Abstract
To evaluate whether α-smooth muscle actin (α-SMA) plays a role in fibroblast contractility, we first compared the contractile activity of rat subcutaneous fibroblasts (SCFs), expressing low levels of α-SMA, with that of lung fibroblasts (LFs), expressing high levels of α-SMA, with the use of silicone substrates of different stiffness degrees. On medium stiffness substrates the percentage of cells producing wrinkles was similar to that of α-SMA–positive cells in each fibroblast population. On high stiffness substrates, wrinkle production was limited to a subpopulation of LFs very positive for α-SMA. In a second approach, we measured the isotonic contraction of SCF- and LF-populated attached collagen lattices. SCFs exhibited 41% diameter reduction compared with 63% by LFs. TGFβ1 increased α-SMA expression and lattice contraction by SCFs to the levels of LFs; TGFβ-antagonizing agents reduced α-SMA expression and lattice contraction by LFs to the level of SCFs. Finally, 3T3 fibroblasts transiently or permanently transfected with α-SMA cDNA exhibited a significantly higher lattice contraction compared with wild-type 3T3 fibroblasts or to fibroblasts transfected with α-cardiac and β- or γ-cytoplasmic actin. This took place in the absence of any change in smooth muscle or nonmuscle myosin heavy-chain expression. Our results indicate that an increased α-SMA expression is sufficient to enhance fibroblast contractile activity.
INTRODUCTION
Early during healing of an open wound, resident dermal fibroblasts proliferate from the wound margin and migrate into the provisional matrix composed of a fibrin clot. About 1 week after wounding, the provisional matrix is replaced by neo-formed connective tissue, known as granulation tissue, essentially composed of small vessels, extracellular matrix, and fibroblastic cells that become activated and modulate into myofibroblasts. The main feature of myofibroblasts is represented by an important contractile apparatus similar to that of smooth muscle (Gabbiani et al., 1971), and in particular by the neo-expression of α-smooth muscle actin (α-SMA), the actin isoform typical of vascular smooth muscle cells (Skalli et al., 1986). Myofibroblasts are recognized to play a central role in closing the wound tissue, through their capacity to produce a strong contractile force (for review see Grinnell, 1994; Rønnov-Jessen et al., 1996; Powell et al., 1999; Serini and Gabbiani, 1999), possibly generated within stress fibers, similar to those present in cultured fibroblasts (Skalli et al., 1986; Serini and Gabbiani, 1999). Because wound contraction takes place when de novo expressed α-SMA is incorporated in stress fibers (Darby et al., 1990), it has been suggested that this actin isoform plays an important role in granulation tissue contraction (for review see Serini and Gabbiani, 1999). Although stress fibers have been proposed to function as contractile organelles (Burridge, 1981; Katoh et al., 1998) and their presence has been correlated with the production of isometric tension (Harris et al., 1981), at present direct evidence of a functional role of α-SMA in fibroblast contraction is lacking. Here, we have used several in vitro models and approaches in order to test the possible correlation between the expression of α-SMA and the contractile activity of fibroblastic cells.
It is more and more accepted that fibroblastic cells are heterogeneous (for review see Komuro, 1990; Serini and Gabbiani, 1999). A marker of fibroblast heterogeneity is the differential expression of cytoskeletal proteins, including actin isoforms (Sappino et al., 1990). When grown in culture, fibroblasts from different organs constantly modulate into myofibroblast-like cells but show various degrees of α-SMA expression (Xu et al., 1997; Dugina et al., 1998). We have exploited this heterogeneity to compare the contractile potential of cultured rat subcutaneous fibroblasts (SCFs), expressing low levels of α-SMA, with lung fibroblasts (LFs), expressing high levels of α-SMA (Xu et al., 1997; Dugina et al., 1998). In addition, we have enhanced the expression of α-SMA in SCFs by treatment with transforming growth factor β (TGFβ; Desmoulière et al., 1993; Rønnov-Jessen and Petersen, 1993). Conversely, we have reduced α-SMA expression in LFs by blocking the activity of endogenous TGFβ1 with a specific neutralizing antibody, the soluble TGFβ-receptor type II (TGFβ-sR) or the recombinant ED-A fragment (rED-A) of cellular fibronectin (FN; Serini et al., 1998). Finally, in a direct approach to induce expression of α-SMA, we have transiently and stably transfected Swiss 3T3 fibroblasts with α-SMA cDNA.
To determine contractility on a single cell level, we used a modification of the silicone elastomer substrate assay originally developed by Harris et al. (1980), combined with a modification of the force quantification method introduced by Lee et al. (1994). The contractility of whole cell populations was quantified with the use of stress-released collagen lattices (Mochitate et al., 1991; Tomasek et al., 1992; Grinnell et al., 1999b). Our results indicate a correlation between the level of α-SMA expression and fibroblast contraction. In addition, we provide new insights into the interdependence of TGFβ1 and ED-A FN in regulating myofibroblast contractile activity.
MATERIALS AND METHODS
Cell Culture
Fibroblasts from explants of rat subcutaneous and lung tissues and 3T3 fibroblasts were cultured as described previously (Desmoulière et al., 1992). Experiments with primary cell cultures between passage 4 and 8 were performed in MEM (Life Technologies AG, Basel, Switzerland), supplemented with 10% FCS or with 4% Monomed, a defined medium in which fibroblasts do not replicate (Commonwealth Serum Laboratories, Melbourne, Australia; Serini et al., 1998). TGFβ1 (R&D Systems, Inc., Minneapolis, MN) or TGFβ2 (10 ng/ml; gift from Dr. A. Cox, Novartis, Basel, Switzerland), TGFβ-sR (Lin et al., 1995; 0.1–100 ng/ml, gift of Biogen Inc., Cambridge, MA, Komesli et al., 1998) and TGFβ1-neutralizing antibody (0.01–10 μg/ml, R&D Systems) were added for 5 d to the culture medium. rED-A (gift of Biogen Inc.) was incorporated at 300 μg/ml (Serini et al., 1998) into collagen lattices (see below).
Deformable Silicone Substrates and Single Cell Force Measurement
Deformable silicone substrates were essentially prepared as described previously (Harris et al., 1980). Fifty microliters of silicone (poly dimethyl siloxane; 30,000 centistokes; Dow Corning, Midland, MI) were deposited onto a 35-mm round glass coverslip, which was placed into a six-well plate and centrifuged at 1000 rpm for 2 min with the use of a swinging rotor. The silicone surface was then cross-linked by passing it through a Bunsen flame. A rubber ring was sealed with a polyvinylsiloxane dental resin (President MicroSystems; Coltène, Altstätten, Switzerland) on the coverslip, resulting in a small chamber containing at the bottom the cross-linked silicone. Silicone substrates were equilibrated with 0.1% gelatin in Tris-HCl buffer, pH 8.4, sterilized by UV light exposure, and left overnight in the incubator at 37°C.
To compare SCFs and LFs for their capacity to produce wrinkles and for α-SMA expression, after preliminary experiments the flaming time of 1 s was used; this restricted wrinkle formation to fibroblasts with high contractile force (see RESULTS). Cells (30,000) were seeded onto the silicone and grown for 5 d in MEM/10% FCS (±TGFβ). The percentage of cells producing wrinkles was related to the number of α-SMA–positive cells grown in parallel on culture dishes (30,000 cells/30 mm dish; Nunc, Life Technologies), as quantified by immunofluorescence. To visualize wrinkling and α-SMA expression simultaneously, fibroblasts were directly fixed on the silicone substrates and immunostained (see below). α-SMA–positive and wrinkling fibroblasts were calculated as percentage of total cells with the use of a program after manual cell selection on a computer screen (KS400; Carl Zeiss Inc., Jena, Germany). Ten random regions of interest were analyzed per experiment (∼25 cells/field), and at least 5 experiments were performed.
To quantify the force exerted by individual fibroblasts, we produced deformable silicone substrates with different degrees of mechanical resistance by modulating the flaming time (0.5–4 s). Before seeding cells, these substrates were mechanically wrinkled with a deflecting flexible microneedle with a stiffness between 90 and 110 nN/μm (Lee et al., 1994; Fray et al., 1998). A second stiff needle (≥50 μN/μm) was used to fix the substrate at a distance of 200 μm, simulating the substrate-pinching of bipolar cells. The force required to produce first wrinkles on different silicone substrates was calculated from the flexible needle stiffness (μN/μm) and deflection (μm; Oliver et al., 1995) on 15 different regions. LFs were then grown on these substrates and immunostained, and the percentage of wrinkling cells of α-SMA–positive LFs and of α-SMA–negative LFs were calculated separately as described above.
Stressed Collagen Lattice Contraction Assay
To correlate the contractile potential and the level of α-SMA expression in cell populations, fibroblasts were grown in attached (Grinnell, 1994) collagen type I lattices (0.75 mg/ml) as previously described (Tomasek et al., 1992; Pilcher et al., 1994; Rayan et al., 1996). Populations were initiated with 0.25–10.0 × 105 cells/ml collagen, depending on the presence of serum, the cell type, and the myofibroblastic differentiation, and cultured for 5 d. Lattices populated with transiently transfected 3T3 fibroblast were cultured for 2 d in order to work at their maximum transgene activity, determined by immunofluorescence and Western blotting (see below). For contraction measurement, lattices were released with the use of a syringe and lattice diameter was measured under dark-field illumination after 5, 10, and 30 min. Only maximum contraction after 30 min is presented. A minimum of five lattices was assayed per experimental condition, mean values were calculated, and lattice diameter reduction was normalized to the lattice diameter before release (= % contraction). Collagen lattices were then digested, and cells were harvested and counted as previously described (Vaughan et al., 2000). Contraction results were only considered if cell concentration ranged between 5.7 × 104 and 6.3 × 104 cells/lattice at the time of release. All experiments were performed at least five times.
Antibodies and Immunofluorescence Microscopy
Cells grown in collagen lattices and on plastic culture dishes were fixed with 3% paraformaldehyde (PFA) in PBS containing 1 mM CaCl2 (PBS/Ca2+) for 15 min and permeabilized with 0.2% Triton X-100 (TX-100) in PBS/Ca2+ for 5 min. To test myosin expression, cells were fixed with 100% ethanol for 30 s. To simultaneously observe wrinkles and α-SMA–positive stress fibers, silicone-coated coverslip chambers were transferred rapidly from culture medium in prewarmed (37°C) 3% PFA in PBS/Ca2+, fixed for 15 min at 37°C, rinsed with PBS, permeabilized with 0.2% TX-100 in PBS/Ca2+ for 5 min, and immunostained. Care was taken that the silicone surface remained wet for the whole procedure.
Cells were stained for α-SMA (anti-αSM-1, IgG2a mAb; Skalli et al., 1986), F-actin (Phalloidin-Alexa 488, Molecular Probe, Eugene, OR), β-cytoplasmic (β74, rbAb; Yao et al., 1995), α-sarcomeric actin (SR-1, IgM mAb; Dako), γ-actins and α-SMA (AAL-20, rbAb), ED-A FN (IST-9, IgG1 mAb, gift from Dr. L. Zardi, National Institute for Cancer Research, Laboratory of Cell Biology, Genoa, Italy; Borsi et al., 1987; Carnemolla et al., 1987), VSV-G-tag (anti-VSV-G-tag, rbAB, a gift of Dr. J.-C. Perriard, ETH, Zürich, CH), the heavy chains (HC) of SMM and NMM (anti-SMMHC and anti-NMMHC, rbAbs; Benzonana et al., 1988) and DNA (DAPI). As secondary antibodies TRITC- and FITC-conjugated goat anti-mouse subclasses IgG1 and IgG2a (Southern Biotechnology Associates Inc., Birmingham, AL), IgG, IgM, and goat anti-rabbit antibodies (Jackson ImmunoResearch Laboratories, West Grove, PA) were used. Cells on plastic and collagen lattices were mounted in polyvinyl alcohol (Lennette, 1978); cells on silicone were covered with PBS, and chambers were sealed with a coverslip with the use of dental resin (President MicroSystems). All samples were observed with an oil immersion objective (Plan-Neofluar 40×/1.3, Ph3; Zeiss) on an Axiophot Zeiss microscope (Zeiss). Digital images were taken with a digital color camera (Coolview; Photonic Science Ltd., Millham, UK) and grabber software (ImageAccess V2.04K; Imagic Bildverarbeitung AG, Glattbrugg, Switzerland).
Inside three-dimensional collagen lattices the number of α-SMA–positive fibroblasts was automatically detected by a color thresholding routine (KS400; Zeiss) on images taken with a 20× objective (Zeiss) and related to the total cell number, determined by DAPI staining. Mean values were calculated from five independent experiments, analyzing five fields per lattice and five lattices per condition. All digital images were processed for printing with the use of Adobe Photoshop and printed with a digital Fujifilm Pictrography 4000 printer (Fuji Photo Film GmbH, Düsseldorf, Germany).
Western Blot Analysis
Fibroblast grown on plastic culture dishes were sampled as previously described (Chaponnier et al., 1995). Fibroblasts harvested from collagen lattices (see above) were suspended in sample buffer (1.25 × 106 cells/ml), sonicated, and boiled for 3 min. Protein content was determined with the use of a 1 μl protein assay (dotMetric; Geno Technology Inc., St. Louis, MO). Equal amounts of total proteins (5–10 μg) were loaded to 10% or 7% SDS-minigels (Bio-Rad Laboratories AG, Glattbrugg, Switzerland), separated by PAGE (Laemmli, 1970), and transferred to nitrocellulose membrane (Protran, BA85; Schleicher & Schuell, Dassel, Germany; Towbin et al., 1979). Membranes were then probed with the same primary antibodies used for immunofluorescence. Total actin was probed with the use of a mixture of antibodies against the different isoforms (see above). Incubations were followed by secondary antibodies goat anti-mouse IgG and goat anti-rabbit IgG, respectively, conjugated with horseradish-peroxidase (HRP; Jackson ImmunoResearch Laboratories). Signals were detected by ECL chemiluminescence (Amersham, Rahn AG, Zürich, Switzerland). Bands were digitized with a scanner (Arcus II; Agfa, Köln, Germany) and the ratio between all band densities of 1 blot was calculated by computer software (ImageQuant V3.3; Molecular Dynamics, Sunnyvale, CA). Relative α-SMA expression was normalized to the respective value for total actin in primary cells; in transfected cells VSV-G-tag expression was normalized to vimentin.
Actin Constructs and Transfection
The actin constructs used in our experiments were described by von Arx et al. (1995) and their sorting in several cell types was reported previously (Mounier et al., 1997). For transient and stable transfections, we used a pCMVβ vector (Clontech Laboratories AG, Basel, CH), in which the neomycin gene was cloned in EcoRI and in which the β-galactosidase gene was replaced by full-length cDNA encoding rat α-SMA, human γ- and β-cytoplasmic actins, and chicken α-cardiac muscle actin. The 3′ untranslated region (3′UTR) of actin isoforms was replaced by cDNA coding for the vesicular stomatitis virus G-protein (VSV-G; Soldati and Perriard, 1991), thus tagging the actins on C terminus. 3T3 fibroblasts were transfected with the use of FuGene 6 (Boehringer Mannheim AG, Mannheim, Germany) according to the manufacturer's protocol. To produce stable clones, transfected cells were split after 24 h and resuspended (1:50) in culture medium containing 1.5 mg/ml selection factor G418 (Promega, Madison, WI). After 7 d, colonies of ≤10 cells were isolated with the use of a 5-mm metal ring, trypsinized, and subcloned under G418 selection. Each clone was tested for expression of the transfected protein by Western blotting and by immunostaining for VSV-G-tag and the respective actin isoform.
Statistical Analysis
Numerical results are presented as the means ± SD of all experiments. Mean values were tested by a two-tailed heteroscedastic Student's t test. Differences were considered to be statistically significant at values of p ≤ 0.01. p values ≤ 0.005 were indicated by an asterisk (*) and with a double-asterisk (**) for p ≤ 0.001. The positive linear correlation between fibroblast contraction and α-SMA expression was statistically tested by calculating the square of the Pearson correlation product (r2 value).
RESULTS
Wrinkle Formation on Deformable Silicone Substrates Correlates with α-SMA Expression
SCFs and LFs, expressing different levels of α-SMA (Figure 1A; Western blot), exhibited different proportions of cells deforming the surface of 1-s flamed silicone (Figure 1). Only 20% of the fibroblasts derived from subcutaneous tissue produced wrinkles on the silicone film after 3 d (Figure 1A). The proportion of wrinkle-producing cells correlated with the proportion of α-SMA–positive SCFs (18%) determined after immunofluorescence staining on plastic dishes. When rat LFs were tested, 79% produced wrinkles, and 80% were α-SMA–positive. To further investigate the correlation between contracting cells and α-SMA–positive cells, SCFs were treated for 3 d with 10 ng/ml TGFβ1, previously shown to increase α-SMA expression in cultured fibroblasts (Desmoulière et al., 1993; Rønnov-Jessen and Petersen, 1993). TGFβ1 significantly enhanced the proportion of SCFs producing wrinkles on silicone to 52%. Concomitantly, 53% of SCFs expressed α-SMA on plastic. TGFβ2 caused similar effects (data not shown). TGFβ treatment of LFs did not enhance wrinkling and α-SMA expression significantly. The correlation factor between the number of wrinkle-producing cells and the number of α-SMA–positive cells was r2 = 0.99. TGFβ did not induce the expression of SMMHC. When α-SMA was stained (Figure 2B) on silicone substrates (1 s flaming), in which wrinkles were preserved (Figure 2A), it became evident that practically only α-SMA–positive cells deformed the silicone surface (Figure 2D). Fixation did not change the proportion of wrinkle-producing cells (Figure 1, A and B). Both, nonwrinkling α-SMA–negative and wrinkling α-SMA–positive fibroblasts exhibited a well-spread morphology and an F-actin–positive cortex. In α-SMA–positive fibroblasts stress fibers were generally more prominent and more organized in parallel bundles throughout the cytoplasm compared with α-SMA–negative cells (Figure 2C). Interestingly, stress fibers appeared to be thinner in fibroblasts on silicone compared with fibroblasts grown on plastic Petri dishes, possibly reflecting the different resistance of the two substrates (Harris et al., 1981). Quantifying the proportion of wrinkle-producing cells that were α-SMA-positive on the 1 s-flamed silicone led to a percentage of 90%, independently of the total number of α-SMA–positive cells (Figure 1B).
Figure 1.
Quantitative correlation between silicone wrinkling fibroblasts and α-SMA expression. (A) The percentage of SCFs and LFs producing wrinkles on silicone substrates is compared with the percentage of α-SMA–positive fibroblasts grown on plastic culture dishes. Expression of α-SMA is further demonstrated by Western blotting, equilibrated for total actin (lanes correspond to columns above). (B) The percentage of α-SMA–positive fibroblasts was evaluated on silicone in which wrinkles were preserved after fixation. The low percentage of wrinkling SCFs compared with LFs was significantly enhanced by TGFβ1 (10 ng/ml). In all cases α-SMA expression correlated with the wrinkling activity. Ninety percent of all silicone-wrinkling fibroblasts were α-SMA positive, independent on cell type or condition. Bars, SD of mean values, calculated from five independent experiments. *p ≤ 0.005, **p ≤ 0.001, compared with control SCFs.
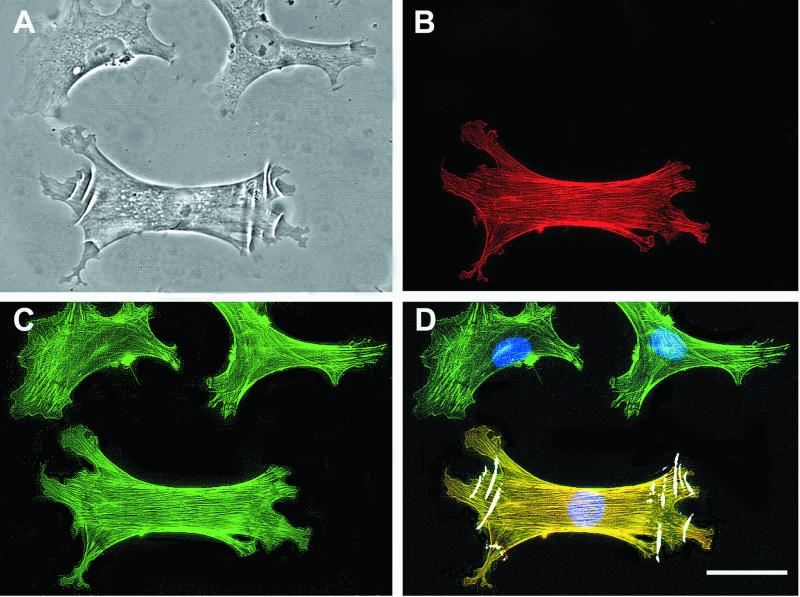
Figure 2.
α-SMA expression by LFs on silicone substrates. LFs (A) were stained on silicone for α-SMA (B, red) and F-actin (C, green). Image overlay (D) demonstrates that wrinkles are restricted to the α-SMA–positive cells (yellow) and absent from α-SMA–negative cells (green). Bar, 50 μm.
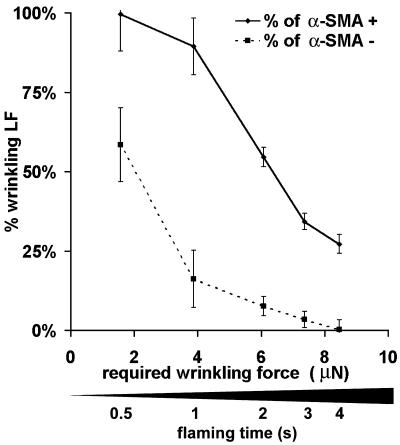
The stiffness of the silicone substrate surface provided a threshold to distinguish between contractile, surface-wrinkling cells and less contractile, nonwrinkling cells. To compare the forces exerted by single α-SMA–positive and –negative fibroblasts, we produced silicone substrates with varying stiffness by gradually increasing the flaming time (Figure 3, bar) and determined the force required to cause wrinkles in these films. Increasing substrate stiffness required approximately linearly increasing wrinkling force and showed little local variance within the same substrate (≤1%). However, the thin silicone film at the substrate periphery showed significantly higher resistance (18 ± 2%) compared with the central part. Hence, the peripheral 5 mm were not considered for quantification. When a force of ∼1.5 μN was required, virtually all α-SMA–positive LFs but only 60% α-SMA–negative LFs produced wrinkles (Figure 3). The largest difference between the percentages of α-SMA–positive and of α-SMA–negative LFs producing wrinkles (89% vs. 16%) was observed on substrates that required a wrinkling force of ∼4.0 μN. By further increasing the substrate stiffness, i.e., when a force of ∼6.0 μN was required, wrinkles were produced essentially by α-SMA–positive and only by <5% of α-SMA–negative LFs. When the stiffness of the substrate required a wrinkling-force of ∼8.5 μN, 27% of α-SMA–positive LFs but none of the α-SMA–negative LFs were capable of producing wrinkles.
Figure 3.
Expression of α-SMA increases the force of individual LFs. By increasing the flaming time of the silicone surface, deformable substrates were produced with increasing stiffness, leading to an increase of the force, which is required to cause substrate wrinkles. The percentage of α-SMA–positive LFs producing wrinkles was related to the wrinkling proportion of α-SMA–negative LFs, as determined by immunostaining. On all substrates α-SMA–positive LFs exhibited a higher proportion of wrinkling cells compared with α-SMA–negative LFs.
Contraction of Fibroblast-populated Collagen Lattices Correlates with α-SMA Expression
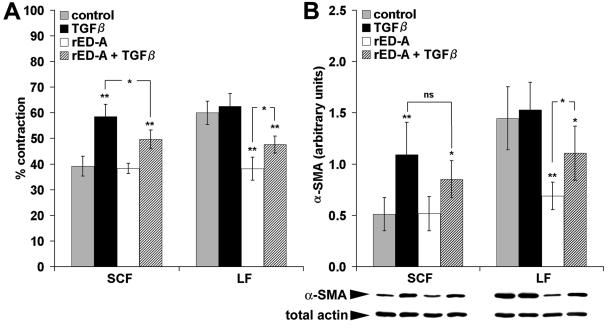
When released from the culture dish after 5 d fibroblast-populated attached collagen lattices contracted rapidly and reached maximal contraction after 30 min. The contractile potential of SCF populations, expressing low levels of α-SMA, was again compared with that of LF populations, expressing high amounts of α-SMA. For a given cell type, the degree of contraction was dependent on the number of fibroblasts populating the lattice at the time of release. However, at any comparable cell number, LFs contracted the lattices significantly more than SCFs. The largest difference was observed at 6 × 104 cells/lattice, where both populations exhibited maximal contraction. SCFs caused a 41% lattice contraction compared with a 63% contraction for LFs (Figure 4A). Treating SCFs with 10 ng/ml TGFβ1 during 5 d increased lattice contraction to 63%, thereby reaching the level of LFs. TGFβ1-treatment showed no enhancing effect on LF contraction (63%). Again, no differences were observed between the effects of TGFβ1 and TGFβ2. In a second approach, the action of endogenous TGFβ was neutralized by applying TGFβ-sR to collagen lattices for 5 d (Figure 4A). Increasing the concentration of TGFβ-sR in steps of one order of magnitude (1.0–100 ng/ml) reduced LF contraction gradually from 63% to a minimum of 38%, thereby reaching the level of control SCFs. The contractility of SCFs was not affected by 100 ng TGFβ-sR. Similar results were obtained by blocking TGFβ action with a TGFβ1-neutralizing antibody in concentrations between 0.1 and 10 μg/ml for 5 d. These results are in accordance with the possibility that LFs in collagen lattices maintain a high level of α-SMA expression by autocrine stimulation with TGFβ.
Figure 4.
TGFβ affects in parallel collagen lattice contraction and α-SMA expression. (A) SCFs and LFs were grown in attached collagen gels in serum-containing medium and treated with 10 ng/ml TGFβ1 or TGFβ-sR (1.0–100 ng/ml) for 5 d. Stressed lattices were then released and maximum contraction was recorded after 30 min. TGFβ1 enhanced SCF contraction but had no effect on that of LFs. TGFβ-sR decreased LFs contraction dose-dependently but did not affect that of SCFs. Each bar represents mean values (±SD) of five independent experiments performed with five lattices per condition, respectively. *p ≤ 0.005, **p ≤ 0.001 compared with the respective control. (B) The level of α-SMA expression in collagen gels was quantified by Western blotting at each condition (lanes 1–3: SCFs; 4–8: LFs; lanes 1 + 4: control; 2 + 5: 10 ng/ml TGFβ1; 6–8: 1.0–100 ng/ml TGFβ-sR). No changes were observed when blots were evaluated for total actin.
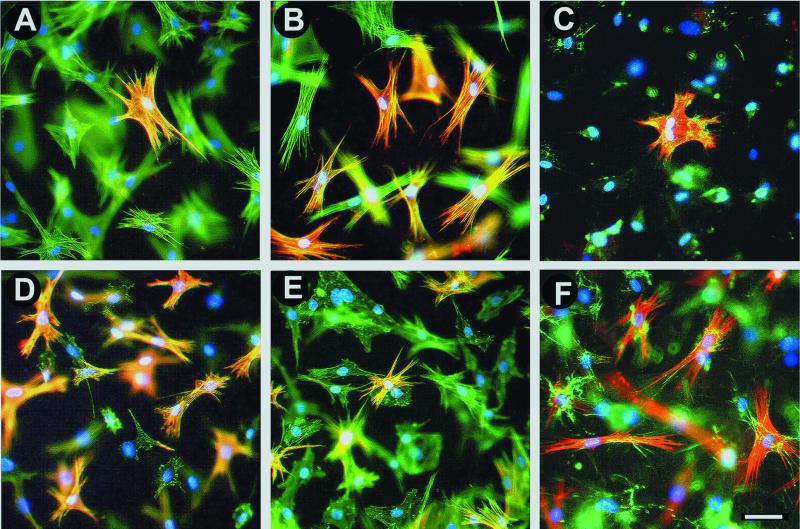
Lattices were digested after contraction experiments, and cellular proteins were blotted to evaluate α-SMA and total actin levels (Figure 4B). α-SMA expression was lower in SCF populations compared with LFs. The level of α-SMA in SCFs was enhanced by TGFβ treatment and reduced in LFs by blocking TGFβ effect with TGFβ-sR (Figure 4B) or anti-TGFβ1 antibody. The level of total actin remained constant at all conditions tested. Protein content analysis was complemented by staining fibroblasts in collagen lattices for α-SMA and F-actin (Figure 5). When grown in lattices, SCFs exhibited broader lamellae (Figure 5A) compared with LFs, which were more elongated and showed prominent stress fibers (Figure 5D). Exposing SCFs to TGFβ increased the proportion of cells expressing α-SMA from 13 ± 2%–69 ± 4% and lead to moderate cell elongation. Treatment of LFs with either TGFβ-sR (Figure 5E) or anti-TGFβ1 antibody reduced the number of LFs expressing α-SMA from 70 ± 5%–12 ± 2% but did not alter F-actin organization. Both blocking factors had no significant effect on SCF morphology and α-SMA expression. Generally, the percentage of α-SMA–positive cells in collagen lattices was ∼1.4-fold lower compared with plastic and silicone substrates, possibly reflecting the different substrate compliance (Arora et al., 1999b).
Figure 5.
TGFβ increases the number of α-SMA–positive fibroblasts in collagen lattices. Collagen lattices with SCFs (A–C) and LFs (D–F) were stained after 5 d for α-SMA (red), F-actin (A, B, D, and E, green), ED-A FN (C and F, green), and cell nuclei (blue). Colocalization of α-SMA with F-actin or ED-A FN, respectively, is indicated by yellow color. Control SCFs populations contained a low number of α-SMA–positive cells (A, C) compared with SCFs treated for 5 d with 10 ng/ml TGFβ (B) or LFs (D and F). TGFβ-sR (100 ng/ml) decreased the number of α-SMA–positive LFs (E). ED-A FN expression by SCFs (C) is lower than that of LFs (F). Bar, 50 μm.
ED-A FN Is Essential for α-SMA Expression and Contraction of Fibroblast-populated Collagen Lattices
We have recently shown that induction of α-SMA expression by TGFβ depends on the presence of ED-A FN in the extracellular matrix (Serini et al., 1998). In the attached collagen lattice model, the levels of endogenous ED-A FN and α-SMA expression at 5 d varied according to the myofibroblastic differentiation (Figure 5). Control SCFs (Figure 5C) and LFs treated with TGF-sR secreted low amounts of ED-A FN. In contrast, TGFβ-stimulated SCFs and control LFs (Figure 5F) produced high amounts of ED-A FN, organized into extracellular fibrils and aligned with intracellular F-actin bundles as observed by confocal imaging.
The importance of ED-A FN in mediating TGFβ-induced fibroblast contraction was then tested by adding rED-A to the collagen matrix of the lattice contraction assay. To exclude effects of plasma factors and plasma FN, all experiments were performed with the use of Monomed without medium change during 5 d. Fibroblast-populated collagen lattices in Monomed contracted similarly (Figure 6A) compared with serum conditions (Figure 4A). rED-A reduced the contraction of LFs to 38% (LF control = 60%) but had no effect on SCF lattice contraction (38%). When fibroblast-populated lattices were simultaneously treated with rED-A and TGFβ, intermediate lattice contraction was obtained: SCF contraction was lower (50%) when treated additionally with rED-A compared with TGFβ treatment only (59%). Treating LFs simultaneously with TGFβ and rED-A partially rescued lattice contraction (48%) compared with rED-A alone (38%).
Figure 6.
Exogenous rED-A reduces collagen lattice contraction and α-SMA expression. (A) rED-A (300 μg/ml) reduced LFs contraction and contraction of TGFβ-treated (10 ng/ml) LFs and SCFs in serum-free conditions. (B) Cell lysates from collagen lattices were immunoblotted against α-SMA and total actin. α-SMA band densities were quantified by image analysis and normalized to total actin measured in the same blot. Changes in α-SMA expression correlated with lattice contraction, whereas total actin remained unchanged in all conditions tested. Each bar represents mean values (±SD) of five independent experiments performed with five lattices per condition, respectively. *p ≤ 0.005, **p ≤ 0.001, ns = not significant, compared with the respective control for each cell type or for selected pairs.
Correlation between increased lattice contraction and increased α-SMA expression was examined by analyzing the amount of α-SMA in the lattice cells (Figure 6B). The level of total actin was not changed by the experimental conditions. A correlation factor of r2 = 0.88 was obtained when contraction mean values were tested against mean values of α-SMA levels (Figure 6B), calculated from Western blot quantitative analysis and normalized to total actin. Immunofluorescence staining of rED-A–containing lattices and subsequent cell counting revealed a decrease in the proportion of α-SMA–positive LFs (13 ± 2%) compared with lattices not containing rED-A (70 ± 4%). LFs exhibited a similar morphology in rED-A lattices as after TGFβ-blocking experiments (Figure 5E). No changes were noticed regarding the number of α-SMA–positive SCFs (13 ± 2%) and their morphology after rED-A, similarly to TGFβ-sR treatment.
Transfection with α-SMA cDNA Enhances Contraction of 3T3 Fibroblasts
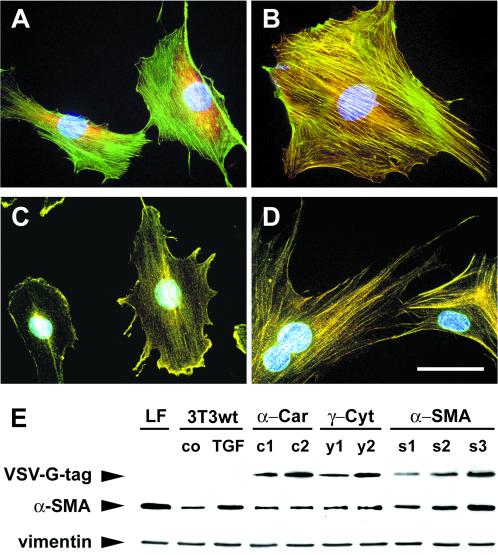
To investigate the direct effect of α-SMA expression on fibroblast contraction, we transiently and stably transfected Swiss 3T3 fibroblasts with α-SMA cDNA and evaluated their ability to contract stressed collagen lattices. 3T3 fibroblasts were chosen because of their high transfection rate (21% in average) compared with primary fibroblasts (2–5%; Mounier et al., 1999) and because of their basal low α-SMA expression (Figure 7A, E). Wild-type 3T3 fibroblasts, grown in attached collagen lattices or on culture dishes, develop stress fibers in the presence of serum (Figure 7A) but only ∼3–5% of the cells are α-SMA positive. Transfected α-SMA was predominantly incorporated into stress fibers as evaluated by double-labeling with antibodies against α-SMA and against the VSV-G-tag at the actin C terminus (Figure 7B). Stably transfected α-cardiac (Figure 7D) and transiently transfected β-cytoplasmic actin were also incorporated into stress fibers, but the latter occasionally accumulated in cytoplasmic aggregates if strongly overexpressed, a phenomenon reported previously for transfected primary cells (von Arx et al., 1995; Mounier et al., 1997). Transfected γ-cytoplasmic actin predominantly localized in lamellipodia and membrane ruffles (Figure 7C) and sporadically appeared in stress fibers of high expressing clones. Increased expression of α-SMA after transfection was demonstrated by Western blotting against VSV-G-tag and α-SMA (Figure 7E). Transfection with other actin isoforms did not change the level of α-SMA expression compared with wild-type fibroblasts. Neither transfected nor wild-type 3T3 fibroblasts expressed SMMHC. Moreover, transfection did not change the level of NMMHC expression as tested by immunofluorescence staining and Western blotting.
Figure 7.
Stable transfection of 3T3 fibroblasts with different actin isoforms. 3T3 fibroblasts (A) were stably transfected with VSV-G–tagged α-SMA (B), γ-cytoplasmic (C) and α-cardiac actin (D) cDNA, respectively. Double-immunostaining of wild-type 3T3 fibroblasts demonstrated low expression of α-SMA (A, red) in F-actin–containing stress fibers (A, green) Transfected α-SMA and α-cardiac actin were predominantly incorporated into stress fibers and γ-cytoplasmic actin into membrane ruffles and lamellipodia, as shown by double-immunostaining of fibroblasts for actin isoforms (B–D, green) and VSV-G-tag (B–D, red). Bar, 50 μm. Western blotting (E) demonstrated low α-SMA expression in wild-type 3T3 fibroblasts (3T3wt, co) compared with TGFβ1-treated 3T3 fibroblasts (3T3wt, TGF) and LFs. Note that enhanced expression of α-SMA correlates with enhanced VSV-G-tag expression in α-SMA transfected clones (s1–s3), but not in clones expressing transfected α-cardiac (α-Car: c1, c2) and γ-cytoplasmic actin (γ-Ccyt: y1, y2). Vimentin was used to demonstrate equal loads of total protein.
With the use of the collagen lattice assay, wild-type 3T3 fibroblasts exhibited 26% contraction, which was 1.6-fold lower compared with SCFs and 2.4-fold lower compared with LFs contraction. 3T3 fibroblast contraction correlated with transient transfection efficiency (r2 = 0.92) and was significantly higher after transfection with α-SMA compared with β-cytoplasmic actin (132 ± 4%). To minimize variations between different transfection experiments, we produced 3T3 fibroblast clones stably expressing α-SMA at different levels and compared their contractile capacity to clones stably expressing various levels of γ-cytoplasmic and α-cardiac actin (Figure 8). Lattice contraction showed small linear increase with increasing expression levels of γ-cytoplasmic actin (r2 = 0.9) compared with wild-type fibroblasts; after transfection with α-cardiac actin, linear increasing contraction (r2 = 0.88) was slightly but significantly more important than after transfection of γ-cytoplasmic actin. Transfection of α-SMA (r2 = 0.79) clearly promoted the highest contraction at comparable expression levels (Figure 8).
Figure 8.
Stable transfection of 3T3 fibroblasts with α-SMA cDNA enhances collagen gel contraction. 3T3 fibroblasts were stably transfected with VSV-G–tagged α-SMA, α-cardiac, and γ-cytoplasmic actin cDNA, respectively, cloned for different VSV-G-tag expression levels and grown in attached collagen gels. Contraction of released collagen gels was normalized to wild-type 3T3 fibroblast contraction. With increasing expression levels, α-SMA–transfected 3T3 fibroblast (▴, continuous line) contracted significantly more than 3T3 fibroblasts transfected with α-cardiac (♦, dashed line) and γ-cytoplasmic actin (●, dotted line). Each data point represents mean collagen lattice contraction of one clone, calculated from three independent experiments. R2 values indicate the correlation between VSV-G-tag expression levels and contraction.
DISCUSSION
In addition to be a well-accepted marker of myofibroblast differentiation, α-SMA has been suggested to play a role in the production of contractile force during wound healing and fibrocontractive diseases (for review see Serini and Gabbiani, 1999). Several works have investigated this possibility at the cellular level (Arora and McCulloch, 1994; Vaughan et al., 2000); however, such function has remained not well established. Our study, exploring the relationship between the level of α-SMA expression in cultured fibroblasts and their efficiency to contract deformable silicone substrates and stress-released collagen gels, shows (1) a significantly higher contraction of LFs, expressing high levels of α-SMA, compared with SCFs, expressing low levels of α-SMA; (2) an enhancement of contractile activity in SCFs treated by TGFβ concomitant with an increase of α-SMA expression. Inversely, decreasing α-SMA expression by blocking the effect of endogenous TGFβ with specific antibodies, TGFβ-sR, or rED-A correlates with a reduction of LFs contractile activity; (3) a clear increase of contraction in 3T3 fibroblasts stably and transiently transfected with α-SMA cDNA compared with nontransfected cells or cells transfected with α-cardiac, γ-cytoplasmic, and β-cytoplasmic actin cDNA, in the absence of changes in the expression of SMMHC and NMMHC. Taken together, these results indicate that α-SMA expression is crucial in determining the extent of in vitro fibroblast contractile activity.
To assess the contractile potential of fibroblast populations we measured the isotonic contraction of attached collagen gels. Isometrically stressed lattices are a more appropriate model of myofibroblast-populated granulation tissue during wound contraction compared with mechanically unloaded floating lattices (Grinnell, 1994). Isometric tension has been shown to be important for the development of myofibroblastic contractile features such as stress fibers (Burridge, 1981; Tomasek et al., 1992; Katoh et al., 1998; Grinnell et al., 1999a) and α-SMA expression in collagen lattices (Arora et al., 1999b). To test whether individual α-SMA–positive fibroblasts contract more efficiently than α-SMA–negative cells under identical conditions, we used a modification of the wrinkling silicone elastomer assay, originally developed by Harris et al. (1980), which is particularly useful in order to visualize overall cell contractile forces. We show here a direct correlation between α-SMA expression and the contractile efficiency of individual fibroblasts by improving the assay with an immunostaining technique that allows the examination of cytoskeletal features and silicone substrate wrinkles in the same cell. By modulating the cross-linking process of the silicone surface, we have generated a range of elastomers with increasing stiffness and determined the forces required to wrinkle these substrates. Wrinkling forces of the same magnitude have been measured for human dermal fibroblasts, with the use of latex bead displacement assays, but were restricted to a silicone film with low stiffness in order to provide a full bead position recovery (Fray et al., 1998). It is conceivable that wrinkle-producing fibroblasts exert only the minimum force necessary to deform these weak surfaces; therefore, the use of elastomers with gradually increasing stiffness is crucial to determine their maximum contractile potential. Highly compliant elastomers have been wrinkled by a large proportion of α-SMA–negative fibroblasts consistent with the wrinkling capability of numerous cell types (Lee et al., 1994; Oliver et al., 1995). With the use of stiffer substrates, we determined a threshold of ∼4 μN that discriminates between these relatively weak traction forces and higher contractile forces (Roy et al., 1999); this threshold is only surpassed by α-SMA–positive fibroblasts. Consequently, expression of α-SMA considerably increases contractile activity but is not mandatory for cells to exert forces on a low level.
In a further attempt to control the relationship between α-SMA expression and fibroblast contractile activity, we modulated the level of α-SMA expression in our cell populations. Among cytokines implicated in myofibroblast differentiation, TGFβ has been proven to directly induce α-SMA expression in vivo and in vitro (Desmoulière et al., 1993; Rønnov-Jessen and Petersen, 1993). A series of studies with the use of free-floating collagen gels reported TGFβ-induced fibroblast contraction, but did not consider the expression of α-SMA (Montesano and Orci, 1988; Finesmith et al., 1990; Tingstrom et al., 1992; Pena et al., 1994; Riikonen et al., 1995; Levi-Schaffer et al., 1999) with two exceptions (Arora and McCulloch, 1994; Kurosaka, 1995; 2154). However, it was shown recently that the mechanisms of mechanically unloaded free-floating gel reduction considerably differ from stressed gel contraction (for review see Grinnell, 2000). Other studies reported a relationship between increased isometric tension in attached collagen gels and increased α-SMA expression after treating fibroblasts with TGFβ, but did not release these gels to test isotonic contraction (Kurosaka et al., 1998; Arora et al., 1999b). Here we show that increasing isotonic contraction of mechanically loaded collagen lattices by TGFβ corresponds to an increase of α-SMA expression in SCFs. Inversely, blocking the effect of TGFβ with specific antibodies, with TGFβ-sR or with rED-A reduced α-SMA expression of LFs and concomitantly lattice contraction. These experiments are compatible with the existence of a TGFβ autocrine loop, maintaining myofibroblast differentiation and contractile activity (Shi et al., 1996; Schmid et al., 1998) and are consistent with similar findings of Vaughan et al. (2000).
The results of rED-A experiments further suggest a functional role of the ED-A FN splice variant in fibroblast contraction. The expression of ED-A FN in healing wounds (Ffrench-Constant et al., 1989; Brown et al., 1993) precedes the appearance of α-SMA–positive myofibroblasts and is essential to mediate TGFβ1-induced α-SMA expression (Serini et al., 1998). The mechanism of specific antibodies and of TGFβ-sR action on TGFβ appears obvious (Komesli et al., 1998); however, the mechanism through which rED-A inhibits TGFβ-mediated α-SMA expression is at present unclear. Because antibodies specific against ED-A FN exert similar effects, rED-A seems to prevent the interaction between fibroblasts and ED-A FN, a possible outside-in signal for myofibroblast differentiation. In addition to serving as a signal, ED-A FN may play a mechanical role by providing efficient cell-matrix attachment, which is important for transmitting intracellular contraction to the matrix (Racine-Samson et al., 1997; Imanaka-Yoshida et al., 1999; Roy et al., 1999). Further studies are needed to understand the mechanism of this ED-A FN activity.
Strong evidence of direct correlation between the level of α-SMA expression and fibroblast contractility is provided by transfection of α-SMA cDNA into 3T3 fibroblasts. Nontransfected fibroblasts also contract collagen gels, but exert considerably lower forces compared with α-SMA–positive fibroblasts, consistent with the wrinkling capacity of α-SMA–negative cells on compliant elastomers. Downregulation of α-SMA expression by antisense mRNA has been previously shown to provoke an increase in cell migratory activity, possibly mediated through a decrease in cell-matrix adhesion sites (Rønnov-Jessen and Petersen, 1993). This possibility is in accordance with the observation that significantly larger focal contacts are present in α-SMA–positive LFs compared with SCFs (Dugina et al., 1998). Further studies should concentrate on the subcellular mechanisms through which α-SMA increases fibroblast contractile activity and in particular investigate whether transfected α-SMA increases induces the formation of specialized focal contacts, typical for α-SMA–positive myofibroblasts (Dugina et al., 1998; Vaughan et al., 2000). For this purpose the use of embedded fluorescent beads in elastic polyacrylamide substrates (Pelham and Wang, 1997) in conjunction with detailed computer analysis (Dembo and Wang, 1999; Pelham and Wang, 1999) will be necessary. This technique can also allow to examine whether α-SMA expression mediates more efficient contraction by increasing the absolute contractile force or by optimizing the spatial distribution of several subcellular forces.
Transfection with α-SMA increased fibroblast contractility in the absence of SMMHC expression, in agreement with the recent demonstration of efficient smooth muscle cell contraction in SMMHC knockout mice (Morano et al., 2000) and notably without increasing NMMHC expression. Furthermore, TGFβ enhances contractile activity of SCFs without changing SMMHC or NMMHC expression. Further studies are needed to determine whether enhanced α-SMA expression influences other structural proteins that are involved in cell contraction in an isoform-specific manner, such as tropomyosin (Schevzov et al., 1993), caldesmon (Bretscher and Lynch, 1985), calponin (Gimona et al., 1992), SM22 (Gimona et al., 1992), and gelsolin (Arora et al., 1999a) or whether it affects regulatory events, including myosin light-chain phosphorylation, small GTPase activation, and calcium signaling (Parizi et al., 2000). The 3′UTRs of α-cardiac and β-cytoplasmic actin have been suggested to facilitate actin isoform sorting in the cytoplasm by localizing to different subcellular compartments (Kislauskis et al., 1993) and the 3′UTR of α-tropomyosin has been shown to exert functional tumor suppressor activity (Rastinejad et al., 1993). However, an effect of the 3′UTR of α-SMA on fibroblast contraction seems to be unlikely, because this region was excluded from our actin cDNA constructs.
In conclusion, we have shown with the use of different approaches that de novo expression of α-SMA in cultured fibroblasts enhances their contractile activity. The direct evidence obtained from α-SMA cDNA transfection experiments is corroborated by the results showing a correlation between levels of α-SMA expression and cell contractility in several other experimental situations. Apparently the α-SMA activity is exerted without any change in myosin HC expression; probably α-SMA provides more competent interaction with NMM either directly or indirectly by generating an actin organization specialized for contraction. Further studies along these lines may provide important information on the cellular mechanisms regulating the contraction of a healing wound and of fibrotic tissues.
ACKNOWLEDGMENTS
We thank Drs. S. Clément, C. von Ballestrem, O. Thoumine, and Pr. Jean-Marc Meyer for their advice in transfection experiments, silicon assays, and on the use of dental resin; A. Maurer-Hiltbrunner for technical assistance; Messrs. J.C. Rumbeli and E. Denkinger for photographic work; and Mrs. S. Josseron for secretarial work. This work was supported by the Swiss National Science Foundation (grants 31–50568.97 and 31–54048.98) and by the Roche Research Foundation, F. Hoffmann-La Roche Ltd. (grant 98–206). Drs. David A. Cox, Victor Koteliansky, Jean-Claude Perriard, and Luciano Zardi are gratefully acknowledged for providing recombinant-human TGFβ2, TGFβ-sR and rED-A, antitag antibodies, and IST-9 antibodies, respectively.
Abbreviations used:
- α-SMA
α-smooth muscle actin
- FN
fibronectin
- HC
heavy chain
- LFs
lung fibroblast
- NMM
nonmuscle myosin
- TGFβ-sR
soluble TGFβ-receptor type II
- PFA
paraformaldehyde
- rED-A
recombinant ED-A
- SCFs
subcutaneous fibroblast
- SMM
smooth muscle myosin
- TX-100
triton X-100
REFERENCES
- Arora PD, McCulloch CA. Dependence of collagen remodelling on alpha-smooth muscle actin expression by fibroblasts. J Cell Physiol. 1994;159:161–175. doi: 10.1002/jcp.1041590120. [DOI] [PubMed] [Google Scholar]
- Arora PD, Janmey PA, McCulloch CA. A role for gelsolin in stress fiber-dependent cell contraction. Exp Cell Res. 1999a;250:155–167. doi: 10.1006/excr.1999.4521. [DOI] [PubMed] [Google Scholar]
- Arora PD, Narani N, McCulloch CA. The compliance of collagen gels regulates transforming growth factor-beta induction of alpha-smooth muscle actin in fibroblasts. Am J Pathol. 1999b;154:871–882. doi: 10.1016/s0002-9440(10)65334-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benzonana G, Skalli O, Gabbiani G. Correlation between the distribution of smooth muscle or non muscle myosins and alpha-smooth muscle actin in normal and pathological soft tissues. Cell Motil Cytoskeleton. 1988;11:260–274. doi: 10.1002/cm.970110405. [DOI] [PubMed] [Google Scholar]
- Borsi L, Carnemolla B, Castellani P, Rosellini C, Vecchio D, Allemanni G, Chang SE, Taylor-Papadimitriou J, Pande H, Zardi L. Monoclonal antibodies in the analysis of fibronectin isoforms generated by alternative splicing of mRNA precursors in normal and transformed human cells. J Cell Biol. 1987;104:595–600. doi: 10.1083/jcb.104.3.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bretscher A, Lynch W. Identification and localization of immunoreactive forms of caldesmon in smooth and nonmuscle cells: a comparison with the distributions of tropomyosin and alpha-actinin. J Cell Biol. 1985;100:1656–1663. doi: 10.1083/jcb.100.5.1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown LF, Dubin D, Lavigne L, Logan B, Dvorak HF, Van de Water L. Macrophages and fibroblasts express embryonic fibronectins during cutaneous wound healing. Am J Pathol. 1993;142:793–801. [PMC free article] [PubMed] [Google Scholar]
- Burridge K. Are stress fibers contractile? Nature. 1981;294:691–692. doi: 10.1038/294691a0. [DOI] [PubMed] [Google Scholar]
- Carnemolla B, Borsi L, Zardi L, Owens RJ, Baralle FE. Localization of the cellular-fibronectin-specific epitope recognized by the monoclonal antibody IST-9 using fusion proteins expressed in E. coli. FEBS Lett. 1987;215:269–273. doi: 10.1016/0014-5793(87)80160-6. [DOI] [PubMed] [Google Scholar]
- Chaponnier C, Goethals M, Janmey PA, Gabbiani F, Gabbiani G, Vandekerckhove J. The specific NH2-terminal sequence Ac-EEED of alpha-smooth muscle actin plays a role in polymerization in vitro and in vivo. J Cell Biol. 1995;130:887–895. doi: 10.1083/jcb.130.4.887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darby I, Skalli O, Gabbiani G. Alpha-smooth muscle actin is transiently expressed by myofibroblasts during experimental wound healing. Lab Invest. 1990;63:21–29. [PubMed] [Google Scholar]
- Dembo M, Wang YL. Stresses at the cell-to-substrate interface during locomotion of fibroblasts. Biophys J. 1999;76:2307–2316. doi: 10.1016/S0006-3495(99)77386-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desmoulière A, Gabbiani G. The role of myofibroblasts in wound healing and fibrocontractive diseases. In: Clark RAF, editor. The Molecular and Cellular Biology of Wound Repair. 2nd ed. New York: Plenum Press; 1996. pp. 391–423. [Google Scholar]
- Desmoulière A, Geinoz A, Gabbiani F, Gabbiani G. Transforming growth factor-beta 1 induces alpha-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol. 1993;122:103–111. doi: 10.1083/jcb.122.1.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desmoulière A, Rubbia-Brandt L, Abdiu A, Walz T, Macieira-Coelho A, Gabbiani G. Alpha-smooth muscle actin is expressed in a subpopulation of cultured and cloned fibroblasts and is modulated by gamma-interferon. Exp Cell Res. 1992;201:64–73. doi: 10.1016/0014-4827(92)90348-c. [DOI] [PubMed] [Google Scholar]
- Dugina V, Alexandrova A, Chaponnier C, Vasiliev J, Gabbiani G. Rat fibroblasts cultured from various organs exhibit differences in alpha-smooth muscle actin expression, cytoskeletal pattern, and adhesive structure organization. Exp Cell Res. 1998;238:481–490. doi: 10.1006/excr.1997.3868. [DOI] [PubMed] [Google Scholar]
- Ffrench-Constant C, Van de Water L, Dvorak HF, Hynes RO. Reappearance of an embryonic pattern of fibronectin splicing during wound healing in the adult rat. J Cell Biol. 1989;109:903–914. doi: 10.1083/jcb.109.2.903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finesmith TH, Broadley KN, Davidson JM. Fibroblasts from wounds of different stages of repair vary in their ability to contract a collagen gel in response to growth factors. J Cell Physiol. 1990;144:99–107. doi: 10.1002/jcp.1041440113. [DOI] [PubMed] [Google Scholar]
- Fray TR, Molloy JE, Armitage MP, Sparrow JC. Quantification of single human dermal fibroblast contraction. Tissue Eng. 1998;4:281–291. doi: 10.1089/ten.1998.4.281. [DOI] [PubMed] [Google Scholar]
- Gabbiani G, Ryan GB, Majno G. Presence of modified fibroblasts in granulation tissue and their possible role in wound contraction. Experientia. 1971;27:549–550. doi: 10.1007/BF02147594. [DOI] [PubMed] [Google Scholar]
- Gimona M, Sparrow MP, Strasser P, Herzog M, Small JV. Calponin and SM 22 isoforms in avian and mammalian smooth muscle. Absence of phosphorylation in vivo. Eur J Biochem. 1992;205:1067–1075. doi: 10.1111/j.1432-1033.1992.tb16875.x. [DOI] [PubMed] [Google Scholar]
- Grinnell F. Fibroblasts, myofibroblasts, and wound contraction. J Cell Biol. 1994;124:401–404. doi: 10.1083/jcb.124.4.401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grinnell F. Fibroblast-collagen-matrix contraction: growth-factor signaling and mechanical loading. Trends Cell Biol. 2000;10:362–365. doi: 10.1016/s0962-8924(00)01802-x. [DOI] [PubMed] [Google Scholar]
- Grinnell F, Ho CH, Lin YC, Skuta G. Differences in the regulation of fibroblast contraction of floating versus stressed collagen matrices. J Biol Chem. 1999a;274:918–923. doi: 10.1074/jbc.274.2.918. [DOI] [PubMed] [Google Scholar]
- Grinnell F, Zhu M, Carlson MA, Abrams JM. Release of mechanical tension triggers apoptosis of human fibroblasts in a model of regressing granulation tissue. Exp Cell Res. 1999b;248:608–619. doi: 10.1006/excr.1999.4440. [DOI] [PubMed] [Google Scholar]
- Harris AK, Stopak D, Wild P. Fibroblast traction as a mechanism for collagen morphogenesis. Nature. 1981;290:249–251. doi: 10.1038/290249a0. [DOI] [PubMed] [Google Scholar]
- Harris AK, Wild P, Stopak D. Silicone rubber substrata: a new wrinkle in the study of cell locomotion. Science. 1980;208:177–179. doi: 10.1126/science.6987736. [DOI] [PubMed] [Google Scholar]
- Imanaka-Yoshida K, Enomoto-Iwamoto M, Yoshida T, Sakakura T. Vinculin, talin, integrin alpha6beta1 and laminin can serve as components of attachment complex mediating contraction force transmission from cardiomyocytes to extracellular matrix. Cell Motil Cytoskeleton. 1999;42:1–11. doi: 10.1002/(SICI)1097-0169(1999)42:1<1::AID-CM1>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- Katoh K, Kano Y, Masuda M, Onishi H, Fujiwara K. Isolation and contraction of stress fibers. Mol Biol Cell. 1998;9:1919–1938. doi: 10.1091/mbc.9.7.1919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kislauskis EH, Li Z, Singer RH, Taneja KL. Isoform-specific 3′-untranslated sequences sort alpha-cardiac and beta-cytoplasmic actin messenger RNAs to different cytoplasmic compartments. J Cell Biol. 1993;123:165–172. doi: 10.1083/jcb.123.1.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komesli S, Vivien D, Dutartre P. Chimeric extracellular domain type II transforming growth factor (TGF)-beta receptor fused to the Fc region of human immunoglobulin as a TGF-beta antagonist. Eur J Biochem. 1998;254:505–513. doi: 10.1046/j.1432-1327.1998.2540505.x. [DOI] [PubMed] [Google Scholar]
- Komuro T. Re-evaluation of fibroblasts and fibroblast-like cells. Anat Embryol. 1990;182:103–112. doi: 10.1007/BF00174011. [DOI] [PubMed] [Google Scholar]
- Kurosaka H, Kurosaka D, Kato K, Mashima Y, Tanaka Y. Transforming growth factor-beta 1 promotes contraction of collagen gel by bovine corneal fibroblasts through differentiation of myofibroblasts. Invest Ophthalmol Vis Sci. 1998;39:699–704. [PubMed] [Google Scholar]
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lee J, Leonard M, Oliver T, Ishihara A, Jacobson K. Traction forces generated by locomoting keratocytes. J Cell Biol. 1994;127:1957–1964. doi: 10.1083/jcb.127.6.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lennette DA. An improved mounting medium for immunofluorescence microscopy. Am J Clin Pathol. 1978;69:647–648. doi: 10.1093/ajcp/69.6.647. [DOI] [PubMed] [Google Scholar]
- Levi-Schaffer F, Garbuzenko E, Rubin A, Reich R, Pickholz D, Gillery P, Emonard H, Nagler A, Maquart FA. Human eosinophils regulate human lung- and skin-derived fibroblast properties in vitro: a role for transforming growth factor beta (TGF- beta) Proc Natl Acad Sci USA. 1999;96:9660–9665. doi: 10.1073/pnas.96.17.9660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin HY, Moustakas A, Knaus P, Wells RG, Henis YI, Lodish HF. The soluble exoplasmic domain of the type II transforming growth factor (TGF)-beta receptor. A heterogeneously glycosylated protein with high affinity and selectivity for TGF-beta ligands. J Biol Chem. 1995;270:2747–2754. doi: 10.1074/jbc.270.6.2747. [DOI] [PubMed] [Google Scholar]
- Mochitate K, Pawelek P, Grinnell F. Stress relaxation of contracted collagen gels: disruption of actin filament bundles, release of cell surface fibronectin, and down-regulation of DNA and protein synthesis. Exp Cell Res. 1991;193:198–207. doi: 10.1016/0014-4827(91)90556-a. [DOI] [PubMed] [Google Scholar]
- Montesano R, Orci L. Transforming growth factor beta stimulates collagen-matrix contraction by fibroblasts: implications for wound healing. Proc Natl Acad Sci USA. 1988;85:4894–4897. doi: 10.1073/pnas.85.13.4894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morano I, Chai GX, Baltas LG, Lamounier-Zepter V, Lutsch G, Kott M, Haase H, Bader M. Smooth-muscle contraction without smooth-muscle myosin. Nat Cell Biol. 2000;2:371–375. doi: 10.1038/35014065. [DOI] [PubMed] [Google Scholar]
- Mounier N, Desmoulière A, Gabbiani G. Subcutaneous tissue fibroblasts transfected with muscle and nonmuscle actins: a good in vitro model to study fibroblastic cell plasticity. Wound Repair Regen. 1999;7:45–52. doi: 10.1046/j.1524-475x.1999.00045.x. [DOI] [PubMed] [Google Scholar]
- Mounier N, Perriard JC, Gabbiani G, Chaponnier C. Transfected muscle and non-muscle actins are differentially sorted by cultured smooth muscle and non-muscle cells. J Cell Sci. 1997;110:839–846. doi: 10.1242/jcs.110.7.839. [DOI] [PubMed] [Google Scholar]
- Oliver T, Dembo M, Jacobson K. Traction forces in locomoting cells. Cell Motil Cytoskelet. 1995;31:225–240. doi: 10.1002/cm.970310306. [DOI] [PubMed] [Google Scholar]
- Parizi M, Howard EW, Tomasek JJ. Regulation of LPA-promoted myofibroblast contraction: role of Rho, myosin light chain kinase, and myosin light chain phosphatase. Exp Cell Res. 2000;254:210–220. doi: 10.1006/excr.1999.4754. [DOI] [PubMed] [Google Scholar]
- Pelham RJ, Jr, Wang YL. Cell locomotion and focal adhesions are regulated by substrate flexibility. Proc Natl Acad Sci USA. 1997;94:13661–13665. doi: 10.1073/pnas.94.25.13661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelham RJ, Jr, Wang YL. High resolution detection of mechanical forces exerted by locomoting fibroblasts on the substrate. Mol Biol Cell. 1999;10:935–945. doi: 10.1091/mbc.10.4.935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pena RA, Jerdan JA, Glaser BM. Effects of TGF-beta and TGF-beta neutralizing antibodies on fibroblast-induced collagen gel contraction: implications for proliferative vitreoretinopathy. Invest Ophthalmol Vis Sci. 1994;35:2804–2808. [PubMed] [Google Scholar]
- Pilcher BK, Kim DW, Carney DH, Tomasek JJ. Thrombin stimulates fibroblast-mediated collagen lattice contraction by its proteolytically activated receptor. Exp Cell Res. 1994;211:368–373. doi: 10.1006/excr.1994.1100. [DOI] [PubMed] [Google Scholar]
- Powell DW, Mifflin RC, Valentich JD, Crowe SE, Saada JI, West AB. Myofibroblasts. I. Paracrine cells important in health and disease. Am J Physiol Cell Physiol. 1999;277:C1–C19. doi: 10.1152/ajpcell.1999.277.1.C1. [DOI] [PubMed] [Google Scholar]
- Racine-Samson L, Rockey DC, Bissell DM. The role of alpha1beta1 integrin in wound contraction. A quantitative analysis of liver myofibroblasts in vivo and in primary culture. J Biol Chem. 1997;272:30911–30917. doi: 10.1074/jbc.272.49.30911. [DOI] [PubMed] [Google Scholar]
- Rastinejad F, Conboy MJ, Rando TA, Blau HM. Tumor suppression by RNA from the 3′ untranslated region of alpha-tropomyosin. Cell. 1993;75:1107–1117. doi: 10.1016/0092-8674(93)90320-p. [DOI] [PubMed] [Google Scholar]
- Rayan GM, Parizi M, Tomasek JJ. Pharmacologic regulation of Dupuytren's fibroblast contraction in vitro. J Hand Surg. 1996;21:1065–1070. doi: 10.1016/S0363-5023(96)80317-0. [DOI] [PubMed] [Google Scholar]
- Riikonen T, Koivisto L, Vihinen P, Heino J. Transforming growth factor-beta regulates collagen gel contraction by increasing alpha 2 beta 1 integrin expression in osteogenic cells. J Biol Chem. 1995;270:376–382. doi: 10.1074/jbc.270.1.376. [DOI] [PubMed] [Google Scholar]
- Rønnov-Jessen L, Petersen OW. Induction of alpha-smooth muscle actin by transforming growth factor-beta 1 in quiescent human breast gland fibroblasts. Implications for myofibroblast generation in breast neoplasia. Lab Invest. 1993;68:696–707. [PubMed] [Google Scholar]
- Rønnov-Jessen L, Petersen OW, Bissell MJ. Cellular changes involved in conversion of normal to malignant breast: importance of the stromal reaction. Physiol Rev. 1996;76:69–125. doi: 10.1152/physrev.1996.76.1.69. [DOI] [PubMed] [Google Scholar]
- Roy P, Petroll WM, Cavanagh HD, Jester JV. Exertion of tractional force requires the coordinated up-regulation of cell contractility and adhesion. Cell Motil Cytoskeleton. 1999;43:23–34. doi: 10.1002/(SICI)1097-0169(1999)43:1<23::AID-CM3>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- Sappino AP, Schurch W, Gabbiani G. Differentiation repertoire of fibroblastic cells: expression of cytoskeletal proteins as marker of phenotypic modulations. Lab Invest. 1990;63:144–161. [PubMed] [Google Scholar]
- Schevzov G, Lloyd C, Hailstones D, Gunning P. Differential regulation of tropomyosin isoform organization and gene expression in response to altered actin gene expression. J Cell Biol. 1993;121:811–821. doi: 10.1083/jcb.121.4.811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmid P, Itin P, Cherry G, Bi C, Cox DA. Enhanced expression of transforming growth factor-beta type I and type II receptors in wound granulation tissue and hypertrophic scar. Am J Pathol. 1998;152:485–493. [PMC free article] [PubMed] [Google Scholar]
- Serini G, Bochaton-Piallat ML, Ropraz P, Geinoz A, Borsi L, Zardi L, Gabbiani G. The fibronectin domain ED-A is crucial for myofibroblastic phenotype induction by transforming growth factor-beta1. J Cell Biol. 1998;142:873–881. doi: 10.1083/jcb.142.3.873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serini G, Gabbiani G. Mechanisms of myofibroblast activity and phenotypic modulation. Exp Cell Res. 1999;250:273–283. doi: 10.1006/excr.1999.4543. [DOI] [PubMed] [Google Scholar]
- Shi Y, O'Brien JE, Jr, Fard A, Zalewski A. Transforming growth factor-beta 1 expression and myofibroblast formation during arterial repair. Arterioscler Thromb Vasc Biol. 1996;16:1298–1305. doi: 10.1161/01.atv.16.10.1298. [DOI] [PubMed] [Google Scholar]
- Skalli O, Ropraz P, Trzeciak A, Benzonana G, Gillessen D, Gabbiani G. A monoclonal antibody against alpha-smooth muscle actin: a new probe for smooth muscle differentiation. J Cell Biol. 1986;103:2787–2796. doi: 10.1083/jcb.103.6.2787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soldati T, Perriard JC. Intracompartmental sorting of essential myosin light chains: molecular dissection and in vivo monitoring by epitope tagging. Cell. 1991;66:277–289. doi: 10.1016/0092-8674(91)90618-9. [DOI] [PubMed] [Google Scholar]
- Tingstrom A, Heldin CH, Rubin K. Regulation of fibroblast-mediated collagen gel contraction by platelet-derived growth factor, interleukin-1 alpha and transforming growth factor-beta 1. J Cell Sci. 1992;102:315–322. doi: 10.1242/jcs.102.2.315. [DOI] [PubMed] [Google Scholar]
- Tomasek JJ, Haaksma CJ, Eddy RJ, Vaughan MB. Fibroblast contraction occurs on release of tension in attached collagen lattices: dependency on an organized actin cytoskeleton and serum. Anat Rec. 1992;232:359–368. doi: 10.1002/ar.1092320305. [DOI] [PubMed] [Google Scholar]
- Towbin H, Staehlin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA. 1979;76:4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughan MB, Howard EW, Tomasek JJ. Transforming growth factor-beta1 promotes the morphological and functional differentiation of the myofibroblast. Exp Cell Res. 2000;257:180–189. doi: 10.1006/excr.2000.4869. [DOI] [PubMed] [Google Scholar]
- von Arx P, Bantle S, Soldati T, Perriard JC. Dominant negative effect of cytoplasmic actin isoproteins on cardiomyocyte cytoarchitecture and function. J Cell Biol. 1995;131:1759–1773. doi: 10.1083/jcb.131.6.1759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu G, Redard M, Gabbiani G, Neuville P. Cellular retinol-binding protein-1 is transiently expressed in granulation tissue fibroblasts and differentially expressed in fibroblasts cultured from different organs. Am J Pathol. 1997;151:1741–1749. [PMC free article] [PubMed] [Google Scholar]
- Yao X, Chaponnier C, Gabbiani G, Forte JG. Polarized distribution of actin isoforms in gastric parietal cells. Mol Biol Cell. 1995;6:541–557. doi: 10.1091/mbc.6.5.541. [DOI] [PMC free article] [PubMed] [Google Scholar]