Abstract
Background & objectives:
The determination of cystatin C (cysC) may be helpful in diagnosis and monitoring of cancer because the pathogenesis of cancer is linked with an increased activity of cysteine peptidases (cathepsins) and a decrease of cysC concentration. This study was aimed to examine the utility of cysC as a marker of bladder cancer (BCa) to be used in the diagnosis.
Methods:
This study was conducted with 90 patients with BCa and 27 healthy people. Patients with other cancers, inflammation process and impaired renal function were excluded from the study. The concentrations of cysC in the plasma and urine were measured by surface plasmon resonance imaging technique.
Results:
The concentration of cysC in the serum taken from the patients with BCa [0.35±0.02 μg/ml (range: 0.20-0.78 μg/ml)] was significantly (P <0.001) lower than the serum cysC concentration of the healthy people [0.68±0.05 μg/ml (range: 0.52-0.89 μg/ml)]. The urinary cysC concentration of the BCa patients [0.19±0.01 μg/ml (range: 0.09-0.34 μg/ml)] was not significantly different from the urinary cysC concentration of the healthy people [0.24±0.02 μg/ml (range: 0.16-0.33 μg/ml)]. Receiver operating characteristic (ROC) curve showed that BCa patients with cysC concentration <0.54 μg/ml [sensitivity: 87%; specificity: 92%; area under the curve (AUC) of ROC: 0.927; P =0.02] could be optimally separated from healthy people. The ROC curve further showed that superficial low-grade patients with cysC concentration lower than 0.36 μg/ml (sensitivity: 0.63%; specificity: 0.58%; AUC of ROC: 0.635; P =0.08) could not be optimally separated from high-risk tumour patients.
Interpretation & conclusions:
BCa patients have lower serum cysC concentration than the control group. Serum cysC may be considered as a potential marker of BCa but not its aggressiveness.
Keywords: Biomarkers, bladder cancer, cystatin C, surface plasmon resonance imaging
The human cystatin C (cysC) is a small molecule protein (13.3 kDa), which consists of 120 amino acids. Its concentration in body fluids does not depend on gender, age or muscle mass. It only depends on the glomerular filtration rate, which makes it a good indicator of urinary tract function. CysC is also known to inhibit and regulate cathepsins activity. Thus, it plays a protective role in inflammation and pathogenesis of cancers by acting as an inhibitor of cathepsins (cysteine proteases)1. The reference value in human serum is 0.57-1.79 μg/ml and in urine 0.03-0.29 μg/ml1,2.
Surface plasmon resonance imaging (SPRI), a label-free technique based on the resonance of plasmons, is used to measure the cysC concentration in the samples. The surface of the metal (gold) is coated with cysteamine and papain. Binding of the cysC with the papain causes changes in the resonance condition, which can be observed as changes in the reflectivity. This technique has already been validated and can be used to measure cysC concentration in biological fluids3.
Bladder cancer (BCa) is the most common urinary tract cancer4,5,6. The treatment of this cancer depends on the stage of the cancer at the time of diagnosis, aggressiveness and degree of differentiation7. The search for new BCa markers is important for early diagnosis and prognosis. Because cysC plays an important role in the pathogenesis of the cancer, it may serve as such a marker. This study was aimed to determine the utility of cysC as a potential BCa biomarker for diagnosis and determination of the aggressiveness of the disease.
Material & Methods
The study included 90 patients with histological confirmation of BCa. Consecutive patients who qualified for transurethral resection of bladder tumour or cystectomy, and were without other recognized tumours or renal insufficiency and who gave consent were included in the study.
Urine and serum samples were taken from patients with BCa during the hospitalization in Urology department of J. Sniadecki Provincial Hospital in Bialystok (Bialystok, Poland) for transurethral resection of the BCa or after cystectomy. The blood samples were collected from the median cubital vein. The blood and urine samples were also taken from 27 healthy people from the Blood Donor Centre of Bialystok, Poland. The age range of the study population was 19-45 years.
Two factors were taken into account: The aggressiveness of the cancer (grading) and the presence of particular clinical parameters. The high-risk patients were characterized by high-grade tumour and the presence of three of the four clinical parameters (size >30 mm, nonpapillary, multifocal, recurrent).
After collection, the serum and urine samples were kept at −70°C. Patients with the other cancer diseases, inflammation process and impaired renal function were excluded from the study. The urine samples were centrifuged at 1850×g for 15 min, and the supernatants were separated. Subsequently, the samples were once filtered through some medium density paper filters before analysis.
The study was approved by the Bioethics Committee of Medical University of Bialystok (R-I 002/409/2014, dated October 30, 2014). The study was conducted between October 2014 and May 2015. Written informed consent was obtained from each patient.
Procedure of cystatin C (cysC) determination: The cysC concentration in the serum and urine samples was measured by the SPRI biosensors (by the SPRI apparatus). The SPRI technique uses a very specific interaction between enzymes and their inhibitors, what enables determination of the tested proteins. All of the necessary steps in the preparation and optimization of the biosensor have been described elsewhere3. In this study, papain (Sigma, Steinheim, Germany) was used as an inhibitor and was immobilized on the biosensor surface. The serum samples taken from both the patients with BCa and the healthy people were two-fold diluted with phosphate-buffered saline (pH=7.4, BIOMED-LUBLIN, Poland). The volume of each sample used to the SPRI measurements was 2 μl. After 10 min of the interaction between cysC from the samples and the papain, the biosensor was washed with water and Hepes-buffered saline-ES buffer solution at pH=7.4 [0.01 M 4-(2-hydroxyethyl) piperazine-1-ethanesulfonic acid, 0.15 M sodium chloride, 0.005% Tween 20, 3 mM EDTA, BIOMED-LUBLIN] to remove other unbound molecules. To control the level of non-specific binding, some of the biosensor sites were covered with buffer. The SPRI signal was measured twice, before the interaction of the cysC with the papain and after this interaction, and was proportional to the amount of the cysC bound to the biosensor surface. The difference between the signals before and after the cysC -papain interaction was used to determine the cysC concentration in the sample. The concentration of serum and urinary cysC was calculated using a previously plotted calibration curve.
Statistical analysis: To compare the obtained data, Student's t test was used. The receiver operating characteristic (ROC) curves were drawn using MedRoc software version 2.0 (StenStat, USA).
Results
Changes in cystatin C (cysC) concentration: SerumcysC concentration in the samples taken from patients with BCa [0.35±0.02 μg/ml (range: 0.20-0.78 μg/ml)] was significantly (P<0.001) lower than serum cysC concentration in the samples taken from healthy people [0.68±0.05 μg/ml, (range: 0.52-0.89 μg/ml)]. The urinary cysC level in the BCa patients was 0.19±0.01 μg/ml (range: 0.09-0.34 μg/ml), which was not significantly different from the urinary cysC level in the healthy people [0.24±0.02 μg/ml (range: 0.16-0.33 μg/ml)]. There was a difference between the urine cysC concentrations in the samples from BCa patients and healthy people, but these results are omitted in the further consideration.
Blood serum analysis: The following parameters were significantly related to a higher cysC concentration: Age <65 yr, male, non-muscle invasive, low-grade and fossil tumours (Table). This indicated that cysC could be used to distinguish healthy individuals from BCa patients by measuring the cysC concentration and comparing it with the listed parameters. On the other hand, the presence of inflammation, multiplicity and recurrence rate were not significant. For this reason, it was not possible to diagnose BCa using cysC concentration alone.
Table.
Diagnostic characteristic of serum cystatin C and cystatin C/mass of a protein ratio compared with various demographic and clinical parameters of bladder cancer

Receiver operating characteristic (ROC) analysis: To determine the diagnostic utility of cysC in BCa, the ROC curve was plotted (Fig. 1). The area under the curve (AUC) was 0.927 (P=0.02). The optimum cut-off was 0.54 μg/ml, which indicated that people with serum cysC concentration lower than that level might have BCa (sensitivity, 87%; specificity, 92%). From the ROC curve (Fig. 2), it was not possible to separate the patients with an aggressive form of BCa from those with non-aggressive BCa (sensitivity, 0.63%; specificity, 0.58%; AUC of ROC: 0.635; P=0.08).
Fig. 1.

Diagnostic efficiency of serum cystatin C concentration in detection of bladder cancer determined by receiver operating characteristic curve.
Figure 2.
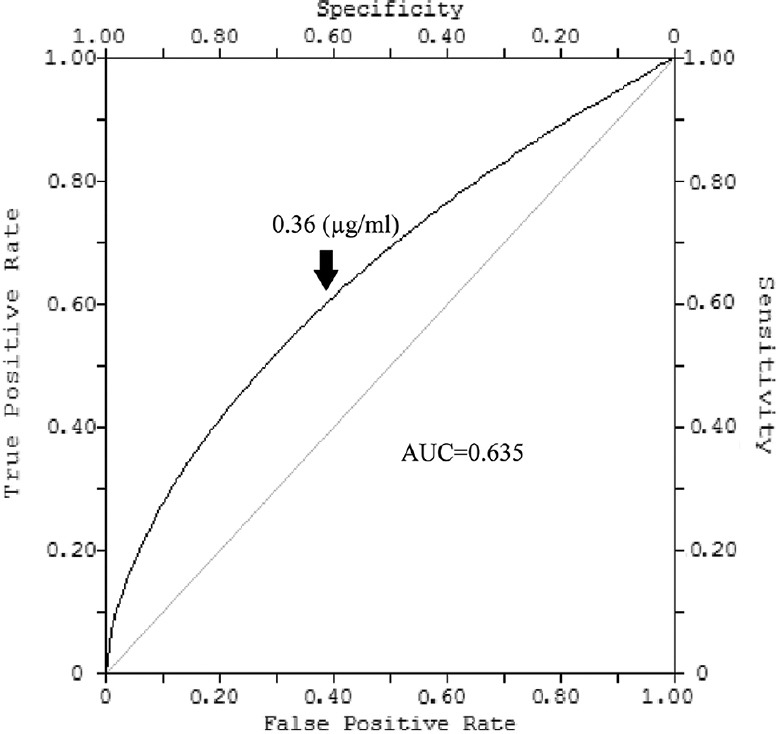
Diagnostic efficiency of serum cystatin C concentration in prediction of superficial transitional cell carcinomas aggressiveness determined by receiver operating characteristic curve.
Discussion
The search for new malignancy markers is important in clinical oncology. CysC is a cysteine protease inhibitor, which plays an important role in the pathogenesis of cancer. This study was designed to explore the utility of the serum cysC level as a biomarker of BCa.
The level of serum cysC in the samples form BCa patients was found to be lower than in healthy individuals. This indicates that cysC may be a potential tumour maker for urinary BCa. CysC expression was found to be declined in glioblastoma8, breast cancer9, colorectal cancer10 and adenocarcinoma of prostate, where this was correlated with poorer diagnosis11. The results obtained in this study were in agreement with the studies which indicated that probably a low level of cysC contributed to the pathogenesis of urinary BCa. However, the role of cysC in pathogenesis of cancer has remained controversial as the patients with non-Hodgkin B cell lymphoma12, multiple myeloma13 and liver carcinoma14 tend to have higher cysC concentration than healthy individuals. Moreover, higher cysC concentration in colorectal15, lung16 and melanoma carcinomas17 was related with the poor outcome.
The ROC curve analysis showed that a cut-off value of 0.54 μg/ml could be used to differentiate BCa patients from a healthy person. The patients with cysC concentration higher than 0.54 (μg/ml) can be regarded as the healthy.
In conclusion, our results showed lower level of cysC in patients with BCa compared to healthy individuals. The results obtained by surface plasmon resonance technique need to be validated by a conventionally acceptable method. The findings need to be confirmed in a larger sample.
Footnotes
Financial support & sponsorship: None
Conflicts of Interest: None.
References
- 1.Onopiuk A, Tokarzewicz A, Gorodkiewicz E. Cystatin C: A kidney function biomarker. Adv Clin Chem. 2015;68:57–69. doi: 10.1016/bs.acc.2014.11.007. [DOI] [PubMed] [Google Scholar]
- 2.Warwas M, Piwowar A. Urinary cystatin C as a biomarker of renal tubular injury. Adv Hyg Exp Med. 2011;65:562–8. doi: 10.5604/17322693.957690. [DOI] [PubMed] [Google Scholar]
- 3.Gorodkiewicz E, Luszczyn J. Surface plasmon resonance imaging (SPRI) sensor for cystatin determination based on immobilized papain. Protein Pept Lett. 2011;18:23–9. doi: 10.2174/092986611794328663. [DOI] [PubMed] [Google Scholar]
- 4.Facio FN, Jr, Facio MFW, Spessoto LCF, Gatti M, Ferraz Arruda PF, Ferraz Arruda JG. Incidental diagnosis of bladder cancer in a 17-year-old patient. Urol Case Rep. 2015;3:96–7. doi: 10.1016/j.eucr.2015.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Azevedo R, Ferreira JA, Peixoto A, Neves M, Sousa N, Lima A, et al. Emerging antibody-based therapeutic strategies for bladder cancer: A systematic review. J Control Release. 2015;214:40–61. doi: 10.1016/j.jconrel.2015.07.002. [DOI] [PubMed] [Google Scholar]
- 6.Conde VR, Oliveira PF, Nunes AR, Rocha CS, Ramalhosa E, Pereira JA, et al. The progression from a lower to a higher invasive stage of bladder cancer is associated with severe alterations in glucose and pyruvate metabolism. Exp Cell Res. 2015;335:91–8. doi: 10.1016/j.yexcr.2015.04.007. [DOI] [PubMed] [Google Scholar]
- 7.Arcangeli G, Arcangeli S, Strigari L. A systematic review and meta-analysis of clinical trials of bladder-sparing trimodality treatment for muscle-invasive bladder cancer (MIBC) Crit Rev Oncol Hematol. 2015;94:105–15. doi: 10.1016/j.critrevonc.2014.11.007. [DOI] [PubMed] [Google Scholar]
- 8.Konduri SD, Yanamandra N, Siddique K, Joseph A, Dinh DH, Olivero WC, et al. Modulation of cystatin C expression impairs the invasive and tumorigenic potential of human glioblastoma cells. Oncogene. 2002;21:8705–12. doi: 10.1038/sj.onc.1205949. [DOI] [PubMed] [Google Scholar]
- 9.Yano M, Hirai K, Naito Z, Yokoyama M, Ishiwata T, Shiraki Y, et al. Expression of cathepsin B and cystatin C in human breast cancer. Surg Today. 2001;31:385–9. doi: 10.1007/s005950170126. [DOI] [PubMed] [Google Scholar]
- 10.Hirai K, Yokoyama M, Asano G, Tanaka S. Expression of cathepsin B and cystatin C in human colorectal cancer. Hum Pathol. 1999;30:680–6. doi: 10.1016/s0046-8177(99)90094-1. [DOI] [PubMed] [Google Scholar]
- 11.Wegiel B, Jiborn T, Abrahamson M, Helczynski L, Otterbein L, Persson JL, et al. Cystatin C is downregulated in prostate cancer and modulates invasion of prostate cancer cells via MAPK/Erk and androgen receptor pathways. PLoS One. 2009;4:e7953. doi: 10.1371/journal.pone.0007953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mulaomerović A, Halilbasić A, Cickusić E, Zavasnik-Bergant T, Begić L, Kos J, et al. Cystatin C as a potential marker for relapse in patients with non-Hodgkin B-cell lymphoma. Cancer Lett. 2007;248:192–7. doi: 10.1016/j.canlet.2006.07.004. [DOI] [PubMed] [Google Scholar]
- 13.Nückel H, Langer C, Herget-Rosenthal S, Wichert M, Assert R, Döhner H, et al. Prognostic significance of serum cystatin C in multiple myeloma. Int J Hematol. 2012;95:545–50. doi: 10.1007/s12185-012-1049-2. [DOI] [PubMed] [Google Scholar]
- 14.He H, Lü RX, Xu J, Liang LB, Zhang MF, An ZM, et al. Preliminary study on the expression of cystatin C in primary liver cancer. Sichuan Da Xue Xue Bao Yi Xue Ban. 2013;44:920–3. [PubMed] [Google Scholar]
- 15.Kos J, Krasovec M, Cimerman N, Nielsen HJ, Christensen IJ, Brünner N, et al. Cysteine proteinase inhibitors stefin A, stefin B, and cystatin C in sera from patients with colorectal cancer: Relation to prognosis. Clin Cancer Res. 2000;6:505–11. [PubMed] [Google Scholar]
- 16.Zhang X, Hou Y, Niu Z, Li W, Meng X, Zhang N, et al. Clinical significance of detection of cathepsin X and cystatin C in the sera of patients with lung cancer. Zhongguo Fei Ai Za Zhi. 2013;16:411–6. doi: 10.3779/j.issn.1009-3419.2013.08.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dikovskaya MA, Trunov AN, Chernykh VV, Korolenko TA. Cystatin C and lactoferrin concentrations in biological fluids as possible prognostic factors in eye tumor development. Int J Circumpolar Health. 2013;72 doi: 10.3402/ijch.v72i0.21087. [DOI] [PMC free article] [PubMed] [Google Scholar]


