Abstract
Cisplatin is used for the treatment of a range of solid malignant tumors; however, with prolonged treatment durations, the sensitivity of tumor cells to the drug decreases owing to an unclear mechanism of drug resistance. The present study aimed to investigate whether breast-cancer-tissue-derived mesenchymal stem cells (BC-MSCs) are involved in mediating the effects of cisplatin on breast cancer cells, and which component of the BC-MSC conditioned medium (BC-MSC-CM) exhibited an anti-apoptotic effect. Cytokines/chemokines in BC-MSC-CM were quantified using a Luminex immunoassay, and reverse transcription-quantitative polymerase chain reaction analysis detected interleukin-6 (IL-6) levels in MCF-7 cells following different treatments. MTT and flow cytometry analysis measured cell vitality and apoptosis, respectively, and activation of signal transduced and activator of transcription 3 (STAT3) was evaluated by western blotting. BC-MSCs reversed the pro-apoptotic effect of cisplatin and enhanced the proliferation of MCF-7 cells more potently than bone-marrow-derived MSCs. Further study revealed that BC-MSCs secreted IL-6 to protect MCF-7 cells from apoptosis and promote their survival. Neutralizing IL-6 with a specific antibody partially inhibited the IL-6/STAT3 signaling pathway and reversed the promoter role of BC-MSCs in MCF-7 cells. Taken together, the findings of the present study indicated that BC-MSCs decreased the level of cisplatin-induced apoptosis in MCF-7 cells by activating the IL-6/STAT3 pathway in cancer cells. BC-MSCs, as important cells in the tumor microenvironment, have a key role in the treatment of breast cancer.
Keywords: cisplatin, breast cancer, breast-cancer-derived mesenchymal stem cells, apoptosis, interleukin-6, tumor microenvironment
Introduction
Breast cancer is the leading cause of cancer-associated mortality among women worldwide (1). Although there are a range of therapeutic methods available to treat breast cancer, including surgery, chemotherapy, endocrine therapy, radiation and targeted therapy, there remains a large proportion therapeutic failures, subsequently resulting in cancer recurrence and metastasis (2).
Cisplatin, a platinum-containing antineoplastic drug, is a chemotherapeutic agent used to treat a range of solid malignant tumors, including ovarian cancer, breast cancer, gastric carcinoma and rectal carcinoma (3,4). Previous studies have revealed that cisplatin exerts its anticancer effects by activating the apoptotic pathway and inducing cell death (5–7). Apoptosis is the main response induced in tumor cells upon treatment chemotherapeutic agents (8). Cisplatin is an antitumor agent with a high rate of success in treating tumors; however, resistance to this therapy can develop. Evidence indicates the involvement of the tumor microenvironment (TME) in acquisition of chemoresistance (9,10). The TME influences the growth of tumors and affects the outcome of chemotherapy (11,12). Whether the TME has a role in cisplatin-triggered chemoresistance in breast cancer is unclear.
The TME is a dynamic system that consists of complex non-malignant cells, extracellular matrix (ECM) and signaling molecules that communicate with cancer cells. The non-malignant cells include fibroblasts, endothelial cells and immune cells that, together with the surrounding ECM, affect tumor activity. Mesenchymal stem cells (MSCs), one of the pivotal components of the TME, are a focus of research in the progression of tumors (13,14). Notably, there is evidence to suggest that MSCs release cytokines and growth factors to influence the behavior of tumor in a paracrine manner (15–17). In particular, owing to the influence on the tumor itself and the inflammatory TME, tumor-tissue-derived MSCs are widely studied (14,18). Previous studies have demonstrated that tumor progression, enhancement of metastatic potential, and resistance to chemo- and radiotherapy may all be attributed to MSCs (19–21). Whether breast-cancer-tissue-derived mesenchymal stem cells (BC-MSCs) are able to promote chemoresistance of cisplatin requires further investigation.
In the present study, it was revealed that BC-MSCs enhanced the proliferation of breast cancer cells and decreased cisplatin triggered apoptosis in MCF-7 cell. Interleukin-6 (IL-6) was released by BC-MSCs, which mediated a reduction in apoptosis induced by cisplatin by activating the signal transducer and activator of transcription 3 (STAT3) signaling pathway. This study revealed a novel mechanism of drug resistance of cisplatin.
Materials and methods
Cells and reagents
The human breast cancer MCF-7 cell line was obtained from the American Type Culture Collection (Manassas, VA, USA) and cultured in Dulbecco's modified Eagle medium (DMEM) (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 15% fetal bovine serum (FBS) (Gibco; Thermo Fisher Scientific, Inc.) at 37°C with 5% CO2 atmosphere in a humidified incubator. Breast cancer tissues were obtained from female patients (n=12) aged 40–60 years (mean 52 years), who underwent surgical operation in the First People's Hospital of Lianyungang from April 2015 to July 2015 (Jiangsu, China), all patients provided written informed consent to participate in the study and all experiment protocols were approved by the Medical Ethics Committee of First People's Hospital of Lianyungang. The cell isolation procedure was typically undertaken within 4 h of tumor removal. In brief, fresh tissue specimens were collected, washed with PBS, cut into 1 mm3-sized pieces and put into culture dishes for 30 min at 37°C. Then, tissue samples were maintained in DMEM supplemented with 10% FBS, 1% penicillin and streptomycin at 37°C with 5% CO2. Medium was replaced every three days after the initial plating. When adherent fibroblast-like cells confluence reached ~80%, the cells were passaged into new flasks for further expansion. Cells at passage 3–4 were used for the evaluation of the experiments. Bone marrow-mesenchymal stem cells (BM-MSCs) from patients (n=12) aged 3–60 years (mean, 22 years) with suspected blood system diseases who were diagnosed with no hematological disease or other cancers at the First People's Hospital of Lianyungang from April 2015 to July 2015 (Jiangsu, China) were chosen as the MSC controls.
Flow cytometry analysis
To investigate the surface antigen markers of different passages of BC-MSCs and BM-MSCs, flow cytometric analysis was performed using fluorescein isothiocyanate (FITC)-conjugated or phycoerythrin (PE)-conjugated antibodies: CD14 (cat. no. 561712; dilution, 1:50), CD34 (cat. no. 555822; dilution, 1:50) and CD45 (cat. no. 560976; dilution, 1:50) were FITC-conjugated, CD29 (cat. no. 561795; dilution, 1:50), CD44 (cat. no. 561858; dilution, 1:50), CD90 (cat. no. 561970; dilution, 1:50) and CD105 (cat. no. 562380; dilution, 1:50) were PE-conjugated. Mouse PE-IgG1 (cat. no. 349043; dilution, 1:50) and FITC-IgG1 (cat. no. 349041; dilution, 1:50) isotypic immunoglobulins were used as controls (BD Biosciences, San Jose, CA, USA). BC-MSCs and BM-MSCs cells (1.0×106) were trypsinized, washed twice in PBS, incubated with monoclonal antibodies for 30 min on ice at 4°C and then washed with PBS. Labeled cells were analyzed using a FACSCanto II flow cytometer (BD Biosciences).
Multidifferentiation capacity
To determine the ability of BC-MSCs to undergo osteogenic and adipogenic differentiation, BC-MSCs were seeded in 35-mm plates at 3×104 cells/cm2 and cultured in L-DMEM containing 15% FBS. The next day, the medium was changed to osteogenic medium or adipogenic medium (both from Cyagen Biosciences, Sunnyvale, CA, USA) in accordance with the culture protocol. After 2 weeks, the osteogenic differentiation of BC-MSCs was examined by alkaline phosphatase staining at room temperature for 15 min. At 3 weeks later, adipocytes were stained with Oil-Red-O at room temperature for 15 min to confirm the ability of adipogenic differentiation of BC-MSCs. A fluorescence-inverted microscope (Olympus Corporation, Tokyo, Japan) was used to observe the staining results at ×100 and ×400 magnification.
Preparation of MSC-conditioned medium
A total of (1.0×106) BC-MSC cells were seeded in 35-mm plates and cultured in L-DMEM with 15% FBS. The following day, the media was removed and the cells were washed with PBS, re-incubated with fresh medium for 48 h. Next, BC-MSC-conditioned medium (BC-MSC-CM) and BM-MSC-conditioned medium (BM-MSC-CM) were collected, and centrifuged at room temperature to remove possible cell debris (1,000 × g for 10 min).
MTT assay
To investigate the effect of cisplatin, BC-MSC-CM combination cisplatin on the viability of MCF-7 cells, an MTT assay was performed. Briefly, MCF-7 cells were plated at a density of 1×104 cells/well in a 96-well rounded bottom plate. After incubation for 12 h, cells were incubated with cisplatin for another 48 h at 37°C by a continuous induction from 2.5 to 80 µM (2.5, 5, 10, 20, 40 and 80 µM) in a stepwise increasing concentration manner in the presence or absence of BC-MSC-CM. Control medium was used as a control. All the cells were cultured for a further 48 h. Subsequently, MTT reagent was added into each well and the cells were incubated for an additional 4 h at 37°C. Following incubation and removal of the supernatant, 150 µl DMSO was added to dissolve the dye and the absorbance was measured at 490 nm with a microplate reader. IC50 was defined as the drug concentration causing a 50% apoptosis relative to the negative control. In this experiment, the IC50 was 20 µM. The experiment was repeated three times; six parallel samples were detected each time.
Measurement of cell apoptosis and vitality
In brief, a total of 5.0×104 MCF-7 cells were seeded into 6-well plates. After 48 h, cells were treated with 20 µM cisplatin diluted in complete medium or BC-MSC-CM. Control cells were treated with medium without the addition of cisplatin. After culturing for 48 h, cells were suspended in PBS and incubated with reagents from the Annexin V & Dead Cell kit and Count & Viability kit (both from Merck KGaA, Darmstadt, Germany) according to manufacturer's protocol. Data was processed using Muse™ smart touch FACS (Merck KGaA) to generate dot plots. The values were exported to GraphPad Prism software (version 6.0; GraphPad Inc., La Jolla, CA, USA) for further analysis.
Luminex immunoassay
To investigate which component of the BC-MSC-CM decreased the level cisplatin-induced apoptosis and promote the viability of MCF-7 cells, the concentration of 6 cytokines and chemokines (IL-17A, IL-7, IL-6, VEGF, EGF, FGF) in BC-MSC-CM were quantified using a Luminex immunoassay. MILLIPLEXH human cytokine 96-well plate assays (cat. no. HCYTOMAG-60K, EMD Millipore, Billerica, MA, USA) were performed according to the manufacturer's protocol.
Antibody blocking assay
To evaluate the effect of IL-6 on cisplatin-induced apoptosis in MCF-7 cells, 10 µg/ml IL-6 neutralization antibody (R&D Systems, Inc., Minneapolis, MN, USA, MAB206.) was added to a total of 5.0×104 MCF-7 cells cultured with BC-MSC-CM. Following incubation at 37°C for 48 h, cells were collected for reverse transcription-quantitative polymerase chain reaction (RT-qPCR) and western blot analyses.
RT-qPCR
Cells were treated as aforementioned and total RNA was extracted using TRIzol reagent (Thermo Fisher Scientific, Inc.). Reverse transcription was performed using Superscript II reverse transcriptase, Oligo(dT) primer (Roche Diagnostics, Indianapolis, IN, USA) in a 40-µl reaction volume. qPCR was performed using the Veriti 96 Well Thermal Cycler (Applied Biosystems, Foster City, CA, USA). The thermocycling conditions were as follows: 94°C for 30 sec, 60°C (primer) for 30 sec and 72°C for 30 sec, with a final extension at 72°C for 10 min, performed for 35 cycles. Primers of human IL-6 and β-actin were designed using the Primer 5.0 Software (Biosoft International, Palo Alto, CA, USA) as shown: IL-6 forward, 5′-GAGGAGACTTGCCTGGTGAA-3′ and reverse, 5′-GCGCAGAATGAGATGAGTTG-3′; β-actin forward, 5′-TGGACTTCGAGCAAGAGATG-3′ and reverse, 5′-GGATGTCCACGTCACACTTC-3′. Data was quantified by the 2−ΔΔCq method (22).
Western blotting analysis
Cells were treated as aforementioned, MCF-7 cells (1.0×106) were lysed in radioimmunoprecipitation assay buffer containing 1 mM phenylmethanesulfonyl fluoride (Beyotime Institute of Biotechnology, Nanjing, China). The protein concentrations were determined using a NanoDrop 2000 Micro-volume spectrophotometer (Thermo Fisher Scientific, Inc., Wilmington, DE, USA). A total of 200 µg protein in each lane was electrophoresed using 12% SDS-PAGE, and the gels were transferred onto a polyvinylidene fluoride membrane. Subsequently, membranes were blocked with 5% skim milk at room temperature for 1 h, and incubated with primary antibodies at 4°C overnight. Then the antibody-bound membranes were washed three times, each time for 10 min. The HRP conjugated goat anti-rabbit secondary antibody (cat. no. A0208; dilution, 1:1,000; Beyotime Institute of Biotechnology) incubated with membranes at 37°C for 1 h. After washed three times, the membranes were visualized by using LuminataTM Crescendo Western HRP substrate (EMD Millipore, USA). Primary antibodies were as follows: Anti-STAT3 (cat. no. MAB1799; dilution, 1:500), anti-phosphorylated (p)-STAT3 (cat. no. MAB4607; dilution, 1:500; R&D Systems, Minneapolis, MN, USA), anti-β-actin (cat. no. sc47778; dilution, 1:1,000) and anti-B-cell lymphoma-associated X (Bax) (cat. no. sc493; dilution, 1:1,000; Santa Cruz Biotechnology, Santa Cruz, CA, USA).
Statistical analysis
All values were expressed as the mean ± standard deviation. Statistically significant differences between groups were assessed by two-way analysis of variance or Student's t-test using GraphPad Prism software (version 6.0; GraphPad, Inc.). P<0.05 was considered to indicate a statistically significant difference.
Results
Characterization and identification of BC-MSCs
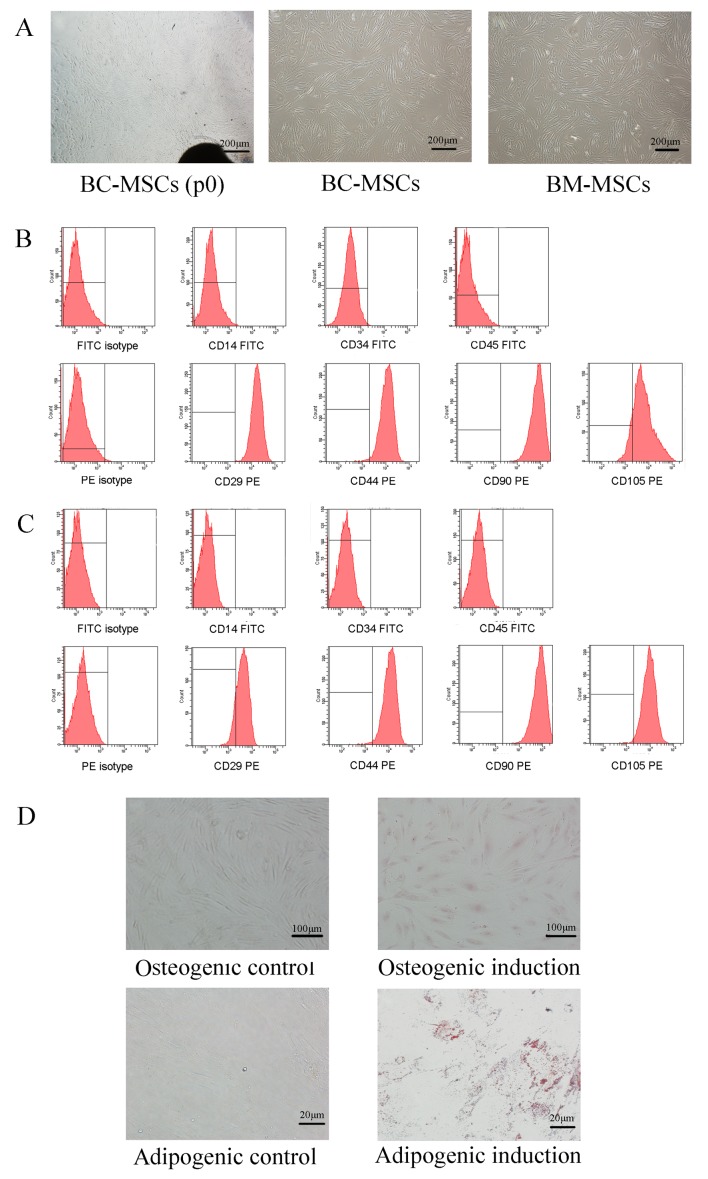
After 10–14 days of primary culture of tissue samples, a small population of fibroblastic cells was observed. When the confluence of adherent fibroblast-like cells reached ~80%, the cells were passaged into new flasks, and spindle-shaped morphologies were observed on BC-MSCs and BM-MSCs (Fig. 1A). Flow cytometric analysis demonstrated that BC-MSCs and BM-MSCs expressed high levels of CD29, CD44, CD90, and CD105, but were negative for CD14, CD34 and CD45 (Fig. 1B and C). In addition, in order to evaluate the pluripotent differentiation potential of BC-MSCs, cells were cultured in osteoblastic-induction medium for two weeks and numerous Alkaline phosphatase staining-positive cells were observed. In addition, a number of BC-MSCs were positive for Oil-Red-O intracellular staining following three weeks of incubation indicating adipogenic induction. Control cells cultured in the complete medium were negative for Oil-Red-O staining (Fig. 1D).
Figure 1.
Characterization of human breast-cancer-derived MSCs. (A) Spindle-shaped cells migrated from breast cancer tissues after 10–14 days of primary culture (BC-MSCs P0), BC-MSCs and BM-MSCs were spindle-shaped and fibroblastic in appearance of passage 3 (magnification, ×40). (B) Flow cytometric characterization of BC-MSCs. BC-MSCs were positive for CD29, CD44, CD90, CD105, but were negative for CD14, CD34 and CD45. (C) Flow cytometric characterization of BC-MSCs. BM-MSCs were positive for CD29, CD44, CD90 and CD105, but were negative for CD14, CD34 and CD45. (D) Adipogenic and osteogenic differentiation of BC-MSCs. Osteogenic differentiation of BC-MSCs was shown by alkaline phosphatase staining (magnification, ×100). Adipogenic differentiation was analyzed by Oil-Red-O staining (magnification, ×400). BC-MSC, breast-cancer-derived mesenchymal stem cell; BM-MSC, bone-marrow-derived MSC; CD, cluster of differentiation; FITC, fluorescein isothiocyanate; PE, phycoerythrin.
BC-MSCs decrease MCF-7 cell apoptosis and promote MCF-7 cell survival from cisplatin
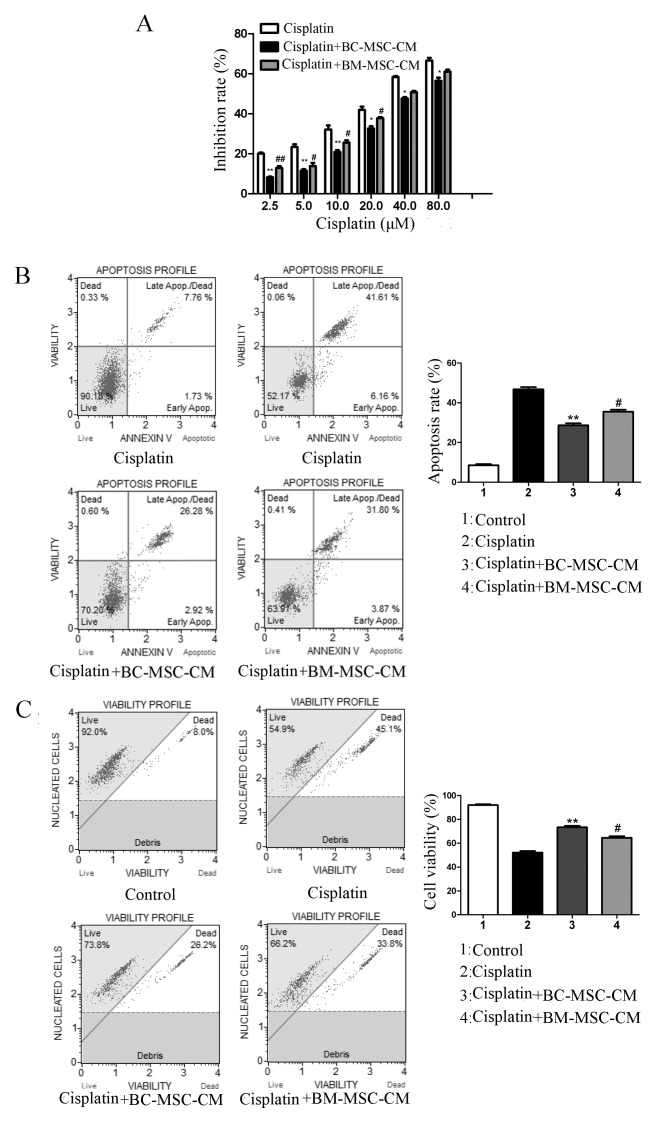
To evaluate the effects of BC-MSCs in the cisplatin-induced MCF-7 cell response, cells were seeded in 96-well plates or 6-well plates for different experiments. As presented in Fig. 2A, the results of an MTT assay revealed that MCF-7 cells exposed to cisplatin in the presence of BC-MSC-CM exhibited significantly higher growth rates compared with those treated with cisplatin alone (P<0.01) and cisplatin plus BM-MSC-CM-treated cells (P<0.05). FACS analysis revealed that the proportion of annexin-V-bound MCF-7 cells cultured with control medium increased significantly in response to cisplatin treatment compared with those cultured with BC-MSC-CM (P<0.01; Fig. 2B). The anti-apoptosis effect of BC-MSC-CM was significantly increased compared with that in the BM-MSC-CM group (P <0.05; Fig. 2B). Co-treatment with BC-MSC-CM and cisplatin revealed that the vitality of MCF-7 cells increased significantly when compared with cells treated with cisplatin alone (Fig. 2C; P<0.01). Furthermore, BC-MSCs had a significantly higher potential to decrease MCF-7 cell apoptosis and promote cell proliferation compared with BM-MSCs (P<0.05; Fig. 2). These results suggested that BC-MSCs weakened the antitumor effect of cisplatin more effectively than BM-MSCs.
Figure 2.
BC-MSCs decreases MCF-7 cell apoptosis and promotes MCF-7 cell survival from cisplatin. (A) The results of the MTT assay revealed that BC-MSCs ameliorated the inhibition of MCF-7 cell from cisplatin, increasing MCF-7 cell proliferation. (B) BC-MSCs observably decreased MCF-7 cell apoptosis. (C) BC-MSCs observably promoted MCF-7 cell survival. *P<0.05, **P<0.01, compared with the cisplatin group; #P<0.05, ##P<0.01, compared with the BC-MSC-CM group. BC-MSC, breast-cancer-derived mesenchymal stem cell; BM-MSC, bone-marrow-derived MSC.
Secretion of IL-6 was higher in BC-MSCs compared with BM-MSCs
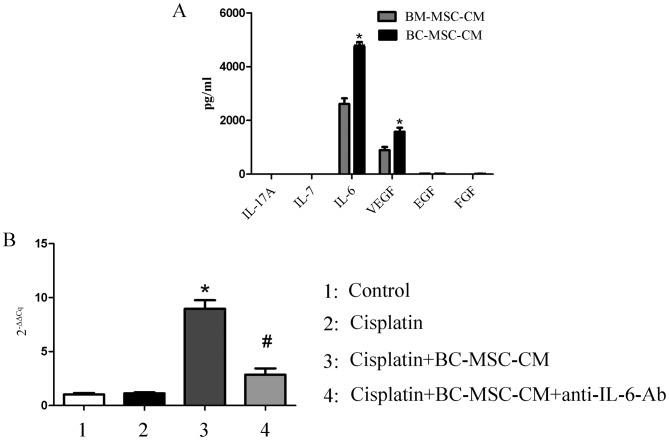
BM-MSCs exhibit marked tropism for tumor sites and have the ability to transition to cancer-associated stromal cells (23). In other words, cancer-associated stromal cells may originate from BM-MSCs. Detecting the different expression of paracrine factors between BC-MSCs and BM-MSCs may reveal which component of the BC-MSC-CM enhanced the survival of MCF-7 cells. The expression of important cytokines was therefore assessed using a Luminex immunoassay, which demonstrated that IL-6 secretion was significantly higher in the BC-MSC-CM group compared with that in the BM-MSC-CM group (P<0.01; Fig. 3A). RT-qPCR confirmed that the level of IL-6 mRNA was significantly increased in MCF-7 cells treated with cisplatin and BC-MSC-CM compared with the control and cisplatin group. IL-6 neutralizing antibody was added to cisplatin+BC-MSC-CM, IL-6 mRNA expression significantly decreased when compared with the cisplatin+BC-MSC-CM group (P<0.01; Fig. 3B). IL-6 secreted by BC-MSCs may therefore have a pivotal role in mediating the enhanced proliferation of MCF-7 cells.
Figure 3.
BC-MSC-secreted IL-6 is higher with increased mRNA expression of IL-6 in MCF-7 cells. (A) Cytokine profile analysis of BC-MSCs and BM-MSCs using a Luminex immunoassay. *P<0.01, compared with the BM-MSCs group. (B) Expression of IL-6 in MCF-7 with different treatment. *P<0.01, compared with the cisplatin group; #P<0.01, compared with the cisplatin/BC-MSC-CM group. IL-6, interleukin-6; BC-MSC-CM, breast-cancer-derived mesenchymal-stem-cell-conditioned medium; BM-MSC, bone-marrow-derived MSC; VEGF, vascular endothelial growth factor; EGF, endothelial growth factor; FGF, follicular growth factor; Ab, antibody.
Neutralizing IL-6 attenuates the action of BC-MSCs on inhibition of MCF-7 cells induced by cisplatin
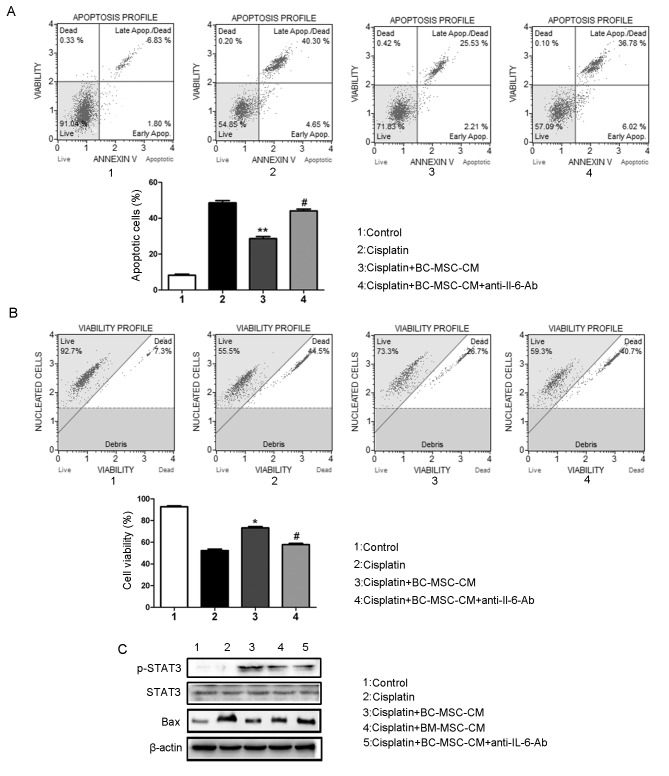
To investigate whether the effect of BC-MSCs secreted IL-6 promoted MCF-7 cell survival from cisplatin, IL-6 neutralization antibody was added to BC-MSC-CM. The resultant depletion of IL-6 recovered cisplatin-induced apoptosis (Fig. 4A). Furthermore, MCF-7 cell viability evidently decreased following IL-6 neutralization antibody treatment (Fig. 4B). To assess whether the change in response of MCF-7 cells to cisplatin treatment caused by BC-MSCs was mediated through activation of the IL-6/STAT3 signaling pathway, western blot analysis was performed (Fig. 4C). Data indicated that in the presence of BC-MSC-CM, levels of p-STAT3 were raised markedly in MCF-7 cells, which could be reversed by incubation with IL-6-neutralizing antibody. The expression of the apoptosis-associated protein Bax was reduced in MCF-7 cells on incubation with BC-MSC-CM compared with cisplatin alone treated cells. The aforementioned data indicated that IL-6 is crucial in the BC-MSC-mediated reduction in apoptosis induced by cisplatin in breast cancer cells.
Figure 4.
Neutralizing IL-6 attenuates the action of BC-MSCs on inhibition of MCF-7 cells induced by cisplatin. (A) IL-6-neutralization antibody recovered cisplatin-induced apoptosis in MCF-7 cells incubated with BC-MSC-CM. **P<0.01, compared with the cisplatin group; #P<0.05, compared with the BC-MSC-CM group. (B) IL-6 neutralization antibody decreased the MCF-7 cell vitality. *P<0.05, compared with the cisplatin group; #P<0.05, compared with the BC-MSC-CM group. (C) Western blot analysis of protein levels of Bax, STAT3, p-STAT3, IL-6 in MCF-7 cells in response to the indicated treatment. IL-6, interleukin-6; BC-MSC, breast-cancer-derived mesenchymal-stem-cell-conditioned medium; p-STAT3, phosphorylated signal transducer and activator of transcription 3; Bax, B-cell lymphoma-associated X.
Discussion
The interactions between the stromal microenvironment and tumor cells have a central role in patient survival, and the response to chemotherapy. MSCs are commonly used as stromal cells for in vitro studies on multiple tumor types, including gastric, breast and ovarian cancer (24–26). Tumor-tissue-derived MSCs have been widely studied owing to their proximity to tumors and the influence they exert on tumors (16,27–30). MSCs act as regulators of apoptosis, proliferation, angiogenesis and immune regulation, and, when in contact with tumor cells, produce a variety of cytokines that affect proliferation, survival and the acquisition of chemoresistance (31,32). The present study focused on the paracrine effects of BC-MSCs on the behavior of MCF-7 cells during cisplatin treatment. MSCs were isolated from human breast cancer tissues and revealed to exhibit a heterogeneous immunophenotype with fibroblastic morphology and the potential to differentiated into multiple cell types. First of all, BC-MSC-CM was prepared for the subsequent experiments. BC-MSC-CM significantly decreased the inhibitory effect of cisplatin treatment on MCF-7 cell growth and promoted MCF-7 cell survival. The results of flow cytometric analysis revealed that in the presence of BC-MSC-CM, the degree of cisplatin-triggered apoptosis was evidently decreased and the proportion of apoptotic cells was more evidently reduced in the presence of BC-MSC-CM compared with in the control medium. A prior study revealed that MSCs protect tumor cells exposed to chemotherapeutic drugs from apoptosis more potently than BM-MSC-CM (33).
To investigate the underlying mechanism by which BC-MSC-CM enhances the survival of the MCF-7 cells and protects them from drug-induced apoptosis, cytokine levels in BC-MSC-CM were examined by a Luminex immunoassay. The present study demonstrated that the level of IL-6 was markedly higher in the BC-MSC-CM compared with in BM-MSC-CM, suggesting that IL-6 may act as a key mediator of the tumor-promoting activity of BC-MSCs. IL-6, as a key mediator of the inflammatory response, has a pathological role in the development of several neoplasms, including malignant mesothelioma, breast tumor, endometrial cancer and lung cancer (16,34,35). It has been reported that bone-marrow- and glioma-derived MSCs enhance cancer cell proliferation via the IL-6/STAT3 signaling pathway (15,36). It is well established that the IL-6/glycoprotein 130/STAT3 signaling pathway further enhances the growth of cancer cells and reduces the sensitivity of cancer cells to antitumor drugs (37,38). The present study was designed to determine the role of BC-MSCs on cisplatin treatment of tumor cells via the IL-6/STAT3 signaling pathway. The current study demonstrated that the degree of proliferation, viability and apoptosis of MCF-7 cells in response to cisplatin treatment regulated by BC-MSCs may be attenuated by incubation with an IL-6 neutralizing antibody. Western blot analysis revealed that incubation of MCF-7 cells with BC-MSC-CM activated the IL-6/STAT3 signaling pathway in MCF-7 cells, markedly decreasing Bax expression. In addition, this effect was partially abolished in the presence of IL-6 neutralizing antibody. The data of the present study indicated that BC-MSC-secreted IL-6 attenuated the function of cisplatin on MCF-7 cells, preventing apoptosis and thus promoting breast cancer growth and survival. A more marked promotion was observed in breast cancer growth and survival when MCF-7 cells were incubated with BC-MSC-CM compared with BM-MSC-CM, suggesting that BC-MSCs have a greater potential to promote breast cancer growth and decrease apoptosis upon exposure to cisplatin than BM-MSCs.
In summary, BC-MSCs significantly enhanced the survival of MCF-7 cells that were exposed to cisplatin, one of the reasons behind the development of drug resistance. Furthermore, IL-6 was demonstrated to contribute to the BC-MSC-induced protection of MCF-7 cells from apoptosis. Therefore, BC-MSC-secreted IL-6 should be considered as a novel therapeutic target to aid the improvement of patient responses to cisplatin.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Haosen Fund Project of Youth Excellence, the First People's Hospital of Lianyungang (grant no. QN140302).
Availability of data and materials
The datasets generated/analyzed during the present study are available on reasonable request from the corresponding author.
Author's contributions
HX and YZ participated in design of the study, performed most of the experiments, analyzed the data, and wrote the paper; WL and BZ contributed to MSCs isolations. HZ, SZ and PZ characterized MSCs and contributed to the molecular analysis. HW and JY designed the experiments, analyzed the data, critically revised the paper and approved the final manuscript.
Ethics approval and consent to participate
The experiment protocols and study were approved by the Medical Ethics Committee of First People's Hospital of Lianyungang (Lianyungang, China). All study participants provided written informed consent.
Consent for publication
All participants provided informed consent for the publication of the present data.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Samavat H, Kurzer MS. Estrogen metabolism and breast cancer. Cancer Lett. 2015;356:231–243. doi: 10.1016/j.canlet.2014.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mao Y, Keller ET, Garfield DH, Shen K, Wang J. Stromal cells in tumor microenvironment and breast cancer. Cancer Metastasis Rev. 2013;32:303–315. doi: 10.1007/s10555-012-9415-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shirmanova MV, Druzhkova IN, Lukina MM, Dudenkova VV, Ignatova NI, Snopova LB, Shcheslavskiy VI, Belousov VV, Zagaynova EV. Chemotherapy with cisplatin: Insights into intracellular pH and metabolic landscape of cancer cells in vitro and in vivo. Sci Rep. 2017;7:8911. doi: 10.1038/s41598-017-09426-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yu WK, Wang Z, Fong CC, Liu D, Yip TC, Au SK, Zhu G, Yang M. Chemoresistant lung cancer stem cells display high DNA repair capability to remove cisplatin-induced DNA damage. Br J Pharmacol. 2017;174:302–313. doi: 10.1111/bph.13690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Shi S, Tan P, Yan B, Gao R, Zhao J, Wang J, Guo J, Li N, Ma Z. ER stress and autophagy are involved in the apoptosis induced by cisplatin in human lung cancer cells. Oncol Rep. 2016;35:2606–2614. doi: 10.3892/or.2016.4680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ethiraj P, Veerappan K, Samuel S, Sivapatham S. Interferon β improves the efficacy of low dose cisplatin by inhibiting NF-κB/p-Akt signaling on HeLa cells. Biomed Pharmacother. 2016;82:124–132. doi: 10.1016/j.biopha.2016.04.058. [DOI] [PubMed] [Google Scholar]
- 7.Matsumoto M, Nakajima W, Seike M, Gemma A, Tanaka N. Cisplatin-induced apoptosis in non-small-cell lung cancer cells is dependent on Bax- and Bak-induction pathway and synergistically activated by BH3-mimetic ABT-263 in p53 wild-type and mutant cells. Biochem Biophys Res Commun. 2016;473:490–496. doi: 10.1016/j.bbrc.2016.03.053. [DOI] [PubMed] [Google Scholar]
- 8.Blanc C, Deveraux QL, Krajewski S, Jänicke RU, Porter AG, Reed JC, Jaggi R, Marti A. Caspase-3 is essential for procaspase-9 processing and cisplatin-induced apoptosis of MCF-7 breast cancer cells. Cancer Res. 2000;60:4386–4390. [PubMed] [Google Scholar]
- 9.Cadamuro M, Brivio S, Spirli C, Joplin RE, Strazzabosco M, Fabris L. Autocrine and paracrine mechanisms promoting chemoresistance in cholangiocarcinoma. Int J Mol Sci. 2017;18 doi: 10.3390/ijms18010149. pii: E149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Li M, Li M, Yin T, Shi H, Wen Y, Zhang B, Chen M, Xu G, Ren K, Wei Y. Targeting of cancer-associated fibroblasts enhances the efficacy of cancer chemotherapy by regulating the tumor microenvironment. Mol Med Rep. 2016;13:2476–2484. doi: 10.3892/mmr.2016.4868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Borriello L, DeClerck YA. Tumor microenvironment and therapeutic resistance process. Med Sci (Paris) 2014;30:445–451. doi: 10.1051/medsci/20143004021. (In French) [DOI] [PubMed] [Google Scholar]
- 12.Flister MJ, Endres BT, Rudemiller N, Sarkis AB, Santarriaga S, Roy I, Lemke A, Geurts AM, Moreno C, Ran S, et al. CXM: A new tool for mapping breast cancer risk in the tumor microenvironment. Cancer Res. 2014;74:6419–6429. doi: 10.1158/0008-5472.CAN-13-3212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gould CM, Courtneidge SA. Regulation of invadopodia by the tumor microenvironment. Cell Adh Migr. 2014;8:226–235. doi: 10.4161/cam.28346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sun Z, Wang S, Zhao RC. The roles of mesenchymal stem cells in tumor inflammatory microenvironment. J Hematol Oncol. 2014;7:14. doi: 10.1186/1756-8722-7-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Scherzad A, Steber M, Gehrke T, Rak K, Froelich K, Schendzielorz P, Hagen R, Kleinsasser N, Hackenberg S. Human mesenchymal stem cells enhance cancer cell proliferation via IL-6 secretion and activation of ERK1/2. Int J Oncol. 2015;47:391–397. doi: 10.3892/ijo.2015.3009. [DOI] [PubMed] [Google Scholar]
- 16.Li W, Zhou Y, Yang J, Zhang X, Zhang H, Zhang T, Zhao S, Zheng P, Huo J, Wu H. Gastric cancer-derived mesenchymal stem cells prompt gastric cancer progression through secretion of interleukin-8. J Exp Clin Cancer Res. 2015;34:52. doi: 10.1186/s13046-015-0172-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ooi YY, Dheen ST, Tay SS. Paracrine effects of mesenchymal stem cells-conditioned medium on microglial cytokines expression and nitric oxide production. Neuroimmunomodulation. 2015;22:233–242. doi: 10.1159/000365483. [DOI] [PubMed] [Google Scholar]
- 18.El-Khattouti A, Sheehan NT, Monico J, Drummond HA, Haikel Y, Brodell RT, Megahed M, Hassan M. CD133+ melanoma subpopulation acquired resistance to caffeic acid phenethyl ester-induced apoptosis is attributed to the elevated expression of ABCB5: Significance for melanoma treatment. Cancer Lett. 2015;357:83–104. doi: 10.1016/j.canlet.2014.10.043. [DOI] [PubMed] [Google Scholar]
- 19.Velletri T, Xie N, Wang Y, Huang Y, Yang Q, Chen X, Chen Q, Shou P, Gan Y, Cao G, et al. P53 functional abnormality in mesenchymal stem cells promotes osteosarcoma development. Cell Death Dis. 2016;7:e2015. doi: 10.1038/cddis.2015.367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhou K, Xia M, Tang B, Yang D, Liu N, Tang D, Xie H, Wang X, Zhu H, Liu C, Zuo C. Isolation and comparison of mesenchymal stem cell-like cells derived from human gastric cancer tissues and corresponding ovarian metastases. Mol Med Rep. 2016;13:1788–1794. doi: 10.3892/mmr.2015.4735. [DOI] [PubMed] [Google Scholar]
- 21.Hendijani F, Javanmard ShH, Rafiee L, Sadeghi-Aliabadi H. Effect of human Wharton's jelly mesenchymal stem cell secretome on proliferation, apoptosis and drug resistance of lung cancer cells. Res Pharm Sci. 2015;10:134–142. [PMC free article] [PubMed] [Google Scholar]
- 22.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 23.Bergfeld SA, DeClerck YA. Bone marrow-derived mesenchymal stem cells and the tumor microenvironment. Cancer Metastasis Rev. 2010;29:249–261. doi: 10.1007/s10555-010-9222-7. [DOI] [PubMed] [Google Scholar]
- 24.Song B, Kim B, Choi SH, Song KY, Chung YG, Lee YS, Park G. Mesenchymal stromal cells promote tumor progression in fibrosarcoma and gastric cancer cells. Korean J Pathol. 2014;48:217–224. doi: 10.4132/KoreanJPathol.2014.48.3.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kim SH, Bang SH, Kang SY, Park KD, Eom JH, Oh IU, Yoo SH, Kim CW, Baek SY. Human amniotic membrane-derived stromal cells (hAMSC) interact depending on breast cancer cell type through secreted molecules. Tissue Cell. 2015;47:10–16. doi: 10.1016/j.tice.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 26.Castells M, Milhas D, Gandy C, Thibault B, Rafii A, Delord JP, Couderc B. Microenvironment mesenchymal cells protect ovarian cancer cell lines from apoptosis by inhibiting XIAP inactivation. Cell Death Dis. 2013;4:e887. doi: 10.1038/cddis.2013.384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chang WW, Hu FW, Yu CC, Wang HH, Feng HP, Lan C, Tsai LL, Chang YC. Quercetin in elimination of tumor initiating stem-like and mesenchymal transformation property in head and neck cancer. Head Neck. 2013;35:413–419. doi: 10.1002/hed.22982. [DOI] [PubMed] [Google Scholar]
- 28.López J, Poitevin A, Mendoza-Martínez V, Pérez-Plasencia C, García-Carrancá A. Cancer-initiating cells derived from established cervical cell lines exhibit stem-cell markers and increased radioresistance. BMC Cancer. 2012;12:48. doi: 10.1186/1471-2407-12-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kansy BA, Dißmann PA, Hemeda H, Bruderek K, Westerkamp AM, Jagalski V, Schuler P, Kansy K, Lang S, Dumitru CA, Brandau S. The bidirectional tumor-mesenchymal stromal cell interaction promotes the progression of head and neck cancer. Stem Cell Res Ther. 2014;5:95. doi: 10.1186/scrt484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Behnan J, Isakson P, Joel M, Cilio C, Langmoen IA, Vik-Mo EO, Badn W. Recruited brain tumor-derived mesenchymal stem cells contribute to brain tumor progression. Stem Cells. 2014;32:1110–1123. doi: 10.1002/stem.1614. [DOI] [PubMed] [Google Scholar]
- 31.Ji R, Zhang B, Zhang X, Xue J, Yuan X, Yan Y, Wang M, Zhu W, Qian H, Xu W. Exosomes derived from human mesenchymal stem cells confer drug resistance in gastric cancer. Cell Cycle. 2015;14:2473–2483. doi: 10.1080/15384101.2015.1005530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lou G, Song X, Yang F, Wu S, Wang J, Chen Z, Liu Y. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J Hematol Oncol. 2015;8:122. doi: 10.1186/s13045-015-0220-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kurtova AV, Balakrishnan K, Chen R, Ding W, Schnabl S, Quiroga MP, Sivina M, Wierda WG, Estrov Z, Keating MJ, et al. Diverse marrow stromal cells protect CLL cells from spontaneous and drug-induced apoptosis: Development of a reliable and reproducible system to assess stromal cell adhesion-mediated drug resistance. Blood. 2009;114:4441–4450. doi: 10.1182/blood-2009-07-233718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.van der Zee M, Sacchetti A, Cansoy M, Joosten R, Teeuwssen M, Heijmans-Antonissen C, Ewing-Graham PC, Burger CW, Blok LJ, Fodde R. IL6/JAK1/STAT3 signaling blockade in endometrial cancer affects the ALDHhi/CD126+ stem-like component and reduces tumor burden. Cancer Res. 2015;75:3608–3622. doi: 10.1158/0008-5472.CAN-14-2498. [DOI] [PubMed] [Google Scholar]
- 35.Hossain A, Gumin J, Gao F, Figueroa J, Shinojima N, Takezaki T, Priebe W, Villarreal D, Kang SG, Joyce C, et al. Mesenchymal stem cells isolated from human gliomas increase proliferation and maintain stemness of glioma stem cells through the IL-6/gp130/STAT3 pathway. Stem Cells. 2015;33:2400–2415. doi: 10.1002/stem.2053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mochizuki D, Adams A, Warner KA, Zhang Z, Pearson AT, Misawa K, McLean SA, Wolf GT, Nör JE. Anti-tumor effect of inhibition of IL-6 signaling in mucoepidermoid carcinoma. Oncotarget. 2015;6:22822–22835. doi: 10.18632/oncotarget.4477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rodriguez-Barrueco R, Yu J, Saucedo-Cuevas LP, Olivan M, Llobet-Navas D, Putcha P, Castro V, Murga-Penas EM, Collaz-Lorduy A, Castillo-Martin M, et al. Inhibition of the autocrine IL-6-JAK2-STAT3-calprotectin axis as targeted therapy for HR-/HER2+ breast cancers. Genes Dev. 2015;29:1631–1648. doi: 10.1101/gad.262642.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ren T, Shan J, Qing Y, Qian C, Li Q, Lu G, Li M, Li C, Peng Y, Luo H, et al. Sequential treatment with AT-101 enhances cisplatin chemosensitivity in human non-small cell lung cancer cells through inhibition of apurinic/apyrimidinic endonuclease 1-activated IL-6/STAT3 signaling pathway. Drug Des Devel Ther. 2014;8:2517–2529. doi: 10.2147/DDDT.S71432. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated/analyzed during the present study are available on reasonable request from the corresponding author.