Abstract
Purpose
This study sought to determine the cortical associates of emotional reactivity and emotion regulation (as indexed by the amplitude of evoked response potentials [ERP]) in young children who do and do not stutter during passive viewing of pleasant, unpleasant and neutral pictures.
Method
Participants were 17 young children who stutter and 22 young children who do not stutter (between 4 years 0 months to 6 years 11 months). The dependent measures were (1) mean amplitude of late positive potential (LPP, an ERP sensitive to emotional stimuli) during passive (i.e., no response required) picture viewing and directed reappraisal tasks and (2) emotional reactivity and regulation related scores on caregiver reports of young children’s temperament (Children’s Behavior Questionnaire, CBQ).
Results
Young CWS, when compared to CWNS, exhibited significantly greater LPP amplitudes when viewing unpleasant pictures, but no significant between-group difference when viewing pleasant pictures and during the emotion regulation condition. There were, however, for CWS, but not CWNS, significant correlations between temperament-related measures of emotion and cortical measures of emotional reactivity and regulation.
Conclusions
Findings provide further empirical support for the notion that emotional processes are associated with childhood stuttering, and that CWS’s inherent temperamental proclivities need to be taken into account when empirically studying or theorizing about this association.
1. Introduction
Over the past several years, numerous empirical studies indicated that emotions play a role in early childhood stuttering (e.g., Anderson, Pellowski, Conture, & Kelly, 2003; Arnold, Conture, Key, & Walden, 2011; Choi, Conture, Walden, Jones & Kim, 2016; Eggers, De Nil, & Van den Bergh, 2009, 2010; Embrechts, Ebben, Franke & van de Poel, 2000; Felsenfeld, van Beijsterveldt & Boomsma, 2010; Johnson, Walden, Conture, & Karrass, 2010). Recently, the Dual Diathesis-Stressor Model (Conture &Walden, 2012) and the Multifactorial Dynamic Pathways Theory (Smith & Weber, 2017) suggest that emotions are associated with childhood stuttering. Taken together, the aforementioned empirical studies and theoretical models involve, at the minimum, two aspects of emotion: (1) emotional reactivity, that is, “the ease by which emotions are aroused, which can involve reactions to novel stimuli, and/or orienting to internal or external stimulation” (Rothbart, 2011) and (2) emotional regulation, that is, “the processes by which we influence which emotions we have, when we have them, and how we experience and express them” (Gross, 1998). Although neither the empirical findings nor theoretical models propose that emotion is the main, primary or sole “cause” of childhood stuttering, they do suggest that any comprehensive account of childhood stuttering should include consideration of emotional processes. Below the present authors provide a brief review of essential findings regarding the association between emotion and childhood stuttering. This review is organized around the various methods used to empirically study this association, including (a) caregiver reports, (b) direct behavioral observations and experimental tests, and (c) psychophysiological measures.
First, evidence from caregiver reports suggests that when compared to children who do not stutter (CWNS), CWS display (a) lower inhibitory control, and higher anger/frustration (Eggers et al., 2010), (b) greater emotional reactivity and greater difficulty in emotion regulation (e.g., Karrass, Walden, Conture, Graham, Arnold, Hartfield & Schwenk, 2006), (c) greater difficulty in flexibly controlling and shifting attention when necessary (Eggers et al, 2010; Felsenfeld et al., 2010; Karrass et al., 2006), and (d) less adaptability to change (Anderson et al., 2003). Overall, these findings, based on parental reports to normed questionnaires, suggest that CWS are perceived as differing from their CWNS peers in emotion reactivity and emotion regulation processes.
In contrast, Kefalianos, Onslow, Ukoumunne, Block, and Reilly (2014), in a study of the temperament of a large cohort of CWS and CWNS at 2, 3, and 4 years of age replicated Anderson et al.’s (2003) and Howell et al.’s (Howell, Davis, Patel, Cuniffe, Downing-Wilson et al. 2004) findings of no significant talker-group differences in approach/withdrawal scores. Lewis and Goldberg (1997) reported that young CWS, when compared to CWNS, were less negative and more adaptable (cf. Anderson et al., 2003) and Williams (2006) reported that CWS were more likely to exhibit the temperamental constellation of an “easy child.” Interestingly, however, Arnold et al. (2011) observed that in the Williams’s study “…a higher proportion of CWS, compared to CWNS, fit the temperamental constellation of “slow to warm up” (p. 277). Perhaps, some of the differences in findings regarding caregiver reports of temperament in children who do and do not stutter relates to the use of different temperament scales. Indeed, these scales vary significantly in content, length, as well as breadth, depth and types of temperamental dimensions measured (e.g., the 30-item Short Temperament Scale for Children used in Kefalianos et al. (2014) study versus the 233-item Children’s Behavior Questionnaire used in Eggers et al. (2010) study).
Second, researchers have used direct behavioral observations and experimental tests to study more variable/state-like/situational aspects of emotion and childhood stuttering. Findings from such studies indicate that when compared to their CWNS peers, CWS exhibit (a) more negative emotional expressions during a disappointing gift procedure (Johnson et al., 2010), (b) difficulty flexibly shifting attention from a stimulus (Bush, 2006); (c) more negative emotion and self-speech during an emotionally frustrating task (Ntourou, Conture, Walden, 2013), (d) less ability to habituate to task-irrelevant environmental stimuli (Schwenk, Conture, & Walden, 2007), (e) less efficiency in the orienting subsystem of the attention network during a computerized attention network task (Eggers, De Nil & Van den Bergh, 2012), and (f) lower inhibitory control during a Go/NoGo task (Eggers, De Nil & Van den Bergh, 2013). In addition, emerging findings have shown that children who stutter and persist, compared to children who recover from stuttering and those who do not stutter, exhibit significantly slower articulation rates following a negative emotion condition (Erdemir, Walden, Jefferson, Choi, & Jones, 2018).
Studies investigating the association between CWS’s emotions and their frequency of stuttering have indicated that young CWS exhibit (a) more emotionally reactive behaviors prior to and during stuttered than fluent utterances (Jones, Conture, & Walden, 2014b), (b) increased stuttering frequency in association with decreased duration and frequency of behavioral regulatory strategies (Arnold et al., 2011), and (c) increased stuttering severity in association with caregiver-reports of lower effortful control skills (Kraft, Ambrose, Chon, 2014). Likewise, Choi, Conture, Walden, Lambert and Tumanova (2013) studied young CWS and CWNS in terms of possible behavioral inhibition (BI). BI is a temperamentally related attribute characterized by initial avoidance and distress in unfamiliar places, situations or the presence of unfamiliar people (Kagan et al., 1984 in Choi et al., 2013). Choi et al. (2013) reported that CWS with higher BI had more stuttering than CWS with lower BI. Overall, evidence from direct behavioral observations and experimental tests indicates that CWS differ from their CWNS peers in the manifestation of emotion regulation and reactivity and that CWS’s increased stuttering is associated with increased emotional reactivity and decreased regulation.
More recently, researchers have used various psychophysiological methods to study the association between emotional processes and childhood stuttering. To date, psychophysiological findings from these studies indicate that when compared to their CWNS peers, young CWS exhibit (1) no significant differences in EEG frontal asymmetries (Arnold et al, 20111); (2) less emotion regulation (as indexed by parasympathetic activity), during a non-speaking baseline condition, as well as more counter-adaptive responses (as indexed by greater co-activation of both sympathetic and parasympathetic responses) during a speaking condition (Jones, Buhr, Conture, Tumanova, Walden, & Porges, 2014a); (3) significantly lower (Ortega & Ambrose, 2011) or no significant differences in salivary cortisol (van der Merwe, Robb, Lewis & Osmond, 2011), (4) significantly higher emotional reactivity (indexed by tonic skin conductance level) in 3-year-old CWS and significantly lower emotional reactivity in 4-year-old CWS (Zengin-Bolatkale, Conture, Walden, 2015), and (5) significantly higher emotional reactivity (indexed by mean tonic skin conductance level) during narratives following negative and positive, compared to baseline, emotional stress conditions (Choi, Conture, Walden, Jones, & Kim, 2016). Most recently, researchers have reported that decreased emotion regulation (indexed by decreased parasympathetic activity) is significantly associated with increased stuttering frequency, with this relation moderated by executive functions (Jones, Walden, Conture, Erdemir, Lambert, & Porges, 2017). Overall, results from psychophysiological studies appear to suggest that CWS and CWNS differ on some measures of emotional reactivity and emotion regulation processes.
It should be noted, however, that measures of autonomic nervous system activity (e.g., skin conductance level) and hypothalamic-pituitary-adrenal (HPA) hormonal secretions (e.g., salivary cortisol) record physiological events on the order of seconds or minutes. Yet, some key emotion reactivity and regulation processes may occur on the order of milliseconds. Scalp-recorded event related potentials (ERPs) have excellent temporal resolution in the order of milliseconds and may allow researchers to better capture very rapid changes in neural activity related to emotional reactivity and regulation. Findings based on ERP measures, together with those from other methodologies mentioned above, would appear to provide converging lines of evidence that contribute to a more comprehensive understanding of the association between emotion and childhood stuttering.
One ERP that appears particularly well suited for examining emotional processes is the late positive-potential (LPP). The LPP emerges approximately 300 ms following stimulus onset, with larger amplitude following the presentation of both pleasant and unpleasant than neutral stimuli (e.g., Cuthbert, Schupp, Bradley, Birbaumer, & Lang, 2000; Foti & Hajcak, 2008; Hajcak, Nieuwenhuis, 2006; Hajcak & Olvet, 2008; Schupp, Cuthbert, Bradley, Cacioppo, Ito, & Lang, 2000). The LPP has been associated with increased attention to motivationally relevant, emotional stimuli (i.e., emotion automatically directs attention) that facilitates subsequent cognitive processing (Bradley et al., 2003; Hajcak, Dunning, Foti, 2009). Recently, Liu et al. (2012) investigated the neural substrate of LPP by recording EEG and fMRI simultaneously and reported that LPP was generated by an extensive brain network that included structures associated with visual and emotional processing such as amygdala, insula, and prefrontal cortex. Previous findings indicate that LPP is sensitive to emotional stimuli in both adults (e.g., Alomari, Fernandez, Banks, Acosta, Tartar, 2015, and Ding, Li, Wang, Luo, 2017 for a review) and children (e.g., Hajcak & Dennis, 2009). Furthermore, in addition to its use in the study of emotion reactivity, LPP amplitude has been used to study children’s emotion regulation (e.g., Babkirk, Rios, Dennis, 2015; DeCicco, Solomon, Dennis, 2012; Dennis & Hajcak, 2009). Findings of such studies of LPP in relation to emotional reactivity and regulation are relevant to the present empirical investigation because childhood stuttering has been shown to be associated with emotional reactivity (e.g., Choi et al., 2016; Zengin-Bolatkale et al., 2015), and emotion regulation (e.g., Arnold et al., 2011; Kraft et al., 2014; Walden et al., 2012).
Indeed, current theories of child temperament and emotional regulation emphasize the different, but inter-related roles of reactivity and regulation (Derryberry & Rothbart, 1997; Posner & Rothbart, 2000). Furthermore, different emotion regulation strategies may influence the strength of emotional reactivity (Campos, Frankel, Camras, 2004; Gross, 2002). Therefore, it is important, when possible, to measure both emotion reactivity and regulation when studying children’s emotions and/or temperament. One emotion regulation strategy is cognitive reappraisal, which is achieved by changing the emotional interpretation of a stimulus or event (Foti & Hajcak, 2008; Gross & John, 2003; Ochsner & Gross, 2005). Late positive potential (i.e., LPP) amplitudes have been shown to be sensitive to emotion regulation strategies such as directed reappraisal in children as young as 5 years old (e.g., Babkirk et al., 2015; Dennis & Hajcak, 2009) and adults (e.g., Foti & Hajcak, 2008; MacNamara, Ochsner, & Hajcak, 2011). Specifically, it has been reported that LPP amplitudes are reduced following the use of an emotion regulation strategy (e.g., directed reappraisal of an unpleasant stimulus in a more positive light as opposed to a more negative light). In essence, systematic research over the past two decades have helped establish the fact that LPP is a replicable and stable measure that varies with emotional reactivity as well as emotion regulation.
Therefore, it was the general purpose of the present investigation to examine the emotional reactivity and regulation of young children who do and do not stutter using (1) their LPP amplitude in response to pleasant, unpleasant, neutral pictures (reactivity) and listening to short reappraisal stories for the unpleasant pictures (regulation) and (2) their parents’ responses to the 195-item version of the Children’s Behavior Questionnaire (CBQ) (Rothbart, Ahadi, Hershey, Fisher, 2001). Based on the findings and theoretical accounts listed above, four research questions/empirical issues were addressed.
Emotional Reactivity to pleasant pictures
The first issue involved whether the cortical associates of emotional reactivity of CWS differs from that of their CWNS peers when viewing pleasant pictures. It was hypothesized that during the viewing of pleasant pictures, CWS, when compared to their CWNS peers, would exhibit greater emotional reactivity (as indexed by LPP amplitude).
Emotional reactivity to unpleasant pictures
The second issue involved whether cortical associates of emotional reactivity of young CWS differs from that of their CWNS peers when viewing unpleasant pictures. It was hypothesized that during the viewing of unpleasant pictures, CWS, when compared to their CWNS peers, would exhibit greater emotional reactivity (as indexed by LPP amplitudes).
Emotion regulation to unpleasant pictures after positive reappraisal
The third issue involved whether the cortical associates of emotion regulation differ between CWS and CWNS during viewing of unpleasant pictures following positive directed reappraisal stories (e.g., “Next is a picture of a snake that is completely harmless. It doesn’t even have teeth.”). It was hypothesized that CWS, when compared to CWNS, would exhibit less emotion regulation (as indexed by reduction of LPP amplitude from unpleasant picture viewing with no reappraisal, and neutral reappraisal) during viewing of unpleasant pictures that followed positive reappraisal stories.
Relation of temperamental and cortical indices of emotion reactivity and regulation
The fourth and final issue involved whether cortical associates (i.e., LPP amplitude) and caregiver report measures of emotion reactivity and emotional regulation were associated. It was hypothesized that for both CWS and CWNS, caregiver reports of emotional reactivity (i.e., Surgency/Extraversion and Negative Affect) and emotion regulation (i.e., Effortful Control) would be correlated with participants’ cortical indices of emotional reactivity and regulation.
2. Method
2.1 Participants
Initially, 21 CWS (5 females), and 30 CWNS (11 females) enrolled in this study. Participants were paid volunteers who either participated in a previous study in our lab, or whose parents learned of the study from advertisements in university news boards, flyers, a free, local monthly parent magazines, or were referred to the university clinic for an evaluation. The study’s protocol was approved by the university’s Institutional Review Board, and for all participants, informed consent by parents and assent by children were obtained.
In general, differing reasons for exclusion of young children (e.g., failure to reach eligibility criteria, movement artifacts, willingness to participate and/or compliance with a particular experimental task) led to varying numbers of participants being included in the final data corpus used to test each of the four a priori hypotheses. For Hypotheses 1 and 2 (Emotional Reactivity task), 1 female CWS and 5 CWNS (1 female) did not agree to participate in the ERP task, 1 male CWS and 2 CWNS (1 female) exhibited excessive movement artifacts in the EEG data, 2 CWS (1 female) and 1 CWNS failed to reach eligibility criteria (described below) for speech and language testing. Thus, 17 children who stutter (CWS, 3 females) and 22 children who do not stutter (CWNS, 7 females) served as participants in the final data corpus for Hypotheses 1 and 2. Children were between 4 years 0 months and 6 years and 11 months of age. CWS’s chronological age (M=63.11, SD=8.76) did not significantly differ from that of CWNS (M=67.59 SD=10.25) t(37)= 1.43, p=.159.
For Hypothesis 3 (Emotion Regulation task) 15 children who stutter (CWS, 3 females) and 22 children who do not stutter (CWNS, 7 females) served as participants in the final data corpus. Two of the 17 CWS who participated in the emotional reactivity portion of the study (Hypotheses 1 and 2) did not agree to participate in the emotion regulation task for Hypothesis 3. Children were between 4 years 0 months and 6 years and 11 months of age. CWS’s chronological age (M=63, SD=9.2) did not significantly differ from that of CWNS (M=67.59 SD=10.25) t(35)= 1.332, p=.192.
For Hypothesis 4, caregiver reports of children’s temperament were not available for 4 of the 17 CWS and 3 of the 22 CWNS due to incorrectly completed, partially or totally incomplete CBQ forms. Thus, 13 children who stutter (CWS, 2 females) and 19 children who do not stutter (CWNS, 6 females) served as participants in the final data corpus for data analyses pertaining to Hypothesis 4. Children were between 4 years 0 months and 6 years and 11 months of age. CWS’s chronological age (M=63.08, SD=10.15) did not significantly differ from that of CWNS (M=67.32 SD=9.98) t(30)= 1.143, p=.262.
Figure 1 depicts the entire experiment from talker group classification through experimental manipulation of emotion.
Figure 1.

A diagram of the current study’s experimental procedures.
Note: As per IAPS instructions, the original IAPS pictures have not been used in this illustration, rather, comparable non-copyrighted pictures were used instead. The loudspeaker (
 ) symbols associated with neutral and pleasant appraisal conditions represent the pre-recorded sentence of reappraisal that each participant heard for each picture presented during the two reappraisal conditions (with the order of presentation of the each of the trials from the two conditions randomized).
) symbols associated with neutral and pleasant appraisal conditions represent the pre-recorded sentence of reappraisal that each participant heard for each picture presented during the two reappraisal conditions (with the order of presentation of the each of the trials from the two conditions randomized).
2.2 Classification and Inclusion Criteria
To reduce the possibility of confounds with clinically significant speech-language and hearing concerns, all participants’ articulation, receptive and expressive language skills, as well as hearing abilities were assessed using standardized measures. Specifically, the “Sounds in Words” subtest of the Goldman-Fristoe Test of Articulation-2 (GFTA-2; Goldman & Fristoe, 2000) was used to measure participants’ speech sound articulation; the Peabody Picture Vocabulary Test-Fourth Edition (PPVT-IV; Dunn & Dunn, 2007) was used to assess receptive vocabulary; and the Test of Early Language Development-3 (TELD-3; Hresko, Reid, & Hamill, 1999) was used to measure receptive and expressive language abilities.
Children who scored below the 16th percentile (i.e., approximately one standard deviation below the mean) on any of these standardized speech or language tests were not included in the current study. In addition, bilateral pure tone hearing screenings were conducted to rule out hearing concerns. Participants were excluded if they did not perform within normal limits on this hearing-screening test (American Speech–Language–Hearing Association, 1990).
Participants were classified as CWS if they (a) exhibited three or more stuttered disfluencies (i.e., sound/syllable repetitions, sound prolongations, or monosyllabic whole-word repetitions) per 100 words of conversational speech (Conture, 2001; Yaruss, 1998), and (b) scored 11 or greater (i.e., severity of at least “mild”) on the Stuttering Severity Instrument-3 (SSI-3; Riley, 1994).
Participants were classified as CWNS if they (a) exhibited two or fewer stuttered disfluencies (i.e., sound/syllable repetitions, sound prolongations, or monosyllabic whole-word repetitions) per 100 words of conversational speech, and (b) scored 10 or lower on the SSI-3 (i.e., severity of less than “mild”).
Measurement Reliability for Stuttering
Approximately 20% of the final data corpus (i.e., 8 participants: 5 CWS and 3 CWNS) was randomly selected and re-coded by a speech-language pathologist independent from the original coder. This independent speech-language pathologist’s sample was used to determine inter-judge reliability for stuttering. Intra-class correlation coefficient (ICC) for stuttered disfluencies was .96, with a 95% confidence interval of .87 – .98, indicating high agreement among coders.
2.3 Caregiver Report of Temperament
Each participant’s caregivers were asked to complete the 195-item Children’s Behavior Questionnaire (CBQ) to assess temperamental factors related to emotional reactivity and emotion regulation (Rothbart, Ahadi, Hershey, Fisher, 2001). For the purposes of this study, and as per Rothbart (2011), the 3-factor level domains of Surgency/Extraversion, Negative affectivity, and Effortful Control factors were used for analysis. Surgency/Extraversion, also known as positive emotional reactivity, was indexed by averaging the scores of the following four CBQ sub-scales: activity level, high intensity pleasure, impulsivity, and shyness. Negative Affectivity, also known as negative emotional reactivity, was indexed by averaging the scores of the following five CBQ sub-scales: anger, discomfort, fear, sadness and soothability. Effortful Control refers to self-regulation of attention, inhibitory control and behavioral inhibition and was indexed by averaging the scores of the following four CBQ sub-scales: attention focusing, inhibitory control, low-intensity pleasure and perceptual sensitivity.
2.4 Stimulus Materials
Stimulus materials were developmentally appropriate pictures from the International Affective Picture System (IAPS; Lang et al., 2005). The IAPS pictures have been widely used for studying emotional processing in children (e.g., Dennis & Hajcak, 2009; Hajcak & Dennis, 2009; Solomon, DeCicco, Dennis, 2012). Pictures used in the current study were identical to those employed by Hajcak and Dennis (2009) in their study of typically developing children (see Appendix A for a list of the IAPS pictures and reappraisal stories used in the present study). Recently, Solomon et al. (2012) successfully used the same pictures to examine emotional processing in children between the ages of 5 to 7 years. Likewise, the positive reappraisal stories used in the present study were adapted from Dennis & Hajcak (2009).
The IAPS pictures used in the present study consisted of (i) 30 pleasant pictures depicting scenes or objects such as ice cream, Disney World, etc.; (ii) 30 unpleasant pictures depicting scenes or objects such as an accident, a snake, etc.; and (iii) 30 neutral pictures depicting scenes or objects such as household objects. All ninety pictures were presented using E-prime 2.0 software (Psychology Software Tools, Pittsburgh, PA) on a PC computer and a 17” monitor, with the screen to participant eyes approximately 25” and pictures were presented in their original size at the center of the computer screen.
Norms for picture valence and arousal ratings have been determined for adult viewers (Lang et al., 2005). The picture categories differ in terms of valence ratings (M= 7.45, SD= 0.58, for pleasant pictures; M= 5.29, SD= 0.74, for neutral pictures; and M= 3.36, SD= 0.73, for unpleasant pictures). In addition, based on adult ratings, the emotional pictures are reliably higher on normative arousal ratings (M= 4.76, SD= 0.75, for pleasant pictures; M= 5.70, SD= 0.69, for unpleasant picture content; and M =2.81, SD= 0.65, for neutral pictures).
Norms for picture valence and arousal are not available for children due to children’s relative lack of reliability in reporting such values (for further details see Dennis & Hajcak, 2009). However, in the present study, after the experimental testing, we administered an abbreviated version of the Self Assessment Manikin scale (SAM, Bradley & Lang, 1994; Lang et al., 2005) to approximately 38% (n = 15) of the participants (6 CWS and 9 CWNS). This scale was used to rate randomly selected 5 neutral, 5 pleasant, and 5 unpleasant pictures for valence. The children were asked to rate each of the images on a 5-point scale based on how happy or sad it made them feel. The valence scale of the abbreviated SAM depicts five characters who range from happy to unhappy, with ‘1’ corresponding to the happiest figure, ‘5’ corresponding to the least happy figure, and ‘3’ located in between.
Valence ratings in the present study were consistent with those reported in 5-to 8-year-old typically developing children (Hajcak &Dennis, 2009). Overall, as predicted, valence ratings increased (i.e., happy [1] to unhappy [5]) from pleasant to neutral and from neutral to unpleasant pictures. Specifically, pleasant pictures (M=1.55; SD=.53) were rated as more pleasant than unpleasant (M=3.67; SD=.84), t(14)= 8.24, p<.0001) and neutral (M=2.47; SD=.62), t(14)= 4.89, p< .0001) pictures. Additionally, neutral pictures were rated as more pleasant than unpleasant pictures, t(14)=3.8, p< .003.
2.5 Procedure
2.5.1 Pre-task set-up
To begin, each child and caregiver was given a brief introduction to the laboratory and the task. Subsequently, children were seated in front of a 17″ computer monitor, where they were given the option to watch cartoons as the electroencephalograph (EEG) electrodes were placed on their scalp. To reduce children’s emotional discomfort and/or concerns they might have about the equipment, set-up, or the experiment, children were told that they were “astronauts getting signals from outer space.”
2.5.2 ERP task: Emotional reactivity
Children were asked to passively view 90 IAPS pictures presented in a randomized fashion. Each picture was displayed once throughout the task for 2500 ms, followed by a random inter-stimulus interval (ISI) between 700 and 1300 ms. Unlike previous studies involving a fixed ISI (e.g., 500 ms used in Hajcak & Dennis, 2009), the ISI in the present study was randomly selected between 700 and 1300 ms. A non-fixed interval was used to avoid possible habituation to the picture presentation onset due to the predictability and/or rhythmicity of a constant ISI, the latter thought to possibly engender boredom and/or inattention on the part of the young participant. On average, the emotional reactivity task took approximately 8 minutes to complete.
Once the first author initiated the task, all trials began automatically; however, the experimenter monitored each participant throughout each task for signs of inattention, uneasiness, excessive muscle tension or movement. Stimulus presentation was suspended if and when the first author noticed such signs, and stimulus presentation was resumed after children were redirected.
2.5.3 ERP task: Emotion regulation
Following the emotional reactivity task, participants performed an emotional regulation task involving directed reappraisal (e.g., DeCicco et al., 2012). Participants viewed 30 unpleasant pictures twice, once following a “neutral” and once following a “positive reappraisal story. The order of the type of reappraisal stories (i.e., neutral or positive reappraisal) was random for each picture for all participants.
Reappraisal stories were provided as the same audio recording for all participants, and one that offered a neutral and a positive interpretation of the unpleasant pictures. Each recording lasted approximately 3-5 seconds while a cartoon image of an ear was presented on the blank screen. Participants heard each reappraisal story followed by a 500 or 700 ms delay prior to picture onset. The pictures associated with the stories were then presented for 2500 ms, with a 1200 or 1500 ms inter-trial interval between each stimulus and the next story. On average, the entire emotion regulation condition (i.e., neutral + positive reappraisals) took approximately 10 minutes to complete.
At the beginning of the reappraisal task, children were instructed, “For our next game, we are going to see some of the same pictures. Listen to the stories and think of the pictures so they match the stories. Remember to stay still and just look at the screen.” Children were reminded to not blink or move around much when viewing the pictures and listening to the stories. EEG was recorded continuously for the whole emotion regulation task.
2.6 Event Related Potential (ERP) Recording (Data Collection) and Analysis
A high-density array of 128 Ag/AgCl electrodes embedded in soft sponges (Geodesic Sensor Net, EGI, Inc., Eugene, OR) was used to record the ERPs. Electrode impedance levels were below 40 kOhm before and after testing. Data were sampled at 250 Hz with the filters set to 0.1-100 Hz throughout the testing. All electrodes were referred to Cz (vertex) during data collection and they were re-referenced to the average reference offline during data analysis (Picton et al., 2000). All bioelectric signals were digitized on a Macintosh computer.
2.6.1 ERP analyses (pre-descriptive and pre-inferential data analysis considerations)
Analysis of ERP data was conducted offline using Net Station 5.3 Software (EGI, Inc., Eugene, OR, USA). Data were band-pass filtered with cut-offs at 0.1 and 30 Hz and referenced to an average of all electrodes. Segmentation of the EEG data for each trial was performed using Net Station software, starting 500 ms prior to each picture onset and continuing for 2500 ms. The average activity in the 500 ms period prior to each picture onset served as the baseline. Eye and other movement artifacts were removed based on the results of Net Station 5.3 software automated screening, followed by manual review. Specifically, to remove from the final data corpus any EEG/ERP recordings confounded by eye or movement artifacts, for the eye channels, voltage exceeding 140 μV was considered as an eye blink and voltage above 55 μV was considered as eyeball movements. “Bad” channels were defined as any channel with voltage exceeding 200 μV. Within each trial (i.e., passive viewing of one picture), if more than 15% of the electrodes were determined to be bad, that trial was excluded from the analyses. The remaining artifact-free trials were averaged within unpleasant, neutral, and pleasant conditions. For a data set to be included in the statistical analyses, individual averages had to be based on at least 8 trials. Moran, Jendrusina, and Moser (2013) found that LPP had good internal consistency and remained stable after 8 trials added to the average.
There were no significant between-group differences in number of trials included in the final data corpus for pleasant, unpleasant, and neutral emotional reactivity conditions: pleasant pictures: CWS (M=13.05, SD=5.01), and CWNS (M=12.45, SD= 5.11), F(1, 37)=.136, p=.714; unpleasant pictures: CWS (M=11.47, SD=4.5), and CWNS (M=12.81, SD=4.08), F(1, 37)= .945, p=.337; neutral pictures: CWS (M=10.94, SD=4.75), and CWNS (M=11.6, SD=4.68), F(1, 37)=.183, p=.672. Similarly, there were no significant between-group differences in the number of trials included in the final data corpus for emotion regulation, that is, positive and neutral reappraisal conditions: positive reappraisal: CWS (M=10.87, SD=3.3), and CWNS (M=13.18, SD=4.03), F(1, 35)= 3.37, p=.075; and neutral reappraisal: CWS (M=10.7, SD=4.1) versus CWNS (M=12.2, SD=5.25), F(1, 35)=.928, p=.342.
Time Windows of Analysis
The LPP is a sustained, rather than a brief response. Therefore, it is common for empirical studies of LPP amplitude to examine three separate time windows across the LPP, from early to late temporal portions of the LPP response. Consistent with this practice, the present study defined LPP amplitude as the average activity in each of 3 consecutive time windows following stimulus onset: early (500-1000 ms), middle (1000-1500 ms), and late (1500-2000 ms.) following the onset of the picture stimulus (e.g., Hajcak & Dennis, 2009). As shown in Appendix B, the specific electrodes for the LPP regions were selected to represent the parietal (i.e., Pz) location, where LPP has been documented to be maximal (e.g., Pastor et al., 2008). Mean LPP amplitudes were computed by averaging across the selected electrodes using a custom package (Moore, 2015) implemented in RStudio (RStudio Team, 2015).
2.7 Statistical analyses
Prior to hypothesis testing, as is the convention in this area of empirical investigation, we examined whether the LPP was sensitive to emotional valence of the stimuli and to the reappraisal manipulation in 4-6-year-old children who do and do not stutter (i.e., CWS+CWNS). These analyses determined whether the LPP was sensitive to emotional valence of the stimuli in 4-6-year-old children who do and do not stutter.
To test the first hypothesis – CWS, when compared to their CWNS peers, would exhibit greater LPP amplitudes during pleasant picture viewing trials – a Group (2: CWS and CWNS) × Time Window (3: early, middle, late) mixed model ANOVA was performed for the pleasant picture trials. In this analysis, LPP amplitude during pleasant picture trials was the dependent variable, Talker Group was an independent variable and Time Window was a repeated measure. The amplitude of LPP to the neutral pictures (in the corresponding time windows) and gender were covariates. Amplitude of LPP during neutral pictures was chosen as a covariate to account for potential baseline differences in picture processing.
To test the second hypothesis – CWS, when compared to their CWNS peers, would exhibit greater LPP amplitudes during unpleasant picture viewing trials -a Group (2: CWS and CWNS) × Time Window (3: early, middle, late) mixed model ANOVA was performed for the unpleasant picture trials. LPP amplitude during unpleasant picture trials was the dependent variable, Talker Group was an independent variable and Time Window was a repeated measure. The amplitude of LPP to the neutral pictures (in the corresponding time windows) and gender were covariates.
Prior to inferential analyses of Hypothesis 3 (Emotion Regulation Task) regarding between-group differences in emotion regulation (as indexed by changes in LPP amplitude), as is the convention in this area of investigation, inferential analyses were conducted for all present participants (i.e., CWS+CWNS). These analyses helped the author determine whether the LPP was sensitive to experimental manipulation of emotion in this population.
To provide a comprehensive analysis of the third hypothesis - CWS, when compared to their CWNS peers, would exhibit less reduction in the LPP amplitude during viewing of the unpleasant pictures following positive reappraisal stories compared to those that followed (a) neutral reappraisal and (b) no reappraisal, two analyses were completed. For both analyses, a Group (2: CWS and CWNS) × Time Window (3: early, middle, late) mixed model ANOVA was performed. LPP amplitude during positive reappraisal trials was the dependent variable; Talker Group was an independent variable, and Condition and Time Window were repeated measures.
For the first analysis of the third hypothesis, the covariates were gender and the amplitude of LPP to unpleasant pictures (in the corresponding time windows) with no reappraisal (i.e., unpleasant pictures during Emotional Reactivity task). The amplitude of LPP to the unpleasant pictures was chosen as a covariate to attempt to quantify emotion regulation by comparing positive reappraisal stories to no reappraisal.
For the second analysis of the third hypothesis, the covariates were gender and the amplitude of LPP to unpleasant pictures (in the corresponding time windows) during neutral reappraisal trials. The neutral reappraisal condition was specifically chosen as a covariate in attempts to quantify the impact of positive stories in comparison to neutral stories on the LPP amplitudes during the Emotion Regulation task.
To test the fourth hypothesis -both CWS and CWNS’s cortical measures of emotional reactivity would be associated with parent reports’ of these processes (i.e., CBQ scores on negative affectivity, surgency/extraversion and effortful control scales) -linear regressions were used. Difference scores for the LPP amplitude were used as the dependent variable for the reactivity and reappraisal conditions (e.g., positive reappraisal - unpleasant reactivity) in attempts to (a) account for potential baseline differences in picture processing, and (b) measure participants’ cognitive reappraisal/emotion regulation relative to their initial emotional reactivity to the unpleasant pictures (i.e., in the absence of a reappraisal story). The p-values of all correlations were adjusted using the SAS Proc Multtest procedure to mitigate family-wise false discovery rate [i.e., Type 1 error; Benjamini & Hochberg, 1995; Westfall, Tobias, Rom, Wolfinger, & Hochberg, 1999]).
3. Results
3.1 Descriptive Analyses
3.1.1 Participants’ stuttered, non-stuttered, total speech disfluencies and stuttering severity
One-way ANOVA compared young CWS and CWNS on measures of speech disfluency. As would be expected based on participant classification criteria, there was a significant difference in stuttered disfluencies per 100 words between CWS (M = 5.38 SD = 2.21) and CWNS (M = .83, SD = 0.7), F (1, 37) = 82.97, p < .0001. There was no significant difference between CWS (M = 4.39, SD = 2.48) and CWNS (M = 3.45, SD = 1.98), F(1, 37) = 1.761, p =.193 in non-stuttered disfluencies per 100 words. There was a significant difference between CWS (M = 9.78, SD = 2.71) and CWNS (M = 4.29 SD = 2.29), F (1, 37) = 46.716, p < .0001 in total disfluencies per 100 words, Likewise, there was a significant difference between SSI-4 scores for CWS (M = 14.94, SD = 3.73) and CWNS (M = 6.09, SD = .43), F (1, 37) = 122.56, p < .0001. These descriptive data pertaining to young CWS and CWNS’s speech (dis)fluency were consistent with comparable measures obtained from a larger scale investigation of these two talker groups (Tumanova, Conture, Lambert & Walden, 2014).
3.1.2 Speech and language
One-way ANOVA compared young CWS and CWNS on standardized speech and language tests. There was no significant difference between CWS (M = 102.31, SD = 9.12) and CWNS (M = 105.6, SD = 8.06) in speech sound articulation, as measured by the GFTA-2 (F(1,37) = 1.37 p= .249. In addition, there was no significant difference between CWS (M= 112, SD=13.45) and CWNS (M=117.77, SD= 9.91) in PPVT-IV (F(1,37) = 2.83, p = .131). Children who stutter (M=113.52 SD=10.05) and CWNS (M=114.58, SD=9.69) did not significantly differ in receptive subtest of TELD-3 F(1,34) =.102, p = .752. Lastly, there was no significant difference between CWS (M=102.43 SD=8.8) and CWNS (M=108.84, SD=10.25) in expressive subtest of TELD-3 F(1,32) =3.54, p = .069. All participants passed the hearing screening.
3.2 Inferential analyses
3.2.1 Emotional reactivity task: All participants’ (CWS+CWNS) sensitivity, as measured by LPP amplitude, to experimental manipulation of emotional reactivity to all picture stimuli.
For the emotional reactivity tasks, Figure 2 (CWS) and Figure 3 (CWNS) depict, the mean stimulus-locked ERPs (i.e., LPP) obtained from the parietal electrode cluster (i.e., Pz) during passive viewing of pleasant, unpleasant, and neutral pictures. For both talker groups, LPP amplitudes across the three time windows (early, middle, & late) were greater for pleasant (M= 1.49, SE=.49) and unpleasant (M=2.44, SE=.44) than for neutral (M= 0094, SE=.47) stimuli F(2, 196)= 6.54, p=0.002, ηp2 =.062. Hence, emotional reactivity, as indexed by LPP amplitude, was greater for all children in emotionally when compared to neutrally valenced pictures.
Figure 2. CWS’s (n=17) LPP Amplitude during Emotional Reactivity Task.

Note: The mean stimulus- locked ERPs (i.e., LPP) shown above were recorded at the parietal electrode cluster (i.e., Pz) during passive viewing of pleasant, unpleasant, and neutral pictures.
Figure 3. CWNS’s (n=22) LPP Amplitude during Emotional Reactivity Task.

Note: The mean stimulus-locked ERPs (i.e., LPP) shown above were recorded at the parietal electrode cluster (i.e., Pz) during passive viewing of pleasant, unpleasant, and neutral pictures.
3.2.1.1 Emotional Reactivity Task (CWS vs. CWNS): A Priori Hypotheses 1 and 2
Hypothesis 1 (H1): CWS, when compared to their CWNS peers, would exhibit greater LPP amplitude to pleasant pictures
There was a significant main effect for Time Window (F(2, 79.46)= 8.01, p= .001), which suggests that for all participants (i.e., CWS + CWNS) there was a significant difference among the early, middle, and late time windows of LPP to pleasant pictures However, there was neither a significant main effect for Talker Group (F(1, 97.55)= .242, p=.624, ηp2 =.002) nor significant interaction effect of Talker Group × Time Window (F(2, 70.59)= .255, p=.776, ηp2 =.007). Thus, findings of inferential analyses of H1 did not support H1.
Hypothesis 2 (H2): CWS, when compared to their CWNS peers, would exhibit greater LPP amplitude to unpleasant pictures
There was a significant main effect for Talker Group F(1, 96.5)= 4.922, p =.029, ηp2 =.048. That is, CWS (M=3.806, SE=.620) exhibited significantly greater LPP to the unpleasant pictures than CWNS (M=2.102, SE=.540) across the three time windows. Likewise, there was a significant main effect for Time Window (F(2, 82.7)= 6.562, p=.002, ηp2 =.14) as well as interaction of Talker Group × Time Window (F(4, 76.8)= 3.866, p=.006, ηp2 =.17).
Simple effects tests were conducted to follow up the significant Talker Group × Time Window interaction effect. No between-group differences were found in either the early window (F(1, 35)= 1.36, p=.251, ηp2 =.037) or late window (F(1, 35)= .086, p=.771, ηp2 =.002). However, in the middle time window of LPP (i.e., 1000-1500 ms), CWS (M=2.816, SE=.895) exhibited significantly greater LPP to the unpleasant pictures compared to CWNS (M=.158, SE=.782) F(1, 35)=4.776, p=.036, ηp2 =.125 (see Figure 4). In brief, findings of inferential analyses supported H2 in the middle time window of LPP activity.
Figure 4. LPP Amplitude to Unpleasant Pictures in Middle Window.

Note: The figure depicts CWS (n=17) and their CWNS peers’ (n=22) estimated marginal means and error bars (standard error of the mean) for LPP amplitudes during unpleasant pictures (covariates = gender and participants’ LPP amplitude while viewing neutral pictures)
3.2.2 Emotion regulation task: All Participants (CWS+CWNS) sensitivity – as measured by LPP amplitude - to experimental manipulation of emotional regulation of all picture stimuli
For the emotion regulation task, Figure 5 (CWS) and Figure 6 (CWNS) depict the mean stimulus-locked ERPs (i.e., LPP) obtained from the parietal electrode cluster (i.e., Pz) during reappraisal conditions. Findings indicated that for the two talker groups, LPP amplitudes were significantly greater for positive (M= 13.24, SD= 8.2) and neutral (M=11.24, SD=7.46) reappraisal conditions versus no reappraisal of unpleasant pictures (M= 7.01, SD=5.14) in the early time window of LPP t(33)= −4.028, p<.0001 and t(33)= −2.95, p= .006, respectively. There were no such significant differences for the middle or the late time windows for all participants (i.e., CWS + CWNS).
Figure 5. CWS’s (n=15) LPP Amplitude during Emotion Regulation Task.
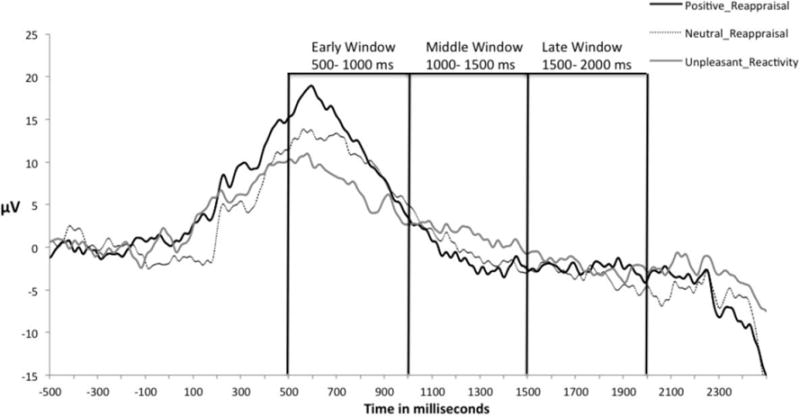
Note: The mean stimulus-locked ERPs (i.e., LPP) shown above were recorded at the parietal electrode cluster (i.e., Pz) while viewing unpleasant pictures with pleasant and neutral reappraisal (i.e., emotion regulation conditions) versus viewing the same pictures without such appraisal (i.e., emotion reactivity conditions) pleasant picture viewing condition during emotional reactivity task.
Figure 6. CWNS’s (n=19) LPP Amplitude during Emotion Regulation Task.

Note: The mean stimulus-locked ERPs (i.e., LPP) at the parietal electrode cluster (i.e., Pz) while viewing unpleasant pictures with pleasant and neutral reappraisal (i.e., emotion regulation conditions) versus viewing the same pictures without such reappraisal (i.e., emotion reactivity conditions) pleasant picture viewing condition during emotional reactivity task.
Hypothesis 3 (H3): CWS, when compared to their CWNS peers, would exhibit less reduction in LPP amplitude during viewing of unpleasant pictures that followed positive reappraisal
To provide a comprehensive analysis of Hypothesis 3, two inferential analyses were conducted. The first analysis used the no-reappraisal condition as a referent, and the second analysis used the neutral reappraisal condition as a referent.
First Analysis re Hypothesis 3 (Positive reappraisal with Unpleasant Reactivity as a covariate): For viewing unpleasant pictures with positive reappraisal with no reappraisal as a referent, the main effect for Time Window (F(2, 62)= 21.65, p< .0001, ηp2 =.411) was significant for all participants (CWS +CWNS). This finding suggests a significant difference among the early, middle, and late time windows of LPP during positive reappraisal condition. However, there was no significant main effect for Talker Group (F(1, 63.8)= 1.28, p= .263, ηp2 =.019) and no interaction of Talker Group × Time Window (F(2, 57.43)= .137, p= .872, ηp2 =.004). Therefore, findings of the inferential First Analysis did not support H3.
Second Analysis re Hypothesis 3 (Positive reappraisal with Neutral reappraisal as a covariate): For viewing unpleasant pictures after listening to positive reappraisal stories with neutral reappraisal as a referent, there was no main effect for Talker Group (F(1, 86.5)= .181, p= .672, ηp2 =.002) and no interaction of Talker Group × Time Window (F(2, 70.47)= .027, p= .973, ηp2 =.0007). In summary, findings of the inferential Second Analysis did not support H3.
3.2.3 Hypothesis 4 (H4): Both CWS and their CWNS peers would exhibit correlations between cortical indices of emotional reactivity and regulation and caregiver reports of temperamental characteristics
As shown in Table 1, for each time window of LPP the effect of picture type on the LPP (LPP unpleasant-neutral and LPP pleasant-neutral) was correlated with temperamentally-related factors (i.e., Surgency/Extraversion, Negative Affectivity and Effortful Control), from Children’s Behavior Questionnaire (CBQ; Table 1 of Rothbart, 2001).
Table 1.
Pearson’s product-moment correlations for those CWS (n=13), and CWNS (n=19) for whom parent-report based CBQ data were available
| Difference Score | Negative Affectivity | Surgency | Effortful Control | ||||
|---|---|---|---|---|---|---|---|
|
| |||||||
| CWS | CWNS | CWS | CWNS | CWS | CWNS | ||
|
| |||||||
| Unpleasant-neutral | early
|
−0.126 | 0.058 | 0.384 | 0.329 | −0.647* | −0.41 |
| middle
|
0.146 | −0.083 | 0.021 | 0.129 | −0.136 | −0.292 | |
| late
|
0.318 | 0.26 | −0.351 | −0.057 | −0.129 | 0.021 | |
|
| |||||||
| Pleasant-neutral | early
|
−0.311 | 0.204 | 0.595 | −0.028 | −0.396 | −0.103 |
| middle
|
−0.190 | 0.192 | −0.039 | −0.061 | 0.363 | −0.189 | |
| late
|
0.145 | 0.076 | −0.291 | 0.107 | 0.283 | −0.318 | |
|
| |||||||
| Positive Reappraisal-Unpleasant Reactivity | early
|
−0.582 | 0.238 | 0.138 | −0.406 | 0.193 | 0.427 |
| middle
|
−0.516 | 0.383 | 0.085 | −0.277 | −0.03 | 0.264 | |
| late
|
−0.709* | 0.035 | 0.151 | −0.194 | −0.246 | 0.215 | |
|
| |||||||
| Positive Reappraisal-Neutral Reappraisal | early
|
−0.683* | 0.561 | 0.455 | −0.5 | 0.32 | 0.465 |
| middle
|
−0.581 | 0.394 | 0.353 | −0.542 | −0.01 | 0.516 | |
| late
|
−0.484 | 0.387 | 0.293 | −0.408 | 0.108 | 0.263 | |
Note: After False Discovery Rate correction (Benjamini, Hochberg, 1995), a correlation with an *indicates p<.05. Difference scores (e.g., unpleasant picture viewing condition – neutral picture viewing condition) for LPP amplitude were as follows: (1) unpleasant-neutral reactivity, (2) pleasant-neutral reactivity, and (3) positive reappraisal-unpleasant reactivity, and (4) positive reappraisal-neutral reappraisal conditions. The SAS Proc Multtest procedure was applied to all correlations in this table to mitigate family-wise false discovery rate (Benjamini & Hochberg, 1995; Westfall et al., 1999).
3.2.3.1 Correlations between Emotional Reactivity and Children’s Temperament for CWS and CWNS
CWS: For the unpleasant picture condition, after correcting for false discovery rate (hereafter referred to as “FDR corrected”), there was one significant negative correlation between CWS’s Effortful Control and LPP in the early window (r=−.647, p=.017). Specifically, greater temperamental effortful control, as measured by parent responses to the CBQ, was associated with lower emotional reactivity as measured by LPP. There were no significant correlations between CWS’s temperament scores on the CBQ and their LPP amplitude for pleasant picture condition.
CWNS: For both the pleasant and unpleasant pictures, after FDR correction, there were no significant correlations between CWNS’s temperament scores on the CBQ and their LPP amplitude.
Between-group comparison of correlations
Using Fisher r-to-z transformation (Fisher, 1921; Howell, 2011), the authors attempted to determine statistical significance of between-group (CWS vs. CWNS) differences in correlation coefficients. This was done only when at least one talker exhibited a statistically significant correlation after FDR correction (i.e., those presented in the paragraphs above). For the unpleasant pictures, there was no significant difference between CWS’s (r=−647) and CWNS’s (r=−.369) correlations of Effortful Control score on CBQ and their LPP in the early window z=−0.95, p=.34.
3.2.3.2 Correlations between Emotional Regulation and Children’s Temperament for CWS and CWNS
CWS: For the positive reappraisal condition, regardless of the referent/baseline (i.e., unpleasant reactivity or neutral reappraisal) and after FDR correction, there were two significant correlations: a significant negative correlation between CWS’ Negative Affectivity score on CBQ and their LPP amplitude for both the early window r= −.683, p=.014 and the late window r=−.709, p=.012. Thus, during positive reappraisal, CWS with greater temperamental negative affectivity exhibited smaller LPP amplitudes to the unpleasant pictures.
CWNS: There were no significant correlations between LPP amplitude during reappraisal conditions and caregiver reports of CWNS’s temperament.
Between-group comparison of correlations
Using Fisher r-to-z transformation (Fisher, 1921; Howell, 2011), the statistical significance of the difference between CWS and CWNS’s correlation coefficients for reappraisal conditions were determined. Again, such between-group comparisons were done for only the statistically significant correlations (i.e., those presented in the paragraphs above).
For the positive reappraisal condition (positive reappraisal-no reappraisal) in the late time window, there was a significant difference between CWS’s (r=−.709) and CWNS’s (r=.11) correlations of Negative Affectivity score on CBQ and their LPP z=−2.47, p=.013. Additionally, for the positive reappraisal condition in the early time window (positive reappraisal-neutral reappraisal), there was a significant difference between CWS’s (r=−.683) and CWNS’s (r=.561) Negative Affectivity score on CBQ and their LPP z=−3.64, p=.0003.
4. Discussion
4.1 Summary of Main Findings
Findings of the present study indicated that CWS exhibited significantly greater LPP amplitude than their CWNS peers in response to unpleasant pictures. In addition, greater LPP amplitudes for unpleasant pictures in the early time window (i.e., 500 to 1000 ms), was associated with lower Effortful Control score on the CBQ in CWS. Last, higher Negative Affectivity scores on the CBQ correlated with lower LPP amplitudes during the positive reappraisal condition in CWS, and this correlation was significantly different from that of their CWNS peers. There were no between-group differences during viewing of pleasant pictures, and during the emotion regulation task. The implications of these findings are discussed below.
4.1.1 Between-group differences in emotional reactivity (as indexed by LPP amplitude) to pleasant pictures (Hypothesis 1)
Contrary to our predictions in Hypothesis 1, CWS did not significantly differ from their CWNS peers in LPP amplitude to pleasant picture stimuli. At least three possible explanations for this finding may be put forth.
First, the present study’s pleasant picture viewing condition may not have elicited positive affect that was strong enough to reveal any existing between group differences. While we were able to acquire valence ratings from children, self-arousal ratings were not available due to young children’s lack of reliability in reporting such measures. Future studies that concurrently acquire LPP data in combination with other objective measures of emotional arousal such as skin conductance level would allow to examine the effectiveness of our positive affect manipulation in greater detail.
A second, alternative, explanation is that CWS and CWNS are not different in their emotional reactivity to pleasant stimuli. Although this account would support the null hypothesis, it would be in contradiction with previous findings that CWS exhibited (1) more emotion regulation following positive than neutral condition (Jones et al., 2014a), and (2) greater speech disfluencies following a positive emotion condition (receiving a desirable gift) than following a negative condition (receiving a disappointing gift, Johnson et al. 2010).
A third possibility is that the “absence of evidence is not the same as evidence of absence” (Leppink, O’Sullivan, Winston, 2017; pp. 117). Therefore, it may simply be the case that the present study lacks the statistical power to detect between group differences in young children’s LPP amplitude associated with pleasant picture stimuli.
4.1.2 Between-group differences in emotional reactivity (as indexed by LPP amplitude) to unpleasant pictures (Hypothesis 2)
Supporting Hypothesis 2, young CWS, when compared to their CWNS peers, exhibited greater LPP amplitude to unpleasant picture stimuli. One possible explanation for this finding is that CWS, compared to CWNS, may exhibit more emotional reactivity to unpleasant stimuli/situations. This explanation is consistent with empirical findings that CWS, relative to CWNS peers, exhibit (a) more negative emotional expressions during a disappointing gift procedure (Johnson et al., 2010), (b) more negative emotion during an emotionally frustrating task (Ntourou et al. 2013), (c) higher anger/frustration (Eggers et al., 2010) as well as (d) greater emotional reactivity based on caregiver reports (Karrass et al., 2006).
An alternative explanation is that CWS, when compared to CWNS, may exhibit less emotion regulation. This account is consistent with findings from empirical studies using caregiver reports. For example, Eggers et al. (2010) reported that CWS, when compared to CWNS, exhibited significantly lower emotional regulation, as indexed by lower inhibitory control (i.e., “the ability to plan and to suppress approach responses under instruction or in uncertain situations,” Rothbart, 2011), but higher anger/frustration. In addition, CWS, when compared to CWNS, have been reported to display higher emotional reactivity but lower emotion regulation (Karrass et al., 2006) and less adaptability to change (Anderson et al., 2003). Likewise, in a study of emotion reactivity and regulation, Jones et al (2014) reported that at baseline, young CWS, compared to CWNS, exhibited lower amplitude respiratory sinus arrhythmia (i.e., lower emotion regulation). The present experimental design (i.e., reappraisal conditions) as well as caregiver report data allows us to further speculate about various possibilities, for example, that CWS exhibit higher emotional reactivity or CWS concurrently exhibit higher reactivity and lower regulation. Such speculations are discussed in the General Discussion section below.
4.1.3 Between-group differences in emotion regulation (as indexed by LPP amplitude) during reappraisal condition (Hypothesis 3)
Contrary to our predictions in Hypothesis 3, there was no significant between-group difference in LPP amplitudes during viewing of unpleasant pictures that followed positive reappraisal. As with Hypothesis 1 above, one may put forth at least two possible explanations for this findings regarding Hypothesis 3.
First, one possible explanation for non-significant findings is that CWS and CWNS do not differ in their emotion regulation skills. While this account would support the null finding for this hypothesis, it would not be consistent with findings from previous reports suggesting that CWS, compared to CWNS, exhibited (1) greater difficulty in emotion regulation per caregiver reports (Karrass et al., 2006); and (2) less emotion regulation (indexed by parasympathetic system activity) during a baseline, nonspeaking condition (Jones et al., 2014a). Further, this account would be in conflict with the findings suggesting that CWS exhibited higher stuttering frequency in association with (1) decreased duration and frequency of behavioral regulatory strategies (Arnold et al., 2011), and (2) caregiver-reports of lower effortful control skills (Kraft, Ambrose, Chon, 2014).
A second possible explanation is that the present study may lack the statistical power to detect any between-group differences in young children’s LPP amplitude during an emotion regulation task. Therefore, one must be very cautious in interpreting these null findings as “evidence of absence” in studies with limited sample sizes (Leppink et al., 2017, pp. 117). Future studies with larger sample sizes, as well as those that replicate this line of research are needed to determine whether the non-significant findings are indeed evidence for a null hypothesis.
4.1.4 Correlations between LPP and parental report of emotional reactivity and emotion regulation (Hypothesis 4)
Emotional reactivity
The support for Hypothesis 4 was only found for CWS. Specifically, CWS’s temperamental Effortful Control (a factor level CBQ construct related to emotional regulation) was negatively correlated with their cortical reactivity to unpleasant pictures. This finding suggests that CWS with greater effortful control were more likely to show less emotional reactivity to unpleasant pictures. This finding is consistent with the notion that effortful control helps an individual down-regulate emotional reactivity to arousing stimuli, events, etc. And although some may question the value of parent reports of children’s temperamental characteristics, it has been suggested that “while parent report measures do contain some subjective parental components, available evidence indicates that these measures also contain a substantial objective component that does accurately assess children’s individual characteristics” (Henderson & Wachs, 2007, p. 402). Even so, it remains unclear why these correlations were only found for CWS; however, subsequently we will speculate about some possibilities.
Emotion regulation
The last finding supportive of Hypothesis 4, was again only found for CWS. Specifically, CWS with greater temperamental negative affectivity showed reduced LPP amplitudes to the unpleasant pictures during positive reappraisal condition. This suggests that CWS with greater temperamental negative affectivity were more likely to benefit from the positive reappraisal stories, enabling them to down-regulate their emotional reactivity to unpleasant pictures. It remains unclear from present findings whether endogenous (i.e., effortful control) versus exogenous (i.e., examiner provided reappraisal stories) sources are more helpful to CWS in down-regulating their emotions, issues that must await future empirical study.
Present findings that CWNS’s temperamental measures of negative affectivity, surgency, and effortful control were not correlated with their cortical associates of emotional reactivity and regulation challenges a straightforward explanation. Perhaps, one might speculate, parents of CWS are somewhat more sensitive to their children’s emotional processes/temperament than parents of CWNS, a suggestion requiring further empirical study to support or refute. It is also possible that the stimuli and experimental manipulation of emotion in the present study were not arousing enough for CWNS to exhibit emotional responses in keeping with their temperament proclivities. Regardless, to the best of the present authors’ knowledge, these findings are the first to have documented significant, meaningful correlations between cortical measures and parental reports of emotional reactivity and regulation in young children who stutter.
4.2 General Discussion
Overall, present findings are consistent with theoretical models regarding the association between emotion and childhood stuttering (Conture & Walden, 2012; Smith & Weber, 2017). This does not mean that emotional reactivity to unpleasantly valenced stimuli causes stuttering, but it does suggest that behavioral, cortical, and psychophysiological indices of emotion warrant further empirical study regarding childhood stuttering.
In addition to present findings supporting some of the assumptions of the aforementioned theoretical models of childhood stuttering, there are several possible accounts for why CWS, when compared to their CWNS peers, exhibited greater LPP amplitudes to unpleasant pictures. In an overview of the relation between emotion, temperament and speech and language disorders, Conture and colleagues (Conture, Kelly & Walden, 2013) discussed the following four possible accounts: (a) the disability model (e.g., experiences with stuttering result in emotional reactivity), (b) the predispositional model (e.g., temperament and emotional differences contribute to the onset of stuttering), (c) the bidirectional model (e.g., stuttering and emotional processes covary), and (d) the correlational model (e.g., a third variable is related to both stuttering and emotional processes). Immediately below we briefly discuss the present study’s findings relative to some of these various models.
The first, or disability model suggests that young CWS’s increased LPP amplitude during unpleasant picture viewing (when compared to that of their CWNS peers) reflects that they have developed greater emotional reactivity (to more negatively valenced stimuli) due to their frequent negative experiences with stuttering. This “spread of reactivity” hypothesis would predict that CWS develop a tendency to exhibit greater emotional reactivity – whether measured behaviorally, cortically, and/or psychophysiologically – because these children frequently experience unpleasant stimuli when they plan to, initiate or are in the process of speaking. These stimuli could be internal (e.g., inability to fluently, rapidly initiate speech, etc.) and/or external (e.g., listeners interrupting and/or talking for them, etc.) in nature (or a complex amalgam of both) and may be relatively often encountered during CWS’s verbal communication.
Certainly, one cannot categorically rule out the possibility that at least some young CWS develop greater negative affectivity due to their experiences with stuttering. One recent review indicated that anxiety related to stuttering increases over time (Smith, Iverach, O’Brian, Kefalianos, & Reilly, 2014). Interestingly, the present authors found no significant correlation between young CWS’ duration of experience with stuttering (i.e., time since onset) and their LPP amplitude to unpleasant pictures (r= .07, p=.8). However, for some of the CWS, it is possible that they develop the negative reactions resulting from their experience(s) with stuttering. For some of the CWS who fall into this category, the “spread of reactivity” might also impact the developmental course of their stuttering frequency and severity. Perhaps, greater levels of anxiety and negative reactions contribute to an increase in their stuttering frequency, placing them at greater risk for stuttering persistence. Of course, such speculation needs to be confirmed or refuted by means of future empirical longitudinal study.
The second, or predispositional model, suggests that between-group differences in LPP amplitude are due to different emotion regulation skills between CWS and CWNS. It should be noted, that emotional reactivity occurs in the context of emotion and attention regulation (Gross, 1998; Rothbart, 2011). Emotion regulation refers to the processes involved in modifying negative and positive emotions in relation to their intensity, duration, how they are expressed and when they occur (Gross, 1998). According to this “regulatory” hypothesis, CWS exhibit higher LPP amplitude when viewing unpleasant pictures because CWS have lower regulatory skills than CWNS.
Although other empirical studies employing behavioral and/or psychophysiological measures would seem to support such conjecture, the present study found no significant between-group differences in reappraisal/emotional regulation in terms of LPP amplitudes. Further, the present study’s use of reappraisal stories – only one of several different emotional regulation strategies that children can possibly use – may have been too developmentally advanced for 4- to 6- year-old children. Perhaps, children of the age of the participants in the present study were not cognitively mature enough to down-regulate their emotion by using the reappraisal stories presented to them. Such a possibility has been raised by others studying LPP in young children (e.g., Dennis & Hajcak, 2009; DeCicco, O’Toole, Dennis, 2014; DeCicco, Solomon, Dennis, 2012; cf. Hua, Han, Zhou, 2015). If such regulatory skills are not adequately developed and/or established in young children (again, suggesting that they are less able to “take advantage” of emotion reappraisal), this regulatory hypothesis may not be the best explanation for between-group differences in LPP amplitude (i.e., emotional reactivity) during unpleasant pictures.
A third possible explanation, also involving the predispositional model, suggests that young CWS, when compared to CWNS, are more emotionally reactive to unpleasant stimuli as a result of their temperamental proclivities. This “temperamental” or “biologically-based filter” hypothesis suggests that young CWS may differ from their CWNS peers in terms of some aspects of their temperament that lead CWS to “filter” (and hence react) differently to unpleasant stimuli. In contrast to the aforementioned “spread of reactivity” hypothesis that emphasizes the amount or duration of children’s experience with stuttering, this “temperamental” hypothesis places the emphasis on children’s “constitutionally” based processes that selectively filter the quantity and quality of events/stimuli the child experiences. Thus, just as experience with stuttering cannot be categorically ruled out as an explanation for CWS’s greater reactivity to unpleasant pictures, neither can one rule out the possibility that inherent temperamental “filters” contribute to these findings. For example, present findings indicate that CWS with lower Effortful Control (i.e., a measure of emotion regulation) exhibited greater LPP amplitudes for unpleasant pictures. Although this finding is correlational, it is consistent with the notion that temperamentally-related emotional regulation may impact other, concurrent processes.
Lastly, the fourth possible explanation, or correlational model, suggests that the association between LPP reactivity and temperament might be due to a third-order (e.g., genetic, environmental factors) variable that impacts both LPP reactivity and temperament. According to this model, a third-order variable that wasn’t measured or investigated in the present study might have influenced the observed significant correlations between CWS’s temperamental characteristics and their LPP reactivity. Again, determining such possibilities must await future empirical study.
While present findings do not categorically refute or support the aforementioned accounts of the association between emotion and stuttering- something that could also be said about empirical studies of the association between brain, linguistic, motoric, or social anxiety processes and stuttering- they do seem consistent with the notion that inherent “filtering” (i.e., temperament) together with environmental stimuli may impact children’s emotional reactivity. Supporting this conjecture is Rothbart’s (2011) description of Escalona’s (1968) notion of effective experience, “…events in children’s lives are experienced only as they are filtered through the individual child’s nervous system, so that an environmental event is not the same for all” (p. 30). Hence, it does not seem unreasonable to suggest that individual differences in such “filtering systems” impact reactions to internal and/or external stimuli, be they pleasant, unpleasant or neutral. As empirical work in this area progresses, perhaps some melding of the “spread of reactivity” and “biologically-based filters” hypotheses may appear to best explain findings like those of the current study. However, at present, such explanations must await the findings of future studies to help determine the relative contributions of these “nature” (temperamental) and “nurture” (spread of reactivity) to emotional processes associated with childhood stuttering.
4.3 Caveats
One limitation of the present study was the relatively small sample size and relatively restricted age range of participants. These may be a limiting factor for generalizing findings to the broader group of CWS and CWNS. Further, due to various reasons discussed in the Methods section (e.g., fatigue, missing data from caregivers), some of the participants tested for Hypotheses 1 and 2 did not provide data needed to test Hypotheses 3 and 4. Perhaps, differences in the number of participants may have resulted in different levels of statistical power for the hypotheses tested. Going forward, a larger sample with a wider age range (e.g., preschool-age children as well as school-age children) would, for example, permit a more robust comparison between younger and older children’s use of reappraisal strategies to down-regulate emotions.
A second limitation relates to the use of directed reappraisal as a means of emotional regulation. Neither young CWS nor their CWNS peers appeared to use positive reappraisal stories to down-regulate their emotions. Others have suggested that children in preschool and kindergarten are still developing their ability to make use of advanced emotion regulation strategies like cognitive reappraisal (e.g., DeCicco et al., 2012). Thus, future research, especially involving preschool-age children, may benefit from augmenting the emotion regulation strategy of reappraisal with other such strategies such as distraction and labeling (e.g., Moyal, Henik, Anholt, 2013). It is important to note that in the present study, positive and neutral reappraisal stories showed a similar impact on children’s LPP to unpleasant pictures. Perhaps the neutral reappraisal stories (e.g., “Next, you will see a picture of an animal in nature. It has thick brown fur”) used in this study were not perceived as being truly “neutral,” and rather were perceived by children as an emotion regulation strategy. Indeed, some researchers reported that manipulating participants’ attention to the neutral aspects of unpleasant pictures was associated with a decreased LPP, in comparison to highlighting more arousing aspects of unpleasant pictures (Dunning and Hajcak, 2009; Hajcak et al., 2009). Thus, besides using emotion regulation strategies other than reappraisal, future studies may also benefit from using other “neutral” stimuli such as reverse speech.
A third limitation is that the present study employed one measure of children’s emotional reactivity and regulation –EEG recordings of LPP amplitude– without consideration of other independent indices of emotional reactivity such as skin conductance (Boucsein, 1992) and emotion regulation such as respiratory sinus arrhythmia (Porges, 2011) to examine the impact of the experimental stimuli and reappraisal strategies.
Finally, the false discovery rate (FDR) correction resulted in a very conservative level of significance for each correlation (e.g., for CWS a relatively strong and significant correlation [r = 0.595] between surgency and LPP during pleasant pictures was non-significant after applying a stringent FDR correction). Perhaps in future studies, one could employ a more planned approach to such correlations, thus reducing the number of correlations and the need for stringent corrections for multiple comparisons.
5. Conclusion
Present findings that CWS exhibited greater LPP amplitude to unpleasant pictures than their CWNS peers are consistent with non-physiological findings suggesting an association between emotion and childhood stuttering. Furthermore, significant correlations between parental reports of various temperament-related constructs and CWS’s LPP amplitude point out the possibility that aspects of CWS’s temperament play a contributing, though not necessarily singular, role in cortical reactivity to emotional stimuli. Thus, present findings provide further empirical support for the notion that emotional processes are associated with childhood stuttering.
In conclusion, to the current authors’ knowledge, the present empirical study was one of the first of its kind to investigate emotional reactivity and emotion regulation in a sample of young children who do and do not stutter using cortical responses (LPP) and to demonstrate the association between neural activity and caregiver reports of participants’ temperamental characteristics. Replication and extension of this initial investigation would be most welcome. For with the pooling of knowledge from this and subsequent similar studies, it should be possible to develop a more comprehensive understanding of the association between emotion and childhood stuttering.
Highlights.
Children who stutter are more reactive to unpleasant stimuli than typical peers
Children who stutter’s temperament correlated with cortical activity of emotion
Emotional processes need inclusion in the study of childhood stuttering
Acknowledgments
The authors would like to thank Dr. Jim Bodfish for reviewing earlier drafts of this article, and for providing valuable feedback regarding content. We also would like to thank Miles Jefferson for his assistance in inter-reliability for disfluency coding. We would like to extend our sincere appreciation to the participants and their families who made this study possible.
This work was supported in part by a CTSA grant UL1 TR000445 from NCATS/NIH, National Stuttering Association Canadeo Family research Award, as well as NIDCD/NIH research grant R01 DC000523. The research reported herein does not reflect the views of the NIH, NCHD, or the University.
Biographies
Hatun Zengin-Bolatkale, PhD CCC-SLP is a Research Assistant Professor at Department of Hearing and Speech Sciences at Vanderbilt University. Dr. Zengin-Bolatkale’s primary research interest is investigating neural correlates of cognitive and emotional contributions to developmental disorders, specifically, childhood stuttering.
Edward G. Conture, PhD CCC-SLP is an Emeritus Professor of Department of Hearing and Speech Sciences at Vanderbilt University. Dr. Conture has published extensively in the field of childhood stuttering. During his 40+ years of career, Dr. Conture has clinically served, trained future clinicians, taught courses and conducted research regarding stuttering, with particular reference to preschool-age children and their families.
Alexandra (Sasha) Key, PhD is a Research Associate Professor at Vanderbilt Kennedy Center and Hearing and Speech Sciences. Dr. Key’s research examines brain mechanisms supporting cognitive strengths and weaknesses in persons with developmental disabilities. Dr. Key’s research interests focus on the psychophysiological indices (EEG/ERP, eye tracking) of sensory and cognitive processes, their use for understanding mechanisms of deficits in intellectual and developmental disabilities and for documenting treatment effects.
Tedra A. Walden, PhD is a Professor of Psychology and Human Development at Vanderbilt University. Her research focuses on social and emotional development of young children and the role of emotions in childhood stuttering.
Robin M. Jones, PhD CCC-SLP is an Assistant Professor at Department of Hearing and Speech Sciences at Vanderbilt University. Dr. Jones’s research focuses on emotional and speech-language contributions to the pathogenesis of childhood stuttering. Dr. Jones is investigating possible mechanisms by which emotion and cognitive processes may disrupt speech-language planning and production and contribute to stuttering.
Appendices
Appendix A
The International Affective Picture System (IAPS; Lang et al., 2005) stimuli for Emotional Reactivity (Table A1) and (2) Emotion Regulation (Table A2) Tasks.
Table A1. Emotional Reactivity Task.
The numbers in this table represent the IAPS number for each picture that was used in the present study
| Unpleasant: | 1050, 1120, 1201, 1300, 1321, 1930, 2120, 2130, 2688, 2780, 2810, 2900, 3022, 3230, 3280, 5970, 6190, 6300, 7380, 9050, 9250, 9404, 9421, 9470, 9480, 9490, 9582, 9594, 9600, 9611 |
| Pleasant: | 1460, 1463, 1601, 1610, 1710, 1750, 1811, 1920, 1999, 2070, 2091, 2165, 2224, 2311, 2340, 2345, 2791, 4603, 5831, 7325, 7330, 7400, 7502, 8031, 8330, 8380, 8461, 8490, 8496, 8620 |
| Neutral: | 5220, 5711, 5740, 5750, 5800, 5820, 7000, 7002, 7004, 7006, 7009, 7010, 7025, 7031, 7035, 7041, 7050, 7080, 7090, 7100, 7140, 7150, 7175, 7190, 7224, 7233, 7235, 7236, 7595, 7950 |
Table A2.
The 30 neutral and 30 positive reappraisal stories used while participants viewed unpleasant pictures during the two Emotion Regulation conditions.
| IAPS Picture | Neutral Reappraisal Stories | Positive Reappraisal Stories |
|---|---|---|
| 1050 | Please look at the following picture. It has green and black colors. | This is a snake that is completely harmless; it doesn’t even have teeth. |
| 1120 | Next, you will see a picture of an animal that has a big mouth. | This snake is also harmless. He is just hungry and begging for a snack. |
| 1201 | Next is a picture of an animal and a person in a white shirt. | This person really likes to let his harmless pet spider crawl on his shoulder. |
| 1300 | Look at the screen. Next picture is in dark brown and black colors. | This dog just went to the dentist – look how clean her teeth are. |
| 1321 | Next, you will see a picture of an animal in nature. It has thick brown fur. | Look at the next picture. This is a friendly and harmless bear that is yawning. |
| 1930 | Please look at the following picture. It is a picture of an animal in water. | This is a well-trained, friendly shark whose keeper is feeding him a fish. |
| 2120 | Look at the following picture of a man wearing a black sweater. | This man has held his breath to make a red face. He is being funny by showing how red his face can get. |
| 2130 | Next is a picture of a woman wearing a coat. She has black hair. | This woman had her picture taken right when she was about to sneeze. |
| 2688 | Next picture shows an animal and a man located far away from the camera. | The doctor is shooting medicine into the bear. It will get better thanks to the friendly doctor. |
| 2780 | Next picture shows a man who painted his face using white and black colors. | This is a friendly actor who is in a play about cats. He is smiling at you. |
| 2810 | This is a picture of a child who is ten years old. He has brown hair, and his eyes are closed. | This boy is playing ‘hide and seek’ and is yelling ‘ready or not here I come’. |
| 2900 | This is a picture of a boy who is in kindergarten. He has blonde hair and a white shirt. | This boy’s dad picked him up after he fell; he got scared, but is now totally okay. |
| 3022 | Look at the following picture. This is a picture of a man with long hair. | Look at the next picture. This person is on a very fun ride and he is so excited. |
| 3230 | Please look at the screen. Next is a picture of an old person. | This man was sick, but he is okay now and on his way to getting all better. |
| 3280 | This is a picture of a woman and a little boy. The woman is wearing glasses and gloves. | This boy is having his teeth cleaned; it feels funny but doesn’t hurt. |
| 5970 | This picture is in gray and black. It is a picture of a storm. | This storm is going away; it is harmless and everyone is safe. |
| 6190 | Please look at the monitor and wait for the next picture. It is a picture of an object. | This is a toy gun; the person holding it is playing cops and robbers. |
| 6300 | Next picture is in black, white and gray colors. It shows a man’s hand. | This man is using the tool to fix the wall. He is being very helpful to his friends. |
| 6370 | Please look at the next picture. It is a picture of a gray mask. | This person is visiting his friend; he is wearing a mask because it is cold. |
| 7380 | This is a picture showing some animals and a plate of food. | These bugs are completely harmless. They’re a little hungry and eating the delicious leftover pizza. |
| 9050 | Next is a picture of a group of people and a plane in the back. | Look at the following picture. Luckily, everyone was okay after the plane crash. |
| 9250 | Please look at the next picture. This is a picture of three women. | These doctors have saved the woman’s life. The woman will be all okay thanks to them. |
| 9421 | Next is a picture that shows two men wearing army uniforms. | This solider is so happy he is crying; the war is over and he is going to his family. |
| 9470 | Next is a picture of a white, big, old building. It used to be a hotel. | This very old building was knocked down; everyone was really happy to see it go. |
| 9480 | Look at the following picture. This picture has brown, white, black and green colors. | These are bones of ancient people. They are very helpful for scientists to learn about the past. |
| 9490 | Following shows a picture of a person covered in something gray. | This is a pretend mummy for a movie set; it is fake and very funny looking. |
| 9582 | Following is a picture of a boy who is closing his eyes and opening his mouth. | The doctor just finished working on his teeth; he feels all better. |
| 9594 | This picture shows a person’s arm and someone wearing white gloves and a blue robe. | This person gives blood all the time because it helps people who are sick. |
| 9600 | Next picture shows a very big, white ship in the big, blue waves of an ocean. | Another ship came just in time to save all these people; everyone will make it just fine. |
| 9611 | Next picture has blue, brown, white, yellow and red colors. | A brand new plane will be built from this old one – people are working on it together. |
Appendix B
Figure B1.

The present study’s measure of LPP amplitude was based on the average evoked response potentials across the 7 electrodes within the red circle. This red-circled cluster of electrodes corresponds to parietal location (Pz).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
It is important to note that although the Arnold et al’s (2011) empirical study of cortical associates of emotion in CWS reported no significant between group differences in these associates, there are significant differences in methodology between the current ERP study and the Arnold et al. (2011) EEG study.
References
- Ambrose NG, Yairi E, Loucks TM, Seery CH, Throneburg R. Relation of motor, linguistic and temperament factors in epidemiologic subtypes of persistent and recovered stuttering: Initial findings. Journal of fluency disorders. 2015 doi: 10.1016/j.jfludis.2015.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Speech–Language–Hearing Association. Guidelines for screening for hearing impairment and middle-ear disorders. ASHA. 1990;32:17–24. [PubMed] [Google Scholar]
- Anderson J, Pellowski M, Conture E, Kelly E. Temperamental characteristics of young children who stutter. Journal of Speech, Language and Hearing Research. 2003;45:20–34. doi: 10.1044/1092-4388(2003/095). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnold HS, Conture EG, Key APF, Walden T. Emotional reactivity, regulation and childhood stuttering: a behavioral and electrophysiological study. Journal of Communication Disorders. 2011;44:276–293. doi: 10.1016/j.jcomdis.2010.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babkirk S, Rios V, Dennis TA. The late positive potential predicts emotion regulation strategy use in school-aged children concurrently and two years later. Developmental Science. 2015;18(5):832–841. doi: 10.1111/desc.12258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the royal statistical society Series B (Methodological) 1995:289–300. [Google Scholar]
- Boucsein W. Electrodermal activity. New York: Plenum Press; 1992. [Google Scholar]
- Bradley MM, Lang PJ. Measuring emotion: the self-assessment manikin and the semantic differential. Journal of behavior therapy and experimental psychiatry. 1994;25(1):49–59. doi: 10.1016/0005-7916(94)90063-9. [DOI] [PubMed] [Google Scholar]
- Bradley MM, Sabatinelli D, Lang PJ, Fitzsimmons JR, King W, Desai P. Activation of the visual cortex in motivated attention. Behavioral neuroscience. 2003;117(2):369–380. doi: 10.1037/0735-7044.117.2.369. [DOI] [PubMed] [Google Scholar]
- Bush A. Effects of childhood stuttering on attention regulation in emotionally arousing situations. Vanderbilt University; Nashville: 2006. [Google Scholar]
- Campos JJ, Frankel CB, Camras L. On the nature of emotion regulation. Child Development. 2004;75:377–394. doi: 10.1111/j.1467-8624.2004.00681.x. [DOI] [PubMed] [Google Scholar]
- Chatham CH, Frank MJ, Munakata Y. Pupillometric and behavioral markers of a developmental shift in the temporal dynamics of cognitive control. Proc Natl Acad Sci USA. 2009;106:5529–5533. doi: 10.1073/pnas.0810002106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi D, Conture E, Walden T, Jones R, Kim H. Emotional diathesis, emotional stress and childhood stuttering. Journal of Speech, Language, and Hearing Research. 2016;59:616–630. doi: 10.1044/2015_JSLHR-S-14-0357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi D, Conture EG, Walden T, Lambert WE, Tumanova V. Behavioral inhibition and childhood stuttering. Journal of Fluency Disorders. 2013;38(2):171–183. doi: 10.1016/j.jfludis.2013.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conture EG. Stuttering: Its nature, diagnosis, and treatment. Needham Heights: MA, Allyn and Bacon; 2001. [Google Scholar]
- Conture E, Walden T. Dual diathesis-stressor model of stuttering. In: Filatova YuO., editor. Theoretical Issues of Fluency Disorders. Moscow: National Book Centre; 2012. pp. 94–127. [Google Scholar]
- Cuthbert BN, Schupp HT, Bradley MM, Birbaumer N, Lang PJ. Brain potentials in affective picture processing: covariation with autonomic arousal and affective report. Biol Psychol. 2000;52:95–111. doi: 10.1016/s0301-0511(99)00044-7. [DOI] [PubMed] [Google Scholar]
- Dennis TA, Hajcak G. The late positive potential: a neurophysiological marker for emotion regulation in children. Journal of Child Psychology and Psychiatry. 2009;50(11):1373–1383. doi: 10.1111/j.1469-7610.2009.02168.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeCicco JM, O’Toole LJ, Dennis TA. Cognitive reappraisal in children: an ERP study using the late positive potential. Developmental neuropsychology. 2014;39(7):497. doi: 10.1080/87565641.2014.959171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeCicco JM, Solomon B, Dennis TA. Neural correlates of cognitive reappraisal in children: an ERP study. Developmental cognitive neuroscience. 2012;2(1):70–80. doi: 10.1016/j.dcn.2011.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derryberry D, Rothbart MK. Reactive and effortful processes in the organization of temperament. Development and Psychopathology. 1997;9:633–652. doi: 10.1017/s0954579497001375. [DOI] [PubMed] [Google Scholar]
- Doré B, Silvers J, Ochsner K. Toward a personalized science of emotion regulation. Social and Personality Psychology Compass. 2016;10(4):171–187. doi: 10.1111/spc3.12240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunn M, Dunn LM. Peabody Picture Vocabulary Test—4. Circle Pines, MN: AGS; 2007. [Google Scholar]
- Dunning JP, Hajcak G. See no evil: directing visual attention within unpleasant images modulates the electrocortical response. Psychophysiology. 2009;46(1):28–33. doi: 10.1111/j.1469-8986.2008.00723.x. [DOI] [PubMed] [Google Scholar]
- Eggers K, De Nil LF, van den Bergh BRH. Factorial Temperament Structure in Stuttering, Voice-Disordered, and Typically Developing Children. Journal of Speech, Language, and Hearing Research. 2009;52:1610–1622. doi: 10.1044/1092-4388(2009/07-0065). [DOI] [PubMed] [Google Scholar]
- Eggers K, De Nil LF, van den Bergh BRH. Temperament dimensions in stuttering and typically developing children. Journal of Fluency Disorders. 2010;35(4):355–372. doi: 10.1016/j.jfludis.2010.10.004. [DOI] [PubMed] [Google Scholar]
- Eggers K, De Nil LF, Van den Bergh BRH. The efficiency of attentional networks in children who stutter. Journal of Speech, Language, and Hearing Research. 2012;55(3):946–959. doi: 10.1044/1092-4388(2011/10-0208). [DOI] [PubMed] [Google Scholar]
- Eggers K, De Nil LF, Van den Bergh BRH. Inhibitory control in childhood stuttering. Journal of Fluency Disorders. 2013;38(1):1–13. doi: 10.1016/j.jfludis.2012.10.001. [DOI] [PubMed] [Google Scholar]
- Embrechts M, Ebben H, Franke P, van de Poel C. Temperament: A comparison between children who stutter and children who do not stutter. In: Bosshardt H-G, Yaruss J, Peters H, editors. Proceedings of the Third World Congress on Fluency Disorders: Theory, research, treatment and self-help. University of Mijmegen; The Netherlands: Nimegen Press; 2000. pp. 557–562. [Google Scholar]
- Erdemir A, Walden TA, Jefferson CM, Choi D, Jones RM. The effect of emotion on articulation rate in persistence and recovery of childhood stuttering. Journal of Fluency Disorders. 2018;56:1–17. doi: 10.1016/j.jfludis.2017.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escalona S. The roots of individuality: Normal patterns of development in infancy. Chicago: Aldine; 1968. [Google Scholar]
- Felsenfeld S, van Beijsterveldt CEM, Boomsma DI. Attentional Regulation in Young Twins With Probable Stuttering, High Nonfluency, and Typical Fluency. Journal of Speech, Language, and Hearing Research. 2010;53(5):1147–1166. doi: 10.1044/1092-4388(2010/09-0164). [DOI] [PubMed] [Google Scholar]
- Fisher RA. On the probable error of a coefficient of correlation deduced from a small sample. Metron. 1921;(1):3–32. [Google Scholar]
- Foti D, Hajcak G. Deconstructing reappraisal: descriptions preceding arousing pictures modulate the subsequent neural response. J Cogn Neurosci. 2008;20:977–988. doi: 10.1162/jocn.2008.20066. [DOI] [PubMed] [Google Scholar]
- Goldin PR, McRae K, Ramel W, Gross JJ. The neural bases of emotion regulation: reappraisal and suppression of negative emotion. Biological psychiatry. 2008;63(6):577–586. doi: 10.1016/j.biopsych.2007.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman R, Fristoe M. Goldman-Fristoe Test of Articulation-2. 2nd. Circle Pines, MN: American Guidance Services, Inc.; 2000. [Google Scholar]
- Gross JJ. The emerging field of emotion regulation: An integrative review. Review of General Psychology. 1998;2:271–299. [Google Scholar]
- Gross JJ. Emotion regulation: Affective, cognitive, and social consequences. Psychophysiology. 2002;39:281–291. doi: 10.1017/s0048577201393198. [DOI] [PubMed] [Google Scholar]
- Gross J, John O. Individual differences in two emotion regulation processes: Implications for affect, relationships and well-being. Journal of Personality and Social Psychology. 2003;85:34–362. doi: 10.1037/0022-3514.85.2.348. [DOI] [PubMed] [Google Scholar]
- Gross J. Emotion regulation: Current status and future prospects. Psychological Inquiry. 2015;26:1–26. [Google Scholar]
- Hajcak G, Dennis TA. Brain potentials during affective picture viewing in children. Biol Psychol. 2009;80:333–338. doi: 10.1016/j.biopsycho.2008.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajcak G, Dunning JP, Foti D. Motivated and controlled attention to emotion: Time-course of the late positive potential. Clinical Neurophysiology. 2009;120(3):505–510. doi: 10.1016/j.clinph.2008.11.028. [DOI] [PubMed] [Google Scholar]
- Hajcak G, MacNamara A, Olvet DM. Event-Related Potentials, Emotion, and Emotion Regulation: An Integrative Review. Developmental Neuropsychology. 2010;35(2):129–155. doi: 10.1080/87565640903526504. [DOI] [PubMed] [Google Scholar]
- Hajcak G, Nieuwenhuis S. Reappraisal modulates the electrocortical response to negative pictures. Cogn Affect Behav Neurosci. 2006;6:291–297. doi: 10.3758/cabn.6.4.291. [DOI] [PubMed] [Google Scholar]
- Hajcak G, Olvet DM. The persistence of attention to emotion: brain potentials during and after picture presentation. Emotion. 2008;8:250–255. doi: 10.1037/1528-3542.8.2.250. [DOI] [PubMed] [Google Scholar]
- Henderson HA, Wachs TD. Temperament theory and the study of cognition–emotion interactions across development. Developmental Review. 2007;27(3):396–427. [Google Scholar]
- Howell DC. Statistical methods for psychology. Cengage Learning 2011 [Google Scholar]
- Howell P, Davis S, Patel H, Cuniffe P, Downing-Wilson D, Au-Yeung J, Williams R. Fluency development and temperament in fluent children and children who stutter. In: Packman A, Meltzer A, Peters HFM, editors. Theory, research and therapy in fluency disorders Proceedings of the fourth world congress on fluency disorders. Nijmegen, The Netherlands: University Press Nijmegen; 2004. pp. 250–256. [Google Scholar]
- Hresko W, Reid D, Hammill D. Test of Early Language Development-3. Austin, TX: Pro-Ed; 1999. [Google Scholar]
- Hua M, Han ZR, Zhou R. Cognitive reappraisal in preschoolers: neuropsychological evidence of emotion regulation from an ERP study. Developmental neuropsychology. 2015;40(5):279–290. doi: 10.1080/87565641.2015.1069827. [DOI] [PubMed] [Google Scholar]
- Johnson KN, Walden T, Conture E, Karass J. Spontaneous regulation of emotions in preschool children who stutter: preliminary findings. Journal of Speech, Language, and Hearing Research. 2010;53(6):1478–95. doi: 10.1044/1092-4388(2010/08-0150). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones R, Walden T, Conture E, Erdemir A, Lambert W, Porges S. Executive functions impact the relation between respiratory sinus arrhythmia and frequency of stuttering in young children who do and do not stutter. Journal of Speech Language and Hearing Research. doi: 10.1044/2017_JSLHR-S-16-0113. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones RM, Buhr AP, Conture EG, Tumanova V, Walden TA, Porges SW. Autonomic nervous system activity of preschool-age children who stutter. Journal of fluency disorders. 2014;41:12–31. doi: 10.1016/j.jfludis.2014.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones R, Conture E, Walden T. Emotional reactivity and regulation initiated prior to and during (dis)fluent utterances of preschool-age children who stutter. Journal of Communication Disorders. 2014b;48:38–51. doi: 10.1016/j.jcomdis.2014.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones RM, Walden TA, Conture EG, Erdemir A, Lambert WE, Porges SW. Executive Functions Impact the Relation Between Respiratory Sinus Arrhythmia and Frequency of Stuttering in Young Children Who Do and Do Not Stutter. Journal of Speech, Language, and Hearing Research. 2017;60(8):2133–2150. doi: 10.1044/2017_JSLHR-S-16-0113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karass J, Walden T, Conture E, Graham C, Arnold H, Hartfield K, et al. Relation of emotional reactivity and regulation to childhood stuttering. Journal of Communication Disorders. 2006;39:402–423. doi: 10.1016/j.jcomdis.2005.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kefalianos E, Onslow M, Ukoumunne O, Block S, Prior M, Reilly S. Stuttering, temperament and anxiety: Data from a community cohort aged 2-4 years. Journal of Speech, Language and Hearing Research. 2014;57:1314–1322. doi: 10.1044/2014_JSLHR-S-13-0069. [DOI] [PubMed] [Google Scholar]
- Kraft SJ, Ambrose N, Chon H. Seminars in speech and language. 2. Vol. 35. Thieme Medical Publishers; 2014. Temperament and environmental contributions to stuttering severity in children: The role of effortful control; pp. 80–94. [DOI] [PubMed] [Google Scholar]
- Lang PJ, Bradley MM, Cuthbert BN. International affective picture system (IAPS): Affective ratings of pictures and instruction manual. University of Florida; Gainesville, FL: 2005. [Google Scholar]
- Leppink J, O’Sullivan P, Winston K. Evidence against vs. in favour of a null hypothesis. Perspectives on medical education. 2017;6(2):115–118. doi: 10.1007/s40037-017-0332-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis K, Goldberg L. Measurements of temperament in the identification of children who stutter. European Journal of Disorders of Communication. 1997;32:441–448. doi: 10.3109/13682829709082258. [DOI] [PubMed] [Google Scholar]
- Liu Y, Huang H, McGinnis-Deweese M, Keil A, Ding M. Neural substrate of the late positive potential in emotional processing. Journal of Neuroscience. 2012;32(42):14563–14572. doi: 10.1523/JNEUROSCI.3109-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacNamara A, Hajcak G. Anxiety and spatial attention moderate the electrocortical response to aversive pictures. Neuropsychologia. 2009;47(13):2975–2980. doi: 10.1016/j.neuropsychologia.2009.06.026. [DOI] [PubMed] [Google Scholar]
- MacNamara A, Ochsner KN, Hajcak G. Previously reappraised: the lasting effect of description type on picture-elicited electrocortical activity. Soc Cogn Affect Neurosci. 2011;6:348–358. doi: 10.1093/scan/nsq053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore T. Event-related potential (ERP) data exploration made easy. 2015 https://CRAN.R-project.org/package=erp.easy.
- Moran TP, Jendrusina AA, Moser JS. The psychometric properties of the late positive potential during emotion processing and regulation. Brain Research. 2013;1516:66–75. doi: 10.1016/j.brainres.2013.04.018. [DOI] [PubMed] [Google Scholar]
- Moyal N, Henik A, Anholt GE. Cognitive strategies to regulate emotions—current evidence and future directions. Frontiers in psychology. 2013;4 doi: 10.3389/fpsyg.2013.01019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ntourou K, Conture EG, Walden TA. Emotional reactivity and regulation in preschool-age children who stutter. Journal of Fluency Disorders. 2013;38(3):260–274. doi: 10.1016/j.jfludis.2013.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ochsner KN, Gross JJ. The cognitive control of emotion. Trends in cognitive sciences. 2005;9(5):242–249. doi: 10.1016/j.tics.2005.03.010. [DOI] [PubMed] [Google Scholar]
- Ortega A, Ambrose N. Developing physiologic stress profiles for school-age children who stutter. Journal of Fluency Disorders. 2011;36:268–273. doi: 10.1016/j.jfludis.2011.04.007. [DOI] [PubMed] [Google Scholar]
- Pastor MC, Bradley MM, Löw A, Versace F, Moltó J, Lang PJ. Affective picture perception: emotion, context, and the late positive potential. Brain research. 2008;1189:145–151. doi: 10.1016/j.brainres.2007.10.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picton TW, Bentin S, Berg P, Donchin E, Hillyard SA, Johnson SA, Johnson R, Miller GR, Ritter W, Ruchkin DS, Rugg MD, Taylor MJ. Guidelines for using human event-related potentials to study cognition: Recording standards and publication criteria. Psychophysiology. 2000;37:127–152. [PubMed] [Google Scholar]
- Porges S. The Polyvagal Theory: Neurophysiological foundations of emotions, attachment, communication and self-regulation. New York: W.W. Norton & Company; 2011. [Google Scholar]
- Posner MI, Rothbart MK. Developing mechanisms of self-regulation. Development and Psychopathology. 2000;12:427–441. doi: 10.1017/s0954579400003096. [DOI] [PubMed] [Google Scholar]
- E-Prime [2.0] Pittsburgh PA: Psychology Software Tools, Inc; 2012. Retrieved from http://www.pstnet.com. [Google Scholar]
- Riley GD. Stuttering severity instrument for children and adults. 3rd. Austin, TX: Pro-Ed; 1994. [DOI] [PubMed] [Google Scholar]
- Rothbart MK. Becoming who we are: Temperament and personality in development. New York: Guilford Press; 2011. [Google Scholar]
- Rothbart MK, Ahadi SA, Evans DE. Temperament and personality: Origins and outcomes. Journal of Personality and Social Psychology. 2000;78(1):122–135. doi: 10.1037//0022-3514.78.1.122. [DOI] [PubMed] [Google Scholar]
- Rothbart MK, Ahadi SA, Hershey KL, Fisher P. Investigations of temperament at 3-7 years: the Children’s Behavior Questionnaire. Child Dev. 2001;72:1394–1408. doi: 10.1111/1467-8624.00355. [DOI] [PubMed] [Google Scholar]
- RStudio Team. RStudio: Integrated Development for R. RStudio, Inc.; Boston, MA: 2015. http://www.rstudio.com/ [Google Scholar]
- Schupp HT, Cuthbert BN, Bradley MM, Cacioppo JT, Ito T, Lang PJ. Affective picture processing: the late positive potential is modulated by motivational relevance. Psychophysiology. 2000;37:257–261. [PubMed] [Google Scholar]
- Schwenk KA, Conture EG, Walden TA. Reaction to background stimulation of preschool children who do and do not stutter. Journal of communication disorders. 2007;40(2):129–141. doi: 10.1016/j.jcomdis.2006.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith KA, Iverach L, O’Brian S, Kefalianos E, Reilly S. Anxiety of children and adolescents who stutter: A review. Journal of Fluency Disorders. 2014;40:22–34. doi: 10.1016/j.jfludis.2014.01.003. [DOI] [PubMed] [Google Scholar]
- Smith A, Weber C. How stuttering develops: The multifactorial dynamic pathways theory. Journal of Speech, Language, and Hearing Research. 2017;60(9):2483–2505. doi: 10.1044/2017_JSLHR-S-16-0343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solomon B, DeCicco JM, Dennis TA. Emotional picture processing in children: An ERP study. Developmental Cognitive Neuroscience. 2012;(2):110–119. doi: 10.1016/j.dcn.2011.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tumanova V, Conture EG, Lambert EW, Walden TA. Speech disfluencies of preschool-age children who do and do not stutter. Journal of communication disorders. 2014;49:25–41. doi: 10.1016/j.jcomdis.2014.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Merwe B, Robb MP, Lewis JG, Osmond T. Anxiety measures and salivary cortisol responses in preschool children who stutter. Contemporary Issues in Communication Science and Disorders. 2011;38:1–10. [Google Scholar]
- Walden TA, Frankel CB, Buhr AP, Johnson KN, Conture EG, Karrass JM. Dual diathesis-stressor model of emotional and linguistic contributions to developmental stuttering. Journal of Abnormal Child Psychology. 2012;40:633–644. doi: 10.1007/s10802-011-9581-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westfall P, Tobias R, Rom D, Wolfinger R, Hochberg Y. Multiple comparisons and multiple tests using SAS. Cary, NC: SAS Institute; 1999. [Google Scholar]
- Williams M. Children who stutter: Easy, difficult, or slow to warm up; 2006, November; Paper presented at the Annual Conference of the American Speech, Language, and Hearing Association; Miami, Florida. [Google Scholar]
- Yairi E, Ambrose N. Early childhood stuttering. Austin: Pro-Ed, Inc.; 2005. [Google Scholar]
- Yaruss JS. Real-Time analysis of speech fluency: Procedures and reliability training. American Journal of Speech-Language Pathology. 1998;7:25–37. [Google Scholar]
- Zengin-Bolatkale H, Conture EG, Walden AT. Sympathetic arousal of preschool-age children who stutter during a stressful picture-naming task. Journal of Fluency Disorders. 2014;46:24–40. doi: 10.1016/j.jfludis.2015.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]


