Abstract
Introduction
Low levels of sex hormones are common in patients with chronic kidney disease (CKD) and may be a contributing factor to bone fragility. We investigated associations between levels of sex hormones and bone mineral density (BMD) in adult kidney transplantation candidates.
Methods
Volumetric BMD of spine and hip were measured by computed tomography. Parathyroid hormone (PTH), testosterone (T), estradiol (E), and sex hormone–binding globulin were measured from fasting morning blood samples. Bioavailable (Bio) T and E were calculated based on constants for protein binding.
Results
A total of 146 patients (102 men and 44 women) were included in the analyses. The median age was 54 years (range, 32−72 years); 32% were diabetic; and 36% received maintenance dialysis therapy. In men, Bio T was positively associated with BMD at the lumbar spine (β = 5.02, P = 0.002), total hip (β = 6.35, P = 0.001), and femoral neck (β = 13.9, P = 0.002), independently of age, body mass index, dialysis, diabetes type 1 and 2, parathyroid hormone, and steroid exposure. Bio E was positively associated with BMD at the lumbar spine (β = 0.23, P = 0.03) and femoral neck (β = 0.61, P = 0.04) using the same fully adjusted model. In postmenopausal women, Bio T was positively correlated with lumbar spine BMD (r = 0.46, P = 0.02).
Conclusion
High endogenous levels of sex hormones are associated with greater BMD in male kidney transplantation candidates. Disturbances in the gonadal axis may contribute to skeletal fragility in men with late-stage CKD.
Keywords: bone density, chronic kidney disease, gonadal sex steroids, hypogonadism, osteoporosis
Gonadal dysfunction is common in chronic kidney disease (CKD), particularly in the late stages.1 Testosterone (T) and estradiol (E) decrease bone resorption in adults,2, 3 and low levels are known to cause osteopenia and osteoporosis in both men and women in the general population.4 Patients with CKD suffer renal bone disease with changes in bone quantity and quality,5 resulting in an increased risk of fractures.6 Disturbances in the gonadal axis could be a contributing factor to bone fragility in these patients.
The effects of sex hormone levels on bone health in CKD has not been well described. Low levels of E are associated with reduced BMD in both men7 and women8, 9 on hemodialysis, and BMD has been shown to improve with hormone replacement therapy in women.10 In male patients, it has been suggested that the action of sex hormones on BMD could be mediated through the receptor activator of nuclear factor−κΒ (RANK) and RANK ligand (RANKL) system,11, 12 although others have found no correlation between levels of T and BMD.7
Only small percentages of T and E circulate unbound. The greater fractions are either tightly bound to sex hormone–binding globulin or more loosely bound to albumin.13 A bioavailable fraction, the sum of free and albumin-bound hormone concentrations, can be calculated based on constants for protein binding.14, 15, 16 It is still unclear whether the biological actions of T and E are best represented by the total, bioavailable, or unbound forms.17 This issue may be particularly relevant in CKD, in which changes in the gonadal axis occur at several levels18 and disturbances in albumin levels are frequent.19
As there is a lack of knowledge of the relationship between sex hormones and bone density in CKD, we investigated the associations between the total and bioavailable fractions of T and E and volumetric bone mineral density (BMD) in a cohort of kidney transplantation candidates. We hypothesized that there would be a positive relationship between endogenous levels of sex hormones and BMD in men and women with late-stage CKD.
Methods
Study Participants
Adult kidney transplantation candidates were consecutively enrolled from 4 centers in Denmark from February 2011 through January 2014. The inclusion criteria were indication for cardiovascular screening before kidney transplantation by 1 or more of the following characteristics: age >40 years, diabetes mellitus (DM), dialysis treatment >5 years, kidney transplant waiting list >3 years, or symptoms of cardiovascular disease. Patients with unstable angina pectoris were excluded.
Written informed consent was obtained from all patients before study participation. The study followed the principles in the Declaration of Helsinki and was registered at ClinicalTrials.gov (NCT01344434). The study protocol was approved by the Central Denmark Region Committee on Health Research Ethics and by the Danish Data Protection Agency.
Bone Density Measurements
An angiographic computed tomography (CT) scan of chest, abdomen, and pelvis was performed on a dual-source CT scanner (SOMATOM Definition Flash; Siemens, Erlangen, Germany). Details of the CT protocol have been published previously.20 Briefly, tube energy was set at 100 or 120 kVp depending on patient size; tube current was dose modulated and set at 250 mAs; slice thickness was 3.0 mm; and images were reconstructed with a standard soft tissue kernel. A set dose of 95 ml of the x-ray contrast media Ioversol (Optiray, Mallinckrodt, Germany) was administered i.v. BMD was analyzed from the contrast-enhanced images. The commercially available software QCT Pro version 5.1 (Mindways Software Inc, Austin, TX) was used to determine the BMD of the lumbar spine and proximal femur. A calibration phantom (Mindways Solid; Mindways Software Inc, Austin, TX) was scanned separately at regular intervals to provide calibration data for asynchronous BMD analysis.21
At the lumbar spine, an oval region of interest was placed in the anterior part of 3 consecutive vertebrae between L1 and L4, keeping clear of the posterior venous plexus. Fractured and visibly deformed vertebrae were excluded from analyses. L1 to L3 was preferred, although in 17 patients L2 to L4 was analyzed. At the proximal femur, the semiautomatic function provided by the software was used for analyses of left hip BMD, with visual control and manual adjustment if needed. In 13 patients, the right hip was analyzed because of previous fracture, metallic prosthesis, or incomplete image of the left hip. Both t and z scores were calculated from the 2-dimensional hip projection (CTXA) compared to reference data supplied by the software manufacturer. In recent guidelines, CTXA was recommended for use in diagnosing osteoporosis.22 Coefficients of variation (CV) were 0.68% at the lumbar spine, 1.85% at the total hip, and 2.30% at the femoral neck.
Fractures
Fracture status was determined by previous clinical fractures and prevalent vertebral fractures. All low-trauma fractures occurring in adult life were included. High-trauma fractures, as well as fractures of the fingers, toes, face, and skull were excluded. Data on clinical fractures were extracted from patient interview and chart review, and all fractures were confirmed by x-ray images or radiology reports. Prevalent vertebral fractures were diagnosed from 2-dimensional reconstructions of CT images of the thoracolumbar spine by an experienced radiologist according to the method of Genant et al.23
Biochemical Measurements
Fasting morning blood samples were collected. Plasma intact parathyroid hormone, phosphate, ionized calcium, and albumin were analyzed throughout the study period. Blood samples for analyses of sex hormones and bone turnover markers were stored at −80°C and analyzed in a single batch at study completion. We analyzed 2 markers of bone formation: bone-specific alkaline phosphatase (BSAP; enzyme-linked immunosorbent assay, CV 10%) and procollagen type 1 N-terminal propeptide trimer (P1NP; radioimmunoassay IDS-iSYS, CV 10%) and 1 marker of bone resorption: tartrate resistant alkaline phosphatase (TRAP5b; enzyme-linked immunosorbent assay, CV 3%). Total T and Total E were measured by high-performance liquid chromatography, with a CV of 10%. Follicle-stimulating hormone (FSH), luteinizing hormone (LH), and sex hormone–binding globulin were measured by sandwich immunometric assay. The limit of detection was 0.12 nmol/l for total T (n = 7 below),l for total/l for total E2 (n = 14 below), and 0.30 IU/l for both LH (n = 1 below), and FSH (none below). Below-detection values were estimated by dividing the limit of detection by the square root of 2. The upper limit of detection was 200 IU/l for both LH (n = 1 above) and FSH (n = 4 above), and values were estimated by adding 10%. Bio T and Free T were calculated based on the algorithm of Vermeulens et al.,14 using the actual concentrations of plasma albumin rather than the suggested set value of 4.3 g/l. Bio E and Free E were similarly calculated based on constants for protein binding.15 As correlation coefficients were close to 1 between the bioavailable and free fractions for both men (Bio T and Free T, r = 0.95; Bio E and Free E, r = 0.93) and women (Bio T and Free T, r = 0.99; Bio E and Free E, r = 0.995), only the bioavailable fractions are reported. Further details on the analytic methods and assays used are given in Supplementary Table S1.
Statistical Analyses
Statistical analyses were performed with the standard software package STATA/IC 13.1 for Windows (StataCorp LP, College Station, TX). Continuous variables were visually evaluated for normality by QQ plots. Skewed variables were transformed to their natural logarithm (BSAP, P1NP, TRAP5b, Total T, Total E2, sex hormone–binding globulin, parathyroid hormone) to enable parametric statistical testing. Severely skewed variables (LH, FSH, prolactin) were not transformed, and nonparametric testing was used. Normally distributed variables are presented as mean ± SD, and skewed variables as median with interquartile range (IQR). Associations between continuous variables were evaluated by pairwise univariate correlation or Spearman rank correlation. Multiple linear regression analysis was then used to adjust for age, BMI, dialysis therapy, type 1 DM, type 2 DM, and steroid exposure. Steroid exposure was defined as previous treatment with either >10 mg prednisolone for 3 months, >5 mg prednisolone for 12 months, or current prednisolone use. All analyses involving sex hormones were stratified by gender and menopausal status. A 2-sided P value <0.05 was considered statistically significant.
Results
Demographic Data
Of 167 included adult kidney transplantation candidates, 10 were excluded because of withdrawn consent (n = 5), a major cardiovascular event (n = 4), or kidney transplantation (n = 1) before the first study visit. Another 11 individuals were excluded because of the inability to draw blood for biochemical analyses (n = 8) or current hormone replacement therapy (2 women and 1 man), leaving a total of 146 patients.
Baseline data including levels of sex hormones are given in Table 1. Normal ranges of sex hormones by gender are given in Supplementary Table S2. Underlying causes of CKD were type 1 or 2 DM (26%), hypertension or glomerulosclerosis (25%), glomerulonephritis or vasculitis (23%), adult polycystic kidney disease (14%), and other/unknown (11%). Of all patients, 53 were on maintenance dialysis therapy (>3 months); 37 received hemodialysis and 16 peritoneal dialysis therapy. The median time on dialysis was 24 months (IQR = 6−66, range = 3−240). Predialysis patients (n = 93) were all considered close to initiating dialysis by their primary clinician. They had a median estimated glomerular filtration rate (eGFR) of 11 ml/min per m3 (IQR = 9−14, range 4−31). Only 4 patients had an eGFR above 20 ml/min per m3 (at 21, 23, 31, and 31); of these, 1 patient had a high variability in serum creatinine, with the lowest eGFR at 14 ml/min per m3; 1 patient had a 24-hour urine creatinine clearance of 15 ml/min; and 1 patient underwent amputation and likely had an overestimated eGFR. The remaining patient had type 1 DM with multiple diabetic complications and was being referred for a simultaneous pancreas−kidney transplantation.
Table 1.
Baseline data of participating kidney transplantation candidates by gender
| Variable | Men (n = 102) | Premenopausal women (n = 16) | Postmenopausal women (n = 28) |
|---|---|---|---|
| Age, yr | 54 (11) | 42 (6) | 60 (7) |
| Body mass index, kg/m2 | 25.7 (3.7) | 25.4 (7.7) | 25.9 (3.7) |
| Active smoker | 34 (33%) | 5 (31%) | 6 (21%) |
| Diabetes mellitus type 1 | 24 (24%) | 7 (44%) | 2 (7%) |
| Diabetes mellitus type 2 | 11 (11%) | 1 (6%) | 1 (4%) |
| Maintenance dialysis therapy | 36 (35%) | 8 (50%) | 9 (32%) |
| Previous kidney transplantation | 14 (14%) | 5 (31%) | 5 (18%) |
| Medical treatment | |||
| Previous steroid exposure | 16 (16%) | 1 (6%) | 5 (18%) |
| Current steroid treatment | 8 (8%) | 4 (25%) | 6 (21%) |
| Phosphate binder | 74 (73%) | 13 (81%) | 16 (57%) |
| Active vitamin D receptor activator | 75 (74%) | 10 (63%) | 20 (77%) |
| Calcium metabolism | |||
| Parathyroid hormone, ρmol/l | 21.5 [14.5, 29.3] | 16.9 [11.9, 27.0] | 19.8 [11.4, 31.4] |
| Phosphate, mmol/l | 1.59 (0.37) | 1.59 (0.44) | 1.48 (0.36) |
| Ionized calcium, mmol/l | 1.22 (0.08) | 1.24 (0.09) | 1.23 (0.06) |
| Bone specific alkaline phosphatase, μ/l | 25 [20−32] | 30 [25–41] | 24 [22–37] |
| Pro-collagen type I N-terminal propeptide, μg/l | 61.0 [40.5–85.3] | 78.1 [37.4–107] | 77.8 [44.9–113] |
| Tartrate resistant alkaline phosphatase type 5b, U/l | 4.1 [2.7–5.8] | 3.5 [2.1–5.2] | 5.0 [3.2–6.3] |
| Sex hormones | |||
| Total testosterone, nmol/l | 13 [10–17] | 0.5 [0.3–0.7] | 0.58 [0.16–0.95] |
| Bioavailable testosterone, nmol/l | 5.71 (2.14) | 0.130 [0.074–0.176] | 0.124 [0.030–0.246] |
| Total estradiol, nmol/l | 114 (42) | 300 [174–692] | 16 [13–47] |
| Bioavailable estradiol, ρmol/l | 74 (33) | 167 [96–270] | 8.3 [5.8–19.8] |
| Sex hormone–binding globulin, nmol/l | 35 [26–48] | 61 [48–81] | 59 [40–84] |
| Bone mineral density | |||
| Lumbar spine vBMD, mg/cm3 | 116 (35) | 167 (30) | 116 (42) |
| Lumbar spine z score | –0.81 (1.20) | 0.42 (1.20) | 0.24 (1.56) |
| Total hip vBMD, mg/cm3 | 227 (41) | 250 (66) | 225 (37) |
| Total hip z scores | –1.11 (1.08) | –1.81 (1.34) | –1.42 (0.66) |
| Femoral neck vBMD, mg/cm3 | 228 (46) | 261 (73) | 233 (48) |
| Femoral neck z score | –0.96 (0.92) | –1.45 (1.17) | –1.26 (0.80) |
| Femoral neck aBMD, mg/cm2 | 0.593 (0.093) | 0.593 (0.143) | 0.541 (0.100) |
aBMD, areal bone mineral density; vBMD, volumetric bone mineral density.
Data are mean (SD), median [interquartile range], or n (%).
One male patient had recently initiated bisphosphonate therapy (1 month before study inclusion), and 2 male patients currently received calcimimetics. A total of 40 patients had been exposed to steroids either by previous treatment (n = 22) or by currently taking prednisolone (n = 18). The majority of patients on active steroid treatment were on a low daily dose of either 2.5 or 5 mg/d (n = 16), most commonly because of previous kidney transplantation (n = 13). Other indications included connective tissue disease (n = 2), glomerulonephritis (n = 2), and lung transplantation (n = 1). Percentages exposed to steroids were as follows: 24% of men versus 36% of women (P = 0.16), and 24% of predialysis patients versus 34% of dialysis patients (P = 0.18). Steroid exposure was associated with significantly reduced z scores at all areas: lumbar spine (−0.99 ± 1.38 vs. −0.28 ± 1.40, P = 0.004), total hip (−1.56 ± 0.86 vs. −1.13 ± 1.12, P = 0.03), and femoral neck (−1.41 ± 0.72 vs. −0.94 ± 0.99, P = 0.008).
Results for Male Patients
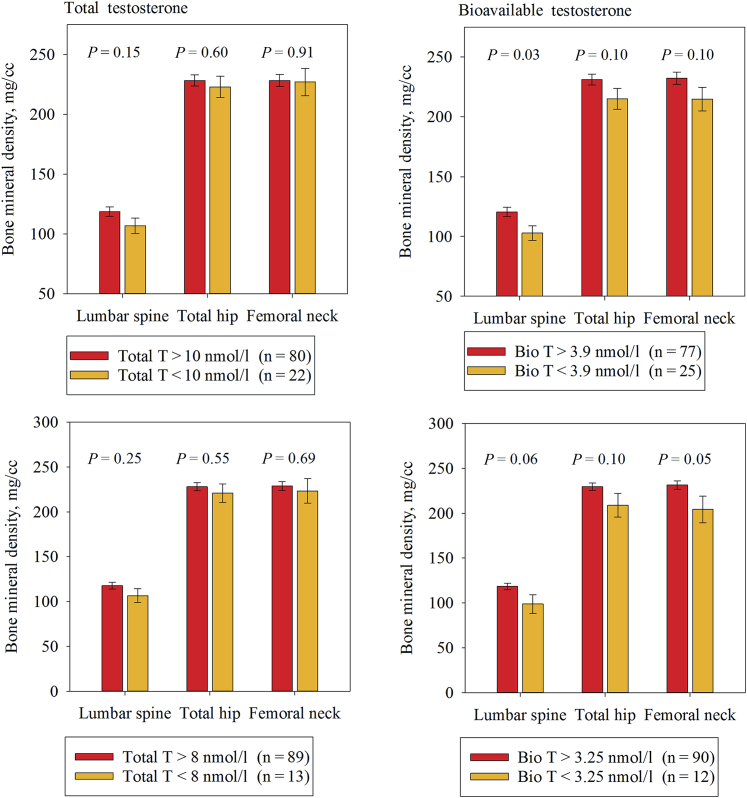
The prevalence of hypogonadism, defined as a Total T < 10 nmol/l, was 22% (n = 22), with levels <8 nmol/l in 13% (n = 13). Men with low levels of Total T had BMD comparable to those with normal T levels (Figure 1). However, a trend toward reduced BMD was seen in men with Bio T levels below the 25th (3.9 nmol/l) or 10th (3.25 nmol/l) percentiles (Figure 1).
Figure 1.
Bone mineral density in male kidney transplantation candidates with low levels of total and bioavailable testosterone. Mean values with standard errors. P values derived from Student t test. Total T, total testosterone; Bio T, bioavailable testosterone.
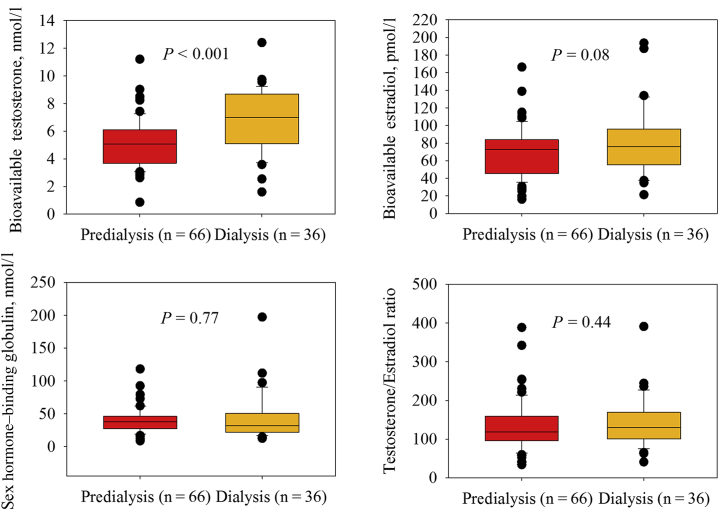
Patients on dialysis had significantly higher levels of T compared to predialysis patients (Figure 2). Patients with type 1 and 2 DM had levels of T similar to those of nondiabetic patients. Bio T was 6.3 ± 2.4 nmol/l in type 1 DM patients, 4.9 ± 2.1 nmol/l in type 2 DM patients, and 5.7 ± 2.0 nmol/l in nondiabetic patients (analysis of variance, P = 0.18). Levels of E were higher in those with DM; Bio E was 89 ± 41 ρmol/l in type 1 DM patients, 84 ± 38 ρmol/l in type 2 DM patients, and 67 ± 26 ρmol/l in nondiabetic patients (analysis of variance, P = 0.01). The difference between type 1 DM patients and nondiabetic patients was significant (p = 0.005) and remained so after adjustment for age, BMI, and dialysis therapy (P = 0.001). The difference between type 2 DM patients and nondiabetic patients was not significant (P = 0.11 and P = 0.10 in the unadjusted and adjusted model, respectively).
Figure 2.
Sex hormone levels in male kidney transplantation candidates, by dialysis status. Box plots with median, interquartile range, and whiskers at 5% and 9%. P values derived from Student t test.
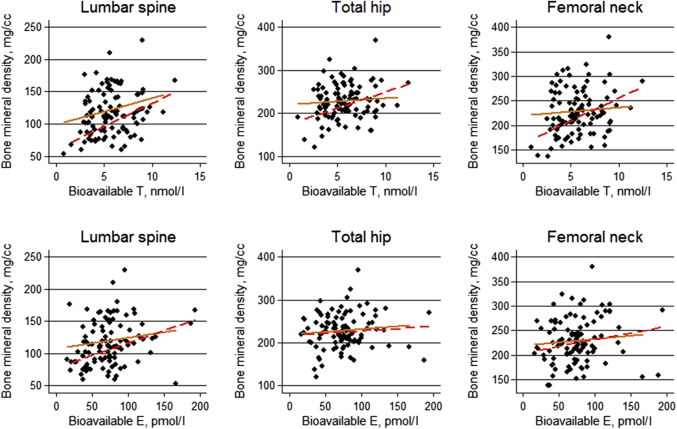
Scatter plots between sex hormones and BMD are shown in Figure 3, and univariate correlations between sex hormones, BMD, and bone turnover markers are shown in Table 2. Both Bio T and Bio E were positively associated with lumbar spine BMD, but not with BMD at the proximal femur. To explore these relationships further, we performed multiple linear regression analyses with BMD as the outcome variable, and the bioavailable fractions of sex hormones as explanatory variables (Table 3). In the adjusted models, Bio T was positively associated with BMD at both the spine and hip, whereas Bio E was positively associated with lumbar spine BMD. Bio T remained significantly associated with BMD at both the spine and hip after adjusting for levels of Bio E (Table 4).
Figure 3.
Scatter plots of the association between bioavailable fractions of testosterone and estradiol and bone mineral density (BMD) in male kidney transplantation candidates. Solid line represents dialysis patients; dotted line represents predialysis patients. Bio E, bioavailable estradiol; Bio T, bioavailable testosterone.
Table 2.
Univariate correlations between levels of sex hormones, bone density, and bone turnover markers in male kidney transplantation candidates
| Variable | Lumbar spine vBMD |
Total hip vBMD |
Femoral neck aBMD |
BSAP |
TRAP5b |
P1NP |
|---|---|---|---|---|---|---|
| rho | rho | rho | rho | rho | rho | |
| Total testosterone, nmol/l | 0.02 | –0.10 | –0.12 | 0.26a | 0.18b | 0.24a |
| Bioavailable testosterone, nmol/l | 0.25a | 0.14 | 0.10 | 0.18b | –0.02 | 0.16 |
| Total estradiol, nmol/l | 0.21a | –0.06 | –0.02 | 0.14 | 0.22a | 0.19b |
| Bioavailable estradiol, ρmol/l | 0.31a | 0.07 | 0.09 | –0.02 | 0.03 | 0.05 |
| Sex hormone–binding globulin, nmol/l | –0.23a | –0.22a | –0.22a | 0.28a | 0.34a | 0.20a |
| Follicle-stimulating hormone, U/l | –0.22a | –0.07 | –0.09 | 0.16 | 0.05 | –0.02 |
| Luteinizing hormone, U/l | –0.09 | –0.08 | –0.11 | 0.03 | –0.01 | –0.09 |
aBMD, areal bone mineral density; BSAP, bone-specific alkaline phosphatase (U/l); P1NP, pro-collagen type 1 N-terminal propeptide (μg/l); TRAP5b, tartrate-resistant alkaline phosphatase (U/l); vBMD, volumetric bone mineral density.
Data are Spearman correlation coefficients (rho).
P < 0.05.
P < 0.10.
Table 3.
Association between bioavailable sex hormones and bone mineral density of spine and hip in male kidney transplantation candidates
| Variable | Lumbar spine vBMD |
Total hip vBMD |
Femoral neck aBMD |
||||||
|---|---|---|---|---|---|---|---|---|---|
| β | 95% CI | P | β | 95% CI | P | β | 95% CI | P | |
| Bioavailable testosterone, nmol/l | |||||||||
| Minimal model | 4.79 | 1.59 to 8.00 | 0.004 | 4.94 | 0.82 to 9.06 | 0.02 | 10.3 | 0.72 to 19.9 | 0.04 |
| Complete model | 5.02 | 1.95 to 8.10 | 0.002 | 6.35 | 2.56 to 10.1 | 0.001 | 13.9 | 5.18 to 22.5 | 0.002 |
| Bioavailable estradiol, nmol/l | |||||||||
| Minimal model | 0.23 | 0.03 to 0.43 | 0.02 | 0.09 | –0.17 to 0.34 | 0.51 | 0.30 | –0.29 to 0.89 | 0.32 |
| Complete model | 0.23 | 0.02 to 0.44 | 0.03 | 0.20 | –0.06 to 0.47 | 0.12 | 0.61 | 0.03 to 1.20 | 0.04 |
aBMD, areal bone mineral density; CI, confidence interval; vBMD, volumetric bone mineral density.
Data are multiple linear regression coefficients (β) with corresponding 95% confidence intervals and –values. Minimal model includes age, body mass index, and dialysis therapy. Complete model includes age, body mass index, dialysis therapy, type 1 diabetes mellitus, type 2 diabetes mellitus, parathyroid hormone, and steroid exposure.
Table 4.
Multiple linear regression analysis of the association between sex hormones and bone density in male kidney transplantation candidates
| Variable | Lumbar spine vBMD (Adj. R2 = 0.27, P < 0.001) |
Total hip vBMD (Adj. R2 = 0.22, P < 0.001) |
Femoral neck aBMD (Adj. R2 = 0.24, P < 0.001) |
||||||
|---|---|---|---|---|---|---|---|---|---|
| β | CI | P | β | CI | P | β | CI | P | |
| Age, yr | –1.28 | –1.93 to –0.63 | <0.001 | –0.61 | –1.41 to 0.19 | 0.14 | –2.05 | –3.88 to –0.23 | 0.03 |
| Body mass index, kg/m2 | 1.46 | –0.26 to 3.19 | 0.10 | 1.75 | –0.40 to 3.90 | 0.11 | 2.15 | –2.77 to 7.07 | 0.39 |
| Dialysis therapy, yes/no | –12.8 | –26.8 to 1.25 | 0.07 | –14.4 | –31.8 to 3.01 | 0.10 | –54.3 | –94.0 to –14.5 | 0.008 |
| Diabetes mellitus type 1, yes/no | 9.04 | –26.4 to 8.36 | 0.31 | –34.7 | –56.2 to –13.2 | 0.002 | –111.8 | –161 to –62.6 | <0.001 |
| Diabetes mellitus type 2, yes/no | 4.77 | –15.4 to 24.9 | 0.64 | 18.3 | –6.62 to 43.2 | 0.15 | 13.0 | –43.2 to 69.1 | 0.65 |
| Steroid exposure, yes/no | –19.3 | –33.7 to –4.80 | 0.01 | –17.3 | –35.2 to 0.59 | 0.06 | –52.6 | –92.8 to –12.3 | 0.01 |
| Parathyroid hormone, ρmol/l | –8.86 | –16.0 to –1.69 | 0.02 | –13.3 | –22.6 to –3.96 | 0.006 | –19.8 | –40.8 to 1.20 | 0.06 |
| Bioavailable testosterone, nmol/l | 4.32 | 0.87 to 7.76 | 0.02 | 6.21 | 1.95 to 10.5 | 0.005 | 12.1 | 2.39 to 21.8 | 0.02 |
| Bioavailable estradiol, ρmol/l | 0.10 | –0.12 to 0.33 | 0.37 | 0.22 | –0.26 to 0.30 | 0.88 | 0.27 | –0.37 to 0.90 | 0.40 |
aBMD, areal bone mineral density; Adj., adjusted; CI, confidence interval; vBMD, volumetric bone mineral density.
Data are multiple linear regression β coefficients with 95% confidence intervals and P values.
Bone turnover markers were not correlated with levels of Bio T or E (Table 3). Total T was positively associated with markers of bone formation (BSAP and P1NP). However, these associations were no longer significant after adjusting for age, BMI, and dialysis therapy (BSAP: P = 0.07, and P1NP: P = 0.18). Similarly, Total E was positively correlated with bone resorption marker TRAP5b, but not significantly so in the adjusted model (P = 0.17).
Men with a previous fragility fracture (n = 17) had Total T 16 [IQR = 9−19] nmol/l, Bio T of 5.3 ± 2.3 nmol/l, and Bio E of 82 ± 44 ρmol/l. This was not significantly different from patients with no history of fracture: Total T 13 [IQR = 10−16] nmol/l (difference: 2%, P = 0.87), Bio T 5.8 ± 2.1 nmol/l (difference: 0.5 nmol/l, P = 0.36), and Bio E 73 ± 30 ρmol/l (difference: 9 ρmol/l, P = 0.30). Levels of sex hormones were also not associated with fracture status in a multivariate logistic regression model adjusted for age, BMI, and dialysis therapy (results not shown).
Results for Female Patients
By patient interview, 12 women were premenopausal, 22 were postmenopausal, and 10 had unknown status. The 10 women with unknown status were categorized as postmenopausal if FSH levels were >100 IU/l (n = 5) or age was >60 years with undetectable Total E (n = 1). The remaining 4 women were 42, 45, 45, and 49 years old, with Total E values well above the postmenopausal range (>200 nmol/l), and they were categorized as premenopausal.
BMD and z scores for pre- and postmenopausal women are shown in Table 1. Lumbar spine BMD was significantly reduced in postmenopausal compared to premenopausal women, with a difference of −52 mg/cm3 (CI = −76 to −28, P < 0.001). In contrast, BMD was comparable at the proximal femur, with a difference of −25 mg/cm3 at the total hip (CI = −57 to 6, P = 0.11), and −28 mg/cm3 at the femoral neck (CI = −65 to 9, P = 0.14). TRAP5b was 31% (CI = 3% to 51%, P = 0.03) higher in postmenopausal women, whereas there were no differences in BSAP (−7%, CI = −35 to 25, P = 0.57) or P1NP (10%, CI = −33 to 40, P = 0.58) based on menopausal status.
Univariate correlations between levels of sex hormones and BMD of spine and hip in women are shown in Table 5. In postmenopausal women, levels of T were positively correlated with BMD of the spine. In premenopausal women, LH was the only hormone significantly correlated with BMD.
Table 5.
Univariate correlations between levels of sex hormones and bone mineral density of spine and hip in female kidney transplantation candidates
| Variable | Premenopausal women (n = 16) |
Postmenopausal women (n = 28) |
||||
|---|---|---|---|---|---|---|
| Lumbar spine vBMD |
Total hip vBMD |
Femoral neck aBMD |
Lumbar spine vBMD |
Total hip vBMD |
Femoral neck aBMD |
|
| rho | rho | rho | rho | rho | rho | |
| Total testosterone, nmol/l | 0.18 | –0.01 | –0.01 | 0.46a | 0.17 | 0.16 |
| Bioavailable testosterone, nmol/l | 0.12 | 0.09 | 0.09 | 0.46a | 0.23 | 0.23 |
| Total estradiol, nmol/l | 0.09 | –0.04 | –0.05 | 0.18 | –0.02 | –0.11 |
| Bioavailable estradiol, nmol/l | 0.22 | 0.14 | 0.12 | 0.17 | –0.00 | –0.02 |
| Sex hormone−binding globulin, nmol/l | 0.02 | –0.15 | –0.14 | 0.06 | –0.05 | –0.04 |
| Follicle-stimulating hormone, U/l | 0.24 | –0.05 | 0.04 | 0.01 | –0.02 | –0.21 |
| Luteinizing hormone, U/l | 0.53a | 0.41 | 0.52a | –0.12 | –0.01 | –0.18 |
aBMD, areal bone mineral density; vBMD, volumetric bone mineral density.
Data are Spearman correlation coefficients (rho).
P < 0.05.
There were no significant correlations between biochemical markers of bone turnover and levels of sex hormones for either pre- or postmenopausal women (data not shown).
Premenopausal women with a previous fragility fracture (n = 4) had lower levels of Bio T than women without fracture (median = 57 [IQR = 52−74] vs. 144 [IQR = 113−208] ρmol/l, P = 0.04), whereas levels of Bio E were comparable (median = 270 [IQR = 140−330] vs. 145 [IQR = 96−215] ρmol/l, P = 0.67). Postmenopausal women with fracture (n = 4) had levels of Bio T (median = 23 [IQR = 18−246] vs. 137 [IQR = 48−246] ρmol/l, P = 0.12) and Bio E (median = 7 [IQR = 6−24] vs. 8 [IQR = 6−20] ρmol/l, P = 0.68), similar to levels in women without fracture.
Discussion
In this study, we demonstrate a positive association between levels of sex hormones and BMD in male kidney transplantation candidates. Specifically, these associations were seen with the biologically active fractions of the hormones. In female patients, testosterone levels were positively correlated with BMD of the spine in postmenopausal, but not premenopausal women.
Male Patients
Surprisingly, patients on maintenance dialysis therapy had elevated levels of T compared to predialysis patients. This is in contrast to previous studies, in which the prevalence of hypogonadism increased with decreasing kidney function.1, 24 Possible explanations include a higher number of type 1 DM patients among predialysis compared to dialysis patients (30% vs. 11%), as these patients had a trend toward higher levels of T. However, including DM 1 and 2 as explanatory variables in the multivariate models did not fully explain the differences in T levels between dialysis and predialysis patients. Albumin levels were lower in predialysis patients; however, this would be expected to affect only Bio T and not Total T-levels. Our maintenance dialysis patients were a highly selected group, as they were all considered kidney transplantation candidates. This was reflected by high proportions of patients having been on dialysis for <12 months (32%) and retaining some residual kidney function (67%). We speculate that an improvement in gonadal axis function may occur with the initiation of dialysis therapy, but this issue will need further investigation.
This is the first study to demonstrate an association between T levels and bone density in male patients with severe CKD. Such a positive relationship is well known in otherwise healthy elderly men,25, 26 in whom low levels of T increase the risk of osteoporosis and are associated with loss of BMD over time.26 In regard to CKD, only 1 study previously reported on this issue. Mirfakhraee et al. found no significant correlations between Total T or Bio T and z scores in male hemodialysis patients.7 The study did find that Free E was positively associated with hip and forearm BMD, with correlations of the same magnitude as seen in our cohort; however, these associations were no longer significant after adjustment for body size. In contrast, the relationship between BMD and both Bio T and Bio E in our cohort were robust through adjustments for several possible confounders, including age, body size, diabetes, maintenance dialysis therapy, hyperparathyroidism, and steroid exposure.
We did not find any differences in sex hormone status between men based on fracture status. However, the cross-sectional nature of our analyses, and the low number of events, may have prevented us from detecting a true association.
The mechanism of T on bone is believed to be mediated through stimulation of the androgen receptor on both osteoblast and osteoclast lineage cells.3 We found positive correlations between levels of sex hormones and bone formation markers, whereas there were no significant associations with bone resorption markers. In addition, a previous study including male hemodialysis patients found a negative association between Total T and RANKL concentrations.11, 12 RANKL is secreted by osteoblasts and binds to its receptor RANK on osteoclasts, stimulating osteoclast proliferation and activation; higher levels are therefore associated with increased bone resorption. Thus, a direct stimulation of osteoblast function, coupled with an indirect effect on osteoclast function through the RANK−RANKL system, may be the possible mechanism of the effect of T on the skeleton in CKD. Unfortunately, we did not have the data to further investigate this hypothesis.
Bio E was positively associated with bone density of the lumbar spine. This is in line with several recent studies in otherwise healthy elderly men, demonstrating strong, positive, associations between endogenous levels of E and BMD, leading the authors to speculate that E could be as powerful a regulator of bone metabolism in men as it is in women.27, 28, 29, 30 This hypothesis is supported by results from interventional studies selectively blocking the effects of T and E and measuring the skeletal response by QCT or bone turnover markers.31, 32
Female Patients
In women, we found the expected differences of reduced BMD and increased bone resorption after menopause. High levels of T were associated with greater BMD at the lumbar spine in postmenopausal women, which is consistent with findings in postmenopausal women with normal kidney function.33 Premenopausal women with previous fragility fractures had lower levels of T, and the same trend was seen in postmenopausal women. Thus, similar to otherwise healthy women, the reduction of endogenous sex hormone levels after menopause could cause a loss of BMD and consequently a reduction in bone strength in CKD. Unfortunately, the low number of women in each group prevented any meaningful multivariate analyses.
Study Strengths and Limitations
We consider our choice of method for measuring BMD a strength, as CT yields high-resolution, 3-dimensional images, enabling precise measurement of volumetric BMD and avoiding artifacts from surrounding tissues. Major limitations include the cross-sectional design, and a relatively small number of patients, particularly women. We used single-measurement of sex hormones in both men and women, and furthermore, we did not time our analyses according to the menstrual cycle in premenopausal women. The equations used to calculate free and bioavailable fractions have not been validated for patients with CKD. There was great heterogeneity in the cohort with regard to the cause, stage, and treatment of CKD; we attempted to adjust for this by including type 1 and type 2 DM, as well as dialysis therapy in our multivariate models. On the other hand, the cohort was a selected population of end-stage kidney disease patients considered for kidney transplantation, which may limit the generalizability of our results. As more than 95% of our cohort was Caucasian/white, results may not be transferrable to other racial/ethnic groups.
In conclusion, a positive relationship between sex hormones and BMD was found in male kidney transplantation candidates. Disturbances in the gonadal axis may contribute to skeletal fragility in men with late-stage CKD.
Disclosure
All the authors declared no competing interests.
Acknowledgments
The results published here have previously been presented in abstract form at ASN Kidney Week, November 15–20, 2016 (Chicago, IL).
The authors thank all study participants and the following co-workers: Birgitte Kildevæld Sahl (Renal Research Laboratory, Aarhus University Hospital) for biochemical analyses and clinical examinations; Birgitte Bang (Department of Nephrology, Aalborg University Hospital) for her long-standing assistance; and staff at Departments of Nephrology, Viborg Hospital, and Holstebro Hospital for including patients.
This study received financial support from the following sources: Aarhus University, Danish Society of Nephrology, Danish Kidney Association, Central Denmark Health Region, Karen Elise Jensen Foundation, Helen and Ejnar Bjørnow Research Fund, Søster and Verner Lippert Research Fund, Cand. polyt. Frode V Nyegaard and Wife Research Fund, and MD Søren Segel and Wife Johanne Wiibroe Segel Research Fund.
Footnotes
Table S1. Details of analytic methods and assays used.
Table S2. Levels of sex hormones with normal ranges by gender.
Supplementary material is linked to the online version of the paper at www.kireports.org.
Supplementary Material
Details of analytic methods and assays used.
Levels of sex hormones with normal ranges by gender.
References
- 1.Carrero J.J., Qureshi A.R., Nakashima A. Prevalence and clinical implications of testosterone deficiency in men with end-stage renal disease. Nephrol Dial Transplant. 2011;26:184–190. doi: 10.1093/ndt/gfq397. [DOI] [PubMed] [Google Scholar]
- 2.Khosla S., Oursler M.J., Monroe D.G. Estrogen and the skeleton. Trends Endocrinol Metab. 2012;23:576–581. doi: 10.1016/j.tem.2012.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Clarke B.L., Khosla S. Androgens and bone. Steroids. 2009;74:296–305. doi: 10.1016/j.steroids.2008.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cauley J.A. Estrogen and bone health in men and women. Steroids. 2015;99:11–15. doi: 10.1016/j.steroids.2014.12.010. [DOI] [PubMed] [Google Scholar]
- 5.Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Update Work Group KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD) Kidney Int. 2009;76(suppl 113):S1–S130. doi: 10.1038/ki.2009.188. [DOI] [PubMed] [Google Scholar]
- 6.Arneson T.J., Li S., Liu J. Trends in hip fracture rates in US hemodialysis patients, 1993–2010. Am J Kidney Dis. 2013;62:747–754. doi: 10.1053/j.ajkd.2013.02.368. [DOI] [PubMed] [Google Scholar]
- 7.Mirfakhraee S., Sakhaee K., Zerwekh J. Risk factors for diminished bone mineral density among male hemodialysis patients—a cross-sectional study. Arch Osteoporos. 2012;7:283–290. doi: 10.1007/s11657-012-0110-3. [DOI] [PubMed] [Google Scholar]
- 8.Weisinger J.R., Gonzalez L., Alvarez H. Role of persistent amenorrhea in bone mineral metabolism of young hemodialyzed women. Kidney Int. 2000;58:331–335. doi: 10.1046/j.1523-1755.2000.00170.x. [DOI] [PubMed] [Google Scholar]
- 9.Sugiya N., Nakashima A., Takasugi N. Endogenous estrogen may prevent bone loss in postmenopausal hemodialysis patients throughout life. Osteoporos Int. 2011;22:1573–1579. doi: 10.1007/s00198-010-1350-y. [DOI] [PubMed] [Google Scholar]
- 10.Matuszkiewicz-Rowinska J., Skorzewska K., Radowicki S. The benefits of hormone replacement therapy in pre-menopausal women with oestrogen deficiency on haemodialysis. Nephrol Dial Transplant. 1999;14:1238–1243. doi: 10.1093/ndt/14.5.1238. [DOI] [PubMed] [Google Scholar]
- 11.Doumouchtsis K.K., Kostakis A.I., Doumouchtsis S.K. Associations between osteoprotegerin and femoral neck BMD in hemodialysis patients. J Bone Miner Metab. 2008;26:66–72. doi: 10.1007/s00774-007-0785-5. [DOI] [PubMed] [Google Scholar]
- 12.Doumouchtsis K.K., Kostakis A.I., Doumouchtsis S.K. The effect of sexual hormone abnormalities on proximal femur bone mineral density in hemodialysis patients and the possible role of RANKL. Hemodial Int Symp Home Hemodial. 2008;12:100–107. doi: 10.1111/j.1542-4758.2008.00249.x. [DOI] [PubMed] [Google Scholar]
- 13.Dunn J.F., Nisula B.C., Rodbard D. Transport of steroid hormones: binding of 21 endogenous steroids to both testosterone-binding globulin and corticosteroid-binding globulin in human plasma. J Clin Endocrinol Metab. 1981;53:58–68. doi: 10.1210/jcem-53-1-58. [DOI] [PubMed] [Google Scholar]
- 14.Vermeulen A., Verdonck L., Kaufman J.M. A critical evaluation of simple methods for the estimation of free testosterone in serum. J Clin Endocrinol Metab. 1999;84:3666–3672. doi: 10.1210/jcem.84.10.6079. [DOI] [PubMed] [Google Scholar]
- 15.Mazer N.A. A novel spreadsheet method for calculating the free serum concentrations of testosterone, dihydrotestosterone, estradiol, estrone and cortisol: with illustrative examples from male and female populations. Steroids. 2009;74:512–519. doi: 10.1016/j.steroids.2009.01.008. [DOI] [PubMed] [Google Scholar]
- 16.Morris P.D., Malkin C.J., Channer K.S., Jones T.H. A mathematical comparison of techniques to predict biologically available testosterone in a cohort of 1072 men. Eur J Endocrinol. 2004;151:241–249. doi: 10.1530/eje.0.1510241. [DOI] [PubMed] [Google Scholar]
- 17.Mendel C.M. The free hormone hypothesis: a physiologically based mathematical model. Endocr Rev. 1989;10:232–274. doi: 10.1210/edrv-10-3-232. [DOI] [PubMed] [Google Scholar]
- 18.Schmidt A., Luger A., Hörl W.H. Sexual hormone abnormalities in male patients with renal failure. Nephrol Dial Transplant. 2002;17:368–371. doi: 10.1093/ndt/17.3.368. [DOI] [PubMed] [Google Scholar]
- 19.Glass A.R., Beach J., Vigersky R.A. Hypogonadotropic hypogonadism in nephrotic rats: increased sensitivity to negative feedback effects of testosterone. Metabolism. 1985;34:574–579. doi: 10.1016/0026-0495(85)90197-0. [DOI] [PubMed] [Google Scholar]
- 20.Winther S., Svensson M., Jørgensen H.S. Diagnostic performance of coronary CT angiography and myocardial perfusion imaging in kidney transplantation candidates. JACC Cardiovasc Imaging. 2015;8:553–562. doi: 10.1016/j.jcmg.2014.12.028. [DOI] [PubMed] [Google Scholar]
- 21.QCT Pro Bone Mineral Densitometry Software—User’s guide [computer program]. Version 5.0. Austin, TX; 2011.
- 22.Engelke K., Lang T., Khosla S. Clinical use of quantitative computed tomography-based advanced techniques in the management of osteoporosis in adults: the 2015 ISCD official positions—part III. J Clin Densitom. 2015;18:393–407. doi: 10.1016/j.jocd.2015.06.010. [DOI] [PubMed] [Google Scholar]
- 23.Genant H.K., Wu C.Y., van Kuijk C., Nevitt M.C. Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res. 1993;8:1137–1148. doi: 10.1002/jbmr.5650080915. [DOI] [PubMed] [Google Scholar]
- 24.Albaaj F., Sivalingham M., Haynes P. Prevalence of hypogonadism in male patients with renal failure. Postgrad Med J. 2006;82:693–696. doi: 10.1136/pgmj.2006.045963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.van den Beld A.W., de Jong F.H., Grobbee D.E. Measures of bioavailable serum testosterone and estradiol and their relationships with muscle strength, bone density, and body composition in elderly men. J Clin Endocrinol Metab. 2000;85:3276–3282. doi: 10.1210/jcem.85.9.6825. [DOI] [PubMed] [Google Scholar]
- 26.Fink H.A., Ewing S.K., Ensrud K.E. Association of testosterone and estradiol deficiency with osteoporosis and rapid bone loss in older men. J Clin Endocrinol Metab. 2006;91:3908–3915. doi: 10.1210/jc.2006-0173. [DOI] [PubMed] [Google Scholar]
- 27.Khosla S., Melton L.J., Atkinson E.J., O’Fallon W.M. Relationship of serum sex steroid levels to longitudinal changes in bone density in young versus elderly men. J Clin Endocrinol Metab. 2001;86:3555–3561. doi: 10.1210/jcem.86.8.7736. [DOI] [PubMed] [Google Scholar]
- 28.Cauley J.A., Ewing S.K., Taylor B.C. Sex steroid hormones in older men: longitudinal associations with 4.5-year change in hip bone mineral density—the Osteoporotic Fractures in Men Study. J Clin Endocrinol Metab. 2010;95:4314–4323. doi: 10.1210/jc.2009-2635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hsu B., Cumming R.G., Seibel M.J. Reproductive hormones and longitudinal change in bone mineral density and incident fracture risk in older men: the Concord Health and Aging in Men Project. J Bone Miner Res. 2015;30:1701–1708. doi: 10.1002/jbmr.2493. [DOI] [PubMed] [Google Scholar]
- 30.Cawthon P.M., Schousboe J.T., Harrison S.L. Sex hormones, sex hormone binding globulin, and vertebral fractures in older men. Bone. 2016;84:271–278. doi: 10.1016/j.bone.2016.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Finkelstein J.S., Lee H., Leder B.Z. Gonadal steroid-dependent effects on bone turnover and bone mineral density in men. J Clin Invest. 2016;126:1114–1125. doi: 10.1172/JCI84137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Falahati-Nini A., Riggs B.L., Atkinson E.J. Relative contributions of testosterone and estrogen in regulating bone resorption and formation in normal elderly men. J Clin Invest. 2000;106:1553–1560. doi: 10.1172/JCI10942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.van Geel T.A.C.M., Geusens P.P., Winkens B. Measures of bioavailable serum testosterone and estradiol and their relationships with muscle mass, muscle strength and bone mineral density in postmenopausal women: a cross-sectional study. Eur J Endocrinol. 2009;160:681–687. doi: 10.1530/EJE-08-0702. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Details of analytic methods and assays used.
Levels of sex hormones with normal ranges by gender.