Abstract
AIM
To examine the relationship between the single nucleotide polymorphism CXCL10 rs1439490 and seronegative occult hepatitis C virus (HCV) infection (OCI).
METHODS
One hundred and three cases of seronegative OCI and 155 cases of seropositive chronic HCV infection (CHC) were diagnosed at five Liver Centers in Northeastern China, from 2012 to 2016. CXCL10 rs1439490, rs1440802, and IL-28B rs12979860 were analyzed by sequencing. Serum CXCL10 was measured by ELISA. Intrahepatic CXCL10 was determined by quantitative PCR and immunohistochemical semi-quantitative scoring. Liver necroinflammation and fibrosis were scored according to the METAVIR system.
RESULTS
CXCL10 rs1439490 G/G was more prevalent in OCI patients (n = 93/103; 90.3%) than in CHC patients (n = 116/155; 74.8%; P = 0.008). OCI patients had lower serum CXCL10 levels than CHC patients (192.91 ± 46.50 pg/mL vs 354.78 ± 102.91 pg/mL, P < 0.0001). Of IL-28B rs12979860 C/C patients, OCI patients with rs1439490 G/G had lower serum and liver levels of CXCL10 and lower levels of liver necroinflammation and fibrosis than non-G/G patients. OCI patients had higher alanine aminotransferase normalization rates after Peg-interferon treatment than CHC patients (P < 0.05) and serum CXCL10 decreased significantly (P < 0.0001). Liver necroinflammation and fibrosis were alleviated in 8 OCI patients after treatment. Multivariate analysis indicated that rs1439490 G/G significantly influenced the occurrence of OCI in HCV infection (OR = 0.31, 95%CI: 0.15-0.66, P = 0.002).
CONCLUSION
CXCL10 rs1439490 G/G is positively associated with OCI in HCV infection and antiviral outcome.
Keywords: Occult hepatitis C virus infection, CXCL10, Single nucleotide polymorphisms, rs1439490
Core tip: We demonstrated that CXCL10 rs1439490 G/G was more prevalent in patients with seronegative occult hepatitis C virus infection (OCI) than in those with seropositive chronic hepatitis C virus (HCV) infection (CHC). Rs1439490 G/G OCI patients had lower serum and liver levels of CXCL10, and lower levels of liver necroinflammation and fibrosis than non-G/G patients. OCI patients had higher alanine aminotransferase normalization rates after Peg-interferon treatment than CHC patients and serum CXCL10 decreased significantly. We, for the first time, showed that CXCL10 rs1439490 G/G may be positively associated with OCI in HCV infection and antiviral outcome.
INTRODUCTION
Occult hepatitis C virus (HCV) infection (OCI) involves the detection of intrahepatic HCV RNA by percutaneous transhepatic liver biopsy in patients with long-standing liver dysfunction who are seronegative for anti-HCV antibodies and RNA[1]. OCI was proposed as a subtype of chronic HCV infection (CHC)[2]. It was found to occur in 8.9% of patients with cryptogenic hepatic disease[3], 1.27% of infectious liver disease-free subjects[4], and in patients without conventional HCV markers but abnormal liver enzyme levels, or healthy subjects with normal alanine aminotransferase (ALT) levels and no clinical evidence of liver disease[1,3-12].
To date, the pathogenesis of OCI remains unclear. It is unknown why OCI patients do not produce anti-HCV antibodies after exposure to HCV and why serum HCV RNA is not detectable. Low detection sensitivity has been implicated as a major reason for OCI[13], which may explain the existence of occult infection in anti-HCV seropositive and HCV RNA seronegative individuals. Mutant HCV strains that may subvert the conventional HCV assays have also been implicated in OCI[14]. However, the 3rd generation HCV antibody detection assays cover most HCV structural and nonstructural antigens and achieve up to 99% sensitivity[15]. Instead, OCI may be the result of “sporadic” exposure to trace amounts of HCV[16] that generate insufficient T cell activation and B cell responses against infection. Consequently, serum anti-HCV activity cannot be detected by current clinical methods. Indeed, persistent low levels of virus-specific T-cell responses have been identified in OCI patients[17,18]. However, this host immune response can only partially suppress HCV replication, but cannot eliminate the virus or viral antigens. Thus, HCV RNA remains detectable in the liver – the primary target tissue.
Patients with OCI have distinct clinical outcomes from those with CHC even of the same genotypes, indicating the role of host factors in OCI pathogenesis. The interleukin-28B (IL-28B) locus has been associated with HCV outcomes and IL-28B C/C was shown to occur more often in OCI than in CHC patients[19]. In OCI patients, intrahepatic HCV RNA load was significantly lower in those with the IL-28B C/C genotype than in those with C/T or T/T genotypes[19]. Thus, IL-28B polymorphisms may affect endogenous IFN-λ levels and be associated with low viral replication in some patients. However, interferon (IFN) has also been shown not to play a determining role in OCI occurrence, and IL-28B C/C OCI patients were found to have lower serum levels of CXC chemokine ligand 10 (CXCL10) than IL-28B C/C CHC patients[19]. Therefore, regulation of OCI and the associated disease progression likely involves additional host immune factors.
The importance of CXCL10 expression during chronic hepatitis B virus (HBV) infection has recently been emphasized. Two single nucleotide polymorphisms (SNPs) of CXCL10 (G-201A and C-1513T) were reported to have high allele frequency in chronic HBV infected Chinese populations. The polymorphism G-201A in the CXCL10 promoter was also implicated in the genetic variation underlying the susceptibility to chronic HBV infection progression[20]. G-201A is located within the CXCL10 promoter region and is proximal to the NF-κB1/2 binding sites. The G-201A SNP is associated with the expression of CXCL10 in peripheral blood mononuclear cells (PBMC), underpinning the mechanism of chronic HBV disease progression. Based on this large cohort study, and the observation that both HCV and HBV promote the development of hepatic lesions and fibrosis by inducing inflammatory infiltration rather than by damaging hepatocytes directly, we hypothesized that CXCL10 G-201A may also affect the disease manifestation of CHC. However, to date, there is no such report.
In this study, we examined the expression frequency of CXCL10 G-201A (rs1439490), C-1513T (rs1440802) and IL-28B rs12979860 SNPs in OCI and CHC patients to investigate whether these polymorphisms are associated with OCI. In addition, we further analyzed the correlation of these SNPs with the serum and liver levels of CXCL10 and liver HCV RNA levels in OCI patients.
MATERIALS AND METHODS
Patients
A total of 1796 patients with liver dysfunction and/or radiographic abnormalities of unknown etiology underwent liver biopsy between 2012 and 2016 at five hospitals in Northeastern China (China-Japan Union Hospital of Jilin University, People’s Hospital of Jilin City, Fourth Affiliated University of Harbin Medical University, People’s Hospital of Hunchun City, and the Second People’s Hospital of Daqing City). All patients were Han Chinese. Subjects seronegative for anti-HCV antibodies and HCV RNA, but with detectable intrahepatic HCV RNA were included in the OCI group (n = 103). One hundred and fifty-five normal CHC patients who underwent liver biopsy prior to antiviral therapy during the same period were included in the CHC control group. Informed consent forms were obtained from all patients. The study was approved by the Institutional Review Boards of the individual centers (registration number: ChiCTR-ONRC-12002207).
Inclusion/exclusion criteria
OCI inclusion criteria were as follows: (1) Serum anti-HCV antibodies and HCV RNA negative in 3 consecutive tests within at least 3 mo, and persistent liver dysfunction and/or radiographic abnormalities; (2) HCV RNA/HBV DNA seronegative after ultracentrifugation and undetectable in PBMC; and (3) HCV RNA-positive in liver tissue. CHC inclusion criteria were as follows: (1) Serum anti-HCV antibodies and HCV RNA positive, and diagnosed with CHC in accordance with the EASL guidelines[21]; and (2) consent to receive hepatic histological evaluation prior to anti-HCV treatment.
Exclusion criteria were as follows: (1) Occult HBV infection, drug-induced liver disease, fatty liver disease, autoimmune liver disease, inherited metabolic liver disease after liver biopsy; (2) co-infection with other types of hepatitis (A, D, E), Epstein-Barr virus, cytomegalovirus, or human immunodeficiency virus; (3) chronic HCV infection complicated with decompensated cirrhosis or primary liver cancer; (4) severe heart, brain or kidney complications; and (5) received or receiving pegylated IFN (Peg-IFN) plus ribavirin (RBV) or IFN treatment.
SNP analyses of CXCL10 rs1439490, rs1440802, and IL-28B rs12979860
Peripheral blood samples were collected from the patients, placed in anticoagulant EDTA-treated tubes, and genomic DNA was extracted using a Puregene SK8224 DNA isolation kit from Sangon Biotech (Shanghai, China), according to the manufacturer’s instructions. The primers targeting specific fragments were designed and synthesized by Sangon Biotech: CXCL10 promoter region G-201A rs1439490[20]: Forward: 5’-TTCAGTAACATAAACCCCAACAA-3’; Reverse: 5’-CACAAAGGAAGACAATAAGGGAG-3’. CXCL10 promoter region C-1513T rs1440802: Forward[20]: 5’-CTCACTTTGTCTCACCAATCTCA-3’; Reverse: 5’-CAGAGAAATGAGAGACCTAAGTGTG-3’. IL-28B rs12979860[22]: Forward: 5’-CCTCTGCACAGTCTGGGATTC-3’; Reverse: 5’-GCTCAGGGTCAATCACAGAAG-3’.
Serum CXCL10 levels
Serum CXCL10 levels were measured by human CXCL10 Quantikine ELISA (R&D, Minneapolis, MN, United States)[20]. The sensitivity of detection was 38-1340 pg/mL.
Liver necroinflammation activity and fibrosis
Liver tissues were obtained by percutaneous transhepatic liver biopsy and routinely stained with hematoxylin and eosin (HE). The biopsies were examined by experienced pathologists. Hepatic necroinflammation activity and fibrosis stages were scored according to the METAVIR scoring system[23] as shown in Tables 1 and 2.
Table 1.
Algorithm for evaluation of histological activity
| Piecemeal necrosis | + | Lobular necrosis | = | Histological activity score |
| 0 (none) | 0 (none or mild) | 0 (none) | ||
| 0 | 1 (moderate) | 1 (mild) | ||
| 0 | 2 (severe) | 2 (moderate) | ||
| 1 (mild) | 0, 1 | 1 | ||
| 1 | 2 | 2 | ||
| 2 (moderate) | 0, 1 | 2 | ||
| 2 | 2 | 3 (severe) | ||
| 3 (severe) | 0, 1, 2 | 3 |
Table 2.
Fibrosis scoring
| Description | Score |
| No fibrosis | 0 |
| Stellate enlargement of portal tract but without septa formation | 1 |
| Enlargement of portal tract with rare septa formation | 2 |
| Numerous septa without cirrhosis | 3 |
| Cirrhosis | 4 |
Liver immunohistochemistry
Biopsied hepatic tissues were immunohistochemically stained with anti-CXCL10 antibody (Abcam, Cambridge, United Kingdom). The staining intensity was assessed in 10 high-power fields based on the following scale: Score 0 – negative (-), staining absent; score 1 – weakly positive (+), yellowish; score 2 – moderately positive (++), brown; score 3 – strongly positive (+++), dark brown. The staining intensity in each field was calculated as: IS (intensity score) = Σ[(0 × F-)+(1 × F+)+(2 × F++)+(3 × F+++)], in which F is the percentage of cells stained at each intensity. The average score of 10 fields was the quantitative result of the whole slide[24]. Sections were scored by two independent observers.
Intrahepatic CXCL10 mRNA
RNA was extracted from biopsied liver tissue and subjected to quantitative real-time PCR using the following primers: Forward: 5’-CTGAATCCAGAATCGAAGGCCATC-3’; Reverse: 5’-TGTAGGGAAGTGATGGGAGAGG-3’. The expression was normalized to the expression of house-keeping gene β-actin using primers as described previously[20].
Antiviral therapy
Patients were treated with Peg-IFNα-2a (180 mg subcutaneous once weekly) plus RBV (1200 mg/d if body weight > 75 kg and 1000 mg/d if ≤ 75 kg for HCV genotype 1; 800 mg/d for non-genotype 1)[21]. Genotype 1 OCI patients were treated for 48 wk and non-genotype 1 patients for 24 wk. For CHC patients, treatment was in accordance with the EASL guidelines[21]. Patients who received more than 80% of the cumulative total planned dose were considered to have completed the treatment[25]. All patients were followed up for 24 wk after treatment and a second liver biopsy was performed 24-96 wk after therapy in patients with informed consent.
Measures and monitoring
A standardized sample collection and data analysis protocol was applied at the five liver centers, including ELISA for serum anti-HCV antibodies (Roche Molecular Diagnostics, NJ, United States), highly sensitive, real-time PCR-based assay for HCV RNAs (LOD 15 IU/mL; COBAS Ampliprep/COBAS TaqMan 48 Analyzer, Roche), direct sequencing of serum or intrahepatic RNA for HCV genotyping (SinoMD, China), and Fibroscan for liver fibrosis (Echosens, France). Serum samples negative in routine HCV RNA tests were further ultracentrifuged and retested. If the ultracentrifuged serum remained HCV negative, PBMC were tested. In OCI patients, HCV RNA levels were re-assessed every 12 wk after initiation of antiviral treatment until the end of treatment or follow up. CHC patients had serum HCV RNA tests at 4 and 12 wk after initiation of treatment, and then every 12 wk until the end of treatment or follow up.
Statistical analysis
Allele frequencies for each SNP were determined by the Hardy-Weinberg equilibrium test and the differences between groups were examined by Chi-square tests. Qualitative results were expressed as frequency and percentage, and statistical analyses were performed using the Chi-square test or Fisher exact probability test. Quantitative data were expressed as mean ± SD and analyzed using the Student t-test. Stepwise binary logistic regression analysis was used to determine the influencing factors. A two-sided P value less than 0.05 was considered statistically significant, and odds ratios (ORs) and 95% confidence intervals (95%CI) were assessed by SPSS software (v19.0; SPSS Inc., Chicago, IL, United States).
RESULTS
Clinical characteristics of seronegative OCI patients compared with seropositive CHC patients
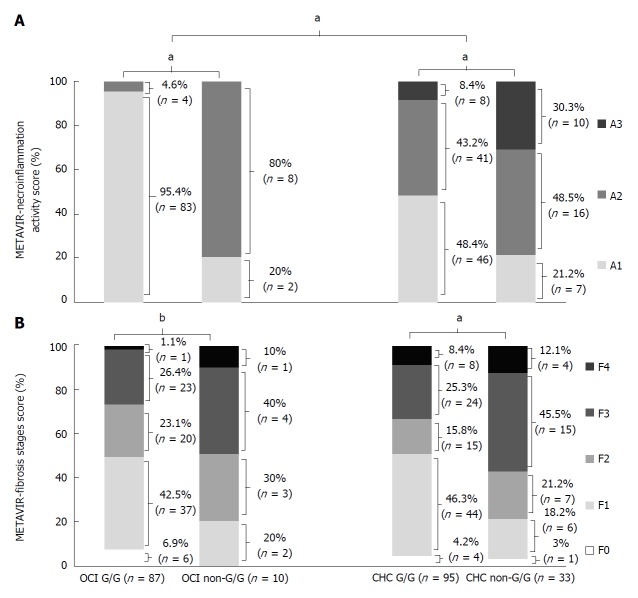
The clinical characteristics of OCI and CHC patients are shown in Table 3. OCI patients were significantly older than CHC patients (52.16 years vs 42.70 years; P < 0.001) and had higher rates of HCV infection history in family members (28.2% vs 12.3%; P < 0.001). In contrast, the OCI group had lower base levels of ALT than the CHC group (61.13 ± 23.54 IU/L vs 93.17 ± 55.39 IU/L; P < 0.001), lower rates of blood transfusion/surgery/tattoo history (7.8% vs 16.8%; P = 0.027), lower levels of intrahepatic HCV RNA (3.19 ± 1.05 vs 5.48 log10 IU/mL ± 1.49 log10 IU/mL; P < 0.001), and lower METAVIR necroinflammation activity scores (1.14 ± 0.34 vs 1.69 ± 0.68; P < 0.001). No significant difference in HCV genotype and METAVIR fibrosis stages were observed between the two groups.
Table 3.
Clinical characteristics of patients enrolled in this study n (%)
| Seronegative OCI patients (n = 103) | Seropositive CHC patients (n = 155) | P value | |
| Gender | 0.664 | ||
| Male | 58 (56.3) | 83 (53.5) | |
| Female | 45 (43.7) | 72 (46.5) | |
| Age (yr) | 52.16 ± 7.64 | 42.70 ± 9.15 | < 0.001 |
| ALT (IU/L) | 61.13 ± 23.54 | 93.17 ± 55.39 | < 0.001 |
| GGT | 56.31 ± 16.63 | 52.86 ± 15.69 | 0.093 |
| BMI | 23.73 ± 2.38 | 24.12 ± 2.36 | 0.192 |
| HOMA-IR | 2.39 ± 0.15 | 2.42 ± 0.18 | 0.219 |
| Transfusion/surgery/tattoo history | 8 (7.8) | 26 (16.8) | 0.027 |
| Family history of HCV infection | 29 (28.2) | 19 (12.3) | < 0.001 |
| Intrahepatic HCV RNA (log10 IU/mL) | 3.19 ± 1.05 | 5.48 ± 1.49 | < 0.001 |
| HCV genotype | 0.89 | ||
| Genotype 1 | 66 (64.1) | 98 (63.2) | |
| Non-genotype 1 | 37 (35.9) | 57 (36.8) | |
| Fibrosis (Fibroscan) | 0.317 | ||
| F0-1 | 65 (63.1) | 104 (67.1) | |
| F2-4 | 38 (36.9) | 51 (32.9) | |
| METAVIR activity score | 1.14 ± 0.34 | 1.69 ± 0.68 | < 0.001 |
| METAVIR fibrosis score | 1.82 ± 0.98 | 1.87 ± 1.07 | 0.673 |
P < 0.05; ALT: Alanine aminotransferase; GGT: Gamma-glutamyl transpeptidase; BMI: Body mass index; HOMA-IR: Homeostasis model assessment of insulin resistance; HCV non-genotype 1: Other HCV genotypes except for genotype 1, including HCV 2-6 genotypes.
Serum CXCL10 levels in OCI patients with different polymorphisms of IL-28B rs12979860
Serum CXCL10 levels in OCI and CHC patients were compared. As shown in Figure 1, the OCI group exhibited significantly lower serum CXCL10 levels than the CHC group (192.91 ± 46.50 pg/mL vs 354.78 ± 102.91 pg/mL, P < 0.0001), irrespective of IL-28B rs12979860 C/C or non-C/C (C/T+ T/T). However, serum CXCL10 levels did not differ significantly between IL-28B rs12979860 polymorphism (OCI: 191.75 ± 45.04 pg/mL vs 211.67 ± 68.56 pg/mL, P = 0.311; CHC: 356.42 ± 106.10 pg/mL vs 347.0 ± 87.50 pg/mL, P = 0.667) (Figure 1).
Figure 1.
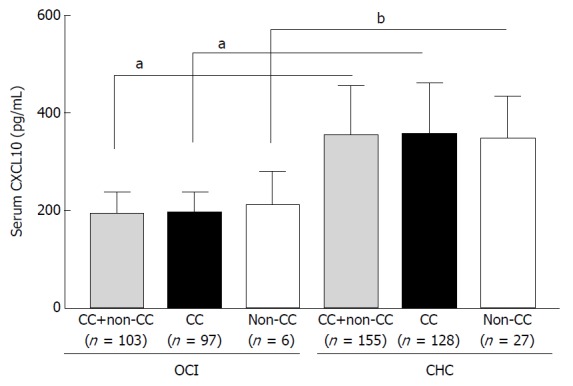
Serum CXCL10 levels in occult hepatitis C virus infection patients with different variants of IL-28B rs12979860 (C/C or non-C/C) as compared to chronic hepatitis C virus infection patients. aP < 0.0001; bP = 0.001. OCI: Occult hepatitis C virus infection; CHC: Chronic hepatitis C virus infection.
CXCL10 polymorphisms in OCI patients compared with CHC patients
CXCL10 G-201A G/G genotype was more prevalent in the OCI group (93 of 103; 90.4%) than in the CHC group (116 out of 155; 74.8%; P = 0.008) (Table 4). The distribution of CXCL10 rs1440802 (C-1513T) did not differ significantly between the two groups (P = 0.733) (Table 4). Of the patients with IL-28B rs12979860 C/C genotype (Table 5), OCI was associated with a higher frequency of CXCL10 G-201A G/G (87 of 97; 89.7%) than CHC (95 of 128; 74.2%; OR = 0.33; 95%CI: 0.15-0.71; P = 0.005).
Table 4.
IL-28B single nucleotide polymorphism rs12979860 and CXCL10 single nucleotide polymorphism G-201A, C-1513T in occult hepatitis C virus infection and chronic hepatitis C virus infection patients n (%)
| Seronegative OCI patients (n=103) | Seropositive CHC patients (n=155) | P value | |
| IL-28BSNP rs12979860 | |||
| C/C | 97 (94.2%) | 128 (82.6%) | 0.009 |
| Non-C/C | 6 (5.8%) | 27 (17.4%) | |
| CXCL10 SNP rs1439490 (G-201A) | |||
| G/G | 93 (90.4%) | 116 (74.8%) | 0.003 |
| G/A | 9 (8.7%) | 35 (22.6%) | |
| A/A | 1 (0.9%) | 4 (2.6%) | |
| CXCL10 SNP rs1440802 (C-1513T) | |||
| C/C | 26 (25.2%) | 40 (25.8%) | 0.733 |
| C/T | 54 (52.4%) | 78 (50.3%) | |
| T/T | 23 (22.4%) | 37 (23.9%) |
P value: OCI group compared with CHC group; P < 0.05; IL-28BSNP rs12979860 non-C/C genotype included IL-28BSNP rs12979860 C/T + T/T genotypes. OCI: Occult hepatitis C virus infection; CHC: Chronic hepatitis C virus infection.
Table 5.
CXCL10 single nucleotide polymorphism G-201A in occult hepatitis C virus infection and chronic hepatitis C virus infection patients with IL-28B rs12979860 C/C n (%)
| Seronegative OCI patients (n = 97) | Seropositive CHC patients (n = 128) | P value | OR (95%CI) | |
| G/G | 87 (89.7) | 95 (74.2) | 0.005 | 0.331 (0.154-0.711) |
| Non-G/G (G/A + A/A) | 10 (10.3) | 33(25.8) |
P value: OCI group compared with CHC group; P < 0.05; OR: odds ratio; 95%CI: 95% confidence interval; CXCL10 rs1439490 non-G/G genotype included CXCL10 rs1439490 G/A + A/A genotypes. OCI: Occult hepatitis C virus infection; CHC: Chronic hepatitis C virus infection.
Serum and liver CXCL10 levels, and HCV RNA levels in OCI patients with different CXCL10 G-201A polymorphisms
The relationship between IL-28B rs12979860 polymorphism and CXCL10 is unclear. We showed that rs12979860 C/C patients had similar serum levels of CXCL10 to non-C/C patients in both the OCI and CHC group (Figure 1, P = 0.311 and 0.667). Due to the prevalence of IL-28B rs12979860 C/C genotype within both groups, we next compared the serum and liver CXCL10 levels and HCV RNA levels in rs12979860 C/C patients with different CXCL10 G-201A SNPs. Rs12979860 C/C OCI patients had lower serum CXCL10 levels, lower levels of intrahepatic CXCL10 mRNA (Figure 2B, 1.18 ± 0.27 vs 2.24 ± 0.65, P < 0.0001) and immunohistological staining scores (Figure 2C and D, 0.94 ± 0.34 vs 2.71 ± 0.52, P < 0.0001), as well as HCV RNA than CHC patients (Figure 2E; 3.20 ± 1.07 log10 IU/mL vs 5.53 ± 1.46 log10 IU/mL, P < 0.0001). In addition, within both groups, CXCL10 rs1439490 G/G patients had lower levels of serum and liver CXCL10 (Figure 2A, OCI: 184.82 ± 39.19 pg/mL vs 252.10 ± 49.52 pg/mL, P < 0.0001; CHC: 333.91 ± 101.01 pg/mL vs 421.24 ± 94.19 pg/mL, P < 0.0001; Figure 2B, OCI: 1.16 ± 0.25 vs 1.42 ± 0.29, P = 0.003; CHC: 2.10 ± 0.61 vs 2.65 ± 0.60, P < 0.0001; Figure 2C and D, OCI: 0.91 ± 0.33 vs 1.20 ± 0.34, P = 0.009; CHC: 2.07 ± 0.51 vs 2.45 ± 0.47, P < 0.0001), except for intrahepatic HCV RNA (Figure 2E; OCI: 3.25 ± 1.09 log10 IU/mL vs 2.76 ± 0.66 log10 IU/mL, P = 0.163; CHC: 5.47 ± 1.42 log10 IU/mL vs 5.71 ± 1.58 log10 IU/mL, P = 0.422).
Figure 2.
Comparison of serum CXCL10 levels (A), liver CXCL10 mRNA levels (B), representative (C) and grouped (D) liver CXCL10 IHC scoring, and liver hepatitis C virus RNA levels (E) among IL-28B rs12979860 C/C patients with different CXCL10 rs1439490 polymorphisms. aP < 0.0001; bP = 0.003; cP = 0.009. OCI: Occult hepatitis C virus infection; CHC: Chronic hepatitis C virus infection.
METAVIR scores in OCI patients with different variants of CXCL10 G-201A
In IL-28B rs12979860 C/C patients, those with OCI had lower hepatic necroinflammation scores than CHC patients, as evaluated by the METAVIR scoring system (P < 0.0001). However, METAVIR scored liver fibrosis stages did not differ significantly (P = 0.67). Necroinflammation activity (OCI: 1.05 ± 0.21 vs 1.80 ±0.42, P < 0.0001; CHC: 1.60 ± 0.64 vs 2.09 ± 0.72, P < 0.0001) and fibrosis stage (OCI: 1.72 ± 0.99 vs 2.40 ± 0.69, P = 0.04; CHC: 1.64 ± 1.03 vs 2.58 ± 1.03, P < 0.0001) scores were both lower in CXCL10 G-201A G/G than in non-G/G SNP patients (Figure 3).
Figure 3.
METAVIR necroinflammation activity (A) and fibrosis stage scores of patients with IL-28B rs12979860 CC genotype (B). aP < 0.0001; bP = 0.04. OCI: Occult hepatitis C virus infection; CHC: Chronic hepatitis C virus infection.
Efficacy of antiviral treatment in OCI patients with different CXCL10 G-201A variants
A total of 73 OCI patients and 90 CHC patients completed more than 80% of the planned antiviral drug doses. After 12 wk of treatment, ALT normalization occurred in 95.9% of OCI and 82.2% of CHC patients (P < 0.05; data not shown). In G-201A G/G OCI patients with lower baseline serum CXCL10, serum CXCL10 levels decreased to 60.90 ± 16.78 pg/mL and 57.2 5 ± 19.51 pg/mL at the endpoint of antiviral treatment and at 24 wk follow up, respectively. In G-201A G/G CHC patients, serum CXCL10 levels decreased to 89.77 ± 35.94 pg/mL and 73.33 ± 22.64 pg/mL at these time points - approximately 1.4-fold higher than in OCI patients (Figure 4).
Figure 4.
Effect of antiviral therapy on serum CXCL10 levels of IL-28 rs12979860 C/C patients with different CXCL10 G-201A single nucleotide polymorphisms. A: The baseline serum CXCL10 levels of IL-28 rs12979860 C/C patients who completed the Peg-IFNα plus ribavirin treatment; B-C: The serum CXCL10 levels of the patients at the endpoint of antiviral treatment (B) and at 24 wk follow up (C); aP < 0.0001; bP = 0.038; cP = 0.008; dP = 0.008; eP = 0.009. OCI: Occult hepatitis C virus infection; CHC: Chronic hepatitis C virus infection.
Eight OCI patients who completed the course of treatment had a second liver biopsy. All these CXCL10 G-201A G/G patients had undetectable intrahepatic HCV RNA. CXCL10 mRNA, necroinflammation activity and fibrosis scores also decreased (Table 6). In contrast, of the 5 CHC patients who finished ≥ 80% of planned doses and achieved SVR, 2 patients (1 G/G and 1 non-G/G) remained intrahepatic HCV RNA detectable and only 3 patients achieved intrahepatic HCV RNA conversion to negative (2 patients with G-201A G/G and 1 patient with non-G/G). Nevertheless, CXCL10 mRNA, necroinflammation activity, and fibrosis scores in liver tissue all decreased after antiviral treatment (Table 7).
Table 6.
Intrahepatic hepatitis C virus RNA, CXCL10 mRNA, and METAVIR scores of 8 seronegative occult hepatitis C virus infection patients who underwent a second liver biopsy
|
Patient ID |
CXCL10
G-201A SNP |
Intrahepatic HCV RNA (log10 IU/mL) |
Relative liver CXCL10 mRNA |
METAVIR
necroinflammation score |
METAVIR
fibrosis scores |
||||
|
Before |
After |
Before |
After |
Before |
After |
Before |
After | ||
| 1 | G/G | 2.17 | (-) | 1.20 | 0.80 | 1 | 0 | 0 | 0 |
| 2 | G/G | 3.57 | (-) | 0.95 | 0.90 | 1 | 0 | 1 | 1 |
| 3 | G/G | 2.72 | (-) | 0.74 | 0.63 | 1 | 1 | 1 | 1 |
| 4 | G/G | 5.08 | (-) | 1.31 | 0.94 | 1 | 0 | 1 | 0 |
| 5 | G/G | 2.83 | (-) | 1.42 | 0.64 | 1 | 0 | 0 | 0 |
| 6 | G/G | 1.97 | (-) | 1.06 | 0.83 | 2 | 1 | 1 | 1 |
| 7 | G/G | 4.64 | (-) | 1.03 | 0.70 | 2 | 0 | 2 | 1 |
| 8 | G/G | 2.94 | (-) | 1.26 | 0.71 | 1 | 1 | 3 | 1 |
SNP: Single nucleotide polymorphism; HCV: Hepatitis C virus.
Table 7.
Intrahepatic hepatitis C virus RNA, CXCL10 mRNA, and METAVIR scores of 5 seropositive chronic hepatitis C virus infection patients who underwent a second liver biopsy
| Patient ID | CXCL10 G-201A SNP |
Intrahepatic HCV RNA (log10 IU/mL) |
Relative Liver CXCL10 mRNA |
METAVIR
necroinflammation score |
METAVIR
fibrosis scores |
||||
| Before | After | Before | After | Before | After | Before | After | ||
| 1 | G/G | 6.18 | (-) | 1.93 | 0.93 | 1 | 0 | 2 | 1 |
| 2 | G/G | 3.9 | (-) | 1.80 | 0.90 | 2 | 1 | 1 | 1 |
| 3 | G/A | 7.74 | 1.94 | 2.78 | 1.30 | 2 | 1 | 3 | 2 |
| 4 | G/A | 7.38 | (-) | 2.39 | 1.24 | 2 | 1 | 2 | 2 |
| 5 | G/G | 5.51 | 1.38 | 1.31 | 1.12 | 1 | 1 | 2 | 1 |
SNP: Single nucleotide polymorphism; HCV: Hepatitis C virus.
Logistic regression analysis of factors associated with OCI
As shown in Table 8, age, blood transfusion, family history of HCV infection, low levels of intrahepatic HCV RNA, IL-28B rs12979860 C/C genotype, and CXCL10 G-201A G/G genotype all influenced OCI occurrence (all P < 0.05). Furthermore, multivariate analysis showed that CXCL10 G-201A G/G genotype (OR = 0.31, 95%CI: 0.15-0.66; P = 0.002) and IL-28B rs12979860 C/C genotype (OR = 0.28, 95%CI: 0.11-0.71; P = 0.008) significantly influenced occult occurrence in patients with HCV infection.
Table 8.
Logistic regression analysis of factors associated with seronegative occult occurrence of hepatitis C virus
| Variable |
Univariate analysis |
Multivariate analysis |
||||
| OR | 95%CI | P value | OR | 95%CI | P value | |
| Age | 1.15 | 1.11-1.19 | < 0.001 | |||
| Blood transfusion | 0.39 | 0.17-0.92 | 0.031 | |||
| Family history of HCV | 3.66 | 1.85-7.25 | < 0.001 | |||
| Intrahepatic HCV RNA level | 0.30 | 0.23-0.39 | < 0.001 | |||
| IL-28B C/C | 0.29 | 0.12-0.74 | 0.009 | 0.28 | 0.11-0.71 | 0.008 |
| CXCL10 G-201A G/G | 0.32 | 0.15-0.68 | 0.003 | 0.31 | 0.15-0.66 | 0.002 |
P < 0.05; OR: Odds ratio
DISCUSSION
CXC chemokine ligand (CXCL-10), also known as IFN-gamma inducible protein (IP-10), is a small and potent cytokine belonging to the C-X-C motif chemokine family. CXCL10 was previously considered an IFN-stimulated gene; however, CXCL10 induction in hepatocytes during acute HCV infection does not require IFNs. Infected hepatocytes and intrahepatic infiltrated lymphocytes secrete CXCL10 within the first days of HCV infection[26]. HCV-associated pathogen-associated molecular patterns (PAMPs) have recently been reported to be capable of directly activating the cellular innate immune pathways[26,27]. HCV RNAs or intermediates during viral replication can directly activate toll-like receptor 3 (TLR3) and retinoic acid-inducible gene-I (RIG-I), and induce the activation of nuclear factor-kappa B (NF-κB) via a myeloid differential protein-88-independent pathway (MyD88-independent pathway). NF-κB was found to positively regulate CXCL10 transcription during HCV infection as well as following exposure to poly(I·C) (a TLR3 agonist) and 5′ poly(U) HCV RNA (a RIG-I agonist) from two viral genotypes[26]. In addition, the transiently nuclear translocated interferon regulatory factor 3 (IRF3) was recruited to the proximal interferon sensitive response element (ISRE) during HCV infection and activated the CXCL10 promoter independently of type I/III IFN signaling. In vitro experiments also demonstrated that during early HCV infection, Huh7-derived cells expressing both TLR3 and RIG-I produced maximal CXCL10 mRNA with negligible induction of type I or III IFN, and neutralization of type I and type III IFN did not affect CXCL10 induction[26,27].
The engagement of CXCL10 with C-X-C motif chemokine receptor 3 (CXCR3) expressed on the surface of CD4+ Th1 cells, natural killer (NK) cells, and CD8+ cytotoxic T cells induces the activation and migration of these cells to inflammatory sites[27,28]. Within the liver, the activated CD4+Th1 cells produce more IFN-γ and tumor necrosis factor alpha (TNF-α), which in turn stimulate further secretion of CXCL10 from liver cells. Thus, the CXCL10-CXCR3 axis creates an amplification feedback loop (second paracrine signaling pathway) and maintains a sustained adaptive immune response, which plays an important role in viral suppression during acute HCV infection. However, this autoimmune process is not able to eliminate the virus in approximately 70% of patients and the virus persists for decades[29]. Instead, the CXCL10-CXCR3 signaling cytokines and cytotoxic factors released from CD4+ Th1 cells contribute to chronic liver inflammation and is termed the CXCL10-mediated non-specific immune response.
This study and that by Bartolomé et al[19] revealed that serum CXCL10 levels in OCI patients were lower than in CHC patients, suggesting an underlying role of CXCL10 in the lower levels of HCV replication in OCI patients and the chronic immune response. IL-28B polymorphisms may affect the endogenous IFN-λ level, and thus are associated with low viral replication. We observed a higher prevalence of IL-28B rs12979860 C/C in OCI than in CHC patients, which is consistent with Bartolomé’s report[19]. This phenomenon could partially explain the suppression of HCV replication in OCI patients; however, low expression of CXCL10 in the context of IL-28B C/C genotype-associated high endogenous IFN expression remains to be understood.
Deng et al[20] recently reported that two CXCL10 SNPs, G-201A and C-1513T, were overrepresented in Chinese populations from Beijing and Chongqing with chronic HBV infection. G-201A locates within the CXCL10 promoter region and is proximal to the NF-κB1/2 binding sites. G-201A SNP was associated with the expression of CXCL10 in PBMC and chronic HBV disease progression[20]. This study inspired us to investigate whether these CXCL10 SNPs were associated with OCI. The general population in the Northeast area of China (Jilin and Heilongjiang Provinces) recruited in our study had a similar allele frequency of CXCL10 G-201A and C-1513T to the general population in Deng’s study (data not shown). However, G-201A G/G genotype, but not C-1513T, was more prevalent in OCI patients. OCI patients with G-201A G/G had not only lower levels of serum CXCL10, but also lower levels of liver mRNA and CXCL10 protein. OCI patients also exhibited less severe liver METAVIR necroinflammation activity and fibrosis. We hypothesize that CXCL10 G-201A may influence the secretion of CXCL10, and subsequently the binding of CXCL10 to CXCR3 on the surface of Th1 cells. As such, the CXCL10-CXCR3 axis-mediated adaptive immune response is compromised. This concession would affect spontaneous clearance of the virus, but may also cause less liver damage. These extremely low levels of HCV replication are not sufficient to elicit anti-HCV antibodies. However, long-term HCV replication still promotes liver disease.
With respect to antiviral treatment, the comprehensive antiviral efficacy was better in OCI patients than in CHC patients, with CXCL10 G-201A G/G OCI patients even better than non-G/G OCI patients. ALT normalization rate increased along with the decrease in serum CXCL10 level. Due to the requirement for intrahepatic biopsy, only 8 OCI patients and 5 CHC patients who finished antiviral treatment consented to receive a second liver biopsy after treatment. Nevertheless, the results showed a decreased tendency of METAVIR liver necroinflammation activity and fibrosis scores along with the decrease in liver CXCL10 mRNA and protein levels. All 8 OCI patients achieved conversion to liver HCV RNA negative and 5 CHC patients achieved seroconversion to HCV RNA negative with 3 cases of conversion to liver HCV RNA negative. The relationship between decreased serum CXCL10 levels in CHC patients and antiviral treatment efficacy is complicated. It remains to be determined whether this is due to the suppression of HCV RNA or direct inhibition of viral replication by CXCL10. High CXLC10 levels were considered to negatively affect the antiviral efficacy of IFN-based treatment. However, CXCL10 levels have also been reported to affect non-IFN therapy for HCV infection, and are implicated as a surrogate marker of intracellular viral replication complex decay[30,31].
In summary, our study revealed a higher prevalence of CXCL10 G-201A (rs1439490) G/G in OCI patients than in CHC patients. OCI patients with G-201A G/G achieved better antiviral efficacy with Peg-IFN plus RBV. CXCL10 G-201A G/G is associated with a seronegative occult response to HCV infection, and may be an independent prognostic factor for IFN-based antiviral treatment. Our results suggest the potential clinical significance of CXCL10 G-201A genotyping in identifying OCI during chronic HCV infection. In addition, clarifying the correlation between CXCL0 rs1439490 and liver necroinflammation or fibrosis stage may also guide IFN-based antiviral treatment of CHC patients. However, given the current availability of direct acting antiviral agents, the relationship between CXCL10 G-201A G/G and IFN-free anti-HCV regimens requires further study.
ARTICLE HIGHLIGHTS
Research background
In the past two decades, some patients with chronic hepatitis C virus (HCV) infection (CHC) have been shown to be seronegative for anti-HCV antibodies and RNA, but have intrahepatic HCV RNA in liver biopsy. However, the etiology of this occult HCV infection (OCI) remains unclear.
Research motivation
Seronegative OCI patients were reported to have significantly lower serum CXCL10 levels than patients with CHC. Polymorphisms in the CXCL10 promoter have been implicated in the genetic variation underlying the susceptibility to chronic HBV infection (CHB) progression in Chinese populations. Moreover, CHC and CHB induce similar liver lesions and fibrosis through continuous infiltration of inflammatory cells, but do not damage hepatocytes directly. These phenomena promoted our interest to examine whether CXCL10 G-201A underlies the disease manifestation of OCI.
Research objectives
To investigate the allele frequency of CXCL10 single nucleotide polymorphisms (SNPs) in patients with OCI and whether they are associated with the low levels of CXCL10 in OCI patients.
Research methods
We characterized the expression frequency of CXCL10 G-201A (rs1439490), C-1513T (rs1440802), and IL-28B rs12979860 in seronegative OCI and seropositive CHC patients in Northeastern China. Serum CXCL10 levels were measured by ELISA. Intrahepatic CXCL10 levels were determined by quantitative PCR and immunohistochemical semi-quantitative scoring. Liver necroinflammation and fibrosis were scored according to the METAVIR system. The associations of CXCL10 rs1439490 with CXCL10 levels and antiviral efficacy in OCI were analyzed.
Research results
CXCL10 G-201A G/G was more prevalent in seronegative OCI patients than in seropositive CHC patients. Serum CXCL10 levels were lower in OCI patients than in CHC patients, but did not differ significantly between IL-28B rs12979860 C/C and non-C/C patients. Of IL-28B rs12979860 C/C patients, OCI patients with CXCL10 G-201A G/G had lower serum and liver levels of CXCL10, and lower levels of liver necroinflammation and fibrosis than non-G/G patients. OCI patients had high ALT normalization rates and serum CXCL10 decreased significantly after Peg-IFNα plus ribavirin treatment, most potently in G-201A G/G patients. Liver necroinflammation and fibrosis were alleviated in 8 OCI patients after treatment. Multivariate analysis indicated that CXCL10 G-201A G/G significantly influenced the occurrence of OCI in HCV infection.
Research conclusions
Our study revealed a higher prevalence of CXCL10 rs1439490 G/G genotype in OCI patients than in CHC patients. OCI patients with rs1439490 G/G genotype achieved better antiviral efficacy with Peg-IFN plus RBV. CXCL10 G-201A genotype is associated with the occurrence of seronegative OCI in patients with CHC, and may be an independent prognostic factor for IFN-based antiviral treatment.
Research perspectives
More paired liver biopsies before and after antiviral treatment are anticipated to examine the correlation of CXCL10 change with clinical outcomes of OCI. In addition, given the current availability of direct acting antiviral agents, the relationship between CXCL10 G-201A G/G and IFN-free anti-HCV regimens requires further study.
Footnotes
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
Supported by the National Natural Science Foundation of China, No. 81670533; the Jilin Provincial Science & Technology Department, No. 2013 0102088JC; and the Jilin Provincial Development and Reform Commission, No. 2013C028-3.
Institutional review board statement: This study was approved by the Institutional Review Boards of individual centers. All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Clinical trial registration statement: Chinese Clinical Trial Registry (Registration number: ChiCTR-ONRC-12002207). The registration information can be found on the following website: http://www.chictr.org.cn/showproj.aspx?proj=7343
Informed consent statement: Written informed consent was obtained from all individual participants included in the study.
Conflict-of-interest statement: The authors declare that they have no conflict of interest.
Data sharing statement: No additional data are available.
CONSORT 2010 statement: The authors have read the CONSORT 2010 Statement, and the manuscript was prepared and revised according to the CONSORT 2010 Statement.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Peer-review started: February 11, 2018
First decision: March 9, 2018
Article in press: May 11, 2018
P- Reviewer: Bourgoin SG, Citores MJ, Cetinkunar S, Kaabachi W, Stefanovic N S- Editor: Wang XJ L- Editor: Webster JR E- Editor: Tan WW
Contributor Information
Xu Wang, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Song Wang, Department of Urology, First Hospital Affiliated to Jilin University, Changchun 130000, Jilin Province, China.
Zhen-Hua Liu, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Wen-Qian Qi, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Qian Zhang, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Yong-Gui Zhang, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
De-Rong Sun, Department of Infectious Disease, the Fourth Affiliated University of Harbin Medical University, Harbin 150001, Heilongjiang Province, China; Department of Digestive, the Second People’s Hospital of Daqing City, Daqing 163461, Heilongjiang Province, China.
Yan Xu, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Hong-Guang Wang, Department of Digestive, People’s Hospital of Jilin City, Changchun 132000, Jilin Province, China.
Zhong-Xie Li, Department of Digestive, People’s Hospital of Hunchun City, Hunchun 133300, Jilin Province, China.
Xian-Ling Cong, Department of Pathology, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Ping Zhao, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Chang-Yu Zhou, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China.
Jiang-Bin Wang, Department of Digestive, China-Japan Union Hospital Affiliated to Jilin University, Changchun 130033, Jilin Province, China. zrlwangjb@163.com.
References
- 1.Castillo I, Pardo M, Bartolomé J, Ortiz-Movilla N, Rodríguez-Iñigo E, de Lucas S, Salas C, Jiménez-Heffernan JA, Pérez-Mota A, Graus J, et al. Occult hepatitis C virus infection in patients in whom the etiology of persistently abnormal results of liver-function tests is unknown. J Infect Dis. 2004;189:7–14. doi: 10.1086/380202. [DOI] [PubMed] [Google Scholar]
- 2.Carreño V, Bartolomé J, Castillo I, Quiroga JA. New perspectives in occult hepatitis C virus infection. World J Gastroenterol. 2012;18:2887–2894. doi: 10.3748/wjg.v18.i23.2887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berasain C, Betés M, Panizo A, Ruiz J, Herrero JI, Civeira MP, Prieto J. Pathological and virological findings in patients with persistent hypertransaminasaemia of unknown aetiology. Gut. 2000;47:429–435. doi: 10.1136/gut.47.3.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Attar BM, Van Thiel D. A New Twist to a Chronic HCV Infection: Occult Hepatitis C. Gastroenterol Res Pract. 2015;2015:579147. doi: 10.1155/2015/579147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Naghdi R, Ranjbar M, Bokharaei-Salim F, Keyvani H, Savaj S, Ossareh S, Shirali A, Mohammad-Alizadeh AH. Occult Hepatitis C Infection Among Hemodialysis Patients: A Prevalence Study. Ann Hepatol. 2017;16:510–513. doi: 10.5604/01.3001.0010.0277. [DOI] [PubMed] [Google Scholar]
- 6.Abdelmoemen G, Khodeir SA, Abou-Saif S, Kobtan A, Abd-Elsalam S. Prevalence of occult hepatitis C virus among hemodialysis patients in Tanta university hospitals: a single-center study. Environ Sci Pollut Res Int. 2018;25:5459–5464. doi: 10.1007/s11356-017-0897-y. [DOI] [PubMed] [Google Scholar]
- 7.Barril G, Castillo I, Arenas MD, Espinosa M, Garcia-Valdecasas J, Garcia-Fernández N, González-Parra E, Alcazar JM, Sánchez C, Diez-Baylón JC, et al. Occult hepatitis C virus infection among hemodialysis patients. J Am Soc Nephrol. 2008;19:2288–2292. doi: 10.1681/ASN.2008030293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bokharaei-Salim F, Keyvani H, Monavari SH, Alavian SM, Madjd Z, Toosi MN, Mohammad Alizadeh AH. Occult hepatitis C virus infection in Iranian patients with cryptogenic liver disease. J Med Virol. 2011;83:989–995. doi: 10.1002/jmv.22044. [DOI] [PubMed] [Google Scholar]
- 9.Castillo I, Bartolomé J, Quiroga JA, Barril G, Carreño V. Hepatitis C virus infection in the family setting of patients with occult hepatitis C. J Med Virol. 2009;81:1198–1203. doi: 10.1002/jmv.21483. [DOI] [PubMed] [Google Scholar]
- 10.Comar M, Dal Molin G, D'Agaro P, Crocè SL, Tiribelli C, Campello C. HBV, HCV, and TTV detection by in situ polymerase chain reaction could reveal occult infection in hepatocellular carcinoma: comparison with blood markers. J Clin Pathol. 2006;59:526–529. doi: 10.1136/jcp.2005.033050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.De Marco L, Gillio-Tos A, Fiano V, Ronco G, Krogh V, Palli D, Panico S, Tumino R, Vineis P, Merletti F, et al. Occult HCV infection: an unexpected finding in a population unselected for hepatic disease. PLoS One. 2009;4:e8128. doi: 10.1371/journal.pone.0008128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cortés-Mancera FM, Restrepo JC, Osorio G, Hoyos S, G C. Occult hepatitis C virus infection in a re-transplanted patients with liver failure of unknown etiology. Rev Col Gastroenterol. 2010;25:72–80. [Google Scholar]
- 13.Bartolomé J, López-Alcorocho JM, Castillo I, Rodríguez-Iñigo E, Quiroga JA, Palacios R, Carreño V. Ultracentrifugation of serum samples allows detection of hepatitis C virus RNA in patients with occult hepatitis C. J Virol. 2007;81:7710–7715. doi: 10.1128/JVI.02750-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pham TN, King D, Macparland SA, McGrath JS, Reddy SB, Bursey FR, Michalak TI. Hepatitis C virus replicates in the same immune cell subsets in chronic hepatitis C and occult infection. Gastroenterology. 2008;134:812–822. doi: 10.1053/j.gastro.2007.12.011. [DOI] [PubMed] [Google Scholar]
- 15.Quiroga JA, Castillo I, Pardo M, Rodríguez-Iñigo E, Carreño V. Combined hepatitis C virus (HCV) antigen-antibody detection assay does not improve diagnosis for seronegative individuals with occult HCV infection. J Clin Microbiol. 2006;44:4559–4560. doi: 10.1128/JCM.01440-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Veerapu NS, Raghuraman S, Liang TJ, Heller T, Rehermann B. Sporadic reappearance of minute amounts of hepatitis C virus RNA after successful therapy stimulates cellular immune responses. Gastroenterology. 2011;140:676–685.e1. doi: 10.1053/j.gastro.2010.10.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rezaee-Zavareh MS, Hadi R, Karimi-Sari H, Hossein Khosravi M, Ajudani R, Dolatimehr F, Ramezani-Binabaj M, Miri SM, Alavian SM. Occult HCV Infection: The Current State of Knowledge. Iran Red Crescent Med J. 2015;17:e34181. doi: 10.5812/ircmj.34181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Quiroga JA, Llorente S, Castillo I, Rodríguez-Iñigo E, Pardo M, Carreño V. Cellular immune responses associated with occult hepatitis C virus infection of the liver. J Virol. 2006;80:10972–10979. doi: 10.1128/JVI.00852-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bartolomé J, Castillo I, Quiroga JA, Carreño V. Interleukin-28B polymorphisms and interferon gamma inducible protein-10 serum levels in seronegative occult hepatitis C virus infection. J Med Virol. 2016;88:268–274. doi: 10.1002/jmv.24322. [DOI] [PubMed] [Google Scholar]
- 20.Deng G, Zhou G, Zhang R, Zhai Y, Zhao W, Yan Z, Deng C, Yuan X, Xu B, Dong X, et al. Regulatory polymorphisms in the promoter of CXCL10 gene and disease progression in male hepatitis B virus carriers. Gastroenterology. 2008;134:716–726. doi: 10.1053/j.gastro.2007.12.044. [DOI] [PubMed] [Google Scholar]
- 21.European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of hepatitis C virus infection. J Hepatol. 2011;55:245–264. doi: 10.1016/j.jhep.2011.02.023. [DOI] [PubMed] [Google Scholar]
- 22.Ge D, Fellay J, Thompson AJ, Simon JS, Shianna KV, Urban TJ, Heinzen EL, Qiu P, Bertelsen AH, Muir AJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature. 2009;461:399–401. doi: 10.1038/nature08309. [DOI] [PubMed] [Google Scholar]
- 23.Bedossa P, Poynard T. An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology. 1996;24:289–293. doi: 10.1002/hep.510240201. [DOI] [PubMed] [Google Scholar]
- 24.Bresalier RS, Ho SB, Schoeppner HL, Kim YS, Sleisenger MH, Brodt P, Byrd JC. Enhanced sialylation of mucin-associated carbohydrate structures in human colon cancer metastasis. Gastroenterology. 1996;110:1354–1367. doi: 10.1053/gast.1996.v110.pm8613039. [DOI] [PubMed] [Google Scholar]
- 25.Furusyo N, Kajiwara E, Takahashi K, Nomura H, Tanabe Y, Masumoto A, Maruyama T, Nakamuta M, Enjoji M, Azuma K, Shimono J, Sakai H, Shimoda S, Hayashi J; Kyushu University Liver Disease Study (KULDS) Group. Association between the treatment length and cumulative dose of pegylated interferon alpha-2b plus ribavirin and their effectiveness as a combination treatment for Japanese chronic hepatitis C patients: project of the Kyushu University Liver Disease Study Group. J Gastroenterol Hepatol. 2008;23:1094–1104. doi: 10.1111/j.1440-1746.2008.05319.x. [DOI] [PubMed] [Google Scholar]
- 26.Brownell J, Bruckner J, Wagoner J, Thomas E, Loo YM, Gale M Jr, Liang TJ, Polyak SJ. Direct, interferon-independent activation of the CXCL10 promoter by NF-κB and interferon regulatory factor 3 during hepatitis C virus infection. J Virol. 2014;88:1582–1590. doi: 10.1128/JVI.02007-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Brownell J, Wagoner J, Lovelace ES, Thirstrup D, Mohar I, Smith W, Giugliano S, Li K, Crispe IN, Rosen HR, et al. Independent, parallel pathways to CXCL10 induction in HCV-infected hepatocytes. J Hepatol. 2013;59:701–708. doi: 10.1016/j.jhep.2013.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brownell J, Polyak SJ. Molecular pathways: hepatitis C virus, CXCL10, and the inflammatory road to liver cancer. Clin Cancer Res. 2013;19:1347–1352. doi: 10.1158/1078-0432.CCR-12-0928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Heim MH. Innate immunity and HCV. J Hepatol. 2013;58:564–574. doi: 10.1016/j.jhep.2012.10.005. [DOI] [PubMed] [Google Scholar]
- 30.Lin JC, Habersetzer F, Rodriguez-Torres M, Afdhal N, Lawitz EJ, Paulson MS, Zhu Y, Subramanian GM, McHutchison JG, Sulkowski M, et al. Interferon γ-induced protein 10 kinetics in treatment-naive versus treatment-experienced patients receiving interferon-free therapy for hepatitis C virus infection: implications for the innate immune response. J Infect Dis. 2014;210:1881–1885. doi: 10.1093/infdis/jiu325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zeremski M, Dimova RB, Benjamin S, Penney MS, Botfield MC, Talal AH. Intrahepatic and Peripheral CXCL10 Expression in Hepatitis C Virus-Infected Patients Treated With Telaprevir, Pegylated Interferon, and Ribavirin. J Infect Dis. 2015;211:1795–1799. doi: 10.1093/infdis/jiu807. [DOI] [PMC free article] [PubMed] [Google Scholar]