptor [9]. Additional experiments using a fusion protein of TGR5 and green fluorescent protein (GFP) identified the localization of TGR5 on the plasma membrane and its internalization upon treatment with taurolithocholic acid. However, this internalization process remains controversial because, to date, it has not been reproduced by other teams, and subsequent data have shown that TGR5 can be repeatedly activated.
3. TGR5 gene and structure
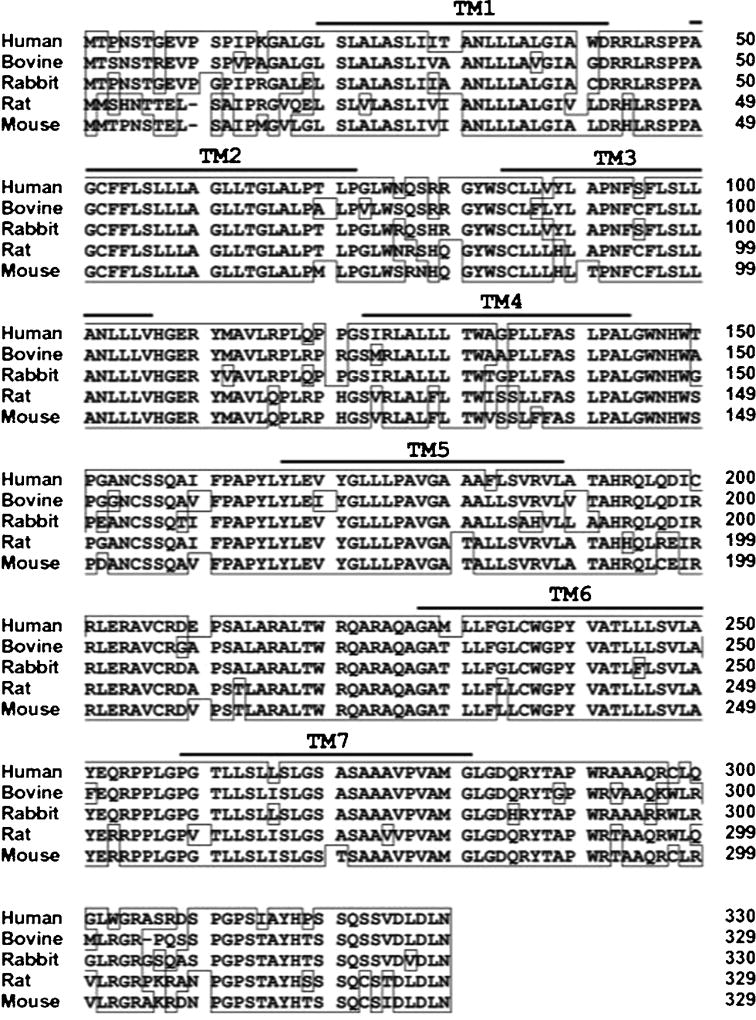
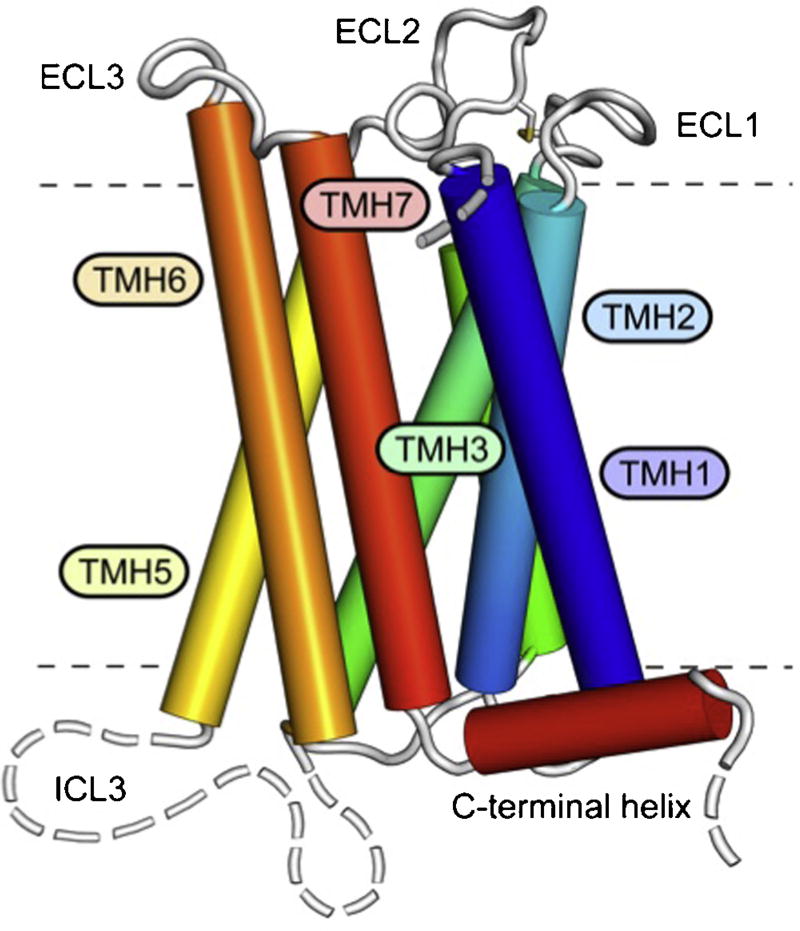
Maruyama et al. as well as Kawamata et al. identified the putative coding sequence using the GenBank DNA database approach to screen amino acid sequences of known GPCRs [8,9]. The coding sequence of the TGR5 gene contains 993 base pairs, encoding a protein of 330 amino acids with the predicted seven transmembrane domains, a feature that is characteristic of GPCRs. In humans, the TGR5 gene is located on chromosome 2q35, and the cDNA sequence shared almost complete homology (>80%) with that of bovid, rabbit, and rodent species, showing a high structural conservation among mammals (Fig. 1) [9]. The crystal structure of TGR5 is not yet available to allow modelling of the binding domain. However, a homology model of the 3D structure of the binding domain has been recently proposed using the TGR5 amino acid sequence (Fig. 2) [15].
Fig. 1. Comparison of amino acid sequences of human, bovine, rabbit, rat, and mouse TGR5.
Reproduce with permission: Copyright: The American Society for Biochemistry and Molecular Biology, Journal of Biological Chemistry, Kawamata et al., J Biol Chem. 2003 Mar 14;278(11):9435-40 [9].
Fig. 2.
3D structure of TGR5. The receptor comprises seven transmembrane helices (TMH1-7), three extracellular loops (ECL1-3), contributing to ligand binding, and three intracellular loops (ICL1-3) involved in mediating the signal to downstream signalling molecules. ICL3 and the N- and C-terminal segments are structurally flexible and disordered (broken lines).
From PLOS one, Hov et al., Mutational characterization of the bile acid receptor TGR5 in primary sclerosing cholangitis. PLoS ONE. 2010;5(8):e12403 [15], with permission.
4. Distribution and regulation of TGR5 gene expression
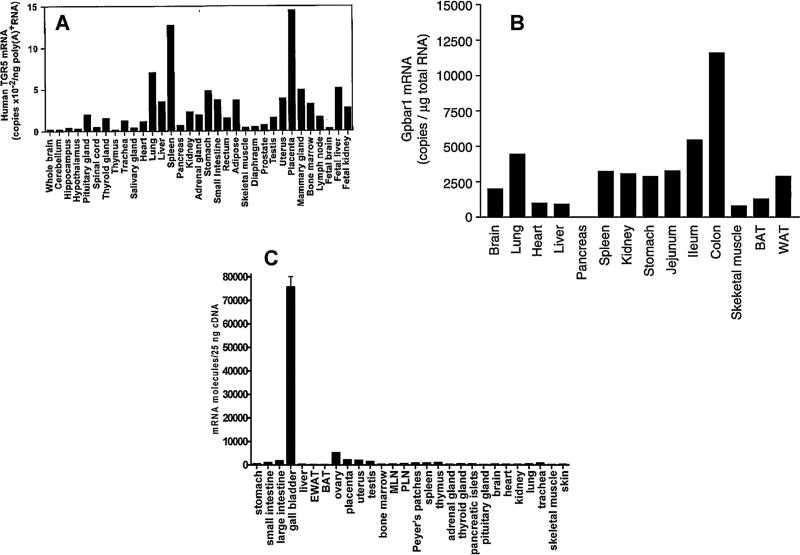
Mapping studies indicate that TGR5 gene expression is ubiquitous, both in animals and humans. The initial expression profile indicated a wide tissue distribution of TGR5 gene expression, including endocrine glands, adipocytes, muscles, as well as immune organs like spleen and lymph nodes (Fig. 3 A) [9]. In C57/BL6 mice, TGR5 mRNA is detected in visceral organs (Fig. 3B) including the gut and the gallbladder (Fig. 3C) [10,16]. TGR5 gene expression is also found in the brain and spinal cord in rodents and humans [10,16–18] and is densely expressed in the enteric nervous system of mice [19].
Fig. 3. (A–C) Comparison of TGR5 mRNA expression in human (A) and C57/BL6 mice (B and C) tissues.
From Copyright: The American Society for Biochemistry and Molecular Biology, Journal of Biological Chemistry, Kawamata et al., J Biol Chem. 2003 Mar 14;278(11):9435-40 [9] (A), “Reproduced with permission”: Maruyama T et al. Targeted disruption of G protein-coupled bile acid receptor 1 (Gpbar1/M-Bar) in mice. J Endocrinol. 2006 Oct;191(1):197-205. Copyright BioScientifica Ltd. [16] (B), Reproduced with permission, from Vassileva G, Golovko A, Markowitz L, Abbondanzo S.J, Zeng M, Yang S, Hoos L, Tetzloff G, Levitan D, Murgolo N.J, Keane K, Davis Jr H.R, Hedrick J and Gustafson E.L (2006) Targeted deletion of Gpbar1 protects mice from cholesterol gallstone formation. Biochemical Journal, 398 423–430 © the Biochemical Society [10] (C), with permission.
At the protein level, an immunohistochemical study in mice showed that TGR5 immunoreactivity is widely distributed throughout the gastrointestinal tract, with a prominent expression in the enteric nervous system [19]. In particular, the submucosal nerve plexus of the small and large intestine display TGR5 immunoreactivity in more than 80% of neurons. In the myenteric plexus, TGR5 immunoreactivity is mainly located in the inhibitory neurons and descending neurons. The enteric localization of TGR5 provides a neuroanatomical support to earlier recognition of the secretory and motor effect of bile acids as a component of the intramural nervous reflex [20,21]. In the rat liver, the TGR5 receptor has been identified on the plasma membrane of liver sinusoidal endothelial cells [22], as well as on the apical membrane and primary cilium of cholangiocytes [23], as well as on biliary epithelial cells [24] and Kupffer cells [25]. In mice spinal cord, the distribution in sacral and lumbar segments has received particular attention, and TGR5 immunoreactivity was found to be located in neuronal cell bodies of the laminae I, II, and V of the dorsal horn, and lamina X [18].
At present, little is known about the regulation of TGR5 expression in the whole organism in response to physiological or pathological conditions, as regulation of expression has been assessed mainly in cultured cells. Ammonium ions down-regulate mRNA expression in cultured astrocytes in a dose and time-dependent manner after 48 h [17]. The steroid 5-beta-pregnan-3-alpha-ol-20-one, which is also a potent TGR5 ligand, can decrease TGR5 gene expression after incubation for 72 h [17].
5. TGR5 agonists
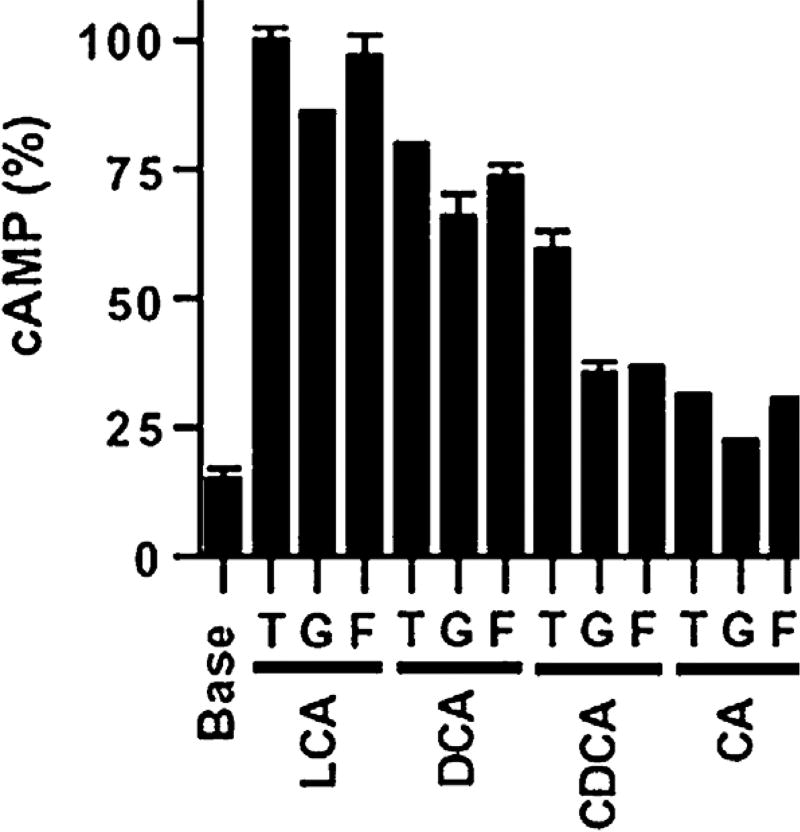
Among the family of steroids, the most powerful endogenous agonists identified for the TGR5 receptor are taurine and glycine-conjugated and unconjugated bile acids. Activation of TGR5 by bile acids was characterized by measuring cAMP production in CHO [9] and HEK293 [4] cell lines transfected with human TGR5 cDNA in an expression vector. TGR5 showed a dose-dependent activation by bile acids, with the following rank order of potency: lithocholic acid ≥ deoxycholic acid > chenodeoxycholic acid > cholic acid. Of note, deoxycholic acid is a secondary bile acid formed by bacterial dehydroxylation of cholic acid; lithocholic acid is formed by the analogous dehydroxylation of chenodeoxycholic acid; cholic and chenodeoxycholic acids are the dominant primary bile acids in humans. Activation was greater for taurine-conjugated than unconjugated bile acids, and these in turn were more potent than glycine-conjugated bile acids. It should be noted that taurine-conjugation of bile acids is the default mode of conjugation in non-mammalian orders. Conversely, glycine conjugation is uncommon, occurring mainly in bovids and primates (Fig. 4) [9]. Oleanolic acid is a triterpene molecule extracted from olive leaves with a shape resembling that of lithocholic acid; it has also been identified as a TGR5 agonist in a luciferase assay based on studies using CHO cell lines transfected with TGR5 expression plasmid [26].
Fig. 4.
Comparative dose response assay of induced by various bile acids in CHO-TGR5 transfected cells. The figure summarizes the production of cAMP in CHO-TGR5 transfected cells by bile acids. LCA: lithocholic acid; DCA: deoxycholic acid; CDCA: chenodeoxycholic acid; CA: cholic acid. T = taurine conjugated; G = glycine conjugated; F = free (unconjugated) bile acid.
Reproduced with permission, Maruyama T et al., Identification of membrane-type receptor for bile acids (M-BAR). Biochem Biophys Res Commun. 2002 Nov 15;298(5):714-9 [8]. Copyright Elsevier.
In addition to bile acids and oleanolic acid, more potent selective TGR5 agonists derived from natural C24 bile acids have also been recently described by researchers at the University of Perugia, Italy [27,28]. So far, the most specific agonist synthesized from cholic acid is the 6α-ethyl-23(S)-methylcholic acid (S-EMCA). Another modified bile acid derivative, INT 767, has been synthesized and activates both TGR5 and FXR [29]. Further evaluation of the dual agonist showed the absence of cytotoxicity in HepG2 cells, its resistance to enzymatic cleavage, and the absence of inhibition of the human ether-á-go-go-related gene (hERG) potassium channel [29]. Additionally, other synthetic agonists have been developed as potential new targets for type 2 diabetes, namely 3-aryl-4-isoxazolecarboxamides. These are non-bile-acid-related compounds that display potent agonist affinity with human TGR5 as shown in a cAMP assay in U2-OS transfected cells. The compounds have satisfactory aqueous solubility, a low molecular weight, and a high membrane permeability [30,31].The class of the 2-aryl-3-aminomethylquinolines has also been tested in the same in vitro assay type [32]. However, none of these agonists have as yet been tested in clinical trials. The antibiotic ciprofloxacin has been proposed as a TGR5 ligand, although its binding affinity and signalling through cAMP assay in cell lines is yet to be demonstrated [33].
6. Intracellular signalling cascades induced by TRG5 activation in eukaryotic cells
Bile acid binding to TGR5 results in the activation of the adenylyl cyclase cAMP signalling pathways [9]. In addition, other cell-specific activation signalling cascades are also activated [22,24,25]. In the pancreatic β-cell line MIN6, TGR5 ligands, oleanolic acid, and S-EMCA selectively activated Gα(s) and increased intracellular cAMP, calcium, and phosphoinositide hydrolysis that was blocked by NF449 (a selective Gα(s) inhibitor) or U 73122 (PI hydrolysis inhibitor) [34]. In cultured neurons, a cAMP increase is accompanied by a rise in intracellular calcium, which may induce the production of reactive nitrogen oxide species [17]. In liver sinusoidal endothelial cells, bile acids activate the phosphorylation of endothelial nitric oxide synthase (NOS) through TGR5 activation and cAMP release, leading to nitric oxide (NO) synthesis [22]. In gallbladder smooth muscle, conversely to neurons and β-cells lines, TGR5 activation lowers the intracellular calcium levels, decreasing the rhythmic discharge of intracellular Ca2+ necessary to induce contraction [35].
7. Phenotype of the TGR5 knockout mice
In the absence of a selective receptor antagonist, the targeted disruption of the TGR5 receptor was performed in C57BL/6 mice [10,16]. TGR5−/− mice display a healthy and fertile phenotype with normal development. Light microscopic examination of organs’ tissue sections did not reveal developmental abnormalities in TGR5−/− compared to TGR5+/+ mice, suggesting that TGR5 is not required for normal organogenesis. Other cellular parameters such as haematological indices and routine serum chemistries blood (bilirubin, low and high density lipoproteins, triglycerides, and bile acids) were similar in knockout and wild type (WT) mice, as were measurements of glucose and other metabolites in the urines [10]. However, the liver mRNA expression of various cytochromes and proteins involved in the hepatic synthesis and transport of bile acids was higher in TGR5−/− than in WT mice, especially Cyp7a1, the rate-limiting enzyme in bile acid synthesis. This should logically lead to an increase of bile acid synthesis. However, the bile acid pool was not increased, suggesting that other homeostatic mechanisms may be involved in the knockout animals [10,16].
Phenotypic sex-specific differences were observed in parameters such as weight and metabolism, as both the heterozygous TGR5−/+ and homozygous TGR5−/− female mice exhibited a significantly higher body weight change than males in response to a high fat diet [16]. This was due to excessive fat accumulation despite an equivalent food intake and locomotor activity, suggesting a decrease in energy expenditure, without any explanation for this sex-related difference [16]. There is evidence that in TGR5 knockout mice fed with a high-fat diet, males exhibit impaired insulin sensitivity while females show improved insulin sensitivity. These data suggest a sex-related difference in the regulation of metabolic disorders through TGR5 [36].
In the gut, where TGR5 is densely expressed, microscopic alterations in the appearance of colonic mucosa occurred in TGR5−/− mice, including the abnormal morphology of colonic mucous cells associated with the altered molecular architecture of epithelial tight junctions. This led to a significant increase in intestinal permeability at the age of 12 months, associated with an overexpression of the main proteins of the tight junctions (zonulin-1 and occluding) in colonic epithelial cells; these characteristics showed a discontinuous pattern of distribution compared to WT animals [33].
8. Biological effects of TGR5 activation
8.1. Anti-inflammatory effects
The initial tissue screening that targeted TGR5 mRNA expression was performed in human monocytes [9]. Convergent findings showed that TGR5 was the receptor involved in the previously known immunosuppressive effect of bile acids in immune cells [14]. Indeed, TGR5 activation lowered the pro-inflammatory cytokines interleukin-1α (IL-1α), IL-1β, IL-6 and tumour necrosis factor-α (TNF-α). These cytokines are known to be synthesized by Kupffer cells in response to a lipopolysaccharide (LPS) challenge [25]. Of significance, the synthesis of pro-inflammatory cytokines after LPS stimulation is higher in isolated macrophages from TGR5−/− than those from TGR5+/+ mice, further supporting the role of TGR5 signalling in the downregulation of the inflammatory response to Gram-negative bacteria [37].
The anti-inflammatory effect of TGR5 is mediated by the inhibition of the pro-inflammatory transcriptional nuclear factor-κB (NF-κB) [37,38]. In macrophages from TGR5 knockout mice, the mRNA levels of various pro-inflammatory genes targeted by NF-κB (inducible NOS, interferon-inducible protein, and IL-1α) are higher than to those in macrophages from WT mice [38]. Likewise, in the macrophage cell line RAW264.7, TGR5 activation inhibited NF-κB activation via the cAMP signalling pathway [37]. When TGR5 was activated or overexpressed, NF-κB transcriptional activity was inhibited after LPS treatment, and was reversed in the presence of cAMP inhibitors [37]. Bile acids’ interaction with TGR5 was also shown to influence the differentiation of subtypes of dendritic cells into cells with an anti-inflammatory phenotype [39]. Dendritic cells are central regulators of the acquired and innate immunity, as antigen presenting cells. During differentiation of monocytes isolated from peripheral human blood into dendritic cells, the addition of bile acids lead to reduced IL-12 production when stimulated by bacterial antigen, in a dose-dependent manner. These effects on differentiation were only induced if bile acids or a specific TGR5-agonist was present at the start of culture, and were not observed in terminally differentiated cells. In addition, the down-regulation of TGR5 expression after differentiation into dendritic cells supports a critical role of the TGR5 expressed on monocytes in the initial phase of differentiation to dendritic cells [39].
8.2. Liver and biliary tree
TGR5 is also found in Kupffer cells and liver sinusoidal endothelial cells, where it activates endothelial NOS and induces NO release [22,25]. To date, TGR5 protein has not been detected in hepatocytes [40] but was found in the biliary tree and gallbladder epithelia [24], as well as in the apical membranes and primary cilium of cholangiocytes [23]. The proximity between synthesis and secretion of ligands (bile acids) by the hepatocytes, and the site of action in the biliary tree, where TGR5 is abundant, represent a paracrine mode of action for bile acids within this organ [41]. At a cellular level, immunohistochemical detection on human gallbladder showed that TGR5 is located in close proximity to the cystic fibrosis transmembrane conductance regulator (CFTR) and the apical sodium dependant bile salt uptake transporter (ASBT) [42]. Data obtained by using the CFTR inhibitor and TGR5 agonist suggested that TGR5 promotes biliary chloride secretion through CFTR [42]. As the primary cilium is a sensor regulating bile flow and composition, TGR5 should be a key player in this sensing mechanism. A recent study indicates that the genetic deletion of TGR5 abrogates the usual increase of bile flow, chloride, and bicarbonates secretion observed after partial hepatectomy in mice, and leads to a slower regeneration of the liver [43]. TGR5 is reported to be localized in different subcellular compartments of the cholangiocytes (primary cilia, plasma membrane, and subapical compartment), probably associated with different effects varying with its complex cellular repartition. Experimentally, after TGR5 stimulation, the ciliary localization is associated with lower cAMP levels and limits ciliated cholangiocytes’ proliferation; this is in contrast with the exact opposite effects in nonciliated cholangiocytes [44]. A functional study in mice showed that TGR5 decreased motility and increased filling of the gallbladder [45]. Likewise, in ex vivo experiments TGR5 activation reduced the gallbladder’s smooth muscle contractility, providing evidence of an important role of TGR5 in stimulating gallbladder smooth muscle relaxation and filling [35].
8.3. Energy homeostasis and metabolism
Watanabe et al.’s seminal observation established that TGR5 enhances energy expenditure, increases oxygen consumption, prevents obesity, and decreases insulin resistance in a mice model of obesity [46]. In human brown adipocytes and skeletal myocytes, bile acids interact with TGR5 and thereby activate the key enzyme iodothyronine deiodinase, that converts inactive thyroxine (T4) into intracellular active tri-iodothyronine (T3), a major component involved in cellular basal metabolism [46]. The protection against insulin resistance has been first attributed to intestinal secretion of glucagon-like peptide 1 (GLP-1) induced through the activation of TGR5. This was demonstrated in an enteroendocrine cell line, where TGR5 agonists increased GLP-1 release [47]. Moreover, evidence indicates that TGR5 is expressed in both mouse and human pancreatic cells islets. In pancreatic β-cells, TGR5 agonists can induce insulin secretion through a cAMP/Ca2+ pathway [34], confirming the critical role of bile acids in glucose homeostasis. Likewise, the TGR5 receptor agonist oleanolic acid, enhanced glucose tolerance by lowering glucose and insulin levels in a mouse model of obesity and diabetes [26]. These convergent findings point to bile acid-TGR5 interaction as a key regulator endpoint of basal metabolism regulation [48,49].
8.4. Cell proliferation, apoptosis, and growth factors
Bile acids have been identified to interact with the epithelial growth factor receptor (EGFR), as demonstrated in the colonic epithelial cell line CaCo-2 cells and in cholangiocytes [50,51]. However, whether this represents an interaction with the TGR5 pathway is still to be established.
Bile acids exert a protective effect against apoptosis in cholangiocytes [52,53]. In isolated sinusoidal endothelial cells, TGR5 activation increases the phosphorylation of the CD95 death receptor, which prevents CD95-dependent apoptosis [22,54]. In isolated hepatic stellate cells from rats, bile acids can trigger both proliferation and apoptosis through EGFR [55]. However, these findings were obtained with sulphated lithocholic acid at a concentration far above that occurring in the space of Disse.
TGR5 is also an activator of mitogen protein kinases. In gastric carcinoma cells treated with TGR5 small interference RNA, there is a decrease in deoxycholic acid-induced EGFR phosphorylation, which is involved in mitogen protein kinases activation [56]. In the chemical mouse model of liver carcinogenesis, using diethyl-nitrosamine to induce hepatocellular carcinoma, TGR5 activation exerts a protective effect against tumour development through the inhibition of the transcription factor STAT3 [57]. Importantly, TGR5 knockout mice had a higher incidence of diethylnitroamine induced hepatocellular carcinoma than WT mice, and in these mice STAT3 phosphorylation was 2-fold higher than in WT mice. At the cellular level, when the human liver cancer cell line HepG2 was transfected with the TGR5 expression plasmid, the ligand-activated TGR5 inhibited cell growth and decreased STAT3 phosphorylation [57]. These findings may be relevant to human diseases, as chronic activation of phosphorylated STAT3 is frequently detected in human liver tumours [58].
8.5. Intestinal motility and secretion
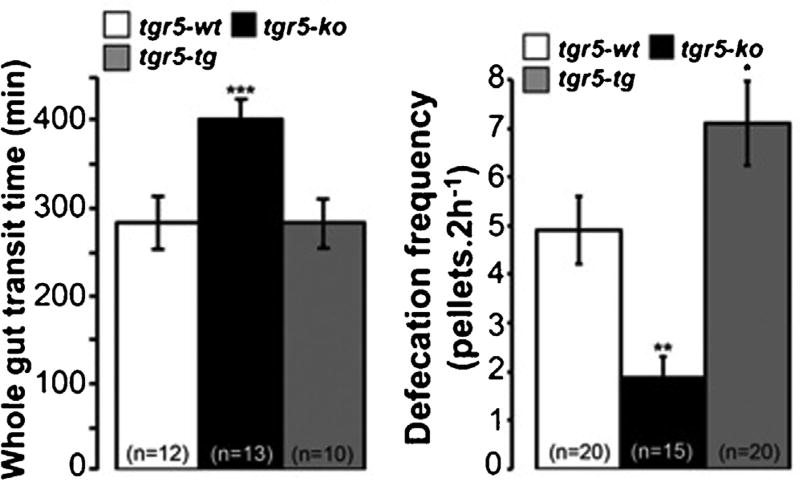
Recent studies by Bunnett and Corvera indicate the involvement of TGR5 in bile acids-induced motility [59]. Ex vivo, full-thickness segments of mouse colon stimulated by various TGR5 agonists induced peristalsis in WT mice, while this effect was absent in knockout mice. In vivo, TGR5−/− mice displayed a delayed colonic transit and a lower defecation frequency compared to WT (Fig. 5). Of mechanistic relevance was the demonstration that bile acids increased the release of the peristaltic transmitters, namely 5-hydroxytryptamine and calcitonin-gene related peptide, which were found in a flat sheet preparation of mice distal colon. Of significance, the mechanically induced peristalsis was not affected by TGR5 deletion, indicating a specific effect of bile acids on colon transit. These findings point to TGR5 as playing a physiological role in the regulation of propulsive motor function [59]. Presumably, this effect is mediated by plasma bile acids, which increase threefold postprandially.
Fig. 5.
Defecation frequency and whole gut transit time in WT and TGR5−/− mice. Reproduced with permission, from Alemi F et al., The receptor TGR5 mediates the prokinetic actions of intestinal bile acids and is required for normal defecation in mice. Gastroenterology. 2013 Jan;144(1):145-54 [59]. Copyright Elsevier.
In the same study, an observation showing that the faecal water content is lower in TGR5 knockout mice suggests that TGR5 may be critical in the regulation of the colonic water and electrolytes efflux. A recent work studying the bile acid-induced chloride secretion in the colon, by using a model of human colorectal cultured cells (T84 cells), showed that the chloride efflux [60] from the apical side uses CFTR as the ion transporter, through an intracellular cAMP signalling pathway: by detecting the cellular TGR5 mRNA expression, the authors suggested that this effect could be mediated through TGR5. However, in the absence of a functional assay using a specific TGR5 inhibitor, it was not possible to demonstrate its effective implication in chloride secretion.
9. TGR5: relevance in diseases
The pleiotropic expression and biological actions induced by the activation of TGR5 that was demonstrated in animal models or differentiated cell lines raise the question of whether TGR5 activation may alleviate or exacerbate specific human pathology as outlined in Table 1 [1–4].
Table 1.
Summary of TGR5 localization, known signalling pathways, biological actions, and potential implications for human disease.
| Reference | Tissue or cell line expressing TGR5 |
TGR5 intracellular actions | TGR5 biological action | Potential implications to human disorder |
|---|---|---|---|---|
| [46] | Brown adipocytes/skeletal myocytes | Iodothyrionine deiodinase activation | Conversion of inactive T4 in active T3 | Enhance basal metabolism |
| [37,64,67] | Arterial | Suppress Inflammation | Reduce development of atheroma plaques | Decreased atherosclerosis |
| [26,47,61–63] | Enteroendocrine cell | Mitochondrial oxidative phosphorylation | GLP-1 release | Multiple GLP-1 actions: improved diabetes control |
| [9,13,14,25,37–39] | Monocytes/macrophages | Decrease expression of NFkB induced genes | Reduction of IL-1 α, IL-1β, IL-6 and TNF-α | Inflammatory diseases improvement |
| [17] | Brain neurons | Increases intracellular calcium | Production of reactive nitrogen and oxygen species | Hepatic encephalopathy (Deleterious?) |
| [22] | Liver Sinusoid Endothelial cell | Phosphorylation of endothelial-NO-synthase | NO synthesis and release in liver | Increased hepatic blood flow |
| [23,24] | Biliary epithelium | CFTR activation | HCO3 secretion in biliary tree | Protection against fibrosing cholangiopathies |
| [19,20,59,77,81] | Enteric nervous system | - | Intestinal secretion and intestinal motility | Exacerbation of irritable bowel syndrome |
| [39,51,33] | Gut dendritic cells | - | Intestinal permeability and immune response | Improvement of inflammatory bowel diseases |
| [35,45] | Gallbladder smooth muscle | K ATP channels | Gallbladder filling | Gallstone formation |
| [73] | Pancreatic acinar cells | Intra acinar cell calcium transients | Activation of digestive zymogens in acini | Aggravation of acute pancreatitis |
| [56,91] | Gastric carcinoma cells | EGFR | Activation of mitogen protein kinases | Upper GI tract carcinogenesis? |
| [90] | Oesophagal adenocarcinoma cells | NADPH oxydase NOX5-S expression | Reactive oxygen species production | |
| [55,57] | Liver carcinoma cells | Decrease STAT 3 phosphorylation | Reduces tumorigenesis and cell growth | Protection against hepatocellular carcinoma |
T4: thyroxine; T3: tri-iodothyronine; GLP-1: glucagon like peptide 1; IL: interleukin; TNF-α: tumour necrosis factor alpha; NO: nitric oxide; CFTR: cystic fibrosis transmembrane conductance regulator; K ATP: adenosine triphosphate sensitive potassium channel; EGFR: epidermal growth factor receptor; NADPH: nicotinamide adenine dinucleotide phosphate.
9.1. Metabolic and cardiovascular disorders
Since the report of its key role in the regulation of the thyroid hormone cellular action and basal metabolism [46], the conditions of obesity, insulin resistance, and type 2 diabetes appear to be critically linked to TGR5 [49]. This is supported by the demonstration that TGR5 activation prevents obesity in mice [46], and treatment with a TGR5 agonist improves glucose tolerance in mice fed with a high-fat diet, as shown by the lowering of plasma glucose levels [26]. The key observations that directly point at TGR5 as a target for the treatment of metabolic disorders are the experimental evidences in animals that implicate TGR5 stimulation in the secretion of both GLP-1 in the gut and insulin in the pancreas [34,61]. In a diet-induced obesity mice model, TGR5 stimulation in the distal colon (using enema with TGR5 agonists) induces a GLP-1 release associated with an increase of GLP-1 in portal blood [62]. These experimental data in animals were confirmed by a clinical study performed in healthy male volunteers, showing that a rectal enema with sodium taurocholate rapidly increases circulating GLP-1 concentrations, suggesting that bile acids activate colonic enteroendocrine cells via TGR5 [63].
TGR5 appears relevant also for the pathogenesis of atherosclerosis, but via a mechanism independent from the well-known increased risk of atheroma development occurring in diabetes [49]. The atheroma plaque composition depends on intimate interactions between monocytes and arterial endothelium, where pro-inflammatory mechanisms first induce differentiation of monocytes in macrophages, and then facilitate the accumulation of lipids in their cytoplasm. TGR5 exerts a protective effect against atheroma development in the mouse model of atherosclerosis (LDL−/− mice) by curtailing inflammation and lipid accumulation [37]. In such mice, TGR5 activation reduces the size of atheroma plaques. A specific semi-synthetic TGR5 agonist attenuates atherosclerosis in LDL−/− TGR5+/+ mice, but not in double knockout mice (LDL−/− TGR5−/−). This is achieved by decreasing the intraplaque inflammation, the macrophage count within the plaque, and the macrophage activity [37]. Experimentally, in bovine and human endothelial cells, the TGR5 agonist taurolithocolate (TLCA) increases the production of NO, considered as a key antiatherogenic molecule [64].
Combined with the same protective effect against atheroma plaque development induced by FXR, TGR5 activation appears to be a new pharmacodynamic approach to treat atherosclerosis [65,66]. Of potential significance, in a recent clinical pilot prospective clinical study one of the authors of this review (H.D) observed differences in the bile acid profiles in sera of patients with or without coronary atheroma; specifically, a decreased plasma concentration of lithocholic acid, known to be the most potent activator of TGR5, was found. Lithocholic acid was the only predictor of coronary atheroma independently of patient gender [67]. It is possible that the decreased lithocholic acid concentration in serum may reduce the TGR5 anti-inflammatory signalling on macrophages, thereby weakening a natural brake against plaque formation. This new concept warrants further investigations to determine the validity of this clinical observation and whether the lithocholic acid concentration in sera can be used as a predictive marker indicating atheroma status or susceptibility in patients with cardiovascular disorders.
Overlapping metabolic and hepatic disorders, such as in nonalcoholic fatty liver disease, are also potential targets of future TGR5 agonists: histopathological analysis of the liver in the obese db/db mice model of obesity, diabetes, and dyslipidemia, shows that the administration of a dual FXR and TGR5 agonist (INT 767) decreases both steatosis and inflammation [68].
9.2. Hepatic and pancreatic disorders
Haemodynamic and vascular hepatic disorders result from alterations of sinusoidal vessels, examples being peliosis hepatis [69] or focal nodular hyperplasia [70]. Such pathologies may involve TGR5, which is known to induce the synthesis of NO, an effective vasodilator, in liver sinusoidal endothelial cells. In cirrhosis, the potential deleterious role of TGR5 in hepatic encephalopathy, a neuropsychological disorder caused by liver failure, seems worth investigating. This is based on the evidence that TGR5 activation by bile acids in cultured astrocytes and neurons has been linked to an intracellular increase in Ca2+, and the generation of reactive oxygen species [17]. These mediators may contribute to the feed-forward loop of hepatic encephalopathy. Interestingly, a marked downregulation of TGR5 mRNA expression has been observed in human cerebral cortex from cirrhotic patients compared to healthy subjects [17]. Ammonia, for long suspected to play a major role in the pathogenesis of hepatic encephalopathy, directly reduces TGR5 expression in cultured astrocytes, suggesting here a downregulation of the receptor in response to chronic elevation of the circulating ligands [17,71,72].
Other studies showed that genetic deletion of TGR5 protects against gallstone formation in mice [10]. We can speculate that TGR5 may play a role in gallbladder lithiasis and its consequences. Indeed, TGR5 activation induced gallbladder filling [45] through the inhibition of biliary smooth muscle cell activity or, possibly, the stimulation of sphincter of Oddi contraction (or both) [35]. However, it is not known whether patients with gallbladder lithiasis have different TGR5 expression in the gallbladder. A recent study suggests that TGR5 should have a protective role in human hepatocellular carcinoma [57].
Acute pancreatitis may also be linked to TGR5. In TGR5-knockout mice, acute biliary pancreatitis is associated with lower “hyperamylasemia, oedema, inflammation, and acinar cell injury” [73]. Whether changes in TGR5 signalling influence the severity of pancreatitis in clinical setting is worth exploring.
TGR5 is present in bile ducts, where its activation stimulates local bicarbonate secretion [24]. However, attempts to find sequence variations in the TGR5 gene associated with primary sclerosing cholangitis (PSC), have not been successful, despite the initially promising results [2,15,74]. The genome-wide association studies performed in patients with PSC, have revealed a statistically significant association between the TGR5 gene locus with PSC and ulcerative colitis. A single-nucleotide polymorphism in the noncoding region of exon 1 was statistically associated with PSC in different cohorts. Moreover, by sequencing the TGR5 gene from 267 patients with PSC, and by cloning epithelial cell lines with mutated TGR5 sequences in patients, Hov et al. identified five mutations able to reduce or abolish TGR5 function [15]. Therefore, TGR5 variants may contribute to disease susceptibility and influence disease course; however, their overall relevance for disease pathogenesis appears to be minor. Chronic cholestatic disorders are also known to induce itching, which is highly distressing for patients. Historically, the cause of such cholestatic pruritus has been attributed to increased plasma and skin concentrations of bile acids, although there is an imperfect correlation between the degree of bile acid retention and the magnitude of itching. A few years ago, the lysophosphatic acid was proposed as the pruritogen. A recent experimental study in mice may open new venues to elucidate the role of bile acids in cholestatic pruritus by showing an implication of TGR5. Bile acids activate mouse dorsal root ganglia via TGR5-mediated activation of gastrin-releasing peptide neurons that mediate pruritus. Interestingly, mice overexpressing TGR5 exhibit spontaneous pruritis, and TGR5-knockout mice itch less than WT mice after intradermal injections of TGR5 agonists. This fascinating study suggests a new approach to the treatment of this distressing problem [18].
9.3. Inflammatory bowel diseases and Irritable bowel syndrome
PSC is strongly linked to inflammatory bowel disease (IBD), mainly ulcerative colitis, affecting up to 80% of the patients with cholangiopathy [74,75]. The anti-inflammatory properties of TGR5 in immune cells suggest that TGR5 activation might be beneficial in inflammatory diseases. In addition, some genetic arguments exist to involve TGR5 in IBD. The TGR5 gene locus is on chromosome 2q35, close to a genetic variant (single-nucleotide polymorphism rs12612347) associated with both PSC and ulcerative colitis [15]. In a recent study, TGR5 has been found to be increased in an experimental model of colitis and in the colon of Crohn’s disease patients [33]. Another work showed that in human lamina propria mononuclear cells isolated from Crohn’s disease patients, the mRNA expression of TGR5 was upregulated in patients with inflammation compared to non-inflamed cases [76]. This was combined with an anti-inflammatory effect of secondary bile acids marked by a decrease in TNF-alpha production of macrophages that should be a compensatory mechanism to counterbalance the vicious circle of inflammation in IBD.
The preferential localization of TGR5 on sensory and motor neurons of the gut seem to indicate its potential involvement in gut motor function and visceral sensitivity. Functional bowel disorders, such as irritable bowel syndrome (IBS) [77], are characterized by abnormal bowel movements [78] and visceral hypersensitivity [79,80] as the hallmark of the disease. TGR5-knockout mice have a higher transit time and a lower defecation rate [59]. In humans, Camilleri et al. tested whether there was an association between single nucleotide polymorphism in the TGR5 sequence and IBS phenotypes (predominantly characterized by diarrhoea and constipation) in 414 patients. Their results indicate a potential association between TGR5 exon 1 polymorphisms and the overall transit time in a subgroup of patients [81]. As previously noted, the TGR5-knockout mouse exhibits microscopic changes in colonic epithelial structure and alteration of epithelial tight junctions, inducing an abnormal increase in epithelial permeability, reported as a contributing mechanism in the pathophysiology of both IBD [62] and IBS [82,83].
Of interest, the two most powerful natural agonists of TGR5 are lithocholic (3α-monohydroxy) and deoxycholic acid (3α,12α-dihydroxy) [9]. As noted before, in humans these bile acids are formed by bacterial 7-dehydroxylation of the primary bile acids, chenodeoxycholic acid (3α7α-dihydroxy) and cholic acid (3α,7α12α-trihydroxy). Ursodeoxycholic acid (3α,7β-dihydroxy) is widely used in the treatment of primary biliary cirrhosis, and in humans this bile acid can also be formed by bacterial epimerization of chenodeoxycholic acid [84]. Bacterial 7-dehydroxylation is mediated by strictly anaerobic bacteria and occurs only in the colon by a complex biochemical pathway involving a Δ4 Δ6 - intermediate. In humans, lithocholic acid and deoxycholic acid are the dominant faecal bile acids and can, in principle, signal to TGR5 that, if expressed, is localized on the apical membrane of colonic epithelial cells. Lithocholic acid, however, has an extremely low aqueous solubility at body temperature, and it seems unlikely that its concentration would be sufficiently high to activate TGR5 and cause GLP-1 release. Moreover, patients who take ursodeoxycholic acid for liver or biliary disease convert it entirely to lithocholic acid, and an improvement in diabetic status has never been reported in these patients.
A small fraction of lithocholic acid is absorbed from the colon and passes to the liver where it is efficiently removed. Upon entering the hepatocyte, lithocholic acid is not only conjugated with taurine or glycine, but is also sulphated, at least in humans. The sulphated lithocholic acid conjugates are secreted into bile and are not absorbed from the small intestine. The plasma concentration of unsulphated (but amidated with taurine or glycine) lithocholic acid is very low - probably less than 0.5 µM - and more than 95% is bound to albumin. Thus, it remains uncertain whether the very low concentration of lithocholyl conjugates in plasma could activate TGR5.
Deoxycholic acid is absorbed to a much greater extent - about 20–50% of that formed in the colon. Deoxycholic acid is efficiently extracted by the liver, conjugated with taurine or glycine, and circulates with the primary bile acids. Postprandial concentrations are in the range of 1–3 µM. In cholestasis, the proportion of deoxycholic acid in the circulating bile acids diminishes as cholestasis worsens.
In mouse, primary bile acids differ from man. There are only two - the taurine-conjugated β-muricholic acid (3α,6β,7β-trihydroxy-) and cholic acid. When the β-muricholic acid undergoes 7-dehydroxylation, murideoxycholic acid (3α,6β-dihydroxy) is formed. Its EC50 for TGR5 activation is 4.9 µM, about 10 times higher than that of lithocholic acid and 4 times higher than that of deoxycholic acid.
A recent study [61] from the laboratories of Auwerx and Schoonjans has presented convincing evidence that release of secondary bile acids from bile acid sequestrants results in TGR5 activation and GLP-1 release in mice, thus providing a mechanism for the well-known antidiabetic effects of bile acid sequestrants.
It seems remarkable that the stimulus for activation of TGR5 should be a secondary bile acid. Yet, in most mammals there is a symbiotic relationship between the whole organism and its microbiome in the distal intestine, and one way for the micro-biome to communicate to its host could be via secondary bile acids. Such an evolutionary development means that there would be less signalling during the first year of life when the anaerobic flora had not developed, or in animals lacking an anaerobic cecum where 7-dehydroxylation occurs.
9.4. Gastrointestinal cancer
Because some, but not all, epidemiological studies have shown an association between high levels of faecal bile acids and colon cancer [85,86], bile acids have been long suspected to be involved in colon carcinogenesis [87]. However, their role still remains controversial based on in vitro dual effects of bile acids on apoptosis and cell proliferation. Nonetheless, a recent study [88] shows that when deoxycholic acid is fed at a level of 0.2% in the diet does induce colon cancer in mice. A possible mechanism is the suppression of miR-199, which targets CAC1, a cell cycle-related protein, and thereby acts as a tumour suppressor [89].
TGR5 has not been associated with colonic carcinogenesis, whereas two in vitro studies have shown an implication of the receptor in gastric [48] and oesophageal carcinoma [90]. In humans, gastroduodenal reflux occurs as a complication of distal gastrectomy. The reflux of bile acids into the stomach and oesophagus is reported to increase the risk of gastric or oesophageal cancer [90–92]. TGR5 is implicated in the activation of carcinogenic pathways in gastric carcinoma cell lines [56]. Moreover, TGR5 is able to induce proliferation of human Barrett cell lines from Barrett’s oesophagus, an oesophageal metaplasic condition well-known for its risk of developing into oesophageal adenocarcinoma [56,90]. A recent clinical study analysing by Kaplan-Meier curves the survival of 171 patients with gastric adenocarcinoma showed that the greater the TGR5 immunoreactivity found in gastric adenocarcinoma specimens, the worse the patient survival [93].
A report from Chen et al. offers perspectives in human liver carcinogenesis, where TGR5 should have a protective role in human hepatocellular carcinoma [58].
10. Conclusion
Since the original characterization of TGR5 in 2002, a growing number of studies have explored not only TGR5 expression at both the gene and protein levels in various tissues but also the results of its activation on several intracellular signalling pathways. Bile acid conjugation with taurine renders bile acids’ membrane impermeable; however, the discovery of TGR5 function provides a mechanism by which conjugated bile acids can influence cellular activity without entering the cell. Several TGR5 signalling pathways activated by bile acids appear to be promising targets for the treatment of gastrointestinal diseases. Decreased pruritus in cholestatic liver disease, improvement of insulin resistance in type 2 diabetes, protection against obesity, and inhibition of atheroma development are pointed out as potential therapeutic approaches based on ligand-TGR5 interaction. Anti-inflammatory effects, especially on TNF-α secretion in monocytes could lead to the development of new molecules to curtail inflammatory diseases, such as rheumatoid arthritis and IBD. Clinical trials are needed to test whether the new semi-synthetic TGR5 agonists have clinical efficacy in any or all of these conditions.
Acknowledgments
We thank Dr Verena Keitel, from the Klinik für Gastroenterologie, Hepatologie und Infektiologie, Universitätsklinikum Düsseldorf, for providing her advice and comments, and for scrutiny of the accuracy of this review.
Footnotes
H.D. was supported by a Robert Tournut Grant from the Societé Nationale Française de Gastroentérologie, a research grant from the Philippe Foundation, a Mobility Grant from Assistance-Publique Hopitaux de Paris. Y.T is a recipient of a Research Career Scientist Award, and NIHDDK R01 57238 and DK-41301 (Animal Core).
Conflict of interest statement
A.F. Hofmann has served as a consultant to Intercept Pharmaceuticals, Inc. that plans to perform clinical trials with a TGR5 agonist. Henri Duboc and Yvette Taché have no conflict of interest to declare.
References
- 1.Chen X, Lou G, Meng Z, Huang W. TGR5: a novel target for weight maintenance and glucose metabolism. Experimental Diabetes Research. 2011;2011:853501. doi: 10.1155/2011/853501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hov JR, Keitel V, Schrumpf E, Häussinger D, Karlsen TH. TGR5 sequence variation in primary sclerosing cholangitis. Digestive Diseases. 2011;29:78–84. doi: 10.1159/000324138. [DOI] [PubMed] [Google Scholar]
- 3.Pols TWH, Noriega LG, Nomura M, Auwerx J, Schoonjans K. The bile acid membrane receptor TGR5 as an emerging target in metabolism and inflammation. Journal of Hepatology. 2011;54:1263–72. doi: 10.1016/j.jhep.2010.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhong M. TGR5 as a therapeutic target for treating obesity. Current Topics in Medicinal Chemistry. 2010;10:386–96. doi: 10.2174/156802610790980576. [DOI] [PubMed] [Google Scholar]
- 5.Wang H, Chen J, Hollister K, Sowers LC, Forman BM. Endogenous bile acids are ligands for the nuclear receptor FXR/BAR. Molecular Cell. 1999;3:543–53. doi: 10.1016/s1097-2765(00)80348-2. [DOI] [PubMed] [Google Scholar]
- 6.Lefebvre P, Cariou B, Lien F, Kuipers F, Staels B. Role of bile acids and bile acid receptors in metabolic regulation. Physiological Reviews. 2009;89:147–91. doi: 10.1152/physrev.00010.2008. [DOI] [PubMed] [Google Scholar]
- 7.Sinal CJ, Tohkin M, Miyata M, Ward JM, Lambert G, Gonzalez FJ. Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell. 2000;102:731–44. doi: 10.1016/s0092-8674(00)00062-3. [DOI] [PubMed] [Google Scholar]
- 8.Maruyama T, Miyamoto Y, Nakamura T, et al. Identification of membrane-type receptor for bile acids (M-BAR) Biochemical and Biophysical Research Communications. 2002;298:714–9. doi: 10.1016/s0006-291x(02)02550-0. [DOI] [PubMed] [Google Scholar]
- 9.Kawamata Y, Fujii R, Hosoya M, et al. A G protein-coupled receptor responsive to bile acids. Journal of Biological Chemistry. 2003;278:9435–40. doi: 10.1074/jbc.M209706200. [DOI] [PubMed] [Google Scholar]
- 10.Vassileva G, Golovko A, Markowitz L, et al. Targeted deletion of Gpbar1 protects mice from cholesterol gallstone formation. Biochemical Journal. 2006;398:423–30. doi: 10.1042/BJ20060537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Müssig K, Staiger H, Machicao F, et al. Preliminary report: genetic variation within the GPBAR1 gene is not associated with metabolic traits in white subjects at an increased risk for type 2 diabetes mellitus. Metabolism - Clinical and Experimental. 2009;58:1809–11. doi: 10.1016/j.metabol.2009.06.012. [DOI] [PubMed] [Google Scholar]
- 12.Gene Symbol Report | HUGO Gene Nomenclature Committee. Available at: http://www.genenames.org/data/hgnc_data.php?hgnc_id=19680.
- 13.Greve JW, Gouma DJ, Buurman WA. Bile acids inhibit endotoxin-induced release of tumor necrosis factor by monocytes: an in vitro study. Hepatology. 1989;10:454–8. doi: 10.1002/hep.1840100409. [DOI] [PubMed] [Google Scholar]
- 14.Calmus Y, Guechot J, Podevin P, Bonnefis MT, Giboudeau J, Poupon R. Differential effects of chenodeoxycholic and ursodeoxycholic acids on interleukin 1, interleukin 6 and tumor necrosis factor-alpha production by monocytes. Hepatology. 1992;16:719–23. doi: 10.1002/hep.1840160317. [DOI] [PubMed] [Google Scholar]
- 15.Hov JR, Keitel V, Laerdahl JK, et al. Mutational characterization of the bile acid receptor TGR5 in primary sclerosing cholangitis. PLoS ONE. 2010;5:e12403. doi: 10.1371/journal.pone.0012403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maruyama T, Tanaka K, Suzuki J, et al. Targeted disruption of G protein-coupled bile acid receptor 1 (Gpbar1/M-Bar) in mice. Journal of Endocrinology. 2006;191:197–205. doi: 10.1677/joe.1.06546. [DOI] [PubMed] [Google Scholar]
- 17.Keitel V, Görg B, Bidmon HJ, et al. The bile acid receptor TGR5 (Gpbar-1) acts as a neurosteroid receptor in brain. Glia. 2010;58:1794–805. doi: 10.1002/glia.21049. [DOI] [PubMed] [Google Scholar]
- 18.Alemi F, Kwon E, Poole DP, et al. The TGR5 receptor mediates bile acid-induced itch and analgesia. Journal of Clinical Investigation. 2013;123:1513–30. doi: 10.1172/JCI64551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Poole DP, Godfrey C, Cattaruzza F, et al. Expression and function of the bile acid receptor GpBAR1 (TGR5) in the murine enteric nervous system. Neurogastroenterology and Motility. 2010;22:814–25. doi: 10.1111/j.1365-2982.2010.01487.x. e227-228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Karlström L, Cassuto J, Jodal M, Lundgren O. The importance of the enteric nervous system for the bile-salt-induced secretion in the small intestine of the rat. Scandinavian Journal of Gastroenterology. 1983;18:117–23. doi: 10.3109/00365528309181570. [DOI] [PubMed] [Google Scholar]
- 21.Karlström L, Cassuto J, Jodal M, Lundgren O. Involvement of the enteric nervous system in the intestinal secretion induced by sodium deoxycholate and sodium ricinoleate. Scandinavian Journal of Gastroenterology. 1986;21:331–40. doi: 10.3109/00365528609003083. [DOI] [PubMed] [Google Scholar]
- 22.Keitel V, Reinehr R, Gatsios P, et al. The G-protein coupled bile salt receptor TGR5 is expressed in liver sinusoidal endothelial cells. Hepatology. 2007;45:695–704. doi: 10.1002/hep.21458. [DOI] [PubMed] [Google Scholar]
- 23.Keitel V, Ullmer C, Häussinger D. The membrane-bound bile acid receptor TGR5 (Gpbar1) is localized in the primary cilium of cholangiocytes. Biological Chemistry. 2010;391:785–9. doi: 10.1515/BC.2010.077. [DOI] [PubMed] [Google Scholar]
- 24.Keitel V, Häussinger D. TGR5 in the biliary tree. Digestive Diseases. 2011;29:45–7. doi: 10.1159/000324127. [DOI] [PubMed] [Google Scholar]
- 25.Keitel V, Donner M, Winandy S, Kubitz R, Häussinger D. Expression and function of the bile acid receptor TGR5 in Kupffer cells. Biochemical and Biophysical Research Communications. 2008;372:78–84. doi: 10.1016/j.bbrc.2008.04.171. [DOI] [PubMed] [Google Scholar]
- 26.Sato H, Genet C, Strehle A, et al. Anti-hyperglycemic activity of a TGR5 agonist isolated from Olea europaea. Biochemical and Biophysical Research Communications. 2007;362:793–8. doi: 10.1016/j.bbrc.2007.06.130. [DOI] [PubMed] [Google Scholar]
- 27.Pellicciari R, Gioiello A, Macchiarulo A, et al. Discovery of 6alpha-ethyl-23(S)-methylcholic acid (S-EMCA, INT-777) as a potent and selective agonist for the TGR5 receptor, a novel target for diabesity. Journal of Medicinal Chemistry. 2009;52:7958–61. doi: 10.1021/jm901390p. [DOI] [PubMed] [Google Scholar]
- 28.Gioiello A, Rosatelli E, Nuti R, et al. Patented TGR5 modulators: a review (2006 - present) Expert Opinion on Therapeutic Patent. 2012;22(12):1399–414. doi: 10.1517/13543776.2012.733000. [DOI] [PubMed] [Google Scholar]
- 29.Rizzo G, Passeri D, De Franco F, et al. Functional characterization of the semisynthetic bile acid derivative INT-767, a dual farnesoid X receptor and TGR5 agonist. Molecular Pharmacology. 2010;78:617–30. doi: 10.1124/mol.110.064501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Budzik BW, Evans KA, Wisnoski DD, et al. Synthesis and structure-activity relationships of a series of 3-aryl-4-isoxazolecarboxamides as a new class of TGR5 agonists. Bioorganic and Medicinal Chemistry Letters. 2010;20:1363–7. doi: 10.1016/j.bmcl.2010.01.003. [DOI] [PubMed] [Google Scholar]
- 31.Evans KA, Budzik BW, Ross SA, et al. Discovery of 3-aryl-4-isoxazolecarboxamides as TGR5 receptor agonists. Journal of Medicinal Chemistry. 2009;52:7962–5. doi: 10.1021/jm901434t. [DOI] [PubMed] [Google Scholar]
- 32.Herbert MR, Siegel DL, Staszewski L, et al. Synthesis and SAR of 2-aryl-3-aminomethylquinolines as agonists of the bile acid receptor TGR5. Bioorganic and Medicinal Chemistry Letters. 2010;20:5718–21. doi: 10.1016/j.bmcl.2010.08.014. [DOI] [PubMed] [Google Scholar]
- 33.Cipriani S, Mencarelli A, Chini MG, et al. The bile acid receptor GPBAR-1 (TGR5) modulates integrity of intestinal barrier and immune response to experimental colitis. PLoS ONE. 2011;6:e25637. doi: 10.1371/journal.pone.0025637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kumar DP, Rajagopal S, Mahavadi S, et al. Activation of transmembrane bile acid receptor TGR5 stimulates insulin secretion in pancreatic β cells. Biochemical and Biophysical Research Communications. 2012;427:600–5. doi: 10.1016/j.bbrc.2012.09.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lavoie B, Balemba OB, Godfrey C, et al. Hydrophobic bile salts inhibit gallbladder smooth muscle function via stimulation of GPBAR1 receptors and activation of KATP channels. Journal of Physiology (London) 2010;588(Pt 17):3295–305. doi: 10.1113/jphysiol.2010.192146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vassileva G, Hu W, Hoos L, et al. Gender-dependent effect of Gpbar1 genetic deletion on the metabolic profiles of diet-induced obese mice. Journal of Endocrinology. 2010;205:225–32. doi: 10.1677/JOE-10-0009. [DOI] [PubMed] [Google Scholar]
- 37.Pols TWH, Nomura M, Harach T, et al. TGR5 activation inhibits atherosclerosis by reducing macrophage inflammation and lipid loading. Cell Metabolism. 2011;14:747–57. doi: 10.1016/j.cmet.2011.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang YD, Chen WD, Yu D, Forman BM, Huang W. The G-protein-coupled bile acid receptor, Gpbar1 (TGR5), negatively regulates hepatic inflammatory response through antagonizing nuclear factor kappa light-chain enhancer of activated B cells (NF-κB) in mice. Hepatology. 2011;54:1421–32. doi: 10.1002/hep.24525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ichikawa R, Takayama T, Yoneno K, et al. Bile acids induce monocyte differentiation toward interleukin-12 hypo-producing dendritic cells via a TGR5-dependent pathway. Immunology. 2012;136:153–62. doi: 10.1111/j.1365-2567.2012.03554.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Keitel V, Häussinger D. Perspective: TGR5 (Gpbar1) in liver physiology and disease. Clinics and Research in Hepatology and Gastroenterology. 2012 doi: 10.1016/j.clinre.2012.03.008. [DOI] [PubMed] [Google Scholar]
- 41.Keitel V, Kubitz R, Häussinger D. Endocrine and paracrine role of bile acids. World Journal of Gastroenterology. 2008;14:5620–9. doi: 10.3748/wjg.14.5620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Keitel V, Cupisti K, Ullmer C, Knoefel WT, Kubitz R, Häussinger D. The membrane-bound bile acid receptor TGR5 is localized in the epithelium of human gallbladders. Hepatology. 2009;50:861–70. doi: 10.1002/hep.23032. [DOI] [PubMed] [Google Scholar]
- 43.Péan N, Doignon I, Garcin I, et al. The receptor TGR5 protects the liver from bile acid overload during liver regeneration in mice. Hepatology. 2013;58(4):1451–60. doi: 10.1002/hep.26463. [DOI] [PubMed] [Google Scholar]
- 44.Masyuk AI, Huang BQ, Radtke BN, et al. Ciliary subcellular localization of TGR5 determines the cholangiocyte functional response to bile acid signaling. American Journal of Physiology - Gastrointestinal and Liver Physiology. 2013;304:G1013–24. doi: 10.1152/ajpgi.00383.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li T, Holmstrom SR, Kir S, et al. The G protein-coupled bile acid receptor, TGR5, stimulates gallbladder filling. Molecular Endocrinology. 2011;25:1066–71. doi: 10.1210/me.2010-0460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Watanabe M, Houten SM, Mataki C, et al. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature. 2006;439:484–9. doi: 10.1038/nature04330. [DOI] [PubMed] [Google Scholar]
- 47.Thomas C, Gioiello A, Noriega L, et al. TGR5-mediated bile acid sensing controls glucose homeostasis. Cell Metabolism. 2009;10:167–77. doi: 10.1016/j.cmet.2009.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pols TWH, Noriega LG, Nomura M, Auwerx J, Schoonjans K. The bile acid membrane receptor TGR5: a valuable metabolic target. Digestive Diseases. 2011;29:37–44. doi: 10.1159/000324126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Porez G, Prawitt J, Gross B, Staels B. Bile acid receptors as targets for the treatment of dyslipidemia and cardiovascular disease. Journal of Lipid Research. 2012;53:1723–37. doi: 10.1194/jlr.R024794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Werneburg NW, Yoon J-H, Higuchi H, Gores GJ. Bile acids activate EGF receptor via a TGF-alpha-dependent mechanism in human cholangiocyte cell lines. American Journal of Physiology - Gastrointestinal and Liver Physiology. 2003;285:G31–6. doi: 10.1152/ajpgi.00536.2002. [DOI] [PubMed] [Google Scholar]
- 51.Raimondi F, Santoro P, Barone MV, et al. Bile acids modulate tight junction structure and barrier function of Caco-2 monolayers via EGFR activation. American Journal of Physiology - Gastrointestinal and Liver Physiology. 2008;294:G906–13. doi: 10.1152/ajpgi.00043.2007. [DOI] [PubMed] [Google Scholar]
- 52.Marzioni M, Ueno Y, Glaser S, et al. Cytoprotective effects of taurocholic acid feeding on the biliary tree after adrenergic denervation of the liver. Liver International. 2007;27:558–68. doi: 10.1111/j.1478-3231.2007.01443.x. [DOI] [PubMed] [Google Scholar]
- 53.Marzioni M, LeSage GD, Glaser S, et al. Taurocholate prevents the loss of intrahepatic bile ducts due to vagotomy in bile duct-ligated rats. American Journal of Physiology - Gastrointestinal and Liver Physiology. 2003;284:G837–52. doi: 10.1152/ajpgi.00398.2002. [DOI] [PubMed] [Google Scholar]
- 54.Reinehr R, Häussinger D. Inhibition of bile salt-induced apoptosis by cyclic AMP involves serine/threonine phosphorylation of CD95. Gastroenterology. 2004;126:249–62. doi: 10.1053/j.gastro.2003.09.044. [DOI] [PubMed] [Google Scholar]
- 55.Sommerfeld A, Reinehr R, Häussinger D. Bile acid-induced epidermal growth factor receptor activation in quiescent rat hepatic stellate cells can trigger both proliferation and apoptosis. Journal of Biological Chemistry. 2009;284:22173–83. doi: 10.1074/jbc.M109.005355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yasuda H, Hirata S, Inoue K, Mashima H, Ohnishi H, Yoshiba M. Involvement of membrane-type bile acid receptor M-BAR/TGR5 in bile acid-induced activation of epidermal growth factor receptor and mitogen-activated protein kinases in gastric carcinoma cells. Biochemical and Biophysical Research Communications. 2007;354:154–9. doi: 10.1016/j.bbrc.2006.12.168. [DOI] [PubMed] [Google Scholar]
- 57.Chen W-D, Yu D, Forman BM, Huang W, Wang YD. The deficiency of G-protein-coupled bile acid receptor gpbar1 (TGR5) enhances chemically-induced liver carcinogenesis. Hepatology. 2012 doi: 10.1002/hep.26019. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 58.He G, Yu G-Y, Temkin V, et al. Hepatocyte IKKbeta/NF-kappaB inhibits tumor promotion and progression by preventing oxidative stress-driven STAT3 activation. Cancer Cells. 2010;17:286–97. doi: 10.1016/j.ccr.2009.12.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Alemi F, Poole DP, Chiu J, et al. The receptor TGR5 mediates the prokinetic actions of intestinal bile acids and is required for normal defecation in mice. Gastroenterology. 2012 doi: 10.1053/j.gastro.2012.09.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ao M, Sarathy J, Domingue J, Alrefai WA, Rao MC. Chenodeoxycholic acid stimulates Cl− secretion via cAMP signaling and increases cystic fibrosis transmembrane conductance regulator phosphorylation in T84 cells. American Journal of Physiology- Cell Physiology. 2013;305:C447–56. doi: 10.1152/ajpcell.00416.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Harach T, Pols TWH, Nomura M, et al. TGR5 potentiates GLP-1 secretion in response to anionic exchange resins. Science Reporter. 2012;2:430. doi: 10.1038/srep00430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Potthoff MJ, Potts A, He T, et al. Colesevelam suppresses hepatic glycogenolysis by TGR5-mediated induction of GLP-1 action in DIO mice. American Journal of Physiology - Gastrointestinal and Liver Physiology. 2013;304:G371–80. doi: 10.1152/ajpgi.00400.2012. http://dx.doi.org/10.1152/ajpgi.00400.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wu T, Bound MJ, Standfield SD, et al. Effects of rectal administration of taurocholic acid on glucagon-like peptide-1 and peptide YY secretion in healthy humans. Diabetes, Obesity and Metabolism. 2013;15:474–7. doi: 10.1111/dom.12043. [DOI] [PubMed] [Google Scholar]
- 64.Kida T, Tsubosaka Y, Hori M, et al. Bile acid receptor TGR5 agonism induces NO production and reduces monocyte adhesion in vascular endothelial cells. Arteriosclerosis, Thrombosis, and Vascular Biology. 2013;33(7):1663–9. doi: 10.1161/ATVBAHA.113.301565. [DOI] [PubMed] [Google Scholar]
- 65.Hartman HB, Gardell SJ, Petucci CJ, Wang S, Krueger JA, Evans MJ. Activation of farnesoid X receptor prevents atherosclerotic lesion formation in LDLR−/−and apoE−/− mice. Journal of Lipid Research. 2009;50:1090–100. doi: 10.1194/jlr.M800619-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mencarelli A, Cipriani S, Renga B, et al. The bile acid sensor FXR protects against dyslipidemia and aortic plaques development induced by the HIV protease inhibitor ritonavir in mice. PLoS ONE. 2010;5:e13238. doi: 10.1371/journal.pone.0013238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Duboc H, Aelion H, Rainteau D, et al. Crosstalk between the hepatologist and the cardiologist: a future place for the lithocholic acid as a coronary atheroma risk factor? Hepatology. 2012;56(6):2426. doi: 10.1002/hep.25839. [DOI] [PubMed] [Google Scholar]
- 68.McMahan RH, Wang XX, Cheng LL, et al. Bile acid receptor activation modulates hepatic monocyte activity and improves nonalcoholic fatty liver disease. Journal of Biological Chemistry. 2013;288:11761–70. doi: 10.1074/jbc.M112.446575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Tzirogiannis KN, Papadimas GK, Kondyli VG, et al. Peliosis hepatis: microscopic and macroscopic type, time pattern, and correlation with liver cell apoptosis in a model of toxic liver injury. Digestive Diseases and Sciences. 2006;51:1998–2006. doi: 10.1007/s10620-006-9242-x. [DOI] [PubMed] [Google Scholar]
- 70.Hartleb M, Gutkowski K, Milkiewicz P. Nodular regenerative hyperplasia: evolving concepts on underdiagnosed cause of portal hypertension. World Journal of Gastroenterology. 2011;17:1400–9. doi: 10.3748/wjg.v17.i11.1400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Reinehr R, Görg B, Becker S, et al. Hypoosmotic swelling and ammonia increase oxidative stress by NADPH oxidase in cultured astrocytes and vital brain slices. Glia. 2007;55:758–71. doi: 10.1002/glia.20504. [DOI] [PubMed] [Google Scholar]
- 72.Görg B, Qvartskhava N, Keitel V, et al. Ammonia induces RNA oxidation in cultured astrocytes and brain in vivo. Hepatology. 2008;48:567–79. doi: 10.1002/hep.22345. [DOI] [PubMed] [Google Scholar]
- 73.Perides G, Laukkarinen JM, Vassileva G, Steer ML. Biliary acute pancreatitis in mice is mediated by the G-protein-coupled cell surface bile acid receptor Gpbar1. Gastroenterology. 2010;138:715–25. doi: 10.1053/j.gastro.2009.10.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Karlsen TH, Franke A, Melum E, et al. Genome-wide association analysis in primary sclerosing cholangitis. Gastroenterology. 2010;138:1102–11. doi: 10.1053/j.gastro.2009.11.046. [DOI] [PubMed] [Google Scholar]
- 75.Loftus EV, Jr, Harewood GC, Loftus CG, et al. PSC-IBD: a unique form of inflammatory bowel disease associated with primary sclerosing cholangitis. Gut. 2005;54:91–6. doi: 10.1136/gut.2004.046615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yoneno K, Hisamatsu T, Shimamura K, et al. TGR5 signalling inhibits the production of pro-inflammatory cytokines by in vitro differentiated inflammatory and intestinal macrophages in Crohn’s disease. Immunology. 2013;139:19–29. doi: 10.1111/imm.12045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Camilleri M. Peripheral mechanisms in irritable bowel syndrome. New England Journal of Medicine. 2012;367:1626–35. doi: 10.1056/NEJMra1207068. [DOI] [PubMed] [Google Scholar]
- 78.Manabe N, Wong BS, Camilleri M, Burton D, McKinzie S, Zinsmeister AR. Lower functional gastrointestinal disorders: evidence of abnormal colonic transit in a 287 patient cohort. Neurogastroenterology and Motility. 2010;22:293–382. doi: 10.1111/j.1365-2982.2009.01442.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sabate J-M, Veyrac M, Mion F, et al. Relationship between rectal sensitivity, symptoms intensity and quality of life in patients with irritable bowel syndrome. Alimentary Pharmacology and Therapeutics. 2008;28:484–90. doi: 10.1111/j.1365-2036.2008.03759.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Coffin B, Bouhassira D, Sabaté J-M, Barbe L, Jian R. Alteration of the spinal modulation of nociceptive processing in patients with irritable bowel syndrome. Gut. 2004;53:1465–70. doi: 10.1136/gut.2003.031310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Camilleri M, Vazquez-Roque MI, Carlson P, Burton D, Wong BS, Zinsmeister AR. Association of bile acid receptor TGR5 variation and transit in health and lower functional gastrointestinal disorders. Neurogastroenterology and Motility. 2011;23:995–9. doi: 10.1111/j.1365-2982.2011.01772.x. e458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Gerova VA, Stoynov SG, Katsarov DS, Svinarov DA. Increased intestinal permeability in inflammatory bowel diseases assessed by iohexol test. World Journal of Gastroenterology. 2011;17:2211–5. doi: 10.3748/wjg.v17.i17.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bertiaux-Vandaële N, Youmba SB, Belmonte L, et al. The expression and the cellular distribution of the tight junction proteins are altered in irritable bowel syndrome patients with differences according to the disease subtype. American Journal of Gastroenterology. 2011;106:2165–73. doi: 10.1038/ajg.2011.257. [DOI] [PubMed] [Google Scholar]
- 84.Ridlon JM, Kang D-J, Hylemon PB. Bile salt biotransformations by human intestinal bacteria. Journal of Lipid Research. 2006;47:241–59. doi: 10.1194/jlr.R500013-JLR200. [DOI] [PubMed] [Google Scholar]
- 85.Hill MJ. Bile acids and colorectal cancer: hypothesis. European Journal of Cancer Prevention. 1991;1(Suppl. 2):69–74. doi: 10.1097/00008469-199110002-00012. [DOI] [PubMed] [Google Scholar]
- 86.Reddy BS, Wynder EL. Metabolic epidemiology of colon cancer. Fecal bile acids and neutral sterols in colon cancer patients and patients with adenomatous polyps. Cancer. 1977;39:2533–9. doi: 10.1002/1097-0142(197706)39:6<2533::aid-cncr2820390634>3.0.co;2-x. [DOI] [PubMed] [Google Scholar]
- 87.Debruyne PR, Bruyneel EA, Li X, Zimber A, Gespach C, Mareel MM. The role of bile acids in carcinogenesis. Mutation Research. 2001;480–481:359–69. doi: 10.1016/s0027-5107(01)00195-6. [DOI] [PubMed] [Google Scholar]
- 88.Bernstein C, Holubec H, Bhattacharyya AK, et al. Carcinogenicity of deoxycholate, a secondary bile acid. Archives of Toxicology. 2011;85:863–71. doi: 10.1007/s00204-011-0648-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kong Y, Bai PS, Sun H, et al. The deoxycholic acid targets miRNA-dependent CAC1 gene expression in multidrug resistance of human colorectal cancer. International Journal of Biochemistry and Cell Biology. 2012;44(12):2321–32. doi: 10.1016/j.biocel.2012.08.006. [DOI] [PubMed] [Google Scholar]
- 90.Hong J, Behar J, Wands J, et al. Role of a novel bile acid receptor TGR5 in the development of oesophageal adenocarcinoma. Gut. 2010;59:170–80. doi: 10.1136/gut.2009.188375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lundegårdh G, Adami HO, Helmick C, Zack M, Meirik O. Stomach cancer after partial gastrectomy for benign ulcer disease. New England Journal of Medicine. 1988;319:195–200. doi: 10.1056/NEJM198807283190402. [DOI] [PubMed] [Google Scholar]
- 92.Lundegårdh G, Adami HO, Helmick C, Zack M. Risk of cancer following partial gastrectomy for benign ulcer disease. British Journal of Surgery. 1994;81:1164–7. doi: 10.1002/bjs.1800810827. [DOI] [PubMed] [Google Scholar]
- 93.Cao W, Tian W, Hong J, et al. Expression of bile acid receptor TGR5 in gastric adenocarcinoma. American Journal of Physiology - Gastrointestinal and Liver Physiology. 2013;304:G322–7. doi: 10.1152/ajpgi.00263.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]