Abstract
Purpose
To investigate the effect of transcorneal electrical stimulation (TcES) on retinal ganglion cell (RGC) function and survival after acute ocular hypertension-related retinal injury in gerbil eyes.
Methods
Gerbil eyes were subjected to acute ocular hypertensive injury (80 mm Hg for 60 minutes). In the treatment group, TcES was applied to the surgical eye immediately and then twice weekly for a total of 1 month. In the control group, sham TcES was given to the surgical eye at the same time points. Retinal function was assessed and compared between groups using flash electroretinography. For histological analysis, the number of RGC and microglial cells were counted by immunofluorescence staining after the gerbils were sacrificed on day 7 and day 28. Real-time polymerase chain reaction and western blot analysis were conducted to compare expression of interleukin (IL)-10, IL-6, COX-2, tumor necrosis factor (TNF)-α, and NF-κB phosphorylation among groups.
Results
TcES-treated eyes had significantly higher RGC survival at 1 month compared to controls. This was associated with RGC function. Furthermore, TcES-treated eyes were shown to have increased IL-10 expression, with a corresponding reduction in IL-6 and COX-2 expression as well as reduction in NF-κB phosphorylation. This was associated with a suppression in microglial cell activation in TcES-treated eyes.
Conclusions
Early treatment with TcES in gerbils protected the RGC from secondary damage and preserved retinal function in acute ocular hypertensive injury through modulation of the microglial-cell activated local inflammatory response.
Translational Relevance
Our study strengthens the argument for translating TcES as a viable treatment in acute glaucoma.
Keywords: transcorneal electrical stimulation, acute ocular hypertensive injury, neuroprotection, anti-inflammatory
Introduction
Primary angle closure glaucoma (PACG) accounts for 91% of bilateral glaucoma blindness in China.1 Eyes with anatomically narrow iridocorneal angles are further at risk of a highly symptomatic variant of PACG known as an acute primary angle closure attack (APAC). The reported incidence of APAC in Asia is 15.5 cases per 100,000 population per year, with a significant number of sufferers developing severe and permanent visual loss.2 The sudden raised intraocular pressure (IOP) exerts damage on the optic nerve head through direct mechanical pressure. After an initial insult in an acute angle closure attack, there is rapid loss of directly injured retinal ganglion cells (RGCs). There is also a delayed but significant secondary loss of RGCs, whose axons are not damaged.3,4 This occurs after the initial attack is aborted. From research work on experimental models of glaucoma, neuro-inflammation has been proposed as a key pathogenetic mechanism for progressive RGC death. Furthermore, there is evidence that suppression of the local inflammatory response confers neuroprotective effects to the retina after injury.5,6 However, in clinical practice, the use of immunosuppressive treatment for retinal neuroprotection is hindered by limitations, including drug side effects like cataracts and glaucoma, as well as difficulties in effective drug delivery to the retina.
Neuronal electrostimulation is a well-established form of treatment in a number of neuropsychiatric and neurodegenerative diseases. More recently, there is emerging evidence that nerve stimulation confers local immunomodulatory effects in neuro-inflammatory disease states, and thus may serve as a potent, noninvasive form of anti-inflammatory treatment for the nervous system.7 In recent years, transcorneal electrical stimulation (TcES) has been used in retinal neuroprotection.8,9 Experiments on animal models have shown that TcES ameliorates degeneration of RGC and photoreceptors via upregulation of neurotrophic factor secretion, including insulin-like growth factor-1 (IGF-1), brain derived neurotrophic factor (BDNF), ciliary neurotrophic factor (CNTF), and B-cell lymphoma 2 (Bcl-2).10–13 TcES has also been shown to exert neuroprotective effects in rat models of optic neuropathies, including mechanical (crush injury) and ischemic types.14 Furthermore several clinical studies have demonstrated improved visual function in patients with traumatic optic neuropathy, anterior ischemic optic neuropathy, and retinal arterial occlusion after treatment with TcES.15,16 Such evidence supports the use of TcES as a method to ameliorate secondary RGC death after acute ocular hypertensive injury. More importantly, the local immunomodulatory effects of TcES have not been investigated in ocular diseases despite emerging evidence to support this from studies on other parts of the nervous system.
The aim of our study is to investigate the potential immunomodulatory and neuroprotective effects of TcES in a gerbil model of acute ocular hypertensive injury.
Materials and Methods
Animals
Mongolian gerbils, of 3 to 5 months old, were used in this experiment and kept in a temperature-controlled animal room subjected to a 12-hour light/12-hour dark cycle provided with water supply and sufficient food. All the animal care and experimental procedures conformed to the Association of Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research and were approved by the Committee on the Use of Live Animals in Teaching and Research (CULATR, 3204-14) of the University of Hong Kong.
Acute Ocular Hypertensive Injury
For our experiments, gerbils were used in our model of acute ocular hypertensive injury. Our choice of rodent was based on our group's previous experience with gerbils in ocular studies as well the inherent translational advantages of this rodent over other models, including having pigmented eyes and having a diurnal rhythm that approximates that of humans.17,18 The right eye of each gerbil was selected for experiments relating to this study. Acute ocular hypertension (AOH) was induced in the gerbil eyes using a protocol adapted from techniques previously described.19 The gerbils were first anesthetized using an intraperitoneal injection of ketamine and xylazine (100 and 10 mg/kg; Alfasan International BV, Woerden, Holland) with a subsequent injection of an additional 1/3 volume for maintenance. One drop of 0.5% proxymetacaine hydrochloride (0.5% Alcaine; Alcon, Fort Worth, TX) was applied to the eye for topical anesthesia and 2 minutes later one drop of tropicamide (1% Mydriacyl; Alcon) was applied for mydriasis. Balance salt solution (BSS; Alcon) was then infused into the anterior chamber of the eye through a 27-gauge needle that was connected to a hanging reservoir (Fig. 1). The elevated IOP was maintained for 60 minutes. During the surgical procedure, the gerbil's body temperature was maintained at 37 ± 0.5°C. The IOP was measured at baseline after anesthesia induction and then at 1 and 58 minutes after initiation of infusion using a TonoLab tonometer (Icare, Finland). These time points are based on similar models of ocular hypertensive injury in existing literature.19,20
Figure 1.
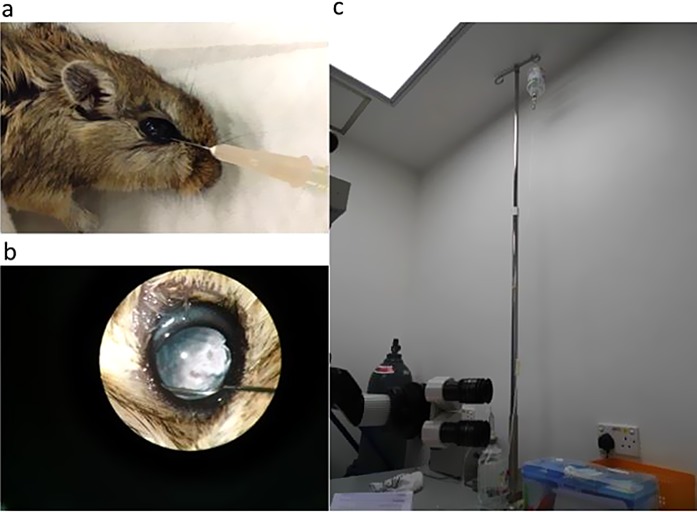
Setting of AOH induction. (a) Anterior chamber infusion of BSS through a needle to the gerbil's eye. (b) Intraocular view of the infusion under microscope. (c) The BSS was hanged at a height of 2.6 m to the gerbil's eye.
Transcorneal Electrical Stimulation
A monopolar contact lens electrode with gold ring mounted in the inner surface (Mayo, Japan) was placed on the surgical eye and was connected to an electrostimulator via an isolator (WPI, Sarasota, FL). The electrical stimulation was monitored by an oscilloscope. Gerbils were anesthetized as above described. Hydroxymethylcellulose gel (1.3%) (Alcon) was used as a coupling agent and for corneal protection. Bipolar rectangular current with pulse duration of 1 ms/phase, intensity of 100 μA, frequency of 20 Hz was delivered to the eye for 1 hour. It was given immediately after IOP elevation (day 1) and then 3 days later (day 4) in the 1 week's duration experiment. And for 1 month's course treatment, TcES was applied on day 1 and day 4 in each additional week to ensure consistency with the previous experiment. The sham group of gerbils received sham stimulation at the same time point in which the electrode was not connected to the electrostimulator.
Full Field Flash Electroretinography (ERG)
Animals were anesthetized as described in previous sections. Eye drops for topical anesthesia and mydriasis were used. ERG signals were recorded by the Espion Diagnosys system (Diagnosys LLC, Littleton, MA) through a pair of gold ring electrodes mounted in the inner surface (Diagnosys LLC). A needle electrode was inserted into the subcutaneous tissue at the midline of the ears to serve as a negative reference to each eye, and another needle electrode was inserted subcutaneously at the end of the tail for ground reference. Both eyes were stimulated simultaneously. Body temperature was maintained by the heating pad in the ERG machine.
After 5-minute light adaptation of using a green 40 cd/m2 background, photopic responses were recorded as averages of 25 responses under intensity of 1.22 log cd.s/m2. For the waveform analysis, a-wave was the first trough after beginning, b-wave was the initial peak after a-wave, and photopic negative response (PhNR) was the first trough following b-wave. All waves were measured from baseline and the exchange frequency between light and dark zones.
Retinal Flat Mount Immunohistochemistry
Gerbils were euthanized by 20% dorminal (240 mg/kg; Alfasan International BV) intraperitoneal injection. The eyes were enucleated quickly and fixed in 4% formaldehyde for 30 minutes. Then it was blocked in a solution containing 4% normal donkey serum (NDS; Jackson Immuno, Linscott's USA, West Grove, PA) in phosphate-buffered saline (PBS) with 0.5% Triton-100 (PBST) for 2 hours. The primary antibodies used were goat anti-Brn3a antibody (1:500; Santa Cruz Biotechnology, Heidelberg, Germany) and rabbit anti-Iba-1 antibody (1:500; Wako Chemicals, Richmond, VA). The retinae were incubated in the primary antibody with diluent solution of 1% NDS at 4°C overnight. The second day, they were rinsed with PBST three times before incubated in the secondary antibody with diluent solution for 2 hours at room temperature. The secondary antibody was conjugated to either Alexa 488 (1:500; Invitrogen, Carlsbad, CA) or Alexa 594 (1:500; Invitrogen) to be visualized by fluorescent microscope. Finally, an anti-fade medium (Dako North America, Carpinteria, CA) was used to mount the retinae on glass slides. Under fluorescence microscope, three fields with a fixed distance along the middle line of each quadrant were captured for cell counting. The counter was blinded during immunohistochemistry review and had no knowledge of the group assignment of the histological specimen.
Retinal Western Blot Analysis
To further understand the underlying mechanisms of how TcES protected RGC from secondary cell death and preserved inner retinal function, we investigated the changes in the local inflammatory response between groups. The NF-κB-p65 signaling pathway, which plays a key role in the inflammation response,21,22 was investigated. Furthermore, proinflammatory proteins including interleukin (IL)-6, COX-2, and anti-inflammatory factor IL-10 were measured by western blot. Retinal lysates were extracted by sonication in ice-cold extraction buffer with 50:1 of radioimmunoprecipitation assay (RIPA) buffer (50 mM TRIS-HCl, 150 mM NaCl, 1% sodium deoxycholate, 1% Triton X-100, 0.1% sodium dodecyl sulfate [SDS], pH 7.4) and protease inhibitor (Roche, Basel, Switzerland). They were then centrifuged with 13,500 rpm at 4°C for 30 minutes. The supernatant was collected. Proteins in retinal lysates were separated by SDS-polyacrylamide gel electrophoresis (PAGE) and then transferred to polyvinylidene difluoride (PVDF) membranes. The membranes were blocked by 5% nonfat milk with nonspecific binding sites in room temperature for 2 hours followed by incubation with primary antibodies of target proteins overnight at 4°C. Primary antibodies used here included the following: rabbit anti-IL-6 (1:1000; molecular weight [MW]: 17 kDa; Abcam, Cambridge, MA); rabbit anti-IL-10 (1:1000; MW: 20 kDa; Abcam); rabbit anti-COX-2 (1:1000; MW: 74 kDa; Abcam); rabbit anti-phosphorylated-NF-κB p65 (Ser536) (pNF-κB; 1:1000; MW: 65 kDa; Cell Signaling Technology, Beverly, MA); mouse anti-NF-κB p65 (1:1000; MW: 65 kDa; Cell Signaling Technology); and actin (1:5000; MW: 42 kDa; Chemicon, Temecula, CA). Secondary HRP-conjugated anti-rabbit (1:2000; Vector Laboratories, Burlingame, CA) and anti-mouse (1:5000; Vector Laboratories) specific to the primary antibodies were applied for 2 hours in room temperature after washing. The signals of target protein were visualized by ECL or ECL Plus (Amersham Pharmacia Biotech, Arlington Heights, IL) by the MyECL Imager (Thermo Fisher Scientific, Waltham, MA) under the chemiluminescence mode. Measurements of the signals on the film were performed using Image J software (National Institute of Mental Health, Bethesda, MD) after scanning them to digital figures. The ratio of the value of the protein product to the internal control (β-actin) or the ratio of the phosphorylated protein to the total form protein were obtained and then expressed as percentage of the normal group.
Retinal Quantitative Real-Time Reverse Transcription–Polymerase Chain Reaction (RT-PCR)
The retinae of animal were harvested for RNA extraction to measure the gene expression of tumor necrosis factor (TNF)-α, IL-6, and IL-10. Total RNA was extracted by the RNeasy Mini Kit (Qiagen, Germany). High Capacity RNA-to-cDNA Kit (Thermo Fisher Scientific) was used for the reverse transcription of the RNA. The samples of cDNA were loaded into the fast optical 96-well reaction plate with the Fast SYBR Green Master Mix (Thermo Fisher Scientific) under a light avoidance condition. The plate was sealed with an adhesive cover after loading and put into the Machine of StepOnePlus™ Real-Time PCR Systems (Thermo Fisher Scientific) for amplification and reading. The glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the internal control. The mRNA level was expressed by the 2(−ΔΔCt) method and compared as relative level of times of the normal control. The primer sequences are as follows:
GAPDH forward, 5′-AACGGCAC AGTCAAGGCTGAGAACG-3′, reverse, 5′-CAACATACTCGGCACC GGCATCG-3′; TNF-α forward, 5′-GCCCCCACCTCGTGCTCCTC AC-3′, reverse, 5′-GGCAGGGGCTCTTGATGGCAGACAG-3′; IL-6 forward, 5′-ATGGCTGAAGTCCAAGACC-3′, reverse, 5′-GGAATGTC CTCAGCTTGGTA-3′; and IL-10 forward, 5′-CAGGGCTCCTGAAAGAGT TA-3′, reverse, 5′-AGAATGAGGTCAGGGGAATC-3′.
Data Analysis
Results were presented as mean ± standard error of measurement (SEM). Multiple comparisons were performed by one-way ANOVA with Tukey post hoc tests (Prism 6; GraphPad Software, Inc, San Diego, CA). A P value of less than 0.05 was considered to be statistically significant.
Results
Ocular Hypertension Was Sustained for 60 Minutes
After anesthesia induction and before intraocular infusion of basic salt solution, baseline IOP was measured at 10.80 ± 0.94 mm Hg (n = 15) in the sham treatment group eyes and 10.80 ± 0.77 mm Hg (n = 15) in the TcES group eyes, without significant differences between groups. IOP was measured at 1 minute after anterior chamber paracentesis and initiation of infusion. The average measured IOP at this time point was 83.93 ± 0.51 mm Hg (n = 15) in the sham treatment group and 84.07 ± 0.64 mm Hg (n = 15) in the TcES group. The average IOP was again determined at 58 minutes after initiation of infusion, and was recorded to be 83.20 ± 0.56 mm Hg (n = 15) and 83.60 ± 0.56 mm Hg (n = 15) in the sham and TcES groups, respectively. There were no significant differences in mean IOP between the two groups as well as the two time points (Fig. 2).
Figure 2.
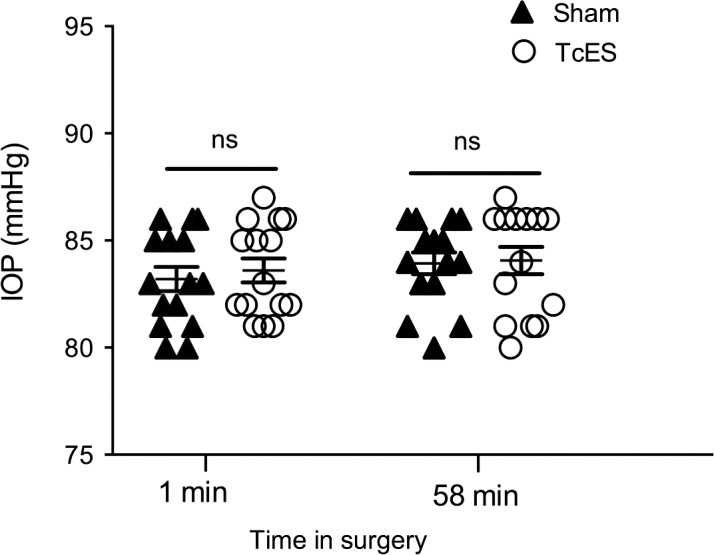
IOP readings during AOH. There were no significant differences (P > 0.05) of the IOP readings between the TcES group and sham group at the beginning or the end of the surgery. ns, no significance.
TcES Preserved Inner Retinal Function
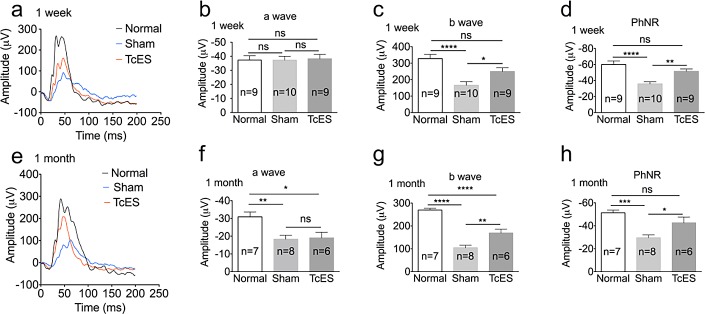
Full field flash ERG was performed 1 week and 1 month after injury. AOH causes transient ocular ischemia by raising IOP, which mimic the acute attack of glaucoma. However, it is reported that the retinal injury from AOH varies from species and different laboratory centers. It is said that outer layers of retina were more vulnerable to ischemia in rabbits23 while the inner layers, including the RGC, are more susceptible to ischemic damage in rats.24 In our experiment, we confirmed that AOH caused firstly inner retinal layers dysfunction by full field flash ERG. There was a 49.4% reduction of amplitude of b-wave (166.2 ± 22.32 μV in sham group versus 328.5 ± 24.71 μV in normal group; P < 0.0001; Fig. 3c) and a 40.3% reduction of amplitude of PhNR in the sham group 1 week after AOH (−35.84 ± 2.634 μV in sham group versus −59.94 ± 4.423 μV in normal group; P < 0.0001; Fig. 3d). However, there were no changes in the a-wave's amplitude (−37.2 ± 2.767 μV in sham group versus −37.32 ± 3.021 μV in normal group; P > 0.05; Fig. 3b) 1 week after AOH. At 1 month after AOH, there was a 40.7% reduction of a-wave amplitude (18.35 ± 2.184 μV in sham group versus −30.93 ± 2.686 μV in normal group; Fig. 3f) and further reduction in b-wave and PhNR's amplitudes in the sham group, which were 61.2% (P < 0.0001; Fig. 3g) and 42.3% (P < 0.001; Fig. 3h) lower than normal eyes, respectively.
Figure 3.
Retinal function reduced after AOH but was improved by TcES. (a), (e) Representative ERG responses at 1 week and 1 month. (b), (c), (d), (f), (g), (h) Averaged amplitudes of 1 week and 1 month a-wave, b-wave, and PhNR. ****P < 0.0001, ***P < 0.001, **P < 0.01, *P < 0.05. n, number of retinas in each group; ns, no significance.
After TcES treatment, there was marked improvement in b-wave and PhNR amplitudes, while there was no beneficial effect on the a-wave amplitude. At 1 week after surgery, the TcES-treated group had a 50.5% higher b-wave amplitude (250.1 ± 22.15 μV; P < 0.05; Fig. 3c) and a 42.9% higher PhNR amplitude (−51.52 ± 2.824 μV; P < 0.01; Fig. 3d) compared to the sham-treated group. Furthermore, at 1 month after surgery, there was further improvement in b-wave and PhNR amplitudes, reported 61.8% (169.2 ± 16.48 μV in TcES group; P < 0.01; Fig. 3g) and 44.1% (−42.60 ± 4.840 μV in TcES group; P < 0.05; Fig. 3h) higher than that in the sham-treated group, respectively.
Significant RGC Cell Survival 1 Month After TcES Treatment
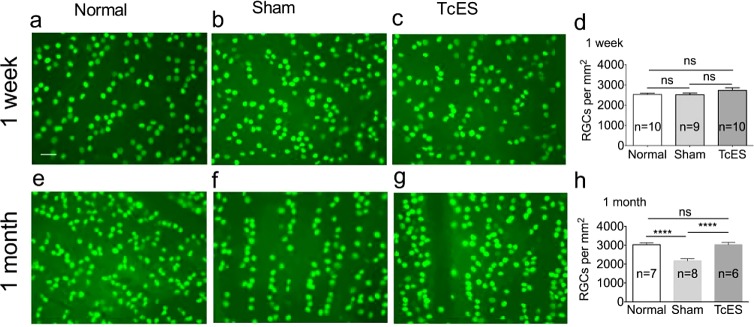
There were no differences of the RGC density in the whole retina among three groups (2526 ± 65 cells/mm2 in the normal group versus 2514 ± 84 cells/mm2 in the sham group versus 2726 ± 131.6 cells/mm2 in the TcES group; P > 0.05; Figs. 4a–d) 7 days after AOH.
Figure 4.
Protection of RGCs by TcES. (a–c) Brn-3a stained RGCs in retinal flat mount of normal, sham, and TcES group at 1 week. (e–g) Brn-3a stained RGCs in retinal flat mount of normal, sham, and TcES group at 1 month. (d, h) Mean density of the RGCs in the retina flat mount of 1 week and 1 month results. Scale bar: 40 μm. ****P < 0.0001. n, number of retinas in each group; ns, no significance.
At 1 month after surgery, the overall RGC density in the sham group was 28.1% less than the normal group (2177 ± 102 cells/mm2 in the sham group versus 3028 ± 101 cells/mm2 in normal group; P < 0.0001; Figs. 4e, 4f, 4h). In the TcES-treated group, which underwent electrical stimulation twice weekly for the entire month, there was a 39.2% higher overall RGC density compared to the sham-treated group (3031 ± 119 cells/mm2 in the TcES group; P < 0.0001; Figs. 4f–h). Accordingly, the TcES significantly ameliorated secondary cell death 4 weeks after the initial injury.
Inhibition of NF-κB Signaling Pathway by TcES Treatment
At 1 week after surgery, the sham-treated group had a 1.67-fold upregulation of phosphorylated NF-κB-p65 compared to normal eyes (P < 0.001; Figs. 5a, 5b) while the TcES-treated group had no significant upregulation of phosphorylated NF-κB-p65 expression (P > 0.05; Figs. 5a, 5b). Moreover, the pro-IL-6 expression in sham-treated eyes increased 1.90-fold compared to normal eyes (P < 0.01; Fig. 5c) while TcES-treated eyes again had no significant upregulation (P > 0.05; Fig. 5c). The expression level of COX-2 was shown to be depressed in TcES-treated eyes, which had a level 82.7% lower than that in sham-treated eyes (P < 0.05; Fig. 5d). Meanwhile, the anti-inflammatory cytokine IL-10 was significantly elevated in TcES-treated eyes to 1.6-fold compared to the sham-treated eyes (P < 0.05; Fig. 5e).
Figure 5.
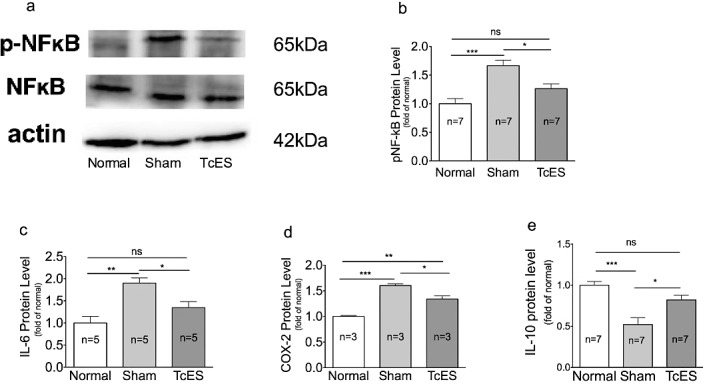
TcES suppressed the inflammatory factors. (a, b) Western blot results of NF-κB phosphorylation level. (c–e) Western blot results of IL-6, COX-2, and IL-10 protein level. ***P < 0.001, **P < 0.01, *P < 0.05. n, number of retinas in each group; ns, no significance.
To determine whether TcES altered the inflammatory response at the gene-expression stage, the mRNA levels of TNF-α, IL-6, and IL-10 were measured by quantitative real-time RT-PCR. The result was that mRNA expression of TNF-α and IL-6 genes was significantly higher in the sham group compared to normal eyes. The expression level of TNF-α mRNA was upregulated 2.18-fold compared to normal eyes (P < 0.01; Fig. 6a) and IL-6 expression was upregulated 2.10-fold compared to normal eyes (P < 0.05; Fig. 6b). Correspondingly, the expression of anti-inflammatory cytokine IL-10 gene in the sham-treated group was reduced to only 40.72% of that in normal eyes (P < 0.01; Fig. 6c). All three genes were neither significantly overexpressed or underexpressed in TcES-treated eyes compared to normal eyes (P > 0.05; Figs. 6a–c).
Figure 6.
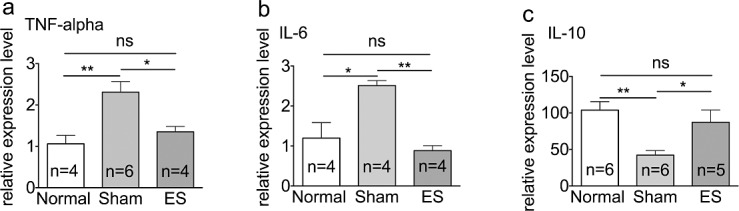
Downregulation of mRNA expression level in inflammatory genes by TcES. (a–c) mRNA expression in TNF-α, IL-6, and IL-10 genes. **P < 0.01, *P < 0.05. n, number of retinas in each group; ns, no significance.
Suppression of Microglial Cells Activation by TcES
We conducted experiments to investigate whether the change in local inflammatory response was a result of microglial cell modulation by TcES. We performed Iba-1 labeling at 1 week and 1 month after surgery and compared the measured densities between groups. Our experiments noted that at 1 week after AOH, the microglial density in the sham-treated group increased to 3.3-fold compared to the normal eyes (335.8 ± 25.38 cells/mm2 in the sham group versus 101.0.7 ± 10.60 cells/mm2 in normal group; P < 0.0001; Fig. 7d) while microglial density was elevated only 2.5-fold in the TcES-treated group (248.8 ± 18.13 cells/mm2 in the TcES group (P < 0.001; Fig. 7d). At 1 month after surgery, the microglial cell density was 1.34-fold higher in the sham-treated group compared to normal eyes (124.6 ± 9.173 cells/mm2 in the sham group versus 92.88 ± 9.390 cells/mm2 in normal group; P < 0.05; Figs. 7a, 7b, 7e). In the TcES-treated eyes, microglial activation was completely suppressed, and density was not significantly different to that found in normal eyes (88.76 ± 7.208 cells/mm2 in the TcES group; P > 0.05; Figs. 7a, 7c, 7e).
Figure 7.
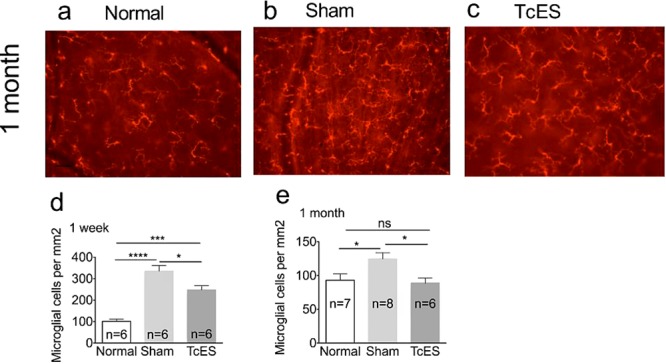
TcES inhibited the activation of microglial cells in RGC layer. (a–c) Fluorescent images of Iba-1 stained microglial cells in retinal whole mounts. (d, e) Quantification of microglia density at 1 week and 1 month after AOH. Scale bar: 50 μm. ****P < 0.0001; ***P < 0.001, *P < 0.05. n, number of retinas in each group; ns, no significance.
Discussion
In our experiments, gerbil eyes that underwent TcES treatment after ocular hypertensive injury had significantly higher RGC survival rates compared to sham-treated eyes, with preservation of RGC function on full-field flash ERG. We further demonstrated that the underlying mechanism of TcES-related RGC neuroprotection was associated with the inhibition of retinal microglial cell activation and suppression of NF-κB pathway.
In our gerbil model with AOH injury, the mean IOP elevation achieved was 82.09 mm Hg for 1 hour. This corresponded with significant retinal electrophysiological dysfunction starting 1 week after injury (Fig. 3) and RGC cell death of 28.1% starting 1 month after injury (Fig. 4). TcES treatment was effective in improving RGC and bipolar cell function, respectively, as evidenced by the recovery of PhNR and b-wave amplitudes in treated eyes, while providing no significant improvement to photoreceptor cell function, as evidenced by a lack of recovery of the a-wave amplitude at 1 month. This differs from previously reported studies on the protective effects of electrical stimulation on photoreceptors in animal models25–27 as well as in cell culture studies.28 This may be due to several potential reasons: first, the animals used in this study were gerbils, which differs from the animal types that were involved in the published literature. Secondly, we only measured the photopic a-wave changes in ERG, which predominantly represents the function of cone photoreceptors but not rod photoreceptors. Due to gerbils possessing a diurnal rhythm, 13% of their retinal photoreceptors are cones compared to only approximately 1% to 3% cones in rats and mice, which are nocturnal animals. The differences in cone photoreceptor concentration between the experimental animals results in the observed differences in photopic electroretinographic responses, as shown by previous experiments conducted by our group.17
The role of the local inflammatory response in secondary retinal degeneration after acute ocular hypertensive injury is an important target for neuroprotection. We examined whether TcES confers an immunomodulatory effect in the context of our experiments. Our study focused on the NF-κB signaling pathway, given its critical role in proinflammatory signaling29 and also in glaucomatous optic neuropathy.30 In our study, acute ocular hypertensive injury resulted in an upregulation of local proinflammatory factors, including IL-6, COX-2, and TNF-α as well as increased NF-κB-p65 phosphorylation in the retina. Conversely, expression of the anti-inflammatory cytokine IL-10 was downregulated after injury. TcES treatment was shown to inhibit the expression of the proinflammatory cytokines, while upregulating the expression of IL-10. This is consistent with existing literature where inhibition of NF-κB has been shown to ameliorate retinal neuronal cell death after injury31 as well as reduce neuroinflammation in the central nervous system.32 It is thus reasonable to believe that TcES protected the RGCs survival through downregulation of NF-κB signaling pathway or at least partly via anti-inflammation mechanism.
Microglial activation is well understood in retinal neurodegenerative diseases.33 In our study, we demonstrated that the density of microglia cells significantly increased after AOH insult. This indicates that the microglia play a prominent role in the inflammatory response in this acute glaucoma model. It is further proved that post injury treatment with TcES suppresses microglial cell activation. Previous studies have shown that the NF-κB pathway is activated by glial cells, thus explained the association between suppression of microglial cell activation and modulation of the local inflammatory response in TcES treated eyes.34,35
Our study has several limitations. In our experiments, the role of electrical stimulation therapy on photoreceptors was not fully investigated. Further work should be undertaken to investigate the detailed role of electrical stimulation on non-RGC cells of the retina. Secondly, experiments on inflammatory cytokine expression would have benefited from more measurement time points. Furthermore, another area of concern is the possibility that the antibodies used in our immunohistochemistry and western blot analysis may not identify gerbil proteins, given the potential for lack of cross-reactivity between species. Our experiments aimed to address this issue firstly by having a “normal eyes” group as a blank control to ensure that the antibodies in the experiments identified gerbil proteins. Furthermore, with reference to previous published work on gerbil models of experimental eye disease, including two papers from our group, antibodies used for rats and mice have been applied in gerbil experiments without issues.36–38 Finally, the relationship between the observed neuroprotective effects and immunomodulatory effects of TcES treatment need to be clarified. It remains to be known whether enhanced inner retinal layer survival and function results in a tempered local inflammatory response or vice versa. Our future work will be focused on further elucidating the potential immunomodulatory effects of TcES through the use of gene-knockout mice.
Conclusion
In conclusion, the present study demonstrated the potential neuroprotective effects of TcES after acute ocular hypertensive injury in gerbils. This effect is partially mediated by modulation of inflammatory cytokines and inhibition of the NF-κB signaling pathway as well as suppression of microglia activation. Our study provides a better understanding of the possible mechanism of TcES and strengthens the possibility of translating its use in clinical trials as a viable treatment strategy for acute ocular hypertensive injury. More importantly, in the wider context of neuro-degenerative and inflammatory diseases, our experiments provide further evidence of the immunomodulatory role of electrical neuro-stimulation. While it was previously thought that neuronal stimulation exerted its beneficial effects solely through growth factor secretion, there is growing weight to the argument for its immunomodulatory effects.10–13 In the long-term, this would serve as a potentially safe alternative to systemic immunosuppressive therapy in neuro-inflammation, which invariably carries significant risk of morbidity and mortality.
Acknowledgments
Disclosure: L. Fu, None; F.K. Fung, None; A.C.-Y. Lo, None; Y.-K. Chan, None; K.-F. So, None; I.Y.-H. Wong, None; K.C. Shih, None; J.S.-M. Lai, None
References
- 1. Quek DT, Koh VT, Tan GS, Perera SA, Wong TT, Aung T. . Blindness and long-term progression of visual field defects in chinese patients with primary angle-closure glaucoma. . 2011; 152: 463– 469. [DOI] [PubMed] [Google Scholar]
- 2. Tan AM, Loon SC, Chew PT. . Outcomes following acute primary angle closure in an Asian population. . 2009; 37: 467– 472. [DOI] [PubMed] [Google Scholar]
- 3. Qu J, Wang D, Grosskreutz CL. . Mechanisms of retinal ganglion cell injury and defense in glaucoma. . 2010; 91: 48– 53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Li HY, Ruan YW, Ren CR, Cui Q, So KF. . Mechanisms of secondary degeneration after partial optic nerve transection. . 2014; 9: 565– 574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Yang X, Hondur G, Tezel G. . Antioxidant treatment limits neuroinflammation in experimental glaucoma. . 2016; 57: 2344– 2354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Soto I, Howell GR. . The complex role of neuroinflammation in glaucoma. . 2014; 4 8: a017269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ulloa L, Quiroz-Gonzalez S, Torres-Rosas R. . Nerve stimulation: immunomodulation and control of inflammation. . 2017; 23: 1103– 1120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Fu L, Lo AC, Lai JS, Shih KC. . The role of electrical stimulation therapy in ophthalmic diseases. . 2015; 253: 171– 176. [DOI] [PubMed] [Google Scholar]
- 9. Sehic A, Guo S, Cho KS, Corraya RM, Chen DF, Utheim TP. . Electrical stimulation as a means for improving vision. . 2016; 186: 2783– 2797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Morimoto TMT, Matsuda S. . Transcorneal electrical stimulation rescues axotomized retinal ganglion cells by activating endogenous retinal IGF-1 system. . 2005; 46: 2147– 2155. [DOI] [PubMed] [Google Scholar]
- 11. Morimoto TFT, Choi J-S. . Transcorneal electrical stimulation promotes the survival of photoreceptors and preserves retinal function in royal college of surgeons rats. . 2007; 48: 4725– 4732. [DOI] [PubMed] [Google Scholar]
- 12. Sato TFT, Lee T-S. . Direct effect of electrical stimulation on induction of brain-derived neurotrophic factor from cultured retinal Müller cells. . 2008; 49: 4641– 4646. [DOI] [PubMed] [Google Scholar]
- 13. Ni YQ GD, Xu HD, Xu GZ, Da CD. . Neuroprotective effect of transcorneal electrical stimulation on light-induced photoreceptor degeneration. . 2009; 219: 439– 452. [DOI] [PubMed] [Google Scholar]
- 14. Miyake KYM, Inoue Y, Hata Y. . Neuroprotective effect of transcorneal electrical stimulation on the acute phase of optic nerve injury. . 2007; 48: 2358– 2361. [DOI] [PubMed] [Google Scholar]
- 15. Inomata KSK, . Ohde H. Transcorneal electrical stimulation of retina to treat longstanding retinal artery occlusion. . 2007; 245: 1773– 1780. [DOI] [PubMed] [Google Scholar]
- 16. Morimoto TFT, Matsushita K. . Evaluation of residual retinal function by pupillary constrictions and phosphenes using transcorneal electrical stimulation in patients with retinal degeneration. . 2006; 244: 1283– 1292. [DOI] [PubMed] [Google Scholar]
- 17. Yang S, Luo X, Xiong G, So KF, Yang H, Xu Y. . The electroretinogram of Mongolian gerbil (Meriones unguiculatus): comparison to mouse. . 2015; 589: 7– 12. [DOI] [PubMed] [Google Scholar]
- 18. Zhang T, Huang L, Zhang L,et al. ON and OFF retinal ganglion cells differentially regulate serotonergic and GABAergic activity in the dorsal raphe nucleus. . 2016; 6: 26060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Wang K, Peng B, Lin B. . Fractalkine receptor regulates microglial neurotoxicity in an experimental mouse glaucoma model. . 2014; 62: 1943– 1954. [DOI] [PubMed] [Google Scholar]
- 20. Mi XS, Feng Q, Lo AC,et al. Protection of retinal ganglion cells and retinal vasculature by Lycium barbarum polysaccharides in a mouse model of acute ocular hypertension. . 2012; 7: e45469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Lentsch AB, Ward PA. . Activation and regulation of NFkappaB during acute inflammation. . 1999; 37: 205– 208. [DOI] [PubMed] [Google Scholar]
- 22. McKenna S, Wright CJ. . Inhibiting IkappaBbeta-NFkappaB signaling attenuates the expression of select pro-inflammatory genes. . 2015; 128: 2143– 2155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Johnson NF, Foulds WS. . The effects of total acute ischaemia on the structure of the rabbit retina. . 1978; 27: 45– 59. [DOI] [PubMed] [Google Scholar]
- 24. Buchi ER, Suivaizdis I, Fu J. . Pressure-induced retinal ischemia in rats: an experimental model for quantitative study. . 1991; 203: 138– 147. [DOI] [PubMed] [Google Scholar]
- 25. Morimoto T, Fujikado T, Choi JS,et al. Transcorneal electrical stimulation promotes the survival of photoreceptors and preserves retinal function in royal college of surgeons rats. . 2007; 48: 4725– 4732. [DOI] [PubMed] [Google Scholar]
- 26. Ni YQ, Gan DK, Xu HD, Xu GZ, Da CD. . Neuroprotective effect of transcorneal electrical stimulation on light-induced photoreceptor degeneration. . 2009; 219: 439– 452. [DOI] [PubMed] [Google Scholar]
- 27. Morimoto T, Kanda H, Kondo M, Terasaki H, Nishida K, Fujikado T. . Transcorneal electrical stimulation promotes survival of photoreceptors and improves retinal function in rhodopsin P347L transgenic rabbits. . 2012; 53: 4254– 4261. [DOI] [PubMed] [Google Scholar]
- 28. Zhou WT, Ni YQ, Jin ZB,et al. Electrical stimulation ameliorates light-induced photoreceptor degeneration in vitro via suppressing the proinflammatory effect of microglia and enhancing the neurotrophic potential of Muller cells. . 2012; 238: 192– 208. [DOI] [PubMed] [Google Scholar]
- 29. Senftleben U, Karin M. . The IKK/NF-kappa B pathway. . 2002; 30: S18– S26. [PubMed] [Google Scholar]
- 30. Agapova OA, Kaufman PL, Hernandez MR. . Androgen receptor and NFkB expression in human normal and glaucomatous optic nerve head astrocytes in vitro and in experimental glaucoma. . 2006; 82: 1053– 1059. [DOI] [PubMed] [Google Scholar]
- 31. Yoshida A, Yoshida S, Ishibashi T, Kuwano M, Inomata H. . Suppression of retinal neovascularization by the NF-kappaB inhibitor pyrrolidine dithiocarbamate in mice. . 1999; 40: 1624– 1629. [PubMed] [Google Scholar]
- 32. Khare P, Datusalia AK, Sharma SS. . Parthenolide, an NF-kappaB inhibitor ameliorates diabetes-induced behavioural deficit, neurotransmitter imbalance and neuroinflammation in type 2 diabetes rat model. . 2017; 19 1: 101– 112. [DOI] [PubMed] [Google Scholar]
- 33. Ramirez AI, de Hoz R, Salobrar-Garcia E,et al. The role of microglia in retinal neurodegeneration: Alzheimer's disease, Parkinson, and glaucoma. . 2017; 9: 214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Dang Y, Xu Y, Wu W,et al. Tetrandrine suppresses lipopolysaccharide-induced microglial activation by inhibiting NF-kappaB and ERK signaling pathways in BV2 cells. . 2014; 9: e102522. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 35. Vuong B, Hogan-Cann AD, Alano CC,et al. NF-kappaB transcriptional activation by TNFalpha requires phospholipase C, extracellular signal-regulated kinase 2 and poly(ADP-ribose) polymerase-1. . 2015; 12: 229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Lau BW, Ren C, Yang J,et al. Light deprivation induces depression-like behavior and suppresses neurogenesis in diurnal mongolian gerbil (Meriones unguiculatus). . 2011; 20: 871– 881. [DOI] [PubMed] [Google Scholar]
- 37. Luan L, Ren C, Lau BW,et al. Y-like retinal ganglion cells innervate the dorsal raphe nucleus in the Mongolian gerbil (Meriones unguiculatus). . 2011; 6: e18938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Shimizu T, Choi E, Petersen CP,et al. Characterization of progressive metaplasia in the gastric corpus mucosa of Mongolian gerbils infected with Helicobacter pylori. . 2016; 239: 399– 410. [DOI] [PMC free article] [PubMed] [Google Scholar]