Abstract
Background:
One of the most significant problems in the treatment of leukemia is the expansion of resistance to chemotherapeutic agents. Therefore, assessing the drug resistance and especially the drug resistance genes of leukemic cells is important in any treatment. The impact of Mesenchymal Stem Cells (MSCs) and hypoxic condition have been observed in the biological performance of majority of leukemic cells.
Methods:
MOLT-4 cells were co-cultured with MSCs in the hypoxic condition induced by Cobalt Chloride (CoCl2) for 6 and 24 hr. Then, apoptosis of cells was analyzed using annexin-V/PI staining and expression of the drug resistance genes including MDR1, MRP, and BCRP along with apoptotic and anti-apoptotic genes, including BAX and BCL2, was evaluated by real-time PCR.
Results:
The hypoxic condition for MOLT-4 cells co-cultured with MSCs could significantly increase the expression of MDR1 and BCRP genes (p<0.05) which are involved in drug resistance. Also, the results indicated that this condition significantly increases the expression of BCL2 (p<0.05) and reduces the apoptosis in MOLT-4 cells co-cultured with MSCs in the hypoxic condition.
Conclusion:
These effects can demonstrate the important role of hypoxia and MSCs on the biological behavior of Acute Lymphoblastic Leukemia (ALL) cells that may lead to particular treatment outcomes.
Keywords: Acute lymphoblastic leukemia, Drug resistance, Hypoxia, Mesenchymal stem cell
Introduction
Childhood malignancies are serious problems that can cause inexpiable damages to families and societies. About 35% of childhood malignancies are childhood leukemia and Acute Lymphoblastic Leukemia (ALL) involves 80–85% of them1. Mesenchymal Stem Cells (MSCs) are multipotent stromal cells that can differentiate into a variety of cells and have a critical role in hematopoiesis regulation and successful hematopoietic stem cell transplantation. There is some evidence that the MSCs have a supporting effect on tumor stroma through various activities such as producing growth factors, amplifying tumor vessel formation and forming niches for tumor stem cells especially in solid tumors like gastric and breast cancer. Thus, it shows that MSCs of tumor microenvironment can promote tumor growth in several ways2. Besides, it has always been debatable that how the MSCs influence the leukemic cells. In some studies, apoptosis induction was supposed to be an answer to this question3. Several studies have shown a proliferation-suppressing effect of MSCs on lymphoid lineage by secreting multiple cytokines that arrest these cells at G0 and G1 phases of cell cycle4. Therefore, more investigations are needed to clarify the MSCs effects in lymphoid leukemia.
Hypoxia-Inducible Factor-1 (HIF-1), a heterodimeric transcription factor, is an essential part of the cellular response to the hypoxic condition5. This element was swiftly destroyed under normoxic condition via proteasomal degradation, but in the hypoxic situation, its degradation is slow, which eventually leads to increased expression of its target genes. Cobalt chloride (CoCl2) has the potential of blocking the HIF-1 degradation and hence it can mimic the hypoxic situation6. Based on many reliable reports in several different kinds of cells, CoCl2 can induce apoptosis7–10.
It is a big challenge to prevent leukemic cells recurrence in the bone marrow to overcome treatment failure in ALL. Bone marrow environment is heterogeneous and has a hypoxic compartment11–13. As a result, it can inhibit the hydroxylation of HIF-1 and give rise to activation of this transcription factor.
HIF-1 target genes are involved in erythropoiesis, angiogenesis, and energy metabolism. In a hypoxic environment, these genes have a lot of critical functions in controlling cellular homeostasis, which makes these genes involved in radio- and chemotherapy resistance of tumors, resulting in tumor survival and more aggressive phenotypes14–16. Several other genes, as mitotic kinase family (Aurora A, B, and C), which are associated with tumor enhancement and cancer progression, are also influenced by hypoxia. Therefore, these genes are new targets for treatment strategies17–19.
Although several different mechanisms have been found, one of the most dangerous problems in cancer patients is the drug resistance that frequently occurs and has a bad prognosis20–22. Some of these mechanisms are the prevention of the drug from entering in to the cell, pumping the drug out of the cell, enzymatic inactivation of drug, mutation or alteration of drug targets, apoptosis or senescence defectiveness, and deficient repair mechanisms. Drug resistance is the primary cause of unsuccessful treatment in patients with acute leukemia23. Multi Drug Resistance (MDR) is a well-known protein that is one of the primary pumps which can actively excrete drugs from the cell24. MDR-Associated Protein (MRP) acts like MDR toward excreting drugs out of the cell and when it is overexpressed in acute leukemia, tumoral cells show resistance against several classes of drugs such as anthracyclines, etoposide, and vinca alkaloids25. In patients for whom MRP was low, overall survival was better than patients with moderate or higher MRP expression26. Another protein that is reported to be involved in MDR phenotype is a newly characterized one and is called Breast Cancer Resistance Protein (BCRP, ABCG2) that is a member of ABC transporter family and seems to develop resistance against commonly used AML drugs27.
In order to have a better understanding of how the hypoxia and MSCs affect leukemia cells, the effects of the hypoxia and MSCs on MOLT-4 cells (ALL cell line) were studied. The proliferation rate, apoptosis, cytotoxicity and expression profiles of the genes involved in the drug resistance process that have very important functions in leukemic cells’ biological behavior were investigated.
Materials and Methods
Cell culture
The MOLT-4 cell line was purchased from Pasteur Institute Cell Bank (Tehran, Iran) and was cultured in RPMI 1640 (Gibco Laboratories, Grand Island, NY). Bone marrow MSCs (positive for CD44, CD90, STRO-1 and Negative for CD14, CD19, CD146) were purchased from Royesh Stem Cell Biotechnology Institute Cell Bank (Tehran, Iran) and were cultured in DMEM (Dulbecco’s Modified Eagle Medium) (Gibco Laboratories). Both media contained 10% fetal bovine serum (FBS) (Gibco Laboratories) and were incubated at 37°C humidified atmosphere in 5% CO2 incubator.
Determining the dose of CoCl2 cytotoxicity
HIF1 induction via CoCl2 is dose-dependent. To reach the maximum levels of HIF1 without significant cell death, the CoCl2 induced cytotoxicity was assessed via cell counting by Trypan blue. To evaluate the CoCl2 cytotoxicity on MOLT-4 cells, different concentrations of cobalt (0, 25, 50, 100, 150, and 200 μM CoCl2) were employed.
Co-culture of MOLT-4 cells with MSCs
Second passage MSCs were seeded in plates containing DMEM at a density of 5×104 cells/cm2. The medium was changed every three days, till the MSCs feeder layer reached confluence. Subsequently, MOLT-4 cells in 2×106 number were added into supernatant and incubated for 6 and 24 hr.
Hypoxic treatment
2×106 MOLT-4 cells, with or without MSCs, were treated with 100 μM CoCl2 in 5% CO2 incubator at 37°C for 6 and 24 hr.
RNA extraction and cDNA synthesis
After removing the medium from plates, MSCs were adherent and MOLT-4 cells were suspended and in the medium just containing MOLT-4 cells, total RNA from leukemic cells was extracted using RNX-Plus solution kit (Sinaclon, Tehran, Iran). After cell lysis (first step based on manufacturer’s instruction), the products were stored at −70°C and thawed when RNA extraction was needed. High capacity kit (Bioneer, Alameda, CA) was used to produce single-stranded cDNA from the extracted RNA.
Real-Time Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
The SYBR1 Green PCR Master Mix (Takara, Clontech, Japan) was used to determine the mRNA levels of BAX, BCL-2, MDR-1, and BCRP genes. The analysis of melting curves was performed using real-time PCR system (Rotor Gene 6000, Corbett Life Science). The supplemental table 1 shows the primers used for BAX, BCL-2, MDR-1, MRP, BCRP, β-actin and GAPDH genes. β-actin and GAPDH were used as an internal control, and duplicate analysis was performed for all samples. The list of the primers is given in table 1.
Table 1.
Summary of primer sequences. All primer sequences are presented in 5′ to 3′ orientation
| Gene | Sequences (5′-3′) | Amplicon length (bp) | Annealing Temp (°C) |
|---|---|---|---|
| MDR1 | F: ATTGCTCACCGCCTGTCCACC R: TGCTGATGCGTGCCATGCTCC |
90 | 57 |
| MRP | F: CGTGTTGGTCTCTGTGTTCCTG R: AGAAAGATGCTCTCTGGGTTTG |
181 | 57 |
| BCRP | F: GGAATCTTGGCTGAGGGTTTGG R: GATGATTCTGACGCACACCTG |
169 | 59 |
| Bax | F: TGCCAGCAAACTGGTGCTCA R: GCACTCCCGCCACAAAGATG |
194 | 59 |
| Bcl2 | F: CCTGTCGATGACTGAGTACC R: GAGACAGCCAGGAGAAATCA |
128 | 55 |
| βactin | F: AGAGCTACGAGCTGCCTGAC R: AGCACTGTGTTGGCGTACAG |
184 | 57 |
| GAPDH | F: GGCGTGAACCACGAGAAGTATAA R: CCCTCCACGATGCCAAAGT |
119 | 59 |
Apoptosis analysis using annexin VFITC/PI
Cells undergoing apoptosis were identified by annexin V and propidium iodide staining (BD Pharmingen, USA) according to the manufacturer’s instructions. Briefly, the cells were washed twice with cold PBS and then were resuspended in 1X binding buffer at a concentration of 1×106 cells/ml. Then, 100 μl of the solution (1×105 cells) was transferred to a 5 ml culture tube. 5 μl of annexin V-FITC and 5 μl of PI were also added. Then, the cells were vortexed gently and incubated for 15 min at RT (25°C) in the dark. Finally, 400 μl of 1×binding buffer was added to each tube and they were analyzed using FACSCalibur flow cytometer (Becton-Dickenson, Mountain View, CA, USA) and FlowJo software.
Statistical analysis
Our results were statistically analyzed by The SPSS v.19. Data statement was as means±SD. One-Way ANOVA was used to assess the observed statistical differences. The GraphPad Prism v.6 was employed for regression analysis of correlation and the response linearity (GraphPad Software Inc). Statistically significant data were considered for p<0.05.
Results
Cell toxicity assay of CoCl2 treated cells
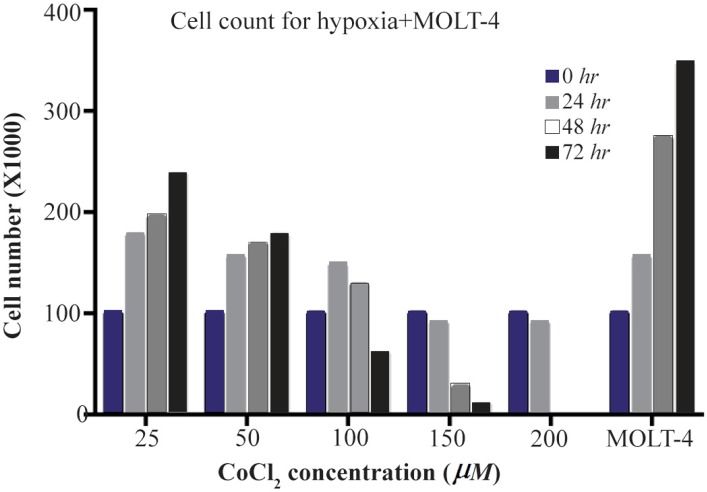
According to our results, with less than 100 μM doses of CoCl2, cell growth was observed at 48 and 72 hr, and at the dose of more than 100 μM, CoCl2 was fatal for cells in each of the designated times. So, the optimum dosage was fixed at 100 μM concentration of CoCl2 within 24 hr (Figure 1).
Figure 1.
The MOLT-4 cells exposed to various doses of CoCl2 (0, 25, 50, 100, 150, 200 μM) during 0, 24, 48 and 72 hr time courses. In these periods, to detect the toxic dose of CoCl2, cells were counted using trypan blue in 1:1 ratio.
Growth curve analysis of MOLT-4 cells co-cultured with MSC under the hypoxic condition
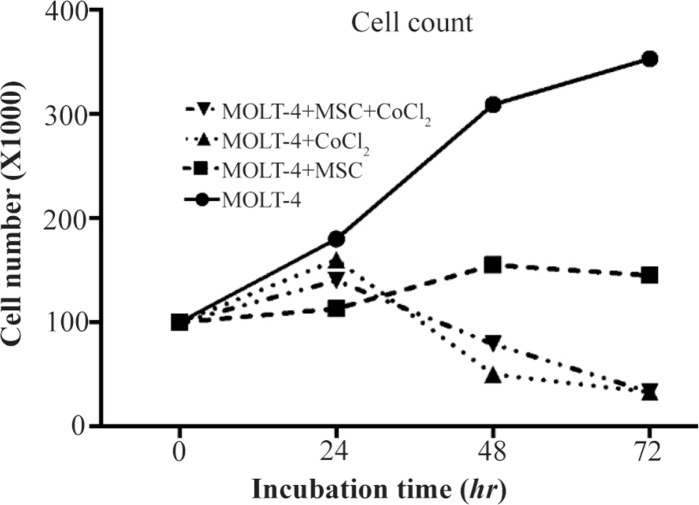
Cobalt exposed (100 μM, 24, 48, and 72 hr) and untreated MOLT-4 cells were cultured in mutual numbers on 6 cm cell culture plates. After 24, 48, and 72 hr, the cells of each plate were counted using trypan blue. The MOLT-4 cells were cultured under different conditions (with MSCs, with CoCl2, with MSCs and CoCl2) and were counted by trypan blue in 1:1 ratio at 24, 48, and 72 hr. Our findings revealed that MOLT-4 cells with MSC and CoCl2 alone, and MSC along with CoCl2 had inhibitory effects on growth of them in 24 hr (Figure 2).
Figure 2.
MOLT-4 cells cultured under different conditions (with MSC, with CoCl2, with MSC and CoCl2) counted by trypan blue at 0, 24, 48, 72 hr.
Comparison of BAX and BCL2 gene expression levels in MOLT-4 cells co-cultured with MSC under hypoxic condition
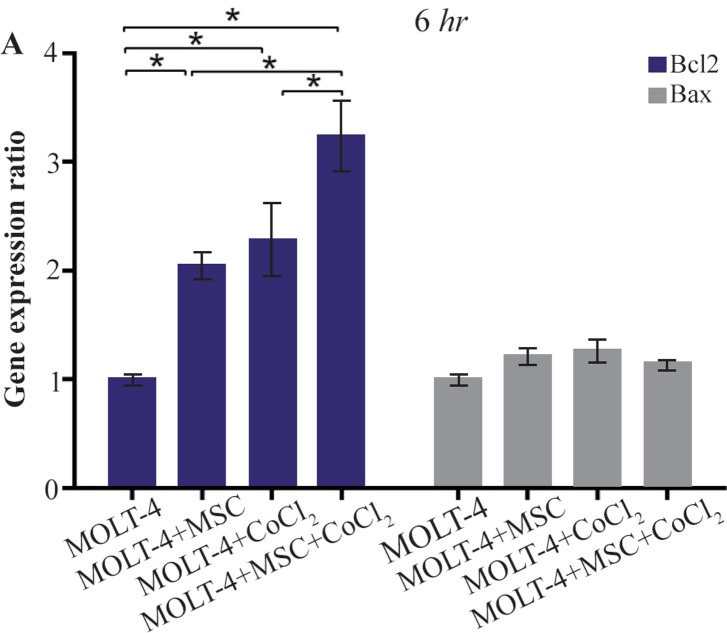
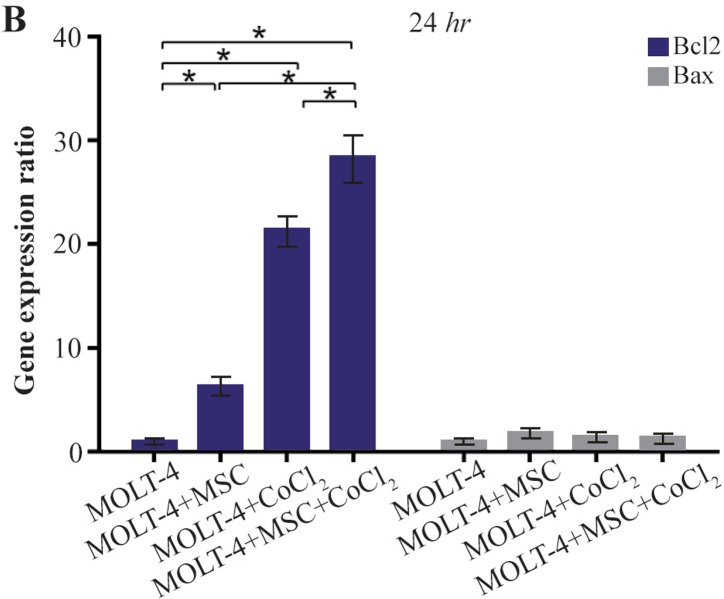
BAX and BCL2 were evaluated to determine apoptotic and anti-apoptotic changes in different conditions (MOLT-4+MSC, MOLT-4+CoCl2 and MOLT-4+MSC+CoCl2). BCL2 expression was increased in the presence of MOLT-4+MSC and MOLT-4+CoCl2 and was the highest in MOLT+MSCs+CoCl2 (p<0.05). However, BAX mRNA expression level did not differ significantly between different groups (Figures 3A and 3B).
Figure 3A.
Real Time-PCR data for BAX and BCL2 expression in MOLT-4 cells under CoCl2 and hypoxia with and without MSC. (A) BAX and BCL2 expression levels were analyzed by Real Time-PCR in MOLT-4 cells under CoCl2 (100 μM) with MSC. RNA was extracted at 6 hr following 100 μM CoCl2 exposure. Data is presented as means±SD of three independent experiments. * Statistically significant difference compared to the respective data of control (untreated cells), p<0.05.
Figure 3B.
Real Time-PCR data for BAX and BCL2 expression in MOLT-4 cells under CoCl2 and hypoxia with and without MSC. (B) BAX and BCL2 expression levels were analyzed by Real Time-PCR in MOLT-4 cells co-cultured with MSC. RNA was extracted at 24 hr following 100 μM CoCl2 exposure. Data is presented as means±SD of three independent experiments. * Statistically significant difference compared to the respective data of control (untreated cells), p<0.05.
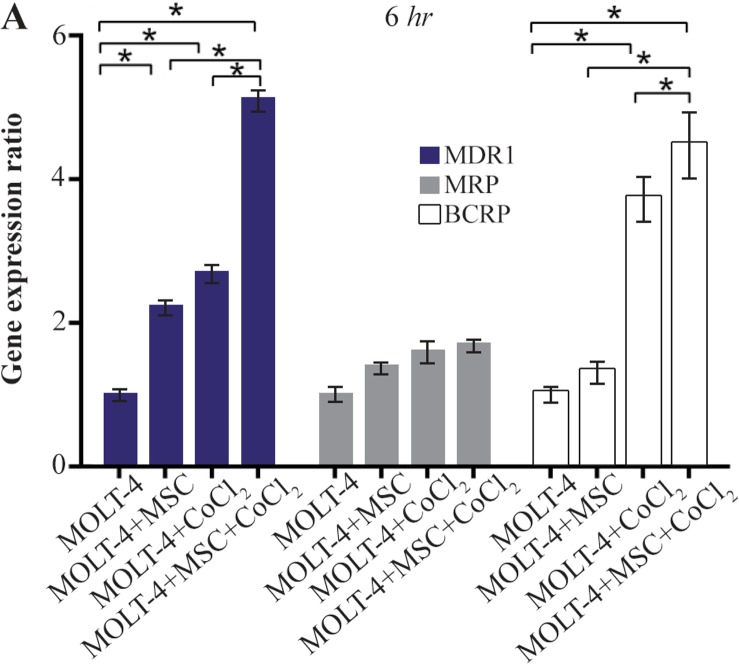
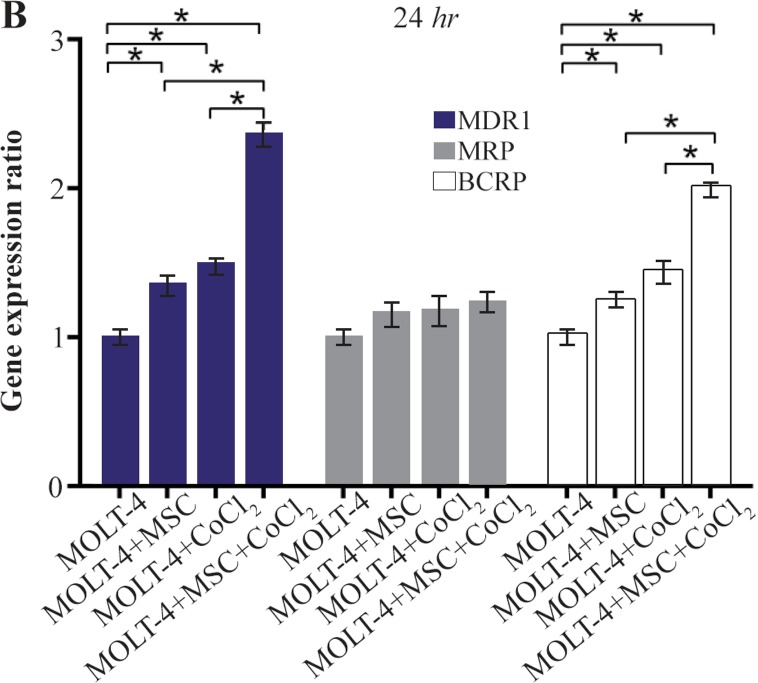
Comparing the multiple drug resistance genes expression levels in MOLT-4 cells co-cultured with MSC under the hypoxic condition
MDR1, MRP, and BCRP were evaluated to determine the expression level changes of drug resistance genes in different conditions (MOLT-4+MSC, MOLT-4+CoCl2, and MOLT-4+MSC+CoCl2). MDR1 expression was increased in the presence of MOLT-4+MSC and MOLT-4+CoCl2 and was the highest in MOLT-4+MSC+CoCl2 (p<0.05).
BCRP expression was increased in the presence of MOLT-4+MSC and MOLT-4+CoCl2 and was the highest in MOLT-4+MSC+CoCl2 (p<0.05). However, MRP mRNA expression level did not differ significantly between different groups (Figures 4A and 4B).
Figure 4A.
Real Time-PCR data for MOLT-4 cells MDR1, MRP, and BCRP genes expression under CoCl2 and hypoxia with and without MSC in different time courses. (A) MDR1, MRP and BCRP genes expression levels were analyzed by Real Time-PCR in MOLT-4 cells under CoCl2 (100 μM) with MSC. RNA was extracted at 6 hr following 100 μM CoCl2 exposure. Data is presented as means±SD of three independent experiments. * Statistically significant difference compared to the respective data of control (untreated cells), p<0.05.
Figure 4B.
Real Time-PCR data for MOLT-4 cells MDR1, MRP, and BCRP genes expression under CoCl2 and hypoxia with and without MSC in different time courses. (B) MDR1, MRP and BCRP genes expression levels were analyzed by Real Time-PCR in MOLT-4 cells cocultured with MSC. RNA was extracted at 24 hr following 100 μM CoCl2 exposure. Data is presented as means±SD of three independent experiments. * Statistically significant difference compared to the respective data of control (untreated cells), p<0.05.
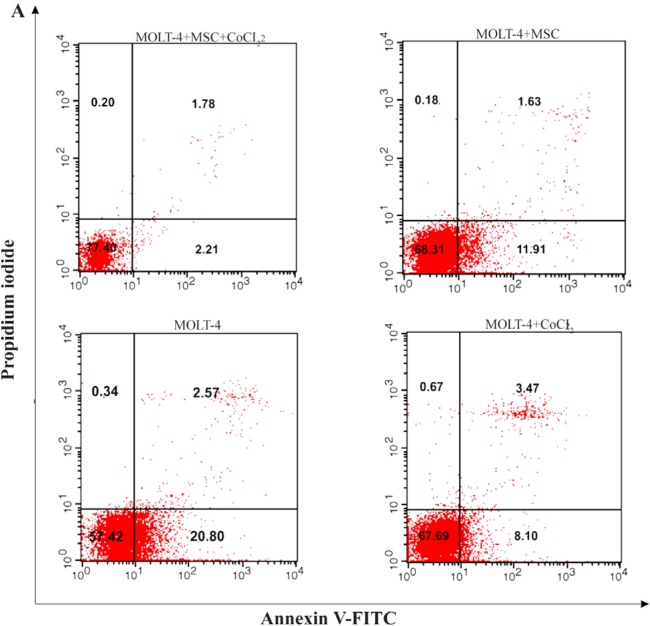
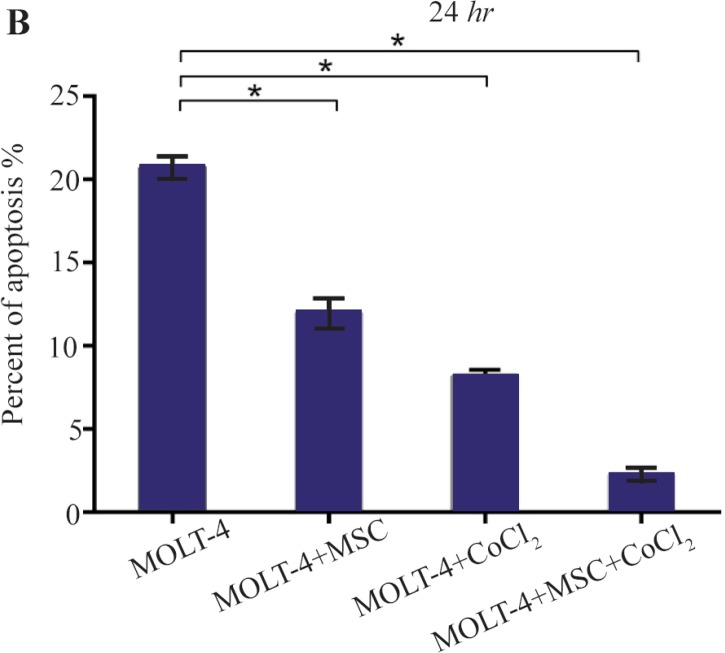
Hypoxia-induced apoptosis of MOLT-4 cells co-cultured with MSC
Hypoxia-induced apoptosis of MOLT-4 cells co-cultured with MSC was assessed using annexin V/PI staining. Treatment of MOLT-4 cells co-cultured with MSC with 100 μM CoCl2 resulted in a statistically significant increase in the number of apoptotic cells after 24 hr. After 24 hr of CoCl2 treatment, apoptotic cells constituted about 20.80±0.66, 11.91±0.95, 8.10±0.44, and 2.21±0.42% of the totally measured cell population, that is MOLT-4, MOLT-4+MSC, MOLT-4+CoCl2 and MOLT-4+MSC+CoCl2, respectively (Figures 5A and 5B).
Figure 5A.
Recognition of apoptosis in MOLT-4 cells co-cultured with MSC treated with CoCl2. Figure 5A depicts a representable sample of assessment of apoptosis using Annexing-V staining and PI. (A) Change of phosphatidylserine externalization induced by CoCl2 in MOLT-4 cells cocultured with MSC (flow cytometry analysis, Annexin V-FITC/PI staining). MOLT-4 cells were incubated with 100 μM CoCl2 for 24 hr. The data presents the three independent experiments. Annexin V positive/PI negative cells (Annexin V+/PI−) showed in the bottom right quadrant of each dot plot signifies cells corresponding to early apoptosis, whereas the Annexin V positive/PI positive (Annexin V+/PI+) cells showed in the upper right quadrant signifies cells corresponding to late apoptotic/necrotic cells.
Figure 5B.
Recognition of apoptosis in MOLT-4 cells co-cultured with MSC treated with CoCl2. (B) Apoptotic cells Percentage (demonstrating phosphatidylserine externalization) induced by CoCl2 (flow cytometry analysis, Annexin V-FITC/PI staining). MOLT-4 cells co-cultured with MSC were incubated with 100 μM CoCl2 for 24 hr. Data is presented as means±SD of three independent experiments. * Statistically significant difference compared to the respective data of control (untreated cells), p<0.05.
Discussion
Today, the hypoxia and drug resistance are considered to play roles in the progression and growth of tumor. There are clear shreds of evidence about the positive effect of HIF1 on the proliferation of leukemic cells, and it is one of the key regulators of the cell response to hypoxia28,29. It seems that hypoxia is a positive regulator of gene expression than a negative one and HIF1 might play a fundamental role in the regulation of the genes related to the hypoxia30. In the field of studies on ALL cells, it has been suggested that mesenchymal cells in co-culture with these cells led to some changes in their signaling process. Some studies have demonstrated that MSCs have inhibitory effects on proliferation of ALL cells in the co-culture experiments1,31. Others have reported that BCL2 gene which was significantly upregulated may play a role in the mechanism of the drug resistance induced by MSCs in the leukemic cells. Our results also showed that in co-culture of MOLT-4 cells, the BCL2 gene expression was increased, whereas BAX gene expression did not show significant changes. Our observations also confirmed previous studies and revealed the inhibitory effect of MSCs on MOLT-4 cell growth1,32. Besides, the impact of hypoxia was examined on MOLT-4 cells co-cultured with MSCs. Our results showed that the hypoxia induced by 100 μM CoCl2 significantly increased the expression of BCL2 gene, whereas the BAX gene expression displayed no significant alteration. Our outcomes presented that hypoxia, as well as co-culture with MSCs, leads to an increased expression of BCL2 gene in MOLT-4 cells. Unsurprisingly, the co-culture of this cancer cell line accompanied by hypoxia had the greatest increase in the BCL2 gene expression.
Several studies have been done on ALL lineage cells co-cultured with MSCs to explore the changes in the expression of drug resistance genes, which represents the influence of co-culture as mentioned above on the expression pattern of drug resistance genes33–35. In this study, an attempt was made to evaluate the changes in the gene expression pattern of the drug resistance genes including MDR1, MRP, and BCRP in MOLT-4 cells co-cultured with MSCs under the hypoxic condition. It is important that the MDR1 and MRP genes are targets for transcription factor HIF136. Our findings revealed that MDR1 and BCRP genes expression levels significantly increased when MOLT-4 cells were co-cultured with MSC in the hypoxic condition, whereas the MRP gene expression did not change significantly in none of the studied situations. It has been demonstrated that the hypoxia increases the expression of anti-apoptotic protein BCL-2, which can result in the drug resistance of leukemic cells and treatment failure37. MSCs have a potential to retard the cell cycle, prevent the proliferation and reduce the apoptosis in leukemic cells. So, MSCs shield these cells against unfavorable conditions and eventually maintain their viability. The data obtained from flow cytometry studies revealed a decline in MOLT-4 cells apoptosis affected by MSCs and hypoxia. Considering the increase of the BCL2 gene expression during the MOLT-4 cells co-cultured with MSCs under the hypoxic condition, the apoptosis lowering was expectable.
A limitation of the present study was using CoCl2 as a hypoxia mimetic agent, that increases HIF1 expression and its DNA binding activity, instead of a low oxygen environment38. Obviously, it doesn’t induce true hypoxia due to the toxic influence of CoCl2 on the cells and it is expectable to get the conflicting results compared with the real hypoxic condition that can cause numerous various changes in the cell biology.
Of course, because of the different study designs, cell line class, oxygen tension, and hypoxia duration, there are disagreements in determining whether CoCl2 acts as a supportive or suppressive agent for leukemic cells development or not38–42.
Conclusion
Our results showed that the hypoxia induced by CoCl2 has an inhibitory effect on proliferation of ALL lineage MOLT-4 cells co-cultured with MSCs that can be a result of CoCl2 toxicity rather than the effect of hypoxia. Also, hypoxia and MSCs can contribute to increased expression of drug resistance genes and less apoptosis in ALL cells. Hence, the induction of anti-apoptotic proteins expression impressed by hypoxia and MSCs can be involved in the leukemic cells’ drug resistance phenomenon and subsequently in the treatment failure. Further studies are required to demonstrate the role of hypoxia and MSCs on the biological behavior of ALL cells, which may lead to particular treatment outcomes.
Acknowledgement
This work was financially supported by the grant of Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran.
Footnotes
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
References
- 1.Bozok Cetintas V, Aktug H, Oltulu F, Keskinoglu A, Erer Del Castello B, Taskiran D. The effects of mesenchymal stem cells on lymphoblastic leukemia cell proliferation. J BUON 2014;19(4):1006–1017. [PubMed] [Google Scholar]
- 2.Roorda BD, ter Elst A, Kamps WA, de Bont ES. Bone marrow-derived cells and tumor growth: contribution of bone marrow-derived cells to tumor micro-environments with special focus on mesenchymal stem cells. Crit Rev Oncol Hematol 2009;69(3):187–198. [DOI] [PubMed] [Google Scholar]
- 3.Tabe Y, Jin L, Tsutsumi-Ishii Y, Xu Y, McQueen T, Priebe W, et al. Activation of integrin-linked kinase is a critical prosurvival pathway induced in leukemic cells by bone marrow-derived stromal cells. Cancer Res 2007;67 (2):684–694. [DOI] [PubMed] [Google Scholar]
- 4.Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, et al. Human mesenchymal stem cells modulate B-cell functions. Blood 2006;107(1):367–372. [DOI] [PubMed] [Google Scholar]
- 5.Semenza GL. Hypoxia-inducible factor 1: master regulator of O2 homeostasis. Curr Opin Genet Dev 1998;8 (5):588–594. [DOI] [PubMed] [Google Scholar]
- 6.An WG, Kanekal M, Simon MC, Maltepe E, Blagosklonny MV, Neckers LM. Stabilization of wild-type p53 by hypoxia-inducible factor 1alpha. Nature 1998;392(6674): 405–408. [DOI] [PubMed] [Google Scholar]
- 7.Lee M, Kang H, Jang SW. CoCl2 induces PC12 cells apoptosis through p53 stability and regulating UNC5B. Brain Res Bull 2013;96:19–27. [DOI] [PubMed] [Google Scholar]
- 8.Yang Z, Yang C, Xiao L, Liao X, Lan A, Wang X, et al. Novel insights into the role of HSP90 in cytoprotection of H2S against chemical hypoxia-induced injury in H9c2 cardiac myocytes. Int J Mol Med 2011;28(3):397–403. [DOI] [PubMed] [Google Scholar]
- 9.Ardyanto TD, Osaki M, Tokuyasu N, Nagahama Y, Ito H. CoCl2-induced HIF-1alpha expression correlates with proliferation and apoptosis in MKN-1 cells: a possible role for the PI3K/Akt pathway. Int J Oncol 2006;29(3): 549–555. [PubMed] [Google Scholar]
- 10.Zou W, Yan M, Xu W, Huo H, Sun L, Zheng Z, et al. Cobalt chloride induces PC12 cells apoptosis through reactive oxygen species and accompanied by AP-1 activation. J Neurosci Res 2001;64(6):646–653. [DOI] [PubMed] [Google Scholar]
- 11.Evans WE, Relling MV. Moving towards individualized medicine with pharmacogenomics. Nature 2004;429 (6990):464–468. [DOI] [PubMed] [Google Scholar]
- 12.Flotho C, Coustan-Smith E, Pei D, Cheng C, Song G, Pui CH, et al. A set of genes that regulate cell proliferation predicts treatment outcome in childhood acute lymphoblastic leukemia. Blood 2007;110(4):1271–1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Scortegagna M, Morris MA, Oktay Y, Bennett M, Garcia JA. The HIF family member EPAS1/HIF-2α is required for normal hematopoiesis in mice. Blood 2003;102(5): 1634–1640. [DOI] [PubMed] [Google Scholar]
- 14.Winter SC, Shah KA, Han C, Campo L, Turley H, Leek R, et al. The relation between hypoxia-inducible factor (HIF)-1 α and HIF-2α expression with anemia and outcome in surgically treated head and neck cancer. Cancer 2006;107(4):757–766. [DOI] [PubMed] [Google Scholar]
- 15.Wu X, Qian C, Yuan K. Correlations of hypoxia-inducible factor-1α/hypoxia-inducible factor-2α expression with angiogenesis factors expression and prognosis in non-small cell lung cancer. Chin Med J 2011;124(1):11–18. [PubMed] [Google Scholar]
- 16.Noguera R, Fredlund E, Piqueras M, Pietras A, Beckman S, Navarro S, et al. HIF-1alpha and HIF-2alpha are differentially regulated in vivo in neuroblastoma: high HIF-1alpha correlates negatively to advanced clinical stage and tumor vascularization. Clin Cancer Res 2009;15(23): 7130–7136. [DOI] [PubMed] [Google Scholar]
- 17.Klein A, Flügel D, Kietzmann T. Transcriptional regulation of serine/threonine kinase-15 (STK15) expression by hypoxia and HIF-1. Mol Biol Cell 2008;19(9):3667–3675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cammareri P, Scopelliti A, Todaro M, Eterno V, Francescangeli F, Moyer MP, et al. Aurora-a is essential for the tumorigenic capacity and chemoresistance of colorectal cancer stem cells. Cancer Res 2010;70(11):4655–4665. [DOI] [PubMed] [Google Scholar]
- 19.Failes TW, Mitic G, Abdel-Halim H, Po’uha ST, Liu M, Hibbs DE, et al. Evolution of resistance to Aurora kinase B inhibitors in leukaemia cells. PLoS One 2012;7(2): e30734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fojo T, Bates S. Strategies for reversing drug resistance. Oncogene 2003;22(47):7512–7523. [DOI] [PubMed] [Google Scholar]
- 21.Kruh GD. Introduction to resistance to anticancer agents. Oncogene 2003;22(47):7262–7264. [DOI] [PubMed] [Google Scholar]
- 22.Bronchud MH. Selecting the right targets for cancer therapy. In: Bronchud MH, Foote MA, Giaccone G, Olopade O, Workman P, eitorts. Principles of molecular oncology. New Jersey: Humana Press; 2008. p. 3–27. [Google Scholar]
- 23.Simon JA, Szankasi P, Nguyen DK, Ludlow C, Dunstan HM, Roberts CJ, et al. Differential toxicities of anticancer agents among DNA repair and checkpoint mutants of Saccharomyces cerevisiae. Cancer Res 2000;60(2):328–333. [PubMed] [Google Scholar]
- 24.List AF. Multidrug resistance: clinical relevance in acute leukemia. Oncology (Williston Park) 1993;7(10):23–28, 32; discussion 32, 35–38. [PubMed] [Google Scholar]
- 25.Galimberti S, Testi R, Guerrini F, Fazzi R, Petrini M. The clinical relevance of the expression of several multi-drug-resistant-related genes in patients with primary acute myeloid leukemia. J Chemother 2003;15(4):374–379. [DOI] [PubMed] [Google Scholar]
- 26.Filipits M, Stranzl T, Pohl G, Suchomel RW, Zöchbauer S, Brunner R, et al. MRP expression in acute myeloid leukemia. An update. Adv Exp Med Biol 1999;457:141–150. [DOI] [PubMed] [Google Scholar]
- 27.Sargent JM, Williamson CJ, Maliepaard M, Elgie AW, Scheper RJ, Taylor CG. Breast cancer resistance protein expression and resistance to daunorubicin in blast cells from patients with acute myeloid leukaemia. Br J Haematol 2001;115(2):257–262. [DOI] [PubMed] [Google Scholar]
- 28.Kaur B, Khwaja FW, Severson EA, Matheny SL, Brat DJ, Van Meir EG. Hypoxia and the hypoxia-inducible-factor pathway in glioma growth and angiogenesis. Neuro Oncol 2005;7(2):134–153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wellmann S, Guschmann M, Griethe W, Eckert C, Stackelberg A, Lottaz C, et al. Activation of the HIF pathway in childhood ALL, prognostic implications of VEGF. Leukemia 2004;18(5):926–933. [DOI] [PubMed] [Google Scholar]
- 30.Greijer A, Van Der Groep P, Kemming D, Shvarts A, Semenza G, Meijer G, et al. Up-regulation of gene expression by hypoxia is mediated predominantly by hypoxia-inducible factor 1 (HIF-1). J Pathol 2005;206(3): 291–304. [DOI] [PubMed] [Google Scholar]
- 31.Ramasamy R, Lam EW, Soeiro I, Tisato V, Bonnet D, Dazzi F. Mesenchymal stem cells inhibit proliferation and apoptosis of tumor cells: impact on in vivo tumor growth. Leukemia 2007;21(2):304–310. [DOI] [PubMed] [Google Scholar]
- 32.Lin YM, Zhang GZ, Leng ZX, Lu ZX, Bu LS, Gao S, et al. Study on the bone marrow mesenchymal stem cells induced drug resistance in the U937 cells and its mechanism. Chin Med J (Engl) 2006;119(11):905–910. [PubMed] [Google Scholar]
- 33.Houthuijzen J, Daenen LG, Roodhart JM, Voest EE. The role of mesenchymal stem cells in anti-cancer drug resistance and tumour progression. Br J Cancer 2012;106 (12):1901–1906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Vianello F, Villanova F, Tisato V, Lymperi S, Ho KK, Gomes AR, et al. Bone marrow mesenchymal stromal cells non-selectively protect chronic myeloid leukemia cells from imatinib-induced apoptosis via the CXCR4/CXCL12 axis. Haematologica 2010;95(7):1081–1089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wong RS. Mesenchymal stem cells: angels or demons? J Biomed Biotechnol 2011;2011:459510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Comerford KM, Wallace TJ, Karhausen J, Louis NA, Montalto MC, Colgan SP. Hypoxia-inducible factor-1-dependent regulation of the multidrug resistance (MDR1) gene. Cancer Res 2002;62(12):3387–3394. [PubMed] [Google Scholar]
- 37.Petit C, Gouel F, Dubus I, Heuclin C, Roget K, Vannier J. Hypoxia promotes chemoresistance in acute lympho-blastic leukemia cell lines by modulating death signaling pathways. BMC Cancer 2016;16(1):746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Huang Y, Du KM, Xue ZH, Yan H, Li D, Liu W, et al. Cobalt chloride and low oxygen tension trigger differentiation of acute myeloid leukemic cells: possible mediation of hypoxia-inducible factor-1alpha. Leukemia 2003;17(11):2065–2073. [DOI] [PubMed] [Google Scholar]
- 39.Yook YJ, Seo YJ, Kang HJ, Ko SH, Shin HY, Lee JJ, et al. Induction of hypoxia-inducible factor-1α inhibits drug-induced apoptosis in the human leukemic cell line HL-60. Korean J Hematol 2010;45(3):158–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zou J, Li P, Lu F, Liu N, Dai J, Ye J, et al. Notch1 is required for hypoxia-induced proliferation, invasion and chemoresistance of T-cell acute lymphoblastic leukemia cells. J Hematol Oncol 2013;6:3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Deynoux M, Sunter N, Hérault O, Mazurier F. Hypoxia and Hypoxia-inducible Factors in Leukemias. Front Oncol 2016;6:41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu W, Guo M, Xu YB, Li D, Zhou ZN, Wu YL, et al. Induction of tumor arrest and differentiation with prolonged survival by intermittent hypoxia in a mouse model of acute myeloid leukemia. Blood 2006;107(2): 698–707. [DOI] [PubMed] [Google Scholar]