Abstract
The present study aimed to observe the expression of circadian gene clock circadian regulator (CLOCK) in ovarian cancer cells and the effects of circadian gene CLOCK on cis-dichlorodiamine platinum (cisplatin) resistance in ovarian cancer cells. The expression of CLOCK mRNA and protein in cisplatin-sensitive A2780 and cisplatin-resistant CP70 cells were detected by quantitative polymerase chain reaction and western blot assay. Cisplatin-sensitive A2780 and cisplatin-resistant CP70 cells were treated with different concentrations of cisplatin for 48 h, and the expression of hCLOCK protein in the two types of cells was detected by western blot assay. RNA interference method was used to knock down the expression of CLOCK in cisplatin-resistant CP70 cells. Subsequently, the cisplatin-resistant CP70 cells were treated with cisplatin. The proliferation of cisplatin-resistant CP70 cells was observed following treatment with cisplatin. The expression of CLOCK mRNA was significantly higher in cisplatin-resistant CP70 cells (1.58±0.49) compared with cisplatin-sensitive A2780 cells (0.44±0.13) (P<0.01). Western blot assay results demonstrated that the expression of CLOCK protein was significantly greater in the cisplatin-resistant CP70 cells (1.47±0.34) compared with the cisplatin-sensitive A2780 cells (0.48±0.15) (P<0.01). Following the treatment of A2780 and CP70 cells with cisplatin, CLOCK protein expression increased with an increased concentration of cisplatin, in a dose-dependent manner (P<0.01). Following the knockdown of CLOCK in cisplatin-resistant CP70 cells by RNA interference, cisplatin treatment was able to significantly inhibit the proliferation of cells and induce apoptosis (P<0.01). The expression of circadian gene CLOCK in ovarian cancer cells was strongly associated with cisplatin resistance. The upregulation of circadian gene CLOCK in ovarian cancer cells may reduce its sensitivity to cisplatin treatment.
Keywords: circadian gene, ovarian cancer, resistance, cisplatin, chemotherapy
Introduction
Ovarian cancer is one of the most common types of cancer in women, and the American cancer society estimated there would be approximately 22,000 new cases of ovarian cancer and 14,000 mortalities caused by the disease in 2017 in the United States, since ovarian cancer causes more deaths than any other cancer of the female reproductive system (1). The most common ovarian cancer types are known as epithelial ovarian cancer, which have the highest mortality rate amongst gynecological malignant tumors in females (2). More than 70% of patients with ovarian cancer have been found in advanced stage (International Federation of Gynecology and Obstetrics, FIGO stage III or IV) (3,4). Cis-dichlorodiamine platinum (cisplatin) is the main treatment for advanced ovarian cancer. However, the majority of patients with cisplatin resistance cannot benefit from chemotherapy (5). A variety of factors are involved in the emergence of cisplatin resistance, including increased drug efflux, abnormal drug targeting, enhanced DNA repair and altered apoptotic pathway (6–11). The basic mechanism for the emergence of chemotherapeutic drug resistance remains poorly understood. At present, there is a lack of effective drugs to reduce resistance to chemotherapy. Circadian genes are important genes, which regulate biological activity and includes period circadian clock (PER)1, PER2, PER3, timeless circadian clock (TIM), clock circadian regulator (CLOCK), brain and muscle Arnt-like protein (BMAL), cryptochrome circadian clock 1 (CRY)1 and CRY2 (11). Of these genes, CLOCK gene was the first gene identified, and has been revealed to be strongly associated with sensitivity of various tumor types to chemotherapy, including gastric cancer, cholangiocarcinoma and colorectal cancer (12–15). There are a limited number of studies that have investigated the association between the CLOCK gene and resistance to chemotherapy in ovarian cancer.
The present study observed CLOCK gene expression in cisplatin-sensitive cell line A2780 and cisplatin-resistant cell line CP70.
The effects of cisplatin on the proliferation and apoptosis of cisplatin-resistant cell line CP70 following the knockdown of CLOCK in cisplatin-resistant CP70 cells were investigated, and the effects of CLOCK on chemotherapy resistance in ovarian cancer were discussed.
Materials and methods
Cells and main reagents
Cisplatin-sensitive A2780 and cisplatin-resistant CP70 cell lines (Shanghai Bogoo Biotechnology Co., Ltd., Shanghai, China) were incubated in Dulbecco's modified Eagle's medium (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) containing 10% fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.). Rat anti-human CLOCK antibody and horseradish peroxidase-labeled goat anti-rat immunoglobulin G (IgG) were purchased from Santa Cruz Biotechnology, Inc., Dallas, TX, USA. Quantitative polymerase chain reaction (qPCR) primers were synthesized by Invitrogen (Thermo Fisher Scientific, Inc.). CytoBuster protein extraction reagent was purchased from Novagen (Merck KGaA, Darmstadt, Germany), and cisplatin was obtained from Sigma-Aldrich (Merck KGaA). Additionally, small-interfering (si)CLOCK small RNA fragments (Ambion; Thermo Fisher Scientific, Inc.), cell transfection reagent (Lonza Group, Ltd., Basel, Switzerland) and protease inhibitor phenylmethanesulfony fluoride (Thermo Fisher Scientific, Inc.) were also used. Phenylmethanesulfony fluoride was added to PBS prior to use.
Reverse transcription (RT)-qPCR
Total RNA was extracted using Trizol reagent (Thermo Fisher Scientific). cDNAs were synthesized using a transcriptor first strand cDNA synthesis kit (Roche Applied Science, Penzberg, Germany), according to the manufacturer's protocol. mRNA levels were measured using using a FastStart Universal SYBR Green Master kit (Roche Applied Science) in a Light Cycler 96 (Roche Applied Science, Penzberg, Germany). Primer sequences were as follows: CLOCK forward, 5′-ACACCCAGAAGGAAGAGCAA-3′ reverse, 5′-GCGAGAACGCTTTGCTTTAG-3′; GAPDH forward, 5′-ATGTCGTGGAGTCTACTGGC-3′, reverse 5′-AGGATGCATTGCTGACAATC-3′. Reaction conditions are as follows: Pre-denaturation at 94°C for 5 min, 40 cycles of 94°C for 30 sec, 60°C for 40 sec and 72°C for 40 sec, followed by 72°C for 10 min. Target gene fragments were amplified with a DNA thermal cycler. GAPDH was used as an internal reference. Relative mRNA levels were calculated using the 2−ΔΔCq method (16). Data were calculated from three independent experiments.
Cell treatment
Cisplatin-sensitive A2780 and cisplatin-resistant cell line CP70 cells (1×105/ml) (Shanghai Gefan Biotechnology Co., Ltd., Shanghai, China) were treated with 0, 32 and 64 µM dipeptidyl peptidase-4 (Exalpha Biologicals, Inc., Shirley, MA, USA) for 48 h at 37°C.
Western blot assay
Total protein was extracted from cisplatin-sensitive A2780 and cisplatin-resistant CP70 cells in the logarithmic phase. Cisplatin-treated cells were digested with 0.25% trypsin, and total protein was extracted using CytoBuster protein extraction reagent. An equal volume of the protein (30 µg) was loaded onto 8% SDS-PAGE separating gel and 5% stacking gel and transferred to nitrocellulose membranes by semi-dry method. The membranes were blocked with Tris-buffered saline and Tween-20 containing 5% bovine serum albumin (Gibco; Thermo Fisher Scientific, Inc.) for 2 h at room temperature, and incubated with rat anti-human CLOCK antibody (1:1,500; cat no. 3896-100; BioVision, Inc., Milpitas, CA, USA) at 4°C overnight. On the following day, the membranes were washed three times with 0.1% Tris-buffered saline and Tween-20 for 5 min each, incubated with horseradish peroxidase-labeled IgG secondary antibody at room temperature for 1 h and washed three times with 0.1% Tris-buffered saline and Tween-20. The membranes were incubated in SuperSignal West Pico substrate (Thermo Fisher Scientific, Inc.) for visualization, and were incubated with β-actin at 55°C for 20 min, densitometry of the western blotting was analyzed with Image Pro Plus 6.0 (cat no. 20910; Media Cybernetics, Inc., Rockville, MD, USA). β-actin (1:1,500; cat no. MAB8929; R&D Systems, Inc., Minneapolis, MN, USA), served as the internal reference. The experiments were conducted at least in triplicate.
Cell transfection
The cell transfection reagent as mentioned previously was used. The cells were transfected with 100 nM of siCLOCK small RNA fragments for 48 h according to the manufacturer's protocol. At 24 h after transfection, an MTT assay was performed.
MTT assay
Following the knockdown of CLOCK protein expression, cisplatin-resistant CP70 cells at a density of 1×105/ml and control cells (cells without gene knockout) at a density of 1×105/ml were incubated in a 96-well plate at 37°C. A total of 20 µl MTT was added in each well for 4 h at 37°C. Following removal of the supernatant, 150 µl dimethyl sulfoxide was added, followed by a low oscillation speed for 10 min. Optical density was measured at 490 nm.
Statistical analysis
All data were analyzed with SPSS software (version 16.0; SPSS, Inc., Chicago, IL, USA). t-test was used to compare the differences in data between two groups. One-way analysis of variance was utilized to compare the differences among multiple groups. P<0.05 was considered to indicate a statistically significant difference.
Results
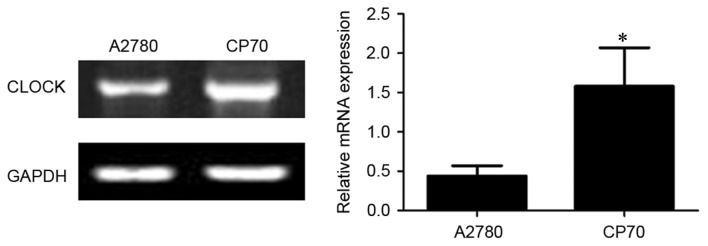
CLOCK mRNA expression in two types of ovarian cancer cells
As shown in Fig. 1, the expression of CLOCK mRNA was significantly higher in cisplatin-resistant CP70 cells (1.58±0.49) compared with cisplatin-sensitive A2780 cells (0.44±0.13) (P<0.01).
Figure 1.
Quantitative polymerase chain reaction for CLOCK mRNA expression in ovarian cancer tissue. CLOCK, clock circadian regulator. *P<0.01 vs. cisplatin-sensitive A2780 cells.
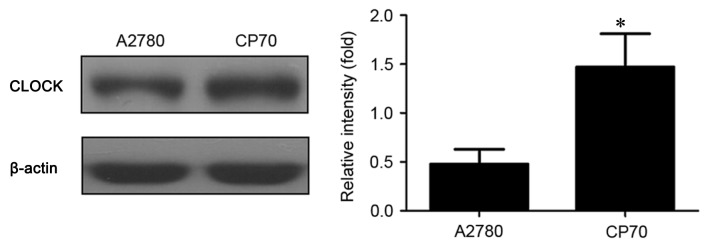
CLOCK protein expression in two types of ovarian cancer cells
As exhibited in Fig. 2, the expression of CLOCK protein was significantly higher in cisplatin-resistant CP70 cells (1.47±0.34) compared with cisplatin-sensitive A2780 cells (0.48±0.15) (P<0.01).
Figure 2.
Western blot assay of CLOCK protein expression in A2780 and CP70 ovarian cancer cells. CLOCK, clock circadian regulator. *P<0.01 vs. cisplatin-sensitive A2780 cells.
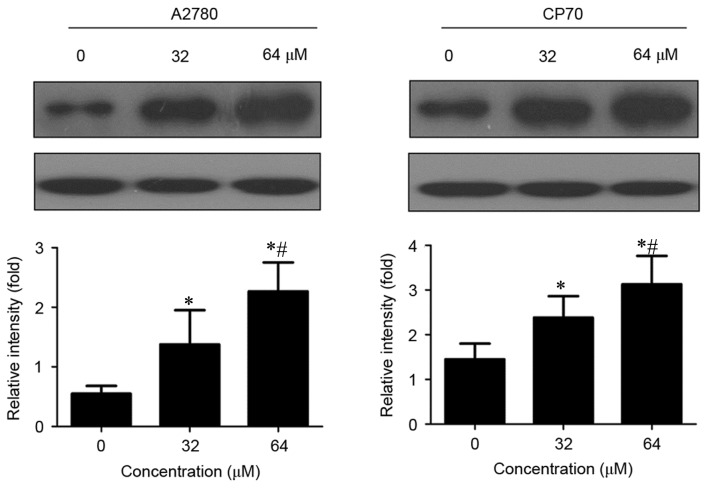
Effects of cisplatin on CLOCK protein expression in two types of ovarian cancer cells
CLOCK protein expression was significantly increased in cisplatin-treated A2780 and CP70 cells compared with untreated cells (P<0.01; Fig. 3). Furthermore, CLOCK protein expression gradually increased with an increased concentration of cisplatin (P<0.01).
Figure 3.
Effects of cisplatin on CLOCK protein expression in A2780 and CP70 cells. CLOCK, clock circadian regulator. *P<0.01 vs. untreated cells. #P<0.01 vs. 32 µM cisplatin-treated cells.
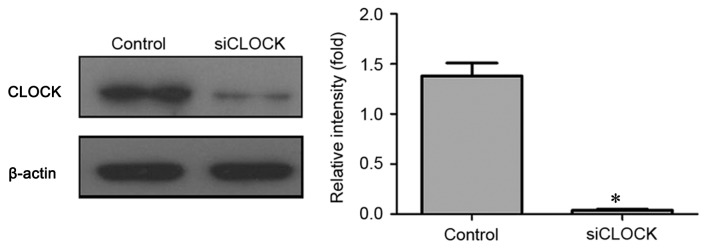
Confirming the effects of CLOCK knockdown on protein expression
As illustrated in Fig. 4, CLOCK siRNA transfection was able to significantly knockdown CLOCK protein expression in cisplatin-resistant CP70 cells compared with control cells (P<0.01).
Figure 4.
Western blot assay of CLOCK protein expression following CLOCK knockdown. CLOCK, clock circadian regulator; si, small-interfering. *P<0.01 vs. control cells.
Proliferation and apoptosis of cisplatin-resistant CP70 cells following CLOCK knockdown
As shown in Fig. 5, treatment with cisplatin did not significantly affect the proliferation and apoptosis of cisplatin-treated CP70 cells (P>0.05). By contrast following CLOCK knockdown, treatment with cisplatin was able to significantly inhibit the proliferation of CP70 cells and induce its apoptosis (P<0.01).
Figure 5.
Proliferation and apoptosis of CP70 cells following CLOCK knockdown, as detected by MTT assay. DDP, cisplatin. *P<0.01, vs. control cells. CLOCK, clock circadian regulator.
Discussion
Ovarian cancer is the fourth most common malignant tumor in women and is the leading cause of mortality from gynecologic malignancies. Due to its high mortality rate, ovarian cancer has become a global public health problem (17–20). The overall 5-year survival rate in the United States is 45%, the 1-year survival rate is 72% and the 10-year survival rate is 35% (21). For cases where a diagnosis is made early in the disease, when the cancer is still confined to the primary site, the 5-year survival rate is 92.7% (22). Approximately 70% of women with the advanced disease respond to initial treatment, a majority of whom attain complete remission, but half of these women experience recurrence 1–4 years following treatment (23). Therefore, if ovarian cancer can be diagnosed at an early stage, patients can get better treatment. Nevertheless, >70% of the patients with ovarian cancer have been diagnosed at advanced stages (FIGO stage III or IV) (4). The main method for advanced ovarian cancer is cisplatin treatment. However, the emergence of cisplatin resistance in the majority of patients with ovarian cancer reduces the effects of the chemotherapeutics (5).
A variety of different factors are involved in the emergence of cisplatin resistance, including increased drug efflux, abnormal drug targeting, enhanced DNA repair and altered apoptotic pathway (6–11). Molecular mechanisms of drug resistance in cisplatin-based chemotherapy remain unclear. There is a lack of effective drugs to reduce the resistance to chemotherapy.
Circadian rhythm is an endogenous adaptation mechanism in the process of long-term biological evolution and a basic characteristic of life activity. Circadian genes are important genes, which regulate biological activity and include PER1, PER2, PER3, TIM, CLOCK, BMAL, CRY1 and CRY2 (11). The CLOCK gene was the first gene discovered and was identified as a circadian clock gene. CLOCK gene is located on the long arm of chromosome 4 (4p12) and contains >20 exons (24). The CLOCK gene is not only expressed in normal tissue and cells. Abnormal expression of CLOCK gene can also be detected in a number of types of tumors (25–27). It was also demonstrated that the knockdown of CLOCK was able to increase the apoptosis of glioma cells (28). CLOCK gene has been revealed to be strongly associated with sensitivity to chemotherapy in various tumors, including gastric cancer, cholangiocarcinoma and colorectal cancer (13–15).
However, there are a limited number of studies that have investigated the association between CLOCK gene and chemotherapy resistance in ovarian cancer. Therefore, the present study observed CLOCK gene expression in cisplatin-sensitive A2780 and cisplatin-resistant CP70 cells and investigated the effects of cisplatin treatment on the proliferation and apoptosis of cisplatin-resistant CP70 cells following CLOCK knockdown. The effects of CLOCK gene on chemotherapy resistance in ovarian cancer were also discussed.
The present study first compared CLOCK mRNA and protein expression in two ovarian cancer cell lines (A2780 and CP70). Results demonstrated that CLOCK mRNA and protein expression was significantly lower in cisplatin-sensitive A2780 cells compared with cisplatin-resistant CP70 cells (P<0.01), indicating that CLOCK gene expression was strongly associated with cisplatin resistance in ovarian cancer cells. To further verify the association between CLOCK gene and cisplatin resistance in ovarian cancer cells, cisplatin-sensitive A2780 and cisplatin-resistant CP70 cells were treated with different concentrations of cisplatin. Results showed that CLOCK protein expression increased with an increased concentration of cisplatin in the two cell lines following cisplatin treatment in a dose-dependent manner (P<0.01), which further suggested that CLOCK gene was associated with cisplatin resistance in ovarian cancer cells. To confirm the precise effect of CLOCK gene on cisplatin resistance in ovarian cancer cells, the present study knocked down the expression of CLOCK protein in cisplatin-resistant CP70 cells by RNA interference. The results showed that cisplatin treatment was able to significantly suppress the proliferation of CP70 cells, and induce its apoptosis following the knockdown of CLOCK protein (P<0.01). In summary, the expression of circadian gene CLOCK was strongly associated with cisplatin resistance in ovarian cancer cells. The increase in the expression of circadian gene CLOCK may reduce the sensitivity to cisplatin treatment in ovarian cancer cells.
Acknowledgements
Not applicable.
Funding
The project was supported by the China Postdoctoral Science Foundation grant (grant no. 2013M542498).
Availability of data and materials
All relevant data are included in the present study.
Authors' contributions
HX conceived and designed the experiments, and wrote the manuscript. ZW, GM and HC conducted the experiments, collected, analyzed and interpreted the data. All authors reviewed the manuscript.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Smith RA, Andrews KS, Brooks D, Fedewa SA, Manassaram-Baptiste D, Saslow D, Brawley OW, Wender RC. Cancer screening in the United States, 2017: A review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin. 2017;67:100–121. doi: 10.3322/caac.21392. [DOI] [PubMed] [Google Scholar]
- 2.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. doi: 10.3322/caac.21166. [DOI] [PubMed] [Google Scholar]
- 3.Knutson KL, Karyampudi L, Lamichhane P, Preston C. Targeted immune therapy of ovarian cancer. Cancer Metastasis Rev. 2015;34:53–74. doi: 10.1007/s10555-014-9540-2. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 4.Killedar A, Stutz MD, Sobinoff AP, Tomlinson CG, Bryan TM, Beesley J, Chenevix-Trench G, Reddel RR, Pickett HA. A common cancer risk-associated allele in the hTERT locus encodes a dominant negative inhibitor of telomerase. PLoS Genet. 2015;11:e1005286. doi: 10.1371/journal.pgen.1005286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yan C, Yang F, Zhou C, Chen X, Han X, Liu X, Ma H, Zheng W. MCT1 promotes the cisplatin-resistance by antagonizing Fas in epithelial ovarian cancer. Int J Clin Exp Pathol. 2015;8:2710–2718. [PMC free article] [PubMed] [Google Scholar]
- 6.Vallo S, Michaelis M, Rothweiler F, Bartsch G, Gust KM, Limbart DM, Rödel F, Wezel F, Haferkamp A, Cinatl J., Jr Drug-resistant urothelial cancer cell lines display diverse sensitivity profiles to potential second-line therapeutics. Transl Oncol. 2015;8:210–216. doi: 10.1016/j.tranon.2015.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Takano M, Kakizoe S, Kawami M, Nagai J, Patanasethnont D, Sripanidkulchai B, Yumoto R. Modulation of P-glycoprotein function and multidrug resistance in cancer cells by Thai plant extracts. Pharmazie. 2014;69:823–828. [PubMed] [Google Scholar]
- 8.Territo PR, Maluccio M, Riley AA, McCarthy BP, Fletcher J, Tann M, Saxena R, Skill NJ. Evaluation of 11C-acetate and 18F-FDG PET/CT in mouse multidrug resistance gene-2 deficient mouse model of hepatocellular carcinoma. BMC Med Imaging. 2015;15:15. doi: 10.1186/s12880-015-0058-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jing X, Zhang H, Hu J, Su P, Zhang W, Jia M, Cheng H, Li W, Zhou G. β-arrestin 2 is associated with multidrug resistance in breast cancer cells through regulating MDR1 gene expression. Int J Clin Exp Pathol. 2015;8:1354–1363. [PMC free article] [PubMed] [Google Scholar]
- 10.Sun Y, Liu JH, Jin L, Sui YX, Han LL, Huang Y. Effect of autophagy-related beclin1 on sensitivity of cisplatin-resistant ovarian cancer cells to chemotherapeutic agents. Asian Pac J Cancer Prev. 2015;16:2785–2791. doi: 10.7314/APJCP.2015.16.7.2785. [DOI] [PubMed] [Google Scholar]
- 11.Sun Y, Liu JH, Jin L, Sui YX, Lai L, Yang Y. Inhibition of Beclin 1 expression enhances cisplatin-induced apoptosis through a mitochondrial-dependent pathway in human ovarian cancer SKOV3/DDP cells. Oncol Res. 2014;21:261–269. doi: 10.3727/096504014X13946388748992. [DOI] [PubMed] [Google Scholar]
- 12.Song JM, Hu X, Fu X, Liu K, Li GM. Effects of circadian genes hClock and hBmal1 on migration and invasion of SGC-7901 cells. Xiandai Zhongliu Yixue. 2013;21:2678–2681. [Google Scholar]
- 13.Hu ML, Yeh KT, Lin PM, Hsu CM, Hsiao HH, Liu YC, Lin HY, Lin SF, Yang MY. Deregulated expression of circadian clock genes in gastric cancer. BMC Gastroenterol. 2014;14:67. doi: 10.1186/1471-230X-14-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Filipski E, Subramanian P, Carrière J, Guettier C, Barbason H, Lévi F. Circadian disruption accelerates liver carcinogenesis in mice. Mutat Res. 2009;680:95–105. doi: 10.1016/j.mrgentox.2009.10.002. [DOI] [PubMed] [Google Scholar]
- 15.Fang L, Yang Z, Zhou J, Tung JY, Hsiao CD, Wang L, Deng Y, Wang P, Wang J, Lee MH. Circadian clock gene cry2 degradation is involved in chemoresistance of colorectal cancer. Mol Cancer Ther. 2015;14:1476–1487. doi: 10.1158/1535-7163.MCT-15-0030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 17.Yan-Hong H, Jing L, Hong L, Shan-Shan H, Yan L, Ju L. Association between alcohol consumption and the risk of ovarian cancer: A meta-analysis of prospective observational studies. BMC Public Health. 2015;15:223. doi: 10.1186/s12889-015-1355-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cui X, Li L, Yan G, Meng K, Lin Z, Nan Y, Jin G, Li C. High expression of NQO1 is associated with poor prognosis in serous ovarian carcinoma. BMC Cancer. 2015;15:244. doi: 10.1186/s12885-015-1271-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gao L, Ye X, Ma RQ, Cheng HY, Han HJ, Cui H, Wei LH, Chang XH. Low programmed cell death 5 expression is a prognostic factor in ovarian cancer. Chin Med J (Engl) 2015;128:1084–1090. doi: 10.4103/0366-6999.155100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lambrechts S, Lambrechts D, Despierre E, Van Nieuwenhuysen E, Smeets D, Debruyne PR, Renard V, Vroman P, Luyten D, Neven P, et al. Genetic variability in drug transport, metabolism or DNA repair affecting toxicity of chemotherapy in ovarian cancer. BMC Pharmacol Toxicol. 2015;16:2. doi: 10.1186/s40360-015-0001-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rooth C. Ovarian cancer: Risk factors, treatment and management. Br J Nurs. 2013;22:S23–S30. doi: 10.12968/bjon.2013.22.Sup17.S23. [DOI] [PubMed] [Google Scholar]
- 22.Zhang Q, Burdette JE, Wang JP. Integrative network analysis of TCGA data for ovarian cancer. BMC Syst Biol. 2014;8:1338. doi: 10.1186/s12918-014-0136-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Longoria TC, Eskander RN. Immune checkpoint inhibition: Therapeutic implications in epithelial ovarian cancer. Recent Pat Anticancer Drug Discov. 2015;10:133–144. doi: 10.2174/1574892810666150504121000. [DOI] [PubMed] [Google Scholar]
- 24.Jung H, Choe Y, Kim H, Park N, Son GH, Khang I, Kim K. Involvement of CLOCK: BMAL1 heterodimer in serum-responsive mPer1 induction. Neuroreport. 2003;14:15–19. doi: 10.1097/00001756-200301200-00003. [DOI] [PubMed] [Google Scholar]
- 25.Bjarnason GA, Jordan RC, Sothern RB. Circadian variation in the expression of cell-cycle proteins in human oral epithelium. Am J Pathol. 1999;154:613–622. doi: 10.1016/S0002-9440(10)65306-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Uth K, Sleigh R. Deregulation of the circadian clock constitutes a significant factor in tumorigenesis: A clockwork cancer. Part II. In vivo studies. Biotechnol Biotechnol Equip. 2014;28:379–386. doi: 10.1080/13102818.2014.925298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tavano F, Pazienza V, Fontana A, Burbaci FP, Panebianco C, Saracino C, Lombardi L, De Bonis A, di Mola FF, di Sebastiano P, et al. SIRT1 and circadian gene expression in pancreatic ductal adenocarcinoma: Effect of starvation. Chronobiol Int. 2015;32:497–512. doi: 10.3109/07420528.2014.1003351. [DOI] [PubMed] [Google Scholar]
- 28.Wang F, Li C, Yongluo, Chen L. The circadian gene clock plays an important role in cell apoptosis and the DNA damage response in vitro. Technol Cancer Res Treat. 2016;15:480–486. doi: 10.1177/1533034615585433. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are included in the present study.