Abstract
Introduction
In an experimental study on carbon monoxide (CO) exposure in swine, we aimed to compare the influence of oxygen therapy using a non-rebreathing mask (NRM) to continuous positive airway pressure (CPAP) and two pressure support ventilation (PSV) devices on the decrease of the terminal elimination half-life of carboxyhemoglobin (COHb t1/2). This was the primary outcome.
Methods
Eight spontaneously breathing pigs were sedated by propofol and exposed to 940 ppm CO several times (n = 25) to obtain COHb levels of 30%. CPAPb (high flow open system, CPAP Boussignac® [7.5 cmH2O]), PSV-Vy (open system, Vylife Boussignac®), and PSV-Leg (closed system, Legendair® [inspiratory/expiratory airway pressure 12/4 cmH2O]) devices were used in a randomized order and compared to NRM (O2 at 15 l min−1) and atmospheric air (AA). The primary outcome was COHb t1/2. Multiple comparisons were performed using Dunn’s tests.
Results
Median FiO2 and minute ventilation were significantly higher in the PSV-Leg group than the NRM group (p < 0.05). Median COHb t1/2 was 251, 85, 82, 93, and 58 min for AA, NRM, CPAPb, PSV-Vy, and PSV-Leg, respectively. All the interventions were superior to AA in terms of CO elimination (p < 0.001), but there was no statistically significant difference between CPAP or PSV and NRM. There was only a trend between PSV-Leg and NRM (p = 0.18). The median AUCs for ln (COHb) × time (h) were 170, 79, 83, 100, and 64 for AA, NRM, CPAPb, PSV-Vy, and PSV-Leg respectively, with a statistically significant difference only between AA and PSV-Leg (p = 0.002).
Conclusion
In conclusion, in our study on CO intoxication in swine, the use of the closed PSV-Leg system led to the shortest COHb t1/2. These results suggest that PSV-Leg can be more efficient than NRM in eliminating CO and support the design of a clinical study to assess this hypothesis.
Electronic supplementary material
The online version of this article (10.1007/s13181-018-0654-8) contains supplementary material, which is available to authorized users.
Keywords: Carbon monoxide, Swine, Positive pressure ventilation, Continuous positive airway pressure (CPAP), Toxicokinetic, Carboxyhemoglobin (COHb), Oxygen therapy
Introduction
Carbon monoxide (CO) is one of the most frequent causes of toxic deaths in the world [1]. The main objective of oxygen therapy for CO poisoning is to increase the CO elimination rate, i.e., to decrease the elimination half-life of carboxyhemoglobin (COHb t1/2). The final objective of this therapeutic approach is to prevent delayed neurological sequelae.
The mode of oxygen administration strongly influences its efficacy: COHb t1/2 ranges from 320 min at atmospheric room air (AA) to 71 min with normobaric oxygen therapy coupled to a non-rebreather mask (NRM) at 15 l min−1 and to 21 min with hyperbaric oxygen (HBO) therapy at a pressure of 1.5 to 3.0 atm absolute (ATA) [2, 3].
Nevertheless, the clinical benefit of HBO therapy for the prevention of neurological sequelae is still under debate. HBO therapy could even expose the victims to deleterious effects [4–6]. While oxygen seems to be the natural antidote for CO poisoning, there is some concern regarding the role of massive oxygen therapy on free-radical production, leading to further cellular damage [4, 6]. Moreover, HBO therapy is not uniformly available in all areas at all times, which leads to significant delays in initiating therapy [7]. This led us to investigate other convenient ways to improve CO elimination. Continuous positive airway pressure (CPAP) with high flow oxygen device and pressure support ventilation (PSV) may positively influence fraction of inspired oxygen (FiO2) and minute ventilation and may be candidates for CO intoxication treatment [8]. Our hypothesis was that these ventilation techniques, which are more convenient and more widely available than HBO, could be more efficient in eliminating CO than NRM. However, as these techniques had not been previously evaluated in this context, a preliminary assessment was required in order to ensure that our hypothesis was plausible and could be safely tested in a clinical setting, find the most promising ventilation support, and estimate the number of patients required for a clinical study. We therefore performed a pilot experimental study on swine in which we compared the effect of CPAP and two PSV devices to NRM and atmospheric air (as a control without treatment) on COHb t1/2.
Materials and Methods
A prospective exploratory randomized trial on swine was conducted using eight male Landrace pigs (77 kg, [72–88]). This animal experimental study was approved by the Université Catholique de Louvain’s ethics committee for animal experimentation (UCL, Brussels, Belgium) (ref 2013/UCL/MD/001 and ref 2015/UCL/MD/28). Formal approval was obtained for the use of the photographs taken during the experiments.
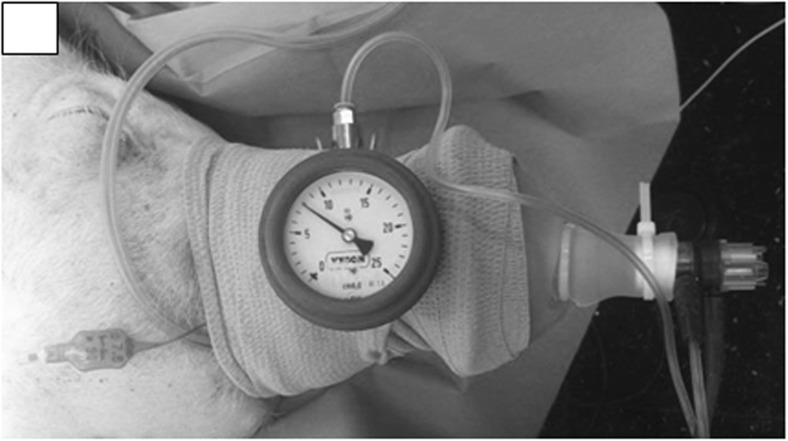
Intubation was performed on the animals using a cuffed endotracheal tube (7.5 to 8.0) 15 to 20 min after they had been pre-medicated with intramuscular injections of tiletamine/zolazepan (Zoletil® 100, Virbac© (6 mg kg−1) and Xylazin (Rompun® 2%, Bayer©) (2 mg kg−1)). The pigs were sedated with a continuous intravenous administration of propofol (1.5 to 2.3 mg kg−1) in order to maintain spontaneous breathing throughout the study. Intubation was required to avoid obstructive apneic episodes during sedation and to record minute ventilation and end-tidal CO2 levels during the experiment. In order to try to reproduce non-invasive positive pressure ventilation (NiPPV), we did not connect the device directly to the endotracheal tube but to a non-invasive nasobuccal mask (Fig. 1).
Fig. 1.
Experimental ventilation interface. The mask was adapted using an elastic strip to cover their labial commissure and limit air leaks
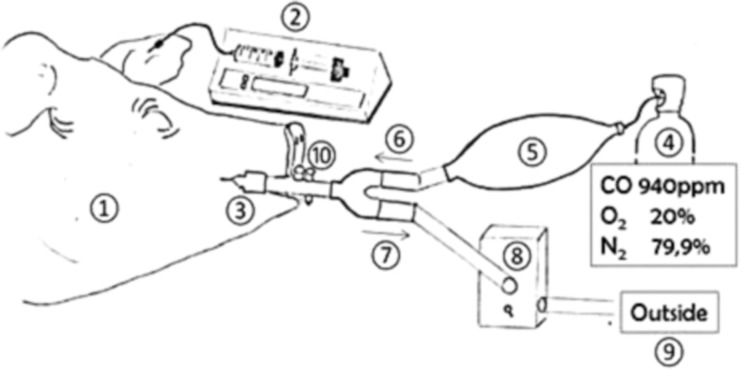
The animals were then placed in the lateral decubitus position for the whole duration of the study. The propofol infusion rate was adjusted to obtain a balance between sedation and awakening, and pCO2 values were maintained between 35 and 40 mmHg. Propofol was also titrated to control hemodynamic changes, spontaneous motor activity and palpebral reflexes, hypoventilation, and responses to noxious stimuli. Target levels of intoxication were obtained by administering a combination of 940 ppm (0.94%) CO gas, 20% O2, and 79.906% N2 (Pulmo1®, Air Liquide Medical systems©). This was administrated via a secured system using the endotracheal tube during spontaneous breathing. All exhaust gases from the respiratory circuit were scavenged and released outside the building (Fig. 2). The CO concentration in the ambient air was continuously tested (PAC7000®, Drager©, Germany).
Fig. 2.
CO intoxication phase. 1 Pig. 2 Anesthesia with propofol 2%. 3 Spontaneous breathing through endotracheal tube. 4 Gas source containing CO 940 ppm. 5 Balloon with gas reserve for inhalation. 6/7 One-direction valve. 8 Exhaled gas scavenger. 9 Gas evacuation outside the room. 10 Pneumotachograph and FiO2 measurement. After the intoxication phase, the endotracheal tube was kept in the same position and was covered by an adapted facial mask connected to the respiratory device (Fig. 1)
Catheters were inserted into the carotid or femoral artery to continuously measure blood pressure and to determine COHb levels (%). All blood samples were analyzed for pH, paCO2 (mmHg), paO2 (mmHg), lactate (mmol l−1), and COHb using the blood gas analyzer ABL90Flex® (Radiometer™, Radiometer Medical Aps, Denmark). For each experiment, minute ventilation (ml min−1), invasive blood pressure (mmHg), FiO2 (%), heart rate (min−1), SpO2 (%), end-tidal CO2 (mmHg), and temperature (°C) were measured continuously using a pneumotachograph and a Datex-Ohmeda® S/5™ light monitor (Datex-Ohmeda©, Finland). Table warming was adjusted to maintain a target temperature of 37.5 °C.
Three different devices were investigated and compared to NRM: CPAP Boussignac® (CPAPb, Vygon©, Ecouen, France) and two PSV devices: Legendair® ventilator (Airox©, France) (PSV-Leg) and Vylife® (Vygon©, Ecouen, France) (PSV-Vy). The CPAPb system provides continuous positive pressure by injecting high-speed gas into a cylinder through angled side channels. This open system is fed by pure oxygen at 25–30 l min−1 and delivers FiO2 values from 0.7 to 1.0, depending on changes in respiratory rate and current volume [9, 10]. The pressure created from the flow of gas was monitored and maintained at 7.0–7.5 cm H2O [11]. PSV-Vy is an open system, whereas PSV-Leg is a closed system. For both devices, positive airway pressure was set at + 12 cm H2O for the inspiratory phase and + 4 cm H2O for the expiratory phase, with pure oxygen, FiO2, at 1.0.
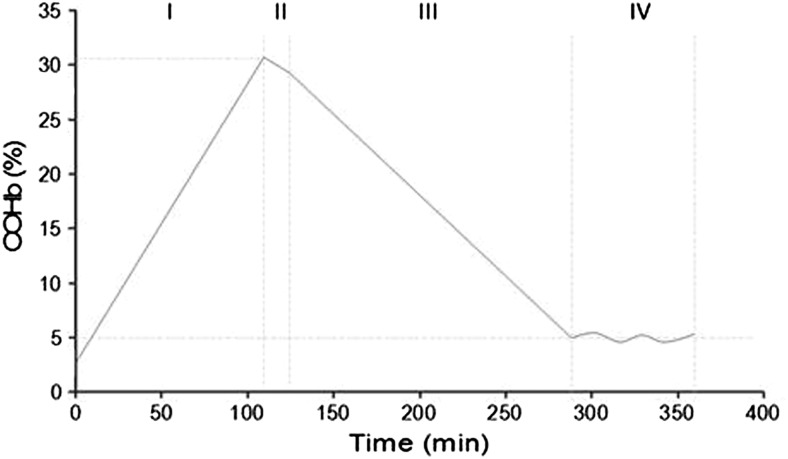
The complete experimental protocol was composed of four consecutive phases (Fig. 3): (I): the intoxication phase with determination of baseline COHb blood level followed by iterative sampling every 10 min until COHb levels reached 30% (± 2%). (II): the “no intervention phase,” with disconnection of the CO circuit. During that period, the animals were placed under ambient air (AA) for 10 min. This corresponded to the virtual delay between the extraction of the victim from the toxic environment and the arrival of the first responders in the context of human exposure. This delay was required for the installation of the respiratory device. (III): the treatment phase, with blood samples collected every 6 min until COHb returned to 5%. (IV): the post-treatment phase without any intervention for 60 min at AA in order to document the decrease in COHb or rule out a rebound.
Fig. 3.
Experimental protocol. Phase I: intoxication; Phase II: no treatment (atmospheric room air); Phase III: treatment phase with different oxygenation/ventilation devices; Phase IV: no treatment (atmospheric room air) to assess COHb levels and verify the absence of a rebound
Each day, one protocol was applied to a single pig, using a randomized treatment that was assigned after the intoxication phase. In order to limit the number of animals required and due to economic constraints, the same animal could be exposed to a new intoxication phase and new randomized treatment using the same protocol the following day.
A minimum time of 12 h was established between experiments and we determined COHb levels before each experiment so as to exclude significant residual exposure, and we measured lactate levels so as to exclude end-organ damage.
Overall, four experiments were conducted with ambient air (AA), eight with NRM, five with CPAPb, five with PSV-Leg, and three with PSV-Vy.
The animals were euthanized at the end of the experiments once the study had been completed using a T-61® (embutramide/mebezonium iodide/tetracaine hydrochloride) injection.
The homogeneity of the intoxication phase was verified by comparing COHb levels and other parameters between the experimental groups at any time. The COHb t1/2 was assessed using non-compartmental analysis. The areas under the curve (AUCs) ln (COHB) × time (h) were calculated. Results were expressed as medians [range], and continuous outcomes were compared using the Kruskal-Wallis test and Dunn’s test for multiple comparisons using GraphPad Prism 4® software (α = 0.05).
Results
The clinical parameters were similar during all the phases of the experiment among the different intervention groups (Supplementary table, online resource). There was no significant increase in lactate levels, and no rebounds in COHb levels were observed up to 1 h after the treatment had ended.
Measurements for delivered oxygen therapy were the highest in the PSV-Leg group in terms of median FiO2 (p < 0.05 when compared to NRM, CPAPb, and PSV-Vy), minute ventilation (p < 0.05 when compared to NRM and CPAPb), and respiratory rate (p < 0.05 when compared to NRM and CPAPb) (Table 1). Median pO2 values were significantly higher in the PSV-Leg group when compared to the CPAPb group (p < 0.05), and there was no difference in median pCO2 values between the intervention groups (Table 1).
Table 1.
Median [range] respiratory parameters in swine during treatment. COHb t1/2 elimination half-life of carboxyhemoglobin, AUC area under the curve, FiO2 fraction of inspired oxygen, pCO2 partial pressure of carbon dioxide, pO2 partial pressure of oxygen, MV minute ventilation in mL/min. AA atmospheric air, NRM high concentration non-rebreathing mask, CPAPb CPAP Boussignac, PSV-Vy PSV-Vylife, PSV-Leg PSV-Legendair
| AA (n = 4) | NRM (n = 8) | CPAP (n = 5) | PSV-Vy (n = 3) | PSV-Leg (n = 5) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| COHb t1/2 (min) | 251 | [130–273] | 85 | [46–116] | 82 | [40–94] | 93 | [92–113] | 58 | [52–79] |
| AUC (ln COHb x time (h)) | 170 | [126–222] | 79 | [57–105] | 83 | [55–95] | 100 | [95–102] | 64 | [60–65] |
| FiO2 (%) | 21 | 72 | [59–83] | 54 | [49–73] | 51 | [51–53] | 92 | [76–93] | |
| pCO2 (mmHg) | 45 | [30–58] | 43.3 | [39.2–52.5] | 47.8 | [46.5–48.3] | 40.6 | [37.1–46.7] | 44.6 | [39.4–48.6] |
| pO2 (mmHg) | 88 | [49–173] | 369.4 | [230.1–418.2] | 281.0 | [234.0–441.2] | 286.2 | [247.1–300.5] | 475.0 | [323.4–478.6] |
| MV (ml min−1) | 6090 | [1800–15,040] | 6539 | [5587–10,312] | 7418 | [6788–8460] | 8096 | [6695–8602] | 9726 | [9377–11,404] |
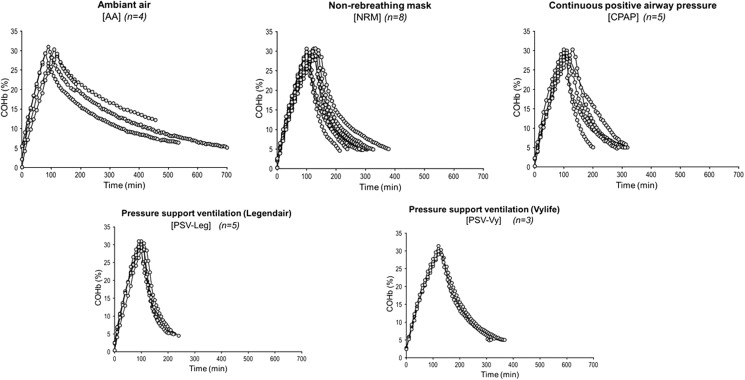
With regard to COHb toxicokinetics, the median terminal half-life of COHb was 251, 85, 82, 93, and 58 min for AA, NRM, CPAPb, PSV-Vy, and PSV-Leg, respectively (Fig. 4 and Table 1). All the interventions (NRM, CPAPb, PSV-Vy, and PSV-Leg) were superior to the reference group (AA) in terms of COHb t1/2 (p < 0.001). However, there was no statistically significant difference between the intervention groups, with a trend only between PSV-Leg (58 min, n = 5) and NRM (85 min, n = 8) (p = 0.18).
Fig. 4.
Toxicokinetic profile of COHb for each treatment group. Atmospheric air (n = 4), CPAPb (n = 5), NRM (n = 8), PSV-Leg (n = 5), and PSV-Vy (n = 3)
The median AUCs for ln (COHb) × time (h) were 170, 79, 83, 100, and 64 for AA, NRM, CPAPb, PSV-Vy, and PSV-Leg, respectively, with a statistically significant difference only between AA and PSV-Leg (p = 0.002).
Discussion
Our pilot experimental study in swine on CO intoxication and non-invasive pressure support ventilation with the closed Legendair® system showed promising results in terms of COHb t1/2 and the AUCs for COHb but did not indicate that there were benefits to using CPAP or PSV-Vy for CO elimination in comparison to NRM.
Non-invasive ventilation modes are widely used to treat acute respiratory failure in pre-hospital settings and emergency units, but, to our knowledge, our study is the first one to evaluate CPAP and PSV specifically in relation to CO intoxication. Several explanations can be hypothesized in light of our results and a few important limitations need to be acknowledged.
Firstly, our results confirm that NRM may not deliver 100% oxygen and, conversely to HBO, all ventilation supports can only lead to a limited value of pO2 and a marginal increase in fractional atmospheric pressure [12]. Indeed, the Haldane equation for the reaction of CO in a blood sample is [HbCO]/[HbO2] = M pCO/pO2, where [HbO2] = oxyhemoglobin rate, pCO = partial pressure in CO dissolved, pO2 = partial pressure in oxygen dissolved, and M = Haldane constant. Nevertheless, several in vivo studies showed the benefit of the influence of ventilation on COHb half-life [7]. Improving ventilation without excessive increasing pO2 could limit the adverse effects of hyper-oxygenation. Moreover, our results suggest that ventilation therapies are not equal in terms of CO elimination. Oxygenation using NRM is considered the first standard treatment for CO poisoning. The FiO2 delivered at 15 l min−1 using NRM is expected to reach up to 100%. However, due to unavoidable leaks around the mask and rebreathing, a maximum concentration of 85% is usually reached [13, 14] . In our study, the mean FiO2 obtained was 72%. For CPAPb, the mean FiO2 was limited to 58% for inspiratory flow in our experimental conditions, which runs contrary to the results of Templier et al.’s bench laboratory test [10]. We observed similar results for PSV-Vy, with a mean FiO2 of 51%. Both CPAPb and PSV-Vy are considered open systems. These open-system devices offer the advantage of more comfort for the patient, but the drawback of this is that atmospheric room air is inspired at the same time, thus resulting in lower FiO2 levels in the gas mixture depending on inspiratory flow. Conversely, the PSV-Leg is considered a closed system and delivered higher levels of FiO2 (92%) than NRM, CPAPb, and PSV-Vy. Interestingly, the PSV-Leg yielded the best results in terms of COHb t1/2. Compared to NRM and CPAP, PSV is an effective ventilation mode for partly overcoming the potentially noxious effects of the sedation which decreases MV. It also improves pO2. Nevertheless, PSV-Vy and PSV-Leg used the same pressure levels and mode, yet the potential benefit of PSV-Leg mainly seems related to its higher levels of oxygen delivery. In addition, the trigger on PSV-Vy is unchangeable compared to that of PSV-Leg, so an inspiratory delay may lead to an increased inspiratory flow and thus decrease FiO2.
Secondly, our study in swine mimicked CO intoxication in humans to a certain extent but differed on several counts. Pigs were selected because they share some physiological cardiorespiratory characteristics with humans [15]. However, the relative affinity of swine hemoglobin for CO compared to O2 (the Haldane constant [M]) at 37 °C is assumed to be 130, whereas the median (M) constant in humans is around 218 [16, 17]. This lower affinity for CO in swine hemoglobin may result in shorter laboratory experiments. We considered 30% COHb a significant level of intoxication because this level is supposed to induce clinically relevant symptoms in human intoxication and require HBO therapy [4]. The animals in the experiment had a body weight comparable to human (72 to 88 kg) and a facial size compatible with CPAP and PSV masks. Nevertheless, we needed to sedate them during the experiment and, as a first pilot experiment had shown numerous obstructive apneic episodes in pigs sedated under propofol, intubation was required. In order to reproduce non-invasive ventilation conditions, we did not directly connect the system to the endotracheal tube but to a nasobuccal mask (Fig. 1).
Thirdly, the power of the statistical analysis was limited by the small number of animals included in each intervention sub-group. This may explain why the difference observed between the PSV-Leg group (n = 5) and the NRM group (n = 8) was not statistically significant. Moreover, in an exploratory setting, we performed several analyses without considering alpha risk inflation.
Finally, we cannot totally exclude the possibility that prior CO poisoning could have affected the terminal elimination half-life of CO in subsequent trials by virtue of end-organ damage. However, another experimental study found that long-term exposure to 200 ppm CO > 24 h did not lead to pathological changes in tissues or organs [18]. Moreover, before each experiment, we determined COHb levels so as to exclude significant residual exposure to CO and measured lactate levels for every COHb sample so as to exclude end-organ damage.
Conclusion
In conclusion, in our study on CO intoxication in swine, the closed PSV-Leg system gave the shortest COHb t1/2 (median 58 vs. 85 min with NRM) and the lowest AUC for COHb (median 63 vs. 79 with NRM). The results of our pilot study suggest that non-invasive ventilation, such as the closed PSV-Leg system, can be more efficient than NRM at eliminating CO when HBO is unavailable and supports the design of a clinical study to evaluate this hypothesis.
Electronic supplementary material
Supplementary table, online resource. Median of different parameters measured in swine during the intoxication and treatment phase. NRM: high concentration non-rebreathing mask; CPAPb: CPAP Boussignac; PSV-Leg: PSV-Legendair; PSV-Vy: PSV- Vylife; AA: atmospheric air; HR: heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; COHb: carboxyhemoglobin; Hb: hemoglobin; T°: body temperature. (PDF 124 kb)
Acknowledgments
The authors wish to thank Sabine Jaumotte (Radiometer©, Medical Aps, Denmark) for their essential technical support and the nurses from the emergency unit (Cliniques universitaires Saint-Luc, UCL, Brussels, Belgium) for their assistance and precious help. We thank Doctor Maximilien Thoma for his assistance in surgical procedures and Johanna Maccioni for reviewing the manuscript.
Funding Information
This study was supported by the Saint-Luc Foundation and the Experimental Surgery and Transplantation unit (CHEX, Université catholique de Louvain, UCL, Brussels, Belgium).
Compliance with Ethical Standards
Conflicts of interest
ND received financial support from Vygon© (France) for this study. Moreover, ND produced pedagogic and promotional medical films for this company. Other authors declare that they have no conflicts of interest.
Footnotes
This work has been partly presented as a poster at the 37th ISICEM Congress in Brussels, Belgium, March 21–24, 2017 and the corresponding abstract was published in Critical Care 2017, 21(Suppl 1):56.
Electronic supplementary material
The online version of this article (10.1007/s13181-018-0654-8) contains supplementary material, which is available to authorized users.
References
- 1.Braubach M, Algoet A, Beaton M, Lauriou S, Heroux ME, Krzyzanowski M. Mortality associated with exposure to carbon monoxide in WHO European member states. Indoor Air. 2013;23(2):115–125. doi: 10.1111/ina.12007. [DOI] [PubMed] [Google Scholar]
- 2.Pace N, Strajman E, Walker EL. Acceleration of carbon monoxide elimination in man by high pressure oxygen. Science. 1950;111(2894):652–654. doi: 10.1126/science.111.2894.652. [DOI] [PubMed] [Google Scholar]
- 3.Jay GD, McKindley DS. Alterations in pharmacokinetics of carboxyhemoglobin produced by oxygen under pressure. Undersea Hyperb Med J Undersea Hyperb Med Soc Inc. 1997;24:165–173. [PubMed] [Google Scholar]
- 4.Buckley NA, Juurlink DN, Isbister G, Bennett MH, Lavonas EJ. Hyperbaric oxygen for carbon monoxide poisoning. Cochrane Database Syst Rev. 2011. 10.1002/14651858.CD002041.pub3. [DOI] [PMC free article] [PubMed]
- 5.Annane D, Chadda K, Gajdos P, Jars-Guincestre MC, Chevret S, Raphael JC. Hyperbaric oxygen therapy for acute domestic carbon monoxide poisoning: two randomized controlled trials. Intensive Care Med. 2011;37(3):486–492. doi: 10.1007/s00134-010-2093-0. [DOI] [PubMed] [Google Scholar]
- 6.Huang CC, Ho CH, Chen YC, Lin HJ, Hsu CC, Wang JJ, Su SB, Guo HR. Hyperbaric oxygen therapy is associated with lower short- and long-term mortality in patients with carbon monoxide poisoning. Chest. 2017;S0012-3692(17):30723–30727. doi: 10.1016/j.chest.2017.03.049. [DOI] [PubMed] [Google Scholar]
- 7.Fisher JA, Iscoe S, Fedorko L, Duffin J. Rapid elimination of CO through the lungs: coming full circle 100 years on. Exp Physiol. 2011;96(12):1262–1269. doi: 10.1113/expphysiol.2011.059428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Thys F, Roeseler J, Delaere S, Palavecino L, El Gariani A, Marion E, et al. Two-level non-invasive positive pressure ventilation in the initial treatment of acute respiratory failure in an emergency department. Eur J Emerg Med. 1999;6(3):207–214. [PubMed] [Google Scholar]
- 9.Spijker EE, de Bont M, Bax M, Sandel M. Practical use, effects and complications of prehospital treatment of acute cardiogenic pulmonary edema using the Boussignac CPAP system. Int J Emerg Med. 2013;6(1):8. doi: 10.1186/1865-1380-6-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Keenan SP, Sinuff T, Burns KE, Muscedere J, Kutsogiannis J, Mehta S, et al. Clinical practice guidelines for the use of noninvasive positive-pressure ventilation and noninvasive continuous positive airway pressure in the acute care setting. CMAJ. 2011;183(3):E195–E214. doi: 10.1503/cmaj.100071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Briganti A, Melanie P, Portela D, Breghi G, Mama K. Continuous positive airway pressure administered via face mask in tranquilized dogs. J Vet Emerg Crit Care (San Antonio) 2010;20(5):503–508. doi: 10.1111/j.1476-4431.2010.00579.x. [DOI] [PubMed] [Google Scholar]
- 12.Wexler HR, Aberman A, Scott AA, Cooper JD. Measurement of intratracheal oxygen concentrations during face mask administration of oxygen: a modification for improved control. Can Anaesth Soc J. 1975;22(4):417–431. doi: 10.1007/BF03004856. [DOI] [PubMed] [Google Scholar]
- 13.Burkhart JE, Jr, Stoller JK. Oxygen and aerosolized drug delivery: matching the device to the patient. Cleve Clin J Med. 1998;65(4):200–208. doi: 10.3949/ccjm.65.4.200. [DOI] [PubMed] [Google Scholar]
- 14.Templier F, Dolveck F, Baer M, Chauvin M, Fletcher D. Laboratory testing measurement of FIO2 delivered by Boussignac CPAP system with an input of 100% oxygen. Ann Fr Anesth Reanim. 2003;22(2):103–107. doi: 10.1016/S0750-7658(02)00859-6. [DOI] [PubMed] [Google Scholar]
- 15.Schomberg DT, Tellez A, Meudt JJ, Brady DA, Dillon KN, Arowolo FK, Wicks J, Rousselle SD, Shanmuganayagam D. Miniature swine for preclinical modeling of complexities of human disease for translational scientific discovery and accelerated development of therapies and medical devices. Toxicol Pathol. 2016;44(3):299–314. doi: 10.1177/0192623315618292. [DOI] [PubMed] [Google Scholar]
- 16.Klimisch HJ, Chevalier HJ, Harke HP, Dontenwill W. Uptake of carbon monoxide in blood of miniture pigs and other mammals. Toxicology. 1975;3(3):301–310. doi: 10.1016/0300-483X(75)90031-1. [DOI] [PubMed] [Google Scholar]
- 17.Rodkey FL, O'Neal JD, Collison HA. Oxygen and carbon monoxide equilibria of human adult hemoglobin at atmospheric and elevated pressure. Blood. 1969;33(1):57–65. [PubMed] [Google Scholar]
- 18.Jones RA, Strickland JA, Stunkard JA, Siegel J. Effects on experimental animals of long-term inhalation exposure to carbon monoxide. Toxicol Appl Pharmacol. 1971;19(1):46–53. doi: 10.1016/0041-008X(71)90188-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary table, online resource. Median of different parameters measured in swine during the intoxication and treatment phase. NRM: high concentration non-rebreathing mask; CPAPb: CPAP Boussignac; PSV-Leg: PSV-Legendair; PSV-Vy: PSV- Vylife; AA: atmospheric air; HR: heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; COHb: carboxyhemoglobin; Hb: hemoglobin; T°: body temperature. (PDF 124 kb)