Abstract
We have previously reported that Ildr2 knockdown via adenovirally-delivered shRNA causes hepatic steatosis in mice. In the present study we investigated hepatic biochemical and anatomic phenotypes of Cre-mediated Ildr2 knock-out mice. Liver-specific Ildr2 knock-out mice were generated in C57BL/6J mice segregating for a floxed (exon 1) allele of Ildr2, using congenital and acute (10-13-week-old male mice) Cre expression. In addition, Ildr2 shRNA was administered to Ildr2 knock-out mice to test the effects of Ildr2 shRNA, per se, in the absence of Ildr2 expression. RNA sequencing was performed on livers of these knockdown and knockout mice. Congenital and acute liver-specific and hepatocyte-specific knockout mice did not develop hepatic steatosis. However, administration of Ildr2 shRNA to Ildr2 knock-out mice did cause hepatic steatosis, indicating that the Ildr2 shRNA had apparent “off-target” effects on gene(s) other than Ildr2. RNA sequencing and BLAST sequence alignment revealed Dgka as a candidate gene mediating these “off-target” effects. Ildr2 shRNA is 63% homologous to the Dgka gene, and Dgka expression decreased only in mice displaying hepatic steatosis. Dgka encodes diacylglycerol kinase (DGK) alpha, one of a family of DGKs which convert diacylglycerides to phosphatidic acid for second messenger signaling. Dgka knockdown mice would be expected to accumulate diacylglyceride, contributing to the observed hepatic steatosis. We conclude that ILDR2 plays a negligible role in hepatic steatosis. Rather, hepatic steatosis observed previously in Ildr2 knockdown mice was likely due to shRNA targeting of Dgka and/or other “off-target” genes. We propose that the gene candidates identified in this follow-up study may lead to identification of novel regulators of hepatic lipid metabolism.
Introduction
Non-alcoholic fatty liver disease (NAFLD) is rapidly becoming the leading cause of liver failure and transplantation in the United States and is predicted to affect ~30% of adults in the US [1]. Often considered the major liver manifestation of the metabolic syndrome, NAFLD is closely associated with obesity, diabetes and insulin resistance [1, 2]. While the simple steatosis that defines NAFLD is relatively benign, it can progress to non-alcoholic steatohepatitis (known as NASH) with inflammatory infiltration and fibrosis [3]. The physiologic and metabolic factors that cause NAFLD and trigger its progression to NASH remain poorly understood.
Recently, we described immunoglobulin-like domain containing receptor 2 (ILDR2) as a novel modulator of NAFLD development [4]. Initially identified by positional genetics as a diabetes-susceptibility gene in mice [5], Ildr2 knockdown via adenovirally-delivered shRNA (ADKD) resulted in gross hepatic steatosis and inflammation within 10 days of infection [4]. Transcript analyses indicated initial increase in expression of genes mediating lipogenesis (3 days post-adenovirus infection), followed by decrease in expression of these transcripts after development of steatosis, and differential expression of genes involved in the unfolded protein response (ER stress) pathways [4].
In this previous study, we used an adenoviral delivery system to target hepatic short hairpin RNA (shRNA) in order to produce an acute liver-specific knockdown of Ildr2 [4]. In the absence (at the time) of any congenital Ildr2 KO mouse models, the Adv-shRNA system allowed us to investigate the effects of acute knockdown of Ildr2 transcripts in the liver. However this system has effects beyond the knockdown of the target gene that confound interpretation: Adv infection is known to trigger hepatic inflammation [6–8] which plays a role in the progression of NAFLD and development of NASH [9, 10]; Adv can also target other tissues, and even though the majority is taken up by the liver [11–13], there are potential consequences for gene expression in those tissues; and, finally, shRNA itself can have “off-target” effects and reduce expression of genes not intentionally targeted [14, 15].
Here we describe liver-specific Ildr2 gene deletion models achieved using the Cre-loxP system. We discuss the development of liver-specific Ildr2 knockout (KO) mice and further characterize them to understand the putative role of Ildr2 in hepatic steatosis. The differing phenotypes observed in Ildr2 Adv-shRNA KD vs. KO models highlight some of the pitfalls of using adenoviruses and shRNA for genetic manipulations; these are discussed below.
Results
Congenital, hepatocyte-specific Ildr2 KO mice do not develop hepatic steatosis
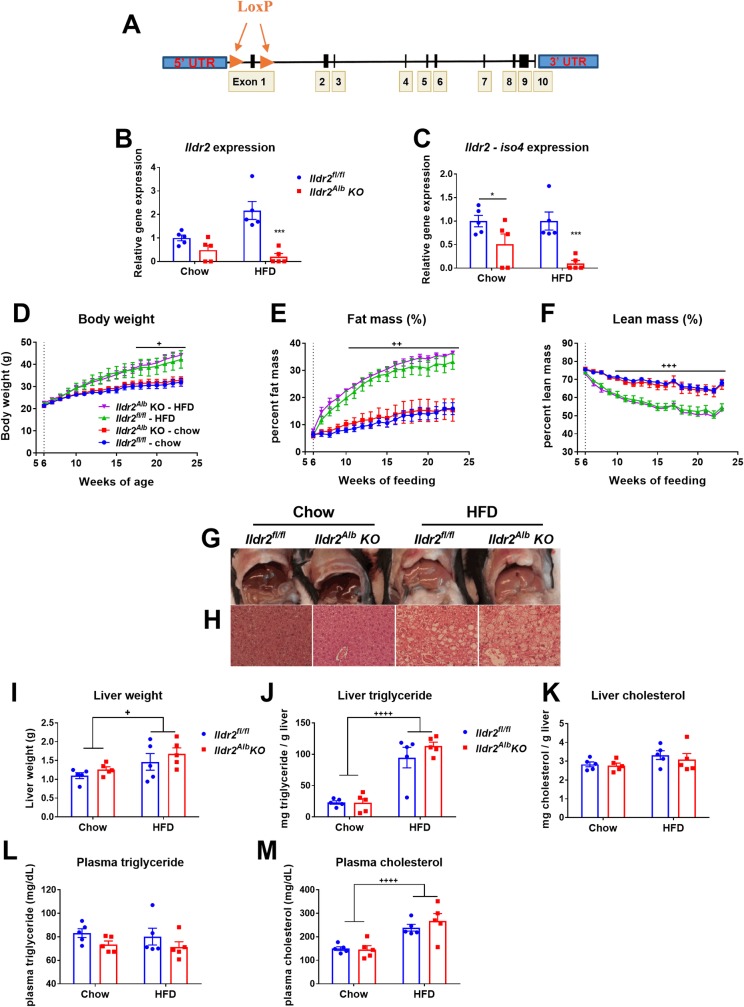
We introduced loxP sites flanking exon 1 of the Ildr2 gene (exon 1 is included in all seven known Ildr2 transcript isoforms [5]) to create an Ildr2 floxed mouse (Ildr2fl/fl) (Fig 1A). To explore the function of ILDR2 in the liver, we crossed Ildr2fl/fl mice with mice expressing Cre recombinase driven by the albumin promoter, obtaining hepatocyte-specific, congenital Ildr2 knockout mice (see Table 1 for nomenclature). Ildr2 liver mRNA expression was reduced >99% in hepatocyte-specific Ildr2 KO mice (Ildr2Alb KO) compared to Ildr2fl/fl littermate controls (Fig 1B and 1C). Although a subset of these mice retained Ildr2 expression–indicating that the albumin-cre was not completely penetrant–these mice displayed no phenotypic differences vs. complete Ildr2Alb KO mice.
Fig 1. Albumin-cre, Ildr2 KO mice do not develop hepatic steatosis.
(A) Schematic of the floxed Ildr2 allele (not to scale). (B,C) Expression of Ildr2, isoforms 1 and 4 in livers of 23-week old Ildr2Alb KO mice and littermate Ildr2fl/fl controls, fed chow or HFD for 17 weeks. Expression was measured by qPCR and normalized to 36b4, actb and Gapdh expression. (D) Body weight curves of HFD and chow-fed, Ildr2Alb KO mice. (E,F) Percent fat mass and lean mass of HFD and chow-fed, Ildr2Alb KO mice measured weekly by NMR. (G) Photographs of livers excised from HFD and chow-fed, Ildr2Alb KO mice at 23 weeks of age. (H) Hematoxylin and eosin staining of representative liver sections at 50X magnification. (I) Liver weight at 23 weeks of age. (J,K) Liver triglyceride and total cholesterol content (measured in duplicate). (L,M) Plasma triglyceride and total cholesterol concentration at 23 weeks of age after a 4hr. fast. n = 4–5 mice per group. Data are represented as mean ± standard error (SEM) * p<0.05, ** p<0.01, *** p<0.001 for Ildr2Alb KO vs. Ildr2fl/fl control. + p<0.05, ++ p<0.01, +++ p<0.001 for chow vs. HFD.
Table 1. Mouse models—nomenclature and abbreviations.
| Mouse model | Abbreviation in text | Cell type(s) targeted | Developmental Timing | Control used | Phenotype without Ildr2 shRNA | Phenotype with Ildr2 shRNA |
|---|---|---|---|---|---|---|
| Adenoviral Ildr2 shRNA | ADKD | All liver cells | Adult; acute | Adenoviral lacZ shRNA | Hepatic steatosis (extreme) and inflammation | |
| Ildr2fl/fl; albumin-cre | Ildr2Alb KO | Hepatocytes | E14.5 (upon albumin expression) | Ildr2fl/fl | No difference from control | Hepatic steatosis (mild) and inflammation |
| Ildr2fl/fl; adeno-associated virus-thyroxine-binding globulin (TBG)-cre | Ildr2AAV KO | Hepatocytes | Adult; acute | Ildr2fl/fl; AAV-TBG-GFP | No difference from control | |
| Ildr2fl/fl; adenoviral-cre | Ildr2Adv KO | All liver cells | Adult; acute | Ildr2fl/fl; Adv-GFP | No difference from control |
When fed, ad libitum, low-fat (9% kcal as fat) chow diet, male, Ildr2Alb KO mice did not differ in body weight or body composition from Ildr2fl/fl littermates (Fig 1D–1F). When fed ad libitum a high-fat diet (HFD, 60% kcal as fat) from 6–23 weeks of age, they increased body weight and fat mass in tandem with their Ildr2fl/fl HFD-fed littermates (Fig 1D–1F).
23-week-old, chow-fed Ildr2Alb KO mice did not exhibit hepatic steatosis by inspection, histology, or quantitative chemical analysis (Fig 1G–1K). They also had normal plasma triglyceride and total cholesterol concentrations (Fig 1L and 1M). 23-week-old, HFD-fed mice showed hepatic lipid accumulation and elevated plasma lipids, but there was no significant difference between Ildr2Alb KO mice and littermate controls fed the same HFD (Fig 1G–1M).
Acute, hepatocyte-specific Ildr2 KO mice do not develop hepatic steatosis
The absence of steatosis in Ildr2Alb KO mice led us to postulate that the congenital nature of the KO may have triggered gene compensation for the lack of ILDR2 during development. The mouse albumin gene is turned on at ~E10.5, about halfway through embryonic development [16], and Cre expression has been detected in fetal mouse hepatocytes from albumin-cre mice as early as E14.5, as immature cells begin to differentiate into hepatocytes [17]. Critical genes deleted at this stage in development may be compensated by functionally similar genes [18–20]. Compensation for the loss of Ildr2 in Ildr2Alb KO mice could theoretically explain the absence of increased steatosis in the Ildr2Alb KO mice.
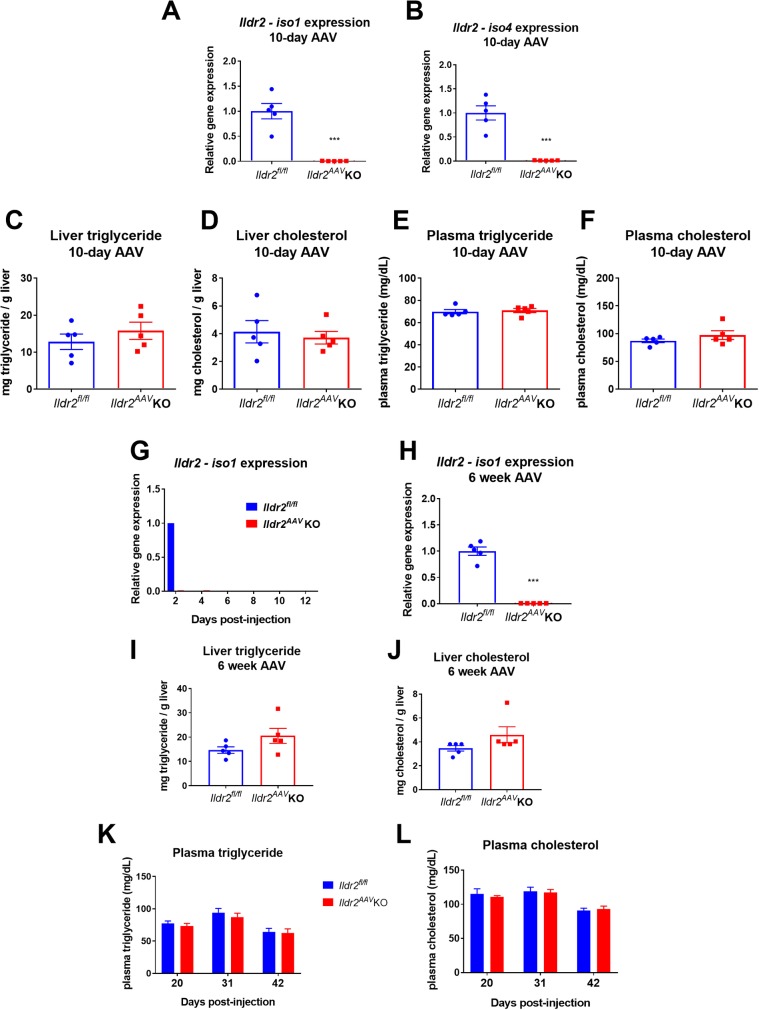
To address this possibility, we designed a mouse model in which Ildr2 could be acutely ablated in the adult animals, similar to the original Adv-shRNA KD mice (ADKD) mice. We utilized an adeno-associated virus (AAV) construct incorporating thyroxine-binding globulin (TBG) promoter-driven Cre or GFP (control). This AAV8-TBG-Cre (developed by the Penn Vector Core) enables acute Cre expression specifically in hepatocytes [21, 22], knocking out Ildr2 (Ildr2AAV KO). We injected AAV8-TBG-Cre intravenously into 13-week-old, chow-fed Ildr2fl/fl mice and examined livers 10 days post-injection, in keeping with the timeline of development of steatosis in ADKD mice [4]. Despite complete KO of Ildr2 (Fig 2A and 2B), livers of Ildr2AAV KO mice were normal, showing neither steatosis nor any lipid metabolic abnormalities when compared to mice injected with the AAV8-TBG-GFP control construct (Fig 2C–2F).
Fig 2. AAV Ildr2 KO mice do not develop hepatic steatosis.
(A,B) qPCR expression of Ildr2, isoforms 1 and 4 in livers of 13-week old mice, 10 days after i.v. injection with AAV-TBG-Cre (Ildr2AAV KO) or AAV-TBG-GFP (Ildr2fl/fl controls). (C,D) Liver triglyceride and total cholesterol content at 10 days. (E,F) Plasma triglyceride and total cholesterol concentration at 10 days. (G) qPCR expression of Ildr2 (isoform 1 unless otherwise noted) in livers of 13-week old mice 2–12 days after i.v. injection with AAV-TBG-Cre (Ildr2AAV KO). AAV-TBG-GFP was only administered for the 2-day timepoint (Ildr2fl/fl controls). (H) qPCR expression of Ildr2 in livers of 18-week old mice, 6 weeks after AAV injection (I,J) Liver triglyceride and total cholesterol measurements. (K,L) Plasma triglyceride and total cholesterol concentration at 20, 31 and 42 days after AAV injection. Blood was collected after a 4hr fast. n = 4–5 mice per group. * p<0.05, ** p<0.01, *** p<0.001 for Ildr2AAV KO vs. Ildr2fl/fl control.
To determine the timing of AAV delivery and gene interruption, we measured hepatic mRNA expression of Ildr2 isoforms 1 through 5 (only isoform 1 is shown) in mice at 2, 4, 6, 8, 10 and 12 days post injection and found that Ildr2 transcription was eliminated as early as 2 days post-injection (Fig 2G). We also followed mice for 6 weeks after AAV infection, measuring plasma lipids at 20 days post-injection, and then at 11-day intervals until sacrifice. Ildr2AAV KO mice maintained normal plasma lipid levels and did not exhibit any hepatic lipid accumulation or metabolic abnormalities at 6 weeks post-injection with AAV (Fig 2H–2L).
Acute, Adv-mediated, liver-specific Ildr2 KO mice do not develop hepatic steatosis
Next, we considered the possibility that loss of Ildr2 in non-parenchymal liver cells may have contributed significantly to the steatosis observed in our original ADKD mice [4]. Both the AAV-TBG-Cre and the albumin-cre were designed to induce recombination and gene knockout specifically in hepatocytes which comprise ~80% of liver tissue. However, the shRNA adenovirus used to produce ADKD mice would have targeted additional liver cell types, such as liver macrophages (Kupffer cells), stellate cells, and epithelial cells. While hepatic steatosis is defined by lipid accumulation in hepatocytes, effects in non-parenchymal liver cells can accelerate the progression of steatosis to more advanced liver disease [23–26]. As resident liver macrophages, Kupffer cells initiate the immune response to metabolic injury, secreting pro-inflammatory chemokines and cytokines such as IL-1β and TNF, stimulating pro-apoptotic signaling pathways in hepatocytes, and recruiting circulating immune cells to the liver [26–28]. Stellate cells play a key role in the induction of fibrosis in liver disease, and can transdifferentiate into myofibroblasts leading to increased production of collagen and extra-cellular matrix (ECM) [25, 28, 29].
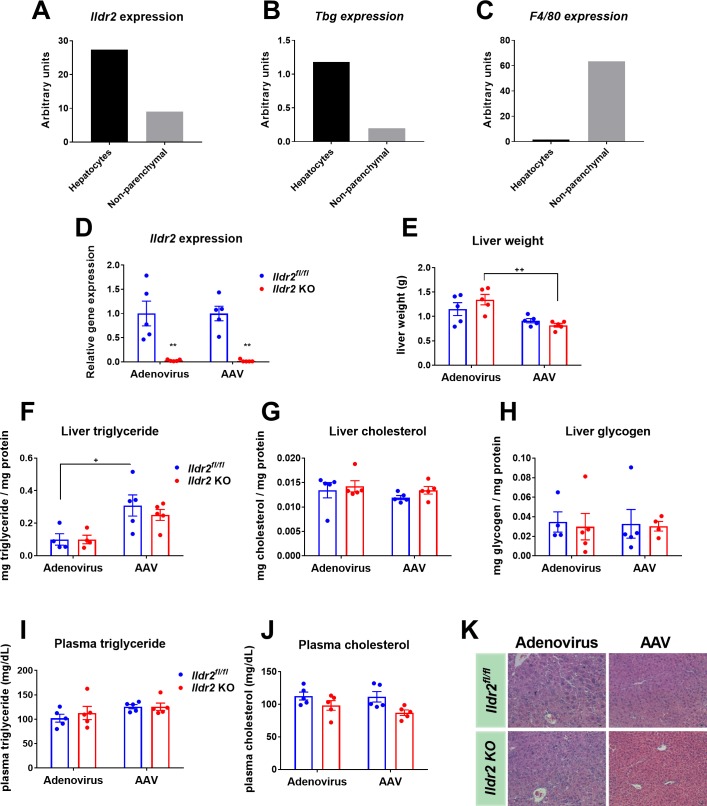
To determine if Ildr2 is expressed in non-parenchymal liver cells, or in hepatocytes only, primary hepatocytes and non-parenchymal cells were isolated from 12-week-old mice using liver collagenase digestion [4]. Hepatocyte and non-hepatocyte cell fractions were separated by centrifugation [30]. Gene expression analysis of liver cell markers was used to confirm the cellular identity of each fraction. Tbg, a hepatocyte-specific marker, and F4/80, a macrophage-specific marker, were highly expressed in the hepatocyte and non-hepatocyte fractions, respectively (Fig 3B and 3C). Ildr2 was expressed in both cell fractions, although at about one-third the level in the non-hepatocyte cell fraction as in the hepatocyte cell fraction (Fig 3A). However, since these cell fractions were sorted by centrifugation there could be cross-contamination as indicated by low-level Tbg expression in the non-hepatocyte fraction, and F4/80 expression in hepatocyte fraction (Fig 3B and 3C). Microarray expression data from Xu, et al. also confirm that Ildr2 is expressed in various populations of macrophages derived from adipose tissue [31]. Taken together, these results suggest that Ildr2 ablation in non-parenchymal liver cells could contribute to the steatotic phenotypes of the ADKD mice, and thus explain the lack of hepatic steatosis in the acute and congenital transgenic hepatocyte-specific KO mice.
Fig 3. Adenoviral Ildr2 KO mice do not develop hepatic steatosis.
(A-C) qPCR expression of Ildr2, Tbg and F4/80 in hepatocyte or non-parenchymal cell fractions isolated from 12-week old wild-type (B6) mice. (D) qPCR expression of Ildr2 in livers of 11-week old mice, 10 days after i.v. injection with adenoviral-Cre (Ildr2Adv KO), adenoviral-GFP (Ildr2fl/fl controls), AAV-TBG-Cre (Ildr2AAV KO) or AAV-TBG-GFP (Ildr2fl/fl controls). (E) Liver weights at sac. (F-H) Liver triglyceride, total cholesterol and glycogen content. (I,J) Plasma triglyceride and total cholesterol concentration at sac following a 12hr fast. (K) Hematoxylin and eosin staining of representative liver sections at 20X magnification. n = 4–5 mice per group. * p<0.05, ** p<0.01, *** p<0.001 for Ildr2 KO vs. Ildr2fl/fl control. + p<0.05, ++ p<0.01, +++ p<0.001 for adenovirus vs. AAV.
To test this possibility, we created another acute Ildr2 KO model by using an adenoviral-Cre construct rather than the AAV-TBG-Cre used previously. While the AAV-TBG-Cre construct is designed to impact only hepatocytes, adenoviral-Cre targets both parenchymal and non-parenchymal liver cells [32, 33]. 11-week-old, male, Ildr2fl/fl mice were injected intravenously with adenovirus-Cre or adenovirus-GFP as a control. Age-matched Ildr2fl/fl mice were infected with AAV-TBG-Cre or AAV-TBG-GFP at the same time for parallel comparison. Ildr2Adv KO mice were euthanized 10 days post-injection. No liver steatosis or dyslipidemia were seen in Ildr2Adv or Ildr2AAV KO mice despite complete Ildr2 ablation in liver (Fig 3D and 3F–3J). Ildr2Adv KO livers were heavier compared to Ildr2AAV KO mice (Fig 3E), and also showed histological evidence of inflammation (Fig 3K). However, as these phenotypes were also present in the Ildr2Adv GFP control mice, we attributed them to the effects of adenovirus treatment as has been documented previously [6–8].
Administration of adenoviral Ildr2 shRNA causes TG accumulation in Ildr2Alb KO mice
We have produced three distinct models of hepatic Ildr2 KO: a congenital, hepatocyte-specific KO (Ildr2Alb KO); an acute, hepatocyte-specific, KO (Ildr2AAV KO); and an acute, liver-specific KO (Ildr2Adv KO). None of these models showed the severe steatohepatitis observed in the adenoviral Ildr2 shRNA (ADKD) model [4]. Thus, we were driven to consider that the steatosis had been caused by a consequence of the shRNA antisense construct–primarily unrelated to the decrease in Ildr2 expression.
The original ADKD model was produced by treating mice with an adenovirally-delivered shRNA. Thus, the adenovirus treatment and/or the shRNA itself may have triggered liver steatosis. Since we showed that adenoviral treatment alone does not cause hepatic steatosis, so we turned our attention to the Ildr2 shRNA. This shRNA was specifically designed to target exon 2 which is present in all isoforms of Ildr2 mRNA; however, the construct may have had “off target” effects on other genes as discussed below [14, 15, 34].
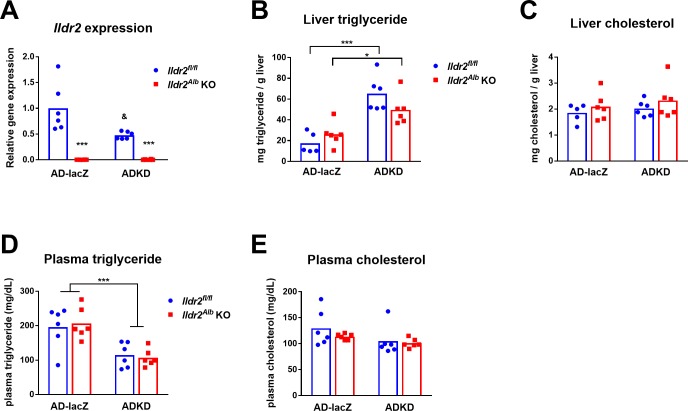
To determine if other targets of the shRNA contributed to the KD liver phenotype, we infected Ildr2Alb KO mice with the original KD adenoviral shRNA [4]. Since these mice do not express Ildr2 in the hepatocytes, any steatosis observed would be the result of shRNA targeting of other genes affecting lipid metabolism. 10-week-old, male, Ildr2Alb KO or Ildr2flfl control mice were injected intravenously with the original adenovirus expressing Ildr2 shRNA (ADKD), or with control adenovirus expressing lacZ shRNA (AD-lacZ) [4]. Mice were euthanized at 10 days post-adenovirus infection following a 24-hour fast. Gene expression analysis by qPCR confirmed that Ildr2 was completely ablated in Ildr2Alb KO mice, regardless of Adv treatment (Fig 4A). In Ildr2flfl mice, Ildr2 shRNA (ADKD) reduced Ildr2 mRNA by about 50% vs. AD-lacZ treated Ildr2flfl mice (Fig 4A). We did not observe gross liver steatosis, but chemical quantification of hepatic lipid content revealed that ADKD-treated mice had significantly increased hepatic TG compared to AD-lacZ treated mice, across both genotypes (3-fold in Ildr2flfl, 1.5-fold in Ildr2Alb KO) (Fig 4B and 4C). Conversely, plasma TG was significantly decreased in Ildr2 shRNA treated Ildr2Alb KO and Ildr2flfl mice vs. AD-lacZ treated mice for both genotypes, although plasma cholesterol was unchanged (Fig 4D and 4E).
Fig 4. Effects of adenoviral Ildr2 shRNA in Ildr2 KO mice.
(A) qPCR expression of Ildr2 in livers of 10-week old mice Ildr2Alb KO mice and littermate Ildr2fl/fl controls, 10 days after i.v. injection with ADKD or AD-lacZ. (B,C) Liver triglyceride and total cholesterol content. (D-E) Plasma triglyceride and total cholesterol concentration at sac following a 24hr fast. n = 5–6 mice per group. * p<0.05, ** p<0.01, *** p<0.001 for Ildr2fl/fl vs. Ildr2Alb KO; & p<0.05 for ADKD vs. AD-lacZ.
These results confirm that the Ildr2 shRNA is sufficient to cause hepatic steatosis despite the preexisting absence of Ildr2. Ildr2 expression was reduced by 50% in Ildr2 shRNA Ildr2flfl mice, indicating that acute partial loss of Ildr2 expression might contribute to the development of steatosis. However, the degree of steatosis and hypotriglyceridemia did not differ between Ildr2 shRNA Ildr2flfl and Ildr2 shRNA Ildr2Alb KO mice, suggesting that Ildr2 expression is either irrelevant to the phenotype or has an equivalent effect at levels below a specific threshold, i.e. below 50%. In either case, the major trigger for hepatic steatosis is the Ildr2 shRNA, not Ildr2 ablation per se.
These results suggest that the Ildr2 shRNA also targets other gene(s) involved in hepatic lipid metabolism, and that KD of these gene(s) is primarily responsible for the gross steatosis in the original Ildr2 shRNA ADKD mice [4] as well as the less striking, but still significantly increased TG accumulation observed here.
RNAseq analysis of Ildr2 shRNA ADKD vs. Ildr2Adv KO livers reveal candidate genes for shRNA off-target effects on hepatic steatosis
The Ildr2 shRNA we used was designed to target exon 2 of the Ildr2 mRNA, which is present in all known Ildr2 isoforms. Analysis of the 19 base pair (bp) shRNA sequence, GTTCAAATCCTACTGCCAG, using NCBI BLAST (Basic Sequence Alignment Search Tool) identified no perfectly matched sequences (other than Ildr2) leaving open the possibility that partially matched sequence(s) could be targeted and cause knockdown of gene(s) essential for hepatic lipid homeostasis [15, 34].
To identify additional gene(s) that might have been knocked down by the Ildr2 shRNA, and thus have contributed to development of steatosis in ADKD mice, we performed RNA sequencing analysis on liver samples from ADKD and AD-lacZ mice (from our previously published ADKD study [4]) and Ildr2Adv KO mice (Fig 3). ADKD and AD-lacZ samples were harvested 3 days post Adv infection to increase the likelihood of detection of primary effects of knocking down the gene rather than secondary gene changes resulting from hepatic steatosis per se.
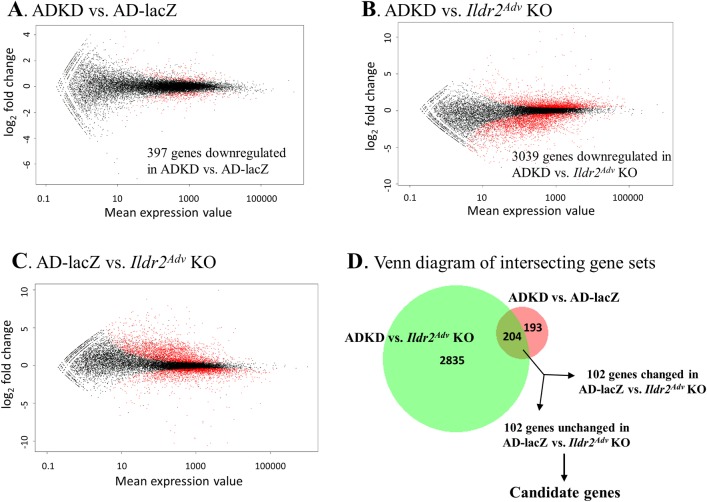
RNAseq count expression data were analyzed with DEseq, a differential expression analysis program based on the negative binomial distribution [35]. Pairwise comparisons were made between Ildr2 shRNA (ADKD) and AD-lacZ; Ildr2 shRNA ADKD and Ildr2Adv KO; and AD-lacZ and Ildr2Adv KO. The Benjamini-Hochberg adjustment [36] was used to correct for the multiple testing problem. Fig 5A–5C are “minus over average” (MA) scatter plots of differential gene expression profiles for each of the 3 comparisons. MA plots display the entire gene set, comparing fold change between samples (y-axis) to mean expression value (x-axis) with significantly differentially expressed genes highlighted in red (Fig 5A–5C). We screened for candidate genes that were: 1) significantly decreased in ADKD vs. AD-lacZ; 2) significantly decreased in ADKD vs. Ildr2Adv KO; and, 3) not significantly changed between AD-lacZ and Ildr2Adv KO livers, indicating a specific effect of the KD Ildr2 shRNA shRNA (Fig 5D). Using these parameters, we identified 102 candidate genes (S1 Table).
Fig 5. RNAseq analysis of AdV-KD vs. Ildr2Adv KO livers identifies candidate genes for shRNA off-target effects.
“Minus over average” (MA) plots showing log2 fold change vs. normalized mean for each comparison. Red dots represent significantly upregulated or downregulated genes in (A) ADKD vs. AD-lacZ mice, (B) ADKD and Ildr2Adv KO mice, (C) AD-lacZ vs. Ildr2Adv KO mice. (D) Venn diagram illustrating how the 102 candidate genes were identified. The intersection of genes downregulated in ADKD mice vs. both AD-lacZ and Ildr2Adv KO was 204. 102 of these genes were not significantly changed in AD-lacZ vs. Ildr2Adv KO. These became the target gene candidates (see S1 Table for complete list). This Venn diagram was created using BioVenn software [37] (www.biovenn.nl).
These candidates were further refined by searching for genes that have been implicated in NAFLD genome-wide association studies (Ppp1ca) [38], genes associated with other liver disease, (Dguok, Ass1) [39, 40], and obesity-related genes (Slc39a1) [41]. Since the shRNA was targeted to Ildr2’s exon 2 which encodes for an IgG domain, we identified genes that are part of the IgG-like family (Neo1, Ptp4a1, Scn8a, Unc13b); additionally, we found a gene located near Ildr2 on chromosome 1 (Pogk) [5].
Initial BLAST searches of the shRNA sequence yielded no complete match apart from Ildr2. However, searching for truncated portions of the 19-bp sequence yielded a partial match in Dgka. Dgka is one of the 102 candidate genes identified by RNAseq analysis (S1 Table), and is 63% homologous to the Ildr2 shRNA sequence. The first 12 bp of the 19 bp shRNA sequence, GTTCAAATCCTA, exactly a sequence in exon 4 of the Dgka mRNA. Dgka expression is downregulated by 50–60% in ADKD livers compared to AD-lacZ and Ildr2Adv KO samples, suggesting that it could be targeted by the Ildr2 shRNA. Dgka encodes diacylglycerol kinase alpha (DGKα), which functions to convert diacylglycerides (DAGs) to phosphatidic acid [42]. DAG species are increased in mouse embryonic fibroblasts from Dgka-null mice [43] similar to increases in DAG species observed in steatotic human liver [44]. These data suggest that loss of Dgka expression could result in the steatosis observed in our ADKD mice.
Discussion
In this study we describe several mouse models developed in an effort to replicate the hepatic steatosis phenotype of adenoviral Ildr2 shRNA KD mice. Using the Cre-loxP system, we created congenital and acute, hepatocyte-only and liver-specific Ildr2 KO mice. However, none of these KO models recapitulated the phenotype of hepatic steatosis observed in the adenoviral Ildr2 shRNA KD mice [4].
RNAi-mediated knockdowns have been effectively used in many experimental settings, and are particularly useful in in vitro studies, and in instances in which a genetic knockout would be prohibitively expensive or difficult to make, or where the knockout is embryonically lethal. KD and KO models are generally quite similar, e.g., Pparα siRNA KD mice phenocopy the null transgenics [45] and Cx43 KO and KD mouse astrocytes have very similar transcriptional profiles [46].
However, discrepancies between RNAi-mediated KD and KO mouse models are not uncommon. As observed in this study and by others, siRNAs and shRNAs can have off-target effects due to sequence similarity to unintended gene targets [14, 15, 34]. RNAi-mediated knockdowns can also exhibit a more severe phenotype than the KO or null mutant due to disruption of the gene at a more mature developmental stage, when functional compensation is difficult [18, 47]. This situation has been documented, for example, for the genes thymosin β4 and Sprn/Prnp in mice, and ABP1 in Arabdopsis thaliana [48–50].
Our studies in which Ildr2 shRNA KD adenovirus was administered to Ildr2Alb KO mice revealed that lipid accumulation occurred with the adenovirus treatment, regardless of Ildr2 genotype of the recipient mouse (Fig 4). These experiments indicate that the hepatic lipid phenotype is due primarily to treatment with the adenovirus shRNA, rather than to loss of Ildr2 expression per se, which suggests that this shRNA targeted genes in addition to Ildr2. We identified Dgka, among other gene candidates, as a potential target of adenoviral Ildr2 shRNA in ADKD mice. Given its homology to the shRNA sequence, its reduced expression in ADKD mice, and its functional role in lipid metabolism [43, 44, 51], we propose that shRNA targeting of Dgka could account for the difference in lipid accumulation between ADKD and KO mice.
Adenovirus is an efficient vector for introduction of gene products into cells both in vitro and in vivo. The adenovirus used in this and our previous study [4] was human adenovirus serotype 5, one of the most commonly used adenoviruses which displays specific liver tropism, and thus is very useful for directing gene products to the liver. However, use of adenoviruses in these contexts can be problematic for several reasons. One overriding concern is that, since they are infectious agents, they can stimulate inflammatory responses in the infected cells [6–8]. This response can mask or confound the effect(s) of whatever biological molecules are being delivered to the cells. Another issue with adenovirus is that its tissue tropism, while specific, is not exclusive, and it can affect tissues other than the target tissue [11]. Additionally, the various methods of measuring adenoviral titer make it difficult to control the amount of active virus that is administered in an experiment, which can lead to significant variation among experiments. The sensitivity of viral activity to temperature changes, i.e. freeze-thaw cycles, also contributes to experimental variability [52, 53].
The experiments described here highlight some of the difficulties in working with adenoviruses. In addition to possible aberrant RNAi gene targeting, the striking phenotype of the original ADKD mice may also have been due to adenovirus-induced inflammation and /or targeting of extra-hepatic tissues. While we confirmed that Ildr2 expression was maintained in other tissues of ADKD mice [4], we cannot rule out that the Adv may have infected other organs. Another concern is that the amount of active Ildr2 shRNA adenovirus used to infect Ildr2Alb KO mice may have decreased from its activity level at the time of ADKD infection. Although the same titer was used in both experiments (3x1011 optical particle units (OPU)/mouse), this titer only measures adenovirus concentration, not viral activity. A reduction in adenoviral activity could also explain the difference in lipid accumulation and severity of steatosis between ADKD mice and Ildr2Alb KO mice infected with Adv-Ildr2 shRNA. Use of appropriate controls enabled us to deconvolute the effects of Ildr2 expression on hepatic steatosis in our various mouse models; however, the confounding effects of using adenovirus as a primary delivery system remain a significant issue.
Conclusions
We have clearly shown that loss of Ildr2—whether specifically in hepatocytes or in all liver cells—is not sufficient to cause hepatic steatosis. These studies indicate that, contrary to the inferences reached based on acute shRNA-mediated KD [4], ILDR2 plays a minimal role in hepatic lipid metabolism. We propose that interruption of other gene(s) significantly contributed to the steatotic phenotype of the original ADKD. RNA-seq identified 102 genes that are significantly reduced in ADKD mice vs. Ildr2Adv KOs or AD-lacZ controls (S1 Table). Among these 102 genes, the most likely candidate is Dgka, which contains a sequence that could be targeted by the Ildr2 shRNA. It is worth noting that hepatic lipid accumulation due to Adv-Ildr2 shRNA treatment was only observed in mice with at least a 50% reduction in Ildr2 expression. Thus, it is possible that KD of candidate gene(s) interacts with Ildr2 hypomorphism to induce steatosis.
Ildr2 was initially identified as modifier of diabetes susceptibility [5], and ongoing work in our lab has confirmed its role in beta cell function, glucose homeostasis, and metabolic partitioning of energy stores. Additionally, ILDR2, along with ILDR1 and ILDR3, are members of the angulin family which maintain membrane integrity at tricellular epithelial tight junctions [54, 55]. Our development of conditional KO mice to clarify the role of ILDR2 in the liver, can now facilitate the study of ILDR2 in various tissues and conditions, enabling a more complete understanding of this novel gene in mammalian biology.
Methods
Ethics statement
All animal experiments were approved by Columbia Institutional Animal Care and Use Committee (Protocol# AAAH0707 and AAAR0416).
Animal care
Mice were housed at room temperature in a 12-hr light/12hr-dark vivarium, with ad libitum access to 5058 Purina PicoLab Mouse Diet 20 (9% kcal from fat) and water. High-fat diet (HFD) fed mice received chow with 60% kcal from fat (Research Diets #D12492i). Where noted, blood was collected by submandibular bleeding. Fat and lean mass were measured with an EchoMRI Analyzer (Bruker Optics), calibrated using mouse carcasses [56]. Mice were euthanized by cervical dislocation for liver tissue collection or prior to primary hepatocyte isolation.
Mouse strains
To generate Ildr2 floxed mice (Ildr2fl/fl), a targeting construct containing Ildr2 exon 1 flanked by loxP sites and a neomycin resistance cassette (transcribed in opposite direction of gene, 1.6 kb from exon 1) was electroporated into V6.5 (129 x B6 F1) ESCs. Electroporated ESCs were selected with G418, picked, and 384 colonies were clonally expanded. Positive integration of the targeting construct was identified by PCR followed by confirmation of correct integration by Southern blot. A positive clone was injected into E3.5 BDF1 blastocytes and transferred into the uteri of pseudopregnant CD1 mice (Crl:CD1; Charles River). Floxed alleles in the pups were confirmed by PCR. Chimeric mice were crossed with C57BL/6J (Jackson Labs) mice and offspring were confirmed carriers by PCR and Southern blot analysis. Mice that carried the floxed allele were backcrossed to C57BL/6J mice (obtained from Jackson Labs) for 10 generations then intercrossed to create B6.129S-Ildr2fl/fl mice. These mice were bred with albumin-Cre mice (B6.Cg-Tg(Alb-cre)21Mgn/J, Jackson Labs stock #003574) until all offspring segregated for two floxed alleles and one or no copies of Cre.
Adenovirus production and administration
Adenovirus expressing Ildr2 shRNA was designed, produced and amplified as previously described [4]. Adenovirus expressing lacZ shRNA was designed and produced as previously described [4], but amplification and purification procedures were performed by Welgen, Inc (Worcester, MA). Mice were administered 3x1011 OPU/mouse via tail vein injection. AAV-TBG-Cre, AAV-TBG-eGFP, adenoviral-Cre, and adenoviral-GFP were obtained from the University of Pennsylvania Vector Core (Philadelphia, PA). Mice were administered 1.3x1011 genome copies/mouse via tail vein injection.
Lipid measurements in tissue and plasma
Capillary blood from submandibular bleeds was collected in heparinized tubes and centrifuged at 200 x g for 20 minutes at 4°C to separate plasma. Lipid extraction from liver was adapted from the Folch method [57]. Approximately 100 mg tissue were homogenized in 3 mL phosphate-buffered saline (PBS). 12 mL 2:1 chloroform: methanol (CHCl3: MeOH) were added and mixture was vortexed twice for 15 seconds each. After centrifuging at 3000 rpm for 10 minutes, the organic lower layer was transferred to a 20-mL glass scintillation vial. An additional 10 mL 2:1 CHCl3: MeOH were added to upper layer and vortexing and centrifugation were repeated. Organic lower layer was added to first extraction in scintillation vial. Solvent was dried down under nitrogen (N2) gas followed by lipid resuspension in 1 mL 15% Triton X-100 in CHCl3. Solvent was dried down again under N2 gas and remaining lipid was resuspended in 1mL H2O. Triglyceride and total cholesterol in plasma and liver extracts were measured with the Infinity Triglycerides (Thermo Scientific) and Cholesterol E (Wako Diagnostics) kits, respectively.
Glycogen measurement
For glycogen extraction 100 mg tissue were homogenized in 1 mL H2O on ice, boiled for 10 minutes, then centrifuged at 13,000 x g for 10 minutes to pellet insoluble material. Supernatant was transferred to a new tube and used for glycogen measurement. Glycogen was measured using a glycogen assay kit from Sigma-Aldrich (#MAK016)
Primary hepatocyte and non-parenchymal cell isolation
Primary hepatocytes isolation was performed as previously described [4] with the exception of euthanasia prior to laparotomy and perfusion. The supernatant from primary hepatocyte centrifugation was collected and spun down at 500 x g, for 10 minutes at 4°C according to a protocol for isolating Kupffer cells by Xu, et al. [30]. The pelleted cells from this centrifugation were considered the non-parenchymal cell fraction.
Hematoxylin and eosin histology
Liver sections were fixed in aqueous zinc-buffered formalin (Anatech, Ltd.), sectioned and visualized by hematoxylin (Fisher) and eosin (Crystalgen) staining. Images were obtained using an Olympus IX73 inverted microscope (Olympus America).
RNA extraction, reverse transcription and quantitative PCR
Tissue and cell samples were homogenized in TRIzol® Reagent (Invitrogen) and extracted using the TRIzol® reagent protocol or the PureLink™ RNA Mini kit (Invitrogen). Reverse transcription was performed using the Transcriptor First Strand cDNA Synthesis kit (Roche). qPCR was performed using a Roche LightCycler® 480 instrument. qPCR primers are listed in Table 2. Tissue-specific standard curves for each gene (primer pair) were used to convert threshold crossing point (Cp) values to relative concentrations, which were then normalized to 36b4, Actb, and/or Gapdh expression. In instances in which standard curves were not used, Cp values are shown, with lower Cp values indicating greater mRNA expression.
Table 2. List of qPCR primers.
| Mouse gene name | Forward primer (5’ to 3’) | Reverse primer (5’ to 3’) |
|---|---|---|
| 36b4 | ACCTCCTTCTTCCAGGCTTTGG | CGAAGGAGAAGGGGGAGATGTT |
| Actb | CGGGCTGTATTCCCCTCCAT | GGGCCTCGTCACCCACATAG |
| Gapdh | CTGGAGAAACCTGCCAAGTATGATG | GAGACAACCTGGTCCTCAGTGTAGC |
| Ildr2 –isoform 1 | GATTATGCCAGAGTGGGTGTTTGTC | CCCTGCTTCATACAAGGCCTGAG |
| Ildr2 –isoform 4 | AACAGGGCTCGACGGTTAC | AACACCCACTCCAACACCAG |
| Tbg | GCAGAAAGGATGGGTTGAATTG | AAGTCAGCACTTTCAGCAAAGG |
| F480 | CTTTGGCTATGGGCTTCCAGTC | GCAAGGAGGACAGAGTTTATCGTG |
RNAseq
RNA was extracted from liver samples as detailed above and sample integrity was assessed with an Agilent 2100 Bioanalyzer with all samples having RIN numbers greater than 8.0. mRNA was isolated using a poly-A pulldown [58] and reverse transcription to generate cDNA. The cDNA was sequenced using single-ended sequencing on a HiSeq2000 according to manufacturer’s recommendations (Illumina; San Diego, CA). The pass filter (PF) reads were mapped to mouse reference genome mm9 using TopHat (version 2.0.4). TopHat infers novel exon-exon junctions ab initio, and combines them with junctions from known mRNA sequences (refgenes) as the reference annotation [59]. For each read, we allowed up to 3 mismatches and 10 multiple hits during the mapping. The software package DESeq [35] in R (Version 3.4.1) was used to conduct the differential gene expression analysis. Multiple comparisons were adjusted by the Benjamini-Hochberg approach, and differentially expressed genes were determined at false discovery rate of 0.01.
Statistical analysis
Statistics were performed using GraphPad Prism 7 software. Data are represented as mean ± standard error (SEM). Statistical comparisons were made using the Student’s two-tailed t-test for 2 groups or two-way analysis of variance (ANOVA) for 4 groups.
Supporting information
(PDF)
Acknowledgments
The authors would like to thank the technical personnel in the Naomi Berrie Diabetes Center histology core for their assistance and members of the Leibel lab for helpful advice and discussion. We are especially grateful to Dr. Domenico Accili, Dr. Henry Ginsberg, Dr. Utpal Pajvani, and Dr. Ira Tabas for providing guidance at multiple stages of this project.
Data Availability
The sequencing data discussed in this paper have been deposited in the National Center for Biotechnology Information (NCBI) Gene Expression Omnibus and are accessible through GEO series accession number GSE114289. (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE114289)
Funding Statement
This work was supported by grants from the United States National Institutes of Health (http://www.nih.gov/): RO1-DK 066518, P30 DK63608-09 (http://derc.cumc.columbia.edu/) and P30 DK26687-31 (http://www.nyorc.org/) to RLL; the National Science Foundation Graduate Research Fellowship Program (http://www.nsfgrfp.org/): DGE 11-44155 and the United Negro College Fund / Merck Graduate Dissertation Fellowship (www.uncf.org) to EJM; the Italian Ministry of Education and University, University of Salerno, Italy (http://www.unisa.it/) and the Russell Berrie Foundation (http://www.russellberriefoundation.org/home.php) to MCDR; the United States National Institutes of Health (http://www.nih.gov/) Pediatric Scientist Development Program to SF; the Japan Society for the Promotion of Science (http://www.jsps.go.jp/english/), the Manpei Suzuki Diabetes Foundation (http://www.suzuki-manpei-df.or.jp/english/) and the Russell Berrie Foundation (http://www.russellberriefoundation.org/home.php) to KW; and the New York Stem Cell Foundation Robertson Prize (https://nyscf.org/) to DE. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Benedict M, Zhang X. Non-alcoholic fatty liver disease: An expanded review. World Journal of Hepatology. 2017;9(16):715–32. doi: 10.4254/wjh.v9.i16.715 PubMed PMID: PMC5468341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease—Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64(1):73–84. doi: 10.1002/hep.28431 [DOI] [PubMed] [Google Scholar]
- 3.Farrell GC, Larter CZ. Nonalcoholic fatty liver disease: From steatosis to cirrhosis. Hepatology. 2006;43(S1):S99–S112. doi: 10.1002/hep.20973 [DOI] [PubMed] [Google Scholar]
- 4.Watanabe K, Watson E, Cremona ML, Millings EJ, Lefkowitch JH, Fischer SG, et al. ILDR2: an endoplasmic reticulum resident molecule mediating hepatic lipid homeostasis. PLoS One. 2013;8(6):e67234 doi: 10.1371/journal.pone.0067234 ; PubMed Central PMCID: PMC3691114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dokmanovic-Chouinard M, Chung WK, Chevre JC, Watson E, Yonan J, Wiegand B, et al. Positional cloning of "Lisch-Like", a candidate modifier of susceptibility to type 2 diabetes in mice. PLoS genetics. 2008;4(7):e1000137 doi: 10.1371/journal.pgen.1000137 ; PubMed Central PMCID: PMC2464733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liu Q, Muruve DA. Molecular basis of the inflammatory response to adenovirus vectors. Gene Ther. 2003;10(11):935–40. doi: 10.1038/sj.gt.3302036 [DOI] [PubMed] [Google Scholar]
- 7.Muruve DA, Barnes MJ, Stillman IE, Libermann TA. Adenoviral Gene Therapy Leads to Rapid Induction of Multiple Chemokines and Acute Neutrophil-Dependent Hepatic Injury in Vivo. Human Gene Therapy. 1999;10(6):965–76. doi: 10.1089/10430349950018364 [DOI] [PubMed] [Google Scholar]
- 8.Worgall S, Wolff G, Falck-Pedersen E, Crystal RG. Innate Immune Mechanisms Dominate Elimination of Adenoviral Vectors Following In Vivo Administration. Human Gene Therapy. 1997;8(1):37–44. doi: 10.1089/hum.1997.8.1-37 [DOI] [PubMed] [Google Scholar]
- 9.Ganz M, Szabo G. Immune and inflammatory pathways in NASH. Hepatology International. 2013;7(2):771–81. doi: 10.1007/s12072-013-9468-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tilg H, Moschen AR. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology. 2010;52(5):1836–46. doi: 10.1002/hep.24001 [DOI] [PubMed] [Google Scholar]
- 11.Wood M, Perotte P, Onishi E, Harper ME, Dinney C, Pagliaro L, et al. Biodistribution of an adenoviral vector carrying the luciferase reporter gene following intravesical or intravenous administration to a mouse. Cancer Gene Therapy. 1999;6(4):367–72. doi: 10.1038/sj.cgt.7700090 [DOI] [PubMed] [Google Scholar]
- 12.Beatty MS, Curiel DT. Adenovirus Strategies for Tissue-Specific Targeting. Advances in cancer research. 2012;115:39–67. doi: 10.1016/B978-0-12-398342-8.00002-1 PubMed PMID: PMC3664935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shayakhmetov DM, Li Z-Y, Ni S, Lieber A. Analysis of Adenovirus Sequestration in the Liver, Transduction of Hepatic Cells, and Innate Toxicity after Injection of Fiber-Modified Vectors. Journal of Virology. 2004;78(10):5368–81. doi: 10.1128/JVI.78.10.5368-5381.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jackson AL, Bartz SR, Schelter J, Kobayashi SV, Burchard J, Mao M, et al. Expression profiling reveals off-target gene regulation by RNAi. Nat Biotech. 2003;21(6):635–7. http://www.nature.com/nbt/journal/v21/n6/suppinfo/nbt831_S1.html. [DOI] [PubMed] [Google Scholar]
- 15.Jackson AL, Burchard J, Schelter J, Chau BN, Cleary M, Lim L, et al. Widespread siRNA “off-target” transcript silencing mediated by seed region sequence complementarity. RNA. 2006;12(7):1179–87. doi: 10.1261/rna.25706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Meehan RR, Barlow DP, Hill RE, Hogan BL, Hastie ND. Pattern of serum protein gene expression in mouse visceral yolk sac and foetal liver. The EMBO journal. 1984;3(8):1881–5. PubMed PMID: PMC557612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Weisend CM, Kundert JA, Suvorova ES, Prigge JR, Schmidt EE. Cre activity in fetal albCre mouse hepatocytes: Utility for developmental studies. genesis. 2009;47(12):789–92. doi: 10.1002/dvg.20568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rossi A, Kontarakis Z, Gerri C, Nolte H, Holper S, Kruger M, et al. Genetic compensation induced by deleterious mutations but not gene knockdowns. Nature. 2015;524(7564):230–3. doi: 10.1038/nature14580 http://www.nature.com/nature/journal/v524/n7564/abs/nature14580.html#supplementary-information. [DOI] [PubMed] [Google Scholar]
- 19.Hagelkruys A, Lagger S, Krahmer J, Leopoldi A, Artaker M, Pusch O, et al. A single allele of Hdac2 but not Hdac1 is sufficient for normal mouse brain development in the absence of its paralog. Development. 2014;141(3):604–16. doi: 10.1242/dev.100487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Deconinck AE, Rafael JA, Skinner JA, Brown SC, Potter AC, Metzinger L, et al. Utrophin-Dystrophin-Deficient Mice as a Model for Duchenne Muscular Dystrophy. Cell. 1997;90(4):717–27. https://doi.org/10.1016/S0092-8674(00)80532-2. [DOI] [PubMed] [Google Scholar]
- 21.Sun Z, Miller RA, Patel RT, Chen J, Dhir R, Wang H, et al. Hepatic Hdac3 promotes gluconeogenesis by repressing lipid synthesis and sequestration. Nature medicine. 2012;18(6):934–42. http://www.nature.com/nm/journal/v18/n6/abs/nm.2744.html#supplementary-information. doi: 10.1038/nm.2744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bell P, Gao G, Haskins ME, Wang L, Sleeper M, Wang H, et al. Evaluation of Adeno-Associated Viral Vectors for Liver-Directed Gene Transfer in Dogs. Human Gene Therapy. 2011;22(8):985–97. doi: 10.1089/hum.2010.194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kolios G, Valatas V, Kouroumalis E. Role of Kupffer cells in the pathogenesis of liver disease. World Journal of Gastroenterology: WJG. 2006;12(46):7413–20. doi: 10.3748/wjg.v12.i46.7413 PubMed PMID: PMC4087584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Carpino G, Renzi A, Onori P, Gaudio E. Role of Hepatic Progenitor Cells in Nonalcoholic Fatty Liver Disease Development: Cellular Cross-Talks and Molecular Networks. International Journal of Molecular Sciences. 2013;14(10):20112–30. doi: 10.3390/ijms141020112 PubMed PMID: PMC3821605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Washington K, Wright K, Shyr Y, Hunter EB, Olson S, Raiford DS. Hepatic stellate cell activation in nonalcoholic steatohepatitis and fatty liver. Human Pathology. 2000;31(7):822–8. http://dx.doi.org/10.1053/hupa.2000.8440. [DOI] [PubMed] [Google Scholar]
- 26.Magee N, Zou A, Zhang Y. Pathogenesis of Nonalcoholic Steatohepatitis: Interactions between Liver Parenchymal and Nonparenchymal Cells. BioMed Research International. 2016;2016:11 doi: 10.1155/2016/5170402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tacke F, Zimmermann HW. Macrophage heterogeneity in liver injury and fibrosis. Journal of Hepatology. 2014;60(5):1090–6. http://dx.doi.org/10.1016/j.jhep.2013.12.025. [DOI] [PubMed] [Google Scholar]
- 28.Luedde T, Schwabe RF. NF-kB in the liver—linking injury, fibrosis and hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2011;8(2):108–18. doi: 10.1038/nrgastro.2010.213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pradere J-P, Kluwe J, De Minicis S, Jiao J-J, Gwak G-Y, Dapito DH, et al. Hepatic macrophages but not dendritic cells contribute to liver fibrosis by promoting the survival of activated hepatic stellate cells in mice. Hepatology. 2013;58(4):1461–73. doi: 10.1002/hep.26429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Xu F, Zhen P, Zheng Y, Lijuan F, Aiting Y, Min C, et al. Preparation of Kupffer cell enriched non-parenchymal liver cells with high yield and reduced damage of surface markers by a modified method for flow cytometry. Cell Biology International. 2013;37(4):284–91. doi: 10.1002/cbin.10035 [DOI] [PubMed] [Google Scholar]
- 31.Xu X, Grijalva A, Skowronski A, van Eijk M, Serlie Mireille J, Ferrante Anthony W Jr. Obesity Activates a Program of Lysosomal-Dependent Lipid Metabolism in Adipose Tissue Macrophages Independently of Classic Activation. Cell metabolism. 2013;18(6):816–30. https://doi.org/10.1016/j.cmet.2013.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Alemany R, Suzuki K, Curiel DT. Blood clearance rates of adenovirus type 5 in mice. Journal of General Virology. 2000;81(11):2605–9. doi: 10.1099/0022-1317-81-11-2605 [DOI] [PubMed] [Google Scholar]
- 33.Tao N, Gao G-P, Parr M, Johnston J, Baradet T, Wilson JM, et al. Sequestration of Adenoviral Vector by Kupffer Cells Leads to a Nonlinear Dose Response of Transduction in Liver. Molecular Therapy. 2001;3(1):28–35. https://doi.org/10.1006/mthe.2000.0227. [DOI] [PubMed] [Google Scholar]
- 34.Birmingham A, Anderson EM, Reynolds A, Ilsley-Tyree D, Leake D, Fedorov Y, et al. 3' UTR seed matches, but not overall identity, are associated with RNAi off-targets. Nat Meth. 2006;3(3):199–204. https://www.nature.com/nmeth/journal/v3/n3/full/nmeth854.html. [DOI] [PubMed] [Google Scholar]
- 35.Anders S, Huber W. Differential expression analysis for sequence count data. Genome Biology. 2010;11(10):R106 doi: 10.1186/gb-2010-11-10-r106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Benjamini Y, Hochberg Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. Journal of the Royal Statistical Society Series B (Methodological). 1995;57(1):289–300. [Google Scholar]
- 37.Hulsen T, de Vlieg J, Alkema W. BioVenn–a web application for the comparison and visualization of biological lists using area-proportional Venn diagrams. BMC Genomics. 2008;9(488). doi: 10.1186/1471-2164-9-492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Speliotes EK, Yerges-Armstrong LM, Wu J, Hernaez R, Kim LJ, Palmer CD, et al. Genome-Wide Association Analysis Identifies Variants Associated with Nonalcoholic Fatty Liver Disease That Have Distinct Effects on Metabolic Traits. PLoS genetics. 2011;7(3):e1001324 doi: 10.1371/journal.pgen.1001324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dimmock DP, Zhang Q, Dionisi-Vici C, Carrozzo R, Shieh J, Tang LY, et al. Clinical and molecular features of mitochondrial DNA depletion due to mutations in deoxyguanosine kinase. Human Mutation. 2008;29(2):330–1. doi: 10.1002/humu.9519 [DOI] [PubMed] [Google Scholar]
- 40.Gao H-Z, Kobayashi K, Tabata A, Tsuge H, Iijima M, Yasuda T, et al. Identification of 16 novel mutations in the argininosuccinate synthetase gene and genotype–phenotype correlation in 38 classical citrullinemia patients. Human Mutation. 2003;22(1):24–34. doi: 10.1002/humu.10230 [DOI] [PubMed] [Google Scholar]
- 41.Speliotes EK, Willer CJ, Berndt SI, Monda KL, Thorleifsson G, Jackson AU, et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat Genet. 2010;42(11):937–48. http://www.nature.com/ng/journal/v42/n11/abs/ng.686.html#supplementary-information. doi: 10.1038/ng.686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xie S, Naslavsky N, Caplan S. Diacylglycerol kinases in membrane trafficking. Cellular Logistics. 2015;5(2):e1078431 doi: 10.1080/21592799.2015.1078431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Milne SB, Ivanova PT, Armstrong MD, Myers DS, Lubarda J, Shulga YV, et al. Dramatic Differences in the Roles in Lipid Metabolism of Two Isoforms of Diacylglycerol Kinase. Biochemistry. 2008;47(36):9372–9. doi: 10.1021/bi800492c [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gorden DL, Ivanova PT, Myers DS, McIntyre JO, VanSaun MN, Wright JK, et al. Increased Diacylglycerols Characterize Hepatic Lipid Changes in Progression of Human Nonalcoholic Fatty Liver Disease; Comparison to a Murine Model. PLOS ONE. 2011;6(8):e22775 doi: 10.1371/journal.pone.0022775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.De Souza AT, Dai X, Spencer AG, Reppen T, Menzie A, Roesch PL, et al. Transcriptional and phenotypic comparisons of Ppara knockout and siRNA knockdown mice. Nucleic Acids Research. 2006;34(16):4486–94. doi: 10.1093/nar/gkl609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Iacobas DA, Iacobas S, Urban-Maldonado M, Scemes E, Spray DC. Similar Transcriptomic Alterations in Cx43 Knockdown and Knockout Astrocytes. Cell Communication & Adhesion. 2008;15(1–2):195–206. doi: 10.1080/15419060802014222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kok Fatma O, Shin M, Ni C-W, Gupta A, Grosse Ann S, van Impel A, et al. Reverse Genetic Screening Reveals Poor Correlation between Morpholino-Induced and Mutant Phenotypes in Zebrafish. Developmental cell. 2015;32(1):97–108. https://doi.org/10.1016/j.devcel.2014.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Daude N, Wohlgemuth S, Brown R, Pitstick R, Gapeshina H, Yang J, et al. Knockout of the prion protein (PrP)-like Sprn gene does not produce embryonic lethality in combination with PrPC-deficiency. Proceedings of the National Academy of Sciences. 2012;109(23):9035–40. doi: 10.1073/pnas.1202130109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Smart N, Riley PR. Thymosin β4 in Vascular Development Response to Research Commentary. Circulation Research. 2013;112(3):e29–e30. doi: 10.1161/CIRCRESAHA.112.300555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gao Y, Zhang Y, Zhang D, Dai X, Estelle M, Zhao Y. Auxin binding protein 1 (ABP1) is not required for either auxin signaling or Arabidopsis development. Proceedings of the National Academy of Sciences. 2015;112(7):2275–80. doi: 10.1073/pnas.1500365112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mannerås‐Holm L, Kirchner H, Björnholm M, Chibalin AV, Zierath JR. mRNA expression of diacylglycerol kinase isoforms in insulin‐sensitive tissues: effects of obesity and insulin resistance. Physiological Reports. 2015;3(4). doi: 10.14814/phy2.12372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ugai H, Watanabe S, Suzuki E, Tsutsui-Nakata H, Yokoyama KK, Murata T. Stability of a Recombinant Adenoviral Vector: Optimization of Conditions for Storage, Transport and Delivery. Japanese Journal of Cancer Research. 2002;93(5):598–603. doi: 10.1111/j.1349-7006.2002.tb01296.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Krajden M, Minor JM, Rifkin O, Comanor L. Effect of Multiple Freeze-Thaw Cycles on Hepatitis B Virus DNA and Hepatitis C Virus RNA Quantification as Measured with Branched-DNA Technology. Journal of Clinical Microbiology. 1999;37(6):1683–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Furuse M, Izumi Y, Oda Y, Higashi T, Iwamoto N. Molecular organization of tricellular tight junctions. Tissue Barriers. 2014;2(3):e28960 doi: 10.4161/tisb.28960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Higashi T, Tokuda S, Kitajiri S-i, Masuda S, Nakamura H, Oda Y, et al. Analysis of the ‘angulin’ proteins LSR, ILDR1 and ILDR2 –tricellulin recruitment, epithelial barrier function and implication in deafness pathogenesis. Journal of Cell Science. 2013;126(4):966–77. doi: 10.1242/jcs.116442 [DOI] [PubMed] [Google Scholar]
- 56.Halldorsdottir S, Carmody J, Boozer CN, Leduc CA, Leibel RL. Reproducibility and accuracy of body composition assessments in mice by dual energy x-ray absorptiometry and time domain nuclear magnetic resonance. International journal of body composition research. 2009;7(4):147–54. PubMed PMID: PMC3169293. [PMC free article] [PubMed] [Google Scholar]
- 57.Folch J, Lees M, Stanley GHS. A simple method for the isolation and purification of total lipides from animal tissues. The Journal of biological chemistry. 1956;226:497. [PubMed] [Google Scholar]
- 58.Wang Z, Gerstein M, Snyder M. RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet. 2009;10(1):57–63. doi: 10.1038/nrg2484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Trapnell C, Pachter L, Salzberg SL. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics. 2009;25(9):1105–11. doi: 10.1093/bioinformatics/btp120 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF)
Data Availability Statement
The sequencing data discussed in this paper have been deposited in the National Center for Biotechnology Information (NCBI) Gene Expression Omnibus and are accessible through GEO series accession number GSE114289. (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE114289)