Abstract
Background
In order to decrease the incidence of ventilator-associated pneumonia (VAP) in Belgium, a national campaign for implementing a VAP bundle involving assessment of sedation, cuff pressure control, oral care with chlorhexidine and semirecumbent position, was launched in 2011–2012. This report will document the impact of this campaign.
Methods
On 1 day, once a year from 2010 till 2016, except in 2012, Belgian ICUs were questioned about their ventilated patients. For each of these, data about the application of the bundle and the possible treatment for VAP were recorded.
Results
Between 36.6 and 54.8% of the 120 Belgian ICUs participated in the successive surveys. While the characteristics of ventilated patients remained similar throughout the years, the percentage of ventilated patients and especially the duration of ventilation significantly decreased before and after the national VAP bundle campaign. Ventilator care also profoundly changed: Controlling cuff pressure, head positioning above 30° were obtained in more than 90% of cases. Oral care was more frequently performed within a day, using more concentrated solutions of chlorhexidine. Subglottic suctioning also was used but in only 24.7% of the cases in the last years. Regarding the prevalence of VAP, it significantly decreased from 28% of ventilated patients in 2010 to 10.1% in 2016 (p ≤ 0.0001).
Conclusion
Although a causal relationship cannot be inferred from these data, the successive surveys revealed a potential impact of the VAP bundle campaign on both the respiratory care of ventilated patients and the prevalence of VAP in Belgian ICUs encouraging them to follow the guidelines.
Keywords: VAP, VAP bundle, Belgian ICUs, VAP survey
Background
Ventilator-associated pneumonia (VAP) is among the most common type of intensive care unit (ICU)-acquired infection and is associated with significant morbidity and mortality [1]. In Europe, the incidence remains higher than in the USA despite the implementation of VAP bundles [2–4]. The need for the implementation of multimodal approach to decrease the incidence of VAP has been recently reemphasized by European guidelines [5] and especially by guidelines coming from the société française d’anesthésie-réanimation and the société de réanimation de langue française [6, 7]. Besides the use of selective digestive decontamination, these guidelines support the use of 6 procedures: avoiding intubation by the use of noninvasive ventilation, avoiding nasotracheal intubation, controlling cuff pressure, reducing the level of sedation, early enteral nutrition and subglottic suctioning.
In Belgium, after having observed high rate of VAP in ICUs from previous surveys, the federal service launched a promotional campaign to implement a national VAP bundle in 2011. This campaign involved several meetings in Brussels (attended by representatives from most Belgian ICUs) where the Belgian VAP bundle was explained and promoted. This campaign was followed by a prospective collect of all the VAP bundle data during 11 months in 2012 from voluntary participating Belgian ICUs. This collect was performed by the federal service. The national VAP bundle involved 4 items: a protocol with daily assessment of sedation, a semirecumbent position of at least 30°, the control of cuff pressure between 20 and 30 cm of H2O and the oral care with chlorhexidine. In addition, the use of subglottic suctioning was encouraged. Before and after this campaign, the college of physicians for intensive care, which also relies on the federal public service for health, food chain safety and environment, has performed surveys to evaluate the prevalence of VAP in Belgian ICUs. The present paper describes the results of the successive surveys and will examine the impact the campaign could have on the compliance of medical teams for implementing the bundle and on the prevalence of VAP. Data from the 2012 national collection study have been already published [8].
Methods
Once a year, from 2010 till 2016, except in 2012, all the 120 adult ICUs in Belgium received an invitation to participate in 1-day survey performed by the college of physicians for intensive care about ventilated patients and the occurrence of VAP. ICUs were asked about their number of beds, their occupancy, the number of ventilated patients. Ventilated patient characteristics included age, sex, primary reason for ICU admission, comorbidities, date of admission to the hospital and to the ICU, date of intubation and cause of ventilation. Regarding ventilation care, the way of intubation (nasal, oral, tracheostomy),the type of cuff (polyvinyl, polyurethane), the current cuff pressure, the type of suctioning system (opened or closed), the current head positioning, the moistening system (heat and moisture exchangers, active devices) and the use of a subglottic suctioning system were recorded for each patient. Regarding the oral care, the type of disinfection (chlorhexidine, polyvidone iodine, other), the rate of disinfection, the use of dental brushing and the type of nutrition tubing (nasogastric, orogastric, postpyloric tube, gastrostomy or jejunostomy) were also recorded for each ventilated patients. If a patient was treated for a VAP, the bacteriological results were asked for and the severity of the infection according to the grade of sepsis was recorded. No follow-up of patients was obtained.
VAP diagnosis was based on new infiltrate on chest X-ray with either fever above 38° or less than 35° or leukocytosis above 10,000 white blood cells/mm3 and either occurrence of purulent tracheal secretions or decrease in PaO2/FiO2. After each survey, all the ICUs, having or not participated in the survey, received a report describing the results and were encouraged to continue to implement the VAP bundle.
Statistical analysis
Quantitative data were summarized as median and interquartile (IQR) values or as mean and SD when normally distributed. Comparisons were made by the Kruskal–Wallis or Student’s t test as appropriate for continuous variables and by Chi-square or Fisher’s exact test for categorical variables. All tests were two-sided, and statistical significance was set at p less than 0.05.
Results
More than 60 adult ICUs participated in the surveys, except in 2016 when there were only 44. Considering that in Belgium there are 120 acute hospitals, these figures correspond to 36.6–54.8% of them. Figure 1 gives the evolution of the number of ICU beds belonging to participating ICUs, the number of patients and among them, the number of ventilated patients. As can be inferred from Fig. 1, the percentage of bed occupancy remained stable, between 75 and 80%, but the percentage of ventilated patients decreased significantly from 44.8% in 2010 to 28.7% in 2016 (p < 0.05). Another impressive difference between the surveys was the decrease in the duration of ventilation from the ICU admission till the day of the survey: The median was 10.5 and 13 days before the campaign, then 7, 5, 5 and 6 days after the campaign (p < 0.001) (Table 1).
Fig. 1.
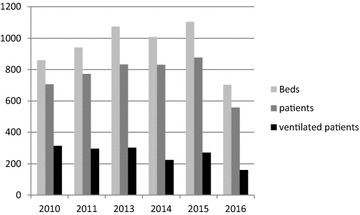
Evolution of the number of ICU beds, patients and ventilated patients in participating ICUs
Table 1.
Characteristics of ventilated patients
| Characteristics of patients | |||||||
|---|---|---|---|---|---|---|---|
| 2010 | 2011 | 2013 | 2014 | 2015 | 2016 | p value | |
| Number | 129 | 293 | 288 | 265 | 271 | 158 | |
| Age median (IQR) | 67 (54–78) | 65 (54–75) | 66 (54–75) | 67 (57–76) | 64 (54–76) | 64 (50–71) | 0.8412 |
| Sex (male) | 88 (68.8%) | 183 (62.5%) | 183 (63.5%) | 162 (61.1%) | 170 (62.7%) | 102 (64.6%) | 0.8326 |
| Pre-ICU hosp. stay | 0 (0–4) | 0 (0–2) | 0 (0–2) | 1 (0–5) | 0 (0–2) | 0 (0–2) | 0.3229 |
| ICU stay median | 10 (4–19) | 13 (7–21) | 8 (3–18) | 7 (2–16) | 7 (2–16) | 8 (2–19) | < 0.0001 |
| Duration of ventilation median | 10.5 (4–17) | 13 (7–21) | 7 (3–16) | 5 (2–14) | 5 (1–15) | 6 (2–14) | < 0.0001 |
| Type of patients | 0.0655 | ||||||
| Medical | 71 (55.8%) | 156 (53.2%) | 155 (53.2%) | 138 (52.1%) | 153 (56.5%) | 94 (59.4%) | |
| Scheduled surgery | 12 (9.3%) | 51 (17.4%) | 57 (19.8%) | 52 (19.6%) | 45 (16.6%) | 18 (11.4%) | |
| Nonscheduled surgery | 32 (24.8%) | 51 (17.4%) | 48 (16.7%) | 59 (22.3%) | 42 (15.5%) | 30 (19%) | |
| Trauma | 13 (10.1%) | 35 (16.1%) | 28 (9.7%) | 16 (6%) | 31 (11.4%) | 16 (10.1%) | |
| Underlying disease | 0.2724 | ||||||
| Smoker | 44 (34.1%) | 102 (47%) | 103 (35.7% | 75 (28.3%) | 81 (29.9%) | 51 (32.3%) | |
| Asthma | 7 (5.4%) | 13 (4.4%) | 5 (1.7%) | 19 (7.2%) | 9(3.3%) | 5 (3.2%) | |
| COPD | 36 (27.9%) | 83 (28.3%) | 89 (30.9%) | 63 (23.8%) | 64 (23.6%) | 38 (24%) | |
| Solid cancer | 4 (3.1%) | 36 (12.3%) | 28 (9.7%) | 28 (10.6%) | 20 (7.4%) | 12 (7.6%) | |
| Hematological cancer | 7 (5.4%) | 7 (2.4%) | 10 (3.5%) | 4 (1.5%) | 9 (3.3%) | 4 (2.5%) | |
| Immunosuppression | 12 (9.3%) | 25 (8.5%) | 20 (6.9%) | 19 (7.2%) | 16 (5.9%) | 6 (3.8%) | |
| Corticotherapy | 9 (7%) | 37 (12.6%) | 28 (9.7%) | 39 (14.7%) | 27 (10%) | 15 (9.5%) | |
| Diabetes | 13 (10.1%) | 38 (13%) | 42 (14.6%) | 34 (12.8%) | 34 (12.5%) | 17 (10.8%) | |
| Other | NA | NA | NA | 26 (9.8%) | 26 (9.6%) | 75 (47.4%) | |
| Cause of ventilation | 0.0012 | ||||||
| Hypoxia | 50 (38.8%) | 79 (27%) | 95 (33%) | 83 (31.3%) | 80 (29.5%) | 55 (34.8%) | |
| Hypercapnia | 7 (5.4%) | 39 (13.3%) | 24 (8.3%) | 19 (7.2%) | 11 (4.1%) | 16 (10.1%) | |
| Central nervous system | 24 (18.6%) | 47 (16%) | 40 (13.9%) | 32 (12.1%) | 45 (16.6%) | 19 (12%) | |
| Peripheral nervous system | 0 (0%) | 1 (0.3%) | 2 (0.7%) | 4 (1.5%) | 5 (1.8%) | 2 (1.3%) | |
| Trauma | 7 (5.4%) | 14 (4.8%) | 12 (4.2%) | 9 (3.4%) | 14 (5.2%) | 10 (6.3%) | |
| Circulatory problem | 17 (13.2%) | 38 (13%) | 40 (17.4%) | 40 (15.1%) | 43 (15.9%) | 17 (10.8%) | |
| Postoperative | 17 (13.2%) | 30 (10.2%) | 59 (20.5%) | 67 (25.2%) | 63 (23.2%) | 25 (15.8%) | |
| Other | 7 (5.4%) | 13 (1.1%) | 16 (5.6%) | 11 (4.1%) | 10 (3.7%) | 14 (8.9%) | |
IQR Interquartile range, ICU intensive care unit, COPD chronic obstruction pulmonary disease, NA not available
The other characteristics of ventilated patients remained quite the same throughout the years of the surveys as shown in Table 1: age, sex, pre-ICU hospitalization stays, types of patients, underlying diseases, none of these characteristics differ between years of survey. However, regarding the causes of ventilation, the differences reached the statistical significance (p = 0.0012).
Regarding ventilatory care, it profoundly changed before and after the national VAP bundle campaign: As shown in Table 2, the cuff pressure measurement, which was not performed in 27% of ventilated patients in 2011, was obtained in more than 90% of cases in 2015. Head positioning above 30° was seen in 2010 only in 54% of ventilated patients, and it was systematically observed in more than 90% of the patients after the campaign except in 2016, when it decreased to 88.6%. Subglottic suctioning also significantly increased, and it was however used in only 24.7% of the cases in 2016. In the same way, oral care was more frequently performed, using more concentrated solutions of chlorhexidine as shown in Table 3. Other oral disinfectants were less often used (from 15.2% in 2010 to 6.3% in 2016), polyvidone iodine solutions remaining at a level of 34.8% in 2016. Dental brushing which was not performed in 25% of the ventilated patients in 2011 was still not done in 17.1% of the patients in 2016.
Table 2.
Ventilatory care
| 2010 | 2011 | 2013 | 2014 | 2015 | 2016 | p value | |
|---|---|---|---|---|---|---|---|
| Previous NIV | 25 (19.4%) | 50 (17.1%) | 52 (18%) | 46 (17.4%) | 44 (16.2%) | 33 (20.9%) | 0.8685 |
| Artificial airway | 0.0663 | ||||||
| Oral intubation | 95 (75.4%) | 244 (83.3%) | 234 (81.2%) | 222 (83.8%) | 226 (83.4%) | 132 (83.5%) | |
| Nasal intubation | 0 | 2 (0.7%) | 4 (1.4%) | 4 (1.6%) | 6 (2.2%) | 2 (1.3%) | |
| Tracheostomy | 34 (26.4%) | 47 (16%) | 50 (17.4%) | 39 (14.7%) | 39 (14.4%) | 24 (15.2%) | |
| Cuff | 0.0318 | ||||||
| Polyvinyl | 74 (64.3%) | 166 (56.6%) | 170 (59%) | 143 (54%) | 158 (58.3%) | 110 (69.6%) | |
| Polyurethane | 41 (36.7%) | 127 (43.3%) | 118 (41%) | 122 (46%) | 113 (41.7%) | 48 (30.4%) | |
| Cuff pressure | < 0.0001 | ||||||
| Not measured | 9 (7%) | 72 (24.6%) | 2 (0.7%) | 10 (3.8%) | 4 (1.5%) | 6 (3.8%) | |
| < 20 cm H2O | 25 (19.4%) | 18 (6.1%) | 9 (3.1%) | 9 (3.4%) | 2 (0.7%) | 7 (4.4%) | |
| 20–30 cm H2O | 91 (70.5%) | 196 (66.9%) | 263 (91%) | 228 (86%) | 251 (92.6%) | 137 (86.7%) | |
| > 30 cm H2O | 4 (3.1%) | 6 (2%) | 10 (3.5%) | 15 (5.7%) | 10 (3.7%) | 5 (3.2%) | |
| Not inflated | 0 | 1 (0.3%) | 4 (1.4%) | 3 (1.1%) | (1.5%) | 3 (1.9%) | |
| Suctioning system | < 0.0001 | ||||||
| Opened | 76 (62.8%) | 236 (80.5%) | 204 (70.8%) | 171 (64.5%) | 205 (75.6%) | 120 (79.7%) | |
| Closed | 45 (37.2%) | 57 (19.5%) | 84 (29.2%) | 94 (35.5%) | 66 (24.4%) | 38 (20.3%) | |
| Subglottic suctioning | < 0.0001 | ||||||
| Yes | 7 (5.8%) | 67 (22.9%) | 94 (32.6%) | 60 (22.6%) | 70 (25.8%) | 39 (24.7%) | |
| No | 114 (94.2%) | 226 (77.1%) | 194 (69.2%) | 205 (77.4%) | 201 (74.2%) | 119 (75.3%) | |
| Head position | < 0.0001 | ||||||
| < 30° | 55 (44.4%) | 43 (14.7%) | 21 (7.3%) | 13 (4.9%) | 25 (9.2%) | 18 (11.4%) | |
| > 30° | 67 (54.0%) | 249 (85%) | 262 (91%) | 249 (94%) | 245 (90.4%) | 140 (88.6%) | |
| Prone position | 2 (1.6%) | 1 (0.3%) | 5 (1.7%) | 3 (1.1%) | 1 (3.7%) | 0 (0%) |
NIV Noninvasive ventilation
Table 3.
Oral care
| Oral disinfection | 2010 | 2011 | 2013 | 2014 | 2015 | 2016 | p value |
|---|---|---|---|---|---|---|---|
| Water | 1 (1%) | 6 (2%) | 1 (0.3%) | 0 (0%) | 0 (0%) | 0 (0%) | |
| CHXD 0.2% | 48 (38.4%) | 126 (43%) | 108 (37.5%) | 132 (49.8%) | 90 (33.2%) | 79 (50%) | < 0.0001 |
| CHXD 0.5% | 13 (10.4%) | 5 (1.7%) | 11 (3.8%) | 7 (2.6%) | 4 (1.5%) | 5 (3.2%) | |
| CHXD 1% | 0 (0%) | 2 (0.7%) | 26 (9%) | 7 (2.6%) | 16 (5.9%) | 3 (1.9%) | |
| CHXD 2% | 0 (0%) | 0 (0%) | 10 (3.5%) | 9 (3.4%) | 15 (5.5%) | 7 (4.4%) | |
| Polyvidone iodine | 44 (35.2%) | 119 (40.6%) | 102 (35.4%) | 93 (35.1%) | 124 (45.8%) | 55 (34.8%) | |
| Other | 19 (15.2%) | 35 (11.9%) | 30 (10.4%) | 17 (6.4%) | 22 (8.11%) | 10 (6.3%) | |
| Rate of disinfection | |||||||
| 1/day | 2 (1.6%) | 31 (10.6%) | 3 (1%) | 9 (3.4%) | 2 (0.7%) | 0 (0%) | < 0.0001 |
| 2/day | 24 (19.2%) | 41 (14%) | 24 (8.3%) | 27 (10.2%) | 22 (8.1%) | 8 (5.1%) | |
| 3/day | 36 (28.8%) | 117 (39.9%) | 158 (54.9%) | 136 (51.3%) | 116 (42.8%) | 75 (47.4%) | |
| > 3/day | 63 (50.4%) | 104 (35.5%) | 103 (35.8%) | 93 (35.1%) | 131 (48.3%) | 75 (47.4%) | |
| Dental brushing | |||||||
| 0/day | NK | 63 (21.5%) | 51 (18.7%) | 32 (12.9%) | 36 (13.3%) | 27 (17.1%) | 0.0235 |
| 1/day | NK | 99 (33.8%) | 103 (35.8%) | 88 (33.3%) | 83 (30.6%) | 63 (39.9%) | |
| 2/day | NK | 70 (23.9%) | 63 (21.9%) | 81 (38.6%) | 84 (31%) | 51 (32.3%) | |
| 2/day | NK | 46 (15.7%) | 47 (16.3%) | 45 (17%) | 53 (19.6%) | 11 (7%) | |
| Not applicable | NK | 15 (5.1%) | 25 (8.7%) | 19 (7.2%) | 15 (5.5%) | 6 (3.8%) |
CHXD Chlorhexidine, NK: not known
Regarding the prevalence of VAP, it significantly decreased from 28% of ventilated patients in 2010 to 10.1% in 2016 as shown in Fig. 2 (p ≤ 0.0001). Interestingly, the associated bacteremia also decreased in absolute terms (from 7 to 2) but not relatively (from 8.9 to 12.5% of the corresponding VAP, p = 0.9625).
Fig. 2.
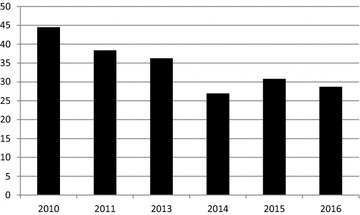
Percentage of ventilated patients treated for VAP the day of the survey
Discussion
This paper reports on the implementation of a VAP bundle in Belgium. It was indeed expected in 2012 by the federal authorities to at least halve the VAP incidence encountered in Belgium. According to Fig. 2, this seems to have been reached and even exceeded, since the prevalence of VAP, as estimated by the surveys, was as high as 28% of the ventilated patients in 2010, reached 11.3% in 2014 and was maintained at this level for the next 2 years. It does not correspond to prevalence levels reported in the USA, but it corresponds to the best levels reported in European centers where VAP bundles are implemented [3, 4].
There is no clear consensus about what a VAP bundle should be [9, 10]. The first VAP bundle from the IHI includes elements which were not directly linked to the management of the airway (prevention of both thromboembolism and digestive hemorrhage) [11]. European experts proposed in 2010 only two procedures in addition to general measures such as hand hygiene, staff education and nonventilatory circuit change: control of sedation with protocol of weaning and oral care with chlorhexidine [12]. The Belgian VAP bundle [8] includes these two procedures into 4 components (sedation protocol, control of cuff pressure, oral disinfection, head of bed elevation) which were also parts of bundles recently published with reported efficacy [3, 4]. It also promotes a fifth procedure: subglottic suctioning which seems to have become the most useful procedure for the prevention of VAP [13–15].
The most striking difference between surveys appeared to be the percentage of ventilated patients and the duration of ventilation from the ICU admission till the day of the survey. Although this latter parameter was not the true duration of ventilation, it reflects a reality which means that from year to year, at one point in the year, fewer patients were ventilated and especially for less time in Belgian ICUs. Of course, this reduction of duration of ventilation reduces the risk for occurrence of VAP, estimated by these 1-day prevalence surveys. It would have been worth correlating this reduction of ventilation to a change in sedation procedures, which, unfortunately, were not directly assessed by the surveys. It may however be inferred that the campaign had an impact because of the clear difference between the duration before and after it.
After the campaign, the compliance with head of bed elevation and control of cuff pressure increased significantly and exceeded 90%. Oral disinfection also improved. The type of antiseptic varied largely, but the rate of application increased: It was done at least 3 times a day in 79.2% in 2010 and in 94.8% in 2016 (p < 0.0001). Chlorhexidine concentration of 1 or 2% was more often used after the campaign, but the low concentration of 0.2% was still used in 50% in 2016. The type and the concentration of antiseptic remain a matter of debate. Chlorhexidine has been shown as effective with concentration as high as 2% in a well-conducted multicenter study in the Netherlands by Koeman in 2006 [16]. Chlorhexidine has been, however, reported to be sometimes not well tolerated by the patients [17]. More worrying is that an increase in mortality in patients receiving chlorhexidine as part of oral disinfection was recently reported by Komplas [18] although this agent is used worldwide [19]. The merit of polyvidone iodine in oral disinfection is supported by very few studies [19], although it remains used in Belgium in one-third of the patients. Regarding dental brushing, it is surprising that as many as 17.1% of patients did not benefit from this care in 2016. They were already 21.5% in 2010, though dental brushing is the only way to eliminate or reduce the dental plaque which can contain a lot of pathogenic bacteria [20]. However, even if dental brushing has been shown to reduce the rate of pneumonia in postoperative patients [21], this was curiously not yet confirmed for VAP in ventilated ICU patients [22]. Subglottic suctioning, which was promoted, remains used in Belgium in a minority of patients. This could be due to the cost of the endotracheal tube which is on average 10 times higher than conventional tubes in Belgium. However, this procedure should still be encouraged because of its efficacy reported as high as 50% reduction of VAP incidence [15].
Thus, the VAP bundle was rather correctly followed and VAP incidence decreased. Was there a clear causal relationship between these two facts? These surveys cannot ascertain that statement, but they were carried out to control the expected impact on VAP prevalence. But the question remains of the reality of this impact, especially because the effect was seen only in 2014, 2015 and 2016, while the campaign was conducted in 2012. All the improvements seen in the application of VAP bundle were, however, already obtained in 2013. Why did the reduction in VAP prevalence not occur in 2013? In fact, this question is not anecdotic, because there may have been a change in the way the VAP was diagnosed and the reduction seen in 2013 onward could be due to this change. As said by Komplas, “an apparent decrease in VAP rate could be achieved by maximally exploiting the subjectivity and inconstancies of VAP definitions” [23]. In our opinion, it was not the case, because the way of diagnosing VAP remains the same, based on radiological findings, the occurrence of fever, change in leukocytosis and bacteriological results. The surveys were answered on a voluntary basis, and there were no reasons to minimize the VAP prevalence at any time. Most medical teams were the same during the surveys. An indirect evidence of the reality in the reduction of VAP prevalence was the corresponding decrease in associated bacteremia, the rate relating to the number of VAP being stable. That means that the same type of infections was taken into account during the surveys. However, Komplas’ concern regarding the data manipulation, even if unconscious, may still be real.
This report is not a true study such as the one recently published about the Spanish experience [24]. It is only a presentation of several surveys supporting the implementation of a VAP bundle in ICU ventilated patients. It gives figures from a large number of ICUs, allowing to describe the average activity of intensive care in Belgium and to encourage Belgian teams to prevent the occurrence of VAP by all valuable means. It is indeed interesting to observe the steady decline in number of ventilated patients in Belgian ICUs and duration of ventilation over time. Data quality may be, however, questioned because data could not be controlled, but remained consistent over time.
Conclusion
The occurrence of VAP was a real issue in 2010 in Belgium. The efforts made by the medical and nurses teams of the different ICUs seem to have successfully contributed to the decrease in VAP prevalence which has now reached a low plateau for several years. However, there could still be room for further improvement.
Authors’ contributions
All authors designed the study, contributed to the interpretation of the results and critically revised the manuscript. All authors made a substantial contributions to the acquisition of the data and revised the manuscript for important intellectual content. LJ and PD made a substantial contribution in writing the manuscript and interpreting the results. PD made a substantial contribution in the study design and critically revised the manuscript for important intellectual content. All authors had full access to the data, take responsibility for the integrity of the data and the accuracy of the analysis. All authors read and approved the final manuscript.
Acknowledgements
None.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
The ethics approval was waived because this survey was observational and organized by the Belgian legal authorities.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- VAP
ventilator-associated pneumonia
- ICU
intensive care unit
- IQR
interquartile range
- SD
standard deviation
- COPD
chronic obstruction pulmonary disease
- NA
not available
- NIV
noninvasive ventilation
- CHXD
chlorhexidine
- NK
not known
Contributor Information
Laurent Jadot, Email: loul7@icloud.com.
Luc Huyghens, Email: luc.huyghens@azbrussel.be.
Annick De Jaeger, Email: annick.dejaeger@ugent.be.
Marc Bourgeois, Email: marc.bourgeois@azsintjan.be.
Dominique Biarent, Email: dominique.biarent@huderf.be.
Adeline Higuet, Email: a.higuet@sintmaria.be.
Koen de Decker, Email: koen.de.Decker@olvz-aalst.be.
Margot Vander Laenen, Email: margot.vanderlaenen@zol.be.
Baudewijn Oosterlynck, Email: baudewijn.oosterlynck@azsintjan.be.
Patrick Ferdinande, Email: patrick.ferdinande@uz.kuleuven.be.
Pascal Reper, Email: pascal.reper@sante.belgique.be.
Serge Brimioulle, Email: serge.brimioulle@ulb.ac.be.
Sophie Van Cromphaut, Email: Sophie.VanCromphaut@uza.be.
Stéphane Clement De Clety, Email: stephan.clementdeclety@uclouvain.be.
Thierry Sottiaux, Email: thierry.sottiaux@cndg.be.
Pierre Damas, Phone: 32-4-366.74.95, Email: pdamas@chuliege.be.
References
- 1.Bekaert M, Timsit JF, Vansteelandt S, et al. Outcomerea Study Group. Attributable mortality of ventilator-associated pneumonia: a reappraisal using causal analysis. Am J Resp Crit Care Med. 2011;184:1133–1139. doi: 10.1164/rccm.201105-0867OC. [DOI] [PubMed] [Google Scholar]
- 2.Hutchins K, Karras G, Erwin J, Sullivan K. Ventilator-associated pneumonia and oral care: a successful quality improvement project. Am J Infect Control. 2009;37:590–597. doi: 10.1016/j.ajic.2008.12.007. [DOI] [PubMed] [Google Scholar]
- 3.Morris AC, Hay AW, Swann DG, et al. Reducing ventilator-associated pneumonia in intensive care: impact of implementing a care bundle. Crit Care Med. 2011;39:2218–2224. doi: 10.1097/CCM.0b013e3182227d52. [DOI] [PubMed] [Google Scholar]
- 4.Bouadma L, Mourvillier B, Deiler V, et al. A multifaceted program to prevent ventilator-associated pneumonia: impact on compliance with preventive measures. Crit Care Med. 2010;38:789–796. doi: 10.1097/CCM.0b013e3181ce21af. [DOI] [PubMed] [Google Scholar]
- 5.Torres A, Niederman M, Chastre J, et al. International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European Respiratory Society (ERS), European Society of Intensive Care Medicine (ESICM), European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and Asociación Latinoamericana del Tórax (ALAT) Eur Respir J. 2017;50:1700582. doi: 10.1183/13993003.00582-2017. [DOI] [PubMed] [Google Scholar]
- 6.SFAR. http://sfar.org/pneumonies-associees-aux-soins-de-reanimation/.
- 7.SRLF. https://www.srlf.org/wp-content/uploads/2017/09/2017_09_RFE-commune_PNEUMONIES-ASSOCIEES-AUX-SOINS-DE-REANIMATION.pdf.
- 8.Reper P, Dicker D, Damas P, et al. Improving the quality of the intensive care follow-up of ventilated patients during a national registration program. Public Health. 2017;148:159–166. doi: 10.1016/j.puhe.2017.03.014. [DOI] [PubMed] [Google Scholar]
- 9.Klompas M. Ventilator-associated pneumonia: Is zero possible? Clin Inf Dis. 2010;51:1123–1126. doi: 10.1086/656738. [DOI] [PubMed] [Google Scholar]
- 10.O’Grady N, Murray PR, Ames N. Preventing ventilator-associated pneumonia. Does the evidence support the practice? JAMA. 2012;307:2534–2539. doi: 10.1001/jama.2012.6445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Resar R, Pronovost P, Haraden C, et al. Using a bundle approach to improve ventilator care processes and reduce ventilator-associated pneumonia. Jt Comm J Qual Patient Saf. 2005;31:243–248. doi: 10.1016/S1553-7250(05)31031-2. [DOI] [PubMed] [Google Scholar]
- 12.Rello J, Lode H, Cornaglia G, et al. The VAP care bundle contributors. A European care bundle for prevention of ventilator-associated pneumonia. Int Care Med. 2010;36:773–780. doi: 10.1007/s00134-010-1841-5. [DOI] [PubMed] [Google Scholar]
- 13.Lacherade JC, De Jonghe B, Guezennec P, et al. Intermittent subglottic secretion drainage and ventilator-associated pneumonia. Am J Resp Crit Care Med. 2010;182:910–917. doi: 10.1164/rccm.200906-0838OC. [DOI] [PubMed] [Google Scholar]
- 14.Damas P, Frippiat F, Ancion A, et al. Prevention of ventilator-associated pneumonia and ventilator-associated conditions: a randomized controlled trial with subglottic secretion suctioning. Crit Care Med. 2015;43:22–30. doi: 10.1097/CCM.0000000000000674. [DOI] [PubMed] [Google Scholar]
- 15.Caroff D, Li L, Muscedere J, Klompas M. Subglottic secretion drainage and objective outcomes: a systematic review and meta-analysis. Crit Care Med. 2016;44:830–840. doi: 10.1097/CCM.0000000000001414. [DOI] [PubMed] [Google Scholar]
- 16.Koeman M, Van der Ven A, Hak E, et al. Oral decontamination with chlorhexidine reduces the incidence of ventilator-associated pneumonia. Am J Resp Crit Care Med. 2006;173:1348–1355. doi: 10.1164/rccm.200505-820OC. [DOI] [PubMed] [Google Scholar]
- 17.Richards D. Chlorhexidine mouthwash plaque levels and gingival health. Evid Based Dent. 2017;18:37–38. doi: 10.1038/sj.ebd.6401232. [DOI] [PubMed] [Google Scholar]
- 18.Klompas M, Li L, Kleinman K, et al. Associations between ventilator bundle components and outcome JAMA. Intern Med. 2016;176:1277–1283. doi: 10.1001/jamainternmed.2016.2427. [DOI] [PubMed] [Google Scholar]
- 19.Shi Z, Xie H, Wang P, et al. Oral hygiene care for critically ill patients to prevent ventilator-associated pneumonia. Cochrane Database Syst Rev. 2013 doi: 10.1002/14651858.CD008367.pub2. [DOI] [PubMed] [Google Scholar]
- 20.Fourrier F, Cau-Pottier E, Boutigny H, et al. Effect of dental plaque antiseptic decontamination on bacterial colonization and nosocomial infection in critically ill patients. Int Care Med. 2000;26:1239–1247. doi: 10.1007/s001340000585. [DOI] [PubMed] [Google Scholar]
- 21.Akutsu Y, Matsubara H, Shuto K, et al. Preoperative dental brushing can reduce the risk of postoperative pneumonia in esophageal cancer patients. Surgery. 2010;147:497–502. doi: 10.1016/j.surg.2009.10.048. [DOI] [PubMed] [Google Scholar]
- 22.Gu WJ, Gong YZ, Pan L, et al. Impact of oral care with versus without toothbrushing on the prevention of ventilator-associated pneumonia: a systematic review and meta-analysis of randomized controlled trials. Crit Care. 2012;16:R90. doi: 10.1186/cc11675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Klompas M. What is new in the prevention of nosocomial pneumonia in the ICU? Curr Opin Crit Care. 2017;23:378–384. doi: 10.1097/MCC.0000000000000443. [DOI] [PubMed] [Google Scholar]
- 24.Alvarez-Lerma F, Palomar-Martinez M, Sanchez-Garcia M, et al. Prevention of ventilator-associated pneumonia: the multimodal approach of the Spanish ICU Pneumonia Zero program. Crit Care Med. 2018;46:181–188. doi: 10.1097/CCM.0000000000002736. [DOI] [PMC free article] [PubMed] [Google Scholar]


